Neuroprotective and Anti-Inflammatory Effects of Low–Moderate Dose Ionizing Radiation in Models of Alzheimer’s Disease
Abstract
1. Introduction
2. Results
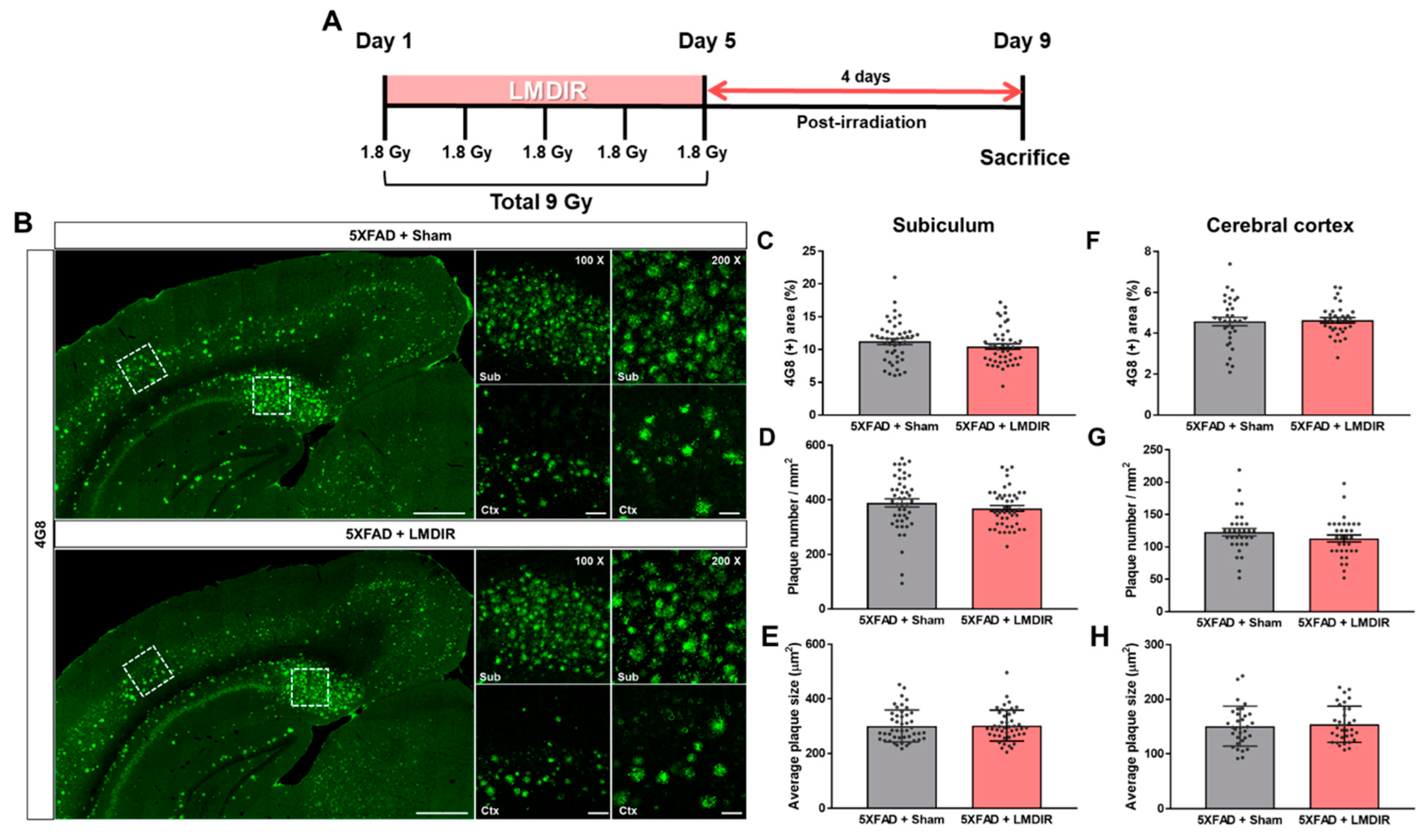
2.1. Short-Term Effect of LMDIR Treatment on Aβ Deposition in the Subiculum and Cerebral Cortex of Aβ-Overexpressing Transgenic Mice
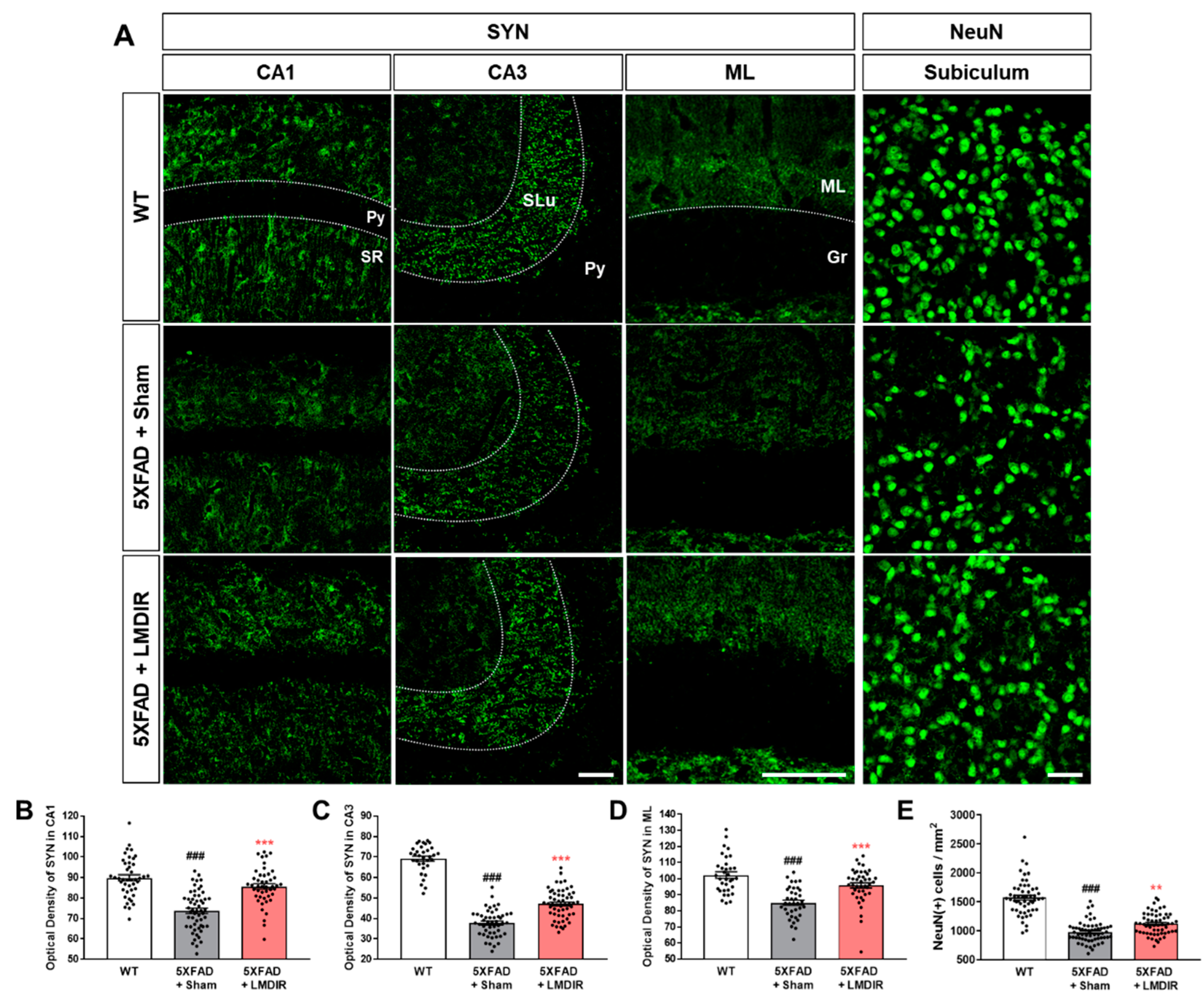
2.2. Inhibitory Effects of LMDIR on Synaptic and Neuronal Loss in the Hippocampal Formation of Aβ-Overexpressing Transgenic Mice
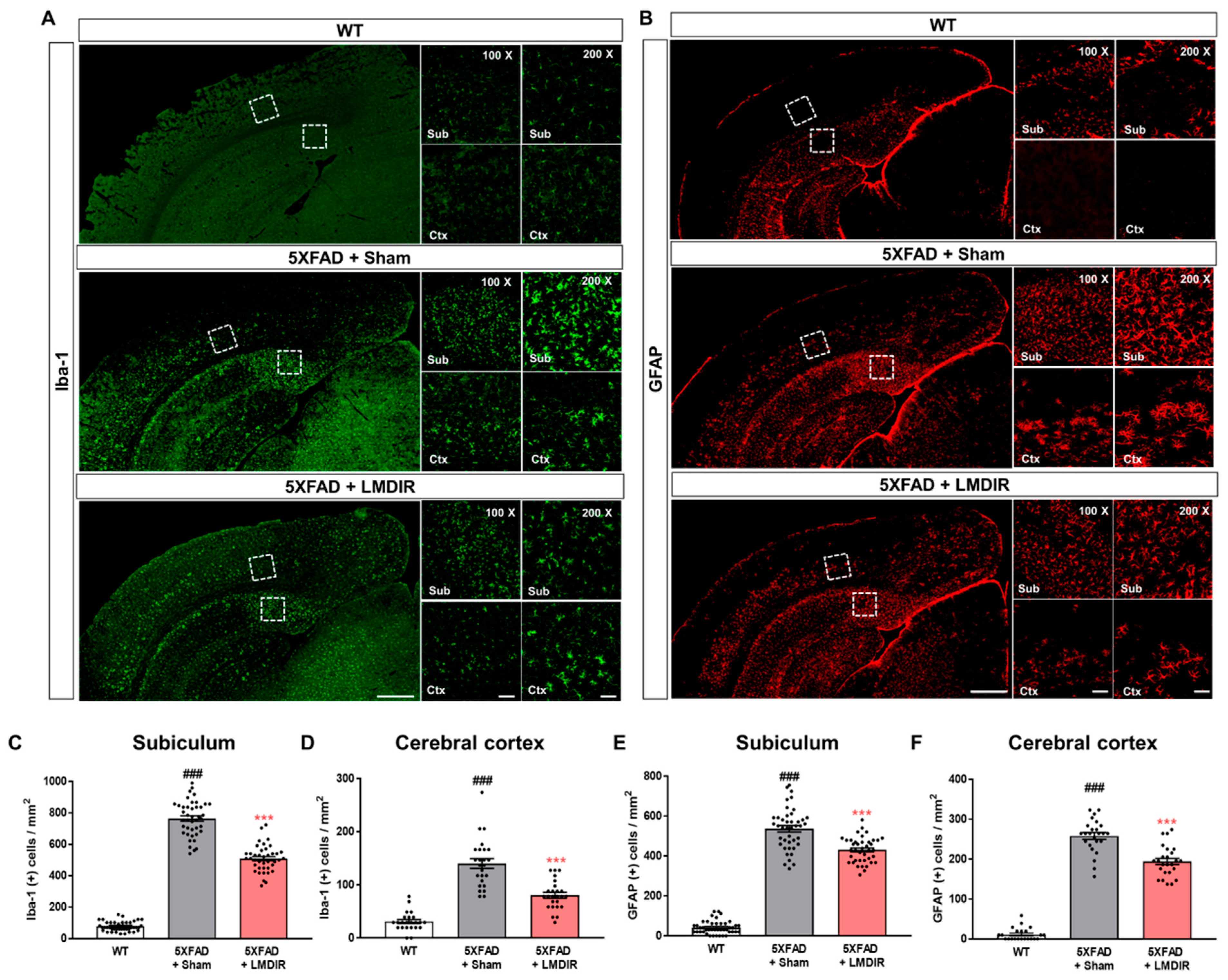
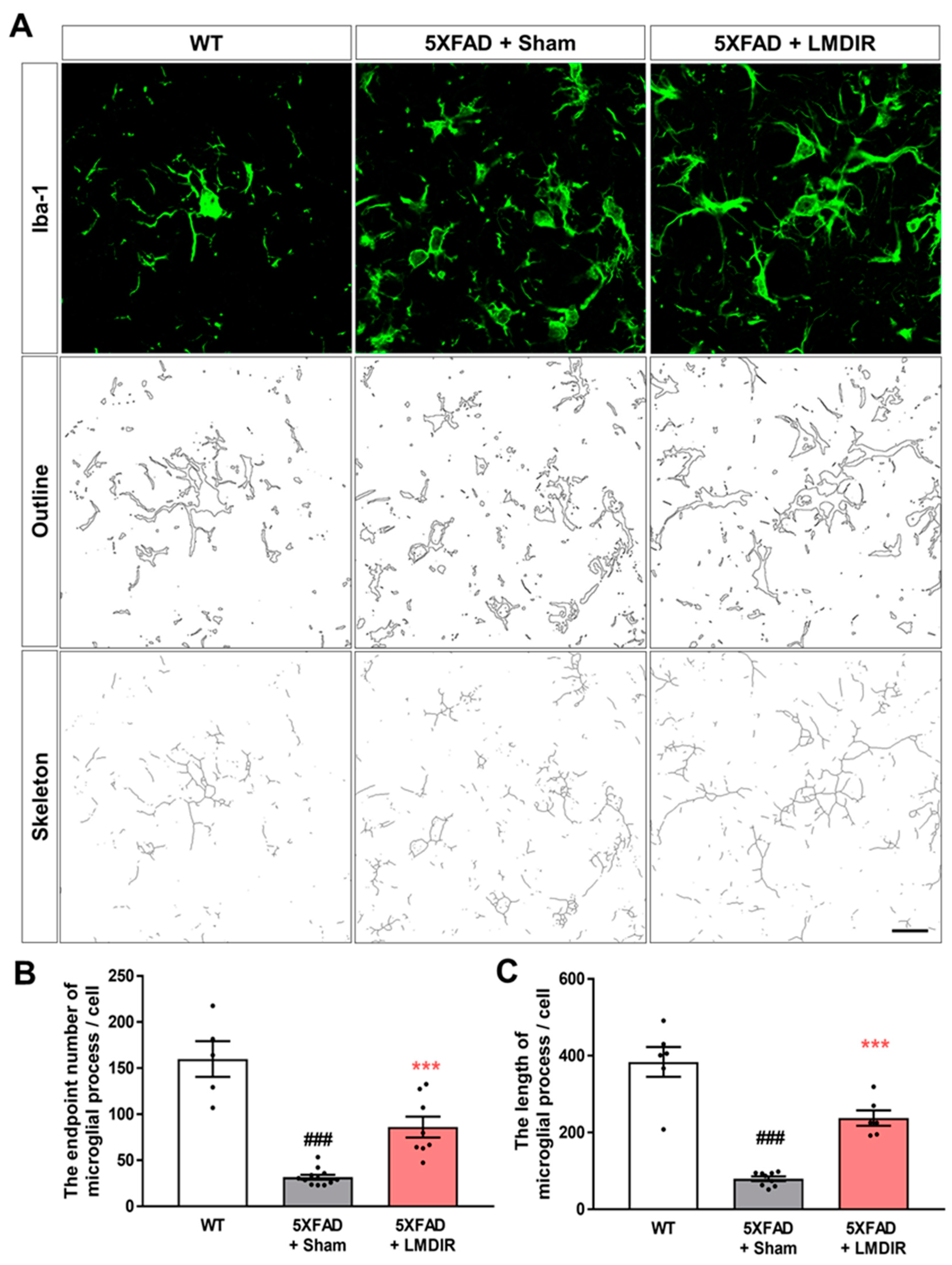
2.3. Attenuating Effects of LMDIR on Neuroinflammation in the Subiculum and Cerebral Cortex of 5XFAD Mice
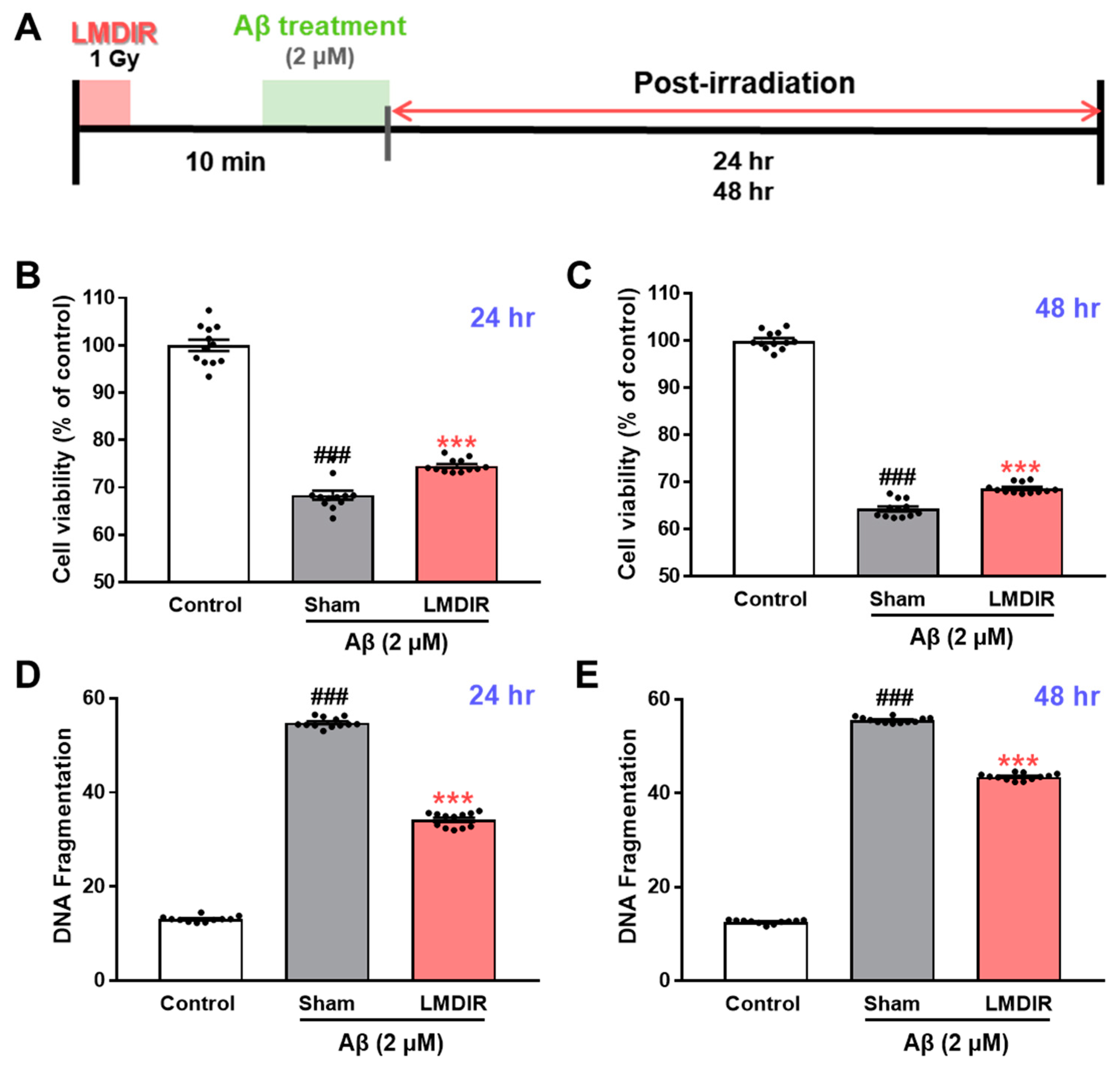
2.4. Direct Effects of LMDIR on Aβ-Induced Cell Death in Cultured Neurons
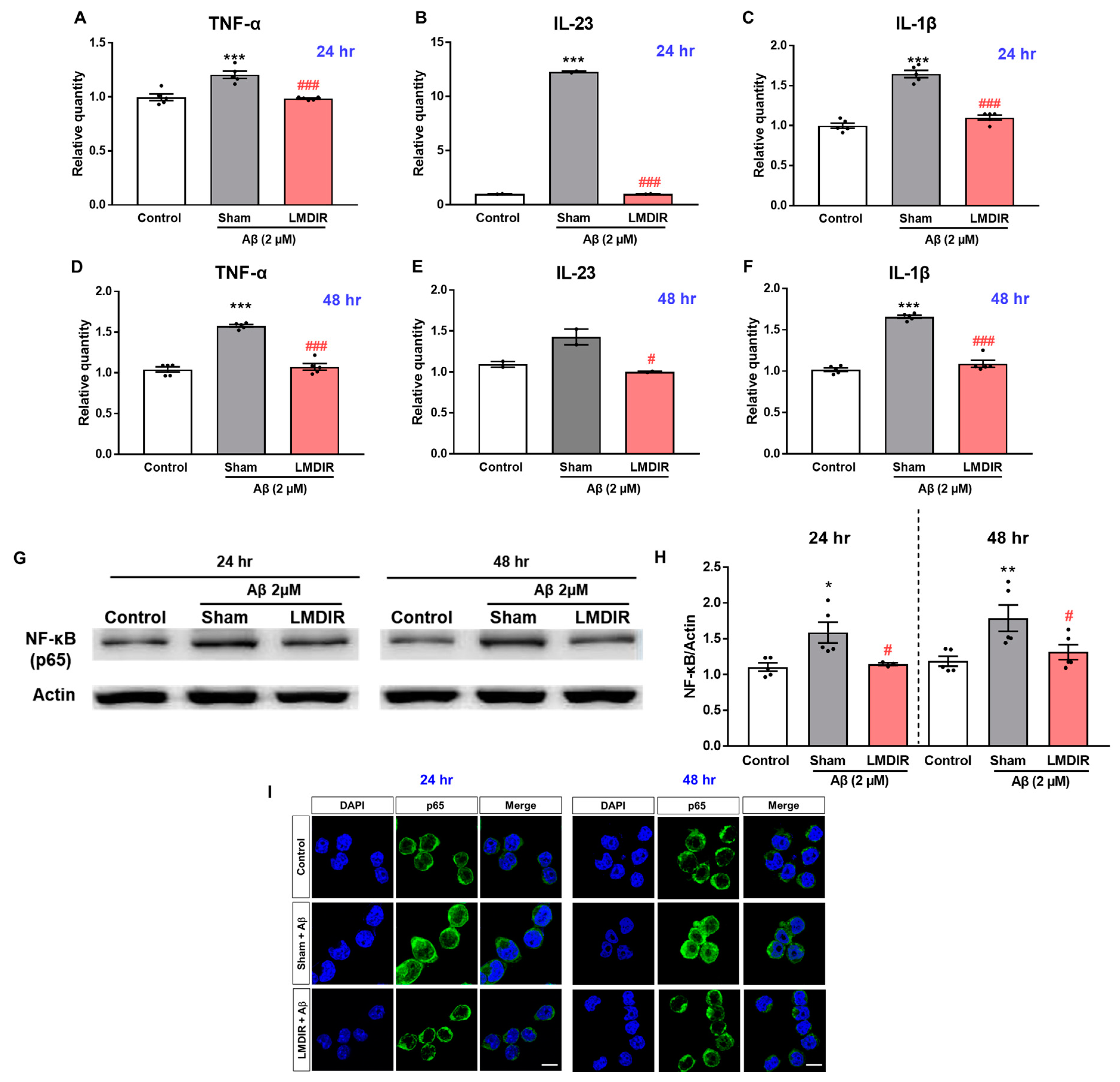
2.5. Alleviatory Effect of LMDIR on Aβ-Induced Neuroinflammatory Cytokine Production and NF-κB Pathway Activation in BV-2 Cells
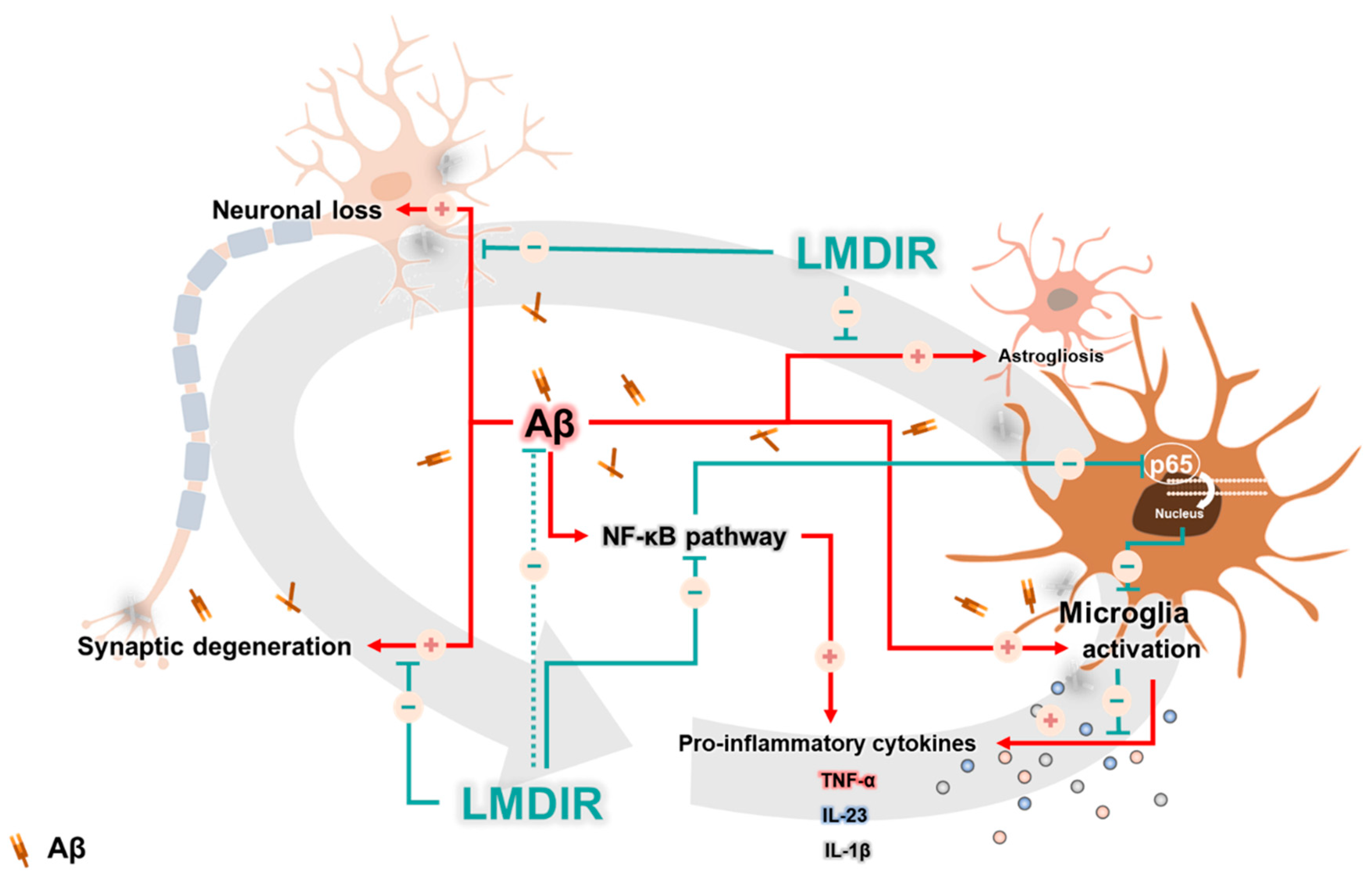
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Cranial Irradiation Procedure
4.3. Preparation of Brain Tissue
4.4. Immunofluorescence Labeling
4.5. Image Acquisition and Analysis
4.6. Skeleton and Fractal Analysis
4.7. Cell Culture
4.8. Measurement of Cell Viability and DNA Fragmentation
4.9. Determination of Pro-Inflammatory Cytokines
4.10. Western Blot Analysis of NF-κB
4.11. Immunocytochemistry of NF-κB
4.12. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| 5XFAD | Five familial AD mutations |
| AD | Alzheimer’s disease |
| Aβ | Amyloid-β |
| DG | Dentate gyrus |
| DMEM | Dulbecco’s Modified Eagle Medium |
| DMSO | Dimethyl sulfoxide |
| GFAP | Glial fibrillary acidic protein |
| Gr | Granular layer |
| Iba-1 | Ionized calcium-binding adaptor molecule 1 |
| IL | Interleukin |
| LDIR | Low-dose ionizing radiation |
| LMDIR | Low–moderate dose ionizing radiation |
| ML | Molecular layer |
| MMP | Metalloproteinase |
| MTT | 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| NeuN | Neuronal nuclei |
| NF-κB | Nuclear factor-kappa B |
| PB | Phosphate buffer |
| PBS | Phosphate-buffered saline |
| PDGF | Platelet-derived growth factor |
| Py | Pyramidal tract |
| ROI | Reactive oxygen intermediates |
| SLu | Stratum lucidum |
| SR | Stratum radiatum |
| Sub | Subiculum |
| SYN | Synaptophysin |
| TNF-α | Tumor necrosis factor-alpha |
References
- Long, J.M.; Holtzman, D.M. Alzheimer Disease: An Update on Pathobiology and Treatment Strategies. Cell 2019, 179, 312–339. [Google Scholar] [CrossRef] [PubMed]
- Winblad, B.; Amouyel, P.; Andrieu, S.; Ballard, C.; Brayne, C.; Brodaty, H.; Cedazo-Minguez, A.; Dubois, B.; Edvardsson, D.; Feldman, H.; et al. Defeating Alzheimer’s disease and other dementias: A priority for European science and society. Lancet Neurol. 2016, 15, 455–532. [Google Scholar] [CrossRef]
- Derouesne, C. Alzheimer and Alzheimer’s disease: The present enlighted by the past. An historical approach. Psychol. Neuropsychiatr. Vieil 2008, 6, 115–128. [Google Scholar] [PubMed]
- Murphy, M.P.; LeVine, H., 3rd. Alzheimer’s disease and the amyloid-beta peptide. J. Alzheimers Dis. 2010, 19, 311–323. [Google Scholar] [CrossRef]
- Moon, M.; Hong, H.S.; Nam, D.W.; Baik, S.H.; Song, H.; Kook, S.Y.; Kim, Y.S.; Lee, J.; Mook-Jung, I. Intracellular amyloid-beta accumulation in calcium-binding protein-deficient neurons leads to amyloid-beta plaque formation in animal model of Alzheimer’s disease. J. Alzheimers Dis. 2012, 29, 615–628. [Google Scholar] [CrossRef]
- Serrano-Pozo, A.; Frosch, M.P.; Masliah, E.; Hyman, B.T. Neuropathological alterations in Alzheimer disease. Cold Spring Harb. Perspect. Med. 2011, 1, a006189. [Google Scholar] [CrossRef]
- McGeer, E.G.; McGeer, P.L. Neuroinflammation in Alzheimer’s disease and mild cognitive impairment: A field in its infancy. J. Alzheimers Dis. 2010, 19, 355–361. [Google Scholar] [CrossRef]
- Querfurth, H.W.; LaFerla, F.M. Alzheimer’s disease. N. Engl. J. Med. 2010, 362, 329–344. [Google Scholar] [CrossRef]
- Herholz, S.C.; Herholz, R.S.; Herholz, K. Non-pharmacological interventions and neuroplasticity in early stage Alzheimer’s disease. Expert. Rev. Neurother. 2013, 13, 1235–1245. [Google Scholar] [CrossRef]
- Martorell, A.J.; Paulson, A.L.; Suk, H.J.; Abdurrob, F.; Drummond, G.T.; Guan, W.; Young, J.Z.; Kim, D.N.; Kritskiy, O.; Barker, S.J.; et al. Multi-sensory Gamma Stimulation Ameliorates Alzheimer’s-Associated Pathology and Improves Cognition. Cell 2019, 177, 256–271. [Google Scholar] [CrossRef]
- Wilson, G.D.; Marples, B. A New Use for an Old Treatment: Radiation Therapy and Alzheimer’s Disease. Radiat. Res. 2016, 185, 443–448. [Google Scholar] [CrossRef] [PubMed]
- Michael, D.B.; Wilson, G.D.; Hanna, A.; Wilson, T.G.; Martinez, A.; Chinnaiyan, P.; Maddens, M.E.; Fontanesi, J. Radiation therapy for the treatment of Alzheimer’s disease. Neurol. Neurosurg. 2019, 2. [Google Scholar] [CrossRef]
- Cuttler, J.M.; Moore, E.R.; Hosfeld, V.D.; Nadolski, D.L. Second Update on a Patient With Alzheimer Disease Treated by CT Scans. Dose Response 2018, 16, 1559325818756461. [Google Scholar] [CrossRef] [PubMed]
- Ceyzeriat, K.; Tournier, B.B.; Millet, P.; Frisoni, G.B.; Garibotto, V.; Zilli, T. Low-Dose Radiation Therapy: A New Treatment Strategy for Alzheimer’s Disease? J. Alzheimers Dis. 2020, 1–9, Preprint. [Google Scholar] [CrossRef] [PubMed]
- Almadana Pacheco, V.; Luque Crespo, E.; Wals Zurita, A.J.; Montemayor Rubio, T. External Beam Radiation Therapy: A Treatment Modality in Diffuse Tracheobronchial Amyloidosis. Arch. Bronconeumol. 2015, 51, 607–608. [Google Scholar] [CrossRef] [PubMed]
- Kalra, S.; Utz, J.P.; Edell, E.S.; Foote, R.L. External-beam radiation therapy in the treatment of diffuse tracheobronchial amyloidosis. Mayo Clin. Proc. 2001, 76, 853–856. [Google Scholar] [CrossRef]
- Liang, Y.; Li, S.; Zou, Q.; Su, B. Potential neuroprotective effect of low dose whole-body gamma-irradiation against 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-induced dopaminergic toxicity in C57 mice. Neurosci. Lett. 2006, 400, 213–217. [Google Scholar] [CrossRef]
- Kipnis, J.; Avidan, H.; Markovich, Y.; Mizrahi, T.; Hauben, E.; Prigozhina, T.B.; Slavin, S.; Schwartz, M. Low-dose gamma-irradiation promotes survival of injured neurons in the central nervous system via homeostasis-driven proliferation of T cells. Eur. J. Neurosci. 2004, 19, 1191–1198. [Google Scholar] [CrossRef]
- Cuttler, J.M.; Moore, E.R.; Hosfeld, V.D.; Nadolski, D.L. Treatment of Alzheimer Disease With CT Scans: A Case Report. Dose Response 2016, 14, 1559325816640073. [Google Scholar] [CrossRef]
- Hwang, S.; Jeong, H.; Hong, E.H.; Joo, H.M.; Cho, K.S.; Nam, S.Y. Low-dose ionizing radiation alleviates Abeta42-induced cell death via regulating AKT and p38 pathways in Drosophila Alzheimer’s disease models. Biol. Open 2019, 8, bio036657. [Google Scholar] [CrossRef]
- Marples, B.; McGee, M.; Callan, S.; Bowen, S.E.; Thibodeau, B.J.; Michael, D.B.; Wilson, G.D.; Maddens, M.E.; Fontanesi, J.; Martinez, A.A. Cranial irradiation significantly reduces beta amyloid plaques in the brain and improves cognition in a murine model of Alzheimer’s Disease (AD). Radiother. Oncol. 2016, 118, 579–580. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Hinshaw, R.G.; Le, K.X.; Park, M.A.; Wang, S.; Belanger, A.P.; Dubey, S.; Frost, J.L.; Shi, Q.; Holton, P.; et al. Space-like (56)Fe irradiation manifests mild, early sex-specific behavioral and neuropathological changes in wildtype and Alzheimer’s-like transgenic mice. Sci. Rep. 2019, 9, 12118. [Google Scholar] [CrossRef] [PubMed]
- Rudobeck, E.; Bellone, J.A.; Szucs, A.; Bonnick, K.; Mehrotra-Carter, S.; Badaut, J.; Nelson, G.A.; Hartman, R.E.; Vlkolinsky, R. Low-dose proton radiation effects in a transgenic mouse model of Alzheimer’s disease - Implications for space travel. PLoS ONE 2017, 12, e0186168. [Google Scholar] [CrossRef] [PubMed]
- Cherry, J.D.; Liu, B.; Frost, J.L.; Lemere, C.A.; Williams, J.P.; Olschowka, J.A.; O’Banion, M.K. Galactic cosmic radiation leads to cognitive impairment and increased abeta plaque accumulation in a mouse model of Alzheimer’s disease. PLoS ONE 2012, 7, e53275. [Google Scholar] [CrossRef] [PubMed]
- Wilson, G.D.; Wilson, T.G.; Hanna, A.; Kulchycki, J.; Buelow, K.; Pruetz, B.L.; Michael, D.B.; Chinnaiyan, P.; Maddens, M.E.; Martinez, A.A.; et al. Low Dose Brain Irradiation Reduces Amyloid-beta and Tau in 3xTg-AD Mice. J. Alzheimers Dis. 2020, 1–7, Preprint. [Google Scholar]
- Choi, S.Y.; Kwon, N.H.; Kim, S.T.; Kim, D.W.; Chung, M.J.; Jahng, G.H.; Chung, W. The Effect of Low Dose Radiation on Alzheimer’s Disease-Induced TG Mice. Int. J. Radiat. Oncol. Biol. Phys. 2018, 102, e210–e211. [Google Scholar] [CrossRef]
- Kempf, S.J.; Janik, D.; Barjaktarovic, Z.; Braga-Tanaka, I., 3rd; Tanaka, S.; Neff, F.; Saran, A.; Larsen, M.R.; Tapio, S. Chronic low-dose-rate ionising radiation affects the hippocampal phosphoproteome in the ApoE−/− Alzheimer’s mouse model. Oncotarget 2016, 7, 71817–71832. [Google Scholar] [CrossRef]
- Villasana, L.E.; Benice, T.S.; Raber, J. Long-term effects of 56Fe irradiation on spatial memory of mice: Role of sex and apolipoprotein E isoform. Int. J. Radiat. Oncol. Biol. Phys. 2011, 80, 567–573. [Google Scholar] [CrossRef]
- Lowe, X.R.; Bhattacharya, S.; Marchetti, F.; Wyrobek, A.J. Early brain response to low-dose radiation exposure involves molecular networks and pathways associated with cognitive functions, advanced aging and Alzheimer’s disease. Radiat. Res. 2009, 171, 53–65. [Google Scholar] [CrossRef]
- Yin, E.; Nelson, D.O.; Coleman, M.A.; Peterson, L.E.; Wyrobek, A.J. Gene expression changes in mouse brain after exposure to low-dose ionizing radiation. Int. J. Radiat. Biol. 2003, 79, 759–775. [Google Scholar] [CrossRef]
- Wang, B.; Tanaka, K.; Ji, B.; Ono, M.; Fang, Y.; Ninomiya, Y.; Maruyama, K.; Izumi-Nakajima, N.; Begum, N.; Higuchi, M.; et al. Low-dose total-body carbon-ion irradiations induce early transcriptional alteration without late Alzheimer’s disease-like pathogenesis and memory impairment in mice. J. Neurosci Res. 2014, 92, 915–926. [Google Scholar] [CrossRef] [PubMed]
- Kimura, R.; Ohno, M. Impairments in remote memory stabilization precede hippocampal synaptic and cognitive failures in 5XFAD Alzheimer mouse model. Neurobiol. Dis. 2009, 33, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Nordengen, K.; Kirsebom, B.E.; Henjum, K.; Selnes, P.; Gisladottir, B.; Wettergreen, M.; Torsetnes, S.B.; Grontvedt, G.R.; Waterloo, K.K.; Aarsland, D.; et al. Glial activation and inflammation along the Alzheimer’s disease continuum. J. Neuroinflammation 2019, 16, 46. [Google Scholar] [CrossRef] [PubMed]
- Meraz-Rios, M.A.; Toral-Rios, D.; Franco-Bocanegra, D.; Villeda-Hernandez, J.; Campos-Pena, V. Inflammatory process in Alzheimer’s Disease. Front. Integr. Neurosci. 2013, 7, 59. [Google Scholar] [CrossRef]
- Wang, W.Y.; Tan, M.S.; Yu, J.T.; Tan, L. Role of pro-inflammatory cytokines released from microglia in Alzheimer’s disease. Ann. Transl. Med. 2015, 3, 136. [Google Scholar]
- Ling, E.A.; Wong, W.C. The origin and nature of ramified and amoeboid microglia: A historical review and current concepts. Glia 1993, 7, 9–18. [Google Scholar] [CrossRef]
- Dheen, S.T.; Kaur, C.; Ling, E.A. Microglial activation and its implications in the brain diseases. Curr. Med. Chem. 2007, 14, 1189–1197. [Google Scholar] [CrossRef]
- Parakalan, R.; Jiang, B.; Nimmi, B.; Janani, M.; Jayapal, M.; Lu, J.; Tay, S.S.; Ling, E.A.; Dheen, S.T. Transcriptome analysis of amoeboid and ramified microglia isolated from the corpus callosum of rat brain. BMC Neurosci. 2012, 13, 64. [Google Scholar] [CrossRef]
- Lawrence, T.; Fong, C. The resolution of inflammation: Anti-inflammatory roles for NF-kappaB. Int. J. Biochem. Cell Biol. 2010, 42, 519–523. [Google Scholar] [CrossRef]
- Eckman, C.B.; Eckman, E.A. An update on the amyloid hypothesis. Neurol. Clin. 2007, 25, 669–682. [Google Scholar] [CrossRef][Green Version]
- Giulian, D.; Haverkamp, L.J.; Yu, J.H.; Karshin, W.; Tom, D.; Li, J.; Kirkpatrick, J.; Kuo, L.M.; Roher, A.E. Specific domains of beta-amyloid from Alzheimer plaque elicit neuron killing in human microglia. J. Neurosci. 1996, 16, 6021–6037. [Google Scholar] [CrossRef] [PubMed]
- Chang, R.; Yee, K.L.; Sumbria, R.K. Tumor necrosis factor alpha Inhibition for Alzheimer’s Disease. J. Cent. Nerv. Syst. Dis. 2017, 9, 1179573517709278. [Google Scholar] [CrossRef]
- Block, M.L.; Zecca, L.; Hong, J.S. Microglia-mediated neurotoxicity: Uncovering the molecular mechanisms. Nat. Rev. Neurosci. 2007, 8, 57–69. [Google Scholar] [CrossRef]
- Bistolfi, F. Localized amyloidosis and Alzheimer’s disease: The rationale for weekly long-term low dose amyloid-based fractionated radiotherapy. Neuroradiol. J. 2008, 21, 683–692. [Google Scholar] [CrossRef]
- Boza-Serrano, A.; Yang, Y.; Paulus, A.; Deierborg, T. Innate immune alterations are elicited in microglial cells before plaque deposition in the Alzheimer’s disease mouse model 5xFAD. Sci. Rep. 2018, 8, 1550. [Google Scholar] [CrossRef] [PubMed]
- Decourt, B.; Lahiri, D.K.; Sabbagh, M.N. Targeting Tumor Necrosis Factor Alpha for Alzheimer’s Disease. Curr. Alzheimer Res. 2017, 14, 412–425. [Google Scholar] [CrossRef] [PubMed]
- Olmos, G.; Llado, J. Tumor necrosis factor alpha: A link between neuroinflammation and excitotoxicity. Mediat. Inflamm. 2014, 2014, 861231. [Google Scholar] [CrossRef]
- McAlpine, F.E.; Tansey, M.G. Neuroinflammation and tumor necrosis factor signaling in the pathophysiology of Alzheimer’s disease. J. Inflamm. Res. 2008, 1, 29–39. [Google Scholar]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.C. NF-kappaB signaling in inflammation. Signal. Transduct Target. 2017, 2, 1–9. [Google Scholar]
- Kim, H.G.; Moon, M.; Choi, J.G.; Park, G.; Kim, A.J.; Hur, J.; Lee, K.T.; Oh, M.S. Donepezil inhibits the amyloid-beta oligomer-induced microglial activation in vitro and in vivo. Neurotoxicology 2014, 40, 23–32. [Google Scholar] [CrossRef]
- Chen, C.H.; Zhou, W.; Liu, S.; Deng, Y.; Cai, F.; Tone, M.; Tone, Y.; Tong, Y.; Song, W. Increased NF-kappaB signalling up-regulates BACE1 expression and its therapeutic potential in Alzheimer’s disease. Int. J. Neuropsychopharmacol. 2012, 15, 77–90. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, M.; Kiyota, T.; Horiba, M.; Buescher, J.L.; Walsh, S.M.; Gendelman, H.E.; Ikezu, T. Interferon-gamma and tumor necrosis factor-alpha regulate amyloid-beta plaque deposition and beta-secretase expression in Swedish mutant APP transgenic mice. Am. J. Pathol. 2007, 170, 680–692. [Google Scholar] [CrossRef] [PubMed]
- Koenigsknecht-Talboo, J.; Landreth, G.E. Microglial phagocytosis induced by fibrillar beta-amyloid and IgGs are differentially regulated by proinflammatory cytokines. J. Neurosci. 2005, 25, 8240–8249. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.; Zhou, T.; Choi, C.; Wang, Z.; Benveniste, E.N. Differential regulation and function of Fas expression on glial cells. J. Immunol. 2000, 164, 1277–1285. [Google Scholar] [CrossRef]
- Frankola, K.A.; Greig, N.H.; Luo, W.; Tweedie, D. Targeting TNF-alpha to elucidate and ameliorate neuroinflammation in neurodegenerative diseases. Cns Neurol. Disord. Drug Targets 2011, 10, 391–403. [Google Scholar] [CrossRef]
- Spanaus, K.S.; Schlapbach, R.; Fontana, A. TNF-alpha and IFN-gamma render microglia sensitive to Fas ligand-induced apoptosis by induction of Fas expression and down-regulation of Bcl-2 and Bcl-xL. Eur. J. Immunol. 1998, 28, 4398–4408. [Google Scholar] [CrossRef]
- Albrecht, U.; Yang, X.; Asselta, R.; Keitel, V.; Tenchini, M.L.; Ludwig, S.; Heinrich, P.C.; Haussinger, D.; Schaper, F.; Bode, J.G. Activation of NF-kappaB by IL-1beta blocks IL-6-induced sustained STAT3 activation and STAT3-dependent gene expression of the human gamma-fibrinogen gene. Cell Signal. 2007, 19, 1866–1878. [Google Scholar] [CrossRef]
- Mise-Omata, S.; Kuroda, E.; Niikura, J.; Yamashita, U.; Obata, Y.; Doi, T.S. A proximal kappaB site in the IL-23 p19 promoter is responsible for RelA- and c-Rel-dependent transcription. J. Immunol. 2007, 179, 6596–6603. [Google Scholar] [CrossRef]
- Liao, Y.F.; Wang, B.J.; Cheng, H.T.; Kuo, L.H.; Wolfe, M.S. Tumor necrosis factor-alpha, interleukin-1beta, and interferon-gamma stimulate gamma-secretase-mediated cleavage of amyloid precursor protein through a JNK-dependent MAPK pathway. J. Biol. Chem. 2004, 279, 49523–49532. [Google Scholar] [CrossRef]
- Kaltschmidt, B.; Sparna, T.; Kaltschmidt, C. Activation of NF-kappa B by reactive oxygen intermediates in the nervous system. Antioxid. Redox Signal. 1999, 1, 129–144. [Google Scholar] [CrossRef]
- Fillit, H.; Ding, W.H.; Buee, L.; Kalman, J.; Altstiel, L.; Lawlor, B.; Wolf-Klein, G. Elevated circulating tumor necrosis factor levels in Alzheimer’s disease. Neurosci. Lett. 1991, 129, 318–320. [Google Scholar] [CrossRef]
- Bevelacqua, J.J.; Mortazavi, S.M.J. Alzheimer ‘s Disease: Possible Mechanisms Behind Neurohormesis Induced by Exposure to Low Doses of Ionizing Radiation. J. Biomed. Phys. Eng. 2018, 8, 153–156. [Google Scholar] [CrossRef] [PubMed]
- Franklin, K.; Paxinos, G. The Mouse Brain in Stereotaxic Coordinates, 4th ed.; Gulf Professional Publishing: Houston, TX, USA, 2012. [Google Scholar]
- Young, K.; Morrison, H. Quantifying Microglia Morphology from Photomicrographs of Immunohistochemistry Prepared Tissue Using ImageJ. J. Vis. Exp. 2018, 136, e57648. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, S.; Nam, Y.; Kim, C.; Lee, H.; Hong, S.; Kim, H.S.; Shin, S.J.; Park, Y.H.; Mai, H.N.; Oh, S.-M.; et al. Neuroprotective and Anti-Inflammatory Effects of Low–Moderate Dose Ionizing Radiation in Models of Alzheimer’s Disease. Int. J. Mol. Sci. 2020, 21, 3678. https://doi.org/10.3390/ijms21103678
Kim S, Nam Y, Kim C, Lee H, Hong S, Kim HS, Shin SJ, Park YH, Mai HN, Oh S-M, et al. Neuroprotective and Anti-Inflammatory Effects of Low–Moderate Dose Ionizing Radiation in Models of Alzheimer’s Disease. International Journal of Molecular Sciences. 2020; 21(10):3678. https://doi.org/10.3390/ijms21103678
Chicago/Turabian StyleKim, Sujin, Yunkwon Nam, Chanyang Kim, Hyewon Lee, Seojin Hong, Hyeon Soo Kim, Soo Jung Shin, Yong Ho Park, Han Ngoc Mai, Sang-Muk Oh, and et al. 2020. "Neuroprotective and Anti-Inflammatory Effects of Low–Moderate Dose Ionizing Radiation in Models of Alzheimer’s Disease" International Journal of Molecular Sciences 21, no. 10: 3678. https://doi.org/10.3390/ijms21103678
APA StyleKim, S., Nam, Y., Kim, C., Lee, H., Hong, S., Kim, H. S., Shin, S. J., Park, Y. H., Mai, H. N., Oh, S.-M., Kim, K. S., Yoo, D.-H., Chung, W. K., Chung, H., & Moon, M. (2020). Neuroprotective and Anti-Inflammatory Effects of Low–Moderate Dose Ionizing Radiation in Models of Alzheimer’s Disease. International Journal of Molecular Sciences, 21(10), 3678. https://doi.org/10.3390/ijms21103678




