Maternal Obesity and the Uterine Immune Cell Landscape: The Shaping Role of Inflammation
Abstract
1. Inflammation and Pregnancy
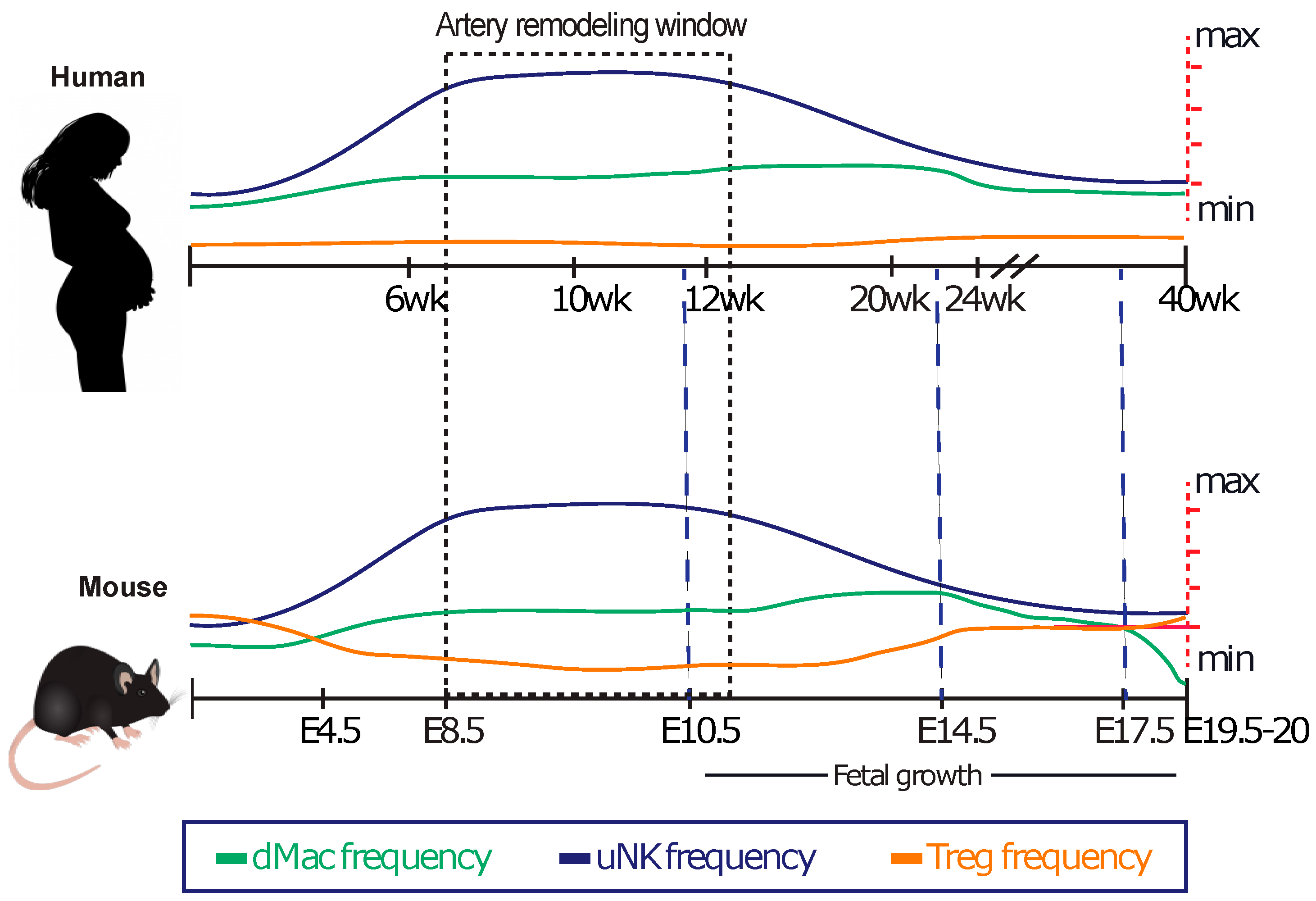
The Uterine Immune Cell Landscape
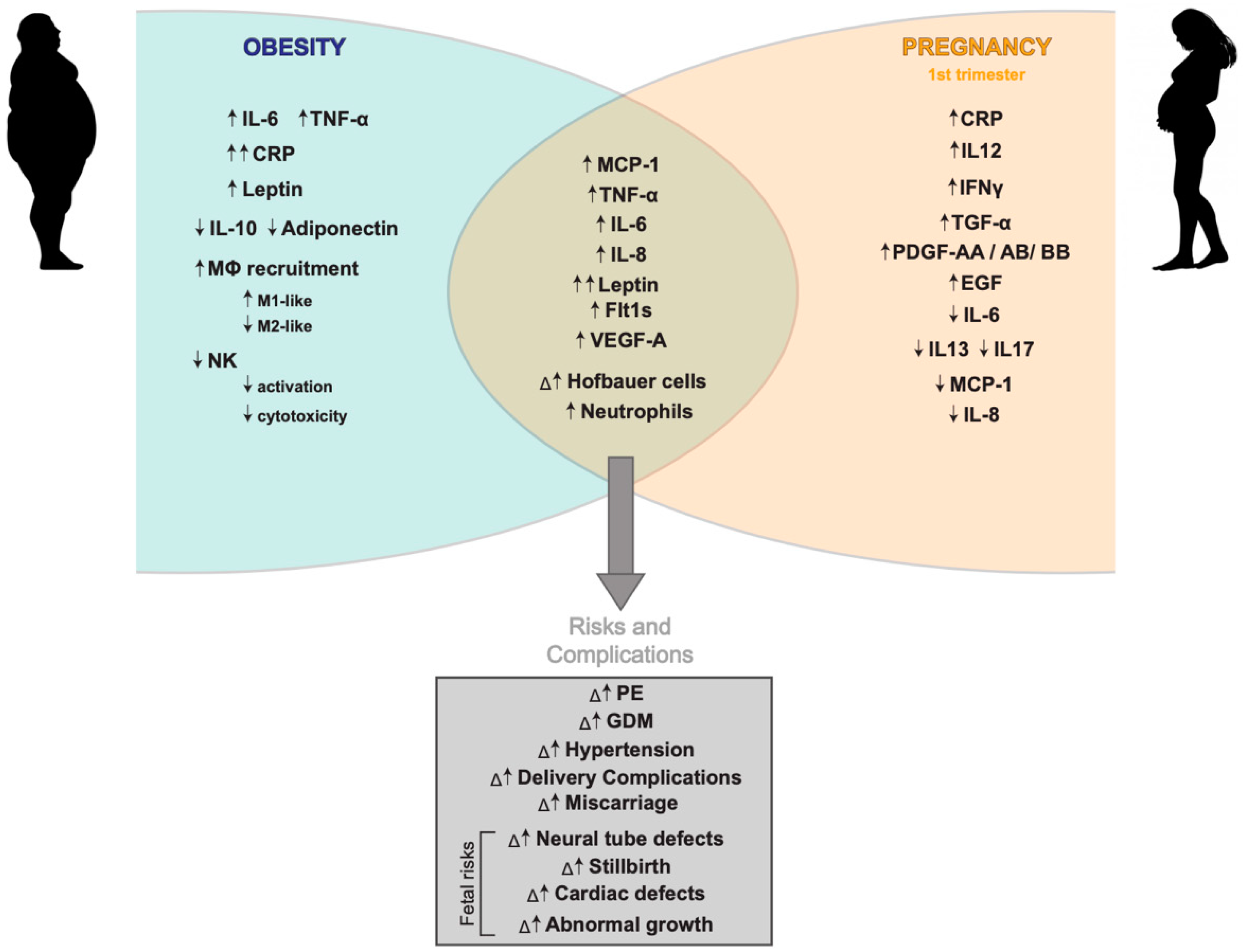
2. Obesity-Driven Inflammation
3. Maternal Obesity
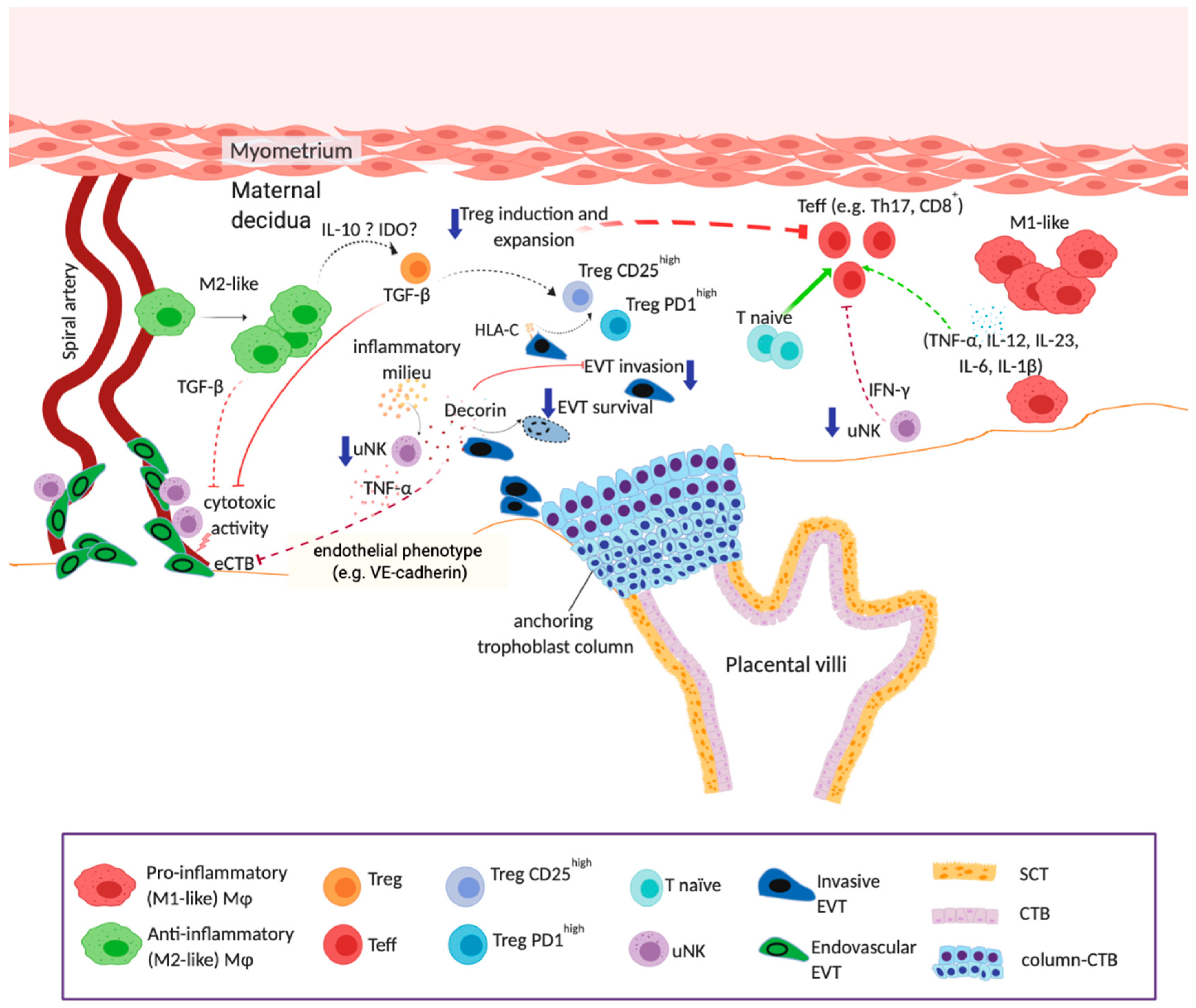
3.1. Impact of Maternal Obesity on Placentation
3.2. Obesity-Driven Uterine Immune Cell Dysfunction
4. Proposed Model of Maternal Obesity in Early Pregnancy
5. Concluding Remarks
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| PRL | prolactin |
| IGFBP | insulin-like growth factor binding protein |
| VEGF | vascular endothelial growth factor |
| TGF | transforming growth factor |
| IL | interleukin |
| uNK | uterine natural killer cell |
| Mφ | macrophage(s) |
| DC | dendritic cell |
| Treg | regulatory T cell |
| ILC | innate lymphoid cell |
| EVT | extravillous trophoblast |
| CXCL | c-x-c motif chemokine |
| IFN | interferon |
| TNF | tumor necrosis factor |
| PlGF | placenta growth factor |
| MMP | matrix metalloproteinase |
| M-CSF | macrophage colony stimulating factor |
| Ang | angiopoietin |
| CD | cluster of differentiation |
| Th | T helper type |
| HLA | human leukocyte antigen |
| HB-EGF | heparin binding-epidermal growth factor |
| NK | natural killer cell |
| GATA3 | GATA-binding protein 3 |
| ROR | retinoic acid receptor-related orphan receptor |
| GM-CSF | granulocyte-macrophage colony stimulating factor |
| dNKp | decidual proliferating natural killer cell |
| dNK | decidual natural killer cell |
| NKR | natural killer cell receptor |
| KIR | killer-cell immunoglobulin-like receptor |
| FFA | free fatty acids |
| MCP-1 | monocyte chemoattractant protein-1 |
| BMI | body mass index |
| AT1 | angiotensin 2 type 1 |
| NOD | non-obese diabetic |
| FGR | fetal-growth restriction |
| i.p. | intraperitoneally |
| LPS | lipopolysaccharide |
| TLR | toll-like receptor |
| Rh | recombinant human |
| RAG | recombinant activating gene |
| MLAp | mesometrial lymphoid aggregate of pregnancy |
| IRF | interferon regulatory factor |
| mr | mouse recombinant |
| Lif | leukemia inhibitory factor gene |
| MHC | major histocompatibility complex |
| HIF | hypoxia-inducible factor |
| HFD | high-fat diet |
| LDL | low density lipoprotein |
| DIO | diet-induced obesity |
| Csf | colony stimulating factor |
| Csfr | colony stimulating factor receptor |
| NCR | natural cytotoxicity triggering receptor |
| UGR | intrauterine growth restriction |
| Teff | T effector cell |
| T naïve | naïve T cell |
| SCT | syncytiotrophoblast |
| CTB | villous cytotrophoblast |
| column-CTB | anchoring column cytotrophoblast |
References
- Mor, G.; Cardenas, I.; Abrahams, V.; Guller, S. Inflammation and pregnancy: The role of the immune system at the implantation site. Ann. N. Y. Acad. Sci. 2011, 1221, 80–87. [Google Scholar] [CrossRef]
- Robson, A.; Harris, L.K.; Innes, B.A.; Lash, G.E.; Aljunaidy, M.M.; Aplin, J.D.; Baker, P.N.; Robson, S.C.; Bulmer, J.N. Uterine natural killer cells initiate spiral artery remodeling in human pregnancy. FASEB J. 2012, 26, 4876–4885. [Google Scholar] [CrossRef]
- Romero, R.; Espinoza, J.; Gonçalves, L.F.; Kusanovic, J.P.; Friel, L.A.; Nien, J.K. Inflammation in preterm and term labour and delivery. Semin. Fetal Neonatal Med. 2006, 11, 317–326. [Google Scholar] [CrossRef]
- Kalagiri, R.R.; Carder, T.; Choudhury, S.; Vora, N.; Ballard, A.R.; Govande, V.; Drever, N.; Beeram, M.R.; Uddin, M.N. Inflammation in Complicated Pregnancy and Its Outcome. Am. J. Perinatol. 2016, 33, 1337–1356. [Google Scholar] [CrossRef]
- Sacks, G.P.; Studena, K.; Sargent, I.L.; Redman, C.W.G. Normal pregnancy and preeclampsia both produce inflammatory changes in peripheral blood leukocytes akin to those of sepsis. Am. J. Obstet. Gynecol. 1998, 179, 80–86. [Google Scholar] [CrossRef]
- Austgulen, R.; Lien, E.; Liabakk, N.B.; Jacobsen, G.; Arntzen, K.J. Increased levels of cytokines and cytokine activity modifiers in normal pregnancy. Eur. J. Obstet. Gynecol. Reprod. Biol. 1994, 57, 149–155. [Google Scholar] [CrossRef]
- Sacks, G.P.; Seyani, L.; Lavery, S.; Trew, G. Maternal C-reactive protein levels are raised at 4 weeks gestation. Hum. Reprod. 2004, 19, 1025–1030. [Google Scholar] [CrossRef]
- Sacks, G.P.; Redman, C.W.G.; Sargent, I.L. Monocytes are primed to produce the Th1 type cytokine IL-12 in normal human pregnancy: An intracellular flow cytometric analysis of peripheral blood mononuclear cells. Clin. Exp. Immunol. 2003, 131, 490–497. [Google Scholar] [CrossRef]
- Borzychowski, A.M.; Croy, B.A.; Chan, W.L.; Redman, C.W.G.; Sargent, I.L. Changes in systemic type 1 and type 2 immunity in normal pregnancy and pre-eclampsia may be mediated by natural killer cells. Eur. J. Immunol. 2005, 35, 3054–3063. [Google Scholar] [CrossRef]
- Aplin, J.D.; Charlton, A.K.; Ayad, S. An immunohistochemical study of human endometrial extracellular matrix during the menstrual cycle and first trimester of pregnancy. Cell Tissue Res. 1988, 253, 231–240. [Google Scholar] [CrossRef]
- Gellersen, B.; Brosens, J.J. Cyclic decidualization of the human endometrium in reproductive health and failure. Endocr. Rev. 2014, 35, 851–905. [Google Scholar] [PubMed]
- Kitaya, K.; Yasuda, J.; Yagi, I.; Tada, Y.; Fushiki, S.; Honjo, H. IL-15 expression at human endometrium and decidua. Biol. Reprod. 2000, 63, 683–687. [Google Scholar] [CrossRef] [PubMed]
- Keskin, D.B.; Allan, D.S.J.; Rybalov, B.; Andzelm, M.M.; Stern, J.N.H.; Kopcow, H.D.; Koopman, L.A.; Strominger, J.L. TGFβ promotes conversion of CD16+ peripheral blood NK cells into CD16- NK cells with similarities to decidual NK cells. Proc. Natl. Acad. Sci. USA 2007, 104, 3378–3383. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Godbole, G.; Modi, D. Decidual control of trophoblast invasion. Am. J. Reprod. Immunol. 2016, 75, 341–350. [Google Scholar] [CrossRef]
- Garrido-Gomez, T.; Dominguez, F.; Quiñonero, A.; Diaz-Gimeno, P.; Kapidzic, M.; Gormley, M.; Ona, K.; Padilla-Iserte, P.; McMaster, M.; Genbacev, O.; et al. Defective decidualization during and after severe preeclampsia reveals a possible maternal contribution to the etiology. Proc. Natl. Acad. Sci. USA 2017, 114, E8468–E8477. [Google Scholar] [CrossRef]
- Bulmer, J.N.; Johnson, P.M. Macrophage populations in the human placenta and amniochorion. Clin. Exp. Immunol. 1984, 57, 393–403. [Google Scholar]
- Bulmer, J.N.; Morrison, L.; Longfellow, M.; Ritson, A.; Pace, D. Granulated lymphocytes in human endometrium: Histochemical and immunohistochemical studies. Hum. Reprod. 1991, 6, 791–798. [Google Scholar] [CrossRef]
- Kammerer, U.; Schoppet, M.; McLellan, A.D.; Kapp, M.; Huppertz, H.I.; Kampgen, E.; Dietl, J. Human decidua contains potent immunostimulatory CD83+ dendritic cells. Am. J. Pathol. 2000, 157, 159–169. [Google Scholar] [CrossRef]
- Amsalem, H.; Kwan, M.; Hazan, A.; Zhang, J.; Jones, R.L.; Whittle, W.; Kingdom, J.C.P.; Croy, B.A.; Lye, S.J.; Dunk, C.E. Identification of a novel neutrophil population: Proangiogenic granulocytes in second-trimester human decidua. J. Immunol. 2014, 193, 3070–3079. [Google Scholar] [CrossRef]
- Vacca, P.; Montaldo, E.; Croxatto, D.; Loiacono, F.; Canegallo, F.; Venturini, P.L.; Moretta, L.; Mingari, M.C. Identification of diverse innate lymphoid cells in human decidua. Mucosal Immunol. 2015, 8, 254–264. [Google Scholar] [CrossRef]
- Carlino, C.; Stabile, H.; Morrone, S.; Bulla, R.; Soriani, A.; Agostinis, C.; Bossi, F.; Mocci, C.; Sarazani, F.; Tedesco, F.; et al. Recruitment of circulating NK cells through decidual tissues: A possible mechanism controlling NK cell accumulation in the uterus during early pregnancy. Blood 2008, 111, 3108–3115. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Zhu, X.-Y.; Du, M.-R.; Li, D.-J. Human trophoblasts recruited T lymphocytes and monocytes into decidua by secretion of chemokine CXCL16 and interaction with CXCR6 in the first-trimester pregnancy. J. Immunol. 2008, 180, 2367–2375. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Lopez, N.; Guilbert, L.J.; Olson, D.M. Invasion of the leukocytes into the fetal-maternal interface during pregnancy. J. Leukoc. Biol. 2010, 88, 625–633. [Google Scholar] [CrossRef] [PubMed]
- Croy, B.A.; Chen, Z.; Hofmann, A.P.; Lord, E.M.; Sedlacek, A.L.; Gerber, S.A. Imaging of vascular development in early mouse decidua and its association with leukocytes and trophoblasts1. Biol. Reprod. 2012, 87, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Lopez, G.E.; Vazquez, J.; Sun, Y.; Chavarria, M.; Lindner, P.N.; Fredrickson, S.; Karst, N.; Stanic, A.K. Decidual-Placental immune landscape during syngeneic murine pregnancy. Front. Immunol. 2018, 9, 2087. [Google Scholar] [CrossRef] [PubMed]
- Erlebacher, A. Leukocyte Population Dynamics and Functions at the Maternal–Fetal Interface. In The Guide to Investigation of Mouse Pregnancy; Elsevier: Amsterdam, The Netherlands, 2014. [Google Scholar]
- Hanna, J.; Goldman-Wohl, D.; Hamani, Y.; Avraham, I.; Greenfield, C.; Natanson-Yaron, S.; Prus, D.; Cohen-Daniel, L.; Arnon, T.I.; Manaster, I.; et al. Decidual NK cells regulate key developmental processes at the human fetal-maternal interface. Nat. Med. 2006, 12, 1065–1074. [Google Scholar] [CrossRef]
- Lash, G.E.; Schiessl, B.; Kirkley, M.; Innes, B.A.; Cooper, A.; Searle, R.F.; Robson, S.C.; Bulmer, J.N. Expression of angiogenic growth factors by uterine natural killer cells during early pregnancy. J. Leukoc. Biol. 2006, 80, 572–580. [Google Scholar] [CrossRef]
- Helige, C.; Ahammer, H.; Moser, G.; Hammer, A.; Dohr, G.; Huppertz, B.; Sedlmayr, P. Distribution of decidual natural killer cells and macrophages in the neighbourhood of the trophoblast invasion front: A quantitative evaluation. Hum. Reprod. 2014, 29, 8–17. [Google Scholar] [CrossRef]
- Smith, S.D.; Dunk, C.E.; Aplin, J.D.; Harris, L.K.; Jones, R.L. Evidence for immune cell involvement in decidual spiral arteriole remodeling in early human pregnancy. Am. J. Pathol. 2009, 174, 1959–1971. [Google Scholar] [CrossRef]
- Hu, Y.; Dutz, J.P.; MacCalman, C.D.; Yong, P.; Tan, R.; von Dadelszen, P. Decidual NK Cells Alter In Vitro First Trimester Extravillous Cytotrophoblast Migration: A Role for IFN-γ. J. Immunol. 2006, 177, 8522–8530. [Google Scholar] [CrossRef]
- Fraser, R.; Whitley, G.S.; Johnstone, A.P.; Host, A.J.; Sebire, N.J.; Thilaganathan, B.; Cartwright, J.E. Impaired decidual natural killer cell regulation of vascular remodelling in early human pregnancies with high uterine artery resistance. J. Pathol. 2012, 228, 322–332. [Google Scholar] [CrossRef] [PubMed]
- Naruse, K.; Lash, G.E.; Innes, B.A.; Otun, H.A.; Searle, R.F.; Robson, S.C.; Bulmer, J.N. Localization of matrix metalloproteinase (MMP)-2, MMP-9 and tissue inhibitors for MMPs (TIMPs) in uterine natural killer cells in early human pregnancy. Hum. Reprod. 2009, 24, 553–561. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, A.P.; Gerber, S.A.; Croy, B.A. Uterine natural killer cells pace early development of mouse decidua basalis. Mol. Hum. Reprod. 2014, 20, 66–76. [Google Scholar] [CrossRef] [PubMed]
- Ashkar, A.A.; Di Santo, J.P.; Croy, B.A. Interferon γ contributes to initiation of uterine vascular modification, decidual integrity, and uterine natural killer cell maturation during normal murine pregnancy. J. Exp. Med. 2000, 192, 259–270. [Google Scholar] [CrossRef]
- LESSIN, D.L.; HUNT, J.S.; KING, C.R.; WOOD, G.W. Antigen expression by cells near the maternal-fetal interface. Am. J. Reprod. Immunol. Microbiol. 1988, 16, 1–7. [Google Scholar] [CrossRef]
- Mosser, D.M.; Edwards, J.P. Exploring the full spectrum of macrophage activation. Nat. Rev. Immunol. 2008, 8, 958–969. [Google Scholar] [CrossRef]
- Macrophage Polarization, Activation and Plasticity: More than Black and White|PromoCell. Available online: https://www.promocell.com/cells-in-action/macrophage-plasticity-black-white/ (accessed on 1 May 2020).
- Mills, C.D.; Kincaid, K.; Alt, J.M.; Heilman, M.J.; Hill, A.M. M-1/M-2 Macrophages and the Th1/Th2 Paradigm. J. Immunol. 2000, 164, 6166–6173. [Google Scholar] [CrossRef]
- Martinez, F.O.; Gordon, S. The M1 and M2 paradigm of macrophage activation: Time for reassessment. F1000Prime Rep. 2014. [Google Scholar] [CrossRef]
- Gustafsson, C.; Mjösberg, J.; Matussek, A.; Geffers, R.; Matthiesen, L.; Berg, G.; Sharma, S.; Buer, J.; Ernerudh, J. Gene expression profiling of human decidual macrophages: Evidence for immunosuppressive phenotype. PLoS ONE 2008, 3, 1–9. [Google Scholar] [CrossRef]
- Svensson-Arvelund, J.; Mehta, R.B.; Lindau, R.; Mirrasekhian, E.; Rodriguez-Martinez, H.; Berg, G.; Lash, G.E.; Jenmalm, M.C.; Ernerudh, J. The human fetal placenta promotes tolerance against the semiallogeneic fetus by inducing regulatory T cells and homeostatic M2 macrophages. J. Immunol. 2015, 194, 1534–1544. [Google Scholar] [CrossRef]
- Lash, G.E.; Pitman, H.; Morgan, H.L.; Innes, B.A.; Agwu, C.N.; Bulmer, J.N. Decidual macrophages: Key regulators of vascular remodeling in human pregnancy. J. Leukoc. Biol. 2016, 100, 315–325. [Google Scholar] [CrossRef] [PubMed]
- Piacentini, M.; Autuori, F. Immunohistochemical localization of tissue transglutaminase and Bcl-2 in rat uterine tissues during embryo implantation and post-partum involution. Differentiation 1994, 57, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Abrahams, V.M.; Kim, Y.M.; Straszewski, S.L.; Romero, R.; Mor, G. Macrophages and apoptotic cell clearance during pregnancy. Am. J. Reprod. Immunol. 2004, 51, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Roth, I.; Fisher, S.J. IL-10 is an autocrine inhibitor of human placental cytotrophoblast MMP- 9 production and invasion. Dev. Biol. 1999, 205, 194–204. [Google Scholar] [CrossRef]
- Bauer, S.; Pollheimer, J.; Hartmann, J.; Husslein, P.; Aplin, J.D.; Knöfler, M. Tumor necrosis factor-α inhibits trophoblast migration through elevation of plasminogen activator inhibitor-1 in first-trimester villous explant cultures. J. Clin. Endocrinol. Metab. 2004, 89, 812–822. [Google Scholar] [CrossRef]
- Jovanović, M.; Stefanoska, I.; Radojčić, L.; Vićovac, L. Interleukin-8 (CXCL8) stimulates trophoblast cell migration and invasion by increasing levels of matrix metalloproteinase (MMP)2 and MMP9 and integrins α5 and β1. Reproduction 2010. [Google Scholar] [CrossRef]
- Houser, B.L.; Tilburgs, T.; Hill, J.; Nicotra, M.L.; Strominger, J.L. Two unique human decidual macrophage populations. J. Immunol. 2011, 186, 2633–2642. [Google Scholar] [CrossRef]
- Geissmann, F.; Manz, M.G.; Jung, S.; Sieweke, M.H.; Merad, M.; Ley, K. Development of monocytes, macrophages, and dendritic cells. Science 2010, 327, 656–661. [Google Scholar] [CrossRef]
- Mellman, I. Dendritic cells: Master regulators of the immune response. Cancer Immunol. Res. 2013. [Google Scholar] [CrossRef]
- Gardner, L. Dendritic cells in the human decidua. Biol. Reprod. 2003, 69, 1438–1446. [Google Scholar] [CrossRef]
- Kämmerer, U.; Eggert, A.O.; Kapp, M.; McLellan, A.D.; Geijtenbeek, T.B.H.H.; Dietl, J.; Van Kooyk, Y.; Kämpgen, E.; Eggert, A.O.; McLellan, A.D.; et al. Unique appearance of proliferating antigen-presenting cells expressing DC-SIGN (CD209) in the decidua of early human pregnancy. Am. J. Pathol. 2003, 162, 887–896. [Google Scholar] [CrossRef]
- Croxatto, D.; Vacca, P.; Canegallo, F.; Conte, R.; Venturini, P.L.; Moretta, L.; Mingari, M.C. Stromal cells from human decidua exert a strong inhibitory effect on NK cell function and dendritic cell differentiation. PLoS ONE 2014, 9, e89006. [Google Scholar] [CrossRef] [PubMed]
- Miyazaki, S.; Tsuda, H.; Sakai, M.; Hori, S.; Sasaki, Y.; Futatani, T.; Miyawaki, T.; Saito, S. Predominance of Th2-promoting dendritic cells in early human pregnancy decidua. J. Leukoc. Biol. 2003, 74, 514–522. [Google Scholar] [CrossRef] [PubMed]
- Plaks, V.; Birnberg, T.; Berkutzki, T.; Sela, S.; BenYashar, A.; Kalchenko, V.; Mor, G.; Keshet, E.; Dekel, N.; Neeman, M.; et al. Uterine DCs are crucial for decidua formation during embryo implantation in mice. J. Clin. Investig. 2008, 118, 3954–3965. [Google Scholar] [CrossRef] [PubMed]
- González, I.T.; Barrientos, G.; Freitag, N.; Otto, T.; Thijssen, V.L.J.L.; Moschansky, P.; von Kwiatkowski, P.; Klapp, B.F.; Winterhager, E.; Bauersachs, S.; et al. Uterine NK cells are critical in shaping DC immunogenic functions compatible with pregnancy progression. PLoS ONE 2012, 7, e46755. [Google Scholar] [CrossRef] [PubMed]
- Blois, S.M.; Barrientos, G.; Garcia, M.G.; Orsal, A.S.; Tometten, M.; Cordo-Russo, R.I.; Klapp, B.F.; Santoni, A.; Fernández, N.; Terness, P.; et al. Interaction between dendritic cells and natural killer cells during pregnancy in mice. J. Mol. Med. 2008, 86, 837. [Google Scholar] [CrossRef]
- Laškarin, G.; Redžović, A.; Rubeša, Ž.; Mantovani, A.; Allavena, P.; Haller, H.; Vlastelić, I.; Rukavina, D. Decidual natural killer cell tuning by autologous dendritic cells. Am. J. Reprod. Immunol. 2008, 59, 433–445. [Google Scholar] [CrossRef]
- Vacca, P.; Cantoni, C.; Vitale, M.; Prato, C.; Canegallo, F.; Fenoglio, D.; Ragni, N.; Moretta, L.; Mingari, M.C. Crosstalk between decidual NK and CD14+ myelomonocytic cells results in induction of Tregs and immunosuppression. Proc. Natl. Acad. Sci. USA 2010, 107, 11918–11923. [Google Scholar] [CrossRef]
- Vassiliadou, N.; Bulmer, J.N. Quantitative analysis of T lymphocyte subsets in pregnant and nonpregnant human endometrium1. Biol. Reprod. 1996, 55, 1017–1022. [Google Scholar] [CrossRef]
- Crespo, Â.C.; van der Zwan, A.; Ramalho-Santos, J.; Strominger, J.L.; Tilburgs, T. Cytotoxic potential of decidual NK cells and CD8+ T cells awakened by infections. J. Reprod. Immunol. 2017, 119, 85–90. [Google Scholar] [CrossRef]
- Tilburgs, T.; Scherjon, S.A.; van der Mast, B.J.; Haasnoot, G.W.; Versteeg-V D Voort-Maarschalk, M.; Roelen, D.L.; van Rood, J.J.; Claas, F.H.J. Fetal-maternal HLA-C mismatch is associated with decidual T cell activation and induction of functional T regulatory cells. J. Reprod. Immunol. 2009, 82, 148–157. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.C.; Li, Y.H.; Piao, H.L.; Hong, X.W.; Zhang, D.; Xu, Y.Y.; Tao, Y.; Wang, Y.; Yuan, M.M.; Li, D.J.; et al. PD-1 and Tim-3 pathways are associated with regulatory CD8+ T-cell function in decidua and maintenance of normal pregnancy. Cell Death Dis. 2015, 6, e1738. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Yamane, H.; Paul, W.E. Differentiation of effector CD4 T cell populations. Annu. Rev. Immunol. 2010, 28, 445–489. [Google Scholar] [CrossRef] [PubMed]
- Burns, W.R.; Wang, Y.; Tang, P.C.Y.; Ranjbaran, H.; Iakimov, A.; Kim, J.; Cuffy, M.; Bai, Y.; Pober, J.S.; Tellides, G. Recruitment of CXCR3+ and CCR5+ T cells and production of interferon-γ-inducible chemokines in rejecting human arteries. Am. J. Transplant. 2005, 5, 1226–1236. [Google Scholar] [CrossRef] [PubMed]
- Kwak-Kim, J.Y.H.; Chung-Bang, H.S.; Ng, S.C.; Ntrivalas, E.I.; Mangubat, C.P.; Beaman, K.D.; Beer, A.E.; Gilman-Sachs, A. Increased T helper 1 cytokine responses by circulating T cells are present in women with recurrent pregnancy losses and in infertile women with multiple implantation failures after IVF. Hum. Reprod. 2003, 18, 767–773. [Google Scholar] [CrossRef] [PubMed]
- Raghupathy, R.; Makhseed, M.; Azizieh, F.; Hassan, N.; Al-Azemi, M.; Al-Shamali, E. Maternal Th1- and Th2-type reactivity to placental antigens in normal human pregnancy and unexplained recurrent spontaneous abortions. Cell. Immunol. 1999, 196, 122–130. [Google Scholar] [CrossRef]
- Ticconi, C.; Pietropolli, A.; Di Simone, N.; Piccione, E.; Fazleabas, A. Endometrial immune dysfunction in recurrent pregnancy loss. Int. J. Mol. Sci. 2019, 20, 5332. [Google Scholar] [CrossRef]
- Wurtz, O.; Bajénoff, M.; Guerder, S. IL-4 mediated inhibition of IFN-γ production by CD4+ T cells proceeds by several developmentally regulated mechanisms. Int. Immunol. 2004, 16, 501–508. [Google Scholar] [CrossRef]
- Lin, H.; Mosmann, T.R.; Guilbert, L.; Tuntipopipat, S.; Wegmann, T.G. Synthesis of T helper 2-type cytokines at the maternal-fetal interface. J. Immunol. 1993, 151, 4562–4573. [Google Scholar]
- Yamada, H.; Morikawa, M.; Furuta, I.; Kato, E.H.; Shimada, S.; Iwabuchi, K.; Minakami, H. Intravenous immunoglobulin treatment in women with recurrent abortions: Increased cytokine levels and reduced Th1/Th2 lymphocyte ratio in peripheral blood. Am. J. Reprod. Immunol. 2003, 49, 84–89. [Google Scholar] [CrossRef]
- Wegmann, T.G.; Lin, H.; Guilbert, L.; Mosmann, T.R. Bidirectional cytokine interactions in the maternal-fetal relationship: Is successful pregnancy a TH2 phenomenon? Immunol. Today 1993, 14, 353–356. [Google Scholar] [CrossRef]
- Makhseed, M.; Raghupathy, R.; Azizieh, F.; Omu, A.; Al-Shamali, E.; Ashkanani, L. Th1 and Th2 cytokine profiles in recurrent aborters with successful pregnancy and with subsequent abortions. Hum. Reprod. 2001, 16, 2219–2226. [Google Scholar] [CrossRef]
- Yuan, J.; Li, J.; Huang, S.Y.; Sun, X. Characterization of the subsets of human NKT-like cells and the expression of Th1/Th2 cytokines in patients with unexplained recurrent spontaneous abortion. J. Reprod. Immunol. 2015, 110, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Sunder-Plassmann, G.; Derfler, K.; Wagner, L.; Stockenhuber, F.; Endler, M.; Nowotny, C.; Balcke, P. Increased serum activity of interleukin-2 in patients with pre-eclampsia. J. Autoimmun. 1989, 2, 203–205. [Google Scholar] [CrossRef]
- Arriaga-Pizano, L.; Jimenez-Zamudio, L.; Vadillo-Ortega, F.; Martinez-Flores, A.; Herrerias-Canedo, T.; Hernandez-Guerrero, C. The predominant Th1 cytokine profile in maternal plasma of preeclamptic women is not reflected in the choriodecidual and fetal compartments. J. Soc. Gynecol. Investig. 2005. [Google Scholar] [CrossRef] [PubMed]
- Peck, A.; Mellins, E.D. Plasticity of T-cell phenotype and function: The T helper type 17 example. Immunology 2010, 129, 147–153. [Google Scholar] [CrossRef]
- Liang, S.C.; Tan, X.Y.; Luxenberg, D.P.; Karim, R.; Dunussi-Joannopoulos, K.; Collins, M.; Fouser, L.A. Interleukin (IL)-22 and IL-17 are coexpressed by Th17 cells and cooperatively enhance expression of antimicrobial peptides. J. Exp. Med. 2006, 203, 2271–2279. [Google Scholar] [CrossRef]
- Wang, W.J.; Hao, C.F.; Yi, L.; Yin, G.J.; Bao, S.H.; Qiu, L.H.; Lin, Q. De Increased prevalence of T helper 17 (Th17) cells in peripheral blood and decidua in unexplained recurrent spontaneous abortion patients. J. Reprod. Immunol. 2010, 129, 147–153. [Google Scholar]
- Liu, Y.S.; Wu, L.; Tong, X.H.; Wu, L.M.; He, G.P.; Zhou, G.X.; Luo, L.H.; Luan, H.B. Study on the relationship between Th17 cells and unexplained recurrent spontaneous abortion. Am. J. Reprod. Immunol. 2011, 65, 503–511. [Google Scholar] [CrossRef]
- Eghbal-Fard, S.; Yousefi, M.; Heydarlou, H.; Ahmadi, M.; Taghavi, S.; Movasaghpour, A.; Jadidi-Niaragh, F.; Yousefi, B.; Dolati, S.; Hojjat-Farsangi, M.; et al. The imbalance of Th17/Treg axis involved in the pathogenesis of preeclampsia. J. Cell. Physiol. 2019, 234, 5106–5116. [Google Scholar] [CrossRef]
- Sakaguchi, S.; Yamaguchi, T.; Nomura, T.; Ono, M. Regulatory T cells and immune tolerance. Cell 2008, 133, 775–787. [Google Scholar] [CrossRef] [PubMed]
- Saito, S.; Nakashima, A.; Shima, T.; Ito, M. Th1/Th2/Th17 and regulatory T-Cell paradigm in pregnancy. Am. J. Reprod. Immunol. 2010, 63, 601–610. [Google Scholar] [CrossRef] [PubMed]
- Aluvihare, V.R.; Kallikourdis, M.; Betz, A.G. Regulatory T cells mediate maternal tolerance to the fetus. Nat. Immunol. 2004, 5, 266–271. [Google Scholar] [CrossRef] [PubMed]
- Zenclussen, A.C.; Gerlof, K.; Zenclussen, M.L.; Sollwedel, A.; Bertoja, A.Z.; Ritter, T.; Kotsch, K.; Leber, J.; Volk, H.D. Abnormal T-cell reactivity against paternal antigens in spontaneous abortion: Adoptive transfer of pregnancy-induced CD4+CD25+ T regulatory cells prevents fetal rejection in a murine abortion model. Am. J. Pathol. 2005, 166, 811–822. [Google Scholar] [CrossRef]
- Witko-Sarsat, V.; Rieu, P.; Descamps-Latscha, B.; Lesavre, P.; Halbwachs-Mecarelli, L. Neutrophils: Molecules, functions and pathophysiological aspects. Lab. Investig. 2000, 80, 617–653. [Google Scholar] [CrossRef]
- Croxatto, D.; Micheletti, A.; Montaldo, E.; Orecchia, P.; Loiacono, F.; Canegallo, F.; Calzetti, F.; Fulcheri, E.; Munari, E.; Zamò, A.; et al. Group 3 innate lymphoid cells regulate neutrophil migration and function in human decidua. Mucosal Immunol. 2016, 9, 1372–1383. [Google Scholar] [CrossRef]
- Nadkarni, S.; Smith, J.; Sferruzzi-Perri, A.N.; Ledwozyw, A.; Kishore, M.; Haas, R.; Mauro, C.; Williams, D.J.; Farsky, S.H.P.; Marelli-Berg, F.M.; et al. Neutrophils induce proangiogenic T cells with a regulatory phenotype in pregnancy. Proc. Natl. Acad. Sci. USA 2016. [Google Scholar] [CrossRef]
- Spits, H.; Cupedo, T. Innate lymphoid cells: Emerging insights in development, lineage relationships, and function. Annu. Rev. Immunol. 2012, 30, 647–675. [Google Scholar] [CrossRef]
- Spits, H.; Artis, D.; Colonna, M.; Diefenbach, A.; Di Santo, J.P.; Eberl, G.; Koyasu, S.; Locksley, R.M.; McKenzie, A.N.J.; Mebius, R.E.; et al. Innate lymphoid cells-a proposal for uniform nomenclature. Nat. Rev. Immunol. 2013, 13, 145–149. [Google Scholar] [CrossRef]
- Boulenouar, S.; Doisne, J.M.; Sferruzzi-Perri, A.; Gaynor, L.M.; Kieckbusch, J.; Balmas, E.; Yung, H.W.; Javadzadeh, S.; Volmer, L.; Hawkes, D.A.; et al. The residual innate lymphoid cells in NFIL3-deficient mice support suboptimal maternal adaptations to pregnancy. Front. Immunol. 2016. [Google Scholar] [CrossRef]
- Li, L.; Tu, J.; Jiang, Y.; Zhou, J.; Schust, D.J. Regulatory T cells decrease invariant natural killer T cell-mediated pregnancy loss in mice. Mucosal Immunol. 2017, 10, 613–623. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Romero, R.; Miller, D.; Silva, P.; Panaitescu, B.; Theis, K.R.; Arif, A.; Hassan, S.S.; Gomez-Lopez, N. Innate lymphoid cells at the human maternal-fetal interface in spontaneous preterm labor. Am. J. Reprod. Immunol. 2018, 79, e12820. [Google Scholar] [CrossRef] [PubMed]
- Huhn, O.; Ivarsson, M.A.; Gardner, L.; Hollinshead, M.; Stinchcombe, J.C.; Chen, P.; Shreeve, N.; Chazara, O.; Farrell, L.E.; Theorell, J.; et al. Distinctive phenotypes and functions of innate lymphoid cells in human decidua during early pregnancy. Nat. Commun. 2020, 11, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Choe, S.S.; Huh, J.Y.; Hwang, I.J.; Kim, J.I.J.B.; Kim, J.I.J.B. Adipose tissue remodeling: Its role in energy metabolism and metabolic disorders. Front. Endocrinol. (Lausanne) 2016, 7. [Google Scholar] [CrossRef]
- Luo, L.; Liu, M. Adipose tissue in control of metabolism. J. Endocrinol. 2016, 231, R77–R99. [Google Scholar] [CrossRef]
- Carvalheira, J.B.C.; Qiu, Y.; Chawla, A. Blood spotlight on leukocytes and obesity. Blood 2013, 122, 3263–3267. [Google Scholar] [CrossRef]
- Castoldi, A.; Naffah de Souza, C.; Câmara, N.O.S.; Moraes-Vieira, P.M. The Macrophage Switch in Obesity Development. Front. Immunol. 2015, 6, 637. [Google Scholar] [CrossRef]
- Lee, H.; Lee, I.S.; Choue, R. Obesity, inflammation and diet. Pediatr. Gastroenterol. Hepatol. Nutr. 2013, 16, 143–152. [Google Scholar] [CrossRef]
- Reilly, S.M.; Saltiel, A.R. Adapting to obesity with adipose tissue inflammation. Nat. Rev. Endocrinol. 2017, 13, 633–643. [Google Scholar] [CrossRef]
- Wang, Q.; Wu, H. T Cells in adipose tissue: Critical players in immunometabolism. Front. Immunol. 2018, 9, 2509. [Google Scholar] [CrossRef]
- Patel, P.S.; Buras, E.D.; Balasubramanyam, A. The role of the immune system in obesity and insulin resistance. J. Obes. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Mclaughlin, T.; Ackerman, S.E.; Shen, L.; Engleman, E. Role of innate and adaptive immunity in obesity-associated metabolic disease. J. Clin. Investig. 2017, 127, 5–13. [Google Scholar] [CrossRef] [PubMed]
- Vahratian, A. Prevalence of overweight and obesity among women of childbearing age: Results from the 2002 national survey of family growth. Matern. Child Health J. 2009, 13, 268–273. [Google Scholar] [CrossRef] [PubMed]
- Leddy, M.A.; Power, M.L.; Schulkin, J. The impact of maternal obesity on maternal and fetal health. Rev. Obstet. Gynecol. 2008, 1, 170–178. [Google Scholar]
- Metwally, M.; Saravelos, S.H.; Ledger, W.L.; Li, T.C. Body mass index and risk of miscarriage in women with recurrent miscarriage. Fertil. Steril. 2010, 94, 290–295. [Google Scholar] [CrossRef]
- Metwally, M.; Ong, K.J.; Ledger, W.L.; Li, T.C. Does high body mass index increase the risk of miscarriage after spontaneous and assisted conception? A meta-analysis of the evidence. Fertil. Steril. 2008, 90, 714–726. [Google Scholar] [CrossRef]
- Bhandari, H.; Tan, B.; Quenby, S. Superfertility is more prevalent in obese women with recurrent early pregnancy miscarriage. BJOG Int. J. Obstet. Gynaecol. 2016, 123, 217–222. [Google Scholar] [CrossRef]
- Kim, Y.M.; Chaiworapongsa, T.; Gomez, R.; Bujold, E.; Yoon, B.H.; Rotmensch, S.; Thaler, H.T.; Romero, R. Failure of physiologic transformation of the spiral arteries in the placental bed in preterm premature rupture of membranes. Am. J. Obstet. Gynecol. 2002, 187, 1137–1142. [Google Scholar] [CrossRef]
- Goldenberg, R.L.; Culhane, J.F.; Iams, J.D.; Romero, R. Epidemiology and causes of preterm birth. Lancet 2008, 371, 75–84. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, L.; Zhang, S.; Yu, Q.; Xiong, F.; Huang, K.; Wang, C.Y.; Yang, P. HMGB1, an innate alarmin, plays a critical role in chronic inflammation of adipose tissue in obesity. Mol. Cell. Endocrinol. 2017, 454, 103–111. [Google Scholar] [CrossRef]
- Tangerås, L.H.; Silva, G.B.; Stødle, G.S.; Gierman, L.M.; Skei, B.; Collett, K.; Beversmark, A.L.; Skråstad, R.B.; Thomsen, L.C.V.; Bjørge, L.; et al. Placental inflammation by HMGB1 activation of TLR4 at the syncytium. Placenta 2018, 72–73, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Shao, J.; Zhao, M.; Tong, M.; Wei, J.; Wise, M.R.; Stone, P.; Chamley, L.; Chen, Q. Increased levels of HMGB1 in trophoblastic debris may contribute to preeclampsia. Reproduction 2016, 152, 775–784. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Koga, K.; Osuga, Y.; Hirata, T.; Saito, A.; Yoshino, O.; Hirota, Y.; Harada, M.; Takemura, Y.; Fujii, T.; et al. High mobility group box 1 (HMGB1) levels in the placenta and in serum in preeclampsia. Am. J. Reprod. Immunol. 2011, 66, 143–148. [Google Scholar] [CrossRef]
- Virdis, A.; Colucci, R.; Bernardini, N.; Blandizzi, C.; Taddei, S.; Masi, S. Microvascular endothelial dysfunction in human obesity: Role of TNF- α. J. Clin. Endocrinol. Metab. 2018, 104, 341–348. [Google Scholar] [CrossRef]
- Aggarwal, R.; Jain, A.K.; Mittal, P.; Kohli, M.; Jawanjal, P.; Rath, G. Association of pro- and anti-inflammatory cytokines in preeclampsia. J. Clin. Lab. Anal. 2019, 33, e22834. [Google Scholar] [CrossRef] [PubMed]
- Bing, C. Is interleukin-1β a culprit in macrophage-adipocyte crosstalk in obesity? Adipocyte 2015, 4, 149–152. [Google Scholar] [CrossRef] [PubMed]
- Chandrasekaran, S.; Hunt, H.; Melhorn, S.; Gammill, H.S.; Schur, E.A. Adipokine profiles in preeclampsia. J. Matern. Neonatal Med. 2019. [Google Scholar] [CrossRef]
- Ma, Y.; Ye, Y.; Zhang, J.; Ruan, C.C.; Gao, P.J. Immune imbalance is associated with the development of preeclampsia. Medicine (Baltimore) 2019, 98, e15080. [Google Scholar] [CrossRef]
- Romero, R.; Gotsch, F.; Pineles, B.; Kusanovic, J.P.; Pedro Kusanovic, J. Inflammation in Pregnancy: Its Roles in Reproductive Physiology, Obstetrical Complications, and Fetal Injury. Nutr. Rev. 2007, 65, 194–202. [Google Scholar] [CrossRef]
- Lisonkova, S.; Joseph, K.S. Incidence of preeclampsia: Risk factors and outcomes associated with early-versus late-onset disease. Am. J. Obstet. Gynecol. 2013, 209, e1–e544. [Google Scholar] [CrossRef]
- Robillard, P.Y.; Dekker, G.; Scioscia, M.; Bonsante, F.; Iacobelli, S.; Boukerrou, M.; Hulsey, T.C. Increased BMI has a linear association with late-onset preeclampsia: A population-based study. PLoS ONE 2019, 14, e0223888. [Google Scholar] [CrossRef]
- Assessment of Pregnancy Outcome in Primigravida: Comparison between Booked and Un-Booked Patients—PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/21702258/ (accessed on 21 May 2020).
- Cardoso, E.M.; Reis, C.; Manzanares-Céspedes, M.C. Chronic periodontitis, inflammatory cytokines, and interrelationship with other chronic diseases. Postgrad. Med. 2018, 130, 98–104. [Google Scholar] [CrossRef]
- Seravalle, G.; Grassi, G. Obesity and hypertension. Pharmacol. Res. 2017, 122, 1–7. [Google Scholar] [CrossRef]
- Parker, V.J.; Solano, M.E.M.E.; Arck, P.C.P.C.; Douglas, A.J. Diet-induced obesity may affect the uterine immune environment in early-mid pregnancy, reducing NK-cell activity and potentially compromising uterine vascularization. Int. J. Obes. 2014, 38, 766–774. [Google Scholar] [CrossRef] [PubMed]
- Gohir, W.; Kennedy, K.M.; Wallace, J.G.; Saoi, M.; Britz-Mckibbin, P.; Petrik, J.J.; Surette, M.G.; Sloboda, D.M. High-fat diet intake modulates maternal intestinal adaptations to pregnancy, and results in placental hypoxia and impaired fetal gut development. Reprod. Dev. 2018, 0, 1–23. [Google Scholar]
- Baltayeva, J.; Konwar, C.; Castellana, B.; Mara, D.L.; Christians, J.K.; Beristain, A.G. Obesogenic diet exposure alters uterine natural killer cell biology and impairs vasculature remodeling in mice†. Biol. Reprod. 2019, 102, 63–75. [Google Scholar] [CrossRef] [PubMed]
- Hayes, E.K.; Tessier, D.R.; Percival, M.E.; Holloway, A.C.; Petrik, J.J.; Gruslin, A.; Raha, S. Trophoblast invasion and blood vessel remodeling are altered in a rat model of lifelong maternal obesity. Reprod. Sci. 2014, 21, 648–657. [Google Scholar] [CrossRef] [PubMed]
- Montoudis, A.; Simoneau, L.; Brissette, L.; Forest, J.C.; Savard, R.; Lafond, J. Impact of a cholesterol enriched diet on maternal and fetal plasma lipids and fetal deposition in pregnant rabbits. Life Sci. 1999, 64, 2439–2450. [Google Scholar] [CrossRef]
- Wallace, K.; Richards, S.; Dhillon, P.; Weimer, A.; Edholm, E.; Bengten, E.; Wilson, M.; Martin, J.N.; LaMarca, B. CD4+ T-Helper Cells Stimulated in Response to Placental Ischemia Mediate Hypertension During Pregnancy. Hypertension 2011, 57, 949–955. [Google Scholar] [CrossRef]
- LaMarca, B.; Wallukat, G.; Llinas, M.; Herse, F.; Dechend, R.; Granger, J.P. Autoantibodies to the angiotensin type I receptor in response to placental ischemia and tumor necrosis factor α in pregnant rats. Hypertension 2008, 52, 1168–1172. [Google Scholar] [CrossRef]
- Novotny, S.R.; Wallace, K.; Heath, J.; Moseley, J.; Dhillon, P.; Weimer, A.; Wallukat, G.; Herse, F.; Wenzel, K.; Martin, J.N.; et al. Activating autoantibodies to the angiotensin II type I receptor play an important role in mediating hypertension in response to adoptive transfer of CD4+ T lymphocytes from placental ischemic rats. Am. J. Physiol. Integr. Comp. Physiol. 2012, 302, R1197–R1201. [Google Scholar] [CrossRef] [PubMed]
- Guzik, T.J.; Hoch, N.E.; Brown, K.A.; McCann, L.A.; Rahman, A.; Dikalov, S.; Goronzy, J.; Weyand, C.; Harrison, D.G. Role of the T cell in the genesis of angiotensin II induced hypertension and vascular dysfunction. J. Exp. Med. 2007, 204, 2449–2460. [Google Scholar] [CrossRef] [PubMed]
- Burke, S.D.; Dong, H.; Hazan, A.D.; Croy, B.A. Aberrant endometrial features of pregnancy in diabetic NOD mice. Diabetes 2007, 56, 2919–2926. [Google Scholar] [CrossRef] [PubMed]
- Larocca, L.; Hauk, V.; Calafat, M.; Roca, V.; Fraccaroli, L.; Franchi, A.; Ramhorst, R.; Leirós, C.P. Modulation of macrophage inflammatory profile in pregnant nonobese diabetic (NOD) mice. Mol. Cell. Endocrinol. 2011, 333, 112–118. [Google Scholar] [CrossRef]
- Hu, Y.; Jin, P.; Peng, J.; Zhang, X.; Wong, F.S.; Wen, L. Different immunological responses to early-life antibiotic exposure affecting autoimmune diabetes development in NOD mice. J. Autoimmun. 2016, 72, 47–56. [Google Scholar] [CrossRef]
- Khan, N.A.; Khan, A.; Savelkoul, H.F.J.; Benner, R. Inhibition of diabetes in NOD mice by human pregnancy factor. Hum. Immunol. 2001, 62, 1315–1323. [Google Scholar] [CrossRef]
- Karimi, K.; Solano, M.E.; Ashkar, A.A.; Ho, H.; Steidle, E.M.; Neufeld, K.A.M.V.; Hecher, K.; Bienenstock, J.; Arck, P.C. Regulation of pregnancy maintenance and fetal survival in mice by CD27 low mature NK cells. J. Mol. Med. 2012, 90, 1047–1057. [Google Scholar] [CrossRef]
- Brien, M.-E.; Duval, C.; Palacios, J.; Boufaied, I.; Hudon-Thibeault, A.-A.; Nadeau-Vallée, M.; Vaillancourt, C.; Sibley, C.P.; Abrahams, V.M.; Jones, R.L.; et al. Uric acid crystals induce placental inflammation and alter trophoblast function via an IL-1–Dependent pathway: Implications for fetal growth restriction. J. Immunol. 2017, 198, 443–451. [Google Scholar] [CrossRef]
- Lee, A.J.; Kandiah, N.; Karimi, K.; Clark, D.A.; Ashkar, A.A. Interleukin-15 is required for maximal lipopolysaccharide-induced abortion. J. Leukoc. Biol. 2013, 93, 905–912. [Google Scholar] [CrossRef]
- Harju, K.; Ojaniemi, M.; Rounioja, S.; Glumoff, V.; Paananen, R.; Vuolteenaho, R.; Hallman, M. Expression of Toll-like receptor 4 and endotoxin responsiveness in mice during perinatal period. Pediatric Res. 2005, 57, 644–648. [Google Scholar] [CrossRef]
- Gibbs, R.S.; McDuffie, R.S.; Kunze, M.; Barr, J.M.; Wolf, D.M.; Sze, C.I.; Shikes, R.; Sherman, M.P. Experimental intrauterine infection with Prevotella bivia in New Zealand White rabbits. Am. J. Obstet. Gynecol. 2004, 190, 1082–1086. [Google Scholar] [CrossRef] [PubMed]
- Cotechini, T.; Komisarenko, M.; Sperou, A.; Macdonald-Goodfellow, S.; Adams, M.A.; Graham, C.H. Inflammation in rat pregnancy inhibits spiral artery remodeling leading to fetal growth restriction and features of preeclampsia. J. Exp. Med. 2014, 211, 165–179. [Google Scholar] [CrossRef] [PubMed]
- Kasawara, K.T.; Cotechini, T.; Macdonald-Goodfellow, S.K.; Surita, F.G.; Pinto e Silva, J.L.; Tayade, C.; Othman, M.; Ozolinš, T.R.S.; Graham, C.H. Moderate exercise attenuates lipopolysaccharide-induced inflammation and associated maternal and fetal morbidities in pregnant rats. PLoS ONE 2016, 11, e0154405. [Google Scholar] [CrossRef] [PubMed]
- Cotechini, T.; Hopman, W.J.; Graham, C.H. Inflammation-induced fetal growth restriction in rats is associated with altered placental morphometrics. Placenta 2014, 35, 575–581. [Google Scholar] [CrossRef] [PubMed]
- Robb, K.P.; Cotechini, T.; Allaire, C.; Sperou, A.; Graham, C.H. Inflammation-induced fetal growth restriction in rats is associated with increased placental HIF-1α accumulation. PLoS ONE 2017, 12, e0175805. [Google Scholar] [CrossRef] [PubMed]
- Ushida, T.; Macdonald-Goodfellow, S.K.; Quadri, A.; Tse, M.Y.; Winn, L.M.; Pang, S.C.; Adams, M.A.; Kotani, T.; Kikkawa, F.; Graham, C.H. Persistence of risk factors associated with maternal cardiovascular disease following aberrant inflammation in rat pregnancy. Biol. Reprod. 2017, 97, 143–152. [Google Scholar] [CrossRef]
- Palei, A.C.; Spradley, F.T.; Granger, J.P. Chronic hyperleptinemia results in the development of hypertension in pregnant rats. Am. J. Physiol. Integr. Comp. Physiol. 2015, 308, R855–R861. [Google Scholar] [CrossRef]
- Kliegman, R.M. Alterations of fasting glucose and fat metabolism in intrauterine growth-retarded newborn dogs. Am. J. Physiol. Endocrinol. Metab. 1989, 256, E380–E385. [Google Scholar] [CrossRef]
- Maxwell, J.R.; Denson, J.L.; Joste, N.E.; Robinson, S.; Jantzie, L.L. Combined in utero hypoxia-ischemia and lipopolysaccharide administration in rats induces chorioamnionitis and a fetal inflammatory response syndrome. Placenta 2015, 36, 1378–1384. [Google Scholar] [CrossRef]
- Celik, H.; Ayar, A. Effects of erythromycin on pregnancy duration and birth weight in lipopolysaccharide-induced preterm labor in pregnant rats. Eur. J. Obstet. Gynecol. Reprod. Biol. 2002, 103, 22–25. [Google Scholar] [CrossRef]
- Bennett, W.A.; Terrone, D.A.; Rinehart, B.K.; Kassab, S.; Martin, J.N.; Granger, J.P. Intrauterine endotoxin infusion in rat pregnancy induces preterm delivery and increases placental prostaglandin F(2α) metabolite levels. Am. J. Obstet. Gynecol. 2000, 182, 1496–1501. [Google Scholar] [CrossRef] [PubMed]
- Kaga, N.; Katsuki, Y.; Obata, M.; Shibutani, Y. Repeated administration of low-dose lipopolysaccharide induces preterm delivery in mice: A model for human preterm parturition and for assessment of the therapeutic ability of drugs against preterm delivery. Am. J. Obstet. Gynecol. 1996, 174, 754–759. [Google Scholar] [CrossRef]
- Lee, P.R.; Kim, S.R.; Jung, B.K.; Kim, K.R.; Chung, J.Y.; Won, H.S.; Kim, A. Therapeutic effect of cyclo-oxygenase inhibitors with different isoform selectivity in lipopolysaccharide-induced preterm birth in mice. Am. J. Obstet. Gynecol. 2003, 189, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Gross, G.; Imamura, T.; Vogt, S.K.; Wozniak, D.F.; Nelson, D.M.; Sadovsky, Y.; Muglia, L.J. Inhibition of cyclooxygenase-2 prevents inflammation-mediated preterm labor in the mouse. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2000, 278, R1415–R1423. [Google Scholar] [CrossRef]
- Buhimschi, I.A.; Buhimschi, C.S.; Weiner, C.P. Protective effect of N-acetylcysteine against fetal death and preterm labor induced by maternal inflammation. Am. J. Obstet. Gynecol. 2003, 188, 203–208. [Google Scholar] [CrossRef]
- Elovitz, M.A.; Wang, Z.; Chien, E.K.; Rychlik, D.F.; Phillippe, M. A new model for inflammation-induced preterm birth: The role of platelet-activating factor and toll-like receptor-4. Am. J. Pathol. 2003, 163, 2103–2111. [Google Scholar] [CrossRef]
- Reznikov, L.L.; Fantuzzi, G.; Selzman, C.H.; Shames, B.D.; Barton, H.A.; Bell, H.; McGregor, J.A.; Dinarello, C.A. Utilization of Endoscopic Inoculation in a Mouse Model of Intrauterine Infection-Induced Preterm Birth: Role of Interleukin 1β1. Biol. Reprod. 1999, 60, 1231–1238. [Google Scholar] [CrossRef] [PubMed]
- Romero, R.; Mazor, M.; Tartakovsky, B. Systemic administration of interleukin-1 induces preterm parturition in mice. Am. J. Obstet. Gynecol. 1991, 165, 969–971. [Google Scholar] [CrossRef]
- Kajikawa, S.; Kaga, N.; Futamura, Y.; Kakinuma, C.; Shibutani, Y. Lipoteichoic acid induces preterm delivery in mice. J. Pharmacol. Toxicol. Methods 1998, 39, 147–154. [Google Scholar] [CrossRef]
- Katsuki, Y. Ability of intrauterine bacterial lipopolysaccharide to cause in situ uterine contractions in pregnant rabbits. Acta Obstet. Gynecol. Scand. 1997, 76, 26–32. [Google Scholar] [CrossRef]
- Davies, J.K.; Shikes, R.H.; Sze, C.I.; Leslie, K.K.; McDuffie, R.S.; Romero, R.; Gibbs, R.S. Histologic inflammation in the maternal and fetal compartments in a rabbit model of acute intra-amniotic infection. Am. J. Obstet. Gynecol. 2000, 183, 1088–1093. [Google Scholar] [CrossRef]
- Schlafer, D.H.; Yuh, B.; Foley, G.L.; Elssaser, T.H.; Sadowsky, D.; Nathanielsz, P.W. Effect of salmonella endotoxin administered to the pregnant sheep at 133–142 days gestation on fetal oxygenation, maternal and fetal adrenocorticotropic hormone and cortisol, and maternal plasma tufnor necrosis factor α Concentrations1. Biol. Reprod. 1994, 50, 1297–1302. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Grigsby, P.L.; Hirst, J.J.; Scheerlinck, J.-P.; Phillips, D.J.; Jenkin, G. Fetal responses to maternal and intra-amniotic lipopolysaccharide administration in Sheep1. Biol. Reprod. 2003, 68, 1695–1702. [Google Scholar] [CrossRef] [PubMed]
- Gravett, M.G.; Haluska, G.J.; Cook, M.J.; Novy, M.J.; Nageotte, M.P.; Lamey, J.; Caillouette, J.C.; Burry, K.; Hobel, C.; Henrichs, L.; et al. Fetal and maternal endocrine responses to experimental intrauterine infection in rhesus monkeys. Am. J. Obstet. Gynecol. 1996, 174, 1725–1733. [Google Scholar] [CrossRef]
- Gravett, M.G.; Witkin, S.S.; Haluska, G.J.; Edwards, J.E.L.; Cook, M.J.; Novy, M.J. An experimental model for intraamniotic infection and preterm labor in rhesus monkeys. Am. J. Obstet. Gynecol. 1994, 171, 1660–1667. [Google Scholar] [CrossRef]
- Ashkar, A.A.; Croy, B.A. Interferon-γ Contributes to the normalcy of murine Pregnancy1. Biol. Reprod. 1999, 61, 493–502. [Google Scholar] [CrossRef] [PubMed]
- Ashkar, A.A.; Black, G.P.; Wei, Q.; He, H.; Liang, L.; Head, J.R.; Croy, B.A. Assessment of requirements for IL-15 and IFN regulatory factors in uterine NK cell differentiation and function during pregnancy. J. Immunol. 2003, 171, 2937–2944. [Google Scholar] [CrossRef]
- Schofield, G.; Kimber, S.J. Leukocyte subpopulations in the uteri of leukemia inhibitory factor knockout mice during early Pregnancy1. Biol. Reprod. 2005, 72, 872–878. [Google Scholar] [CrossRef]
- Kieckbusch, J.; Gaynor, L.M.; Moffett, A.; Colucci, F. MHC-dependent inhibition of uterine NK cells impedes fetal growth and decidual vascular remodelling. Nat. Commun. 2014, 5, 3359. [Google Scholar] [CrossRef]
- Madeja, Z.; Yadi, H.; Apps, R.; Boulenouar, S.; Roper, S.J.; Gardner, L.; Moffett, A.; Colucci, F.; Hemberger, M. Paternal MHC expression on mouse trophoblast affects uterine vascularization and fetal growth. Proc. Natl. Acad. Sci. USA 2011, 108, 4012–4017. [Google Scholar] [CrossRef]
- Brouwers, L.; Franx, A.; Vogelvang, T.E.; Houben, M.L.; van Rijn, B.B.; Nikkels, P.G.J. Association of maternal prepregnancy body mass index with placental histopathological characteristics in uncomplicated term pregnancies. Pediatr. Dev. Pathol. 2019, 22, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Wallace, J.M.; Horgan, G.W.; Bhattacharya, S. Placental weight and efficiency in relation to maternal body mass index and the risk of pregnancy complications in women delivering singleton babies. Placenta 2012, 33, 611–618. [Google Scholar] [CrossRef] [PubMed]
- Challier, J.C.C.; Basu, S.; Bintein, T.; Minium, J.; Hotmire, K.; Catalano, P.M.M.; Hauguel-de Mouzon, S. Obesity in pregnancy stimulates macrophage accumulation and inflammation in the placenta. Placenta 2008, 29, 274–281. [Google Scholar] [CrossRef] [PubMed]
- Wallace, A.E.; Fraser, R.; Cartwright, J.E. Extravillous trophoblast and decidual natural killer cells: A remodelling partnership. Hum. Reprod. Update 2012, 18, 458–471. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Curran, P.; Raker, C.; Martin, S.; Larson, L.; Bourjeily, G. Placental findings associated with maternal obesity at early pregnancy. Pathol. Res. Pract. 2016, 212, 282–287. [Google Scholar] [CrossRef] [PubMed]
- Saben, J.; Lindsey, F.; Zhong, Y.; Thakali, K.; Badger, T.M.M.; Andres, A.; Gomez-Acevedo, H.; Shankar, K. Maternal obesity is associated with a lipotoxic placental environment. Placenta 2014, 35, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Loardi, C.; Falchetti, M.; Prefumo, F.; Facchetti, F.; Frusca, T. Placental morphology in pregnancies associated with pregravid obesity. J. Matern. Neonatal Med. 2016, 29, 2611–2616. [Google Scholar] [CrossRef]
- Ye, K.; Li, L.; Zhang, D.; Li, Y.; Wang, H.Q.; Lai, H.L.; Hu, C.L. Effect of maternal obesity on fetal growth and expression of placental fatty acid transporters. JCRPE J. Clin. Res. Pediatr. Endocrinol. 2017, 9, 300–307. [Google Scholar]
- Nam, H.K.; Lee, K.H. Small for gestational age and obesity: Epidemiology and general risks. Ann. Pediatr. Endocrinol. Metab. 2018, 23, 9–13. [Google Scholar] [CrossRef]
- Radulescu, L.; Munteanu, O.; Popa, F.; Cirstoiu, M. The implications and consequences of maternal obesity on fetal intrauterine growth restriction. J. Med. Life 2013, 6, 292–298. [Google Scholar]
- Stuart, T.J.; O’Neill, K.; Condon, D.; Sasson, I.; Sen, P.; Xia, Y.; Simmons, R.A. Diet-induced obesity alters the maternal metabolome and early placenta transcriptome and decreases placenta vascularity in the mouse. Biol. Reprod. 2018, 98, 795–809. [Google Scholar] [CrossRef] [PubMed]
- Perdu, S.; Castellana, B.; Kim, Y.; Chan, K.; DeLuca, L.; Beristain, A.G. Maternal obesity drives functional alterations in uterine NK cells. JCI Insight 2016, 1, e85560. [Google Scholar] [CrossRef] [PubMed]
- Roberts, K.A.A.; Riley, S.C.C.; Reynolds, R.M.M.; Barr, S.; Evans, M.; Statham, A.; Hor, K.; Jabbour, H.N.N.; Norman, J.E.E.; Denison, F.C.C. Placental structure and inflammation in pregnancies associated with obesity. Placenta 2011, 32, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Rhee, J.S.; Saben, J.L.; Mayer, A.L.; Schulte, M.B.; Asghar, Z.; Stephens, C.; Chi, M.M.Y.; Moley, K.H. Diet-induced obesity impairs endometrial stromal cell decidualization: A potential role for impaired autophagy. Hum. Reprod. 2016, 31, 1315–1326. [Google Scholar] [CrossRef] [PubMed]
- Pavan, L.; Tsatsaris, V.; Hermouet, A.; Therond, P.; Evain-Brion, D.; Fournier, T. Oxidized low-density lipoproteins inhibit trophoblastic cell invasion. J. Clin. Endocrinol. Metab. 2004, 89, 1969–1972. [Google Scholar] [CrossRef]
- Liu, H.; Wu, Y.; Qiao, F.; Gong, X. Effect of leptin on cytotrophoblast proliferation and invasion. J. Huazhong Univ. Sci. Technol. [Medical Sci.] 2009, 29, 631–636. [Google Scholar] [CrossRef]
- Moffett, A.; Loke, C. Immunology of placentation in eutherian mammals. Nat. Rev. Immunol. 2006, 6, 584–594. [Google Scholar] [CrossRef]
- Lim, H.J.; Wang, H. Uterine disorders and pregnancy complications: Insights from mouse models. J. Clin. Investig. 2010, 120, 1004–1015. [Google Scholar] [CrossRef]
- Sato, Y. Endovascular trophoblast and spiral artery remodeling. Mol. Cell. Endocrinol. 2020, 503, 110699. [Google Scholar] [CrossRef]
- O’Rourke, R.W.; Gaston, G.D.; Meyer, K.A.; White, A.E.; Marks, D.L. Adipose tissue NK cells manifest an activated phenotype in human obesity. Metabolism 2013, 62, 1557–1561. [Google Scholar] [CrossRef]
- Wouters, K.; Gaens, K.; Bijnen, M.; Verboven, K.; Jocken, J.; Wetzels, S.; Wijnands, E.; Hansen, D.; Van Greevenbroek, M.; Duijvestijn, A.; et al. Circulating classical monocytes are associated with CD11c+ macrophages in human visceral adipose tissue. Sci. Rep. 2017, 7, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, S.; Koh, V.; Kua, L.-F.; So, J.; Davide, L.; Lim, K.S.; Petersen, S.H.; Yong, W.-P.; Shabbir, A.; Kono, K. Accumulation of CD11c + CD163 + Adipose tissue macrophages through upregulation of intracellular 11β-HSD1 in human obesity. J. Immunol. 2016, 197, 3735–3745. [Google Scholar] [CrossRef] [PubMed]
- Fujisaka, S.; Usui, I.; Bukhari, A.; Ikutani, M.; Oya, T.; Kanatani, Y.; Tsuneyama, K.; Nagai, Y.; Takatsu, K.; Urakaze, M.; et al. Regulatory mechanisms for adipose tissue M1 and M2 macrophages in diet-induced obese mice. Diabetes 2009, 58, 2574–2582. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Perrard, X.D.; Wang, Q.; Perrard, J.L.; Polsani, V.R.; Jones, P.H.; Smith, C.W.; Ballantyne, C.M. CD11c expression in adipose tissue and blood and its role in diet-induced obesity. Arterioscler. Thromb. Vasc. Biol. 2010, 30, 186–192. [Google Scholar] [CrossRef]
- Endo, Y.; Asou, H.K.; Matsugae, N.; Hirahara, K.; Shinoda, K.; Tumes, D.J.; Tokuyama, H.; Yokote, K.; Nakayama, T. Obesity drives Th17 cell differentiation by inducing the lipid metabolic kinase, ACC1. Cell Rep. 2015, 12, 1042–1055. [Google Scholar] [CrossRef]
- Strissel, K.J.; DeFuria, J.; Shaul, M.E.; Bennett, G.; Greenberg, A.S.; Obin, M.S. T-cell recruitment and Th1 polarization in adipose tissue during diet-induced obesity in C57BL/6 mice. Obesity (Silver Spring) 2010, 18, 1918–1925. [Google Scholar] [CrossRef]
- Lumeng, C.N.; Bodzin, J.L.; Saltiel, A.R.; Lumeng, C.N.; Bodzin, J.L.; Saltiel, A.R. Obesity induces a phenotypic switch in adipose tissue macrophage polarization. J. Clin. Investig. 2007, 117, 175–184. [Google Scholar] [CrossRef]
- Wensveen, F.M.; Jelenčić, V.; Valentić, S.; Šestan, M.; Wensveen, T.T.; Theurich, S.; Glasner, A.; Mendrila, D.; Štimac, D.; Wunderlich, F.T.; et al. NK cells link obesity-induced adipose stress to inflammation and insulin resistance. Nat. Immunol. 2015, 16, 376–385. [Google Scholar] [CrossRef]
- Guleria, I.; Sayegh, M.H. Maternal Acceptance of the Fetus: True Human Tolerance. J. Immunol. 2007, 178, 3345–3351. [Google Scholar] [CrossRef]
- Rajagopalan, S.; Long, E.O. KIR2DL4 (CD158d): An activation receptor for HLA-G. Front. Immunol. 2012, 3. [Google Scholar] [CrossRef]
- King, A.; Allan, D.S.J.; Bowen, M.; Powis, S.J.; Joseph, S.; Verma, S.; Hiby, S.E.; McMichael, A.J.; Wai Loke, Y.; Braud, V.M. HLA-E is expressed on trophoblast and interacts with CD94 / NKG2 receptors on decidual NK cells. Eur. J. Immunol. 2000, 30, 1623–1631. [Google Scholar] [CrossRef]
- Braud, V.M.; Allan, D.S.J.; O’Callaghan, C.A.; Soderstrom, K.; D’Andrea, A.; Ogg, G.S.; Lazetic, S.; Young, N.T.; Bell, J.I.; Phillips, J.H.; et al. HLA-E binds to natural killer cell receptors CD94/NKG2A, B and C. Nature 1998, 391, 795–799. [Google Scholar] [CrossRef] [PubMed]
- Rieger, L. Th1- and Th2-like cytokine production by first trimester decidual large granular lymphocytes is influenced by HLA-G and HLA-E. Mol. Hum. Reprod. 2002, 8, 255–261. [Google Scholar] [CrossRef] [PubMed]
- Kanai, T.; Fujii, T.; Unno, N.; Yamashita, T.; Hyodo, H.; Miki, A.; Hamai, Y.; Kozuma, S.; Taketani, Y. Human leukocyte antigen-G-expressing cells differently modulate the release of cytokines from mononuclear cells present in the decidua versus peripheral blood. Am. J. Reprod. Immunol. 2001, 45, 94–99. [Google Scholar] [CrossRef]
- Rouas-Freiss, N.; Marchal, R.E.; Kirszenbaum, M.; Dausset, J.; Carosella, E.D. The α1 domain of HLA-G1 and HLA-G2 inhibits cytotoxicity induced by natural killer cells: Is HLA-G the public ligand for natural killer cell inhibitory receptors? Proc. Natl. Acad. Sci. USA 1997, 94, 5249–5254. [Google Scholar] [CrossRef]
- Hiby, S.E.; Apps, R.; Sharkey, A.M.; Farrell, L.E.; Gardner, L.; Mulder, A.; Claas, F.H.; Walker, J.J.; Redman, C.C.; Morgan, L.; et al. Maternal activating KIRs protect against human reproductive failure mediated by fetal HLA-C2. J. Clin. Investig. 2010, 120, 4102–4110. [Google Scholar] [CrossRef]
- Xiong, S.; Sharkey, A.M.; Kennedy, P.R.; Gardner, L.; Farrell, L.E.; Chazara, O.; Bauer, J.; Hiby, S.E.; Colucci, F.; Moffett, A. Maternal uterine NK cell-activating receptor KIR2DS1 enhances placentation. J. Clin. Investig. 2013, 123, 4264–4272. [Google Scholar] [CrossRef]
- Salvany-Celades, M.; van der Zwan, A.; Benner, M.; Setrajcic-Dragos, V.; Bougleux Gomes, H.A.; Iyer, V.; Norwitz, E.R.; Strominger, J.L.; Tilburgs, T. Three types of functional regulatory T Cells control T Cell responses at the human maternal-fetal interface. Cell Rep. 2019, 27, 2537–2547.e5. [Google Scholar] [CrossRef]
- McIntire, R.H.; Morales, P.J.; Petroff, M.G.; Colonna, M.; Hunt, J.S. Recombinant HLA-G5 and -G6 drive U937 myelomonocytic cell production of TGF-β1. J. Leukoc. Biol. 2004, 76, 1220–1228. [Google Scholar] [CrossRef]
- Pollheimer, J.; Vondra, S.; Baltayeva, J.; Beristain, A.G.; Knöfler, M. Regulation of placental extravillous trophoblasts by the maternal uterine environment. Front. Immunol. 2018, 9, 1–18. [Google Scholar] [CrossRef]
- Onodera, T.; Fukuhara, A.; Jang, M.H.; Shin, J.; Aoi, K.; Kikuta, J.; Otsuki, M.; Ishii, M.; Shimomura, I. Adipose tissue macrophages induce PPARγ-high FOXP3+ regulatory T cells. Sci. Rep. 2015, 5, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Deiuliis, J.; Shah, Z.; Shah, N.; Needleman, B.; Mikami, D.; Narula, V.; Perry, K.; Hazey, J.; Kampfrath, T.; Kollengode, M.; et al. Visceral adipose inflammation in obesity is associated with critical alterations in tregulatory cell numbers. PLoS ONE 2011, 6, e16376. [Google Scholar] [CrossRef]
- Ghiringhelli, F.; Ménard, C.; Terme, M.; Flament, C.; Taieb, J.; Chaput, N.; Puig, P.E.; Novault, S.; Escudier, B.; Vivier, E.; et al. CD4+CD25+ regulatory T cells inhibit natural killer cell functions in a transforming growth factor-beta-dependent manner. J. Exp. Med. 2005, 202, 1075–1085. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, Z.; Tian, M.; Hu, X.; Wang, L.; Ji, J.; Liao, A. The altered PD-1/PD-L1 pathway delivers the ‘one-two punch’ effects to promote the Treg/Th17 imbalance in pre-eclampsia. Cell. Mol. Immunol. 2018, 15, 710–723. [Google Scholar] [CrossRef] [PubMed]
- McClymont, S.A.; Putnam, A.L.; Lee, M.R.; Esensten, J.H.; Liu, W.; Hulme, M.A.; Hoffmüller, U.; Baron, U.; Olek, S.; Bluestone, J.A.; et al. Plasticity of human regulatory T Cells in healthy subjects and patients with type 1 diabetes. J. Immunol. 2011, 186, 3918–3926. [Google Scholar] [CrossRef] [PubMed]
- Bähr, I.; Goritz, V.; Doberstein, H.; Hiller, G.G.R.; Rosenstock, P.; Jahn, J.; Pörtner, O.; Berreis, T.; Mueller, T.; Spielmann, J.; et al. Diet-Induced Obesity Is Associated with an Impaired NK Cell Function and an Increased Colon Cancer Incidence. J. Nutr. Metab. 2017, 2017, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Viel, S.; Besson, L.; Charrier, E.; Marçais, A.; Disse, E.; Bienvenu, J.; Walzer, T.; Dumontet, C. Alteration of Natural Killer cell phenotype and function in obese individuals. Clin. Immunol. 2017, 177, 12–17. [Google Scholar] [CrossRef]
- Bähr, I.; Jahn, J.; Zipprich, A.; Pahlow, I.; Spielmann, J.; Kielstein, H. Impaired natural killer cell subset phenotypes in human obesity. Immunol. Res. 2018, 66, 234–244. [Google Scholar] [CrossRef]
- Lobo, T.F.; Borges, C.d.M.; Mattar, R.; Gomes, C.P.; Angelo, d.A.G.S.; Pendeloski, K.P.T.; Daher, S. Impaired Treg and NK cells profile in overweight women with gestational diabetes mellitus. Am. J. Reprod. Immunol. 2018, 79, 3–7. [Google Scholar] [CrossRef]
- Tobin, L.M.; Mavinkurve, M.; Carolan, E.; Kinlen, D.; O’Brien, E.C.; Little, M.A.; Finlay, D.K.; Cody, D.; Hogan, A.E.; O’Shea, D. NK cells in childhood obesity are activated, metabolically stressed, and functionally deficient. JCI Insight 2017, 2, 1–9. [Google Scholar] [CrossRef]
- Castellana, B.; Perdu, S.; Kim, Y.; Chan, K.; Atif, J.; Marziali, M.; Beristain, A.G. Maternal obesity alters uterine NK activity through a functional KIR2DL1/S1 imbalance. Immunol. Cell Biol. 2018, 96, 805–819. [Google Scholar] [CrossRef] [PubMed]
- Geng, Y.-J.; Wu, Q.; Muszynski, M.; Hansson, G.K.; Libby, P. Apoptosis of vascular smooth muscle cells induced by In Vitro stimulation with Interferon-γ, tumor necrosis Factor–α, and Interleukin-1β. Arterioscler. Thromb. Vasc. Biol. 1996, 16, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Solano, M.E. Decidual immune cells: Guardians of human pregnancies. Best Pract. Res. Clin. Obstet. Gynaecol. 2019, 60, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Vacca, P.; Cantoni, C.; Prato, C.; Fulcheri, E.; Moretta, A.; Moretta, L.; Mingari, M.C. Regulatory role of NKp44, NKp46, DNAM-1 and NKG2D receptors in the interaction between NK cells and trophoblast cells. Evidence for divergent functional profiles of decidual versus peripheral NK cells. Int. Immunol. 2008, 20, 1395–1405. [Google Scholar] [CrossRef]
- Yougbaré, I.; Tai, W.-S.S.; Zdravic, D.; Oswald, B.E.; Lang, S.; Zhu, G.; Leong-Poi, H.; Qu, D.; Yu, L.; Dunk, C.; et al. Activated NK cells cause placental dysfunction and miscarriages in fetal alloimmune thrombocytopenia. Nat. Commun. 2017, 8, 224. [Google Scholar] [CrossRef]
- Murphy, S.P.; Fast, L.D.; Hanna, N.N.; Sharma, S. Uterine NK cells mediate inflammation-induced fetal demise in IL-10-Null mice. J. Immunol. 2005, 175, 4084–4090. [Google Scholar] [CrossRef]
- Lumeng, C.N. Innate immune activation in obesity. Mol. Asp. Med. 2013, 34, 12–29. [Google Scholar] [CrossRef]
- Travers, R.L.; Motta, A.C.; Betts, J.A.; Bouloumié, A.; Thompson, D. The impact of adiposity on adipose tissue-resident lymphocyte activation in humans. Int. J. Obes. (Lond.) 2015, 39, 762–769. [Google Scholar] [CrossRef]
- Hill, D.A.; Lim, H.W.; Kim, Y.H.; Ho, W.Y.; Foong, Y.H.; Nelson, V.L.; Nguyen, H.C.B.; Chegireddy, K.; Kim, J.; Habertheuer, A.; et al. Distinct macrophage populations direct inflammatory versus physiological changes in adipose tissue. Proc. Natl. Acad. Sci. USA 2018, 115, E5096–E5105. [Google Scholar] [CrossRef]
- Wallace, J.G.; Bellissimo, C.J.; Yeo, E.; Fei Xia, Y.; Petrik, J.J.; Surette, M.G.; Bowdish, D.M.E.; Sloboda, D.M. Obesity during pregnancy results in maternal intestinal inflammation, placental hypoxia, and alters fetal glucose metabolism at mid-gestation. Sci. Rep. 2019, 9, 17621. [Google Scholar] [CrossRef]
- Schonkeren, D.; Van Der Hoorn, M.L.; Khedoe, P.; Swings, G.; Van Beelen, E.; Claas, F.; Van Kooten, C.; De Heer, E.; Scherjon, S. Differential distribution and phenotype of decidual macrophages in preeclamptic versus control pregnancies. Am. J. Pathol. 2011, 178, 709–717. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Piao, L.; Chen, C.P.; Wu, X.; Yeh, C.C.; Masch, R.; Chang, C.C.; Huang, S.J. Modulation of decidual macrophage polarization by macrophage colony-stimulating factor derived from first-trimester decidual cells: Implication in preeclampsia. Am. J. Pathol. 2016, 186, 1258–1266. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Romero, R.; Miller, D.; Kadam, L.; Mial, T.N.; Plazyo, O.; Garcia-Flores, V.; Hassan, S.S.; Xu, Z.; Tarca, A.L.; et al. An M1-like macrophage polarization in decidual tissue during spontaneous preterm labor that is attenuated by rosiglitazone treatment. J. Immunol. 2016, 196, 2476–2491. [Google Scholar] [CrossRef] [PubMed]
- Laskewitz, A.; Benthem, K.L.V.; Kieffer, T.E.C.; Faas, M.M.; Verkaik-Schakel, R.N.; Plösch, T.; Scherjon, S.A.; Prins, J.R. The influence of maternal obesity on macrophage subsets in the human decidua. Cell. Immunol. 2019, 336, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Sisino, G.; Bouckenooghe, T.; Aurientis, S.; Fontaine, P.; Storme, L.; Vambergue, A. Diabetes during pregnancy influences Hofbauer cells, a subtype of placental macrophages, to acquire a pro-inflammatory phenotype. Biochim. Biophys. Acta Mol. Basis Dis. 2013, 1832, 1959–1968. [Google Scholar] [CrossRef]
- Wu, Z.-M.; Yang, H.; Li, M.; Yeh, C.-C.; Schatz, F.; Lockwood, C.J.; Di, W.; Huang, S.J. Pro-inflammatory cytokine-stimulated first trimester decidual cells enhance macrophage-induced apoptosis of extravillous trophoblasts. Placenta 2012, 33, 188–194. [Google Scholar] [CrossRef]
- Reister, F.; Frank, H.-G.; Kingdom, J.C.P.; Heyl, W.; Kaufmann, P.; Rath, W.; Huppertz, B. Macrophage-Induced apoptosis limits endovascular trophoblast invasion in the uterine wall of preeclamptic women. Lab. Investig. 2001, 81, 1143–1152. [Google Scholar] [CrossRef]
- Renaud, S.J.; Postovit, L.-M.; Macdonald-Goodfellow, S.K.; McDonald, G.T.; Caldwell, J.D.; Graham, C.H. Activated macrophages inhibit human cytotrophoblast invasiveness In Vitro1. Biol. Reprod. 2005, 73, 237–243. [Google Scholar] [CrossRef]
- Felker, A.M.; Chen, Z.; Foster, W.G.; Croy, B.A. Receptors for non-MHC ligands contribute to uterine natural killer cell activation during pregnancy in mice. Placenta 2013, 34, 757–764. [Google Scholar] [CrossRef][Green Version]
- Chakraborty, D.; Rumi, M.A.K.; Konno, T.; Soares, M.J. Natural killer cells direct hemochorial placentation by regulating hypoxia-inducible factor dependent trophoblast lineage decisions. Proc. Natl. Acad. Sci. USA 2011, 108, 16295–16300. [Google Scholar] [CrossRef]
- Milosevic-Stevanovic, J.; Krstic, M.; Radovic-Janosevic, D.; Popovic, J.; Tasic, M.; Stojnev, S. Number of decidual natural killer cells & macrophages in pre-eclampsia. Indian J. Med. Res. 2016, 144, 823–830. [Google Scholar] [PubMed]
- Merline, R.; Moreth, K.; Beckmann, J.; Nastase, M.V.; Zeng-Brouwers, J.; Tralhão, J.G.; Lemarchand, P.; Pfeilschifter, J.; Schaefer, R.M.; Iozzo, R.V.; et al. Signaling by the matrix proteoglycan decorin controls inflammation and cancer through PDCD4 and MicroRNA-21. Sci. Signal. 2011, 4, ra75. [Google Scholar] [CrossRef] [PubMed]
- Fu, B.; Li, X.; Sun, R.; Tong, X.; Ling, B.; Tian, Z.; Wei, H. Natural killer cells promote immune tolerance by regulating inflammatory TH17 cells at the human maternal-fetal interface. Proc. Natl. Acad. Sci. USA 2013, 110, E231–E240. [Google Scholar] [CrossRef] [PubMed]
- Tiemessen, M.M.; Jagger, A.L.; Evans, H.G.; van Herwijnen, M.J.C.; John, S.; Taams, L.S. CD4+CD25+Foxp3+ regulatory T cells induce alternative activation of human monocytes/macrophages. Proc. Natl. Acad. Sci. USA 2007, 104, 19446–19451. [Google Scholar] [CrossRef] [PubMed]
- Obesity and Overweight. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 9 April 2020).
| Model | Animal | Experimental Conditions | Results | Source |
|---|---|---|---|---|
| Obesity | Mouse | High-fat diet | ∆ gut microbiota composition with ↓ gut barrier integrity ↓ artery remodeling and placentation ↑ placental inflammation, hypoxia, uNK cytotoxicity, fetal demise | [127,128,129] |
| Rat | High-fat diet | ↑ trophoblast invasion, MMP9 ↓ artery remodeling | [130] | |
| Rabbit | Cholesterol enriched diet | ↑ concentration of total-cholesterol and lipoproteins ↓ fetal weight | [131] | |
| Hypertension | Rat | Reduced uterine perfusion pressure procedure | ↑ hypertension (mediated through Th cells and AT1) ↑ RORγ+CD4+ T cells and secretion of inflammatory factors | [132,133,134] |
| Mouse | Angiotensin II infusion | ↑ T cell composition in aorta, and aortic hypertrophy | [135] | |
| Type 1 Diabetes | Mouse | Non-obese diabetic (NOD) mice | ↑ placental weights, IFN-γ, tolerogenic antigen presenting cells ↓ uNK, vascular remodeling, fetal weights, T cells in offspring | [136,137,138,139] |
| Viral infection | Mouse | Allogeneically mated female injected poly I:C | ↑ fetal loss, cytotoxicity of CD27posDX5posCD3neg NK ↓ proportion of CD27lowDX5posCD3neg NK | [140] |
| Sterile inflammation | Mouse | Administration of monosodium urate crystals during late gestation | ↑ placental inflammation and FGR ↓ fetal weight = placental weight, placental morphology | [141] |
| Bacterial infection | Mouse | Mice were administered lipopolysaccharide (LPS) intraperitoneally (i.p.) | ↑ uNK, % NK1.1+CD27+ uNK, and NK-mediated fetal demise ↑ pulmonary and placental cytokine production | [142,143] |
| Rabbit | Transcervical P. bivea inoculation | ↑ preterm delivery, chronic intrauterine, and fetal infection | [144] | |
| FGR | Rat | 4-day low-dose LPS injections i.p. | ↓ placental development, vascular remodeling, fetal growth | [145,146,147,148,149] |
| Chronic leptin treatment | ↑ TNF-α expression in placenta ↓ maternal, fetal, and placental weights | [150] | ||
| Canine | Maternal nutrient deprivation | ↓ fetal weight, maternal blood glucose, ketone bodies, and FFA | [151] | |
| Preterm labor | Rat | Intrauterine injury induced by transient hypoxia-ischemia and LPS injection | ↑ acute and subacute placental injury ↑ inflammatory factors in the placenta | [152] |
| i.p. injection of LPS | ↓ birth weights (alleviated with erythromycin treatment) | [153,154] | ||
| Mouse | i.p. injection of LPS | ↑ TLR-4-mediated preterm birth with no neonatal mortality | [155,156,157,158,159,160] | |
| 3 injections of rhIL-1 | Parturition occurred within 24 h | [161] | ||
| Lipoteichoic acid i.p. | ↑ incidence of preterm delivery | [162] | ||
| Rabbit | LPS administered via catheter to uteri five times at 1-h intervals | ↑ uterine contractions ↑ prostaglandin production | [163] | |
| Endocervical E. coli inoculation | ↑ Inflammation in uterus, placenta, and fetal lung | [164] | ||
| Sheep | Intravenous LPS | ↑ fetal hypoxemia ↑ fetal and maternal hypothalamo-hypophyseal-adrenal axis | [165,166] | |
| Monkey | Streptococci inoculation | ↑ spontaneous parturition, steroid hormones | [167,168] | |
| Genetic models of inflammation | Mouse | IFN-γ-/- or IFN-γRα−/− mated with BALB/c male | ↑ uNK, fluid, cellularity, and necrosis in decidua ↓ uNK granularity and decidual artery remodeling | [169] |
| RAG-2-/-yc-/- or IL-15-/- pregnant females received bone marrow from IL-15-/- or C57/BL6 pregnant females IRF-1-/- females treated with mrIL-15 on GD5 for 5 days | RAG-2-/-yc-/- + bone marrow from IL-15-/- ↑ uNK, uNK differentiation, decidual artery remodeling RAG-2-/-yc-/-, or IL-15-/- + bone marrow from C57/BL6 ↓ uNK, MLAp, decidual cellularity and artery remodeling IRF1-/- + mrIL-15 ↓ uNK differentiation, artery remodeling, placental and fetal weight | [170] | ||
| Lif-/- females mated with MF-1 males | ↓ macrophages and NKs in mesometrial stroma ↑ eosinophils, NKs in uterus | [171] | ||
| D8 male father (containing H2-Dd MHC allele) | ↓ NK function, uterine arterial remodeling, fetal growth | [172] | ||
| BALB/cx C57/BL6 (FxM) compared to C57/BL6xBALB/c | ↑ decidual artery diameters | [173] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
St-Germain, L.E.; Castellana, B.; Baltayeva, J.; Beristain, A.G. Maternal Obesity and the Uterine Immune Cell Landscape: The Shaping Role of Inflammation. Int. J. Mol. Sci. 2020, 21, 3776. https://doi.org/10.3390/ijms21113776
St-Germain LE, Castellana B, Baltayeva J, Beristain AG. Maternal Obesity and the Uterine Immune Cell Landscape: The Shaping Role of Inflammation. International Journal of Molecular Sciences. 2020; 21(11):3776. https://doi.org/10.3390/ijms21113776
Chicago/Turabian StyleSt-Germain, Lauren E., Barbara Castellana, Jennet Baltayeva, and Alexander G. Beristain. 2020. "Maternal Obesity and the Uterine Immune Cell Landscape: The Shaping Role of Inflammation" International Journal of Molecular Sciences 21, no. 11: 3776. https://doi.org/10.3390/ijms21113776
APA StyleSt-Germain, L. E., Castellana, B., Baltayeva, J., & Beristain, A. G. (2020). Maternal Obesity and the Uterine Immune Cell Landscape: The Shaping Role of Inflammation. International Journal of Molecular Sciences, 21(11), 3776. https://doi.org/10.3390/ijms21113776





