Increased Thyroid Cancer Incidence in Volcanic Areas: A Role of Increased Heavy Metals in the Environment?
Abstract
1. Introduction
2. Volcanic Environment and Thyroid Cancer
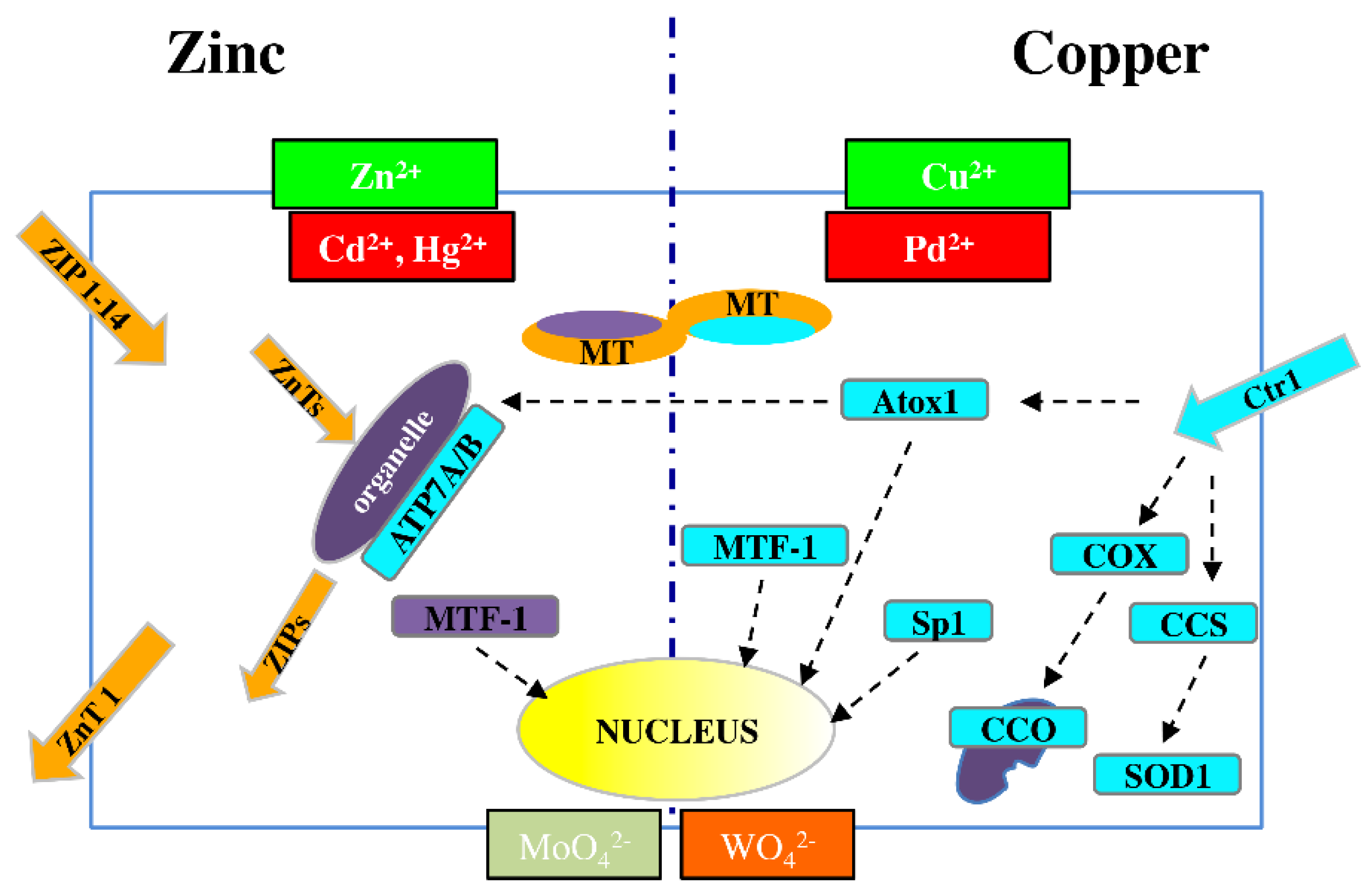
3. Biological Bases of Metal Homeostasis
4. Metal Carcinogenicity and the Thyroid
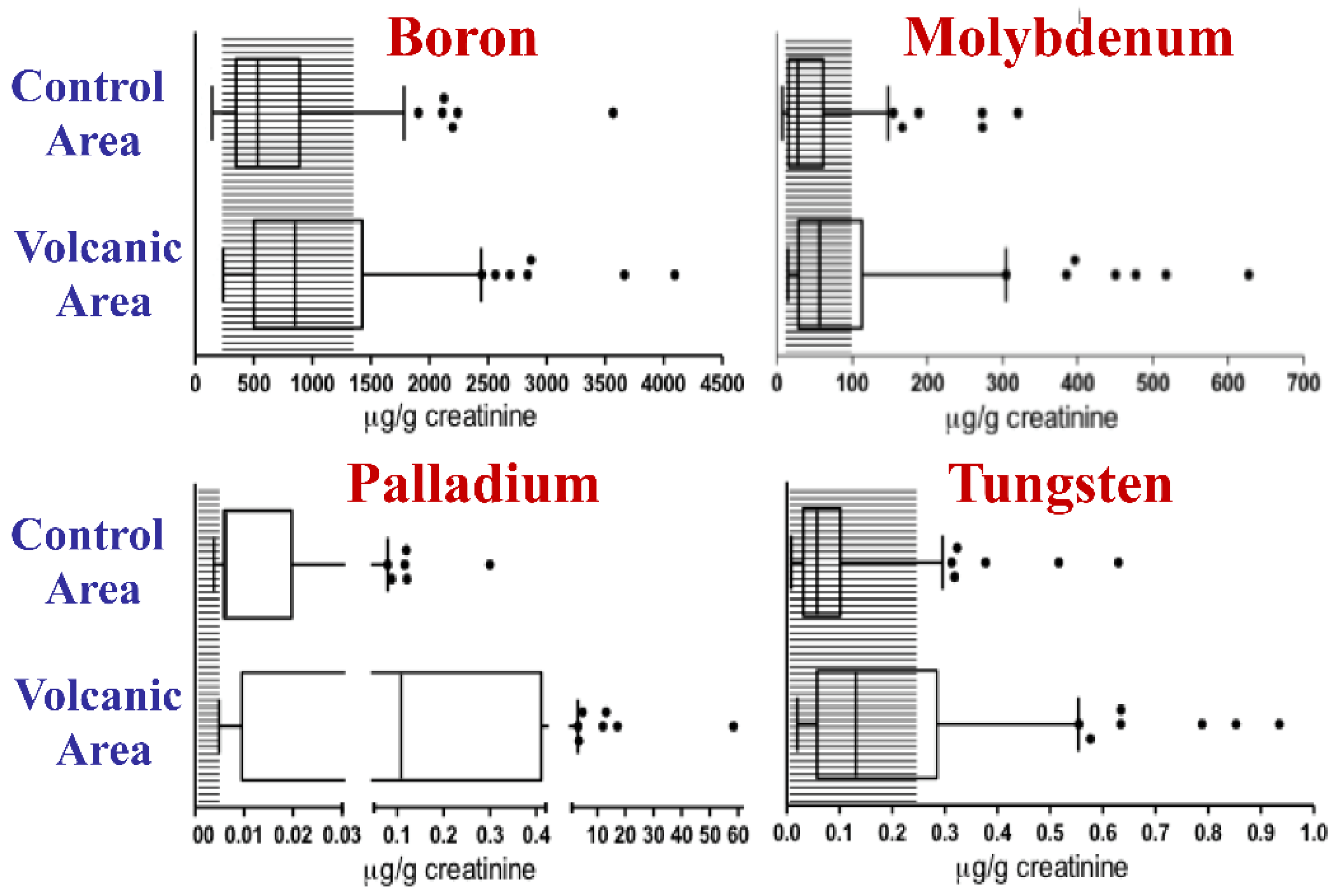
5. Heavy Metals in the Mt. Etna Volcanic Area and Resident Biocontamination
6. Increases in Heavy Metals and Thyroid Cancer: A Cause–Effect Relationship?
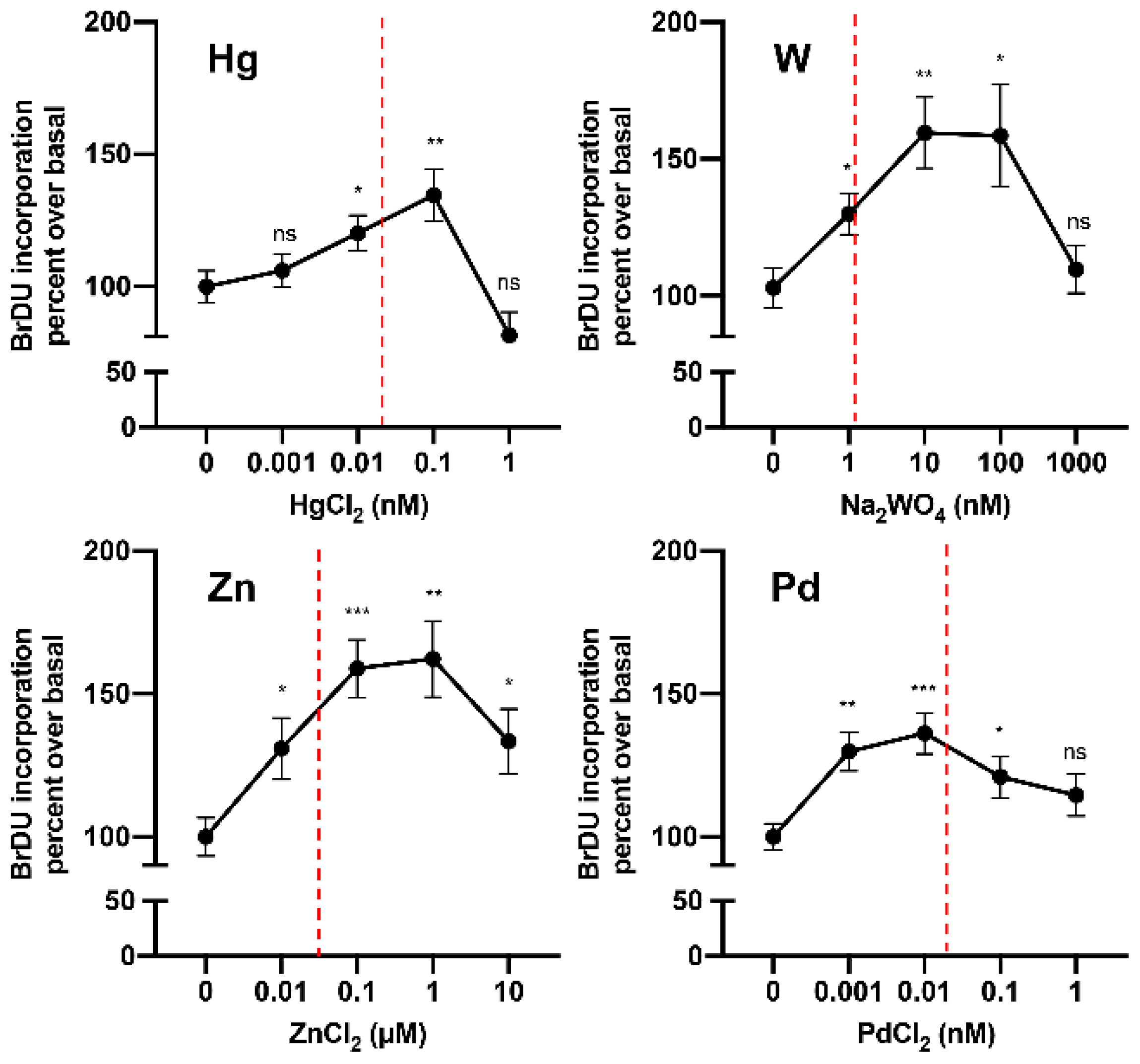
6.1. Hormesis Effect
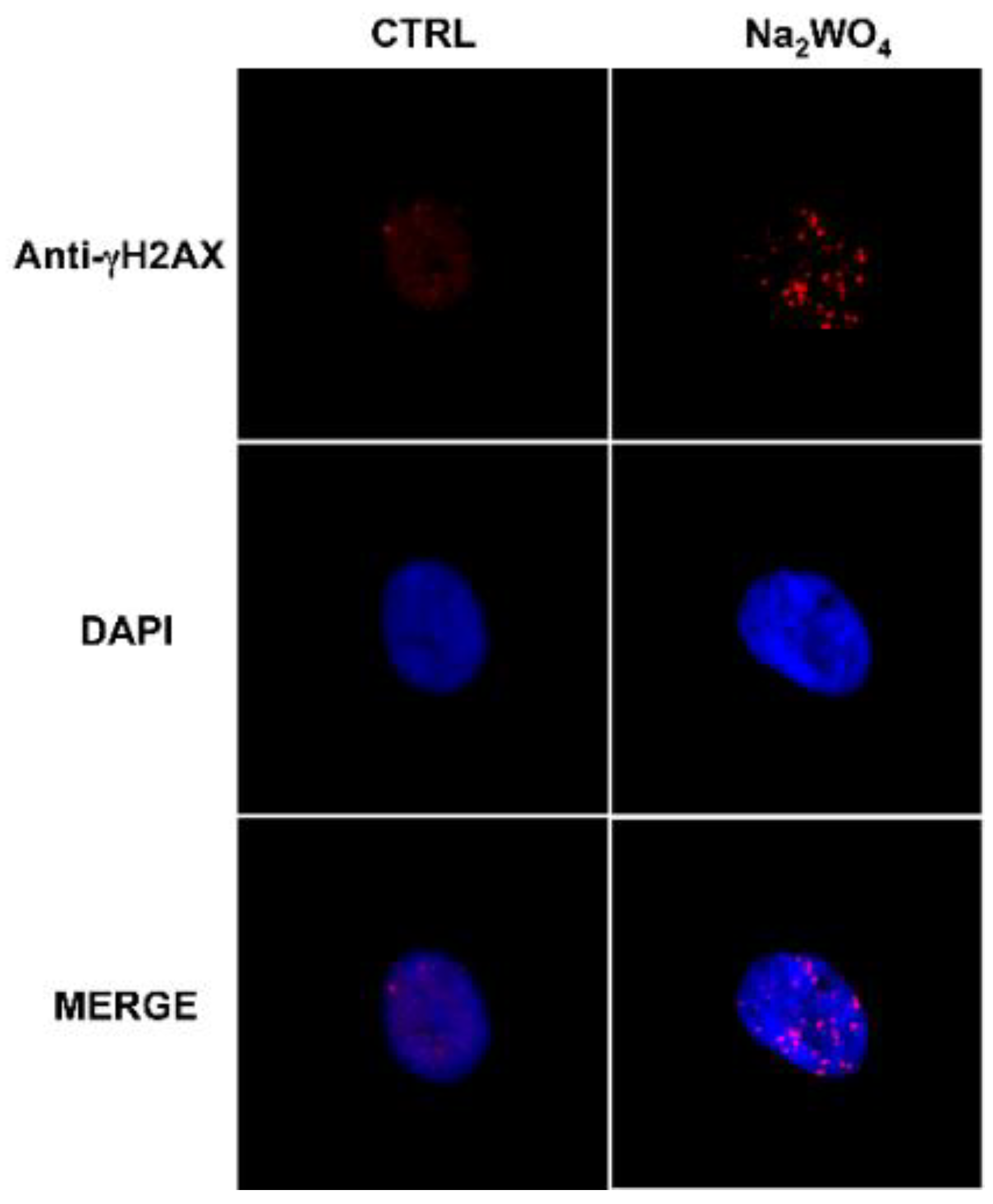
6.2. Is Metal Accumulation in the Thyroid a Possible Mechanism Contributing to the Increase in Thyroid Cancer?
6.3. Metals and Thyroid-Specific Biology
6.4. In Vivo Data in Experimental Animals
7. Concluding Remarks
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ito, Y.; Nikiforov, Y.E.; Schlumberger, M.; Vigneri, R. Increasing incidence of thyroid cancer: Controversies explored. Nat. Rev. Endocrinol. 2013, 9, 178–184. [Google Scholar] [CrossRef]
- Vigneri, R.; Malandrino, P.; Vigneri, P. The changing epidemiology of thyroid cancer: Why is incidence increasing? Curr. Opin. Oncol. 2015, 27, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Colombet, M.; Soerjomataram, I.; Mathers, C.; Parkin, D.M.; Piñeros, M.; Znaor, A.; Bray, F. Estimating the global cancer incidence and mortality in 2018: GLOBOCAN sources and methods. Int. J. Cancer 2019, 144, 1941–1953. [Google Scholar] [CrossRef]
- Kilfoy, B.A.; Zheng, T.; Holford, T.R.; Han, X.; Ward, M.H.; Sjodin, A.; Zhang, Y.; Bai, Y.; Zhu, C.; Guo, G.L.; et al. International patterns and trends in thyroid cancer incidence, 1973–2002. Cancer Causes Control 2009, 20, 525–531. [Google Scholar] [CrossRef]
- Pellegriti, G.; Frasca, F.; Regalbuto, C.; Squatrito, S.; Vigneri, R. Worldwide Increasing Incidence of Thyroid Cancer: Update on Epidemiology and Risk Factors. Available online: https://www.hindawi.com/journals/jce/2013/965212/?viewType=Print&%3BviewClass=Print&%3BB1 (accessed on 10 April 2020).
- Davies, L.; Welch, H.G. Current Thyroid Cancer Trends in the United States. JAMA Otolaryngol. Neck Surg. 2014, 140, 317–322. [Google Scholar] [CrossRef] [PubMed]
- Mao, Y.; Xing, M. Recent incidences and differential trends of thyroid cancer in the USA. Endocr. Relat. Cancer 2016, 23, 313–322. [Google Scholar] [CrossRef] [PubMed]
- Lim, H.; Devesa, S.S.; Sosa, J.A.; Check, D.; Kitahara, C.M. Trends in Thyroid Cancer Incidence and Mortality in the United States, 1974–2013. JAMA 2017, 317, 1338–1348. [Google Scholar] [CrossRef] [PubMed]
- Elisei, R. Molecular Profiles of Papillary Thyroid Tumors Have Been Changing in the Last Decades: How Could We Explain It? J. Clin. Endocrinol. Metab. 2014, 99, 412–414. [Google Scholar] [CrossRef] [PubMed]
- Jung, C.K.; Little, M.P.; Lubin, J.H.; Brenner, A.V.; Wells, S.A.; Sigurdson, A.J.; Nikiforov, Y.E. The Increase in Thyroid Cancer Incidence During the Last Four Decades Is Accompanied by a High Frequency of BRAF Mutations and a Sharp Increase in RAS Mutations. J. Clin. Endocrinol. Metab. 2014, 99, E276–E285. [Google Scholar] [CrossRef] [PubMed]
- Iglesias, M.L.; Schmidt, A.; Ghuzlan, A.A.; Lacroix, L.; De Vathaire, F.; Chevillard, S.; Schlumberger, M. Radiation exposure and thyroid cancer: A review. Arch. Endocrinol. Metab. 2017, 61, 180–187. [Google Scholar] [CrossRef]
- Han, M.A.; Kim, J.H. Diagnostic X-Ray Exposure and Thyroid Cancer Risk: Systematic Review and Meta-Analysis. Thyroid 2017, 28, 220–228. [Google Scholar] [CrossRef]
- Dong, W.; Zhang, H.; Zhang, P.; Li, X.; He, L.; Wang, Z.; Liu, Y. The changing incidence of thyroid carcinoma in Shenyang, China before and after universal salt iodization. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2013, 19, 49. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.-L.; Cheng, S.-P.; Lin, H.-W.; Lai, Y.-L. Risk of thyroid cancer in patients with thyroiditis: A population-based cohort study. Ann. Surg. Oncol. 2014, 21, 843–849. [Google Scholar] [CrossRef] [PubMed]
- Oh, C.-M.; Park, S.; Lee, J.Y.; Won, Y.-J.; Shin, A.; Kong, H.-J.; Choi, K.-S.; Lee, Y.J.; Chung, K.-W.; Jung, K.-W. Increased prevalence of chronic lymphocytic thyroiditis in Korean patients with papillary thyroid cancer. PLoS ONE 2014, 9, e99054. [Google Scholar] [CrossRef] [PubMed]
- Marcello, M.A.; Malandrino, P.; Almeida, J.F.M.; Martins, M.B.; Cunha, L.L.; Bufalo, N.E.; Pellegriti, G.; Ward, L.S. The influence of the environment on the development of thyroid tumors: A new appraisal. Endocr. Relat. Cancer 2014, 21, T235–T254. [Google Scholar] [CrossRef] [PubMed]
- Benedetti, M.; Zona, A.; Beccaloni, E.; Carere, M.; Comba, P. Incidence of Breast, Prostate, Testicular, and Thyroid Cancer in Italian Contaminated Sites with Presence of Substances with Endocrine Disrupting Properties. Int. J. Environ. Res. Public. Health 2017, 14, 355. [Google Scholar] [CrossRef]
- Kilfoy, B.A.; Zhang, Y.; Park, Y.; Holford, T.R.; Schatzkin, A.; Hollenbeck, A.; Ward, M.H. Dietary nitrate and nitrite and the risk of thyroid cancer in the NIH-AARP Diet and Health Study. Int. J. Cancer 2011, 129, 160–172. [Google Scholar] [CrossRef]
- Ward, M.H.; Kilfoy, B.A.; Weyer, P.J.; Anderson, K.E.; Folsom, A.R.; Cerhan, J.R. Nitrate intake and the risk of thyroid cancer and thyroid disease. Epidemiol. Camb. Mass 2010, 21, 389. [Google Scholar] [CrossRef]
- Fiore, M.; Oliveri Conti, G.; Caltabiano, R.; Buffone, A.; Zuccarello, P.; Cormaci, L.; Cannizzaro, M.A.; Ferrante, M. Role of Emerging Environmental Risk Factors in Thyroid Cancer: A Brief Review. Int. J. Environ. Res. Public. Health 2019, 16, 1185. [Google Scholar] [CrossRef]
- Huang, H.; Sjodin, A.; Chen, Y.; Ni, X.; Ma, S.; Yu, H.; Ward, M.H.; Udelsman, R.; Rusiecki, J.; Zhang, Y. Polybrominated Diphenyl Ethers, Polybrominated Biphenyls, and Risk of Papillary Thyroid Cancer: A Nested Case-Control Study. Am. J. Epidemiol. 2020, 189, 120–132. [Google Scholar] [CrossRef]
- Marotta, V.; Malandrino, P.; Russo, M.; Panariello, I.; Ionna, F.; Chiofalo, M.G.; Pezzullo, L. Fathoming the link between anthropogenic chemical contamination and thyroid cancer. Crit. Rev. Oncol. Hematol. 2020, 150, 102950. [Google Scholar] [CrossRef] [PubMed]
- Kung, T.M.; Ng, W.L.; Gibson, J.B. Volcanoes and Carcinoma of the Thyroid: A Possible Association. Arch. Environ. Health Int. J. 1981, 36, 265–267. [Google Scholar] [CrossRef] [PubMed]
- Goodman, M.T.; Yoshizawa, C.N.; Kolonel, L.N. Descriptive epidemiology of thyroid cancer in Hawaii. Cancer 1988, 61, 1272–1281. [Google Scholar] [CrossRef]
- Kolonel, L.N.; Hankin, J.H.; Wilkens, L.R.; Fukunaga, F.H.; Hinds, M.W. An epidemiologic study of thyroid cancer in Hawaii. Cancer Causes Control 1990, 1, 223–234. [Google Scholar] [CrossRef]
- Arnbjörnsson, E.; Arnbjörnsson, A.; Ólafsson, A. Thyroid Cancer Incidence in Relation to Volcanic Activity. Arch. Environ. Health Int. J. 1986, 41, 36–40. [Google Scholar] [CrossRef]
- Hrafnkelsson, J.; Tulinius, H.; Jonasson, J.G.; Ólafsdottir, G.; Sigvaldason, H. Papillary Thyroid Carcinoma in Iceland: A study of the Occurrence in families and the coexistence of other primary tumours. Acta Oncol. 1989, 28, 785–788. [Google Scholar] [CrossRef]
- Paksoy, N.; Montaville, B.; McCarthy, S. Cancer Occurrence in Vanuatu in the South Pacific, 1980–1986. Asia Pac. J. Public Health 1989, 3, 231–236. [Google Scholar] [CrossRef]
- Curado, M.P.; Edwards, B.; Shin, H.R.; Storm, H.; Ferlay, J.; Heanue, M. Cancer Incidence in Five Continents Vol. IX. IARC Scientific Publication No. 160. 2007; IARC Press: Lyon, France, 2007. [Google Scholar]
- Truong, T.; Rougier, Y.; Dubourdieu, D.; Guihenneuc-Jouyaux, C.; Orsi, L.; Hémon, D.; Guénel, P. Time trends and geographic variations for thyroid cancer in New Caledonia, a very high incidence area (1985–1999). Eur. J. Cancer Prev. 2007, 16, 62–70. [Google Scholar] [CrossRef][Green Version]
- Duntas, L.H.; Doumas, C. The ‘rings of fire’ and thyroid cancer. Hormones 2009, 8, 249–253. [Google Scholar] [CrossRef]
- Kristbjornsdottir, A.; Rafnsson, V. Cancer incidence among population utilizing geothermal hot water: A census-based cohort study. Int. J. Cancer 2013, 133, 2944–2952. [Google Scholar] [CrossRef]
- Goyal, N.; Camacho, F.; Mangano, J.; Goldenberg, D. Evaluating for a geospatial relationship between radon levels and thyroid cancer in Pennsylvania. Laryngoscope 2015, 125, E45–E49. [Google Scholar] [CrossRef] [PubMed]
- Oakland, C.; Meliker, J.R. County-Level Radon and Incidence of Female Thyroid Cancer in Iowa, New Jersey, and Wisconsin, USA. Toxics 2018, 6, 17. [Google Scholar] [CrossRef] [PubMed]
- Pellegriti, G.; De Vathaire, F.; Scollo, C.; Attard, M.; Giordano, C.; Arena, S.; Dardanoni, G.; Frasca, F.; Malandrino, P.; Vermiglio, F.; et al. Papillary Thyroid Cancer Incidence in the Volcanic Area of Sicily. JNCI J. Natl. Cancer Inst. 2009, 101, 1575–1583. [Google Scholar] [CrossRef] [PubMed]
- Russo, M.; Malandrino, P.; Moleti, M.; D’Angelo, A.; Tavarelli, M.; Sapuppo, G.; Gianì, F.; Richiusa, P.; Squatrito, S.; Vigneri, R.; et al. Thyroid Cancer in the Pediatric Age in Sicily: Influence of the Volcanic Environment. Anticancer Res. 2017, 37, 1515–1522. [Google Scholar] [PubMed]
- Dahal, B.M.; Fuerhacker, M.; Mentler, A.; Karki, K.B.; Shrestha, R.R.; Blum, W.E.H. Arsenic contamination of soils and agricultural plants through irrigation water in Nepal. Environ. Pollut. 2008, 155, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Abiye, T.A.; Sulaiman, H.; Hailu, A. Metal concentration in vegetables grown in the hydrothermally affected area in Ethiopia. J. Geogr. Geol. 2011, 3, 86. [Google Scholar]
- World Health Organization; International Atomic Energy Agency; Food and Agriculture Organization of the United Nations. Trace Elements in Human Nutrition and Health. 1996. Available online: https://apps.who.int/iris/handle/10665/37931 (accessed on 24 April 2020).
- Outten, C.E.; O’Halloran, T.V. Femtomolar Sensitivity of Metalloregulatory Proteins Controlling Zinc Homeostasis. Science 2001, 292, 2488–2492. [Google Scholar] [CrossRef]
- Finney, L.A.; O’Halloran, T.V. Transition Metal Speciation in the Cell: Insights from the Chemistry of Metal Ion Receptors. Science 2003, 300, 931–936. [Google Scholar] [CrossRef]
- Trusso Sfrazzetto, G.; Satriano, C.; Tomaselli, G.A.; Rizzarelli, E. Synthetic fluorescent probes to map metallostasis and intracellular fate of zinc and copper. Coord. Chem. Rev. 2016, 311, 125–167. [Google Scholar] [CrossRef]
- Dittmer, P.J.; Miranda, J.G.; Gorski, J.A.; Palmer, A.E. Genetically Encoded Sensors to Elucidate Spatial Distribution of Cellular Zinc. J. Biol. Chem. 2009, 284, 16289–16297. [Google Scholar] [CrossRef]
- Changela, A.; Chen, K.; Xue, Y.; Holschen, J.; Outten, C.E.; O’Halloran, T.V.; Mondragón, A. Molecular Basis of Metal-Ion Selectivity and Zeptomolar Sensitivity by CueR. Science 2003, 301, 1383–1387. [Google Scholar] [CrossRef] [PubMed]
- Milardi, D.; Rizzarelli, E. Neurodegeneration: Metallostasis and Proteostasis; Royal Society of Chemistry: London, UK, 2011; ISBN 978-1-84973-301-4. [Google Scholar]
- Grasso, G.; Giuffrida, M.L.; Rizzarelli, E. Metallostasis and amyloid β-degrading enzymes. Metallomics 2012, 4, 937–949. [Google Scholar] [CrossRef] [PubMed]
- Nyquist, M.D.; Prasad, B.; Mostaghel, E.A. Harnessing Solute Carrier Transporters for Precision Oncology. Molecules 2017, 22, 539. [Google Scholar] [CrossRef] [PubMed]
- Si, M.; Lang, J. The roles of metallothioneins in carcinogenesis. J. Hematol. Oncol. 2018, 11, 107. [Google Scholar] [CrossRef] [PubMed]
- Calvo, J.; Jung, H.; Meloni, G. Copper metallothioneins. IUBMB Life 2017, 69, 236–245. [Google Scholar] [CrossRef] [PubMed]
- Rutherford, J.C.; Bird, A.J. Metal-Responsive Transcription Factors That Regulate Iron, Zinc, and Copper Homeostasis in Eukaryotic Cells. Eukaryot. Cell 2004, 3, 1–13. [Google Scholar] [CrossRef]
- Jung, J.; Lee, S.J. Biochemical and Biodiversity Insights into Heavy Metal Ion-Responsive Transcription Regulators for Synthetic Biological Heavy Metal Sensors. J. Microbiol. Biotechnol. 2019, 29, 1522–1542. [Google Scholar] [CrossRef]
- Sies, H. Glutathione and its role in cellular functions. Free Radic. Biol. Med. 1999, 27, 916–921. [Google Scholar] [CrossRef]
- Fukada, T.; Yamasaki, S.; Nishida, K.; Murakami, M.; Hirano, T. Zinc homeostasis and signaling in health and diseases. JBIC J. Biol. Inorg. Chem. 2011, 16, 1123–1134. [Google Scholar] [CrossRef]
- Kambe, T.; Yamaguchi-Iwai, Y.; Sasaki, R.; Nagao, M. Overview of mammalian zinc transporters. Cell. Mol. Life Sci. CMLS 2004, 61, 49–68. [Google Scholar] [CrossRef]
- La Mendola, D.; Giacomelli, C.; Rizzarelli, E. Intracellular Bioinorganic Chemistry and Cross Talk Among Different–Omics. Available online: https://www.ingentaconnect.com/content/ben/ctmc/2016/00000016/00000027/art00008 (accessed on 12 April 2020).
- Seve, M.; Chimienti, F.; Devergnas, S.; Favier, A. In silico identification and expression of SLC30 family genes: An expressed sequence tag data mining strategy for the characterization of zinc transporters’ tissue expression. BMC Genom. 2004, 5, 32. [Google Scholar] [CrossRef] [PubMed]
- Dong, G.; Chen, H.; Qi, M.; Dou, Y.; Wang, Q. Balance between metallothionein and metal response element binding transcription factor 1 is mediated by zinc ions (Review). Mol. Med. Rep. 2015, 11, 1582–1586. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, M.C.; Palmer, A.E. Native and engineered sensors for Ca2+ and Zn2+: Lessons from calmodulin and MTF1. Essays Biochem. 2017, 61, 237–243. [Google Scholar]
- Nose, Y.; Kim, B.-E.; Thiele, D.J. Ctr1 drives intestinal copper absorption and is essential for growth, iron metabolism, and neonatal cardiac function. Cell Metab. 2006, 4, 235–244. [Google Scholar] [CrossRef] [PubMed]
- Festa, R.A.; Thiele, D.J. Copper: An essential metal in biology. Curr. Biol. 2011, 21, R877–R883. [Google Scholar] [CrossRef] [PubMed]
- Harrison, M.D.; Jones, C.E.; Dameron, C.T. Copper chaperones: Function, structure and copper-binding properties. JBIC J. Biol. Inorg. Chem. 1999, 4, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Palumaa, P. Copper chaperones. The concept of conformational control in the metabolism of copper. FEBS Lett. 2013, 587, 1902–1910. [Google Scholar] [CrossRef]
- Devi, S.R.B.; Dhivya M, A.; Sulochana, K.N. Copper transporters and chaperones: Their function on angiogenesis and cellular signalling. J. Biosci. 2016, 41, 487–496. [Google Scholar] [CrossRef]
- Yuan, S.; Chen, S.; Xi, Z.; Liu, Y. Copper-finger protein of Sp1: The molecular basis of copper sensing. Metallomics 2017, 9, 1169–1175. [Google Scholar] [CrossRef]
- Hughes Michael, F. Biomarkers of Exposure: A Case Study with Inorganic Arsenic. Environ. Health Perspect. 2006, 114, 1790–1796. [Google Scholar] [CrossRef]
- Sun, H.; Xiang, P.; Luo, J.; Hong, H.; Lin, H.; Li, H.-B.; Ma, L.Q. Mechanisms of arsenic disruption on gonadal, adrenal and thyroid endocrine systems in humans: A review. Environ. Int. 2016, 95, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, S.; Konishi, Y.; Matsuda, T.; Murai, T.; Shibata, M.-A.; Matsui-Yuasa, I.; Otani, S.; Kuroda, K.; Endo, G.; Fukushima, S. Cancer Induction by an Organic Arsenic Compound, Dimethylarsinic Acid (Cacodylic Acid), in F344/DuCrj Rats after Pretreatment with Five Carcinogens. Cancer Res. 1995, 55, 1271–1276. [Google Scholar] [PubMed]
- Fröhlich, E.; Czarnocka, B.; Brossart, P.; Wahl, R. Antitumor Effects of Arsenic Trioxide in Transformed Human Thyroid Cells. Thyroid 2008, 18, 1183–1193. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Ge, X.; Zheng, J.; Li, D.; Liu, X.; Wang, L.; Jiang, C.; Shi, Z.; Qin, L.; Liu, J.; et al. Role and mechanism of miR-222 in arsenic-transformed cells for inducing tumor growth. Oncotarget 2016, 7, 17805–17814. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Jazdzewski, K.; Li, W.; Liyanarachchi, S.; Nagy, R.; Volinia, S.; Calin, G.A.; Liu, C.; Franssila, K.; Suster, S.; et al. The role of microRNA genes in papillary thyroid carcinoma. Proc. Natl. Acad. Sci. USA 2005, 102, 19075–19080. [Google Scholar] [CrossRef]
- Zidane, M.; Ren, Y.; Xhaard, C.; Leufroy, A.; Côte, S.; Dewailly, E.; Noël, L.; Guérin, T.; Bouisset, P.; Bernagout, S.; et al. Non-Essential Trace Elements Dietary Exposure in French Polynesia: Intake Assessment, Nail Bio Monitoring and Thyroid Cancer Risk. Asian Pac. J. Cancer Prev. APJCP 2019, 20, 355–367. [Google Scholar] [CrossRef]
- Iavicoli, I.; Fontana, L.; Bergamaschi, A. The Effects of Metals as Endocrine Disruptors. J. Toxicol. Environ. Health Part B 2009, 12, 206–223. [Google Scholar] [CrossRef]
- Hartwig, A.; Asmuss, M.; Blessing, H.; Hoffmann, S.; Jahnke, G.; Khandelwal, S.; Pelzer, A.; Bürkle, A. Interference by toxic metal ions with zinc-dependent proteins involved in maintaining genomic stability. Food Chem. Toxicol. 2002, 40, 1179–1184. [Google Scholar] [CrossRef]
- Buha, A.; Matovic, V.; Antonijevic, B.; Bulat, Z.; Curcic, M.; Renieri, E.A.; Tsatsakis, A.M.; Schweitzer, A.; Wallace, D. Overview of Cadmium Thyroid Disrupting Effects and Mechanisms. Int. J. Mol. Sci. 2018, 19, 1501. [Google Scholar] [CrossRef]
- Beyersmann, D.; Hartwig, A. Carcinogenic metal compounds: Recent insight into molecular and cellular mechanisms. Arch. Toxicol. 2008, 82, 493. [Google Scholar] [CrossRef]
- Huff, J.; Lunn, R.M.; Waalkes, M.P.; Tomatis, L.; Infante, P.F. Cadmium-induced Cancers in Animals and in Humans. Int. J. Occup. Environ. Health 2007, 13, 202–212. [Google Scholar] [CrossRef] [PubMed]
- Prozialeck, W.C.; Lamar, P.C.; Lynch, S.M. Cadmium alters the localization of N-cadherin, E-cadherin, and β-catenin in the proximal tubule epithelium. Toxicol. Appl. Pharmacol. 2003, 189, 180–195. [Google Scholar] [CrossRef]
- Zhu, P.; Liao, L.-Y.; Zhao, T.-T.; Mo, X.-M.; Chen, G.G.; Liu, Z.-M. GPER/ERK&AKT/NF-κB pathway is involved in cadmium-induced proliferation, invasion and migration of GPER-positive thyroid cancer cells. Mol. Cell. Endocrinol. 2017, 442, 68–80. [Google Scholar] [CrossRef] [PubMed]
- Deheyn, D.D.; Gendreau, P.; Baldwin, R.J.; Latz, M.I. Evidence for enhanced bioavailability of trace elements in the marine ecosystem of Deception Island, a volcano in Antarctica. Mar. Environ. Res. 2005, 60, 1–33. [Google Scholar] [CrossRef]
- Fiorentino, C.E.; Paoloni, J.D.; Sequeira, M.E.; Arosteguy, P. The presence of vanadium in groundwater of southeastern extreme the pampean region Argentina: Relationship with other chemical elements. J. Contam. Hydrol. 2007, 93, 122–129. [Google Scholar] [CrossRef]
- Malandrino, P.; Russo, M.; Ronchi, A.; Minoia, C.; Cataldo, D.; Regalbuto, C.; Giordano, C.; Attard, M.; Squatrito, S.; Trimarchi, F.; et al. Increased thyroid cancer incidence in a basaltic volcanic area is associated with non-anthropogenic pollution and biocontamination. Endocrine 2016, 53, 471–479. [Google Scholar] [CrossRef]
- Wolterbeek, B. Biomonitoring of trace element air pollution: Principles, possibilities and perspectives. Environ. Pollut. 2002, 120, 11–21. [Google Scholar] [CrossRef]
- Augusto, S.; Máguas, C.; Branquinho, C. Guidelines for biomonitoring persistent organic pollutants (POPs), using lichens and aquatic mosses—A review. Environ. Pollut. 2013, 180, 330–338. [Google Scholar] [CrossRef]
- World Health Organization. Guidelines for Drinking-Water Quality, 4th ed.; WHO Chronicle: Geneva, Switzerland, 2011; Volume 38. [Google Scholar]
- Varrica, D.; Tamburo, E.; Dongarrà, G.; Sposito, F. Trace elements in scalp hair of children chronically exposed to volcanic activity (Mt. Etna, Italy). Sci. Total Environ. 2014, 470–471, 117–126. [Google Scholar] [CrossRef]
- Rodrigues, A.S.; Arruda, M.S.C.; Garcia, P.V. Evidence of DNA damage in humans inhabiting a volcanically active environment: A useful tool for biomonitoring. Environ. Int. 2012, 49, 51–56. [Google Scholar] [CrossRef]
- Russo, M.; Malandrino, P.; Addario, W.P.; Dardanoni, G.; Vigneri, P.; Pellegriti, G.; Squatrito, S.; Vigneri, R. Several Site-specific Cancers are Increased in the Volcanic Area in Sicily. Anticancer Res. 2015, 35, 3995–4001. [Google Scholar] [PubMed]
- Calabrese, E.J. Hormesis: A revolution in toxicology, risk assessment and medicine. EMBO Rep. 2004, 5, S37–S40. [Google Scholar] [CrossRef]
- Hao, C.; Hao, W.; Wei, X.; Xing, L.; Jiang, J.; Shang, L. The role of MAPK in the biphasic dose-response phenomenon induced by cadmium and mercury in HEK293 cells. Toxicol. Vitr. 2009, 23, 660–666. [Google Scholar] [CrossRef]
- Llabjani, V.; Hoti, V.; Pouran, H.M.; Martin, F.L.; Zhang, H. Bimodal responses of cells to trace elements: Insights into their mechanism of action using a biospectroscopy approach. Chemosphere 2014, 112, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Mantha, M.; Jumarie, C. Cadmium-induced hormetic effect in differentiated Caco-2 cells: ERK and p38 activation without cell proliferation stimulation. J. Cell. Physiol. 2010, 224, 250–261. [Google Scholar] [CrossRef]
- Damelin, L.H.; Vokes, S.; Whitcutt, J.M.; Damelin, S.B.; Alexander, J.J. Hormesis: A stress response in cells exposed to low levels of heavy metals. Hum. Exp. Toxicol. 2000, 19, 420–430. [Google Scholar] [CrossRef] [PubMed]
- Nascarella, M.A.; Stoffolano, J.G.; Stanek, E.J.; Kostecki, P.T.; Calabrese, E.J. Hormesis and stage specific toxicity induced by cadmium in an insect model, the queen blowfly, Phormia regina Meig. Environ. Pollut. 2003, 124, 257–262. [Google Scholar] [CrossRef]
- Renieri, E.A.; Sfakianakis, D.G.; Alegakis, A.A.; Safenkova, I.V.; Buha, A.; Matović, V.; Tzardi, M.; Dzantiev, B.B.; Divanach, P.; Kentouri, M.; et al. Nonlinear responses to waterborne cadmium exposure in zebrafish. An in vivo study. Environ. Res. 2017, 157, 173–181. [Google Scholar] [CrossRef]
- Zhang, W.Z.; Sun, N.N.; Ma, S.Q.; Zhao, Z.C.; Cao, Y.; Zhang, C. Hormetic Effects of Yttrium on Male Sprague-Dawley Rats. Biomed. Environ. Sci. BES 2018, 31, 777–780. [Google Scholar] [CrossRef]
- Maggisano, V.; Bulotta, S.; Celano, M.; Maiuolo, J.; Lepore, S.M.; Abballe, L.; Iannone, M.; Russo, D. Low Doses of Methylmercury Induce the Proliferation of Thyroid Cells In Vitro Through Modulation of ERK Pathway. Int. J. Mol. Sci. 2020, 21, 1556. [Google Scholar] [CrossRef]
- Schmidt, C.M.; Cheng, C.N.; Marino, A.; Konsoula, R.; Barile, F.A. Hormesis effect of trace metals on cultured normal and immortal human mammary cells. Toxicol. Ind. Health 2004, 20, 57–68. [Google Scholar] [CrossRef] [PubMed]
- Tyne, W.; Little, S.; Spurgeon, D.J.; Svendsen, C. Hormesis depends upon the life-stage and duration of exposure: Examples for a pesticide and a nanomaterial. Ecotoxicol. Environ. Saf. 2015, 120, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Hadacek, F.; Bachmann, G.; Engelmeier, D.; Chobot, V. Hormesis and a chemical raison d’ětre for secondary plant metabolites. Dose Response 2011, 9, 79–116. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.; He, X.-Q.; Peng, L.; Li, A.-P.; Wang, X.-R.; Zhou, J.-W.; Liu, Q.-Z. The Role of Oxidative Stress in Hormesis Induced by Sodium Arsenite in Human Embryo Lung Fibroblast (HELF) Cellular Proliferation Model. J. Toxicol. Environ. Health A 2007, 70, 976–983. [Google Scholar] [CrossRef]
- Tan, Q.; Liu, Z.; Li, H.; Liu, Y.; Xia, Z.; Xiao, Y.; Usman, M.; Du, Y.; Bi, H.; Wei, L. Hormesis of mercuric chloride-human serum albumin adduct on N9 microglial cells via the ERK/MAPKs and JAK/STAT3 signaling pathways. Toxicology 2018, 408, 62–69. [Google Scholar] [CrossRef]
- Singh, K.B.; Maret, W. The interactions of metal cations and oxyanions with protein tyrosine phosphatase 1B. BioMetals 2017, 30, 517–527. [Google Scholar] [CrossRef]
- Gianì, F.; Pandini, G.; Scalisi, N.M.; Vigneri, P.; Fazzari, C.; Malandrino, P.; Russo, M.; Masucci, R.; Belfiore, A.; Pellegriti, G.; et al. Effect of low-dose tungsten on human thyroid stem/precursor cells and their progeny. Endocr. Relat. Cancer 2019, 26, 713–725. [Google Scholar] [CrossRef]
- Jiang, G.; Duan, W.; Xu, L.; Song, S.; Zhu, C.; Wu, L. Biphasic effect of cadmium on cell proliferation in human embryo lung fibroblast cells and its molecular mechanism. Toxicol. Vitr. 2009, 23, 973–978. [Google Scholar] [CrossRef]
- Tsui, M.T.K.; Wang, W.-X. Influences of maternal exposure on the tolerance and physiological performance of Daphnia magna under mercury stress. Environ. Toxicol. Chem. 2005, 24, 1228–1234. [Google Scholar] [CrossRef]
- Carvalho, M.E.A.; Castro, P.R.C.; Azevedo, R.A. Hormesis in plants under Cd exposure: From toxic to beneficial element? J. Hazard. Mater. 2020, 384, 121434. [Google Scholar] [CrossRef]
- Malandrino, P.; Russo, M.; Ronchi, A.; Moretti, F.; Gianì, F.; Vigneri, P.; Masucci, R.; Pellegriti, G.; Belfiore, A.; Vigneri, R. Concentration of Metals and Trace Elements in the Normal Human and Rat Thyroid: Comparison with Muscle and Adipose Tissue and Volcanic Versus Control Areas. Thyroid 2019, 30, 290–299. [Google Scholar] [CrossRef]
- Kopp, P. Thyroid hormone synthesis. In Werner and Ingbar’s the Thyroid: A Fundamental and Clinical Text, 10th ed.; Cooper, D., Ed.; Lippincott Williams Wilkins: Philadelphia, PA, USA, 2012; pp. 48–74. [Google Scholar]
- Maier, J.; Van Steeg, H.; Van Oostrom, C.; Karger, S.; Paschke, R.; Krohn, K. Deoxyribonucleic Acid Damage and Spontaneous Mutagenesis in the Thyroid Gland of Rats and Mice. Endocrinology 2006, 147, 3391–3397. [Google Scholar] [CrossRef]
- Kawada, J.; Shirakawa, Y.; Yoshimura, Y.; Nishida, M. Thyroid xanthine oxidase and its role in thyroid iodine metabolism in the rat: Difference between effects of allopurinol and tungstate. J. Endocrinol. 1982, 95, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Brady, D.C.; Crowe, M.S.; Turski, M.L.; Hobbs, G.A.; Yao, X.; Chaikuad, A.; Knapp, S.; Xiao, K.; Campbell, S.L.; Thiele, D.J. Copper is required for oncogenic BRAF signalling and tumorigenesis. Nature 2014, 509, 492–496. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Casio, M.; Range, D.E.; Sosa, J.A.; Counter, C.M. Copper chelation as targeted therapy in a mouse model of oncogenic BRAF-driven papillary thyroid cancer. Clin. Cancer Res. 2018, 24, 4271–4281. [Google Scholar] [CrossRef] [PubMed]
- Assem, F.L.; Holmes, P.; Levy, L.S. The mutagenicity and carcinogenicity of inorganic manganese compounds: A synthesis of the evidence. J. Toxicol. Environ. Health Part B 2011, 14, 537–570. [Google Scholar] [CrossRef]
- Sun, H.-J.; Li, S.-W.; Li, C.; Wang, W.-Q.; Li, H.-B.; Ma, L.Q. Thyrotoxicity of arsenate and arsenite on juvenile mice at organism, subcellular, and gene levels under low exposure. Chemosphere 2017, 186, 580–587. [Google Scholar] [CrossRef]
- Velický, J.; TItlbach, M.; Dušková, J.; Vobecký, M.; Štrbák, V.; Raška, I. Potassium bromide and the thyroid gland of the rat: Morphology and immunohistochemistry, RIA and INAA analysis. Ann. Anat. Anat. Anz. 1997, 179, 421–431. [Google Scholar] [CrossRef]
- Velický, J.; Titlbach, M.; Lojda, Z.; Dušková, J.; Vobecký, M.; Štrbák, V.; Raška, I. Long-term action of potassium bromide on the rat thyroid gland. Acta Histochem. 1998, 100, 11–23. [Google Scholar] [CrossRef]
- DeAngelo, A.B.; George, M.H.; Kilburn, S.R.; Moore, T.M.; Wolf, D.C. Carcinogenicity of potassium bromate administered in the drinking water to male B6C3F1 mice and F344/N rats. Toxicol. Pathol. 1998, 26, 587–594. [Google Scholar] [CrossRef]
- Kanno, J.; Matsuoka, C.; Furuta, K.; Onodera, H.; Miyajima, H.; Maekawa, A.; Hayashi, Y. Tumor promoting effect of goitrogens on the rat thyroid. Toxicol. Pathol. 1990, 18, 239–246. [Google Scholar] [CrossRef] [PubMed]
- Luca, E.; Fici, L.; Ronchi, A.; Marandino, F.; Rossi, E.D.; Caristo, M.E.; Malandrino, P.; Russo, M.; Pontecorvi, A.; Vigneri, R. Intake of Boron, Cadmium, and Molybdenum enhances rat thyroid cell transformation. J. Exp. Clin. Cancer Res. 2017, 36, 73. [Google Scholar] [CrossRef] [PubMed]
- Vareda, J.P.; Valente, A.J.; Durães, L. Assessment of heavy metal pollution from anthropogenic activities and remediation strategies: A review. J. Environ. Manag. 2019, 246, 101–118. [Google Scholar] [CrossRef] [PubMed]
| Environment | Inhabitants (millions) | Thyroid Cancer Incidence (ASRw) | Papillary/Follicular Ratio | |
|---|---|---|---|---|
| F | M | |||
| Volcanic area (Catania province) | 1116 | 31.7 | 6.4 | 25.9 |
| Control area (all Sicily without Catania) | 3853 | 14.1 | 3.0 | 9.8 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Malandrino, P.; Russo, M.; Gianì, F.; Pellegriti, G.; Vigneri, P.; Belfiore, A.; Rizzarelli, E.; Vigneri, R. Increased Thyroid Cancer Incidence in Volcanic Areas: A Role of Increased Heavy Metals in the Environment? Int. J. Mol. Sci. 2020, 21, 3425. https://doi.org/10.3390/ijms21103425
Malandrino P, Russo M, Gianì F, Pellegriti G, Vigneri P, Belfiore A, Rizzarelli E, Vigneri R. Increased Thyroid Cancer Incidence in Volcanic Areas: A Role of Increased Heavy Metals in the Environment? International Journal of Molecular Sciences. 2020; 21(10):3425. https://doi.org/10.3390/ijms21103425
Chicago/Turabian StyleMalandrino, Pasqualino, Marco Russo, Fiorenza Gianì, Gabriella Pellegriti, Paolo Vigneri, Antonino Belfiore, Enrico Rizzarelli, and Riccardo Vigneri. 2020. "Increased Thyroid Cancer Incidence in Volcanic Areas: A Role of Increased Heavy Metals in the Environment?" International Journal of Molecular Sciences 21, no. 10: 3425. https://doi.org/10.3390/ijms21103425
APA StyleMalandrino, P., Russo, M., Gianì, F., Pellegriti, G., Vigneri, P., Belfiore, A., Rizzarelli, E., & Vigneri, R. (2020). Increased Thyroid Cancer Incidence in Volcanic Areas: A Role of Increased Heavy Metals in the Environment? International Journal of Molecular Sciences, 21(10), 3425. https://doi.org/10.3390/ijms21103425








