Peritoneal Fluid Cytokines Reveal New Insights of Endometriosis Subphenotypes
Abstract
1. Introduction
2. Results
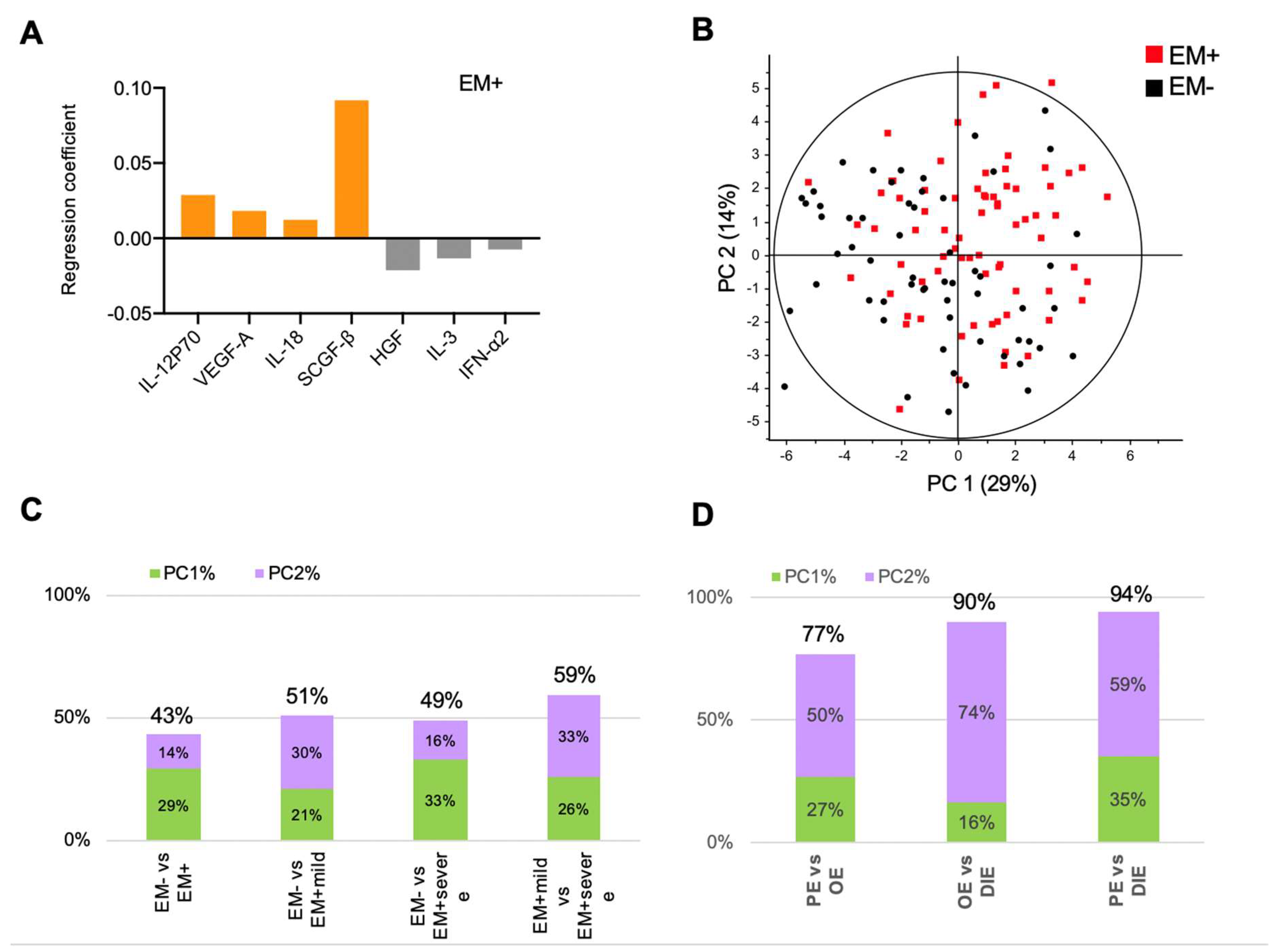
2.1. Peritoneal Fluid Cytokines Inadequately Describe Endometriosis Stages and Their Heterogeneity
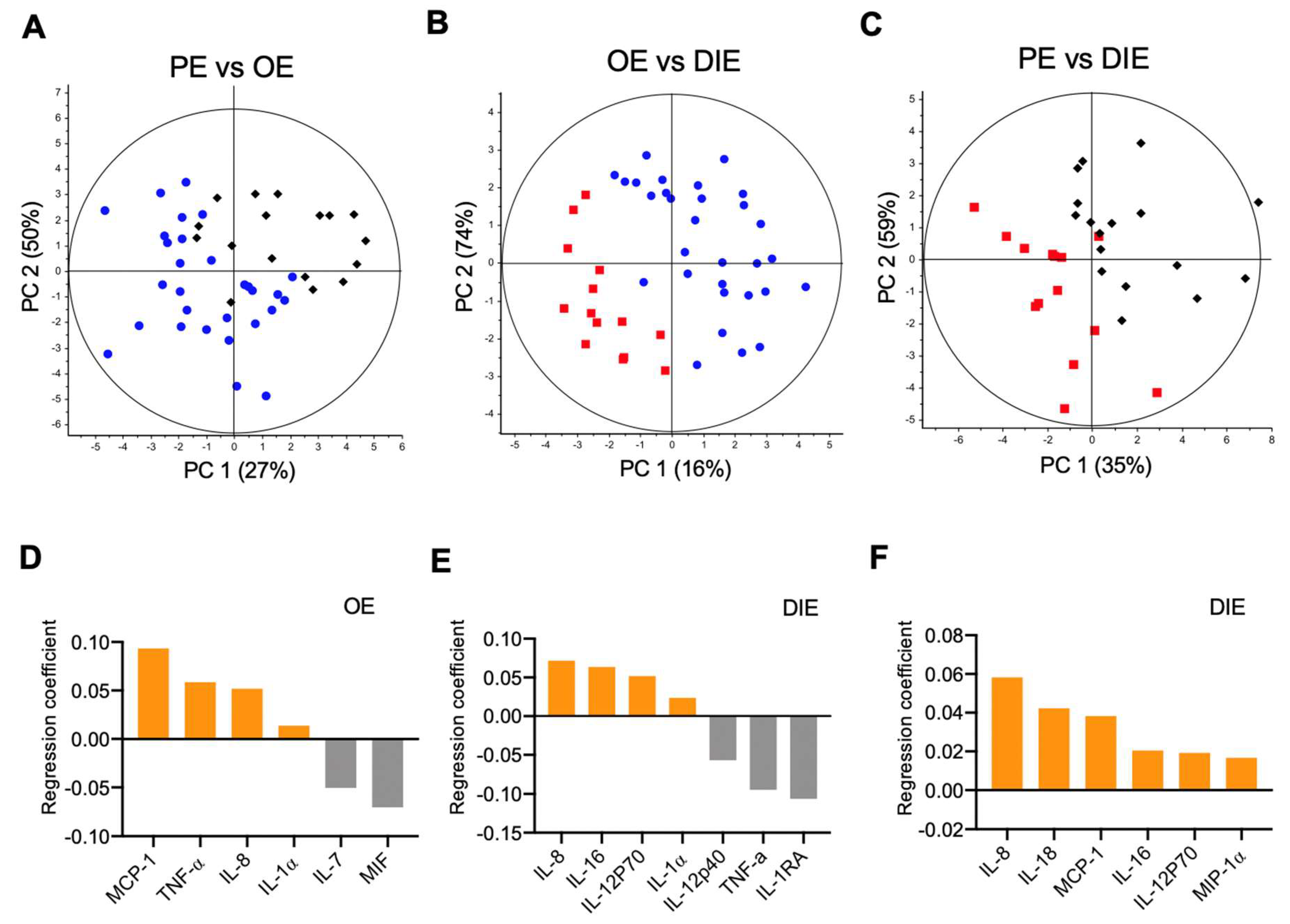
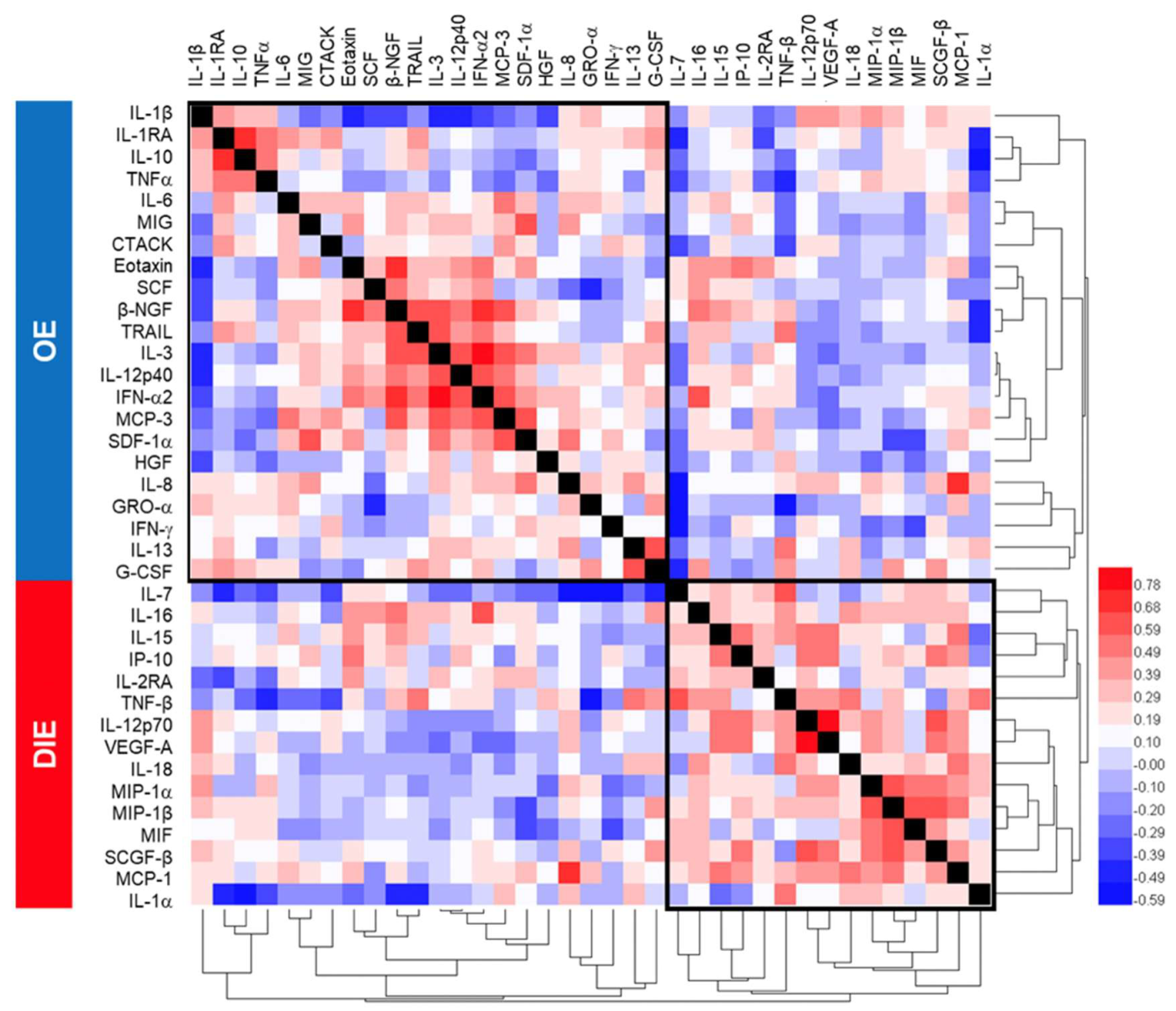
2.2. Peritoneal Fluid Cytokine Signatures Delineate Endometriosis Subphenotypes
2.3. Subphenotype Cytokine Signatures Are Associated with Different Biological Processes.
3. Discussion
4. Materials and Methods
4.1. Subjects and Sample Collection
4.2. Multiplex Immunoassay Analysis
4.3. Sample Size Calculation
4.4. Statistical Analysis
4.5. Pathway Enrichment Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ASRM | American Society for Reproductive Medicine |
| DIE | Deep infiltrating endometriosis |
| EM- | Control without endometriosis |
| EM+ | Case with endometriosis |
| OE | Ovarian endometriosis |
| PC | Principal Component |
| PE | Peritoneal endometriosis |
| PF | Peritoneal fluid |
| PLSR | Partial least squares regression |
References
- Practice Committee of the American Society for Reproductive Medicine. Endometriosis and infertility. Fertil. Steril. 2006, 86, S156–S160. [Google Scholar] [CrossRef] [PubMed]
- Bulun, S.E. Endometriosis. N. Engl. J. Med. 2009, 360, 268–279. [Google Scholar] [CrossRef] [PubMed]
- Nisolle, M.; Donnez, J. Peritoneal endometriosis, ovarian endometriosis, and adenomyotic nodules of the rectovaginal septum are three different entities. Fertil. Steril. 1997, 68, 585–596. [Google Scholar] [CrossRef]
- Stegmann, B.J.; Sinaii, N.; Liu, S.; Segars, J.; Merino, M.; Nieman, L.K.; Stratton, P. Using location, color, size, and depth to characterize and identify endometriosis lesions in a cohort of 133 women. Fertil. Steril. 2008, 89, 1632–1636. [Google Scholar] [CrossRef] [PubMed]
- Burney, R.O.; Giudice, L.C. Pathogenesis and pathophysiology of endometriosis. Fertil. Steril. 2012, 98, 511–519. [Google Scholar] [CrossRef] [PubMed]
- van Langendonckt, A.; Casanas-Roux, F.; Donnez, J. Oxidative stress and peritoneal endometriosis. Fertil. Steril. 2002, 77, 861–870. [Google Scholar] [CrossRef]
- Yamaguchi, K.; Mandai, M.; Toyokuni, S.; Hamanishi, J.; Higuchi, T.; Takakura, K.; Fujii, S. Contents of endometriotic cysts, especially the high concentration of free iron, are a possible cause of carcinogenesis in the cysts through the iron-induced persistent oxidative stress. Clin. Cancer Res. 2008, 14. [Google Scholar] [CrossRef]
- Guo, S.W.; Ding, D.; Shen, M.; Liu, X. Dating endometriotic ovarian cysts based on the content of cyst fluid and its potential clinical implications. Reprod. Sci. 2015, 22, 873–883. [Google Scholar] [CrossRef]
- Koninckx, P.R.; Meuleman, C.; Demeyere, S.; Lesaffre, E.; Cornillie, F.J. Suggestive evidence that pelvic endometriosis is a progressive disease, whereas deeply infiltrating endometriosis is associated with pelvic pain. Fertil. Steril. 1991, 55, 759–765. [Google Scholar] [CrossRef]
- Chapron, C.; Fauconnier, A.; Vieira, M.; Barakat, H.; Dousset, B.; Pansini, V.; Vacher-Lavenu, M.C.; Dubuisson, J.B. Anatomical distribution of deeply infiltrating endometriosis: Surgical implications and proposition for a classification. Hum. Reprod. 2003, 18, 157–161. [Google Scholar] [CrossRef]
- Koninckx, P.R.; Kennedy, S.H.; Barlow, D.H. Endometriotic disease: The role of peritoneal fluid. Hum. Reprod. Update 1998, 4, 741–751. [Google Scholar] [CrossRef] [PubMed]
- Laganà, A.S.; Garzon, S.; Götte, M.; Viganò, P.; Franchi, M.; Ghezzi, F.; Martin, D.C. The Pathogenesis of Endometriosis: Molecular and Cell Biology Insights. Int. J. Mol. Sci. 2019, 20, 5615. [Google Scholar] [CrossRef] [PubMed]
- Riemma, G.; Laganà, A.S.; Schiattarella, A.; Garzon, S.; Cobellis, L.; Autiero, R.; Licciardi, F.; della Corte, L.; la Verde, M.; de Franciscis, P. Ion Channels in The Pathogenesis of Endometriosis: A Cutting-Edge Point of View. Int. J. Mol. Sci. 2020, 21, 1114. [Google Scholar] [CrossRef] [PubMed]
- Gordts, S.; Koninckx, P.; Brosens, I. Pathogenesis of deep endometriosis. Fertil. Steril. 2017, 108, 872–885. [Google Scholar] [CrossRef]
- Flores, V.A.; Vanhie, A.; Dang, T.; Taylor, H.S. Progesterone Receptor Status Predicts Response to Progestin Therapy in Endometriosis. J. Clin. Endocrinol. Metab. 2018, 103, 4561–4568. [Google Scholar] [CrossRef]
- Zondervan, K.T.; Becker, C.M.; Missmer, S.A. Endometriosis. N. Engl. J. Med. 2020, 382, 1244–1256. [Google Scholar] [CrossRef]
- Herington, J.L.; Bruner-Tran, K.L.; Lucas, J.A.; Osteen, K.G. Immune interactions in endometriosis. Expert Rev. Clin. Immunol. 2011, 7, 611–626. [Google Scholar] [CrossRef]
- Lee, Y.H.; Tan, C.W.; Venkatratnam, A.; Tan, C.S.; Cui, L.; Loh, S.F.; Griffith, L.; Tannenbaum, S.R.; Chan, J.K.Y. Dysregulated sphingolipid metabolism in endometriosis. J. Clin. Endocrinol. Metab. 2014, 99, E1913–E1921. [Google Scholar] [CrossRef]
- Lee, Y.H.; Cui, L.; Fang, J.; Chern, B.S.M.; Tan, H.H.; Chan, J.K.Y. Limited value of pro-inflammatory oxylipins and cytokines as circulating biomarkers in endometriosis—A targeted ‘omics study. Sci. Rep. 2016, 6, 26117. [Google Scholar] [CrossRef]
- Bhat, R.; Bissell, M.J. Of plasticity and specificity: Dialectics of the microenvironment and macroenvironment and the organ phenotype, Wiley Interdiscip. Rev. Dev. Biol. 2014, 3, 147–163. [Google Scholar] [CrossRef]
- Schmitz, M.L.; Weber, A.; Roxlau, T.; Gaestel, M.; Kracht, M. Signal integration, crosstalk mechanisms and networks in the function of inflammatory cytokines. Biochim. Biophys. Acta Mol. Cell Res. 2011, 1813, 2165–2175. [Google Scholar] [CrossRef] [PubMed]
- Pinheiro, M.B.; Martins-Filho, O.A.; Mota, A.P.L.; Alpoim, P.N.; Godoi, L.C.; Silveira, A.C.O.; Teixeira-Carvalho, A.; Gomes, K.B.; Dusse, L.M. Severe preeclampsia goes along with a cytokine network disturbance towards a systemic inflammatory state. Cytokine 2013, 62, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Siedentopf, F.; Tariverdian, N.; Rücke, M.; Kentenich, H.; Arck, P.C. Immune Status, Psychosocial Distress and Reduced Quality of Life in Infertile Patients with Endometriosis. Am. J. Reprod. Immunol. 2008, 60, 449–461. [Google Scholar] [CrossRef] [PubMed]
- Khan, K.N.; Masuzaki, H.; Fujishita, A.; Kitajima, M.; Sekine, I.; Ishimaru, T. Differential macrophage infiltration in early and advanced endometriosis and adjacent peritoneum. Fertil. Steril. 2004, 81, 652–661. [Google Scholar] [CrossRef]
- Beste, M.T.; Pfäffle-Doyle, N.; Prentice, E.A.; Morris, S.N.; Lauffenburger, D.A.; Isaacson, K.B.; Griffith, L.G. Molecular network analysis of endometriosis reveals a role for c-Jun-regulated macrophage activation. Sci. Transl. Med. 2014, 6, 222ra16. [Google Scholar] [CrossRef]
- Gottschalk, P.G.; Dunn, J.R. The five-parameter logistic: A characterization and comparison with the four-parameter logistic. Anal. Biochem. 2005, 343, 54–65. [Google Scholar] [CrossRef]
- Rakhila, H.; Al-Akoum, M.; Bergeron, M.-E.; Leboeuf, M.; Lemyre, M.; Akoum, A.; Pouliot, M. Promotion of angiogenesis and proliferation cytokines patterns in peritoneal fluid from women with endometriosis. J. Reprod. Immunol. 2016, 116, 1–6. [Google Scholar] [CrossRef]
- Vercellini, P.; Viganò, P.; Somigliana, E.; Fedele, L. Endometriosis: Pathogenesis and treatment. Nat. Rev. Endocrinol. 2013, 10, 261–275. [Google Scholar] [CrossRef]
- Bersinger, N.A.; Dechaud, H.; McKinnon, B.; Mueller, M.D. Analysis of cytokines in the peritoneal fluid of endometriosis patients as a function of the menstrual cycle stage using the Bio-Plex® platform. Arch. Physiol. Biochem. 2012, 118, 210–218. [Google Scholar] [CrossRef]
- Jørgensen, H.; Hill, A.S.; Beste, M.T.; Kumar, M.P.; Chiswick, E.; Fedorcsak, P.; Isaacson, K.B.; Lauffenburger, D.A.; Griffith, L.G.; Qvigstad, E. Peritoneal fluid cytokines related to endometriosis in patients evaluated for infertility. Fertil. Steril. 2017, 107, 1191–1199. [Google Scholar] [CrossRef]
- Mahnke, J.L.; Dawood, M.Y.; Huang, J.C. Vascular endothelial growth factor and interleukin-6 in peritoneal fluid of women with endometriosis. Fertil. Steril. 2000, 73, 166–170. [Google Scholar] [CrossRef]
- Knific, T.; Fishman, D.; Vogler, A.; Gstöttner, M.; Wenzl, R.; Peterson, H.; Rižner, T.L. Multiplex analysis of 40 cytokines do not allow separation between endometriosis patients and controls. Sci. Rep. 2019, 9, 16738. [Google Scholar] [CrossRef] [PubMed]
- Cornillie, F.J.; Oosterlynck, D.; Lauweryns, J.M.; Koninckx, P.R. Deeply infiltrating pelvic endometriosis: Histology and clinical significance. Fertil. Steril. 1990, 53, 978–983. [Google Scholar] [CrossRef]
- Sun, L.; He, C.; Nair, L.; Yeung, J.; Egwuagu, C.E. Interleukin 12 (IL-12) family cytokines: Role in immune pathogenesis and treatment of CNS autoimmune disease. Cytokine 2015, 75, 249–255. [Google Scholar] [CrossRef]
- van Kaam, K.J.A.F.; Schouten, J.P.; Nap, A.W.; Dunselman, G.A.J.; Groothuis, P.G. Fibromuscular differentiation in deeply infiltrating endometriosis is a reaction of resident fibroblasts to the presence of ectopic endometrium. Hum. Reprod. 2008, 23, 2692–2700. [Google Scholar] [CrossRef]
- Carmona, F.; Chapron, C.; Martínez-Zamora, M.-Á.; Santulli, P.; Rabanal, A.; Martínez-Florensa, M.; Lozano, F.; Balasch, J. Ovarian endometrioma but not deep infiltrating endometriosis is associated with increased serum levels of interleukin-8 and interleukin-6. J. Reprod. Immunol. 2012, 95, 80–86. [Google Scholar] [CrossRef]
- Santulli, P.; Chouzenoux, S.; Fiorese, M.; Marcellin, L.; Lemarechal, H.; Millischer, A.E.; Batteux, F.; Borderie, D.; Chapron, C. Protein oxidative stress markers in peritoneal fluids of women with deep infiltrating endometriosis are increased. Hum. Reprod. 2015, 30, 49–60. [Google Scholar] [CrossRef]
- Fagotti, A. Analysis of cyclooxygenase-2 (COX-2) expression in different sites of endometriosis and correlation with clinico-pathological parameters. Hum. Reprod. 2004, 19, 393–397. [Google Scholar] [CrossRef]
- Anaf, V.; Simon, P.; el Nakadi, I.; Fayt, I.; Simonart, T.; Buxant, F.; Noel, J.-C. Hyperalgesia, nerve infiltration and nerve growth factor expression in deep adenomyotic nodules, peritoneal and ovarian endometriosis. Hum. Reprod. 2002, 17, 1895–1900. [Google Scholar] [CrossRef]
- Borghese, B.; Barbaux, S.; Mondon, F.; Santulli, P.; Pierre, G.; Vinci, G.; Chapron, C.; Vaiman, D. Research Resource: Genome-Wide Profiling of Methylated Promoters in Endometriosis Reveals a Subtelomeric Location of Hypermethylation. Mol. Endocrinol. 2010, 24, 1872–1885. [Google Scholar] [CrossRef]
- Bedaiwy, M.A.; Alfaraj, S.; Yong, P.; Casper, R. New developments in the medical treatment of endometriosis. Fertil. Steril. 2017, 107, 555–565. [Google Scholar] [CrossRef] [PubMed]
- Dunselman, G.A.J.; Vermeulen, N.; Becker, C.; Calhaz-Jorge, C.; D’Hooghe, T.; de Bie, B.; Heikinheimo, O.; Horne, A.W.; Kiesel, L.; Nap, A.; et al. ESHRE guideline: Management of women with endometriosis. Hum. Reprod. 2014, 29, 400–412. [Google Scholar] [CrossRef] [PubMed]
- Altintas, D.; Kokcu, A.; Kandemir, B.; Cetinkaya, M.B.; Tosun, M. Efficacy of imiquimod, an immunomodulatory agent, on experimental endometriosis. Fertil. Steril. 2008, 90, 401–405. [Google Scholar] [CrossRef] [PubMed]
- Uygur, D.; Aytan, H.; Zergeroglu, S.; Batioglu, S. Leflunomide—An Immunomodulator—Induces Regression of Endometrial Explants in a Rat Model of Endometriosis. J. Soc. Gynecol. Investig. 2006, 13, 378–383. [Google Scholar] [CrossRef]
- Barrier, B.F.; Bates, G.W.; Leland, M.M.; Leach, D.A.; Robinson, R.D.; Propst, A.M. Efficacy of anti-tumor necrosis factor therapy in the treatment of spontaneous endometriosis in baboons. Fertil. Steril. 2004, 81, 775–779. [Google Scholar] [CrossRef]
- Koninckx, P.R.; Craessaerts, M.; Timmerman, D.; Cornillie, F.; Kennedy, S. Anti-TNF-treatment for deep endometriosis-associated pain: A randomized placebo-controlled trial. Hum. Reprod. 2008, 23, 2017–2023. [Google Scholar] [CrossRef]
- Itoh, H.; Sashihara, T.; Hosono, A.; Kaminogawa, S.; Uchida, M. Interleukin-12 inhibits development of ectopic endometriotic tissues in peritoneal cavity via activation of NK cells in a murine endometriosis model. Cytotechnology 2011, 63, 133–141. [Google Scholar] [CrossRef]
- Oosterlynck, D.J.; Meuleman, C.; Waer, M.; Koninckx, P.R.; Vandeputte, M. Immunosuppressive activity of peritoneal fluid in women with endometriosis. Obstet. Gynecol. 1993, 82, 206–212. [Google Scholar]
- Ścieżyńska, A.; Komorowski, M.; Soszyńska, M.; Malejczyk, J. NK Cells as Potential Targets for Immunotherapy in Endometriosis. J. Clin. Med. 2019, 8, 1468. [Google Scholar] [CrossRef]
- Durairaj, R.R.P.; Aberkane, A.; Polanski, L.; Maruyama, Y.; Baumgarten, M.; Lucas, E.S.; Quenby, S.; Chan, J.K.Y.; Raine-Fenning, N.; Brosens, J.J.; et al. Deregulation of the endometrial stromal cell secretome precedes embryo implantation failure. MHR Basic Sci. Reprod. Med. 2017, 23, 478–487. [Google Scholar] [CrossRef]
- Symons, L.K.; Miller, J.E.; Kay, V.R.; Marks, R.M.; Liblik, K.; Koti, M.; Tayade, C. The Immunopathophysiology of Endometriosis. Trends Mol. Med. 2018, 24, 748–762. [Google Scholar] [CrossRef] [PubMed]
- Vallvé-Juanico, J.; Houshdaran, S.; Giudice, L.C. The endometrial immune environment of women with endometriosis. Hum. Reprod. Update 2019, 25, 565–592. [Google Scholar] [CrossRef] [PubMed]
- Ahn, S.H.; Singh, V.; Tayade, C. Biomarkers in endometriosis: Challenges and opportunities. Fertil. Steril. 2017, 107, 523–532. [Google Scholar] [CrossRef] [PubMed]
- May, K.E.; Villar, J.; Kirtley, S.; Kennedy, S.H.; Becker, C.M. Endometrial alterations in endometriosis: A systematic review of putative biomarkers. Hum. Reprod. Update 2011, 17, 637–653. [Google Scholar] [CrossRef] [PubMed]
- Anastasiu, C.V.; Moga, M.A.; Neculau, A.E.; Bălan, A.; Scârneciu, I.; Dragomir, R.M.; Dull, A.-M.; Chicea, L.-M. Biomarkers for the Noninvasive Diagnosis of Endometriosis: State of the Art and Future Perspectives. Int. J. Mol. Sci. 2020, 21, 1750. [Google Scholar] [CrossRef]
- Wimalachandra, D.; Yang, J.X.; Zhu, L.; Tan, E.; Asada, H.; Chan, J.Y.K. Lee, Y.H. Long-chain glucosylceramides crosstalk with LYN mediates endometrial cell migration. Biochim. Biophys Acta. Mol. Cell Biol. Lipids. 2018, 1863, 71–80. [Google Scholar] [CrossRef]
- Huhtinen, K.; Desai, R.; Ståhle, M.; Salminen, A.; Handelsman, D.J.; Perheentupa, A.; Poutanen, M. Endometrial and Endometriotic Concentrations of Estrone and Estradiol Are Determined by Local Metabolism Rather than Circulating Levels. J. Clin. Endocrinol. Metab. 2012, 97, 4228–4235. [Google Scholar] [CrossRef]
- Kyama, C.; Overbergh, L.; Debrock, S.; Valckx, D.; Vanderperre, S.; Meuleman, C.; Mihalyi, A.; Mwenda, J.; Mathieu, C.; Dhooghe, T. Increased peritoneal and endometrial gene expression of biologically relevant cytokines and growth factors during the menstrual phase in women with endometriosis. Fertil. Steril. 2006, 85, 1667–1675. [Google Scholar] [CrossRef]
- Kyama, C.M.; Overbergh, L.; Mihalyi, A.; Meuleman, C.; Mwenda, J.M.; Mathieu, C.; D’Hooghe, T.M. Endometrial and peritoneal expression of aromatase, cytokines, and adhesion factors in women with endometriosis. Fertil. Steril. 2008, 89, 301–310. [Google Scholar] [CrossRef]
- Young, V.J.; Brown, J.K.; Saunders, P.T.K.; Horne, A.W. The role of the peritoneum in the pathogenesis of endometriosis. Hum. Reprod. Update 2013, 19, 558–569. [Google Scholar] [CrossRef]
- Tani, H.; Sato, Y.; Ueda, M.; Miyazaki, Y.; Suginami, K.; Horie, A.; Konishi, I.; Shinomura, T. Role of Versican in the Pathogenesis of Peritoneal Endometriosis. J. Clin. Endocrinol. Metab. 2016, 101, 4349–4356. [Google Scholar] [CrossRef] [PubMed]
- Scutiero, G.; Iannone, P.; Bernardi, G.; Bonaccorsi, G.; Spadaro, S.; Volta, C.A.; Greco, P.; Nappi, L. Oxidative Stress and Endometriosis: A Systematic Review of the Literature. Oxid. Med. Cell. Longev. 2017, 2017, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Vitale, S.G.; Capriglione, S.; Peterlunger, I.; la Rosa, V.L.; Vitagliano, A.; Noventa, M.; Valenti, G.; Sapia, F.; Angioli, R.; Lopez, S.; et al. The Role of Oxidative Stress and Membrane Transport Systems during Endometriosis: A Fresh Look at a Busy Corner. Oxid. Med. Cell. Longev. 2018, 2018, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.H.; Yang, J.X.; Allen, J.C.; Tan, C.S.; Chern, B.S.M.; Tan, T.Y.; Tan, H.H.; Mattar, C.N.Z.; Chan, J.K.Y. Elevated peritoneal fluid ceramides in human endometriosis-associated infertility and their effects on mouse oocyte maturation. Fertil. Steril. 2018, 110, 767–777. [Google Scholar] [CrossRef]
- Leconte, M.; Nicco, C.; Ngô, C.; Arkwright, S.; Chéreau, C.; Guibourdenche, J.; Weill, B.; Chapron, C.; Dousset, B.; Batteux, F. Antiproliferative Effects of Cannabinoid Agonists on Deep Infiltrating Endometriosis. Am. J. Pathol. 2010, 177, 2963–2970. [Google Scholar] [CrossRef]
- Chapron, C.; Souza, C.; de Ziegler, D.; Lafay-Pillet, M.-C.; Ngô, C.; Bijaoui, G.; Goffinet, F.; Borghese, B. Smoking habits of 411 women with histologically proven endometriosis and 567 unaffected women. Fertil. Steril. 2010, 94, 2353–2355. [Google Scholar] [CrossRef]
- Straussman, R.; Morikawa, T.; Shee, K.; Barzily-Rokni, M.; Qian, Z.R.; Du, J.; Davis, A.; Mongare, M.M.; Gould, J.; Frederick, D.T.; et al. Tumour micro-environment elicits innate resistance to RAF inhibitors through HGF secretion. Nature 2012, 487, 500–504. [Google Scholar] [CrossRef]
- Suryawanshi, S.; Huang, X.; Elishaev, E.; Budiu, R.A.; Zhang, L.; Kim, S.; Donnellan, N.; Mantia-Smaldone, G.; Ma, T.; Tseng, G.; et al. Complement Pathway Is Frequently Altered in Endometriosis and Endometriosis-Associated Ovarian Cancer. Clin. Cancer Res. 2014, 20, 6163–6174. [Google Scholar] [CrossRef]
- Zamani, M.R.; Salmaninejad, A.; Asbagh, F.A.; Masoud, A.; Rezaei, N. STAT4 single nucleotide gene polymorphisms and susceptibility to endometriosis-related infertility. Eur. J. Obstet. Gynecol. Reprod. Biol. 2016, 203, 20–24. [Google Scholar] [CrossRef]
- Bianco, B.; Fernandes, R.F.M.; Trevisan, C.M.; Christofolini, D.M.; Sanz-Lomana, C.M.; Bernabe, J.V.; Barbosa, C.P. Influence of STAT4 gene polymorphisms in the pathogenesis of endometriosis. Ann. Hum. Genet. 2019, 83, 249–255. [Google Scholar] [CrossRef]
- Fasciani, A. High concentrations of the vascular endothelial growth factor and interleukin-8 in ovarian endometriomata. Mol. Hum. Reprod. 2000, 6, 50–54. [Google Scholar] [CrossRef] [PubMed]
- Jana, S.; Chatterjee, K.; Ray, A.K.; DasMahapatra, P.; Swarnakar, S. Regulation of Matrix Metalloproteinase-2 Activity by COX-2-PGE2-pAKT Axis Promotes Angiogenesis in Endometriosis. PLoS ONE 2016, 11, e0163540. [Google Scholar] [CrossRef] [PubMed]
- Thiruchelvam, U.; Wingfield, M.; O’Farrelly, C. Natural Killer Cells: Key Players in Endometriosis. Am. J. Reprod. Immunol. 2015, 74, 291–301. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, Y.; Mori, T.; Ito, F.; Koshiba, A.; Takaoka, O.; Kataoka, H.; Maeda, E.; Okimura, H.; Mori, T.; Kitawaki, J. Exacerbation of Endometriosis Due to Regulatory T-Cell Dysfunction. J. Clin. Endocrinol. Metab. 2017, 102, 3206–3217. [Google Scholar] [CrossRef]
- Ferrero, S.; Evangelisti, G.; Barra, F. Current and emerging treatment options for endometriosis. Expert Opin. Pharmacother. 2018, 19, 1109–1125. [Google Scholar] [CrossRef]
- Rahmioglu, N.; Fassbender, A.; Vitonis, A.F.; Tworoger, S.S.; Hummelshoj, L.; D’Hooghe, T.M.; Adamson, G.D.; Giudice, L.C.; Becker, C.M.; Zondervan, K.T.; et al. World Endometriosis Research Foundation Endometriosis Phenome and Biobanking Harmonization Project: III. Fluid biospecimen collection, processing, and storage in endometriosis research. Fertil. Steril. 2014, 102, 1233–1243. [Google Scholar] [CrossRef]
- ASRM. Revised American Society for Reproductive Medicine classification of endometriosis: 1996. Fertil Steril. 1997, 67, 817–821. [Google Scholar] [CrossRef]
- AFS. Revised American Fertility Society classification of endometriosis: 1985. Fertil. Steril. 1985, 43, 351–352. [Google Scholar] [CrossRef]
- Somigliana, E.; Infantino, M.; Candiani, M.; Vignali, M.; Chiodini, A.; Busacca, M.; Vignali, M. Association rate between deep peritoneal endometriosis and other forms of the disease: Pathogenetic implications. Hum. Reprod. 2004, 19, 168–171. [Google Scholar] [CrossRef]
- Cochran, W.G. Methodological problems in the study of human populations. Ann. N. Y. Acad. Sci. 2006, 107, 476–489. [Google Scholar] [CrossRef]
- Huang, D.W.; Sherman, B.T.; Lempicki, R.A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc. 2009, 4, 44–57. [Google Scholar] [CrossRef] [PubMed]
- Yoav, B.; Daniel, Y. The control of the false discovery rate in multiple testing under dependency. Ann. Stat. 2001, 29, 1165–1188. [Google Scholar]
| Characteristics | EM- (n = 59) | EM+ (n = 73) | p-Value † |
|---|---|---|---|
| Age, y | 0.844 | ||
| Mean | 35 | 35 | |
| Range | 22–51 | 25–45 | |
| ASRM Stage | |||
| I–II | NA | 31 | |
| III–IV | NA | 42 | |
| Subtype | |||
| Peritoneal | NA | 17 | |
| Endometrioma | NA | 30 | |
| Deep infiltrating | NA | 14 | |
| Undetermined | NA | 12 | |
| Preoperative pain symptomsa | <0.0001 | ||
| Dysmenorrhea | 20 | 56 | |
| Dyspareunia b | 5 | 20 | |
| Menstrual phase | 0.290 | ||
| Proliferative | 27 | 41 | |
| Secretory | 32 | 32 | |
| Race | 0.650 | ||
| Chinese | 36 | 46 | |
| Malay | 14 | 13 | |
| Others c | 9 | 14 |
| (A) OE vs. PE | ||
|---|---|---|
| Term Identifier | Fold Enrichment | p-Value |
| immune response | 29.9 | 1.90 × 10−3 |
| positive regulation of B-cell proliferation | 215.3 | 7.00 × 10−3 |
| inflammatory response | 33.2 | 1.50 × 10−3 |
| negative regulation of extrinsic apoptotic signaling pathway in absence of ligand | 226.9 | 6.60 × 10−3 |
| positive regulation of angiogenesis | 73 | 2.00 × 10−2 |
| positive regulation of ERK1 and ERK2 cascade | 48 | 3.10 × 10−2 |
| cell proliferation | 22.9 | 6.40 × 10−2 |
| negative regulation of apoptotic process | 18.5 | 7.90 × 10−2 |
| (B) OE vs. DIE | ||
| Term Identifier | Fold Enrichment | p-Value |
| immune response | 31.9 | 6.10 × 10−5 |
| positive regulation of natural killer cell activation | 1343.4 | 1.20 × 10−3 |
| positive regulation of NK T-cell activation | 1343.4 | 1.20 × 10−3 |
| positive regulation of tyrosine phosphorylation of Stat4 protein | 1679.2 | 9.50 × 10−4 |
| positive regulation of lymphocyte proliferation | 959.5 | 1.70 × 10−3 |
| positive regulation of natural killer cell mediated cytotoxicity directed against tumor cell target | 959.5 | 1.70 × 10−3 |
| positive regulation of mononuclear cell proliferation | 2238.9 | 7.10 × 10−4 |
| response to UV-B | 746.3 | 2.10 × 10−3 |
| positive regulation of smooth muscle cell apoptotic process | 746.3 | 2.10 × 10−3 |
| negative regulation of interleukin-17 production | 610.6 | 2.60 × 10−3 |
| positive regulation of T-cell-mediated cytotoxicity | 516.7 | 3.10 × 10−3 |
| defense response to protozoan | 353.5 | 4.50 × 10−3 |
| negative regulation of smooth muscle cell proliferation | 231.6 | 6.90 × 10−3 |
| positive regulation of interferon-gamma production | 146 | 1.10 × 10−2 |
| positive regulation of cell adhesion | 156.2 | 1.00 × 10−2 |
| positive regulation of T-cell proliferation | 111.9 | 1.40 × 10−2 |
| cellular response to lipopolysaccharide | 59.4 | 2.70 × 10−2 |
| cytokine-mediated signaling pathway | 51.3 | 3.10 × 10−2 |
| cell cycle arrest | 47.6 | 3.30 × 10−2 |
| cell migration | 39.1 | 4.00 × 10−2 |
| (C) PE vs. DIE | ||
| Term Identifier | Fold Enrichment | p-Value |
| immune response | 39.9 | 1.60 × 10−5 |
| positive regulation of protein kinase B signaling | 100 | 1.50 × 10−2 |
| positive regulation of inflammatory response | 115 | 1.30 × 10−2 |
| cellular response to organic cyclic compound | 142.3 | 1.10 × 10−2 |
| positive regulation of interferon-gamma production | 182.5 | 8.20 × 10−3 |
| lipopolysaccharide-mediated signaling pathway | 262.4 | 5.70 × 10−3 |
| MAPK cascade | 32 | 4.60 × 10−2 |
| cell–cell signaling | 33.1 | 4.50 × 10−2 |
| inflammatory response | 22.2 | 6.60 × 10−2 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, J.; Chern, B.S.M.; Barton-Smith, P.; Phoon, J.W.L.; Tan, T.Y.; Viardot-Foucault, V.; Ku, C.W.; Tan, H.H.; Chan, J.K.Y.; Lee, Y.H. Peritoneal Fluid Cytokines Reveal New Insights of Endometriosis Subphenotypes. Int. J. Mol. Sci. 2020, 21, 3515. https://doi.org/10.3390/ijms21103515
Zhou J, Chern BSM, Barton-Smith P, Phoon JWL, Tan TY, Viardot-Foucault V, Ku CW, Tan HH, Chan JKY, Lee YH. Peritoneal Fluid Cytokines Reveal New Insights of Endometriosis Subphenotypes. International Journal of Molecular Sciences. 2020; 21(10):3515. https://doi.org/10.3390/ijms21103515
Chicago/Turabian StyleZhou, Jieliang, Bernard Su Min Chern, Peter Barton-Smith, Jessie Wai Leng Phoon, Tse Yeun Tan, Veronique Viardot-Foucault, Chee Wai Ku, Heng Hao Tan, Jerry Kok Yen Chan, and Yie Hou Lee. 2020. "Peritoneal Fluid Cytokines Reveal New Insights of Endometriosis Subphenotypes" International Journal of Molecular Sciences 21, no. 10: 3515. https://doi.org/10.3390/ijms21103515
APA StyleZhou, J., Chern, B. S. M., Barton-Smith, P., Phoon, J. W. L., Tan, T. Y., Viardot-Foucault, V., Ku, C. W., Tan, H. H., Chan, J. K. Y., & Lee, Y. H. (2020). Peritoneal Fluid Cytokines Reveal New Insights of Endometriosis Subphenotypes. International Journal of Molecular Sciences, 21(10), 3515. https://doi.org/10.3390/ijms21103515





