Plant-Derived Substances in the Fight Against Infections Caused by Candida Species
Abstract
1. Introduction
2. Virulence Factors of Candida Species to be Considered for the Development of New Antifungal Strategies
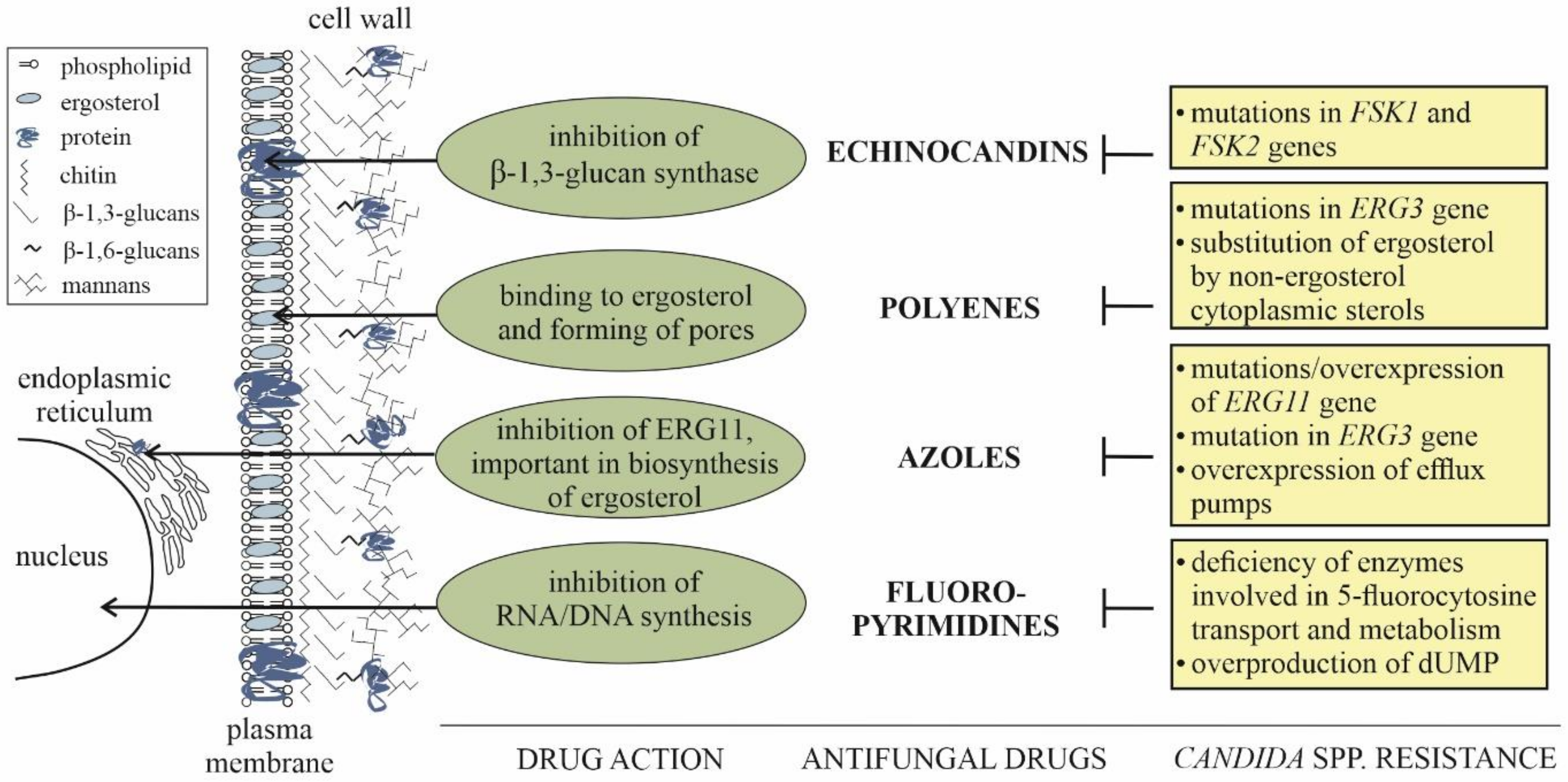
3. Current Methods for Treatment and Management of Superficial and Invasive Candidiases
4. A New Perspective for Plants’ Components in Antifungal Therapy
4.1. Mechanisms of the Action of Plant Metabolites on the Fungal Cell Wall, Plasma Membrane and Biofilm Extracellular Matrix
4.2. Mechanisms of Antivirulence Activity of Plant Metabolites on Candida Species
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Casadevall, A. Fungi and the rise of mammals. PLoS Pathog. 2012, 8, e1002808. [Google Scholar] [CrossRef] [PubMed]
- Anwar, K.P.; Malik, A.; Subhan, K.H. Profile of candidiasis in HIV infected patients. Iran. J. Microbiol. 2012, 4, 204–209. [Google Scholar] [PubMed]
- Dimopoulos, G.; Karabinis, A.; Samonis, G.; Falagas, M.E. Candidemia in immunocompromised and immunocompetent critically ill patients: A prospective comparative study. Eur. J. Clin. Microbiol. Infect. Dis. 2007, 26, 377–384. [Google Scholar] [CrossRef]
- Köhler, J.R.; Casadevall, A.; Perfect, J. The spectrum of fungi that infects humans. Cold Spring Harb. Perspect. Med. 2014, 5, a019273. [Google Scholar] [CrossRef] [PubMed]
- Lamoth, F.; Lockhart, S.R.; Berkow, E.L.; Calandra, T. Changes in the epidemiological landscape of invasive candidiasis. J. Antimicrob. Chemother. 2018, 73, i4–i13. [Google Scholar] [CrossRef] [PubMed]
- Ramos-Martínez, A.; Vicente-López, N.; Sánchez-Romero, I.; Padilla, B.; Merino-Amador, P.; Garnacho-Montero, J.; Ruiz-Camps, I.; Montejo, M.; Salavert, M.; Mensa, J. Members of the CANDIPOP Project from GEIH-GEMICOMED (SEIMC) and REIPI. Epidemiology and prognosis of candidaemia in elderly patients. Mycoses 2017, 60, 808–817. [Google Scholar] [CrossRef]
- Vasileiou, E.; Apsemidou, A.; Vyzantiadis, T.A.; Tragiannidis, A. Invasive candidiasis and candidemia in pediatric and neonatal patients: A review of current guidelines. Curr. Med. Mycol. 2018, 4, 28–33. [Google Scholar] [CrossRef]
- Kullberg, B.J.; Arendrup, M.C. Invasive Candidiasis. N. Engl. J. Med. 2015, 373, 1445–1456. [Google Scholar] [CrossRef]
- Barbosa, A.; Araújo, D.; Ribeiro, E.; Henriques, M.; Silva, S. Candida albicans Adaptation on Simulated Human Body Fluids under Different pH. Microorganisms 2020, 8, 511. [Google Scholar] [CrossRef]
- MacCallum, D.M.; Castillo, L.; Nather, K.; Munro, C.A.; Brown, A.J.; Gow, N.A.; Odds, F.C. Property differences among the four major Candida albicans strain clades. Eukaryot. Cell 2009, 8, 373–387. [Google Scholar] [CrossRef]
- Scordino, F.; Giuffrè, L.; Felice, M.R.; Orlando, M.G.; Medici, M.A.; Marino Merlo, F.; Romeo, O. Genetic diversity of Candida albicans isolates recovered from hospital environments and patients with severe acquired brain injuries. Infect. Genet. Evol. 2019, 76, 104068. [Google Scholar] [CrossRef] [PubMed]
- Diongue, K.; Baha, Z.; Seck, M.C.; Ndiaye, M.; Diallo, M.A.; Ndiaye, D. Cases of skin and nail candidiasis diagnosed at the parasitology and mycology laboratory of Le Dantec University Hospital in Dakar, 2008–2015. Candidoses cutanéo-unguéales diagnostiquées au laboratoire de parasitologie et mycologie du CHU Le Dantec de Dakar de 2008 à 2015. Med. Sante Trop. 2018, 28, 390–394. [Google Scholar] [CrossRef] [PubMed]
- Martins, N.; Ferreira, I.C.; Barros, L.; Silva, S.; Henriques, M. Candidiasis: Predisposing factors, prevention, diagnosis and alternative treatment. Mycopathologia 2014, 177, 223–240. [Google Scholar] [CrossRef] [PubMed]
- Millsop, J.W.; Fazel, N. Oral candidiasis. Clin. Dermatol. 2016, 34, 487–494. [Google Scholar] [CrossRef]
- Qiao, G.L.; Ling, J.; Wong, T.; Yeung, S.N.; Iovieno, A. Candida Keratitis: Epidemiology, Management, and Clinical Outcomes. Cornea 2020, 39, 801–805. [Google Scholar] [CrossRef]
- Yano, J.; Sobel, J.D.; Nyirjesy, P.; Sobel, R.; Williams, V.L.; Yu, Q.; Noverr, M.C.; Fidel, P.L., Jr. Current patient perspectives of vulvovaginal candidiasis: Incidence, symptoms, management and post-treatment outcomes. BMC Womens Health 2019, 19, 48. [Google Scholar] [CrossRef]
- Adams, E.; Quinn, M.; Tsay, S.; Poirot, E.; Chaturvedi, S.; Southwick, K.; Greenko, J.; Fernandez, R.; Kallen, A.; Vallabhaneni, S.; et al. Candida auris in Healthcare Facilities, New York, USA, 2013–2017. Emerg. Infect. Dis. 2018, 24, 1816–1824. [Google Scholar] [CrossRef]
- Hashemi Fesharaki, S.; Aghili, S.R.; Shokohi, T.; Boroumand, M.A. Catheter-related candidemia and identification of causative Candida species in patients with cardiovascular disorder. Curr. Med. Mycol. 2018, 4, 7–13. [Google Scholar] [CrossRef]
- Mamtani, S.; Aljanabi, N.M.; Gupta Rauniyar, R.P.; Acharya, A.; Malik, B.H. Candida Endocarditis: A Review of the Pathogenesis, Morphology, Risk Factors, and Management of an Emerging and Serious Condition. Cureus 2020, 12, e6695. [Google Scholar] [CrossRef]
- Mesini, A.; Mikulska, M.; Giacobbe, D.R.; Del Puente, F.; Gandolfo, N.; Codda, G.; Orsi, A.; Tassinari, F.; Beltramini, S.; Marchese, A. Changing epidemiology of candidaemia: Increase in fluconazole-resistant Candida parapsilosis. Mycoses 2020, 63, 361–368. [Google Scholar] [CrossRef]
- Pappas, P.G.; Lionakis, M.S.; Arendrup, M.C.; Ostrosky-Zeichner, L.; Kullberg, B.J. Invasive candidiasis. Nat. Rev. Dis. Primers 2018, 4, 18026. [Google Scholar] [CrossRef] [PubMed]
- Seok, H.; Huh, K.; Cho, S.Y.; Kang, C.I.; Chung, D.R.; Huh, W.S.; Park, J.B.; Peck, K.R. Invasive Fungal Diseases in Kidney Transplant Recipients: Risk Factors for Mortality. J. Clin. Med. 2020, 9, 1824. [Google Scholar] [CrossRef] [PubMed]
- Ture, Z.; Alp, E. Infection control measures to prevent hospital transmission of Candida. Hosp. Pract. (1995) 2018, 46, 253–257. [Google Scholar] [CrossRef] [PubMed]
- Bitew, A.; Abebaw, Y. Vulvovaginal candidiasis: Species distribution of Candida and their antifungal susceptibility pattern. BMC Womens Health 2018, 18, 94. [Google Scholar] [CrossRef]
- Brunetti, G.; Navazio, A.S.; Giuliani, A.; Giordano, A.; Proli, E.M.; Antonelli, G.; Raponi, G. Candida blood stream infections observed between 2011 and 2016 in a large Italian University Hospital: A time-based retrospective analysis on epidemiology, biofilm production, antifungal agents consumption and drug-susceptibility. PLoS ONE 2019, 14, e0224678. [Google Scholar] [CrossRef]
- Rhodes, J.; Fisher, M.C. Global epidemiology of emerging Candida auris. Curr. Opin. Microbiol. 2019, 52, 84–89. [Google Scholar] [CrossRef]
- Sardi, J.; Scorzoni, L.; Bernardi, T.; Fusco-Almeida, A.M.; Mendes Giannini, M. Candida species: Current epidemiology, pathogenicity, biofilm formation, natural antifungal products and new therapeutic options. J. Med. Microbiol. 2013, 62, 10–24. [Google Scholar] [CrossRef]
- Sadeghi, G.; Ebrahimi-Rad, M.; Mousavi, S.F.; Shams-Ghahfarokhi, M.; Razzaghi-Abyaneh, M. Emergence of non-Candida albicans species: Epidemiology, phylogeny and fluconazole susceptibility profile. J. Mycol. Med. 2018, 28, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Suryana, K.; Suharsono, H.; Antara, I. Factors Associated with Oral Candidiasis in People Living with HIV/AIDS: A Case Control Study. HIV/AIDS (Auckl) 2020, 12, 33–39. [Google Scholar] [CrossRef]
- Pfaller, M.A.; Diekema, D.J.; Turnidge, J.D.; Castanheira, M.; Jones, R.N. Twenty Years of the SENTRY Antifungal Surveillance Program: Results for Candida Species From 1997–2016. Open Forum Infect. Dis. 2019, 6, S79–S94. [Google Scholar] [CrossRef]
- Sudbery, P.; Gow, N.; Berman, J. The distinct morphogenic states of Candida albicans. Trends Microbiol. 2004, 12, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Satala, D.; Karkowska-Kuleta, J.; Zelazna, A.; Rapala-Kozik, M.; Kozik, A. Moonlighting Proteins at the Candidal Cell Surface. Microorganisms 2020, 8, 1046. [Google Scholar] [CrossRef] [PubMed]
- Murciano, C.; Moyes, D.L.; Runglall, M.; Tobouti, P.; Islam, A.; Hoyer, L.L.; Naglik, J.R. Evaluation of the role of Candida albicans agglutinin-like sequence (Als) proteins in human oral epithelial cell interactions. PLoS ONE 2012, 7, e33362. [Google Scholar] [CrossRef] [PubMed]
- Verstrepen, K.J.; Klis, F.M. Flocculation, adhesion and biofilm formation in yeasts. Mol. Microbiol. 2006, 60, 5–15. [Google Scholar] [CrossRef] [PubMed]
- Staab, J.F.; Bradway, S.D.; Fidel, P.L.; Sundstrom, P. Adhesive and mammalian transglutaminase substrate properties of Candida albicans Hwp1. Science 1999, 283, 1535–1538. [Google Scholar] [CrossRef] [PubMed]
- Naglik, J.R.; Moyes, D.L.; Wächtler, B.; Hube, B. Candida albicans interactions with epithelial cells and mucosal immunity. Microbes Infect. 2011, 13, 963–976. [Google Scholar] [CrossRef]
- Fanning, S.; Mitchell, A.P. Fungal biofilms. PLoS Pathog. 2012, 8, e1002585. [Google Scholar] [CrossRef]
- Mayer, F.L.; Wilson, D.; Hube, B. Candida albicans pathogenicity mechanisms. Virulence 2013, 4, 119–128. [Google Scholar] [CrossRef]
- Nobile, C.J.; Nett, J.E.; Hernday, A.D.; Homann, O.R.; Deneault, J.S.; Nantel, A.; Andes, D.R.; Johnson, A.D.; Mitchell, A.P. Biofilm matrix regulation by Candida albicans Zap1. PLoS Biol. 2009, 7, e1000133. [Google Scholar] [CrossRef]
- Taff, H.T.; Nett, J.E.; Zarnowski, R.; Ross, K.M.; Sanchez, H.; Cain, M.T.; Hamaker, J.; Mitchell, A.P.; Andes, D.R. A Candida biofilm-induced pathway for matrix glucan delivery: Implications for drug resistance. PLoS Pathog. 2012, 8, e1002848. [Google Scholar] [CrossRef]
- Theiss, S.; Ishdorj, G.; Brenot, A.; Kretschmar, M.; Lan, C.Y.; Nichterlein, T.; Hacker, J.; Nigam, S.; Agabian, N.; Köhler, G.A. Inactivation of the phospholipase B gene PLB5 in wild-type Candida albicans reduces cell-associated phospholipase A2 activity and attenuates virulence. Int. J. Med. Microbiol. 2006, 296, 405–420. [Google Scholar] [CrossRef] [PubMed]
- Ying, S.; Chunyang, L. Correlation between phospholipase of Candida albicans and resistance to fluconazole. Mycoses 2012, 55, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Gácser, A.; Stehr, F.; Kröger, C.; Kredics, L.; Schäfer, W.; Nosanchuk, J.D. Lipase 8 affects the pathogenesis of Candida albicans. Infect. Immun. 2007, 75, 4710–4718. [Google Scholar] [CrossRef] [PubMed]
- Bochenska, O.; Rapala-Kozik, M.; Wolak, N.; Kamysz, W.; Grzywacz, D.; Aoki, W.; Ueda, M.; Kozik, A. Inactivation of human kininogen-derived antimicrobial peptides by secreted aspartic proteases produced by the pathogenic yeast Candida albicans. Biol. Chem. 2015, 396, 1369–1375. [Google Scholar] [CrossRef]
- Naglik, J.R.; Challacombe, S.J.; Hube, B. Candida albicans secreted aspartyl proteinases in virulence and pathogenesis. Microbiol. Mol. Biol. Rev. 2003, 67, 400–428. [Google Scholar] [CrossRef]
- Rapala-Kozik, M.; Bochenska, O.; Zawrotniak, M.; Wolak, N.; Trebacz, G.; Gogol, M.; Ostrowska, D.; Aoki, W.; Ueda, M.; Kozik, A. Inactivation of the antifungal and immunomodulatory properties of human cathelicidin LL-37 by aspartic proteases produced by the pathogenic yeast Candida albicans. Infect. Immun. 2015, 83, 2518–2530. [Google Scholar] [CrossRef]
- Singh, D.K.; Tóth, R.; Gácser, A. Mechanisms of Pathogenic Candida Species to Evade the Host Complement Attack. Front. Cell Infect. Microbiol. 2020, 10, 94. [Google Scholar] [CrossRef]
- Kozik, A.; Gogol, M.; Bochenska, O.; Karkowska-Kuleta, J.; Wolak, N.; Kamysz, W.; Aoki, W.; Ueda, M.; Faussner, A.; Rapala-Kozik, M. Kinin release from human kininogen by 10 aspartic proteases produced by pathogenic yeast Candida albicans. BMC Microbiol. 2015, 15, 60. [Google Scholar] [CrossRef]
- Bondaryk, M.; Kurzątkowski, W.; Staniszewska, M. Antifungal agents commonly used in the superficial and mucosal candidiasis treatment: Mode of action and resistance development. Postepy Dermatol. Alergol. 2013, 30, 293–301. [Google Scholar] [CrossRef]
- Barker, K.S.; Rogers, P.D. Recent insights into the mechanisms of antifungal resistance. Curr. Infect. Dis. Rep. 2006, 8, 449–456. [Google Scholar] [CrossRef]
- Ripeau, J.S.; Aumont, F.; Belhumeur, P.; Ostrosky-Zeichner, L.; Rex, J.H.; de Repentigny, L. Effect of the echinocandin caspofungin on expression of Candida albicans secretory aspartyl proteinases and phospholipase in vitro. Antimicrob. Agents Chemother. 2002, 46, 3096–3100. [Google Scholar] [CrossRef] [PubMed]
- Stan, C.D.; Tuchiluş, C.; Stan, C.I. Echinocandins--new antifungal agents. Rev. Med. Chir. Soc. Med. Nat. Iasi 2014, 118, 528–536. [Google Scholar]
- Garcia-Effron, G.; Katiyar, S.K.; Park, S.; Edlind, T.D.; Perlin, D.S. A naturally occurring proline-to-alanine amino acid change in Fks1p in Candida parapsilosis, Candida orthopsilosis, and Candida metapsilosis accounts for reduced echinocandin susceptibility. Antimicrob. Agents Chemother. 2008, 52, 2305–2312. [Google Scholar] [CrossRef] [PubMed]
- Orasch, C.; Marchetti, O.; Garbino, J.; Schrenzel, J.; Zimmerli, S.; Mühlethaler, K.; Pfyffer, G.; Ruef, C.; Fehr, J.; Zbinden, R. FUNGINOS. Candida species distribution and antifungal susceptibility testing according to European Committee on Antimicrobial Susceptibility Testing and new vs. old Clinical and Laboratory Standards Institute clinical breakpoints: A 6-year prospective candidaemia survey from the fungal infection network of Switzerland. Clin. Microbiol. Infect. 2014, 20, 698–705. [Google Scholar] [CrossRef]
- Garcia-Effron, G.; Chua, D.J.; Tomada, J.R.; DiPersio, J.; Perlin, D.S.; Ghannoum, M.; Bonilla, H. Novel FKS mutations associated with echinocandin resistance in Candida species. Antimicrob. Agents Chemother. 2010, 54, 2225–2227. [Google Scholar] [CrossRef] [PubMed]
- Kordalewska, M.; Lee, A.; Park, S.; Berrio, I.; Chowdhary, A.; Zhao, Y.; Perlin, D.S. Understanding Echinocandin Resistance in the Emerging Pathogen Candida auris. Antimicrob. Agents Chemother. 2018, 62, e00238-18. [Google Scholar] [CrossRef]
- Taff, H.T.; Mitchell, K.F.; Edward, J.A.; Andes, D.R. Mechanisms of Candida biofilm drug resistance. Future Microbiol. 2013, 8, 1325–1337. [Google Scholar] [CrossRef]
- Pappas, P.G.; Kauffman, C.A.; Andes, D.R.; Clancy, C.J.; Marr, K.A.; Ostrosky-Zeichner, L.; Reboli, A.C.; Schuster, M.G.; Vazquez, J.A.; Walsh, T.J. Clinical Practice Guideline for the Management of Candidiasis: 2016 Update by the Infectious Diseases Society of America. Clin. Infect. Dis. 2016, 62, e1–e50. [Google Scholar] [CrossRef]
- Bersani, I.; Piersigilli, F.; Goffredo, B.M.; Santisi, A.; Cairoli, S.; Ronchetti, M.P.; Auriti, C. Antifungal Drugs for Invasive Candida Infections (ICI) in Neonates: Future Perspectives. Front. Pediatr. 2019, 7, 375. [Google Scholar] [CrossRef]
- Aparicio, J.F.; Barreales, E.G.; Payero, T.D.; Vicente, C.M.; de Pedro, A.; Santos-Aberturas, J. Biotechnological production and application of the antibiotic pimaricin: Biosynthesis and its regulation. Appl. Microbiol. Biotechnol. 2016, 100, 61–78. [Google Scholar] [CrossRef]
- Fjaervik, E.; Zotchev, S.B. Biosynthesis of the polyene macrolide antibiotic nystatin in Streptomyces noursei. Appl. Microbiol. Biotechnol. 2005, 67, 436–443. [Google Scholar] [CrossRef] [PubMed]
- Odds, F.C.; Brown, A.J.; Gow, N.A. Antifungal agents: Mechanisms of action. Trends Microbiol. 2003, 11, 272–279. [Google Scholar] [CrossRef]
- Ahmad Khan, M.S.; Alshehrei, F.; Al-Ghamdi, S.B.; Bamaga, M.A.; Al-Thubiani, A.S.; Alam, M.Z. Virulence and biofilms as promising targets in developing antipathogenic drugs against candidiasis. Future Sci. OA 2020, 6, FSO440. [Google Scholar] [CrossRef] [PubMed]
- Laniado-Laborín, R.; Cabrales-Vargas, M.N. Amphotericin B: Side effects and toxicity. Rev. Iberoam. Micol. 2009, 26, 223–227. [Google Scholar] [CrossRef]
- Salazar, S.B.; Simões, R.S.; Pedro, N.A.; Pinheiro, M.J.; Carvalho, M.; Mira, N.P. An Overview on Conventional and Non-Conventional Therapeutic Approaches for the Treatment of Candidiasis and Underlying Resistance Mechanisms in Clinical Strains. J. Fungi 2020, 6, 23. [Google Scholar] [CrossRef]
- Arendrup, M.C.; Dzajic, E.; Jensen, R.H.; Johansen, H.K.; Kjaeldgaard, P.; Knudsen, J.D.; Kristensen, L.; Leitz, C.; Lemming, L.E.; Nielsen, L. Epidemiological changes with potential implication for antifungal prescription recommendations for fungaemia: Data from a nationwide fungaemia surveillance programme. Clin. Microbiol. Infect. 2013, 19, E343–E353. [Google Scholar] [CrossRef]
- Vandeputte, P.; Ferrari, S.; Coste, A.T. Antifungal resistance and new strategies to control fungal infections. Int. J. Microbiol. 2012, 713687. [Google Scholar] [CrossRef]
- De Oliveira Santos, G.C.; Vasconcelos, C.C.; Lopes, A.; de Sousa Cartágenes, M.; Filho, A.; do Nascimento, F.; Ramos, R.M.; Pires, E.; de Andrade, M.S.; Rocha, F. Candida Infections and Therapeutic Strategies: Mechanisms of Action for Traditional and Alternative Agents. Front. Microbiol. 2018, 9, 1351. [Google Scholar] [CrossRef]
- Hoekstra, W.J.; Garvey, E.P.; Moore, W.R.; Rafferty, S.W.; Yates, C.M.; Schotzinger, R.J. Design and optimization of highly-selective fungal CYP51 inhibitors. Bioorg. Med. Chem. Lett. 2014, 24, 3455–3458. [Google Scholar] [CrossRef]
- Healey, K.R.; Kordalewska, M.; Jiménez Ortigosa, C.; Singh, A.; Berrío, I.; Chowdhary, A.; Perlin, D.S. Limited ERG11 Mutations Identified in Isolates of Candida auris Directly Contribute to Reduced Azole Susceptibility. Antimicrob. Agents Chemother. 2018, 62, e01427-18. [Google Scholar] [CrossRef]
- Autmizguine, J.; Smith, P.B.; Prather, K.; Bendel, C.; Natarajan, G.; Bidegain, M.; Kaufman, D.A.; Burchfield, D.J.; Ross, A.S.; Pandit, P.; et al. Effect of fluconazole prophylaxis on Candida fluconazole susceptibility in premature infants. J. Antimicrob. Chemother. 2018, 73, 3482–3487. [Google Scholar] [CrossRef]
- Bennett, J.E.; Izumikawa, K.; Marr, K.A. Mechanism of increased fluconazole resistance in Candida glabrata during prophylaxis. Antimicrob. Agents Chemother. 2004, 48, 1773–1777. [Google Scholar] [CrossRef] [PubMed]
- Fisher, M.C.; Hawkins, N.J.; Sanglard, D.; Gurr, S.J. Worldwide emergence of resistance to antifungal drugs challenges human health and food security. Science 2018, 360, 739–742. [Google Scholar] [CrossRef] [PubMed]
- Desnos-Ollivier, M.; Bretagne, S.; Bernède, C.; Robert, V.; Raoux, D.; Chachaty, E.; Forget, E.; Lacroix, C.; Dromer, F.; Yeasts Group. Clonal population of flucytosine-resistant Candida tropicalis from blood cultures, Paris, France. Emerg. Infect. Dis. 2008, 14, 557–565. [Google Scholar] [CrossRef]
- Lockhart, S.R. Candida auris and multidrug resistance: Defining the new normal. Fungal Genet. Biol. 2019, 131, 103243. [Google Scholar] [CrossRef] [PubMed]
- Aldholmi, M.; Marchand, P.; Ourliac-Garnier, I.; Le Pape, P.; Ganesan, A. A Decade of Antifungal Leads from Natural Products: 2010–2019. Pharmaceuticals 2019, 12, 182. [Google Scholar] [CrossRef]
- Felice, M.R.; Giuffrè, L.; El Aamri, L.; Hafidi, M.; Criseo, G.; Romeo, O.; Scordino, F. Looking for New Antifungal Drugs from Flavonoids: Impact of the Genetic Diversity of Candida albicans on the in-vitro Response. Curr. Med. Chem. 2019, 26, 5108–5123. [Google Scholar] [CrossRef]
- Martins, N.; Barros, L.; Henriques, M.; Silva, S.; Ferreira, I.C.R.F. Activity of phenolic compounds from plant origin against Candida species. Ind. Crops Prod. 2015, 74, 648–670. [Google Scholar] [CrossRef]
- Noguti, J.; Rajinia, M.; Zancope, B.R.; Marquezin, M.; Seleem, D.; Pardi, V.; Murata, R.M. Antifungal Activity of Alkaloids Against Candida albicans. J. Calif. Dent. Assoc. 2016, 44, 493–498. [Google Scholar]
- Palande, V.; Jaitly, P.; Kunchiraman, B.N. Plants with anti-Candida activity and their mechanism of action: A review. J. Environ. Res. Develop. 2015, 9, 1189–1196. [Google Scholar]
- Teodoro, G.R.; Ellepola, K.; Seneviratne, C.J.; Koga-Ito, C.Y. Potential Use of Phenolic Acids as Anti-Candida Agents: A Review. Front. Microbiol. 2015, 6, 1420. [Google Scholar] [CrossRef]
- Wojtunik-Kulesza, K.A.; Kasprzak, K.; Oniszczuk, T.; Oniszczuk, A. Natural Monoterpenes: Much More than Only a Scent. Chem. Biodivers. 2019, 16, e1900434. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, K.F.; Zarnowski, R.; Andes, D.R. The Extracellular Matrix of Fungal Biofilms. Adv. Exp. Med. Biol. 2016, 931, 21–35. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, K.F.; Zarnowski, R.; Sanchez, H.; Edward, J.A.; Reinicke, E.L.; Nett, J.E.; Mitchell, A.P.; Andes, D.R. Community participation in biofilm matrix assembly and function. Proc. Natl. Acad. Sci. USA 2015, 112, 4092–4097. [Google Scholar] [CrossRef] [PubMed]
- Farisa Banu, S.; Rubini, D.; Shanmugavelan, P.; Murugan, R.; Gowrishankar, S.; Karutha Pandian, S.; Nithyanand, P. Effects of patchouli and cinnamon essential oils on biofilm and hyphae formation by Candida species. J. Mycol. Med. 2018, 28, 332–339. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.S.; Kim, Y. Aucklandia lappa causes cell wall damage in Candida albicans by reducing chitin and (1,3)-β-D-glucan. J. Microbiol. Biotechnol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.S.; Kim, Y. Paeonia lactiflora Inhibits Cell Wall Synthesis and Triggers Membrane Depolarization in Candida albicans. J. Microbiol. Biotechnol. 2017, 27, 395–404. [Google Scholar] [CrossRef] [PubMed]
- Qian, W.; Yang, M.; Li, X.; Sun, Z.; Li, Y.; Wang, X.; Wang, T. Anti-microbial and anti-biofilm activities of combined chelerythrine-sanguinarine and mode of action against Candida albicans and Cryptococcus neoformans in vitro. Colloids Surf. B Biointerfaces 2020, 191, 111003. [Google Scholar] [CrossRef]
- Gupta, P.; Gupta, S.; Sharma, M.; Kumar, N.; Pruthi, V.; Poluri, K.M. Effectiveness of Phytoactive Molecules on Transcriptional Expression, Biofilm Matrix, and Cell Wall Components of Candida glabrata and Its Clinical Isolates. ACS Omega 2018, 3, 12201–12214. [Google Scholar] [CrossRef]
- Lee, K.K.; Kubo, K.; Abdelaziz, J.A.; Cunningham, I.; de Silva Dantas, A.; Chen, X.; Okada, H.; Ohya, Y.; Gow, N. Yeast species-specific, differential inhibition of β-1,3-glucan synthesis by poacic acid and caspofungin. Cell Surf. 2018, 3, 12–25. [Google Scholar] [CrossRef]
- Piotrowski, J.S.; Okada, H.; Lu, F.; Li, S.C.; Hinchman, L.; Ranjan, A.; Smith, D.L.; Higbee, A.J.; Ulbrich, A.; Coon, J.J. Plant-derived antifungal agent poacic acid targets β-1,3-glucan. Proc. Natl. Acad. Sci. USA 2015, 112, E1490–E1497. [Google Scholar] [CrossRef]
- Lee, J.H.; Kim, Y.G.; Khadke, S.K.; Yamano, A.; Watanabe, A.; Lee, J. Inhibition of Biofilm Formation by Candida albicans and Polymicrobial Microorganisms by Nepodin via Hyphal-Growth Suppression. ACS Infect. Dis. 2019, 5, 1177–1187. [Google Scholar] [CrossRef] [PubMed]
- Sharma, Y.; Khan, L.A.; Manzoor, N. Anti-Candida activity of geraniol involves disruption of cell membrane integrity and function. J. Mycol. Med. 2016, 26, 244–254. [Google Scholar] [CrossRef] [PubMed]
- Sharma, Y.; Rastogi, S.K.; Ahmedi, S.; Manzoor, N. Antifungal activity of β-citronellol against two non-albicans Candida species. J. Essent. Oil Res. 2020, 32, 198–208. [Google Scholar] [CrossRef]
- Xu, J.; Liu, R.; Sun, F.; An, L.; Shang, Z.; Kong, L.; Yang, M. Eucalyptal D Enhances the Antifungal Effect of Fluconazole on Fluconazole-Resistant Candida albicans by Competitively Inhibiting Efflux Pump. Front. Cell. Infect. Microbiol. 2019, 9, 211. [Google Scholar] [CrossRef]
- Essid, R.; Hammami, M.; Gharbi, D.; Karkouch, I.; Hamouda, T.B.; Elkahoui, S.; Limam, F.; Tabbene, O. Antifungal mechanism of the combination of Cinnamomum verum and Pelargonium graveolens essential oils with fluconazole against pathogenic Candida strains. Appl. Microbiol. Biotechnol. 2017, 101, 6993–7006. [Google Scholar] [CrossRef]
- Patel, M.; Srivastava, V.; Ahmad, A. Dodonaea viscosa var angustifolia derived 5,6,8-trihydroxy-7,4′ dimethoxy flavone inhibits ergosterol synthesis and the production of hyphae and biofilm in Candida albicans. J. Ethnopharmacol. 2020, 259, 112965, Advance online publication. [Google Scholar] [CrossRef]
- Muthamil, S.; Balasubramaniam, B.; Balamurugan, K.; Pandian, S.K. Synergistic Effect of Quinic Acid Derived From Syzygium cumini and Undecanoic Acid Against Candida spp. Biofilm and Virulence. Front. Microbiol. 2018, 9, 2835. [Google Scholar] [CrossRef]
- Shao, J.; Shi, G.; Wang, T.; Wu, D.; Wang, C. Antiproliferation of Berberine in Combination with Fluconazole from the Perspectives of Reactive Oxygen Species, Ergosterol and Drug Efflux in a Fluconazole-Resistant Candida tropicalis Isolate. Front. Microbiol. 2016, 7, 1516. [Google Scholar] [CrossRef]
- Bondaryk, M.; Staniszewska, M.; Zielińska, P.; Urbańczyk-Lipkowska, Z. Natural Antimicrobial Peptides as Inspiration for Design of a New Generation Antifungal Compounds. J. Fungi 2017, 3, 46. [Google Scholar] [CrossRef]
- Girardot, M.; Imbert, C. Natural Sources as Innovative Solutions Against Fungal Biofilms. Adv. Exp. Med. Biol. 2016, 931, 105–125. [Google Scholar] [CrossRef] [PubMed]
- Soliman, S.; Alnajdy, D.; El-Keblawy, A.A.; Mosa, K.A.; Khoder, G.; Noreddin, A.M. Plants’ Natural Products as Alternative Promising Anti-Candida Drugs. Pharmacogn. Rev. 2017, 11, 104–122. [Google Scholar] [CrossRef] [PubMed]
- Barbosa Pelegrini, P.; Del Sarto, R.P.; Silva, O.N.; Franco, O.L.; Grossi-de-Sa, M.F. Antibacterial peptides from plants: What they are and how they probably work. Biochem. Res. Int. 2011, 250349. [Google Scholar] [CrossRef] [PubMed]
- Vriens, K.; Cammue, B.P.; Thevissen, K. Antifungal plant defensins: Mechanisms of action and production. Molecules 2014, 19, 12280–12303. [Google Scholar] [CrossRef]
- Vriens, K.; Cools, T.L.; Harvey, P.J.; Craik, D.J.; Braem, A.; Vleugels, J.; De Coninck, B.; Cammue, B.P.; Thevissen, K. The radish defensins RsAFP1 and RsAFP2 act synergistically with caspofungin against Candida albicans biofilms. Peptides 2016, 75, 71–79. [Google Scholar] [CrossRef]
- Vriens, K.; Cools, T.L.; Harvey, P.J.; Craik, D.J.; Spincemaille, P.; Cassiman, D.; Braem, A.; Vleugels, J.; Nibbering, P.H.; Drijfhout, J.W. Synergistic Activity of the Plant Defensin HsAFP1 and Caspofungin against Candida albicans Biofilms and Planktonic Cultures. PLoS ONE 2015, 10, e0132701. [Google Scholar] [CrossRef]
- Cools, T.L.; Struyfs, C.; Drijfhout, J.W.; Kucharíková, S.; Lobo Romero, C.; Van Dijck, P.; Ramada, M.; Bloch, C., Jr.; Cammue, B.; Thevissen, K. A Linear 19-Mer Plant Defensin-Derived Peptide Acts Synergistically with Caspofungin against Candida albicans Biofilms. Front. Microbiol. 2017, 8, 2051. [Google Scholar] [CrossRef]
- Taveira, G.B.; Carvalho, A.O.; Rodrigues, R.; Trindade, F.G.; Da Cunha, M.; Gomes, V.M. Thionin-like peptide from Capsicum annuum fruits: Mechanism of action and synergism with fluconazole against Candida species. BMC Microbiol. 2016, 16, 12. [Google Scholar] [CrossRef]
- Park, S.C.; Kim, I.R.; Hwang, J.E.; Kim, J.Y.; Jung, Y.J.; Choi, W.; Lee, Y.; Jang, M.K.; Lee, J.R. Functional Mechanisms Underlying the Antimicrobial Activity of the Oryza sativa Trx-like Protein. Int. J. Mol. Sci. 2019, 20, 1413. [Google Scholar] [CrossRef]
- Loo, S.; Kam, A.; Xiao, T.; Tam, J.P. Bleogens: Cactus-Derived Anti-Candida Cysteine-Rich Peptides with Three Different Precursor Arrangements. Front. Plant. Sci. 2017, 8, 2162. [Google Scholar] [CrossRef]
- Muthamil, S.; Prasath, K.G.; Priya, A.; Precilla, P.; Pandian, S.K. Global proteomic analysis deciphers the mechanism of action of plant derived oleic acid against Candida albicans virulence and biofilm formation. Sci. Rep. 2020, 10, 5113. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Shan, M.; Li, S.; Wang, Y.; Yang, H.; Chen, Y.; Gu, B.; Zhu, Z. Teasaponin suppresses Candida albicans filamentation by reducing the level of intracellular cAMP. Ann. Transl. Med. 2020, 8, 175. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Liao, K.; Wang, D. Effects of magnolol and honokiol on adhesion, yeast-hyphal transition, and formation of biofilm by Candida albicans. PLoS ONE 2015, 10, e0117695. [Google Scholar] [CrossRef] [PubMed]
- Zhong, H.; Hu, D.D.; Hu, G.H.; Su, J.; Bi, S.; Zhang, Z.E.; Wang, Z.; Zhang, R.L.; Xu, Z.; Jiang, Y.Y. Activity of Sanguinarine against Candida albicans Biofilms. Antimicrob. Agents Chemother. 2017, 61, e02259-16. [Google Scholar] [CrossRef] [PubMed]
- Manoharan, R.K.; Lee, J.H.; Lee, J. Antibiofilm and Antihyphal Activities of Cedar Leaf Essential Oil, Camphor, and Fenchone Derivatives against Candida albicans. Front. Microbiol. 2017, 8, 1476. [Google Scholar] [CrossRef] [PubMed]
- Manoharan, R.K.; Lee, J.H.; Kim, Y.G.; Lee, J. Alizarin and Chrysazin Inhibit Biofilm and Hyphal Formation by Candida albicans. Front. Cell. Infect. Microbiol. 2017, 7, 447. [Google Scholar] [CrossRef]
- Yan, Y.; Tan, F.; Miao, H.; Wang, H.; Cao, Y. Effect of Shikonin Against Candida albicans Biofilms. Front. Microbiol. 2019, 10, 1085. [Google Scholar] [CrossRef]
- Khan, S.N.; Khan, S.; Iqbal, J.; Khan, R.; Khan, A.U. Enhanced Killing and Antibiofilm Activity of Encapsulated Cinnamaldehyde against Candida albicans. Front. Microbiol. 2017, 8, 1641. [Google Scholar] [CrossRef]
- Lee, J.H.; Kim, Y.G.; Choi, P.; Ham, J.; Park, J.G.; Lee, J. Antibiofilm and Antivirulence Activities of 6-Gingerol and 6-Shogaol Against Candida albicans Due to Hyphal Inhibition. Front. Cell. Infect. Microbiol. 2018, 8, 299. [Google Scholar] [CrossRef]
- Lin, M.Y.; Yuan, Z.L.; Hu, D.D.; Hu, G.H.; Zhang, R.L.; Zhong, H.; Yan, L.; Jiang, Y.Y.; Su, J.; Wang, Y. Effect of loureirin A against Candida albicans biofilms. Chin. J. Nat. Med. 2019, 17, 616–623. [Google Scholar] [CrossRef]
- Xu, K.; Wang, J.L.; Chu, M.P.; Jia, C. Activity of coumarin against Candida albicans biofilms. J. Mycol. Med. 2019, 29, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Shin, D.S.; Eom, Y.B. Efficacy of zerumbone against dual-species biofilms of Candida albicans and Staphylococcus aureus. Microb. Pathog. 2019, 137, 103768. [Google Scholar] [CrossRef] [PubMed]
- Shirtliff, M.E.; Krom, B.P.; Meijering, R.A.; Peters, B.M.; Zhu, J.; Scheper, M.A.; Harris, M.L.; Jabra-Rizk, M.A. Farnesol-induced apoptosis in Candida albicans. Antimicrob. Agents Chemother. 2009, 53, 2392–2401. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Wang, Z.; Liu, L.; Qu, S.; Mao, Y.; Peng, X.; Li, Y.X.; Tian, J. Cinnamaldehyde inhibits Candida albicans growth by causing apoptosis and its treatment on vulvovaginal candidiasis and oropharyngeal candidiasis. Appl. Microbiol. Biotechnol. 2019, 103, 9037–9055. [Google Scholar] [CrossRef]
- Hwang, J.H.; Choi, H.; Kim, A.R.; Yun, J.W.; Yu, R.; Woo, E.R.; Lee, D.G. Hibicuslide C-induced cell death in Candida albicans involves apoptosis mechanism. J. Appl. Microbiol. 2014, 117, 1400–1411. [Google Scholar] [CrossRef]
- Wang, T.; Shi, G.; Shao, J.; Wu, D.; Yan, Y.; Zhang, M.; Cui, Y.; Wang, C. In vitro antifungal activity of baicalin against Candida albicans biofilms via apoptotic induction. Microb. Pathog. 2015, 87, 21–29. [Google Scholar] [CrossRef]
- Yun, D.G.; Lee, D.G. Silibinin triggers yeast apoptosis related to mitochondrial Ca2+ influx in Candida albicans. Int. J. Biochem. Cell Biol. 2016, 80, 1–9. [Google Scholar] [CrossRef]
- Choi, H.; Lee, D.G. Lycopene induces apoptosis in Candida albicans through reactive oxygen species production and mitochondrial dysfunction. Biochimie 2015, 115, 108–115. [Google Scholar] [CrossRef]
- Araújo, N.; Dias, L.P.; Costa, H.; Sousa, D.; Vasconcelos, I.M.; de Morais, G.A.; Oliveira, J. ClTI, a Kunitz trypsin inhibitor purified from Cassia leiandra Benth. seeds, exerts a candidicidal effect on Candida albicans by inducing oxidative stress and necrosis. Biochim. Biophys. Acta. Biomembr. 2019, 1861, 183032. [Google Scholar] [CrossRef]
- Da Silva, P.M.; de Moura, M.C.; Gomes, F.S.; da Silva Trentin, D.; Silva de Oliveira, A.P.; de Mello, G.; da Rocha Pitta, M.G.; de Melo Rego, M.; Coelho, L.; Macedo, A.J. PgTeL, the lectin found in Punica granatum juice, is an antifungal agent against Candida albicans and Candida krusei. Int. J. Biol. Macromol. 2018, 108, 391–400. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guevara-Lora, I.; Bras, G.; Karkowska-Kuleta, J.; González-González, M.; Ceballos, K.; Sidlo, W.; Rapala-Kozik, M. Plant-Derived Substances in the Fight Against Infections Caused by Candida Species. Int. J. Mol. Sci. 2020, 21, 6131. https://doi.org/10.3390/ijms21176131
Guevara-Lora I, Bras G, Karkowska-Kuleta J, González-González M, Ceballos K, Sidlo W, Rapala-Kozik M. Plant-Derived Substances in the Fight Against Infections Caused by Candida Species. International Journal of Molecular Sciences. 2020; 21(17):6131. https://doi.org/10.3390/ijms21176131
Chicago/Turabian StyleGuevara-Lora, Ibeth, Grazyna Bras, Justyna Karkowska-Kuleta, Miriam González-González, Kinga Ceballos, Wiktoria Sidlo, and Maria Rapala-Kozik. 2020. "Plant-Derived Substances in the Fight Against Infections Caused by Candida Species" International Journal of Molecular Sciences 21, no. 17: 6131. https://doi.org/10.3390/ijms21176131
APA StyleGuevara-Lora, I., Bras, G., Karkowska-Kuleta, J., González-González, M., Ceballos, K., Sidlo, W., & Rapala-Kozik, M. (2020). Plant-Derived Substances in the Fight Against Infections Caused by Candida Species. International Journal of Molecular Sciences, 21(17), 6131. https://doi.org/10.3390/ijms21176131




