Anti-Aging Effect of the Ketone Metabolite β-Hydroxybutyrate in Drosophila Intestinal Stem Cells
Abstract
1. Introduction
2. Results
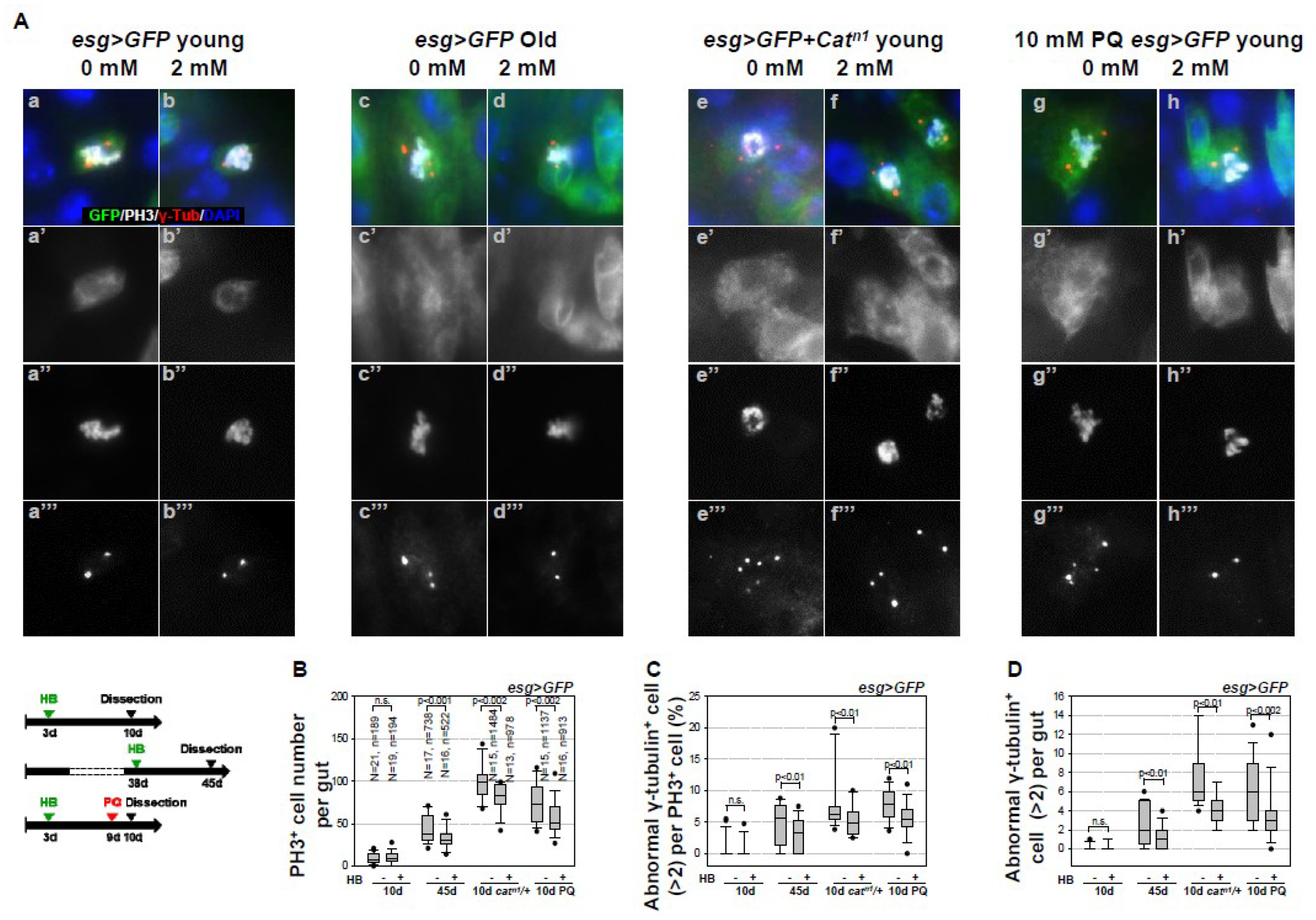
2.1. Inhibitory Effect of β-HB on Age- and Oxidative Stress-Related Centrosome Amplification in Drosophila Midgut ISCs
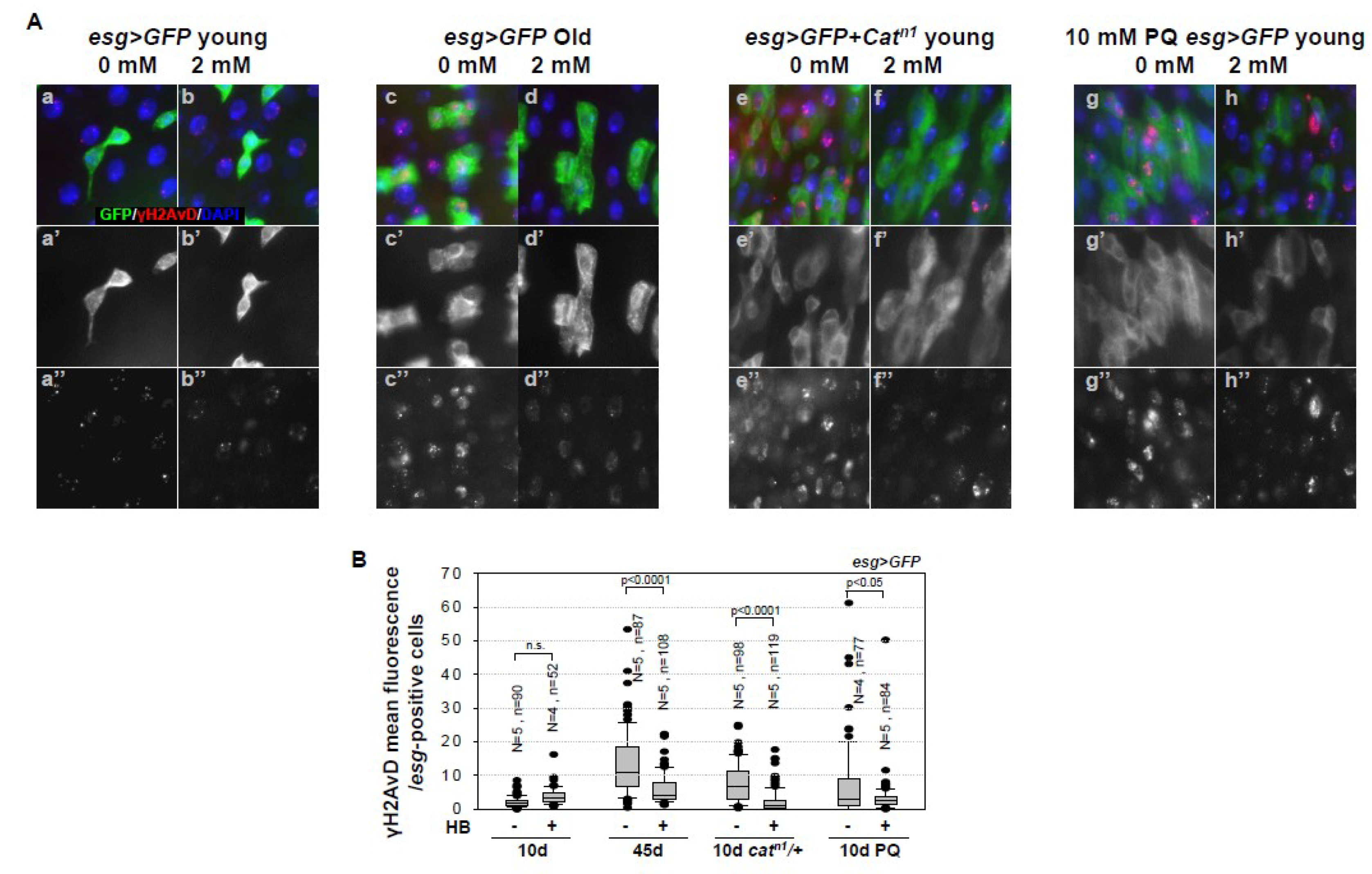
2.2. Inhibitory Effect of β-HB on Age-Related Increases in DNA Damage Accumulation in Midgut ISCs
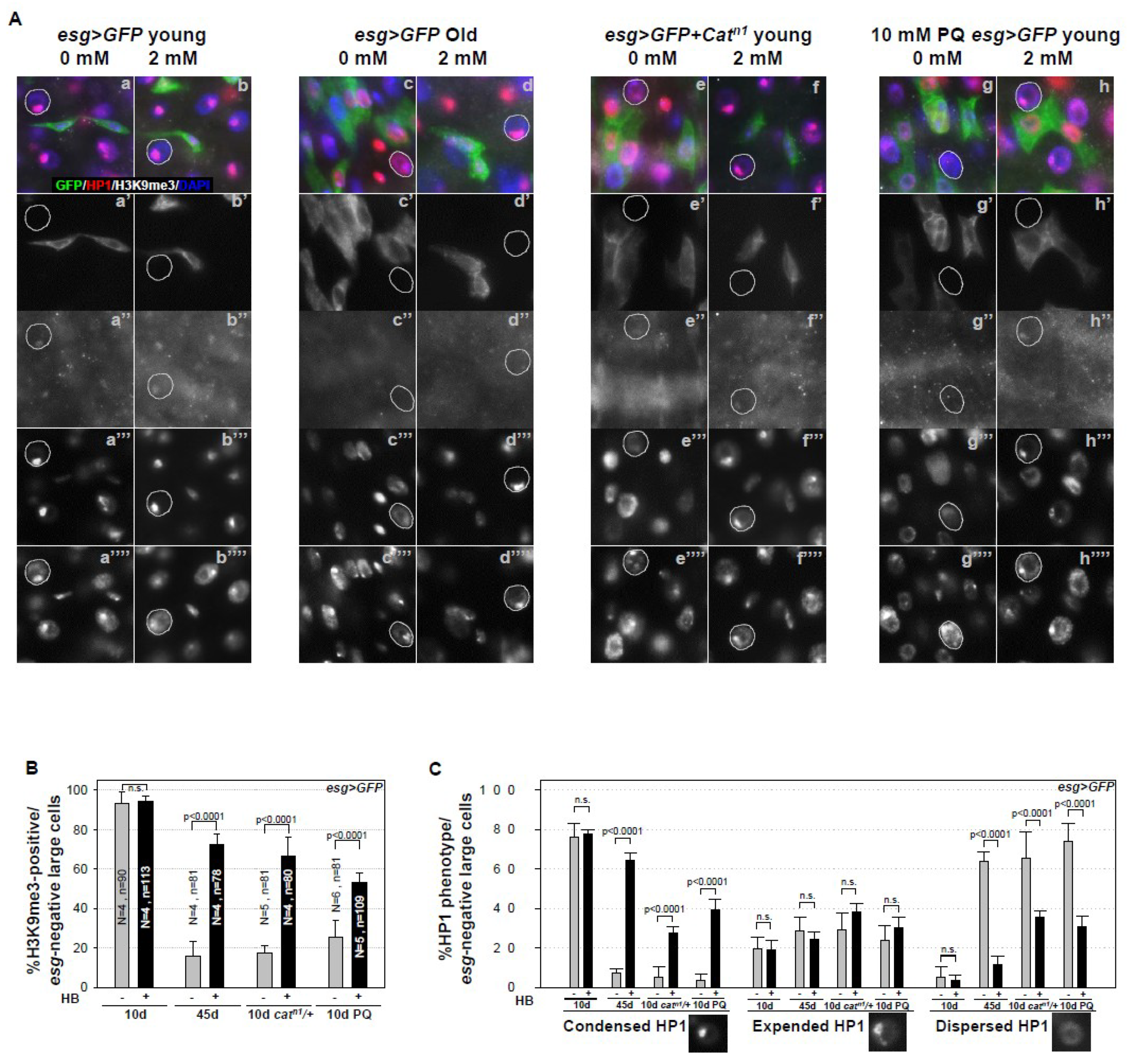
2.3. Inhibitory Effect of β-HB on Age-Related Loss of Heterochromatin Stability in Midgut ECs, ISC Niche Cells
3. Discussion
4. Material and Methods
4.1. Fly Stock
4.2. Immunochemistry
4.3. Antisera
4.4. β-Hydroxybutyrate Feeding Assay
4.5. β-Hydroxybutyrate Pre-Feeding Assay
4.6. Paraquat Feeding Assay
4.7. Quantitative Analysis
4.8. Quantification of γH2AvD Fluorescence Levels (Means)
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Jiang, H.; Edgar, B.A. Intestinal stem cells in the adult Drosophila midgut. Exp. Cell. Res. 2011, 317, 2780–2788. [Google Scholar] [CrossRef] [PubMed]
- Micchelli, C.A.; Perrimon, N. Evidence that stem cells reside in the adult Drosophila midgut epithelium. Nature 2006, 439, 475–479. [Google Scholar] [CrossRef] [PubMed]
- Ohlstein, B.; Spradling, A. The adult Drosophila posterior midgut is maintained by pluripotent stem cells. Nature 2006, 439, 470–474. [Google Scholar] [CrossRef] [PubMed]
- Ohlstein, B.; Spradling, A. Multipotent Drosophila intestinal stem cells specify daughter cell fates by differential notch signaling. Science 2007, 315, 988–992. [Google Scholar] [CrossRef]
- Apidianakis, Y.; Rahme, L.G. Drosophila melanogaster as a model for human intestinal infection and pathology. Dis. Model. Mech. 2011, 4, 21–30. [Google Scholar] [CrossRef]
- Biteau, B.; Hochmuth, C.E.; Jasper, H. JNK activity in somatic stem cells causes loss of tissue homeostasis in the aging Drosophila gut. Cell Stem Cell 2008, 3, 442–455. [Google Scholar] [CrossRef]
- Choi, N.H.; Kim, J.G.; Yang, D.J.; Kim, Y.S.; Yoo, M.A. Age-related changes in Drosophila midgut are associated with PVF2, a PDGF/VEGF-like growth factor. Aging Cell 2008, 7, 318–334. [Google Scholar] [CrossRef]
- Park, J.S.; Kim, Y.S.; Yoo, M.A. The role of p38b MAPK in age-related modulation of intestinal stem cell proliferation and differentiation in Drosophila. Aging 2009, 1, 637–651. [Google Scholar] [CrossRef]
- Rando, T.A. Stem cells, ageing and the quest for immortality. Nature 2006, 441, 1080–1086. [Google Scholar] [CrossRef]
- Spradling, A.; Drummond-Barbosa, D.; Kai, T. Stem cells find their niche. Nature 2001, 414, 98–104. [Google Scholar] [CrossRef]
- Mana, M.D.; Kuo, E.Y.; Yilmaz, Ö.H. Dietary regulation of adult stem cells. Curr. Stem Cell Rep. 2017, 3, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Longo, V.D.; Fontana, L. Calorie restriction and cancer prevention: Metabolic and molecular mechanisms. Trends Pharmacol. Sci. 2010, 31, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Maharajan, N.; Vijayakumar, K.; Jang, C.H.; Cho, G.W. Caloric restriction maintains stem cells through niche and regulates stem cell aging. J. Mol. Med. 2020, 98, 25–37. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.M.; Bedarida, T.; Ding, Y.; Somba, B.K.; Lu, Q.; Wang, Q.; Song, P.; Zou, M.H. β-Hydroxybutyrate Prevents Vascular Senescence through hnRNP A1-Mediated Upregulation of Oct. Mol. Cell. 2018, 71, 1064–1078. [Google Scholar] [CrossRef] [PubMed]
- Norwitz, N.G.; Hu, M.T.; Clarke, K. The mechanisms by which the ketone body d-β-hydroxybutyrate may improve the multiple cellular pathologies of parkinson’s disease. Front. Nutr. 2019, 6, 63. [Google Scholar] [CrossRef]
- Rodrigues, L.M.; Uribe-Lewis, S.; Madhu, B.; Honess, D.J.; Stubbs, M.; Griffiths, J.R. The action of β-hydroxybutyrate on the growth, metabolism and global histone H3 acetylation of spontaneous mouse mammary tumours: Evidence of a β-hydroxybutyrate paradox. Cancer Metab. 2017, 5, 4. [Google Scholar] [CrossRef]
- Newman, J.C.; Verdin, E. β-Hydroxybutyrate: A Signaling Metabolite. Annu. Rev. Nutr. 2017, 37, 51–76. [Google Scholar] [CrossRef]
- Koeslag, J.H.; Noakes, T.D.; Sloan, A.W. Post-exercise ketosis. J. Physiol. 1980, 301, 79–90. [Google Scholar] [CrossRef]
- Rogina, B.; Helfand, S.L. Sir2 mediates longevity in the fly through a pathway related to calorie restriction. Proc. Natl. Acad. Sci. USA 2004, 101, 15998–16003. [Google Scholar] [CrossRef]
- Veech, R.L.; Bradshaw, P.C.; Clarke, K.; Curtis, W.; Pawlosky, R.; King, M.T. Ketone bodies mimic the life span extending properties of caloric restriction. IUBMB Life 2017, 69, 305–314. [Google Scholar] [CrossRef]
- Veech, R.L. The therapeutic implications of ketone bodies: The effects of ketone bodies in pathological conditions: Ketosis, ketogenic diet, redox states, insulin resistance, and mitochondrial metabolism. Prostaglandins Leukot. Essent. Fat. Acids 2004, 70, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Edwards, C.; Canfield, J.; Copes, N.; Rehan, M.; Lipps, D.; Bradshaw, P.C. D-beta-hydroxybutyrate extends lifespan in C. elegans. Aging 2014, 6, 621–644. [Google Scholar] [CrossRef] [PubMed]
- Newman, J.C.; Milman, S.; Hashmi, S.K.; Austad, S.N.; Kirkland, J.L.; Halter, J.B.; Barzilai, N. Strategies and Challenges in Clinical Trials Targeting Human Aging. J. Gerontol. A Biol. Sci. Med. Sci. 2016, 71, 1424–1434. [Google Scholar] [CrossRef] [PubMed]
- Carabotti, M.; Scirocco, A.; Maselli, M.A.; Severi, C. The gut-brain axis: Interactions between enteric microbiota, central and enteric nervous systems. Ann. Gastroenterol. 2015, 28, 203–209. [Google Scholar] [PubMed]
- Buchon, N.; Broderick, N.A.; Poidevin, M.; Pradervand, S.; Lemaitre, B. Drosophila intestinal response to bacterial infection: Activation of host defense and stem cell proliferation. Cell Host Microbe. 2009, 5, 200–211. [Google Scholar] [CrossRef]
- Buchon, N.; Broderick, N.A.; Chakrabarti, S.; Lemaitre, B. Invasive and indigenous microbiota impact intestinal stem cell activity through multiple pathways in Drosophila. Genes Dev. 2009, 23, 2333–2344. [Google Scholar] [CrossRef]
- Lee, W.J. Bacterial-modulated host immunity and stem cell activation for gut homeostasis. Genes Dev. 2009, 23, 2260–2265. [Google Scholar] [CrossRef]
- Park, J.S.; Lee, S.H.; Na, H.J.; Pyo, J.H.; Kim, Y.S.; Yoo, M.A. Age- and oxidative stress-induced DNA damage in Drosophila intestinal stem cells as marked by Gamma-H2AX. Exp. Gerontol. 2012, 47, 401–405. [Google Scholar] [CrossRef]
- Na, H.J.; Park, J.S.; Pyo, J.H.; Lee, S.H.; Jeon, H.J.; Kim, Y.S.; Yoo, M.A. Mechanism of metformin: Inhibition of DNA damage and proliferative activity in Drosophila midgut stem cell. Mech. Ageing Dev. 2013, 134, 381–390. [Google Scholar] [CrossRef]
- Park, J.S.; Pyo, J.H.; Na, H.J.; Jeon, H.J.; Kim, Y.S.; Arking, R.; Yoo, M.A. Increased centrosome amplification in aged stem cells of the Drosophila midgut. Biochem. Biophys. Res. Commun. 2014, 450, 961–965. [Google Scholar] [CrossRef]
- Park, J.S.; Jeon, H.J.; Pyo, J.H.; Kim, Y.S.; Yoo, M.A. Deficiency in DNA damage response of enterocytes accelerates intestinal stem cell aging in Drosophila. Aging 2018, 10, 322–338. [Google Scholar] [CrossRef] [PubMed]
- Beebe, K.; Lee, W.C.; Micchelli, C.A. JAK/STAT signaling coordinates stem cell proliferation and multilineage differentiation in the Drosophila intestinal stem cell lineage. Dev. Biol. 2010, 338, 28–37. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Grenley, M.O.; Bravo, M.J.; Blumhagen, R.Z.; Edgar, B.A. EGFR/Ras/MAPK signaling mediates adult midgut epithelial homeostasis and regeneration in Drosophila. Cell Stem Cell 2011, 8, 84–95. [Google Scholar] [CrossRef] [PubMed]
- Bond, D.; Foley, E. Autocrine platelet-derived growth factor-vascular endothelial growth factor receptor-related (Pvr) pathway activity controls intestinal stem cell proliferation in the adult Drosophila midgut. J. Biol. Chem. 2012, 287, 27359–27370. [Google Scholar] [CrossRef]
- Ren, F.; Wang, B.; Yue, T.; Yun, E.Y.; Ip, Y.T.; Jiang, J. Hippo signaling regulates Drosophila intestine stem cell proliferation through multiple pathways. Proc. Natl. Acad. Sci. USA 2010, 107, 21064–21069. [Google Scholar] [CrossRef]
- Shaw, R.L.; Kohlmaier, A.; Polesello, C.; Veelken, C.; Edgar, B.A.; Tapon, N. The Hippo pathway regulates intestinal stem cell proliferation during Drosophila adult midgut regeneration. Development 2010, 137, 4147–4158. [Google Scholar] [CrossRef]
- Karpowicz, P.; Perez, J.; Perrimon, N. The Hippo tumor suppressor pathway regulates intestinal stem cell regeneration. Development 2010, 137, 4135–4145. [Google Scholar] [CrossRef]
- Choi, N.H.; Lucchetta, E.; Ohlstein, B. Nonautonomous regulation of Drosophila midgut stem cell proliferation by the insulin-signaling pathway. Proc. Natl. Acad. Sci. USA 2011, 108, 18702–18707. [Google Scholar] [CrossRef]
- Na, H.J.; Park, J.S.; Pyo, J.H.; Jeon, H.J.; Kim, Y.S.; Arking, R.; Yoo, M.A. Metformin inhibits age-related centrosome amplification in Drosophila midgut stem cells through AKT/TOR pathway. Mech. Ageing Dev. 2015, 149, 8–18. [Google Scholar] [CrossRef]
- Zhou, J.; Florescu, S.; Boettcher, A.L.; Luo, L.; Dutta, D.; Kerr, G.; Cai, Y.; Edgar, B.A.; Boutros, M. Dpp/Gbb signaling is required for normal intestinal regeneration during infection. Dev. Biol. 2015, 399, 189–203. [Google Scholar] [CrossRef]
- Osman, D.; Buchon, N.; Chakrabarti, S.; Huang, Y.T.; Su, W.C.; Poidevin, M.; Tsai, Y.C.; Lemaitre, B. Autocrine and paracrine unpaired signaling regulate intestinal stem cell maintenance and division. J. Cell. Sci. 2012, 125 Pt 24, 5944–5949. [Google Scholar] [CrossRef]
- Li, V.S.; Clevers, H. Intestinal regeneration: YAP-tumor suppressor and oncoprotein? Curr. Biol. 2013, 23, R110–R112. [Google Scholar] [CrossRef] [PubMed]
- Lin, G.; Xu, N.; Xi, R. Paracrine Wingless signalling controls self-renewal of Drosophila intestinal stem cells. Nature 2008, 455, 1119–1123. [Google Scholar] [CrossRef] [PubMed]
- Biteau, B.; Jasper, H. EGF signaling regulates the proliferation of intestinal stem cells in Drosophila. Development 2011, 138, 1045–1055. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Zhang, Y.; Han, L.; Shi, L.; Lin, X. Trachea-derived dpp controls adult midgut homeostasis in Drosophila. Dev. Cell 2013, 24, 133–143. [Google Scholar] [CrossRef]
- Nigg, E.A. Centrosome aberrations: Cause or consequence of cancer progression? Nat. Rev. Cancer 2002, 2, 815–825. [Google Scholar] [CrossRef]
- Xu, X.; Weaver, Z.; Linke, S.P.; Li, C.; Gotay, J.; Wang, X.W.; Harris, C.C.; Ried, T.; Deng, C.X. Centrosome amplification and a defective G2-M cell cycle checkpoint induce genetic instability in BRCA1 exon 11 isoform-deficient cells. Mol. Cell 1999, 3, 389–395. [Google Scholar] [CrossRef]
- Jeon, H.J.; Kim, Y.S.; Kim, J.G.; Heo, K.; Pyo, J.H.; Yamaguchi, M.; Park, J.S.; Yoo, M.A. Effect of heterochromatin stability on intestinal stem cell aging in Drosophila. Mech. Ageing Dev. 2018, 173, 50–60. [Google Scholar] [CrossRef]
- Lachner, M.; O’Carroll, D.; Rea, S.; Mechtler, K.; Jenuwein, T. Methylation of histone H3 lysine 9 creates a binding site for HP1 proteins. Nature 2001, 410, 116–120. [Google Scholar] [CrossRef]
- Cordero, J.B.; Stefanatos, R.K.; Scopelliti, A.; Vidal, M.; Sansom, O.J. Inducible progenitor-derived Wingless regulates adult midgut regeneration in Drosophila. EMBO J. 2012, 31, 3901–3917. [Google Scholar] [CrossRef]
- Lin, G.; Xi, R. Intestinal stem cell, muscular niche and Wingless signaling. Fly 2008, 2, 310–312. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jiang, H.; Patel, P.H.; Kohlmaier, A.; Grenley, M.O.; McEwen, D.G.; Edgar, B.A. Cytokine/Jak/Stat signaling mediates regeneration and homeostasis in the Drosophila midgut. Cell 2009, 137, 1343–1355. [Google Scholar] [CrossRef] [PubMed]
- Buchon, N.; Broderick, N.A.; Kuraishi, T.; Lemaitre, B. Drosophila EGFR pathway coordinates stem cell proliferation and gut remodeling following infection. BMC Biol. 2010, 8, 152. [Google Scholar] [CrossRef]
- Jiang, H.; Edgar, B.A. EGFR signaling regulates the proliferation of Drosophila adult midgut progenitors. Development 2009, 136, 483–493. [Google Scholar] [CrossRef]
- O’Brien, L.E.; Soliman, S.S.; Li, X.; Bilder, D. Altered modes of stem cell division drive adaptive intestinal growth. Cell 2011, 147, 603–614. [Google Scholar] [CrossRef]
- Biteau, B.; Karpac, J.; Supoyo, S.; Degennaro, M.; Lehmann, R.; Jasper, H. Lifespan extension by preserving proliferative homeostasis in Drosophila. PLoS Genet. 2010, 6, e1001159. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Halicka, H.D.; Traganos, F.; Darzynkiewicz, Z. Phosphorylation of histone H2AX on Ser 139 and activation of ATM during oxidative burst in phorbol ester-treated human leukocytes. Cell Cycle 2006, 5, 2671–2675. [Google Scholar] [CrossRef] [PubMed]
- Pyo, J.H.; Park, J.S.; Na, H.J.; Jeon, H.J.; Lee, S.H.; Kim, J.G.; Park, S.Y.; Jin, Y.W.; Kim, Y.S.; Yoo, M.A. Functional modification of Drosophila intestinal stem cells by ionizing radiation. Radiat. Res. 2014, 181, 376–386. [Google Scholar] [CrossRef]
- Shimazu, T.; Hirschey, M.D.; Newman, J.; He, W.; Shirakawa, K.; Le Moan, N.; Grueter, C.A.; Lim, H.; Saunders, L.R.; Stevens, R.D.; et al. Suppression of oxidative stress by β-hydroxybutyrate, an endogenous histone deacetylase inhibitor. Science 2013, 339, 211–214. [Google Scholar] [CrossRef]
- Tian, A.; Jiang, J. Intestinal epithelium-derived BMP controls stem cell self-renewal in Drosophila adult midgut. Elife 2014, 3, e01857. [Google Scholar] [CrossRef]
- Jiang, H.; Tian, A.; Jiang, J. Intestinal stem cell response to injury: Lessons from Drosophila. Cell. Mol. Life Sci. 2016, 73, 3337–3349. [Google Scholar] [CrossRef] [PubMed]
- Kux, K.; Pitsoul, I.C. Tissue communication in regenerative inflammatory signaling: Lessons from the fly gut. Front. Cell. Infect. Microbiol. 2014, 4, 49. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Zhou, Y.; Rychahou, P.; Fan, T.W.; Lane, A.N.; Weiss, H.L.; Evers, B.M. Ketogenesis contributes to intestinal cell differentiation. Cell Death Differ. 2017, 24, 458–468. [Google Scholar] [CrossRef] [PubMed]
- Griswold, C.M.; Matthews, A.L.; Bewley, K.E.; Mahaffey, J.W. Molecular characterization and rescue of acatalasemic mutants of Drosophila melanogaster. Genetics 1993, 134, 781–788. [Google Scholar] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, J.-S.; Kim, Y.-J. Anti-Aging Effect of the Ketone Metabolite β-Hydroxybutyrate in Drosophila Intestinal Stem Cells. Int. J. Mol. Sci. 2020, 21, 3497. https://doi.org/10.3390/ijms21103497
Park J-S, Kim Y-J. Anti-Aging Effect of the Ketone Metabolite β-Hydroxybutyrate in Drosophila Intestinal Stem Cells. International Journal of Molecular Sciences. 2020; 21(10):3497. https://doi.org/10.3390/ijms21103497
Chicago/Turabian StylePark, Joung-Sun, and Yung-Jin Kim. 2020. "Anti-Aging Effect of the Ketone Metabolite β-Hydroxybutyrate in Drosophila Intestinal Stem Cells" International Journal of Molecular Sciences 21, no. 10: 3497. https://doi.org/10.3390/ijms21103497
APA StylePark, J.-S., & Kim, Y.-J. (2020). Anti-Aging Effect of the Ketone Metabolite β-Hydroxybutyrate in Drosophila Intestinal Stem Cells. International Journal of Molecular Sciences, 21(10), 3497. https://doi.org/10.3390/ijms21103497




