Clinical Significance of Expression Changes and Promoter Methylation of PLA2R1 in Tissues of Breast Cancer Patients
Abstract
1. Introduction
2. Results
2.1. Downregulation of PLA2R1 mRNA Expression Is Associated with High Histological-Grade Breast Cancer
2.2. Lower Levels of PLA2R1 mRNA Expression Were Significantly Associated with Triple Negative Breast Cancers (TNBC)
2.3. PLA2R1 Promoter Hypermethylation Was Associated with Aggressive Subtypes of Breast Cancer
2.4. PLA2R1 Promoter Methylation Outperformed PLA2R1 Expression as a Diagnostic and Prognostic Marker of Breast Cancer
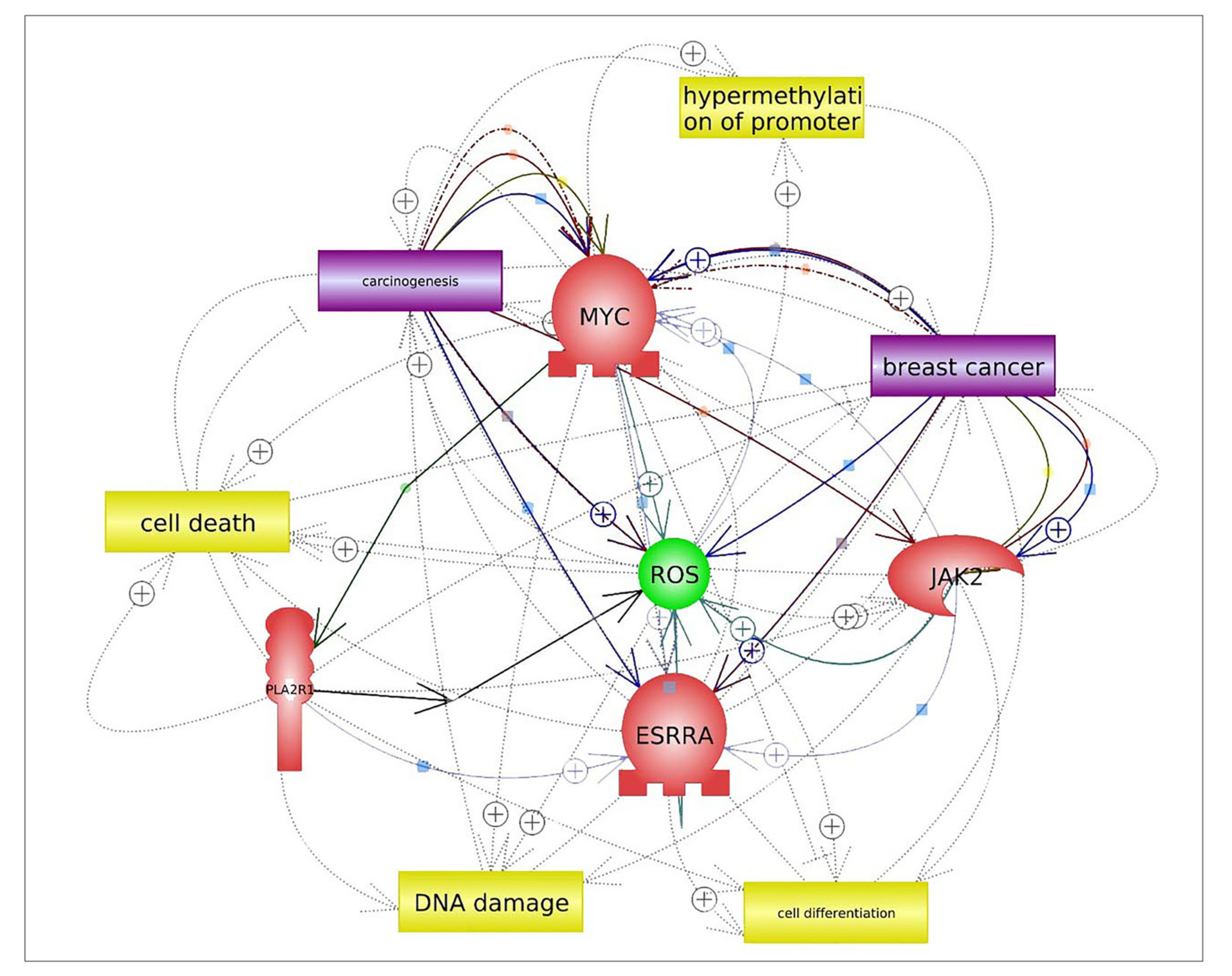
3. Discussion
4. Materials and Methods
4.1. Bioinformatics Analysis
4.2. Patients and Tissue Samples
4.3. Histological and Immunohistochemical Investigation
4.4. Molecular Biology Examinations
4.4.1. RNA Extraction and Reverse Transcription
4.4.2. Quantitative Real-Time PCR (qRT-PCR)
4.4.3. Genomic DNA Extraction and DNA Methylation
4.5. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AUC | Area under the curve |
| ASCO/CAP | American Society of Clinical Oncology/ College of American Pathologists |
| CCLE | Cancer cell line encyclopedia |
| cDNA | Complementary DNA |
| c-MYC | Cellular myelocytomatosis |
| Ct | Cycle threshold |
| ER | Estrogen receptor |
| ESRRA | Estrogen-related receptor alpha1 |
| FFPE | Formalin-fixed paraffin-embedded |
| GAPDH | Glyceraldehyde 3-phosphate dehydrogenase |
| HER2 | Human epidermal growth factor receptor 2 |
| JAK2 | Janus kinase 2 |
| PLA2R1 | Phospholipase A2 receptor 1 |
| PR | Progesterone receptor |
| qRT-PCR | Quantitative Real-time PCR |
| ROC | Receiver operating characteristic |
| RT | Reverse transcription |
| SBR-EE | Scarff-Bloom-Richardson-Elston-Ellis grading system |
| SD | Standard deviation |
| sPLA2 | Secretory phospholipase A2 |
| TNBC | Triple negative breast cancer |
References
- Liu, W.; Gao, C.; Dai, H.; Zheng, Y.; Dong, Z.; Gao, Y.; Liu, F.; Zhang, Z.; Liu, Z.; Liu, J.W.; et al. Immunological pathogenesis of membranous nephropathy: Focus on PLA2R1 and Its role. Front. Immunol. 2019, 10, 1809. [Google Scholar] [CrossRef] [PubMed]
- Sukocheva, O.; Menschikowski, M.; Hagelgans, A.; Yarla, N.S.; Siegert, G.; Reddanna, P.; Bishayee, A. Current insights into functions of phospholipase A2 receptor in normal and cancer cells: More questions than answers. Semin. Cancer Biol. 2019, 56, 116–127. [Google Scholar] [CrossRef] [PubMed]
- Bernard, D.; Vindrieux, D. PLA2R1: Expression and function in cancer. Biochim. Biophys. Acta Rev. Cancer 2014, 1846, 40–44. [Google Scholar] [CrossRef] [PubMed]
- Menschikowski, M.; Platzbecker, U.; Hagelgans, A.; Vogel, M.; Thiede, C.; Schönefeldt, C.; Lehnert, R.; Eisenhofer, G.; Siegert, G. Aberrant methylation of the M-type phospholipase A 2 receptor gene in leukemic cells. BMC Cancer 2012, 12, 576. [Google Scholar] [CrossRef]
- Augert, A.; Vindrieux, D.; Girard, C.A.; Le Calvé, B.; Gras, B.; Ferrand, M.; Bouchet, B.P.; Puisieux, A.; de Launoit, Y.; Simonnet, H.; et al. PLA2R1 kills cancer cells by inducing mitochondrial stress. Free Radic. Biol. Med. 2013, 65, 969–977. [Google Scholar] [CrossRef]
- Vindrieux, D.; Devailly, G.; Augert, A.; Le Calve, B.; Ferrand, M.; Pigny, P.; Payen, L.; Lambeau, G.; Perrais, M.; Aubert, S.; et al. Repression of PLA2R1 by c-MYC and HIF-2alpha promotes cancer growth. Oncotarget 2014, 5, 1004–1013. [Google Scholar] [CrossRef]
- Brennan, K.; Holsinger, C.; Dosiou, C.; Sunwoo, J.B.; Akatsu, H.; Haile, R.; Gevaert, O. Development of prognostic signatures for intermediate-risk papillary thyroid cancer. BMC Cancer 2016, 16, 736. [Google Scholar] [CrossRef]
- Quach, N.; Mock, J.; Scholpa, N.; Eggert, M.; Payré, C.; Lambeau, G.; Arnold, R.; Cummings, B. Role of the phospholipase A2 receptor in liposome drug delivery in prostate cancer cells. Mol. Pharm. 2014, 11, 3443–3451. [Google Scholar] [CrossRef]
- Humphries, B.; Wang, Z.; Yang, C. MicroRNA regulation of epigenetic modifiers in breast Cancer. Cancers 2019, 11, 897. [Google Scholar] [CrossRef]
- Menschikowski, M.; Hagelgans, A.; Nacke, B.; Jandeck, C.; Sukocheva, O.; Siegert, G. Epigenetic control of phospholipase A2 receptor expression in mammary cancer cells. BMC Cancer 2015, 15, 971. [Google Scholar] [CrossRef]
- Menschikowski, M.; Jandeck, C.; Friedemann, M.; Richter, S.; Thiem, D.; Lange, B.S.; Suttorp, M. Identification and quantification of heterogeneously-methylated DNA fragments using epiallele-sensitive droplet digital polymerase chain reaction (EAST-ddPCR). Cancer Genomics Proteomics 2018, 15, 299–312. [Google Scholar] [CrossRef] [PubMed]
- WHO Breast Cancer Report. Available online: https://www.who.int/cancer/prevention/diagnosis-screening/breast-cancer/en/ (accessed on 3 June 2020).
- Turashvili, G.; Brogi, E. Tumor Heterogeneity in Breast Cancer. Front. Med. (Lausanne) 2017, 4, 227. [Google Scholar] [CrossRef] [PubMed]
- Russnes, H.G.; Lingjærde, O.C.; Borresen-Dale, A.L.; Caldas, C. Breast Cancer Molecular Stratification: From Intrinsic Subtypes to Integrative Clusters. Am. J. Pathol. 2017, 187, 2152–2162. [Google Scholar] [CrossRef] [PubMed]
- Integerated Center for Oncology. Available online: http://bcgenex.centregauducheau.fr/BC-GEM/GEM-Accueil.php?js=1 (accessed on 9 December 2019).
- Kondov, B.; Milenkovikj, Z.; Kondov, G.; Petrushevska, G.; Basheska, N.; Bogdanovska-Todorovska, M.; Tolevska, N.; Ivkovski, L. Presentation of the molecular subtypes of breast cancer detected by immunohistochemistry in surgically treated patients. OAMJMS 2018, 6, 961. [Google Scholar] [CrossRef]
- Goldhirsch, A.; Winer, E.P.; Coates, A.; Gelber, R.; Piccart-Gebhart, M.; Thürlimann, B.; Senn, H.-J.; Members, P.; Albain, K.S.; André, F. Personalizing the treatment of women with early breast cancer: Highlights of the St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2013. Ann. Oncol. 2013, 24, 2206–2223. [Google Scholar] [CrossRef]
- Cancer Line Encyclopedia. Available online: https://portals.broadinstitute.org/ccle (accessed on 20 December 2019).
- Jiang, G.; Zhang, S.; Yazdanparast, A.; Li, M.; Pawar, A.V.; Liu, Y.; Inavolu, S.M.; Cheng, L. Comprehensive comparison of molecular portraits between cell lines and tumors in breast cancer. BMC Genomics 2016, 17, 525. [Google Scholar] [CrossRef]
- Dai, X.; Cheng, H.; Bai, Z.; Li, J. Breast Cancer Cell Line Classification and Its Relevance with Breast Tumor Subtyping. J. Cancer 2017, 8, 3131–3141. [Google Scholar] [CrossRef]
- Kao, J.; Salari, K.; Bocanegra, M.; Choi, Y.L.; Girard, L.; Gandhi, J.; Kwei, K.A.; Hernandez-Boussard, T.; Wang, P.; Gazdar, A.F.; et al. Molecular profiling of breast cancer cell lines defines relevant tumor models and provides a resource for cancer gene discovery. PLoS ONE 2009, 4, e6146. [Google Scholar] [CrossRef]
- Vindrieux, D.; Augert, A.; Girard, C.A.; Gitenay, D.; Lallet-Daher, H.; Wiel, C.; Le Calvé, B.; Gras, B.; Ferrand, M.; Verbeke, S.; et al. PLA2R1 mediates tumor suppression by activating JAK2. Cancer Res. 2013, 73, 6334–6345. [Google Scholar] [CrossRef]
- Friedemann, M.; Nacke, B.; Hagelgans, A.; Jandeck, C.; Bechmann, N.; Ullrich, M.; Belter, B.; Neuber, C.; Sukocheva, O.; Pietzsch, J.; et al. Diverse effects of phospholipase A2 receptor expression on LNCaP and PC-3 prostate cancer cell growth in vitro and in vivo. Oncotarget 2018, 9, 35983. [Google Scholar] [CrossRef][Green Version]
- Augert, A.; Payré, C.; De Launoit, Y.; Gil, J.; Lambeau, G.; Bernard, D. The M-type receptor PLA2R regulates senescence through the p53 pathway. EMBO Rep. 2009, 10, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Tamaru, S.; Mishina, H.; Watanabe, Y.; Watanabe, K.; Fujioka, D.; Takahashi, S.; Suzuki, K.; Nakamura, T.; Obata, J.-e.; Kawabata, K.; et al. Deficiency of phospholipase A2 receptor exacerbates ovalbumin-induced lung inflammation. J. Immunol. 2013, 191, 1021–1028. [Google Scholar] [CrossRef] [PubMed]
- PathwayStudio. Available online: https://www.pathwaystudio.com/ (accessed on 5 May 2020).
- Griveau, A.; Devailly, G.; Eberst, L.; Navaratnam, N.; Le Calvé, B.; Ferrand, M.; Faull, P.; Augert, A.; Dante, R.; Vanacker, J.; et al. The PLA2R1-JAK2 pathway upregulates ERRα and its mitochondrial program to exert tumor-suppressive action. Oncogene 2016, 35, 5033–5042. [Google Scholar] [CrossRef] [PubMed]
- Toiyama, Y.; Okugawa, Y.; Goel, A. DNA methylation and microRNA biomarkers for noninvasive detection of gastric and colorectal cancer. Biochem. Biophys. Res. Commun. 2014, 455, 43–57. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; He, C.; Wang, M.; Ma, X.; Mo, F.; Yang, S.; Han, J.; Wei, X. Targeting epigenetic regulators for cancer therapy: Mechanisms and advances in clinical trials. Signal Transduct. Target. Ther. 2019, 4, 1–39. [Google Scholar] [CrossRef]
- Teschendorff, A.E.; Gao, Y.; Jones, A.; Ruebner, M.; Beckmann, M.W.; Wachter, D.L.; Fasching, P.A.; Widschwendter, M. DNA methylation outliers in normal breast tissue identify field defects that are enriched in cancer. Nat. Commun. 2016, 7, 10478. [Google Scholar] [CrossRef]
- Boxer, L.M.; Dang, C.V. Translocations involving c-myc and c-myc function. Oncogene 2001, 20, 5595–5610. [Google Scholar] [CrossRef]
- Hanby, A.M.; Walker, C. Tavassoli FA, Devilee P: Pathology and Genetics: Tumours of the Breast and Female Genital Organs. WHO Classification of Tumours series—volume IV.; Lyon, France: IARC Press. Breast Cancer Res. 2004, 6, 133. [Google Scholar] [CrossRef]
- Fitzgibbons, P.L.; Dillon, D.A.; Alsabeh, R.; Berman, M.A.; Hayes, D.F.; Hicks, D.G.; Hughes, K.S.; Nofech-Mozes, S. Template for reporting results of biomarker testing of specimens from patients with carcinoma of the breast. Arch. Pathol. Lab. Med. 2014, 138, 595–601. [Google Scholar] [CrossRef]
- Fitzgibbons, P.L.; Bartley, A.N.; Connolly, J.L. Template for reporting results of biomarker testing of specimens from patients with carcinoma of the breast. In 2018 CAP Accreditation Program Checklist and the CAP/ASCO Guidelines; Version: BreastBiomarkers 1.2.0.1; College of American Pathologists (CAP): Northfield, IL, USA, 2018. [Google Scholar]
- Fawcett, T. An introduction to ROC analysis. Pattern Recognit. Lett. 2006, 27, 861–874. [Google Scholar] [CrossRef]


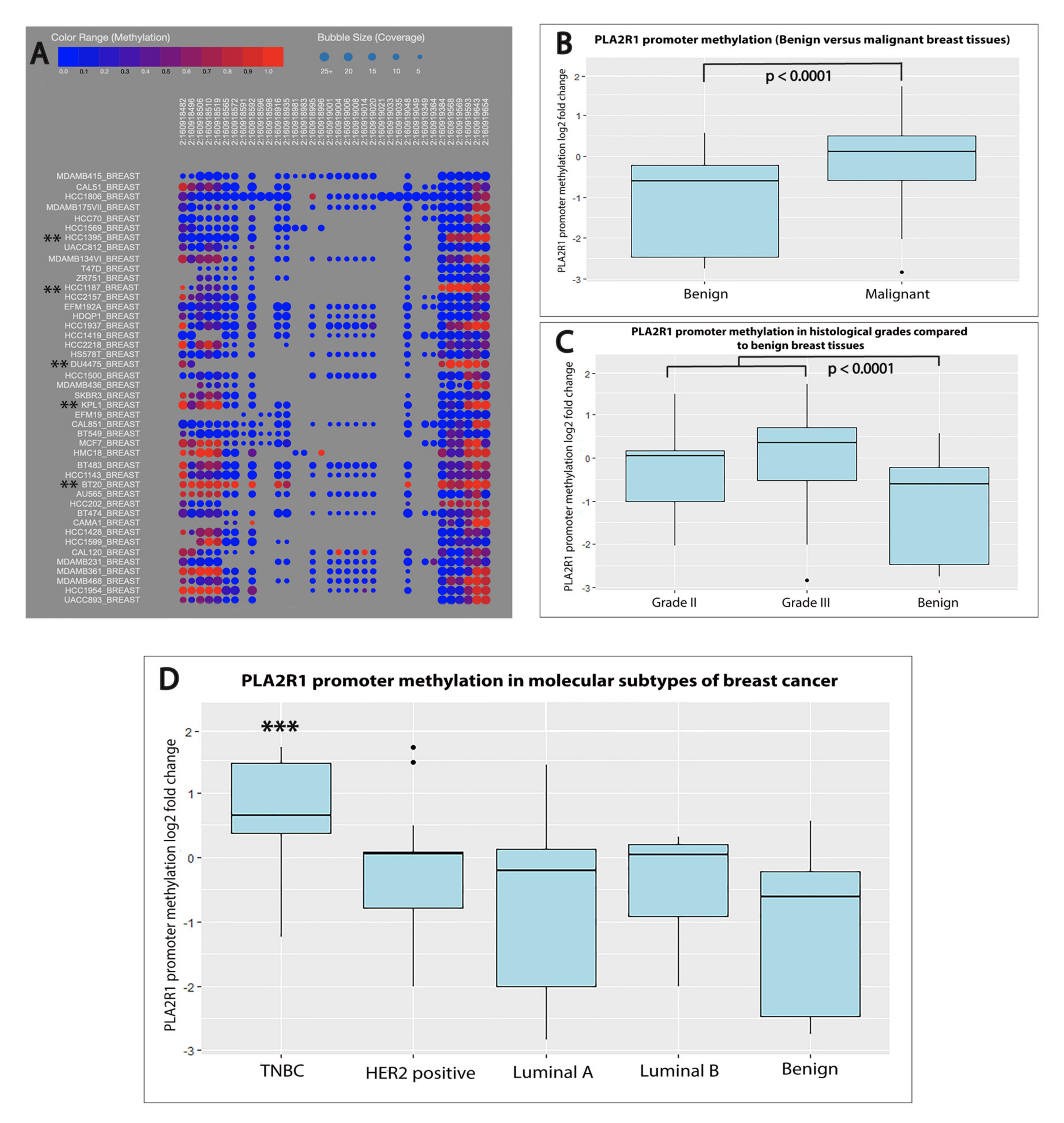

| Variable | N (%) |
|---|---|
| Benign fibroadenomas (control) | 30 (30%) |
| Infiltrating ductal carcinomas | 70 (70%) |
| Tumor Grades | |
| II | 24 (34.2%) |
| III | 46 (65.71%) |
| ER Status | |
| Negative | 47 (67.14%) |
| Positive | 23(32.85%) |
| PR Status | |
| Negative | 47 (67.14%) |
| Positive | 23 (32.85%) |
| HER2 Expression | |
| Negative | 44 (62.8%) |
| Positive | 26 (37.14) |
| Molecular Subtypes | |
| TNBC | 25 (35.71%) |
| HER2-positive | 22 (31.4%) |
| Luminal A | 12 (17.14%) |
| Luminal B | 11 (15.71%) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mitwally, N.; Yousef, E.; Abd Al Aziz, A.; Taha, M. Clinical Significance of Expression Changes and Promoter Methylation of PLA2R1 in Tissues of Breast Cancer Patients. Int. J. Mol. Sci. 2020, 21, 5453. https://doi.org/10.3390/ijms21155453
Mitwally N, Yousef E, Abd Al Aziz A, Taha M. Clinical Significance of Expression Changes and Promoter Methylation of PLA2R1 in Tissues of Breast Cancer Patients. International Journal of Molecular Sciences. 2020; 21(15):5453. https://doi.org/10.3390/ijms21155453
Chicago/Turabian StyleMitwally, Noha, Einas Yousef, Ahmad Abd Al Aziz, and Mohamed Taha. 2020. "Clinical Significance of Expression Changes and Promoter Methylation of PLA2R1 in Tissues of Breast Cancer Patients" International Journal of Molecular Sciences 21, no. 15: 5453. https://doi.org/10.3390/ijms21155453
APA StyleMitwally, N., Yousef, E., Abd Al Aziz, A., & Taha, M. (2020). Clinical Significance of Expression Changes and Promoter Methylation of PLA2R1 in Tissues of Breast Cancer Patients. International Journal of Molecular Sciences, 21(15), 5453. https://doi.org/10.3390/ijms21155453





