Tuning Adipogenic Differentiation in ADSCs by Metformin and Vitamin D: Involvement of miRNAs
Abstract
1. Introduction
2. Results
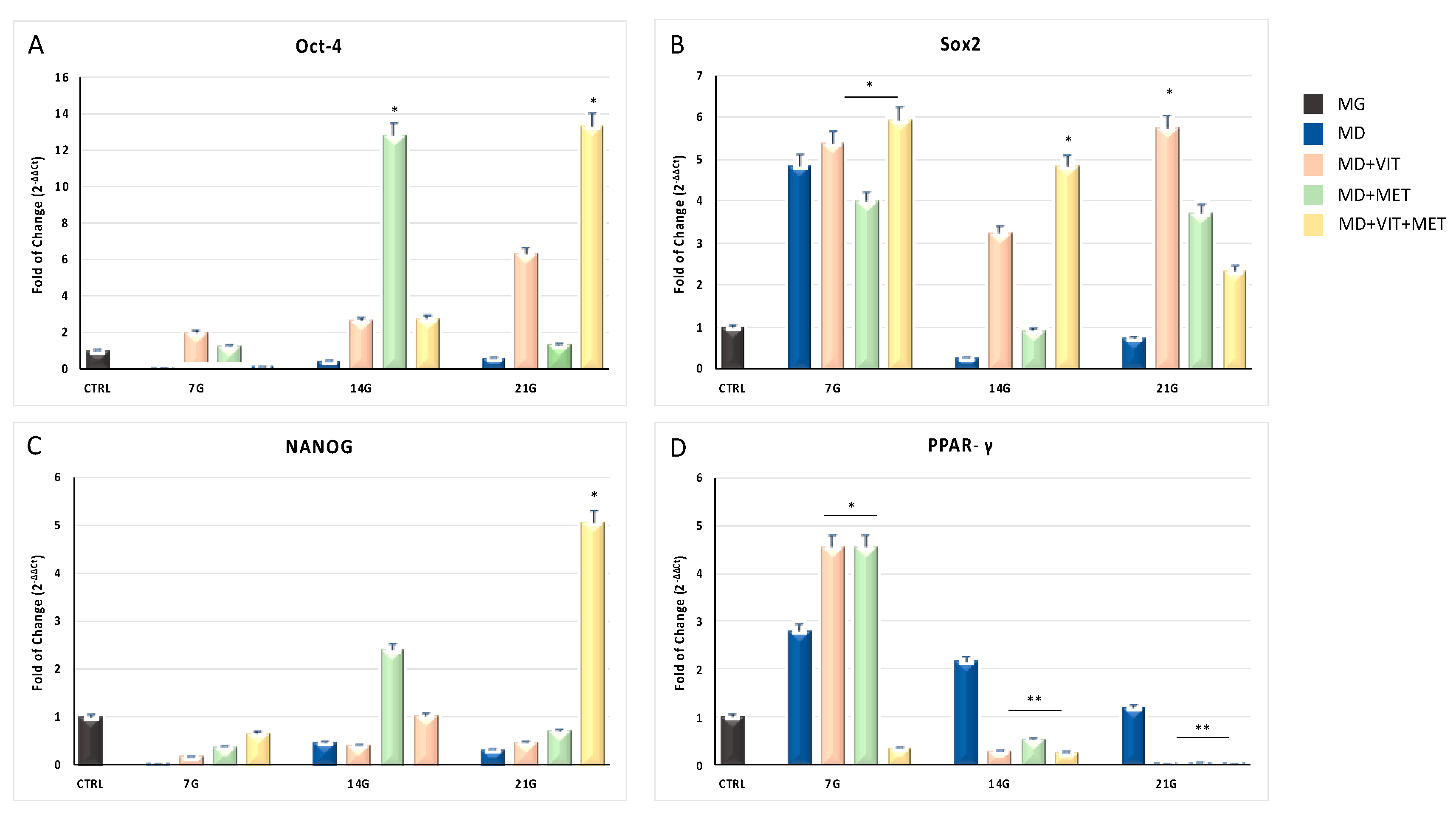
2.1. Exposure to Metformin with or without Vitamin D Increases Stem Cell Potency Reducing the Expression of Adipogenic Markers
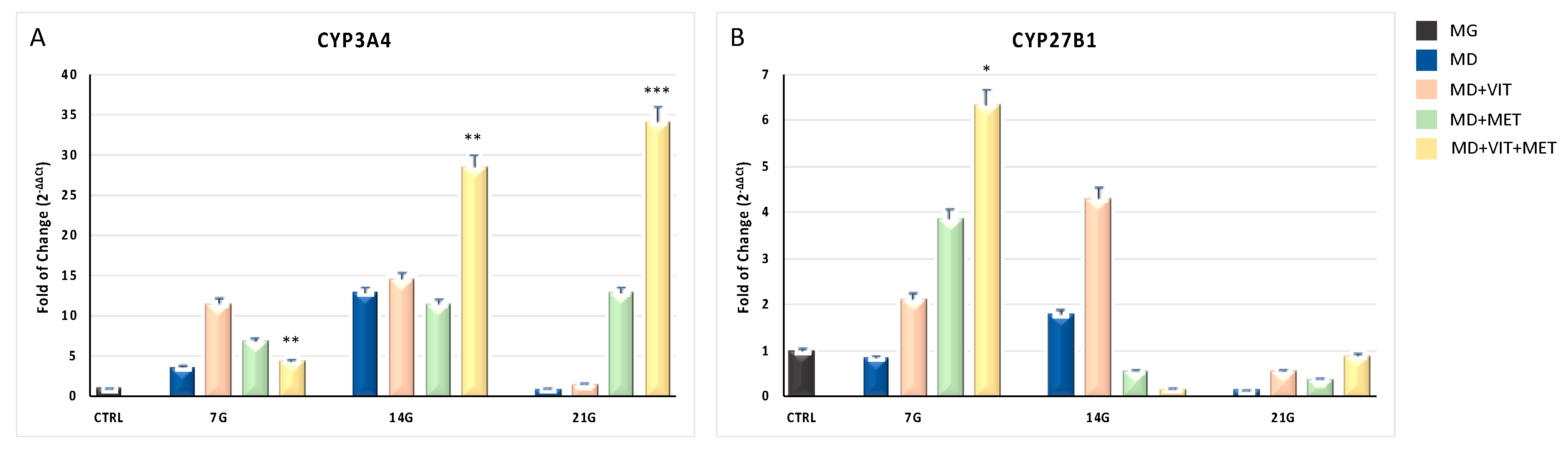
2.2. Metformin and Vitamin D Are Able to Modulate the Expression of CYP
2.3. Metformin Inhibits Adipogenesis Recruiting Epigenetic Modulating Genes
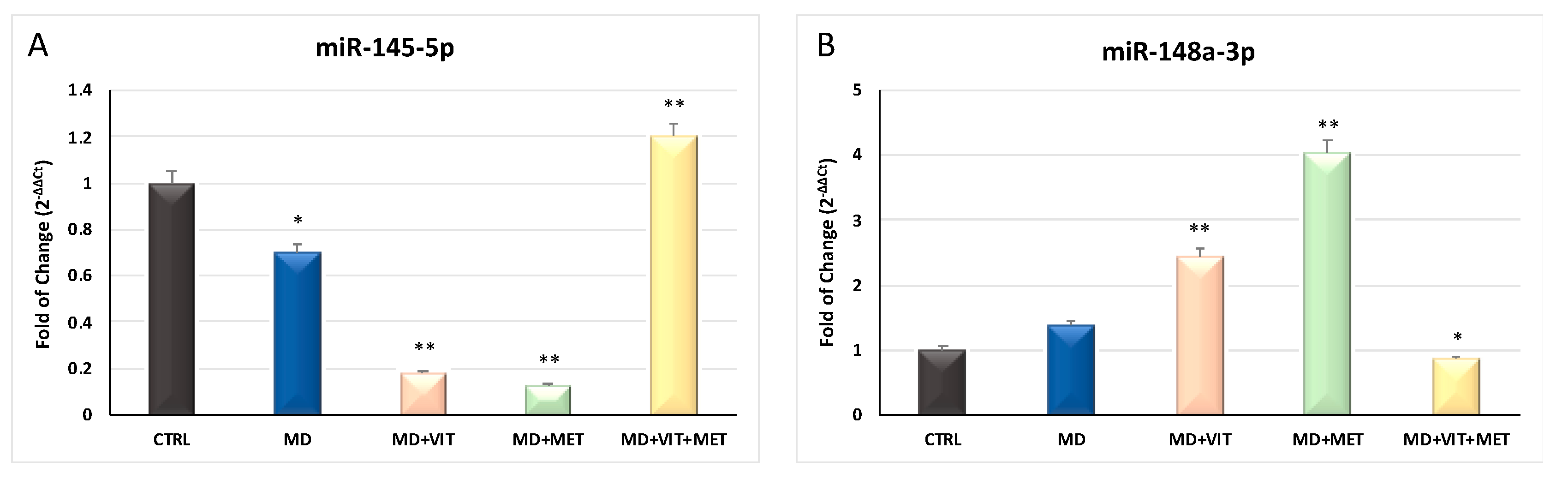
2.4. miRNAs Are Modulated by Metformin
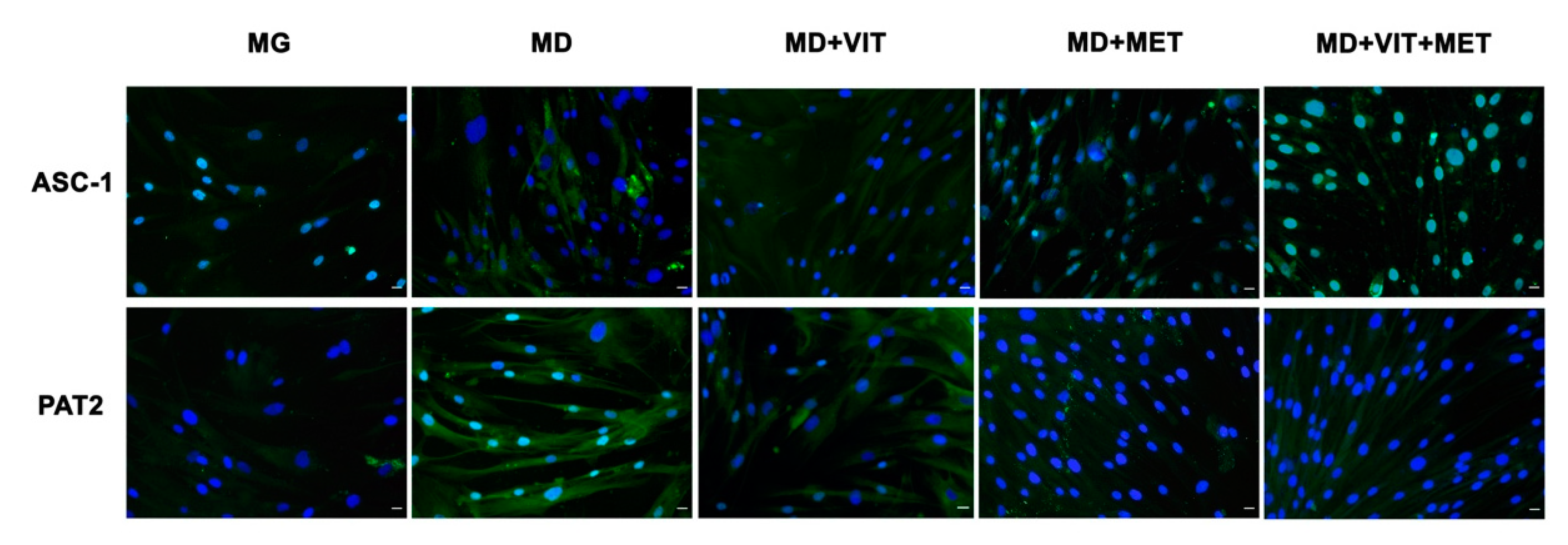
2.5. Metformin Counteract ADSC Adipogenic Differentiation Despite the Presence of a Specific Conditioned Medium
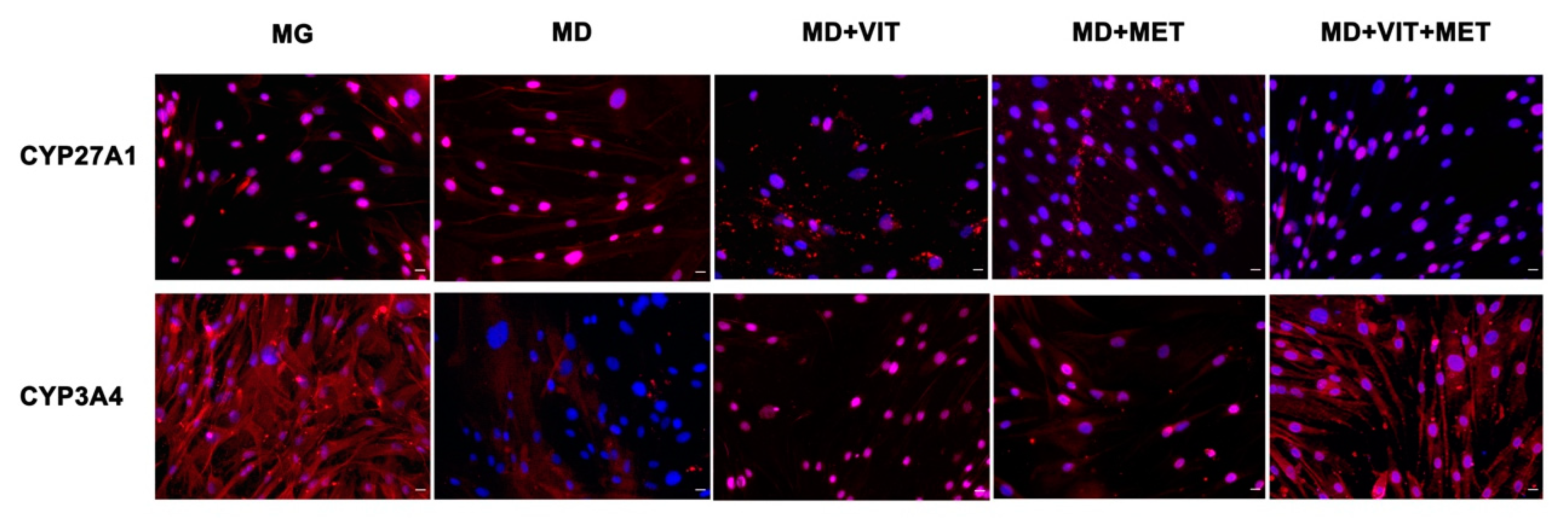
2.6. The Syngergy between the Two Molecules Ameliorate the Expression of CYP450
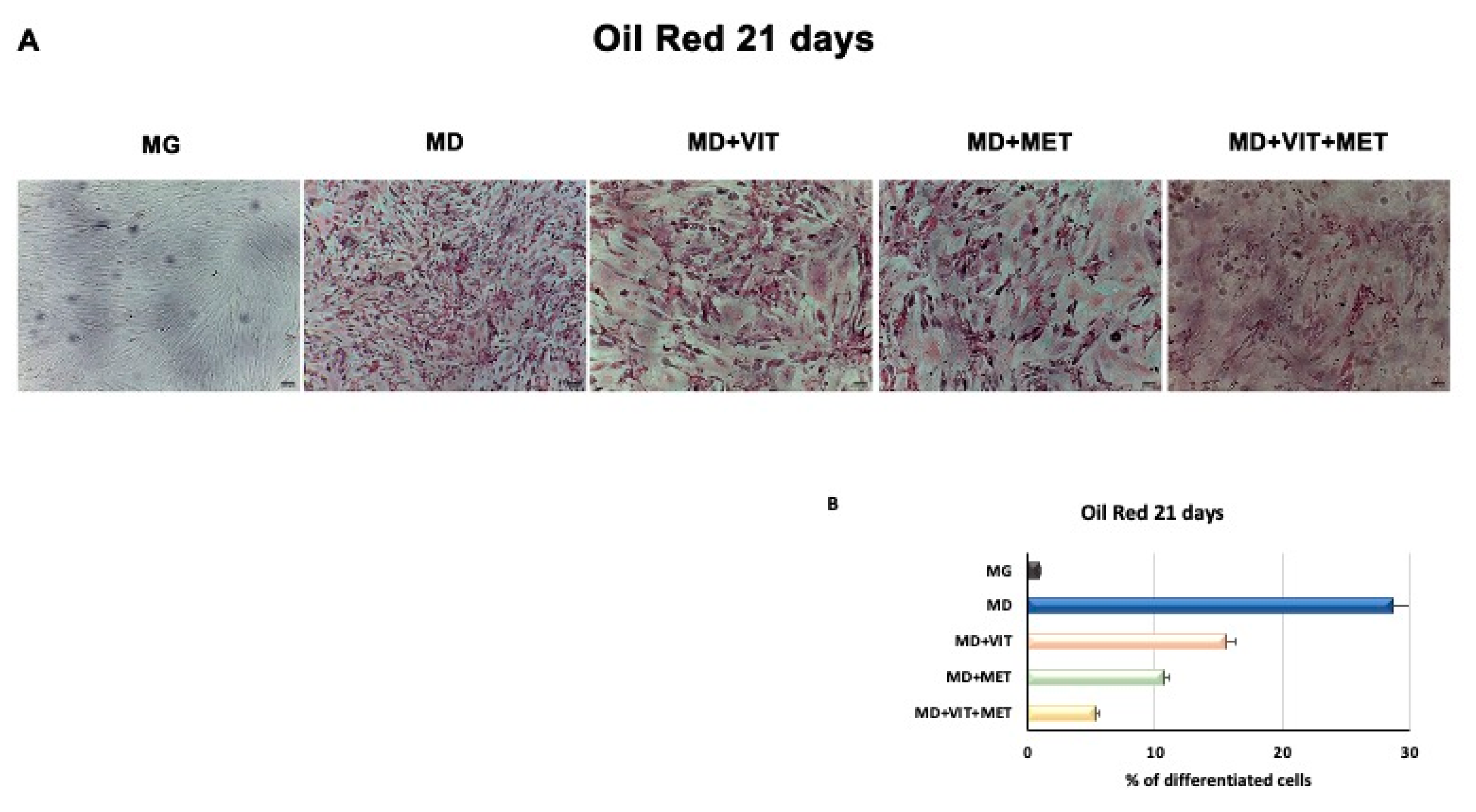
2.7. Metformin Counteract Fat Droplet Accumulation
3. Discussion
4. Materials and Methods
4.1. Cell Isolation and Treatment
4.2. Gene Expression Analysis
4.3. miRNA Expression
4.4. Immunostaining
4.5. Oil Red Staining
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- El-Badawy, A.; Amer, M.; Abdelbaset, R.; Sherif, S.N.; Abo-Elela, M.; Ghallab, Y.H.; Elhamid, H.A.; Ismail, Y.; El-Badri, N. Adipose Stem Cells Display Higher Regenerative Capacities and More Adaptable Electro-Kinetic Properties Compared to Bone Marrow-Derived Mesenchymal Stromal Cells. Sci. Rep. 2016, 6, 37801. [Google Scholar] [CrossRef]
- Cruciani, S.; Santaniello, S.; Montella, A.; Ventura, C.; Maioli, M. Orchestrating stem cell fate: Novel tools for regenerative medicine. World J. Stem Cells 2019, 11, 464–475. [Google Scholar] [CrossRef]
- Tang, Q.-Q.; Lane, M.D. Adipogenesis: From Stem Cell to Adipocyte. Annu. Rev. Biochem. 2012, 81, 715–736. [Google Scholar] [CrossRef]
- Kozlowska, U.; Krawczenko, A.; Futoma, K.; Jurek, T.; Rorat, M.; Patrzalek, D.; Klimczak, A. Similarities and differences between mesenchymal stem/progenitor cells derived from various human tissues. World J. Stem Cells 2019, 11, 347–374. [Google Scholar] [CrossRef]
- Rinaldi, S.; Maioli, M.; Santaniello, S.; Pigliaru, G.; Ventura, C.; Montela, A.; Sanna, R.; Bandiera, P.; Bagella, L.; Delitala, A.P.; et al. Amniotic fluid stem cells morph into a cardiovascular lineage: Analysis of a chemically induced cardiac and vascular commitment. Drug Des. Dev. Ther. 2013, 7, 1063–1073. [Google Scholar] [CrossRef][Green Version]
- Lefterova, M.I.; Haakonsson, A.K.; Lazar, M.A.; Mandrup, S. PPARγ and the global map of adipogenesis and beyond. Trends Endocrinol. Metab. 2014, 25, 293–302. [Google Scholar] [CrossRef]
- Ussar, S.; Lee, K.; Dankel, S.N.; Boucher, J.; Haering, M.-F.; Kleinridders, A.; Thomou, T.; Xue, R.; Macotela, Y.; Cypess, A.M.; et al. ASC-1, PAT2, and P2RX5 are cell surface markers for white, beige, and brown adipocytes. Sci. Transl. Med. 2014, 6, 247ra103. [Google Scholar] [CrossRef]
- Ma, X.; Lee, P.; Chisholm, D.J.; James, D.E. Control of Adipocyte Differentiation in Different Fat Depots; Implications for Pathophysiology or Therapy. Front. Endocrinol. 2015, 6, 1. [Google Scholar] [CrossRef] [PubMed]
- Choe, S.S.; Huh, J.Y.; Hwang, I.J.; Kim, J.I.; Kim, J.B. Adipose Tissue Remodeling: Its Role in Energy Metabolism and Metabolic Disorders. Front. Endocrinol. 2016, 7, 9. [Google Scholar] [CrossRef]
- Pereira-Santos, M.; Costa, P.R.D.F.; Assis, A.M.O.; Santos, C.A.S.T.; Santos, D.B. Obesity and vitamin D deficiency: A systematic review and meta-analysis. Obes. Rev. 2015, 16, 341–349. [Google Scholar] [CrossRef]
- Abbas, M.A. Physiological functions of Vitamin D in adipose tissue. J. Steroid Biochem. Mol. Boil. 2017, 165, 369–381. [Google Scholar] [CrossRef]
- Jones, G.; Prosser, D.E.; Kaufmann, M. Cytochrome P450-mediated metabolism of vitamin D. J. Lipid Res. 2013, 55, 13–31. [Google Scholar] [CrossRef]
- McGregor, R.A.; Choi, M.-S. microRNAs in the Regulation of Adipogenesis and Obesity. Curr. Mol. Med. 2011, 11, 304–316. [Google Scholar] [CrossRef]
- Iacomino, G.; Siani, A. Role of microRNAs in obesity and obesity-related diseases. Genes Nutr. 2017, 12, 23. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, S.; Cheng, G.; Mei, C.; Li, S.; Zhang, W.; Junjvlieke, Z.; Zan, L.; Zainaguli, J. MiR-145 reduces the activity of PI3K/Akt and MAPK signaling pathways and inhibits adipogenesis in bovine preadipocytes. Genomics 2020, 112, 2688–2694. [Google Scholar] [CrossRef]
- Guo, Y.; Chen, Y.; Zhang, Y.; Zhang, Y.; Chen, L.; Mo, D. Up-regulated miR-145 Expression Inhibits Porcine Preadipocytes Differentiation by Targeting IRS1. Int. J. Boil. Sci. 2012, 8, 1408–1417. [Google Scholar] [CrossRef]
- Shi, C.; Zhang, M.; Tong, M.; Yang, L.; Pang, L.; Chen, L.; Xu, G.; Chi, X.; Hong, Q.; Ni, Y.; et al. miR-148a is Associated with Obesity and Modulates Adipocyte Differentiation of Mesenchymal Stem Cells through Wnt Signaling. Sci. Rep. 2015, 5, 9930. [Google Scholar] [CrossRef]
- Ferrari, A.; Longo, R.; Peri, C.; Coppi, L.; Caruso, D.; Mai, A.; Mitro, N.; De Fabiani, E.; Crestani, M. Inhibition of class I HDACs imprints adipogenesis toward oxidative and brown-like phenotype. Biochim. Biophys. Acta BBA Mol. Cell Boil. Lipids 2020, 1865, 158594. [Google Scholar] [CrossRef]
- Zhou, Y.; Song, T.; Peng, J.; Zhou, Z.; Wei, H.; Zhou, R.; Jiang, S.; Peng, J. SIRT1 suppresses adipogenesis by activating Wnt/β-catenin signaling in vivo and in vitro. Oncotarget 2016, 7, 77707–77720. [Google Scholar] [CrossRef] [PubMed]
- Bäckesjö, C.-M.; Li, Y.; Lindgren, U.; Haldosén, L.-A. Activation of Sirt1 Decreases Adipocyte Formation during Osteoblast Differentiation of Mesenchymal Stem Cells. Cells Tissues Organs 2008, 189, 93–97. [Google Scholar] [CrossRef]
- Wang, F.; Tong, Q. SIRT2 Suppresses Adipocyte Differentiation by Deacetylating FOXO1 and Enhancing FOXO1’s Repressive Interaction with PPARγ. Mol. Boil. Cell 2009, 20, 801–808. [Google Scholar] [CrossRef]
- Alexandre, K.B.; Smit, A.M.; Gray, I.P.; Crowther, N.J. Metformin inhibits intracellular lipid accumulation in the murine pre-adipocyte cell line, 3T3-L1. Diabetes Obes. Metab. 2008, 10, 688–690. [Google Scholar] [CrossRef]
- Chen, D.; Wang, Y.; Wu, K.; Wang, X. Dual Effects of Metformin on Adipogenic Differentiation of 3T3-L1 Preadipocyte in AMPK-Dependent and Independent Manners. Int. J. Mol. Sci. 2018, 19, 1547. [Google Scholar] [CrossRef] [PubMed]
- Fujita, K.; Iwama, H.; Oura, K.; Tadokoro, T.; Hirose, K.; Watanabe, M.; Sakamoto, T.; Katsura, A.; Mimura, S.; Nomura, T.; et al. Metformin-suppressed differentiation of human visceral preadipocytes: Involvement of microRNAs. Int. J. Mol. Med. 2016, 38, 1135–1140. [Google Scholar] [CrossRef] [PubMed]
- Mazini, L.; Rochette, L.; Amine, M.; Malka, G. Regenerative Capacity of Adipose Derived Stem Cells (ADSCs), Comparison with Mesenchymal Stem Cells (MSCs). Int. J. Mol. Sci. 2019, 20, 2523. [Google Scholar] [CrossRef] [PubMed]
- Kolf, C.M.; Cho, E.; Tuan, R.S. Mesenchymal stromal cells. Biology of adult mesenchymal stem cells: Regulation of niche, self-renewal and differentiation. Arthritis Res. Ther. 2007, 9, 204. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Panina, Y.A.; Yakimov, A.S.; Komleva, Y.K.; Morgun, A.V.; Lopatina, O.L.; Malinovskaya, N.A.; Shuvaev, A.N.; Salmin, V.V.; Taranushenko, T.E.; Salmina, A.B. Plasticity of Adipose Tissue-Derived Stem Cells and Regulation of Angiogenesis. Front. Physiol. 2018, 9, 30. [Google Scholar] [CrossRef] [PubMed]
- Trivisonno, A.; Alexander, R.W.; Baldari, S.; Cohen, S.R.; Di Rocco, G.; Gentile, P.; Magalon, G.; Magalon, J.; Miller, R.B.; Womack, H.; et al. Intraoperative Strategies for Minimal Manipulation of Autologous Adipose Tissue for Cell- and Tissue-Based Therapies: Concise Review. STEM CELLS Transl. Med. 2019, 8, 1265–1271. [Google Scholar] [CrossRef] [PubMed]
- Aronowitz, J.A.; Lockhart, R.A.; Hakakian, C.S. Mechanical versus enzymatic isolation of stromal vascular fraction cells from adipose tissue. SpringerPlus 2015, 4, 713. [Google Scholar] [CrossRef]
- Bianchi, F.; Maioli, M.; Leonardi, E.; Olivi, E.; Pasquinelli, G.; Valente, S.; Mendez, A.J.; Ricordi, C.; Raffaini, M.; Tremolada, C.; et al. A New Nonenzymatic Method and Device to Obtain a Fat Tissue Derivative Highly Enriched in Pericyte-Like Elements by Mild Mechanical Forces from Human Lipoaspirates. Cell Transplant. 2013, 22, 2063–2077. [Google Scholar] [CrossRef]
- Rodriguez, A.-M.; Elabd, C.; Amri, E.-Z.; Ailhaud, G.; Dani, C. The human adipose tissue is a source of multipotent stem cells. Biochimie 2005, 87, 125–128. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Ojeda, F.J.; Rupérez, A.I.; Gomez-Llorente, C.; Gil, Á.; Aguilera, C.M. Cell Models and Their Application for Studying Adipogenic Differentiation in Relation to Obesity: A Review. Int. J. Mol. Sci. 2016, 17, 1040. [Google Scholar] [CrossRef] [PubMed]
- Basoli, V.; Santaniello, S.; Cruciani, S.; Ginesu, G.C.; Cossu, M.L.; Delitala, A.P.; Serra, P.A.; Ventura, C.; Maioli, M. Melatonin and Vitamin D Interfere with the Adipogenic Fate of Adipose-Derived Stem Cells. Int. J. Mol. Sci. 2017, 18, 981. [Google Scholar] [CrossRef] [PubMed]
- Santaniello, S.; Cruciani, S.; Basoli, V.; Balzano, F.; Bellu, E.; Garroni, G.; Ginesu, G.C.; Cossu, M.L.; Facchin, F.; Delitala, A.P.; et al. Melatonin and Vitamin D Orchestrate Adipose Derived Stem Cell Fate by Modulating Epigenetic Regulatory Genes. Int. J. Med Sci. 2018, 15, 1631–1639. [Google Scholar] [CrossRef] [PubMed]
- Desilets, A.R.; Dhakal-Karki, S.; Dunican, K.C. Role of Metformin for Weight Management in Patients Without Type 2 Diabetes. Ann. Pharmacother. 2008, 42, 817–826. [Google Scholar] [CrossRef] [PubMed]
- Park, M.H.; Kinra, S.; Ward, K.J.; White, B.; Viner, R.M. Metformin for Obesity in Children and Adolescents: A Systematic Review. Diabetes Care 2009, 32, 1743–1745. [Google Scholar] [CrossRef]
- Entezari, M.H.; Khosravi, Z.S.; Kafeshani, M.; Tavasoli, P.; Zadeh, A.H. Effect of Vitamin D supplementation on weight loss, glycemic indices, and lipid profile in obese and overweight women: A clinical trial study. Int. J. Prev. Med. 2018, 9, 63. [Google Scholar] [CrossRef]
- Earthman, C.P.; Beckman, L.M.; Masodkar, K.; Sibley, S.D. The link between obesity and low circulating 25-hydroxyvitamin D concentrations: Considerations and implications. Int. J. Obes. 2011, 36, 387–396. [Google Scholar] [CrossRef]
- Wortsman, J.; Matsuoka, L.Y.; Chen, T.C.; Lu, Z.; Holick, M.F. Decreased bioavailability of vitamin D in obesity. Am. J. Clin. Nutr. 2000, 72, 690–693. [Google Scholar] [CrossRef]
- Szymczak-Pajor, I.; Śliwińska, A. Analysis of Association between Vitamin D Deficiency and Insulin Resistance. Nutrients 2019, 11, 794. [Google Scholar] [CrossRef]
- Gupta, R.P.; Hollis, B.W.; Patel, S.; Patrick, K.S.; Bell, N.H. CYP3A4 is a Human Microsomal Vitamin D 25-Hydroxylase. J. Bone Miner. Res. 2003, 19, 680–688. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Schuetz, E.G.; Xu, Y.; Thummel, K.E. Interplay between vitamin D and the drug metabolizing enzyme CYP3A4. J. Steroid Biochem. Mol. Boil. 2012, 136, 54–58. [Google Scholar] [CrossRef] [PubMed]
- Schuster, I. Cytochromes P450 are essential players in the vitamin D signaling system. Biochim. Biophys. Acta BBA Proteins Proteom. 2011, 1814, 186–199. [Google Scholar] [CrossRef] [PubMed]
- Prosser, D.E.; Jones, G. Enzymes involved in the activation and inactivation of vitamin D. Trends Biochem. Sci. 2004, 29, 664–673. [Google Scholar] [CrossRef]
- Maioli, M.; Rinaldi, S.; Santaniello, S.; Castagna, A.; Pigliaru, G.; Delitala, A.P.; Bianchi, F.; Tremolada, C.; Fontani, V.; Ventura, C. Radioelectric Asymmetric Conveyed Fields and Human Adipose-Derived Stem Cells Obtained with a Nonenzymatic Method and Device: A Novel Approach to Multipotency. Cell Transplant. 2014, 23, 1489–1500. [Google Scholar] [CrossRef]
- Perrini, S.; Porro, S.; Nigro, P.; Cignarelli, A.; Caccioppoli, C.; Genchi, V.A.; Martines, G.; De Fazio, M.; Capuano, P.; Natalicchio, A.; et al. Reduced SIRT1 and SIRT2 expression promotes adipogenesis of human visceral adipose stem cells and associates with accumulation of visceral fat in human obesity. Int. J. Obes. 2019, 44, 307–319. [Google Scholar] [CrossRef]
- Yoo, E.J.; Chung, J.-J.; Choe, S.S.; Kim, K.H.; Kim, J.B. Down-regulation of Histone Deacetylases Stimulates Adipocyte Differentiation. J. Boil. Chem. 2006, 281, 6608–6615. [Google Scholar] [CrossRef]
- He, H.; Cai, M.; Zhu, J.; Xiao, W.; Liu, B.; Shi, Y.; Yang, X.; Liang, X.; Zheng, T.; Hu, S.; et al. miR-148a-3p promotes rabbit preadipocyte differentiation by targeting PTEN. Vitr. Cell. Dev. Boil. Anim. 2018, 54, 241–249. [Google Scholar] [CrossRef]
- Ferguson, B.S.; Nam, H.; Hopkins, R.G.; Morrison, R.F. Impact of Reference Gene Selection for Target Gene Normalization on Experimental Outcome Using Real-Time qRT-PCR in Adipocytes. PLoS ONE 2010, 5, e15208. [Google Scholar] [CrossRef]
- Balzano, F.; Campesi, I.; Cruciani, S.; Garroni, G.; Bellu, E.; Giudici, S.D.; Angius, A.; Oggiano, A.; Rallo, V.; Capobianco, G.; et al. Epigenetics, Stem Cells, and Autophagy: Exploring a Path Involving miRNA. Int. J. Mol. Sci. 2019, 20, 5091. [Google Scholar] [CrossRef]

| Primer Name | Forward | Reverse |
|---|---|---|
| hGAPDH | GAGTCAACGGAATTTGGTCGT | GACAAGCTTCCCGTTCTCAG |
| Oct-4 | GAGGAGTCCCAGGCAATCAA | CATCGGCCTGTGTATATCCC |
| Sox2 | CCGTTCATGTAGGTCTCGGAGCTG | CAACGGCAGCTACAGCTAGATGC |
| NANOG | CATGAGTGTGGATCCAGCT | CCTGAATAAGCAGATCCAT |
| PPAR-γ | AATCCGTCTTCATCCACAGG | GTGAAGACCAGCCTCTTTGC |
| HDAC1 | ACTGCTAAAGTATCACCAGAGGG | CACACTTGGCGTGTCCTTTG |
| SIRT1 | CATTTTCCATGGCGCTGAGG | TGCTGGTGGAACAATTCCTGT |
| SIRT2 | TTGCTGAGCTCCTTGGATGG | GGGGAGGGAGCTGTAAGAGA |
| CYP3A4 | TAGCCCAGCAAAGAGCAACA | CAAAAGGCCTCCGGTTTGTG |
| CYP27B1 | CCTGAACCAGACCATGACCC | GAGCCTTTGCCATTCTTCGC |
| Accession ID Number | Symbol | Sequence |
|---|---|---|
| MIMAT0000437 | hsa-miR-145-5p | GUCCAGUUUUCCCAGGAAUCCCU |
| MIMAT0000243 | hsa-miR-148a-3p | UCAGUGCACUACAGAACUUUGU |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cruciani, S.; Garroni, G.; Balzano, F.; Pala, R.; Bellu, E.; Cossu, M.L.; Ginesu, G.C.; Ventura, C.; Maioli, M. Tuning Adipogenic Differentiation in ADSCs by Metformin and Vitamin D: Involvement of miRNAs. Int. J. Mol. Sci. 2020, 21, 6181. https://doi.org/10.3390/ijms21176181
Cruciani S, Garroni G, Balzano F, Pala R, Bellu E, Cossu ML, Ginesu GC, Ventura C, Maioli M. Tuning Adipogenic Differentiation in ADSCs by Metformin and Vitamin D: Involvement of miRNAs. International Journal of Molecular Sciences. 2020; 21(17):6181. https://doi.org/10.3390/ijms21176181
Chicago/Turabian StyleCruciani, Sara, Giuseppe Garroni, Francesca Balzano, Renzo Pala, Emanuela Bellu, Maria Laura Cossu, Giorgio Carlo Ginesu, Carlo Ventura, and Margherita Maioli. 2020. "Tuning Adipogenic Differentiation in ADSCs by Metformin and Vitamin D: Involvement of miRNAs" International Journal of Molecular Sciences 21, no. 17: 6181. https://doi.org/10.3390/ijms21176181
APA StyleCruciani, S., Garroni, G., Balzano, F., Pala, R., Bellu, E., Cossu, M. L., Ginesu, G. C., Ventura, C., & Maioli, M. (2020). Tuning Adipogenic Differentiation in ADSCs by Metformin and Vitamin D: Involvement of miRNAs. International Journal of Molecular Sciences, 21(17), 6181. https://doi.org/10.3390/ijms21176181






