The In Vitro Effect of Prostaglandin E2 and F2α on the Chemerin System in the Porcine Endometrium during Gestation
Abstract
1. Introduction
2. Results
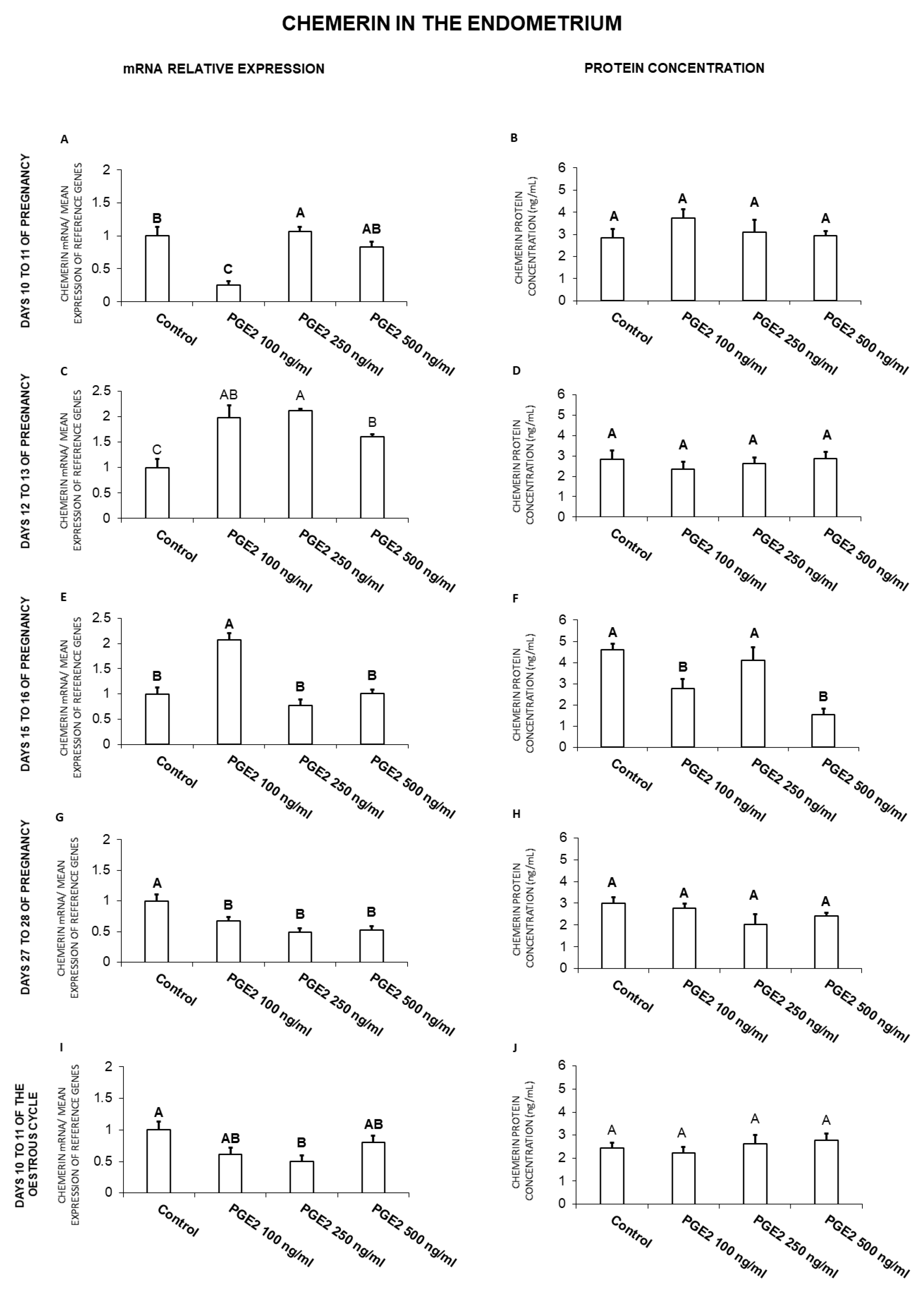
2.1. The Effect of PGE2 on Chemerin Gene Expression and Protein Secretion by the Endometrial Tissue Explants
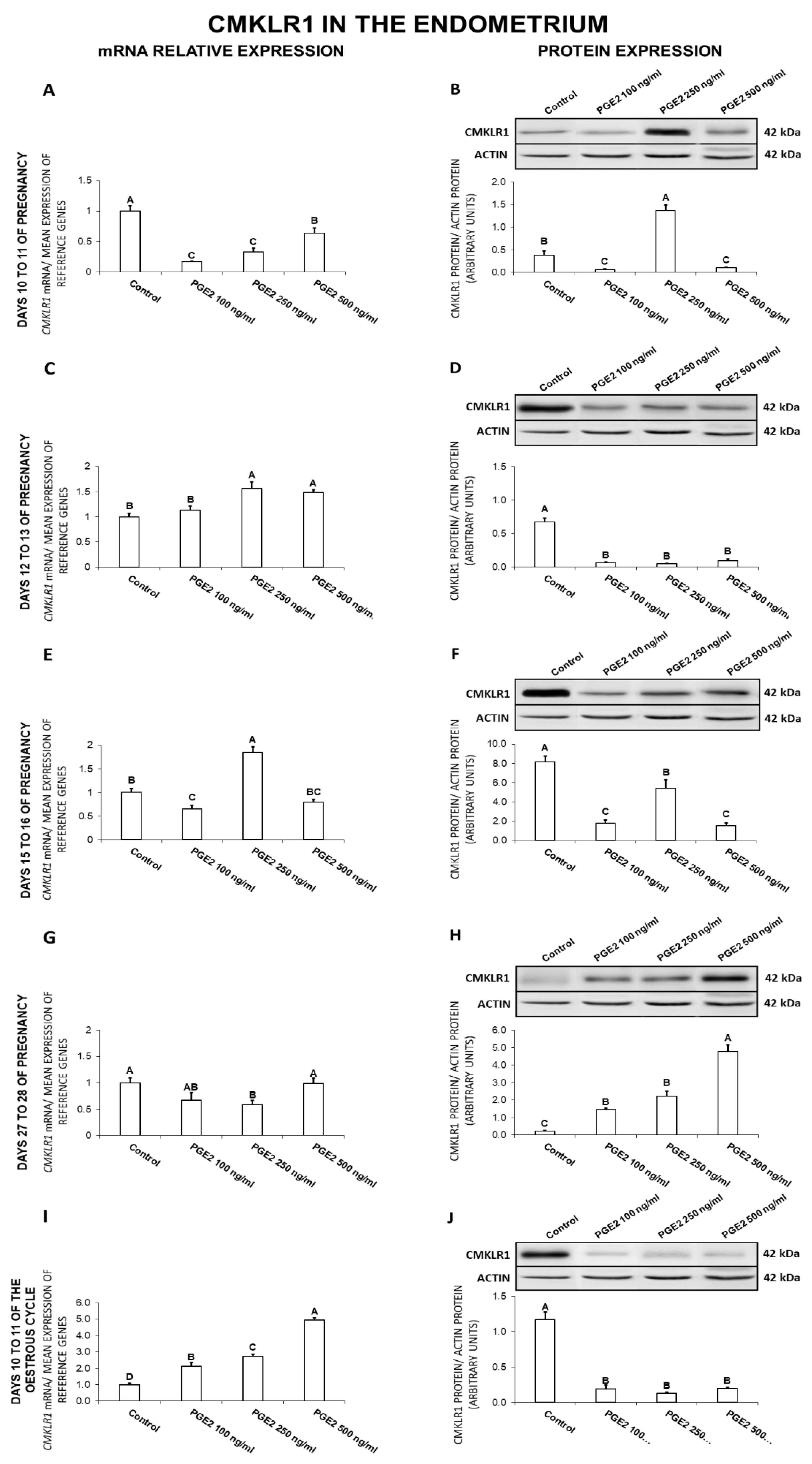
2.2. The Effect of PGE2 on CMKLR1 Gene and Protein Expression in the Endometrial Tissue Explants
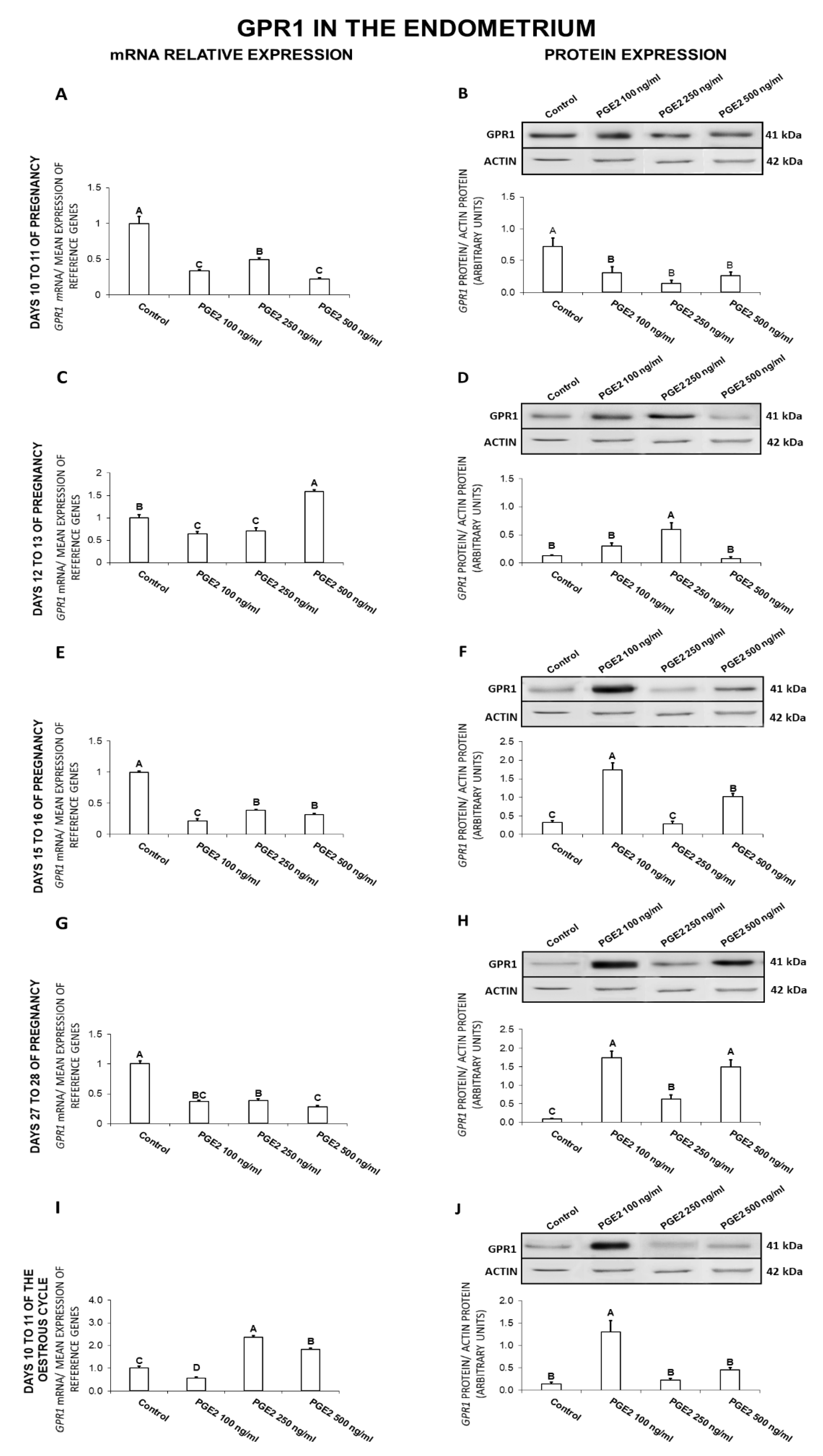
2.3. The Effect of PGE2 on GPR1 Gene and Protein Expression in the Endometrial Tissue Explants
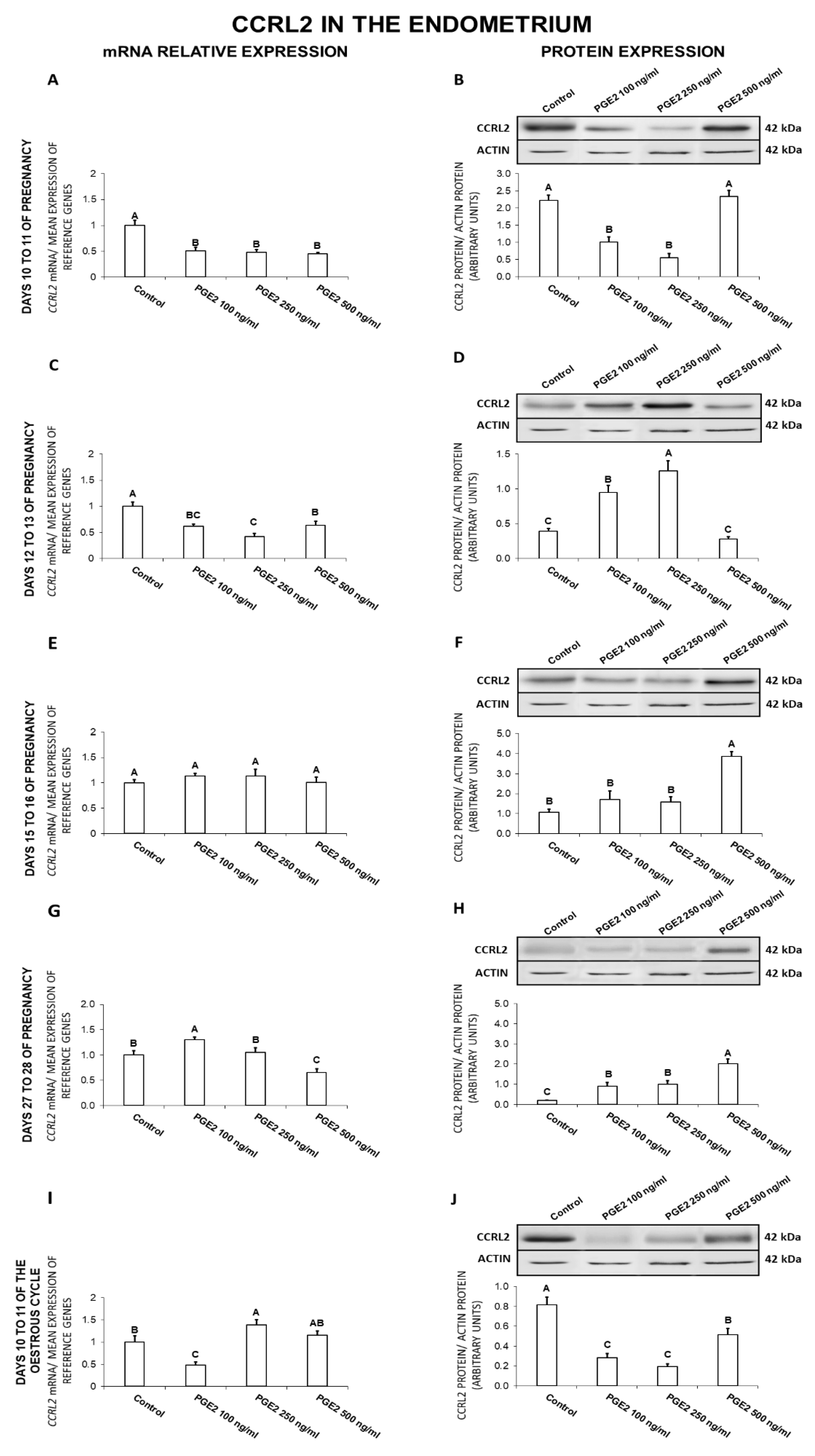
2.4. The Effect of PGE2 on CCRL2 Gene and Protein Expression in the Endometrial Tissue Explants
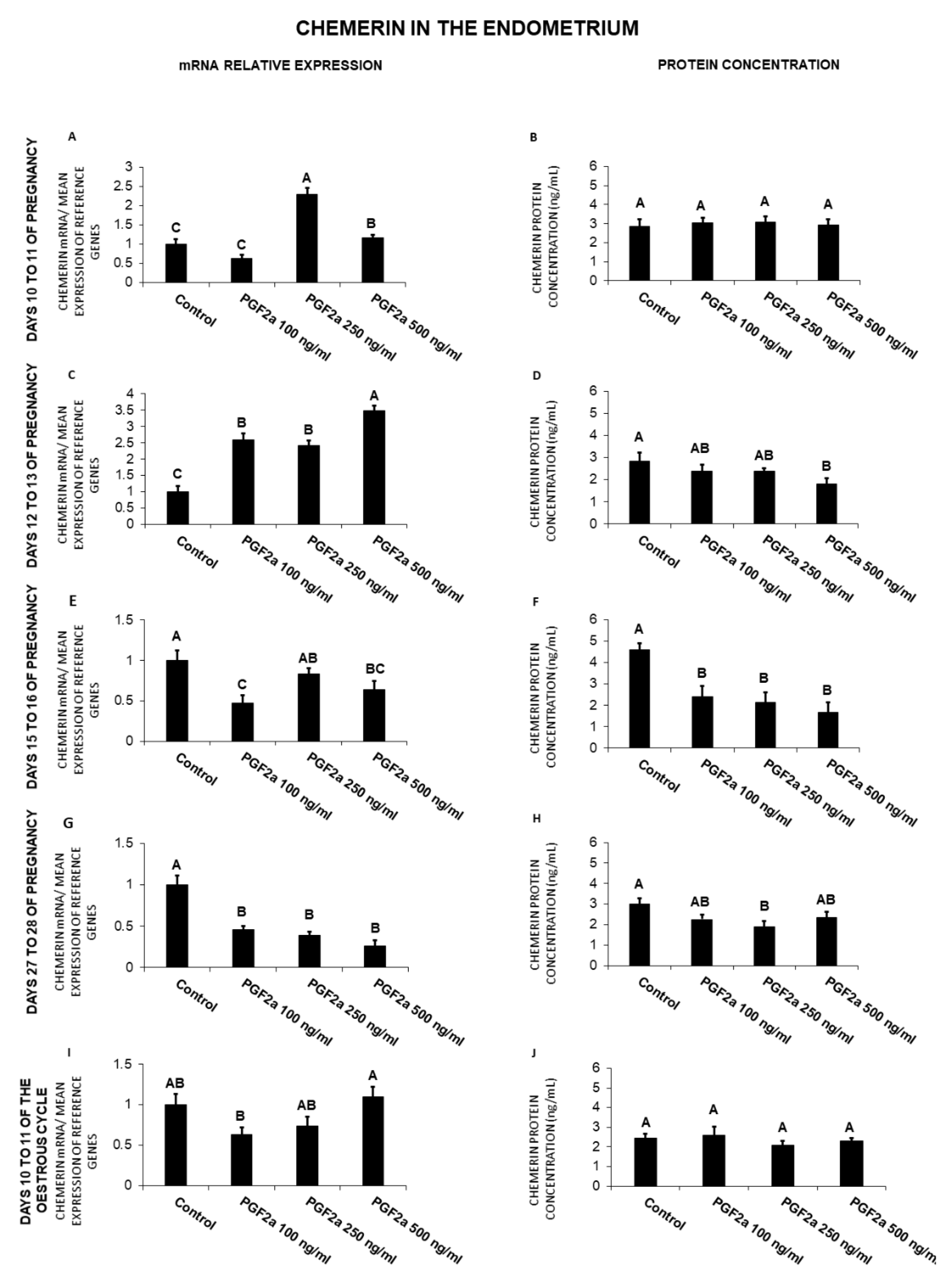
2.5. The Effect of PGF2α on Chemerin Gene Expression and Protein Secretion by the Endometrial Tissue Explants
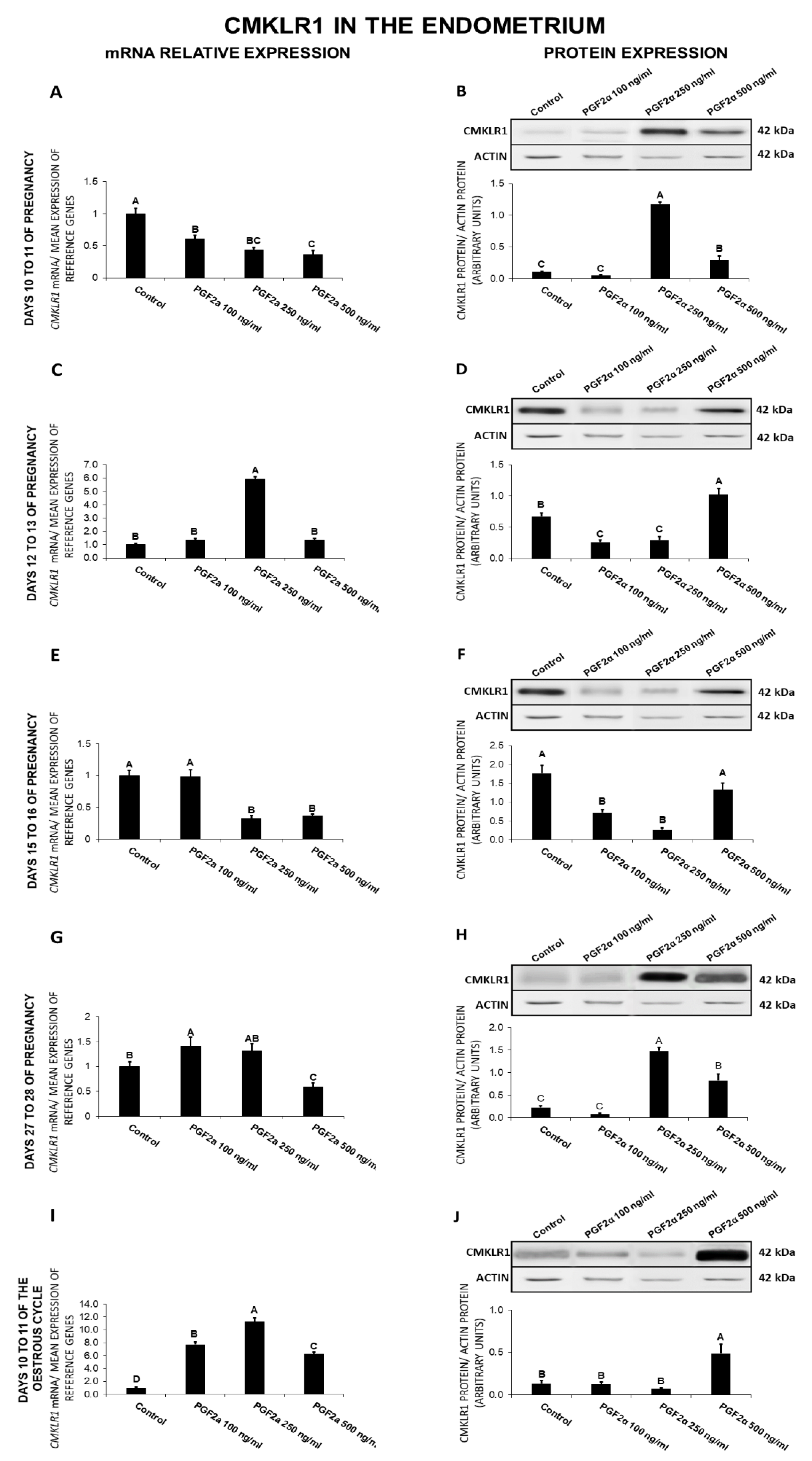
2.6. The Effect of PGF2α on CMKLR1 Gene and Protein Expression in the Endometrial Tissue Explants
2.7. The Effect of PGF2α on GPR1 Gene and Protein Expression in the Endometrial Tissue Explants
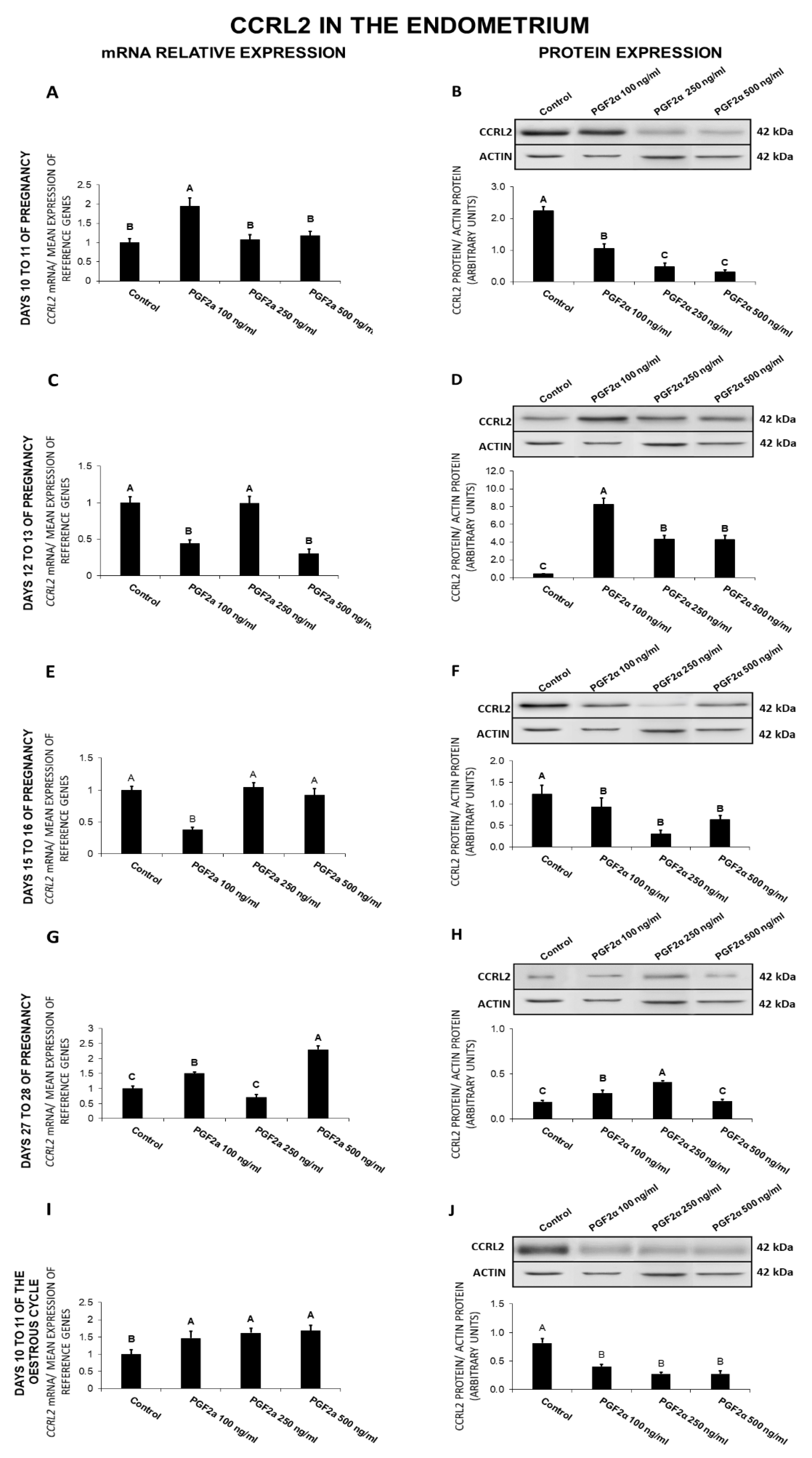
2.8. The Effect of PGF2α on CCRL2 Gene and Protein Expression in the Endometrial Tissue Explants
3. Discussion
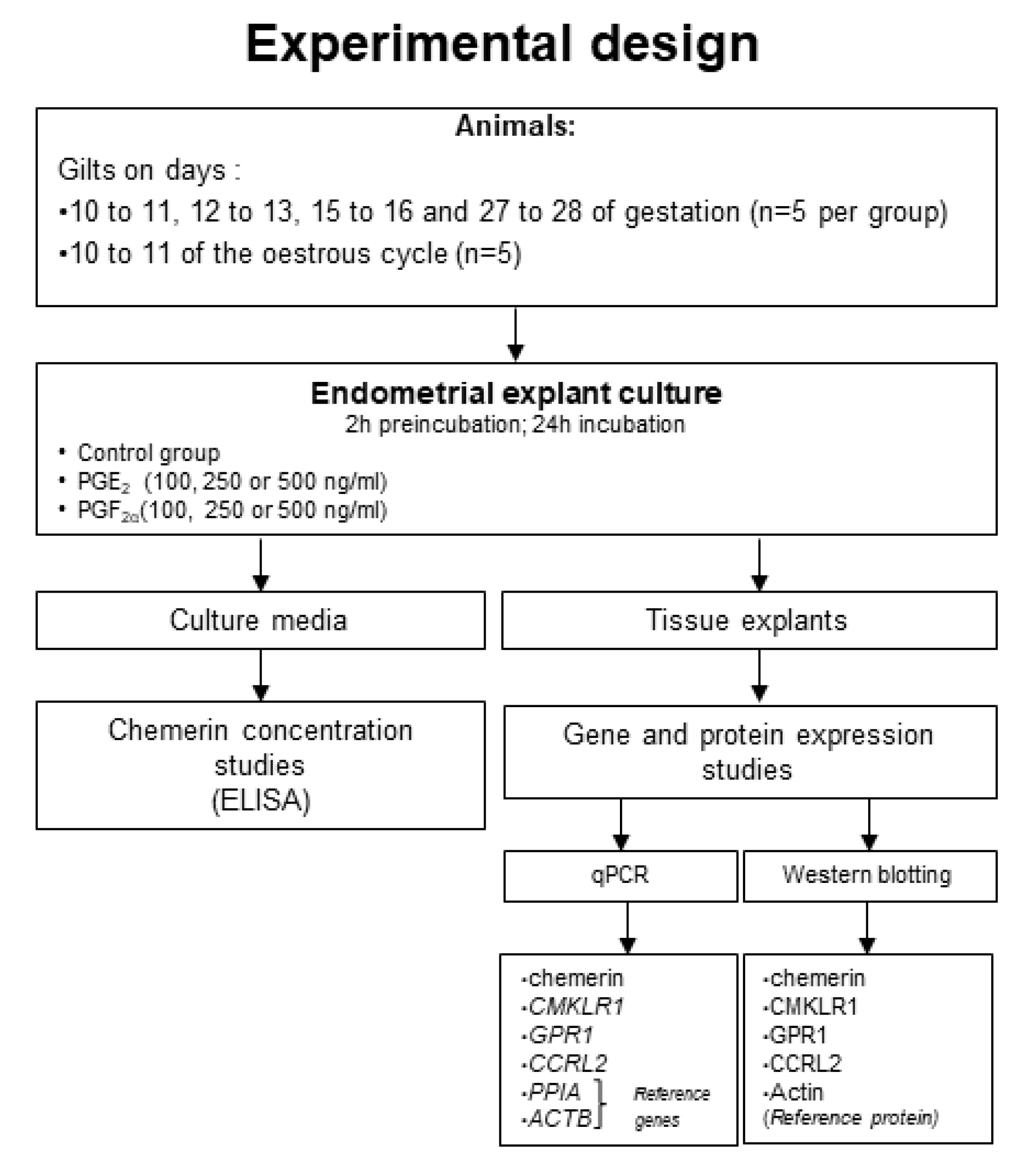
4. Materials and Methods
4.1. Animals and Tissue Collection
4.2. Endometrial Explant Cultures
4.3. Total RNA Isolation and Reverse Transcription
4.4. Quantitative Real-Time PCR Analysis
4.5. Enzyme-Linked Immunosorbent Assay (ELISA) of Chemerin
4.6. Protein Isolation and Western Blotting
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Animal Welfare Statement
References
- Goralski, K.B.; McCarthy, T.C.; Hanniman, E.A.; Zabel, B.A.; Butcher, E.C.; Parlee, S.D.; Muruganandan, S.; Sinal, C.J. Chemerin, a novel adipokine that regulates adipogenesis and adipocyte metabolism. J. Biol. Chem. 2007, 282, 28175–28188. [Google Scholar] [CrossRef] [PubMed]
- Zabel, B.A.; Zuniga, L.; Ohyama, T.; Allen, S.J.; Cichy, J.; Handel, T.M.; Butcher, E.C. Chemoattractants, extracellular proteases, and the integrated host defense response. Exp. Hematol. 2006, 34, 1021–1032. [Google Scholar] [CrossRef] [PubMed]
- Mattern, A.; Zellmann, T.; Beck-Sickinger, A.G. Processing, signaling, and physiological function of chemerin. IUBMB Life 2014, 66, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Brunetti, L.; Orlando, G.; Ferrante, C.; Recinella, L.; Leone, S.; Chiavaroli, A.; Di Nisio, C.; Shohreh, R.; Manippa, F.; Ricciuti, A.; et al. Peripheral chemerin administration modulates hypothalamic control of feeding. Peptides 2014, 51, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Ernst, M.C.; Sinal, C.J. Chemerin: At the crossroads of inflammation and obesity. Trends Endocrinol. Metab. 2010, 21, 660–667. [Google Scholar] [CrossRef] [PubMed]
- Wittamer, V.; Franssen, J.D.; Vulcano, M.; Mirjolet, J.F.; Le Poul, E.; Migeotte, I.; Brézillon, S.; Tyldesley, R.; Blanpain, C.; Detheux, M.; et al. Specific recruitment of antigen-presenting cells by chemerin, a novel processed ligand from human inflammatory fluids. J. Exp. Med. 2003, 198, 977–985. [Google Scholar] [CrossRef]
- Edinger, A.L.; Hoffman, T.L.; Sharron, M.; Lee, B.; O’Dowd, B.; Doms, R.W. Use of GPR1, GPR15, and STRL33 as coreceptors by diverse human immunodeficiency virus type 1 and simian immunodeficiency virus envelope proteins. Virology 1998, 249, 367–378. [Google Scholar] [CrossRef][Green Version]
- Shimizu, N.; Soda, Y.; Kanbe, K.; Liu, H.Y.; Jinno, A.; Hoshino, H.; Kitamura, T. An orphan G protein-coupled receptor, GPR1, acts as a coreceptor to allow replication of human immunodeficiency virus types 1 and 2 in brain- derived cells. J. Virol. 1999, 73, 5231–5239. [Google Scholar] [CrossRef]
- Reverchon, M.; Ramé, C.; Bertoldo, M.; Dupont, J. Adipokines and the Female Reproductive Tract. Int. J. Endocrinol. 2014, 2014, 232454. [Google Scholar]
- Zabel, B.A.; Nakae, S.; Zúñiga, L.; Kim, J.Y.; Ohyama, T.; Alt, C.; Pan, J.; Suto, H.; Soler, D.; Allen, S.J.; et al. Mast cell-expressed orphan receptor CCRL2 binds chemerin and is required for optimal induction of IgE-mediated passive cutaneous anaphylaxis. J. Exp. Med. 2008, 205, 2207–2220. [Google Scholar] [CrossRef]
- Migeotte, I.; Franssen, J.D.; Goriely, S.; Willems, F.; Parmentier, M. Distribution and regulation of expression of the putative human chemokine receptor HCR in leukocyte populations. Eur. J. Immunol. 2002, 32, 494–501. [Google Scholar] [CrossRef]
- Yang, X.; Yao, J.; Wei, Q.; Ye, J.; Yin, X.; Quan, X.; Lan, Y.; Xing, H. Role of chemerin/CMKLR1 in the maintenance of early pregnancy. Front. Med. 2018, 12, 525–532. [Google Scholar] [CrossRef] [PubMed]
- Helfer, G.; Ross, A.W.; Thomson, L.M.; Mayer, C.D.; Stoney, P.N.; McCaffery, P.J.; Morgan, P.J. A neuroendocrine role for chemerin in hypothalamic remodelling and photoperiodic control of energy balance. Sci. Rep. 2016, 6, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Smolinska, N.; Kiezun, M.; Dobrzyn, K.; Rytelewska, E.; Kisielewska, K.; Gudelska, M.; Zaobidna, E.; Bogus-Nowakowska, K.; Wyrebek, J.; Bors, K.; et al. Expression of chemerin and its receptors in the porcine hypothalamus and plasma chemerin levels during the oestrous cycle and early pregnancy. Int. J. Mol. Sci. 2019, 20, 3887. [Google Scholar] [CrossRef] [PubMed]
- Kisielewska, K.; Rytelewska, E.; Gudelska, M.; Kiezun, M.; Dobrzyn, K.; Bogus-Nowakowska, K.; Kaminska, B.; Smolinska, N.; Kaminski, T. Relative abundance of chemerin mRNA transcript and protein in pituitaries of pigs during the estrous cycle and early pregnancy and associations with LH and FSH secretion during the estrous cycle. Anim. Reprod. Sci. 2020, 219, 106532. [Google Scholar] [CrossRef]
- Song, S.H.; Fukui, K.; Nakajima, K.; Kozakai, T.; Sasaki, S.; Roh, S.G.; Katoh, K. Cloning, expression analysis, and regulatory mechanisms of bovine chemerin and chemerin receptor. Domest. Anim. Endocrinol. 2010, 39, 97–105. [Google Scholar] [CrossRef]
- Brunetti, L.; Di Nisio, C.; Recinella, L.; Chiavaroli, A.; Leone, S.; Ferrante, C.; Orlando, G.; Vacca, M. Effects of vaspin, chemerin and omentin-1 on feeding behavior and hypothalamic peptide gene expression in the rat. Peptides 2011, 32, 1866–1871. [Google Scholar] [CrossRef]
- Carlino, C.; Trotta, E.; Stabile, H.; Morrone, S.; Bulla, R.; Soriani, A.; Iannitto, M.L.; Agostinis, C.; Mocci, C.; Minozzi, M.; et al. Chemerin regulates NK cell accumulation and endothelial cell morphogenesis in the decidua during early pregnancy. J. Clin. Endocrinol. Metab. 2012, 97, 3603–3612. [Google Scholar] [CrossRef]
- Jin, C.H.; Yi, K.W.; Ha, Y.R.; Shin, J.-H.; Park, H.T.; Kim, T.; Hur, J.-Y. Chemerin Expression in the Peritoneal Fluid, Serum, and Ovarian Endometrioma of Women with Endometriosis. Am. J. Reprod. Immunol. 2015, 74, 379–386. [Google Scholar] [CrossRef]
- Garces, M.F.; Sanchez, E.; Acosta, B.J.; Angel, E.; Ruíz, A.I.; Rubio-Romero, J.A.; Diéguez, C.; Nogueiras, R.; Caminos, J.E. Expression and regulation of chemerin during rat pregnancy. Placenta 2012, 33, 373–378. [Google Scholar] [CrossRef]
- Reverchon, M.; Cornuau, M.; Ram, C.; Guerif, F.; Royre, D.; Dupont, J. Chemerin inhibits IGF-1-induced progesterone and estradiol secretion in human granulosa cells. Hum. Reprod. 2012, 27, 1790–1800. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Kim, J.Y.; Xue, K.; Liu, J.Y.; Leader, A.; Tsang, B.K. Chemerin, a novel regulator of follicular steroidogenesis and its potential involvement in polycystic ovarian syndrome. Endocrinology 2012, 153, 5600–5611. [Google Scholar] [CrossRef] [PubMed]
- Gudelska, M.; Dobrzyn, K.; Kiezun, M.; Rytelewska, E.; Kisielewska, K.; Kaminska, B.; Kaminski, T.; Smolinska, N. The expression of chemerin and its receptors (CMKLR1, GPR1, CCRL2) in the porcine uterus during the oestrous cycle and early pregnancy and in trophoblasts and conceptuses. Animal 2020, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Rytelewska, E.; Kisielewska, K.; Kiezun, M.; Kamil, D.; Marlena, G.; Rak, A.; Dupont, J.; Kaminska, B.; Kaminski, T.; Smolinska, N. Expression of chemerin and its receptors in the ovaries of prepubertal and mature gilts. Mol. Reprod. Dev. 2020, in press. [Google Scholar] [CrossRef]
- Reverchon, M.; Bertoldo, M.J.; Ramé, C.; Froment, P.; Dupont, J. CHEMERIN (RARRES2) Decreases In Vitro Granulosa Cell Steroidogenesis and Blocks Oocyte Meiotic Progression in Bovine Species1. Biol. Reprod. 2014, 90, 1–15. [Google Scholar] [CrossRef]
- Yang, Y.L.; Ren, L.R.; Sun, L.F.; Huang, C.; Xiao, T.X.; Wang, B.B.; Chen, J.; Zabel, B.A.; Ren, P.; Zhang, J.V. The role of GPR1 signaling in mice corpus luteum. J. Endocrinol. 2016, 230, 55–65. [Google Scholar] [CrossRef][Green Version]
- Schwanhüusser, B.; Busse, D.; Li, N.; Dittmar, G.; Schuchhardt, J.; Wolf, J.; Chen, W.; Selbach, M. Global quantification of mammalian gene expression control. Nature 2011, 473, 337–342. [Google Scholar] [CrossRef]
- Gry, M.; Rimini, R.; Strömberg, S.; Asplund, A.; Pontén, F.; Uhlén, M.; Nilsson, P. Correlations between RNA and protein expression profiles in 23 human cell lines. BMC Genomics 2009, 10. [Google Scholar] [CrossRef]
- De Sousa Abreu, R.; Penalva, L.O.; Marcotte, E.M.; Vogel, C. Global signatures of protein and mRNA expression levels. Mol. Biosyst. 2009, 5, 1512–1526. [Google Scholar] [CrossRef]
- Liu, J. Control of protein synthesis and mRNA degradation by microRNAs. Curr. Opin. Cell Biol. 2008, 20, 214–221. [Google Scholar] [CrossRef]
- Yang, Z.-M.; Das, S.K.; Wang, J.; Sugimoto, Y.; Ichikawa, A.; Dey, S.K. Potential Sites of Prostaglandin Actions in the Periimplantation Mouse Uterus: Differential Expression and Regulation of Prostaglandin Receptor Genes1. Biol. Reprod. 1997, 56, 368–379. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kaczynski, P.; Waclawik, A. Effect of conceptus on expression of prostaglandin F2α receptor in the porcine endometrium. Theriogenology 2013, 79, 784–790. [Google Scholar] [CrossRef] [PubMed]
- Waclawik, A.; Jabbour, H.N.; Blitek, A.; Ziecik, A.J. Estradiol-17beta, prostaglandin E2 (PGE2), and the PGE2 receptor are involved in PGE2 positive feedback loop in the porcine endometrium. Endocrinology 2009, 150, 3823–3832. [Google Scholar] [CrossRef] [PubMed]
- Waclawik, A.; Ziecik, A.J. Differential expression of prostaglandin (PG) synthesis enzymes in conceptus during peri-implantation period and endometrial expression of carbonyl reductase/PG 9-ketoreductase in the pig. J. Endocrinol. 2007, 194, 499–510. [Google Scholar] [CrossRef]
- Perry, J.S.; Heap, R.B.; Amoroso, E.C. Steroid hormone production by pig blastocysts. Nature 1973, 245, 45–47. [Google Scholar] [CrossRef]
- Bazer, F.W.; Thatcher, W.W. Theory of maternal recognition of pregnancy in swine based on estrogen controlled endocrine versus exocrine secretion of prostaglandin F2alpha by the uterine endometrium. Prostaglandins 1977, 14, 397–400. [Google Scholar] [CrossRef]
- Akinlosotu, B.A.; Diehl, J.R.; Gimenez, T. Prostaglandin E2 counteracts the effects of PGF2 alpha in indomethacin treated cycling gilts. Prostaglandins 1988, 35, 81–93. [Google Scholar] [CrossRef]
- Christenson, L.K.; Anderson, L.H.; Ford, S.P.; Farley, D.B. Luteal maintenance during early pregnancy in the pig: Role for prostaglandin E2. Prostaglandins 1994, 47, 61–75. [Google Scholar] [CrossRef]
- Kennedy, T.G.; Lukash, L.A. Induction of Decidualization in Rats by the Intrauterine Infusion of Prostaglandins1. Biol. Reprod. 1982, 27, 253–260. [Google Scholar] [CrossRef]
- Tobert, J.A. A study of the possible role of prostaglandins in decidualization using a nonsurgical method for the instillation of fluids into the rat uterine lumen. J. Reprod. Fertil. 1976, 47, 391–393. [Google Scholar] [CrossRef]
- Nakamura, N.; Naruse, K.; Kobayashi, Y.; Miyabe, M.; Saiki, T.; Enomoto, A.; Takahashi, M.; Matsubara, T. Chemerin promotes angiogenesis in vivo. Physiol. Rep. 2018, 6, e13962. [Google Scholar] [CrossRef] [PubMed]
- Kaur, J.; Adya, R.; Tan, B.K.; Chen, J.; Randeva, H.S. Identification of chemerin receptor (ChemR23) in human endothelial cells: Chemerin-induced endothelial angiogenesis. Biochem. Biophys. Res. Commun. 2010, 391, 1762–1768. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, A.J.; Yang, P.; Read, C.; Kuc, R.E.; Yang, L.; Taylor, E.J.A.; Taylor, C.W.; Maguire, J.J.; Davenport, A.P. Chemerin Elicits Potent Constrictor Actions via Chemokine-Like Receptor 1 (CMKLR1), not G-Protein-Coupled Receptor 1 (GPR1), in Human and Rat Vasculature. J. Am. Heart Assoc. 2016, 5. [Google Scholar] [CrossRef] [PubMed]
- Rourke, J.L.; Muruganandan, S.; Dranse, H.J.; McMullen, N.M.; Sinal, C.J. Gpr1 is an active chemerin receptor influencing glucose homeostasis in obese mice. J. Endocrinol. 2014, 222, 201–215. [Google Scholar] [CrossRef]
- Huang, B.; Huang, C.; Zhao, H.; Zhu, W.; Wang, B.; Wang, H.; Chen, J.; Iao, T.; Niu, J.; Zhang, J. Impact of GPR1 signaling on maternal high-fat feeding and placenta metabolism in mice. Am. J. Physiol. Endocrinol. Metab. 2019, 316, E987–E997. [Google Scholar] [CrossRef]
- Anegon, I.; Cuturi, M.C.; Godard, A.; Moreau, M.; Terqui, M.; Martinat-Botté, F.; Soulillou, J.P. Presence of leukaemia inhibitory factor and interleukin 6 in porcine uterine secretions prior to conceptus attachment. Cytokine 1994, 6, 493–499. [Google Scholar] [CrossRef]
- Vaughan, T.J.; James, P.S.; Pascall, J.C.; Brown, K.D. Expression of the genes for TGF alpha, EGF and the EGF receptor during early pig development. Development 1992, 116, 663–669. [Google Scholar]
- Jaeger, L.A.; Spiegel, A.K.; Ing, N.H.; Johnson, G.A.; Bazer, F.W.; Burghardt, R.C. Functional effects of transforming growth factor beta on adhesive properties of porcine trophectoderm. Endocrinology 2005, 146, 3933–3942. [Google Scholar] [CrossRef]
- Cencič, A.; Guillomot, M.; Koren, S.; La Bonnardière, C. Trophoblastic interferons: Do they modulate uterine cellular markers at the time of conceptus attachment in the pig? Placenta 2003, 24, 862–869. [Google Scholar] [CrossRef]
- Jeong, W.; Kim, J.; Bazer, F.W.; Song, G.; Kim, J. Stimulatory effects of interleukin-1 beta on development of porcine uterine epithelial cell are mediated by activation of the ERK1/2 MAPK cell signaling cascade. Mol. Cell. Endocrinol. 2016, 419, 225–234. [Google Scholar] [CrossRef]
- Hayden, M.S.; Ghosh, S. Regulation of NF-κB by TNF family cytokines. Semin. Immunol. 2014, 26, 253–266. [Google Scholar] [CrossRef] [PubMed]
- Geisert, R.D.; Lucy, M.C.; Whyte, J.J.; Ross, J.W.; Mathew, D.J. Cytokines from the pig conceptus: Roles in conceptus development in pigs. J. Anim. Sci. Biotechnol. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Parhar, R.S.; Yagel, S.; Lala, P.K. PGE2-mediated immunosuppression by first trimester human decidual cells blocks activation of maternal leukocytes in the decidua with potential anti-trophoblast activity. Cell. Immunol. 1989, 120, 61–74. [Google Scholar] [CrossRef]
- Pollard, J.K.; Mitchell, M.D. Intrauterine infection and the effects of inflammatory mediators on prostaglandin production by myometrial cells from pregnant women. Am. J. Obstet. Gynecol. 1996, 174, 682–686. [Google Scholar] [CrossRef]
- Cash, J.L.; Hart, R.; Russ, A.; Dixon, J.P.C.; Colledge, W.H.; Doran, J.; Hendrick, A.G.; Carlton, M.B.L.; Greaves, D.R. Synthetic chemerin-derived peptides suppress inflammation through ChemR23. J. Exp. Med. 2008, 205, 767–775. [Google Scholar] [CrossRef] [PubMed]
- Geisert, R.D.; Yelich, J. V Regulation of conceptus development and attachment in pigs. J. Reprod. Fertil. Suppl. 1997, 52, 133–149. [Google Scholar] [PubMed]
- Akins, E.L.; Morrissette, M.C. Oferta. Am. J. Vet. Res. 1968, 29, 1953–1957. [Google Scholar]
- Anderson, L.L. Growth, protein content and distribution of early pig embryos. Anat. Rec. 1978, 190, 143–153. [Google Scholar] [CrossRef]
- Nitkiewicz, A.; Smolinska, N.; Maleszka, A.; Kiezun, M.; Kaminski, T. Localization of orexin A and orexin B in the porcine uterus. Reprod. Biol. 2012, 12, 135–155. [Google Scholar] [CrossRef]
- Dobrzyn, K.; Smolinska, N.; Szeszko, K.; Kiezun, M.; Maleszka, A.; Rytelewska, E.; Kaminski, T. Effect of progesterone on adiponectin system in the porcine uterus during early pregnancy. J. Anim. Sci. 2017, 95, 338–352. [Google Scholar] [CrossRef]
- Smolinska, N.; Dobrzyn, K.; Kiezun, M.; Szeszko, K.; Maleszka, A.; Kaminski, T. Effect of adiponectin on the steroidogenic acute regulatory protein, P450 side chain cleavage enzyme and 3β-hydroxysteroid dehydrogenase gene expression, progesterone and androstenedione production by the porcine uterus during early pregnancy. J. Physiol. Pharmacol. 2016, 67, 443–456. [Google Scholar] [PubMed]
- Morgan, G.L.; Geisert, R.D.; Zavy, M.T.; Shawley, R.V.; Fazleabas, A.T. Development of pig blastocysts in a uterine environment advanced by exogenous oestrogen. J. Reprod. Fertil. 1987, 80, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Gregoraszczuk, E.L.; Michas, N. Progesterone and estradiol secretion by porcine luteal cells is influenced by individual and combined treatment with prostaglandins E2 and F(2a) throughout the estrus cycle. Prostaglandins Other Lipid Mediat. 1999, 57, 231–241. [Google Scholar] [CrossRef]
- Nitkiewicz, A.; Smolinska, N.; Przala, J.; Kaminski, T. Expression of orexin receptors 1 (OX1R) and 2 (OX2R) in the porcine ovary during the oestrous cycle. Regul. Pept. 2010, 165, 186–190. [Google Scholar] [CrossRef] [PubMed]
- Spagnuolo-Weaver, M.; Fuerst, R.; Campbell, S.T.; Meehan, B.M.; McNeilly, F.; Adair, B.; Allan, G. A fluorimeter-based RT-PCR method for the detection and quantitation of porcine cytokines. J. Immunol. Methods 1999, 230, 19–27. [Google Scholar] [CrossRef]

| Gene | Primers Sequences | Accession Number | Primer, nM | Reaction Conditions | Reference | |
|---|---|---|---|---|---|---|
| RARRES2 | F: 5′-TGGAGGAGTTCCACAAGCAC-3′ | EU660865 | 500 | 40 | [14] | |
| R: 5′-GCTTTCTTCCAGTCCCTCTTC-3′ | 500 | |||||
| CCRL2 | F: 5′-GAGCAGCAGCTACTTACTTCC-3′ | NM_001001617.1 | 200 | 40 | [14] | |
| R: 5′-CTGCCCACTGACCGAGTTC-3′ | 200 | |||||
| CMKLR1 | F: 5′-GGACTACCACTGGGTGTTCG-3′ | EU660866 | 200 | 40 | [14] | |
| R: 5′-GCCATGTAAGCCAGTCGGA-3′ | 200 | |||||
| GPR1 | F: 5′-ACCGACTTGGAGGAGAAAGC -3’ | FJ234899.1 | 200 | 40 | [14] | |
| R: 5′-ATTGAGGAACCAGAGCGTGG -3’ | 200 | |||||
| PPIA | F: 5′-GCACTGGTGGCAAGTCCAT-3’ | U48832 | 300 | 40 | [64] | |
| R: 5′-AGGACCCGTATGCTTCAGGA-3’ | 300 | |||||
| ACTB | F: 5′-ACATCAAGGAGAAGCTCTGCTACG-3’ | U07786 | 500 | 40 | [65] | |
| R: 5′-GAGGGGCGATGATCTTGATCTTCA-3’ | 500 | |||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dobrzyn, K.; Kiezun, M.; Zaobidna, E.; Kisielewska, K.; Rytelewska, E.; Gudelska, M.; Kopij, G.; Bors, K.; Szymanska, K.; Kaminska, B.; et al. The In Vitro Effect of Prostaglandin E2 and F2α on the Chemerin System in the Porcine Endometrium during Gestation. Int. J. Mol. Sci. 2020, 21, 5213. https://doi.org/10.3390/ijms21155213
Dobrzyn K, Kiezun M, Zaobidna E, Kisielewska K, Rytelewska E, Gudelska M, Kopij G, Bors K, Szymanska K, Kaminska B, et al. The In Vitro Effect of Prostaglandin E2 and F2α on the Chemerin System in the Porcine Endometrium during Gestation. International Journal of Molecular Sciences. 2020; 21(15):5213. https://doi.org/10.3390/ijms21155213
Chicago/Turabian StyleDobrzyn, Kamil, Marta Kiezun, Ewa Zaobidna, Katarzyna Kisielewska, Edyta Rytelewska, Marlena Gudelska, Grzegorz Kopij, Kinga Bors, Karolina Szymanska, Barbara Kaminska, and et al. 2020. "The In Vitro Effect of Prostaglandin E2 and F2α on the Chemerin System in the Porcine Endometrium during Gestation" International Journal of Molecular Sciences 21, no. 15: 5213. https://doi.org/10.3390/ijms21155213
APA StyleDobrzyn, K., Kiezun, M., Zaobidna, E., Kisielewska, K., Rytelewska, E., Gudelska, M., Kopij, G., Bors, K., Szymanska, K., Kaminska, B., Kaminski, T., & Smolinska, N. (2020). The In Vitro Effect of Prostaglandin E2 and F2α on the Chemerin System in the Porcine Endometrium during Gestation. International Journal of Molecular Sciences, 21(15), 5213. https://doi.org/10.3390/ijms21155213










