VEGF-A in Cardiomyocytes and Heart Diseases
Abstract
1. Vascular Endothelial Growth Factor
2. Cardiomyocytes
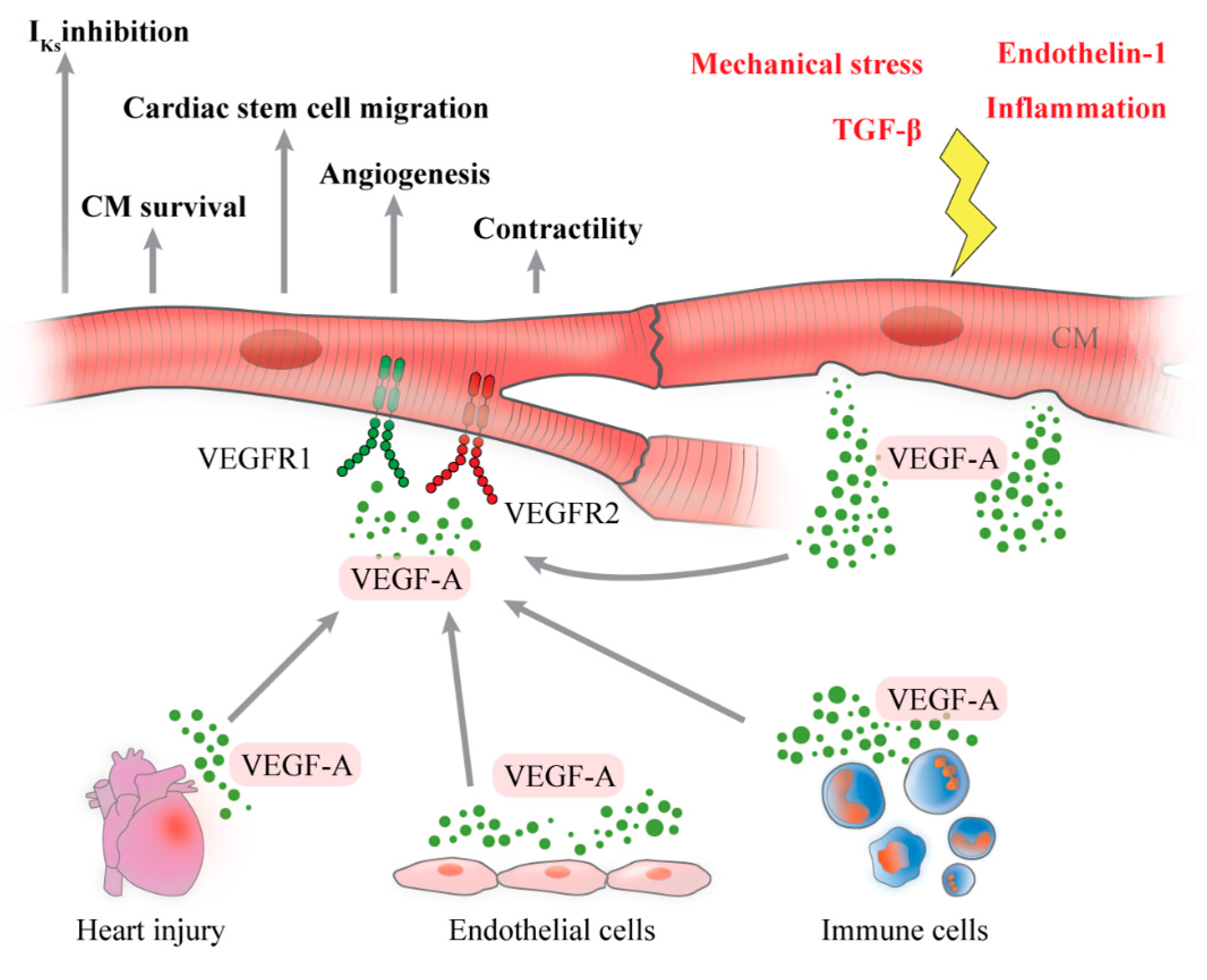
3. Cardiomyocytes as Producers of VEGF-A
4. Cardiomyocytes as Target of VEGF-A
5. VEGF-A and Angiogenesis in Cardiovascular Diseases
5.1. Ischemic Heart Disease
5.2. Atherosclerosis
6. Conclusions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ANGPT | angiopoietin |
| ATE | arterial thromboembolic events |
| CM | cardiomyocytes |
| CVD | cardiovascular disease |
| CAD | coronary artery disease |
| EC | endothelial cells |
| ECM | extracellular matrix |
| IL- | interleukin |
| IHD | ischemic heart disease |
| MI | myocardial infarction |
| NO | nitric oxide |
| sVEGF-A | soluble vascular endothelial growth factor receptor |
| TGF-β | transforming growth factor β |
| TNF | tumor necrosis factor |
| VEGF | vascular endothelial growth |
| VSP | VEGF-A signaling pathway |
| VEGFR | vascular endothelial growth factor receptor |
References
- Senger, D.R.; Galli, S.J.; Dvorak, A.M.; Perruzzi, C.A.; Harvey, V.S.; Dvorak, H.F. Tumor cells secrete a vascular permeability factor that promotes accumulation of ascites fluid. Science 1983, 219, 983–985. [Google Scholar] [CrossRef]
- Ferrara, N.; Gerber, H.-P.; LeCouter, J. The biology of VEGF and its receptors. Nat. Med. 2003, 9, 669–676. [Google Scholar] [CrossRef]
- Ferrara, N.; Davis-Smyth, T. The Biology of Vascular Endothelial Growth Factor. Endocr. Rev. 1997, 18, 4–25. [Google Scholar] [CrossRef]
- Moens, S.; Goveia, J.; Stapor, P.C.; Cantelmo, A.R.; Carmeliet, P. The multifaceted activity of VEGF in angiogenesis—Implications for therapy responses. Cytokine Growth Factor Rev. 2014, 25, 473–482. [Google Scholar] [CrossRef]
- Varricchi, G.; Loffredo, S.; Galdiero, M.R.; Marone, G.; Cristinziano, L.; Granata, F.; Marone, G. Innate effector cells in angiogenesis and lymphangiogenesis. Curr. Opin. Immunol. 2018, 53, 152–160. [Google Scholar] [CrossRef] [PubMed]
- Koch, A.E.; Harlow, L.A.; Haines, G.K.; Amento, E.P.; Unemori, E.N.; Wong, W.L.; Pope, R.M.; Ferrara, N. Vascular endothelial growth factor. A cytokine modulating endothelial function in rheumatoid arthritis. J. Immunol. Baltim. Md 1950 1994, 152, 4149–4156. [Google Scholar]
- Mandriota, S.J.; Seghezzi, G.; Vassalli, J.-D.; Ferrara, N.; Wasi, S.; Mazzieri, R.; Mignatti, P.; Pepper, M.S. Vascular Endothelial Growth Factor Increases Urokinase Receptor Expression in Vascular Endothelial Cells. J. Biol. Chem. 1995, 270, 9709–9716. [Google Scholar] [CrossRef] [PubMed]
- Unemori, E.N.; Ferrara, N.; Bauer, E.A.; Amento, E.P. Vascular endothelial growth factor induces interstitial collagenase expression in human endothelial cells. J. Cell. Physiol. 1992, 153, 557–562. [Google Scholar] [CrossRef]
- Hu, G.-J.; Feng, Y.-G.; Lu, W.-P.; Li, H.-T.; Xie, H.-W.; Li, S.-F. Effect of combined VEGF165/ SDF-1 gene therapy on vascular remodeling and blood perfusion in cerebral ischemia. J. Neurosurg. 2017, 127, 670–678. [Google Scholar] [CrossRef]
- Kazi, A.S.; Lotfi, S.; Goncharova, E.A.; Tliba, O.; Amrani, Y.; Krymskaya, V.P.; Lazaar, A.L. Vascular endothelial growth factor-induced secretion of fibronectin is ERK dependent. Am. J. Physiol. Lung Cell. Mol. Physiol. 2004, 286, L539–L545. [Google Scholar] [CrossRef]
- Morales-Ruiz, M.; Fulton, D.; Sowa, G.; Languino, L.R.; Fujio, Y.; Walsh, K.; Sessa, W.C. Vascular endothelial growth factor-stimulated actin reorganization and migration of endothelial cells is regulated via the serine/threonine kinase Akt. Circ. Res. 2000, 86, 892–896. [Google Scholar] [CrossRef] [PubMed]
- Lundkvist, A.; Lee, S.; Iruela-Arispe, L.; Betsholtz, C.; Gerhardt, H. Growth factor gradients in vascular patterning. Novartis Found. Symp. 2007, 283, 194–201; discussion 201. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Jilani, S.M.; Nikolova, G.V.; Carpizo, D.; Iruela-Arispe, M.L. Processing of VEGF-A by matrix metalloproteinases regulates bioavailability and vascular patterning in tumors. J. Cell Biol. 2005, 169, 681–691. [Google Scholar] [CrossRef] [PubMed]
- Vempati, P.; Popel, A.S.; Mac Gabhann, F. Extracellular regulation of VEGF: Isoforms, proteolysis, and vascular patterning. Cytokine Growth Factor Rev. 2014, 25, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Tischer, E.; Mitchell, R.; Hartman, T.; Silva, M.; Gospodarowicz, D.; Fiddes, J.C.; Abraham, J.A. The human gene for vascular endothelial growth factor. Multiple protein forms are encoded through alternative exon splicing. J. Biol. Chem. 1991, 266, 11947–11954. [Google Scholar]
- Venables, J.P. Unbalanced alternative splicing and its significance in cancer. BioEssays News Rev. Mol. Cell. Dev. Biol. 2006, 28, 378–386. [Google Scholar] [CrossRef]
- Azimi-Nezhad, M. Vascular endothelial growth factor from embryonic status to cardiovascular pathology. Rep. Biochem. Mol. Biol. 2014, 2, 59–69. [Google Scholar]
- Ferrara, N. Role of vascular endothelial growth factor in regulation of physiological angiogenesis. Am. J. Physiol. Cell Physiol. 2001, 280, C1358–C1366. [Google Scholar] [CrossRef]
- Gorenjak, V.; Vance, D.R.; Petrelis, A.M.; Stathopoulou, M.G.; Dadé, S.; El Shamieh, S.; Murray, H.; Masson, C.; Lamont, J.; Fitzgerald, P.; et al. Peripheral blood mononuclear cells extracts VEGF protein levels and VEGF mRNA: Associations with inflammatory molecules in a healthy population. PLoS ONE 2019, 14, e0220902. [Google Scholar] [CrossRef]
- Peach, C.J.; Mignone, V.W.; Arruda, M.A.; Alcobia, D.C.; Hill, S.J.; Kilpatrick, L.E.; Woolard, J. Molecular Pharmacology of VEGF-A Isoforms: Binding and Signalling at VEGFR2. Int. J. Mol. Sci. 2018, 19, 1264. [Google Scholar] [CrossRef]
- Houck, K.A.; Leung, D.W.; Rowland, A.M.; Winer, J.; Ferrara, N. Dual regulation of vascular endothelial growth factor bioavailability by genetic and proteolytic mechanisms. J. Biol. Chem. 1992, 267, 26031–26037. [Google Scholar] [PubMed]
- Park, J.E.; Keller, G.A.; Ferrara, N. The vascular endothelial growth factor (VEGF) isoforms: Differential deposition into the subepithelial extracellular matrix and bioactivity of extracellular matrix-bound VEGF. Mol. Biol. Cell 1993, 4, 1317–1326. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Bao, L.; Zhao, M.; Cao, J.; Zheng, H. Progress in Research on the Role of FGF in the Formation and Treatment of Corneal Neovascularization. Front. Pharmacol. 2020, 11, 111. [Google Scholar] [CrossRef] [PubMed]
- Ferrara, N. Vascular endothelial growth factor as a target for anticancer therapy. The Oncologist 2004, 9 (Suppl. 1), 2–10. [Google Scholar] [CrossRef]
- Nicolas, S.; Abdellatef, S.; Haddad, M.A.; Fakhoury, I.; El-Sibai, M. Hypoxia and EGF Stimulation Regulate VEGF Expression in Human Glioblastoma Multiforme (GBM) Cells by Differential Regulation of the PI3K/Rho-GTPase and MAPK Pathways. Cells 2019, 8, 1397. [Google Scholar] [CrossRef] [PubMed]
- Saryeddine, L.; Zibara, K.; Kassem, N.; Badran, B.; El-Zein, N. EGF-Induced VEGF Exerts a PI3K-Dependent Positive Feedback on ERK and AKT through VEGFR2 in Hematological In Vitro Models. PLoS ONE 2016, 11, e0165876. [Google Scholar] [CrossRef] [PubMed]
- Seghezzi, G.; Patel, S.; Ren, C.J.; Gualandris, A.; Pintucci, G.; Robbins, E.S.; Shapiro, R.L.; Galloway, A.C.; Rifkin, D.B.; Mignatti, P. Fibroblast growth factor-2 (FGF-2) induces vascular endothelial growth factor (VEGF) expression in the endothelial cells of forming capillaries: An autocrine mechanism contributing to angiogenesis. J. Cell Biol. 1998, 141, 1659–1673. [Google Scholar] [CrossRef]
- Wang, H.; Han, X.; Wittchen, E.S.; Hartnett, M.E. TNF-α mediates choroidal neovascularization by upregulating VEGF expression in RPE through ROS-dependent β-catenin activation. Mol. Vis. 2016, 22, 116–128. [Google Scholar]
- Ruhrberg, C.; Gerhardt, H.; Golding, M.; Watson, R.; Ioannidou, S.; Fujisawa, H.; Betsholtz, C.; Shima, D.T. Spatially restricted patterning cues provided by heparin-binding VEGF-A control blood vessel branching morphogenesis. Genes Dev. 2002, 16, 2684–2698. [Google Scholar] [CrossRef]
- Stalmans, I.; Ng, Y.-S.; Rohan, R.; Fruttiger, M.; Bouché, A.; Yuce, A.; Fujisawa, H.; Hermans, B.; Shani, M.; Jansen, S.; et al. Arteriolar and venular patterning in retinas of mice selectively expressing VEGF isoforms. J. Clin. Investig. 2002, 109, 327–336. [Google Scholar] [CrossRef]
- Koch, S.; Tugues, S.; Li, X.; Gualandi, L.; Claesson-Welsh, L. Signal transduction by vascular endothelial growth factor receptors. Biochem. J. 2011, 437, 169–183. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.A.; Fearnley, G.W.; Tomlinson, D.C.; Harrison, M.A.; Ponnambalam, S. The cellular response to vascular endothelial growth factors requires co-ordinated signal transduction, trafficking and proteolysis. Biosci. Rep. 2015, 35. [Google Scholar] [CrossRef] [PubMed]
- Alexander, S.P.; Fabbro, D.; Kelly, E.; Marrion, N.; Peters, J.A.; Benson, H.E.; Faccenda, E.; Pawson, A.J.; Sharman, J.L.; Southan, C.; et al. The Concise Guide to PHARMACOLOGY 2015/16: Catalytic receptors. Br. J. Pharmacol. 2015, 172, 5979–6023. [Google Scholar] [CrossRef] [PubMed]
- Detoraki, A.; Staiano, R.I.; Granata, F.; Giannattasio, G.; Prevete, N.; de Paulis, A.; Ribatti, D.; Genovese, A.; Triggiani, M.; Marone, G. Vascular endothelial growth factors synthesized by human lung mast cells exert angiogenic effects. J. Allergy Clin. Immunol. 2009, 123, 1142–1149.e5. [Google Scholar] [CrossRef] [PubMed]
- Ishida, A.; Murray, J.; Saito, Y.; Kanthou, C.; Benzakour, O.; Shibuya, M.; Wijelath, E.S. Expression of vascular endothelial growth factor receptors in smooth muscle cells. J. Cell. Physiol. 2001, 188, 359–368. [Google Scholar] [CrossRef] [PubMed]
- Kabrun, N.; Bühring, H.J.; Choi, K.; Ullrich, A.; Risau, W.; Keller, G. Flk-1 expression defines a population of early embryonic hematopoietic precursors. Dev. Camb. Engl. 1997, 124, 2039–2048. [Google Scholar]
- Witmer, A.N.; Dai, J.; Weich, H.A.; Vrensen, G.F.J.M.; Schlingemann, R.O. Expression of vascular endothelial growth factor receptors 1, 2, and 3 in quiescent endothelia. J. Histochem. Cytochem. Off. J. Histochem. Soc. 2002, 50, 767–777. [Google Scholar] [CrossRef]
- Meyer, R.D.; Mohammadi, M.; Rahimi, N. A single amino acid substitution in the activation loop defines the decoy characteristic of VEGFR-1/FLT-1. J. Biol. Chem. 2006, 281, 867–875. [Google Scholar] [CrossRef]
- Terman, B.I.; Dougher-Vermazen, M.; Carrion, M.E.; Dimitrov, D.; Armellino, D.C.; Gospodarowicz, D.; Böhlen, P. Identification of the KDR tyrosine kinase as a receptor for vascular endothelial cell growth factor. Biochem. Biophys. Res. Commun. 1992, 187, 1579–1586. [Google Scholar] [CrossRef]
- De Vries, C.; Escobedo, J.A.; Ueno, H.; Houck, K.; Ferrara, N.; Williams, L.T. The fms-like tyrosine kinase, a receptor for vascular endothelial growth factor. Science 1992, 255, 989–991. [Google Scholar] [CrossRef]
- Waltenberger, J.; Claesson-Welsh, L.; Siegbahn, A.; Shibuya, M.; Heldin, C.H. Different signal transduction properties of KDR and Flt1, two receptors for vascular endothelial growth factor. J. Biol. Chem. 1994, 269, 26988–26995. [Google Scholar] [PubMed]
- Hornig, C.; Weich, H.A. Soluble VEGF receptors. Angiogenesis 1999, 3, 33–39. [Google Scholar] [CrossRef]
- Shibuya, M.; Claesson-Welsh, L. Signal transduction by VEGF receptors in regulation of angiogenesis and lymphangiogenesis. Exp. Cell Res. 2006, 312, 549–560. [Google Scholar] [CrossRef] [PubMed]
- Woodcock, E.A.; Matkovich, S.J. Cardiomyocytes structure, function and associated pathologies. Int. J. Biochem. Cell Biol. 2005, 37, 1746–1751. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, A.J.S.; Ang, Y.-S.; Fu, J.-D.; Rivas, R.N.; Mohamed, T.M.A.; Higgs, G.C.; Srivastava, D.; Pruitt, B.L. Contractility of single cardiomyocytes differentiated from pluripotent stem cells depends on physiological shape and substrate stiffness. Proc. Natl. Acad. Sci. USA 2015, 112, 12705–12710. [Google Scholar] [CrossRef] [PubMed]
- Pittman, R.N. Regulation of Tissue Oxygenation; Integrated Systems Physiology: From Molecule to Function to Disease; Morgan & Claypool Life Sciences: San Rafael, CA, USA, 2011. [Google Scholar]
- Walker, C.A.; Spinale, F.G. The structure and function of the cardiac myocyte: A review of fundamental concepts. J. Thorac. Cardiovasc. Surg. 1999, 118, 375–382. [Google Scholar] [CrossRef]
- Frank, J.S.; Langer, G.A.; Nudd, L.M.; Seraydarian, K. The myocardial cell surface, its histochemistry, and the effect of sialic acid and calcium removal on its stucture and cellular ionic exchange. Circ. Res. 1977, 41, 702–714. [Google Scholar] [CrossRef]
- Frank, J.S.; Langer, G.A. The myocardial interstitium: Its structure and its role in ionic exchange. J. Cell Biol. 1974, 60, 586–601. [Google Scholar] [CrossRef]
- Yang, H.; Borg, T.K.; Wang, Z.; Ma, Z.; Gao, B.Z. Role of the Basement Membrane in Regulation of Cardiac Electrical Properties. Ann. Biomed. Eng. 2014, 42, 1148–1157. [Google Scholar] [CrossRef]
- Langer, G.A.; Frank, J.S.; Rich, T.L.; Orner, F.B. Calcium exchange, structure, and function in cultured adult myocardial cells. Am. J. Physiol. 1987, 252, H314–H324. [Google Scholar] [CrossRef]
- Langer, G.A.; Frank, J.S.; Philipson, K.D. Ultrastructure and calcium exchange of the sarcolemma, sarcoplasmic reticulum and mitochondria of the myocardium. Pharmacol. Ther. 1982, 16, 331–376. [Google Scholar] [CrossRef]
- Hong, T.; Shaw, R.M. Cardiac T-Tubule Microanatomy and Function. Physiol. Rev. 2017, 97, 227–252. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Qiu, Y.; Zhang, H.M.; Yang, D. Intercalated discs: Cellular adhesion and signaling in heart health and diseases. Heart Fail. Rev. 2019, 24, 115–132. [Google Scholar] [CrossRef] [PubMed]
- McCain, M.L.; Parker, K.K. Mechanotransduction: The role of mechanical stress, myocyte shape, and cytoskeletal architecture on cardiac function. Pflüg. Arch.- Eur. J. Physiol. 2011, 462, 89–104. [Google Scholar] [CrossRef]
- Guhathakurta, P.; Prochniewicz, E.; Thomas, D.D. Actin-Myosin Interaction: Structure, Function and Drug Discovery. Int. J. Mol. Sci. 2018, 19, 2628. [Google Scholar] [CrossRef]
- Powers, J.D.; Yuan, C.-C.; McCabe, K.J.; Murray, J.D.; Childers, M.C.; Flint, G.V.; Moussavi-Harami, F.; Mohran, S.; Castillo, R.; Zuzek, C.; et al. Cardiac myosin activation with 2-deoxy-ATP via increased electrostatic interactions with actin. Proc. Natl. Acad. Sci. USA 2019, 116, 11502–11507. [Google Scholar] [CrossRef]
- Yamada, Y.; Namba, K.; Fujii, T. Cardiac muscle thin filament structures reveal calcium regulatory mechanism. Nat. Commun. 2020, 11, 153. [Google Scholar] [CrossRef]
- Kuznetsov, A.V.; Javadov, S.; Grimm, M.; Margreiter, R.; Ausserlechner, M.J.; Hagenbuchner, J. Crosstalk between Mitochondria and Cytoskeleton in Cardiac Cells. Cells 2020, 9, 222. [Google Scholar] [CrossRef]
- Pohjoismäki, J.L.; Goffart, S. The role of mitochondria in cardiac development and protection. Free Radic. Biol. Med. 2017, 106, 345–354. [Google Scholar] [CrossRef]
- Solarewicz, J.; Manly, A.; Kokoszka, S.; Sleiman, N.; Leff, T.; Cala, S. Adiponectin secretion from cardiomyocytes produces canonical multimers and partial co-localization with calsequestrin in junctional SR. Mol. Cell. Biochem. 2019, 457, 201–214. [Google Scholar] [CrossRef]
- Frangogiannis, N.G. The Reparative Function of Cardiomyocytes in the Infarcted Myocardium. Cell Metab. 2015, 21, 797–798. [Google Scholar] [CrossRef] [PubMed]
- Estrada, D.; Specker, G.; Martínez, A.; Dias, P.P.; Hissa, B.; Andrade, L.O.; Radi, R.; Piacenza, L. Cardiomyocyte diffusible redox mediators control Trypanosoma cruzi infection: Role of parasite mitochondrial iron superoxide dismutase. Biochem. J. 2018, 475, 1235–1251. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Zhang, X.; He, Z.; Wu, Z.; Rao, L.; Li, Y. Metabolites of Hypoxic Cardiomyocytes Induce the Migration of Cardiac Fibroblasts. Cell. Physiol. Biochem. 2017, 41, 413–421. [Google Scholar] [CrossRef] [PubMed]
- Ancey, C.; Corbi, P.; Froger, J.; Delwail, A.; Wijdenes, J.; Gascan, H.; Potreau, D.; Lecron, J.-C. Secretion of IL-6, IL-11 and lif by human cardiomyocytes in primary culture. Cytokine 2002, 18, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Aoyagi, T.; Matsui, T. The Cardiomyocyte as a Source of Cytokines in Cardiac Injury. J. Cell Sci. Ther. 2011, 2012. [Google Scholar] [CrossRef]
- Hsieh, P.C.H.; Davis, M.E.; Lisowski, L.K.; Lee, R.T. Endothelial-cardiomyocyte interactions in cardiac development and repair. Annu. Rev. Physiol. 2006, 68, 51–66. [Google Scholar] [CrossRef]
- Ziegler, T.; Horstkotte, J.; Schwab, C.; Pfetsch, V.; Weinmann, K.; Dietzel, S.; Rohwedder, I.; Hinkel, R.; Gross, L.; Lee, S.; et al. Angiopoietin 2 mediates microvascular and hemodynamic alterations in sepsis. J. Clin. Investig. 2013. [Google Scholar] [CrossRef]
- Kuwabara, M.; Kakinuma, Y.; Ando, M.; Katare, R.G.; Yamasaki, F.; Doi, Y.; Sato, T. Nitric Oxide Stimulates Vascular Endothelial Growth Factor Production in Cardiomyocytes Involved in Angiogenesis. J. Physiol. Sci. 2006, 56, 95–101. [Google Scholar] [CrossRef]
- Abhinand, C.S.; Raju, R.; Soumya, S.J.; Arya, P.S.; Sudhakaran, P.R. VEGF-A/VEGFR2 signaling network in endothelial cells relevant to angiogenesis. J. Cell Commun. Signal. 2016, 10, 347–354. [Google Scholar] [CrossRef]
- Giordano, F.J.; Gerber, H.P.; Williams, S.P.; VanBruggen, N.; Bunting, S.; Ruiz-Lozano, P.; Gu, Y.; Nath, A.K.; Huang, Y.; Hickey, R.; et al. A cardiac myocyte vascular endothelial growth factor paracrine pathway is required to maintain cardiac function. Proc. Natl. Acad. Sci. USA 2001, 98, 5780–5785. [Google Scholar] [CrossRef]
- Smart, N.; Dubé, K.N.; Riley, P.R. Coronary vessel development and insight towards neovascular therapy. Int. J. Exp. Pathol. 2009, 90, 262–283. [Google Scholar] [CrossRef] [PubMed]
- Carmeliet, P.; Ng, Y.S.; Nuyens, D.; Theilmeier, G.; Brusselmans, K.; Cornelissen, I.; Ehler, E.; Kakkar, V.V.; Stalmans, I.; Mattot, V.; et al. Impaired myocardial angiogenesis and ischemic cardiomyopathy in mice lacking the vascular endothelial growth factor isoforms VEGF164 and VEGF188. Nat. Med. 1999, 5, 495–502. [Google Scholar] [CrossRef] [PubMed]
- Dor, Y.; Camenisch, T.D.; Itin, A.; Fishman, G.I.; McDonald, J.A.; Carmeliet, P.; Keshet, E. A novel role for VEGF in endocardial cushion formation and its potential contribution to congenital heart defects. Dev. Camb. Engl. 2001, 128, 1531–1538. [Google Scholar]
- Dor, Y.; Klewer, S.E.; McDonald, J.A.; Keshet, E.; Camenisch, T.D. VEGF modulates early heart valve formation. Anat. Rec. A. Discov. Mol. Cell. Evol. Biol. 2003, 271, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Miquerol, L.; Langille, B.L.; Nagy, A. Embryonic development is disrupted by modest increases in vascular endothelial growth factor gene expression. Dev. Camb. Engl. 2000, 127, 3941–3946. [Google Scholar]
- Lambrechts, D.; Carmeliet, P. Sculpting heart valves with NFATc and VEGF. Cell 2004, 118, 532–534. [Google Scholar] [CrossRef][Green Version]
- Chang, C.-P.; Neilson, J.R.; Bayle, J.H.; Gestwicki, J.E.; Kuo, A.; Stankunas, K.; Graef, I.A.; Crabtree, G.R. A field of myocardial-endocardial NFAT signaling underlies heart valve morphogenesis. Cell 2004, 118, 649–663. [Google Scholar] [CrossRef]
- Seko, Y.; Seko, Y.; Takahashi, N.; Shibuya, M.; Yazaki, Y. Pulsatile stretch stimulates vascular endothelial growth factor (VEGF) secretion by cultured rat cardiac myocytes. Biochem. Biophys. Res. Commun. 1999, 254, 462–465. [Google Scholar] [CrossRef]
- Semenza, G.L. Hypoxia-inducible factor 1 and cardiovascular disease. Annu. Rev. Physiol. 2014, 76, 39–56. [Google Scholar] [CrossRef]
- Shen, J.; Xie, Y.; Liu, Z.; Zhang, S.; Wang, Y.; Jia, L.; Wang, Y.; Cai, Z.; Ma, H.; Xiang, M. Increased myocardial stiffness activates cardiac microvascular endothelial cell via VEGF paracrine signaling in cardiac hypertrophy. J. Mol. Cell. Cardiol. 2018, 122, 140–151. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, A.; Hou, R.; Zhang, J.; Jia, X.; Jiang, W.; Chen, J. Salidroside protects cardiomyocyte against hypoxia-induced death: A HIF-1α-activated and VEGF-mediated pathway. Eur. J. Pharmacol. 2009, 607, 6–14. [Google Scholar] [CrossRef] [PubMed]
- Leychenko, A.; Konorev, E.; Jijiwa, M.; Matter, M.L. Stretch-induced hypertrophy activates NFkB-mediated VEGF secretion in adult cardiomyocytes. PLoS ONE 2011, 6, e29055. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Hampton, T.; Morgan, J.P.; Simons, M. Stretch-induced VEGF expression in the heart. J. Clin. Investig. 1997, 100, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Shimojo, N.; Jesmin, S.; Zaedi, S.; Otsuki, T.; Maeda, S.; Yamaguchi, N.; Aonuma, K.; Hattori, Y.; Miyauchi, T. Contributory role of VEGF overexpression in endothelin-1-induced cardiomyocyte hypertrophy. Am. J. Physiol. Heart Circ. Physiol. 2007, 293, H474–H481. [Google Scholar] [CrossRef] [PubMed]
- Rysä, J.; Tenhunen, O.; Serpi, R.; Soini, Y.; Nemer, M.; Leskinen, H.; Ruskoaho, H. GATA-4 is an angiogenic survival factor of the infarcted heart. Circ. Heart Fail. 2010, 3, 440–450. [Google Scholar] [CrossRef]
- Heineke, J.; Auger-Messier, M.; Xu, J.; Oka, T.; Sargent, M.A.; York, A.; Klevitsky, R.; Vaikunth, S.; Duncan, S.A.; Aronow, B.J.; et al. Cardiomyocyte GATA4 functions as a stress-responsive regulator of angiogenesis in the murine heart. J. Clin. Investig. 2007, 117, 3198–3210. [Google Scholar] [CrossRef]
- Van Berlo, J.H.; Elrod, J.W.; van den Hoogenhof, M.M.G.; York, A.J.; Aronow, B.J.; Duncan, S.A.; Molkentin, J.D. The transcription factor GATA-6 regulates pathological cardiac hypertrophy. Circ. Res. 2010, 107, 1032–1040. [Google Scholar] [CrossRef]
- Nam, Y.S.; Kim, Y.; Joung, H.; Kwon, D.-H.; Choe, N.; Min, H.-K.; Kim, Y.S.; Kim, H.-S.; Kim, D.-K.; Cho, Y.K.; et al. Small heterodimer partner blocks cardiac hypertrophy by interfering with GATA6 signaling. Circ. Res. 2014, 115, 493–503. [Google Scholar] [CrossRef]
- Van Berlo, J.H.; Aronow, B.J.; Molkentin, J.D. Parsing the roles of the transcription factors GATA-4 and GATA-6 in the adult cardiac hypertrophic response. PLoS ONE 2013, 8, e84591. [Google Scholar] [CrossRef] [PubMed]
- Curry, J.M.; Eubank, T.D.; Roberts, R.D.; Wang, Y.; Pore, N.; Maity, A.; Marsh, C.B. M-CSF signals through the MAPK/ERK pathway via Sp1 to induce VEGF production and induces angiogenesis in vivo. PLoS ONE 2008, 3, e3405. [Google Scholar] [CrossRef]
- Lin, H.-H.; Lai, S.-C.; Chau, L.-Y. Heme oxygenase-1/carbon monoxide induces vascular endothelial growth factor expression via p38 kinase-dependent activation of Sp1. J. Biol. Chem. 2011, 286, 3829–3838. [Google Scholar] [CrossRef]
- Rose, B.A.; Yokota, T.; Chintalgattu, V.; Ren, S.; Iruela-Arispe, L.; Khakoo, A.Y.; Minamisawa, S.; Wang, Y. Cardiac myocyte p38α kinase regulates angiogenesis via myocyte-endothelial cell cross-talk during stress-induced remodeling in the heart. J. Biol. Chem. 2017, 292, 12787–12800. [Google Scholar] [CrossRef]
- Chen, Y.; Amende, I.; Hampton, T.G.; Yang, Y.; Ke, Q.; Min, J.-Y.; Xiao, Y.-F.; Morgan, J.P. Vascular endothelial growth factor promotes cardiomyocyte differentiation of embryonic stem cells. Am. J. Physiol.-Heart Circ. Physiol. 2006, 291, H1653–H1658. [Google Scholar] [CrossRef]
- Neufeld, G.; Cohen, T.; Gengrinovitch, S.; Poltorak, Z. Vascular endothelial growth factor (VEGF) and its receptors. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 1999, 13, 9–22. [Google Scholar] [CrossRef]
- Pearlman, J.D.; Hibberd, M.G.; Chuang, M.L.; Harada, K.; Lopez, J.J.; Gladstone, S.R.; Friedman, M.; Sellke, F.W.; Simons, M. Magnetic resonance mapping demonstrates benefits of VEGF-induced myocardial angiogenesis. Nat. Med. 1995, 1, 1085–1089. [Google Scholar] [CrossRef]
- Taimeh, Z.; Loughran, J.; Birks, E.J.; Bolli, R. Vascular endothelial growth factor in heart failure. Nat. Rev. Cardiol. 2013, 10, 519–530. [Google Scholar] [CrossRef]
- Tang, J.; Wang, J.; Kong, X.; Yang, J.; Guo, L.; Zheng, F.; Zhang, L.; Huang, Y.; Wan, Y. Vascular endothelial growth factor promotes cardiac stem cell migration via the PI3K/Akt pathway. Exp. Cell Res. 2009, 315, 3521–3531. [Google Scholar] [CrossRef]
- Friehs, I.; Barillas, R.; Vasilyev, N.V.; Roy, N.; McGowan, F.X.; del Nido, P.J. Vascular endothelial growth factor prevents apoptosis and preserves contractile function in hypertrophied infant heart. Circulation 2006, 114, I290–I295. [Google Scholar] [CrossRef]
- Tao, Z.; Chen, B.; Tan, X.; Zhao, Y.; Wang, L.; Zhu, T.; Cao, K.; Yang, Z.; Kan, Y.W.; Su, H. Coexpression of VEGF and angiopoietin-1 promotes angiogenesis and cardiomyocyte proliferation reduces apoptosis in porcine myocardial infarction (MI) heart. Proc. Natl. Acad. Sci. USA 2011, 108, 2064–2069. [Google Scholar] [CrossRef]
- Conway, E. Molecular mechanisms of blood vessel growth. Cardiovasc. Res. 2001, 49, 507–521. [Google Scholar] [CrossRef]
- Borgers, M.; Voipio-Pulkki, L.-M.; Izumo, S. Apoptosis. Cardiovasc. Res. 2000, 45, 525–527. [Google Scholar] [CrossRef][Green Version]
- Madonna, R.; De Caterina, R. VEGF receptor switching in heart development and disease. Cardiovasc. Res. 2009, 84, 4–6. [Google Scholar] [CrossRef]
- Zentilin, L.; Puligadda, U.; Lionetti, V.; Zacchigna, S.; Collesi, C.; Pattarini, L.; Ruozi, G.; Camporesi, S.; Sinagra, G.; Pepe, M.; et al. Cardiomyocyte VEGFR-1 activation by VEGF-B induces compensatory hypertrophy and preserves cardiac function after myocardial infarction. FASEB J. 2010, 24, 1467–1478. [Google Scholar] [CrossRef]
- Guo, H.; Cui, G.; Yang, J.; Wang, C.; Zhu, J.; Zhang, L.; Jiang, J.; Shao, S. Sustained delivery of VEGF from designer self-assembling peptides improves cardiac function after myocardial infarction. Biochem. Biophys. Res. Commun. 2012, 424, 105–111. [Google Scholar] [CrossRef]
- Rottbauer, W. VEGF-PLC 1 pathway controls cardiac contractility in the embryonic heart. Genes Dev. 2005, 19, 1624–1634. [Google Scholar] [CrossRef]
- Arsic, N.; Zentilin, L.; Zacchigna, S.; Santoro, D.; Stanta, G.; Salvi, A.; Sinagra, G.; Giacca, M. Induction of functional neovascularization by combined VEGF and angiopoietin-1 gene transfer using AAV vectors. Mol. Ther. J. Am. Soc. Gene Ther. 2003, 7, 450–459. [Google Scholar] [CrossRef]
- Poltorak, Z.; Cohen, T.; Neufeld, G. The VEGF splice variants: Properties, receptors, and usage for the treatment of ischemic diseases. Herz 2000, 25, 126–129. [Google Scholar] [CrossRef]
- Ren, G.; Dewald, O.; Frangogiannis, N. Inflammatory Mechanisms in Myocardial Infarction. Curr. Drug Target -Inflamm. Allergy 2003, 2, 242–256. [Google Scholar] [CrossRef]
- Tang, J.-M.; Wang, J.-N.; Zhang, L.; Zheng, F.; Yang, J.-Y.; Kong, X.; Guo, L.-Y.; Chen, L.; Huang, Y.-Z.; Wan, Y.; et al. VEGF/SDF-1 promotes cardiac stem cell mobilization and myocardial repair in the infarcted heart. Cardiovasc. Res. 2011, 91, 402–411. [Google Scholar] [CrossRef] [PubMed]
- Zhao, T.; Zhao, W.; Chen, Y.; Liu, L.; Ahokas, R.A.; Sun, Y. Differential expression of vascular endothelial growth factor isoforms and receptor subtypes in the infarcted heart. Int. J. Cardiol. 2013, 167, 2638–2645. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Brown, L.F.; Hibberd, M.G.; Grossman, J.D.; Morgan, J.P.; Simons, M. VEGF, flk-1, and flt-1 expression in a rat myocardial infarction model of angiogenesis. Am. J. Physiol. 1996, 270, H1803–H1811. [Google Scholar] [CrossRef] [PubMed]
- Zhao, T.; Zhao, W.; Chen, Y.; Ahokas, R.A.; Sun, Y. Vascular endothelial growth factor (VEGF)-A: Role on cardiac angiogenesis following myocardial infarction. Microvasc. Res. 2010, 80, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Touyz, R.M.; Herrmann, J. Cardiotoxicity with vascular endothelial growth factor inhibitor therapy. NPJ Precis. Oncol. 2018, 2, 13. [Google Scholar] [CrossRef]
- Ucuzian, A.A.; Gassman, A.A.; East, A.T.; Greisler, H.P. Molecular mediators of angiogenesis. J. Burn Care Res. Off. Publ. Am. Burn Assoc. 2010, 31, 158–175. [Google Scholar] [CrossRef]
- Carmeliet, P.; Jain, R.K. Angiogenesis in cancer and other diseases. Nature 2000, 407, 249–257. [Google Scholar] [CrossRef]
- Feng, Y.-M.; Verfaillie, C.; Yu, H. Vascular Diseases and Metabolic Disorders. Stem Cells Int. 2016, 2016, 5810358. [Google Scholar] [CrossRef]
- Moran, A.; Forouzanfar, M.; Sampson, U.; Chugh, S.; Feigin, V.; Mensah, G. The epidemiology of cardiovascular diseases in sub-Saharan Africa: The Global Burden of Diseases, Injuries and Risk Factors 2010 Study. Prog. Cardiovasc. Dis. 2013, 56, 234–239. [Google Scholar] [CrossRef]
- Mozaffarian, D.; Benjamin, E.J.; Go, A.S.; Arnett, D.K.; Blaha, M.J.; Cushman, M.; Das, S.R.; de Ferranti, S.; Després, J.-P.; Fullerton, H.J.; et al. Heart Disease and Stroke Statistics-2016 Update: A Report From the American Heart Association. Circulation 2016, 133, e38-360. [Google Scholar] [CrossRef]
- Semenkovich, C.F. We Know More Than We Can Tell About Diabetes and Vascular Disease: The 2016 Edwin Bierman Award Lecture. Diabetes 2017, 66, 1735–1741. [Google Scholar] [CrossRef]
- Lauer, M.S. Advancing cardiovascular research. Chest 2012, 141, 500–505. [Google Scholar] [CrossRef]
- Luo, K.; Long, H.; Xu, B. Reduced apoptosis after acute myocardial infarction by simvastatin. Cell Biochem. Biophys. 2015, 71, 735–740. [Google Scholar] [CrossRef]
- Hung, J.; Teng, T.-H.K.; Finn, J.; Knuiman, M.; Briffa, T.; Stewart, S.; Sanfilippo, F.M.; Ridout, S.; Hobbs, M. Trends from 1996 to 2007 in incidence and mortality outcomes of heart failure after acute myocardial infarction: A population-based study of 20,812 patients with first acute myocardial infarction in Western Australia. J. Am. Heart Assoc. 2013, 2, e000172. [Google Scholar] [CrossRef] [PubMed]
- Mendis, S.; Thygesen, K.; Kuulasmaa, K.; Giampaoli, S.; Mähönen, M.; Ngu Blackett, K.; Lisheng, L. Writing group on behalf of the participating experts of the WHO consultation for revision of WHO definition of myocardial infarction World Health Organization definition of myocardial infarction: 2008-09 revision. Int. J. Epidemiol. 2011, 40, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Dalen, J.E.; Alpert, J.S.; Goldberg, R.J.; Weinstein, R.S. The epidemic of the 20(th) century: Coronary heart disease. Am. J. Med. 2014, 127, 807–812. [Google Scholar] [CrossRef] [PubMed]
- Patel, N.R.; Patel, D.V.; Murumkar, P.R.; Yadav, M.R. Contemporary developments in the discovery of selective factor Xa inhibitors: A review. Eur. J. Med. Chem. 2016, 121, 671–698. [Google Scholar] [CrossRef]
- Antithrombotic Trialists’ (ATT) Collaboration; Baigent, C.; Blackwell, L.; Collins, R.; Emberson, J.; Godwin, J.; Peto, R.; Buring, J.; Hennekens, C.; Kearney, P.; et al. Aspirin in the primary and secondary prevention of vascular disease: Collaborative meta-analysis of individual participant data from randomised trials. Lancet Lond. Engl. 2009, 373, 1849–1860. [Google Scholar] [CrossRef]
- Jackson, L.R.; Ju, C.; Zettler, M.; Messenger, J.C.; Cohen, D.J.; Stone, G.W.; Baker, B.A.; Effron, M.; Peterson, E.D.; Wang, T.Y. Outcomes of Patients With Acute Myocardial Infarction Undergoing Percutaneous Coronary Intervention Receiving an Oral Anticoagulant and Dual Antiplatelet Therapy: A Comparison of Clopidogrel Versus Prasugrel From the TRANSLATE-ACS Study. JACC Cardiovasc. Interv. 2015, 8, 1880–1889. [Google Scholar] [CrossRef]
- Anisimov, A.; Tvorogov, D.; Alitalo, A.; Leppänen, V.-M.; An, Y.; Han, E.C.; Orsenigo, F.; Gaál, E.I.; Holopainen, T.; Koh, Y.J.; et al. Vascular endothelial growth factor-angiopoietin chimera with improved properties for therapeutic angiogenesis. Circulation 2013, 127, 424–434. [Google Scholar] [CrossRef]
- Albrecht-Schgoer, K.; Schgoer, W.; Holfeld, J.; Theurl, M.; Wiedemann, D.; Steger, C.; Gupta, R.; Semsroth, S.; Fischer-Colbrie, R.; Beer, A.G.E.; et al. The angiogenic factor secretoneurin induces coronary angiogenesis in a model of myocardial infarction by stimulation of vascular endothelial growth factor signaling in endothelial cells. Circulation 2012, 126, 2491–2501. [Google Scholar] [CrossRef]
- Cheng, C.; Li, P.; Wang, Y.-G.; Bi, M.-H.; Wu, P.-S. Study on the expression of VEGF and HIF-1α in infarct area of rats with AMI. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 115–119. [Google Scholar]
- Seko, Y.; Fukuda, S.; Nagai, R. Serum levels of endostatin, vascular endothelial growth factor (VEGF) and hepatocyte growth factor (HGF) in patients with acute myocardial infarction undergoing early reperfusion therapy. Clin. Sci. Lond. Engl. 1979 2004, 106, 439–442. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Wan, J.; Pan, W.; Zou, J. Expression of vascular endothelial growth factor in cardiac repair: Signaling mechanisms mediating vascular protective effects. Int. J. Biol. Macromol. 2018, 113, 179–185. [Google Scholar] [CrossRef]
- Yu, F.; Lu, J.; Li, Z.; Zhou, X.; Zeng, S.; Zhan, Q.; Yuan, M.; Yang, Q.; Xia, J. Correlation of Plasma Vascular Endothelial Growth Factor and Endostatin Levels with Symptomatic Intra- and Extracranial Atherosclerotic Stenosis in a Chinese Han Population. J. Stroke Cerebrovasc. Dis. Off. J. Natl. Stroke Assoc. 2017, 26, 1061–1070. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.-M.; Zhang, X.-B.; Jiang, W.; Wang, H.-D.; Zhang, Y.-N. Astragalosides promote angiogenesis via vascular endothelial growth factor and basic fibroblast growth factor in a rat model of myocardial infarction. Mol. Med. Rep. 2015, 12, 6718–6726. [Google Scholar] [CrossRef] [PubMed]
- Shinohara, K. The expression and the role of vascular endothelial growth factor (VEGF) in human normal and myocardial infarcted heart. Hokkaido Igaku Zasshi 1994, 69, 978–993. [Google Scholar] [PubMed]
- Eržen, B.; Šilar, M.; Šabovič, M. Stable phase post-MI patients have elevated VEGF levels correlated with inflammation markers, but not with atherosclerotic burden. BMC Cardiovasc. Disord. 2014, 14, 166. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Banquet, S.; Gomez, E.; Nicol, L.; Edwards-Lévy, F.; Henry, J.-P.; Cao, R.; Schapman, D.; Dautreaux, B.; Lallemand, F.; Bauer, F.; et al. Arteriogenic therapy by intramyocardial sustained delivery of a novel growth factor combination prevents chronic heart failure. Circulation 2011, 124, 1059–1069. [Google Scholar] [CrossRef] [PubMed]
- Iadecola, C. The pathobiology of vascular dementia. Neuron 2013, 80, 844–866. [Google Scholar] [CrossRef]
- Del Zoppo, G.J.; Mabuchi, T. Cerebral microvessel responses to focal ischemia. J. Cereb. Blood Flow Metab. Off. J. Int. Soc. Cereb. Blood Flow Metab. 2003, 23, 879–894. [Google Scholar] [CrossRef]
- Fan, Y.; Yang, G.-Y. Therapeutic angiogenesis for brain ischemia: A brief review. J. Neuroimmune Pharmacol. Off. J. Soc. NeuroImmune Pharmacol. 2007, 2, 284–289. [Google Scholar] [CrossRef]
- Beck, H.; Plate, K.H. Angiogenesis after cerebral ischemia. Acta Neuropathol. (Berl.) 2009, 117, 481–496. [Google Scholar] [CrossRef] [PubMed]
- Li, T.-S.; Cheng, K.; Malliaras, K.; Smith, R.R.; Zhang, Y.; Sun, B.; Matsushita, N.; Blusztajn, A.; Terrovitis, J.; Kusuoka, H.; et al. Direct comparison of different stem cell types and subpopulations reveals superior paracrine potency and myocardial repair efficacy with cardiosphere-derived cells. J. Am. Coll. Cardiol. 2012, 59, 942–953. [Google Scholar] [CrossRef] [PubMed]
- Talwar, T.; Srivastava, M.V.P. Role of vascular endothelial growth factor and other growth factors in post-stroke recovery. Ann. Indian Acad. Neurol. 2014, 17, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Zhao, B.-Q.; Wang, S.; Kim, H.-Y.; Storrie, H.; Rosen, B.R.; Mooney, D.J.; Wang, X.; Lo, E.H. Role of matrix metalloproteinases in delayed cortical responses after stroke. Nat. Med. 2006, 12, 441–445. [Google Scholar] [CrossRef] [PubMed]
- Carmeliet, P.; Collen, D. Molecular basis of angiogenesis. Role of VEGF and VE-cadherin. Ann. N. Y. Acad. Sci. 2000, 902, 249–262; discussion 262. [Google Scholar] [CrossRef] [PubMed]
- Lutsenko, S.V.; Kiselev, S.M.; Severin, S.E. Molecular mechanisms of tumor angiogenesis. Biochem. Biokhimiia 2003, 68, 286–300. [Google Scholar] [CrossRef]
- Kobiyama, K.; Ley, K. Atherosclerosis. Circ. Res. 2018, 123, 1118–1120. [Google Scholar] [CrossRef]
- Chistiakov, D.A.; Melnichenko, A.A.; Myasoedova, V.A.; Grechko, A.V.; Orekhov, A.N. Role of lipids and intraplaque hypoxia in the formation of neovascularization in atherosclerosis. Ann. Med. 2017, 49, 661–677. [Google Scholar] [CrossRef]
- Holm, P.W.; Slart, R.H.J.A.; Zeebregts, C.J.; Hillebrands, J.L.; Tio, R.A. Atherosclerotic plaque development and instability: A dual role for VEGF. Ann. Med. 2009, 41, 257–264. [Google Scholar] [CrossRef]
- Asahara, T.; Chen, D.; Tsurumi, Y.; Kearney, M.; Rossow, S.; Passeri, J.; Symes, J.F.; Isner, J.M. Accelerated restitution of endothelial integrity and endothelium-dependent function after phVEGF165 gene transfer. Circulation 1996, 94, 3291–3302. [Google Scholar] [CrossRef]
- Camaré, C.; Pucelle, M.; Nègre-Salvayre, A.; Salvayre, R. Angiogenesis in the atherosclerotic plaque. Redox Biol. 2017, 12, 18–34. [Google Scholar] [CrossRef] [PubMed]
- Barleon, B.; Sozzani, S.; Zhou, D.; Weich, H.A.; Mantovani, A.; Marmé, D. Migration of human monocytes in response to vascular endothelial growth factor (VEGF) is mediated via the VEGF receptor flt-1. Blood 1996, 87, 3336–3343. [Google Scholar] [CrossRef] [PubMed]
- Jaipersad, A.S.; Lip, G.Y.H.; Silverman, S.; Shantsila, E. The Role of Monocytes in Angiogenesis and Atherosclerosis. J. Am. Coll. Cardiol. 2014, 63, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.; Moon, S.O.; Kim, S.H.; Kim, H.J.; Koh, Y.S.; Koh, G.Y. Vascular endothelial growth factor expression of intercellular adhesion molecule 1 (ICAM-1), vascular cell adhesion molecule 1 (VCAM-1), and E-selectin through nuclear factor-kappa B activation in endothelial cells. J. Biol. Chem. 2001, 276, 7614–7620. [Google Scholar] [CrossRef] [PubMed]
- Hong, K.H.; Ryu, J.; Han, K.H. Monocyte chemoattractant protein-1–induced angiogenesis is mediated by vascular endothelial growth factor-A. Blood 2005, 105, 1405–1407. [Google Scholar] [CrossRef] [PubMed]
- Ylä-Herttuala, S.; Rissanen, T.T.; Vajanto, I.; Hartikainen, J. Vascular Endothelial Growth Factors. J. Am. Coll. Cardiol. 2007, 49, 1015–1026. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.-M.; Deng, X.-T.; Qi, R.-M.; Xiao, L.-Y.; Yang, C.-Q.; Gong, T. Mechanism of Chronic Stress-induced Reduced Atherosclerotic Medial Area and Increased Plaque Instability in Rabbit Models of Chronic Stress. Chin. Med. J. (Engl.) 2018, 131, 161–170. [Google Scholar] [CrossRef]
- Figueira, L.; González, J.C. Effect of resveratrol on seric vascular endothelial growth factor concentrations during atherosclerosis. Clin. E Investig. En Arterioscler. Publicacion Soc. Espanola Arterioscler. 2018, 30, 209–216. [Google Scholar] [CrossRef]
- Moulton, K.S.; Heller, E.; Konerding, M.A.; Flynn, E.; Palinski, W.; Folkman, J. Angiogenesis inhibitors endostatin or TNP-470 reduce intimal neovascularization and plaque growth in apolipoprotein E-deficient mice. Circulation 1999, 99, 1726–1732. [Google Scholar] [CrossRef]
- Choueiri, T.K.; Schutz, F.A.B.; Je, Y.; Rosenberg, J.E.; Bellmunt, J. Risk of arterial thromboembolic events with sunitinib and sorafenib: A systematic review and meta-analysis of clinical trials. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2010, 28, 2280–2285. [Google Scholar] [CrossRef]
- Sandhofer, A.; Tatarczyk, T.; Kirchmair, R.; Iglseder, B.; Paulweber, B.; Patsch, J.R.; Schratzberger, P. Are plasma VEGF and its soluble receptor sFlt-1 atherogenic risk factors? Cross-sectional data from the SAPHIR study. Atherosclerosis 2009, 206, 265–269. [Google Scholar] [CrossRef]
- Libby, P.; Loscalzo, J.; Ridker, P.M.; Farkouh, M.E.; Hsue, P.Y.; Fuster, V.; Hasan, A.A.; Amar, S. Inflammation, Immunity, and Infection in Atherothrombosis: JACC Review Topic of the Week. J. Am. Coll. Cardiol. 2018, 72, 2071–2081. [Google Scholar] [CrossRef] [PubMed]
- Döring, Y.; Soehnlein, O.; Weber, C. Neutrophil Extracellular Traps in Atherosclerosis and Atherothrombosis. Circ. Res. 2017, 120, 736–743. [Google Scholar] [CrossRef] [PubMed]
- Badimon, L.; Suades, R.; Crespo, J.; Padro, T.; Chiva-Blanch, G. Diet, microparticles and atherothrombosis. Front. Biosci. Landmark Ed. 2018, 23, 432–457. [Google Scholar] [CrossRef] [PubMed]
- Daigo, K.; Inforzato, A.; Barajon, I.; Garlanda, C.; Bottazzi, B.; Meri, S.; Mantovani, A. Pentraxins in the activation and regulation of innate immunity. Immunol. Rev. 2016, 274, 202–217. [Google Scholar] [CrossRef] [PubMed]
- Arrillaga-Romany, I.; Norden, A.D. Antiangiogenic therapies for glioblastoma. CNS Oncol. 2014, 3, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Puranik, R.; Celermajer, D.S. Smoking and endothelial function. Prog. Cardiovasc. Dis. 2003, 45, 443–458. [Google Scholar] [CrossRef]
- Cooney, M.M.; Tserng, K.-Y.; Makar, V.; McPeak, R.J.; Ingalls, S.T.; Dowlati, A.; Overmoyer, B.; McCrae, K.; Ksenich, P.; Lavertu, P.; et al. A phase IB clinical and pharmacokinetic study of the angiogenesis inhibitor SU5416 and paclitaxel in recurrent or metastatic carcinoma of the head and neck. Cancer Chemother. Pharmacol. 2005, 55, 295–300. [Google Scholar] [CrossRef]
- Oppelt, P.; Betbadal, A.; Nayak, L. Approach to chemotherapy-associated thrombosis. Vasc. Med. Lond. Engl. 2015, 20, 153–161. [Google Scholar] [CrossRef]
- Zangari, M.; Fink, L.M.; Elice, F.; Zhan, F.; Adcock, D.M.; Tricot, G.J. Thrombotic events in patients with cancer receiving antiangiogenesis agents. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2009, 27, 4865–4873. [Google Scholar] [CrossRef]
- Mir, O.; Coriat, R.; Cabanes, L.; Ropert, S.; Billemont, B.; Alexandre, J.; Durand, J.-P.; Treluyer, J.-M.; Knebelmann, B.; Goldwasser, F. An observational study of bevacizumab-induced hypertension as a clinical biomarker of antitumor activity. Oncologist 2011, 16, 1325–1332. [Google Scholar] [CrossRef] [PubMed]
- Semeniuk-Wojtaś, A.; Lubas, A.; Stec, R.; Szczylik, C.; Niemczyk, S. Influence of Tyrosine Kinase Inhibitors on Hypertension and Nephrotoxicity in Metastatic Renal Cell Cancer Patients. Int. J. Mol. Sci. 2016, 17, 2073. [Google Scholar] [CrossRef] [PubMed]
- Rini, B.I.; Cohen, D.P.; Lu, D.R.; Chen, I.; Hariharan, S.; Gore, M.E.; Figlin, R.A.; Baum, M.S.; Motzer, R.J. Hypertension as a biomarker of efficacy in patients with metastatic renal cell carcinoma treated with sunitinib. J. Natl. Cancer Inst. 2011, 103, 763–773. [Google Scholar] [CrossRef] [PubMed]
- Touyz, R.M.; Herrmann, S.M.S.; Herrmann, J. Vascular toxicities with VEGF inhibitor therapies-focus on hypertension and arterial thrombotic events. J. Am. Soc. Hypertens. 2018, 12, 409–425. [Google Scholar] [CrossRef]
- Van Hinsbergh, V.W.M.; Eringa, E.C.; Daemen, M.J.A.P. Neovascularization of the atherosclerotic plaque: Interplay between atherosclerotic lesion, adventitia-derived microvessels and perivascular fat. Curr. Opin. Lipidol. 2015, 26, 405–411. [Google Scholar] [CrossRef]
- Pilarczyk, K.; Sattler, K.J.E.; Galili, O.; Versari, D.; Olson, M.L.; Meyer, F.B.; Zhu, X.-Y.; Lerman, L.O.; Lerman, A. Placenta growth factor expression in human atherosclerotic carotid plaques is related to plaque destabilization. Atherosclerosis 2008, 196, 333–340. [Google Scholar] [CrossRef]
- Sluimer, J.C.; Kolodgie, F.D.; Bijnens, A.P.J.J.; Maxfield, K.; Pacheco, E.; Kutys, B.; Duimel, H.; Frederik, P.M.; van Hinsbergh, V.W.M.; Virmani, R.; et al. Thin-walled microvessels in human coronary atherosclerotic plaques show incomplete endothelial junctions relevance of compromised structural integrity for intraplaque microvascular leakage. J. Am. Coll. Cardiol. 2009, 53, 1517–1527. [Google Scholar] [CrossRef]
- Michel, J.-B.; Virmani, R.; Arbustini, E.; Pasterkamp, G. Intraplaque haemorrhages as the trigger of plaque vulnerability. Eur. Heart J. 2011, 32, 1977–1985, 1985a, 1985b, 1985c. [Google Scholar] [CrossRef]
- Tziakas, D.N.; Kaski, J.C.; Chalikias, G.K.; Romero, C.; Fredericks, S.; Tentes, I.K.; Kortsaris, A.X.; Hatseras, D.I.; Holt, D.W. Total cholesterol content of erythrocyte membranes is increased in patients with acute coronary syndrome: A new marker of clinical instability? J. Am. Coll. Cardiol. 2007, 49, 2081–2089. [Google Scholar] [CrossRef]
- Moulton, K.S.; Vakili, K.; Zurakowski, D.; Soliman, M.; Butterfield, C.; Sylvin, E.; Lo, K.-M.; Gillies, S.; Javaherian, K.; Folkman, J. Inhibition of plaque neovascularization reduces macrophage accumulation and progression of advanced atherosclerosis. Proc. Natl. Acad. Sci. USA 2003, 100, 4736–4741. [Google Scholar] [CrossRef]
- Winnik, S.; Lohmann, C.; Siciliani, G.; von Lukowicz, T.; Kuschnerus, K.; Kraenkel, N.; Brokopp, C.E.; Enseleit, F.; Michels, S.; Ruschitzka, F.; et al. Systemic VEGF inhibition accelerates experimental atherosclerosis and disrupts endothelial homeostasis--implications for cardiovascular safety. Int. J. Cardiol. 2013, 168, 2453–2461. [Google Scholar] [CrossRef] [PubMed]
- Cho, M.-L.; Jung, Y.O.; Moon, Y.-M.; Min, S.-Y.; Yoon, C.-H.; Lee, S.-H.; Park, S.-H.; Cho, C.-S.; Jue, D.-M.; Kim, H.-Y. Interleukin-18 induces the production of vascular endothelial growth factor (VEGF) in rheumatoid arthritis synovial fibroblasts via AP-1-dependent pathways. Immunol. Lett. 2006, 103, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Meng, R.; Pei, Z.; Chen, B.; Ma, R.; Zhang, C.; Chen, B.; Zhang, A.; Wu, T.; Liu, D.; Dong, Y. Age-related change of serum angiogenic factor levels in patients with coronary artery disease. Acta Cardiol. 2009, 64, 735–740. [Google Scholar] [CrossRef] [PubMed]
- Vm, M.; Al, S.; Aa, A.; As, Z.; Av, K.; Rs, O.; Im, M.; Ga, K. Circulating interleukin-18: Association with IL-8, IL-10 and VEGF serum levels in patients with and without heart rhythm disorders. Int. J. Cardiol. 2016, 215, 105–109. [Google Scholar] [CrossRef] [PubMed]
- Kucukardali, Y.; Aydogdu, S.; Ozmen, N.; Yonem, A.; Solmazgul, E.; Ozyurt, M.; Cingozbay, Y.; Aydogdu, A. The relationship between severity of coronary artery disease and plasma level of vascular endothelial growth factor. Cardiovasc. Revasc. Med. 2008, 9, 66–70. [Google Scholar] [CrossRef]
- Henning, R.J. Therapeutic angiogenesis: Angiogenic growth factors for ischemic heart disease. Future Cardiol. 2016, 12, 585–599. [Google Scholar] [CrossRef]
- Laham, R.J.; Chronos, N.A.; Pike, M.; Leimbach, M.E.; Udelson, J.E.; Pearlman, J.D.; Pettigrew, R.I.; Whitehouse, M.J.; Yoshizawa, C.; Simons, M. Intracoronary basic fibroblast growth factor (FGF-2) in patients with severe ischemic heart disease: Results of a phase I open-label dose escalation study. J. Am. Coll. Cardiol. 2000, 36, 2132–2139. [Google Scholar] [CrossRef]
- Mitsos, S.; Katsanos, K.; Koletsis, E.; Kagadis, G.C.; Anastasiou, N.; Diamantopoulos, A.; Karnabatidis, D.; Dougenis, D. Therapeutic angiogenesis for myocardial ischemia revisited: Basic biological concepts and focus on latest clinical trials. Angiogenesis 2012, 15, 1–22. [Google Scholar] [CrossRef]
- Grines, C.L.; Watkins, M.W.; Helmer, G.; Penny, W.; Brinker, J.; Marmur, J.D.; West, A.; Rade, J.J.; Marrott, P.; Hammond, H.K.; et al. Angiogenic Gene Therapy (AGENT) trial in patients with stable angina pectoris. Circulation 2002, 105, 1291–1297. [Google Scholar] [CrossRef]
- Grines, C.L.; Watkins, M.W.; Mahmarian, J.J.; Iskandrian, A.E.; Rade, J.J.; Marrott, P.; Pratt, C.; Kleiman, N. Angiogene GENe Therapy (AGENT-2) Study Group A randomized, double-blind, placebo-controlled trial of Ad5FGF-4 gene therapy and its effect on myocardial perfusion in patients with stable angina. J. Am. Coll. Cardiol. 2003, 42, 1339–1347. [Google Scholar] [CrossRef]
- Henry, T.D.; Grines, C.L.; Watkins, M.W.; Dib, N.; Barbeau, G.; Moreadith, R.; Andrasfay, T.; Engler, R.L. Effects of Ad5FGF-4 in patients with angina: An analysis of pooled data from the AGENT-3 and AGENT-4 trials. J. Am. Coll. Cardiol. 2007, 50, 1038–1046. [Google Scholar] [CrossRef] [PubMed]
- Afzal, M.R.; Samanta, A.; Shah, Z.I.; Jeevanantham, V.; Abdel-Latif, A.; Zuba-Surma, E.K.; Dawn, B. Adult Bone Marrow Cell Therapy for Ischemic Heart Disease: Evidence and Insights From Randomized Controlled Trials. Circ. Res. 2015, 117, 558–575. [Google Scholar] [CrossRef] [PubMed]
- Epelman, S.; Liu, P.P.; Mann, D.L. Role of innate and adaptive immune mechanisms in cardiac injury and repair. Nat. Rev. Immunol. 2015, 15, 117–129. [Google Scholar] [CrossRef] [PubMed]
- Sattler, S.; Rosenthal, N. The neonate versus adult mammalian immune system in cardiac repair and regeneration. Biochim. Biophys. Acta 2016, 1863, 1813–1821. [Google Scholar] [CrossRef]
- Tompkins, B.A.; Natsumeda, M.; Balkan, W.; Hare, J.M. What Is the Future of Cell-Based Therapy for Acute Myocardial Infarction. Circ. Res. 2017, 120, 252–255. [Google Scholar] [CrossRef]
- Duran, J.M.; Makarewich, C.A.; Sharp, T.E.; Starosta, T.; Zhu, F.; Hoffman, N.E.; Chiba, Y.; Madesh, M.; Berretta, R.M.; Kubo, H.; et al. Bone-derived stem cells repair the heart after myocardial infarction through transdifferentiation and paracrine signaling mechanisms. Circ. Res. 2013, 113, 539–552. [Google Scholar] [CrossRef]
- Gray, W.D.; French, K.M.; Ghosh-Choudhary, S.; Maxwell, J.T.; Brown, M.E.; Platt, M.O.; Searles, C.D.; Davis, M.E. Identification of therapeutic covariant microRNA clusters in hypoxia-treated cardiac progenitor cell exosomes using systems biology. Circ. Res. 2015, 116, 255–263. [Google Scholar] [CrossRef]
- Sahoo, S.; Losordo, D.W. Exosomes and cardiac repair after myocardial infarction. Circ. Res. 2014, 114, 333–344. [Google Scholar] [CrossRef]
- Vicencio, J.M.; Yellon, D.M.; Sivaraman, V.; Das, D.; Boi-Doku, C.; Arjun, S.; Zheng, Y.; Riquelme, J.A.; Kearney, J.; Sharma, V.; et al. Plasma exosomes protect the myocardium from ischemia-reperfusion injury. J. Am. Coll. Cardiol. 2015, 65, 1525–1536. [Google Scholar] [CrossRef]
| Cardiovascular Diseases | VEGF-A | Effects | References |
|---|---|---|---|
| Ischemic heart disease: →Myocardial infarction | ↑ | microvessel density in the infarcted area, myocardial remodeling angiogenesis, neovascularization, destabilization of inflamed plaques. | [131,132,133,134,135,136,137] |
| →Stroke | ↑ | vasodilation, vascular permeability, endothelial cells migration, dissociation of smooth muscle cells, loosening of the extracellular matrix. | [146] |
| Atherosclerosis: →Atherothrombosis | ↓ | vascular toxicity, arterial thrombosis. | [169,170,171] |
| →Coronary artery disease | ↑ | angiogenesis, growth and vulnerability of plaques, intraplaque hemorrhage, inflammatory cell and erythrocyte recruitment, coronary artery revascularization. | [176,177,178,179,180] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Braile, M.; Marcella, S.; Cristinziano, L.; Galdiero, M.R.; Modestino, L.; Ferrara, A.L.; Varricchi, G.; Marone, G.; Loffredo, S. VEGF-A in Cardiomyocytes and Heart Diseases. Int. J. Mol. Sci. 2020, 21, 5294. https://doi.org/10.3390/ijms21155294
Braile M, Marcella S, Cristinziano L, Galdiero MR, Modestino L, Ferrara AL, Varricchi G, Marone G, Loffredo S. VEGF-A in Cardiomyocytes and Heart Diseases. International Journal of Molecular Sciences. 2020; 21(15):5294. https://doi.org/10.3390/ijms21155294
Chicago/Turabian StyleBraile, Mariantonia, Simone Marcella, Leonardo Cristinziano, Maria Rosaria Galdiero, Luca Modestino, Anne Lise Ferrara, Gilda Varricchi, Giancarlo Marone, and Stefania Loffredo. 2020. "VEGF-A in Cardiomyocytes and Heart Diseases" International Journal of Molecular Sciences 21, no. 15: 5294. https://doi.org/10.3390/ijms21155294
APA StyleBraile, M., Marcella, S., Cristinziano, L., Galdiero, M. R., Modestino, L., Ferrara, A. L., Varricchi, G., Marone, G., & Loffredo, S. (2020). VEGF-A in Cardiomyocytes and Heart Diseases. International Journal of Molecular Sciences, 21(15), 5294. https://doi.org/10.3390/ijms21155294






