AMPK and the Need to Breathe and Feed: What’s the Matter with Oxygen?
Abstract
1. Introduction
2. The AMP-Activated Protein Kinase and Cellular Metabolic Homeostasis
3. Phosphorylation by AMPK of Non-Metabolic Targets That Modulate Physiological Systems
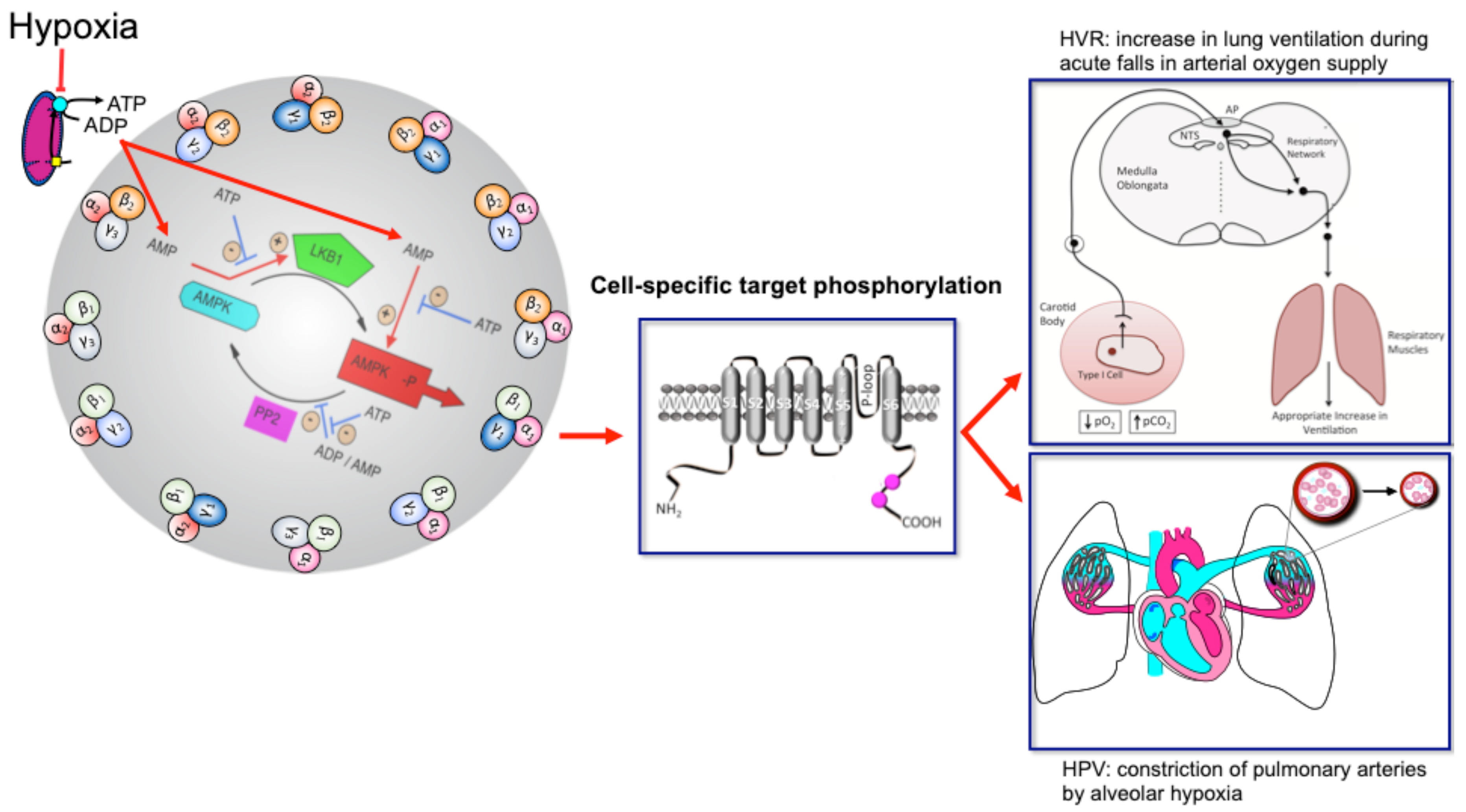
4. AMPK Aids HPV and Thus Gaseous Exchange at The Lungs
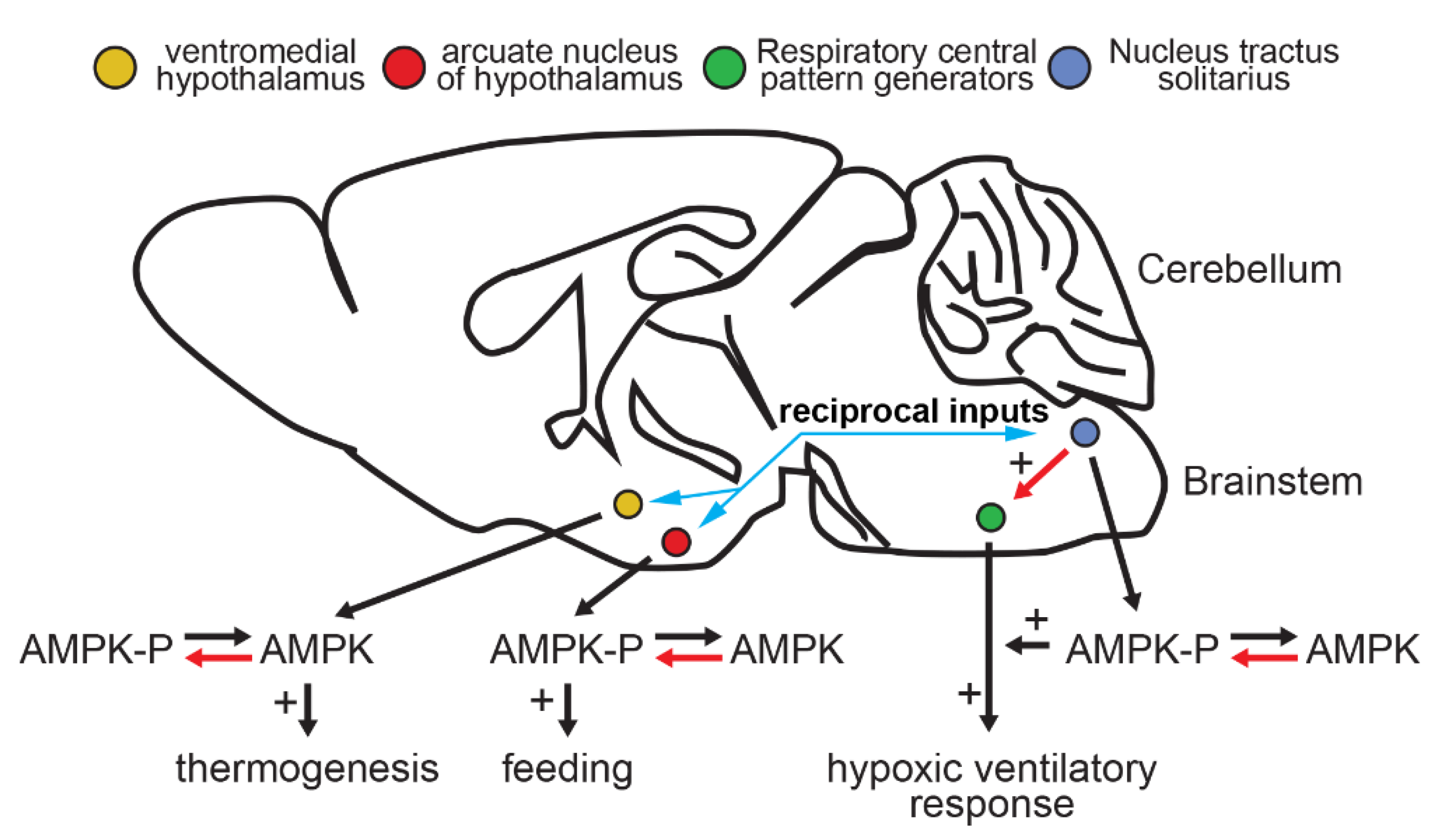
5. AMPK and Central Neural Control Mechanisms
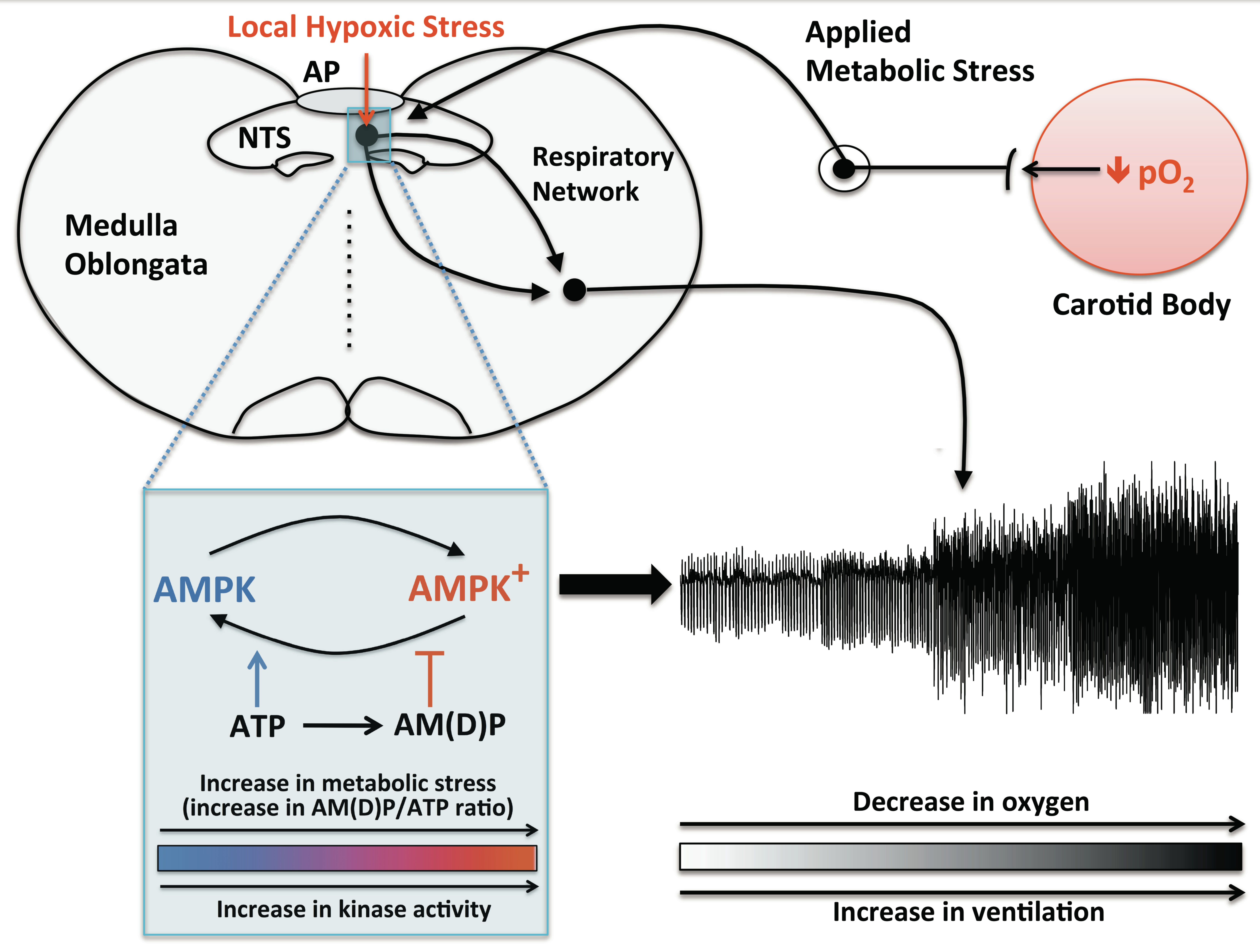
6. AMPK, HVR and Oxygen Supply
7. The Role of Hypothalamic AMPK in Regulating Appetite and Feeding Behaviour
8. AMPK and Food Choice
9. Hypothalamic AMPK and Energy Expenditure through Thermogenesis
10. Regulation of Oxygen Supply, Feeding and Food Choice during Adaptive Acclimation to Hypoxia at Altitude
11. Pathological Links between AMPK, Oxygen Supply and Food Choice
12. Clinical Perspective
- Idiopathic neonatal pulmonary hypertension affects 60 per million, with 50%–75% survival at five years [222];
- Sudden infant death syndrome, which has been linked to hypoventilation and apnoea of pre-term infants during hypoxia [194], is the leading cause of death in otherwise healthy infants, and currently accounts for approximately 300 deaths per annum in the UK (UK Office for National Statistics) and 2000 deaths per annum in the USA [224];
Funding
Acknowledgments
Conflicts of Interest
References
- Smith, J.C.; Abdala, A.P.; Borgmann, A.; Rybak, I.A.; Paton, J.F. Brainstem respiratory networks: Building blocks and microcircuits. Trends Neurosci. 2013, 36, 152–162. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.F.; Feldman, J.L. Efferent projections of excitatory and inhibitory prebotzinger complex neurons. J. Comp. Neurol. 2018, 526, 1389–1402. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Prabhakar, N.R. Peripheral chemoreceptors: Function and plasticity of the carotid body. Compr. Physiol. 2012, 2, 141–219. [Google Scholar] [PubMed]
- Nurse, C.A. Synaptic and paracrine mechanisms at carotid body arterial chemoreceptors. J. Physiol. 2014, 592, 3419–3426. [Google Scholar] [CrossRef]
- Zera, T.; Moraes, D.J.A.; Da Silva, M.P.; Fisher, J.P.; Paton, J.F.R. The logic of carotid body connectivity to the brain. Physiology 2019, 34, 264–282. [Google Scholar] [CrossRef]
- Papamatheakis, D.G.; Blood, A.B.; Kim, J.H.; Wilson, S.M. Antenatal hypoxia and pulmonary vascular function and remodeling. Curr. Vasc. Pharm. 2013, 11, 616–640. [Google Scholar] [CrossRef]
- Von Euler, U.S.; Liljestrand, G. Observations on the pulmonary arterial blood pressure in the cat. Acta Physiol. Scand. 1946, 12, 301–320. [Google Scholar] [CrossRef]
- Sylvester, J.T.; Shimoda, L.A.; Aaronson, P.I.; Ward, J.P. Hypoxic pulmonary vasoconstriction. Physiol. Rev. 2012, 92, 367–520. [Google Scholar] [CrossRef]
- Roy, C.S.; Sherrington, C.S. On the regulation of the blood-supply of the brain. J. Physiol. 1890, 11, 85–185. [Google Scholar] [CrossRef]
- Evans, A.M. Ampk breathing and oxygen supply. Respir. Physiol. Neurobiol. 2019, 265, 112–120. [Google Scholar] [CrossRef]
- Evans, A.M. Amp-activated protein kinase and the regulation of ca2+ signalling in o2-sensing cells. J. Physiol. 2006, 574, 113–123. [Google Scholar] [CrossRef] [PubMed]
- Evans, A.M. Hypoxia, cell metabolism, and cadpr accumulation; Springer: Boston, MA, USA, 2004. [Google Scholar]
- Mills, E.; Jobsis, F.F. Simultaneous measurement of cytochrome a3 reduction and chemoreceptor afferent activity in the carotid body. Nature 1970, 225, 1147–1149. [Google Scholar] [CrossRef] [PubMed]
- Mills, E.; Jobsis, F.F. Mitochondrial respiratory chain of carotid body and chemoreceptor response to changes in oxygen tension. J. Neurophysiol. 1972, 35, 405–428. [Google Scholar] [CrossRef] [PubMed]
- Firth, A.L.; Yuill, K.H.; Smirnov, S.V. Mitochondria-dependent regulation of kv currents in rat pulmonary artery smooth muscle cells. Am. J. Physiol. Lung Cell Mol. Physiol. 2008, 295, L61–L70. [Google Scholar] [CrossRef][Green Version]
- Post, J.M.; Hume, J.R.; Archer, S.L.; Weir, E.K. Direct role for potassium channel inhibition in hypoxic pulmonary vasoconstriction. Am. J. Physiol. 1992, 262, C882–C890. [Google Scholar] [CrossRef]
- Duchen, M.R.; Biscoe, T.J. Relative mitochondrial membrane potential and [ca2+]i in type i cells isolated from the rabbit carotid body. J. Physiol. 1992, 450, 33–61. [Google Scholar] [CrossRef]
- Duchen, M.R.; Biscoe, T.J. Mitochondrial function in type i cells isolated from rabbit arterial chemoreceptors. J. Physiol. 1992, 450, 13–31. [Google Scholar] [CrossRef]
- Buckler, K.J.; Turner, P.J. Oxygen sensitivity of mitochondrial function in rat arterial chemoreceptor cells. J. Physiol. 2013, 591, 3549–3563. [Google Scholar] [CrossRef]
- Wyatt, C.N.; Buckler, K.J. The effect of mitochondrial inhibitors on membrane currents in isolated neonatal rat carotid body type i cells. J. Physiol. 2004, 556, 175–191. [Google Scholar] [CrossRef]
- Tello, D.; Balsa, E.; Acosta-Iborra, B.; Fuertes-Yebra, E.; Elorza, A.; Ordonez, A.; Corral-Escariz, M.; Soro, I.; Lopez-Bernardo, E.; Perales-Clemente, E.; et al. Induction of the mitochondrial ndufa4l2 protein by hif-1alpha decreases oxygen consumption by inhibiting complex i activity. Cell Metab. 2011, 14, 768–779. [Google Scholar] [CrossRef]
- Fukuda, R.; Zhang, H.; Kim, J.W.; Shimoda, L.; Dang, C.V.; Semenza, G.L. Hif-1 regulates cytochrome oxidase subunits to optimize efficiency of respiration in hypoxic cells. Cell 2007, 129, 111–122. [Google Scholar] [CrossRef] [PubMed]
- Huttemann, M.; Kadenbach, B.; Grossman, L.I. Mammalian subunit iv isoforms of cytochrome c oxidase. Gene 2001, 267, 111–123. [Google Scholar] [CrossRef]
- Zhou, T.; Chien, M.S.; Kaleem, S.; Matsunami, H. Single cell transcriptome analysis of mouse carotid body glomus cells. J. Physiol. 2016, 594, 4225–4251. [Google Scholar] [CrossRef] [PubMed]
- Aras, S.; Pak, O.; Sommer, N.; Finley, R., Jr.; Huttemann, M.; Weissmann, N.; Grossman, L.I. Oxygen-dependent expression of cytochrome c oxidase subunit 4-2 gene expression is mediated by transcription factors rbpj, cxxc5 and chchd2. Nucleic Acids Res. 2013, 41, 2255–2266. [Google Scholar] [CrossRef]
- Huttemann, M.; Lee, I.; Gao, X.; Pecina, P.; Pecinova, A.; Liu, J.; Aras, S.; Sommer, N.; Sanderson, T.H.; Tost, M.; et al. Cytochrome c oxidase subunit 4 isoform 2-knockout mice show reduced enzyme activity, airway hyporeactivity, and lung pathology. FASEB J. 2012, 26, 3916–3930. [Google Scholar] [CrossRef]
- Moreno-Dominguez, A.; Ortega-Saenz, P.; Gao, L.; Colinas, O.; Garcia-Flores, P.; Bonilla-Henao, V.; Aragones, J.; Huttemann, M.; Grossman, L.I.; Weissmann, N.; et al. Acute o2 sensing through hif2alpha-dependent expression of atypical cytochrome oxidase subunits in arterial chemoreceptors. Sci. Signal. 2020, 13, eaay9452. [Google Scholar] [CrossRef]
- Sommer, N.; Huttemann, M.; Pak, O.; Scheibe, S.; Knoepp, F.; Sinkler, C.; Malczyk, M.; Gierhardt, M.; Esfandiary, A.; Kraut, S.; et al. Mitochondrial complex iv subunit 4 isoform 2 is essential for acute pulmonary oxygen sensing. Circ. Res. 2017, 121, 424–438. [Google Scholar] [CrossRef]
- Pajuelo Reguera, D.; Cunatova, K.; Vrbacky, M.; Pecinova, A.; Houstek, J.; Mracek, T.; Pecina, P. Cytochrome c oxidase subunit 4 isoform exchange results in modulation of oxygen affinity. Cells 2020, 9, 443. [Google Scholar] [CrossRef]
- Horvat, S.; Beyer, C.; Arnold, S. Effect of hypoxia on the transcription pattern of subunit isoforms and the kinetics of cytochrome c oxidase in cortical astrocytes and cerebellar neurons. J. Neurochem. 2006, 99, 937–951. [Google Scholar] [CrossRef]
- Carreau, A.; El Hafny-Rahbi, B.; Matejuk, A.; Grillon, C.; Kieda, C. Why is the partial oxygen pressure of human tissues a crucial parameter? Small molecules and hypoxia. J. Cell. Mol. Med. 2011, 15, 1239–1253. [Google Scholar] [CrossRef]
- Kocha, K.M.; Reilly, K.; Porplycia, D.S.; McDonald, J.; Snider, T.; Moyes, C.D. Evolution of the oxygen sensitivity of cytochrome c oxidase subunit 4. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2015, 308, R305–R320. [Google Scholar] [CrossRef] [PubMed]
- Dzeja, P.P.; Terzic, A. Phosphotransfer networks and cellular energetics. J. Exp. Biol. 2003, 206, 2039–2047. [Google Scholar] [CrossRef] [PubMed]
- Panayiotou, C.; Solaroli, N.; Karlsson, A. The many isoforms of human adenylate kinases. Int. J. Biochem. Cell Biol. 2014, 49, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Auciello, F.R.; Ross, F.A.; Ikematsu, N.; Hardie, D.G. Oxidative stress activates ampk in cultured cells primarily by increasing cellular amp and/or adp. FEBS Lett. 2014, 588, 3361–3366. [Google Scholar] [CrossRef] [PubMed]
- Hawley, S.A.; Ross, F.A.; Chevtzoff, C.; Green, K.A.; Evans, A.; Fogarty, S.; Towler, M.C.; Brown, L.J.; Ogunbayo, O.A.; Evans, A.M.; et al. Use of cells expressing gamma subunit variants to identify diverse mechanisms of ampk activation. Cell Metab. 2010, 11, 554–565. [Google Scholar] [CrossRef]
- Andersen, M.N.; Skibsbye, L.; Tang, C.; Petersen, F.; MacAulay, N.; Rasmussen, H.B.; Jespersen, T. Pkc and ampk regulation of kv1.5 potassium channels. Channels 2015, 9, 121–128. [Google Scholar] [CrossRef]
- Mia, S.; Munoz, C.; Pakladok, T.; Siraskar, G.; Voelkl, J.; Alesutan, I.; Lang, F. Downregulation of kv1.5 k channels by the amp-activated protein kinase. Cell. Physiol. Biochem. Int. J. Exp. Cell. Physiol. Biochem. Pharmacol. 2012, 30, 1039–1050. [Google Scholar] [CrossRef]
- Moral-Sanz, J.; Mahmoud, A.D.; Ross, F.A.; Eldstrom, J.; Fedida, D.; Hardie, D.G.; Evans, A.M. Amp-activated protein kinase inhibits kv 1.5 channel currents of pulmonary arterial myocytes in response to hypoxia and inhibition of mitochondrial oxidative phosphorylation. J. Physiol. 2016, 594, 4901–4915. [Google Scholar] [CrossRef]
- Lipton, A.J.; Johnson, M.A.; Macdonald, T.; Lieberman, M.W.; Gozal, D.; Gaston, B. S-nitrosothiols signal the ventilatory response to hypoxia. Nature 2001, 413, 171–174. [Google Scholar] [CrossRef]
- Murphy, B.A.; Fakira, K.A.; Song, Z.; Beuve, A.; Routh, V.H. Amp-activated protein kinase and nitric oxide regulate the glucose sensitivity of ventromedial hypothalamic glucose-inhibited neurons. Am. J. Physiol. Cell Physiol. 2009, 297, C750–C758. [Google Scholar] [CrossRef]
- Zhang, J.; Dong, J.; Martin, M.; He, M.; Gongol, B.; Marin, T.L.; Chen, L.; Shi, X.; Yin, Y.; Shang, F.; et al. Ampk phosphorylation of ace2 in endothelium mitigates pulmonary hypertension. Am. J. Respir. Crit. Care Med. 2018, 198, 509–520. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, M.; Roy, R. Ampk acts as a molecular trigger to coordinate glutamatergic signals and adaptive behaviours during acute starvation. eLife 2016, 5, e16349. [Google Scholar] [CrossRef] [PubMed]
- Schneider, H.; Schubert, K.M.; Blodow, S.; Kreutz, C.P.; Erdogmus, S.; Wiedenmann, M.; Qiu, J.; Fey, T.; Ruth, P.; Lubomirov, L.T.; et al. Ampk dilates resistance arteries via activation of serca and bkca channels in smooth muscle. Hypertension 2015, 66, 108–116. [Google Scholar] [CrossRef] [PubMed]
- Ross, F.A.; Rafferty, J.N.; Dallas, M.L.; Ogunbayo, O.; Ikematsu, N.; McClafferty, H.; Tian, L.; Widmer, H.; Rowe, I.C.; Wyatt, C.N.; et al. Selective expression in carotid body type i cells of a single splice variant of the large conductance calcium- and voltage-activated potassium channel confers regulation by amp-activated protein kinase. J. Biol. Chem. 2011, 286, 11929–11936. [Google Scholar] [CrossRef]
- Ikematsu, N.; Dallas, M.L.; Ross, F.A.; Lewis, R.W.; Rafferty, J.N.; David, J.A.; Suman, R.; Peers, C.; Hardie, D.G.; Evans, A.M. Phosphorylation of the voltage-gated potassium channel kv2.1 by amp-activated protein kinase regulates membrane excitability. Proc. Natl. Acad. Sci. USA 2011, 108, 18132–18137. [Google Scholar] [CrossRef]
- Hardie, D.G. Amp-activated protein kinase: A key regulator of energy balance with many roles in human disease. J. Intern. Med. 2014, 276, 543–559. [Google Scholar] [CrossRef]
- Ross, F.A.; MacKintosh, C.; Hardie, D.G. Amp-activated protein kinase: A cellular energy sensor that comes in twelve flavours. FEBS J. 2016, 283, 2987–3001. [Google Scholar] [CrossRef]
- Zong, Y.; Zhang, C.S.; Li, M.; Wang, W.; Wang, Z.; Hawley, S.A.; Ma, T.; Feng, J.W.; Tian, X.; Qi, Q.; et al. Hierarchical activation of compartmentalized pools of ampk depends on severity of nutrient or energy stress. Cell Res. 2019, 29, 460–473. [Google Scholar] [CrossRef]
- Moral-Sanz, J.; Lewis, S.A.; MacMillan, S.; Ross, F.A.; Thomson, A.; Viollet, B.; Foretz, M.; Moran, C.; Hardie, D.G.; Evans, A.M. The lkb1-ampk-alpha1 signaling pathway triggers hypoxic pulmonary vasoconstriction downstream of mitochondria. Sci. Signal. 2018, 11, eaau0296. [Google Scholar] [CrossRef]
- Gu, X.; Yan, Y.; Novick, S.J.; Kovach, A.; Goswami, D.; Ke, J.; Tan, M.H.E.; Wang, L.; Li, X.; De Waal, P.W.; et al. Deconvoluting amp-activated protein kinase (ampk) adenine nucleotide binding and sensing. J. Biol. Chem. 2017, 292, 12653–12666. [Google Scholar] [CrossRef]
- Xiao, B.; Heath, R.; Saiu, P.; Leiper, F.C.; Leone, P.; Jing, C.; Walker, P.A.; Haire, L.; Eccleston, J.F.; Davis, C.T.; et al. Structural basis for amp binding to mammalian amp-activated protein kinase. Nature 2007, 449, 496–500. [Google Scholar] [CrossRef] [PubMed]
- Gowans, G.J.; Hawley, S.A.; Ross, F.A.; Hardie, D.G. Amp is a true physiological regulator of amp-activated protein kinase by both allosteric activation and enhancing net phosphorylation. Cell Metab. 2013, 18, 556–566. [Google Scholar] [CrossRef] [PubMed]
- Ross, F.; Jensen, T.E.; Hardie, D.G. Differential regulation by amp and adp of ampk complexes containing different gamma subunit isoforms. Biochem. J. 2015, 473, 189–199. [Google Scholar] [CrossRef] [PubMed]
- Oakhill, J.S.; Steel, R.; Chen, Z.P.; Scott, J.W.; Ling, N.; Tam, S.; Kemp, B.E. Ampk is a direct adenylate charge-regulated protein kinase. Science 2011, 332, 1433–1435. [Google Scholar] [CrossRef]
- Xiao, B.; Sanders, M.J.; Underwood, E.; Heath, R.; Mayer, F.V.; Carmena, D.; Jing, C.; Walker, P.A.; Eccleston, J.F.; Haire, L.F.; et al. Structure of mammalian ampk and its regulation by adp. Nature 2011, 472, 230–233. [Google Scholar] [CrossRef]
- Sakamoto, K.; Goransson, O.; Hardie, D.G.; Alessi, D.R. Activity of lkb1 and ampk-related kinases in skeletal muscle: Effects of contraction, phenformin, and aicar. Am. J. Physiol. Endocrinol. Metab. 2004, 287, E310–E317. [Google Scholar] [CrossRef]
- Hawley, S.A.; Pan, D.A.; Mustard, K.J.; Ross, L.; Bain, J.; Edelman, A.M.; Frenguelli, B.G.; Hardie, D.G. Calmodulin-dependent protein kinase kinase-beta is an alternative upstream kinase for amp-activated protein kinase. Cell Metab. 2005, 2, 9–19. [Google Scholar] [CrossRef]
- Woods, A.; Dickerson, K.; Heath, R.; Hong, S.P.; Momcilovic, M.; Johnstone, S.R.; Carlson, M.; Carling, D. Ca2+/calmodulin-dependent protein kinase kinase-beta acts upstream of amp-activated protein kinase in mammalian cells. Cell Metab. 2005, 2, 21–33. [Google Scholar] [CrossRef]
- Hardie, D.G. Ampk—Sensing energy while talking to other signaling pathways. Cell Metab. 2014, 20, 939–952. [Google Scholar] [CrossRef]
- Hardie, D.G. Ampk: Positive and negative regulation, and its role in whole-body energy homeostasis. Curr. Opin. Cell Biol. 2014, 33C, 1–7. [Google Scholar] [CrossRef]
- Hardie, D.G. Amp-activated/snf1 protein kinases: Conserved guardians of cellular energy. Nat. Rev. Mol. Cell Biol. 2007, 8, 774–785. [Google Scholar] [CrossRef] [PubMed]
- Emerling, B.M.; Weinberg, F.; Snyder, C.; Burgess, Z.; Mutlu, G.M.; Viollet, B.; Budinger, G.R.; Chandel, N.S. Hypoxic activation of ampk is dependent on mitochondrial ros but independent of an increase in amp/atp ratio. Free Radic. Biol. Med. 2009, 46, 1386–1391. [Google Scholar] [CrossRef] [PubMed]
- Zmijewski, J.W.; Banerjee, S.; Bae, H.; Friggeri, A.; Lazarowski, E.R.; Abraham, E. Exposure to hydrogen peroxide induces oxidation and activation of amp-activated protein kinase. J. Biol. Chem. 2010, 285, 33154–33164. [Google Scholar] [CrossRef] [PubMed]
- Hardie, D.G. Keeping the home fires burning: Amp-activated protein kinase. J. R. Soc. Interface 2018, 15, 20170774. [Google Scholar] [CrossRef] [PubMed]
- Hardie, D.G.; Schaffer, B.E.; Brunet, A. Ampk: An energy-sensing pathway with multiple inputs and outputs. Trends Cell Biol. 2016, 26, 190–201. [Google Scholar] [CrossRef] [PubMed]
- Evans, A.M.; Hardie, D.G.; Peers, C.; Wyatt, C.N.; Viollet, B.; Kumar, P.; Dallas, M.L.; Ross, F.; Ikematsu, N.; Jordan, H.L.; et al. Ion channel regulation by ampk: The route of hypoxia-response coupling in thecarotid body and pulmonary artery. Ann. N. Y. Acad. Sci. 2009, 1177, 89–100. [Google Scholar] [CrossRef]
- Hardie, D.G.; Carling, D.; Carlson, M. The amp-activated/snf1 protein kinase subfamily: Metabolic sensors of the eukaryotic cell? Annu. Rev. Biochem. 1998, 67, 821–855. [Google Scholar] [CrossRef]
- Klein, H.; Garneau, L.; Trinh, N.T.; Prive, A.; Dionne, F.; Goupil, E.; Thuringer, D.; Parent, L.; Brochiero, E.; Sauve, R. Inhibition of the kca3.1 channels by amp-activated protein kinase in human airway epithelial cells. Am. J. Physiol. 2009, 296, C285–C295. [Google Scholar] [CrossRef]
- Chang, T.J.; Chen, W.P.; Yang, C.; Lu, P.H.; Liang, Y.C.; Su, M.J.; Lee, S.C.; Chuang, L.M. Serine-385 phosphorylation of inwardly rectifying k+ channel subunit (kir6.2) by amp-dependent protein kinase plays a key role in rosiglitazone-induced closure of the k(atp) channel and insulin secretion in rats. Diabetologia 2009, 52, 1112–1121. [Google Scholar] [CrossRef]
- Tan, C.D.; Smolenski, R.T.; Harhun, M.I.; Patel, H.K.; Ahmed, S.G.; Wanisch, K.; Yanez-Munoz, R.J.; Baines, D.L. Amp-activated protein kinase (ampk)-dependent and -independent pathways regulate hypoxic inhibition of transepithelial na+ transport across human airway epithelial cells. Br. J. Pharm. 2012, 167, 368–382. [Google Scholar] [CrossRef]
- Evans, A.M.; Mustard, K.J.; Wyatt, C.N.; Peers, C.; Dipp, M.; Kumar, P.; Kinnear, N.P.; Hardie, D.G. Does amp-activated protein kinase couple inhibition of mitochondrial oxidative phosphorylation by hypoxia to calcium signaling in o2-sensing cells? J. Biol. Chem. 2005, 280, 41504–41511. [Google Scholar] [CrossRef] [PubMed]
- Bergofsky, E.H.; Haas, F.; Porcelli, R. Determination of the sensitive vascular sites from which hypoxia and hypercapnia elicit rises in pulmonary arterial pressure. Fed. Proc. 1968, 27, 1420–1425. [Google Scholar] [PubMed]
- Dipp, M.; Nye, P.C.; Evans, A.M. Hypoxic release of calcium from the sarcoplasmic reticulum of pulmonary artery smooth muscle. Am. J. Physiol. Lung Cell Mol. Physiol. 2001, 281, L318–L325. [Google Scholar] [CrossRef] [PubMed]
- Robertson, T.P.; Ward, J.P.; Aaronson, P.I. Hypoxia induces the release of a pulmonary-selective, ca(2+)-sensitising, vasoconstrictor from the perfused rat lung. Cardiovasc Res. 2001, 50, 145–150. [Google Scholar] [CrossRef]
- Gaine, S.P.; Hales, M.A.; Flavahan, N.A. Hypoxic pulmonary endothelial cells release a diffusible contractile factor distinct from endothelin. Am. J. Physiol. 1998, 274, L657–L664. [Google Scholar] [CrossRef] [PubMed]
- Naeije, R.; Lejeune, P.; Leeman, M.; Melot, C.; Closset, J. Pulmonary vascular responses to surgical chemodenervation and chemical sympathectomy in dogs. J. Appl. Physiol. 1989, 66, 42–50. [Google Scholar] [CrossRef]
- Robin, E.D.; Theodore, J.; Burke, C.M.; Oesterle, S.N.; Fowler, M.B.; Jamieson, S.W.; Baldwin, J.C.; Morris, A.J.; Hunt, S.A.; Vankessel, A.; et al. Hypoxic pulmonary vasoconstriction persists in the human transplanted lung. Clin. Sci. 1987, 72, 283–287. [Google Scholar] [CrossRef]
- Dipp, M.; Evans, A.M. Cyclic adp-ribose is the primary trigger for hypoxic pulmonary vasoconstriction in the rat lung in situ. Circ. Res. 2001, 89, 77–83. [Google Scholar] [CrossRef]
- Clement, A.M.; Long, J.A. Air-breathing adaptation in a marine devonian lungfish. Biol. Lett. 2010, 6, 509–512. [Google Scholar] [CrossRef]
- West, J.B. Comparative physiology of the pulmonary circulation. Compr. Physiol. 2011, 1, 1525–1539. [Google Scholar]
- Farmer, C.G. The evolution of unidirectional pulmonary airflow. Physiology 2015, 30, 260–272. [Google Scholar] [CrossRef] [PubMed]
- Porteus, C.; Hedrick, M.S.; Hicks, J.W.; Wang, T.; Milsom, W.K. Time domains of the hypoxic ventilatory response in ectothermic vertebrates. J. Comp. Physiol. B 2011, 181, 311–333. [Google Scholar] [CrossRef] [PubMed]
- Hawley, S.A.; Boudeau, J.; Reid, J.L.; Mustard, K.J.; Udd, L.; Makela, T.P.; Alessi, D.R.; Hardie, D.G. Complexes between the lkb1 tumor suppressor, strad alpha/beta and mo25 alpha/beta are upstream kinases in the amp-activated protein kinase cascade. J. Biol. 2003, 2, 28. [Google Scholar] [CrossRef] [PubMed]
- Owen, M.R.; Doran, E.; Halestrap, A.P. Evidence that metformin exerts its anti-diabetic effects through inhibition of complex 1 of the mitochondrial respiratory chain. Biochem. J. 2000, 348 Pt 3, 607–614. [Google Scholar] [CrossRef]
- Corton, J.M.; Gillespie, J.G.; Hawley, S.A.; Hardie, D.G. 5-aminoimidazole-4-carboxamide ribonucleoside. A specific method for activating amp-activated protein kinase in intact cells? Eur. J. Biochem. 1995, 229, 558–565. [Google Scholar] [CrossRef]
- Robertson, T.P.; Mustard, K.J.; Lewis, T.H.; Clark, J.H.; Wyatt, C.N.; Blanco, E.A.; Peers, C.; Hardie, D.G.; Evans, A.M. Amp-activated protein kinase and hypoxic pulmonary vasoconstriction. Eur. J. Pharmacol. 2008, 595, 39–43. [Google Scholar] [CrossRef][Green Version]
- Bain, J.; Plater, L.; Elliott, M.; Shpiro, N.; Hastie, C.J.; McLauchlan, H.; Klevernic, I.; Arthur, J.S.; Alessi, D.R.; Cohen, P. The selectivity of protein kinase inhibitors: A further update. Biochem. J. 2007, 408, 297–315. [Google Scholar] [CrossRef]
- Yuan, X.J.; Goldman, W.F.; Tod, M.L.; Rubin, L.J.; Blaustein, M.P. Hypoxia reduces potassium currents in cultured rat pulmonary but not mesenteric arterial myocytes. Am. J. Physiol. 1993, 264, L116–L123. [Google Scholar] [CrossRef]
- Yuan, X.J.; Sugiyama, T.; Goldman, W.F.; Rubin, L.J.; Blaustein, M.P. A mitochondrial uncoupler increases kca currents but decreases kv currents in pulmonary artery myocytes. Am. J. Physiol. 1996, 270, C321–C331. [Google Scholar] [CrossRef]
- Rajamohan, F.; Reyes, A.R.; Frisbie, R.K.; Hoth, L.R.; Sahasrabudhe, P.; Magyar, R.; Landro, J.A.; Withka, J.M.; Caspers, N.L.; Calabrese, M.F.; et al. Probing the enzyme kinetics, allosteric modulation and activation of alpha-1 and alpha-2 subunit containing amp-activated protein kinase (ampk) heterotrimeric complexes by pharmacological and physiological activators. Biochem. J. 2015, 473, 581–592. [Google Scholar] [CrossRef]
- Goransson, O.; McBride, A.; Hawley, S.A.; Ross, F.A.; Shpiro, N.; Foretz, M.; Viollet, B.; Hardie, D.G.; Sakamoto, K. Mechanism of action of a-769662, a valuable tool for activation of amp-activated protein kinase. J. Biol. Chem. 2007, 282, 32549–32560. [Google Scholar] [CrossRef] [PubMed]
- Hunter, R.W.; Foretz, M.; Bultot, L.; Fullerton, M.D.; Deak, M.; Ross, F.A.; Hawley, S.A.; Shpiro, N.; Viollet, B.; Barron, D.; et al. Mechanism of action of compound-13: An alpha1-selective small molecule activator of ampk. Chem. Biol. 2014, 21, 866–879. [Google Scholar] [CrossRef] [PubMed]
- Bigham, A.W.; Julian, C.G.; Wilson, M.J.; Vargas, E.; Browne, V.A.; Shriver, M.D.; Moore, L.G. Maternal prkaa1 and ednra genotypes are associated with birth weight, and prkaa1 with uterine artery diameter and metabolic homeostasis at high altitude. Physiol. Genom. 2014, 46, 687–697. [Google Scholar] [CrossRef] [PubMed]
- Penaloza, D.; Arias-Stella, J. The heart and pulmonary circulation at high altitudes: Healthy highlanders and chronic mountain sickness. Circulation 2007, 115, 1132–1146. [Google Scholar] [CrossRef] [PubMed]
- Misonou, H.; Mohapatra, D.P.; Trimmer, J.S. Kv2.1: A voltage-gated k+ channel critical to dynamic control of neuronal excitability. Neurotoxicology 2005, 26, 743–752. [Google Scholar] [CrossRef] [PubMed]
- Bielanska, J.; Hernandez-Losa, J.; Moline, T.; Somoza, R.; Ramon, Y.C.S.; Condom, E.; Ferreres, J.C.; Felipe, A. Voltage-dependent potassium channels kv1.3 and kv1.5 in human fetus. Cell Physiol. Biochem. 2010, 26, 219–226. [Google Scholar] [CrossRef]
- Epperson, A.; Bonner, H.P.; Ward, S.M.; Hatton, W.J.; Bradley, K.K.; Bradley, M.E.; Trimmer, J.S.; Horowitz, B. Molecular diversity of k(v) alpha- and beta-subunit expression in canine gastrointestinal smooth muscles. Am. J. Physiol. 1999, 277, G127–G136. [Google Scholar]
- Kues, W.A.; Wunder, F. Heterogeneous expression patterns of mammalian potassium channel genes in developing and adult rat brain. Eur. J. Neurosci. 1992, 4, 1296–1308. [Google Scholar] [CrossRef]
- Attwell, D.; Laughlin, S.B. An energy budget for signaling in the grey matter of the brain. J. Cereb. Blood Flow Metab. 2001, 21, 1133–1145. [Google Scholar] [CrossRef]
- Nagy, S.; Maurer, G.W.; Hentze, J.L.; Rose, M.; Werge, T.M.; Rewitz, K. Ampk signaling linked to the schizophrenia-associated 1q21.1 deletion is required for neuronal and sleep maintenance. PLoS Genet. 2018, 14, e1007623. [Google Scholar] [CrossRef]
- Krieger, J. Respiratory Physiology: Breathing in Normal Subjects; WB Saunders: Philadelphia, PA, USA, 2005; pp. 232–244. [Google Scholar]
- O’Halloran, K.D. Chronic intermittent hypoxia creates the perfect storm with calamitous consequences for respiratory control. Respir. Physiol. Neurobiol. 2016, 226, 63–67. [Google Scholar] [CrossRef] [PubMed]
- Schober, A.K.; Neurath, M.F.; Harsch, I.A. Prevalence of sleep apnoea in diabetic patients. Clin. Respir. J. 2011, 5, 165–172. [Google Scholar] [CrossRef] [PubMed]
- West, S.D.; Nicoll, D.J.; Stradling, J.R. Prevalence of obstructive sleep apnoea in men with type 2 diabetes. Thorax 2006, 61, 945–950. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, A.M.; Holmes, A.P.; MacMillan, S.; Ogunbayo, O.A.; Wyatt, C.N.; Dallas, M.D.; Kumar, P.; Foretz, M.; Viollet, B.; Evans, A.M. The hypoxic ventilatory response is facilitated by the activation of lkb1-ampk signalling pathways downstream of the carotid bodies. bioRxiv 2019, 604900. [Google Scholar]
- Mahmoud, A.D.; Lewis, S.; Juricic, L.; Udoh, U.A.; Hartmann, S.; Jansen, M.A.; Ogunbayo, O.A.; Puggioni, P.; Holmes, A.P.; Kumar, P.; et al. Amp-activated protein kinase deficiency blocks the hypoxic ventilatory response and thus precipitates hypoventilation and apnea. Am. J. Respir. Crit. Care Med. 2016, 193, 1032–1043. [Google Scholar] [CrossRef]
- Heymans, C.; Bouckaert, J.J.; Dautrebande, L. Sinus carotidien et refléxes respiratoires. Ii. Influences respiratoires réflexes de l’acidose, de l’alcalose, de l’anhydride carbonique, de l’ion hydrogéne et de l’anoxémie: Sinus carotidiens et échanges respiratoires dans les poumons et au dela poumons. Arch. Int. Pharmacodyn. Ther. 1930, 39, 400–408. [Google Scholar]
- Verna, A.; Roumy, M.; Leitner, L.M. Loss of chemoreceptive properties of the rabbit carotid body after destruction of the glomus cells. Brain Res. 1975, 100, 13–23. [Google Scholar] [CrossRef]
- Prabhakar, N.R. Oxygen sensing by the carotid body chemoreceptors. J. Appl. Physiol. 2000, 88, 2287–2295. [Google Scholar] [CrossRef]
- Kim, D.; Kang, D.; Martin, E.A.; Kim, I.; Carroll, J.L. Effects of modulators of amp-activated protein kinase on task-1/3 and intracellular ca(2+) concentration in rat carotid body glomus cells. Respir. Physiol. Neurobiol. 2014, 195, 19–26. [Google Scholar] [CrossRef]
- Evans, A.M.; Mahmoud, A.D.; Moral-Sanz, J.; Hartmann, S. The emerging role of ampk in the regulation of breathing and oxygen supply. Biochem. J. 2016, 473, 2561–2572. [Google Scholar] [CrossRef]
- Teppema, L.J.; Dahan, A. The ventilatory response to hypoxia in mammals: Mechanisms, measurement, and analysis. Physiol. Rev. 2010, 90, 675–754. [Google Scholar] [CrossRef] [PubMed]
- Gourine, A.V.; Funk, G.D. On the existence of a central respiratory oxygen sensor. J. Appl. Physiol. 2017, 123, 1344–1349. [Google Scholar] [CrossRef] [PubMed]
- Wade, J.G.; Larson, C.P., Jr.; Hickey, R.F.; Ehrenfeld, W.K.; Severinghaus, J.W. Effect of carotid endarterectomy on carotid chemoreceptor and baroreceptor function in man. N. Engl. J. Med. 1970, 282, 823–829. [Google Scholar] [CrossRef] [PubMed]
- Curran, A.K.; Rodman, J.R.; Eastwood, P.R.; Henderson, K.S.; Dempsey, J.A.; Smith, C.A. Ventilatory responses to specific cns hypoxia in sleeping dogs. J. Appl. Physiol. 2000, 88, 1840–1852. [Google Scholar] [CrossRef] [PubMed]
- Roux, J.C.; Villard, L. Biogenic amines in rett syndrome: The usual suspects. Behav. Genet. 2010, 40, 59–75. [Google Scholar] [CrossRef]
- MacMillan, S.; Evans, A.M. The hypoxic ventilatory response is blocked by ampk deletion in catecholaminergic, but not adrenergic cells. FASEB J. 2019, 33, 551.12. [Google Scholar]
- Guyenet, P.G. Regulation of breathing and autonomic outflows by chemoreceptors. Compr. Physiol. 2014, 4, 1511–1562. [Google Scholar]
- Smith, J.C.; Ellenberger, H.H.; Ballanyi, K.; Richter, D.W.; Feldman, J.L. Pre-botzinger complex: A brainstem region that may generate respiratory rhythm in mammals. Science 1991, 254, 726–729. [Google Scholar] [CrossRef]
- Bucher, E.S.; Fox, M.E.; Kim, L.; Kirkpatrick, D.C.; Rodeberg, N.T.; Belle, A.M.; Wightman, R.M. Medullary norepinephrine neurons modulate local oxygen concentrations in the bed nucleus of the stria terminalis. J. Cereb. Blood Flow Metab. 2014, 34, 1128–1137. [Google Scholar] [CrossRef]
- MacMillan, S.; Evans, A.M. Ampk-alpha1 or ampk-alpha2 deletion in smooth muscles does not affect the hypoxic ventilatory response or systemic arterial blood pressure regulation during hypoxia. Front Physiol. 2018, 9, 655. [Google Scholar] [CrossRef]
- Angelova, P.R.; Kasymov, V.; Christie, I.; Sheikhbahaei, S.; Turovsky, E.; Marina, N.; Korsak, A.; Zwicker, J.; Teschemacher, A.G.; Ackland, G.L.; et al. Functional oxygen sensitivity of astrocytes. J. Neurosci. 2015, 35, 10460–10473. [Google Scholar] [CrossRef] [PubMed]
- Marina, N.; Tang, F.; Figueiredo, M.; Mastitskaya, S.; Kasimov, V.; Mohamed-Ali, V.; Roloff, E.; Teschemacher, A.G.; Gourine, A.V.; Kasparov, S. Purinergic signalling in the rostral ventro-lateral medulla controls sympathetic drive and contributes to the progression of heart failure following myocardial infarction in rats. Basic Res. Cardiol. 2013, 108, 317. [Google Scholar] [CrossRef] [PubMed]
- Magistretti, P.J.; Allaman, I. Lactate in the brain: From metabolic end-product to signalling molecule. Nat Rev. Neurosci. 2018, 19, 235–249. [Google Scholar] [CrossRef] [PubMed]
- Voss, C.M.; Andersen, J.V.; Jakobsen, E.; Siamka, O.; Karaca, M.; Maechler, P.; Waagepetersen, H.S. Amp-activated protein kinase (ampk) regulates astrocyte oxidative metabolism by balancing tca cycle dynamics. Glia 2020. [Google Scholar] [CrossRef] [PubMed]
- Andersson, U.; Filipsson, K.; Abbott, C.R.; Woods, A.; Smith, K.; Bloom, S.R.; Carling, D.; Small, C.J. Amp-activated protein kinase plays a role in the control of food intake. J. Biol. Chem. 2004, 279, 12005–12008. [Google Scholar] [CrossRef]
- Kola, B.; Hubina, E.; Tucci, S.A.; Kirkham, T.C.; Garcia, E.A.; Mitchell, S.E.; Williams, L.M.; Hawley, S.A.; Hardie, D.G.; Grossman, A.B.; et al. Cannabinoids and ghrelin have both central and peripheral metabolic and cardiac effects via amp-activated protein kinase. J. Biol. Chem. 2005, 280, 25196–25201. [Google Scholar] [CrossRef]
- Minokoshi, Y.; Alquier, T.; Furukawa, N.; Kim, Y.B.; Lee, A.; Xue, B.; Mu, J.; Foufelle, F.; Ferre, P.; Birnbaum, M.J.; et al. Amp-kinase regulates food intake by responding to hormonal and nutrient signals in the hypothalamus. Nature 2004, 428, 569–574. [Google Scholar] [CrossRef]
- Claret, M.; Smith, M.A.; Batterham, R.L.; Selman, C.; Choudhury, A.I.; Fryer, L.G.; Clements, M.; Al-Qassab, H.; Heffron, H.; Xu, A.W.; et al. Ampk is essential for energy homeostasis regulation and glucose sensing by pomc and agrp neurons. J. Clin. Invest. 2007, 117, 2325–2336. [Google Scholar] [CrossRef]
- Zhang, C.S.; Hawley, S.A.; Zong, Y.; Li, M.; Wang, Z.; Gray, A.; Ma, T.; Cui, J.; Feng, J.W.; Zhu, M.; et al. Fructose-1,6-bisphosphate and aldolase mediate glucose sensing by ampk. Nature 2017, 548, 112–116. [Google Scholar] [CrossRef]
- Yang, Y.; Atasoy, D.; Su, H.H.; Sternson, S.M. Hunger states switch a flip-flop memory circuit via a synaptic ampk-dependent positive feedback loop. Cell 2011, 146, 992–1003. [Google Scholar] [CrossRef]
- Anderson, K.A.; Ribar, T.J.; Lin, F.; Noeldner, P.K.; Green, M.F.; Muehlbauer, M.J.; Witters, L.A.; Kemp, B.E.; Means, A.R. Hypothalamic camkk2 contributes to the regulation of energy balance. Cell Metab. 2008, 7, 377–388. [Google Scholar] [CrossRef] [PubMed]
- Lopez, M.; Lage, R.; Saha, A.K.; Perez-Tilve, D.; Vazquez, M.J.; Varela, L.; Sangiao-Alvarellos, S.; Tovar, S.; Raghay, K.; Rodriguez-Cuenca, S.; et al. Hypothalamic fatty acid metabolism mediates the orexigenic action of ghrelin. Cell Metab. 2008, 7, 389–399. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, S.; Martins, L.; Jacas, J.; Carrasco, P.; Pozo, M.; Clotet, J.; Serra, D.; Hegardt, F.G.; Dieguez, C.; Lopez, M.; et al. Hypothalamic ceramide levels regulated by cpt1c mediate the orexigenic effect of ghrelin. Diabetes 2013, 62, 2329–2337. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, S.; Sato, T.; Tateyama, M.; Kageyama, H.; Maejima, Y.; Nakata, M.; Hirako, S.; Matsuo, T.; Kyaw, S.; Shiuchi, T.; et al. Activation of ampk-regulated crh neurons in the pvh is sufficient and necessary to induce dietary preference for carbohydrate over fat. Cell Rep. 2018, 22, 706–721. [Google Scholar] [CrossRef]
- Ruyle, B.C.; Klutho, P.J.; Baines, C.P.; Heesch, C.M.; Hasser, E.M. Hypoxia activates a neuropeptidergic pathway from the paraventricular nucleus of the hypothalamus to the nucleus tractus solitarii. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2018, 315, R1167–R1182. [Google Scholar] [CrossRef]
- Coldren, K.M.; Li, D.P.; Kline, D.D.; Hasser, E.M.; Heesch, C.M. Acute hypoxia activates neuroendocrine, but not presympathetic, neurons in the paraventricular nucleus of the hypothalamus: Differential role of nitric oxide. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2017, 312, R982–R995. [Google Scholar] [CrossRef]
- King, T.L.; Heesch, C.M.; Clark, C.G.; Kline, D.D.; Hasser, E.M. Hypoxia activates nucleus tractus solitarii neurons projecting to the paraventricular nucleus of the hypothalamus. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2012, 302, R1219–R1232. [Google Scholar] [CrossRef]
- King, T.L.; Ruyle, B.C.; Kline, D.D.; Heesch, C.M.; Hasser, E.M. Catecholaminergic neurons projecting to the paraventricular nucleus of the hypothalamus are essential for cardiorespiratory adjustments to hypoxia. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2015, 309, R721–R731. [Google Scholar] [CrossRef]
- Martinez de Morentin, P.B.; Gonzalez-Garcia, I.; Martins, L.; Lage, R.; Fernandez-Mallo, D.; Martinez-Sanchez, N.; Ruiz-Pino, F.; Liu, J.; Morgan, D.A.; Pinilla, L.; et al. Estradiol regulates brown adipose tissue thermogenesis via hypothalamic ampk. Cell Metab. 2014, 20, 41–53. [Google Scholar] [CrossRef]
- Lopez, M.; Varela, L.; Vazquez, M.J.; Rodriguez-Cuenca, S.; Gonzalez, C.R.; Velagapudi, V.R.; Morgan, D.A.; Schoenmakers, E.; Agassandian, K.; Lage, R.; et al. Hypothalamic ampk and fatty acid metabolism mediate thyroid regulation of energy balance. Nat. Med. 2010, 16, 1001–1008. [Google Scholar] [CrossRef]
- Beiroa, D.; Imbernon, M.; Gallego, R.; Senra, A.; Herranz, D.; Villarroya, F.; Serrano, M.; Ferno, J.; Salvador, J.; Escalada, J.; et al. Glp-1 agonism stimulates brown adipose tissue thermogenesis and browning through hypothalamic ampk. Diabetes 2014, 63, 3346–3358. [Google Scholar] [CrossRef] [PubMed]
- Martins, L.; Seoane-Collazo, P.; Contreras, C.; Gonzalez-Garcia, I.; Martinez-Sanchez, N.; Gonzalez, F.; Zalvide, J.; Gallego, R.; Dieguez, C.; Nogueiras, R.; et al. A functional link between ampk and orexin mediates the effect of bmp8b on energy balance. Cell Rep. 2016, 16, 2231–2242. [Google Scholar] [CrossRef] [PubMed]
- Whittle, A.J.; Carobbio, S.; Martins, L.; Slawik, M.; Hondares, E.; Vazquez, M.J.; Morgan, D.; Csikasz, R.I.; Gallego, R.; Rodriguez-Cuenca, S.; et al. Bmp8b increases brown adipose tissue thermogenesis through both central and peripheral actions. Cell 2012, 149, 871–885. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Sanchez, N.; Seoane-Collazo, P.; Contreras, C.; Varela, L.; Villarroya, J.; Rial-Pensado, E.; Buque, X.; Aurrekoetxea, I.; Delgado, T.C.; Vazquez-Martinez, R.; et al. Hypothalamic ampk-er ctress-jnk1 axis mediates the central actions of thyroid hormones on energy balance. Cell Metab. 2017, 26, 212–229 e212. [Google Scholar]
- Seoane-Collazo, P.; Roa, J.; Rial-Pensado, E.; Linares-Pose, L.; Beiroa, D.; Ruiz-Pino, F.; Lopez-Gonzalez, T.; Morgan, D.A.; Pardavila, J.A.; Sanchez-Tapia, M.J.; et al. Sf1-specific ampkalpha1 deletion protects against diet-induced obesity. Diabetes 2018, 67, 2213–2226. [Google Scholar] [CrossRef]
- Pollard, A.E.; Martins, L.; Muckett, P.J.; Khadayate, S.; Bornot, A.; Clausen, M.; Admyre, T.; Bjursell, M.; Fiadeiro, R.; Wilson, L.; et al. Ampk activation protects against diet induced obesity through ucp1-independent thermogenesis in subcutaneous white adipose tissue. Nat. Metab. 2019, 1, 340–349. [Google Scholar] [CrossRef]
- Bastias-Perez, M.; Zagmutt, S.; Soler-Vazquez, M.C.; Serra, D.; Mera, P.; Herrero, L. Impact of adaptive thermogenesis in mice on the treatment of obesity. Cells 2020, 9, 316. [Google Scholar] [CrossRef]
- Ainslie, P.N.; Lucas, S.J.; Burgess, K.R. Breathing and sleep at high altitude. Respir. Physiol. Neurobiol. 2013, 188, 233–256. [Google Scholar] [CrossRef]
- Cheng, X.; Prange-Barczynska, M.; Fielding, J.W.; Zhang, M.; Burrell, A.L.; Lima, J.D.; Eckardt, L.; Argles, I.; Pugh, C.W.; Buckler, K.J.; et al. Marked and rapid effects of pharmacological hif-2alpha antagonism on hypoxic ventilatory control. J. Clin. Investig. 2020, 130. [Google Scholar] [CrossRef]
- Takakura, A.C.; Moreira, T.S.; Colombari, E.; West, G.H.; Stornetta, R.L.; Guyenet, P.G. Peripheral chemoreceptor inputs to retrotrapezoid nucleus (rtn) co2-sensitive neurons in rats. J. Physiol. 2006, 572, 503–523. [Google Scholar] [CrossRef]
- McMurtry, I.F.; Petrun, M.D.; Reeves, J.T. Lungs from chronically hypoxic rats have decreased pressor response to acute hypoxia. Am. J. Physiol. 1978, 235, H104–H109. [Google Scholar] [CrossRef]
- Durmowicz, A.G.; Hofmeister, S.; Kadyraliev, T.K.; Aldashev, A.A.; Stenmark, K.R. Functional and structural adaptation of the yak pulmonary circulation to residence at high altitude. J. Appl. Physiol. 1993, 74, 2276–2285. [Google Scholar] [CrossRef] [PubMed]
- Salminen, A.; Kaarniranta, K.; Kauppinen, A. Ampk and hif signaling pathways regulate both longevity and cancer growth: The good news and the bad news about survival mechanisms. Biogerontology 2016, 17, 655–680. [Google Scholar] [CrossRef] [PubMed]
- Berquin, P.; Bodineau, L.; Gros, F.; Larnicol, N. Brainstem and hypothalamic areas involved in respiratory chemoreflexes: A fos study in adult rats. Brain Res. 2000, 857, 30–40. [Google Scholar] [CrossRef]
- Berquin, P.; Cayetanot, F.; Gros, F.; Larnicol, N. Postnatal changes in fos-like immunoreactivity evoked by hypoxia in the rat brainstem and hypothalamus. Brain Res. 2000, 877, 149–159. [Google Scholar] [CrossRef]
- Trapp, S.; Cork, S.C. Ppg neurons of the lower brain stem and their role in brain glp-1 receptor activation. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2015, 309, R795–R804. [Google Scholar] [CrossRef]
- Henry, J.L.; Sessle, B.J. Vasopressin and oxytocin express excitatory effects on respiratory and respiration-related neurones in the nuclei of the tractus solitarius in the cat. Brain Res. 1989, 491, 150–155. [Google Scholar] [CrossRef]
- Leng, G.; Sabatier, N. Oxytocin—The sweet hormone? Trends Endocrinol. Metab. 2017, 28, 365–376. [Google Scholar] [CrossRef]
- Uchoa, E.T.; Aguilera, G.; Herman, J.P.; Fiedler, J.L.; Deak, T.; De Sousa, M.B. Novel aspects of glucocorticoid actions. J. Neuroendocr. 2014, 26, 557–572. [Google Scholar] [CrossRef]
- Vargas, V.E.; Gurung, S.; Grant, B.; Hyatt, K.; Singleton, K.; Myers, S.M.; Saunders, D.; Njoku, C.; Towner, R.; Myers, D.A. Gestational hypoxia disrupts the neonatal leptin surge and programs hyperphagia and obesity in male offspring in the sprague-dawley rat. PLoS ONE 2017, 12, e0185272. [Google Scholar] [CrossRef]
- Liu, X.W.; Yin, J.; Ma, Q.S.; Qi, C.C.; Mu, J.Y.; Zhang, L.; Gao, L.P.; Jing, Y.H. Role of arcuate nucleus in the regulation of feeding behavior in the process of altitude acclimatization in rats. High Alt. Med. Biol. 2017, 18, 234–241. [Google Scholar] [CrossRef]
- Liu, Y.N.; Ma, Q.S.; Wu, Q. Preliminary investigation for effects of hypothalamic leptin/ghrelin and arcuate nucleus pro-opiomelanocortin system on regulation of high-altitude acclimatization. Biomed. Pharmacother. 2017, 89, 1462–1466. [Google Scholar] [CrossRef] [PubMed]
- Kelly, K.R.; Williamson, D.L.; Fealy, C.E.; Kriz, D.A.; Krishnan, R.K.; Huang, H.; Ahn, J.; Loomis, J.L.; Kirwan, J.P. Acute altitude-induced hypoxia suppresses plasma glucose and leptin in healthy humans. Metabolism 2010, 59, 200–205. [Google Scholar] [CrossRef] [PubMed]
- Roberts, A.C.; Butterfield, G.E.; Cymerman, A.; Reeves, J.T.; Wolfel, E.E.; Brooks, G.A. Acclimatization to 4300-m altitude decreases reliance on fat as a substrate. J. Appl. Physiol. 1996, 81, 1762–1771. [Google Scholar] [CrossRef] [PubMed]
- Brooks, G.A.; Butterfield, G.E.; Wolfe, R.R.; Groves, B.M.; Mazzeo, R.S.; Sutton, J.R.; Wolfel, E.E.; Reeves, J.T. Decreased reliance on lactate during exercise after acclimatization to 4300 m. J. Appl. Physiol. 1991, 71, 333–341. [Google Scholar] [CrossRef]
- Brooks, G.A.; Butterfield, G.E.; Wolfe, R.R.; Groves, B.M.; Mazzeo, R.S.; Sutton, J.R.; Wolfel, E.E.; Reeves, J.T. Increased dependence on blood glucose after acclimatization to 4300 m. J. Appl. Physiol. 1991, 70, 919–927. [Google Scholar] [CrossRef]
- Moore, L.G. Human genetic adaptation to high altitudes: Current status and future prospects. Quat. Int. 2017, 461, 4–13. [Google Scholar] [CrossRef]
- Holden, J.E.; Stone, C.K.; Clark, C.M.; Brown, W.D.; Nickles, R.J.; Stanley, C.; Hochachka, P.W. Enhanced cardiac metabolism of plasma glucose in high-altitude natives: Adaptation against chronic hypoxia. J. Appl. Physiol. 1995, 79, 222–228. [Google Scholar] [CrossRef]
- Richardson, A.; Watt, P.; Maxwell, N. Hydration and the physiological responses to acute normobaric hypoxia. Wilderness Environ. Med. 2009, 20, 212–220. [Google Scholar] [CrossRef]
- Ge, R.L.; Simonson, T.S.; Gordeuk, V.; Prchal, J.T.; McClain, D.A. Metabolic aspects of high-altitude adaptation in tibetans. Exp. Physiol. 2015, 100, 1247–1255. [Google Scholar] [CrossRef]
- Horscroft, J.A.; Kotwica, A.O.; Laner, V.; West, J.A.; Hennis, P.J.; Levett, D.Z.H.; Howard, D.J.; Fernandez, B.O.; Burgess, S.L.; Ament, Z.; et al. Metabolic basis to sherpa altitude adaptation. Proc. Natl. Acad. Sci. USA 2017, 114, 6382–6387. [Google Scholar] [CrossRef]
- Wu, T.; Kayser, B. High altitude adaptation in tibetans. High Alt. Med. Biol. 2006, 7, 193–208. [Google Scholar] [CrossRef] [PubMed]
- Woolcott, O.O.; Ader, M.; Bergman, R.N. Glucose homeostasis during short-term and prolonged exposure to high altitudes. Endocr. Rev. 2015, 36, 149–173. [Google Scholar] [CrossRef] [PubMed]
- Hochachka, P.W.; Clark, C.M.; Holden, J.E.; Stanley, C.; Ugurbil, K.; Menon, R.S. 31p magnetic resonance spectroscopy of the sherpa heart: A phosphocreatine/adenosine triphosphate signature of metabolic defense against hypobaric hypoxia. Proc. Natl. Acad. Sci. USA 1996, 93, 1215–1220. [Google Scholar] [CrossRef] [PubMed]
- Narravula, S.; Colgan, S.P. Hypoxia-inducible factor 1-mediated inhibition of peroxisome proliferator-activated receptor alpha expression during hypoxia. J. Immunol. 2001, 166, 7543–7548. [Google Scholar] [CrossRef] [PubMed]
- Bartsch, P.; Mairbaurl, H.; Maggiorini, M.; Swenson, E.R. Physiological aspects of high-altitude pulmonary edema. J. Appl. Physiol. 2005, 98, 1101–1110. [Google Scholar] [CrossRef]
- Huez, S.; Faoro, V.; Vachiery, J.L.; Unger, P.; Martinot, J.B.; Naeije, R. Images in cardiovascular medicine. High-altitude-induced right-heart failure. Circulation 2007, 115, e308–e309. [Google Scholar] [CrossRef]
- Reichmuth, K.J.; Austin, D.; Skatrud, J.B.; Young, T. Association of sleep apnea and type ii diabetes: A population-based study. Am. J. Respir. Crit. Care Med. 2005, 172, 1590–1595. [Google Scholar] [CrossRef]
- Chau, E.H.; Lam, D.; Wong, J.; Mokhlesi, B.; Chung, F. Obesity hypoventilation syndrome: A review of epidemiology, pathophysiology, and perioperative considerations. Anesthesiology 2012, 117, 188–205. [Google Scholar] [CrossRef]
- Ussavarungsi, K.; Thomas, C.S.; Burger, C.D. Prevalence of metabolic syndrome in patients with pulmonary hypertension. Clin. Respir. J. 2015, 11, 721–726. [Google Scholar] [CrossRef]
- Robbins, I.M.; Newman, J.H.; Johnson, R.F.; Hemnes, A.R.; Fremont, R.D.; Piana, R.N.; Zhao, D.X.; Byrne, D.W. Association of the metabolic syndrome with pulmonary venous hypertension. Chest 2009, 136, 31–36. [Google Scholar] [CrossRef]
- Ruderman, N.B.; Carling, D.; Prentki, M.; Cacicedo, J.M. Ampk, insulin resistance, and the metabolic syndrome. J. Clin. Invest. 2013, 123, 2764–2772. [Google Scholar] [CrossRef] [PubMed]
- Gauthier, M.S.; O’Brien, E.L.; Bigornia, S.; Mott, M.; Cacicedo, J.M.; Xu, X.J.; Gokce, N.; Apovian, C.; Ruderman, N. Decreased amp-activated protein kinase activity is associated with increased inflammation in visceral adipose tissue and with whole-body insulin resistance in morbidly obese humans. Biochem. Biophys. Res. Commun. 2011, 404, 382–387. [Google Scholar] [CrossRef] [PubMed]
- Voss, J.D.; Allison, D.B.; Webber, B.J.; Otto, J.L.; Clark, L.L. Lower obesity rate during residence at high altitude among a military population with frequent migration: A quasi experimental model for investigating spatial causation. PLoS ONE 2014, 9, e93493. [Google Scholar] [CrossRef] [PubMed]
- Lindgarde, F.; Ercilla, M.B.; Correa, L.R.; Ahren, B. Body adiposity, insulin, and leptin in subgroups of peruvian amerindians. High Alt. Med. Biol. 2004, 5, 27–31. [Google Scholar] [CrossRef]
- Baracco, R.; Mohanna, S.; Seclen, S. A comparison of the prevalence of metabolic syndrome and its components in high and low altitude populations in peru. Metab. Syndr. Relat. Disord. 2007, 5, 55–62. [Google Scholar] [CrossRef]
- Matsubayashi, K.; Kimura, Y.; Sakamoto, R.; Wada, T.; Ishimoto, Y.; Hirosaki, M.; Konno, A.; Chen, W.; Ishine, M.; Kosaka, Y.; et al. Comprehensive geriatric assessment of elderly highlanders in qinghai, china i: Activities of daily living, quality of life and metabolic syndrome. Geriatr. Gerontol. Int. 2009, 9, 333–341. [Google Scholar] [CrossRef]
- Wang, Z.; Dang, S.; Yan, H. Nutrient intakes of rural tibetan mothers: A cross-sectional survey. BMC Public Health 2010, 10, 801. [Google Scholar] [CrossRef]
- Sherpa, L.Y.; Stigum, H.; Chongsuvivatwong, V.; Thelle, D.S.; Bjertness, E. Obesity in tibetans aged 30-70 living at different altitudes under the north and south faces of mt. Everest. Int. J. Environ. Res. Public Health 2010, 7, 1670–1680. [Google Scholar] [CrossRef]
- Wang, Z.; Khor, S.; Cai, D. Age-dependent decline of hypothalamic hif2alpha in response to insulin and its contribution to advanced age-associated metabolic disorders in mice. J. Biol. Chem. 2019, 294, 4946–4955. [Google Scholar] [CrossRef]
- Punjabi, N.M.; Caffo, B.S.; Goodwin, J.L.; Gottlieb, D.J.; Newman, A.B.; O’Connor, G.T.; Rapoport, D.M.; Redline, S.; Resnick, H.E.; Robbins, J.A.; et al. Sleep-disordered breathing and mortality: A prospective cohort study. PLoS Med. 2009, 6, e1000132. [Google Scholar] [CrossRef]
- Gozal, D. The energy crisis revisited: Amp-activated protein kinase and the mammalian hypoxic ventilatory response. Am. J. Respir. Crit. Care Med. 2016, 193, 945–946. [Google Scholar] [CrossRef] [PubMed]
- Goncharov, D.A.; Kudryashova, T.V.; Ziai, H.; Ihida-Stansbury, K.; DeLisser, H.; Krymskaya, V.P.; Tuder, R.M.; Kawut, S.M.; Goncharova, E.A. Mammalian target of rapamycin complex 2 (mtorc2) coordinates pulmonary artery smooth muscle cell metabolism, proliferation, and survival in pulmonary arterial hypertension. Circulation 2014, 129, 864–874. [Google Scholar] [CrossRef] [PubMed]
- Omura, J.; Satoh, K.; Kikuchi, N.; Satoh, T.; Kurosawa, R.; Nogi, M.; Otsuki, T.; Kozu, K.; Numano, K.; Suzuki, K.; et al. Protective roles of endothelial amp-activated protein kinase against hypoxia-induced pulmonary hypertension in mice. Circ. Res. 2016, 119, 197–209. [Google Scholar] [CrossRef] [PubMed]
- Dunlop, E.A.; Tee, A.R. The kinase triad, ampk, mtorc1 and ulk1, maintains energy and nutrient homoeostasis. Biochem. Soc. Trans. 2013, 41, 939–943. [Google Scholar] [CrossRef]
- Fogarty, S.; Ross, F.A.; Vara Ciruelos, D.; Gray, A.; Gowans, G.J.; Hardie, D.G. Ampk causes cell cycle arrest in lkb1-deficient cells via activation of camkk2. Mol. Cancer Res. 2016, 14, 683–695. [Google Scholar] [CrossRef]
- Rena, G.; Hardie, D.G.; Pearson, E.R. The mechanisms of action of metformin. Diabetologia 2017, 60, 1577–1585. [Google Scholar] [CrossRef]
- Goncharov, D.A.; Goncharova, E.A.; Tofovic, S.P.; Hu, J.; Baust, J.J.; Pena, A.Z.; Ray, A.; Rode, A.; Vanderpool, R.R.; Mora, A.L.; et al. Metformin therapy for pulmonary hypertension associated with hfpef versus pah. Am. J. Respir. Crit. Care Med. 2018, 198, 681–684. [Google Scholar] [CrossRef]
- Hunter, R.W.; Hughey, C.C.; Lantier, L.; Sundelin, E.I.; Peggie, M.; Zeqiraj, E.; Sicheri, F.; Jessen, N.; Wasserman, D.H.; Sakamoto, K. Metformin reduces liver glucose production by inhibition of fructose-1-6-bisphosphatase. Nat. Med. 2018, 24, 1395–1406. [Google Scholar] [CrossRef]
- Cameron, A.R.; Logie, L.; Patel, K.; Erhardt, S.; Bacon, S.; Middleton, P.; Harthill, J.; Forteath, C.; Coats, J.T.; Kerr, C.; et al. Metformin selectively targets redox control of complex i energy transduction. Redox Biol. 2018, 14, 187–197. [Google Scholar] [CrossRef]
- Nicola, T.; Ambalavanan, N.; Zhang, W.; James, M.L.; Rehan, V.; Halloran, B.; Olave, N.; Bulger, A.; Oparil, S.; Chen, Y.F. Hypoxia-induced inhibition of lung development is attenuated by the peroxisome proliferator-activated receptor-gamma agonist rosiglitazone. Am. J. Physiol. Lung Cell Mol. Physiol. 2011, 301, L125–L134. [Google Scholar] [CrossRef]
- Kool, H.; Mous, D.; Tibboel, D.; De Klein, A.; Rottier, R.J. Pulmonary vascular development goes awry in congenital lung abnormalities. Birth Defects Res. C Embryo Today 2014, 102, 343–358. [Google Scholar] [CrossRef] [PubMed]
- Thompson, B.T.; Hassoun, P.M.; Kradin, R.L.; Hales, C.A. Acute and chronic hypoxic pulmonary hypertension in guinea pigs. J. Appl. Physiol. 1989, 66, 920–928. [Google Scholar] [CrossRef] [PubMed]
- Schwenke, D.O.; Bolter, C.P.; Cragg, P.A. Are the carotid bodies of the guinea-pig functional? Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2007, 146, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Thompson, B.T.; Steigman, D.M.; Spence, C.L.; Janssens, S.P.; Hales, C.A. Chronic hypoxic pulmonary hypertension in the guinea pig: Effect of three levels of hypoxia. J. Appl. Physiol. 1993, 74, 916–921. [Google Scholar] [CrossRef] [PubMed]
- Lucking, E.F.; O’Connor, K.M.; Strain, C.R.; Fouhy, F.; Bastiaanssen, T.F.S.; Burns, D.P.; Golubeva, A.V.; Stanton, C.; Clarke, G.; Cryan, J.F.; et al. Chronic intermittent hypoxia disrupts cardiorespiratory homeostasis and gut microbiota composition in adult male guinea-pigs. EBioMedicine 2018, 38, 191–205. [Google Scholar] [CrossRef] [PubMed]
- Docio, I.; Olea, E.; Prieto, L.J.; Gallego-Martin, T.; Obeso, A.; Gomez-Nino, A.; Rocher, A. Guinea pig as a model to study the carotid body mediated chronic intermittent hypoxia effects. Front Physiol 2018, 9, 694. [Google Scholar] [CrossRef]
- Basnyat, B.; Starling, J.M. Infectious diseases at high altitude. Microbiol. Spectr. 2015, 3. [Google Scholar] [CrossRef]
- Arias-Reyes, C.; Zubieta-DeUrioste, N.; Poma-Machicao, L.; Aliaga-Raduan, F.; Carvajal-Rodriguez, F.; Dutschmann, M.; Schneider-Gasser, E.M.; Zubieta-Calleja, G.; Soliz, J. Does the pathogenesis of sars-cov-2 virus decrease at high-altitude? Respir Physiol. Neurobiol. 2020, 277, 103443. [Google Scholar] [CrossRef]
- Watts, E.R.; Walmsley, S.R. Inflammation and hypoxia: Hif and phd isoform selectivity. Trends Mol. Med. 2019, 25, 33–46. [Google Scholar] [CrossRef]
- Silwal, P.; Kim, J.K.; Yuk, J.M.; Jo, E.K. Amp-activated protein kinase and host defense against infection. Int. J. Mol. Sci. 2018, 19, 3495. [Google Scholar] [CrossRef]
- Moreira, D.; Silvestre, R.; Cordeiro-da-Silva, A.; Estaquier, J.; Foretz, M.; Viollet, B. Amp-activated protein kinase as a target for pathogens: Friends or foes? Curr. Drug Targets 2016, 17, 942–953. [Google Scholar] [CrossRef]
- Sorbara, M.T.; Girardin, S.E. Emerging themes in bacterial autophagy. Curr. Opin. Microbiol. 2015, 23, 163–170. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, K.M.; Lucking, E.F.; Golubeva, A.V.; Strain, C.R.; Fouhy, F.; Cenit, M.C.; Dhaliwal, P.; Bastiaanssen, T.F.S.; Burns, D.P.; Stanton, C.; et al. Manipulation of gut microbiota blunts the ventilatory response to hypercapnia in adult rats. EBioMedicine 2019, 44, 618–638. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Indias, I.; Torres, M.; Montserrat, J.M.; Sanchez-Alcoholado, L.; Cardona, F.; Tinahones, F.J.; Gozal, D.; Poroyko, V.A.; Navajas, D.; Queipo-Ortuno, M.I.; et al. Intermittent hypoxia alters gut microbiota diversity in a mouse model of sleep apnoea. Eur. Respir J. 2015, 45, 1055–1065. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Indias, I.; Torres, M.; Sanchez-Alcoholado, L.; Cardona, F.; Almendros, I.; Gozal, D.; Montserrat, J.M.; Queipo-Ortuno, M.I.; Farre, R. Normoxic recovery mimicking treatment of sleep apnea does not reverse intermittent hypoxia-induced bacterial dysbiosis and low-grade endotoxemia in mice. Sleep 2016, 39, 1891–1897. [Google Scholar] [CrossRef]
- Tripathi, A.; Melnik, A.V.; Xue, J.; Poulsen, O.; Meehan, M.J.; Humphrey, G.; Jiang, L.; Ackermann, G.; McDonald, D.; Zhou, D.; et al. Intermittent hypoxia and hypercapnia, a hallmark of obstructive sleep apnea, alters the gut microbiome and metabolome. mSystems 2018, 3. [Google Scholar] [CrossRef]
- Round, J.L.; Palm, N.W. Causal effects of the microbiota on immune-mediated diseases. Sci. Immunol. 2018, 3, eaao1603. [Google Scholar] [CrossRef]
- Noureldein, M.H.; Eid, A.A. Gut microbiota and mtor signaling: Insight on a new pathophysiological interaction. Microb. Pathog. 2018, 118, 98–104. [Google Scholar] [CrossRef]
- Ivy, D.D.; Abman, S.H.; Barst, R.J.; Berger, R.M.; Bonnet, D.; Fleming, T.R.; Haworth, S.G.; Raj, J.U.; Rosenzweig, E.B.; Schulze Neick, I.; et al. Pediatric pulmonary hypertension. J. Am. Coll. Cardiol. 2013, 62, D117–D126. [Google Scholar] [CrossRef]
- Galie, N.; Hoeper, M.M.; Humbert, M.; Torbicki, A.; Vachiery, J.L.; Barbera, J.A.; Beghetti, M.; Corris, P.; Gaine, S.; Gibbs, J.S.; et al. Guidelines for the diagnosis and treatment of pulmonary hypertension: The task force for the diagnosis and treatment of pulmonary hypertension of the european society of cardiology (esc) and the european respiratory society (ers), endorsed by the international society of heart and lung transplantation (ishlt). Eur. Heart J. 2009, 30, 2493–2537. [Google Scholar]
- Gauda, E.B.; Shirahata, M.; Mason, A.; Pichard, L.E.; Kostuk, E.W.; Chavez-Valdez, R. Inflammation in the carotid body during development and its contribution to apnea of prematurity. Respir. Physiol. Neurobiol. 2013, 185, 120–131. [Google Scholar] [CrossRef] [PubMed]
- Young, T.; Peppard, P.E.; Gottlieb, D.J. Epidemiology of obstructive sleep apnea: A population health perspective. Am. J. Respir Crit. Care Med. 2002, 165, 1217–1239. [Google Scholar] [CrossRef] [PubMed]
- Tomashefski, J.F., Jr. Pulmonary pathology of acute respiratory distress syndrome. Clin. Chest Med. 2000, 21, 435–466. [Google Scholar] [CrossRef]
- Tomashefski, J.F., Jr.; Davies, P.; Boggis, C.; Greene, R.; Zapol, W.M.; Reid, L.M. The pulmonary vascular lesions of the adult respiratory distress syndrome. Am. J. Pathol. 1983, 112, 112–126. [Google Scholar]
- Karnatovskaia, L.V.; Lee, A.S.; Bender, S.P.; Talmor, D.; Festic, E.; Illness, U.S.C.; Injury Trials Group: Lung Injury Prevention Study, I. Obstructive sleep apnea, obesity, and the development of acute respiratory distress syndrome. J. Clin. Sleep Med. 2014, 10, 657–662. [Google Scholar] [CrossRef]
- Wang, G.; Song, Y.; Feng, W.; Liu, L.; Zhu, Y.; Xie, X.; Pan, Y.; Ke, R.; Li, S.; Li, F.; et al. Activation of ampk attenuates lps-induced acute lung injury by upregulation of pgc1alpha and sod1. Exp. Ther. Med. 2016, 12, 1551–1555. [Google Scholar] [CrossRef]
- Perlman, S.; Netland, J. Coronaviruses post-sars: Update on replication and pathogenesis. Nat. Rev. Microbiol. 2009, 7, 439–450. [Google Scholar] [CrossRef]
- Zangrillo, A.; Beretta, L.; Scandroglio, A.M.; Monti, G.; Fominskiy, E.; Colombo, S.; Morselli, F.; Belletti, A.; Silvani, P.; Crivellari, M.; et al. Characteristics, treatment, outcomes and cause of death of invasively ventilated patients with covid-19 ards in milan, italy. Crit. Care Resusc. 2020. ePub ahead of print. [Google Scholar]
- Van den Berghe, G. On the neuroendocrinopathy of critical illness. Perspectives for feeding and novel treatments. Am. J. Respir Crit. Care Med. 2016, 194, 1337–1348. [Google Scholar] [CrossRef]
- Schneider, M.; Bernasch, D.; Weymann, J.; Holle, R.; Bartsch, P. Acute mountain sickness: Influence of susceptibility, preexposure, and ascent rate. Med. Sci. Sports Exerc. 2002, 34, 1886–1891. [Google Scholar] [CrossRef]
- Imray, C.; Wright, A.; Subudhi, A.; Roach, R. Acute mountain sickness: Pathophysiology, prevention, and treatment. Prog. Cardiovasc. Dis. 2010, 52, 467–484. [Google Scholar] [CrossRef] [PubMed]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Evans, A.M.; Hardie, D.G. AMPK and the Need to Breathe and Feed: What’s the Matter with Oxygen? Int. J. Mol. Sci. 2020, 21, 3518. https://doi.org/10.3390/ijms21103518
Evans AM, Hardie DG. AMPK and the Need to Breathe and Feed: What’s the Matter with Oxygen? International Journal of Molecular Sciences. 2020; 21(10):3518. https://doi.org/10.3390/ijms21103518
Chicago/Turabian StyleEvans, A. Mark, and D. Grahame Hardie. 2020. "AMPK and the Need to Breathe and Feed: What’s the Matter with Oxygen?" International Journal of Molecular Sciences 21, no. 10: 3518. https://doi.org/10.3390/ijms21103518
APA StyleEvans, A. M., & Hardie, D. G. (2020). AMPK and the Need to Breathe and Feed: What’s the Matter with Oxygen? International Journal of Molecular Sciences, 21(10), 3518. https://doi.org/10.3390/ijms21103518




