Synthetic Strategies and Computational Inhibition Activity Study for Triazinyl-Substituted Benzenesulfonamide Conjugates with Polar and Hydrophobic Amino Acids as Inhibitors of Carbonic Anhydrases
Abstract
1. Introduction
2. Results and Discussion
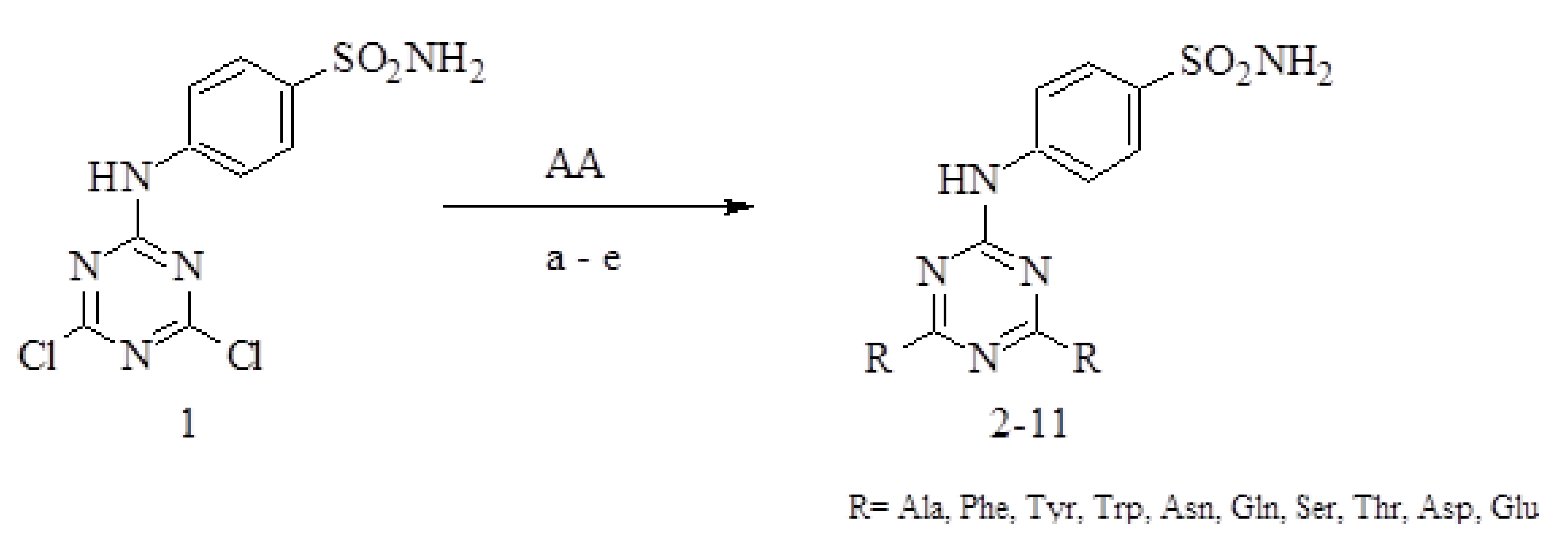
2.1. Chemistry
2.2. QSAR Study of Inhibition Activities of Sulfonamide Derivatives against hCA (I, II, IV, IX) Isoforms
2.2.1. Data Set
2.2.2. Molecular Optimization and Descriptor Calculation
2.2.3. QSAR Modelling
2.2.4. Application of Artificial Neural Networks
2.2.5. Evaluation of Predicted Inhibition Activities of New Sulfonamide Derivatives
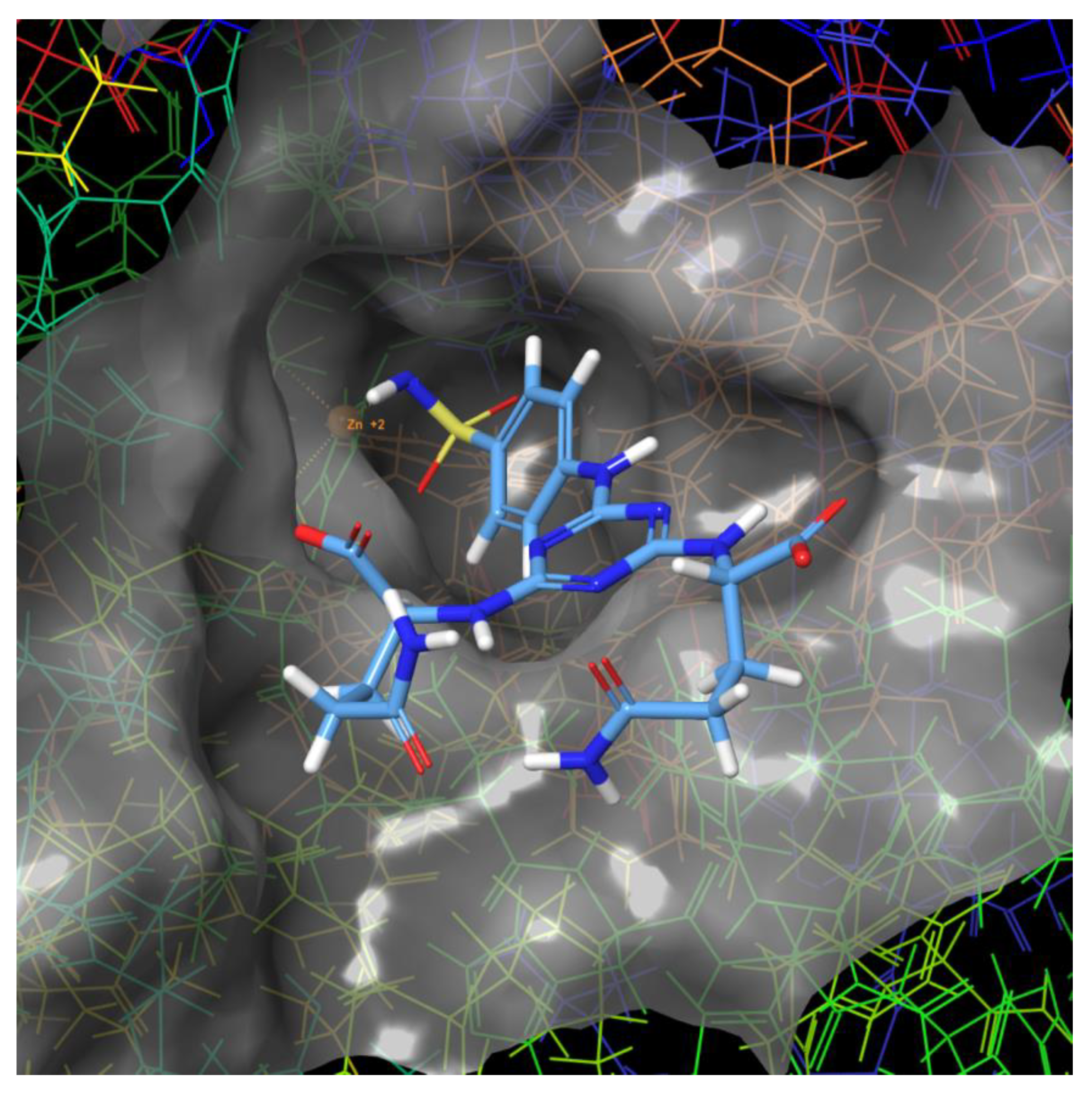
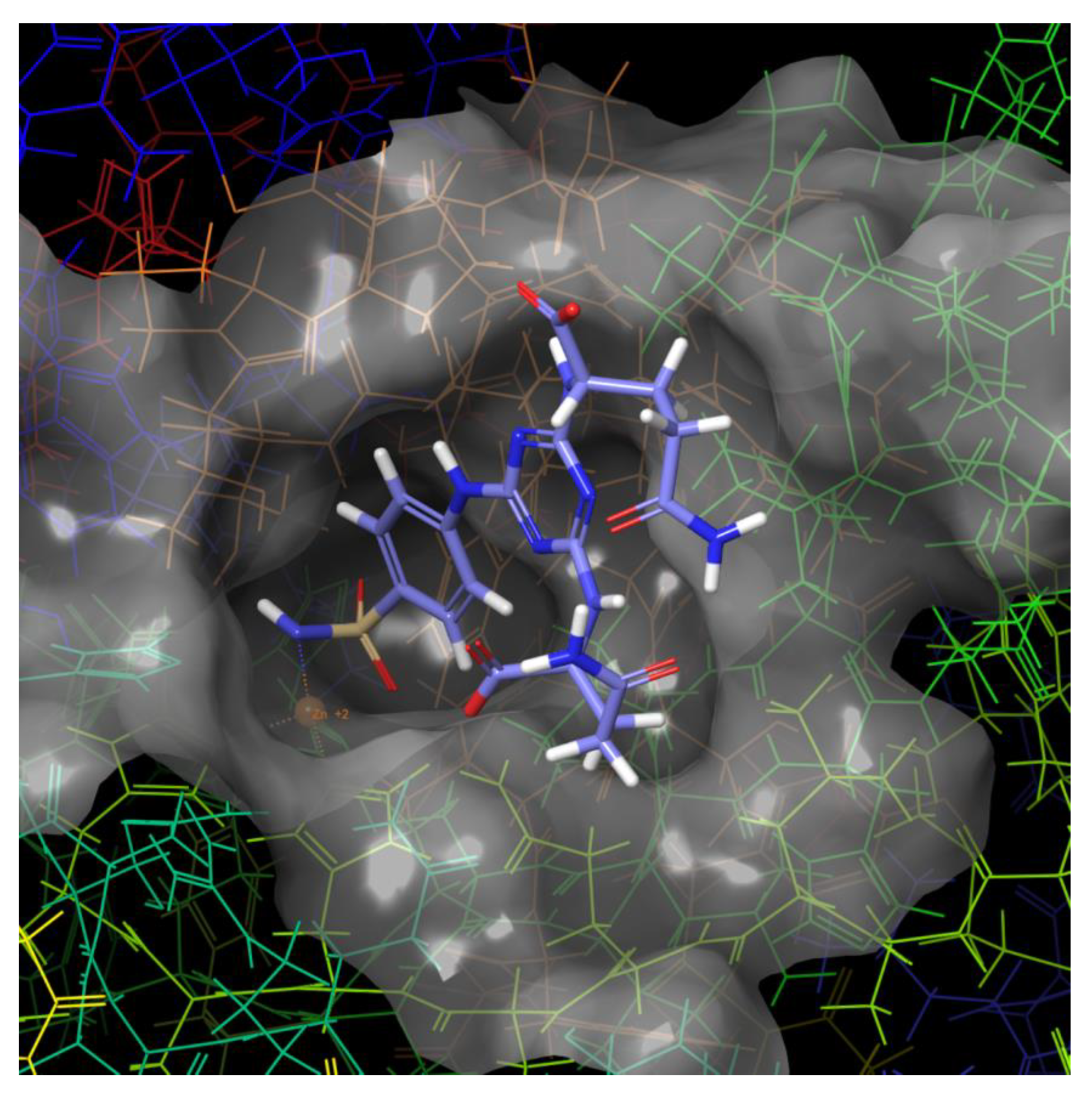
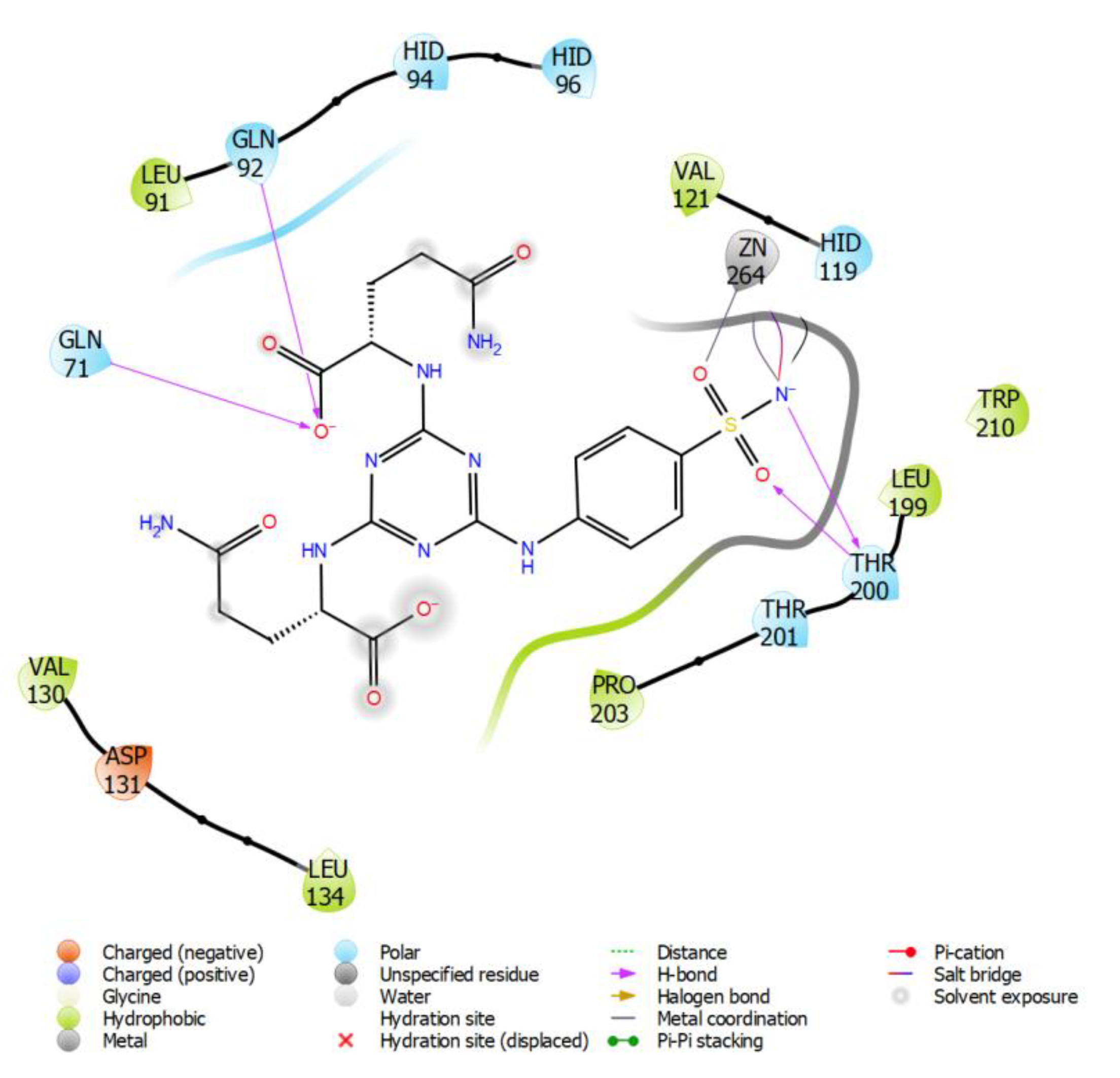
2.3. Molecular Modeling
3. Materials and Methods
3.1. Materials and Instruments for the Synthesis and Analysis
3.2. Synthetic Procedures and Products Characterization
3.2.1. General Methods for the Synthesis of Disubstituted Derivatives of 4-(4′,6′-dichloro-1′,3′,5′-triazine-2′-ylamino)benzenesulfonamide 2–11
3.2.2. Pure Products Characterization
3.3. Program and Software for the Computational Study of Sulfonamide Derivatives
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| hCA | human carbonic anhydrase |
| hCAi | human carbonic anhydrase inhibitors |
| PG | proteoglycan |
| HIF | hypoxia inducible factor |
| VHL | Von Hippel-Lindau (tumor suppressor) |
| QSAR | Quantitative structure-activity relationship |
| MMFF | Molecular mechanics force field |
| ANN | Artificial neural networks |
| 3-MLP | 3-layers of Multilayer perceptron neural networks |
| RMSE | root mean squared error |
| TSA | triazinyl-aminobenzenesulfonamide derivative |
| TSAM | triazinyl-aminomethylbenzenesulfonamide derivative |
| TSAE | triazinyl-aminoethylbenzenesulfonamide derivative |
References
- Rezuchova, I.; Pastorek, J. Carbonic anhydrase IX and hypoxia. In Carbonic Anhydrases: Biochemistry, Mechanism of Action and Therapeutic Applications; Penttinen, J., Ed.; Nova Science Publishers: New York, NY, USA, 2018; pp. 1–69. [Google Scholar]
- Pastorek, J.; Pastoreková, S.; Callebaut, I.; Mornon, J.P.; Zelník, V.; Opavský, R.; Zaťovičová, M.; Liao, S.; Portelle, D.; Stanbridge, E.J. Cloning and characterization of MN, a human tumor-associated protein with a domain homologous to carbonic anhydrase and putative helix-loop-helix DNA binding segment. Oncogene 1994, 9, 2877–2888. [Google Scholar]
- Pastoreková, S.; Gillies, R.J. The role of carbonic anhydrase IX in cancer development: Links to hypoxia, acidosis, and beyond. Cancer Metast. Rev. 2019, 38, 65–77. [Google Scholar] [CrossRef]
- Švastová, E.; Zilka, N.; Zaťovičová, M.; Gibadulinová, A.; Ciampor, F.; Pastorek, J.; Pastoreková, S. Carbonic anhydrase IX reduces E-cadherin-mediated adhesion of MDCK cells via interaction with beta-catenin. Exp. Cell. Res. 2003, 290, 332–345. [Google Scholar] [CrossRef]
- Iwasaki, K.; Yabushita, H.; Ueno, T.; Wakatsuki, A. Role of hypoxia-inducible factor-1α, carbonic anhydrase-IX, glucose transporter-1 and vascular endothelial growth factor associated with lymph node metastasis and recurrence in patients with locally advanced cervical cancer. Oncol. Lett. 2015, 10, 1970–1978. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Supuran, C.T. Carbonic Anhydrase Inhibition and the Management of Hypoxic Tumors. Metabolites 2017, 7, 48. [Google Scholar] [CrossRef] [PubMed]
- Wykoff, C.C.; Beasley, N.; Watson, P.H.; Campo, L.; Chia, S.K.; English, R.; Pastorek, J.; Sly, W.S.; Ratcliffe, P.; Harris, A.L. Expression of the hypoxia-inducible and tumor-associated carbonic anhydrases in ductal carcinoma in situ of the breast. Am. J. Pathol. 2001, 158, 1011–1019. [Google Scholar] [CrossRef]
- Beasley, N.J.P.; Cox, G.J.; Wykoff, C.C.; Watson, P.H.; Leek, R.; Turley, H.; Harris, A.L.; Gatter, K.; Pastorek, J.; Ratcliffe, P. Carbonic anhydrase IX, an endogenous hypoxia marker, expression in head and neck squamous cell carcinoma and its relationship to hypoxia, necrosis, and microvessel density. Cancer Res. 2001, 61, 5262–5267. [Google Scholar] [PubMed]
- Giatromanolaki, A.; Koukourakis, M.I.; Sivridis, E.; Pastorek, J.; Wykoff, C.C.; Gatter, K.C.; Harris, A.L. Expression of hypoxia-inducible carbonic anhydrase-9 relates to angiogenic pathways and independently to poor outcome in non-small cell lung cancer. Cancer Res. 2001, 61, 7992–7998. [Google Scholar]
- Hynninen, P.; Vaskivuo, L.; Saarnio, J.; Haapasalo, H.; Kivelä, J.; Pastoreková, S.; Pastorek, J.; Waheed, A.; Sly, W.S.; Puistola, U.; et al. Expression of transmembrane carbonic anhydrases IX and XII in ovarian tumours. Histopathology 2006, 49, 594–602. [Google Scholar] [CrossRef]
- Turner, K.J.; Crew, J.P.; Wykoff, C.C.; Watson, P.H.; Poulsom, R.; Pastorek, J.; Ratcliffe, P.J.; Cranston, D.; Harris, A.L. The hypoxia-inducible genes VEGF and CA9 are differentially regulated in superficial vs invasive bladder cancer. Brit. J. Cancer 2002, 86, 1276–1282. [Google Scholar] [CrossRef]
- Loncaster, J.A.; Harris, A.L.; Davidson, S.E.; Logue, J.P.; Hunter, R.D.; Wycoff, C.C.; Pastorek, J.; Ratcliffe, P.J.; Stratford, I.J.; West, C.M.L. Carbonic anhydrase (CA IX) expression, a potential new intrinsic marker of hypoxia: Correlations with tumor oxygen measurements and prognosis in locally advanced carcinoma of the cervix. Cancer Res. 2001, 61, 6394–6399. [Google Scholar] [PubMed]
- Hui, E.P.; Chan, A.T.C.; Pezzella, F.; Turley, H.; To, K.-F.; Poon, T.C.W.; Zee, B.; Mo, F.; Teo, P.M.L.; Huang, D.P.; et al. Coexpression of hypoxia-inducible factors 1α and 2α, carbonic anhydrase IX, and vascular endothelial growth factor in nasopharyngeal carcinoma and relationship to survival. Clin. Cancer Res. 2002, 8, 2595–2604. [Google Scholar] [PubMed]
- Proescholdt, M.A.; Mayer, C.; Kubitza, M.; Schubert, T.; Liao, S.-Y.; Stanbridge, E.J.; Ivanov, S.; Oldfield, E.H.; Brawanski, A.; Merrill, M.J. Expression of hypoxia-inducible carbonic anhydrases in brain tumors. Neuro-Oncol. 2005, 7, 465–475. [Google Scholar] [CrossRef] [PubMed]
- Van Laarhoven, H.W.M.; Kaanders, J.H.A.M.; Lok, J.; Peeters, W.J.M.; Rijken, P.F.J.W.; Wiering, B.; Ruers, T.J.M.; Punt, C.J.A.; Heerschap, A.; Van Der Kogel, A.J. Hypoxia in relation to vasculature and proliferation in liver metastases in patients with colorectal cancer. Int. J. Radiat. Oncol. Biol. Phys. 2006, 64, 473–482. [Google Scholar] [CrossRef]
- Wiesener, M.S.; Münchenhagen, P.M.; Berger, I.; Morgan, N.V.; Roigas, J.; Schwiertz, A.; Jürgensen, J.S.; Gruber, G.; Maxwell, P.H.; Löning, S.A.; et al. Constitutive activation of hypoxia-inducible genes related to overexpression of hypoxia-inducible factor-1alpha in clear cell renal carcinomas. Cancer Res. 2001, 61, 5215–5222. [Google Scholar]
- Tripodi, S.A.; Del Vecchio, M.T.; Supuran, C.T.; Scozzafava, A.; Gabrielli, M.G.; Pastorekova, S.; Rossi, R.; Fasolis, G.; Puccetti, L. Redox state and carbonic anhydrase isozyme IX expression in human renal cell carcinoma: Biochemical and morphological investigations. J. Enzyme Inhib. Med. Chem. 2004, 19, 287–291. [Google Scholar] [CrossRef]
- Pastoreková, S.; Parkkila, S.; Parkkila, A.K.; Opavsky, R.; Zelnik, V.; Saarnio, J.; Pastorek, J. Carbonic anhydrase IX, MN/CA IX: Analysis of stomach complementary DNA sequence and expression in human and rat alimentary tracts. Gastroenterology 1997, 112, 398–408. [Google Scholar] [CrossRef]
- Supuran, C.T. Carbonic anhydrase inhibitors as emerging agents for the treatment and imaging of hypoxic tumors. Expert Opin. Investig. Drugs 2018, 27, 963–970. [Google Scholar] [CrossRef]
- Pastorek, J.; Pastoreková, S. Hypoxia-induced carbonic anhydrase IX as a target for cancer therapy: From biology to clinical use. Semin. Cancer Biol. 2015, 31, 52–64. [Google Scholar] [CrossRef]
- El-Azab, A.S.; Abdel-Aziz, A.A.M.; Bua, S.; Nocentini, A.; ALSaif, N.A.; Almehizia, A.A.; Alanazi, M.M.; Hefnawy, M.M.; Supuran, C.T. New anthranilic acid-incorporating N-benzenesulfonamidophthalimides as potent inhibitors of carbonic anhydrases I, II, IX, and XII: Synthesis, in vitro testing, and in silico assessment. Eur. J. Med. Chem. 2019, 181, 111573. [Google Scholar] [CrossRef]
- Eldehna, W.M.; Fares, M.; Ceruso, M.; Ghabbour, H.A.; Abou-Seri, S.M.; Abdel-Aziz, H.A.; Abou El Ella, D.A.; Supuran, C.T. Amido/ureidosubstituted benzenesulfonamides-isatin conjugates as low nanomolar/subnanomolar inhibitors of the tumor-associated carbonic anhydrase isoform XII. Eur. J. Med. Chem. 2016, 110, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Ceruso, M.; Antel, S.; Scozzafava, A.; Supuran, C.T. Synthesis and inhibition potency of novel ureido benzenesulfonamides incorporating GABA as tumor-associated carbonic anhydrase IX and XII inhibitors. J. Enzyme Inhib. Med. Chem. 2016, 31, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Abdel Gawad, N.M.; Amin, N.H.; Elsaadi, M.T.; Mohamed, F.M.M.; Angeli, A.; De Luca, V.; Capasso, C.; Supuran, C.T. Synthesis of 4-(thiazol-2-ylamino)-benzenesulfonamides with carbonic anhydrase I, II and IX inhibitory activity and cytotoxic effects against breast cancer cell lines. Bioorg. Med. Chem. 2016, 24, 3043–3051. [Google Scholar] [CrossRef] [PubMed]
- Chandak, N.; Ceruso, M.; Supuran, C.T.; Sharma, P.K. Novel sulfonamide bearing coumarin scaffolds as selective inhibitors of tumor associated carbonic anhydrase isoforms IX and XII. Bioorg. Med. Chem. 2016, 24, 2882–2886. [Google Scholar] [CrossRef]
- Nocentini, A.; Ferraroni, M.; Carta, F.; Ceruso, M.; Gratteri, P.; Lanzi, C.; Masini, E.; Supuran, C.T. Benzenesulfonamides Incorporating Flexible Triazole Moieties Are Highly Effective Carbonic Anhydrase Inhibitors: Synthesis and Kinetic, Crystallographic, Computational, and Intraocular Pressure Lowering Investigations. J. Med. Chem. 2016, 59, 10692–10704. [Google Scholar] [CrossRef]
- Kumar, R.; Sharma, V.; Bua, S.; Supuran, C.T.; Sharma, P.K. Synthesis and biological evaluation of benzenesulphonamide-bearing 1,4,5-trisubstituted-1,2,3-triazoles possessing human carbonic anhydrase I, II, IV, and IX inhibitory activity. J. Enzyme Inhib. Med. Chem. 2017, 32, 1187–1194. [Google Scholar] [CrossRef]
- Angeli, A.; Tanini, D.; Capperucci, A.; Supuran, C.T. Synthesis of Novel Selenides Bearing Benzenesulfonamide Moieties as Carbonic Anhydrase I, II, IV, VII, and IX Inhibitors. ACS Med. Chem. Lett. 2017, 8, 1213–1217. [Google Scholar] [CrossRef]
- Krasavin, M.; Shetnev, A.; Sharonova, T.; Baykov, S.; Tuccinardi, T.; Kalinin, S.; Angeli, A.; Supuran, C.T. Heterocyclic periphery in the design of carbonic anhydrase inhibitors: 1,2,4-Oxadiazol-5-yl benzenesulfonamides as potent and selective inhibitors of cytosolic hCA II and membrane-bound hCA IX isoforms. Bioorg. Chem. 2018, 76, 88–97. [Google Scholar] [CrossRef]
- Krasavin, M.; Shetnev, A.; Baykov, S.; Kalinin, S.; Nocentini, A.; Sharoyko, V.; Poli, G.; Tuccinardi, T.; Korsakov, M.; Tennikova, T.B.; et al. Pyridazinone-substituted benzenesulfonamides display potent inhibition of membrane-bound human carbonic anhydrase IX and promising antiproliferative activity against cancer cell lines. Eur. J. Med. Chem. 2019, 168, 301–314. [Google Scholar] [CrossRef]
- Winum, J.-Y.; Vullo, D.; Casini, A.; Montero, J.-L.; Scozzafava, A.; Supuran, C.T. Carbonic Anhydrase Inhibitors: Inhibition of Transmembrane, Tumor-Associated Isozyme IX, and Cytosolic Isozymes I and II with Aliphatic Sulfamates. J. Med. Chem. 2003, 46, 5471–5477. [Google Scholar] [CrossRef]
- Garaj, V.; Puccetti, L.; Fasolis, G.; Winum, J.-Y.; Montero, J.-L.; Scozzafava, A.; Vullo, D.; Innocenti, A.; Supuran, C.T. Carbonic anhydrase inhibitors: Synthesis and inhibition of cytosolic/tumor-associated carbonic anhydrase isozymes I, II, and IX with sulfonamides incorporating 1,2,4-triazine moieties. Bioorg. Med. Chem. Lett. 2004, 14, 5427–5433. [Google Scholar] [CrossRef] [PubMed]
- Winum, J.-Y.; Dogné, J.-M.; Casini, A.; De Leval, X.; Montero, J.-L.; Scozzafava, A.; Vullo, D.; Innocenti, A.; Supuran, C.T. Carbonic anhydrase inhibitors: Synthesis and inhibition of cytosolic/membrane-associated carbonic anhydrase isozymes I, II, and IX with sulfonamides incorporating hydrazino moieties. J. Med. Chem. 2005, 48, 2121–2125. [Google Scholar] [CrossRef] [PubMed]
- Cecchi, A.; Hulikova, A.; Pastorek, J.; Pastoreková, S.; Scozzafava, A.; Winum, J.Y.; Montero, J.L.; Supuran, C.T. Carbonic Anhydrase Inhibitors. Design of Fluorescent Sulfonamides as Probes of Tumor-Associated Carbonic Anhydrase IX That Inhibit Isozyme IX-Mediated Acidification of Hypoxic Tumors. J. Med. Chem. 2005, 48, 4834–4841. [Google Scholar] [CrossRef] [PubMed]
- Carta, F.; Garaj, V.; Maresca, A.; Wagner, J.; Avvaru, B.S.; Robbins, A.H.; Scozzafava, A.; McKenna, R.; Supuran, C.T. Sulfonamides incorporating 1,3,5-triazine moieties selectively and potently inhibit carbonic anhydrase transmembrane isoforms IX, XII and XIV over cytosolic isoforms I and II: Solution and X-ray crystallographic studies. Bioorg. Med. Chem. 2011, 19, 3105–3119. [Google Scholar] [CrossRef] [PubMed]
- Garaj, V.; Puccetti, L.; Fasolis, G.; Winum, J.Y.; Montero, J.L.; Scozzafava, A.; Vullo, D.; Innocenti, A.; Supuran, C.T. Carbonic anhydrase inhibitors: Novel sulfonamides incorporating 1,3,5-triazine moieties as inhibitors of the cytosolic and tumour-associated carbonic anhydrase isozymes I, II and IX. Bioorg. Med. Chem. Lett. 2005, 15, 3102–3108. [Google Scholar] [CrossRef]
- Ceruso, M.; Vullo, D.; Scozzafava, A.; Supuran, C.T. Inhibition of human carbonic anhydrase isoforms I-XIV with sulfonamides incorporating fluorine and 1,3,5-triazine moieties. Bioorg. Med. Chem. 2013, 21, 6929–6936. [Google Scholar] [CrossRef]
- Saluja, A.K.; Tiwari, M.; Vullo, D.; Supuran, C.T. Substituted benzene sulfonamides incorporating 1,3,5-triazinyl moieties potently inhibit human carbonic anhydrases II, IX and XII. Bioorg. Med. Chem. Lett. 2014, 24, 1310–1314. [Google Scholar] [CrossRef]
- Lolak, N.; Akocak, S.; Bua, S.; Sanku, R.K.K.; Supuran, C.T. Discovery of new ureido benzenesulfonamides incorporating 1,3,5-triazine moieties as carbonic anhydrase I, II, IX and XII inhibitors. Bioorg. Med. Chem. 2019, 27, 1588–1594. [Google Scholar] [CrossRef]
- Havránková, E.; Csollei, J.; Vullo, D.; Garaj, V.; Pazdera, P.; Supuran, C.T. Novel sulfonamide incorporating piperazine, aminoalcohol and 1,3,5-triazine structural motifs with carbonic anhydrase I, II and IX inhibitory action. Bioorg. Chem. 2018, 77, 25–37. [Google Scholar] [CrossRef]
- Havránková, E.; Csöllei, J.; Pazdera, P. New Approach for the One-Pot Synthesis of 1,3,5-Triazine Derivatives: Application of Cu(I) Supported on a Weakly Acidic Cation-Exchanger Resin in a Comparative Study. Molecules 2019, 24, 3586. [Google Scholar] [CrossRef]
- Krajčiová, D.; Pecher, D.; Garaj, V.; Mikuš, P. Optimization and Comparison of Synthetic Procedures for a Group of Triazinyl-substituted Benzene-Sulfonamides Conjugates with Amino Acids. Molecules 2017, 22, 1533. [Google Scholar] [CrossRef]
- Mikuš, P.; Krajčiová, D.; Mikulová, M.; Horváth, B.; Pecher, D.; Garaj, V.; Bua, S.; Angeli, A.; Supuran, T. Novel sulfonamides incorporating 1,3,5-triazine and amino acid structural motifs as inhibitors of the physiological carbonic anhydrase isozymes I, II and IV and tumor-associated isozyme IX. Bioorg. Chem. 2018, 81, 241–252. [Google Scholar] [CrossRef] [PubMed]
- Wei, D.; Zhang, A.; Wu, C.; Han, S.; Wang, L. Progressive study and robustness test of QSAR model based on quantum chemical parameters for predicting BCF of selected polychlorinated organic compounds (PCOCs). Chemosphere 2001, 44, 1421–1428. [Google Scholar] [CrossRef]
- Wold, S.; Eriksson, L. Statistical Validation of QSAR Results. In Chemometric Methods in Molecular Design; Waterbeemd, H., Ed.; VCH Verlagsgesellschaft mbH: Weinheim, Germany, 1995; p. 379. [Google Scholar]
- ChemSketch 2018; Advanced Chemistry Development, Inc.: Toronto, ON, Canada, 2019.
- Spartan ’14; Wavefunction, Inc.: Irvine, CA, USA, 2013.
- Dragon 6; Talete Srl: Milano, Italy, 2010.
- Statistica 12; TIBCO Software, Inc.: Palo Alto, CA, USA, 2013.
- Angeli, A.; Tanini, D.; Capperucci, A.; Malevolti, G.; Turco, F.; Ferrraroni, M.; Supuran, C.T. Synthesis of different thio-scaffolds bearing sulfonamide with subnanomolar carbonic anhydrase II and IX inhibitory properties and X-ray investigations for their inhibitory mechanism. Bioorg. Chem. 2018, 81, 642–648. [Google Scholar] [CrossRef] [PubMed]
- Salerno, S.; Barresi, E.; Amendola, G.; Berrino, E.; Milite, C.; Marini, A.M.; Da Settimo, F.; Novellino, E.; Supuran, C.T.; Cosconati, S.; et al. 4-Substituted Benzenesulfonamides Incorporating Bi/Tricyclic Moieties Act as Potent and Isoform-Selective Carbonic Anhydrase II/IX Inhibitors. J. Med. Chem. 2018, 61, 5765–5770. [Google Scholar] [CrossRef] [PubMed]
- Alafeefy, A.M.; Isik, S.; Abdel-Aziz, H.A.; Ashour, A.E.; Vullo, D.; Al-Jaber, N.A.; Supuran, C.T. Carbonic anhydrase inhibitors: Benzenesulfonamides incorporating cyanoacrylamide moieties are low nanomolar/subnanomolar inhibitors of the tumor-associated isoforms IX and XII. Bioorg. Med. Chem. 2013, 21, 1396–1403. [Google Scholar] [CrossRef] [PubMed]
- Eldehna, W.M.; Abo-Ashour, M.F.; Berrino, E.; Vullo, D.; Ghabbour, H.A.; Al-Rashood, S.T.; Hassan, G.S.; Alkahtani, H.M.; Almehizia, A.A.; Alharbi, A.; et al. SLC-0111 enaminone analogs, 3/4-(3-aryl-3-oxopropenyl) aminobenzenesulfonamides, as novel selective subnanomolar inhibitors of the tumor-associated carbonic anhydrase isoform IX. Bioorg. Chem. 2019, 83, 549–558. [Google Scholar] [CrossRef] [PubMed]
- Krasavin, M.; Shetnev, A.; Sharonova, T.; Baykov, S.; Kalinin, S.; Nocentini, A.; Sharoyko, V.; Poli, G.; Tuccinardi, T.; Presnukhina, S.; et al. Continued exploration of 1,2,4-oxadiazole periphery for carbonic anhydrase-targeting primary arene sulfonamides: Discovery of subnanomolar inhibitors of membrane-bound hCA IX isoform that selectively kill cancer cells in hypoxic environment. Eur. J. Med. Chem. 2019, 164, 92–105. [Google Scholar] [CrossRef]
- Schrödinger Release 2019-4: Maestro; Schrödinger, LLC: New York, NY, USA, 2019.
- Schrödinger Release 2019-4: LigPrep; Schrödinger, LLC: New York, NY, USA, 2019.
- Schrödinger Release 2019-4: Protein Preparation Wizard; Epik, Schrödinger, LLC: New York, NY, USA, 2016.
- Schrödinger Release 2019-4: MacroModel; Schrödinger, LLC: New York, NY, USA, 2019.
- MS Office 2007; Microsoft Corp.: Redmond, WA, USA, 2007.

| (a)-the products with hydrophobic amino acids | ||||
| Product (Mr) | Parameter | Na2CO3 | NaHCO3 | TEA |
| 2 Ala | yield of crude product (%) | 99 | 73 | 48 |
| (425.11 g/mol) | RP-HPLC-UV purity (%) | 92.55 | 93.34 | 92.18 |
| MS [M + H]+ found | 426.1196 (7.25 min) | 426.1213 (7.29 min) | 426.1210 (7.27 min) | |
| disubst. AA + OH (%) | x | x | 1.19 | |
| 3 Phe | yield of crude product (%) | 94 | 94 | 12 |
| (577.17g/mol) | RP-HPLC-UV purity (%) | 96.81 | 97.19 | 20.43 |
| MS [M + H]+ found | 578.1829 (4.38 min) | 578.1831 (4.39 min) | 578.1862 (4.36 min) | |
| 4 Tyr | yield of crude product (%) | 87 | 77 | 1.7 |
| (609.16 g/mol) | RP-HPLC-UV purity (%) | 32.72 | 27.47 | 10.57 |
| MS [M + H]+ found | 610.1727 (7.04 min) | 610.1721 (7.04 min) | 610.1766 (7.03 min) | |
| disubst. AA + OH (%) | 32.23 | 1.06 | x | |
| disubst. OH + OH (%) | 2.7 | x | 1.22 | |
| unreacted initial compounds (%) | x | 23.34 (182.0812) | 15.46 (182.0813) | |
| 5 Trp | yield of crude product (%) | 94 | 96 | 70 |
| (655.19 g/mol) | RP-HPLC-UV purity (%) | 93.12 | 87.44 | 77.82 |
| MS [M + H]+ found | 656.2051 (5.5 min) | 656.2046 (5.51 min) | 656.2050 (5.51 min) | |
| disubst. AA + OH (%) | x | 1.78 | x | |
| disubst. OH + OH (%) | x | 0.79 | x | |
| unreacted initial compounds (%) | 1.63 (205.0793) | 8.94 (205.0975) | x | |
| (b)-the products with polar uncharged amino acids | ||||
| Product (Mr) | Parameter | Na2CO3 | NaHCO3 | TEA |
| 6 Asn | yield of crude product (%) | 97 | 71 | 9.0 |
| (511.12 g/mol) | RP-HPLC-UV purity (%) | 49.66 | 88.47 | 16.31 |
| MS [M + H]+ found | 512.1320 (9.63 min) | 512.1320 (9.63 min) | 512.1340 (9.65 min) | |
| disubst. AA + OH (%) | 0.42 | 1.33 | 23.45 | |
| monosubst. AA (%) | x | x | 26.29 | |
| disubst. OH + OH (%) | 1.30 | x | 2.20 | |
| 7 Gln | yield of crude product (%) | 78 | 68 | 18 |
| (539.15 g/mol) | RP-HPLC-UV purity (%) | 21.58 | 58.04 | 55.23 |
| MS [M + H]+ found | 540.1635 (9 min) | 540.1634 (9.01 min) | 540.1650 (9.03 min) | |
| disubst. AA + OH (%) | 9.42 | 3.41 | 2.64 | |
| monosubst. AA (%) | 4.91 | 19.76 | 3.42 | |
| disubst. OH + OH (%) | 1.17 | x | x | |
| 8 Ser | yield of crude product (%) | 73 | 82 | 41 |
| (457.42 g/mol) | RP-HPLC-UV purity (%) | 87.09 | 94.86 | 4.08 |
| MS [M + H]+ found | 458.1095 (9.13 min) | 458.1095 (9.13 min) | 458.1092 (9.13 min) | |
| disubst. AA + OH (%) | 3.71 | 2.23 | 50.33 | |
| monosubst. AA (%) | x | x | 7.91 | |
| disubst. OH + OH (%) | x | x | 7.27 | |
| 9 Thr | yield of crude product (%) | 93 | 92 | 14 |
| (485.37 g/mol) | RP-HPLC-UV purity (%) | 94.42 | 96.56 | 20.93 |
| MS [M + H]+ found | 486.1411 (7.7 min) | 486.1414 (7.71 min) | 486.1415 (7.73 min) | |
| disubst. AA + OH (%) | 1.50 | 1.43 | 22.17 | |
| disubst. OH + OH (%) | x | x | 4.25 | |
| (c)-the products with acidic (negatively charged) amino acids | ||||
| Product (Mr) | Parameter | Na2CO3 | NaHCO3 | TEA |
| 10 Asp (513.09 g/mol) | yield of crude product (%) | 90 | 46 | 16 |
| RP-HPLC-UV purity (%) | 93.07 | 17.57 | 37.68 | |
| MS [M + H]+ found | 514.1003 (10.71 min) | 514.1000 (10.74 min) | 514.1023 (10.75 min) | |
| disubst. AA + OH (%) | 5.37 | 21.96 | 6.92 | |
| monosubst. AA (%) | x | 50.99 | x | |
| disubst. OH + OH (%) | 0.32 | 1.36 | 9.99 | |
| 11 Glu (541.12 g/mol) | yield of crude product (%) | 75 | 69 | 38 |
| RP-HPLC-UV purity (%) | 89.53 | 16.68 | 58.00 | |
| MS [M + H]+ found | 542.1313 (10.76 min) | 542.1310 (10.77 min) | 542.1340 (10.77 min) | |
| disubst. AA + OH (%) | 3.70 | 19.26 | 10.07 | |
| monosubst. AA (%) | x | 50.04 | x | |
| disubst. OH + OH (%) | x | 1.84 | 3.40 | |
| ANN | hCA I | hCA II | hCA IV | hCA IX | |
|---|---|---|---|---|---|
| Architecture * | 3-MLP | 30-17-1 | 30-13-1 | 30-19-1 | 30-12-1 |
| Activation function | Hidden layer | tanh | tanh | tanh | tanh |
| Output layer | exp | log | log | log | |
| Training set | Performance ** | 0.9643 | 0.9972 | 0.9994 | 0.9927 |
| Error *** | 0.0039 | 0.0003 | 0.0001 | 0.0011 | |
| Validation set | Performance ** | 0.9411 | 0.9531 | 0.9441 | 0.9748 |
| Error *** | 0.0111 | 0.0064 | 0.0097 | 0.0066 | |
| Name | Linker | Model | hCA I (KI, nM) | hCA II (KI, nM) | hCA IV (KI, nM) | hCA IX (KI, nM) | hCA II/IX | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Measured | Predicted | Measured | Predicted | Measured | Predicted | Measured | Predicted | Measured | Predicted | |||
| TSA Asp | 0 | Train | 8259.8 | 8257.0 | 6219.1 | 6201.2 | 10000.0 | 10000.0 | 211.1 | 135.0 | 29.5 | 45.9 |
| TSAM Asp | 1 | Train | 3700.0 | 3682.6 | 5953.7 | 5693.6 | 10000.0 | 9999.6 | 2364.1 | 2159.9 | 2.5 | 2.6 |
| TSAE Asp | 2 | Validation | 10000.0 | 9866.0 | 5519.6 | 5931.6 | 10000.0 | 10000.0 | 25.8 | 44.7 | 213.9 | 132.7 |
| TSA B-Ala | 0 | Train | 673.0 | 364.2 | 368.0 | 235.8 | 9596.0 | 9990.0 | 8.9 | 12.5 | 41.3 | 18.9 |
| TSAM B-Ala | 1 | Train | 655.4 | 309.4 | 661.6 | 239.6 | 4476.0 | 4519.5 | 1818.5 | 1395.9 | 0.4 | 0.2 |
| TSAE B-Ala | 2 | Train | 960.7 | 1396.7 | 892.1 | 752.2 | 10000.0 | 9999.8 | 134.2 | 8.5 | 6.6 | 88.7 |
| TSA Glu | 0 | Validation | 9260.8 | 8976.3 | 7125.0 | 6203.0 | 10000.0 | 9999.8 | 202.4 | 65.7 | 35.2 | 94.4 |
| TSAM Glu | 1 | Validation | 6214.1 | 7938.1 | 695.2 | 771.3 | 4125.8 | 5622.2 | 193.2 | 19.5 | 3.6 | 39.6 |
| TSAE Glu | 2 | Train | 10000.0 | 10035.4 | 2284.3 | 2269.0 | 10000.0 | 9910.8 | 27.1 | 28.3 | 84.3 | 80.3 |
| TSA Gly | 0 | Train | 4550.0 | 277.0 | 376.0 | 235.7 | 10000.0 | 9978.9 | 8.4 | 8.8 | 44.8 | 26.8 |
| TSAM Gly | 1 | Validation | 9362.4 | 5603.6 | 478.4 | 235.7 | 867.3 | 1651.2 | 145.8 | 10.5 | 3.3 | 22.4 |
| TSAE Gly | 2 | Train | 4023.8 | 4032.9 | 428.1 | 235.7 | 3415.2 | 3465.8 | 330.4 | 8.4 | 1.3 | 28.1 |
| TSA Ile | 0 | Train | 10000.0 | 9724.8 | 2948.9 | 2952.7 | 4021.2 | 4024.9 | 92.6 | 47.1 | 31.8 | 62.7 |
| TSAM Ile | 1 | Train | 664.5 | 914.8 | 628.5 | 807.6 | 76.3 | 154.9 | 164.3 | 22.1 | 3.8 | 36.5 |
| TSAE Ile | 2 | Train | 7337.2 | 7292.8 | 1556.2 | 1644.5 | 1421.3 | 1301.5 | 189.3 | 19.9 | 8.2 | 82.8 |
| TSA Leu | 0 | Train | 4191.9 | 4392.1 | 4528.8 | 4505.6 | 2380.9 | 2482.7 | 24.0 | 25.2 | 188.7 | 178.6 |
| TSAM Leu | 1 | Validation | 96.9 | 387.6 | 396.0 | 588.8 | 516.8 | 537.2 | 167.3 | 288.6 | 2.4 | 2.0 |
| TSAE Leu | 2 | Train | 4854.5 | 4960.0 | 912.5 | 511.3 | 367.1 | 171.1 | 123.8 | 32.0 | 7.4 | 16.0 |
| TSA Met | 0 | Train | 346.7 | 798.1 | 803.7 | 565.5 | 3294.0 | 3040.8 | 2222.2 | 1960.4 | 0.4 | 0.3 |
| TSAM Met | 1 | Train | 531.3 | 459.8 | 3170.6 | 3153.4 | 45.6 | 64.0 | 1274.9 | 1962.3 | 2.5 | 1.6 |
| TSAE Met | 2 | Validation | 4527.8 | 1988.3 | 5017.9 | 4957.6 | 2336.1 | 60.1 | 2592.4 | 1951.0 | 1.9 | 2.5 |
| TSA Phe | 0 | Train | 305.4 | 210.1 | 866.1 | 870.8 | 9387.0 | 9998.7 | 191.5 | 131.5 | 4.5 | 6.6 |
| TSAM Phe | 1 | Train | 67.1 | 364.1 | 235.7 | 394.3 | 61.9 | 47.9 | 119.6 | 107.9 | 2.0 | 3.7 |
| TSAE Phe | 2 | Train | 4893.8 | 4847.3 | 6161.8 | 5956.5 | 374.5 | 47.5 | 223.1 | 22.9 | 27.6 | 259.8 |
| TSA Pro | 0 | Validation | 87.0 | 188.4 | 3112.8 | 3168.2 | 9315.0 | 8607.2 | 295.0 | 56.1 | 10.6 | 56.4 |
| TSAM Pro | 1 | Train | 256.4 | 204.1 | 773.1 | 759.2 | 350.4 | 329.7 | 265.5 | 158.4 | 2.9 | 4.8 |
| TSAE Pro | 2 | Validation | 958.4 | 398.8 | 1070.8 | 667.0 | 2206.9 | 719.9 | 25.7 | 47.9 | 41.7 | 13.9 |
| TSA Val | 0 | Train | 398.7 | 498.5 | 5335.4 | 5503.5 | 10000.0 | 9942.7 | 2111.1 | 2241.1 | 2.5 | 2.5 |
| TSAM Val | 1 | Validation | 377.0 | 308.8 | 839.3 | 1050.1 | 656.8 | 418.9 | 1371.8 | 457.2 | 0.6 | 2.3 |
| TSAE Val | 2 | Train | 932.2 | 613.9 | 804.1 | 857.2 | 476.3 | 677.7 | 130.9 | 179.2 | 6.1 | 4.8 |
| KI (nM) | Selectivity | ||||
|---|---|---|---|---|---|
| TSA Derivative | hCA I | hCA II | hCA IV | hCA IX | hCA II/IX |
| 2 Ala | 239.6 | 235.8 | 8629.1 | 8.4 | 28.0 |
| 4 Tyr | 376.0 | 6198.0 | 9998.5 | 192.9 | 32.1 |
| 5 Trp | 10085.9 | 6217.8 | 9837.2 | 2218.0 | 2.8 |
| 6 Asn | 8892.8 | 924.6 | 9930.3 | 720.1 | 1.3 |
| 7 Gln | 9996.2 | 4111.2 | 2328.9 | 29.6 | 139.1 |
| 8 Ser | 4711.8 | 250.8 | 10000.0 | 8.7 | 28.8 |
| 9 Thr | 7271.6 | 555.3 | 3099.5 | 13.1 | 42.3 |
| Product | Stationary Phase | Mobile Phase A | Mobile Phase B | %B |
|---|---|---|---|---|
| 2 Ala | RP-C18 | 100 mM NH4HCO3 | Methanol | 12.5 |
| 3 Phe | RP-C18 | 100 mM NH4HCO3 | Methanol | 45.0 |
| 4 Tyr | RP-C18 | 100 mM NH4HCO3 | Methanol | 25.0 |
| 5 Trp | RP-C18 | 100 mM NH4HCO3 | Methanol | 40.0 |
| 6 Asn | RP-C18 | 50 mM NH4HCO3 | Methanol | 5.0 |
| 7 Gln | RP-C18 | 100 mM NH4HCO3 | Methanol | 7.5 |
| 8 Ser | RP-C18 | 50 mM NH4HCO3 | Methanol | 5.0 |
| 9 Thr | RP-C18 | 100 mM NH4HCO3 | Methanol | 10.0 |
| 10 Asp | HILIC | 100 mM NH4HCO3 | Acetonitrile | 72.5 |
| 11 Glu | HILIC | 100 mM NH4HCO3 | Acetonitrile | 75.0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mikulová, M.B.; Kružlicová, D.; Pecher, D.; Supuran, C.T.; Mikuš, P. Synthetic Strategies and Computational Inhibition Activity Study for Triazinyl-Substituted Benzenesulfonamide Conjugates with Polar and Hydrophobic Amino Acids as Inhibitors of Carbonic Anhydrases. Int. J. Mol. Sci. 2020, 21, 3661. https://doi.org/10.3390/ijms21103661
Mikulová MB, Kružlicová D, Pecher D, Supuran CT, Mikuš P. Synthetic Strategies and Computational Inhibition Activity Study for Triazinyl-Substituted Benzenesulfonamide Conjugates with Polar and Hydrophobic Amino Acids as Inhibitors of Carbonic Anhydrases. International Journal of Molecular Sciences. 2020; 21(10):3661. https://doi.org/10.3390/ijms21103661
Chicago/Turabian StyleMikulová, Mária Bodnár, Dáša Kružlicová, Daniel Pecher, Claudiu T. Supuran, and Peter Mikuš. 2020. "Synthetic Strategies and Computational Inhibition Activity Study for Triazinyl-Substituted Benzenesulfonamide Conjugates with Polar and Hydrophobic Amino Acids as Inhibitors of Carbonic Anhydrases" International Journal of Molecular Sciences 21, no. 10: 3661. https://doi.org/10.3390/ijms21103661
APA StyleMikulová, M. B., Kružlicová, D., Pecher, D., Supuran, C. T., & Mikuš, P. (2020). Synthetic Strategies and Computational Inhibition Activity Study for Triazinyl-Substituted Benzenesulfonamide Conjugates with Polar and Hydrophobic Amino Acids as Inhibitors of Carbonic Anhydrases. International Journal of Molecular Sciences, 21(10), 3661. https://doi.org/10.3390/ijms21103661







