Signaling Molecules Regulating Pancreatic Endocrine Development from Pluripotent Stem Cell Differentiation
Abstract
1. Introduction
2. Molecules Promoting the Generation of Functional β-Cells from Human Pluripotent Stem Cells
3. Signaling Pathways Involved in In Vitro Islet β-Cell Development
4. Microenvironments That Facilitate the Formation of β-Cells from Stem Cell Differentiation
5. Molecules Critical for the Generation of Functional α-Cells from hPSC Differentiation
6. Generation of Other Types of Islet Cells
7. Approaches for the Generation of Islet Organoids from hPSCs
7.1. Suspension Cultures Enhance the Differentiation of Islet Organoids
7.2. ECM Signaling Enhances the Differentiation of Islet Organoids
8. Perspective
Author Contributions
Funding
Conflicts of Interest
References
- Jin, S. Regeneration of Islet beta-Cells from Stem Cells and Progenitors. J. Stem. Cell. Res. Transplant. 2014, 1, 1001–1004. [Google Scholar]
- Rezania, A.; Bruin, J.E.; Arora, P.; Rubin, A.; Batushansky, I.; Asadi, A.; O’Dwyer, S.; Quiskamp, N.; Mojibian, M.; Albrecht, T.; et al. Reversal of diabetes with insulin-producing cells derived in vitro from human pluripotent stem cells. Nat. Biotechnol. 2014, 32, 1121–1133. [Google Scholar] [CrossRef]
- Pagliuca, F.W.; Millman, J.R.; Gurtler, M.; Segel, M.; Van Dervort, A.; Ryu, J.H.; Peterson, Q.P.; Greiner, D.; Melton, D.A. Generation of functional human pancreatic beta cells in vitro. Cell 2014, 159, 428–439. [Google Scholar] [CrossRef]
- Zhang, N.; Jiang, W.; Liu, M.; Sui, X.; Yin, X.; Chen, S.; Shi, Y.; Deng, H. Highly efficient differentiation of human ES cells and iPS cells into mature pancreatic insulin-producing cells. Cell Res. 2009, 19, 429–438. [Google Scholar] [CrossRef]
- Jiang, J.; Au, M.; Lu, K.; Eshpeter, A.; Korbutt, G.; Fisk, G.; Majumdar, A.S. Generation of Insulin-Producing Islet-Like Clusters from Human Embryonic Stem Cells. Stem Cells 2007, 25, 1940–1953. [Google Scholar] [CrossRef]
- Wang, W.; Jin, S.; Ye, K. Development of Islet Organoids from H9 Human Embryonic Stem Cells in Biomimetic 3D Scaffolds. Stem Cells Dev. 2017, 26, 394–404. [Google Scholar] [CrossRef] [PubMed]
- Bi, H.; Karanth, S.S.; Ye, K.; Stein, R.; Jin, S. Decellularized Tissue Matrix Enhances Self-Assembly of Islet Organoids from Pluripotent Stem Cell Differentiation. ACS Biomater. Sci. Eng. 2020. [Google Scholar] [CrossRef]
- Bi, H.; Ye, K.; Jin, S. Proteomic analysis of decellularized pancreatic matrix identifies collagen V as a critical regulator for islet organogenesis from human pluripotent stem cells. Biomaterials 2020, 233, 119673. [Google Scholar] [CrossRef]
- Seymour, P.A.; Sander, M. Historical perspective: Beginnings of the β-cell: Current perspectives in β-cell development. Diabetes 2011, 2, 364–376. [Google Scholar] [CrossRef] [PubMed]
- Marroquí, L.; Alonso-Magdalena, P.; Merino, B.; Fuentes, E.; Nadal, A.; Quesada, I. Nutrient regulation of glucagon secretion: involvement in metabolism and diabetes. Nutr. Res. Rev. 2014, 27, 48–62. [Google Scholar] [CrossRef] [PubMed]
- Aragón, F.; Karaca, M.; Novials, A.; Maldonado, R.; Maechler, P.; Rubi, B. Pancreatic polypeptide regulates glucagon release through PPYR1 receptors expressed in mouse and human alpha-cells. Biochim. Biophys. Acta (BBA) Gen. Subj. 2015, 1850, 343–351. [Google Scholar] [CrossRef]
- Hauge-Evans, A.C.; King, A.; Carmignac, D.; Richardson, C.C.; Robinson, I.C.; Low, M.J.; Christie, M.R.; Persaud, S.J.; Jones, P. Somatostatin Secreted by Islet δ-Cells Fulfills Multiple Roles as a Paracrine Regulator of Islet Function. Diabetes 2008, 58, 403–411. [Google Scholar] [CrossRef] [PubMed]
- Siller, R.; Greenhough, S.; Naumovska, E.; Sullivan, G.J. Small-Molecule-Driven Hepatocyte Differentiation of Human Pluripotent Stem Cells. Stem Cell Rep. 2015, 4, 939–952. [Google Scholar] [CrossRef] [PubMed]
- Osafune, K.; Caron, L.; Borowiak, M.; Martinez, R.J.; Fitz-Gerald, C.S.; Sato, Y.; Cowan, C.; Chien, K.R.; Melton, D.A. Marked differences in differentiation propensity among human embryonic stem cell lines. Nat. Biotechnol. 2008, 26, 313–315. [Google Scholar] [CrossRef]
- Takeuchi, H.; Nakatsuji, N.; Suemori, H. Endodermal differentiation of human pluripotent stem cells to insulin-producing cells in 3D culture. Sci. Rep. 2014, 4, 4488. [Google Scholar] [CrossRef]
- Kim, S.K.; Melton, U.A. Pancreas development is promoted by cyclopamine, a Hedgehog signaling inhibitor. Proc. Natl. Acad. Sci. USA 1998, 95, 13036–13041. [Google Scholar] [CrossRef]
- Kaneto, H.; Matsuoka, T.A.; Kawashima, S.K.; Yamamoto, K.; Kato, T.; Miyatsuka, N.; Matsuhisa, M. Role of MafA in pancreatic beta-cells. Adv. Drug. Deliv. Rev. 2009, 61, 489–496. [Google Scholar] [CrossRef]
- Mfopou, J.K.; Chen, B.; Sui, L.; Sermon, K.; Bouwens, L. Recent Advances and Prospects in the Differentiation of Pancreatic Cells From Human Embryonic Stem Cells. Diabetes 2010, 59, 2094–2101. [Google Scholar] [CrossRef]
- Zhang, C.; Moriguchi, T.; Kajihara, M.; Esaki, R.; Harada, A.; Shimohata, H.; Oishi, H.; Hamada, M.; Morito, N.; Hasegawa, K.; et al. MafA Is a Key Regulator of Glucose-Stimulated Insulin Secretion. Mol. Cell. Boil. 2005, 25, 4969–4976. [Google Scholar] [CrossRef]
- El Khattabi, I.; Sharma, A. Proper activation of MafA is required for optimal differentiation and maturation of pancreatic beta-cells. Best Pract. Res. Clin. Endocrinol. Metab. 2015, 6, 821–831. [Google Scholar] [CrossRef]
- Zhu, Y.; Liu, Q.; Zhou, Z.; Ikeda, Y. PDX1, Neurogenin-3, and MAFA: critical transcription regulators for beta cell development and regeneration. Stem Cell Res. Ther. 2017, 8, 240. [Google Scholar] [CrossRef] [PubMed]
- Kataoka, K.; Han, S.I.; Shioda, S.M.; Hirai, M.; Nishizawa, H.; Handa, M. A is a glucose-regulated and pancreatic beta-cell-specific transcriptional activator for the insulin gene. J. Biol. Chem. 2002, 51, 49903–49910. [Google Scholar] [CrossRef] [PubMed]
- Aguayo-Mazzucato, C.; DiIenno, A.; Hollister-Lock, J.; Cahill, C.; Sharma, A.; Weir, G.; Colton, C.; Bonner-Weir, S. MAFA and T3 Drive Maturation of Both Fetal Human Islets and Insulin-Producing Cells Differentiated From hESC. J. Clin. Endocrinol. Metab. 2015, 100, 3651–3659. [Google Scholar] [CrossRef] [PubMed]
- Schaffer, A.; Taylor, B.L.; Benthuysen, J.; Liu, J.; Thorel, F.; Yuan, W.; Jiao, Y.; Kaestner, K.H.; Herrera, P.L.; Magnuson, M.A.; et al. Nkx6.1 Controls a Gene Regulatory Network Required for Establishing and Maintaining Pancreatic Beta Cell Identity. PLoS Genet. 2013, 9, e1003274. [Google Scholar] [CrossRef] [PubMed]
- Taylor, B.L.; Liu, F.-F.; Sander, M. Nkx6.1 is essential for maintaining the functional state of pancreatic beta cells. Cell Rep. 2013, 4, 1262–1275. [Google Scholar] [CrossRef] [PubMed]
- Schisler, J.C.; Jensen, P.B.; Taylor, D.G.; Becker, T.C.; Knop, F.K.; Takekawa, S.; German, M.; Weir, G.C.; Lu, D.; Mirmira, R.G.; et al. The Nkx6.1 homeodomain transcription factor suppresses glucagon expression and regulates glucose-stimulated insulin secretion in islet beta cells. Proc. Natl. Acad. Sci. USA 2005, 102, 7297–7302. [Google Scholar] [CrossRef]
- Memon, B.; Karam, M.; Al-Khawaga, S.; Abdelalim, E.M. Enhanced differentiation of human pluripotent stem cells into pancreatic progenitors co-expressing PDX1 and NKX6. 1. Stem Cell Res. Ther. 2018, 9, 15. [Google Scholar] [CrossRef]
- Tran, R.; Moraes, C.; Hoesli, C.A. Controlled clustering enhances PDX1 and NKX6.1 expression in pancreatic endoderm cells derived from pluripotent stem cells. Sci. Rep. 2020, 10, 1–12. [Google Scholar] [CrossRef]
- Spaeth, J.M.; Gupte, M.; Perelis, M.; Yang, Y.P.; Cyphert, H.; Guo, S.; Liu, J.H.; Guo, M.; Bass, J.; Magnuson, M.A.; et al. Stein, Defining a Novel Role for the Pdx1 Transcription Factor in Islet beta-Cell Maturation and Proliferation During Weaning. Diabetes 2017, 66, 2830–2839. [Google Scholar] [CrossRef]
- Kumar, M.; Jordan, N.; Melton, D.A.; Grapin-Botton, A. Signals from lateral plate mesoderm instruct endoderm toward a pancreatic fate. Dev. Boil. 2003, 259, 109–122. [Google Scholar] [CrossRef]
- Vanhoose, A.M.; Samaras, S.; Artner, I.; Henderson, E.; Hang, Y.; Stein, R. MafA and MafB RegulatePdx1Transcription through the Area II Control Region in Pancreatic β Cells. J. Boil. Chem. 2008, 283, 22612–22619. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Pan, F.C.; Brandes, N.; Afelik, S.; Sölter, M.; Pieler, T. Retinoic acid signaling is essential for pancreas development and promotes endocrine at the expense of exocrine cell differentiation in Xenopus. Dev. Boil. 2004, 271, 144–160. [Google Scholar] [CrossRef] [PubMed]
- Johannesson, M.; Ståhlberg, A.; Ameri, J.; Sand, F.W.; Norrman, K.; Semb, H. FGF4 and Retinoic Acid Direct Differentiation of hESCs into PDX1-Expressing Foregut Endoderm in a Time- and Concentration-Dependent Manner. PLoS ONE 2009, 4, e4794. [Google Scholar] [CrossRef] [PubMed]
- Kuo, Y.C.; Liu, Y.C.; Rajesh, R. Pancreatic differentiation of induced pluripotent stem cells in activin A-grafted gelatin-poly(lactide-co-glycolide) nanoparticle scaffolds with induction of LY294002 and retinoic acid. Mater. Sci. Eng. C 2017, 77, 384–393. [Google Scholar] [CrossRef]
- Idris, I.; Patiag, D.; Gray, S.; Donnelly, R. Exendin-4 increases insulin sensitivity via a PI-3-kinase-dependent mechanism: Contrasting effects of GLP-1. Biochem. Pharmacol. 2002, 63, 993–996. [Google Scholar] [CrossRef]
- Arakawa, M.; Ebato, C.; Mita, T.; Hirose, T.; Kawamori, R.; Fujitani, Y.; Watada, H. Effects of exendin-4 on glucose tolerance, insulin secretion, and beta-cell proliferation depend on treatment dose, treatment duration and meal contents. Biochem. Biophys. Res. Commun. 2009, 390, 809–814. [Google Scholar] [CrossRef]
- Alarçon, C.; Wicksteed, B.; Rhodes, C.J. Exendin 4 controls insulin production in rat islet beta cells predominantly by potentiation of glucose-stimulated proinsulin biosynthesis at the translational level. Diabetologia 2006, 49, 2920–2929. [Google Scholar] [CrossRef]
- Movassat, J.; Beattie, G.M.; Lopez, A.D.; Hayek, A. Exendin 4 Up-Regulates Expression of PDX 1 and Hastens Differentiation and Maturation of Human Fetal Pancreatic Cells. J. Clin. Endocrinol. Metab. 2002, 87, 4775–4781. [Google Scholar] [CrossRef]
- Massumi, M.; Pourasgari, F.; Nalla, A.; Batchuluun, B.; Nagy, K.; Neely, E.; Gull, R.; Nagy, A.; Wheeler, M.B. An Abbreviated Protocol for In Vitro Generation of Functional Human Embryonic Stem Cell-Derived Beta-Like Cells. PLoS ONE 2016, 11, e0164457. [Google Scholar] [CrossRef]
- Russ, A.H.; Parent, A.V.; Ringler, J.J.; Hennings, T.G.; Nair, G.G.; Shveygert, M.; Guo, T.; Puri, S.; Haataja, L.; Cirulli, V.; et al. Controlled induction of human pancreatic progenitors produces functional beta-like cells in vitro. EMBO J. 2015, 34, 1759–1772. [Google Scholar] [CrossRef]
- Vethe, H.; Ghila, L.; Berle, M.; Hoareau, L.; Haaland, Ø.A.; Scholz, H.; Paulo, J.A.; Chera, S.; Ræder, H. The Effect of Wnt Pathway Modulators on Human iPSC-Derived Pancreatic Beta Cell Maturation. Front. Endocrinol. 2019, 10, 293. [Google Scholar] [CrossRef] [PubMed]
- Civelek, V.N.; Deeney, J.T.; Kubik, K.; Schultz, V.; Tornheim, K.; Corkey, B.E. Temporal sequence of metabolic and ionic events in glucose-stimulated clonal pancreatic β-cells (HIT). Biochem. J. 1996, 315, 1015–1019. [Google Scholar] [CrossRef] [PubMed]
- Ashcroft, F.; Rorsman, P. Diabetes mellitus and the β cell: the last ten years. Cell 2012, 148, 1160–1171. [Google Scholar] [CrossRef] [PubMed]
- Trexler, A.J.; Taraska, J.W. Regulation of insulin exocytosis by calcium-dependent protein kinase C in beta cells. Cell Calcium 2017, 67, 1–10. [Google Scholar] [CrossRef]
- Hivelin, C.; Béraud-Dufour, S.; DeVader, C.; Abderrahmani, A.; Moreno, S.; Maati, H.M.O.; Djillani, A.; Heurteaux, C.; Borsotto, M.; Mazella, J.; et al. Potentiation of Calcium Influx and Insulin Secretion in Pancreatic Beta Cell by the Specific TREK-1 Blocker Spadin. J. Diabetes Res. 2016, 2016, 1–9. [Google Scholar] [CrossRef]
- Kenty, J.H.R.; Melton, D.A. Testing Pancreatic Islet Function at the Single Cell Level by Calcium Influx with Associated Marker Expression. PLOS ONE 2015, 10, e0122044. [Google Scholar] [CrossRef]
- Bader, E.; Migliorini, A.; Gegg, M.; Moruzzi, N.; Gerdes, J.; Roscioni, S.S.; Bakhti, M.; Brandl, E.; Irmler, M.; Beckers, J.; et al. Identification of proliferative and mature beta-cells in the islets of Langerhans. Nature 2016, 535, 430–434. [Google Scholar] [CrossRef]
- Uzan, B.; Figeac, F.; Portha, B.; Movassat, J. Mechanisms of KGF Mediated Signaling in Pancreatic Duct Cell Proliferation and Differentiation. PLOS ONE 2009, 4, e4734. [Google Scholar] [CrossRef]
- Movassat, J.; Beattie, G.M.; Lopez, A.D.; Portha, B.; Hayek, A. Keratinocyte growth factor and beta-cell differentiation in human fetal pancreatic endocrine precursor cells. Diabetologia 2003, 46, 822–829. [Google Scholar] [CrossRef]
- Wagner, M.; Koschnick, S.; Beilke, S.; Frey, M.; Adler, G.; Schmid, R.M. Selective expansion of the beta-cell compartment in the pancreas of keratinocyte growth factor transgenic mice. Am. J. Physiol. Gastrointest. Liver Physiol. 2008, 294, G1139–G1147. [Google Scholar] [CrossRef]
- Toyoda, T.; Mae, S.-I.; Tanaka, H.; Kondo, Y.; Funato, M.; Hosokawa, Y.; Sudo, T.; Kawaguchi, Y.; Osafune, K. Cell aggregation optimizes the differentiation of human ESCs and iPSCs into pancreatic bud-like progenitor cells. Stem Cell Res. 2015, 14, 185–197. [Google Scholar] [CrossRef]
- Schulz, T.C.; Young, H.Y.; Agulnick, A.D.; Babin, M.J.; Baetge, E.E.; Bang, A.G.; Bhoumik, A.; Cepa, I.; Cesario, R.M.; Haakmeester, C.; et al. A Scalable System for Production of Functional Pancreatic Progenitors from Human Embryonic Stem Cells. PLoS ONE 2012, 7, e37004. [Google Scholar] [CrossRef] [PubMed]
- Ostrom, M.; Loffler, K.A.; Edfalk, S.; Selander, L.; Dahl, U.; Ricordi, C.; Jeon, J.; Correa-Medina, J.; Diez, H. Edlund, Retinoic acid promotes the generation of pancreatic endocrine progenitor cells and their further differentiation into beta-cells. PLoS ONE 2008, 3, e2841. [Google Scholar] [CrossRef] [PubMed]
- Abazari, M.F.; Soleimanifar, F.; Aleagha, M.N.; Torabinejad, S.; Nasiri, N.; Khamisipour, G.; Mahabadi, J.A.; Mahboudi, H.; Enderami, S.E.; Saburi, E.; et al. PCL/PVA nanofibrous scaffold improve insulin-producing cells generation from human induced pluripotent stem cells. Gene 2018, 671, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Shahjalal, H.M.; Dayem, A.A.; Lim, K.M.; Jeon, T.I.; Cho, S.G. Generation of pancreatic β cells for treatment of diabetes: advances and challenges. Stem Cell Res. Ther. 2018, 9, 355. [Google Scholar] [CrossRef] [PubMed]
- Tao, T.; Wang, Y.; Chen, W.; Li, Z.; Su, W.; Guo, Y.; Deng, P.; Qin, J. Engineering human islet organoids from iPSCs using an organ-on-chip platform. Lab Chip 2019, 19, 948–958. [Google Scholar] [CrossRef]
- Brissova, M.; Fowler, M.J.; Nicholson, W.E.; Chu, A.; Hirshberg, B.; Harlan, D.M.; Powers, A.C. Assessment of Human Pancreatic Islet Architecture and Composition by Laser Scanning Confocal Microscopy. J. Histochem. Cytochem. 2005, 53, 1087–1097. [Google Scholar] [CrossRef]
- Wendt, A.; Eliasson, L. Pancreatic α-cells – The unsung heroes in islet function. Semin. Cell Dev. Boil. 2020, 103, 41–50. [Google Scholar] [CrossRef]
- Pipeleers, D.; Veld, P.I.I.; Maes, E.; Van De Winkel, M. Glucose-induced insulin release depends on functional cooperation between islet cells. Proc. Natl. Acad. Sci. USA 1982, 79, 7322–7325. [Google Scholar] [CrossRef]
- Rodriguez-Diaz, R.; Tamayo, A.; Hara, M.; Caicedo, A. The Local Paracrine Actions of the Pancreatic α-Cell. Diabetes 2019, 69, 550–558. [Google Scholar] [CrossRef]
- Brissova, M.; Haliyur, R.; Saunders, D.; Shrestha, S.; Dai, C.; Blodgett, D.M.; Bottino, R.; Campbell-Thompson, M.; Aramandla, R.; Poffenberger, G.; et al. α Cell Function and Gene Expression Are Compromised in Type 1 Diabetes. Cell Rep. 2018, 22, 2667–2676. [Google Scholar] [CrossRef] [PubMed]
- Rezania, A.; Bruin, J.E.; Riedel, M.J.; Mojibian, M.; Asadi, A.; Xu, J.; Gauvin, R.; Narayan, K.; Karanu, F.; O’Neil, J.J.; et al. Maturation of Human Embryonic Stem Cell–Derived Pancreatic Progenitors Into Functional Islets Capable of Treating Pre-existing Diabetes in Mice. Diabetes 2012, 61, 2016–2029. [Google Scholar] [CrossRef] [PubMed]
- Peterson, Q.P.; Veres, A.; Chen, L.; Slama, M.Q.; Kenty, J.H.R.; Hassoun, S.; Brown, M.R.; Dou, H.; Duffy, C.D.; Zhou, Q.; et al. A method for the generation of human stem cell-derived alpha cells. Nat. Commun. 2020, 11, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Banerjee, A.; Herring, C.A.; Attalla, J.; Hu, R.; Xu, Y.; Shao, Q.; Simmons, A.J.; Dadi, P.K.; Wang, S.; et al. Neurog3-Independent Methylation Is the Earliest Detectable Mark Distinguishing Pancreatic Progenitor Identity. Dev. Cell 2019, 48, 49–63.e7. [Google Scholar] [CrossRef]
- Chakravarthy, H.; Gu, X.; Enge, M.; Dai, X.; Wang, Y.; Damond, N.; Downie, C.; Liu, K.; Wang, J.; Xing, Y.; et al. Converting Adult Pancreatic Islet alpha Cells into beta Cells by Targeting Both Dnmt1 and Arx. Cell Metab. 2017, 25, 622–634. [Google Scholar] [CrossRef]
- Courtney, M.; Pfeifer, A.; Gjernes, E.; Vieira, A.; Collombat, P.; Al-Hasani, K.; Ben-Othman, N. In vivo conversion of adult α-cells into β-like cells: a new research avenue in the context of type 1 diabetes. Diabetes Obes. Metab. 2011, 13, 47–52. [Google Scholar] [CrossRef]
- Collombat, P.; Mansouri, A.; Hecksher-Sørensen, J.; Serup, P.; Krull, J.; Gradwohl, G.; Gruss, P. Opposing actions of Arx and Pax4 in endocrine pancreas development. Genes Dev. 2003, 17, 2591–2603. [Google Scholar] [CrossRef]
- Rezania, A.; Riedel, M.J.; Wideman, R.D.; Karanu, F.; Ao, Z.; Warnock, G.L.; Kieffer, T.J. Production of Functional Glucagon-Secreting α-Cells From Human Embryonic Stem Cells. Diabetes 2010, 60, 239–247. [Google Scholar] [CrossRef]
- Baskin, D.G.; Gorray, K.C.; Fujimoto, W.Y. Immunocytochemical identification of cells containing insulin, glucagon, somatostatin, and pancreatic polypetide in the islets of langerhans of the guinea pig pancreas with light and electron microscopy. Anat. Rec. Adv. Integr. Anat. Evol. Boil. 1984, 208, 567–578. [Google Scholar] [CrossRef]
- Grube, D.; Bohn, R. The Microanatomy of Human Islets of Langerhans, with Special Reference to Somatostatin (D-) Cells. Arch. Histol. Cytol. 1983, 46, 327–353. [Google Scholar] [CrossRef]
- Brereton, M.F.; Vergari, E.; Zhang, Q.; Clark, A. Alpha-, Delta- and PP-cells. J. Histochem. Cytochem. 2015, 63, 575–591. [Google Scholar] [CrossRef] [PubMed]
- Yue, J.T.; Riddell, M.C.; Burdett, E.; Coy, D.H.; Efendic, S.; Vranic, M. Amelioration of Hypoglycemia Via Somatostatin Receptor Type 2 Antagonism in Recurrently Hypoglycemic Diabetic Rats. Diabetes 2013, 62, 2215–2222. [Google Scholar] [CrossRef] [PubMed]
- Karimian, N. Somatostatin Receptor Type 2 Antagonism Improves Glucagon Counter-regulation Biobreeding Diabetes-Prone Rats. Diabetes 2013, 62, 2968–2977. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Yu, Q.; Ahooghalandari, P.; Gribble, F.M.; Reimann, F.; Tengholm, A.; Gylfe, E. Submembrane ATP and Ca2+ kinetics in α-cells: unexpected signaling for glucagon secretion. FASEB J. 2015, 29, 3379–3388. [Google Scholar] [CrossRef] [PubMed]
- Gerich, J.E.; Lorenzi, M.; Bier, D.M.; Schneider, V.; Tsalikian, E.; Karam, J.H.; Forsham, P.H. Prevention of Human Diabetic Ketoacidosis by Somatostatin. N. Engl. J. Med. 1975, 292, 985–989. [Google Scholar] [CrossRef] [PubMed]
- Grieco, F.A.; Moretti, M.; Sebastiani, G.; Galleri, L.; Spagnuolo, I.; Scafetta, G.; Gulino, A.; De Smaele, E.; Maroder, M.; Dotta, F. Delta-cell-specific expression of hedgehog pathway Ptch1 receptor in murine and human endocrine pancreas. Diabetes/Metabolism Res. Rev. 2011, 27, 755–760. [Google Scholar] [CrossRef]
- Jonsson, J.; Carlsson, L.; Edlund, T.; Edlund, H. Insulin-promoter-factor 1 is required for pancreas development in mice. Nature 1994, 371, 606–609. [Google Scholar] [CrossRef]
- Peshavaria, M.; Gamer, L.; Henderson, E.; Teitelman, G.; Wright, C.; Stein, R. XIHbox 8, an endoderm-specific Xenopus homeodomain protein, is closely related to a mammalian insulin gene transcription factor. Mol. Endocrinol. 1994, 8, 806–816. [Google Scholar]
- Itkin-Ansari, P.; Demeterco, C.; Bossie, S.; Dufayet de la Tour, D.; Beattie, G.M.; Movassat, J.; Mally, M.I.; Hayek, A.; Levine, F. PDX-1 and cell-cell contact act in synergy to promote δ-cell development in a human pancreatic endocrine precursor cell line. Mol. Endocrinol. 2000, 14, 814–822. [Google Scholar]
- Orci, L.; Malaisse-Lagae, F.; Baetens, D.; Perrelet, A. Pancreatic-Polypeptide-Rich Regions In Human Pancreas. Lancet 1978, 312, 1200–1201. [Google Scholar] [CrossRef]
- Larhammar, D. Structural diversity of receptors for neuropeptide Y, peptide YY and pancreatic polypeptide. Regul. Pept. 1996, 65, 165–174. [Google Scholar] [CrossRef]
- Rabiee, A.; Galiatsatos, P.; Salas-Carrillo, R.; Thompson, M.J.; Andersen, D.K.; Elahi, D. Pancreatic Polypeptide Administration Enhances Insulin Sensitivity and Reduces the Insulin Requirement of Patients on Insulin Pump Therapy. J. Diabetes Sci. Technol. 2011, 5, 1521–1528. [Google Scholar] [CrossRef] [PubMed]
- Gettys, T.W.; Garcia, R.; Savage, K.; Whitcomb, D.C.; Kanayama, S.; Taylor, I.L. Insulin-Sparing Effects of Pancreatic Polypeptide in Congenitally Obese Rodents. Pancreas 1991, 6, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Mashima, H.; Ohnishi, H.; Wakabayashi, K.; Mine, T.; Miyagawa, J.; Hanafusa, T.; Seno, M.; Yamada, H.; Kojima, I. Betacellulin and activin A coordinately convert amylase-secreting pancreatic AR42J cells into insulin-secreting cells. J. Clin. Investig. 1996, 97, 1647–1654. [Google Scholar] [CrossRef]
- D’Amour, K.A.; Bang, A.G.; Eliazer, S.; Kelly, O.G.; Agulnick, A.D.; Smart, N.G.; Baetge, E.E. Production of pancreatic hormone–expressing endocrine cells from human embryonic stem cells. Nat. Biotechnol. 2006, 24, 1392–1401. [Google Scholar] [CrossRef]
- Kroon, E.; Martinson, L.; Kadoya, K.; Bang, A.G.; Kelly, O.G.; Eliazer, S.; Young, H.; Richardson, M.; Smart, N.G.; Cunningham, J.; et al. Pancreatic endoderm derived from human embryonic stem cells generates glucose-responsive insulin-secreting cells in vivo. Nat. Biotechnol. 2008, 26, 443–452. [Google Scholar] [CrossRef]
- Rickels, M.R. Stem Cell-Derived Islets: Next Steps for Histologic and Functional Assessment During Development as a Cellular Therapy for the Treatment of Diabetes. Diabetes 2019, 68, 901–903. [Google Scholar] [CrossRef]
- Rorsman, P.; Braun, M. Regulation of Insulin Secretion in Human Pancreatic Islets. Annu. Rev. Physiol. 2013, 75, 155–179. [Google Scholar] [CrossRef]
- Benninger, R.K.P.; Piston, D.W. Cellular communication and heterogeneity in pancreatic islet insulin secretion dynamics. Trends Endocrinol. Metab. 2014, 25, 399–406. [Google Scholar] [CrossRef]
- Kim, Y.; Kim, H.; Ko, U.H.; Oh, Y.; Lim, A.; Sohn, J.-W.; Shin, J.H.; Kim, H.; Han, Y.-M. Islet-like organoids derived from human pluripotent stem cells efficiently function in the glucose responsiveness in vitro and in vivo. Sci. Rep. 2016, 6, 35145. [Google Scholar] [CrossRef]
- Hirano, K.; Konagaya, S.; Turner, A.; Noda, Y.; Kitamura, S.; Kotera, H.; Iwata, H. Closed-channel culture system for efficient and reproducible differentiation of human pluripotent stem cells into islet cells. Biochem. Biophys. Res. Commun. 2017, 487, 344–350. [Google Scholar] [CrossRef] [PubMed]
- Ida, H.; Akiyama, T.; Ishiguro, K.I.; Goparaju, S.K.; Nakatake, Y.; Chikazawa-Nohtomi, N.; Sato, S.; Kimura, H.; Yokoyama, Y.; Nagino, M.; et al. Establishment of a rapid and footprint-free protocol for differentiation of human embryonic stem cells into pancreatic endocrine cells with synthetic mRNAs encoding transcription factors. Stem Cell Res. Ther. 2018, 9, 277. [Google Scholar] [CrossRef]
- Mao, G.H.; Lu, P.; Wang, Y.N.; Tian, C.G.; Huang, X.H.; Feng, Z.G.; Zhang, J.L.; Chang, H.Y. Role of PI3K p110beta in the differentiation of human embryonic stem cells into islet-like cells. Biochem. Biophys. Res. Commun. 2017, 1, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.Y.; Tsai, C.C.; Chen, L.L.; Chiou, S.H.; Wang, Y.J.; Hung, S.C. Fibronectin and laminin promote differentiation of human mesenchymal stem cells into insulin producing cells through activating Akt and ERK. J. Biomed. Sci. 2010, 17, 56. [Google Scholar] [CrossRef] [PubMed]
- Brafman, D.A.; Phung, C.; Kumar, N.; Willert, K. Regulation of endodermal differentiation of human embryonic stem cells through integrin-ECM interactions. Cell Death Differ. 2012, 20, 369–381. [Google Scholar] [CrossRef]
- Wang, X.; Ye, K. Three-Dimensional Differentiation of Embryonic Stem Cells into Islet-like Insulin-Producing Clusters. Tissue Eng. Part. A 2009, 15, 1941–1952. [Google Scholar] [CrossRef]
- Pokrywczyńska, M.; Lewandowska, M.A.; Krzyzanowska, S.; Jundziłł, A.; Rasmus, M.; Warda, K.; Gagat, M.; Deptula, A.; Helmin-Basa, A.; Hołysz, M.; et al. Transdifferentiation of Bone Marrow Mesenchymal Stem Cells into the Islet-Like Cells: The Role of Extracellular Matrix Proteins. Arch. Immunol. Ther. Exp. 2015, 63, 377–384. [Google Scholar] [CrossRef]
- Chao, K.C.; Chao, K.F.; Fu, Y.S.; Liu, S.-H. Islet-Like Clusters Derived from Mesenchymal Stem Cells in Wharton’s Jelly of the Human Umbilical Cord for Transplantation to Control Type 1 Diabetes. PLoS ONE 2008, 3, e1451. [Google Scholar] [CrossRef]
- Zhang, Y.; Shen, W.; Hua, J.; Lei, A.; Lv, C.; Wang, H.; Yang, C.; Gao, Z.; Dou, Z. Pancreatic Islet-Like Clusters from Bone Marrow Mesenchymal Stem Cells of Human First-Trimester Abortus Can Cure Streptozocin-Induced Mouse Diabetes. Rejuvenation Res. 2010, 13, 695–706. [Google Scholar] [CrossRef]
- Nair, G.G.; Liu, J.S.; Russ, H.A.; Tran, S.; Saxton, M.S.; Chen, R.; Juang, C.; Li, M.L.; Nguyen, V.Q.; Giacometti, S.; et al. Recapitulating endocrine cell clustering in culture promotes maturation of human stem-cell-derived beta cells. Nat. Cell. Biol. 2019, 21, 263–274. [Google Scholar]
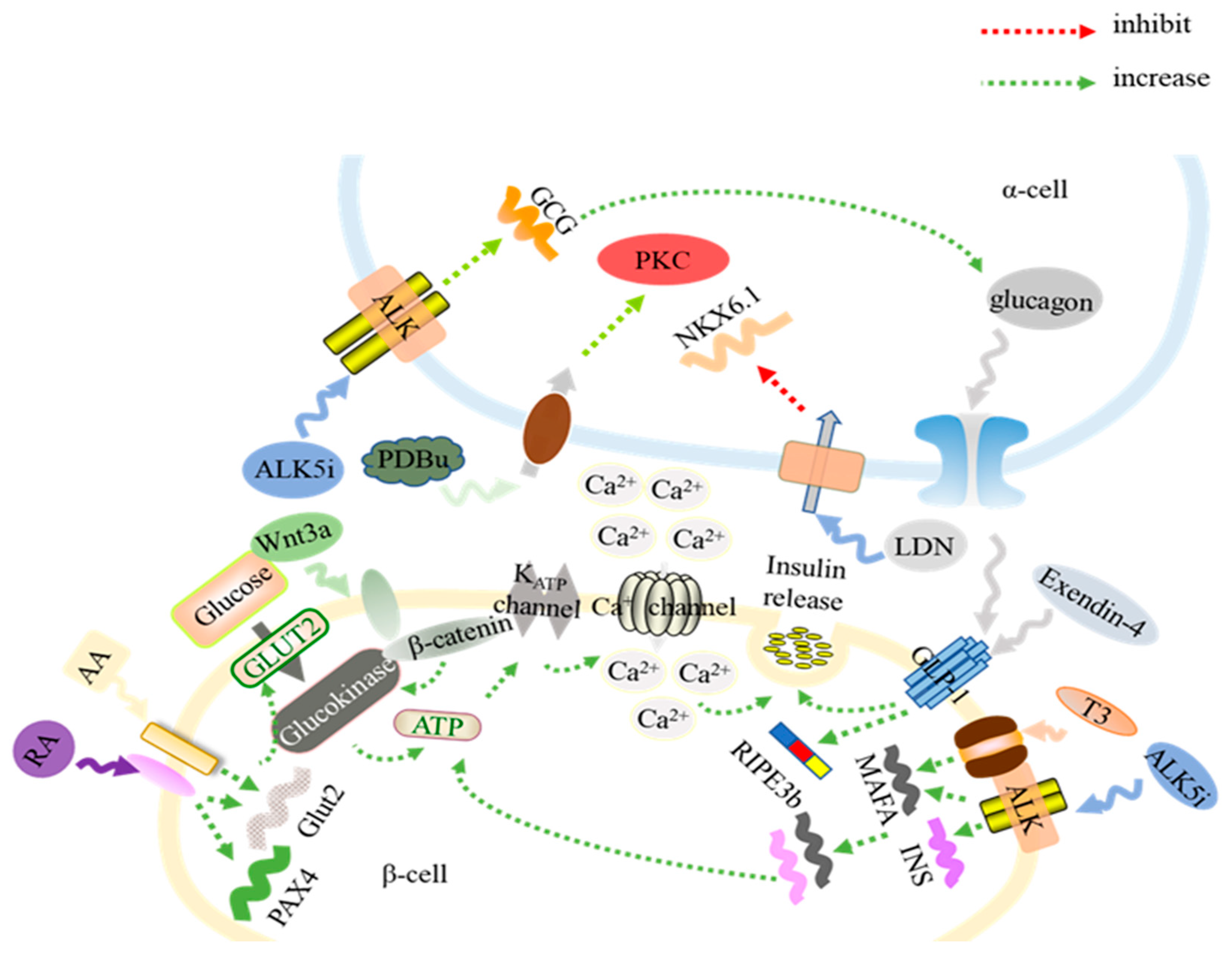
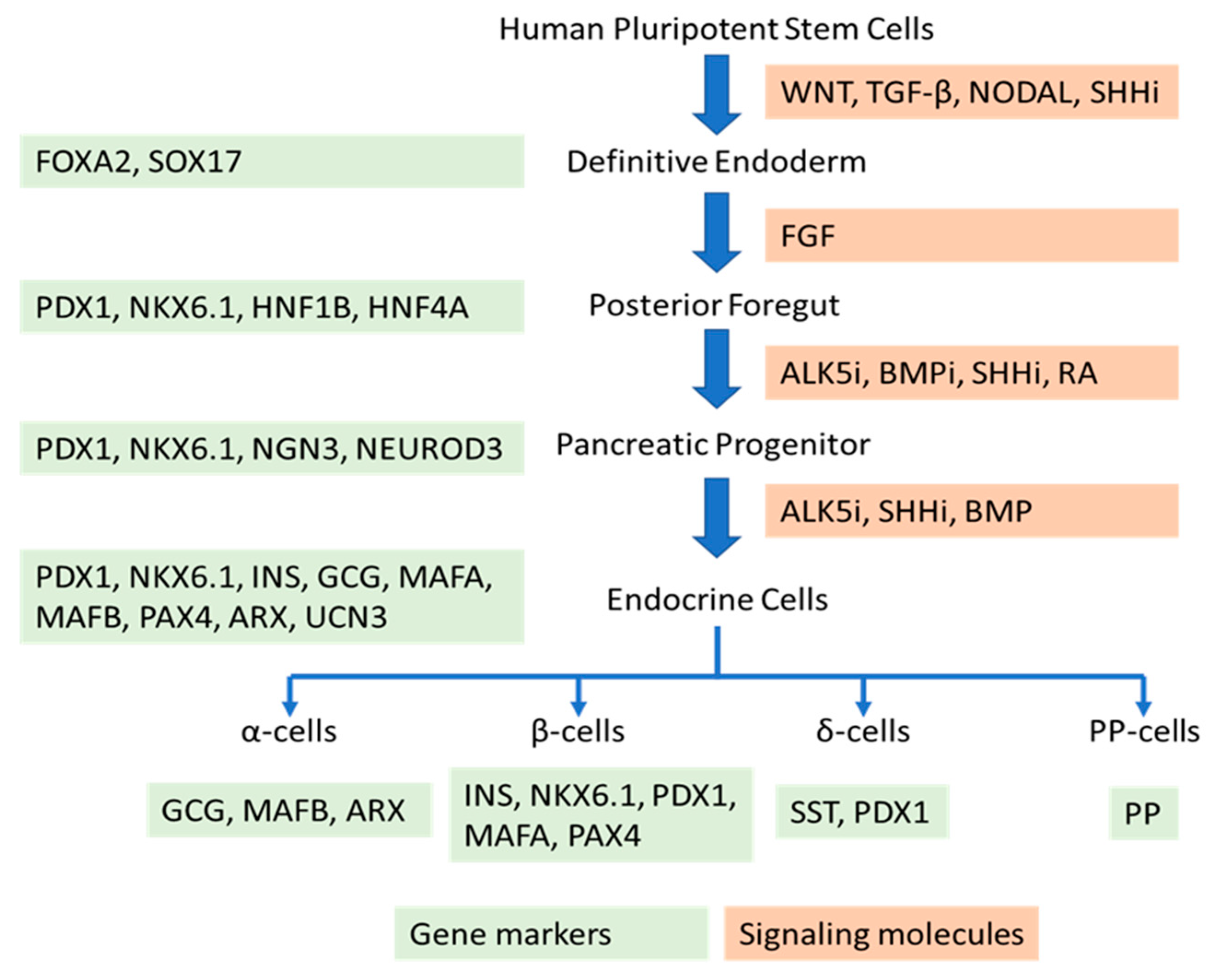
| Molecule | Function | Period | Cell Line | Timeline | Reference |
|---|---|---|---|---|---|
| T3 | Increase MAFA expression | Stage 5 | CyT49 | Five stages (21+ days) | [23] |
| ALK5 inhibitor | Increase MAFA expression | Stage 5~7 | H1 | Seven stages (~40 days) | [2] |
| Exendin-4 | Improve β-cell maturation | Stage 4 | H9, H1 | Four stages (22 days) | [4] |
| Stage 5 | H1, Epi-9, iPS1-10 | Five stages (30 days) | [39] | ||
| RA | Increase PDX1, PAX4, and GLUT2 expression | Stage 3 | MEL1/INSGFP/W | Five stages (20 days) | [40] |
| Stage 3~5 | HUES8, iPSC-1, iPSC-2 | Five stages (~35 days) | [3] | ||
| Tankyrase inhibitor G007-LK | Inhibit Wnt signaling | Stage 7 | ND41866 | Seven stages (~40 days) | [41] |
| Molecules | Function | Period | Cell Line | Timeline | Reference |
|---|---|---|---|---|---|
| LDN | Suppress NKX6.1 expression | Stage 3 | HUES8, 1016 | Five stages (48 days) | [63] |
| PDBu | Activator of PKC | Stage 5 | HUES8, 1016 | Five stages (48 days) | [63] |
| Noggin | Inhibit BMP signaling | Stage 2~3 | H1 | Six stages (26 days) | [68] |
| Reference | Stages (Days) | Strategy | Cell Line | GSIS (Fold Change) | GSGS (Fold Change) | δ Cells | PP Cells | In Vivo Study |
|---|---|---|---|---|---|---|---|---|
| Jiang et al., 2007 [5] | Four-stage (36) | 2D | H1, H7, H9 | 3.3 | N/A | N/A | N/A | N/A |
| Bi et al., 2020 [7] | Five-stage (28) | 2D for 18 days; 3D suspension for 10 days | IMR90, H9 | ~2.8 | 2 | Detected | Detected | N/A |
| Bi et al., 2020 [8] | Five-stage (28) | 2D for 18 days; 3D suspension for 10 days | IMR90 | ~2.3 | ~4 | Detected | Detected | N/A |
| Peterson et al., 2020 [63] | Five-stage (48) | Suspension culture | HUES8, 1016 cell | ~2 | ~2 | N/A | N/A | N/A |
| Massumi et al., 2016 [39] | Five-stage (30) | 2D | H1, Epi-9, iPS1-10 | ~5.2 | N/A | Detected | N/A | N/A |
| Wang et al., 2017 [6] | Four-stage (23) | 3D collagen scaffold | H9 | ~3.5 | N/A | Detected | Detected | N/A |
| Kim et al., 2016 [90] | Four-stage (17) | Suspension culture on the last day of differentiation | H1, CHA15 | 2.5 | N/A | N/A | Not detected | Function in diabetic mice for 12 days |
| Hirano et al., 2017 [91] | Four-stage (31) | Suspension culture with closed-channel device | 253G1 | ~4 | N/A | Detected | N/A | N/A |
| Rezania et al., 2011 [68] | Seven-stage (26) | 2D | H1 | N/A | ~2 | Detected | N/A | Function in diabetic mice for over 90 days |
| Rezania et al., 2014 [2] | Seven-stage (30~40) | 2D at Stage 1~4; 3D suspension at Stage 5~7 | H1, homemade iPSCs | 1.7 | N/A | Detected | Not detected | Function in diabetic mice for 40 days |
| Pagliuca et al., 2014 [3] | Six-stage (30~37) | Suspension culture in spinner flasks | HUES8, homemade iPSCs | ~3.0 | N/A | N/A | N/A | Function in diabetic mice within 2 weeks |
| Mao et al., 2017 [93] | Five-stage (35) | 2D | PKU1.1 | N/A | N/A | Detected | N/A | Function in diabetic mice |
| Lin et al., 2010 [94] | Four-stage (10) | Suspension culture | Homemade hMSCs | ~2 | N/A | N/A | N/A | N/A |
| Chao et al., 2008 [98] | Four-stage (34) | 2D | Homemade hMSCs | ~5 | N/A | N/A | N/A | Function in diabetic rats for over 21 weeks |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, H.; Bader, T.N.; Jin, S. Signaling Molecules Regulating Pancreatic Endocrine Development from Pluripotent Stem Cell Differentiation. Int. J. Mol. Sci. 2020, 21, 5867. https://doi.org/10.3390/ijms21165867
Huang H, Bader TN, Jin S. Signaling Molecules Regulating Pancreatic Endocrine Development from Pluripotent Stem Cell Differentiation. International Journal of Molecular Sciences. 2020; 21(16):5867. https://doi.org/10.3390/ijms21165867
Chicago/Turabian StyleHuang, Hui, Taylor N. Bader, and Sha Jin. 2020. "Signaling Molecules Regulating Pancreatic Endocrine Development from Pluripotent Stem Cell Differentiation" International Journal of Molecular Sciences 21, no. 16: 5867. https://doi.org/10.3390/ijms21165867
APA StyleHuang, H., Bader, T. N., & Jin, S. (2020). Signaling Molecules Regulating Pancreatic Endocrine Development from Pluripotent Stem Cell Differentiation. International Journal of Molecular Sciences, 21(16), 5867. https://doi.org/10.3390/ijms21165867





