Lipids and Lipid Mediators Associated with the Risk and Pathology of Ischemic Stroke
Abstract
1. Introduction
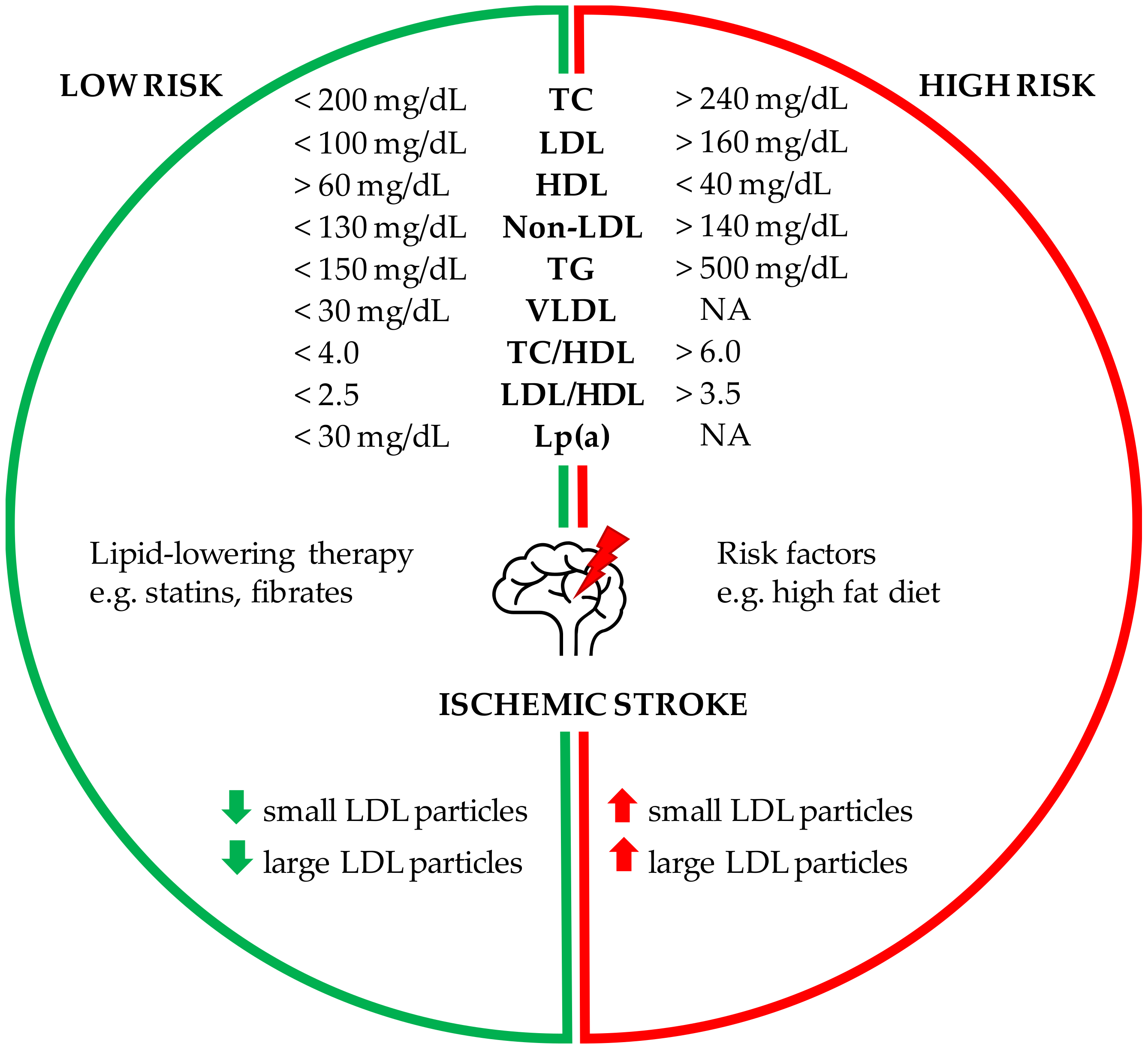
2. Association of Serum Lipids with the Risk of Stroke
2.1. Cholesterol and the Risk of Stroke
2.2. Hypertriglyceridemia and Ischemic Stroke
2.3. Non-Traditional Lipid Profiles as Stroke Predictors
2.4. Number, Size and Composition of Lipoprotein Particle
2.5. Elevated Lipoprotein(a) and the Risk of Ischemic Stroke
2.6. Polyunsaturated Fatty Acids and Risk of Ischemic Stroke
2.7. Fatty Acids and Cardioembolic Stroke
2.8. The Role of Fish-Derived Fatty Acids in Stroke
2.9. Lipid-Lowering Therapy for Prevention of Ischemic Stroke
3. Lipids of the Brain during Ischemic Stroke
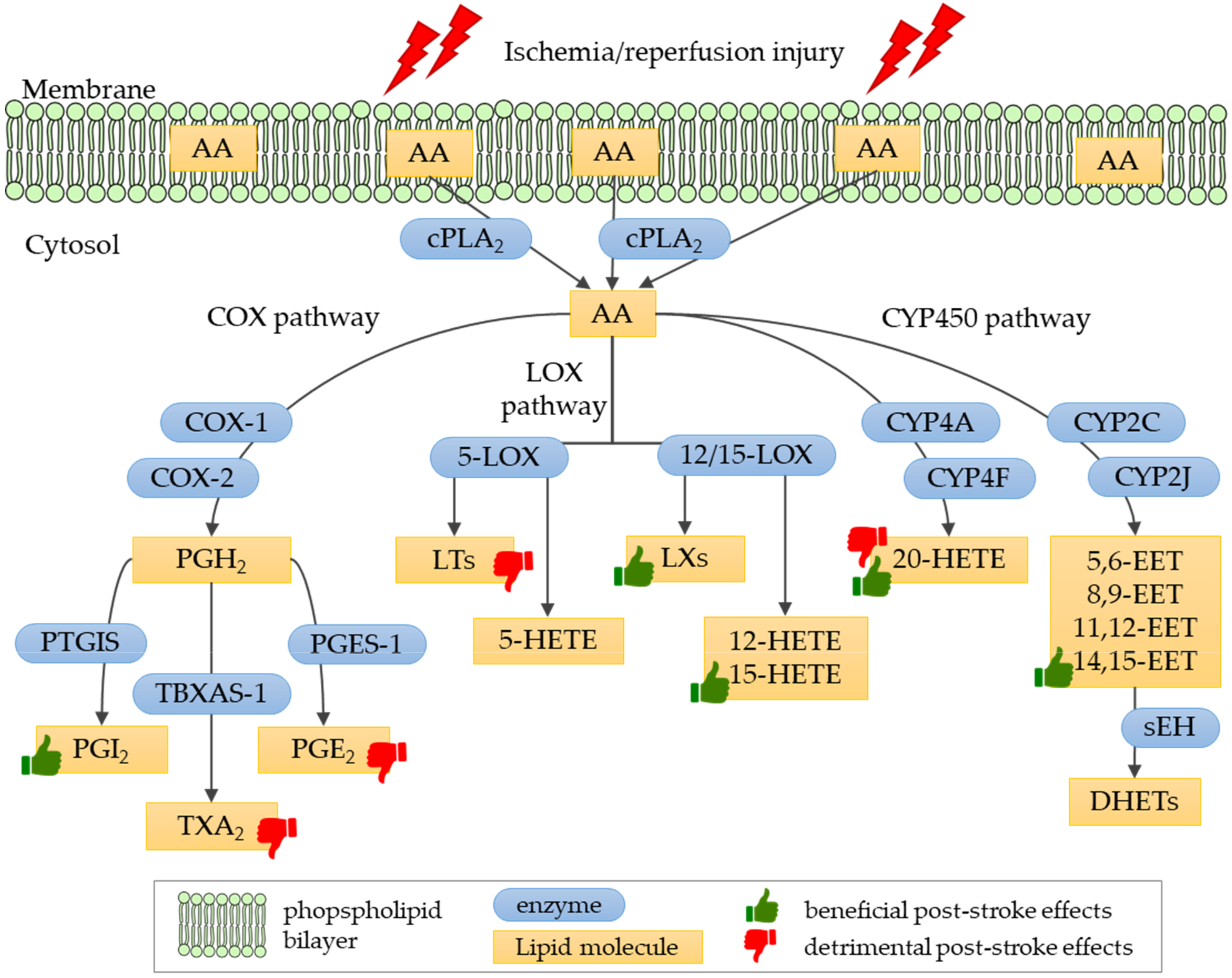
4. Eicosanoids in Ischemic Stroke
4.1. Biosynthesis and Physiological Role of Eicosanoids
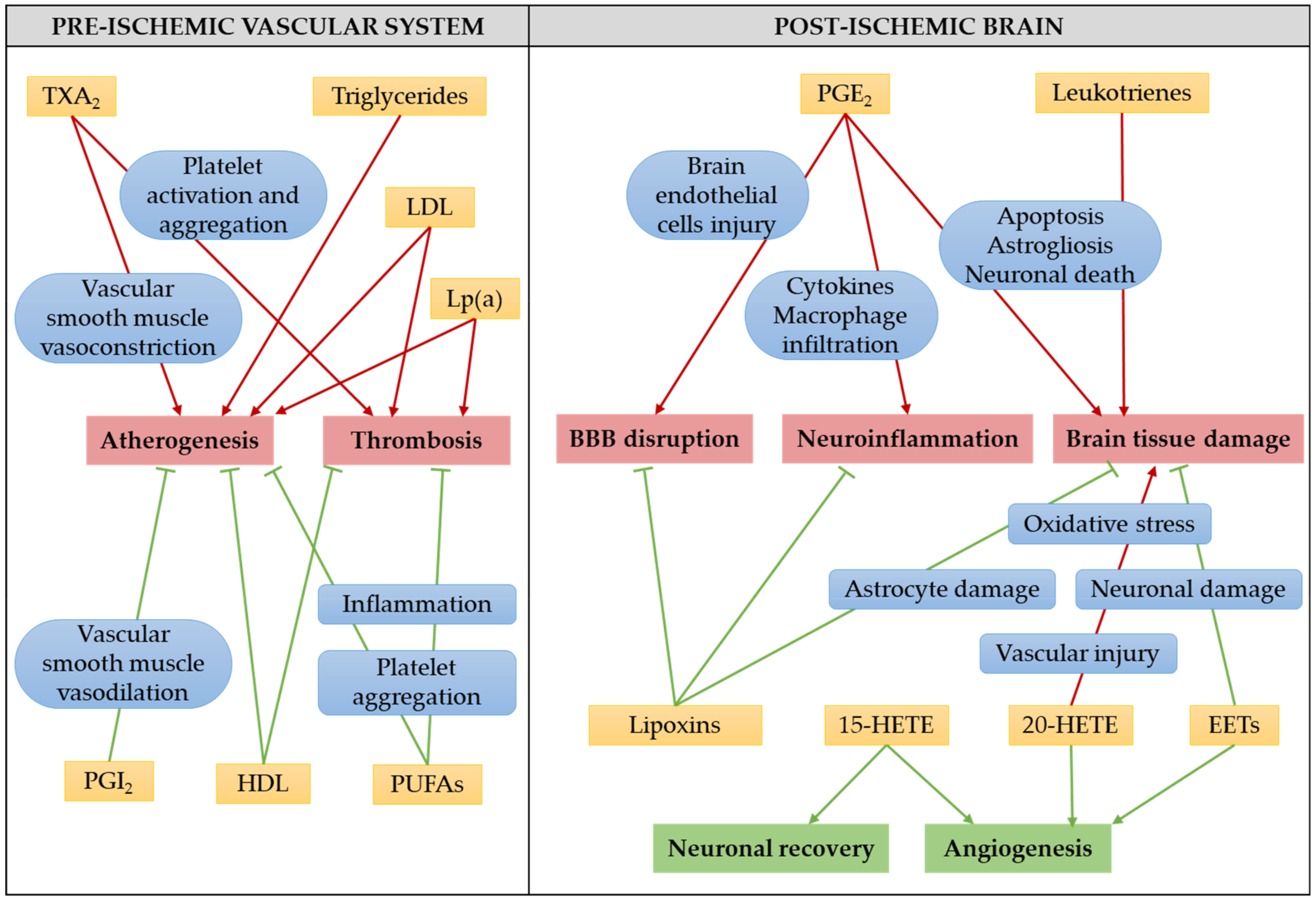
4.2. The Role of Eicosanoids in Ischemic Stroke Pathology
4.2.1. Prostanoids in Ischemic Stroke
4.2.2. Leukotrienes in Ischemic Stroke
4.2.3. Lipoxin A4 in Ischemic Stroke
4.2.4. Hydroxyeicosatetraenoic Acids in Ischemic Stroke
4.2.5. Epoxyeicosatrienoic Acids in Ischemic Stroke
4.3. Eicosanoids as the Target for Ischemic Stroke Treatment
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AA | arachidonic acid |
| CAD | coronary artery disease |
| CE | cardioembolic |
| CETP | cholesterylester transfer protein |
| COX | cyclooxygenase |
| cPLA2 | cytosolic phospholipase A2 |
| CVD | cardiovascular disease |
| CYP | cytochrome P450 |
| CysLT | cysteinyl leukotriene |
| DHA | docosahexaenoic acid |
| DPA | docosapentaenoic acid |
| EET | epoxyeicosatrienoic acid |
| END | early neurological deterioration |
| EPA | eicosapentaenoic acid |
| FA | fatty acid |
| FLAP | 5-LOX-activating protein |
| HDL | high-density lipoprotein |
| HDL-C | high-density lipoprotein cholesterol |
| HETE | hydroxyeicosatetraenoic acid |
| HS | hemorrhagic stroke |
| IDL | intermediate-density lipoprotein |
| IS | ischemic stroke |
| LA | linoleic acid |
| LDL | low-density lipoprotein |
| LDL-C | low-density lipoprotein cholesterol |
| LOX | lipoxygenase |
| Lp(a) | lipoprotein(a) |
| LT | leukotriene |
| LX | lipoxin |
| LXA4ME | lipoxin A4 methyl ester |
| NMR | nuclear magnetic resonance |
| non-HDL-C | non-high-density lipoprotein cholesterol |
| NPD1 | neuroprotectin D1 |
| OGD | oxygen-glucose deprivation |
| PAF | platelet-activating factor |
| PCSK9 | proprotein convertase subtilisin/kexin type-9 |
| PG | prostaglandin |
| PGE2 | prostaglandin E2 |
| PGI2 | prostaglandin I2, prostacyclin |
| PUFA | polyunsaturated fatty acid |
| sEH | soluble epoxide hydrolase |
| TC | total cholesterol |
| TG | triglyceride |
| TXA2 | thromboxane A2 |
| VEGF | vascular endothelial growth factor |
| VLDL | very low-density lipoprotein |
References
- James, S.L.; Abate, D.; Hassen Abate, K.; Abay, S.M.; Abbafati, C.; Abbasi, N.; Abbastabar, H.; Abd-Allah, F.; Abdela, J.; Abdelalim, A.; et al. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1789–1858. [Google Scholar] [CrossRef]
- World Health Organization. Global Health Estimates 2016: Disease burden by Cause, Age, Sex, by Country and by Region, 2000–2016; WHO: Geneva, Switzerland, 2018. [Google Scholar]
- Avan, A.; Digaleh, H.; Di Napoli, M.; Stranges, S.; Behrouz, R.; Shojaeianbabaei, G.; Amiri, A.; Tabrizi, R.; Mokhber, N.; Spence, J.D.; et al. Socioeconomic status and stroke incidence, prevalence, mortality, and worldwide burden: An ecological analysis from the Global Burden of Disease Study 2017. BMC Med. 2019, 17, 191. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, M.J.; Xavier, D.; Liu, L.; Zhang, H.; Chin, S.L.; Rao-Melacini, P.; Rangarajan, S.; Islam, S.; Pais, P.; McQueen, M.J.; et al. Risk factors for ischaemic and intracerebral haemorrhagic stroke in 22 countries (the INTERSTROKE study): A case-control study. Lancet 2010, 376, 112–123. [Google Scholar] [CrossRef]
- Gorelick, P.B.; Mazzone, T. Plasma Lipids and Stroke. Eur. J. Cardiovasc. Risk 1999, 6, 217–221. [Google Scholar] [CrossRef]
- Tirschwell, D.L.; Smith, N.L.; Heckbert, S.R.; Lemaitre, R.N.; Longstreth, W.T.; Psaty, B.M. Association of cholesterol with stroke risk varies in stroke subtypes and patient subgroups. Neurology 2004, 63, 1868–1875. [Google Scholar] [CrossRef]
- Jain, M.; Jain, A.; Yerragondu, N.; Brown, R.D.; Rabinstein, A.; Jahromi, B.S.; Vaidyanathan, L.; Blyth, B.; Ganti Stead, L. The Triglyceride Paradox in Stroke Survivors: A Prospective Study. Corp. Neurosci. J. 2013, 2013. [Google Scholar] [CrossRef]
- Park, J.H.; Lee, J.; Ovbiagele, B. Nontraditional serum lipid variables and recurrent stroke risk. Stroke 2014, 45, 3269–3274. [Google Scholar] [CrossRef]
- Holmes, M.V.; Millwood, I.Y.; Kartsonaki, C.; Hill, M.R.; Bennett, D.A.; Boxall, R.; Guo, Y.; Xu, X.; Bian, Z.; Hu, R.; et al. Lipids, Lipoproteins, and Metabolites and Risk of Myocardial Infarction and Stroke. J. Am. Coll. Cardiol. 2018, 71, 620–632. [Google Scholar] [CrossRef]
- Goldstein, L.B.; Bushnell, C.D.; Adams, R.J.; Appel, L.J.; Braun, L.T.; Chaturvedi, S.; Creager, M.A.; Culebras, A.; Eckel, R.H.; Hart, R.G.; et al. Guidelines for the primary prevention of stroke: A Guideline for Healthcare Professionals from the American Heart Association/American Stroke Association. Stroke 2011, 42, 517–584. [Google Scholar] [CrossRef]
- Shahar, E.; Chambless, L.E.; Rosamond, W.D.; Boland, L.L.; Ballantyne, C.M.; McGovern, P.G.; Sharrett, A.R. Plasma lipid profile and incident ischemic stroke: The Atherosclerosis Risk in Communities (ARIC) Study. Stroke 2003, 34, 623–631. [Google Scholar] [CrossRef]
- Suh, I.; Jee, S.H.; Kim, H.C.; Nam, C.M.; Kim, I.S.; Appel, L.J. Low serum cholesterol and haemorrhagic stroke in men: Korea Medical Insurance Corporation Study. Lancet 2001, 357, 922–925. [Google Scholar] [CrossRef]
- Hart, C.L.; Hole, D.J.; Smith, G.D. Risk factors and 20-year stroke mortality in men and women in the Renfrew/Paisley study in Scotland. Stroke 1999, 30, 1999–2007. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tanizaki, Y.; Kiyohara, Y.; Kato, I.; Iwamoto, H.; Nakayama, K.; Shinohara, N.; Arima, H.; Tanaka, K.; Ibayashi, S.; Fujishima, M. Incidence and risk factors for subtypes of cerebral infarction in a general population: The Hisayama study. Stroke 2000, 31, 2616–2622. [Google Scholar] [CrossRef] [PubMed]
- Sacco, R.L.; Benson, R.T.; Kargman, D.E.; Boden-Albala, B.; Tuck, C.; Lin, I.F.; Cheng, J.F.; Paik, M.C.; Shea, S.; Berglund, L. High-density lipoprotein cholesterol and ischemic stroke in the elderly the northern manhattan stroke study. J. Am. Med. Assoc. 2001, 285, 2729–2735. [Google Scholar] [CrossRef]
- Spence, J.D. Statins for prevention of stroke. Lancet 1998, 352, 909. [Google Scholar] [CrossRef]
- Zhang, Y.; Tuomilehto, J.; Jousilahti, P.; Wang, Y.; Antikainen, R.; Hu, G. Total and high-density lipoprotein cholesterol and stroke risk. Stroke 2012, 43, 1768–1774. [Google Scholar] [CrossRef]
- Wang, X.; Dong, Y.; Qi, X.; Huang, C.; Hou, L. Cholesterol levels and risk of hemorrhagic stroke: A systematic review and meta-analysis. Stroke 2013, 44, 1833–1839. [Google Scholar] [CrossRef]
- Di Angelantonio, E.; Sarwar, N.; Perry, P.; Kaptoge, S.; Ray, K.K.; Thompson, A.; Wood, A.M.; Lewington, S.; Sattar, N.; Packard, C.J.; et al. Major lipids, apolipoproteins, and risk of vascular disease. JAMA J. Am. Med. Assoc. 2009, 302, 1993–2000. [Google Scholar]
- Ali, K.M.; Wonnerth, A.; Huber, K.; Wojta, J. Cardiovascular disease risk reduction by raising HDL cholesterol—Current therapies and future opportunities. Br. J. Pharmacol. 2012, 167, 1177–1194. [Google Scholar] [CrossRef]
- Ballantyne, C.M.; Herd, J.A.; Ferlic, L.L.; Dunn, J.K.; Farmer, J.A.; Jones, P.H.; Schein, J.R.; Gotto, A.M. Influence of low HDL on progression of coronary artery disease and response to fluvastatin therapy. Circulation 1999, 99, 736–743. [Google Scholar] [CrossRef]
- Santos-Gallego, C.G.; Badimón, J.J. High-Density Lipoprotein and Cardiovascular Risk Reduction: Promises and Realities. Rev. Española Cardiol. 2012, 65, 305–308. [Google Scholar] [CrossRef] [PubMed]
- Imamura, T.; Doi, Y.; Arima, H.; Yonemoto, K.; Hata, J.; Kubo, M.; Tanizaki, Y.; Ibayashi, S.; Iida, M.; Kiyohara, Y. LDL cholesterol and the development of stroke subtypes and coronary heart disease in a general Japanese population the Hisayama study. Stroke 2009, 40, 382–388. [Google Scholar] [CrossRef] [PubMed]
- Gu, X.; Li, Y.; Chen, S.; Yang, X.; Liu, F.; Li, Y.; Li, J.; Cao, J.; Liu, X.; Chen, J.; et al. Association of Lipids With Ischemic and Hemorrhagic Stroke: A Prospective Cohort Study Among 267 500 Chinese. Stroke 2019, 50, 3376–3384. [Google Scholar] [CrossRef] [PubMed]
- Dziedzic, T.; Slowik, A.; Gryz, E.A.; Szczudlik, A. Lower serum triglyceride level is associated with increased stroke severity. Stroke 2004, 35, e151–e152. [Google Scholar] [CrossRef]
- Simundic, A.M.; Nikolac, N.; Topic, E.; Basic-Kes, V.; Demarin, V. Are serum lipids measured on stroke admission prognostic? Clin. Chem. Lab. Med. 2008, 46, 1163–1167. [Google Scholar] [CrossRef]
- Berger, J.S.; McGinn, A.P.; Howard, B.V.; Kuller, L.; Manson, J.E.; Otvos, J.; Curb, J.D.; Eaton, C.B.; Kaplan, R.C.; Lynch, J.K.; et al. Lipid and lipoprotein biomarkers and the risk of ischemic stroke in postmenopausal women. Stroke 2012, 43, 958–966. [Google Scholar] [CrossRef]
- Ebinger, M.; Sievers, C.; Klotsche, J.; Schneider, H.J.; Leonards, C.O.; Pieper, L.; Wittchen, H.U.; Stalla, G.K.; Endres, M. Triglycerides and stroke risk prediction: Lessons from a prospective cohort study in German primary care patients. Front. Neurol. 2010. [Google Scholar] [CrossRef]
- Leonards, C.; Ebinger, M.; Batluk, J.; Malzahn, U.; Heuschmann, P.; Endres, M. The role of fasting versus non-fasting triglycerides in ischemic stroke: A systematic review. Front. Neurol. 2010, 1, 133. [Google Scholar] [CrossRef][Green Version]
- Bansal, S.; Buring, J.E.; Rifai, N.; Mora, S.; Sacks, F.M.; Ridker, P.M. Fasting compared with nonfasting triglycerides and risk of cardiovascular events in women. J. Am. Med. Assoc. 2007, 298, 309–316. [Google Scholar] [CrossRef]
- Freiberg, J.J.; Tybjærg-Hansen, A.; Jensen, J.S.; Nordestgaard, B.G. Nonfasting triglycerides and risk of ischemic stroke in the general population. JAMA J. Am. Med. Assoc. 2008, 300, 2142–2152. [Google Scholar] [CrossRef]
- Weir, C.J.; Sattar, N.; Walters, M.R.; Lees, K.R. Low triglyceride, not low cholesterol concentration, independently predicts poor outcome following acute stroke. Cerebrovasc. Dis. 2003, 16, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Chapman, M.J.; Ginsberg, H.N.; Amarenco, P.; Andreotti, F.; Borén, J.; Catapano, A.L.; Descamps, O.S.; Fisher, E.; Kovanen, P.T.; Kuivenhoven, J.A.; et al. Triglyceride-rich lipoproteins and high-density lipoprotein cholesterol in patients at high risk of cardiovascular disease: Evidence and guidance for management. Eur. Heart J. 2011, 32, 1345–1361. [Google Scholar] [CrossRef] [PubMed]
- Grundy, S.M. Hypertriglyceridemia, atherogenic dyslipidemia, and the metabolic syndrome. Am. J. Cardiol. 1998, 81, 18B–25B. [Google Scholar] [CrossRef]
- Arsenault, B.J.; Rana, J.S.; Stroes, E.S.G.; Després, J.P.; Shah, P.K.; Kastelein, J.J.P.; Wareham, N.J.; Boekholdt, S.M.; Khaw, K.T. Beyond Low-Density Lipoprotein Cholesterol. Respective Contributions of Non-High-Density Lipoprotein Cholesterol Levels, Triglycerides, and the Total Cholesterol/High-Density Lipoprotein Cholesterol Ratio to Coronary Heart Disease Risk in Apparently Healt. J. Am. Coll. Cardiol. 2009, 55, 35–41. [Google Scholar] [CrossRef]
- Ridker, P.M.; Rifai, N.; Cook, N.R.; Bradwin, G.; Buring, J.E. Non-HDL cholesterol, apolipoproteins A-I and B 100, standard lipid measures, lipid ratios, and CRP as risk factors for cardiovascular disease in women. J. Am. Med. Assoc. 2005, 294, 326–333. [Google Scholar] [CrossRef]
- Mathews, S.C.; Mallidi, J.; Kulkarni, K.; Toth, P.P.; Jones, S.R. Achieving Secondary Prevention Low-Density Lipoprotein Particle Concentration Goals Using Lipoprotein Cholesterol-Based Data. PLoS ONE 2012, 7, e33692. [Google Scholar] [CrossRef]
- Elshazly, M.B.; Quispe, R.; Michos, E.D.; Sniderman, A.D.; Toth, P.P.; Banach, M.; Kulkarni, K.R.; Coresh, J.; Blumenthal, R.S.; Jones, S.R.; et al. Patient-level discordance in population percentiles of the total cholesterol to high-density lipoprotein cholesterol ratio in comparison with low-density lipoprotein cholesterol and non-high-density lipoprotein cholesterol: The very large database of lipi. Circulation 2015, 132, 667–676. [Google Scholar] [CrossRef]
- Katakami, N.; Kaneto, H.; Osonoi, T.; Saitou, M.; Takahara, M.; Sakamoto, F.; Yamamoto, K.; Yasuda, T.; Matsuoka, T.A.; Matsuhisa, M.; et al. Usefulness of lipoprotein ratios in assessing carotid atherosclerosis in Japanese type 2 diabetic patients. Atherosclerosis 2011, 214, 442–447. [Google Scholar] [CrossRef]
- De la Riva, P.; Zubikarai, M.; Sarasqueta, C.; Tainta, M.; Muñoz-Lopetegui, A.; Andrés-Marín, N.; González, F.; Díez, N.; de Arce, A.; Bergareche, A.; et al. Nontraditional Lipid Variables Predict Recurrent Brain Ischemia in Embolic Stroke of Undetermined Source. J. Stroke Cerebrovasc. Dis. 2017, 26, 1670–1677. [Google Scholar] [CrossRef]
- Zheng, J.; Sun, Z.; Zhang, X.; Li, Z.; Guo, X.; Xie, Y.; Sun, Y.; Zheng, L. Non-traditional lipid profiles associated with ischemic stroke not hemorrhagic stroke in hypertensive patients: Results from an 8.4 years follow-up study. Lipids Health Dis. 2019, 18, 9. [Google Scholar] [CrossRef]
- Guo, X.; Li, Z.; Sun, G.; Guo, L.; Zheng, L.; Yu, S.; Yang, H.; Pan, G.; Zhang, Y.; Sun, Y. Comparison of four nontraditional lipid profiles in relation to ischemic stroke among hypertensive Chinese population. Int. J. Cardiol. 2015, 201, 123–125. [Google Scholar] [CrossRef] [PubMed]
- Würtz, P.; Havulinna, A.S.; Soininen, P.; Tynkkynen, T.; Prieto-Merino, D.; Tillin, T.; Ghorbani, A.; Artati, A.; Wang, Q.; Tiainen, M.; et al. Metabolite profiling and cardiovascular event risk: A prospective study of 3 population-based cohorts. Circulation 2015, 131, 774–785. [Google Scholar] [CrossRef] [PubMed]
- Björnheden, T.; Babyi, A.; Bondjers, G.; Wiklund, O. Accumulation of lipoprotein fractions and subfractions in the arterial wall, determined in an in vitro perfusion system. Atherosclerosis 1996, 123, 43–56. [Google Scholar] [CrossRef]
- Jeyarajah, E.J.; Cromwell, W.C.; Otvos, J.D. Lipoprotein Particle Analysis by Nuclear Magnetic Resonance Spectroscopy. Clin. Lab. Med. 2006, 26, 847–870. [Google Scholar] [CrossRef] [PubMed]
- Hachinski, V.; Graffagnino, C.; Beaudry, M.; Bernier, G.; Buck, C.; Donner, A.; Spence, J.D.; Doig, G.; Wolfe, B.M.J. Lipids and stroke: A paradox resolved. Arch. Neurol. 1996, 53, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Glader, C.A.; Stegmayr, B.; Boman, J.; Stenlund, H.; Weinehall, L.; Hallmans, G.; Dahlén, G.H. Chlamydia pneumoniae antibodies and high lipoprotein(a) levels do not predict ischemic cerebral infarctions. Results from a nested case-control study in Northern Sweden. Stroke 1999, 30, 2013–2018. [Google Scholar] [CrossRef] [PubMed]
- Alfthan, G.; Pekkanen, J.; Jauhiainen, M.; Pitkäniemi, J.; Karvonen, M.; Tuomilehto, J.; Salonen, J.T.; Ehnholm, C. Relation of serum homocysteine and lipoprotein(a) concentrations to atherosclerotic disease in a prospective Finnish population based study. Atherosclerosis 1994, 106, 9–19. [Google Scholar] [CrossRef]
- Tipping, R.W.; Ford, C.E.; Simpson, L.M.; Walldius, G.; Jungner, I.; Folsom, A.R.; Chambless, L.; Panagiotakos, D.; Pitsavos, C.; Chrysohoou, C.; et al. Lipoprotein(a) concentration and the risk of coronary heart disease, stroke, and nonvascular mortality. JAMA J. Am. Med. Assoc. 2009, 302, 412–423. [Google Scholar]
- Nave, A.H.; Lange, K.S.; Leonards, C.O.; Siegerink, B.; Doehner, W.; Landmesser, U.; Steinhagen-Thiessen, E.; Endres, M.; Ebinger, M. Lipoprotein (a) as a risk factor for ischemic stroke: A meta-analysis. Atherosclerosis 2015, 242, 496–503. [Google Scholar] [CrossRef]
- Journal, B.; Lan, Y.; Zhao, X.; Wang, X.; Song, X.; Chen, J.; Zhang, Y.; Zhang, Z. Lipoprotein(a) as a Risk Factor for Predicting Coronary Artery Disease Events: A Meta-analysis. Biomark J. 2018, 4, 17. [Google Scholar]
- Yaghi, S.; Elkind, M.S.V. Lipids and Cerebrovascular Disease. Stroke 2015, 46, 3322–3328. [Google Scholar] [CrossRef] [PubMed]
- Nordestgaard, B.G.; Chapman, M.J.; Ray, K.; Boré, J.; Andreotti, F.; Watts, G.F.; Ginsberg, H.; Amarenco, P.; Catapano, A.; Descamps, O.S.; et al. Lipoprotein(a) as a cardiovascular risk factor: Current status. Eur. Heart J. 2010, 31, 2844–2853. [Google Scholar] [CrossRef] [PubMed]
- Bu, J.; Dou, Y.; Tian, X.; Wang, Z.; Chen, G. The Role of Omega-3 Polyunsaturated Fatty Acids in Stroke. Oxid. Med. Cell. Lomgev. 2016. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D.; Wu, J.H.Y. Omega-3 fatty acids and cardiovascular disease: Effects on risk factors, molecular pathways, and clinical events. J. Am. Coll. Cardiol. 2011, 58, 2047–2067. [Google Scholar] [CrossRef]
- Rebiger, L.; Lenzen, S.; Mehmeti, I. Susceptibility of brown adipocytes to pro-inflammatory cytokine toxicity and reactive oxygen species. Biosci. Rep. 2016, 36, e00306. [Google Scholar] [CrossRef]
- Geleijnse, J.M.; Giltay, E.J.; Grobbee, D.E.; Donders, A.R.T.; Kok, F.J. Blood pressure response to fish oil supplementation: Metaregression analysis of randomized trials. J. Hypertens. 2002, 20, 1493–1499. [Google Scholar] [CrossRef]
- Mozaffarian, D.; Wu, J.H.Y. (n-3) Fatty Acids and Cardiovascular Health: Are Effects of EPA and DHA Shared or Complementary? J. Nutr. 2012, 142, 614S–625S. [Google Scholar] [CrossRef]
- Wallström, P.; Sonestedt, E.; Hlebowicz, J.; Ericson, U.; Drake, I.; Persson, M.; Gullberg, B.; Hedblad, B.; Wirfält, E. Dietary fiber and saturated fat intake associations with cardiovascular disease differ by sex in the Malmö diet and cancer cohort: A prospective study. PLoS ONE 2012, 7, e31637. [Google Scholar] [CrossRef]
- Yaemsiri, S.; Sen, S.; Tinker, L.F.; Robinson, W.R.; Evans, R.W.; Rosamond, W.; Wasserthiel-Smoller, S.; He, K. Serum fatty acids and incidence of ischemic stroke among postmenopausal women. Stroke 2013, 44, 2710–2717. [Google Scholar] [CrossRef]
- Tanaka, K.; Ishikawa, Y.; Yokoyama, M.; Origasa, H.; Matsuzaki, M.; Saito, Y.; Matsuzawa, Y.; Sasaki, J.; Oikawa, S.; Hishida, H.; et al. Reduction in the recurrence of stroke by eicosapentaenoic acid for hypercholesterolemic patients: Subanalysis of the JELIS trial. Stroke 2008, 39, 2052–2058. [Google Scholar] [CrossRef]
- Nishizaki, Y.; Shimada, K.; Tani, S.; Ogawa, T.; Ando, J.; Takahashi, M.; Yamamoto, M.; Shinozaki, T.; Miyauchi, K.; Nagao, K.; et al. Significance of imbalance in the ratio of serum n-3 to n-6 polyunsaturated fatty acids in patients with acute coronary syndrome. Am. J. Cardiol. 2014, 113, 441–445. [Google Scholar] [CrossRef] [PubMed]
- Thies, F.; Garry, J.M.C.; Yaqoob, P.; Rerkasem, K.; Williams, J.; Shearman, C.P.; Gallagher, P.J.; Calder, P.C.; Grimble, R.F. Association of n-3 polyunsaturated fatty acids with stability of atherosclerotic plaques: A randomised controlled trial. Lancet 2003, 361, 477–485. [Google Scholar] [CrossRef]
- Ajami, M.; Eghtesadi, S.; Razaz, J.M.; Kalantari, N.; Habibey, R.; Nilforoushzadeh, M.A.; Zarrindast, M.; Pazoki-Toroudi, H. Expression of Bcl-2 and Bax after hippocampal ischemia in DHA + EPA treated rats. Neurol. Sci. 2011, 32, 811–818. [Google Scholar] [CrossRef] [PubMed]
- Suda, S.; Katsumata, T.; Okubo, S.; Kanamaru, T.; Suzuki, K.; Watanabe, Y.; Katsura, K.I.; Katayama, Y. Low serum n-3 polyunsaturated fatty acid/n-6 polyunsaturated fatty acid ratio predicts neurological deterioration in japanese patients with acute ischemic stroke. Cerebrovasc. Dis. 2013, 36, 388–393. [Google Scholar] [CrossRef]
- Alawneh, J.A.; Moustafa, R.R.; Baron, J.C. Hemodynamic factors and perfusion abnormalities in early neurological deterioration. Stroke 2009, 40, e443–e450. [Google Scholar] [CrossRef]
- Huang, Z.X.; Wang, Q.Z.; Dai, Y.Y.; Lu, H.K.; Liang, X.Y.; Hu, H.; Liu, X.T. Early neurological deterioration in acute ischemic stroke: A propensity score analysis. J. Chinese Med. Assoc. 2018, 81, 865–870. [Google Scholar] [CrossRef]
- Poirier, P.; Giles, T.D.; Bray, G.A.; Hong, Y.; Stern, J.S.; Pi-Sunyer, F.X.; Eckel, R.H. Obesity and cardiovascular disease: Pathophysiology, evaluation, and effect of weight loss: An update of the 1997 American Heart Association Scientific Statement on obesity and heart disease from the Obesity Committee of the Council on Nutrition, Physical. Circulation 2006, 113, 898–918. [Google Scholar] [CrossRef]
- Fagot-Campagna, A.; Balkau, B.; Simon, D.; Warnet, J.-M.; Claude, J.-R.; Ducimetiere, P.; Eschwege, E. High free fatty acid concentration: An independent risk factor for hypertension in the Paris Prospective Study. Int. J. Epidemiol. 1998, 27, 808–813. [Google Scholar] [CrossRef]
- Bays, H.; Mandarino, L.; DeFronzo, R.A. Role of the Adipocyte, Free Fatty Acids, and Ectopic Fat in Pathogenesis of Type 2 Diabetes Mellitus: Peroxisomal Proliferator-Activated Receptor Agonists Provide a Rational Therapeutic Approach. J. Clin. Endocrinol. Metab. 2004, 89, 463–478. [Google Scholar] [CrossRef]
- O’Donoghue, M.; De Lemos, J.A.; Morrow, D.A.; Murphy, S.A.; Buros, J.L.; Cannon, C.P.; Sabatine, M.S. Prognostic utility of heart-type fatty acid binding protein in patients with acute coronary syndromes. Circulation 2006, 114, 550–557. [Google Scholar] [CrossRef]
- Cocco, G.; Chu, D. Drug points: Rimonabant may induce atrial fibrillation. BMJ 2009, 339, 296. [Google Scholar]
- Khawaja, O.; Bartz, T.M.; Ix, J.H.; Heckbert, S.R.; Kizer, J.R.; Zieman, S.J.; Mukamal, K.J.; Tracy, R.P.; Siscovick, D.S.; Djoussé, L. Plasma free fatty acids and risk of atrial fibrillation (from the Cardiovascular Health Study). Am. J. Cardiol. 2012, 110, 212–216. [Google Scholar] [CrossRef] [PubMed]
- Iso, H.; Sato, S.; Umemura, U.; Kudo, M.; Koike, K.; Kitamura, A.; Imano, H.; Okamura, T.; Naito, Y.; Shimamoto, T. Linoleic acid, other fatty acids, and the risk of stroke. Stroke 2002, 33, 2086–2093. [Google Scholar] [CrossRef] [PubMed]
- Saber, H.; Yakoob, M.Y.; Shi, P.; Longstreth, W.T.; Lemaitre, R.N.; Siscovick, D.; Rexrode, K.M.; Willett, W.C.; Mozaffarian, D. Omega-3 fatty acids and incident ischemic stroke and its atherothrombotic and cardioembolic subtypes in 3 US cohorts. Stroke 2017, 48, 2678–2685. [Google Scholar] [CrossRef]
- Choi, J.Y.; Kim, J.S.; Kim, J.H.; Oh, K.; Koh, S.B.; Seo, W.K. High free fatty acid level is associated with recurrent stroke in cardioembolic stroke patients. Neurology 2014, 82, 1142–1148. [Google Scholar] [CrossRef]
- Seo, W.K.; Jung, J.M.; Kim, J.H.; Koh, S.B.; Bang, O.Y.; Oh, K. Free fatty acid is associated with thrombogenicity in cardioembolic stroke. Cerebrovasc. Dis. 2017, 44, 160–168. [Google Scholar] [CrossRef]
- Erkkilä, A.T.; Lehto, S.; Pyörälä, K.; Uusitupa, M.I.J. n-3 Fatty acids and 5-y risks of death and cardiovascular disease events in patients with coronary artery disease. Am. J. Clin. Nutr. 2003, 78, 65–71. [Google Scholar] [CrossRef]
- Watanabe, Y.; Tatsuno, I. Omega-3 polyunsaturated fatty acids for cardiovascular diseases: Present, past and future. Expert Rev. Clin. Pharmacol. 2017, 10, 865–873. [Google Scholar] [CrossRef]
- Owen, A.J.; Magliano, D.J.; O’Dea, K.; Barr, E.L.M.; Shaw, J.E. Polyunsaturated fatty acid intake and risk of cardiovascular mortality in a low fish-consuming population: A prospective cohort analysis. Eur. J. Nutr. 2016, 55, 1605–1613. [Google Scholar] [CrossRef]
- Wennberg, M.; Jansson, J.-H.; Norberg, M.; Skerfving, S.; Strömberg, U.; Wiklund, P.-G.; Bergdahl, I.A. Fish consumption and risk of stroke: A second prospective case-control study from northern Sweden. Nutr. J. 2016, 15, 98. [Google Scholar] [CrossRef]
- Mensink, R.P. Effects of Saturated Fatty Acids on Serum Lipids and Lipoproteins: A Systematic Review and Regression Analysis; World Health Organization: Geneva, Switzerland, 2016; Available online: https://apps.who.int/iris/bitstream/handle/10665/246104/9789241565349-eng.pdf (accessed on 11 May 2020).
- McEwen, B.J.; Morel-Kopp, M.C.; Chen, W.; Tofler, G.H.; Ward, C.M. Effects of omega-3 polyunsaturated fatty acids on platelet function in healthy subjects and subjects with cardiovascular disease. Semin. Thromb. Hemost. 2013, 39, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Cohen, M.G.; Rossi, J.S.; Garbarino, J.; Bowling, R.; Motsinger-Reif, A.A.; Schuler, C.; Dupont, A.G.; Gabriel, D. Insights into the inhibition of platelet activation by omega-3 polyunsaturated fatty acids: Beyond aspirin and clopidogrel. Thromb. Res. 2011, 128, 335–340. [Google Scholar] [CrossRef] [PubMed]
- Browning, L.M.; Walker, C.G.; Mander, A.P.; West, A.L.; Madden, J.; Gambell, J.M.; Young, S.; Wang, L.; Jebb, S.A.; Calder, P.C. Incorporation of eicosapentaenoic and docosahexaenoic acids into lipid pools when given as supplements providing doses equivalent to typical intakes of oily fish. Am. J. Clin. Nutr. 2012, 96, 748–758. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi Fard, S.; Wang, F.; Sinclair, A.J.; Elliott, G.; Turchini, G.M. How does high DHA fish oil affect health? A systematic review of evidence. Crit. Rev. Food Sci. Nutr. 2019, 59, 1684–1727. [Google Scholar] [CrossRef] [PubMed]
- Elvevoll, E.O.; Barstad, H.; Breimo, E.S.; Brox, J.; Eilertsen, K.E.; Lund, T.; Olsen, J.O.; Østerud, B. Enhanced incorporation of n-3 fatty acids from fish compared with fish oils. Lipids 2006, 41, 1109–1114. [Google Scholar] [CrossRef] [PubMed]
- Vidgren, H.M.; Ågren, J.J.; Schwab, U.; Rissanen, T.; Hänninen, O.; Uusitupa, M.I.J. Incorporation of n-3 fatty acids into plasma lipid fractions, and erythrocyte membranes and platelets during dietary supplementation with fish, fish oil, and docosahexaenoic acid-rich oil among healthy young men. Lipids 1997, 32, 697–705. [Google Scholar] [CrossRef] [PubMed]
- Saposnik, G.; Fonarow, G.C.; Pan, W.; Liang, L.; Hernandez, A.F.; Schwamm, L.H.; Smith, E.E. Guideline-directed low-density lipoprotein management in high-risk patients with ischemic stroke: Findings from get with the guidelines-stroke 2003 to 2012. Stroke 2014, 45, 3343–3351. [Google Scholar] [CrossRef]
- Amarenco, P.; Lavallée, P.; Touboul, P.J. Stroke prevention, blood cholesterol, and statins. Lancet Neurol. 2004, 3, 271–278. [Google Scholar] [CrossRef]
- Wafa, H.A.; Wolfe, C.D.A.; Rudd, A.; Wang, Y. Long-term trends in incidence and risk factors for ischaemic stroke subtypes: Prospective population study of the South London Stroke Register. PLoS Med. 2018, 15, e1002669. [Google Scholar] [CrossRef]
- Tramacere, I.; Boncoraglio, G.B.; Banzi, R.; Del Giovane, C.; Kwag, K.H.; Squizzato, A.; Moja, L. Comparison of statins for secondary prevention in patients with ischemic stroke or transient ischemic attack: A systematic review and network meta-analysis. BMC Med. 2019, 17, 67. [Google Scholar] [CrossRef]
- Tziomalos, K.; Giampatzis, V.; Bouziana, S.D.; Spanou, M.; Kostaki, S.; Papadopoulou, M.; Angelopoulou, S.M.; Konstantara, F.; Savopoulos, C.; Hatzitolios, A.I. Comparative effects of more versus less aggressive treatment with statins on the long-term outcome of patients with acute ischemic stroke. Atherosclerosis 2015, 243, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Amarenco, P.; Bogousslavsky, J.; Callahan, A.; Goldstein, L.B.; Hennerici, M.; Rudolph, A.E.; Sillesen, H.; Simunovic, L.; Szarek, M.; Welch, K.M.A.; et al. High-dose atorvastatin after stroke or transient ischemic attack. N. Engl. J. Med. 2006, 355, 549–559. [Google Scholar] [CrossRef] [PubMed]
- Muscari, A.; Puddu, G.M.; Santoro, N.; Serafini, C.; Cenni, A.; Rossi, V.; Zoli, M. The atorvastatin during ischemic stroke study: A pilot randomized controlled trial. Clin. Neuropharmacol. 2011, 34, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Tuttolomondo, A.; Di Raimondo, D.; Pecoraro, R.; Maida, C.; Arnao, V.; Corte, V.D.; Simonetta, I.; Corpora, F.; Di Bona, D.; Maugeri, R.; et al. Early high-dosage atorvastatin treatment improved serum immune-inflammatory markers and functional outcome in acute ischemic strokes classified as large artery atherosclerotic stroke: A randomized trial. Medicine 2016, 95, e3186. [Google Scholar] [CrossRef]
- Yu, Y.; Zhu, C.; Liu, C.; Gao, Y. Clinical Study Effect of Prior Atorvastatin Treatment on the Frequency of Hospital Acquired Pneumonia and Evolution of Biomarkers in Patients with Acute Ischemic Stroke: A Multicenter Prospective Study. BioMed Res. Int. 2017, 2017, 5642704. [Google Scholar] [CrossRef]
- Szarek, M.; Amarenco, P.; Callahan, A.; DeMicco, D.; Fayyad, R.; Goldstein, L.B.; Laskey, R.; Sillesen, H.; Welch, K.M. Atorvastatin Reduces First and Subsequent Vascular Events Across Vascular Territories: The SPARCL Trial. J. Am. Coll. Cardiol. 2020, 75, 2110–2118. [Google Scholar] [CrossRef]
- Sabatine, M.S.; Giugliano, R.P.; Keech, A.C.; Honarpour, N.; Wiviott, S.D.; Murphy, S.A.; Kuder, J.F.; Wang, H.; Liu, T.; Wasserman, S.M.; et al. Evolocumab and clinical outcomes in patients with cardiovascular disease. N. Engl. J. Med. 2017, 376, 1713–1722. [Google Scholar] [CrossRef]
- Van Poelgeest, E.P.; Hodges, M.R.; Moerland, M.; Tessier, Y.; Levin, A.A.; Persson, R.; Lindholm, M.W.; Dumong Erichsen, K.; Ørum, H.; Cohen, A.F.; et al. Antisense-mediated reduction of proprotein convertase subtilisin/kexin type 9 (PCSK9): A first-in-human randomized, placebo-controlled trial. Br. J. Clin. Pharmacol. 2015, 80, 1350–1361. [Google Scholar] [CrossRef]
- Ray, K.K.; Landmesser, U.; Leiter, L.A.; Kallend, D.; Dufour, R.; Karakas, M.; Hall, T.; Troquay, R.P.T.; Turner, T.; Visseren, F.L.J.; et al. Inclisiran in patients at high cardiovascular risk with elevated LDL cholesterol. N. Engl. J. Med. 2017, 376, 1430–1440. [Google Scholar] [CrossRef]
- Lintner, N.G.; McClure, K.F.; Petersen, D.; Londregan, A.T.; Piotrowski, D.W.; Wei, L.; Xiao, J.; Bolt, M.; Loria, P.M.; Maguire, B.; et al. Selective stalling of human translation through small-molecule engagement of the ribosome nascent chain. PLoS Biol. 2017, 15, e2001882. [Google Scholar] [CrossRef]
- Alghamdi, R.H.; O’Reilly, P.; Lu, C.; Gomes, J.; Lagace, T.A.; Basak, A. LDL-R promoting activity of peptides derived from human PCSK9 catalytic domain (153–421): Design, synthesis and biochemical evaluation. Eur. J. Med. Chem. 2015, 92, 890–907. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, T.; Chao, G.; Sitkoff, D.; Lo, F.; Monshizadegan, H.; Meyers, D.; Low, S.; Russo, K.; DiBella, R.; Denhez, F.; et al. Pharmacologic profile of the adnectin BMS-962476, a small protein biologic alternative to PCSK9 antibodies for low-density lipoprotein lowering. J. Pharmacol. Exp. Ther. 2014, 350, 412–424. [Google Scholar] [CrossRef] [PubMed]
- Landlinger, C.; Pouwer, M.G.; Juno, C.; van der Hoorn, J.W.A.; Pieterman, E.J.; Jukema, J.W.; Staffler, G.; Princen, H.M.G.; Galabova, G. The AT04A vaccine against proprotein convertase subtilisin/kexin type 9 reduces total cholesterol, vascular inflammation, and atherosclerosis in APOE*3Leiden.CETP mice. Eur. Heart J. 2017, 38, 2499–2507. [Google Scholar] [CrossRef] [PubMed]
- Meschia, J.F.; Bushnell, C.; Boden-Albala, B.; Braun, L.T.; Bravata, D.M.; Chaturvedi, S.; Creager, M.A.; Eckel, R.H.; Elkind, M.S.; Fornage, M.; et al. Guidelines for the primary prevention of stroke: A statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2014, 45, 3754–3832. [Google Scholar] [CrossRef] [PubMed]
- Bloomfield Rubins, H.; Davenport, J.; Babikian, V.; Brass, L.M.; Collins, D.; Wexler, L.; Wagner, S.; Papademetriou, V.; Rutan, G.; Robins, S.J. Reduction in Stroke With Gemfibrozil in Men With Coronary Heart Disease and Low HDL Cholesterol. Circulation 2001, 103, 2828–2833. [Google Scholar] [CrossRef] [PubMed]
- Van Capelleveen, J.C.; Van Der Valk, F.M.; Stroes, E.S.G. Current therapies for lowering lipoprotein (a). J. Lipid Res. 2016, 57, 1612–1618. [Google Scholar] [CrossRef]
- Santos, R.D.; Raal, F.J.; Catapano, A.L.; Witztum, J.L.; Steinhagen-Thiessen, E.; Tsimikas, S. Mipomersen, an Antisense Oligonucleotide to Apolipoprotein B-100, Reduces Lipoprotein(a) in Various Populations with Hypercholesterolemia: Results of 4 Phase III Trials. Arterioscler. Thromb. Vasc. Biol. 2015, 35, 689–699. [Google Scholar] [CrossRef]
- Hamilton, J.A.; Hillard, C.J.; Spector, A.A.; Watkins, P.A. Brain uptake and utilization of fatty acids, lipids and lipoproteins: Application to neurological disorders. J. Mol. Neurosci. 2007, 33, 2–11. [Google Scholar] [CrossRef]
- Tracey, T.J.; Steyn, F.J.; Wolvetang, E.J.; Ngo, S.T. Neuronal Lipid Metabolism: Multiple Pathways Driving Functional Outcomes in Health and Disease. Front. Mol. Neurosci. 2018, 11, 10. [Google Scholar] [CrossRef]
- Ebert, D.; Haller, R.G.; Walton, M.E. Energy contribution of octanoate to intact rat brain metabolism measured by 13C nuclear magnetic resonance spectroscopy. J. Neurosci. 2003, 23, 5928–5935. [Google Scholar] [CrossRef]
- Hankin, J.A.; Farias, S.E.; Barkley, R.M.; Heidenreich, K.; Frey, L.C.; Hamazaki, K.; Kim, H.-Y.; Murphy, R.C. MALDI mass spectrometric imaging of lipids in rat brain injury models. J. Am. Soc. Mass Spectrom. 2011, 22, 1014–1021. [Google Scholar] [CrossRef] [PubMed]
- Mulder, I.A.; Ogrinc Potocnik, N.; Broos, L.A.M.; Prop, A.; Wermer, M.J.H.; Heeren, R.M.A.; van den Maagdenberg, A.M.J.M. Distinguishing core from penumbra by lipid profiles using Mass Spectrometry Imaging in a transgenic mouse model of ischemic stroke. Sci. Rep. 2019, 9, 1090. [Google Scholar] [CrossRef] [PubMed]
- Rapoport, S.I. Arachidonic acid and the brain. J. Nutr. 2008, 138, 2515–2520. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Liu, H.; Zuo, F.; Zhang, L. Adenoviruses-mediated RNA interference targeting cytosolic phospholipase A2alpha attenuates focal ischemic brain damage in mice. Mol. Med. Rep. 2018, 17, 5601–5610. [Google Scholar]
- Liu, H.; Zuo, F.; Wu, H. Blockage of cytosolic phospholipase A2 alpha by monoclonal antibody attenuates focal ischemic brain damage in mice. Biosci. Trends 2017, 11, 439–449. [Google Scholar] [CrossRef][Green Version]
- Belayev, L.; Khoutorova, L.; Atkins, K.D.; Eady, T.N.; Hong, S.; Lu, Y.; Obenaus, A.; Bazan, N.G. Docosahexaenoic Acid therapy of experimental ischemic stroke. Transl. Stroke Res. 2011, 2, 33–41. [Google Scholar] [CrossRef]
- Eady, T.N.; Belayev, L.; Khoutorova, L.; Atkins, K.D.; Zhang, C.; Bazan, N.G. Docosahexaenoic acid signaling modulates cell survival in experimental ischemic stroke penumbra and initiates long-term repair in young and aged rats. PLoS ONE 2012, 7, e46151. [Google Scholar] [CrossRef]
- De Brito Toscano, E.C.; Silva, B.C.; Victoria, E.C.G.; de Souza Cardoso, A.C.; de Miranda, A.S.; Sugimoto, M.A.; Sousa, L.P.; de Carvalho, B.A.; Kangussu, L.M.; da Silva, D.G.; et al. Platelet-activating factor receptor (PAFR) plays a crucial role in experimental global cerebral ischemia and reperfusion. Brain Res. Bull. 2016, 124, 55–61. [Google Scholar] [CrossRef]
- Belayev, L.; Eady, T.N.; Khoutorova, L.; Atkins, K.D.; Obenaus, A.; Cordoba, M.; Vaquero, J.J.; Alvarez-Builla, J.; Bazan, N.G. Superior Neuroprotective Efficacy of LAU-0901, a Novel Platelet-Activating Factor Antagonist, in Experimental Stroke. Transl. Stroke Res. 2012, 3, 154–163. [Google Scholar] [CrossRef]
- Smyth, E.M.; Grosser, T.; Wang, M.; Yu, Y.; FitzGerald, G.A. Prostanoids in health and disease. J. Lipid Res. 2009, 50, S423–S428. [Google Scholar] [CrossRef]
- Huang, H.; Al-Shabrawey, M.; Wang, M.-H. Cyclooxygenase- and cytochrome P450-derived eicosanoids in stroke. Prostaglandins Other Lipid Mediat. 2016, 122, 45–53. [Google Scholar] [CrossRef] [PubMed]
- The Role of Bioactive Lipids in Cancer, Inflammation and Related Diseases, 1st ed.; Honn, K.V., Zeldin, D.C., Eds.; Springer International Publishing: Cham, Switzerland, 2019; ISBN 978-3-030-21636-8. [Google Scholar]
- Nandakishore, R.; Yalavarthi, P.R.; Kiran, Y.R.; Rajapranathi, M. Selective cyclooxygenase inhibitors: Current status. Curr. Drug Discov. Technol. 2014, 11, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Trettin, A.; Bohmer, A.; Suchy, M.-T.; Probst, I.; Staerk, U.; Stichtenoth, D.O.; Frolich, J.C.; Tsikas, D. Effects of paracetamol on NOS, COX, and CYP activity and on oxidative stress in healthy male subjects, rat hepatocytes, and recombinant NOS. Oxid. Med. Cell. Longev. 2014, 2014, 212576. [Google Scholar] [CrossRef] [PubMed]
- Ruan, K.-H.; Deng, H.; So, S.-P. Engineering of a protein with cyclooxygenase and prostacyclin synthase activities that converts arachidonic acid to prostacyclin. Biochemistry 2006, 45, 14003–14011. [Google Scholar] [CrossRef] [PubMed]
- Ling, Q.-L.; Mohite, A.J.; Murdoch, E.; Akasaka, H.; Li, Q.-Y.; So, S.-P.; Ruan, K.-H. Creating a mouse model resistant to induced ischemic stroke and cardiovascular damage. Sci. Rep. 2018, 8, 1653. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Liang, X.; Wang, Q.; Wilson, E.N.; Lam, R.; Wang, J.; Kong, W.; Tsai, C.; Pan, T.; Larkin, P.B.; et al. PGE2 signaling via the neuronal EP2 receptor increases injury in a model of cerebral ischemia. Proc. Natl. Acad. Sci. USA 2019, 116, 10019–10024. [Google Scholar] [CrossRef] [PubMed]
- Frankowski, J.C.; DeMars, K.M.; Ahmad, A.S.; Hawkins, K.E.; Yang, C.; Leclerc, J.L.; Dore, S.; Candelario-Jalil, E. Detrimental role of the EP1 prostanoid receptor in blood-brain barrier damage following experimental ischemic stroke. Sci. Rep. 2015, 5, 17956. [Google Scholar] [CrossRef]
- Zhou, Y.; Wei, E.-Q.; Fang, S.-H.; Chu, L.-S.; Wang, M.-L.; Zhang, W.-P.; Yu, G.-L.; Ye, Y.-L.; Lin, S.-C.; Chen, Z. Spatio-temporal properties of 5-lipoxygenase expression and activation in the brain after focal cerebral ischemia in rats. Life Sci. 2006, 79, 1645–1656. [Google Scholar] [CrossRef]
- Chan, S.J.; Ng, M.P.E.; Zhao, H.; Ng, G.J.L.; De Foo, C.; Wong, P.T.-H.; Seet, R.C.S. Early and Sustained Increases in Leukotriene B4 Levels Are Associated with Poor Clinical Outcome in Ischemic Stroke Patients. Neurotherapeutics 2020, 17, 282–293. [Google Scholar] [CrossRef]
- Shi, S.; Yang, W.; Tu, X.; Wang, C.; Chen, C.; Chen, Y. 5-Lipoxygenase inhibitor zileuton inhibits neuronal apoptosis following focal cerebral ischemia. Inflammation 2013, 36, 1209–1217. [Google Scholar] [CrossRef]
- Silva, B.C.; de Miranda, A.S.; Rodrigues, F.G.; Silveira, A.L.M.; Resende, G.H.d.S.; Moraes, M.F.D.; de Oliveira, A.C.P.; Parreiras, P.M.; Barcelos, L.d.S.; Teixeira, M.M.; et al. The 5-lipoxygenase (5-LOX) Inhibitor Zileuton Reduces Inflammation and Infarct Size with Improvement in Neurological Outcome Following Cerebral Ischemia. Curr. Neurovasc. Res. 2015, 12, 398–403. [Google Scholar] [CrossRef] [PubMed]
- Tu, X.; Yang, W.; Wang, C.; Shi, S.; Zhang, Y.; Chen, C.; Yang, Y.; Jin, C.; Wen, S. Zileuton reduces inflammatory reaction and brain damage following permanent cerebral ischemia in rats. Inflammation 2010, 33, 344–352. [Google Scholar] [CrossRef] [PubMed]
- Fang, S.H.; Wei, E.Q.; Zhou, Y.; Wang, M.L.; Zhang, W.P.; Yu, G.L.; Chu, L.S.; Chen, Z. Increased expression of cysteinyl leukotriene receptor-1 in the brain mediates neuronal damage and astrogliosis after focal cerebral ischemia in rats. Neuroscience 2006, 140, 969–979. [Google Scholar] [CrossRef] [PubMed]
- Chandrasekharan, J.A.; Sharma-Walia, N. Lipoxins: Nature’s way to resolve inflammation. J. Inflamm. Res. 2015, 8, 181–192. [Google Scholar] [PubMed]
- Wu, L.; Miao, S.; Zou, L.-B.; Wu, P.; Hao, H.; Tang, K.; Zeng, P.; Xiong, J.; Li, H.-H.; Wu, Q.; et al. Lipoxin A4 inhibits 5-lipoxygenase translocation and leukotrienes biosynthesis to exert a neuroprotective effect in cerebral ischemia/reperfusion injury. J. Mol. Neurosci. 2012, 48, 185–200. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Liu, Z.J.; Miao, S.; Zou, L.B.; Cai, L.; Wu, P.; Ye, D.Y.; Wu, Q.; Li, H.H. Lipoxin A4 ameliorates cerebral ischaemia/reperfusion injury through upregulation of nuclear factor erythroid 2-related factor 2. Neurol. Res. 2013, 35, 968–975. [Google Scholar] [CrossRef]
- Wu, L.; Li, H.-H.; Wu, Q.; Miao, S.; Liu, Z.-J.; Wu, P.; Ye, D.-Y. Lipoxin A4 Activates Nrf2 Pathway and Ameliorates Cell Damage in Cultured Cortical Astrocytes Exposed to Oxygen-Glucose Deprivation/Reperfusion Insults. J. Mol. Neurosci. 2015, 56, 848–857. [Google Scholar] [CrossRef]
- Powell, W.S.; Rokach, J. Biosynthesis, biological effects, and receptors of hydroxyeicosatetraenoic acids (HETEs) and oxoeicosatetraenoic acids (oxo-ETEs) derived from arachidonic acid. Biochim. Biophys. Acta 2015, 1851, 340–355. [Google Scholar] [CrossRef]
- Usui, M.; Asano, T.; Takakura, K. Identification and quantitative analysis of hydroxy-eicosatetraenoic acids in rat brains exposed to regional ischemia. Stroke 1987, 18, 490–494. [Google Scholar] [CrossRef]
- Wang, D.; Liu, Y.; Chen, L.; Li, P.; Qu, Y.; Zhu, Y.; Zhu, Y. Key role of 15-LO/15-HETE in angiogenesis and functional recovery in later stages of post-stroke mice. Sci. Rep. 2017, 7, 46698. [Google Scholar] [CrossRef]
- Chen, L.; Zhu, Y.-M.; Li, Y.-N.; Li, P.-Y.; Wang, D.; Liu, Y.; Qu, Y.-Y.; Zhu, D.-L.; Zhu, Y.-L. The 15-LO-1/15-HETE system promotes angiogenesis by upregulating VEGF in ischemic brains. Neurol. Res. 2017, 39, 795–802. [Google Scholar] [CrossRef] [PubMed]
- Ward, N.C.; Croft, K.D.; Blacker, D.; Hankey, G.J.; Barden, A.; Mori, T.A.; Puddey, I.B.; Beer, C.D. Cytochrome P450 metabolites of arachidonic acid are elevated in stroke patients compared with healthy controls. Clin. Sci. 2011, 121, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Yi, X.; Han, Z.; Zhou, Q.; Lin, J.; Liu, P. 20-Hydroxyeicosatetraenoic Acid as a Predictor of Neurological Deterioration in Acute Minor Ischemic Stroke. Stroke 2016, 47, 3045–3047. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, Y.; Omura, T.; Fukasawa, M.; Horiuchi, N.; Miyata, N.; Minagawa, T.; Yoshida, S.; Nakaike, S. Continuous inhibition of 20-HETE synthesis by TS-011 improves neurological and functional outcomes after transient focal cerebral ischemia in rats. Neurosci. Res. 2007, 59, 475–480. [Google Scholar] [CrossRef] [PubMed]
- Dunn, K.M.; Renic, M.; Flasch, A.K.; Harder, D.R.; Falck, J.; Roman, R.J. Elevated production of 20-HETE in the cerebral vasculature contributes to severity of ischemic stroke and oxidative stress in spontaneously hypertensive rats. Am. J. Physiol. Heart Circ. Physiol. 2008, 295, H2455–H2465. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, Y.; Zhan, M.; Liu, Y.; Li, Z.; Li, J.; Cheng, G.; Teng, G.; Lu, L. Astrocytic cytochrome P450 4A/20-hydroxyeicosatetraenoic acid contributes to angiogenesis in the experimental ischemic stroke. Brain Res. 2019, 1708, 160–170. [Google Scholar] [CrossRef]
- Qu, Y.; Liu, Y.; Zhu, Y.; Chen, L.; Sun, W.; Zhu, Y. Epoxyeicosatrienoic Acid Inhibits the Apoptosis of Cerebral Microvascular Smooth Muscle Cells by Oxygen Glucose Deprivation via Targeting the JNK/c-Jun and mTOR Signaling Pathways. Mol. Cells 2017, 40, 837–846. [Google Scholar]
- Qu, Y.-Y.; Yuan, M.-Y.; Liu, Y.; Xiao, X.-J.; Zhu, Y.-L. The protective effect of epoxyeicosatrienoic acids on cerebral ischemia/reperfusion injury is associated with PI3K/Akt pathway and ATP-sensitive potassium channels. Neurochem. Res. 2015, 40, 1–14. [Google Scholar] [CrossRef]
- Bansal, S.; Sangha, K.S.; Khatri, P. Drug treatment of acute ischemic stroke. Am. J. Cardiovasc. Drugs 2013, 13, 57–69. [Google Scholar] [CrossRef]
- Ahmad, M.; Zhang, Y.; Liu, H.; Rose, M.E.; Graham, S.H. Prolonged opportunity for neuroprotection in experimental stroke with selective blockade of cyclooxygenase-2 activity. Brain Res. 2009, 1279, 168–173. [Google Scholar] [CrossRef]
- Pirlamarla, P.; Bond, R.M. FDA labeling of NSAIDs: Review of nonsteroidal anti-inflammatory drugs in cardiovascular disease. Trends Cardiovasc. Med. 2016, 26, 675–680. [Google Scholar] [CrossRef] [PubMed]
- Stitham, J.; Midgett, C.; Martin, K.A.; Hwa, J. Prostacyclin: An inflammatory paradox. Front. Pharmacol. 2011, 2, 24. [Google Scholar] [CrossRef] [PubMed]
- Gryglewski, R.J.; Nowak, S.; Kostka-Trabka, E.; Kusmiderski, J.; Dembinska-Kiec, A.; Bieron, K.; Basista, M.; Blaszczyk, B. Treatment of ischaemic stroke with prostacyclin. Stroke 1983, 14, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Zhang, X.; Dong, L.; Wen, Y.; Cui, L. The Many Roles of Statins in Ischemic Stroke. Curr. Neuropharmacol. 2014, 12, 564–574. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Yang, J.; Zhong, J.; Luo, Y.; Du, W.; Hu, C.; Xia, H.; Li, Y.; Zhang, J.; Li, M.; et al. MicroRNA-193b-3p alleviates focal cerebral ischemia and reperfusion-induced injury in rats by inhibiting 5-lipoxygenase expression. Exp. Neurol. 2020, 327, 113223. [Google Scholar] [CrossRef] [PubMed]
- Corser-Jensen, C.E.; Goodell, D.J.; Freund, R.K.; Serbedzija, P.; Murphy, R.C.; Farias, S.E.; Dell’Acqua, M.L.; Frey, L.C.; Serkova, N.; Heidenreich, K.A. Blocking leukotriene synthesis attenuates the pathophysiology of traumatic brain injury and associated cognitive deficits. Exp. Neurol. 2014, 256, 7–16. [Google Scholar] [CrossRef]
- Shi, Q.J.; Wang, H.; Liu, Z.X.; Fang, S.H.; Song, X.M.; Lu, Y.B.; Zhang, W.P.; Sa, X.Y.; Ying, H.Z.; Wei, E.Q. HAMI 3379, a CysLT2R antagonist, dose- and time-dependently attenuates brain injury and inhibits microglial inflammation after focal cerebral ischemia in rats. Neuroscience 2015, 291, 53–69. [Google Scholar] [CrossRef]
- Marumo, T.; Eto, K.; Wake, H.; Omura, T.; Nabekura, J. The inhibitor of 20-HETE synthesis, TS-011, improves cerebral microcirculatory autoregulation impaired by middle cerebral artery occlusion in mice. Br. J. Pharmacol. 2010, 161, 1391–1402. [Google Scholar] [CrossRef]
- Renic, M.; Klaus, J.A.; Omura, T.; Kawashima, N.; Onishi, M.; Miyata, N.; Koehler, R.C.; Harder, D.R.; Roman, R.J. Effect of 20-HETE inhibition on infarct volume and cerebral blood flow after transient middle cerebral artery occlusion. J. Cereb. Blood Flow Metab. 2009, 29, 629–639. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, D.; Wang, H.; Qu, Y.; Xiao, X.; Zhu, Y. The protective effect of HET0016 on brain edema and blood-brain barrier dysfunction after cerebral ischemia/reperfusion. Brain Res. 2014, 1544, 45–53. [Google Scholar] [CrossRef]
- Hawkins, K.E.; DeMars, K.M.; Alexander, J.C.; de Leon, L.G.; Pacheco, S.C.; Graves, C.; Yang, C.; McCrea, A.O.; Frankowski, J.C.; Garrett, T.J.; et al. Targeting resolution of neuroinflammation after ischemic stroke with a lipoxin A4 analog: Protective mechanisms and long-term effects on neurological recovery. Brain Behav. 2017, 7, e00688. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, Y.-P.; Guo, P.; Ye, X.-H.; Wang, J.; Yuan, S.-Y.; Yao, S.-L.; Shang, Y. A lipoxin A4 analog ameliorates blood-brain barrier dysfunction and reduces MMP-9 expression in a rat model of focal cerebral ischemia-reperfusion injury. J. Mol. Neurosci. 2012, 46, 483–491. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.-B.; Pang, X.-B.; Zhao, Y.; Wang, Y.-H.; Zhang, L.; Yang, X.-Y.; Fang, L.-H.; Du, G.-H. Protection of salvianolic acid A on rat brain from ischemic damage via soluble epoxide hydrolase inhibition. J. Asian Nat. Prod. Res. 2012, 14, 1084–1092. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Hong, G.; Lee, K.S.S.; Hammock, B.D.; Gebremedhin, D.; Harder, D.R.; Koehler, R.C.; Sapirstein, A. Inhibition of soluble epoxide hydrolase augments astrocyte release of vascular endothelial growth factor and neuronal recovery after oxygen-glucose deprivation. J. Neurochem. 2017, 140, 814–825. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Xu, X.; Chen, C.; Yu, X.; Edin, M.L.; Degraff, L.M.; Lee, C.R.; Zeldin, D.C.; Wang, D.W. Cytochrome P450 2J2 is protective against global cerebral ischemia in transgenic mice. Prostaglandins Other Lipid Mediat. 2012, 99, 68–78. [Google Scholar] [CrossRef]
- Liu, Y.; Wan, Y.; Fang, Y.; Yao, E.; Xu, S.; Ning, Q.; Zhang, G.; Wang, W.; Huang, X.; Xie, M. Epoxyeicosanoid Signaling Provides Multi-target Protective Effects on Neurovascular Unit in Rats After Focal Ischemia. J. Mol. Neurosci. 2016, 58, 254–265. [Google Scholar] [CrossRef]
- Hackam, D.G.; Spence, J.D. Antiplatelet therapy in ischemic stroke and transient ischemic attack: An overview of major trials and meta-analyses. Stroke 2019, 50, 773–778. [Google Scholar] [CrossRef]
| Therapeutic Intervention 1 | Effect | Application 2 | Reference |
|---|---|---|---|
| Lipid-lowering therapies for stroke prevention | |||
| Lowering cholesterol with statins | Anti-atherogenic: LDL cholesterol decrease | Clinical use | [91,92,97] |
| Lowering cholesterol with PCSK9 inhibitors, siRNAs, mimetic peptides, adnectins, vaccinations | Anti-atherogenic: increased LDL-C clearance from circulation | Experimental | [99,100,101,102,103,104,105] |
| Reduction of hypertriglyceridemia with fibric acid and derivatives | Anti-atherogenic: triglyceride decrease; HDL cholesterol increase | Clinical use | [106,107] |
| Lowering Lp(a) levels with niacin, PCSK9 or CETP inhibitors, or antisense oligonucleotides | Anti-atherogenic: reduced Lp(a) in circulation | Experimental | [108,109] |
| Eicosanoid-targeted therapies for post-ischemic brain tissue rescue | |||
| Lowering prostaglandins level with COX inhibitors | Anti-atherogenic: anti-platelet effect of aspirin Neuroprotection: anti-inflammatory actions | Clinical use Experimental | [152,153,170] |
| Increasing prostacyclin levels with intravenous infusions | Vasodilation of cerebral microvessels | Experimental | [156] |
| Lowering TXA2 levels with statins | Anti-thrombogenic: TXA2 level decrease, anti-platelet activity | Experimental | [157] |
| Decreasing leukotriene B4 synthesis with 5-LOX inhibitors, microRNAs, FLAP inhibitors, or CysLT receptors antagonists | Neuroprotection: anti-inflammatory and anti-apoptotic actions, reduced neuronal loss | Experimental | [133,134,135,136,158,159,160] |
| Increasing lipoxin A4 levels with LXA4 or LXA4ME analogues | Neuroprotection: anti-inflammatory action, blood–brain barrier rescue | Experimental | [164,165] |
| Lowering 20-HETE levels with inhibitors or antagonists | Neuroprotection: improved microcirculation | Experimental | [147,161,162,163] |
| Increasing EETs levels with sEH inhibitors | Neuroprotection: anti-apoptotic, anti-inflammatory, pro-angiogenic, astrogliosis-preventive actions | Experimental | [166,167,168,169] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kloska, A.; Malinowska, M.; Gabig-Cimińska, M.; Jakóbkiewicz-Banecka, J. Lipids and Lipid Mediators Associated with the Risk and Pathology of Ischemic Stroke. Int. J. Mol. Sci. 2020, 21, 3618. https://doi.org/10.3390/ijms21103618
Kloska A, Malinowska M, Gabig-Cimińska M, Jakóbkiewicz-Banecka J. Lipids and Lipid Mediators Associated with the Risk and Pathology of Ischemic Stroke. International Journal of Molecular Sciences. 2020; 21(10):3618. https://doi.org/10.3390/ijms21103618
Chicago/Turabian StyleKloska, Anna, Marcelina Malinowska, Magdalena Gabig-Cimińska, and Joanna Jakóbkiewicz-Banecka. 2020. "Lipids and Lipid Mediators Associated with the Risk and Pathology of Ischemic Stroke" International Journal of Molecular Sciences 21, no. 10: 3618. https://doi.org/10.3390/ijms21103618
APA StyleKloska, A., Malinowska, M., Gabig-Cimińska, M., & Jakóbkiewicz-Banecka, J. (2020). Lipids and Lipid Mediators Associated with the Risk and Pathology of Ischemic Stroke. International Journal of Molecular Sciences, 21(10), 3618. https://doi.org/10.3390/ijms21103618






