Stimulus-Responsive Nanomedicines for Disease Diagnosis and Treatment
Abstract
1. Introduction
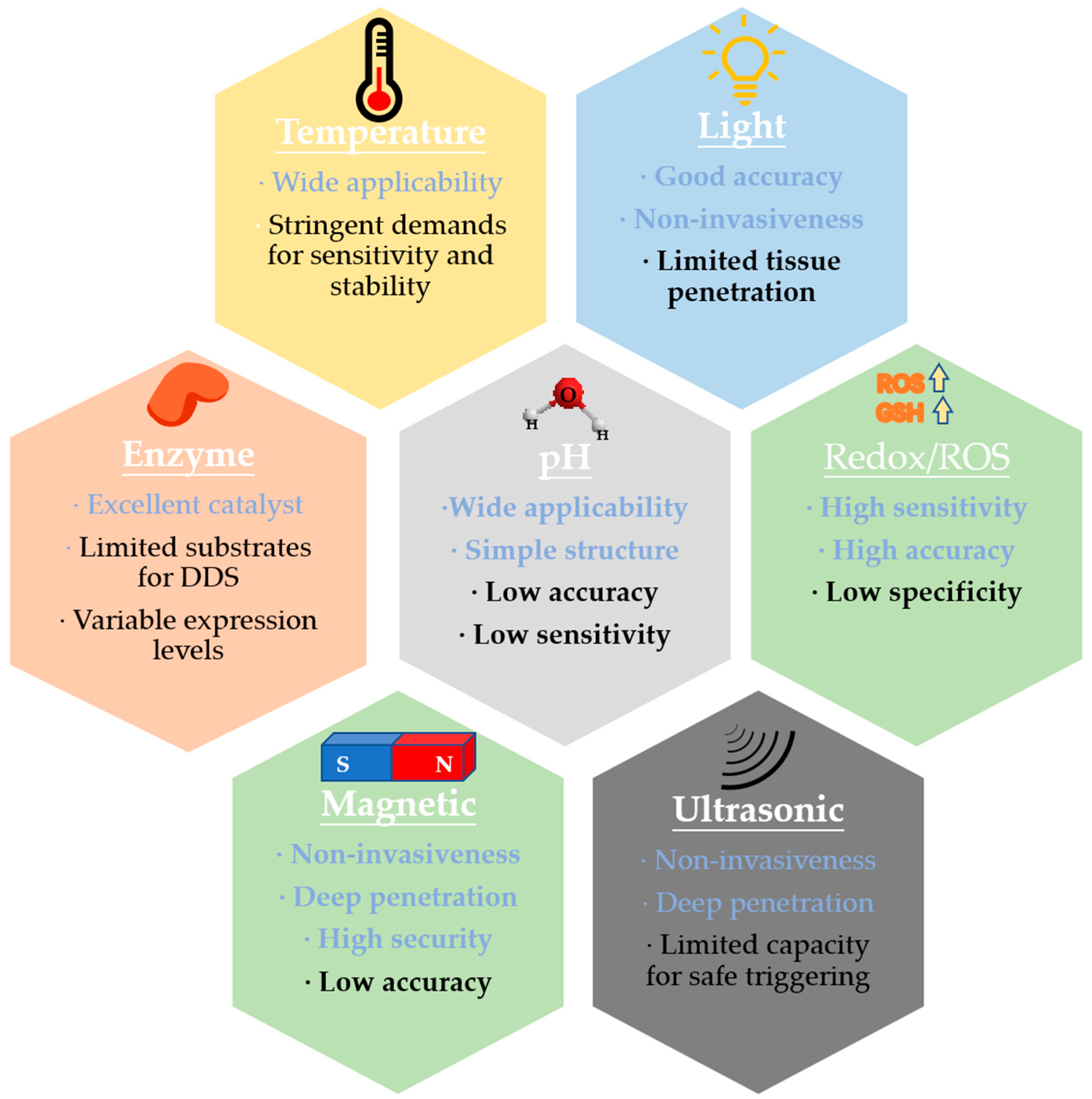
2. Stimulus-Responsive Systems
2.1. pH Stimulus
2.2. Redox Stimulus
2.3. Enzyme Stimulus
2.4. Light Stimulus
2.5. Temperature Stimulus
2.6. Magnetic and Ultrasound Stimulus
2.7. Self-Immolative Structures
3. Applications of Stimulus-Responsive Systems
3.1. Therapeutic Applications
3.1.1. Cancers
3.1.2. Inflammation
3.1.3. Oral Medications
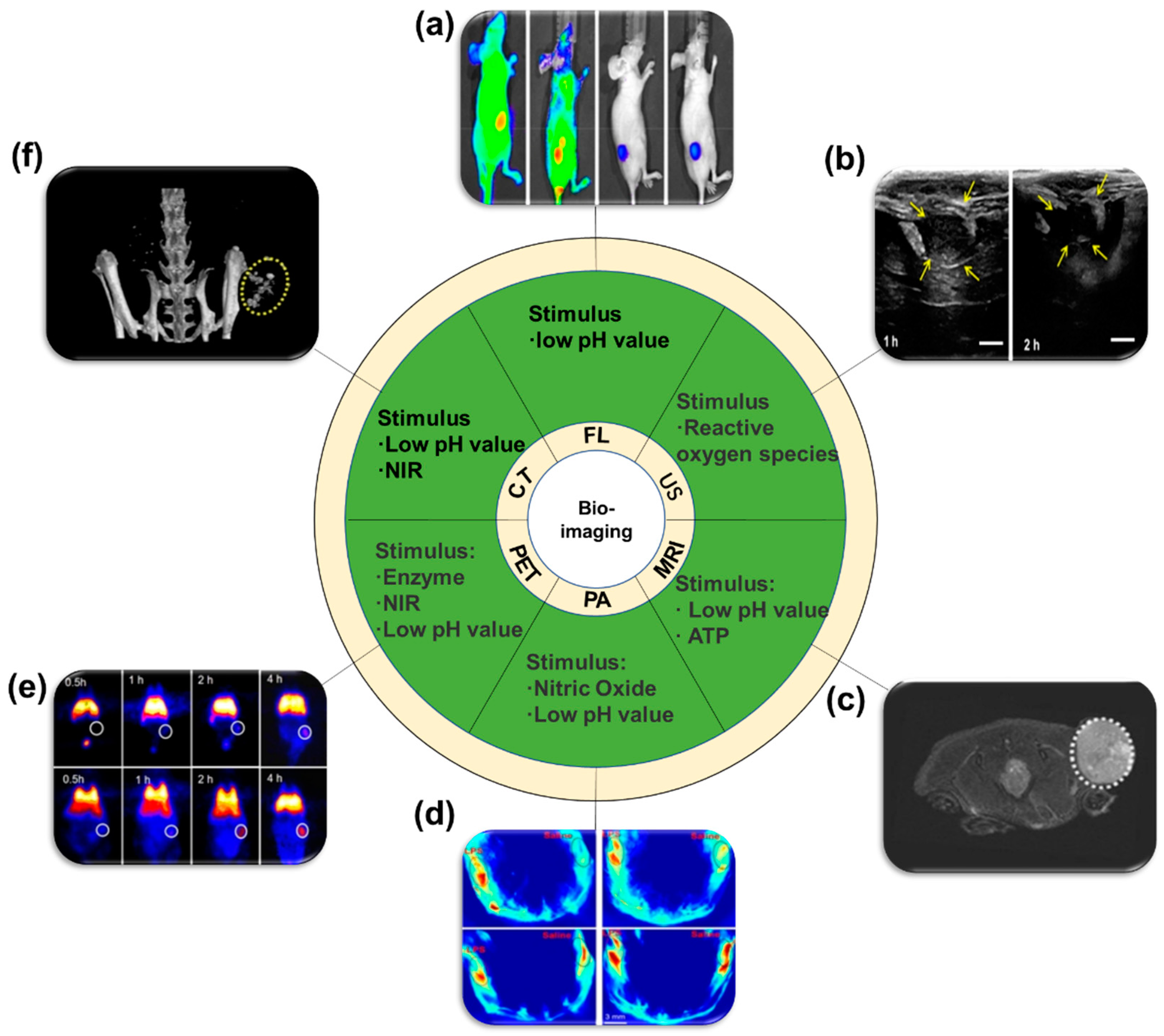
3.2. Applications for Bio-Imaging
4. Conclusions
Funding
Conflicts of Interest
Abbreviations
| API | Active pharmaceutical ingredient |
| AAV2 | Adeno-associated virus 2 |
| APA | Apatite |
| ACP | Acetylated-chondroitin sulfate-protoporphyrin |
| BSA | Bovine serum albumin |
| CDDP | Cisplatin |
| CPT | Camptothecin |
| CS | Chitosan |
| CT | Computed tomography |
| COVID-19 | Coronavirus disease 2019 |
| DSS | Dextran sodium sulphate |
| DPPC | 1,2-dipalmitoyl-sn-glycerin-3-phosphocholine |
| DOX | Doxorubicin |
| DAEs | Diarylethenes |
| Epo B | Epothilone B |
| FI | Fluorescence imaging |
| FA | Folic acid |
| FPRR | Fluoro-photoacoustic polymeric renal reporter |
| GSH | Glutathione |
| GIT | Gastrointestinal tract |
| GNS | Gold nanostars |
| GOx | Glucose oxidase |
| Hf | Hafnium |
| HPMCAS | Hydroxypropyl methylcellulose acetate succinate |
| HPPH | Hexyloxyethy-pyropheophorbide |
| ICG | Indocyanine green |
| IBD | Inflammatory bowel disease |
| Ins | Insulin |
| LCST | Lower critical solubility temperature |
| MRI | Magnetic resonance imaging |
| MTX | Methotrexate |
| MC | Merocyanine |
| MSNs | Mesoporous silica nanoparticles |
| MBs | Microbubbles |
| NCPs | Nanoscale coordination polymers |
| NQO1 | NAD(P)H, quinone oxidoreductase isoenzyme I |
| Nap | Naproxen |
| NPCS | N-palmitoyl chitosan |
| NIPAAm | N-isopropylacrylamide |
| NIRF | Near-infrared fluorescence |
| NLC | Nanostructured lipid carrier |
| OEGMA | Poly (ethylene glycol) methyl ether methacrylate |
| OSPE | Organic semiconducting pro-nanoenzyme |
| OxiDEX | Oxidatively sensitive dextran |
| OPV | Oligo (p-phenylene vinylene) derivative |
| ONB | O-nitrobenzyl |
| OEG | Oligo (ethylene glycol) |
| PBA | Phenylboronic acid |
| PBE | Phenylboronic ester |
| PDT | Photodynamic therapy |
| PLGA | Poly (d,l-lactic-co-glycolic acid) |
| PEGDA | Poly (ethylene glycol) diacrylate |
| PS | Polystyrene |
| PEG | Polyethylene glycol |
| PEI | Polyethylenimine |
| PAMAM | Poly(amidoamine) |
| PNP-UC | PEG-NMAB-PLA-UCNPs |
| PTX | Paclitaxel |
| PDP | Pentadecylphenol |
| pTSL | Polymer-modified temperature-sensitive liposome |
| PAA | Propylacrylic acid |
| PAI | Photoacoustic imaging |
| PGA | Penicillin G amidase |
| PET | Positron emission tomography |
| P-gp | P-glycoprotein |
| ROS | Reactive oxygen species |
| RAFT | Reversible addition-break chain transfer |
| SPN | Semiconductor polymer nanoparticles |
| sPLA2 | Phospholipase A2 |
| sUN | Solid ultrasound nanosensor |
| SP | Spiropyran |
| TK | Thioketal |
| TL | Thioketal linker |
| TGMS | Triglyceride monostearate |
| TPA | Two-photon absorption |
| TSL | Temperature-sensitive liposomes |
| US | Ultrasound |
| UC | Upconversion |
| UCST | Upper critical solubility temperature |
| VEGF | Vascular endothelial growth factor |
| W/O/W | Water/oil/water |
References
- Blum, A.P.; Kammeyer, J.K.; Rush, A.M.; Callmann, C.E.; Hahn, M.E.; Gianneschi, N.C. Stimuli-Responsive Nanomaterials for Biomedical Applications. J. Am. Chem. Soc. 2015, 137, 2140–2154. [Google Scholar] [CrossRef]
- Timko, B.P.; Dvir, T.; Kohane, D.S. Remotely Triggerable Drug Delivery Systems. Adv. Mater. 2010, 22, 4925–4943. [Google Scholar] [CrossRef]
- Wang, Y.; Kohane, D.S. External triggering and triggered targeting strategies for drug delivery. Nat. Rev. Mater. 2017, 2, 17020. [Google Scholar] [CrossRef]
- Manzoor, A.A.; Lindner, L.H.; Landon, C.D.; Park, J.-Y.; Simnick, A.J.; Dreher, M.R.; Das, S.; Hanna, G.; Park, W.; Chilkoti, A.; et al. Overcoming Limitations in Nanoparticle Drug Delivery: Triggered, Intravascular Release to Improve Drug Penetration into Tumors. Cancer Res. 2012, 72, 5566. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.-X.; Li, Z.-H.; Liu, X.-H.; Zheng, D.-W.; Chen, Y.; Zhang, X.-Z. Bacteria-Mediated Tumor Therapy Utilizing Photothermally-Controlled TNF-α Expression via Oral Administration. Nano Lett. 2018, 18, 2373–2380. [Google Scholar] [CrossRef] [PubMed]
- Tibbitt, M.W.; Dahlman, J.E.; Langer, R. Emerging Frontiers in Drug Delivery. J. Am. Chem. Soc. 2016, 138, 704–717. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T. Collapse of Gels and Critical Endpoint. Phys. Rev. Lett. 1978, 40, 820–823. [Google Scholar] [CrossRef]
- Yatvin, M.B.; Weinstein, J.N.; Dennis, W.H.; Blumenthal, R. Design of liposomes for enhanced local release of drugs by hyperthermia. Science 1978, 202, 1290. [Google Scholar] [CrossRef]
- Gao, W.; Chan, J.M.; Farokhzad, O.C. pH-Responsive Nanoparticles for Drug Delivery. Mol. Pharm. 2010, 7, 1913–1920. [Google Scholar] [CrossRef]
- Yin, W.; Li, J.; Ke, W.; Zha, Z.; Ge, Z. Integrated Nanoparticles to Synergistically Elevate Tumor Oxidative Stress and Suppress Antioxidative Capability for Amplified Oxidation Therapy. ACS Appl. Mater. Interfaces 2017, 9, 29538–29546. [Google Scholar] [CrossRef]
- Ramezani, P.; Abnous, K.; Taghdisi, S.M.; Zahiri, M.; Ramezani, M.; Alibolandi, M. Targeted MMP-2 responsive chimeric polymersomes for therapy against colorectal cancer. Colloids Surf. B Biointerfaces 2020, 193, 111135. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Kim, E.-J.; Shi, H.; Lee, J.Y.; Chung, B.G.; Kim, J.S. Development of a theranostic prodrug for colon cancer therapy by combining ligand-targeted delivery and enzyme-stimulated activation. Biomaterials 2018, 155, 145–151. [Google Scholar] [CrossRef]
- Luo, L.; Xu, F.; Peng, H.; Luo, Y.; Tian, X.; Battaglia, G.; Zhang, H.; Gong, Q.; Gu, Z.; Luo, K. Stimuli-responsive polymeric prodrug-based nanomedicine delivering nifuroxazide and doxorubicin against primary breast cancer and pulmonary metastasis. J. Control. Release 2020, 318, 124–135. [Google Scholar] [CrossRef] [PubMed]
- Darvishi, B.; Farahmand, L.; Majidzadeh-A, K. Stimuli-Responsive Mesoporous Silica NPs as Non-viral Dual siRNA/Chemotherapy Carriers for Triple Negative Breast Cancer. Mol. Ther. Nucleic Acids 2017, 7, 164–180. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, P.; Hira, S.K.; Sharma, A.; Kashif, M.; Srivastava, P.; Srivastava, D.N.; Singh, R.A.; Manna, P.P. Telomerase Responsive Delivery of Doxorubicin from Mesoporous Silica Nanoparticles in Multiple Malignancies: Therapeutic Efficacies against Experimental Aggressive Murine Lymphoma. Bioconjugate Chem. 2018, 29, 2107–2119. [Google Scholar] [CrossRef] [PubMed]
- Hira, S.K.; Mitra, K.; Srivastava, P.; Singh, S.; Vishwakarma, S.; Singh, R.; Ray, B.; Manna, P.P. Doxorubicin loaded pH responsive biodegradable ABA-type Amphiphilic PEG-b-aliphatic Polyketal-b-PEG block copolymer for therapy against aggressive murine lymphoma. Nanomed. Nanotechnol. Biol. Med. 2020, 24, 102128. [Google Scholar] [CrossRef]
- Nguyen, C.T.H.; Webb, R.I.; Lambert, L.K.; Strounina, E.; Lee, E.C.; Parat, M.-O.; McGuckin, M.A.; Popat, A.; Cabot, P.J.; Ross, B.P. Bifunctional Succinylated ε-Polylysine-Coated Mesoporous Silica Nanoparticles for pH-Responsive and Intracellular Drug Delivery Targeting the Colon. ACS Appl. Mater. Interfaces 2017, 9, 9470–9483. [Google Scholar] [CrossRef]
- Wang, X.; Yan, J.-J.; Wang, L.; Pan, D.; Yang, R.; Xu, Y.; Sheng, J.; Huang, Q.; Zhao, H.; Yang, M. Rational Design of Polyphenol-Poloxamer Nanovesicles for Targeting Inflammatory Bowel Disease Therapy. Chem. Mater. 2018, 30, 4073–4080. [Google Scholar] [CrossRef]
- Ding, S.; Cao, S.; Liu, Y.; Lian, Y.; Zhu, A.; Shi, G. Rational Design of a Stimuli-Responsive Polymer Electrode Interface Coupled with in Vivo Microdialysis for Measurement of Sialic Acid in Live Mouse Brain in Alzheimer’s Disease. ACS Sens. 2017, 2, 394–400. [Google Scholar] [CrossRef]
- Tong, Z.; Zhou, J.; Zhong, J.; Tang, Q.; Lei, Z.; Luo, H.; Ma, P.; Liu, X. Glucose- and H2O2-Responsive Polymeric Vesicles Integrated with Microneedle Patches for Glucose-Sensitive Transcutaneous Delivery of Insulin in Diabetic Rats. ACS Appl. Mater. Interfaces 2018, 10, 20014–20024. [Google Scholar] [CrossRef]
- Shamaeli, E.; Alizadeh, N. Functionalized gold nanoparticle-polypyrrole nanobiocomposite with high effective surface area for electrochemical/pH dual stimuli-responsive smart release of insulin. Colloids Surf. B: Biointerfaces 2015, 126, 502–509. [Google Scholar] [CrossRef] [PubMed]
- Poon, R.T.; Borys, N. Lyso-thermosensitive liposomal doxorubicin: An adjuvant to increase the cure rate of radiofrequency ablation in liver cancer. Future Oncol. 2011, 7, 937–945. [Google Scholar] [CrossRef] [PubMed]
- Shaffer, S.A.; Baker-Lee, C.; Kennedy, J.; Lai, M.S.; de Vries, P.; Buhler, K.; Singer, J.W. In vitro and in vivo metabolism of paclitaxel poliglumex: Identification of metabolites and active proteases. Cancer Chemother. Pharmacol. 2007, 59, 537–548. [Google Scholar] [CrossRef] [PubMed]
- Maier-Hauff, K.; Ulrich, F.; Nestler, D.; Niehoff, H.; Wust, P.; Thiesen, B.; Orawa, H.; Budach, V.; Jordan, A. Efficacy and safety of intratumoral thermotherapy using magnetic iron-oxide nanoparticles combined with external beam radiotherapy on patients with recurrent glioblastoma multiforme. J. Neuro-Oncol. 2011, 103, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, J.A.; Shetty, A.M.; Price, R.E.; Stafford, R.J.; Wang, J.C.; Uthamanthil, R.K.; Pham, K.; McNichols, R.J.; Coleman, C.L.; Payne, J.D. Feasibility Study of Particle-Assisted Laser Ablation of Brain Tumors in Orthotopic Canine Model. Cancer Res. 2009, 69, 1659. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Huang, Y.; Kumar, A.; Tan, A.; Jin, S.; Mozhi, A.; Liang, X.J. pH-sensitive nano-systems for drug delivery in cancer therapy. Biotechnol. Adv. 2014, 32, 693–710. [Google Scholar] [CrossRef]
- Wike-Hooley, J.L.; Haveman, J.; Reinhold, H.S. The relevance of tumour pH to the treatment of malignant disease. Radiother. Oncol. 1984, 2, 343–366. [Google Scholar] [CrossRef]
- Engin, K.; Leeper, D.B.; Cater, J.R.; Thistlethwaite, A.J.; Tupchong, L.; McFarlane, J.D. Extracellular pH distribution in human tumours. Int. J. Hyperth. 1995, 11, 211–216. [Google Scholar] [CrossRef]
- Stubbs, M.; McSheehy, P.M.J.; Griffiths, J.R.; Bashford, C.L. Causes and consequences of tumour acidity and implications for treatment. Mol. Med. Today 2000, 6, 15–19. [Google Scholar] [CrossRef]
- Cao, Z.; Wang, X.; Pang, Y.; Cheng, S.; Liu, J. Biointerfacial self-assembly generates lipid membrane coated bacteria for enhanced oral delivery and treatment. Nat. Commun. 2019, 10, 5783. [Google Scholar] [CrossRef]
- Narang, A.S.; Mahato, R.I.; Ajit, S. Targeted Delivery of Small and Macromolecular Drugs; CRC Press/Taylor & Francis Group: Milton Park, UK, 2010. [Google Scholar]
- Zhou, L.; Wang, H.; Li, Y. Stimuli-Responsive Nanomedicines for Overcoming Cancer Multidrug Resistance. Theranostics 2018, 8, 1059–1074. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.A.; Firestone, B.A. pH-dependent equilibrium swelling properties of hydrophobic polyelectrolyte copolymer gels. Macromolecules 1988, 21, 3254–3259. [Google Scholar] [CrossRef]
- Liu, J.; Wang, H.; Yi, X.; Chao, Y.; Geng, Y.; Xu, L.; Yang, K.; Liu, Z. pH-Sensitive Dissociable Nanoscale Coordination Polymers with Drug Loading for Synergistically Enhanced Chemoradiotherapy. Adv. Funct. Mater. 2017, 27, 1703832. [Google Scholar] [CrossRef]
- Sun, L.; Zhou, Y.; Zhou, X.; Ma, L.; Wang, B.; Yu, C.; Wei, H. Synthesis of a Triple-Responsive Double Hydrophilic Block Copolymer Prodrug Using a Reducible RAFT–ATRP Double-Head Agent. ACS Appl. Polym. Mater. 2020. [Google Scholar] [CrossRef]
- Xue, X.; Jin, S.; Zhang, C.; Yang, K.; Huo, S.; Chen, F.; Zou, G.; Liang, X.-J. Probe-Inspired Nano-Prodrug with Dual-Color Fluorogenic Property Reveals Spatiotemporal Drug Release in Living Cells. ACS Nano 2015, 9, 2729–2739. [Google Scholar] [CrossRef]
- Deng, Z.; Zhen, Z.; Hu, X.; Wu, S.; Xu, Z.; Chu, P.K. Hollow chitosan–silica nanospheres as pH-sensitive targeted delivery carriers in breast cancer therapy. Biomaterials 2011, 32, 4976–4986. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.; Cheng, R.; Meng, F.; Deng, C.; Zhong, Z. Micelles Based on Acid Degradable Poly(acetal urethane): Preparation, pH-Sensitivity, and Triggered Intracellular Drug Release. Biomacromolecules 2015, 16, 2228–2236. [Google Scholar] [CrossRef]
- Lan, S.; Liu, Y.; Shi, K.; Ma, D. Acetal-Functionalized Pillar (5)arene: A pH-Responsive and Versatile Nanomaterial for the Delivery of Chemotherapeutic Agents. ACS Appl. Bio Mater. 2020, 3, 2325–2333. [Google Scholar] [CrossRef]
- Yoo, H.S.; Lee, E.A.; Park, T.G. Doxorubicin-conjugated biodegradable polymeric micelles having acid-cleavable linkages. J. Control. Release 2002, 82, 17–27. [Google Scholar] [CrossRef]
- Li, L.; Xu, Y.; Milligan, I.; Fu, L.; Franckowiak, E.A.; Du, W. Synthesis of Highly pH-Responsive Glucose Poly(orthoester). Angew. Chem. Int. Ed. 2013, 52, 13699–13702. [Google Scholar] [CrossRef]
- Cheng, N.; Wang, Y.; Wu, F. Facile fabrication of double-walled polymeric hollow spheres with independent temperature and pH dual-responsiveness for synergetic drug delivery. J. Appl. Polym. Sci. 2016, 133. [Google Scholar] [CrossRef]
- Parrott, M.C.; Finniss, M.; Luft, J.C.; Pandya, A.; Gullapalli, A.; Napier, M.E.; DeSimone, J.M. Incorporation and Controlled Release of Silyl Ether Prodrugs from PRINT Nanoparticles. J. Am. Chem. Soc. 2012, 134, 7978–7982. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.; Zhuang, Y.; Shen, H.; Yang, F.; Wang, X.; Wu, D. Acetal-linked PEGylated paclitaxel prodrugs forming free-paclitaxel-loaded pH-responsive micelles with high drug loading capacity and improved drug delivery. Mater. Sci. Eng. C 2018, 82, 60–68. [Google Scholar] [CrossRef] [PubMed]
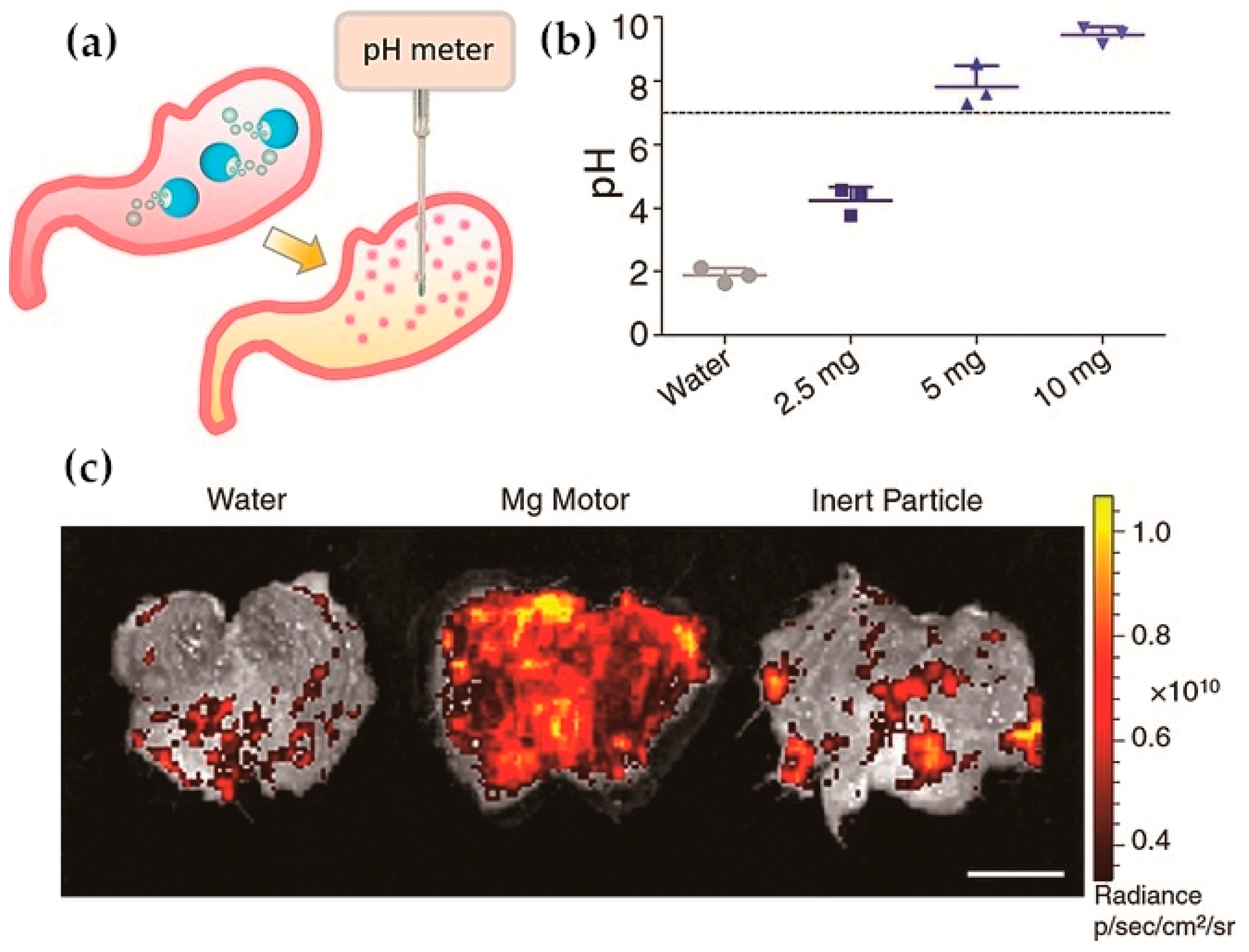
- Li, J.; Angsantikul, P.; Liu, W.; Esteban-Fernández de Ávila, B.; Thamphiwatana, S.; Xu, M.; Sandraz, E.; Wang, X.; Delezuk, J.; Gao, W.; et al. Micromotors Spontaneously Neutralize Gastric Acid for pH-Responsive Payload Release. Angew. Chem. Int. Ed. 2017, 56, 2156–2161. [Google Scholar] [CrossRef] [PubMed]
- Zhao, T.; Huang, G.; Li, Y.; Yang, S.; Ramezani, S.; Lin, Z.; Wang, Y.; Ma, X.; Zeng, Z.; Luo, M.; et al. A transistor-like pH nanoprobe for tumour detection and image-guided surgery. Nat. Biomed. Eng. 2016, 1, 0006. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Liao, S.; Guo, S.; Zheng, X.; Wang, B.; Duan, Z.; Zhang, H.; Gong, Q.; Luo, K. Multistimuli-responsive PEGylated polymeric bioconjugate-based nano-aggregate for cancer therapy. Chem. Eng. J. 2019. [Google Scholar] [CrossRef]
- Wu, L.; Zou, Y.; Deng, C.; Cheng, R.; Meng, F.; Zhong, Z. Intracellular release of doxorubicin from core-crosslinked polypeptide micelles triggered by both pH and reduction conditions. Biomaterials 2013, 34, 5262–5272. [Google Scholar] [CrossRef]
- Alvarez-Lorenzo, C.; Concheiro, A. Smart drug delivery systems: From fundamentals to the clinic. Chem. Commun. 2014, 50, 7743–7765. [Google Scholar] [CrossRef]
- Kuppusamy, P.; Li, H.; Ilangovan, G.; Cardounel, A.J.; Zweier, J.L.; Yamada, K.; Krishna, M.C.; Mitchell, J.B. Noninvasive imaging of tumor redox status and its modification by tissue glutathione levels. Cancer Res. 2002, 62, 307–312. [Google Scholar]
- Shi, F.; Ding, J.; Xiao, C.; Zhuang, X.; He, C.; Chen, L.; Chen, X. Intracellular microenvironment responsive PEGylated polypeptide nanogels with ionizable cores for efficient doxorubicin loading and triggered release. J. Mater. Chem. 2012, 22. [Google Scholar] [CrossRef]
- Wu, G.; Fang, Y.Z.; Yang, S.; Lupton, J.R.; Turner, N.D. Glutathione Metabolism and Its Implications for Health. J. Nutr. 2004, 134, 489–492. [Google Scholar] [CrossRef] [PubMed]
- Jia, X.; He, J.; Shen, L.; Chen, J.; Wei, Z.; Qin, X.; Niu, D.; Li, Y.; Shi, J. Gradient Redox-Responsive and Two-Stage Rocket-Mimetic Drug Delivery System for Improved Tumor Accumulation and Safe Chemotherapy. Nano Lett. 2019, 19, 8690–8700. [Google Scholar] [CrossRef] [PubMed]
- Kildahl, N.K. Bond Energy Data Summarized. J. Chem. Educ. 1995, 72, 423. [Google Scholar] [CrossRef]
- Wei, C.; Zhang, Y.; Song, Z.; Xia, Y.; Xu, H.; Lang, M. Enhanced bioreduction-responsive biodegradable diselenide-containing poly(ester urethane) nanocarriers. Biomater. Sci. 2017, 5, 669–677. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.; He, W.; Xia, Q.; Zhou, W.; Yao, C.; Li, X. Thioether Phosphatidylcholine Liposomes: A Novel ROS-Responsive Platform for Drug Delivery. ACS Appl. Mater. Interfaces 2019, 11, 37411–37420. [Google Scholar] [CrossRef]
- Luo, C.; Sun, J.; Liu, D.; Sun, B.; Miao, L.; Musetti, S.; Li, J.; Han, X.; Du, Y.; Li, L.; et al. Self-Assembled Redox Dual-Responsive Prodrug-Nanosystem Formed by Single Thioether-Bridged Paclitaxel-Fatty Acid Conjugate for Cancer Chemotherapy. Nano Lett. 2016, 16, 5401–5408. [Google Scholar] [CrossRef]
- Engman, L. Synthetic applications of organotellurium chemistry. Acc. Chem. Res. 1985, 18, 274–279. [Google Scholar] [CrossRef]
- Cao, W.; Zhang, X.; Miao, X.; Yang, Z.; Xu, H. γ-Ray-Responsive Supramolecular Hydrogel Based on a Diselenide-Containing Polymer and a Peptide. Angew. Chem. Int. Ed. 2013, 52, 6233–6237. [Google Scholar] [CrossRef]
- Cao, W.; Gu, Y.; Li, T.; Xu, H. Ultra-sensitive ROS-responsive tellurium-containing polymers. Chem. Commun. 2015, 51, 7069–7071. [Google Scholar] [CrossRef]
- Li, F.; Li, T.; Cao, W.; Wang, L.; Xu, H. Near-infrared light stimuli-responsive synergistic therapy nanoplatforms based on the coordination of tellurium-containing block polymer and cisplatin for cancer treatment. Biomaterials 2017, 133, 208–218. [Google Scholar] [CrossRef]
- Liu, C.; Zhu, X.; Wang, X.; Miao, D.; Liang, X.; Wang, C.; Pang, L.; Sun, H.; Kong, D.; Yang, J. Hydrogen peroxide-responsive micelles self-assembled from a peroxalate ester-containing triblock copolymer. Biomater. Sci. 2016, 4, 255–257. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.; Khaja, S.; Velasquez-Castano, J.C.; Dasari, M.; Sun, C.; Petros, J.; Taylor, W.R.; Murthy, N. In vivo imaging of hydrogen peroxide with chemiluminescent nanoparticles. Nat. Mater. 2007, 6, 765–769. [Google Scholar] [CrossRef]
- Saravanakumar, G.; Lee, J.; Kim, J.; Kim, W.J. Visible light-induced singlet oxygen-mediated intracellular disassembly of polymeric micelles co-loaded with a photosensitizer and an anticancer drug for enhanced photodynamic therapy. Chem. Commun. 2015, 51, 9995–9998. [Google Scholar] [CrossRef]
- Xia, X.; Yang, X.; Huang, P.; Yan, D. ROS-Responsive Nanoparticles Formed from RGD–Epothilone B Conjugate for Targeted Cancer Therapy. ACS Appl. Mater. Interfaces 2020, 12, 18301–18308. [Google Scholar] [CrossRef]
- Zhang, T.; Chen, X.; Xiao, C.; Zhuang, X.; Chen, X. Synthesis of a phenylboronic ester-linked PEG-lipid conjugate for ROS-responsive drug delivery. Polym. Chem. 2017, 8, 6209–6216. [Google Scholar] [CrossRef]
- Gong, M.; Yang, J.; Li, Y.; Gu, J. Glutathione-responsive nanoscale MOFs for effective intracellular delivery of the anticancer drug 6-mercaptopurine. Chem. Commun. 2020. [Google Scholar] [CrossRef] [PubMed]
- Mura, S.; Nicolas, J.; Couvreur, P. Stimuli-responsive nanocarriers for drug delivery. Nat. Mater. 2013, 12, 991–1003. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Yang, F.; Xiong, F.; Gu, N. The Smart Drug Delivery System and Its Clinical Potential. Theranostics 2016, 6, 1306–1323. [Google Scholar] [CrossRef]
- Li, M.; Ning, Y.; Chen, J.; Duan, X.; Song, N.; Ding, D.; Su, X.; Yu, Z. Proline Isomerization-Regulated Tumor Microenvironment-Adaptable Self-Assembly of Peptides for Enhanced Therapeutic Efficacy. Nano Lett. 2019, 19, 7965–7976. [Google Scholar] [CrossRef]
- Lee, S.H.; Boire, T.C.; Lee, J.B.; Gupta, M.K.; Zachman, A.L.; Rath, R.; Sung, H.-J. ROS-cleavable proline oligomer crosslinking of polycaprolactone for pro-angiogenic host response. J. Mater. Chem. B 2014, 2, 7109–7113. [Google Scholar] [CrossRef]
- Wilson, D.S.; Dalmasso, G.; Wang, L.; Sitaraman, S.V.; Merlin, D.; Murthy, N. Orally delivered thioketal nanoparticles loaded with TNF-α–siRNA target inflammation and inhibit gene expression in the intestines. Nat. Mater. 2010, 9, 923–928. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, F.; Ke, W.; Mukerabigwi, J.F.; M. Japir, A.A.-W.M.; Ibrahim, A.; Wang, Y.; Zha, Z.; Lu, N.; Zhou, M.; Ge, Z. ROS-Responsive Polymeric Nanocarriers with Photoinduced Exposure of Cell-Penetrating Moieties for Specific Intracellular Drug Delivery. ACS Appl. Mater. Interfaces 2019, 11, 31681–31692. [Google Scholar] [CrossRef] [PubMed]
- Ghadiali, J.E.; Stevens, M.M. Enzyme-Responsive Nanoparticle Systems. Adv. Mater. 2008, 20, 4359–4363. [Google Scholar] [CrossRef]
- López-Otín, C.; Bond, J.S. Proteases: Multifunctional Enzymes in Life and Disease. J. Biol. Chem. 2008, 283, 30433–30437. [Google Scholar] [CrossRef]
- Secret, E.; Kelly, S.J.; Crannell, K.E.; Andrew, J.S. Enzyme-Responsive Hydrogel Microparticles for Pulmonary Drug Delivery. ACS Appl. Mater. Interfaces 2014, 6, 10313–10321. [Google Scholar] [CrossRef]
- Satchi, R.; Connors, T.A.; Duncan, R. PDEPT: Polymer-directed enzyme prodrug therapy. I. HPMA copolymer-cathepsin B and PK1 as a model combination. Br. J. Cancer 2001, 85, 1070. [Google Scholar] [CrossRef]
- Wang, W.; Liang, G.; Zhang, W.; Xing, D.; Hu, X. Cascade-Promoted Photo-Chemotherapy against Resistant Cancers by Enzyme-Responsive Polyprodrug Nanoplatforms. Chem. Mater. 2018, 30, 3486–3498. [Google Scholar] [CrossRef]
- Zhu, L.; Kate, P.; Torchilin, V.P. Matrix Metalloprotease 2-Responsive Multifunctional Liposomal Nanocarrier for Enhanced Tumor Targeting. ACS Nano 2012, 6, 3491–3498. [Google Scholar] [CrossRef]
- Ji, T.; Li, S.; Zhang, Y.; Lang, J.; Ding, Y.; Zhao, X.; Zhao, R.; Li, Y.; Shi, J.; Hao, J.; et al. An MMP-2 Responsive Liposome Integrating Antifibrosis and Chemotherapeutic Drugs for Enhanced Drug Perfusion and Efficacy in Pancreatic Cancer. ACS Appl. Mater. Interfaces 2016, 8, 3438–3445. [Google Scholar] [CrossRef]
- Han, M.; Huang-Fu, M.-Y.; Guo, W.-W.; Guo, N.-N.; Chen, J.; Liu, H.-N.; Xie, Z.-Q.; Lin, M.-T.; Wei, Q.-C.; Gao, J.-Q. MMP-2-Sensitive HA End-Conjugated Poly(amidoamine) Dendrimers via Click Reaction to Enhance Drug Penetration into Solid Tumor. ACS Appl. Mater. Interfaces 2017, 9, 42459–42470. [Google Scholar] [CrossRef]
- Li, J.; Xiao, S.; Xu, Y.; Zuo, S.; Zha, Z.; Ke, W.; He, C.; Ge, Z. Smart Asymmetric Vesicles with Triggered Availability of Inner Cell-Penetrating Shells for Specific Intracellular Drug Delivery. ACS Appl. Mater. Interfaces 2017, 9, 17727–17735. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Ge, Z.; Liu, S. PEG-sheddable polyplex micelles as smart gene carriers based on MMP-cleavable peptide-linked block copolymers. Chem. Commun. 2013, 49, 6974–6976. [Google Scholar] [CrossRef] [PubMed]
- Ke, W.; Li, J.; Zhao, K.; Zha, Z.; Han, Y.; Wang, Y.; Yin, W.; Zhang, P.; Ge, Z. Modular Design and Facile Synthesis of Enzyme-Responsive Peptide-Linked Block Copolymers for Efficient Delivery of Doxorubicin. Biomacromolecules 2016, 17, 3268–3276. [Google Scholar] [CrossRef]
- He, L.; Fan, D.; Liang, W.; Wang, Q.; Fang, J. Matrix Metalloproteinase-Responsive PEGylated Lipid Nanoparticles for Controlled Drug Delivery in the Treatment of Rheumatoid Arthritis. ACS Appl. Bio Mater. 2020, 3, 3276–3284. [Google Scholar] [CrossRef]
- Dennis, E.A.; Cao, J.; Hsu, Y.-H.; Magrioti, V.; Kokotos, G. Phospholipase A2 Enzymes: Physical Structure, Biological Function, Disease Implication, Chemical Inhibition, and Therapeutic Intervention. Chem. Rev. 2011, 111, 6130–6185. [Google Scholar] [CrossRef]
- Graff, J.R.; Konicek, B.W.; Deddens, J.A.; Chedid, M.; Hurst, B.M.; Colligan, B.; Neubauer, B.L.; Carter, H.W.; Carter, J.H. Expression of Group IIa Secretory Phospholipase A2 Increases with Prostate Tumor Grade. Clin. Cancer Res. 2001, 7, 3857. [Google Scholar]
- Yamashita, S.I.; Yamashita, J.I.; Ogawa, M. Overexpression of group II phospholipase A2 in human breast cancer tissues is closely associated with their malignant potency. Br. J. Cancer 1994, 69, 1166–1170. [Google Scholar] [CrossRef]
- Lilian, L.; Astrid, J.F.; Monika, Ø.; Arild, F.; Berit, J. Presence of secretory group IIa and V phospholipase A2 and cytosolic group IVα phospholipase A2 in chondrocytes from patients with rheumatoid arthritis. Clin. Chem. Lab. Med. 2004, 42, 602–610. [Google Scholar] [CrossRef]
- Cummings, B.S.; McHowat, J.; Schnellmann, R.G. Phospholipase A2s in Cell Injury and Death. J. Pharmacol. Exp. Ther. 2000, 294, 793. [Google Scholar]
- Shchegravina, E.S.; Tretiakova, D.S.; Alekseeva, A.S.; Galimzyanov, T.R.; Utkin, Y.N.; Ermakov, Y.A.; Svirshchevskaya, E.V.; Negrebetsky, V.V.; Karpechenko, N.Y.; Chernikov, V.P.; et al. Phospholipidic Colchicinoids as Promising Prodrugs Incorporated into Enzyme-Responsive Liposomes: Chemical, Biophysical, and Enzymological Aspects. Bioconjugate Chem. 2019, 30, 1098–1113. [Google Scholar] [CrossRef]
- Arouri, A.; Mouritsen, O.G. Phospholipase A2-susceptible liposomes of anticancer double lipid-prodrugs. Eur. J. Pharm. Sci. 2012, 45, 408–420. [Google Scholar] [CrossRef] [PubMed]
- Hatzinikolaou, D.G.; Hansen, O.C.; Macris, B.J. A new glucose oxidase fromAspergillus niger: Characterization and regulation studies of enzyme and gene. Appl. Microbiol. Biotechnol. 1996, 46, 371–381. [Google Scholar] [PubMed]
- Pluschkell, S.; Hellmuth, K.; Rinas, U. Kinetics of glucose oxidase excretion by recombinant Aspergillus niger. Biotechnol. Bioeng. 1996, 51, 215–220. [Google Scholar] [CrossRef]
- Volpatti, L.R.; Matranga, M.A.; Cortinas, A.B.; Delcassian, D.; Daniel, K.B.; Langer, R.; Anderson, D.G. Glucose-Responsive Nanoparticles for Rapid and Extended Self-Regulated Insulin Delivery. ACS Nano 2020, 14, 488–497. [Google Scholar] [CrossRef]
- Fu, Y.; Liu, W.; Wang, L.-y.; Zhu, B.-y.; Qu, M.-k.; Yang, L.-q.; Sun, X.; Gong, T.; Zhang, Z.-R.; Lin, Q.; et al. Erythrocyte-Membrane-Camouflaged Nanoplatform for Intravenous Glucose-Responsive Insulin Delivery. Adv. Funct. Mater. 2018, 28, 1802250. [Google Scholar] [CrossRef]
- Chen, W.-H.; Luo, G.-F.; Vázquez-González, M.; Cazelles, R.; Sohn, Y.S.; Nechushtai, R.; Mandel, Y.; Willner, I. Glucose-Responsive Metal–Organic-Framework Nanoparticles Act as “Smart” Sense-and-Treat Carriers. ACS Nano 2018, 12, 7538–7545. [Google Scholar] [CrossRef]
- Belinsky, M.; Jaiswal, A.K. NAD(P)H:Quinone oxidoreductase1 (DT-diaphorase) expression in normal and tumor tissues. Cancer Metastasis Rev. 1993, 12, 103–117. [Google Scholar] [CrossRef]
- Ough, M.; Lewis, A.; Bey, E.A.; Gao, J.; Ritchie, J.M.; Bornmann, W.; Boothman, D.A.; Oberley, L.W.; Cullen, J.J. Efficacy of beta-lapachone in pancreatic cancer treatment: Exploiting the novel, therapeutic target NQO1. Cancer Biol. Ther. 2005, 4, 102–109. [Google Scholar] [CrossRef]
- Yao, C.; Li, Y.; Wang, Z.; Song, C.; Hu, X.; Liu, S. Cytosolic NQO1 Enzyme-Activated Near-Infrared Fluorescence Imaging and Photodynamic Therapy with Polymeric Vesicles. ACS Nano 2020, 14, 1919–1935. [Google Scholar] [CrossRef]
- Weitzel, J.N.; Pooler, P.A.; Mohammed, R.; Levitt, M.D.; Eckfeldt, J.H. A unique case of breast carcinoma producing pancreatic-type isoamylase. Gastroenterology 1988, 94, 519–520. [Google Scholar] [CrossRef]
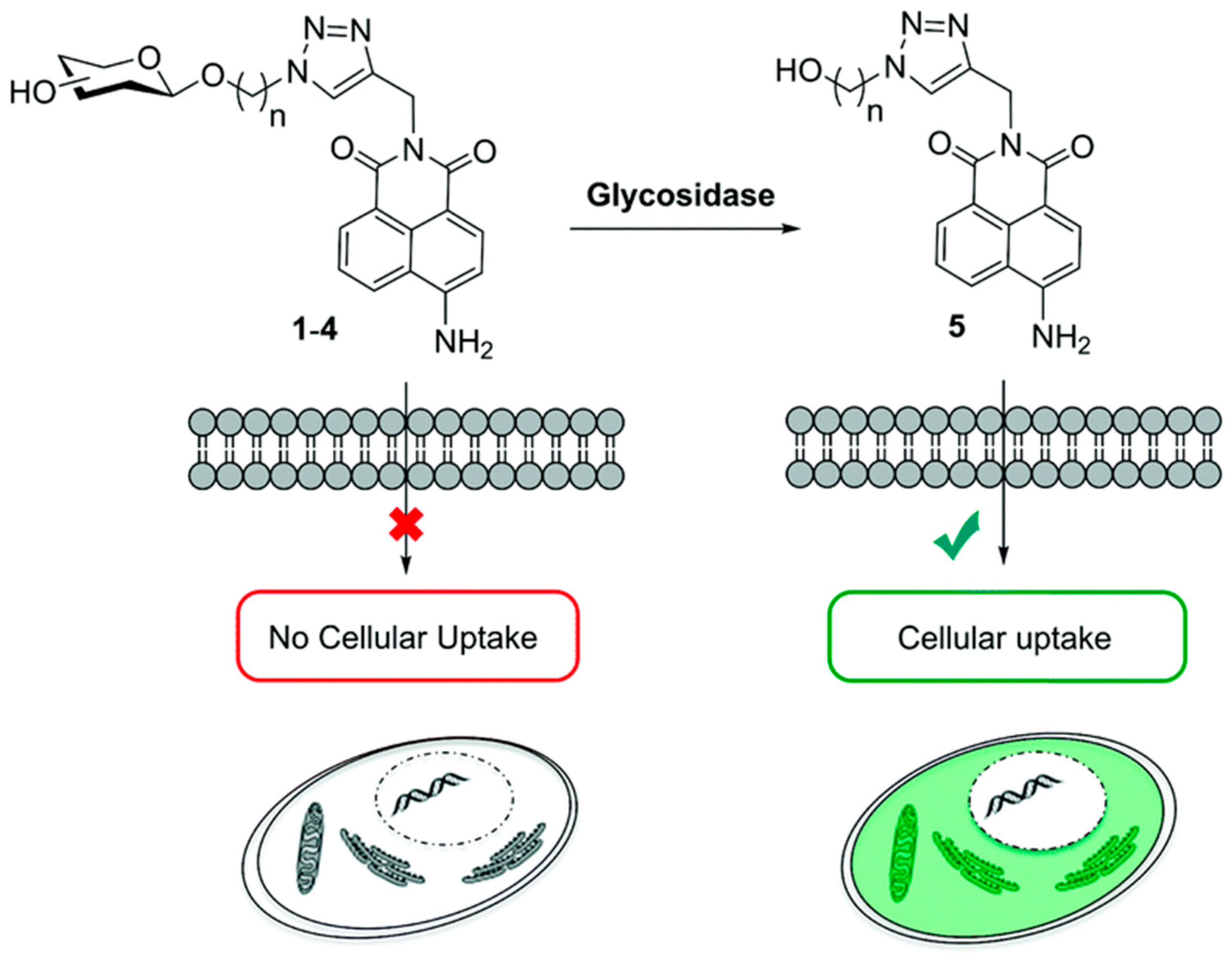
- Calatrava-Pérez, E.; Bright, S.A.; Achermann, S.; Moylan, C.; Senge, M.O.; Veale, E.B.; Williams, D.C.; Gunnlaugsson, T.; Scanlan, E.M. Glycosidase activated release of fluorescent 1,8-naphthalimide probes for tumor cell imaging from glycosylated ‘pro-probes’. Chem. Commun. 2016, 52, 13086–13089. [Google Scholar] [CrossRef] [PubMed]
- Ernsting, M.J.; Murakami, M.; Roy, A.; Li, S.-D. Factors controlling the pharmacokinetics, biodistribution and intratumoral penetration of nanoparticles. J. Control. Release 2013, 172, 782–794. [Google Scholar] [CrossRef]
- Urano, D. Thermal Effects on Cells and Tissues; VSP: Leiden, The Netherlands, 1988. [Google Scholar]
- Cheng, L.; Wang, C.; Feng, L.; Yang, K.; Liu, Z. Functional Nanomaterials for Phototherapies of Cancer. Chem. Rev. 2014, 114, 10869–10939. [Google Scholar] [CrossRef] [PubMed]
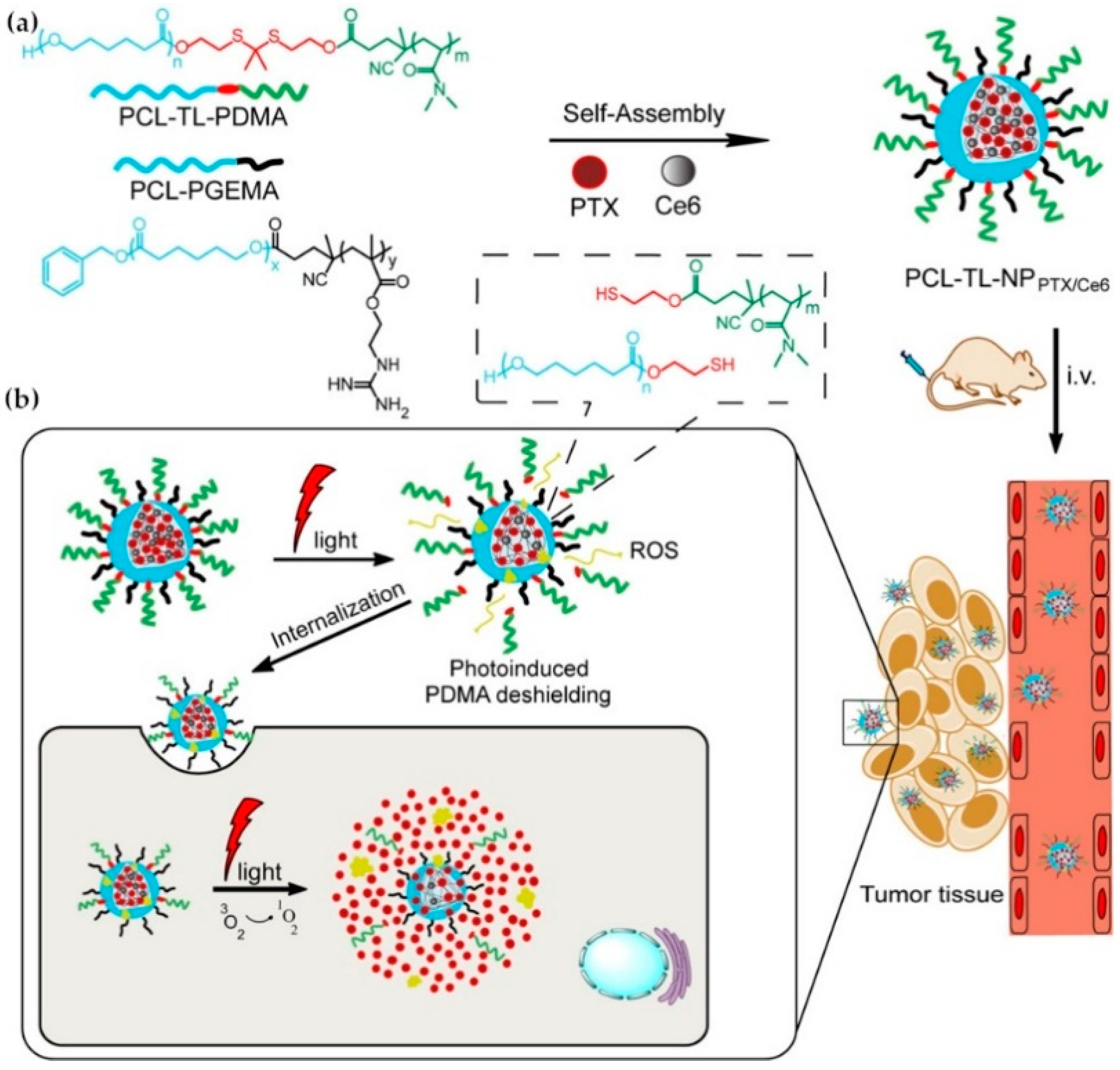
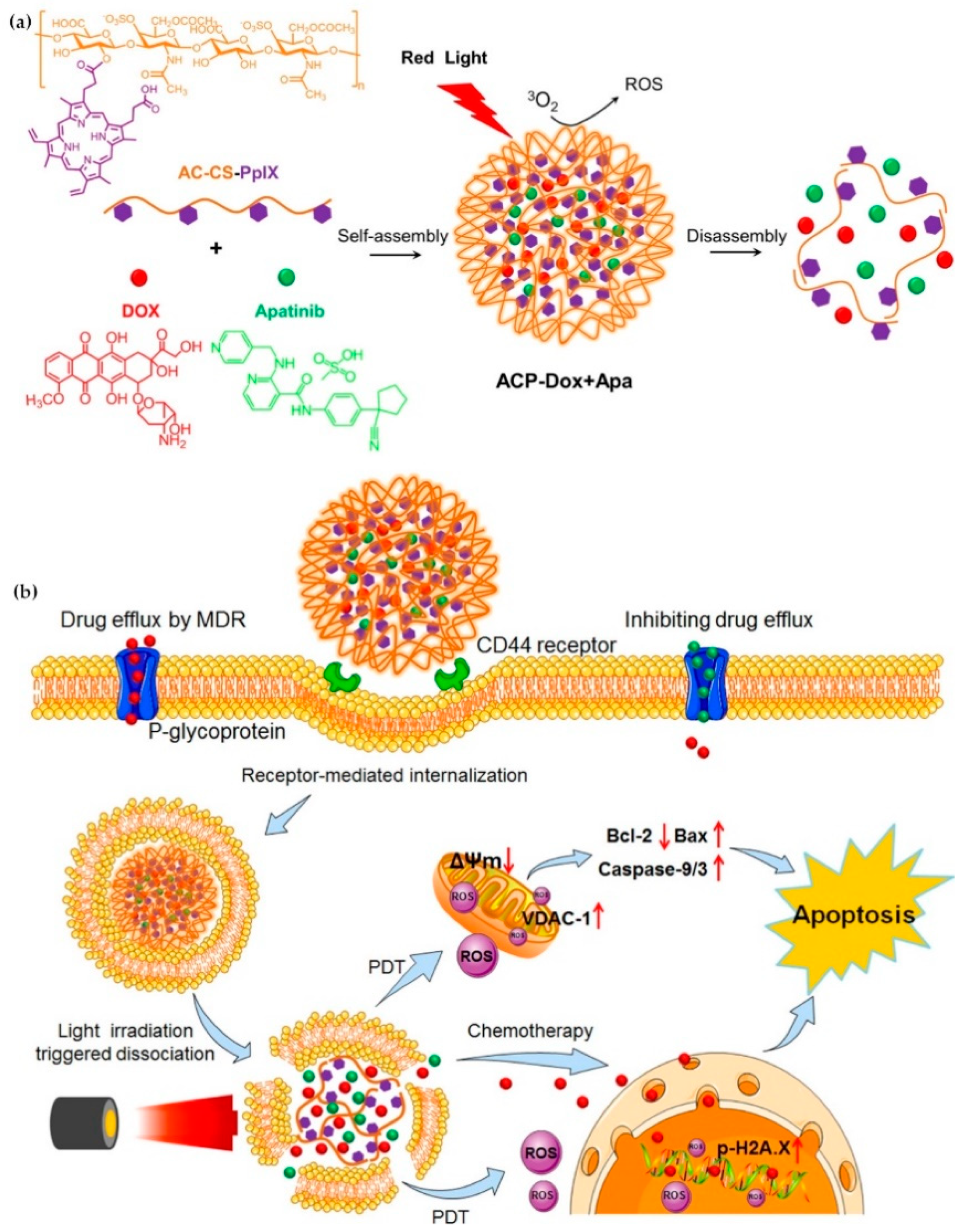
- Wei, X.; Liu, L.; Guo, X.; Wang, Y.; Zhao, J.; Zhou, S. Light-Activated ROS-Responsive Nanoplatform Codelivering Apatinib and Doxorubicin for Enhanced Chemo-Photodynamic Therapy of Multidrug-Resistant Tumors. ACS Appl. Mater. Interfaces 2018, 10, 17672–17684. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Zhao, Y. Applications of Light-Responsive Systems for Cancer Theranostics. ACS Appl. Mater. Interfaces 2018, 10, 21021–21034. [Google Scholar] [CrossRef]
- Mayer, G.; Heckel, A. Biologically Active Molecules with a “Light Switch”. Angew. Chem. Int. Ed. 2006, 45, 4900–4921. [Google Scholar] [CrossRef] [PubMed]
- Carter, K.A.; Shao, S.; Hoopes, M.I.; Luo, D.; Ahsan, B.; Grigoryants, V.M.; Song, W.; Huang, H.; Zhang, G.; Pandey, R.K.; et al. Porphyrin–phospholipid liposomes permeabilized by near-infrared light. Nat. Commun. 2014, 5, 3546. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhang, H.-Y.; Zhang, Z.-Y.; Liu, Y. Tunable Luminescent Lanthanide Supramolecular Assembly Based on Photoreaction of Anthracene. J. Am. Chem. Soc. 2017, 139, 7168–7171. [Google Scholar] [CrossRef]
- Wang, H.; Miao, W.; Wang, F.; Cheng, Y. A Self-Assembled Coumarin-Anchored Dendrimer for Efficient Gene Delivery and Light-Responsive Drug Delivery. Biomacromolecules 2018, 19, 2194–2201. [Google Scholar] [CrossRef]
- Jana, A.; Saha, B.; Banerjee, D.R.; Ghosh, S.K.; Nguyen, K.T.; Ma, X.; Qu, Q.; Zhao, Y.; Singh, N.D.P. Photocontrolled Nuclear-Targeted Drug Delivery by Single Component Photoresponsive Fluorescent Organic Nanoparticles of Acridin-9-Methanol. Bioconjugate Chem. 2013, 24, 1828–1839. [Google Scholar] [CrossRef]
- Ye, Q.; Huo, M.; Zeng, M.; Liu, L.; Peng, L.; Wang, X.; Yuan, J. Photoinduced Reversible Worm-to-Vesicle Transformation of Azo-Containing Block Copolymer Assemblies Prepared by Polymerization-Induced Self-Assembly. Macromolecules 2018, 51, 3308–3314. [Google Scholar] [CrossRef]
- Huang, F.; Liao, W.-C.; Sohn, Y.S.; Nechushtai, R.; Lu, C.-H.; Willner, I. Light-Responsive and pH-Responsive DNA Microcapsules for Controlled Release of Loads. J. Am. Chem. Soc. 2016, 138, 8936–8945. [Google Scholar] [CrossRef] [PubMed]
- LeValley, P.J.; Neelarapu, R.; Sutherland, B.P.; Dasgupta, S.; Kloxin, C.J.; Kloxin, A.M. Photolabile Linkers: Exploiting Labile Bond Chemistry to Control Mode and Rate of Hydrogel Degradation and Protein Release. J. Am. Chem. Soc. 2020, 142, 4671–4679. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.K.; Verma, M.; Silpe, J.; Moody, R.E.; Tang, K.; Hanson, J.J.; Baker, J.R. A photochemical approach for controlled drug release in targeted drug delivery. Bioorganic Med. Chem. 2012, 20, 1281–1290. [Google Scholar] [CrossRef]
- Zhu, X.; Su, Q.; Feng, W.; Li, F. Anti-Stokes shift luminescent materials for bio-applications. Chem. Soc. Rev. 2017, 46, 1025–1039. [Google Scholar] [CrossRef]
- Auzel, F. Upconversion and Anti-Stokes Processes with f and d Ions in Solids. Chem. Rev. 2004, 104, 139–174. [Google Scholar] [CrossRef]
- Zhang, Y.; Lu, G.; Yu, Y.; Zhang, H.; Gao, J.; Sun, Z.; Lu, Y.; Zou, H. NIR-Responsive Copolymer Upconversion Nanocomposites for Triggered Drug Release in Vitro and in Vivo. ACS Appl. Bio Mater. 2019, 2, 495–503. [Google Scholar] [CrossRef]
- Brieke, C.; Rohrbach, F.; Gottschalk, A.; Mayer, G.; Heckel, A. Light-Controlled Tools. Angew. Chem. Int. Ed. 2012, 51, 8446–8476. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, Y.; Song, G.; He, Y.; Zhang, X.; Liu, Y.; Ju, H. A DNA–Azobenzene Nanopump Fueled by Upconversion Luminescence for Controllable Intracellular Drug Release. Angew. Chem. Int. Ed. 2019, 58, 18207–18211. [Google Scholar] [CrossRef]
- Chen, S.; Bian, Q.; Wang, P.; Zheng, X.; Lv, L.; Dang, Z.; Wang, G. Photo, pH and redox multi-responsive nanogels for drug delivery and fluorescence cell imaging. Polym. Chem. 2017, 8, 6150–6157. [Google Scholar] [CrossRef]
- Irie, M. Diarylethenes for Memories and Switches. Chem. Rev. 2000, 100, 1685–1716. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.; Zhao, J.; Zhou, Y.; Ma, J.; Zhao, Y. Reversible Photoswitching of Triplet–Triplet Annihilation Upconversion Using Dithienylethene Photochromic Switches. J. Am. Chem. Soc. 2014, 136, 9256–9259. [Google Scholar] [CrossRef] [PubMed]
- Shirinian, V.Z.; Lvov, A.G.; Krayushkin, M.M.; Lubuzh, E.D.; Nabatov, B.V. Synthesis and Comparative Photoswitching Studies of Unsymmetrical 2,3-Diarylcyclopent-2-en-1-ones. J. Org. Chem. 2014, 79, 3440–3451. [Google Scholar] [CrossRef] [PubMed]
- Kutsunugi, Y.; Kawai, S.; Nakashima, T.; Kawai, T. Photochromic properties of terarylene derivatives having a π-conjugation unit on central aromatic ring. New J. Chem. 2009, 33, 1368–1373. [Google Scholar] [CrossRef]
- Neilson, B.M.; Bielawski, C.W. Photoswitchable Organocatalysis: Using Light to Modulate the Catalytic Activities of N-Heterocyclic Carbenes. J. Am. Chem. Soc. 2012, 134, 12693–12699. [Google Scholar] [CrossRef]
- Wu, T.; Tang, H.; Bohne, C.; Branda, N.R. Reporting the Release of Caged Species by a Combination of Two Sequential Photoreactions, a Molecular Switch, and One Color of Light. Angew. Chem. Int. Ed. 2012, 51, 2741–2744. [Google Scholar] [CrossRef]
- Deng, X.; Liebeskind, L.S. A Contribution to the Design of Molecular Switches: Novel Acid-Mediated Ring-Closing−Photochemical Ring-Opening of 2,3-Bis(heteroaryl)quinones (Heteroaryl = Thienyl, Furanyl, Pyrrolyl). J. Am. Chem. Soc. 2001, 123, 7703–7704. [Google Scholar] [CrossRef]
- Suzuki, K.; Ubukata, T.; Yokoyama, Y. Dual-mode fluorescence switching of photochromic bisthiazolylcoumarin. Chem. Commun. 2012, 48, 765–767. [Google Scholar] [CrossRef]
- Ding, H.; Liu, W.; Pu, S. Synthesis and Photochromism of a Novel Diarylethene Compound Containing Terminal Benzothiazole Unit. In Proceedings of the 2012 Symposium on Photonics and Optoelectronics, Shanghai, China, 21–23 May 2012; pp. 1–3. [Google Scholar]
- Shell, T.A.; Lawrence, D.S. Vitamin B12: A Tunable, Long Wavelength, Light-Responsive Platform for Launching Therapeutic Agents. Acc. Chem. Res. 2015, 48, 2866–2874. [Google Scholar] [CrossRef] [PubMed]
- Marvin, C.M.; Ding, S.; White, R.E.; Orlova, N.; Wang, Q.; Zywot, E.M.; Vickerman, B.M.; Harr, L.; Tarrant, T.K.; Dayton, P.A.; et al. On Command Drug Delivery via Cell-Conveyed Phototherapeutics. Small 2019, 15, 1901442. [Google Scholar] [CrossRef]
- Sun, W.; Thiramanas, R.; Slep, L.D.; Zeng, X.; Mailänder, V.; Wu, S. Photoactivation of Anticancer Ru Complexes in Deep Tissue: How Deep Can We Go? Chem. A Eur. J. 2017, 23, 10832–10837. [Google Scholar] [CrossRef] [PubMed]
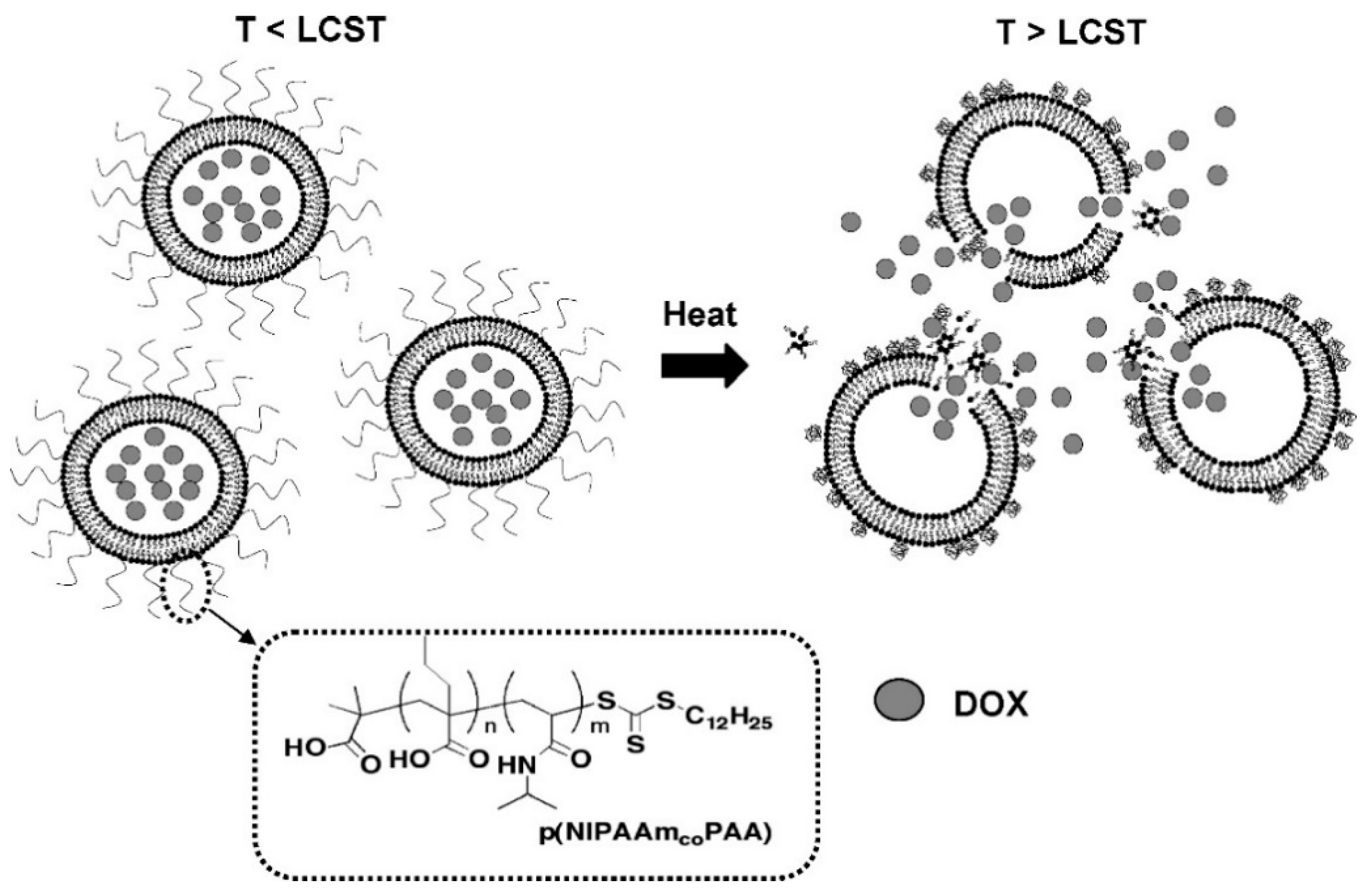
- Karimi, M.; Sahandi Zangabad, P.; Ghasemi, A.; Amiri, M.; Bahrami, M.; Malekzad, H.; Ghahramanzadeh Asl, H.; Mahdieh, Z.; Bozorgomid, M.; Ghasemi, A.; et al. Temperature-Responsive Smart Nanocarriers for Delivery of Therapeutic Agents: Applications and Recent Advances. ACS Appl. Mater. Interfaces 2016, 8, 21107–21133. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Yang, P.; Dai, Y.; Ma, P.a.; Li, X.; Cheng, Z.; Hou, Z.; Kang, X.; Li, C.; Lin, J. Multifunctional Up-Converting Nanocomposites with Smart Polymer Brushes Gated Mesopores for Cell Imaging and Thermo/pH Dual-Responsive Drug Controlled Release. Adv. Funct. Mater. 2013, 23, 4067–4078. [Google Scholar] [CrossRef]
- Coughlan, D.C.; Corrigan, O.I. Release Kinetics of Benzoic Acid and its Sodium Salt from a Series of Poly(N-Isopropylacrylamide) Matrices with Various Percentage Crosslinking. J. Pharm. Sci. 2008, 97, 318–330. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Chen, H.; Xu, L.; Gu, Y. The targeted behavior of thermally responsive nanohydrogel evaluated by NIR system in mouse model. J. Control. Release 2008, 131, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Beija, M.; Marty, J.-D.; Destarac, M. Thermoresponsive poly(N-vinyl caprolactam)-coated gold nanoparticles: Sharp reversible response and easy tunability. Chem. Commun. 2011, 47, 2826–2828. [Google Scholar] [CrossRef]
- Xu, J.-W.; Ge, X.; Lv, L.-H.; Xu, F.; Luo, Y.-L. Dual-Stimuli-Responsive Paclitaxel Delivery Nanosystems from Chemically Conjugate Self-Assemblies for Carcinoma Treatment. Macromol. Rapid Commun. 2018, 39, 1800628. [Google Scholar] [CrossRef]
- Kashyap, S.; Singh, N.; Surnar, B.; Jayakannan, M. Enzyme and Thermal Dual Responsive Amphiphilic Polymer Core–Shell Nanoparticle for Doxorubicin Delivery to Cancer Cells. Biomacromolecules 2016, 17, 384–398. [Google Scholar] [CrossRef]
- Ta, T.; Convertine, A.J.; Reyes, C.R.; Stayton, P.S.; Porter, T.M. Thermosensitive Liposomes Modified with Poly(N-isopropylacrylamide-co-propylacrylic acid) Copolymers for Triggered Release of Doxorubicin. Biomacromolecules 2010, 11, 1915–1920. [Google Scholar] [CrossRef]
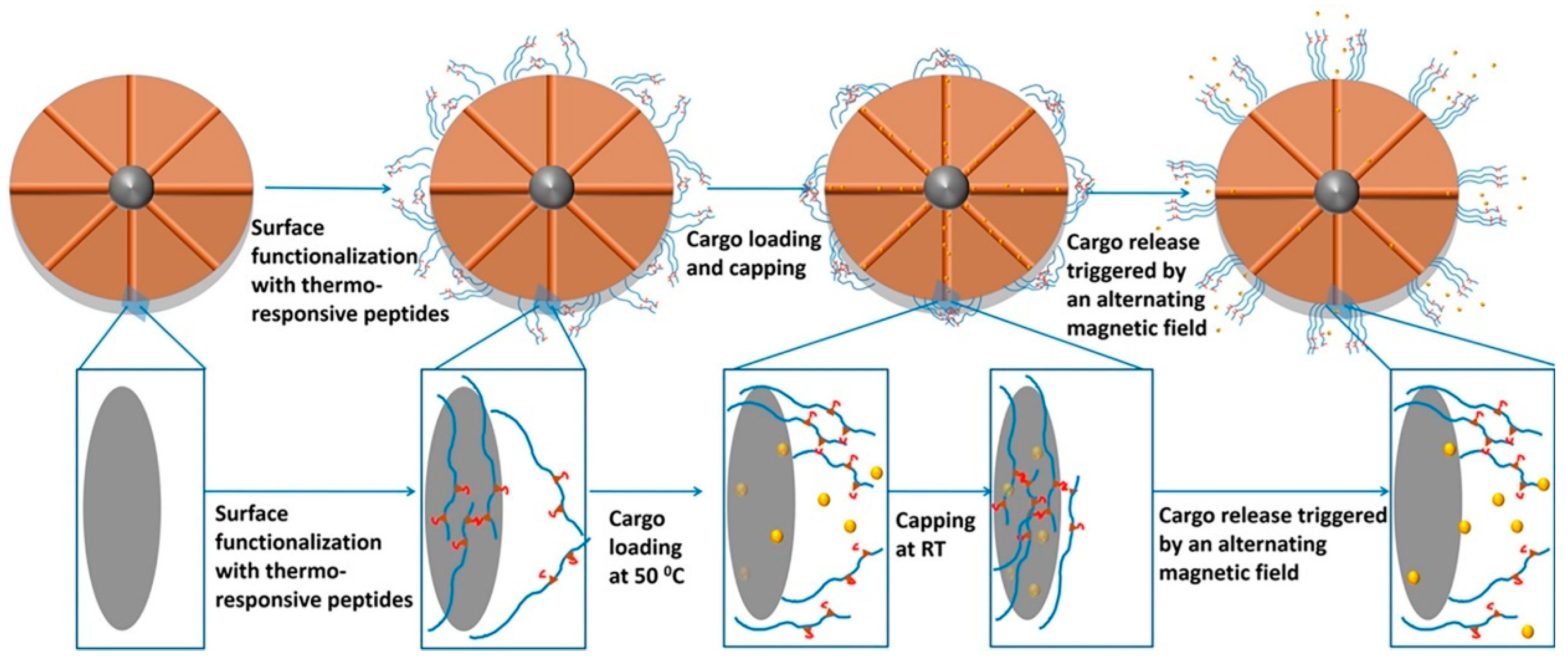
- Freeman, M.W.; Arrott, A.; Watson, J.H.L. Magnetism in Medicine. J. Appl. Phys. 1960, 31, S404–S405. [Google Scholar] [CrossRef]
- Louguet, S.; Rousseau, B.; Epherre, R.; Guidolin, N.; Goglio, G.; Mornet, S.; Duguet, E.; Lecommandoux, S.; Schatz, C. Thermoresponsive polymer brush-functionalized magnetic manganite nanoparticles for remotely triggered drug release. Polym. Chem. 2012, 3, 1408–1417. [Google Scholar] [CrossRef]
- Pernia Leal, M.; Torti, A.; Riedinger, A.; La Fleur, R.; Petti, D.; Cingolani, R.; Bertacco, R.; Pellegrino, T. Controlled Release of Doxorubicin Loaded within Magnetic Thermo-responsive Nanocarriers under Magnetic and Thermal Actuation in a Microfluidic Channel. ACS Nano 2012, 6, 10535–10545. [Google Scholar] [CrossRef] [PubMed]
- Ruan, L.; Chen, W.; Wang, R.; Lu, J.; Zink, J.I. Magnetically Stimulated Drug Release Using Nanoparticles Capped by Self-Assembling Peptides. ACS Appl. Mater. Interfaces 2019, 11, 43835–43842. [Google Scholar] [CrossRef]
- Widder, K.J.; Senyei, A.E.; Scarpelli, D.G. Magnetic Microspheres: A Model System for Site Specific Drug Delivery in Vivo. Proc. Soc. Exp. Biol. Med. 1978, 158, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Mah, C.; Fraites, T.J.; Zolotukhin, I.; Song, S.; Flotte, T.R.; Dobson, J.; Batich, C.; Byrne, B.J. Improved Method of Recombinant AAV2 Delivery for Systemic Targeted Gene Therapy. Mol. Ther. 2002, 6, 106–112. [Google Scholar] [CrossRef] [PubMed]
- Scherer, F.; Anton, M.; Schillinger, U.; Henke, J.; Bergemann, C.; Krüger, A.; Gänsbacher, B.; Plank, C. Magnetofection: Enhancing and targeting gene delivery by magnetic force in vitro and in vivo. Gen. Ther. 2002, 9, 102–109. [Google Scholar] [CrossRef]
- Apfel, R.E. Acoustic cavitation: A possible consequence of biomedical uses of ultrasound. Br. J. Cancer Suppl. 1982, 5, 140–146. [Google Scholar]
- Klibanov, A.L. Microbubble Contrast Agents: Targeted Ultrasound Imaging and Ultrasound-Assisted Drug-Delivery Applications. Investig. Radiol. 2006, 41, 354–362. [Google Scholar] [CrossRef]
- Chen, Y.; Liang, Y.; Jiang, P.; Li, F.; Yu, B.; Yan, F. Lipid/PLGA Hybrid Microbubbles as a Versatile Platform for Noninvasive Image-Guided Targeted Drug Delivery. ACS Appl. Mater. Interfaces 2019, 11, 41842–41852. [Google Scholar] [CrossRef]
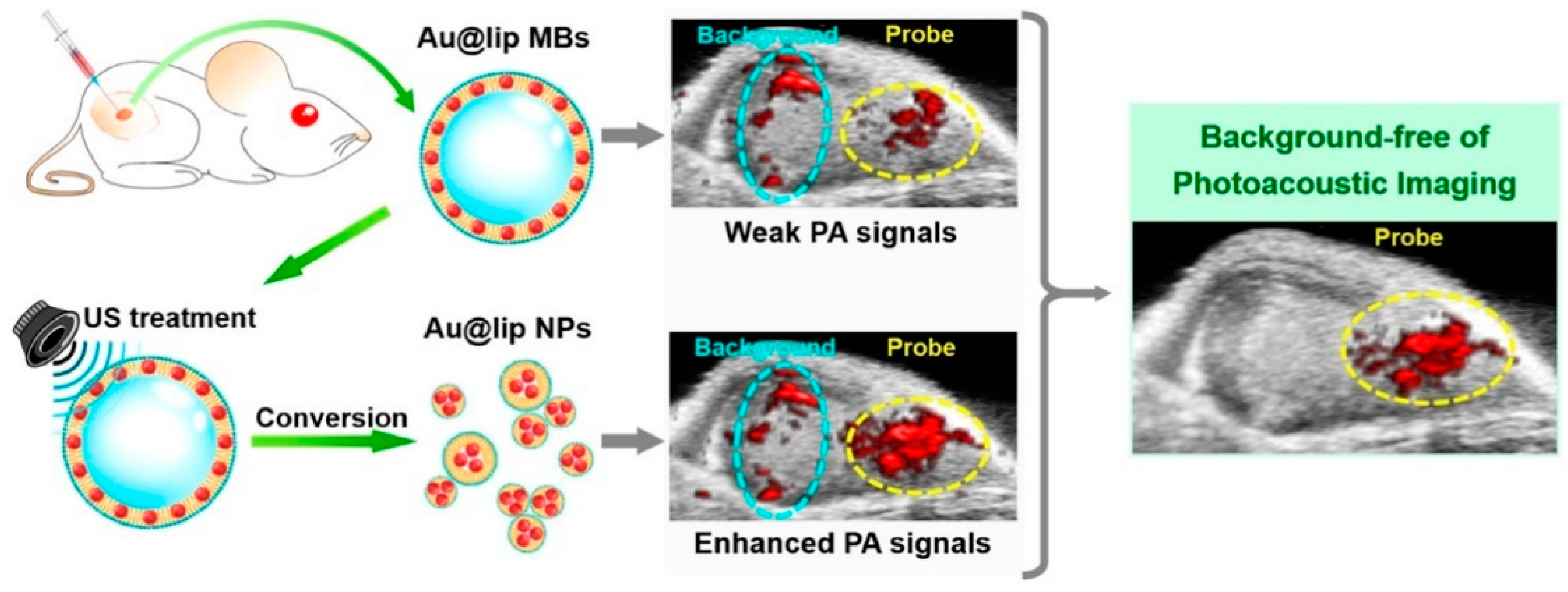
- Huynh, E.; Leung, B.Y.C.; Helfield, B.L.; Shakiba, M.; Gandier, J.-A.; Jin, C.S.; Master, E.R.; Wilson, B.C.; Goertz, D.E.; Zheng, G. In situ conversion of porphyrin microbubbles to nanoparticles for multimodality imaging. Nat. Nanotechnol. 2015, 10, 325–332. [Google Scholar] [CrossRef]
- Meng, Z.; Zhou, X.; She, J.; Zhang, Y.; Feng, L.; Liu, Z. Ultrasound-Responsive Conversion of Microbubbles to Nanoparticles to Enable Background-Free in Vivo Photoacoustic Imaging. Nano Lett. 2019, 19, 8109–8117. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.-Y.; Sha, W.-H.; Zhou, Y.-J.; Nie, Y.-Q. Short and long term efficacy of high intensity focused ultrasound therapy for advanced hepatocellular carcinoma. J. Gastroenterol. Hepatol. 2007, 22, 2148–2154. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.; Wang, Z.-B.; Chen, W.-Z.; Wang, W.; Gui, Y.; Zhang, M.; Zheng, G.; Zhou, Y.; Xu, G.; Li, M.; et al. Extracorporeal high intensity focused ultrasound ablation in the treatment of 1038 patients with solid carcinomas in China: An overview. Ultrason. Sonochem. 2004, 11, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Carl, P.L.; Chakravarty, P.K.; Katzenellenbogen, J.A. A novel connector linkage applicable in prodrug design. J. Med. Chem. 1981, 24, 479–480. [Google Scholar] [CrossRef] [PubMed]
- Dillon, K.M.; Carrazzone, R.J.; Wang, Y.; Powell, C.R.; Matson, J.B. Polymeric Persulfide Prodrugs: Mitigating Oxidative Stress through Controlled Delivery of Reactive Sulfur Species. ACS Macro Lett. 2020, 606–612. [Google Scholar] [CrossRef]
- Khodade, V.S.; Pharoah, B.M.; Paolocci, N.; Toscano, J.P. Alkylamine-Substituted Perthiocarbamates: Dual Precursors to Hydropersulfide and Carbonyl Sulfide with Cardioprotective Actions. J. Am. Chem. Soc. 2020, 142, 4309–4316. [Google Scholar] [CrossRef]
- Scales, S.J.; Tsai, S.P.; Zacharias, N.; Cruz-Chuh, J.D.; Bullen, G.; Velasquez, E.; Chang, J.; Bruguera, E.; Kozak, K.R.; Sadowsky, J. Development of a Cysteine-Conjugatable Disulfide FRET Probe: Influence of Charge on Linker Cleavage and Payload Trafficking for an Anti-HER2 Antibody Conjugate. Bioconjugate Chem. 2019, 30, 3046–3056. [Google Scholar] [CrossRef]
- Singh, H.; Tiwari, K.; Tiwari, R.; Pramanik, S.K.; Das, A. Small Molecule as Fluorescent Probes for Monitoring Intracellular Enzymatic Transformations. Chem. Rev. 2019, 119, 11718–11760. [Google Scholar] [CrossRef]
- Gisbert-Garzarán, M.; Berkmann, J.C.; Giasafaki, D.; Lozano, D.; Spyrou, K.; Manzano, M.; Steriotis, T.; Duda, G.N.; Schmidt-Bleek, K.; Charalambopoulou, G.; et al. Engineered pH-Responsive Mesoporous Carbon Nanoparticles for Drug Delivery. ACS Appl. Mater. Interfaces 2020, 12, 14946–14957. [Google Scholar] [CrossRef]
- Jäger, E.; Sincari, V.; Albuquerque, L.J.C.; Jäger, A.; Humajova, J.; Kucka, J.; Pankrac, J.; Paral, P.; Heizer, T.; Janouskova, O.; et al. Reactive Oxygen Species (ROS)-Responsive Polymersomes with Site-Specific Chemotherapeutic Delivery into Tumors via Spacer Design Chemistry. Biomacromolecules 2020, 21, 1437–1449. [Google Scholar] [CrossRef]
- Jacquemet, A.; Rihn, S.; Ulrich, G.; Renard, P.Y.; Romieu, A.; Ziessel, R. Rational Design of Latent Fluorophores from Water-Soluble Hydroxyphenyltriazine Dyes Suitable for Lipase Sensing. Eur. J. Org. Chem. 2015, 2015, 1664–1669. [Google Scholar] [CrossRef]
- Alouane, A.; Labruère, R.; Silvestre, K.J.; Le Saux, T.; Schmidt, F.; Jullien, L. Disassembly Kinetics of Quinone-Methide-Based Self-Immolative Spacers that Contain Aromatic Nitrogen Heterocycles. Chem. Asian J. 2014, 9, 1334–1340. [Google Scholar] [CrossRef]
- Sáez, J.A.; Escuder, B.; Miravet, J.F. Supramolecular hydrogels for enzymatically triggered self-immolative drug delivery. Tetrahedron 2010, 66, 2614–2618. [Google Scholar] [CrossRef]
- Gnaim, S.; Shabat, D. ChemInform Abstract: Quinone-Methide Species, A Gateway to Functional Molecular Systems: From Self-Immolative Dendrimers to Long-Wavelength Fluorescent Dyes. Acc. Chem. Res. 2015, 45, 2970. [Google Scholar] [CrossRef]
- Senter, P.D.; Pearce, W.E.; Greenfield, R.S. Development of a drug-release strategy based on the reductive fragmentation of benzyl carbamate disulfides. J. Org. Chem. 1990, 55, 2975–2978. [Google Scholar] [CrossRef]
- Paciotti, G.F.; Zhao, J.; Cao, S.; Brodie, P.J.; Tamarkin, L.; Huhta, M.; Myer, L.D.; Friedman, J.; Kingston, D.G.I. Synthesis and Evaluation of Paclitaxel-Loaded Gold Nanoparticles for Tumor-Targeted Drug Delivery. Bioconjug. Chem. 2016, 27, 2646–2657. [Google Scholar] [CrossRef]
- Weinstain, R.; Segal, E.; Satchi-Fainaro, R.; Shabat, D. Real-time monitoring of drug release. Chem. Commun. 2010, 46, 553–555. [Google Scholar] [CrossRef]
- Shamis, M.; Prof, D.S. Single-Triggered AB6 Self-Immolative Dendritic Amplifiers. Chemistry 2007, 13, 4523–4528. [Google Scholar] [CrossRef]
- de Groot, F.M.H.; Loos, W.J.; Koekkoek, R.; Van Berkom, L.W.A.; Busscher, G.F.; Seelen, A.E.; Albrecht, C.; De Bruijn, P.; Scheeren, H.W. Elongated Multiple Electronic Cascade and Cyclization Spacer Systems in Activatible Anticancer Prodrugs for Enhanced Drug Release. J. Org. Chem. 2001, 66, 8815–8830. [Google Scholar] [CrossRef]
- Shan, D.; Nicolaou, M.G.; Borchardt, R.T.; Wang, B. Prodrug strategies based on intramolecular cyclization reactions. J. Pharm. Sci. 1997, 86, 765–767. [Google Scholar] [CrossRef]
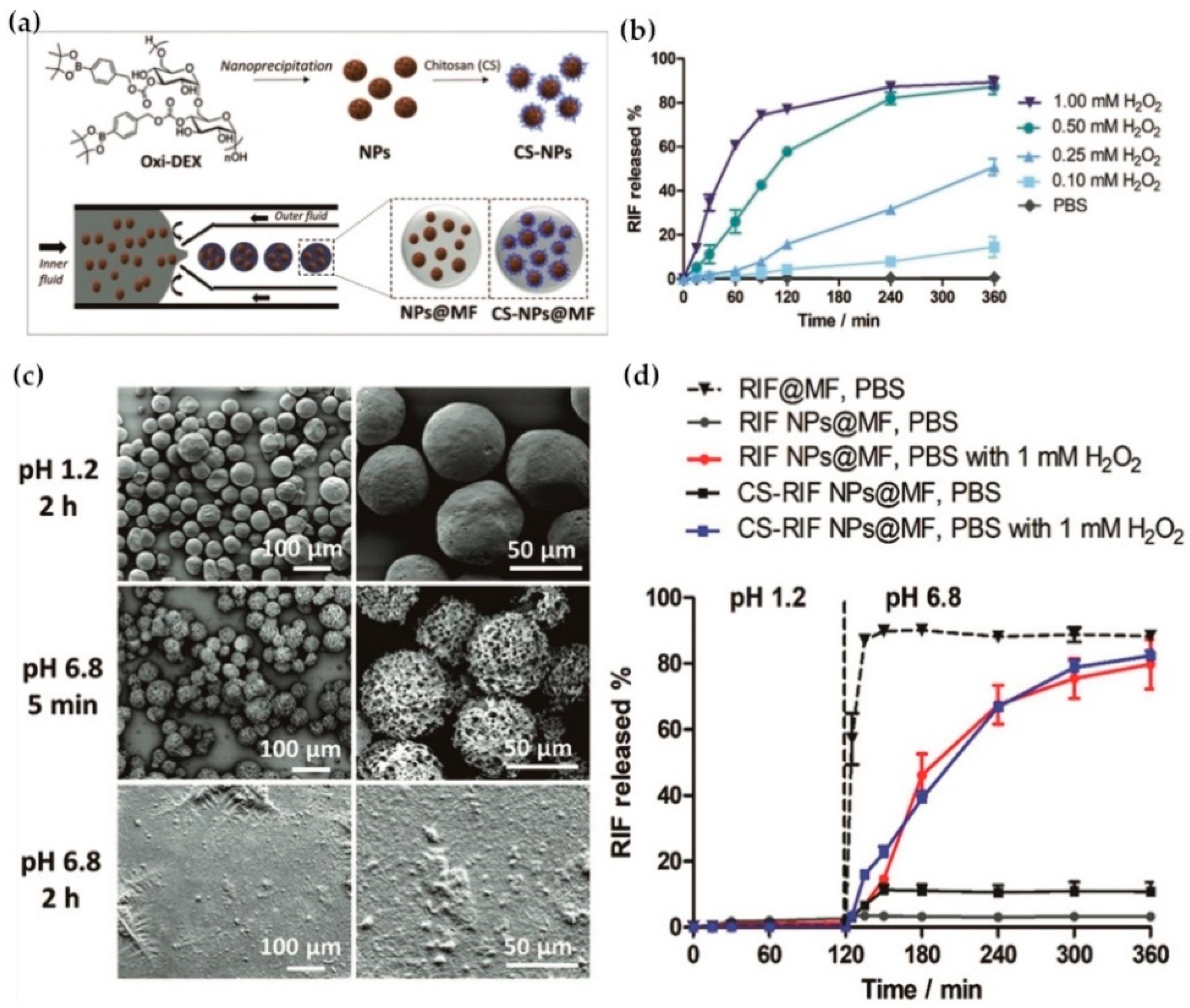
- Qiu, F.Y.; Zhang, M.; Du, F.S.; Li, Z.C. Oxidation Degradable Aliphatic Polycarbonates with Pendent Phenylboronic Ester. Macromolecules 2017, 50, 23–34. [Google Scholar] [CrossRef]
- Huvelle, S.; Alouane, A.; Saux, T.L.; Jullien, L.; Schmidt, F. Syntheses and kinetic studies of cyclisation-based self-immolative spacers. Org. Biomol. Chem. 2017, 15, 3435–3443. [Google Scholar] [CrossRef] [PubMed]
- Esser-Kahn, A.P.; Sottos, N.R.; White, S.R.; Moore, J.S. Programmable Microcapsules from Self-Immolative Polymers. J. Am. Chem. Soc. 2010, 132, 10266–10268. [Google Scholar] [CrossRef] [PubMed]
- Juárez, L.A.; Añón, E.; Giménez, C.; Sancenón, F.; Martínez-Máñez, R.; Costero, A.M.; Gaviña, P.; Parra, M.; Bernardos, A. Self-Immolative Linkers as Caps for the Design of Gated Silica Mesoporous Supports. Chem. Eur. J. 2016, 22, 14126–14130. [Google Scholar] [CrossRef]
- de Gracia Lux, C.; Almutairi, A. Intramolecular Cyclization for Stimuli-Controlled Depolymerization of Polycaprolactone Particles Leading to Disassembly and Payload Release. ACS Macro Lett. 2013, 2, 432–435. [Google Scholar] [CrossRef][Green Version]
- Cerda, M.M.; Mancuso, J.L.; Mullen, E.J.; Hendon, C.H.; Pluth, M.D. Use of Dithiasuccinoyl-Caged Amines Enables COS/H2S Release Lacking Electrophilic Byproducts. Chem. Eur. J. 2020, 26, 5374–5380. [Google Scholar] [CrossRef]
- Warnecke, A.; Kratz, F. 2,4-Bis(hydroxymethyl)aniline as a Building Block for Oligomers with Self-Eliminating and Multiple Release Properties. J. Org. Chem. 2008, 73, 1546–1552. [Google Scholar] [CrossRef]
- Polaske, N.W.; Szalai, M.L.; Shanahan, C.S.; McGrath, D.V. Convergent Synthesis of Geometrically Disassembling Dendrimers using Cu(I)-Catalyzed C−O Bond Formation. Org. Lett. 2010, 12, 4944–4947. [Google Scholar] [CrossRef]
- Deng, Z.; Hu, J.; Liu, S. Disulfide-Based Self-Immolative Linkers and Functional Bioconjugates for Biological Applications. Macromol. Rapid Commun. 2020, 41, e1900531. [Google Scholar] [CrossRef]
- Daniel, K.B.; Callmann, C.E.; Gianneschi, N.C.; Cohen, S.M. Dual-responsive nanoparticles release cargo upon exposure to matrix metalloproteinase and reactive oxygen species. Chem. Commun. 2016, 52, 2126–2128. [Google Scholar] [CrossRef]
- Pallu, J.; Rabin, C.; Creste, G.; Branca, M.; Mavré, F.; Limoges, B. Exponential Molecular Amplification by H2O2-Mediated Autocatalytic Deprotection of Boronic Ester Probes to Redox Cyclers. Chem. Eur. J. 2019, 25, 7534–7546. [Google Scholar] [CrossRef] [PubMed]
- Robbins, J.S.; Schmid, K.M.; Phillips, S.T. Effects of Electronics, Aromaticity, and Solvent Polarity on the Rate of Azaquinone–Methide-Mediated Depolymerization of Aromatic Carbamate Oligomers. J. Org. Chem. 2013, 78, 3159–3169. [Google Scholar] [CrossRef] [PubMed]
- Peiró Cadahía, J.; Bondebjerg, J.; Hansen, C.A.; Previtali, V.; Hansen, A.E.; Andresen, T.L.; Clausen, M.H. Synthesis and Evaluation of Hydrogen Peroxide Sensitive Prodrugs of Methotrexate and Aminopterin for the Treatment of Rheumatoid Arthritis. J. Med. Chem. 2018, 61, 3503–3515. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Shen, Y.; Meng, X.; Wu, Y.; Zhao, Y.; Wu, C. Stabilizing p-Dithiobenzyl Urethane Linkers without Rate-Limiting Self-Immolation for Traceless Drug Release. ChemMedChem 2019, 14, 1196–1203. [Google Scholar] [CrossRef]
- Sitnikov, N.S.; Malysheva, Y.B.; Fedorov, A.Y.; Schmalz, H.-G. Design and Synthesis of New Protease-Triggered CO-Releasing Peptide–Metal-Complex Conjugates. Eur. J. Org. Chem. 2019, 2019, 6830–6837. [Google Scholar] [CrossRef]
- Sagi, A.; Weinstain, R.; Karton, N.; Shabat, D. Self-Immolative Polymers. J. Am. Chem. Soc. 2008, 130, 5434–5435. [Google Scholar] [CrossRef]
- Gnaim, S.; Shabat, D. Activity-Based Optical Sensing Enabled by Self-Immolative Scaffolds: Monitoring of Release Events by Fluorescence or Chemiluminescence Output. Acc. Chem. Res. 2019, 52, 2806–2817. [Google Scholar] [CrossRef]
- Duimstra, J.A.; Femia, F.J.; Meade, T.J. A Gadolinium Chelate for Detection of β-Glucuronidase: A Self-Immolative Approach. J. Am. Chem. Soc. 2005, 127, 12847–12855. [Google Scholar] [CrossRef]
- Prijovich, Z.M.; Burnouf, P.-A.; Chou, H.-C.; Huang, P.-T.; Chen, K.-C.; Cheng, T.-L.; Leu, Y.-L.; Roffler, S.R. Synthesis and Antitumor Properties of BQC-Glucuronide, a Camptothecin Prodrug for Selective Tumor Activation. Mol. Pharm. 2016, 13, 1242–1250. [Google Scholar] [CrossRef]
- Leu, Y.-L.; Chen, C.-S.; Wu, Y.-J.; Chern, J.-W. Benzyl Ether-Linked Glucuronide Derivative of 10-Hydroxycamptothecin Designed for Selective Camptothecin-Based Anticancer Therapy. J. Med. Chem. 2008, 51, 1740–1746. [Google Scholar] [CrossRef]
- Park, S.; Kim, S.Y.; Cho, J.; Jung, D.; Seo, D.; Lee, J.; Lee, S.; Yun, S.; Lee, H.; Park, O.; et al. Aryl Sulfate is a Useful Motif for Conjugating and Releasing Phenolic Molecules: Sulfur Fluorine Exchange Click Chemistry Enables Discovery of Ortho-Hydroxy-Protected Aryl Sulfate Linker. Bioconjugate Chem. 2019, 30, 1957–1968. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Kim, Y. An improved fluorogenic substrate for the detection of alkaline phosphatase activity. Bioorg. Med. Chem. Lett. 2013, 23, 2332–2335. [Google Scholar] [CrossRef] [PubMed]
- Subbaiah, M.A.M.; Mandlekar, S.; Desikan, S.; Ramar, T.; Subramani, L.; Annadurai, M.; Desai, S.D.; Sinha, S.; Jenkins, S.M.; Krystal, M.R.; et al. Design, Synthesis, and Pharmacokinetic Evaluation of Phosphate and Amino Acid Ester Prodrugs for Improving the Oral Bioavailability of the HIV-1 Protease Inhibitor Atazanavir. J. Med. Chem. 2019, 62, 3553–3574. [Google Scholar] [CrossRef] [PubMed]
- Alouane, A.; Labruère, R.; Le Saux, T.; Aujard, I.; Dubruille, S.; Schmidt, F.; Jullien, L. Light Activation for the Versatile and Accurate Kinetic Analysis of Disassembly of Self-Immolative Spacers. Chem. A Eur. J. 2013, 19, 11717–11724. [Google Scholar] [CrossRef]
- Procházková, E.; Filo, J.; Cigáň, M.; Baszczyňski, O. Sterically-Controlled Self-Immolation in Phosphoramidate Linkers Triggered by Light. Eur. J. Org. Chem. 2020, 2020, 897–906. [Google Scholar] [CrossRef]
- de Gracia Lux, C.; McFearin, C.L.; Joshi-Barr, S.; Sankaranarayanan, J.; Fomina, N.; Almutairi, A. Single UV or Near IR Triggering Event Leads to Polymer Degradation into Small Molecules. ACS Macro Lett. 2012, 1, 922–926. [Google Scholar] [CrossRef]
- Yardley, R.E.; Gillies, E.R. Multi-stimuli-responsive self-immolative polymer assemblies. J. Polym. Sci. Part A Polym. Chem. 2018, 56, 1868–1877. [Google Scholar] [CrossRef]
- Zeng, X.; Zhou, X.; Wu, S. Red and Near-Infrared Light-Cleavable Polymers. Macromol. Rapid Commun. 2018, 39, 1800034. [Google Scholar] [CrossRef]
- Fomina, N.; McFearin, C.L.; Almutairi, A. Increasing materials’ response to two-photon NIR light via self-immolative dendritic scaffolds. Chem. Commun. 2012, 48, 9138–9140. [Google Scholar] [CrossRef]
- Hong, M.-S.; Lim, S.-J.; Oh, Y.-K.; Kim, C.-K. pH-sensitive, serum-stable and long-circulating liposomes as a new drug delivery system. J. Pharm. Pharmacol. 2002, 54, 51–58. [Google Scholar] [CrossRef]
- Zhao, X.B.; Lee, R.J. Tumor-selective targeted delivery of genes and antisense oligodeoxyribonucleotides via the folate receptor. Adv. Drug Deliv. Rev. 2004, 56, 1193–1204. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Wang, Z.; Zhu, D.; Zong, S.; Yang, L.; Zhong, Y.; Cui, Y. pH and Thermo Dual-Stimuli-Responsive Drug Carrier Based on Mesoporous Silica Nanoparticles Encapsulated in a Copolymer–Lipid Bilayer. ACS Appl. Mater. Interfaces 2013, 5, 10895–10903. [Google Scholar] [CrossRef] [PubMed]
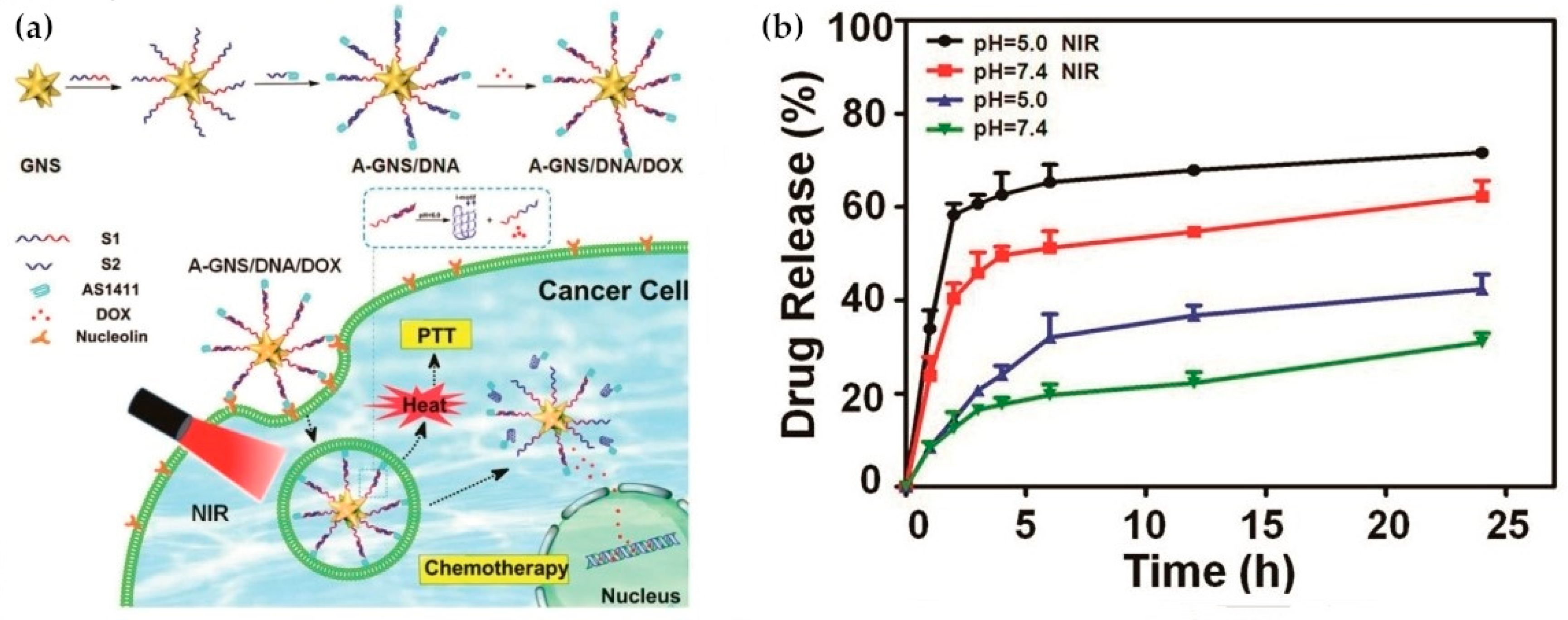
- Miao, D.; Yu, Y.; Chen, Y.; Liu, Y.; Su, G. Facile Construction of i-Motif DNA-Conjugated Gold Nanostars as Near-Infrared and pH Dual-Responsive Targeted Drug Delivery Systems for Combined Cancer Therapy. Mol. Pharm. 2020, 17, 1127–1138. [Google Scholar] [CrossRef]
- Mei, X.; Yang, S.; Chen, D.; Li, N.; Li, H.; Xu, Q.; Ge, J.; Lu, J. Light-triggered reversible assemblies of azobenzene-containing amphiphilic copolymer with β-cyclodextrin-modified hollow mesoporous silica nanoparticles for controlled drug release. Chem. Commun. 2012, 48, 10010–10012. [Google Scholar] [CrossRef]
- Bian, Q.; Wang, W.; Wang, S.; Wang, G. Light-Triggered Specific Cancer Cell Release from Cyclodextrin/Azobenzene and Aptamer-Modified Substrate. ACS Appl. Mater. Interfaces 2016, 8, 27360–27367. [Google Scholar] [CrossRef]
- Yi, S.; Zheng, J.; Lv, P.; Zhang, D.; Zheng, X.; Zhang, Y.; Liao, R. Controlled Drug Release from Cyclodextrin-Gated Mesoporous Silica Nanoparticles Based on Switchable Host–Guest Interactions. Bioconjugate Chem. 2018, 29, 2884–2891. [Google Scholar] [CrossRef]
- Zhang, J.; Zhou, Z.-H.; Li, L.; Luo, Y.-L.; Xu, F.; Chen, Y. Dual Stimuli-Responsive Supramolecular Self-Assemblies Based on the Host–Guest Interaction between β-Cyclodextrin and Azobenzene for Cellular Drug Release. Mol. Pharm. 2020, 17, 1100–1113. [Google Scholar] [CrossRef]
- Luo, Y.; Yin, X.; Yin, X.; Chen, A.; Zhao, L.; Zhang, G.; Liao, W.; Huang, X.; Li, J.; Zhang, C.Y. Dual pH/Redox-Responsive Mixed Polymeric Micelles for Anticancer Drug Delivery and Controlled Release. Pharmaceutics 2019, 11, 176. [Google Scholar] [CrossRef]
- Lei, Z.; Ju, Y.; Lin, Y.; Bai, X.; Hu, W.; Wang, Y.; Luo, H.; Tong, Z. Reactive Oxygen Species Synergistic pH/H2O2-Responsive Poly(l-lactic acid)-block-poly(sodium 4-styrenesulfonate)/Citrate-Fe(III)@ZIF-8 Hybrid Nanocomposites for Controlled Drug Release. ACS Appl. Bio Mater. 2019, 2, 3648–3658. [Google Scholar] [CrossRef]
- Liu, T.; Jin, R.; Yuan, P.; Bai, Y.; Cai, B.; Chen, X. Intracellular Enzyme-Triggered Assembly of Amino Acid-Modified Gold Nanoparticles for Accurate Cancer Therapy with Multimode. ACS Appl. Mater. Interfaces 2019, 11, 28621–28630. [Google Scholar] [CrossRef]
- Gao, X.; Yu, Z.; Liu, B.; Yang, J.; Yang, X.; Yu, Y. A smart drug delivery system responsive to pH/enzyme stimuli based on hydrophobic modified sodium alginate. Eur. Polym. J. 2020, 133, 109779. [Google Scholar] [CrossRef]
- Pan, X.; Chen, J.; Yang, M.; Wu, J.; He, G.; Yin, Y.; He, M.; Xu, W.; Xu, P.; Cai, W.; et al. Enzyme/pH dual-responsive polymer prodrug nanoparticles based on 10-hydroxycamptothecin-carboxymethylchitosan for enhanced drug stability and anticancer efficacy. Eur. Polym. J. 2019, 117, 372–381. [Google Scholar] [CrossRef]
- Yin, W.; Ke, W.; Lu, N.; Wang, Y.; Japir, A.A.-W.M.M.; Mohammed, F.; Wang, Y.; Pan, Y.; Ge, Z. Glutathione and Reactive Oxygen Species Dual-Responsive Block Copolymer Prodrugs for Boosting Tumor Site-Specific Drug Release and Enhanced Antitumor Efficacy. Biomacromolecules 2020, 21, 921–929. [Google Scholar] [CrossRef]
- Bai, S.; Ma, X.; Shi, X.; Shao, J.; Zhang, T.; Wang, Y.; Cheng, Y.; Xue, P.; Kang, Y.; Xu, Z. Smart Unimolecular Micelle-Based Polyprodrug with Dual-Redox Stimuli Response for Tumor Microenvironment: Enhanced in Vivo Delivery Efficiency and Tumor Penetration. ACS Appl. Mater. Interfaces 2019, 11, 36130–36140. [Google Scholar] [CrossRef]
- Li, J.; Li, X.; Liu, H.; Ren, T.; Huang, L.; Deng, Z.; Yang, Y.; Zhong, S. GSH and light dual stimuli-responsive supramolecular polymer drug carriers for cancer therapy. Polym. Degrad. Stab. 2019, 168, 108956. [Google Scholar] [CrossRef]
- Sokolovskaya, E.; Rahmani, S.; Misra, A.C.; Bräse, S.; Lahann, J. Dual-Stimuli-Responsive Microparticles. ACS Appl. Mater. Interfaces 2015, 7, 9744–9751. [Google Scholar] [CrossRef]
- Yue, C.; Liu, P.; Zheng, M.; Zhao, P.; Wang, Y.; Ma, Y.; Cai, L. IR-780 dye loaded tumor targeting theranostic nanoparticles for NIR imaging and photothermal therapy. Biomaterials 2013, 34, 6853–6861. [Google Scholar] [CrossRef]
- Zhang, C.; Wang, S.; Xiao, J.; Tan, X.; Zhu, Y.; Su, Y.; Cheng, T.; Shi, C. Sentinel lymph node mapping by a near-infrared fluorescent heptamethine dye. Biomaterials 2010, 31, 1911–1917. [Google Scholar] [CrossRef]
- Wang, C.; Huang, B.; Yang, G.; Ouyang, Y.; Tian, J.; Zhang, W. NIR-Triggered Multifunctional and Degradable Nanoplatform Based on an ROS-Sensitive Block Copolymer for Imaging-Guided Chemo-Phototherapy. Biomacromolecules 2019, 20, 4218–4229. [Google Scholar] [CrossRef]
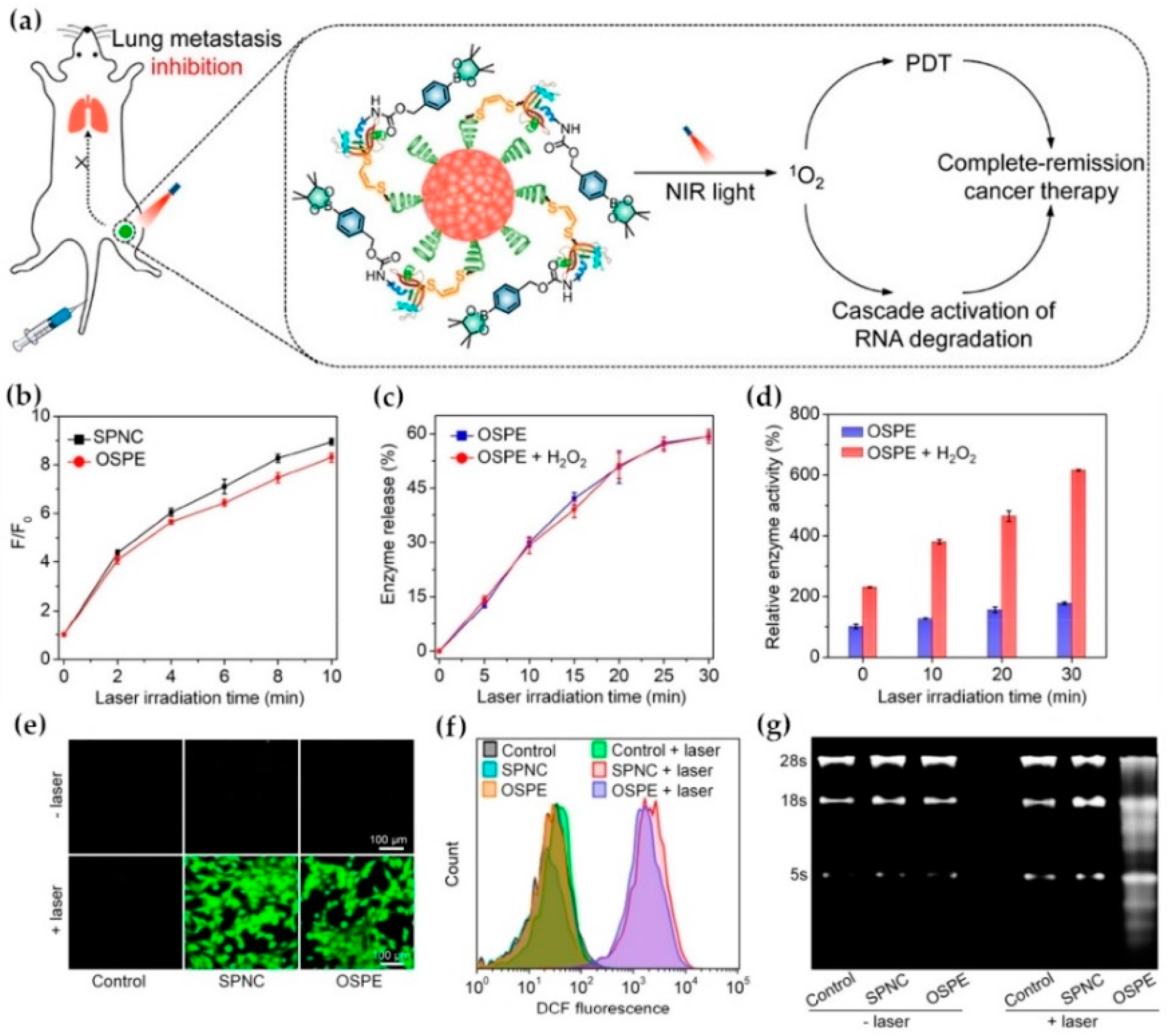
- Li, J.; Huang, J.; Lyu, Y.; Huang, J.; Jiang, Y.; Xie, C.; Pu, K. Photoactivatable Organic Semiconducting Pro-nanoenzymes. J. Am. Chem. Soc. 2019, 141, 4073–4079. [Google Scholar] [CrossRef]
- Liu, T.-I.; Lu, T.-Y.; Chang, S.-H.; Shen, M.-Y.; Chiu, H.-C. Dual stimuli-guided lipid-based delivery system of cancer combination therapy. J. Control. Release 2020, 318, 16–24. [Google Scholar] [CrossRef]
- Wang, D.; Xie, W.; Gao, Q.; Yan, H.; Zhang, J.; Lu, J.; Liaw, B.; Guo, Z.; Gao, F.; Yin, L.; et al. Non-Magnetic Injectable Implant for Magnetic Field-Driven Thermochemotherapy and Dual Stimuli-Responsive Drug Delivery: Transformable Liquid Metal Hybrid Platform for Cancer Theranostics. Small 2019, 15, 1900511. [Google Scholar] [CrossRef]
- Zhao, Q.; Wang, X.; Yang, M.; Li, X.; Mao, Y.; Guan, X.; Di, D.; Wang, S. Multi-stimuli responsive mesoporous carbon nano-platform gated by human serum albumin for cancer thermo-chemotherapy. Colloids Surf. B: Biointerfaces 2019, 184, 110532. [Google Scholar] [CrossRef]
- Qu, J.; Wang, Q.-Y.; Chen, K.-L.; Luo, J.-B.; Zhou, Q.-H.; Lin, J. Reduction/temperature/pH multi-stimuli responsive core cross-linked polypeptide hybrid micelles for triggered and intracellular drug release. Colloids Surf. B Biointerfaces 2018, 170, 373–381. [Google Scholar] [CrossRef]
- Zhang, K.; Liu, J.; Guo, Y.; Li, Y.; Ma, X.; Lei, Z. Synthesis of temperature, pH, light and dual-redox quintuple-stimuli-responsive shell-crosslinked polymeric nanoparticles for controlled release. Mater. Sci. Eng. C 2018, 87, 1–9. [Google Scholar] [CrossRef]
- Nathan, C.; Ding, A. Nonresolving Inflammation. Cell 2010, 140, 871–882. [Google Scholar] [CrossRef]
- Zetoune, F.S.; Serhan, C.N.; Ward, P.A. Inflammatory Disorders. In Reference Module in Biomedical Sciences; Elsevier: Amsterdam, The Netherlands, 2014. [Google Scholar] [CrossRef]
- Mittal, M.; Siddiqui, M.R.; Tran, K.; Reddy, S.P.; Malik, A.B. Reactive Oxygen Species in Inflammation and Tissue Injury. Antioxid. Redox Signal. 2013, 20, 1126–1167. [Google Scholar] [CrossRef]
- Mahmoud, E.A.; Sankaranarayanan, J.; Morachis, J.M.; Kim, G.; Almutairi, A. Inflammation Responsive Logic Gate Nanoparticles for the Delivery of Proteins. Bioconjugate Chem. 2011, 22, 1416–1421. [Google Scholar] [CrossRef]
- Mavuso, S.; Choonara, Y.E.; Marimuthu, T.; Kumar, P.; du Toit, L.C.; Kondiah, P.P.D.; Pillay, V. A dual pH/Redox responsive copper-ligand nanoliposome bioactive complex for the treatment of chronic inflammation. Int. J. Pharm. 2016, 509, 348–359. [Google Scholar] [CrossRef]
- Zhang, R.; Liu, R.; Liu, C.; Pan, L.; Qi, Y.; Cheng, J.; Guo, J.; Jia, Y.; Ding, J.; Zhang, J.; et al. A pH/ROS dual-responsive and targeting nanotherapy for vascular inflammatory diseases. Biomaterials 2020, 230, 119605. [Google Scholar] [CrossRef]
- Lee, S.; Stubelius, A.; Hamelmann, N.; Tran, V.; Almutairi, A. Inflammation-Responsive Drug-Conjugated Dextran Nanoparticles Enhance Anti-Inflammatory Drug Efficacy. ACS Appl. Mater. Interfaces 2018, 10, 40378–40387. [Google Scholar] [CrossRef]
- Soliman, M.; Allen, S.; Davies, M.C.; Alexander, C. Responsive polyelectrolyte complexes for triggered release of nucleic acid therapeutics. Chem. Commun. 2010, 46, 5421–5433. [Google Scholar] [CrossRef]
- Pan, T.-T.; He, W.-D.; Li, L.-Y.; Jiang, W.-X.; He, C.; Tao, J. Dual thermo- and pH-sensitive network-grafted hydrogels formed by macrocrosslinker as drug delivery system. J. Polym. Sci. Part A Polym. Chem. 2011, 49, 2155–2164. [Google Scholar] [CrossRef]
- Zhao, X.-P.; Wang, S.-S.; Younis, M.R.; Xia, X.-H.; Wang, C. Thermo and pH Dual Actuating Smart Porous Anodic Aluminum for Controllable Drug Release. Adv. Mater. Interfaces 2018, 5, 1800185. [Google Scholar] [CrossRef]
- Chiang, W.-L.; Hu, Y.-C.; Liu, H.-Y.; Hsiao, C.-W.; Sureshbabu, R.; Yang, C.-M.; Chung, M.-F.; Chia, W.-T.; Sung, H.-W. Injectable Microbeads with a Thermo-Responsive Shell and a pH-Responsive Core as a Dual-Switch-Controlled Release System. Small 2014, 10, 4100–4105. [Google Scholar] [CrossRef]
- Kaser, A.; Zeissig, S.; Blumberg, R.S. Inflammatory Bowel Disease. Annu. Rev. Immunol. 2010, 28, 573–621. [Google Scholar] [CrossRef]
- Kotla, N.G.; Rana, S.; Sivaraman, G.; Sunnapu, O.; Vemula, P.K.; Pandit, A.; Rochev, Y. Bioresponsive drug delivery systems in intestinal inflammation: State-of-the-art and future perspectives. Adv. Drug Deliv. Rev. 2019, 146, 248–266. [Google Scholar] [CrossRef]
- Bertoni, S.; Machness, A.; Tiboni, M.; Bártolo, R.; Santos, H.A. Reactive oxygen species responsive nanoplatforms as smart drug delivery systems for gastrointestinal tract targeting. Biopolymers 2020, 111, e23336. [Google Scholar] [CrossRef] [PubMed]
- Bertoni, S.; Liu, Z.; Correia, A.; Martins, J.P.; Rahikkala, A.; Fontana, F.; Kemell, M.; Liu, D.; Albertini, B.; Passerini, N.; et al. pH and Reactive Oxygen Species-Sequential Responsive Nano-in-Micro Composite for Targeted Therapy of Inflammatory Bowel Disease. Adv. Funct. Mater. 2018, 28, 1806175. [Google Scholar] [CrossRef]
- Zhang, S.; Langer, R.; Traverso, G. Nanoparticulate drug delivery systems targeting inflammation for treatment of inflammatory bowel disease. Nano Today 2017, 16, 82–96. [Google Scholar] [CrossRef]
- Lautenschläger, C.; Schmidt, C.; Fischer, D.; Stallmach, A. Drug delivery strategies in the therapy of inflammatory bowel disease. Adv. Drug Deliv. Rev. 2014, 71, 58–76. [Google Scholar] [CrossRef] [PubMed]
- Loftus, E.V., Jr.; Kane, S.V.; Bjorkman, D. Short-term adverse effects of 5-aminosalicylic acid agents in the treatment of ulcerative colitis. Aliment. Pharmacol. Ther. 2004, 19, 179–189. [Google Scholar] [CrossRef]
- Wee Yong, V. Inflammation in Neurological Disorders: A Help or a Hindrance? Neuroscience 2010, 16, 408–420. [Google Scholar] [CrossRef] [PubMed]
- Amor, S.; Puentes, F.; Baker, D.; Van Der Valk, P. Inflammation in neurodegenerative diseases. Immunology 2010, 129, 154–169. [Google Scholar] [CrossRef] [PubMed]
- Fakhoury, M. Role of Immunity and Inflammation in the Pathophysiology of Neurodegenerative Diseases. Neurodegener. Dis. 2015, 15, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.-Q.; Chen, Q.; Wang, X.; Wang, Q.-C.; Wang, Y.; Cheng, H.-P.; Guo, C.; Sun, Q.; Chen, Q.; Tang, T.-S. Dysregulation of Mitochondrial Calcium Signaling and Superoxide Flashes Cause Mitochondrial Genomic DNA Damage in Huntington Disease. J. Biol. Chem. 2013, 288, 3070–3084. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, Y.; Ji, W.; Lu, Z.; Liu, L.; Shi, Y.; Ma, G.; Zhang, X. Positively Charged Polyprodrug Amphiphiles with Enhanced Drug Loading and Reactive Oxygen Species-Responsive Release Ability for Traceable Synergistic Therapy. J. Am. Chem. Soc. 2018, 140, 4164–4171. [Google Scholar] [CrossRef]
- Yoo, D.; Magsam, A.W.; Kelly, A.M.; Stayton, P.S.; Kievit, F.M.; Convertine, A.J. Core-Cross-Linked Nanoparticles Reduce Neuroinflammation and Improve Outcome in a Mouse Model of Traumatic Brain Injury. ACS Nano 2017, 11, 8600–8611. [Google Scholar] [CrossRef]
- Choonara, B.F.; Choonara, Y.E.; Kumar, P.; Bijukumar, D.; du Toit, L.C.; Pillay, V. A review of advanced oral drug delivery technologies facilitating the protection and absorption of protein and peptide molecules. Biotechnol. Adv. 2014, 32, 1269–1282. [Google Scholar] [CrossRef]
- Joslin, E.P.; Gray, H.; Root, H.F. Insulin in hospital and home. J. Metab. Res. 1922, 2, 651–699. [Google Scholar]
- Liu, L.; Zhang, Y.; Yu, S.; Yang, Z.; He, C.; Chen, X. Dual Stimuli-Responsive Nanoparticle-Incorporated Hydrogels as an Oral Insulin Carrier for Intestine-Targeted Delivery and Enhanced Paracellular Permeation. ACS Biomater. Sci. Eng. 2018, 4, 2889–2902. [Google Scholar] [CrossRef]
- Lin, Y.-H.; Chang, C.-H.; Wu, Y.-S.; Hsu, Y.-M.; Chiou, S.-F.; Chen, Y.-J. Development of pH-responsive chitosan/heparin nanoparticles for stomach-specific anti-Helicobacter pylori therapy. Biomaterials 2009, 30, 3332–3342. [Google Scholar] [CrossRef] [PubMed]
- Rajpoot, K.; Jain, S.K. Oral delivery of pH-responsive alginate microbeads incorporating folic acid-grafted solid lipid nanoparticles exhibits enhanced targeting effect against colorectal cancer: A dual-targeted approach. Int. J. Biol. Macromol. 2020, 151, 830–844. [Google Scholar] [CrossRef] [PubMed]
- Puranik, A.S.; Pao, L.P.; White, V.M.; Peppas, N.A. In Vitro Evaluation of pH-Responsive Nanoscale Hydrogels for the Oral Delivery of Hydrophobic Therapeutics. Ind. Eng. Chem. Res. 2016, 55, 10576–10590. [Google Scholar] [CrossRef]
- Cheng, L.; Deng, B.; Luo, W.; Nie, S.; Liu, X.; Yin, Y.; Liu, S.; Wu, Z.; Zhan, P.; Zhang, L.; et al. pH-Responsive Lignin-Based Nanomicelles for Oral Drug Delivery. J. Agric. Food Chem. 2020, 68, 5249–5258. [Google Scholar] [CrossRef] [PubMed]
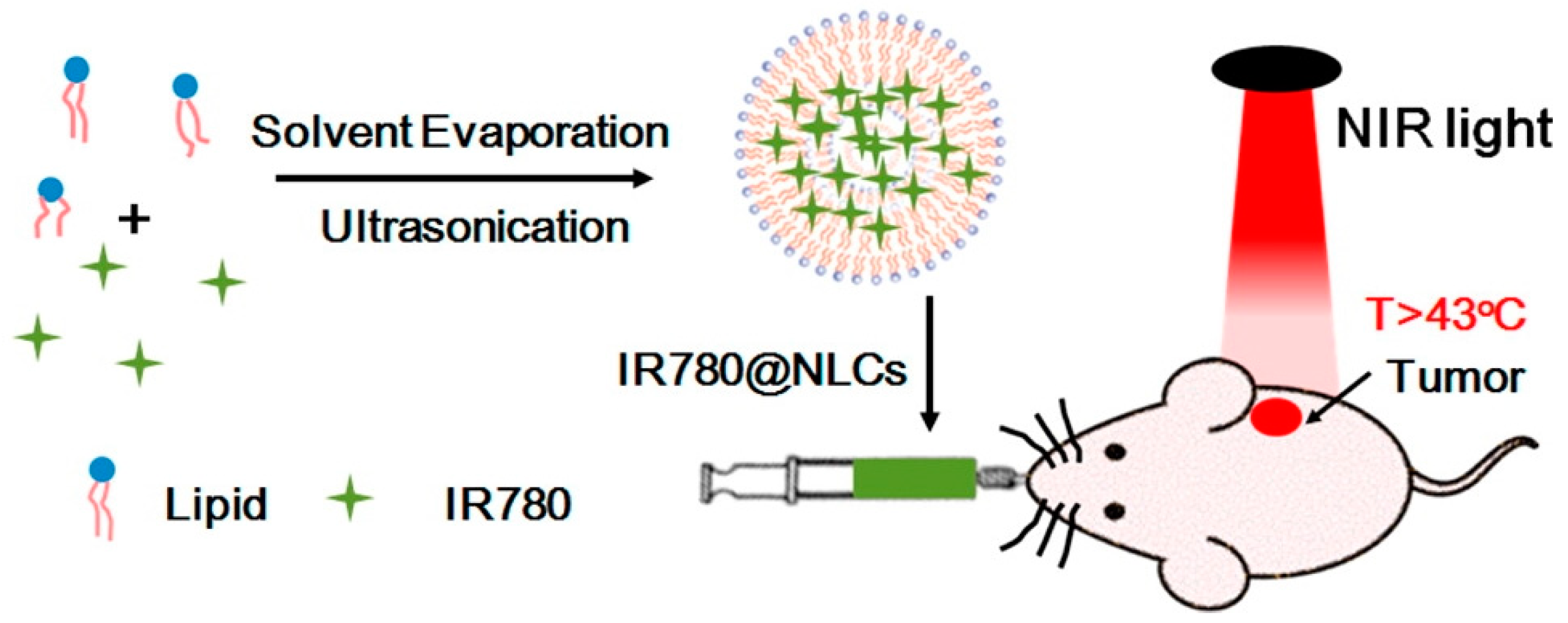
- Chen, G.; Wang, K.; Zhou, Y.; Ding, L.; Ullah, A.; Hu, Q.; Sun, M.; Oupický, D. Oral Nanostructured Lipid Carriers Loaded with Near-Infrared Dye for Image-Guided Photothermal Therapy. ACS Appl. Mater. Interfaces 2016, 8, 25087–25095. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Bellinger, A.M.; Glettig, D.L.; Barman, R.; Lee, Y.-A.L.; Zhu, J.; Cleveland, C.; Montgomery, V.A.; Gu, L.; Nash, L.D.; et al. A pH-responsive supramolecular polymer gel as an enteric elastomer for use in gastric devices. Nat. Mater. 2015, 14, 1065–1071. [Google Scholar] [CrossRef]
- Jiang, Y.; Cui, D.; Fang, Y.; Zhen, X.; Upputuri, P.K.; Pramanik, M.; Ding, D.; Pu, K. Amphiphilic semiconducting polymer as multifunctional nanocarrier for fluorescence/photoacoustic imaging guided chemo-photothermal therapy. Biomaterials 2017, 145, 168–177. [Google Scholar] [CrossRef]
- Zhao, X.; Yang, C.-X.; Chen, L.-G.; Yan, X.-P. Dual-stimuli responsive and reversibly activatable theranostic nanoprobe for precision tumor-targeting and fluorescence-guided photothermal therapy. Nat. Commun. 2017, 8, 14998. [Google Scholar] [CrossRef]
- Chen, S.; Sun, B.; Miao, H.; Wang, G.; Sun, P.; Li, J.; Wang, W.; Fan, Q.; Huang, W. NIR-II Dye-Based Multifunctional Telechelic Glycopolymers for NIR-IIa Fluorescence Imaging-Guided Stimuli-Responsive Chemo-Photothermal Combination Therapy. ACS Mater. Lett. 2020, 2, 174–183. [Google Scholar] [CrossRef]
- Li, P.; Sun, M.; Xu, Z.; Liu, X.; Zhao, W.; Gao, W. Site-Selective in Situ Growth-Induced Self-Assembly of Protein–Polymer Conjugates into pH-Responsive Micelles for Tumor Microenvironment Triggered Fluorescence Imaging. Biomacromolecules 2018, 19, 4472–4479. [Google Scholar] [CrossRef]
- Wang, X.; Niu, D.; Li, P.; Wu, Q.; Bo, X.; Liu, B.; Bao, S.; Su, T.; Xu, H.; Wang, Q. Dual-Enzyme-Loaded Multifunctional Hybrid Nanogel System for Pathological Responsive Ultrasound Imaging and T2-Weighted Magnetic Resonance Imaging. ACS Nano 2015, 9, 5646–5656. [Google Scholar] [CrossRef]
- Song, X.-R.; Li, S.-H.; Dai, J.; Song, L.; Huang, G.; Lin, R.; Li, J.; Liu, G.; Yang, H.-H. Polyphenol-Inspired Facile Construction of Smart Assemblies for ATP- and pH-Responsive Tumor MR/Optical Imaging and Photothermal Therapy. Small 2017, 13, 1603997. [Google Scholar] [CrossRef] [PubMed]
- Teng, L.; Song, G.; Liu, Y.; Han, X.; Li, Z.; Wang, Y.; Huan, S.; Zhang, X.-B.; Tan, W. Nitric Oxide-Activated “Dual-Key–One-Lock” Nanoprobe for in Vivo Molecular Imaging and High-Specificity Cancer Therapy. J. Am. Chem. Soc. 2019, 141, 13572–13581. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.; Yin, W.; Zhang, X.; Zhu, S.; He, X.; Yu, J.; Xie, J.; Guo, Z.; Yan, L.; Liu, X.; et al. Intelligent MoS2 Nanotheranostic for Targeted and Enzyme-/pH-/NIR-Responsive Drug Delivery To Overcome Cancer Chemotherapy Resistance Guided by PET Imaging. ACS Appl. Mater. Interfaces 2018, 10, 4271–4284. [Google Scholar] [CrossRef]
- Li, Z.; Hu, Y.; Miao, Z.; Xu, H.; Li, C.; Zhao, Y.; Li, Z.; Chang, M.; Ma, Z.; Sun, Y.; et al. Dual-Stimuli Responsive Bismuth Nanoraspberries for Multimodal Imaging and Combined Cancer Therapy. Nano Lett. 2018, 18, 6778–6788. [Google Scholar] [CrossRef]
- Lindner, J.R. Microbubbles in medical imaging: Current applications and future directions. Nat. Rev. Drug Discov. 2004, 3, 527–533. [Google Scholar] [CrossRef] [PubMed]
- Schutt, E.G.; Klein, D.H.; Mattrey, R.M.; Riess, J.G. Injectable Microbubbles as Contrast Agents for Diagnostic Ultrasound Imaging: The Key Role of Perfluorochemicals. Angew. Chem. Int. Ed. 2003, 42, 3218–3235. [Google Scholar] [CrossRef] [PubMed]
- Walker, J.A.-T.; Wang, X.; Peter, K.; Kempe, K.; Corrie, S.R. Dynamic Solid-State Ultrasound Contrast Agent for Monitoring pH Fluctuations In Vivo. ACS Sens. 2020, 5, 1190–1197. [Google Scholar] [CrossRef]
- Winter, P.M.; Caruthers, S.D.; Wickline, S.A.; Lanza, G.M. Molecular imaging by MRI. Curr. Cardiol. Rep. 2006, 8, 65–69. [Google Scholar] [CrossRef]
- Hu, X.; Norris, D.G. Advances in High-Field Magnetic Resonance Imaging. Annu. Rev. Biomed. Eng. 2004, 6, 157–184. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.; Zhang, M.; Lu, H.; Liang, D.; Huang, Y.; Xia, Y.; Hu, Y.; Hu, S.; Wang, J.; Yi, X.; et al. Triple Stimuli-Responsive Magnetic Hollow Porous Carbon-Based Nanodrug Delivery System for Magnetic Resonance Imaging-Guided Synergistic Photothermal/Chemotherapy of Cancer. ACS Appl. Mater. Interfaces 2018, 10, 21939–21949. [Google Scholar] [CrossRef] [PubMed]
- Song, S.; Chong, Y.; Fu, H.; Ning, X.; Shen, H.; Zhang, Z. HP-β-CD Functionalized Fe3O4/CNPs-Based Theranostic Nanoplatform for pH/NIR Responsive Drug Release and MR/NIRFL Imaging-Guided Synergetic Chemo/Photothermal Therapy of Tumor. ACS Appl. Mater. Interfaces 2018, 10, 33867–33878. [Google Scholar] [CrossRef]
- Wang, X.; Pang, Y.; Ku, G.; Xie, X.; Stoica, G.; Wang, L.V. Noninvasive laser-induced photoacoustic tomography for structural and functional in vivo imaging of the brain. Nat. Biotechnol. 2003, 21, 803–806. [Google Scholar] [CrossRef]
- Cheng, P.; Chen, W.; Li, S.; He, S.; Miao, Q.; Pu, K. Fluoro-Photoacoustic Polymeric Renal Reporter for Real-Time Dual Imaging of Acute Kidney Injury. Adv. Mater. 2020, 32, 1908530. [Google Scholar] [CrossRef] [PubMed]
- Phelps, M.E. PET: The Merging of Biology and Imaging into Molecular Imaging. J. Nucl. Med. 2000, 41, 661–681. [Google Scholar]
- Valk, P.E.; Delbeke, D.; Bailey, D.L.; Townsend, D.W.; Maisey, M.N. Positron Emission Tomography Basic Science of PET and PET/CT; Springer: New York, NY, USA, 2006. [Google Scholar]
- Song, J.; Lin, L.; Yang, Z.; Zhu, R.; Zhou, Z.; Li, Z.-W.; Wang, F.; Chen, J.; Yang, H.; Chen, X. Self-Assembled Responsive Bilayered Vesicles with Adjustable Oxidative Stress for Enhanced Cancer Imaging and Therapy. J. Am. Chem. Soc. 2019, 141, 8158–8170. [Google Scholar] [CrossRef]
- Ai, T.; Yang, Z.; Hou, H.; Zhan, C.; Chen, C.; Lv, W.; Tao, Q.; Sun, Z.; Xia, L. Correlation of Chest CT and RT-PCR Testing in Coronavirus Disease 2019 (COVID-19) in China: A Report of 1014 Cases. Radiology 2020, 296, 200642. [Google Scholar] [CrossRef]
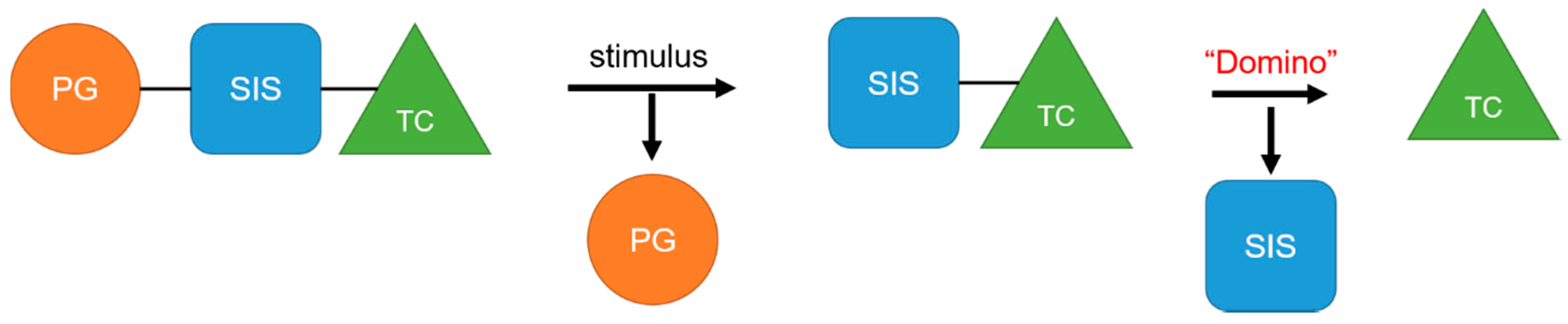
| Function Group | Mechanism | Ref |
|---|---|---|
| Imine |  | [34] |
| Hydrazone | 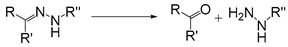 | [35,36] |
| Ester | 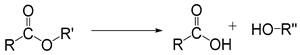 | [37] |
| Acetal | 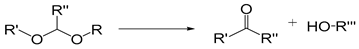 | [38,39] |
| Cis-aconityl | 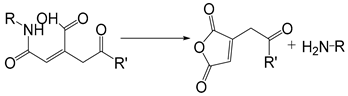 | [40] |
| Orthoester | 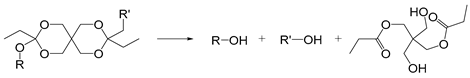 | [41,42] |
| Silyl ether | 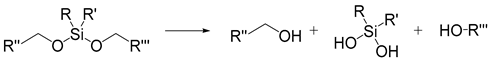 | [43] |
| Function Group | Mechanism | Ref |
|---|---|---|
| Disulfide | 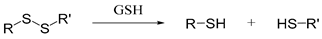 | [53] |
| Diselenium | 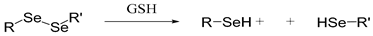 | [54,55] |
| Thioether Selenium Tellurium | 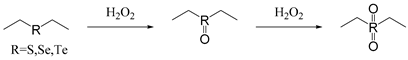 | [56,57,58,59,60,61] |
| Oxalate ester | 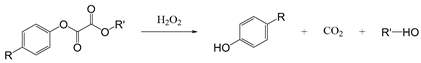 | [62,63] |
| Vinyldithioether | 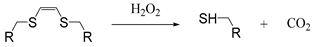 | [64] |
| Thioketal | 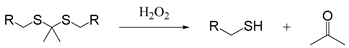 | [65] |
| PBA/PBE | 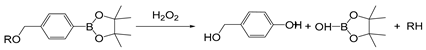 | [66] |
| Function Groups | Mechanisms | Ref |
|---|---|---|
| Anthracene | 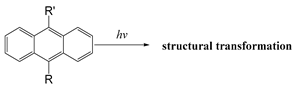 | [110] |
| Coumarinyl ester | 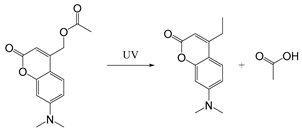 | [111] |
| Arylmethyl | 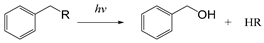 | [112] |
| Pyrenylmethyl ester | 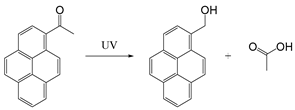 | [113] |
| O-nitrobenzyl | 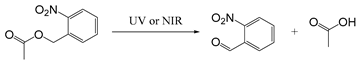 | [114,115,116] |
| Azobenzene | 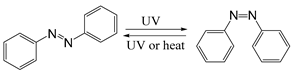 | [120,121] |
| Spiropyan | 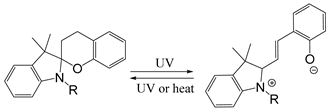 | [122] |
| Diarylethene | 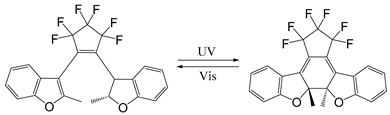 | [123,124,125,126,127,128,129,130,131] |
| Structure Responsive to Stimulus | Stimulus | Triggers | Ref |
|---|---|---|---|
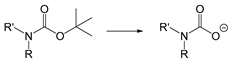 | H+ | pH | [176,177] |
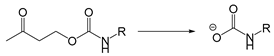 | Piperidine/ Morpholine | pH | [178] |
 | Zn/AcOH | Redox | [180] |
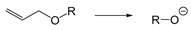 | Pd (PPh3)4 | Redox | [179,181] |
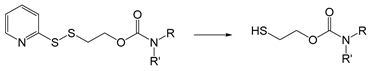 | Dithiothreitol | Redox | [182,187] |
 | Thiols | Redox | [182] |
 | H2O2 | Redox | [183,184,185,186] |
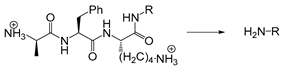 | Plasmin | Enzyme | [172,188] |
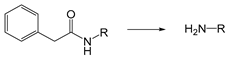 | Penicillin-G-amidase (PGA) | Enzyme | [190] |
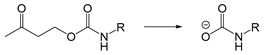 | BSA | Enzyme | [189] |
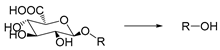 | β-Glucuronidase | Enzyme | [191,192,193] |
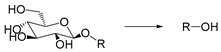 | β-Galactosidase | Enzyme | [194] |
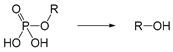 | Alkaline phosphatase | Enzyme | [195,196] |
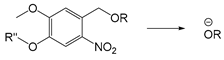 | UV light/ NIR light | Light | [197,198,199,200] |
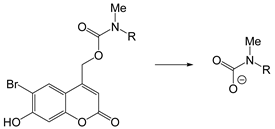 | NIR light | Light | [201,202] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, G.; Lovell, J.F.; Zhang, L.; Zhang, Y. Stimulus-Responsive Nanomedicines for Disease Diagnosis and Treatment. Int. J. Mol. Sci. 2020, 21, 6380. https://doi.org/10.3390/ijms21176380
Liu G, Lovell JF, Zhang L, Zhang Y. Stimulus-Responsive Nanomedicines for Disease Diagnosis and Treatment. International Journal of Molecular Sciences. 2020; 21(17):6380. https://doi.org/10.3390/ijms21176380
Chicago/Turabian StyleLiu, Gengqi, Jonathan F. Lovell, Lei Zhang, and Yumiao Zhang. 2020. "Stimulus-Responsive Nanomedicines for Disease Diagnosis and Treatment" International Journal of Molecular Sciences 21, no. 17: 6380. https://doi.org/10.3390/ijms21176380
APA StyleLiu, G., Lovell, J. F., Zhang, L., & Zhang, Y. (2020). Stimulus-Responsive Nanomedicines for Disease Diagnosis and Treatment. International Journal of Molecular Sciences, 21(17), 6380. https://doi.org/10.3390/ijms21176380






