Insulin Resistance Does Not Impair Mechanical Overload-Stimulated Glucose Uptake, but Does Alter the Metabolic Fate of Glucose in Mouse Muscle
Abstract
1. Introduction
2. Results
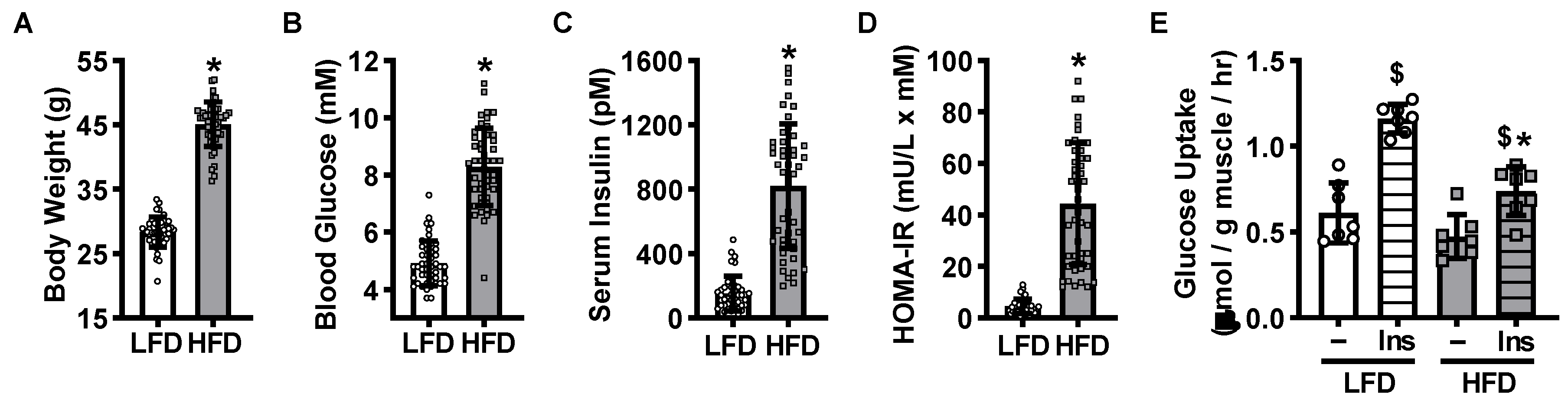
2.1. High-Fat Diet-Induced Mouse Model of Insulin Resistance
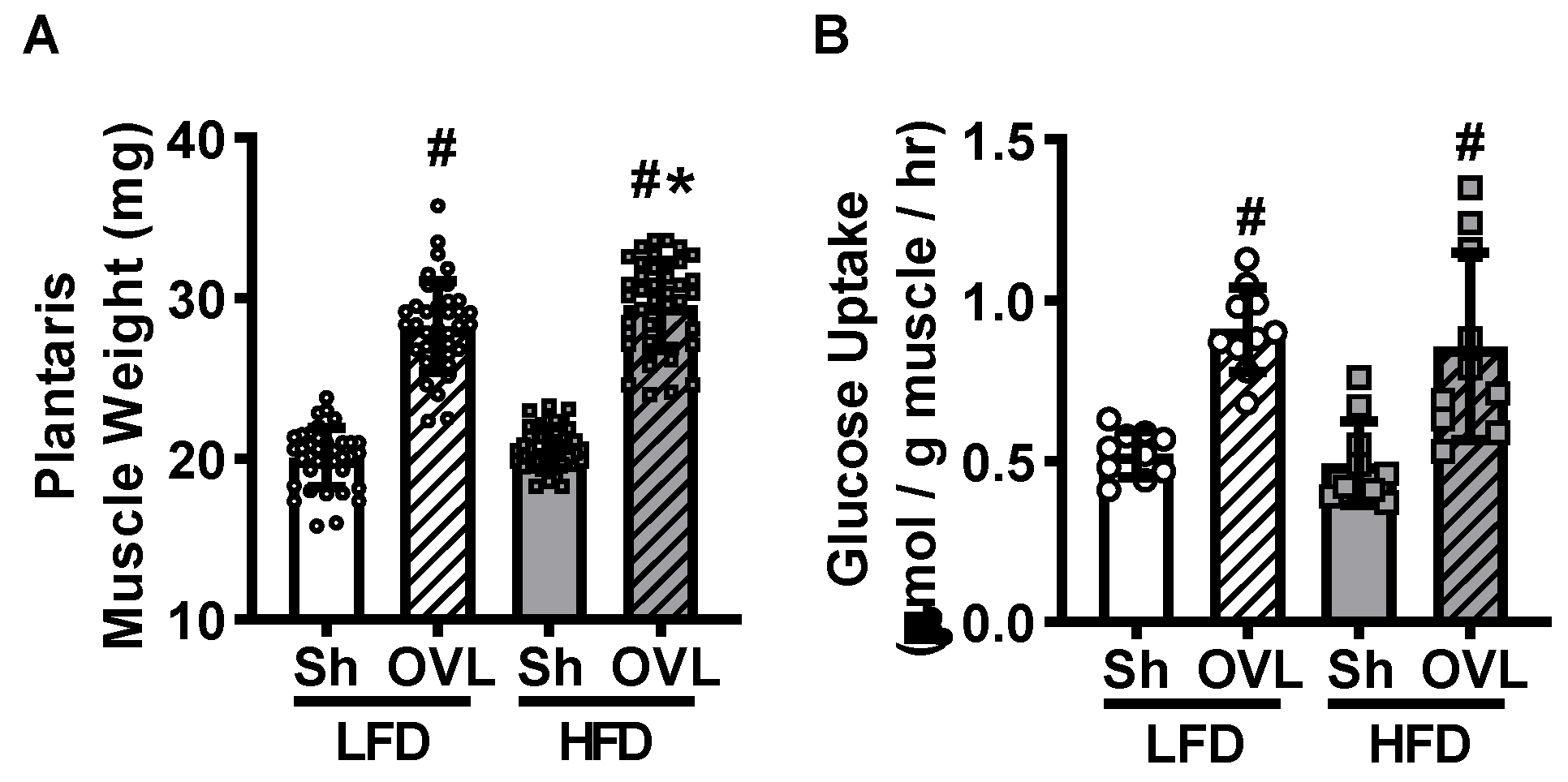
2.2. Insulin Resistance Does Not Impair Overload-Stimulated Muscle Glucose Uptake
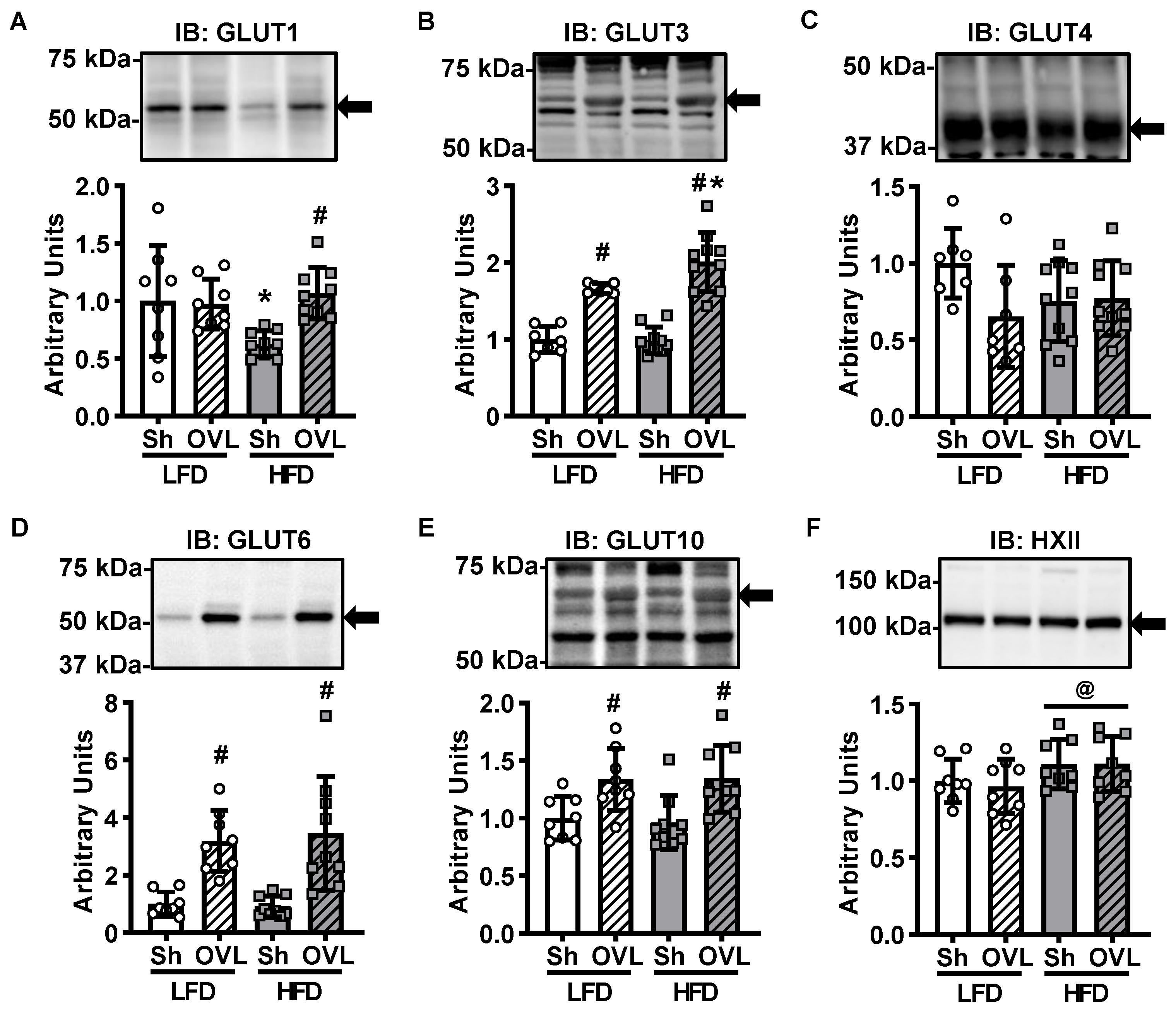
2.3. Insulin Resistance Does Not Impair Overload-Stimulated Glucose Transporter Expression
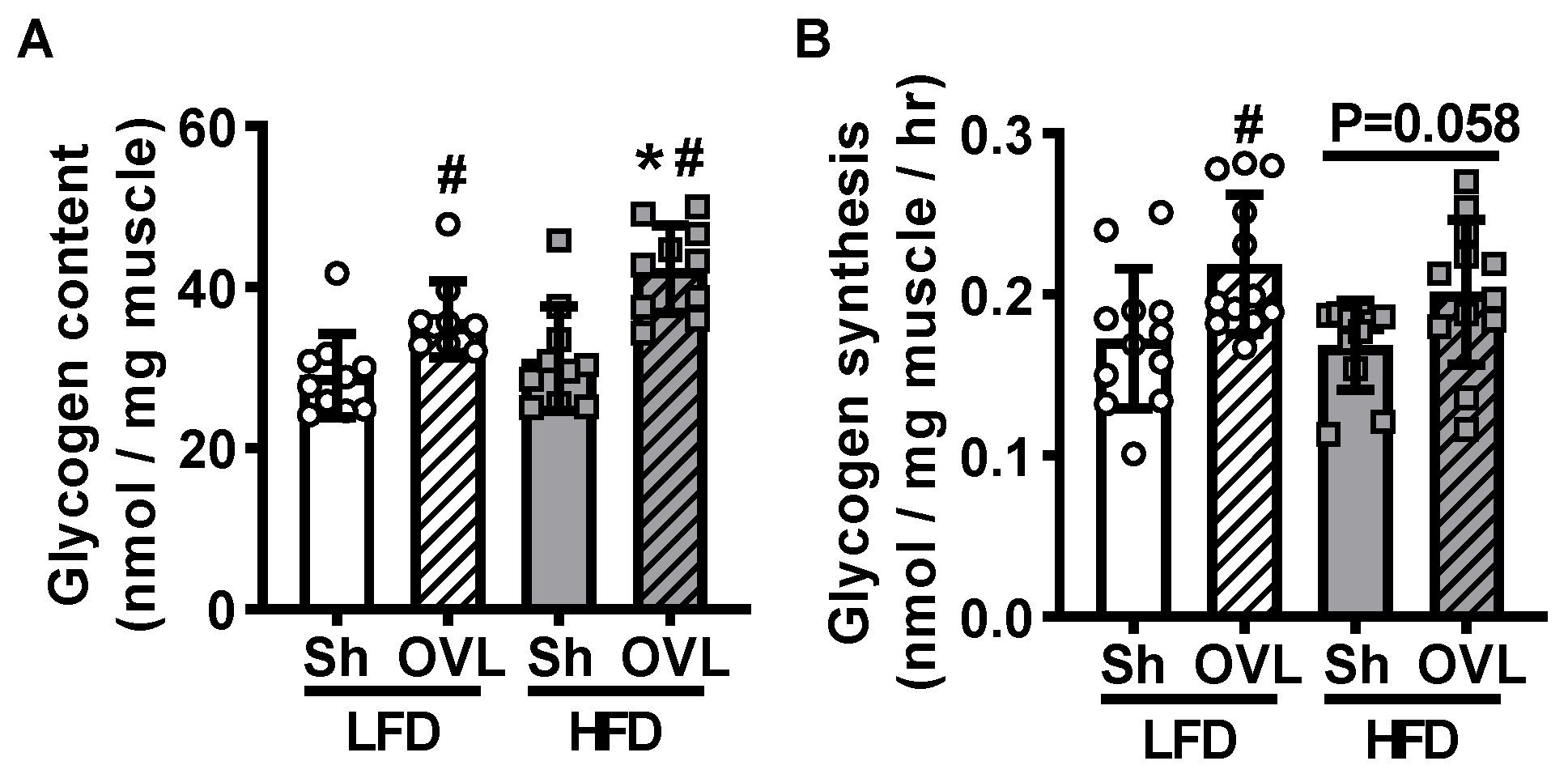
2.4. Insulin Resistance Enhances Overload-Stimulated Accumulation of Muscle Glycogen
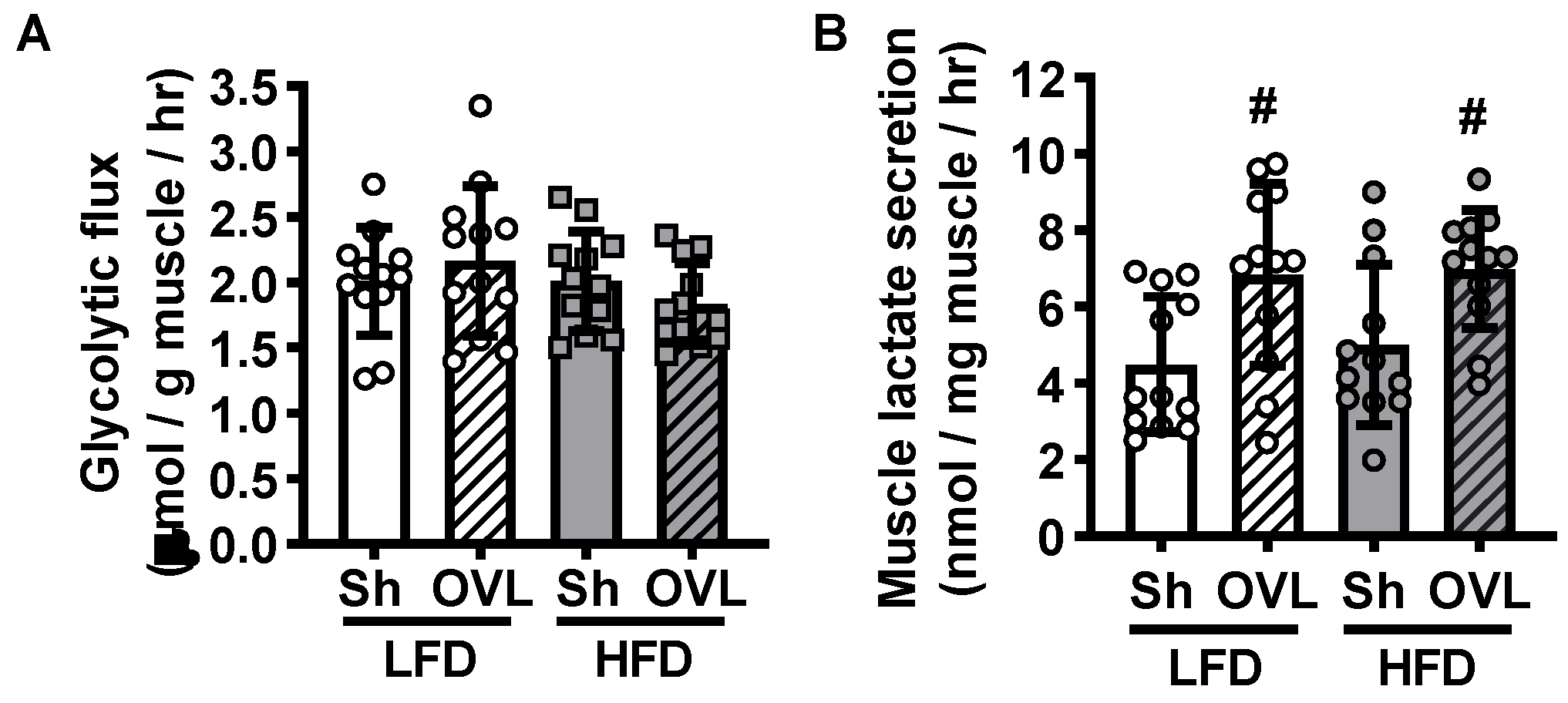
2.5. Overload Does Not Alter Glycolytic Flux in Insulin Sensitive or Insulin Resistant Muscle
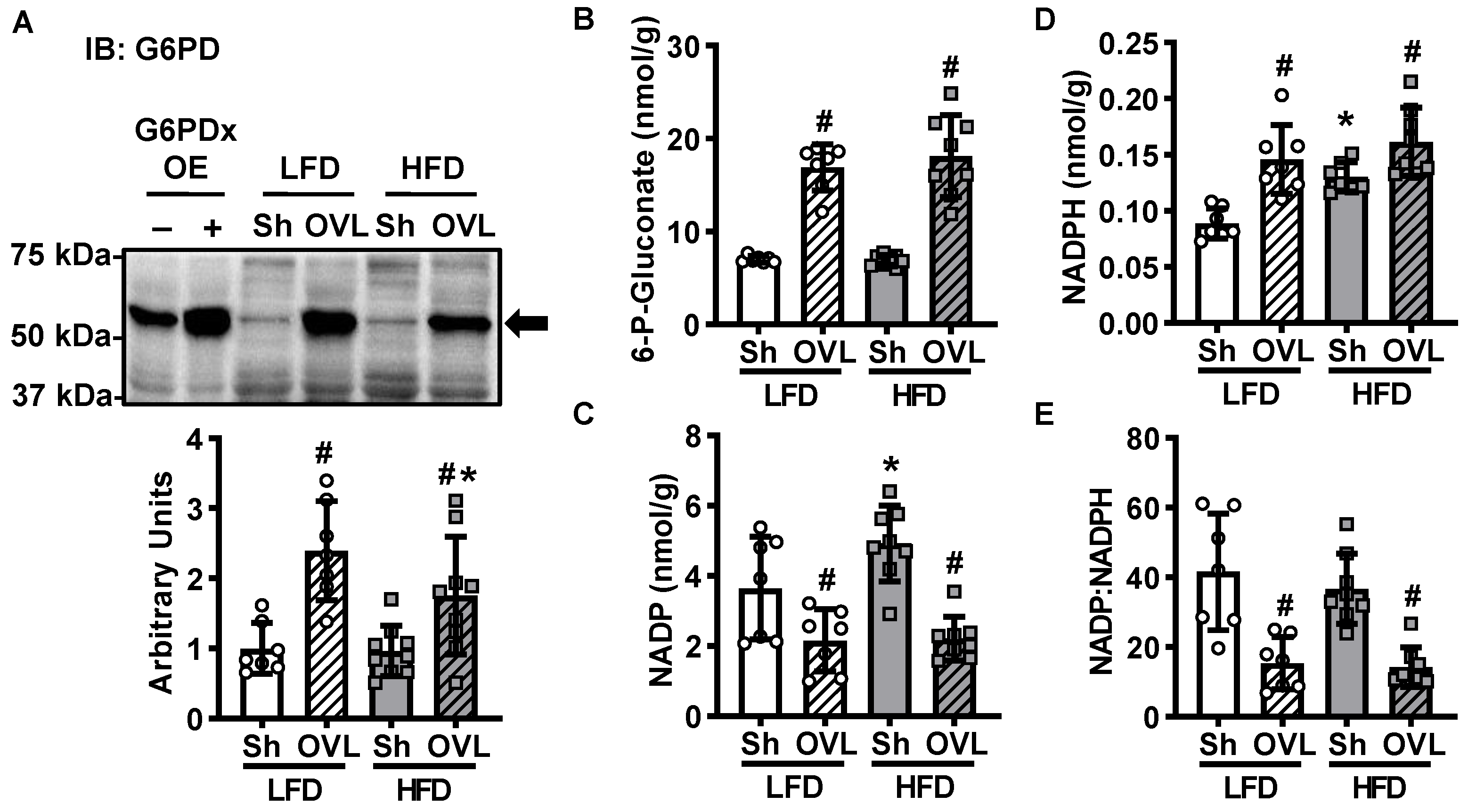
2.6. Overload-Stimulated Activation of the Pentose Phosphate Pathway Is Not Impaired by Insulin Resistance
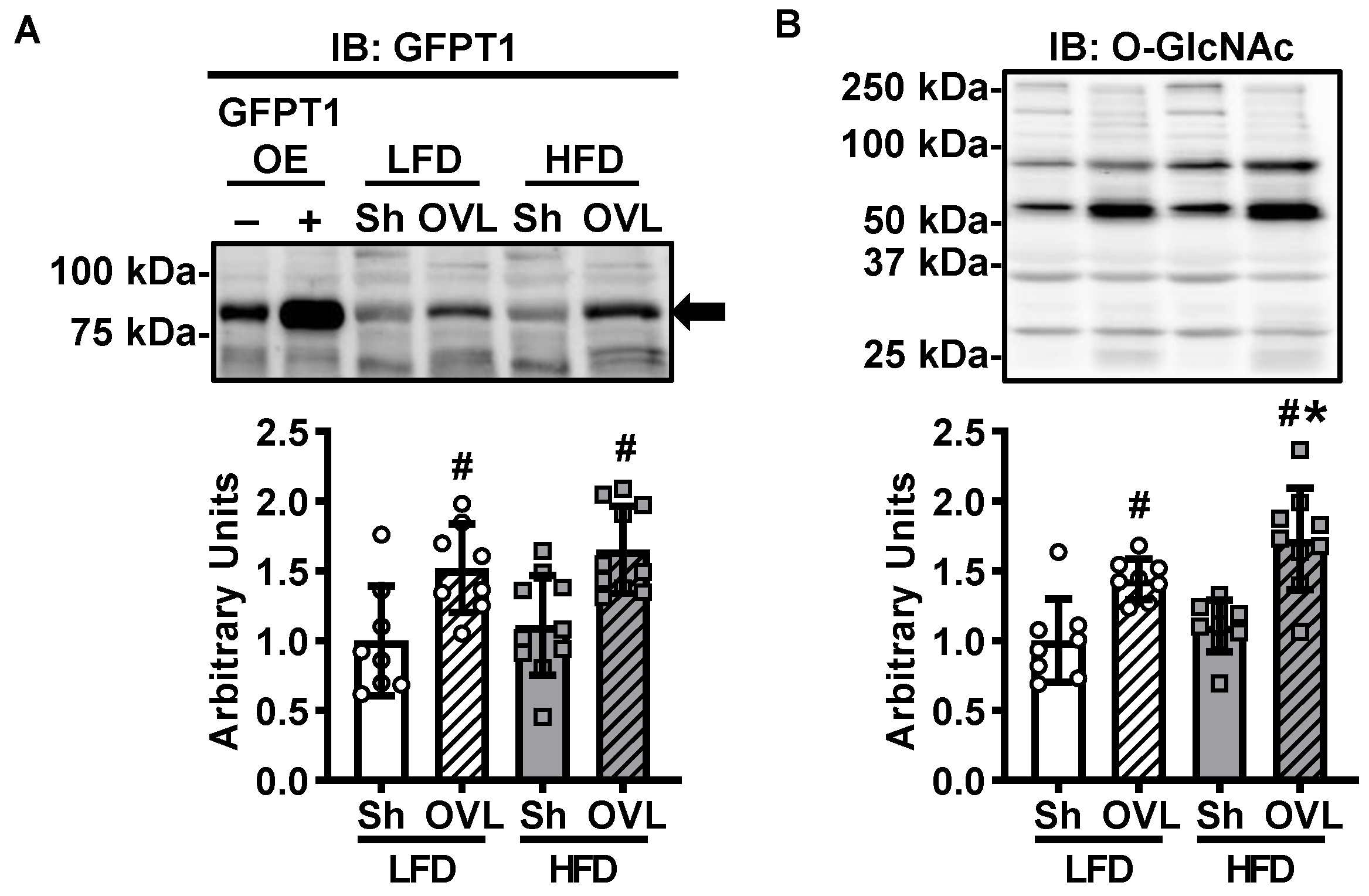
2.7. Overload-Stimulated Activation of the Hexosamine Pathway Is Enhanced by Insulin Resistance
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Systemic Insulin Resistance Assessment
4.3. Unilateral Synergist Ablation Surgery
4.4. Muscle [3H]-2-Deoxyglucose Uptake
4.5. Muscle Glycogen Content
4.6. Muscle Glycolytic Flux and Lactate Secretion
4.7. Muscle Glycogen Synthesis
4.8. Pentose Phosphate Pathway Metabolites
4.9. Immunoblot Analyses
4.10. Statistical Analyses.
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| G6PD | Glucose-6-phosphate dehydrogenase |
| GFPT1 | Glutamine fructose-6-phosphate transaminase 1 |
| GLUT | Glucose transporter |
| HFD | High-fat diet |
| HOMA-IR | Homeostatic model assessment of insulin resistance |
| KRB | Krebs-Ringer-Bicarbonate |
| LFD | Low-fat diet |
| NADP(H) | Nicotinamide adenine dinucleotide phosphate |
| SDS-PAGE | Sodium dodecyl sulfate-polyacrylamide gel electrophoresis |
| UPLC-MRM/MS | Ultra performance liquid chromatography-multiple reaction monitoring mass spectrometry |
References
- Knowler, W.C.; Barrett-Connor, E.; Fowler, S.E.; Hamman, R.F.; Lachin, J.M.; Walker, E.A.; Nathan, D.M. Diabetes Prevention Program Research Group Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N. Engl. J. Med. 2002, 346, 393–403. [Google Scholar]
- Colberg, S.R.; Sigal, R.J.; Fernhall, B.; Regensteiner, J.G.; Blissmer, B.J.; Rubin, R.R.; Chasan-Taber, L.; Albright, A.L.; Braun, B.; American College of Sports Medicine; et al. Exercise and type 2 diabetes: The American College of Sports Medicine and the American Diabetes Association: Joint position statement. Diabetes Care 2010, 33, e147–e167. [Google Scholar] [CrossRef] [PubMed]
- Jorge, M.L.M.P.; de Oliveira, V.N.; Resende, N.M.; Paraiso, L.F.; Calixto, A.; Diniz, A.L.D.; Resende, E.S.; Ropelle, E.R.; Carvalheira, J.B.; Espindola, F.S.; et al. The effects of aerobic, resistance, and combined exercise on metabolic control, inflammatory markers, adipocytokines, and muscle insulin signaling in patients with type 2 diabetes mellitus. Metabolism 2011, 60, 1244–1252. [Google Scholar] [CrossRef] [PubMed]
- Bacchi, P.E.; Moghetti, N.C.; Zanolin, M.E.; Milanese, C.; Faccioli, N.; Trombetta, M.; Zoppini, G.; Cevese, A.; Bonadonna, R.C.; Schena, F.; et al. Metabolic effects of aerobic training and resistance training in type 2 diabetic subjects. Diabetes Care 2012, 35, 676–682. [Google Scholar] [CrossRef] [PubMed]
- Baldi, J.; Snowling, N. Resistance training improves glycaemic control in obese type 2 diabetic men. Int. J. Sports Med. 2003, 24, 419–423. [Google Scholar]
- Castaneda, F.; Layne, J.E.; Castaneda, C. Skeletal muscle sodium glucose co-transporters in older adults with type 2 diabetes undergoing resistance training. Int. J. Med. Sci. 2006, 3, 84–91. [Google Scholar] [CrossRef]
- Ferrara, C.M.; Goldberg, A.P.; Ortmeyer, H.K.; Ryan, A.S. Effects of aerobic and resistive exercise training on glucose disposal and skeletal muscle metabolism in older men. J. Gerontol. Ser. A 2006, 61, 480–487. [Google Scholar] [CrossRef]
- Ishii, T.; Yamakita, T.; Sato, T.; Tanaka, S.; Fujii, S. Resistance training improves insulin sensitivity in NIDDM subjects without altering maximal oxygen uptake. Diabetes Care 1998, 21, 1353–1355. [Google Scholar] [CrossRef]
- Augert, G.; Van de Werve, G.; Le Marchand-Brustel, Y. Effect of work-induced hypertrophy on muscle glucose metabolism in lean and obese mice. Diabetologia 1985, 28, 295–301. [Google Scholar] [CrossRef]
- Holten, M.K.; Zacho, M.; Gaster, M.; Juel, C.; Wojtaszewski, J.F.P.; Dela, F. Strength Training Increases Insulin-Mediated Glucose Uptake, GLUT4 Content, and Insulin Signaling in Skeletal Muscle in Patients With Type 2 Diabetes. Diabetes 2004, 53, 294–305. [Google Scholar] [CrossRef]
- Patti, M.E.; Virkamäki, A.; Landaker, E.J.; Kahn, C.R.; Yki-Järvinen, H. Activation of the hexosamine pathway by glucosamine in vivo induces insulin resistance of early postreceptor insulin signaling events in skeletal muscle. Diabetes 1999, 48, 1562–1571. [Google Scholar] [CrossRef] [PubMed]
- Cooksey, R.C.; McClain, D.A. Transgenic mice overexpressing the rate-limiting enzyme for hexosamine synthesis in skeletal muscle or adipose tissue exhibit total body insulin resistance. Ann. N. Y. Acad. Sci. 2006, 967, 102–111. [Google Scholar] [CrossRef]
- Gulve, E.A.; Ren, J.M.; Marshall, B.A.; Gao, J.; Hansen, P.A.; Holloszy, J.O.; Mueckler, M. Glucose transport activity in skeletal muscles from transgenic mice overexpressing GLUT1: Increased basal transport is associated with a defective response to diverse stimuli that activate GLUT4. J. Biol. Chem. 1994, 269, 18366–18370. [Google Scholar] [PubMed]
- Barnes, B.R.; Marklund, S.; Steiler, T.L.; Walter, M.; Hjälm, G.; Amarger, V.; Mahlapuu, M.; Leng, Y.; Johansson, C.; Galuska, D.; et al. The 5′-AMP-activated protein kinase γ3 isoform has a key role in carbohydrate and lipid metabolism in glycolytic skeletal muscle. J. Biol. Chem. 2004, 279, 38441–38447. [Google Scholar] [CrossRef] [PubMed]
- Buse, M.G.; Robinson, K.A.; Marshall, B.A.; Mueckler, M. Differential effects of GLUT1 or GLUT4 overexpression on hexosamine biosynthesis by muscles of transgenic mice. J. Biol. Chem. 1996, 271, 23197–23202. [Google Scholar] [CrossRef] [PubMed]
- Kuo, W.; Lin, J.; Tang, T.K. Human glucose-6-phosphate dehydrogenase (G6PD) gene transforms NIH 3T3 cells and induces tumors in nude mice. Int. J. Cancer 2000, 85, 857–864. [Google Scholar] [CrossRef]
- Tian, W.N.; Braunstein, L.D.; Pang, J.; Stuhlmeier, K.M.; Xi, Q.C.; Tian, X.; Stanton, R.C. Importance of glucose-6-phosphate dehydrogenase activity for cell growth. J. Biol. Chem. 1998, 273, 10609–10617. [Google Scholar] [CrossRef] [PubMed]
- Tixier, V.; Bataille, L.; Etard, C.; Jagla, T.; Weger, M.; DaPonte, J.P.; Strahle, U.; Dickmeis, T.; Jagla, K. Glycolysis supports embryonic muscle growth by promoting myoblast fusion. Proc. Natl. Acad. Sci. USA 2013, 110, 18982–18987. [Google Scholar] [CrossRef] [PubMed]
- Tanner, L.B.; Goglia, A.G.; Wei, M.H.; Sehgal, T.; Parsons, L.R.; Park, J.O.; White, E.; Toettcher, J.E.; Rabinowitz, J.D. Four key steps control glycolytic flux in mammalian cells. Cell Syst. 2018, 7, 49–62. [Google Scholar] [CrossRef]
- Castaneda, C.; Layne, J.E.; Munoz-Orians, L.; Gordon, P.L.; Walsmith, J.; Foldvari, M.; Roubenoff, R.; Tucker, K.L.; Nelson, M.E. A randomized controlled trial of resistance exercise training to improve glycemic control in older adults with type 2 diabetes. Diabetes Care 2002, 25, 2335–2341. [Google Scholar] [CrossRef]
- Haun, C.T.; Vann, C.G.; Osburn, S.C.; Mumford, P.W.; Roberson, P.A.; Romero, M.A.; Fox, C.D.; Johnson, C.A.; Parry, H.A.; Kavazis, A.N.; et al. Muscle fiber hypertrophy in response to 6 weeks of high-volume resistance training in trained young men is largely attributed to sarcoplasmic hypertrophy. PLoS ONE 2019, 14, e0215267. [Google Scholar] [CrossRef] [PubMed]
- Haun, C.T.; Vann, C.G.; Mobley, C.B.; Osburn, S.C.; Mumford, P.W.; Roberson, P.A.; Romero, M.A.; Fox, C.D.; Parry, H.A.; Kavazis, A.N.; et al. Pre-training skeletal muscle fiber size and predominant fiber type best predict hypertrophic responses to 6 weeks of resistance training in previously trained young men. Front. Physiol. 2019, 10, 297. [Google Scholar] [CrossRef] [PubMed]
- Yaspelkis, B.B.; Singh, M.K.; Trevino, B.; Krisan, A.D.; Collins, D.E. Resistance training increases glucose uptake and transport in rat skeletal muscle. Acta Physiol. Scand. 2002, 175, 315–323. [Google Scholar] [CrossRef] [PubMed]
- McMillin, S.L.; Schmidt, D.L.; Kahn, B.B.; Witczak, C.A. GLUT4 is not necessary for overload-induced glucose uptake or hypertrophic growth in mouse skeletal muscle. Diabetes 2017, 66, 1491–1500. [Google Scholar] [CrossRef] [PubMed]
- Chaillou, T.; Lee, J.D.; England, J.H.; Esser, K.A.; McCarthy, J.J. Time course of gene expression during mouse skeletal muscle hypertrophy. J. Appl. Physiol. 2013, 115, 1065–1074. [Google Scholar] [CrossRef] [PubMed]
- Steensberg, A.; van Hall, G.; Keller, C.; Osada, T.; Schjerling, P.; Pedersen, B.K.; Saltin, B.; Febbraio, M.A. Muscle glycogen content and glucose uptake during exercise in humans: Influence of prior exercise and dietary manipulation. J. Physiol. 2002, 541, 273–281. [Google Scholar] [CrossRef] [PubMed]
- Guillet-Deniau, I.; Leturque, A.; Girard, J. Expression and cellular localization of glucose transporters (GLUT1, GLUT3, GLUT4) during differentiation of myogenic cells isolated from rat foetuses. J. Cell Sci. 1994, 107, 487–496. [Google Scholar] [PubMed]
- Burant, C.F.; Bell, G.I. Mammalian Facilitative Glucose Transporters: Evidence for Similar Substrate Recognition Sites in Functionally Monomeric Proteins. Biochemistry 1992, 31, 10414–10420. [Google Scholar] [CrossRef] [PubMed]
- Colville, C.A.; Seatter, M.J.; Jess, T.J.; Gould, G.W.; Thomas, H.M. Kinetic analysis of the liver-type (GLUT2) and brain-type (GLUT3) glucose transporters in Xenopus oocytes: Substrate specificities and effects of transport inhibitors. Biochem. J. 1993, 290, 701–706. [Google Scholar] [CrossRef]
- Maedera, S.; Mizuno, T.; Ishiguro, H.; Ito, T.; Soga, T.; Kusuhara, H. GLUT6 is a lysosomal transporter that is regulated by inflammatory stimuli and modulates glycolysis in macrophages. FEBS Lett. 2019, 593, 195–208. [Google Scholar] [CrossRef]
- Lisinski, I.; Schurmann, A.; Joost, H.-G.; Cushman, S.W.; Al-Hasani, H. Targeting of GLUT6 (formerly GLUT9) and GLUT8 in rat adipose cells. Biochem J. 2001, 358, 517–522. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.-C.; Huang, H.-Y.; Chang, C.-J.; Cheng, C.-H.; Chen, Y.-T. Mitochondrial GLUT10 facilitates dehydroascorbic acid import and protects cells against oxidative stress: Mechanistic insight into arterial tortuosity syndrome. Hum. Mol. Genet. 2010, 19, 3721–3733. [Google Scholar] [CrossRef] [PubMed]
- Scheffer, D.L.; Silva, L.A.; Tromm, C.B.; da Rosa, G.L.; Silveira, P.C.L.; de Souza, C.T.; Latini, A.; Pinho, R.A. Impact of different resistance training protocols on muscular oxidative stress parameters. Appl. Physiol. Nutr. Metab. 2012, 37, 1239–1246. [Google Scholar] [CrossRef]
- Schwane, J.A.; Armstrong, R.B. Effect of training on skeletal muscle injury from downhill running in rats. J. Appl. Physiol. 1983, 55, 969–975. [Google Scholar] [CrossRef] [PubMed]
- Tsuda, S.; Hayashi, T.; Egawa, T. The effects of caffeine on metabolomic responses to muscle contraction in rat skeletal muscle. Nutrients 2019, 11, 1819. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Munk, A.; Nielsen, T.S.; Daughtry, M.R.; Larsson, L.; Li, S.; Høyer, K.F.; Geisler, H.W.; Sulek, K.; Kjøbsted, R.; et al. Skeletal muscle O-GlcNAc transferase is important for muscle energy homeostasis and whole-body insulin sensitivity. Mol. Metab. 2018, 11, 160–177. [Google Scholar] [CrossRef]
- Garg, N.; Thakur, S.; Alex McMahan, C.; Adamo, M.L. High fat diet induced insulin resistance and glucose intolerance are gender-specific in IGF-1R heterozygous mice. Biochem. Biophys. Res. Commun. 2011, 413, 476–480. [Google Scholar] [CrossRef]
- Hong, J.; Stubbins, R.E.; Smith, R.R.; Harvey, A.E.; Núẽz, N.P. Differential susceptibility to obesity between male, female and ovariectomized female mice. Nutr. J. 2009, 8, 11. [Google Scholar] [CrossRef]
- Pettersson, U.S.; Waldén, T.B.; Carlsson, P.-O.; Jansson, L.; Phillipson, M. Female mice are protected against high-fat diet induced metabolic syndrome and increase the regulatory T cell population in adipose tissue. PLoS ONE 2012, 7, e46057. [Google Scholar] [CrossRef]
- Salinero, A.E.; Anderson, B.M.; Zuloaga, K.L. Sex differences in the metabolic effects of diet-induced obesity vary by age of onset. Int. J. Obes. 2018, 42, 1088–1091. [Google Scholar] [CrossRef]
- Ferey, J.L.A.; Brault, J.J.; Smith, C.A.S.; Witczak, C.A. Constitutive activation of CaMKK signaling is sufficient but not necessary for mTORC1 activation and growth in mouse skeletal muscle. AJP Endocrinol. Metab. 2014, 307, E686–E694. [Google Scholar] [CrossRef] [PubMed]
- Hinkley, J.M.; Ferey, J.L.; Brault, J.J.; Smith, C.A.S.; Gilliam, L.A.A.; Witczak, C.A. Constitutively active CaMKKα stimulates skeletal muscle glucose uptake in insulin-resistant mice in vivo. Diabetes 2014, 63, 142–151. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hammerstent, R.H. The use of dowex-l-borate from to separate 3HOH from 2-3h-glucose. Anal. Biochem. 1973, 56, 292–293. [Google Scholar] [CrossRef]
- Bouskila, M.; Hirshman, M.F.; Jensen, J.; Goodyear, L.J.; Sakamoto, K. Insulin promotes glycogen synthesis in the absence of GSK3 phosphorylation in skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 2008, 294, E28–E35. [Google Scholar] [CrossRef]
| Antigen | Blocking | 1° Antibody | 1° Antibody RRID | 2° Antibody | ECL Reagent |
|---|---|---|---|---|---|
| G6PD | 5% BSA | 1:2000 in 5% BSA, cat#8866, lot#3, Cell Signaling Technology, Danvers, MA, USA | AB_10827744 | 1:2000 Rabbit-HRP | Western Lightning™ |
| GFPT1 | 5% milk | 1:1000 in 5% BSA, cat#14132-1-AP, Proteintech, Rosemont, IL, USA | AB_2110155 | 1:2000 Rabbit-HRP | Super Signal™ |
| GLUT1 | 5% milk | 1:4000 in 5% BSA, cat#07-1401, lot#2882724, Millipore, St. Louis, MO, USA | AB_1587074 | 1:2000 Rabbit-HRP | Super Signal™ |
| GLUT3 | 5% milk | 1:5000 in 5% BSA, cat#AB1344, lot#2943583, Millipore, St. Louis, MO, USA | AB_1587078 | 1:2000 Rabbit-HRP | Western Lightning™ |
| GLUT4 | 5% BSA | 1:2000 in 5% BSA, cat#07-1404, lot#2890837, Millipore, St. Louis, MO, USA | AB_1587080 | 1:2000 Rabbit-HRP | Western Lightning™ |
| GLUT6 | 5% BSA | 1:1000 in 5% BSA, cat#TA500639, lot#A001, OriGene Technologies, Rockville, MD, USA | AB_2270444 | 1:5000 Mouse-HRP | Super Signal™ |
| GLUT10 | 5% milk | 1:5000 in 5% BSA, cat#sc-21635, lot#G0214, Santa Cruz Biotechnology, Dallas, TX, USA | AB_10989951 | 1:5000 Goat-HRP | Western Lightning™ |
| HXII | 5% BSA | 1:1000 in 5% BSA, cat#sc-6521, lot#H2611, Santa Cruz Biotechnology, Dallas, TX, USA | AB_648073 | 1:5000 Goat-HRP | Western Lightning™ |
| O-GlcNAc | 5% milk | 1:2000 in 5% BSA, cat#9875, lot#4, Cell Signaling Technology, Danvers, MA, USA | AB_10950973 | 1:5000 Mouse-HRP | Western Lightning™ |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Weyrauch, L.A.; McMillin, S.L.; Witczak, C.A. Insulin Resistance Does Not Impair Mechanical Overload-Stimulated Glucose Uptake, but Does Alter the Metabolic Fate of Glucose in Mouse Muscle. Int. J. Mol. Sci. 2020, 21, 4715. https://doi.org/10.3390/ijms21134715
Weyrauch LA, McMillin SL, Witczak CA. Insulin Resistance Does Not Impair Mechanical Overload-Stimulated Glucose Uptake, but Does Alter the Metabolic Fate of Glucose in Mouse Muscle. International Journal of Molecular Sciences. 2020; 21(13):4715. https://doi.org/10.3390/ijms21134715
Chicago/Turabian StyleWeyrauch, Luke A., Shawna L. McMillin, and Carol A. Witczak. 2020. "Insulin Resistance Does Not Impair Mechanical Overload-Stimulated Glucose Uptake, but Does Alter the Metabolic Fate of Glucose in Mouse Muscle" International Journal of Molecular Sciences 21, no. 13: 4715. https://doi.org/10.3390/ijms21134715
APA StyleWeyrauch, L. A., McMillin, S. L., & Witczak, C. A. (2020). Insulin Resistance Does Not Impair Mechanical Overload-Stimulated Glucose Uptake, but Does Alter the Metabolic Fate of Glucose in Mouse Muscle. International Journal of Molecular Sciences, 21(13), 4715. https://doi.org/10.3390/ijms21134715





