Dietary Fat and Cancer—Which Is Good, Which Is Bad, and the Body of Evidence
Abstract
1. Introduction
Source of Data
2. The Connection Between Fat, Gut Microbiota, and Inflammatory Diseases
3. HFD, Oxidative Stress, and Inflammation
4. Xenobiotics in the Obesity-Cancer Link
5. Mediterranean Diet and Cancer Risk
6. Dietary Fat and Cancer Risk
6.1. Total Fat
6.2. Saturated Fat
6.2.1. The Role of SFAs in Signalling Pathways Involved in Cancer
Palmitic Acid
Stearic Acid
Lauric Acid (LaA)
Myristic Acid (MA)
The Impact of Other SFAs on Carcinogenesis
6.3. Unsaturated Fat
6.3.1. MUFAs
6.3.2. PUFAs
Ω-6 PUFAs
Ω-3 PUFAs
Trans Fatty Acids (TFAs)
6.3.3. iTFAs
6.3.4. rTFAs
| In Vitro | In Vivo | Human Data | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Cell Line, Fat Specification | Outcome | Reference | Model, Fat Specification | Outcome | Reference | Cancer Type, Fat Specification | Outcome | Reference | |
| SATURATED FAT | |||||||||
| All cancers | Positive association between high SFAs intake and cancer risk and mortality, respectively | [123,124] | |||||||
| HER2/neu-positive breast cancer cells, PA | Induction of cell cycle delay and apoptosis | [151] | Spontaneous mammary tumours, C3H mice, diet supplemented with PA, SA, MA, and LaA, respectively | No effect of diet supplemented with PA, MA or LaA, respectively | [304] | Breast cancer, high SFAs intake | Positive association | [103,124] | |
| Breast cancer, PA and SA intake | Positive association | [154] | |||||||
| Hs578T human breast cancer cells, SA | Growth suppression via cell cycle inhibition | [161] | Breast cancer, PA intake | No association | [155] | ||||
| NMU-induced mammary tumours, Sprague-Dawley rats, HFD rich in SA | Decreased tumour incidence and increased latency after SA supplementation | [161] | |||||||
| SkBr3 breast cancer cells, LaA | Inhibition of proliferation, apoptosis stimulation | [167] | |||||||
| MDA-MB-231 breast cancer cells, capric, caprylic and caproic acids | Cell growth inhibition and apoptosis stimulation | [178] | MDA-MB-435 xenografts, athymic mice, HFD rich in SA | Decreased incidence and multiplicity of tumours | [305] | ||||
| Spontaneous mammary tumours, A/ST mice, HFD rich in SA | Growth suppression, increased tumour latency | [306] | |||||||
| HCT-15 colon cancer cells, LaA | Apoptosis induction | [9] | Azoxymethane-induced colorectal cancer, F344 rats, HFD rich in SFAs | Increased incidence and multiplicity of colon tumours, induction of colonic inflammation | [307] | Colon cancer, SFAs intake | No association | [112] | |
| Caco-2 human colon cancer cells, LaA | Suppression of proliferation | [168] | HCT116 colorectal cancer xenografts, nude mice, HFD rich in PA | Tumour growth stimulation | [8] | ||||
| CT26 mouse colon cancer cells, LaA | Suppression of proliferation, increase in oxidative stress | [169] | |||||||
| HCT-116 colorectal cancer cells, capric, caprylic and caproic acids | Cell growth inhibition, apoptosis stimulation | [178] | |||||||
| Hep3B, SW480, SW620, AGS, BGC-823, HGC-27, 97H, and LM3 hepatocarcinoma cells, PA | Reduced cell proliferation, impaired cell invasiveness | [152] | LM3 hepatocarcinoma xenografts, athymic mice, PA (via gavage) | Tumour growth suppression | [152] | ||||
| PNT1A and PC3 prostate cancer cell lines, PA | Increased proliferation and migration | [147] | PC-3 prostate cancer xenografts, SCID mice, HFD rich in PA | Stimulated proliferation | [98] | Prostate cancer, SFAs intake | Positive association | [125] | |
| Prostate cancer, PA intake | Positive association | [154] | |||||||
| Prostate cancer, PA intake | No association | [155] | |||||||
| Prostate cancer, MA intake | Positive association | [176] | |||||||
| AsPC-1 pancreatic cancer cells, PA | Increased invasiveness | [148] | Nude mice, HPAF pancreatic cancer xenografts, HFD rich in SFAs | Increased tumour viability | [308] | Pancreatic cancer, SFAs intake, PA and SA intake | Negative association | [127] | |
| MIA PaCa-2, PANC-1 and CFPAC pancreatic cancer cells, PA, SA, LaA | Growth inhibition | [309] | |||||||
| Gastric cancer cell lines, PA | Promotion of metastasis | [150] | |||||||
| Oral carcinoma cell lines PA | Increased metastasis | [149] | |||||||
| Ovarian cancer, SFAs intake | Positive association | [158] | |||||||
| No association | [126] | ||||||||
| Ischikawa endometrial cancer cells, LaA | Inhibition of proliferation, apoptosis stimulation | [167] | |||||||
| A-431 skin cancer cells, capric, caprylic and caproic acids | Cell growth inhibition, apoptosis stimulation | [178] | |||||||
| UNSATURATED FAT | |||||||||
| MUFAs | |||||||||
| Isocaloric replacement of SFAs with plant MUFAs | Decreased cancer mortality | [123] | |||||||
| Isocaloric replacement of animal MUFAs with plant MUFAs | [192] | ||||||||
| MCF-7 breast cancer cells, OA | Stimulation of proliferation | [180] | Breast cancer, olive oil consumption, highest vs lowest intake | Decreased risk | [191] | ||||
| Suppressed growth and survival | [182] | ||||||||
| Increased invasiveness | [184] | ||||||||
| MDA-MB-231, OA | Stimulation of growth and migration | [182] | |||||||
| Increased invasiveness | [184] | ||||||||
| BT-474 and SK-Br3 breast cancer cells, OA | Inhibition of Her-2/neu expression | [181] | |||||||
| Caco-2 colon cancer cell line, OA | Growth promotion | [186] | Colon cancer, MUFAs intake | No association | [112] | ||||
| SGC 7901gastric carcinoma cells, OA | Suppressed growth and survival | [182] | GIT cancer, MUFAs intake | Decreased risk | [124] | ||||
| HGC-27 gastric carcinoma line, OA | Stimulation of growth and migration | [182] | GIT cancer, olive oil consumption, highest vs lowest intake | [191] | |||||
| MKN-45 and AGS gastric cancer cell lines, OA | Increased invasiveness | [185] | |||||||
| Prostate cancer, MUFAs intake | Positive association | [125] | |||||||
| Ovarian cancer, MUFAs intake | No association | [126] | |||||||
| HeLa cervical cancer xenografts, BALB/c mice, diet high in OA | Increased growth and metastasis | [188] | |||||||
| Basal cell carcinoma, MUFAs intake | Inverse association between intake and risk | [113] | |||||||
| 786-O renal cancer cells, OA | Increased invasiveness | [187] | |||||||
| CAL27 and UM1 tongue squamous cell carcinomas, OA | Induction of apoptosis and autophagy | [189] | |||||||
| PUFAs | |||||||||
| ω-6 PUFAs | |||||||||
| Isocaloric replacement of SFAs with LA | Decrease in cancer mortality | [123] | |||||||
| Colon cancer, PUFAs intake | No association | [112] | |||||||
| MDA-MB-231 breast cancer cells, LA | Promotion of migration and invasion | [212] | DMBA-induced mammary tumours, Sprague-Dawley rats, diet high in LA | Stimulation of DMBA-DNA adducts formation in mammary gland | [221] | Breast cancer, ω-6 PUFAs intake | No association | [110] | |
| Breast cancer, higher dietary ω-3 PUFAs / ω-6 PUFAs ratio | Lower risk in Asian countries | [222] | |||||||
| RKO and LOVO colon cancer cell lines, LA | Growth stimulation by low concentrations, grow inhibition by high concentrations | [209] | C57BL/6J mice, diet high in LA | Epigenetic alterations associated with colonic inflammation and cancer | [220] | ||||
| SW480 and SW620 colon cancer cells, LA | Decreased cell proliferation and viability | [210] | |||||||
| AGS gastric adenocarcinoma cells, LA | Growth inhibition | [211] | CUM-2MD3 gastric carcinoma transplants, NCr-nu/nu mice, HFD rich in LA | Stimulation of invasion and metastasis | [218] | ||||
| OCUM-2MD3 gastric carcinoma transplants, athymic nude mice, HFD rich in LA | Enhanced tumour growth and angiogenesis | [219] | |||||||
| Oral carcinomas induced by DMBA and betel quid extract, hamsters, high dietary ω-6 PUFAs / ω-3 PUFAs ratio | Tumour growth promotion | [216] | |||||||
| MIA PaCa-2, PANC-1 and CFPAC pancreatic cancer cells, LA | Growth inhibition | [309] | HPAF pancreatic cancer xenografts, nude mice, HFD rich in ω-6 PUFAs | Increased tumour viability, stimulation of liver metastasis | [308] | ||||
| Pancreatic neoplasia, KRAS transgenic mice, diet high in ω-6 PUFAs | Shortened tumour latency | [217] | |||||||
| PC-3 and C4-2 prostatic cancer cells, AA and LA | Reduced cell proliferation and viability | [207] | |||||||
| T98G glioblastoma cells, AA | Growth inhibition | [180] | |||||||
| ω-3 PUFAs | |||||||||
| MCF-7 mammary cancer cells, ALA or ALA combined with EPA and DHA | Decreased viability | [223] | 4T1 mammary tumour transplants, BALB/c mice, ω-3 PUFAs enriched diet | Decrease in proliferation and angiogenesis, stimulation of apoptosis | [243] | Breast cancer, highest ω-3 PUFAs intake vs lowest ω-3 PUFAs intake / high ω-6 PUFAs intake | Decreased risk | [254] | |
| MCF-7 cells, DHA | Reduced proliferation | [226] | |||||||
| LM3 mammary transplants, BALB/c mice, ALA enriched diet | Inhibition of tumour growth and metastasis | [244] | Breast cancer, fish ω-3 PUFAs intake | Decreased risk in Asian patients | [255] | ||||
| MDA-MB-231 cells DHA | Pyroptosis induction | [225] | |||||||
| DMBA-induced mammary tumours in offspring of rats fed with diet enriched with ALA or DHA and EPA, respectively, C57BL/6J mice | Tumour growth inhibition, reduced proliferation and stimulation of apoptosis | [245] | |||||||
| HT-29 and CaCo-2 colorectal cancer cells, DHA | Decreased viability | [227] | Azoxymethane-induced colorectal cancer, F344 rats, HFD rich in ω-3 PUFAs | Decreased incidence and multiplicity of colon tumours in comparison with HFD rich in SFAs | [307] | Colorectal cancer, long-chained ω-3 PUFAs | Inverse association between intake and risk | [256] | |
| HCT-116 and Caco-2 cells, DHA | Anti-angiogenic activity | [228] | |||||||
| HCT-116, HT-29, SW620, DLD-1 colorectal cancer cells, DHA | Decreased proliferation, enhancement of autophagy induced by oxaliplatin | [231] | HCT116 xenografts, BALB/c mice, DHA (i.p.) | Enhancement of autophagy induced by oxaliplatin | [231] | ||||
| N-methyl phosphite nitrourea-induced colorectal cancer, rats, ω-3 PUFAs enriched diet | Tumour growth inhibition | [246] | |||||||
| Colorectal neoplasia, transgenic Apc Min/+mice, dietary fish-oil ω-3 PUFAs | Decreased colorectal carcinoma growth | [247] | |||||||
| MC38 colorectal carcinoma, C57BL/6 mice, ω-3 PUFAs enriched diet | Tumour growth suppression | [248] | |||||||
| MIA PaCa-2, PANC-1 and CFPAC pancreatic cancer cells, ALA, DHA, EPA | Growth inhibition | [308] | HPAF pancreatic cancer xenografts, nude mice, HFD rich in ω-3 PUFAs | Decreased tumour viability | [309] | ||||
| Pancreatic carcinoma, KRAS mice, fish oil ω-3 PUFAs enriched diet | Tumour growth inhibition, reduced proliferation | [249] | |||||||
| PANC-1 pancreatic cancer cells, DHA | Apoptosis induction | [232] | |||||||
| SW1990, PANC-1 pancreatic cancer cells, EPA, DHA | Growth inhibition | [235] | PANC02 transplants, fat-1 transgenic mice | Tumour growth inhibition, apoptosis induction | [235] | ||||
| MHCC 97-L metastatic hepatocarcinoma line | Decreased proliferation, DHA | [236] | |||||||
| Prostate carcinoma, Pten-knockout mice, diet enriched with ALA | Tumour growth inhibition | [250] | Prostate cancer risk, ω-3 PUFAs intake | No effect | [258] | ||||
| Endometrial cancer xenografts, BALB/c mice, dietary ω-3 PUFAs | Tumour growth inhibition | [251,252] | Breast cancer, long-chain ω-3 PUFAs intake | Decreased risk in women with normal BMI | [257] | ||||
| SKOV-3 ovarian cancer line, EPA | Apoptosis induction | [237] | Ovarian cancer, PUFAs intake | No association | [126] | ||||
| SKOV3, A2780, HO8910 ovarian cancer cells, ALA, DHA | Decreased viability by ALA and DHA, inhibition of invasion and metastasis by DHA | [238] | |||||||
| A549 non-small lung cancer cells, DHA | Inhibition of proliferation | [233,234] | |||||||
| LLC murine lung cancer cells, DHA | [234] | ||||||||
| LA-N-1 neuroblastoma cells, DHA, EPA | Cell cycle arrest and induction of apoptosis | [239] | GL261 glioma transplants, fat-1 transgenic mice | Induction of apoptosis and autophagy | [240] | ||||
| D54MG, U87MG and U251MG glioblastoma cells, DHA | Induction of apoptosis and autophagy | [240] | |||||||
| G1a, ML-2, HL-60, THP-1, U937 and MOLM-13 acute myeloid leukaemia cell lines, DHA and EPA | Decrease in cell viability | [241] | |||||||
| Molt-4 acute lymphoblastic leukaemia cells, DHA | Apoptosis induction | [242] | |||||||
| TFAs | |||||||||
| iTFAs | |||||||||
| Ehrlich tumour, CBA mice, dietary EA | Tumour growth promotion, decreased survival | [273] | Oestrogen-receptor negative breast cancer risk, serum level of iTFAs | Positive association | [302] | ||||
| CT-26 and HT-29 colorectal cancer cells, EA | Enhanced growth and metastasis | [270,271] | Colon cancer risk, TFAs intake | Positive association | [298,299] | ||||
| Attenuation of 5-fluorouracil cytotoxicity | [271] | CT26 and HT29 transplants, BALB/c mice, dietary EA | Increased tumour growth and metastasis | [270,272] | Rectal cancer risk, fish TFAs intake | Positive association | [269] | ||
| Caco-2 colorectal cancer cells, EA | No effect on growth | [186] | |||||||
| CMT93 murine rectal carcinoma cell line, EA | Increased stemness, attenuation of 5-fluorouracil cytotoxicity | [271] | |||||||
| Stomach cancer risk, fish TFAs intake | Positive association | [269] | |||||||
| Prostate cancer risk, total TFAs intake | Positive association | [125] | |||||||
| Prostate cancer risk, fish TFAs intake | Negative association | [269] | |||||||
| Pancreatic cancer risk, vegetable TFAs intake | Negative association in men | [269] | |||||||
| Pancreatic risk, serum level of iTFAs | Positive association in men | [303] | |||||||
| Ovarian cancer risk, TFAs intake | Positive association | [301] | |||||||
| SH-SY5Y neuroblastoma cells, EA | Growth inhibition, apoptosis induction | [274] | CNS cancer risk | Negative association in women | [269] | ||||
| LL2 murine lung cancer cell line, EA | Increased stemness, attenuation of 5-fluorouracil cytotoxicity | [271] | Lung cancer risk | Negative association in women | [269] | ||||
| Non-Hodgkin lymphoma risk, vegetable TFAs intake | Negative association | [269] | |||||||
| Multiple myeloma, fish TFAs intake | Positive association | ||||||||
| Bladder cancer risk, fish TFAs intake | Negative association | [269] | |||||||
| rTFAs | |||||||||
| MCF-7 mammary carcinoma, VA | Inhibition of proliferation | [275] | Mammary tumour growth | Growth inhibition | Reviewed in [287] | Breast cancer risk, CLA intake | No association | [297] | |
| MCF-10A mammary cancer cells, VA | No effect | [276] | DMBA-induced mammary tumours in Sprague-Dawley rat offspring, maternal diet enriched with CLA | Decreased susceptibility to tumour induction | [291] | Post-menopausal breast cancer, rTFAs intake | Positive association | [269] | |
| MCF-7 and MDA-MB-231 cells, CLA | Growth inhibition | [278,281] | |||||||
| Potentiation of docetaxel effect | [279] | ||||||||
| MCF-7 cells, CLA-gemcitabine conjugate | Growth inhibition | [292] | MCF-7 xenografts, BALB/c mice, CLA-gemcitabine conjugate | Suppression of tumour growth | [292] | ||||
| SW480 colon carcinoma, VA | Inhibition of proliferation | [275] | CT29 xenografts, BALB/c mice, dietary CLA | Metastasis inhibition | [283] | ||||
| HCT-116 and HT-29 colorectal carcinoma, CLA | Isomer-dependent inhibition of proliferation, induction of apoptosis, | [282] | |||||||
| 1,2-dimethylhydrazine-induced colon cancer, Sprague-Dawley rats, dietary CLA | Apoptosis induction | [310] | |||||||
| SW480 colon cancer cells, CLA | Isomer-dependent effect on cell invasiveness | [283] | |||||||
| Azoxymethane-induced colon cancer, Sprague-Dawley rats, dietary CLA | Decrease in aberrant crypt foci formation, apoptosis induction | [311] | |||||||
| Azoxymethane and dextransodium sulfate-induced colorectal cancer, 57BL/6 mice, dietary CLA | Tumour growth promotion | [312] | |||||||
| Mouth/pharynx cancer risk, rTFAs | Positive association | [269] | |||||||
| DU145 prostate carcinoma cells, CLA | Cell cycle inhibition | [285] | DU-145 transplants, SCID mice, dietary CLA | Inhibition of tumour growth and metastasis | [313] | ||||
| R-3327-AT-1 transplants, Copenhagen rats, dietary CLA | No effect on tumour growth | [314] | |||||||
| SKOV-3 and A2780 ovarian cancer cells, CLA | Isomer-dependent suppression of proliferation and migration | [284] | |||||||
| RL 95-2 endometrial cancer cells, CLA | Apoptosis induction | [286] | |||||||
| 5-8F and CNE-2 human nasopharyngeal carcinoma | Inhibition of proliferation, induction of apoptosis | [277] | |||||||
| B16-F10 melanoma, liposomes containing CLA and paclitaxel | Growth inhibition | [293] | B16-F10 melanoma transplants, C57BL6/N mice, liposomes containing CLA and paclitaxel (i.v.) | Tumour growth inhibition | [293] | Malignant melanoma risk, rTFAs intake | Negative association in women | [269] | |
| Non-melanoma cancer risk, rTFAs intake | Positive association | ||||||||
| Multiple myeloma risk, rTFAs intake | Negative association | [269] | |||||||
| Non-Hodgkin’s lymphoma risk, rTFAs intake | Positive association | ||||||||
7. Targeting Lipid Metabolism in Cancer Treatment
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AA | Arachidonic acid |
| ALA | Alpha-linolenic acid |
| AMPK | Adenosine monophosphate-activated protein kinase |
| AR | Adrenergic receptor |
| ATF | Activating transcription factor |
| aP2 | Adipocyte fatty acid-binding protein |
| CD36 | Cluster of differentiation 36 |
| CDK2 | Cyclin-dependent kinase 2 |
| CHOP | C/EBP homologous protein, DNA damage-inducible transcript 3 |
| CI | Confidence interval |
| CLA | Conjugated linoleic acid |
| DHA | Docosahexaenoic acid |
| DMBA | 9,10-dimethyl-1,2-benz[a]-anthracene |
| EA | Elaidic acid |
| EGFR | Epidermal growth factor receptor |
| EMT | Epithelial-mesenchymal transition |
| EPA | Eicosapentaenoic acid |
| EPIC | European Prospective Investigation into Cancer and Nutrition |
| ER | Endoplasmic reticulum |
| ERK1/2 | Extracellular signal-regulated kinase 1/2 |
| FAK | Focal adhesion kinase |
| FAs | Fatty acids |
| GIT | Gastrointestinal tract |
| GPR | G-protein-coupled receptor |
| GSK-3β | Glycogen synthase kinase-3 beta |
| HDAC | Histone deacetylase |
| HDACi | Histone deacetylase inhibitor |
| HFD | High-fat diet |
| HMGB1 | High mobility group box 1 protein |
| HR | Hazard ratio |
| IARC | International Agency for Research on Cancer |
| IL | Interleukin |
| iTFAs | Industrially produced trans fatty acids |
| LaA | Lauric acid |
| LA | Linoleic acid |
| LPS | Lipopolysaccharides |
| MA | Myristic acid |
| MAPKs | Mitogen-activated protein kinases |
| MCP-1 | Monocyte chemoattractant protein-1 |
| MMP | Matrix metalloproteinase |
| mTOR | Mammalian target of rapamycin |
| MUFAs | Monounsaturated fatty acids |
| NF-κB | Nuclear factor kappa B |
| NMU | N-methyl-N-nitrosourea |
| NOX | Nicotinamide adenine dinucleotide phosphate oxidase |
| Nrf2 | Nuclear factor erythroid 2-related factor 2 |
| OA | Oleic acid |
| OR | Odds ratio |
| p21(CIP1/WAF1) | Cyclin-dependent kinase inhibitor 1 |
| p27 (KIP) | Cyclin-dependent kinase inhibitor 1B |
| PA | Palmitic acid |
| PCNA | Proliferating cell nuclear antigen |
| PI3K | Phosphatidylinositol 3-kinase |
| PPAR | Peroxisome proliferator-activated receptor |
| PUFAs | Polyunsaturated fatty acids |
| Q | Quintile |
| RCT | Randomised controlled trial |
| RNS | Reactive nitrogen species |
| ROS | Reactive oxygen species |
| RVLM | Rostral ventral lateral medulla |
| RR | Relative risk |
| rTFAs | Ruminant trans fatty acids |
| SA | Stearic acid |
| SREBP | Sterol regulatory element-binding protein |
| SCD | Stearoyl-CoA desaturase |
| SFAs | Saturated fatty acids |
| STAT | Signal transducer and activator of transcription |
| T | Tertile |
| TFAs | Trans fatty acids |
| TGF-β | Transforming growth factor beta |
| TLR-4 | Toll-like receptor 4 |
| TMA | Trimethylamine |
| TMAO | Trimethylamine-N-oxide |
| TNFα | Tumour necrosis factor alpha |
| UPR | Unfolded protein response |
| VA | Vaccenic acid |
| XBP1 | X-box binding protein 1 |
References
- Eyre, H.; Kahn, R.; Robertson, R.M.; Clark, N.G.; Doyle, C.; Gansler, T.; Glynn, T.; Hong, Y.; Smith, R.A.; Taubert, K.; et al. Preventing Cancer, Cardiovascular Disease, and Diabetes: A Common Agenda for the American Cancer Society, the American Diabetes Association, and the American Heart Association. CA Cancer J. Clin. 2004, 54, 190–207. [Google Scholar] [CrossRef] [PubMed]
- Duan, Y.; Zeng, L.; Zheng, C.; Song, B.; Li, F.; Kong, X.; Xu, K. Inflammatory Links Between High Fat Diets and Diseases. Front. Immunol. 2018, 9, 2649. [Google Scholar] [CrossRef] [PubMed]
- Font-Burgada, J.; Sun, B.; Karin, M. Obesity and Cancer: The Oil that Feeds the Flame. Cell Metab. 2016, 23, 48–62. [Google Scholar] [CrossRef] [PubMed]
- Murphy, E.A.; Velazquez, K.T.; Herbert, K.M. Influence of high-fat diet on gut microbiota: A driving force for chronic disease risk. Curr. Opin. Clin. Nutr. Metab. Care 2015, 18, 515–520. [Google Scholar] [CrossRef]
- Le Lay, S.; Simard, G.; Martinez, M.C.; Andriantsitohaina, R. Oxidative Stress and Metabolic Pathologies: From an Adipocentric Point of View. Oxid. Med. Cell. Longev. 2014, 2014, 1–18. [Google Scholar] [CrossRef]
- Bai, Y.; Sun, Q. Macrophage recruitment in obese adipose tissue. Obes. Rev. 2015, 16, 127–136. [Google Scholar] [CrossRef]
- Maslov, L.N.; Naryzhnaya, N.V.; Boshchenko, A.A.; Popov, S.V.; Ivanov, V.V.; Oeltgen, P.R. Is oxidative stress of adipocytes a cause or a consequence of the metabolic syndrome? J. Clin. Transl. Endocrinol. 2019, 15, 1–5. [Google Scholar] [CrossRef]
- Fatima, S.; Hu, X.; Huang, C.; Zhang, W.; Cai, J.; Huang, M.; Gong, R.-H.; Chen, M.; Ho, A.H.M.; Su, T.; et al. High-fat diet feeding and palmitic acid increase CRC growth in β2AR-dependent manner. Cell Death Dis. 2019, 10, 711. [Google Scholar] [CrossRef]
- Sheela, D.; Narayanankutty, A.; Nazeem, P.; Raghavamenon, A.; Muthangaparambil, S. Lauric acid induce cell death in colon cancer cells mediated by the epidermal growth factor receptor downregulation: An in silico and in vitro study. Hum. Exp. Toxicol. 2019, 38, 753–761. [Google Scholar] [CrossRef]
- Zitvogel, L.; Galluzzi, L.; Viaud, S.; Vétizou, M.; Daillère, R.; Merad, M.; Kroemer, G. Cancer and the gut microbiota: An unexpected link. Sci. Transl. Med. 2015, 7, 271ps1. [Google Scholar] [CrossRef]
- Tang, W.H.W.; Kitai, T.; Hazen, S.L. Gut microbiota in cardiovascular health and disease. Circ. Res. 2017, 120, 1183–1196. [Google Scholar] [CrossRef] [PubMed]
- Caesar, R.; Tremaroli, V.; Kovatcheva-Datchary, P.; Cani, P.D.; Bäckhed, F. Crosstalk between gut microbiota and dietary lipids aggravates WAT inflammation through TLR signaling. Cell Metab. 2015, 22, 658–668. [Google Scholar] [CrossRef] [PubMed]
- Patterson, E.; O’Doherty, R.M.; Murphy, E.F.; Wall, R.; O’Sullivan, O.; Nilaweera, K.; Fitzgerald, G.F.; Cotter, P.D.; Ross, R.P.; Stanton, C. Impact of dietary fatty acids on metabolic activity and host intestinal microbiota composition in C57BL/6J mice. Br. J. Nutr. 2014, 111, 1905–1917. [Google Scholar] [CrossRef] [PubMed]
- Lam, Y.Y.; Ha, C.W.Y.; Hoffmann, J.M.A.; Oscarsson, J.; Dinudom, A.; Mather, T.J.; Cook, D.I.; Hunt, N.H.; Caterson, I.D.; Holmes, A.J.; et al. Effects of dietary fat profile on gut permeability and microbiota and their relationships with metabolic changes in mice. Obesity 2015, 23, 1429–1439. [Google Scholar] [CrossRef]
- Qiao, Y.; Sun, J.; Ding, Y.; Le, G.; Shi, Y. Alterations of the gut microbiota in high-fat diet mice is strongly linked to oxidative stress. Appl. Microbiol. Biotechnol. 2013, 97, 1689–1697. [Google Scholar] [CrossRef]
- Reuter, S.; Gupta, S.C.; Chaturvedi, M.M.; Aggarwal, B.B. Oxidative stress, inflammation, and cancer: How are they linked? Free Radic. Biol. Med. 2010, 49, 1603–1616. [Google Scholar] [CrossRef]
- Carmody, R.N.; Gerber, G.K.; Luevano, J.M.; Gatti, D.M.; Somes, L.; Svenson, K.L.; Turnbaugh, P.J. Diet dominates host genotype in shaping the murine gut microbiota. Cell Host Microbe 2015, 17, 72–84. [Google Scholar] [CrossRef]
- Cândido, F.G.; Valente, F.X.; Grześkowiak, Ł.M.; Moreira, A.P.B.; Rocha, D.M.U.P.; Alfenas, R.D.C.G. Impact of dietary fat on gut microbiota and low-grade systemic inflammation: Mechanisms and clinical implications on obesity. Int. J. Food Sci. Nutr. 2018, 69, 125–143. [Google Scholar] [CrossRef]
- Coelho, O.G.L.; Cândido, F.G.; Alfenas, R.D.C.G. Dietary fat and gut microbiota: Mechanisms involved in obesity control. Crit. Rev. Food Sci. Nutr. 2019, 59, 3045–3053. [Google Scholar] [CrossRef]
- Schoeler, M.; Caesar, R. Dietary lipids, gut microbiota and lipid metabolism. Rev. Endocr. Metab. Disord. 2019, 20, 461–472. [Google Scholar] [CrossRef]
- Cani, P.D.; Amar, J.; Iglesias, M.A.; Poggi, M.; Knauf, C.; Bastelica, D.; Neyrinck, A.M.; Fava, F.; Tuohy, K.M.; Chabo, C.; et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes 2007, 56, 1761–1772. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, R.; Andersen, A.D.; Hermann-Bank, M.L.; Stagsted, J.; Boye, M. The effect of high-fat diet on the composition of the gut microbiota in cloned and non-cloned pigs of lean and obese phenotype. Gut Microbes 2013, 4, 371–381. [Google Scholar] [CrossRef] [PubMed]
- Raetz, C.R.H.; Whitfield, C. Lipopolysaccharide Endotoxins. Annu. Rev. Biochem. 2002, 71, 635–700. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.C. NF-κB signaling in inflammation. Signal Transduct. Target. 2017, 2, 17023. [Google Scholar] [CrossRef]
- Shi, H.; Kokoeva, M.V.; Inouye, K.; Tzameli, I.; Yin, H.; Flier, J.S. TLR4 links innate immunity and fatty acid-induced insulin resistance. J. Clin. Invest. 2006, 116, 3015–3025. [Google Scholar] [CrossRef] [PubMed]
- Rohr, M.W.; Narasimhulu, C.A.; Rudeski-Rohr, T.A.; Parthasarathy, S. Negative Effects of a High-Fat Diet on Intestinal Permeability: A Review. Adv. Nutr. 2019, 11, 77–91. [Google Scholar] [CrossRef]
- Do, M.H.; Lee, E.; Oh, M.J.; Kim, Y.; Park, H.Y. High-glucose or-fructose diet cause changes of the gut microbiota and metabolic disorders in mice without body weight change. Nutrients 2018, 10, 761. [Google Scholar] [CrossRef]
- Chan, C.W.H.; Law, B.M.H.; Waye, M.M.Y.; Chan, J.Y.W.; Wei So, W.K.; Chow, K.M. Trimethylamine-N-oxide as one hypothetical link for the relationship between intestinal microbiota and cancer—Where we are and where shall we go? J. Cancer 2019, 10, 5874–5882. [Google Scholar] [CrossRef]
- D’Hennezel, E.; Abubucker, S.; Murphy, L.O.; Cullen, T.W. Total Lipopolysaccharide from the Human Gut Microbiome Silences Toll-Like Receptor Signaling. mSystems 2017, 2, e00046-17. [Google Scholar] [CrossRef]
- Wisniewski, P.J.; Dowden, R.A.; Campbell, S.C. Role of dietary lipids in modulating inflammation through the gut microbiota. Nutrients 2019, 11, E117. [Google Scholar] [CrossRef]
- Furukawa, S.; Fujita, T.; Shimabukuro, M.; Iwaki, M.; Yamada, Y.; Nakajima, Y.; Nakayama, O.; Makishima, M.; Matsuda, M.; Shimomura, I. Increased oxidative stress in obesity and its impact on metabolic syndrome. J. Clin. Invest. 2004, 114, 1752–1761. [Google Scholar] [CrossRef] [PubMed]
- Nagpal, R.; Newman, T.; Wang, S.; Jain, S.; Lovato, J.; Yadav, H. Obesity-Linked Gut Microbiome Dysbiosis Associated With Derangements in Gut Permeability and Intestinal Cellular Homeostasis Independent of Diet. J. Diabetes Res. 2018, 2018, 3462092. [Google Scholar] [CrossRef] [PubMed]
- Zak-Gołab, A.; Kocełak, P.; Aptekorz, M.; Zientara, M.; Juszczyk, Ł.; Martirosian, G.; Chudek, J.; Olszanecka-Glinianowicz, M. Gut microbiota, microinflammation, metabolic profile, and zonulin concentration in obese and normal weight subjects. Int. J. Endocrinol. 2013, 2013, 674106. [Google Scholar] [CrossRef] [PubMed]
- Gummesson, A.; Carlsson, L.M.S.; Storlien, L.H.; Bäckhed, F.; Lundin, P.; Löfgren, L.; Stenlöf, K.; Lam, Y.Y.; Fagerberg, B.; Carlsson, B. Intestinal permeability is associated with visceral adiposity in healthy women. Obesity 2011, 19, 2280–2282. [Google Scholar] [CrossRef]
- Seki, E.; Brenner, D.A. Toll-like receptors and adaptor molecules in liver disease: Update. Hepatology 2008, 48, 322–335. [Google Scholar] [CrossRef]
- Crispe, I.N. The Liver as a Lymphoid Organ. Annu. Rev. Immunol. 2009, 27, 147–163. [Google Scholar] [CrossRef]
- Chen, G.Y.; Nuñez, G. Sterile inflammation: Sensing and reacting to damage. Nat. Rev. Immunol. 2010, 10, 826–837. [Google Scholar] [CrossRef]
- Guo, J.; Friedman, S.L. Toll-like receptor 4 signaling in liver injury and hepatic fibrogenesis. Fibrogenes. Tissue Repair 2010, 3, 21. [Google Scholar] [CrossRef]
- Arab, J.P.; Martin-Mateos, R.M.; Shah, V.H. Gut–liver axis, cirrhosis and portal hypertension: The chicken and the egg. Hepatol. Int. 2018, 12, 24–33. [Google Scholar] [CrossRef]
- Pradere, J.P.; Troeger, J.S.; Dapito, D.H.; Mencin, A.A.; Schwabe, R.F. Toll-like receptor 4 and hepatic fibrogenesis. Semin. Liver Dis. 2010, 30, 232–244. [Google Scholar] [CrossRef]
- Zangara, M.T.; McDonald, C. How diet and the microbiome shape health or contribute to disease: A mini-review of current models and clinical studies. Exp. Biol. Med. 2019, 244, 484–493. [Google Scholar] [CrossRef] [PubMed]
- Heisel, T.; Montassier, E.; Johnson, A.; Al-Ghalith, G.; Lin, Y.-W.; Wei, L.-N.; Knights, D.; Gale, C.A. High-Fat Diet Changes Fungal Microbiomes and Interkingdom Relationships in the Murine Gut. mSphere 2017, 2, e00351-17. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.K.; Brzezinska-Slebodzinska, E.; Madsen, F.C. Oxidative Stress, Antioxidants, and Animal Function. J. Dairy Sci. 1993, 76, 2812–2823. [Google Scholar] [CrossRef]
- Weidinger, A.; Kozlov, A.V. Biological activities of reactive oxygen and nitrogen species: Oxidative stress versus signal transduction. Biomolecules 2015, 5, 472–484. [Google Scholar] [CrossRef] [PubMed]
- Di Meo, S.; Reed, T.T.; Venditti, P.; Victor, V.M. Role of ROS and RNS Sources in Physiological and Pathological Conditions. Oxid. Med. Cell. Longev. 2016, 2016, 1245049. [Google Scholar] [CrossRef]
- Birben, E.; Sahiner, U.M.; Sackesen, C.; Erzurum, S.; Kalayci, O. Oxidative stress and antioxidant defense. World Allergy Organ. J. 2012, 5, 9–19. [Google Scholar] [CrossRef]
- Prasad, S.; Gupta, S.C.; Tyagi, A.K. Reactive oxygen species (ROS) and cancer: Role of antioxidative nutraceuticals. Cancer Lett. 2017, 387, 95–105. [Google Scholar] [CrossRef]
- Houstis, N.; Rosen, E.D.; Lander, E.S. Reactive oxygen species have a causal role in multiple forms of insulin resistance. Nature 2006, 440, 944–948. [Google Scholar] [CrossRef]
- Fonseca-Alaniz, M.H.; Takada, J.; Alonso-Vale, M.I.C.; Lima, F.B. Adipose tissue as an endocrine organ: From theory to practice. J. Pediatr. 2007, 83, S192–S203. [Google Scholar] [CrossRef]
- Han, C.Y. Roles of reactive oxygen species on insulin resistance in adipose tissue. Diabetes Metab. J. 2016, 40, 272–279. [Google Scholar] [CrossRef]
- Curtis, J.M.; Grimsrud, P.A.; Wright, W.S.; Xu, X.; Foncea, R.E.; Graham, D.W.; Brestoff, J.R.; Wiczer, B.M.; Ilkayeva, O.; Cianflone, K.; et al. Downregulation of adipose glutathione S-tansferase A4 leads to increased protein carbonylation, oxidative stress, and mitochondrial dysfunction. Diabetes 2010, 59, 1132–1142. [Google Scholar] [CrossRef] [PubMed]
- Schoettl, T.; Fischer, I.P.; Ussar, S. Heterogeneity of adipose tissue in development and metabolic function. J. Exp. Biol. 2018, 121 (Suppl. 1), jeb162958. [Google Scholar] [CrossRef]
- Sun, K.; Kusminski, C.M.; Scherer, P.E. Adipose tissue remodeling and obesity. J. Clin. Invest. 2011, 121, 2094–2101. [Google Scholar] [CrossRef] [PubMed]
- Harman-Boehm, I.; Blüher, M.; Redel, H.; Sion-Vardy, N.; Ovadia, S.; Avinoach, E.; Shai, I.; Klöting, N.; Stumvoll, M.; Bashan, N.; et al. Macrophage infiltration into omental versus subcutaneous fat across different populations: Effect of regional adiposity and the comorbidities of obesity. J. Clin. Endocrinol. Metab. 2007, 92, 2240–2247. [Google Scholar] [CrossRef] [PubMed]
- Lumeng, C.N.; Bodzin, J.L.; Saltiel, A.R. Obesity induces a phenotypic switch in adipose tissue macrophage polarization. J. Clin. Invest. 2007, 117, 175–184. [Google Scholar] [CrossRef]
- Tan, H.Y.; Wang, N.; Li, S.; Hong, M.; Wang, X.; Feng, Y. The Reactive Oxygen Species in Macrophage Polarization: Reflecting Its Dual Role in Progression and Treatment of Human Diseases. Oxid. Med. Cell. Longev. 2016, 2016, 2795090. [Google Scholar] [CrossRef]
- Jung, U.J.; Choi, M.S. Obesity and its metabolic complications: The role of adipokines and the relationship between obesity, inflammation, insulin resistance, dyslipidemia and nonalcoholic fatty liver disease. Int. J. Mol. Sci. 2014, 15, 6184–6223. [Google Scholar] [CrossRef]
- Schapira, D.V.; Clark, R.A.; Wolff, P.A.; Jarrett, A.R.; Kumar, N.B.; Aziz, N.M. Visceral obesity and breast cancer risk. Cancer 1994, 74, 632–639. [Google Scholar] [CrossRef]
- Beddy, P.; Howard, J.; McMahon, C.; Knox, M.; De Blacam, C.; Ravi, N.; Reynolds, J.V.; Keogan, M.T. Association of visceral adiposity with oesophageal and junctional adenocarcinomas. Br. J. Surg. 2010, 97, 1028–1034. [Google Scholar] [CrossRef]
- Schoen, R.E.; Tangen, C.M.; Kuller, L.H.; Burke, G.L.; Cushman, M.; Tracy, R.P.; Dobs, A.; Savage, P.J. Increased blood glucose and insulin, body size, and incident colorectal cancer. J. Natl. Cancer Inst. 1999, 91, 1147–1154. [Google Scholar] [CrossRef]
- Marseglia, L.; Manti, S.; D’Angelo, G.; Nicotera, A.; Parisi, E.; Di Rosa, G.; Gitto, E.; Arrigo, T. Oxidative stress in obesity: A critical component in human diseases. Int. J. Mol. Sci. 2015, 16, 378–400. [Google Scholar] [CrossRef] [PubMed]
- Moon, H.G.; Ju, Y.T.; Jeong, C.Y.; Jung, E.J.; Lee, Y.J.; Hong, S.C.; Ha, W.S.; Park, S.T.; Choi, S.K. Visceral obesity may affect oncologic outcome in patients with colorectal cancer. Ann. Surg. Oncol. 2008, 15, 1918–1922. [Google Scholar] [CrossRef] [PubMed]
- Clark, W.; Siegel, E.M.; Chen, Y.A.; Zhao, X.; Parsons, C.M.; Hernandez, J.M.; Weber, J.; Thareja, S.; Choi, J.; Shibata, D. Quantitative measures of visceral adiposity and body mass index in predicting rectal cancer outcomes after neoadjuvant chemoradiation. J. Am. Coll. Surg. 2013, 216, 1070–1081. [Google Scholar] [CrossRef] [PubMed]
- Grignol, V.P.; Smith, A.D.; Shlapak, D.; Zhang, X.; Del Campo, S.M.; Carson, W.E. Increased visceral to subcutaneous fat ratio is associated with decreased overall survival in patients with metastatic melanoma receiving anti-angiogenic therapy. Surg. Oncol. 2015, 24, 353–358. [Google Scholar] [CrossRef]
- Okamura, A.; Watanabe, M.; Mine, S.; Nishida, K.; Imamura, Y.; Kurogochi, T.; Kitagawa, Y.; Sano, T. Clinical Impact of Abdominal Fat Distribution on Prognosis After Esophagectomy for Esophageal Squamous Cell Carcinoma. Ann. Surg. Oncol. 2016, 23, 1387–1394. [Google Scholar] [CrossRef]
- Mauland, K.K.; Eng, Ø.; Ytre-Hauge, S.; Tangen, I.L.; Berg, A.; Salvesen, H.B.; Salvesen, Ø.O.; Krakstad, C.; Trovik, J.; Hoivik, E.A.; et al. High visceral fat percentage is associated with poor outcome in endometrial cancer. Oncotarget 2017, 8, 105184–105195. [Google Scholar] [CrossRef]
- Thaler, J.P.; Yi, C.X.; Schur, E.A.; Guyenet, S.J.; Hwang, B.H.; Dietrich, M.O.; Zhao, X.; Sarruf, D.A.; Izgur, V.; Maravilla, K.R.; et al. Obesity is associated with hypothalamic injury in rodents and humans. J. Clin. Invest. 2012, 122, 153–162. [Google Scholar] [CrossRef]
- Yi, C.X.; Al-Massadi, O.; Donelan, E.; Lehti, M.; Weber, J.; Ress, C.; Trivedi, C.; Müller, T.D.; Woods, S.C.; Hofmann, S.M. Exercise protects against high-fat diet-induced hypothalamic inflammation. Physiol. Behav. 2012, 106, 485–490. [Google Scholar] [CrossRef]
- Buckman, L.B.; Thompson, M.M.; Moreno, H.N.; Ellacott, K.L.J. Regional astrogliosis in the mouse hypothalamus in response to obesity. J. Comp. Neurol. 2013, 521, 1322–1333. [Google Scholar] [CrossRef]
- Hsuchou, H.; He, Y.; Kastin, A.J.; Tu, H.; Markadakis, E.N.; Rogers, R.C.; Fossier, P.B.; Pan, W. Obesity induces functional astrocytic leptin receptors in hypothalamus. Brain 2009, 132, 889–902. [Google Scholar] [CrossRef]
- Wu, K.L.H.; Chan, S.H.H.; Chan, J.Y.H. Neuroinflammation and oxidative stress in rostral ventrolateral medulla contribute to neurogenic hypertension induced by systemic inflammation. J. Neuroinflammation 2012, 9, 212. [Google Scholar] [CrossRef] [PubMed]
- Winklewski, P.J.; Radkowski, M.; Wszedybyl-Winklewska, M.; Demkow, U. Brain inflammation and hypertension: The chicken or the egg? J. Neuroinflammation 2015, 12, 85. [Google Scholar] [CrossRef] [PubMed]
- Winklewski, P.J.; Radkowski, M.; Demkow, U. Neuroinflammatory mechanisms of hypertension: Potential therapeutic implications. Curr. Opin. Nephrol. Hypertens. 2016, 25, 410–416. [Google Scholar] [CrossRef] [PubMed]
- Cole, S.W.; Sood, A.K. Molecular pathways: Beta-adrenergic signaling in cancer. Clin. Cancer Res. 2012, 18, 1201–1206. [Google Scholar] [CrossRef] [PubMed]
- Cole, S.W.; Nagaraja, A.S.; Lutgendorf, S.K.; Green, P.A.; Sood, A.K. Sympathetic nervous system regulation of the tumour microenvironment. Nat. Rev. Cancer 2015, 15, 563–572. [Google Scholar] [CrossRef]
- Regulska, K.; Regulski, M.; Karolak, B.; Murias, M.; Stanisz, B. Can cardiovascular drugs support cancer treatment? The rationale for drug repurposing. Drug Discov. Today 2019, 24, 1059–1065. [Google Scholar] [CrossRef]
- Diakos, C.I.; Charles, K.A.; McMillan, D.C.; Clarke, S.J. Cancer-related inflammation and treatment effectiveness. Lancet Oncol. 2014, 15, e493–e503. [Google Scholar] [CrossRef]
- Lambert, G.W.; Schlaich, M.P.; Eikelis, N.; Lambert, E.A. Sympathetic activity in obesity: A brief review of methods and supportive data. Ann. N. Y. Acad. Sci. 2019, 1454, 56–67. [Google Scholar] [CrossRef]
- Simó, M.; Navarro, X.; Yuste, V.J.; Bruna, J. Autonomic nervous system and cancer. Clin. Auton. Res. 2018, 28, 301–314. [Google Scholar] [CrossRef]
- Regulska, K.; Regulski, M.; Karolak, B.; Michalak, M.; Murias, M.; Stanisz, B. Beyond the boundaries of cardiology: Still untapped anticancer properties of the cardiovascular system-related drugs. Pharm. Res. 2019, 147, 104326. [Google Scholar] [CrossRef]
- Grün, F.; Blumberg, B. Environmental obesogens: Organotins and endocrine disruption via nuclear receptor signaling. Endocrinology 2006, 147, S50–S55. [Google Scholar] [CrossRef] [PubMed]
- Soto, A.M.; Sonnenschein, C. Environmental causes of cancer: Endocrine disruptors as carcinogens. Nat. Rev. Endocrinol. 2010, 6, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Tareke, E.; Rydberg, P.; Karlsson, P.; Eriksson, S.; Törnqvist, M. Analysis of acrylamide, a carcinogen formed in heated foodstuffs. J. Agric. Food Chem. 2002, 50, 4998–5006. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.W.; Pyo, S. Acrylamide induces adipocyte differentiation and obesity in mice. Chem. Biol. Interact. 2019, 298, 24–34. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.; Zhuang, P.; Jiao, J.; Wang, J.; Zhang, Y. Association of acrylamide hemoglobin biomarkers with obesity, abdominal obesity and overweight in general US population: NHANES 2003–2006. Sci. Total Enviorn. 2018, 31–632, 589–596. [Google Scholar] [CrossRef]
- Newbold, R.R. Impact of environmental endocrine disrupting chemicals on the development of obesity. Hormones 2010, 9, 206–217. [Google Scholar] [CrossRef]
- Heindel, J.J.; Blumberg, B. Environmental Obesogens: Mechanisms and Controversies. Annu. Rev. Pharm. Toxicol. 2019, 59, 89–106. [Google Scholar] [CrossRef]
- Mentella, M.C.; Scaldaferri, F.; Ricci, C.; Gasbarrini, A.; Miggiano, G.A.D. Cancer and Mediterranean Diet: A Review. Nutrients 2019, 11, 2059. [Google Scholar] [CrossRef]
- Davis, C.; Bryan, J.; Hodgson, J.; Murphy, K. Definition of the Mediterranean diet: A Literature Review. Nutrients 2015, 7, 9139–9153. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Schwedhelm, C.; Galbete, C.; Hoffmann, G. Adherence to Mediterranean Diet and Risk of Cancer: An Updated Systematic Review and Meta-Analysis. Nutrients 2017, 9, E1063. [Google Scholar] [CrossRef]
- Witlox, W.J.A.; van Osch, F.H.M.; Brinkman, M.; Jochems, S.; Goossens, M.E.; Weiderpass, E.; White, E.; van den Brandt, P.A.; Giles, G.G.; Milne, R.L.; et al. An inverse association between the Mediterranean diet and bladder cancer risk: A pooled analysis of 13 cohort studies. Eur. J. Nutr. 2019, 59, 287–296. [Google Scholar] [CrossRef] [PubMed]
- Hernáez, Á.; Estruch, R. The Mediterranean Diet and Cancer: What Do Human and Molecular Studies Have to Say About It? Nutrients 2019, 11, E2155. [Google Scholar] [CrossRef]
- IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. In Alcohol Consumption and EthylCarbamate; International Agency for Research on Cancer: Lyon, France, 2010.
- Watson, A.F.; Mellanby, E. Tar Cancer in Mice. II. The Condition of the Skin, when Modified by External Treatment or Diet, as a Factor in Influencing the Cancerous Reaction. Br. J. Exp. Pathol. 1930, 11, 311–322. [Google Scholar]
- Albanes, D. Total Calories, Body Weight, and Tumor Incidence in Mice. Cancer Res. 1987, 47, 1987–1992. [Google Scholar]
- Cohen, L.A.; Thompson, D.O.; Maeura, Y.; Choi, K.; Blank, M.E.; Rose, D.P. Dietary fat and mammary cancer. I. Promoting effects of different dietary fats on N-nitrosomethylurea-induced rat mammary tumorigenesis. J. Natl. Cancer Inst. 1986, 77, 33–42. [Google Scholar]
- Takeshita, M.; Ueda, H.; Shirabe, K.; Higuchi, Y.; Yoshida, S. Lack of promotion of colon carcinogenesis by high-oleic safflower oil. Cancer 1997, 79, 1487–1493. [Google Scholar] [CrossRef]
- Kim, S.; Yang, X.; Yin, A.; Zha, J.; Beharry, Z.; Bai, A.; Bielawska, A.; Bartlett, M.G.; Yin, H.; Cai, H. Dietary palmitate cooperates with Src kinase to promote prostate tumor progression. Prostate 2019, 79, 896–908. [Google Scholar] [CrossRef]
- Hardy, S.; El-Assaad, W.; Przybytkowski, E.; Joly, E.; Prentki, M.; Langelier, Y. Saturated fatty acid-induced apoptosis in MDA-MB-231 breast cancer cells. A role for cardiolipin. J. Biol. Chem. 2003, 278, 31861–31870. [Google Scholar] [CrossRef]
- Chénais, B.; Blanckaert, V. The Janus Face of Lipids in Human Breast Cancer: How Polyunsaturated Fatty Acids Affect Tumor Cell Hallmarks. Int. J. Breast Cancer 2012, 2012, 1–8. [Google Scholar] [CrossRef]
- Arnold, M.; Pandeya, N.; Byrnes, G.; Renehan, A.G.; Stevens, G.A.; Ezzati, M.; Ferlay, J.; Miranda, J.J.; Romieu, I.; Dikshit, R.; et al. Global burden of cancer attributable to high body-mass index in 2012: A population-based study. Lancet Oncol. 2015, 16, 36–46. [Google Scholar] [CrossRef]
- Sung, H.; Siegel, R.L.; Torre, L.A.; Pearson-Stuttard, J.; Islami, F.; Fedewa, S.A.; Goding Sauer, A.; Shuval, K.; Gapstur, S.M.; Jacobs, E.J.; et al. Global patterns in excess body weight and the associated cancer burden. CA Cancer J. Clin. 2019, 69, 88–112. [Google Scholar] [CrossRef]
- Gonzalez, C.A.; Riboli, E. Diet and cancer prevention: Contributions from the European Prospective Investigation into Cancer and Nutrition (EPIC) study. Eur. J. Cancer 2010, 46, 2555–2562. [Google Scholar] [CrossRef]
- Barberio, A.M.; Alareeki, A.; Viner, B.; Pader, J.; Vena, J.E.; Arora, P.; Friedenreich, C.M.; Brenner, D.R. Central body fatness is a stronger predictor of cancer risk than overall body size. Nat. Commun. 2019, 10, 383. [Google Scholar] [CrossRef] [PubMed]
- Kushi, L.; Giovannucci, E. Dietary fat and cancer. Am. J. Med. 2002, 113 (Suppl. 2), 63S–70S. [Google Scholar] [CrossRef]
- Deng, T.; Lyon, C.J.; Bergin, S.; Caligiuri, M.A.; Hsueh, W.A. Obesity, Inflammation, and Cancer. Annu. Rev. Pathol. Mech. Dis. 2016, 11, 421–449. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, V.; Tuli, H.S.; Varol, A.; Thakral, F.; Yerer, M.B.; Sak, K.; Varol, M.; Jain, A.; Khan, M.A.; Sethi, G. Role of reactive oxygen species in cancer progression: Molecular mechanisms and recent advancements. Biomolecules 2019, 9, E735. [Google Scholar] [CrossRef]
- Schüz, J.; Espina, C.; Villain, P.; Herrero, R.; Leon, M.E.; Minozzi, S.; Romieu, I.; Segnan, N.; Wardle, J.; Wiseman, M.; et al. European Code against Cancer 4th Edition: 12 ways to reduce your cancer risk. Cancer Epidemiol. 2015, 39, S1–S10. [Google Scholar] [CrossRef]
- Anderson, A.S.; Key, T.J.; Norat, T.; Scoccianti, C.; Cecchini, M.; Berrino, F.; Boutron-Ruault, M.C.; Espina, C.; Leitzmann, M.; Powers, H.; et al. European Code against Cancer 4th Edition: Obesity, body fatness and cancer. Cancer Epidemiol. 2015, 39, S34–S45. [Google Scholar] [CrossRef]
- Cao, Y.Y.; Hou, L.; Wang, W. Dietary total fat and fatty acids intake, serum fatty acids and risk of breast cancer: A meta-analysis of prospective cohort studies. Int. J. Cancer 2016, 138, 1894–1904. [Google Scholar] [CrossRef]
- Shen, Q.W.; Yao, Q.Y. Total fat consumption and pancreatic cancer risk: A meta-analysis of epidemiologic studies. Eur. J. Cancer Prev. 2015, 24, 278–285. [Google Scholar] [CrossRef]
- Kim, M.; Park, K. Dietary Fat Intake and Risk of Colorectal Cancer: A Systematic Review and Meta-Analysis of Prospective Studies. Nutrients 2018, 10, 1963. [Google Scholar] [CrossRef] [PubMed]
- Ruan, L.; Cheng, S.P.; Zhu, Q.X. Dietary Fat Intake and the Risk of Skin Cancer: A Systematic Review and Meta-Analysis of Observational Studies. Nutr. Cancer 2019, 2019, 1637910. [Google Scholar] [CrossRef] [PubMed]
- Brennan, S.F.; Woodside, J.V.; Lunny, P.M.; Cardwell, C.R.; Cantwell, M.M. Dietary fat and breast cancer mortality: A systematic review and meta-analysis. Crit. Rev. Food Sci. Nutr. 2017, 57, 1999–2008. [Google Scholar] [CrossRef] [PubMed]
- Woods, S.C.; Seeley, R.J.; Rushing, P.A.; D’Alessio, D.; Tso, P. A Controlled High-Fat Diet Induces an Obese Syndrome in Rats. J. Nutr. 2003, 133, 1081–1087. [Google Scholar] [CrossRef]
- Estrany, M.E.; Proenza, A.M.; Lladó, I.; Gianotti, M. Isocaloric intake of a high-fat diet modifies adiposity and lipid handling in a sex dependent manner in rats. Lipids Health Dis. 2011, 10, 52. [Google Scholar] [CrossRef]
- Meisinger, C.; Rospleszcz, S.; Wintermeyer, E.; Lorbeer, R.; Thorand, B.; Bamberg, F.; Peters, A.; Schlett, C.L.; Linseisen, J. Isocaloric substitution of dietary carbohydrate intake with fat intake and MRI-determined total volumes of visceral, subcutaneous and hepatic fat content in middle-aged adults. Nutrients 2019, 11, 1151. [Google Scholar] [CrossRef]
- Veum, V.L.; Laupsa-Borge, J.; Eng, Ø.; Rostrup, E.; Larsen, T.H.; Nordrehaug, J.E.; Nygård, O.K.; Sagen, J.V.; Gudbrandsen, O.A.; Dankel, S.N.; et al. Visceral adiposity and metabolic syndrome after very high–fat and low-fat isocaloric diets: A randomized controlled trial. Am. J. Clin. Nutr. 2017, 105, 85–99. [Google Scholar] [CrossRef]
- Hariri, N.; Thibault, L. High-fat diet-induced obesity in animal models. Nutr. Res. Rev. 2010, 23, 270–299. [Google Scholar] [CrossRef]
- Doerner, S.K.; Leung, E.S.; Ko, J.S.; Nadeau, J.H.; Berger, N.A. Differential effects of specific dietary fat sources on inflammation and intestinal tumorigenesis. Cancer Res. 2013, 73, 2859. [Google Scholar] [CrossRef]
- Beyaz, S.; Mana, M.D.; Roper, J.; Kedrin, D.; Saadatpour, A.; Hong, S.-J.; Bauer-Rowe, K.E.; Xifaras, M.E.; Akkad, A.; Arias, E.; et al. High-fat diet enhances stemness and tumorigenicity of intestinal progenitors. Nature 2016, 531, 53–58. [Google Scholar] [CrossRef]
- Kim, S.; Yang, X.; Li, Q.; Wu, M.; Costyn, L.; Beharry, Z.; Bartlett, M.G.; Cai, H. Myristoylation of Src kinase mediates Src-induced and high-fat diet–accelerated prostate tumor progression in mice. J. Biol. Chem. 2017, 292, 18422–18433. [Google Scholar] [CrossRef]
- Zhuang, P.; Zhang, Y.; He, W.; Chen, X.; Chen, J.; He, L.; Mao, L.; Wu, F.; Jiao, J. Dietary Fats in Relation to Total and Cause-Specific Mortality in a Prospective Cohort of 521 120 Individuals With 16 Years of Follow-Up. Circ. Res. 2019, 124, 757–768. [Google Scholar] [CrossRef]
- Sellem, L.; Srour, B.; Guéraud, F.; Pierre, F.; Kesse-Guyot, E.; Fiolet, T.; Lavalette, C.; Egnell, M.; Latino-Martel, P.; Fassier, P.; et al. Saturated, mono- and polyunsaturated fatty acid intake and cancer risk: Results from the French prospective cohort NutriNet-Santé. Eur. J. Nutr. 2019, 58, 1515–1527. [Google Scholar] [CrossRef]
- Liss, M.A.; Al-Bayati, O.; Gelfond, J.; Goros, M.; Ullevig, S.; DiGiovanni, J.; Hamilton-Reeves, J.; O’Keefe, D.; Bacich, D.; Weaver, B.; et al. Higher baseline dietary fat and fatty acid intake is associated with increased risk of incident prostate cancer in the SABOR study. Prostate Cancer Prostatic Dis. 2019, 22, 244–251. [Google Scholar] [CrossRef]
- Hou, R.; Wu, Q.J.; Gong, T.T.; Jiang, L. Dietary fat and fatty acid intake and epithelial ovarian cancer risk: Evidence from epidemiological studies. Oncotarget 2015, 6, 43099–43119. [Google Scholar] [CrossRef]
- Nkondjock, A.; Krewski, D.; Johnson, K.C.; Ghadirian, P.; Paulse, B.; Dewar, R.; Dryer, D.; Kreiger, N.; Kliewer, E.; Robson, D.; et al. Specific fatty acid intake and the risk of pancreatic cancer in Canada. Br. J. Cancer 2005, 92, 971–977. [Google Scholar] [CrossRef]
- Zhao, Z.; Feng, Q.; Yin, Z.; Shuang, J.; Bai, B.; Yu, P.; Guo, M.; Zhao, Q. Red and processed meat consumption and colorectal cancer risk: A systematic review and meta-analysis. Oncotarget 2017, 8, 83306–83314. [Google Scholar] [CrossRef]
- Bastide, N.M.; Pierre, F.H.F.; Corpet, D.E. Heme iron from meat and risk of colorectal cancer: A meta-analysis and a review of the mechanisms involved. Cancer Prev. Res. 2011, 4, 177–184. [Google Scholar] [CrossRef]
- Demeyer, D.; Mertens, B.; De Smet, S.; Ulens, M. Mechanisms Linking Colorectal Cancer to the Consumption of (Processed) Red Meat: A Review. Crit. Rev. Food Sci. Nutr. 2016, 56, 2747–2766. [Google Scholar] [CrossRef]
- Nogacka, A.M.; Gómez-Martín, M.; Suárez, A.; González-Bernardo, O.; de los Reyes-Gavilán, C.G.; González, S. Xenobiotics formed during food processing: Their relation with the intestinal microbiota and colorectal cancer. Int. J. Mol. Sci. 2019, 20, E2051. [Google Scholar] [CrossRef]
- López-Suárez, A. Burden of cancer attributable to obesity, type 2 diabetes and associated risk factors. Metabolism 2019, 92, 136–146. [Google Scholar] [CrossRef] [PubMed]
- Castelló, A.; Amiano, P.; Fernández de Larrea, N.; Martín, V.; Alonso, M.H.; Castaño-Vinyals, G.; Pérez-Gómez, B.; Olmedo-Requena, R.; Guevara, M.; Fernandez-Tardon, G.; et al. Low adherence to the western and high adherence to the mediterranean dietary patterns could prevent colorectal cancer. Eur. J. Nutr. 2019, 58, 1495–1505. [Google Scholar] [CrossRef]
- Jeyaraman, M.M.; Abou-Setta, A.M.; Grant, L.; Farshidfar, F.; Copstein, L.; Lys, J.; Gottschalk, T.; Desautels, D.; Czaykowski, P.; Pitz, M.; et al. Dairy product consumption and development of cancer: An overview of reviews. BMJ Open 2019, 9, e023625. [Google Scholar] [CrossRef] [PubMed]
- Sadeghi, A.; Shab-Bidar, S.; Parohan, M.; Djafarian, K. Dietary Fat Intake and Risk of Ovarian Cancer: A Systematic Review and Dose–Response Meta-Analysis of Observational Studies. Nutr. Cancer 2019, 71, 939–953. [Google Scholar] [CrossRef]
- Mazidi, M.; Mikhailidis, D.P.; Sattar, N.; Howard, G.; Graham, I.; Banach, M. Lipid and Blood Pressure Meta-analysis Collaboration (LBPMC) Group Consumption of dairy product and its association with total and cause specific mortality—A population-based cohort study and meta-analysis. Clin. Nutr. 2019, 38, 2833–2845. [Google Scholar] [CrossRef]
- Yang, W.; Sui, J.; Ma, Y.; Simon, T.G.; Chong, D.; Meyerhardt, J.A.; Willett, W.C.; Giovannucci, E.L.; Chan, A.T.; Zhang, X. A prospective study of dairy product intake and the risk of hepatocellular carcinoma in U.S. men and women. Int. J. Cancer 2019, 146, 1241–1249. [Google Scholar] [CrossRef]
- Haskins, C.P.; Henderson, G.; Champ, C.E. Meat, eggs, full-fat dairy, and nutritional boogeymen: Does the way in which animals are raised affect health differently in humans? Crit. Rev. Food Sci. Nutr. 2019, 59, 2709–2719. [Google Scholar] [CrossRef]
- Mancini, A.; Imperlini, E.; Nigro, E.; Montagnese, C.; Daniele, A.; Orrù, S.; Buono, P. Biological and Nutritional Properties of Palm Oil and Palmitic Acid: Effects on Health. Molecules 2015, 20, 17339–17361. [Google Scholar] [CrossRef]
- Gunstone, F.D. Fatty acids—Nomenclature, structure, isolation and structure determination, biosynthesis and chemical synthesis. In Fatty Acid and Lipid Chemistry; Springer US: Boston, MA, USA, 1996; pp. 1–34. [Google Scholar]
- Sette, S.; Le Donne, C.; Piccinelli, R.; Arcella, D.; Turrini, A.; Leclercq, C. The third Italian National Food Consumption Survey, INRAN-SCAI 2005-06—Part 1: Nutrient intakes in Italy. Nutr. Metab. Cardiovasc. Dis. 2011, 21, 922–932. [Google Scholar] [CrossRef]
- Carta, G.; Murru, E.; Banni, S.; Manca, C. Palmitic Acid: Physiological Role, Metabolism and Nutritional Implications. Front. Physiol. 2017, 8, 902. [Google Scholar] [CrossRef]
- Fatima, S.; Hu, X.; Gong, R.H.; Huang, C.; Chen, M.; Wong, H.L.X.; Bian, Z.; Kwan, H.Y. Palmitic acid is an intracellular signaling molecule involved in disease development. Cell. Mol. Life Sci. 2019, 76, 2547–2557. [Google Scholar] [CrossRef]
- Conibear, E.; Davis, N.G. Palmitoylation and depalmitoylation dynamics at a glance. J. Cell Sci. 2010, 123, 4007–4010. [Google Scholar] [CrossRef]
- Ko, P.; Dixon, S.J. Protein palmitoylation and cancer. EMBO Rep. 2018, 19, e46666. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.T.S.; Davis, N.G.; Conibear, E. Targeting the Ras palmitoylation/depalmitoylation cycle in cancer. Biochem. Soc. Trans. 2017, 45, 913–921. [Google Scholar] [CrossRef] [PubMed]
- Landim, B.C.; de Jesus, M.M.; Bosque, B.P.; Zanon, R.G.; da Silva, C.V.; Góes, R.M.; Ribeiro, D.L. Stimulating effect of palmitate and insulin on cell migration and proliferation in PNT1A and PC3 prostate cells: Counteracting role of metformin. Prostate 2018, 78, 731–742. [Google Scholar] [CrossRef] [PubMed]
- Binker-Cosen, M.J.; Richards, D.; Oliver, B.; Gaisano, H.Y.; Binker, M.G.; Cosen-Binker, L.I. Palmitic acid increases invasiveness of pancreatic cancer cells AsPC-1 through TLR4/ROS/NF-κB/MMP-9 signaling pathway. Biochem. Biophys. Res. Commun. 2017, 484, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Pascual, G.; Avgustinova, A.; Mejetta, S.; Martín, M.; Castellanos, A.; Attolini, C.S.-O.; Berenguer, A.; Prats, N.; Toll, A.; Hueto, J.A.; et al. Targeting metastasis-initiating cells through the fatty acid receptor CD36. Nature 2017, 541, 41–45. [Google Scholar] [CrossRef]
- Pan, J.; Fan, Z.; Wang, Z.; Dai, Q.; Xiang, Z.; Yuan, F.; Yan, M.; Zhu, Z.; Liu, B.; Li, C. CD36 mediates palmitate acid-induced metastasis of gastric cancer via AKT/GSK-3β/β-catenin pathway. J. Exp. Clin. Cancer Res. 2019, 38, 52. [Google Scholar] [CrossRef]
- Baumann, J.; Wong, J.; Sun, Y.; Conklin, D.S. Palmitate-induced ER stress increases trastuzumab sensitivity in HER2/neu-positive breast cancer cells. BMC Cancer 2016, 16, 551. [Google Scholar] [CrossRef]
- Lin, L.; Ding, Y.; Wang, Y.; Wang, Z.; Yin, X.; Yan, G.; Zhang, L.; Yang, P.; Shen, H. Functional lipidomics: Palmitic acid impairs hepatocellular carcinoma development by modulating membrane fluidity and glucose metabolism. Hepatology 2017, 66, 432–448. [Google Scholar] [CrossRef]
- Fiorentino, M.; Zadra, G.; Palescandolo, E.; Fedele, G.; Bailey, D.; Fiore, C.; Nguyen, P.L.; Migita, T.; Zamponi, R.; Di Vizio, D.; et al. Overexpression of fatty acid synthase is associated with palmitoylation of Wnt1 and cytoplasmic stabilization of β-catenin in prostate cancer. Lab. Investig. 2008, 88, 1340–1348. [Google Scholar] [CrossRef] [PubMed]
- Sczaniecka, A.K.; Brasky, T.M.; Lampe, J.W.; Patterson, R.E.; White, E. Dietary intake of specific fatty acids and breast cancer risk among postmenopausal women in the VITAL cohort. Nutr. Cancer 2012, 64, 1131–1142. [Google Scholar] [CrossRef] [PubMed]
- Bassett, J.K.; Hodge, A.M.; English, D.R.; MacInnis, R.J.; Giles, G.G. Plasma phospholipids fatty acids, dietary fatty acids, and breast cancer risk. Cancer Causes Control 2016, 27, 759–773. [Google Scholar] [CrossRef] [PubMed]
- Bassett, J.K.; Severi, G.; Hodge, A.M.; MacInnis, R.J.; Gibson, R.A.; Hopper, J.L.; English, D.R.; Giles, G.G. Plasma phospholipid fatty acids, dietary fatty acids and prostate cancer risk. Int. J. Cancer 2013, 133, 1882–1891. [Google Scholar] [CrossRef] [PubMed]
- Crowe, F.L.; Allen, N.E.; Appleby, P.N.; Overvad, K.; Aardestrup, I.V.; Johnsen, N.F.; Tjønneland, A.; Linseisen, J.; Kaaks, R.; Boeing, H.; et al. Fatty acid composition of plasma phospholipids and risk of prostate cancer in a case-control analysis nested within the European Prospective Investigation into Cancer and Nutrition. Am. J. Clin. Nutr. 2008, 88, 1353–1363. [Google Scholar] [CrossRef]
- Merritt, M.A.; Tzoulaki, I.; Van Den Brandt, P.A.; Schouten, L.J.; Tsilidis, K.K.; Weiderpass, E.; Patel, C.J.; Tjønneland, A.; Hansen, L.; Overvad, K.; et al. Nutrient-wide association study of 57 foods/nutrients and epithelial ovarian cancer in the European Prospective Investigation into Cancer and Nutrition study and the Netherlands Cohort Study. Am. J. Clin. Nutr. 2016, 103, 161–167. [Google Scholar] [CrossRef]
- Marangoni, F.; Galli, C.; Ghiselli, A.; Lercker, G.; La Vecchia, C.; Maffeis, C.; Agostoni, C.; Ballardini, D.; Brignoli, O.; Faggiano, P.; et al. Palm oil and human health. Meeting report of NFI: Nutrition Foundation of Italy symposium. Int. J. Food Sci. Nutr. 2017, 68, 643–655. [Google Scholar] [CrossRef]
- Gesteiro, E.; Guijarro, L.; Sánchez-Muniz, F.J.; Del Carmen Vidal-Carou, M.; Troncoso, A.; Venanci, L.; Jimeno, V.; Quilez, J.; Anadón, A.; González-Gross, M. Palm oil on the edge. Nutrients 2019, 11, E2008. [Google Scholar] [CrossRef]
- Li, C.; Zhao, X.; Toline, E.C.; Siegal, G.P.; Evans, L.M.; Ibrahim-Hashim, A.; Desmond, R.A.; Hardy, R.W. Prevention of carcinogenesis and inhibition of breast cancer tumor burden by dietary stearate. Carcinogenesis 2011, 32, 1251–1258. [Google Scholar] [CrossRef][Green Version]
- Shen, M.C.; Zhao, X.; Siegal, G.P.; Desmond, R.; Hardy, R.W. Dietary stearic acid leads to a reduction of visceral adipose tissue in athymic nude mice. PLoS ONE 2014, 9, e104083. [Google Scholar] [CrossRef]
- Qiu, Q.; Li, C.; Song, Y.; Shi, T.; Luo, X.; Zhang, H.; Hu, L.; Yan, X.; Zheng, H.; Liu, M.; et al. Targeted delivery of ibrutinib to tumor-associated macrophages by sialic acid-stearic acid conjugate modified nanocomplexes for cancer immunotherapy. Acta Biomater. 2019, 92, 184–195. [Google Scholar] [CrossRef] [PubMed]
- Amin, H.H.; Meghani, N.M.; Park, C.; Nguyen, V.H.; Tran, T.T.D.; Tran, P.H.L.; Lee, B.J. Fattigation-platform nanoparticles using apo-transferrin stearic acid as a core for receptor-oriented cancer targeting. Colloids Surf. B Biointerfaces 2017, 159, 571–579. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Kumar, V.; Kumar, B.; Deeba, F.; Bano, S.; Kulshreshtha, A.; Gopinath, P.; Negi, Y.S. Lipophilic 5-fluorouracil prodrug encapsulated xylan-stearic acid conjugates nanoparticles for colon cancer therapy. Int. J. Biol. Macromol. 2019, 128, 204–213. [Google Scholar] [CrossRef]
- Jeong, G.W.; Jeong, Y.I.; Nah, J.W. Triggered doxorubicin release using redox-sensitive hyaluronic acid-g-stearic acid micelles for targeted cancer therapy. Carbohydr. Polym. 2019, 209, 161–171. [Google Scholar] [CrossRef]
- Lappano, R.; Sebastiani, A.; Cirillo, F.; Rigiracciolo, D.C.; Galli, G.R.; Curcio, R.; Malaguarnera, R.; Belfiore, A.; Cappello, A.R.; Maggiolini, M. The lauric acid-activated signaling prompts apoptosis in cancer cells. Cell Death Discov. 2017, 3, 17063. [Google Scholar] [CrossRef]
- Fauser, J.K.; Matthews, G.M.; Cummins, A.G.; Howarth, G.S. Induction of Apoptosis by the Medium-Chain Length Fatty Acid Lauric Acid in Colon Cancer Cells due to Induction of Oxidative Stress. Chemotherapy 2013, 59, 214–224. [Google Scholar] [CrossRef]
- Kadochi, Y.; Mori, S.; Fujiwara-Tani, R.; Luo, Y.; Nishiguchi, Y.; Kishi, S.; Fujii, K.; Ohmori, H.; Kuniyasu, H. Remodeling of energy metabolism by a ketone body and medium-chain fatty acid suppressed the proliferation of CT26 mouse colon cancer cells. Oncol. Lett. 2017, 14, 673–680. [Google Scholar] [CrossRef]
- Weng, W.H.; Leung, W.H.; Pang, Y.J.; Hsu, H.H. Lauric acid can improve the sensitization of Cetuximab in KRAS/BRAF mutated colorectal cancer cells by retrievable microRNA-378 expression. Oncol. Rep. 2016, 35, 107–116. [Google Scholar] [CrossRef]
- De Matteis, V.; Cascione, M.; De Giorgi, M.L.; Leporatti, S.; Rinaldi, R. Encapsulation of Thermo-Sensitive Lauric Acid in Silica Shell: A Green Derivate for Chemo-Thermal Therapy in Breast Cancer Cell. Molecules 2019, 24, E2034. [Google Scholar] [CrossRef]
- Jiang, L.; Liang, X.; Liu, G.; Zhou, Y.; Ye, X.; Chen, X.; Miao, Q.; Gao, L.; Zhang, X.; Mei, L. The mechanism of lauric acid-modified protein nanocapsules escape from intercellular trafficking vesicles and its implication for drug delivery. Drug Deliv. 2018, 25, 985–994. [Google Scholar] [CrossRef]
- Silva, M.O.D.; Carneiro, M.L.B.; Siqueira, J.L.N.; Báo, S.N.; Souza, A.R. Development of a Promising Antitumor Compound Based on Rhodium(II) Succinate Associated with Iron Oxide Nanoparticles Coated with Lauric Acid/Albumin Hybrid: Synthesis, Colloidal Stability and Cytotoxic Effect in Breast Carcinoma Cells. J. Nanosci. Nanotechnol. 2017, 18, 3832–3843. [Google Scholar] [CrossRef]
- Zaloga, J.; Feoktystov, A.; Garamus, V.M.; Karawacka, W.; Ioffe, A.; Brückel, T.; Tietze, R.; Alexiou, C.; Lyer, S. Studies on the adsorption and desorption of mitoxantrone to lauric acid/albumin coated iron oxide nanoparticles. Colloids Surf. B Biointerfaces 2018, 161, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Udenwobele, D.I.; Su, R.C.; Good, S.V.; Ball, T.B.; Shrivastav, S.V.; Shrivastav, A. Myristoylation: An important protein modification in the immune response. Front. Immunol. 2017, 8, 751. [Google Scholar] [CrossRef]
- Kurahashi, N.; Inoue, M.; Iwasaki, M.; Sasazuki, S.; Tsugane, S. Dairy product, saturated fatty acid, and calcium intake and prostate cancer in a prospective cohort of Japanese men. Cancer Epidemiol. Biomark. Prev. 2008, 17, 930–937. [Google Scholar] [CrossRef]
- Ying, M.; Wang, S.; Zhang, M.; Wang, R.; Zhu, H.; Ruan, H.; Ran, D.; Chai, Z.; Wang, X.; Lu, W. Myristic Acid-Modified DA7R Peptide for Whole-Process Glioma-Targeted Drug Delivery. Acs Appl. Mater. Interfaces 2018, 10, 19473–19482. [Google Scholar] [CrossRef]
- Narayanan, A.; Baskaran, S.A.; Amalaradjou, M.A.R.; Venkitanarayanan, K. Anticarcinogenic properties of medium chain fatty acids on human colorectal, skin and breast cancer cells in vitro. Int. J. Mol. Sci. 2015, 16, 5014–5027. [Google Scholar] [CrossRef]
- McNabney, S.M.; Henagan, T.M. Short chain fatty acids in the colon and peripheral tissues: A focus on butyrate, colon cancer, obesity and insulin resistance. Nutrients 2017, 9, E1348. [Google Scholar] [CrossRef]
- Gastón, R.; María Eugenia, P.; Das, U.N.; Eynard, A.R. Polyunsaturated Fatty Acids Differentially Modulate Cell Proliferation and Endocannabinoid System in Two Human Cancer Lines. Arch. Med. Res. 2017, 48, 46–54. [Google Scholar] [CrossRef]
- Menendez, J.A.; Vellon, L.; Colomer, R.; Lupu, R. Oleic acid, the main monounsaturated fatty acid of olive oil, suppresses Her-2/neu (erbB-2) expression and synergistically enhances the growth inhibitory effects of trastuzumab (HerceptinTM) in breast cancer cells with Her-2/neu oncogene amplification. Ann. Oncol. 2005, 16, 359–371. [Google Scholar] [CrossRef]
- Li, S.; Zhou, T.; Li, C.; Dai, Z.; Che, D.; Yao, Y.; Li, L.; Ma, J.; Yang, X.; Gao, G. High metastatic gastric and breast cancer cells consume oleic acid in an AMPK dependent manner. PLoS ONE 2014, 9, e97330. [Google Scholar] [CrossRef]
- Angelucci, C.; D’Alessio, A.; Iacopino, F.; Proietti, G.; Di Leone, A.; Masetti, R.; Sica, G. Pivotal role of human stearoyl-CoA desaturases (SCD1 and 5) in breast cancer progression: Oleic acid-based effect of SCD1 on cell migration and a novel pro-cell survival role for SCD5. Oncotarget 2018, 9, 24364–24380. [Google Scholar] [CrossRef] [PubMed]
- Marcial-Medina, C.; Ordoñez-Moreno, A.; Gonzalez-Reyes, C.; Cortes-Reynosa, P.; Salazar, E.P. Oleic acid induces migration through a FFAR1/4, EGFR and AKT-dependent pathway in breast cancer cells. Endocr. Connect. 2019, 8, 252–265. [Google Scholar] [CrossRef]
- Xiang, F.; Wu, K.; Liu, Y.; Shi, L.; Wang, D.; Li, G.; Tao, K.; Wang, G. Omental adipocytes enhance the invasiveness of gastric cancer cells by oleic acid-induced activation of the PI3K-Akt signaling pathway. Int. J. Biochem. Cell Biol. 2017, 84, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Storniolo, C.E.; Martínez-Hovelman, N.; Martínez-Huélamo, M.; Lamuela-Raventos, R.M.; Moreno, J.J. Extra Virgin Olive Oil Minor Compounds Modulate Mitogenic Action of Oleic Acid on Colon Cancer Cell Line. J. Agric. Food Chem. 2019, 67, 11420–11427. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.H.; Qing, C.P.; Sheng, Z.Z.; Li, G.Z.; Man, L.B.; Xu, T. Oleic acid promotes cell invasion through an integrin-linked kinase signaling pathway in renal cell carcinoma. Nutr. Cancer 2019, 2019, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.; Su, C.; Luo, X.; Zeng, H.; Zhao, L.; Wei, L.; Zhang, X.; Varghese, Z.; Moorhead, J.F.; Chen, Y.; et al. Dietary oleic acid-induced CD36 promotes cervical cancer cell growth and metastasis via up-regulation Src/ERK pathway. Cancer Lett. 2018, 438, 76–85. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Wang, W.; He, Q.; Wu, Y.; Lu, Z.; Sun, J.; Liu, Z.; Shao, Y.; Wang, A. Oleic acid induces apoptosis and autophagy in the treatment of tongue squamous cell carcinomas. Sci. Rep. 2017, 7, 11277. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Quesada, C.; López-Biedma, A.; Warleta, F.; Campos, M.; Beltrán, G.; Gaforio, J.J. Bioactive properties of the main triterpenes found in olives, virgin olive oil, and leaves of olea europaea. J. Agric. Food Chem. 2013, 61, 12173–12182. [Google Scholar] [CrossRef]
- Psaltopoulou, T.; Kosti, R.I.; Haidopoulos, D.; Dimopoulos, M.; Panagiotakos, D.B. Olive oil intake is inversely related to cancer prevalence: A systematic review and a meta-analysis of 13800 patients and 23340 controls in 19 observational studies. Lipids Health Dis. 2011, 10, 127. [Google Scholar] [CrossRef]
- Guasch-Ferré, M.; Zong, G.; Willett, W.C.; Zock, P.L.; Wanders, A.J.; Hu, F.B.; Sun, Q. Associations of Monounsaturated Fatty Acids From Plant and Animal Sources With Total and Cause-Specific Mortality in Two US Prospective Cohort Studies. Circ. Res. 2019, 124, 1266–1275. [Google Scholar] [CrossRef]
- Gaforio, J.J.; Visioli, F.; Alarcón-de-la-Lastra, C.; Castañer, O.; Delgado-Rodríguez, M.; Fitó, M.; Hernández, A.F.; Huertas, J.R.; Martínez-González, M.A.; Menendez, J.A.; et al. Virgin olive oil and health: Summary of the iii international conference on virgin olive oil and health consensus report, JAEN (Spain) 2018. Nutrients 2019, 11, E2039. [Google Scholar] [CrossRef] [PubMed]
- Ros, E. Health benefits of nut consumption. Nutrients 2010, 2, 652–682. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Shin, A.; Oh, J.H.; Kim, J. The relationship between nut intake and risk of colorectal cancer: A case control study. Nutr. J. 2018, 17, 37. [Google Scholar] [CrossRef] [PubMed]
- Obón-Santacana, M.; Luján-Barroso, L.; Freisling, H.; Naudin, S.; Boutron-Ruault, M.; Mancini, F.R.; Rebours, V.; Kühn, T.; Katzke, V.; Boeing, H.; et al. Consumption of nuts and seeds and pancreatic ductal adenocarcinoma risk in the European Prospective Investigation into Cancer and Nutrition. Int. J. Cancer 2020, 146, 76–84. [Google Scholar] [CrossRef] [PubMed]
- Flax Council of Canada. Flax—A Healthy Food. Available online: https://flaxcouncil.ca/resources/nutrition/general-nutrition-information/flax-a-healthy-food/ (accessed on 5 July 2019).
- Saini, R.K.; Keum, Y.S. Omega-3 and omega-6 polyunsaturated fatty acids: Dietary sources, metabolism, and significance—A review. Life Sci. 2018, 203, 255–267. [Google Scholar] [CrossRef] [PubMed]
- Glaser, C.; Heinrich, J.; Koletzko, B. Role of FADS1 and FADS2 polymorphisms in polyunsaturated fatty acid metabolism. Metabolism 2010, 59, 993–999. [Google Scholar] [CrossRef]
- Calder, P.C. Omega-3 fatty acids and inflammatory processes. Nutrients 2010, 2, 355–374. [Google Scholar] [CrossRef]
- Innes, J.K.; Calder, P.C. Omega-6 fatty acids and inflammation. Prostaglandins Leukot. Essent. Fat. Acids 2018, 132, 41–48. [Google Scholar] [CrossRef]
- Dinicolantonio, J.J.; O’Keefe, J.H. Importance of maintaining a low omega-6/omega-3 ratio for reducing inflammation. Open Hear. 2018, 5, e000946. [Google Scholar] [CrossRef]
- Simopoulos, A.P. The Importance of the Omega-6/Omega-3 Fatty Acid Ratio in Cardiovascular Disease and Other Chronic Diseases. Exp. Biol. Med. 2008, 233, 674–688. [Google Scholar] [CrossRef]
- Stanford, J.L.; Herrinton, L.J.; Schwartz, S.M.; Weiss, N.S. Breast cancer incidence in asian migrants to the United States and their descendants. Epidemiology 1995, 6, 181–183. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, H.; Ross, R.K.; Bernstein, L.; Henderson, B.E.; Mack, T.M.; Yatani, R. Cancers of the prostate and breast among japanese and white immigrants in los angeles county. Br. J. Cancer 1991, 63, 963–966. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Demissie, K.; Lu, S.E.; Rhoads, G.G. Cancer incidence among Korean-American immigrants in the United States and native Koreans in South Korea. Cancer Control 2007, 14, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Bratton, B.A.; Maly, I.V.; Hofmann, W.A. Effect of polyunsaturated fatty acids on proliferation and survival of prostate cancer cells. PLoS ONE 2019, 14, e0219822. [Google Scholar] [CrossRef]
- Meng, H.; Shen, Y.; Shen, J.; Zhou, F.; Shen, S.; Das, U.N. Effect of n-3 and n-6 unsaturated fatty acids on prostate cancer (PC-3) and prostate epithelial (RWPE-1) cells in vitro. Lipids Health Dis. 2013, 12, 160. [Google Scholar] [CrossRef]
- Lu, X.; Yu, H.; Ma, Q.; Shen, S.; Das, U.N. Linoleic acid suppresses colorectal cancer cell growth by inducing oxidant stress and mitochondrial dysfunction. Lipids Health Dis. 2010, 9, 106. [Google Scholar] [CrossRef]
- Mielczarek-Puta, M.; Otto-Ślusarczyk, D.; Chrzanowska, A.; Filipek, A.; Graboń, W. Telmisartan Influences the Antiproliferative Activity of Linoleic Acid in Human Colon Cancer Cells. Nutr. Cancer 2020, 72, 98–109. [Google Scholar] [CrossRef]
- Choi, Y.H. Linoleic Acid-Induced Growth Inhibition of Human Gastric Epithelial Adenocarcinoma AGS Cells is Associated with Down-Regulation of Prostaglandin E2 Synthesis and Telomerase Activity. J. Cancer Prev. 2014, 19, 31–38. [Google Scholar] [CrossRef]
- Diaz-Aragon, R.; Ramirez-Ricardo, J.; Cortes-Reynosa, P.; Simoni-Nieves, A.; Gomez-Quiroz, L.E.; Perez Salazar, E. Role of phospholipase D in migration and invasion induced by linoleic acid in breast cancer cells. Mol. Cell. Biochem. 2019, 457, 119–132. [Google Scholar] [CrossRef]
- Gonzalez-Reyes, C.; Marcial-Medina, C.; Cervantes-Anaya, N.; Cortes-Reynosa, P.; Salazar, E.P. Migration and invasion induced by linoleic acid are mediated through fascin in MDA-MB-231 breast cancer cells. Mol. Cell. Biochem. 2018, 443, 1–10. [Google Scholar] [CrossRef]
- Serna-Marquez, N.; Diaz-Aragon, R.; Reyes-Uribe, E.; Cortes-Reynosa, P.; Salazar, E.P. Linoleic acid induces migration and invasion through FFAR4- and PI3K-/Akt-dependent pathway in MDA-MB-231 breast cancer cells. Med. Oncol. 2017, 34, 111. [Google Scholar] [CrossRef] [PubMed]
- Espinosa-Neira, R.; Mejia-Rangel, J.; Cortes-Reynosa, P.; Salazar, E.P. Linoleic acid induces an EMT-like process in mammary epithelial cells MCF10A. Int. J. Biochem. Cell Biol. 2011, 43, 1782–1791. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.C.; Liang, A.; Lin, Y.H.; Guo, Y.R.; Huang, S.Y. Low dietary n-6/n-3 polyunsaturated fatty acid ratio prevents induced oral carcinoma in a hamster pouch model. Prostaglandins Leukot. Essent. Fat. Acids 2018, 136, 67–75. [Google Scholar] [CrossRef]
- Cheon, E.C.; Strouch, M.J.; Barron, M.R.; Ding, Y.; Melstrom, L.G.; Krantz, S.B.; Mullapudi, B.; Adrian, K.; Rao, S.; Adrian, T.E.; et al. Alteration of strain background and a high omega-6 fat diet induces earlier onset of pancreatic neoplasia in EL-Kras transgenic mice. Int. J. Cancer 2011, 128, 2783–2792. [Google Scholar] [CrossRef]
- Matsuoka, T.; Adair, J.E.; Lih, F.B.; Hsi, L.C.; Rubino, M.; Eling, T.E.; Tomer, K.B.; Yashiro, M.; Hirakawa, K.; Olden, K.; et al. Elevated dietary linoleic acid increases gastric carcinoma cell invasion and metastasis in mice. Br. J. Cancer 2010, 103, 1182–1191. [Google Scholar] [CrossRef]
- Nishioka, N.; Matsuoka, T.; Yashiro, M.; Hirakawa, K.; Olden, K.; Roberts, J.D. Linoleic acid enhances angiogenesis through suppression of angiostatin induced by plasminogen activator inhibitor 1. Br. J. Cancer 2011, 105, 1750–1758. [Google Scholar] [CrossRef][Green Version]
- Romagnolo, D.F.; Donovan, M.G.; Doetschman, T.C.; Selmin, O.I. N-6 linoleic acid induces epigenetics alterations associated with colonic inflammation and cancer. Nutrients 2019, 11, 171. [Google Scholar] [CrossRef]
- Manzanares, M.Á.; de Miguel, C.; Ruiz de Villa, M.C.; Santella, R.M.; Escrich, E.; Solanas, M. Dietary lipids differentially modulate the initiation of experimental breast carcinogenesis through their influence on hepatic xenobiotic metabolism and DNA damage in the mammary gland. J. Nutr. Biochem. 2017, 43, 68–77. [Google Scholar] [CrossRef]
- Nindrea, R.D.; Aryandono, T.; Lazuardi, L.; Dwiprahasto, I. Association of dietary intake ratio of n-3/n-6 polyunsaturated fatty acids with breast cancer risk in Western and Asian countries: A meta-analysis. Asian Pac. J. Cancer Prev. 2019, 20, 1321–1327. [Google Scholar] [CrossRef]
- LeMay-Nedjelski, L.; Mason-Ennis, J.K.; Taibi, A.; Comelli, E.M.; Thompson, L.U. Omega-3 polyunsaturated fatty acids time-dependently reduce cell viability and oncogenic microRNA-21 expression in estrogen receptor-positive breast cancer cells (MCF-7). Int. J. Mol. Sci. 2018, 19, 244. [Google Scholar] [CrossRef]
- Siddiqui, R.A.; Harvey, K.A.; Xu, Z.; Bammerlin, E.M.; Walker, C.; Altenburg, J.D. Docosahexaenoic acid: A natural powerful adjuvant that improves efficacy for anticancer treatment with no adverse effects. BioFactors 2011, 37, 399–412. [Google Scholar] [CrossRef]
- Pizato, N.; Luzete, B.C.; Kiffer, L.F.M.V.; Corrêa, L.H.; De Oliveira Santos, I.; Assumpção, J.A.F.; Ito, M.K.; Magalhães, K.G. Omega-3 docosahexaenoic acid induces pyroptosis cell death in triple-negative breast cancer cells. Sci. Rep. 2018, 8, 1952. [Google Scholar] [CrossRef]
- Huang, L.H.; Chung, H.Y.; Su, H.M. Docosahexaenoic acid reduces sterol regulatory element binding protein-1 and fatty acid synthase expression and inhibits cell proliferation by inhibiting pAkt signaling in a human breast cancer MCF-7 cell line. BMC Cancer 2017, 17, 890. [Google Scholar] [CrossRef]
- Murad, L.B.; da Silva Nogueira, P.; de Araújo, W.M.; Sousa-Squiavinato, A.C.M.; Rocha, M.R.; de Souza, W.F.; de-Freitas-Junior, J.; Barcellos-de-Souza, P.; Bastos, L.G.; Morgado-Díaz, J.A. Docosahexaenoic acid promotes cell cycle arrest and decreases proliferation through WNT/β-catenin modulation in colorectal cancer cells exposed to γ-radiation. BioFactors 2019, 45, 24–34. [Google Scholar] [CrossRef] [PubMed]
- Moradi Sarabi, M.; Zahedi, S.A.; Pajouhi, N.; Khosravi, P.; Bagheri, S.; Ahmadvand, H.; Shahryarhesami, S. The effects of dietary polyunsaturated fatty acids on miR-126 promoter DNA methylation status and VEGF protein expression in the colorectal cancer cells. Genes Nutr. 2018, 13, 32. [Google Scholar] [CrossRef]
- Tylichová, Z.; Neča, J.; Topinka, J.; Milcová, A.; Hofmanová, J.; Kozubík, A.; Machala, M.; Vondráček, J. n-3 Polyunsaturated fatty acids alter benzo[a]pyrene metabolism and genotoxicity in human colon epithelial cell models. Food Chem. Toxicol. 2019, 124, 374–384. [Google Scholar] [CrossRef]
- Serini, S.; Ottes Vasconcelos, R.; Fasano, E.; Calviello, G. Epigenetic regulation of gene expression and M2 macrophage polarization as new potential omega-3 polyunsaturated fatty acid targets in colon inflammation and cancer. Expert Opin. Targets 2016, 20, 843–858. [Google Scholar] [CrossRef]
- Jeong, S.; Kim, D.Y.; Kang, S.H.; Yun, H.K.; Kim, J.L.; Kim, B.R.; Park, S.H.; Na, Y.J.; Jo, M.J.; Jeong, Y.A.; et al. Docosahexaenoic acid enhances oxaliplatin-induced autophagic cell death via the ER stress/sesn2 pathway in colorectal cancer. Cancers 2019, 11, 982. [Google Scholar] [CrossRef]
- Park, M.; Lim, J.W.; Kim, H. Docoxahexaenoic acid induces apoptosis of pancreatic cancer cells by suppressing activation of STAT3 and Nf-κB. Nutrients 2018, 10, 1621. [Google Scholar] [CrossRef]
- Yin, Y.; Sui, C.; Meng, F.; Ma, P.; Jiang, Y. The omega-3 polyunsaturated fatty acid docosahexaenoic acid inhibits proliferation and progression of non-small cell lung cancer cells through the reactive oxygen species-mediated inactivation of the PI3K /Akt pathway. Lipids Health Dis. 2017, 16, 87. [Google Scholar] [CrossRef]
- Bai, X.; Shao, J.; Zhou, S.; Zhao, Z.; Li, F.; Xiang, R.; Zhao, A.Z.; Pan, J. Inhibition of lung cancer growth and metastasis by DHA and its metabolite, RvD1, through miR-138-5p/FOXC1 pathway. J. Exp. Clin. Cancer Res. 2019, 38, 479. [Google Scholar] [CrossRef] [PubMed]
- Song, K.S.; Jing, K.; Kim, J.S.; Yun, E.J.; Shin, S.; Seo, K.S.; Park, J.H.; Heo, J.Y.; Kang, J.X.; Suh, K.S.; et al. Omega-3-polyunsaturated fatty acids suppress pancreatic cancer cell growth in vitro and in vivo via downregulation of Wnt/beta-catenin signaling. Pancreatology 2011, 11, 574–584. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.Y.-K.; Sit, W.-H.; Fan, S.-T.; Man, K.; Jor, I.W.-Y.; Wong, L.L.-Y.; Wan, M.L.-Y.; Tan-Un, K.C.; Wan, J.M.-F. The cell cycle effects of docosahexaenoic acid on human metastatic hepatocellular carcinoma proliferation. Int. J. Oncol. 2010, 36, 991–998. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Han, L.; Zhang, Y.; Meng, M.; Cheng, D.; Wang, C. Eicosapentaenoic acid induced SKOV-3 cell apoptosis through ERK1/2-mTOR-NF-κB pathways. Anticancer. Drugs 2016, 27, 635–642. [Google Scholar] [CrossRef]
- Wang, Y.C.; Wu, Y.N.; Wang, S.L.; Lin, Q.H.; He, M.F.; Liu, Q.L.; Wang, J.H. Docosahexaenoic Acid Modulates Invasion and Metastasis of Human Ovarian Cancer via Multiple Molecular Pathways. Int. J. Gynecol. Cancer 2016, 26, 994–1003. [Google Scholar] [CrossRef] [PubMed]
- So, W.W.; Liu, W.N.; Leung, K.N. Omega-3 polyunsaturated fatty acids trigger cell cycle arrest and induce apoptosis in human neuroblastoma LA-N-1 cells. Nutrients 2015, 7, 6956–6973. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Jing, K.; Shin, S.; Jeong, S.; Han, S.H.; Oh, H.; Yoo, Y.S.; Han, J.; Jeon, Y.J.; Heo, J.Y.; et al. ω3-polyunsaturated fatty acids induce cell death through apoptosis and autophagy in glioblastoma cells: In vitro and in vivo. Oncol. Rep. 2018, 39, 239–246. [Google Scholar] [CrossRef]
- Picou, F.; Debeissat, C.; Bourgeais, J.; Gallay, N.; Ferrié, E.; Foucault, A.; Ravalet, N.; Maciejewski, A.; Vallet, N.; Ducrocq, E.; et al. n-3 Polyunsaturated fatty acids induce acute myeloid leukemia cell death associated with mitochondrial glycolytic switch and Nrf2 pathway activation. Pharm. Res. 2018, 136, 45–55. [Google Scholar] [CrossRef]
- Sam, M.R.; Esmaeillou, M.; Shokrgozar, M.A. Fish-Oil-Derived DHA-mediated Enhancement of Apoptosis in Acute Lymphoblastic Leukemia Cells is Associated with Accumulation of p53, Downregulation of Survivin, and Caspase-3 Activation. Nutr. Cancer 2017, 69, 64–73. [Google Scholar] [CrossRef]
- Khadge, S.; Thiele, G.M.; Sharp, J.G.; McGuire, T.R.; Klassen, L.W.; Black, P.N.; DiRusso, C.C.; Cook, L.; Talmadge, J.E. Long-chain omega-3 polyunsaturated fatty acids decrease mammary tumor growth, multiorgan metastasis and enhance survival. Clin. Exp. Metastasis 2018, 35, 797–818. [Google Scholar] [CrossRef]
- Vara-Messler, M.; Pasqualini, M.E.; Comba, A.; Silva, R.; Buccellati, C.; Trenti, A.; Trevisi, L.; Eynard, A.R.; Sala, A.; Bolego, C.; et al. Increased dietary levels of α-linolenic acid inhibit mammary tumor growth and metastasis. Eur. J. Nutr. 2017, 56, 509–519. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Li, K.; Gao, J.; Guo, X.; Lu, M.; Li, Z.; Li, D. Maternal exposure to an n-3 polyunsaturated fatty acid diet decreases mammary cancer risk of female offspring in adulthood. Food Funct. 2018, 9, 5768–5777. [Google Scholar] [CrossRef] [PubMed]
- Huang, Q.; Wen, J.; Chen, G.; Ge, M.; Gao, Y.; Ye, X.; Liu, C.; Cai, C. Omega-3 polyunsaturated fatty acids inhibited tumor growth via preventing the decrease of genomic DNA methylation in colorectal cancer rats. Nutr. Cancer 2016, 68, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Notarnicola, M.; Tutino, V.; De Nunzio, V.; Dituri, F.; Caruso, M.G.; Giannelli, G. Dietary ω-3 polyunsaturated fatty acids inhibit tumor growth in transgenic Apcmin/+ mice, correlating with CB1 receptor up-regulation. Int. J. Mol. Sci. 2017, 18, 485. [Google Scholar] [CrossRef]
- Wang, W.; Yang, J.; Nimiya, Y.; Lee, K.S.S.; Sanidad, K.; Qi, W.; Sukamtoh, E.; Park, Y.; Liu, Z.; Zhang, G. ω-3 Polyunsaturated fatty acids and their cytochrome P450-derived metabolites suppress colorectal tumor development in mice. J. Nutr. Biochem. 2017, 48, 29–35. [Google Scholar] [CrossRef]
- Ding, Y.; Mullapudi, B.; Torres, C.; Mascariñas, E.; Mancinelli, G.; Diaz, A.M.; McKinney, R.; Barron, M.; Schultz, M.; Heiferman, M.; et al. Omega-3 fatty acids prevent early pancreatic carcinogenesis via repression of the AKT pathway. Nutrients 2018, 10, 1289. [Google Scholar] [CrossRef]
- Li, J.; Gu, Z.; Pan, Y.; Wang, S.; Chen, H.; Zhang, H.; Chen, W.; Chen, Y.Q. Dietary supplementation of α-linolenic acid induced conversion of n-3 LCPUFAs and reduced prostate cancer growth in a mouse model. Lipids Health Dis. 2017, 16, 136. [Google Scholar] [CrossRef]
- Zheng, H.; Tang, H.; Liu, M.; He, M.; Lai, P.; Dong, H.; Lin, J.; Jia, C.; Zhong, M.; Dai, Y.; et al. Inhibition of endometrial cancer by n-3 polyunsaturated fatty acids in preclinical models. Cancer Prev. Res. 2014, 7, 824–834. [Google Scholar] [CrossRef]
- Lu, X.; Ding, X.; Jing, L. Effect of Mechanism of Action of Different ω-6/ω-3 Polyunsaturated Fatty Acids Ratio on the Growth of Endometrial Carcinoma Mice. Cell Biochem. Biophys. 2015, 71, 1671–1676. [Google Scholar] [CrossRef]
- Serini, S.; Cassano, R.; Trombino, S.; Calviello, G. Nanomedicine-based formulations containing ω-3 polyunsaturated fatty acids: Potential application in cardiovascular and neoplastic diseases. Int. J. Nanomed. 2019, 14, 2809–2828. [Google Scholar] [CrossRef]
- Witte, T.R.; Hardman, W.E. The Effects of Omega-3 Polyunsaturated Fatty Acid Consumption on Mammary Carcinogenesis. Lipids 2015, 50, 437–446. [Google Scholar] [CrossRef] [PubMed]
- Nindrea, R.D.; Aryandono, T.; Lazuardi, L.; Dwiprahasto, I. Protective effect of omega-3 fatty acids in fish consumption against breast cancer in Asian patients: A meta-analysis. Asian Pac. J. Cancer Prev. 2019, 20, 327–332. [Google Scholar] [CrossRef] [PubMed]
- Aglago, E.K.; Huybrechts, I.; Murphy, N.; Casagrande, C.; Nicolas, G.; Pischon, T.; Fedirko, V.; Severi, G.; Boutron-Ruault, M.-C.; Fournier, A.; et al. Consumption of Fish and Long-chain n-3 Polyunsaturated Fatty Acids Is Associated With Reduced Risk of Colorectal Cancer in a Large European Cohort. Clin. Gastroenterol. Hepatol. 2020, 18, 654–666. [Google Scholar] [CrossRef] [PubMed]
- Brasky, T.M.; Rodabough, R.J.; Liu, J.; Kurta, M.L.; Wise, L.A.; Orchard, T.S.; Cohn, D.E.; Belury, M.A.; White, E.; Manson, J.E.; et al. Long-chain ω-3 fatty acid intake and endometrial cancer risk in the Women’s Health Initiative. Am. J. Clin. Nutr. 2015, 101, 824–834. [Google Scholar] [CrossRef]
- Dinwiddie, M.T.; Terry, P.D.; Whelan, J.; Patzer, R.E. Omega-3 Fatty Acid Consumption and Prostate Cancer: A Review of Exposure Measures and Results of Epidemiological Studies. J. Am. Coll. Nutr. 2016, 35, 452–468. [Google Scholar] [CrossRef]
- D’Eliseo, D.; Velotti, F. Omega-3 Fatty Acids and Cancer Cell Cytotoxicity: Implications for Multi-Targeted Cancer Therapy. J. Clin. Med. 2016, 5, 15. [Google Scholar] [CrossRef] [PubMed]
- Elbarbary, N.S.; Ismail, E.A.R.; Farahat, R.K.; El-Hamamsy, M. ω-3 fatty acids as an adjuvant therapy ameliorates methotrexate-induced hepatotoxicity in children and adolescents with acute lymphoblastic leukemia: A randomized placebo-controlled study. Nutrition 2016, 32, 41–47. [Google Scholar] [CrossRef]
- Hashemipour, M.A.; Barzegari, S.; Kakoie, S.; Aghahi, R.H. Effects of Omega-3 Fatty Acids Against Chemotherapy-induced Mucositis: A Double-blind Randomized Clinical Trial. Wounds 2017, 29, 360–366. [Google Scholar]
- Roche, H.M.; Noone, E.; Sewter, C.; McBennett, S.M.; Savage, D.; Gibney, M.J.; O’Rahilly, S.; Vidal-Puig, A.J. Isomer-dependent metabolic effects of conjugated linoleic acid: Insights from molecular markers sterol regulatory element-binding protein-1c and LXRα. Diabetes 2002, 51, 2037–2044. [Google Scholar] [CrossRef]
- Stender, S.; Astrup, A.; Dyerberg, J. Ruminant and industrially produced trans fatty acids: Health aspects. Food Nutr. Res. 2008, 52, 1651. [Google Scholar] [CrossRef]
- Coulston, A.M.; Boushey, C.J.; Ferruzzi, M.G. Nutrition in the Prevention and Treatment of Disease; Coulston, A.M., Boushey, C.J., Ferruzzi, M.G., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2013; ISBN 9780123918840. [Google Scholar]
- Alonso, L.; Fontecha, J.; Lozada, L.; Fraga, M.J.; Juárez, M. Fatty Acid Composition of Caprine Milk: Major, Branched-Chain, and Trans Fatty Acids. J. Dairy Sci. 1999, 82, 878–884. [Google Scholar] [CrossRef]
- World Health Organization. WHO Plan to Eliminate Industrially-Produced Trans-Fatty Acids from Global Food Supply. Available online: https://www.who.int/news-room/detail/14-05-2018-who-plan-to-eliminate-industrially-produced-trans-fatty-acids-from-global-food-supply (accessed on 5 July 2019).
- Islam, M.A.; Amin, M.N.; Siddiqui, S.A.; Hossain, M.P.; Sultana, F.; Kabir, M.R. Trans fatty acids and lipid profile: A serious risk factor to cardiovascular disease, cancer and diabetes. Diabetes Metab. Syndr. Clin. Res. Rev. 2019, 13, 1643–1647. [Google Scholar] [CrossRef] [PubMed]
- Den Hartigh, L.J. Conjugated linoleic acid effects on cancer, obesity, and atherosclerosis: A review of pre-clinical and human trials with current perspectives. Nutrients 2018, 11, 370. [Google Scholar] [CrossRef] [PubMed]
- Laake, I.; Carlsen, M.H.; Pedersen, J.I.; Weiderpass, E.; Selmer, R.; Kirkhus, B.; Thune, I.; Veierød, M.B. Intake of trans fatty acids from partially hydrogenated vegetable and fish oils and ruminant fat in relation to cancer risk. Int. J. Cancer 2013, 132, 1389–1403. [Google Scholar] [CrossRef] [PubMed]
- Ohmori, H.; Fujii, K.; Kadochi, Y.; Mori, S.; Nishiguchi, Y.; Fujiwara, R.; Kishi, S.; Sasaki, T.; Kuniyasu, H. Elaidic acid, a trans-fatty acid, enhances the metastasis of colorectal cancer cells. Pathobiology 2017, 84, 144–151. [Google Scholar] [CrossRef]
- Tanabe, E.; Kitayoshi, M.; Fujii, K.; Ohmori, H.; Luo, Y.; Kadochi, Y.; Mori, S.; Fujiwara, R.; Nishiguchi, Y.; Sasaki, T.; et al. Fatty acids inhibit anticancer effects of 5-fluorouracil in mouse cancer cell lines. Oncol. Lett. 2017, 14, 681–686. [Google Scholar] [CrossRef]
- Fujii, K.; Luo, Y.; Fujiwara-Tani, R.; Kishi, S.; He, S.; Yang, S.; Sasaki, T.; Ohmori, H.; Kuniyasu, H. Pro-metastatic intracellular signaling of the elaidic trans fatty acid. Int. J. Oncol. 2017, 50, 85–92. [Google Scholar] [CrossRef]
- Awad, A.B. Trans fatty acids in tumor development and the host survival. J. Natl. Cancer Inst. 1981, 67, 189–192. [Google Scholar]
- Ma, W.W.; Zhao, L.; Yuan, L.H.; Yu, H.L.; Wang, H.; Gong, X.Y.; Wei, F.; Xiao, R. Elaidic acid induces cell apoptosis through induction of ROS accumulation and endoplasmic reticulum stress in SH-SY5Y cells. Mol. Med. Rep. 2017, 16, 9337–9346. [Google Scholar] [CrossRef]
- Miller, A.; McGrath, E.; Stanton, C.; Devery, R. Vaccenic acid (t11-18:1) is converted to c9,t11-CLA in MCF-7 and SW480 cancer cells. Lipids 2003, 38, 623–632. [Google Scholar] [CrossRef]
- Lim, J.N.; Oh, J.J.; Wang, T.; Lee, J.S.; Kim, S.H.; Kim, Y.J.; Lee, H.G. Trans-11 18:1 vaccenic acid (TVA) has a direct anti-carcinogenic effect on MCF-7 human mammary adenocarcinoma cells. Nutrients 2014, 6, 627–636. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Wang, Y.; Fan, X.; Wu, H.; Han, J.; Yang, M.; Lu, L.; Nie, G. Trans-vaccenic acid inhibits proliferation and induces apoptosis of human nasopharyngeal carcinoma cells via a mitochondrial-mediated apoptosis pathway. Lipids Health Dis. 2019, 18, 46. [Google Scholar] [CrossRef] [PubMed]
- Bocca, C.; Bozzo, F.; Cannito, S.; Colombatto, S.; Miglietta, A. CLA reduces breast cancer cell growth and invasion through ERα and PI3K/Akt pathways. Chem. Biol. Interact. 2010, 183, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Fite, A.; Goua, M.; Wahle, K.W.J.; Schofield, A.C.; Hutcheon, A.W.; Heys, S.D. Potentiation of the anti-tumour effect of docetaxel by conjugated linoleic acids (CLAs) in breast cancer cells in vitro. Prostaglandins Leukot. Essent. Fat. Acids 2007, 77, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.; Park, Y.; Storkson, J.M.; Pariza, M.W.; Ntambi, J.M. Inhibition of stearoyl-CoA desaturase activity by the cis-9, trans-11 isomer and the trans-10, cis-12 isomer of conjugated linoleic acid in MDA-MB-231 and MCF-7 human breast cancer cells. Biochem. Biophys. Res. Commun. 2002, 294, 785–790. [Google Scholar] [CrossRef]
- El Roz, A.; Bard, J.M.; Huvelin, J.M.; Nazih, H. The anti-proliferative and pro-apoptotic effects of the trans9,trans11 conjugated linoleic acid isomer on MCF-7 breast cancer cells are associated with LXR activation. Prostaglandins Leukot. Essent. Fat. Acids 2013, 88, 265–272. [Google Scholar] [CrossRef]
- Lee, S.H.; Yamaguchi, K.; Kim, J.S.; Eling, T.E.; Safe, S.; Park, Y.; Baek, S.J. Conjugated linoleic acid stimulates an anti-tumorigenic protein NAG-1 in an isomer specific manner. Carcinogenesis 2006, 27, 972–981. [Google Scholar] [CrossRef]
- Soel, S.M.; Choi, O.S.; Bang, M.H.; Yoon Park, J.H.; Kim, W.K. Influence of conjugated linoleic acid isomers on the metastasis of colon cancer cells in vitro and in vivo. J. Nutr. Biochem. 2007, 18, 650–657. [Google Scholar] [CrossRef]
- Shahzad, M.M.K.; Felder, M.; Ludwig, K.; Van Galder, H.R.; Anderson, M.L.; Kim, J.; Cook, M.E.; Kapur, A.K.; Patankar, M.S. Trans10,cis12 conjugated linoleic acid inhibits proliferation and migration of ovarian cancer cells by inducing ER stress, autophagy, and modulation of Src. PLoS ONE 2018, 13, e0189524. [Google Scholar] [CrossRef]
- Eun, J.K.; Shin, H.K.; Jin, S.C.; Sang, K.L.; Moo, H.W.; Jong, W.K.; Park, J.H.Y. trans-10,cis-12 conjugated linoleic acid inhibits the G1-S cell cycle progression in DU145 human prostate carcinoma cells. J. Med. Food 2006, 9, 293–299. [Google Scholar] [CrossRef]
- Wang, J.; Liu, X.; Zhang, X.; Liu, J.; Ye, S.; Xiao, S.; Chen, H.; Wang, H. Induction of apoptosis by c9, t11-CLA in human endometrial cancer RL 95-2 cells via ERα-mediated pathway. Chem. Phys. Lipids 2013, 175–176, 27–32. [Google Scholar] [CrossRef]
- Amarù, D.L.; Biondo, P.D.; Field, C.J. The Role of Conjugated Linoleic Acid in Breast Cancer Growth and Development. Open Nutraceuticals J. 2010, 3, 30–46. [Google Scholar] [CrossRef]
- Kelley, N.S.; Hubbard, N.E.; Erickson, K.L. Conjugated linoleic acid isomers and cancer. J. Nutr. 2007, 137, 2599–2607. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, A.; Banu, J.; Rahman, M.; Causey, J.; Fernandes, G. Biological effects of conjugated linoleic acids in health and disease. J. Nutr. Biochem. 2006, 17, 789–810. [Google Scholar] [CrossRef] [PubMed]
- Gebauer, S.K.; Chardigny, J.-M.; Jakobsen, M.U.; Lamarche, B.; Lock, A.L.; Proctor, S.D.; Baer, D.J. Effects of Ruminant trans Fatty Acids on Cardiovascular Disease and Cancer: A Comprehensive Review of Epidemiological, Clinical, and Mechanistic Studies. Adv. Nutr. 2011, 2, 332–354. [Google Scholar] [CrossRef] [PubMed]
- Białek, A.; Jelińska, M.; Tokarz, A. Influence of maternal diet enrichment with conjugated linoleic acids on lipoxygenase metabolites of polyunsaturated fatty acids in serum of their offspring with 7,12-dimethylbenz[a]anthracene induced mammary tumors. Prostaglandins Other Lipid Mediat. 2015, 116–117, 10–18. [Google Scholar] [CrossRef]
- Tao, X.M.; Wang, J.C.; Wang, J.B.; Feng, Q.; Gao, S.Y.; Zhang, L.R.; Zhang, Q. Enhanced anticancer activity of gemcitabine coupling with conjugated linoleic acid against human breast cancer in vitro and in vivo. Eur. J. Pharm. Biopharm. 2012, 82, 401–409. [Google Scholar] [CrossRef]
- Du, R.; Zhong, T.; Zhang, W.Q.; Song, P.; Song, W.D.; Zhao, Y.; Wang, C.; Tang, Y.-Q.; Zhang, X.; Zhang, Q. Antitumor effect of iRGD-modified liposomes containing conjugated linoleic acid-paclitaxel (CLA-PTX) on B16-F10 melanoma. Int. J. Nanomed. 2014, 9, 3091–3105. [Google Scholar] [CrossRef]
- Da Silva, M.S.; Julien, P.; Bilodeau, J.F.; Barbier, O.; Rudkowska, I. Trans Fatty Acids Suppress TNF-α-Induced Inflammatory Gene Expression in Endothelial (HUVEC) and Hepatocellular Carcinoma (HepG2) Cells. Lipids 2017, 52, 315–325. [Google Scholar] [CrossRef]
- Hirata, Y.; Takahashi, M.; Kudoh, Y.; Kano, K.; Kawana, H.; Makide, K.; Shinoda, Y.; Yabuki, Y.; Fukunaga, K.; Aoki, J.; et al. Trans-Fatty acids promote proinflammatory signaling and cell death by stimulating the apoptosis signal-regulating kinase 1 (ASK1)-p38 pathway. J. Biol. Chem. 2017, 292, 8174–8185. [Google Scholar] [CrossRef]
- Monguchi, T.; Hara, T.; Hasokawa, M.; Nakajima, H.; Mori, K.; Toh, R.; Irino, Y.; Ishida, T.; Hirata, K.-I.; Shinohara, M. Excessive intake of trans fatty acid accelerates atherosclerosis through promoting inflammation and oxidative stress in a mouse model of hyperlipidemia. J. Cardiol. 2017, 70, 121–127. [Google Scholar] [CrossRef]
- Anjom-Shoae, J.; Sadeghi, O.; Larijani, B.; Esmaillzadeh, A. Dietary intake and serum levels of trans fatty acids and risk of breast cancer: A systematic review and dose-response meta-analysis of prospective studies. Clin. Nutr. 2020, 39, 755–764. [Google Scholar] [CrossRef]
- Vinikoor, L.C.; Millikan, R.C.; Satia, J.A.; Schroeder, J.C.; Martin, C.F.; Ibrahim, J.G.; Sandler, R.S. Trans-Fatty Acid Consumption and its Association with Distal Colorectal Cancer in the North Carolina Colon Cancer Study II. Cancer Causes Control 2010, 21, 171–180. [Google Scholar] [CrossRef]
- Hu, J.; La Vecchia, C.; Negri, E.; Mery, L. Nutrients and Risk of Colon Cancer. Cancers 2010, 2, 51–67. [Google Scholar] [CrossRef]
- Slattery, M.L.; Benson, J.; Ma, K.-N.; Schaffer, D.; Potter, J.D. Trans-Fatty Acids and Colon Cancer. Nutr. Cancer 2001, 39, 170–175. [Google Scholar] [CrossRef]
- Qiu, W.; Lu, H.; Qi, Y.; Wang, X. Dietary fat intake and ovarian cancer risk: A meta-analysis of epidemiological studies. Oncotarget 2016, 7, 37390–37406. [Google Scholar] [CrossRef]
- Chajès, V.; Assi, N.; Biessy, C.; Ferrari, P.; Rinaldi, S.; Slimani, N.; Lenoir, G.M.; Baglietto, L.; His, M.; Boutron-Ruault, M.C.; et al. A prospective evaluation of plasma phospholipid fatty acids and breast cancer risk in the EPIC study. Ann. Oncol. 2017, 28, 2836–2842. [Google Scholar] [CrossRef]
- Matejcic, M.; Lesueur, F.; Biessy, C.; Renault, A.L.; Mebirouk, N.; Yammine, S.; Keski-Rahkonen, P.; Li, K.; Hémon, B.; Weiderpass, E.; et al. Circulating plasma phospholipid fatty acids and risk of pancreatic cancer in a large European cohort. Int. J. Cancer 2018, 143, 2437–2448. [Google Scholar] [CrossRef]
- Tinsley, I.J.; Schmitz, J.A.; Pierce, D.A. Influence of dietary fatty acids on the incidence of mammary tumors in the C3H mouse. Cancer Res. 1981, 41, 1460–1465. [Google Scholar]
- Evans, L.M.; Toline, E.C.; Desmond, R.; Siegal, G.P.; Hashim, A.I.; Hardy, R.W. Dietary stearate reduces human breast cancer metastasis burden in athymic nude mice. Clin. Exp. Metastasis 2009, 26, 415–424. [Google Scholar] [CrossRef]
- Bennett, A.S. Effect of dietary stearic acid on the genesis of spontaneous mammary adenocarcinomas in strain A/ST mice. Int. J. Cancer 1984, 34, 529–533. [Google Scholar] [CrossRef]
- Rao, C.V.; Hirose, Y.; Indranie, C.; Reddy, B.S. Modulation of experimental colon tumorigenesis by types and amounts of dietary fatty acids. Cancer Res. 2001, 61, 1927–1933. [Google Scholar]
- Yu, M.; Liu, H.; Duan, Y.; Zhang, D.; Li, S.; Wang, F. Four types of fatty acids exert differential impact on pancreatic cancer growth. Cancer Lett. 2015, 360, 187–194. [Google Scholar] [CrossRef]
- Falconer, J.S.; Ross, J.A.; Fearon, K.C.H.; Hawkins, R.A.; O’Riordain, M.G.; Carter, D.C. Effect of eicosapentaenoic acid and other fatty acids on the growth in vitro of human pancreatic cancer cell lines. Br. J. Cancer 1994, 69, 826–832. [Google Scholar] [CrossRef]
- Park, H.S.; Ryu, J.H.; Ha, Y.L.; Park, J.H.Y. Dietary conjugated linoleic acid (CLA) induces apoptosis of colonic mucosa in 1,2-dimethylhydrazine-treated rats: A possible mechanism of the anticarcinogenic effect by CLA. Br. J. Nutr. 2001, 86, 549–555. [Google Scholar] [CrossRef]
- Shiraishi, R.; Iwakiri, R.; Fujise, T.; Kuroki, T.; Kakimoto, T.; Takashima, T.; Sakata, Y.; Tsunada, S.; Nakashima, Y.; Yanagita, T.; et al. Conjugated linoleic acid suppresses colon carcinogenesis in azoxymethane-pretreated rats with long-term feeding of diet containing beef tallow. J. Gastroenterol. 2010, 45, 625–635. [Google Scholar] [CrossRef]
- Moreira, T.G.; Horta, L.S.; Gomes-Santos, A.C.; Oliveira, R.P.; Queiroz, N.M.G.P.; Mangani, D.; Daniel, B.; Vieira, A.T.; Liu, S.; Rodrigues, A.M.; et al. CLA-supplemented diet accelerates experimental colorectal cancer by inducing TGF-β-producing macrophages and T cells. Mucosal Immunol. 2019, 12, 188–199. [Google Scholar] [CrossRef]
- Cesano, A.; Visonneau, S.; Scimeca, J.A.; Kritchevsky, D.; Santoli, D. Opposite effects of linoleic acid and conjugated linoleic acid on human prostatic cancer in SCID mice. Anticancer Res. 1998, 18, 1429–1434. [Google Scholar]
- Cohen, L.A.; Zhao, Z.; Pittman, B.; Scimeca, J. Effect of soy protein isolate and conjugated linoleic acid on the growth of dunning R-3327-AT-1 rat prostate tumors. Prostate 2003, 54, 169–180. [Google Scholar] [CrossRef]
- Glatz, J.F.C.; Luiken, J. From fat to FAT (CD36/SR-B2): Understanding the regulation of cellular fatty acid uptake. Biochimie 2017, 136, 21–26. [Google Scholar] [CrossRef]
- Zhao, J.; Zhi, Z.; Wang, C.; Xing, H.; Song, G.; Yu, X.; Zhu, Y.; Wang, X.; Zhang, X.; Di, Y. Exogenous lipids promote the growth of breast cancer cells via CD36. Oncol. Rep. 2017, 38, 2105–2115. [Google Scholar] [CrossRef]
- Liang, Y.; Han, H.; Liu, L.; Duan, Y.; Yang, X.; Ma, C.; Zhu, Y.; Han, J.; Li, X.; Chen, Y. CD36 plays a critical role in proliferation, migration and tamoxifen-inhibited growth of ER-positive breast cancer cells. Oncogenesis 2018, 7, 98. [Google Scholar] [CrossRef]
- Deng, M.; Cai, X.; Long, L.; Xie, L.; Ma, H.; Zhou, Y.; Liu, S.; Zeng, C. CD36 promotes the epithelial-mesenchymal transition and metastasis in cervical cancer by interacting with TGF-β. J. Transl. Med. 2019, 17, 352. [Google Scholar] [CrossRef]
- Miki, Y.; Yashiro, M.; Okuno, T.; Kuroda, K.; Togano, S.; Hirakawa, K.; Ohira, M. Clinico-pathological significance of exosome marker CD63 expression on cancer cells and stromal cells in gastric cancer. PLoS ONE 2018, 13, e0202956. [Google Scholar] [CrossRef]
- Feng, W.W.; Wilkins, O.; Bang, S.; Ung, M.; Li, J.; An, J.; del Genio, C.; Canfield, K.; DiRenzo, J.; Wells, W.; et al. CD36-Mediated Metabolic Rewiring of Breast Cancer Cells Promotes Resistance to HER2-Targeted Therapies. Cell Rep. 2019, 29, 3405–3420.e5. [Google Scholar] [CrossRef]
- Xu, W.H.; Qu, Y.Y.; Wang, J.; Wang, H.K.; Wan, F.N.; Zhang, H.L.; Ye, D.W.; Zhao, J.Y. Elevated CD36 expression correlates with increased visceral adipose tissue and predicts poor prognosis in ccRCC patients. J. Cancer 2019, 10, 4522–4531. [Google Scholar] [CrossRef]
- Jia, S.; Zhou, L.; Shen, T.; Zhou, S.; Ding, G.; Cao, L. Down-expression of CD36 in pancreatic adenocarcinoma and its correlation with clinicopathological features and prognosis. J. Cancer 2018, 9, 578–583. [Google Scholar] [CrossRef]
- Flowers, M.T.; Ntambi, J.M. Role of stearoyl-coenzyme A desaturase in regulating lipid metabolism. Curr. Opin. Lipidol. 2008, 19, 248–256. [Google Scholar] [CrossRef]
- Wang, J.; Yu, L.; Schmidt, R.E.; Su, C.; Huang, X.; Gould, K.; Cao, G. Characterization of HSCD5, a novel human stearoyl-CoA desaturase unique to primates. Biochem. Biophys. Res. Commun. 2005, 332, 735–742. [Google Scholar] [CrossRef]
- Wang, J.; Xu, Y.; Zhu, L.; Zou, Y.; Kong, W.; Dong, B.; Huang, J.; Chen, Y.; Xue, W.; Huang, Y.; et al. High expression of Stearoyl-CoA desaturase 1 Predicts Poor Prognosis in Patients with Clear-Cell Renal Cell Carcinoma. PLoS ONE 2016, 11, e0166231. [Google Scholar] [CrossRef]
- Noto, A.; De Vitis, C.; Pisanu, M.E.; Roscilli, G.; Ricci, G.; Catizone, A.; Sorrentino, G.; Chianese, G.; Taglialatela-Scafati, O.; Trisciuoglio, D.; et al. Stearoyl-CoA-desaturase 1 regulates lung cancer stemness via stabilization and nuclear localization of YAP/TAZ. Oncogene 2017, 36, 4573–4584. [Google Scholar] [CrossRef] [PubMed]
- Ran, H.; Zhu, Y.; Deng, R.; Zhang, Q.; Liu, X.; Feng, M.; Zhong, J.; Lin, S.; Tong, X.; Su, Q. Stearoyl-CoA desaturase-1 promotes colorectal cancer metastasis in response to glucose by suppressing PTEN. J. Exp. Clin. Cancer Res. 2018, 37, 54. [Google Scholar] [CrossRef] [PubMed]
- Tracz-Gaszewska, Z.; Dobrzyn, P. Stearoyl-CoA desaturase 1 as a therapeutic target for the treatment of cancer. Cancers 2019, 11, E948. [Google Scholar] [CrossRef]
- Liu, Q.; Luo, Q.; Halim, A.; Song, G. Targeting lipid metabolism of cancer cells: A promising therapeutic strategy for cancer. Cancer Lett. 2017, 401, 39–45. [Google Scholar] [CrossRef]
- Pizato, N.; Kiffer, L.F.M.V.; Luzete, B.C.; Assumpção, J.A.F.; Correa, L.H.; De Melo, H.A.B.; De Sant’Ana, L.P.; Ito, M.K.; Magalhães, K.G. Omega 3-DHA and delta-tocotrienol modulate lipid droplet biogenesis and lipophagy in breast cancer cells: The impact in cancer aggressiveness. Nutrients 2019, 11, 1199. [Google Scholar] [CrossRef]
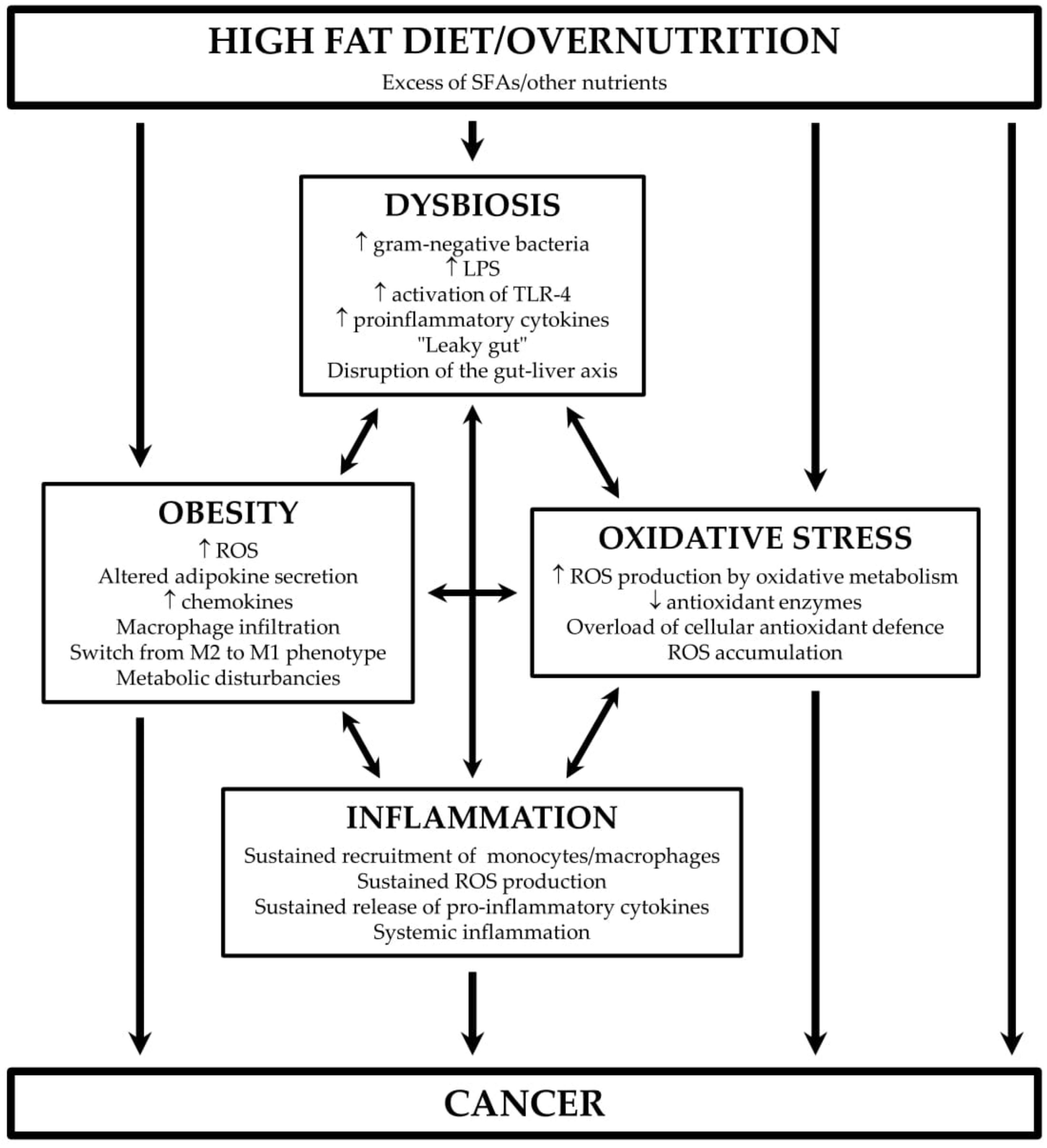
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bojková, B.; Winklewski, P.J.; Wszedybyl-Winklewska, M. Dietary Fat and Cancer—Which Is Good, Which Is Bad, and the Body of Evidence. Int. J. Mol. Sci. 2020, 21, 4114. https://doi.org/10.3390/ijms21114114
Bojková B, Winklewski PJ, Wszedybyl-Winklewska M. Dietary Fat and Cancer—Which Is Good, Which Is Bad, and the Body of Evidence. International Journal of Molecular Sciences. 2020; 21(11):4114. https://doi.org/10.3390/ijms21114114
Chicago/Turabian StyleBojková, Bianka, Pawel J. Winklewski, and Magdalena Wszedybyl-Winklewska. 2020. "Dietary Fat and Cancer—Which Is Good, Which Is Bad, and the Body of Evidence" International Journal of Molecular Sciences 21, no. 11: 4114. https://doi.org/10.3390/ijms21114114
APA StyleBojková, B., Winklewski, P. J., & Wszedybyl-Winklewska, M. (2020). Dietary Fat and Cancer—Which Is Good, Which Is Bad, and the Body of Evidence. International Journal of Molecular Sciences, 21(11), 4114. https://doi.org/10.3390/ijms21114114





