Molecular Mechanism Contributing to Malnutrition and Sarcopenia in Patients with Liver Cirrhosis
Abstract
1. Introduction
Disease-Related Malnutrition and Sarcopenia
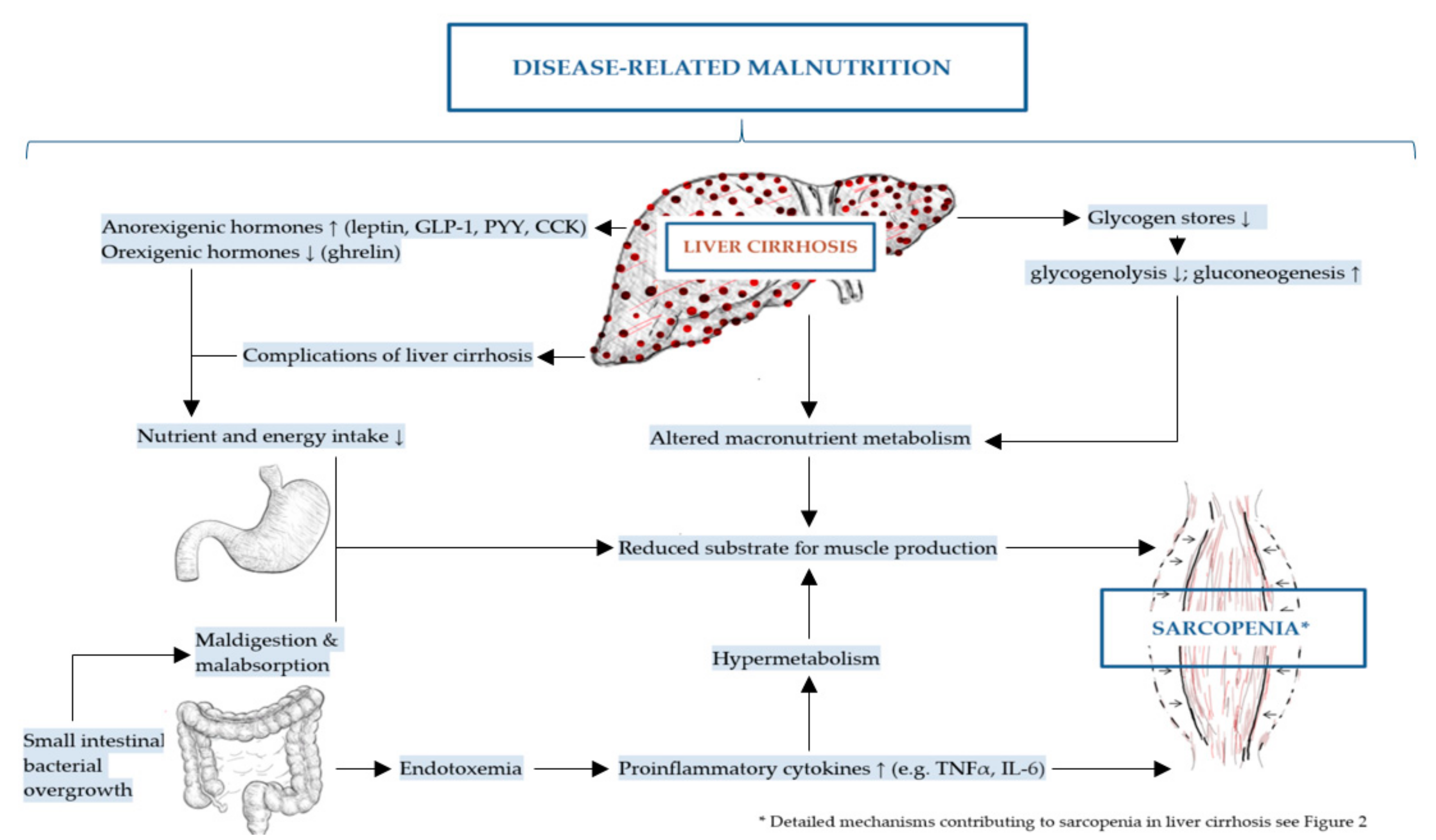
2. Molecular Mechanisms Contributing to Disease-Related Malnutrition (DRM) and Sarcopenia in Liver Cirrhosis
2.1. Energy-Associated Mechanisms
2.1.1. Reduced Nutrient and Energy Intake
Regulators of Appetite and Satiety
Ghrelin
Leptin
Cholecystokinin (CCK), Glucagon Like peptide-1 (GLP-1), Peptide YY (PYY)
Proinflammatory Cytokines
Regulators of Taste and Smell
Dietary Restrictions
2.1.2. Maldigestion and Malabsorption
Cholestasis
Small Intestinal Bacterial Overgrowth and Gut Microbiome
2.1.3. Altered Macronutrient Metabolism
Glucose Metabolism
Alterations in Protein Turnover
Hypermetabolism
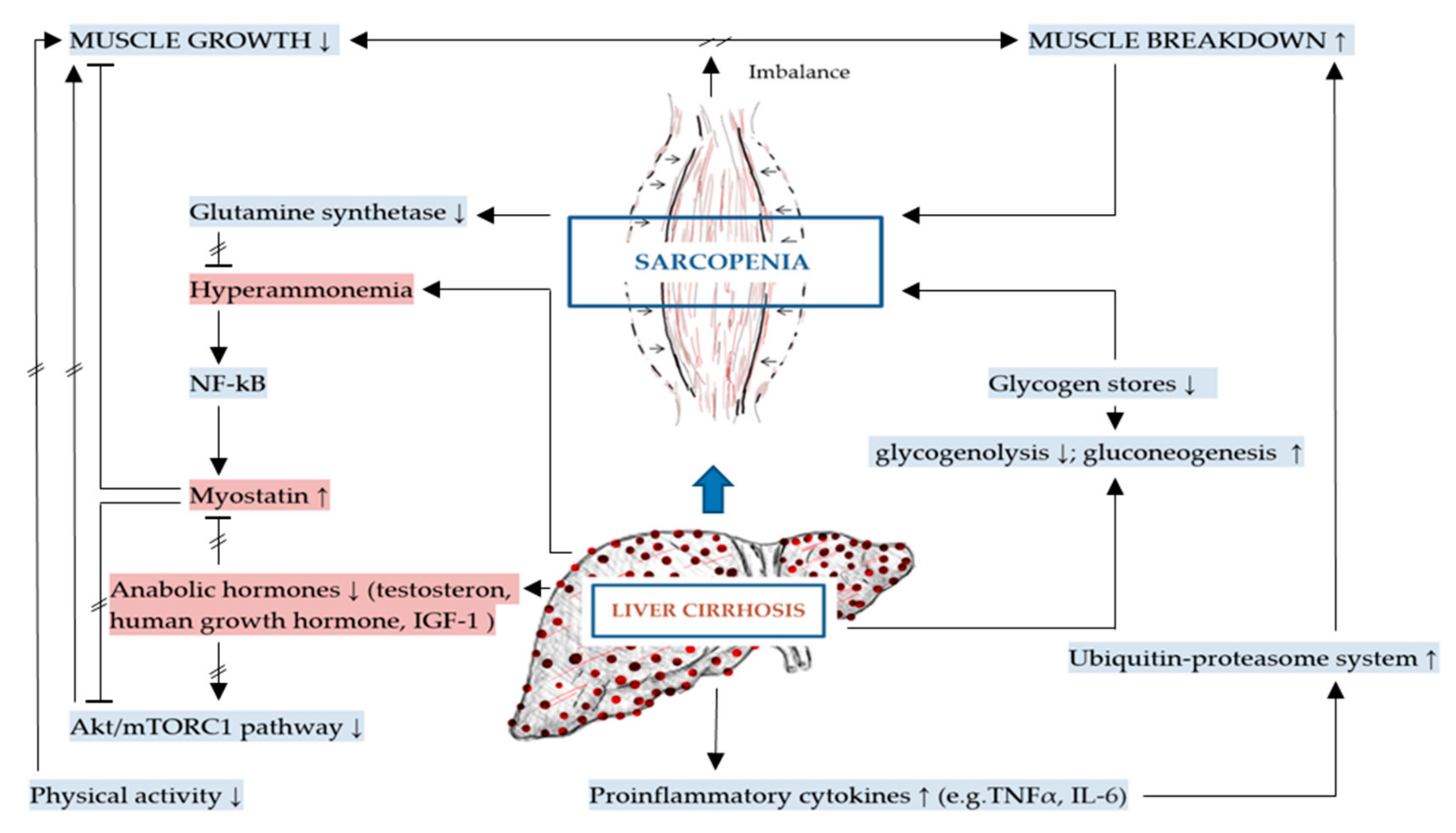
2.2. Additional to DRM: Mechanisms Involved in Sarcopenia in Liver Cirrhosis
2.2.1. Inhibition of Muscle Growth and Elevated Myostatin
Hormonal Regulation of Muscle Homeostasis
2.2.2. Increased Muscle Breakdown
Ubiquitin–Proteasome System and Autophagy
2.2.3. Physical Activity
3. DRM and Sarcopenia Management Strategies Based on Molecular Mechanisms
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AAAs | Aromatic amino acids |
| AGRP | Agouti-related protein |
| ATP | Adenosine triphosphate |
| BCAAs | Branched-chained amino acids |
| BMI | Body mass index |
| CCK | Cholecystokinin |
| DRM | Disease-related malnutrition |
| E3 | Ubiquitin protein ligase |
| EWGSOP2 | European working group on sarcopenia in older people |
| GH | Growth hormone |
| GLIM | Global leadership initiative on malnutrition |
| GLP-1 | Glucagon like peptide-1 |
| HBV | Chronic hepatitis B virus |
| HCV | Chronic hepatitis C virus |
| IGF-1 | Insulin-like growth factor |
| IGT | Impaired glucose tolerance |
| IL-1 β | Interleukin 1 beta |
| IL-12 | Interleukin 12 |
| IL-6 | Interleukin 6 |
| kDa | Kilo-dalton |
| MAFbx | Muscle atrophy F-box |
| MHC | Myosin heavy chain |
| MSS | Malnutrition sarcopenia syndrome |
| mTORC1 | Mammalian target of rapamycin complex-1 |
| MuRF-1 | Muscle specific ring-finger protein 1 |
| NASH | Non-alcoholic steatohepatitis |
| NF-κB | Nuclear factor kappa B |
| NPY | Neuropeptide Y |
| PAMPs | Pathogen associated molecular patterns |
| PBC | Primary biliary cholangitis |
| PP | Pancreatic polypeptide |
| PSC | Primary sclerosing cholangitis |
| PYY | Peptide YY |
| REE | Resting energy expenditure |
| SIBO | Small intestinal bacterial overgrowth |
| SNS | Sympathetic nervous system |
| TCA | Tricarboxylic acid cycle |
| TEE | Total energy expenditure |
| TGF-β | Transforming growth factor beta |
| TNF-α | Tumor necrosis factor-alpha |
| UPP | Ubiquitin-proteasome pathway |
| UPS | Ubiquitin-proteasome system |
References
- Asrani, S.K.; Devarbhavi, H.; Eaton, J.; Kamath, P.S. Burden of liver diseases in the world. J. Hepatol. 2019, 70, 151–171. [Google Scholar] [CrossRef] [PubMed]
- Palmer, L.B.; Kuftinec, G.; Pearlman, M.; Green, C.H. Nutrition in Cirrhosis. Curr. Gastroenterol. Rep. 2019, 21, 38. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.N.; Chen, J.; Cai, F.F.; Lu, Y.Y.; Chen, Q.L.; Zhang, Y.Y.; Liu, P.; Su, S.B. A metabolic mechanism analysis of Fuzheng-Huayu formula for improving liver cirrhosis with traditional Chinese medicine syndromes. Acta Pharmacol. Sin. 2018, 39, 942–951. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Global Hepatitis Report 2017. Available online: http://www.who.int/hepatitis/publications/global-hepatitis-report2017/en/ (accessed on 3 July 2020).
- Statistisches Bundesamt (DESTATIS), Gesundheit. Diagnosedaten der Patienten und Patientinnen in Krankenh-usern (einschl. Sterbe- und Stundenfälle), in Fachserie 12 Reihe 6.2.1). Available online: https://www.destatis.de/DE/Themen/Gesellschaft-Umwelt/Gesundheit/Krankenhaeuser/Publikationen/Downloads-Krankenhaeuser/diagnosedaten-krankenhaus-2120621167004.html (accessed on 10 July 2020).
- Parkash, O.; Jafri, W.; Munir, S.M.; Iqbal, R. Assessment of malnutrition in patients with liver cirrhosis using protein calorie malnutrition (PCM) score verses bio-electrical impedance analysis (BIA). BMC Res. Notes 2018, 11, 545. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Aguilera, J.R.; De Vaca, R.P.-C.; Guerrero-Celis, N.; Velasco-Loyden, G.; Domínguez-López, M.; Recillas-Targa, F.; De Sánchez, V.C. Molecular and Cellular Aspects of Cirrhosis and How an Adenosine Derivative Could Revert Fibrosis. In Liver Cirrhosis—Debates and Current Challenges; Tsoulfas, G., Ed.; IntechOpen: London, UK, 2019; Available online: https://www.intechopen.com/books/liver-cirrhosis-debates-and-current-challenges (accessed on 6 July 2020).
- Buechler, C.; Haberl, E.M.; Rein-Fischboeck, L.; Aslanidis, C. Adipokines in Liver Cirrhosis. Int. J. Mol. Sci. 2017, 18, 1392. [Google Scholar] [CrossRef]
- Pimpin, L.; Cortez-Pinto, H.; Negro, F.; Corbould, E.; Lazarus, J.; Webber, L.; Sheron, N. Burden of liver disease in Europe: Epidemiology and analysis of risk factors to identify prevention policies. J. Hepatol. 2018, 69, 718–735. [Google Scholar] [CrossRef]
- Rungta, S.; Deep, A.; Swaroop, S. Malnutrition in Liver Cirrhosis: A Review. J. Clin. Diag. Res. 2019, 13, OE01–OE05. [Google Scholar] [CrossRef]
- Nishikawa, H.; Shiraki, M.; Hiramatsu, A.; Moriya, K.; Hino, K.; Nishiguchi, S. Japan Society of Hepatology guidelines for sarcopenia in liver disease (1st edition): Recommendation from the working group for creation of sarcopenia assessment criteria. Hepatol. Res. 2016, 46, 951–963. [Google Scholar] [CrossRef]
- Bémeur, C.; Desjardins, P.; Butterworth, R. Role of Nutrition in the Management of Hepatic Encephalopathy in End-Stage Liver Failure. J. Nutr. Metab. 2010, 2010, 1–12. [Google Scholar] [CrossRef]
- Bémeur, C.; Butterworth, R.F. Reprint of: Nutrition in the Management of Cirrhosis and its Neurological Complications. J. Clin. Exp. Hepatol. 2015, 5, S131–S140. [Google Scholar] [CrossRef] [PubMed]
- Theodoridis, X.; Grammatikopoulou, M.G.; Petalidou, A.; Kontonika, S.-M.; Potamianos, S.P.; Bogdanos, D.P. A Systematic Review of Medical Nutrition Therapy Guidelines for Liver Cirrhosis: Do We Agree? Nutr. Clin. Pr. 2019, 35, 98–107. [Google Scholar] [CrossRef] [PubMed]
- Anand, A.C. Nutrition and Muscle in Cirrhosis. J. Clin. Exp. Hepatol. 2017, 7, 340–357. [Google Scholar] [CrossRef] [PubMed]
- Kwarta, E.; Nagle, S.; Welstead, L. Update on Malnutrition in Liver Cirrhosis: Assessment and Treatment. Curr. Hepatol. Rep. 2014, 13, 24–34. [Google Scholar] [CrossRef]
- Fan, J.G.; Cao, H.X. Malnutrition and nutritional support. In Cirrhosis: A Practical Guide to Management; Samuel, S.L., Richard, M., Eds.; John Wiley & Sons, Ltd.: Chichester, UK, 2015; pp. 124–136. [Google Scholar]
- Ockenga, J.; Bischoff, S.C.; Tillmann, H.L.; Rifai, K.; Widjaja, A.; Böker, K.H.; Manns, M.P.; Brabant, G. Elevated bound leptin correlates with energy expenditure in cirrhotics. Gastroenterology 2000, 119, 1656–1662. [Google Scholar] [CrossRef]
- Bunchorntavakul, C.; Reddy, K. Review article: Malnutrition/sarcopenia and frailty in patients with cirrhosis. Aliment. Pharmacol. Ther. 2019, 51, 64–77. [Google Scholar] [CrossRef] [PubMed]
- Thandassery, R.B.; Montano-Loza, A. Role of Nutrition and Muscle in Cirrhosis. Curr. Treat. Options Gastroenterol. 2016, 14, 257–273. [Google Scholar] [CrossRef]
- Rachakonda, V.; Borhani, A.A.; Dunn, M.A.; Andrzejewski, M.; Martin, K.; Behari, J. Serum Leptin Is a Biomarker of Malnutrition in Decompensated Cirrhosis. PLoS ONE 2016, 11, e0159142. [Google Scholar] [CrossRef]
- Cederholm, T.; Jensen, G.; Correia, M.; Gonzalez, M.; Fukushima, R.; Higashiguchi, T.; Baptista, G.; Barazzoni, R.; Blaauw, R.; Coats, A.; et al. GLIM criteria for the diagnosis of malnutrition—A consensus report from the global clinical nutrition community. Clin. Nutr. 2019, 38, 1–9. [Google Scholar] [CrossRef]
- Eghtesad, S.; Poustchi, H.; Malekzadeh, R. Malnutrition in Liver Cirrhosis: The Influence of Protein and Sodium. Middle East J. Dig. Dis. 2013, 5, 65–75. [Google Scholar]
- Chawla, Y.; Bodh, V. Clinical clues to the diagnosis of cirrhosis. In Cirrhosis: A Practical Guide to Management; Samuel, S.L., Richard, M., Eds.; John Wiley & Sons, Ltd.: Chichester, UK, 2015; pp. 1–11. [Google Scholar]
- Georgiou, A.; Papatheodoridis, G.V.; Alexopoulou, A.; Deutsch, M.; Vlachogiannakos, I.; Ioannidou, P.; Papageorgiou, M.-V.; Papadopoulos, N.; Tsibouris, P.; Prapa, A.; et al. Evaluation of the effectiveness of eight screening tools in detecting risk of malnutrition in cirrhotic patients: The KIRRHOS study. Br. J. Nutr. 2019, 122, 1–9. [Google Scholar] [CrossRef]
- Ney, M.; Li, S.; VanderMeer, B.; Gramlich, L.; Ismond, K.P.; Raman, M.; Tandon, P. Systematic review with meta-analysis: Nutritional screening and assessment tools in cirrhosis. Liver Int. 2019, 40, 664–673. [Google Scholar] [CrossRef]
- Meyer, F.; Valentini, L. Disease-Related Malnutrition and Sarcopenia as Determinants of Clinical Outcome. Visc. Med. 2019, 35, 282–291. [Google Scholar] [CrossRef] [PubMed]
- Kalafateli, M.; Mantzoukis, K.; Yau, Y.C.; Mohammad, A.O.; Arora, S.; Rodrigues, S.; De Vos, M.; Papadimitriou, K.; Thorburn, U.; O’Beirne, J.; et al. Malnutrition and sarcopenia predict post-liver transplantation outcomes independently of the Model for End-stage Liver Disease score. J. Cachex. Sarcopenia Muscle 2016, 8, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Barve, A. Malnutrition in Cirrhosis. Vidarbha J. Int. Med. 2018, 25, 41–53. [Google Scholar]
- Dasarathy, S. The Pathogenesis of Physical Frailty and Sarcopenia; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2019; pp. 33–53. [Google Scholar]
- Lin, S.-Y.; Chen, W.-Y.; Lee, F.-Y.; Huang, C.-J.; Sheu, W.H.-H. Activation of ubiquitin-proteasome pathway is involved in skeletal muscle wasting in a rat model with biliary cirrhosis: Potential role of TNF-α. Am. J. Physiol. Metab. 2005, 288, E493–E501. [Google Scholar] [CrossRef] [PubMed]
- Bojko, M. Causes of Sarcopenia in Liver Cirrhosis. Clin. Liver Dis. 2019, 14, 167–170. [Google Scholar] [CrossRef]
- Dasarathy, S. Consilience in sarcopenia of cirrhosis. J. Cachex. Sarcopenia Muscle 2012, 3, 225–237. [Google Scholar] [CrossRef]
- Mazurak, V.C.; Tandon, P.; Montano-Loza, A. Nutrition and the transplant candidate. Liver Transplant. 2017, 23, 1451–1464. [Google Scholar] [CrossRef] [PubMed]
- Cederholm, T.; Barazzoni, R.; Austin, P.D.; Ballmer, P.E.; Biolo, G.; Bischoff, S.C.; Compher, C.; Correia, M.I.T.D.; Higashiguchi, T.; Holst, M.; et al. ESPEN guidelines on definitions and terminology of clinical nutrition. Clin. Nutr. 2016, 36, 49–64. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyère, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A. Writing Group for the European Working Group on Sarcopenia in Older People 2 (EWGSOP2), and the Extended Group for EWGSOP2. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef]
- Zhuang, C.-L.; Shen, X.; Zou, H.-B.; Dong, Q.-T.; Cai, H.-Y.; Chen, X.-L.; Yu, Z.; Wang, S.-L. EWGSOP2 versus EWGSOP1 for sarcopenia to predict prognosis in patients with gastric cancer after radical gastrectomy: Analysis from a large-scale prospective study. Clin. Nutr. 2019, 39. [Google Scholar] [CrossRef] [PubMed]
- Bamba, S.; Sasaki, M.; Takaoka, A.; Takahashi, K.; Imaeda, H.; Nishida, A.; Inatomi, O.; Sugimoto, M.; Andoh, A. Sarcopenia is a predictive factor for intestinal resection in admitted patients with Crohn’s disease. PLoS ONE 2017, 12, e0180036. [Google Scholar] [CrossRef]
- Ebadi, M.; Bhanji, R.A.; Mazurak, V.C.; Montano-Loza, A. Sarcopenia in cirrhosis: From pathogenesis to interventions. J. Gastroenterol. 2019, 54, 845–859. [Google Scholar] [CrossRef] [PubMed]
- Rachakonda, V.; Argemi, J.; Borhani, A.A.; Bataller, R.; Tevar, A.; Behari, J. Reduced Serum Sphingolipids Constitute a Molecular Signature of Malnutrition in Hospitalized Patients with Decompensated Cirrhosis. Clin. Transl. Gastroenterol. 2019, 10, e00013. [Google Scholar] [CrossRef] [PubMed]
- Merli, M.; Iebba, V.; Giusto, M. What is new about diet in hepatic encephalopathy. Metab. Brain Dis. 2015, 31, 1289–1294. [Google Scholar] [CrossRef]
- Dasarathy, S.; Merli, M. Sarcopenia from mechanism to diagnosis and treatment in liver disease. J. Hepatol. 2016, 65, 1232–1244. [Google Scholar] [CrossRef]
- Kalaitzakis, E.; Bosaeus, I.; Öhman, L.; Björnsson, E. Altered postprandial glucose, insulin, leptin, and ghrelin in liver cirrhosis: Correlations with energy intake and resting energy expenditure. Am. J. Clin. Nutr. 2007, 85, 808–815. [Google Scholar] [CrossRef]
- Tandon, P.; Raman, M.; Mourtzakis, M.; Merli, M. A practical approach to nutritional screening and assessment in cirrhosis. Hepatology 2017, 65, 1044–1057. [Google Scholar] [CrossRef] [PubMed]
- Dhaliwal, A.; Towey, J.; Lord, J.M.; Armstrong, M.; Elsharkawy, A.M. Nutrition in liver cirrhosis: A case-based overview. Front. Gastroenterol. 2019, 11, 155–161. [Google Scholar] [CrossRef]
- Chen, H.-W.; Dunn, M.A. Arresting frailty and sarcopenia in cirrhosis: Future prospects. Clin. Liver Dis. 2018, 11, 52–57. [Google Scholar] [CrossRef] [PubMed]
- Sinclair, M.; Gow, P.J.; Grossmann, M.; Angus, P.W. Review article: Sarcopenia in cirrhosis—Aetiology, implications and potential therapeutic interventions. Aliment. Pharmacol. Ther. 2016, 43, 765–777. [Google Scholar] [CrossRef] [PubMed]
- Chao, A.; Waitzberg, D.; De Jesus, R.P.; Bueno, A.A.; Kha, V.; Allen, K.; Kappus, M.; Medici, V. Malnutrition and Nutritional Support in Alcoholic Liver Disease: A Review. Curr. Gastroenterol. Rep. 2016, 18, 65. [Google Scholar] [CrossRef] [PubMed]
- Tsiaousi, E.T.; Hatzitolios, A.; Trygonis, S.K.; Savopoulos, C.G. Malnutrition in end stage liver disease: Recommendations and nutritional support. J. Gastroenterol. Hepatol. 2008, 23, 527–533. [Google Scholar] [CrossRef] [PubMed]
- Ataseven, H.; Bahcecioglu, I.H.; Kuzu, N.; Yalniz, M.; Celebi, S.; Erensoy, A.; Üstündağ, B. The Levels of Ghrelin, Leptin, TNF-α, and IL-6 in Liver Cirrhosis and Hepatocellular Carcinoma due to HBV and HDV Infection. Mediat. Inflamm. 2006, 2006, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Dibb, M. Nutritional Management of End-Stage Liver Disease. In Clinical Cardiac Electrophysiology in Clinical Practice; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2016; Volume 21, pp. 71–87. [Google Scholar]
- Wang, T.; Shen, J. Usefulness of Simplified Nutritional Appetite Questionnaire (SNAQ) in Appetite Assessment in Elder Patients with Liver Cirrhosis. J. Nutr. Health Aging 2018, 22, 911–915. [Google Scholar] [CrossRef] [PubMed]
- Laviano, A.; Meguid, M.; Fanelli, F.R. Anorexia. In Cachexia and Wasting: A Modern Approach; Anker, S.D., Inui, A., Morley, J.E., Fanelli, F.R., Scevola, D., Schuster, M.W., Yeh, S.-S., Eds.; Springer: Milano, Italy, 2006; pp. 139–148. [Google Scholar]
- Marchesini, G.; Bianchi, G.; Lucidi, P.; Villanova, N.; Zoli, M.; De Feo, P. Plasma Ghrelin Concentrations, Food Intake, and Anorexia in Liver Failure. J. Clin. Endocrinol. Metab. 2004, 89, 2136–2141. [Google Scholar] [CrossRef]
- Koga, A.; Toda, K.; Tatsushima, K.; Matsuubayashi, S.; Tamura, N.; Imamura, M.; Kawai, K. Portal hypertension in prolonged anorexia nervosa with laxative abuse: A case report of three patients. Int. J. Eat. Disord. 2019, 52, 211–215. [Google Scholar] [CrossRef]
- Valentini, L.; Schuetz, T.; Omar, A.; Gläser, S.; Kasim, E.; Nowotny, P.; Kröncke, T.; Ockenga, J. Abnormal plasma peptide YY3–36 levels in patients with liver cirrhosis. Nutrition 2011, 27, 880–884. [Google Scholar] [CrossRef]
- Calmet, F.; Martin, P.; Pearlman, M. Nutrition in Patients with Cirrhosis. Gastroenterol. Hepatol. 2019, 15, 248–254. [Google Scholar]
- Small, C.; Bloom, S. Gut hormones and the control of appetite. Trends Endocrinol. Metab. 2004, 15, 259–263. [Google Scholar] [CrossRef]
- Crooks, B.; Stamataki, N.S.; McLaughlin, J.T. Appetite, the enteroendocrine system, gastrointestinal disease and obesity. Proc. Nutr. Soc. 2020, 1–9. [Google Scholar] [CrossRef]
- Bohórquez, D.V.; Liddle, R.A. Gastrointestinal hormones and neurotransmitters. In Sleisenger and Fordtran’s gastrointestinal and Liver Disease E-Book: Pathophysiology, Diagnosis, Management; Feldman, M., Friedman, L.S., Brandt, L.J., Eds.; Elsevier Health Sciences: Amsterdam, The Netherlands, 2016; pp. 36–56. [Google Scholar]
- Gardiner, J.V.; Jayasena, C.N.; Bloom, S.R. Gut Hormones: A Weight Off Your Mind. J. Neuroendocr. 2008, 20, 834–841. [Google Scholar] [CrossRef] [PubMed]
- Moreno, M.; Chaves, F.J.; Sancho-Bru, P.; Ramalho, L.N.Z.; Ramalho, L.N.Z.; Mansego, M.L.; Ivorra, C.; Dominguez, M.; Conde, L.; Millán, C.; et al. Ghrelin attenuates hepatocellular injury and liver fibrogenesis in rodents and influences fibrosis progression in humans. Hepatology 2010, 51, 974–985. [Google Scholar] [CrossRef]
- Freire-Regatillo, A.; Argente-Arizón, P.; Argente, J.; Garcia-Segura, L.M.; Chowen, J. Non-Neuronal Cells in the Hypothalamic Adaptation to Metabolic Signals. Front. Endocrinol. 2017, 8, 51. [Google Scholar] [CrossRef] [PubMed]
- Bluher, S.; Mantzoros, C.S. The role of leptin in regulating neuroendocrine function in humans. J. Nutr. 2004, 134, 2469S–2474S. [Google Scholar] [CrossRef]
- Nolte, W.; Wirtz, M.; Rossbach, C.; Buchwald, A.B.; Scholz, K.H.; Leonhardt, U.; Ramadori, G. TIPS Implantation Raises Leptin Levels in Patients with Liver Cirrhosis. Exp. Clin. Endocrinol. Diabetes 2003, 111, 435–442. [Google Scholar] [CrossRef] [PubMed]
- Campillo, B.; Sherman, E.; Richardet, J.; Bories, P. Serum leptin levels in alcoholic liver cirrhosis: Relationship with gender, nutritional status, liver function and energy metabolism. Eur. J. Clin. Nutr. 2001, 55, 980–988. [Google Scholar] [CrossRef] [PubMed]
- Schaible, U.E.; Kaufmann, S.H. Malnutrition and Infection: Complex Mechanisms and Global Impacts. PLoS Med. 2007, 4, e115. [Google Scholar] [CrossRef]
- Cheung, K.; Lee, S.S.; Raman, M. Prevalence and Mechanisms of Malnutrition in Patients with Advanced Liver Disease, and Nutrition Management Strategies. Clin. Gastroenterol. Hepatol. 2012, 10, 117–125. [Google Scholar] [CrossRef]
- Henkel, A.S.; Buchman, A.L. Nutritional support in patients with chronic liver disease. Nat. Clin. Pr. Gastroenterol. Hepatol. 2006, 3, 202–209. [Google Scholar] [CrossRef]
- Fakhry, J.; Wang, J.; Martins, P.; Fothergill, L.J.; Hunne, B.; Prieur, P.; Shulkes, A.; Rehfeld, J.F.; Callaghan, B.; Furness, J. Distribution and characterisation of CCK containing enteroendocrine cells of the mouse small and large intestine. Cell Tissue Res. 2017, 369, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Keire, D.A.; Mannon, P.; Kobayashi, M.; Walsh, J.H.; Solomon, T.E.; Reeve, J.R. Primary structures of PYY, [Pro(34)]PYY, and PYY-(3-36) confer different conformations and receptor selectivity. Am. J. Physiol. Liver Physiol. 2000, 279, G126–G131. [Google Scholar] [CrossRef]
- Bahr, M.J.; Ockenga, J.; Böker, K.H.W.; Manns, M.P.; Tietge, U.J.F. Elevated resistin levels in cirrhosis are associated with the proinflammatory state and altered hepatic glucose metabolism but not with insulin resistance. Am. J. Physiol. Metab. 2006, 291, E199–E206. [Google Scholar] [CrossRef] [PubMed]
- Aranda-Michel, J. Nutrition in hepatic failure and liver transplantation. Curr. Gastroenterol. Rep. 2001, 3, 362–370. [Google Scholar] [CrossRef] [PubMed]
- Wiest, R.; Lawson, M.A.; Geuking, M.B. Pathological bacterial translocation in liver cirrhosis. J. Hepatol. 2014, 60, 197–209. [Google Scholar] [CrossRef]
- Fukui, H. Gut-liver axis in liver cirrhosis: How to manage leaky gut and endotoxemia. World J. Hepatol. 2015, 7, 425–442. [Google Scholar] [CrossRef]
- Kapil, S.; Duseja, A.; Sharma, B.K.; Singla, B.; Chakraborti, A.; Das, A.; Ray, P.; Dhiman, R.K.; Chawla, Y. Small intestinal bacterial overgrowth and toll-like receptor signaling in patients with non-alcoholic fatty liver disease. J. Gastroenterol. Hepatol. 2015, 31, 213–221. [Google Scholar] [CrossRef]
- Heiser, C.; Haller, B.; Sohn, M.; Hofauer, B.; Knopf, A.; Muhling, T.; Freiherr, J.; Bender, M.; Tiller, M.; Schmidt, A.; et al. Olfactory Function is Affected in Patients with Cirrhosis Depending on the Severity of Hepatic Encephalopathy. Ann. Hepatol. 2018, 17, 822–829. [Google Scholar] [CrossRef]
- Sevastianos, V.A.; Sp, D. Malnutrition and Sarcopenia in Advanced Liver Disease. J. Nutr. Food Sci. 2016, 6, 2. [Google Scholar] [CrossRef]
- Fernandes, S.A.; Bona, S.; Cerski, C.T.S.; Marroni, N.P.; Marroni, N.P. Alteration of taste buds in experimental cirrhosis. Is there correlation with human hypogeusia? Arq. Gastroenterol. 2016, 53, 278–284. [Google Scholar] [CrossRef]
- Schiavo, L.; Busetto, L.; Cesaretti, M.; Zelber-Sagi, S.; Deutsch, L.; Iannelli, A. Nutritional issues in patients with obesity and cirrhosis. World J. Gastroenterol. 2018, 24, 3330–3346. [Google Scholar] [CrossRef] [PubMed]
- Chang, K.-V.; Chen, J.-D.; Wu, W.-T.; Huang, K.-C.; Lin, H.-Y.; Han, D.-S. Is sarcopenia associated with hepatic encephalopathy in liver cirrhosis? A systematic review and meta-analysis. J. Formos. Med Assoc. 2019, 118, 833–842. [Google Scholar] [CrossRef] [PubMed]
- Moss, O. Nutrition Priorities: Diet Recommendations in Liver Cirrhosis. Clin. Liver Dis. 2019, 14, 146–148. [Google Scholar] [CrossRef] [PubMed]
- Plauth, M.; Bernal, W.; Dasarathy, S.; Merli, M.; Plank, L.D.; Schütz, T.; Bischoff, S.C. ESPEN guideline on clinical nutrition in liver disease. Clin. Nutr. 2019, 38, 485–521. [Google Scholar] [CrossRef]
- McClain, C.J. Nutrition in Patients with Cirrhosis. Gastroenterol. Hepatol. 2016, 12, 507–510. [Google Scholar]
- Morando, F.; Rosi, S.; Gola, E.; Nardi, M.; Piano, S.; Fasolato, S.; Stanco, M.; Cavallin, M.; Romano, A.; Sticca, A.; et al. Adherence to a moderate sodium restriction diet in outpatients with cirrhosis and ascites: A real-life cross-sectional study. Liver Int. 2014, 35, 1508–1515. [Google Scholar] [CrossRef]
- Aghdassi, A.A.; Schneider, A.; Kahl, M.; Schütte, K.; Kuliaviene, I.; Salacone, P.; Lutz, J.; Tukiainen, E.; Simon, P.; Schauer, B.; et al. Analysis of lifestyle factors in patients with concomitant chronic pancreatitis and liver cirrhosis. Pancreatol. 2017, 17, 698–705. [Google Scholar] [CrossRef]
- Perri, R.E. Nutrition in Patients with Diseases of the Liver and Pancreas. In Nutrition Guide for Physicians and Related Healthcare Professionals; Temple, N.J., Wilson, T., Bray, G.A., Eds.; Springer: Berlin/Heidelberg, Germany, 2017; pp. 139–144. [Google Scholar]
- Chiang, J.Y. Bile acid metabolism and signaling. Compr. Physiol. 2013, 3, 1191–1212. [Google Scholar] [CrossRef]
- Kalaitzakis, E. Gastrointestinal dysfunction in liver cirrhosis. World J. Gastroenterol. 2014, 20, 14686–14695. [Google Scholar] [CrossRef]
- Augustyn, M.; Grys, I.; Kukla, M. Small intestinal bacterial overgrowth and nonalcoholic fatty liver disease. Clin. Exp. Hepatol. 2019, 5, 1–10. [Google Scholar] [CrossRef]
- El Kurdi, B.; Babar, S.; El Iskandarani, M.; Bataineh, A.; Lerch, M.M.; Young, M.; Singh, V.P. Factors That Affect Prevalence of Small Intestinal Bacterial Overgrowth in Chronic Pancreatitis. Clin. Transl. Gastroenterol. 2019, 10, e00072. [Google Scholar] [CrossRef] [PubMed]
- Fukui, H.; Wiest, R. Changes of Intestinal Functions in Liver Cirrhosis. Inflamm. Intest. Dis. 2016, 1, 24–40. [Google Scholar] [CrossRef] [PubMed]
- Bauer, T.M.; Steinbruckner, B.; Brinkmann, F.E.; Ditzen, A.K.; Schwacha, H.; Aponte, J.J.; Pelz, K.; Kist, M.; Blum, H.E. Small intestinal bacterial overgrowth in patients with cirrhosis: Prevalence and relation with spontaneous bacterial peritonitis. Am. J. Gastroenterol. 2001, 96, 2962–2967. [Google Scholar] [CrossRef] [PubMed]
- Frost, F.; Kacprowski, T.; Rühlemann, M.C.; Bülow, R.; Kühn, J.-P.; Franke, A.; Heinsen, F.-A.; Pietzner, M.; Nauck, M.; Volker, U.; et al. Impaired Exocrine Pancreatic Function Associates With Changes in Intestinal Microbiota Composition and Diversity. Gastroenterology 2019, 156, 1010–1015. [Google Scholar] [CrossRef] [PubMed]
- Bajaj, J.S.; Heuman, U.M.; Hylemon, P.B.; Sanyal, A.J.; White, M.B.; Monteith, P.; Noble, N.A.; Unser, A.B.; Daita, K.; Fisher, A.R.; et al. Altered profile of human gut microbiome is associated with cirrhosis and its complications. J. Hepatol. 2013, 60, 940–947. [Google Scholar] [CrossRef] [PubMed]
- Moriwaki, H. Protein-energy malnutrition in liver cirrhosis. J. Gastroenterol. 2002, 37, 578–579. [Google Scholar] [CrossRef]
- Nishikawa, H.; Enomoto, H.; Iwata, Y.; Kishino, K.; Shimono, Y.; Hasegawa, K.; Nakano, C.; Takata, R.; Ishii, A.; Nishimura, T.; et al. Prognostic significance of nonprotein respiratory quotient in patients with liver cirrhosis. Medicine 2017, 96, e5800. [Google Scholar] [CrossRef]
- Valentini, L.; Gläser, S.; Schuetz, T.; Omar, A.; Kasim, E.; Kroencke, T.J.; Tietge, U.J.; Lochs, H.; Schulzke, J.-D.; Brabant, G.; et al. Serum bile acids and leptin interact with glucose metabolism in patients with liver cirrhosis. Clin. Nutr. 2013, 32, 122–129. [Google Scholar] [CrossRef]
- Košuta, I.; Mrzljak, A.; Kolarić, B.; Lovrencic, M.V. Leptin as a Key Player in Insulin Resistance of Liver Cirrhosis? A Cross-Sectional Study in Liver Transplant Candidates. J. Clin. Med. 2020, 9, 560. [Google Scholar] [CrossRef]
- Changani, K.K.; Jalan, R.; Cox, I.J.; Ala-Korpela, M.; Bhakoo, K.; Taylor-Robinson, S.; Bell, J. Evidence for altered hepatic gluconeogenesis in patients with cirrhosis using in vivo 31-phosphorus magnetic resonance spectroscopy. Gut 2001, 49, 557–564. [Google Scholar] [CrossRef]
- Swart, G.R.; Berg, J.W.O.V.D.; Wattimena, J.L.D.; Rietveld, T.; Van Vuure, J.K.; Frenkel, M. Elevated protein requirements in cirrhosis of the liver investigated by whole body protein turnover studies. Clin. Sci. 1988, 75, 101–107. [Google Scholar] [CrossRef]
- Park, J.G.; Tak, W.Y.; Park, S.Y.; Kweon, Y.O.; Jang, S.Y.; Lee, Y.R.; Bae, S.H.; Jang, J.Y.; Kim, D.Y.; Lee, J.S.; et al. Effects of branched-chain amino acids (BCAAs) on the progression of advanced liver disease. Medicine 2017, 96, e6580. [Google Scholar] [CrossRef] [PubMed]
- Grungreiff, K.; Reinhold, D.; Wedemeyer, H. The role of zinc in liver cirrhosis. Ann. Hepatol. 2016, 15, 7–16. [Google Scholar] [CrossRef]
- Kuntz, E.; Kuntz, H.-D. Clinical aspects of liver disease. In Hepatology, Principles and Practice: History, Morphology, Biochemistry, Diagnostics, Clinic, Therapy; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2006; pp. 716–745. [Google Scholar]
- Müller, M.J.; Böttcher, J.; Selberg, O.; Weselmann, S.; Böker, K.H.; Schwarze, M.; Mühlen, A.V.Z.; Manns, M.P. Hypermetabolism in clinically stable patients with liver cirrhosis. Am. J. Clin. Nutr. 1999, 69, 1194–1201. [Google Scholar] [CrossRef] [PubMed]
- Ehlers, L.; Bannert, K.; Rohde, S.; Berlin, P.; Reiner, J.; Wiese, M.; Doller, J.; Lerch, M.M.; Aghdassi, A.A.; Meyer, F.; et al. Preclinical insights into the gut-skeletal muscle axis in chronic gastrointestinal diseases. J. Cell. Mol. Med. 2020. [Google Scholar] [CrossRef]
- Butterworth, R.F. L-Ornithine L-Aspartate for the Treatment of Sarcopenia in Chronic Liver Disease: The Taming of a Vicious Cycle. Can. J. Gastroenterol. Hepatol. 2019, 2019, 8182195-6. [Google Scholar] [CrossRef] [PubMed]
- Qiu, J.; Thapaliya, S.; Runkana, A.; Yang, Y.; Tsien, C.; Mohan, M.L.; Narayanan, A.; Eghtesad, B.; Mozdziak, P.; McDonald, C.; et al. Hyperammonemia in cirrhosis induces transcriptional regulation of myostatin by an NF- B-mediated mechanism. Proc. Natl. Acad. Sci. USA 2013, 110, 18162–18167. [Google Scholar] [CrossRef]
- Dasarathy, S.; Mookerjee, R.; Rackayova, V.; Thrane, V.R.; Vairappan, B.; Ott, P.; Rose, C. Ammonia toxicity: From head to toe? Metab. Brain Dis. 2016, 32, 529–538. [Google Scholar] [CrossRef]
- McPherron, A.C.; Lawler, A.M.; Lee, S.-J. Regulation of skeletal muscle mass in mice by a new TGF-p superfamily member. Nature 1997, 387, 83–90. [Google Scholar] [CrossRef]
- Dasarathy, S.; Hatzoglou, M. Hyperammonemia and proteostasis in cirrhosis. Curr. Opin. Clin. Nutr. Metab. Care 2018, 21, 30–36. [Google Scholar] [CrossRef]
- Bhanji, R.; Moctezuma-Velazquez, C.; Duarte-Rojo, A.; Ebadi, M.; Ghosh, S.; Rose, C.F.; Montano-Loza, A.J. Myosteatosis and sarcopenia are associated with hepatic encephalopathy in patients with cirrhosis. Hepatol. Int. 2018, 12, 377–386. [Google Scholar] [CrossRef] [PubMed]
- Bémeur, C.; Rose, C.F. Hepatic Encephalopathy, Sarcopenia, and Frailty. In Frailty and Sarcopenia in Cirrhosis; Tandon, P., Montano-Loza, A.J., Eds.; Springer: Cham, Switzerland, 2020; pp. 247–263. [Google Scholar]
- Hey, P.; Sinclair, M. Upcoming Pharmacological and Interventional Therapies for the Treatment of Physical Frailty and Sarcopenia. In Frailty and Sarcopenia in Cirrhosis; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2019; pp. 211–232. [Google Scholar]
- Sinclair, M.; Grossmann, M.; Hoermann, R.; Angus, P.W.; Gow, P.J. Testosterone therapy increases muscle mass in men with cirrhosis and low testosterone: A randomised controlled trial. J. Hepatol. 2016, 65, 906–913. [Google Scholar] [CrossRef]
- Conchillo, M.; De Knegt, R.J.; Payeras, M.; Quiroga, J.; Sangro, B.; Herrero, J.-I.; Castilla-Cortazar, I.; Frystyk, J.; Flyvbjerg, A.; Yoshizawa, C.; et al. Insulin-like growth factor I (IGF-I) replacement therapy increases albumin concentration in liver cirrhosis: Results of a pilot randomized controlled clinical trial. J. Hepatol. 2005, 43, 630–636. [Google Scholar] [CrossRef] [PubMed]
- Mitch, W.E. Mechanisms accelerating muscle atrophy in catabolic diseases. Trans. Am. Clin. Clim. Assoc. 2000, 111, 258–270. [Google Scholar]
- Campos, F.; Abrigo, J.; Aguirre, F.; Garcés, B.; Arrese, M.; Karpen, S.; Cabrera, D.; Andia, M.E.; Simon, F.; Cabello-Verrugio, C. Sarcopenia in a mice model of chronic liver disease: Role of the ubiquitin–proteasome system and oxidative stress. Pflügers Archiv. 2018, 470, 1503–1519. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Yu, Y.; Wang, J. Protein Nutritional Support: The Classical and Potential New Mechanisms in the Prevention and Therapy of Sarcopenia. J. Agric. Food Chem. 2020, 68, 4098–4108. [Google Scholar] [CrossRef]
- Qiu, J.; Tsien, C.; Thapalaya, S.; Narayanan, A.; Weihl, C.C.; Ching, J.K.; Eghtesad, B.; Singh, K.; Fu, X.; Dubyak, G.R.; et al. Hyperammonemia-mediated autophagy in skeletal muscle contributes to sarcopenia of cirrhosis. Am. J. Physiol. Metab. 2012, 303, E983–E993. [Google Scholar] [CrossRef]
- Les, I.; Doval, E.; García-Martínez, R.; Planas, M.; Cárdenas, G.; Gómez, P.; Flavià, M.; Jacas, C.; Mínguez, B.; Vergara, M.; et al. Effects of Branched-Chain Amino Acids Supplementation in Patients With Cirrhosis and a Previous Episode of Hepatic Encephalopathy: A Randomized Study. Am. J. Gastroenterol. 2011, 106, 1081–1088. [Google Scholar] [CrossRef]
- Tsien, C.; Davuluri, G.; Singh, D.; Allawy, A.; Have, G.A.T.; Thapaliya, S.; Schulze, J.M.; Barnes, D.; McCullough, A.J.; Engelen, M.P.; et al. Metabolic and molecular responses to leucine-enriched branched chain amino acid supplementation in the skeletal muscle of alcoholic cirrhosis. Hepatology 2015, 61, 2018–2029. [Google Scholar] [CrossRef]
- Merli, M.; Berzigotti, A.; Zelber-Sagi, S.; Dasarathy, S.; Montagnese, S.; Genton, L.; Plauth, M.; Parés, A. EASL Clinical Practice Guidelines on nutrition in chronic liver disease. J. Hepatol. 2019, 70, 172–193. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meyer, F.; Bannert, K.; Wiese, M.; Esau, S.; Sautter, L.F.; Ehlers, L.; Aghdassi, A.A.; Metges, C.C.; Garbe, L.-A.; Jaster, R.; et al. Molecular Mechanism Contributing to Malnutrition and Sarcopenia in Patients with Liver Cirrhosis. Int. J. Mol. Sci. 2020, 21, 5357. https://doi.org/10.3390/ijms21155357
Meyer F, Bannert K, Wiese M, Esau S, Sautter LF, Ehlers L, Aghdassi AA, Metges CC, Garbe L-A, Jaster R, et al. Molecular Mechanism Contributing to Malnutrition and Sarcopenia in Patients with Liver Cirrhosis. International Journal of Molecular Sciences. 2020; 21(15):5357. https://doi.org/10.3390/ijms21155357
Chicago/Turabian StyleMeyer, Fatuma, Karen Bannert, Mats Wiese, Susanne Esau, Lea F. Sautter, Luise Ehlers, Ali A. Aghdassi, Cornelia C. Metges, Leif-A. Garbe, Robert Jaster, and et al. 2020. "Molecular Mechanism Contributing to Malnutrition and Sarcopenia in Patients with Liver Cirrhosis" International Journal of Molecular Sciences 21, no. 15: 5357. https://doi.org/10.3390/ijms21155357
APA StyleMeyer, F., Bannert, K., Wiese, M., Esau, S., Sautter, L. F., Ehlers, L., Aghdassi, A. A., Metges, C. C., Garbe, L.-A., Jaster, R., Lerch, M. M., Lamprecht, G., & Valentini, L. (2020). Molecular Mechanism Contributing to Malnutrition and Sarcopenia in Patients with Liver Cirrhosis. International Journal of Molecular Sciences, 21(15), 5357. https://doi.org/10.3390/ijms21155357




