Ectopic Expression of Grapevine Gene VaRGA1 in Arabidopsis Improves Resistance to Downy Mildew and Pseudomonas syringae pv. tomato DC3000 But Increases Susceptibility to Botrytis cinerea
Abstract
1. Introduction
2. Results
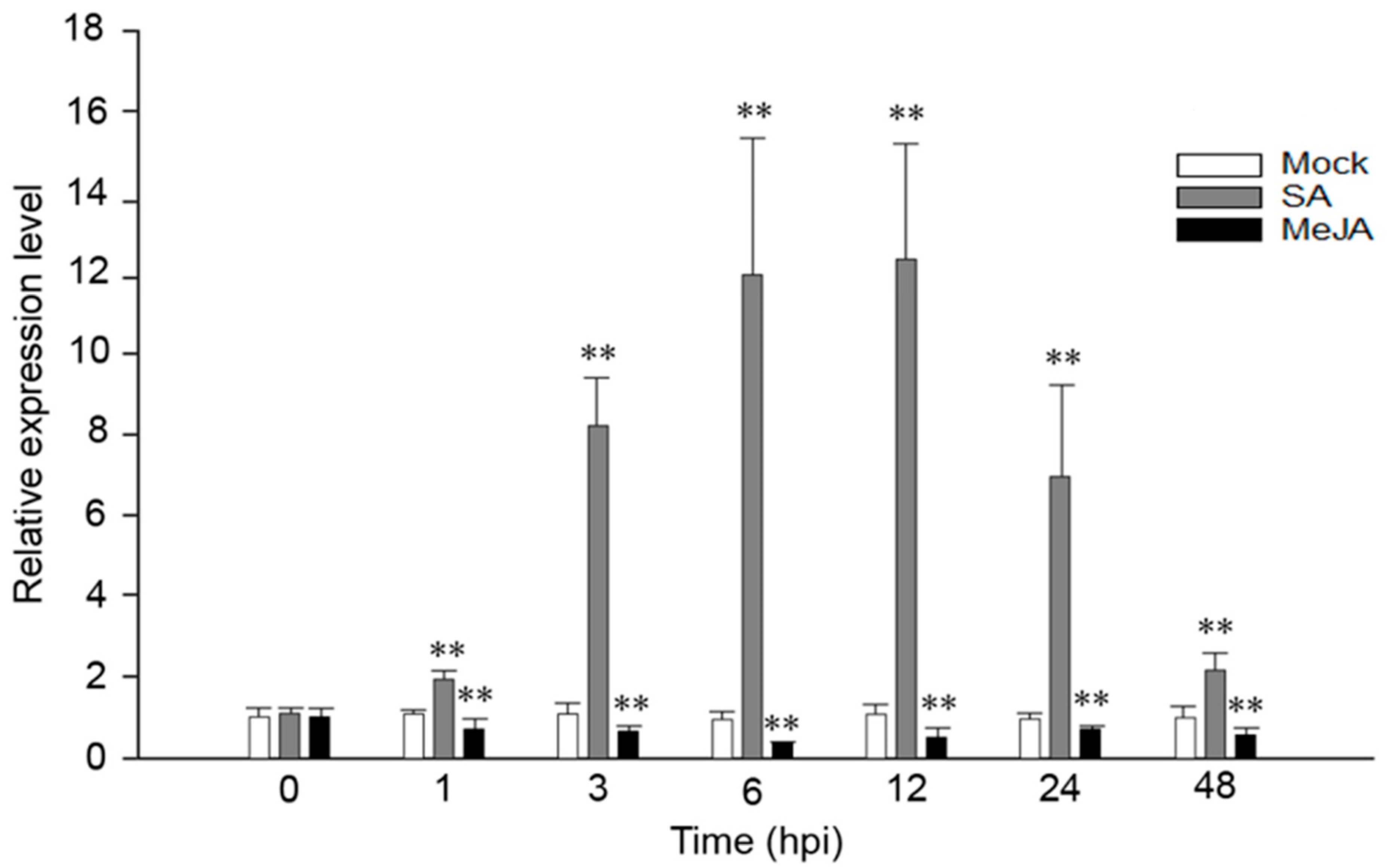
2.1. VaRGA1 Expression Is Affected by SA and JA in Grape
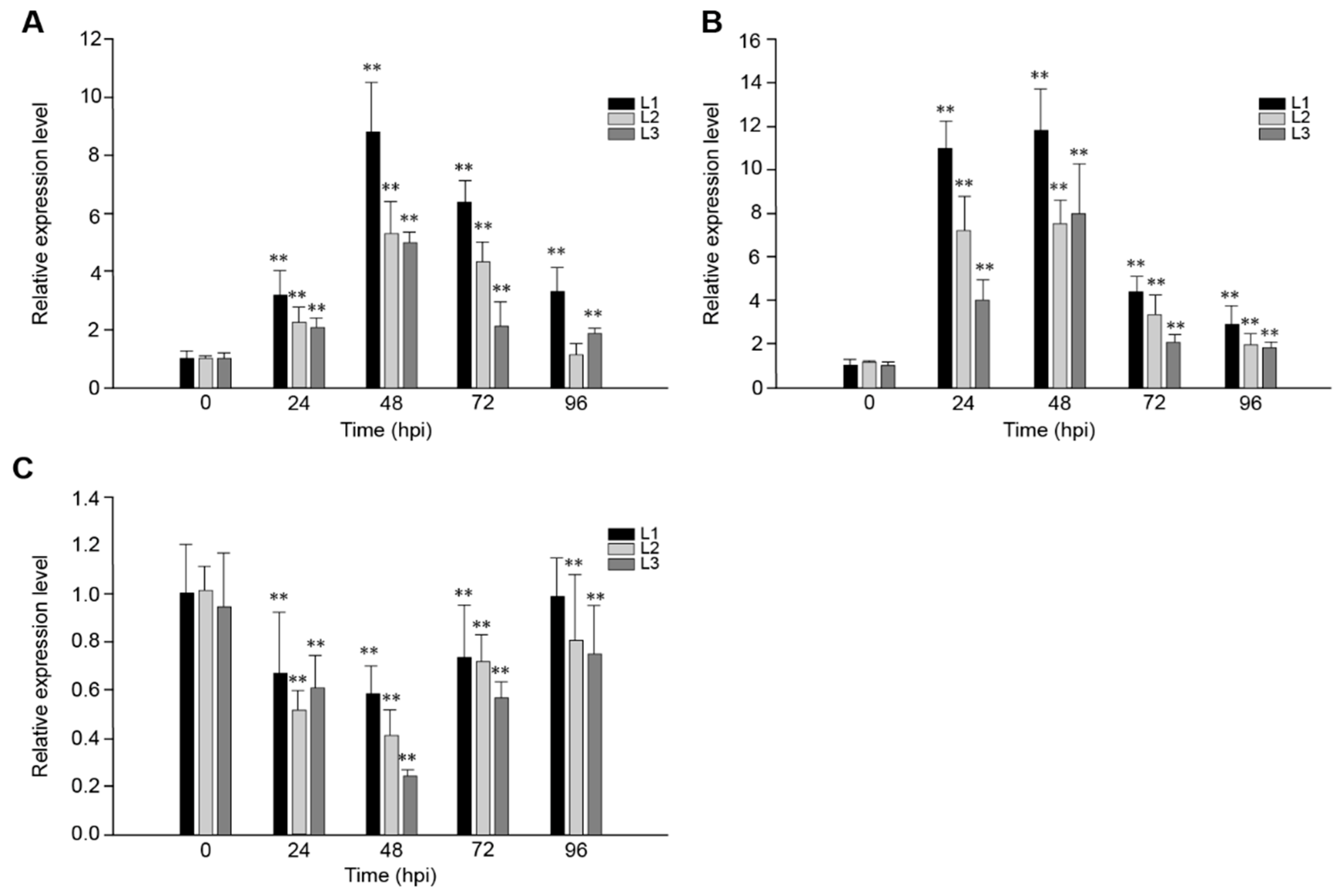
2.2. The Response of VaRGA1 in Transgenic Arabidopsis Lines to Different Pathogens
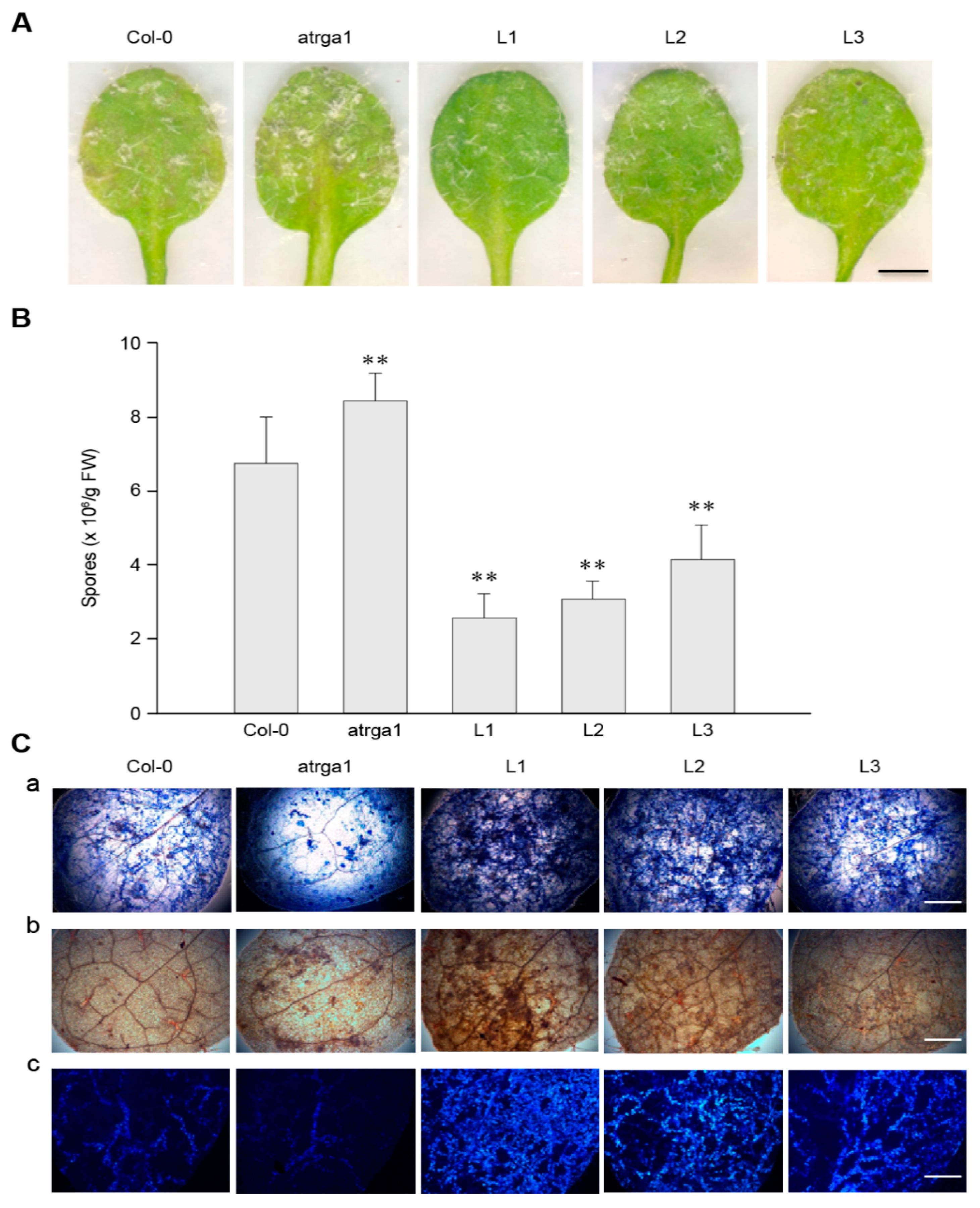
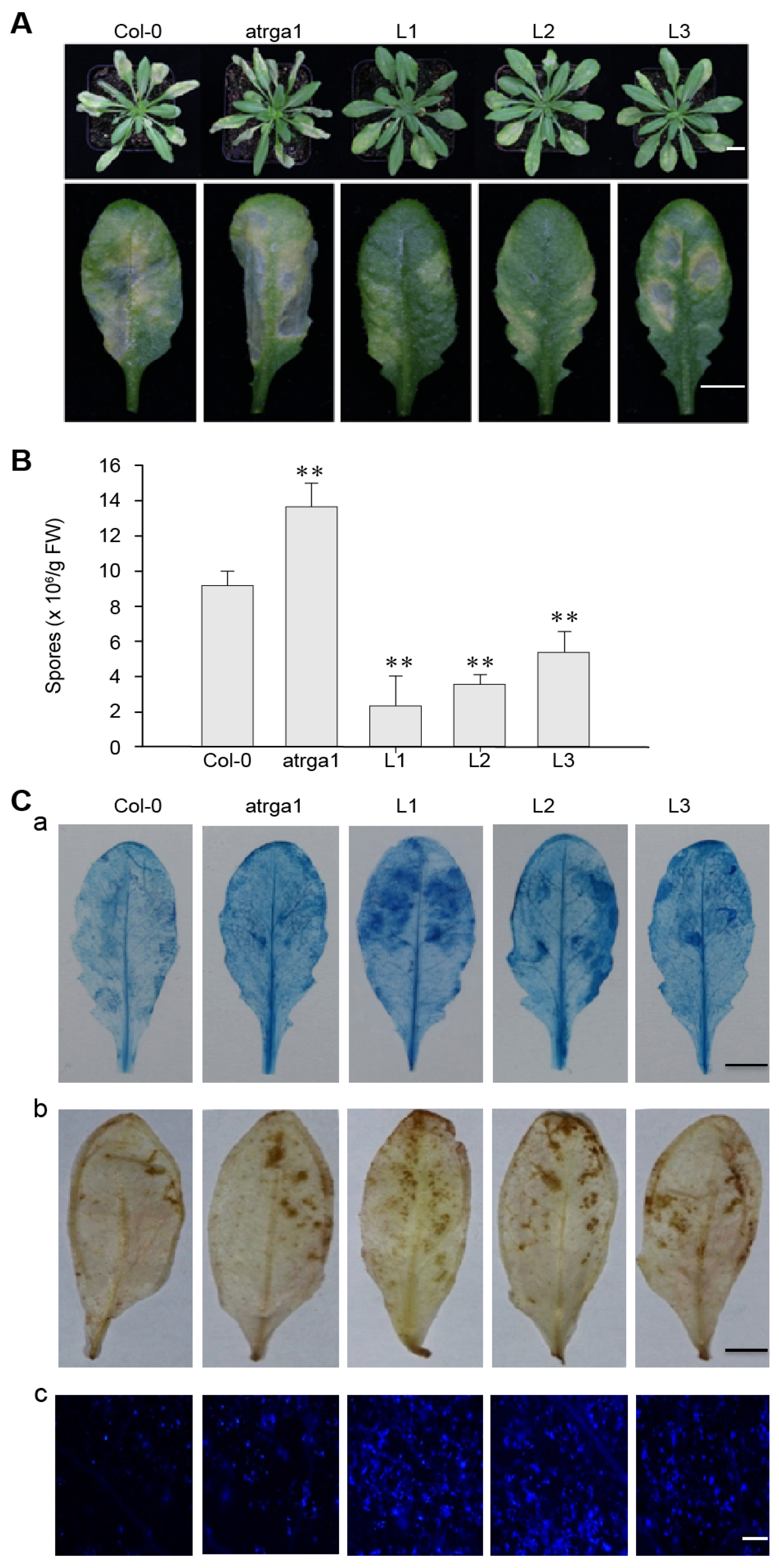
2.3. Expression of VaRGA1 in Arabidopsis Improves Resistance to Hpa
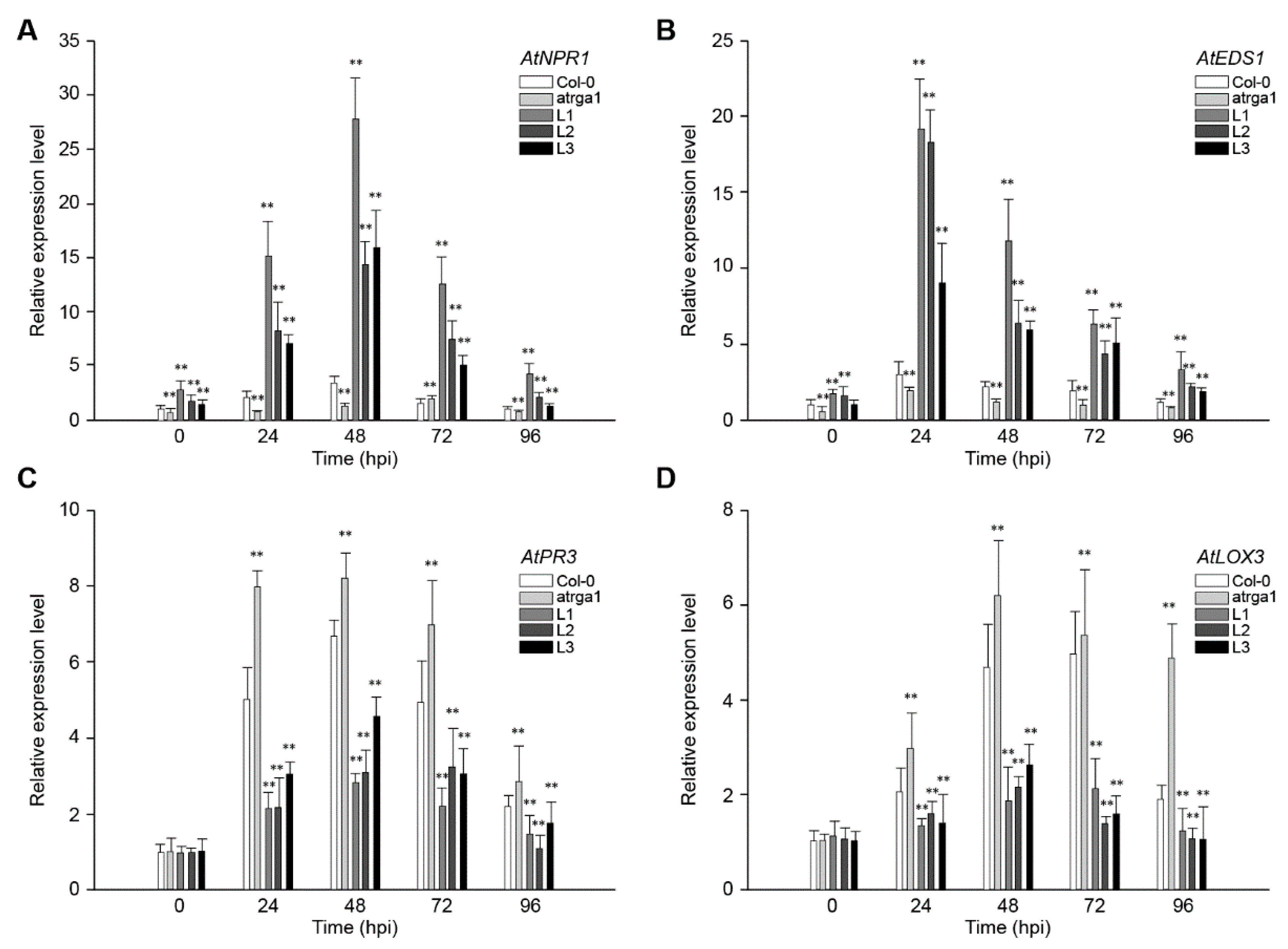
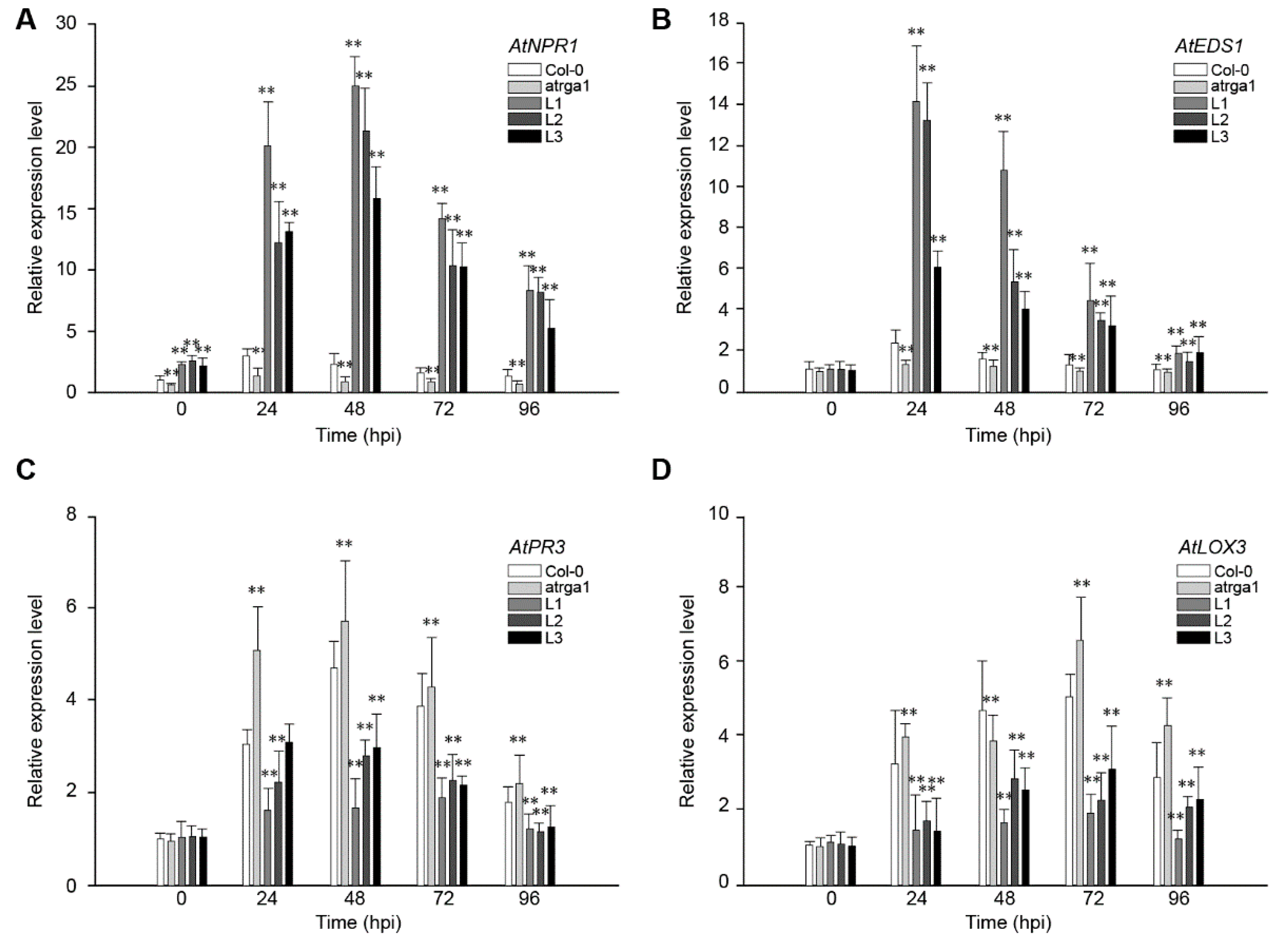
2.4. Assessment of Defense-Related Gene Expression After Inoculating Hpa
2.5. Expression of VaRGA1 in Arabidopsis Improves Resistance to PstDC3000
2.6. Assessment of the Expression of Defense-Related Genes after Inoculating PstDC3000
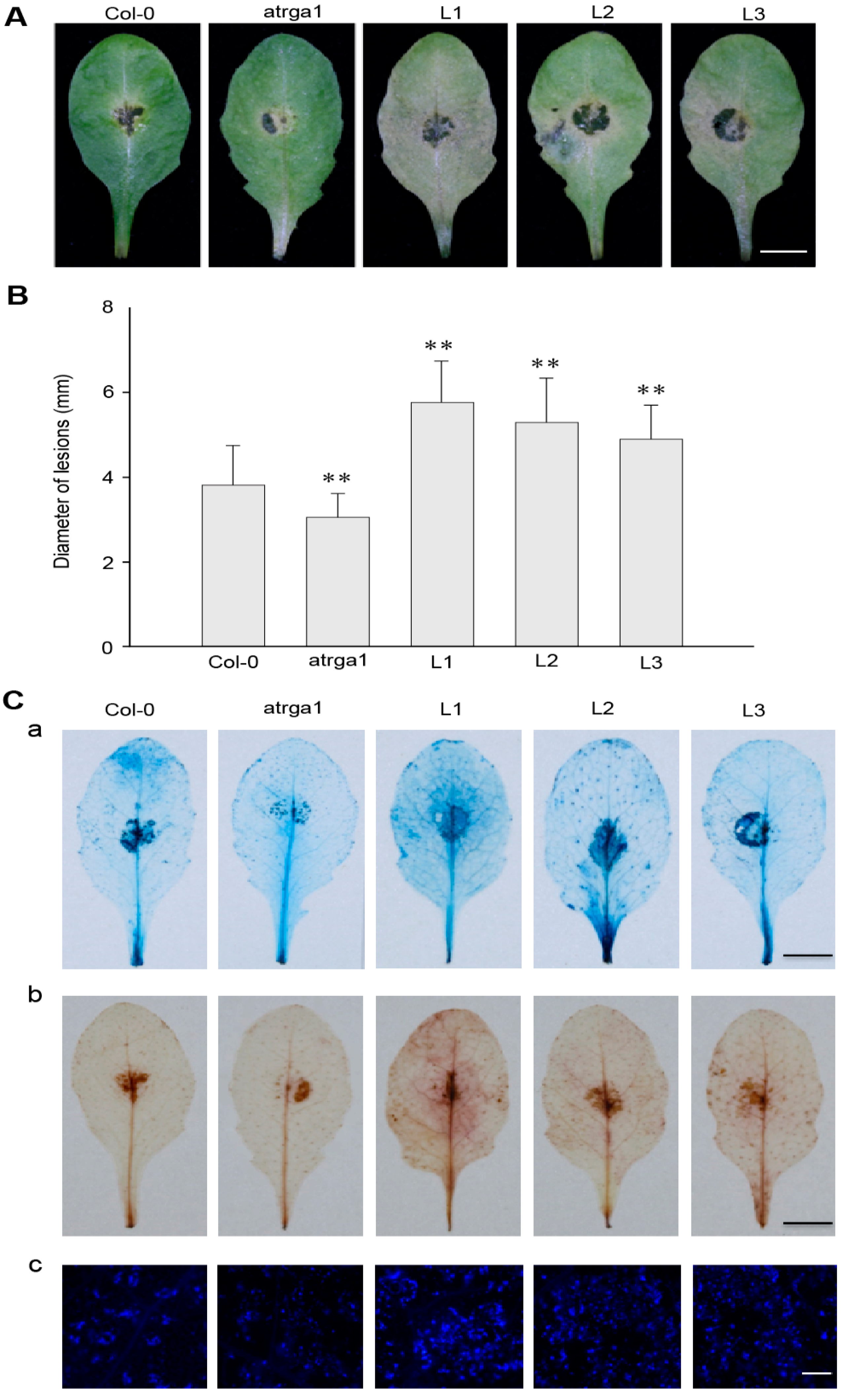
2.7. Expression of VaRGA1 in Arabidopsis Decreases Resistance to B. cinerea
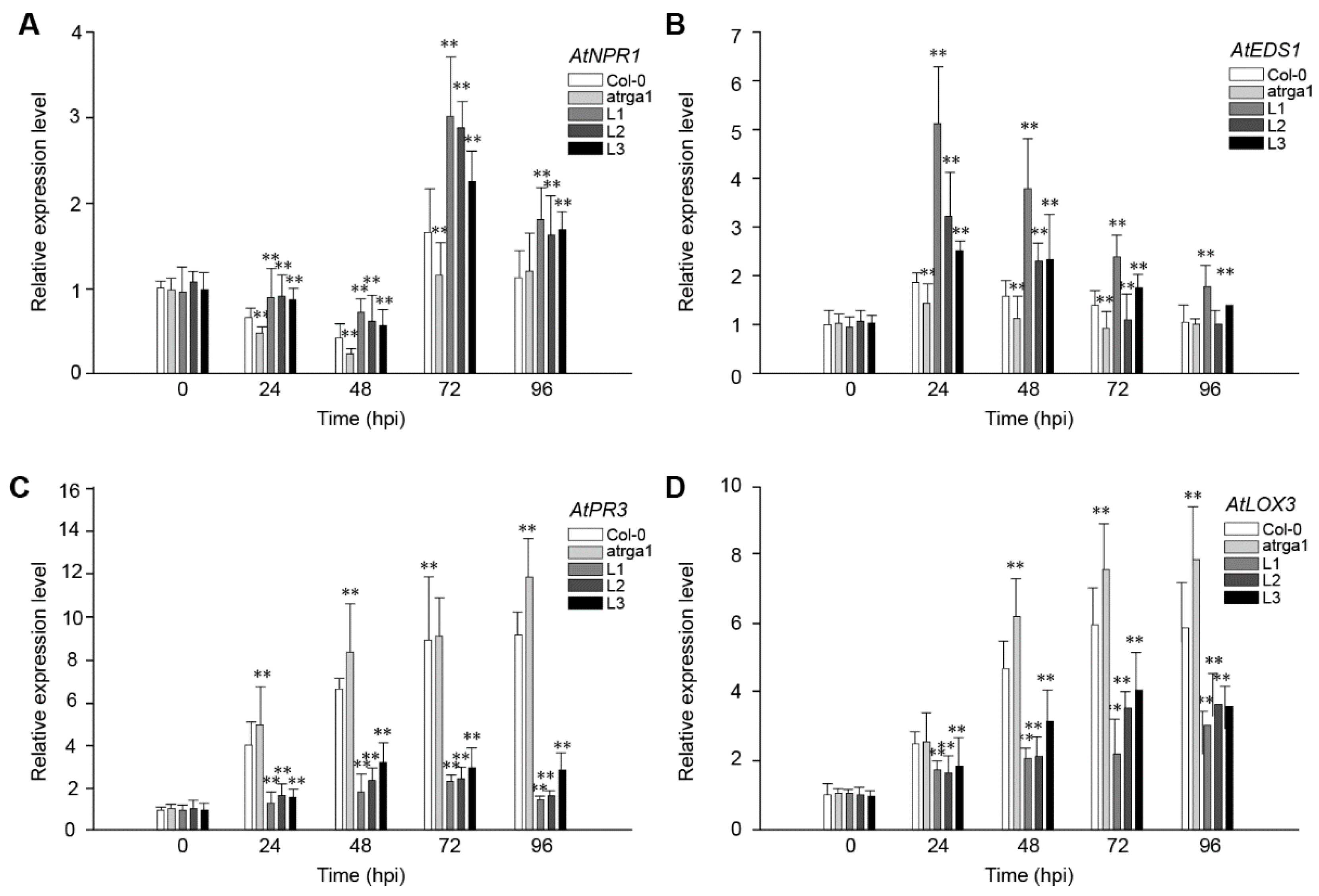
2.8. Assessment of the Expression of Defense-Related Genes After Inoculating B. cinerea
3. Discussion
4. Materials and Methods
4.1. Plant Materials and Pathogens
4.2. Hormone Treatments in Grape
4.3. Generation of Transgenic Plants
4.4. Inoculation of Pathogens
4.5. Expression Analysis of Related Genes by Quantitative Real-Time PCR
4.6. Plant DNA Extraction and VaRGA1 Promoter Cloning
4.7. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| PstDC3000 | Pseudomonas syringae pv. tomato DC3000 |
| B. cinerea | Botrytis cinerea |
| SA | salicylic acid |
| JA | jasmonic acid |
| qRT-PCR | quantitative real-time PCR |
| NBS | nucleotide binding sites |
| LRR | leucine-rich repeats |
| HR | hypersensitive response |
| ROS | reactive oxygen species |
| ET | ethylene |
| ABA | abscisic acid |
| hpi | hours post inoculation |
| PCR | polymerase chain reaction |
| DAB | diaminobenzidine |
| NBT | Nitro blue tetrazolium |
| NPR1 | nonexpressor of pathogenesis-related gene |
| PR1 | pathogenesis-related gene 1 |
| EDS1 | enhanced disease susceptibility 1 |
| LOX3 | Lipoxygenase 3 |
| HR | hypersensitive response |
| ROS | reactive oxygen species |
References
- Mzid, R.; Marchive, C.; Blancard, D.; Deluc, L.; Barrieu, F.; Corio-Costet, M.; Drira, N.; Hamdi, S.; Lauvergeat, V. Overexpression of VvWRKY2 in tobacco enhances broad resistance to necrotrophic fungal pathogens. Physiol. Plant. 2010, 131, 434–447. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Peng, Q.; Li, K.; Xie, D.Y. Molecular Cloning and Functional Characterization of a Dihydroflavonol 4-Reductase from Vitis bellula. Molecules 2018, 23, 861. [Google Scholar] [CrossRef] [PubMed]
- Bag, M.K.; Yadav, M.; Mukherjee, A.K. Bioefficacy of Strobilurin Based Fungicides against Rice Sheath Blight Disease. Transcr. Open Access 2016, 4. [Google Scholar] [CrossRef]
- Yoon, M.Y.; Cha, B.; Kim, J.C. Recent trends in studies on botanical fungicides in agriculture. Plant Pathol. J. 2013, 29, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Banerjee, A.; Tarafdar, J.; Senapati, B.K.; Dasgupta, I. Transfer of transgenes for resistance to rice tungro into high-yielding rice cultivars through gene-based marker-assisted selection. J. Agric. Sci. 2012, 150, 610–618. [Google Scholar] [CrossRef]
- Zhu, Z.; Shi, J.; Cao, J.; He, M.; Wang, Y. VpWRKY3, a biotic and abiotic stress-related transcription factor from the Chinese wild Vitis pseudoreticulata. Plant Cell Rep. 2012, 31, 2109–2120. [Google Scholar] [CrossRef]
- Huang, L.; Zhang, S.; Singer, S.D.; Yin, X.; Yang, J.; Wang, Y.; Wang, X. Expression of the Grape VqSTS21 Gene in Arabidopsis Confers Resistance to Osmotic Stress and Biotrophic Pathogens but Not Botrytis cinerea. Front. Plant Sci. 2016, 7, 1379. [Google Scholar] [CrossRef]
- Jones, J.D.; Dangl, J.L. The plant immune system. Nature 2006, 444, 323–329. [Google Scholar] [CrossRef]
- Shamrai, S.N. Plant immune system: Basal immunity. Cytol. Genet. 2014, 48, 258–271. [Google Scholar] [CrossRef]
- Gururani, M.A.; Venkatesh, J.; Upadhyaya, C.P.; Nookaraju, A.; Pandey, S.K.; Park, S.W. Plant disease resistance genes: Current status and future directions. Physiol. Mol. Plant Pathol. 2012, 78, 51–65. [Google Scholar] [CrossRef]
- Hammond-Kosack, K.E.; Parker, J.E. Deciphering plant–pathogen communication: Fresh perspectives for molecular resistance breeding. Curr. Opin. Biotechnol. 2003, 14, 177–193. [Google Scholar] [CrossRef]
- Yu, D.; Mpina, M.H.; Birch, P.R.J.; Klaas, B.; Francine, G. Phytophthora infestans RXLR Effector AVR1 Interacts with Exocyst Component Sec5 to Manipulate Plant Immunity. Plant Physiol. 2015, 169, 1975–1990. [Google Scholar]
- Yahiaoui, N.; Brunner, S.; Keller, B. Rapid generation of new powdery mildew resistance genes after wheat domestication. Plant J. 2010, 47, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.K. Genome-wide mapping of NBS-LRR genes and their association with disease resistance in soybean. BMC Plant Biol. 2012, 12, 139. [Google Scholar]
- Glazebrook, J. Contrasting Mechanisms of Defense against Biotrophic and Necrotrophic Pathogens. Annu. Rev. Phytopathol. 2005, 43, 205–227. [Google Scholar] [CrossRef]
- Li, N.Y.; Zhou, L.; Zhang, D.D.; Klosterman, S.J.; Li, T.G.; Gui, Y.J.; Kong, Z.Q.; Ma, X.F.; Short, D.P.G.; Zhang, W.Q. Heterologous Expression of the Cotton NBS-LRR GeneGbaNA1Enhances Verticillium Wilt Resistance inArabidopsis. Front. Plant Sci. 2018, 9, 119. [Google Scholar] [CrossRef]
- Karpiński, S.; Szechyńskahebda, M.; Wituszyńska, W.; Burdiak, P. Light acclimation, retrograde signalling, cell death and immune defences in plants. Plant Cell Environ. 2013, 36, 736–744. [Google Scholar] [CrossRef]
- Lee, M.W.; Huffaker, A.; Crippen, D.; Robbins, R.T.; Goggin, F.L. Plant elicitor peptides promote plant defences against nematodes in soybean. Mol. Plant Pathol. 2017, 19, 858–869. [Google Scholar] [CrossRef]
- Wu, L.; Huang, Z.; Li, X.; Ma, L.; Gu, Q.; Wu, H.; Liu, J.; Borriss, R.; Wu, Z.; Gao, X. Stomatal Closure and SA-, JA/ET-Signaling Pathways Are Essential for Bacillus amyloliquefaciens FZB42 to Restrict Leaf Disease Caused by Phytophthora nicotianae in Nicotiana benthamiana. Front. Microbiol. 2018, 9, 847. [Google Scholar] [CrossRef]
- Jin, Y.; Zheng, H.Y.; Jin, X.J.; Zhang, Y.X.; Ren, C.Y. Effect of ABA, SA and JA on Soybean Growth Under Drought Stress and Re-watering. Soybean Sci. 2016, 6, 958–963. [Google Scholar]
- Eshraghi, L.; Anderson, J.P.; Aryamanesh, N.; Mccomb, J.A.; Shearer, B.; Hardy, G.E.S.J. Defence Signalling Pathways Involved in Plant Resistance and Phosphite-Mediated Control of Phytophthora Cinnamomi. Plant Mol. Biol. Report. 2013, 32, 342–356. [Google Scholar] [CrossRef]
- Chen, L.; Zhang, L.; Yu, D. Wounding-Induced WRKY8 Is Involved in Basal Defense in Arabidopsis. Mol. Plant Microbe Interact. 2010, 23, 558–565. [Google Scholar] [CrossRef] [PubMed]
- Ke, Y.; Liu, H.; Li, X.; Xiao, J.; Wang, S. Rice OsPAD4 functions differently from Arabidopsis AtPAD4 in host-pathogen interactions. Plant J. Cell Mol. Biol. 2014, 78, 619–631. [Google Scholar] [CrossRef] [PubMed]
- Feys, B.J.; Moisan, L.J.; Newman, M.A.; Parker, J.E. Direct interaction between the Arabidopsis disease resistance signaling proteins, EDS1 and PAD4. Embo J. 2014, 20, 5400–5411. [Google Scholar] [CrossRef] [PubMed]
- Thomma, B.P.; Eggermont, K.; Penninckx, I.A.; Mauch-Mani, B.; Vogelsang, R.; Cammue, B.P.; Broekaert, W.F. Separate jasmonate-dependent and salicylate-dependent defense-response pathways in Arabidopsis are essential for resistance to distinct microbial pathogens. Proc. Natl. Acad. Sci. USA 1998, 95, 15107–15111. [Google Scholar] [CrossRef] [PubMed]
- Kolosova, N.; Bohlmann, J. Conifer Defense against Insects and Fungal Pathogens. In Growth and Defence in Plants; Springer: Berlin/Heidelberg, Germany, 2012. [Google Scholar]
- Kunkel, B.N.; Brooks, D.M. Cross talk between signaling pathways in pathogen defense. Curr. Opin. Plant Biol. 2002, 5, 325–331. [Google Scholar] [CrossRef]
- Xu, W.; Yu, Y.; Ding, J.; Hua, Z.; Wang, Y. Characterization of a novel stilbene synthase promoter involved in pathogen- and stress-inducible expression from Chinese wild Vitis pseudoreticulata. Planta 2010, 231, 475–487. [Google Scholar] [CrossRef]
- Li, J.; Li, M.; Liang, D.; Cui, M.; Ma, F. Expression patterns and promoter characteristics of the gene encoding Actinidia deliciosal-galactose-1-phosphate phosphatase involved in the response to light and abiotic stresses. Mol. Biol. Rep. 2013, 40, 1473–1485. [Google Scholar] [CrossRef]
- Lai, G.; Fu, P.; Liu, Y.; Xiang, J.; Lu, J. Molecular Characterization and Overexpression of VpRPW8s from Vitis pseudoreticulata Enhances Resistance to Phytophthora capsici in Nicotiana benthamiana. Int. J. Mol. Sci. 2018, 19, 839. [Google Scholar] [CrossRef]
- Stokes, T.L.; Kunkel, B.N.; Richards, E.J. Epigenetic variation in Arabidopsis disease resistance. Genes Dev. 2002, 16, 171. [Google Scholar] [CrossRef]
- Oldroyd, G.E.; Staskawicz, B.J. Genetically engineered broad-spectrum disease resistance in tomato. Proc. Natl. Acad. Sci. USA 1998, 95, 10300–10305. [Google Scholar] [CrossRef] [PubMed]
- Michael Weaver, L.; Swiderski, M.R.; Li, Y.; Jones, J.D. The Arabidopsis thaliana TIR-NB-LRR R-protein, RPP1A; protein localization and constitutive activation of defence by truncated alleles in tobacco and Arabidopsis. Plant J. 2010, 47, 829–840. [Google Scholar] [CrossRef] [PubMed]
- Jens, S.; Maria, K.; Ellen, D.; Mattias, P.; Christina, D. RLM3, a TIR domain encoding gene involved in broad-range immunity of Arabidopsis to necrotrophic fungal pathogens. Plant J. 2010, 55, 188–200. [Google Scholar]
- Li, X.; Zhang, Y.; Yin, L.; Lu, J. Overexpression of pathogen-induced grapevine TIR-NB-LRR gene VaRGA1 enhances disease resistance and drought and salt tolerance in Nicotiana benthamiana. Protoplasma 2017, 254, 957–969. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Y.; Zhang, X.; Cheng, Z.-M. Lineage-specific duplications of NBS-LRR genes occurring before the divergence of six Fragaria species. BMC Genom. 2018, 19, 128. [Google Scholar] [CrossRef] [PubMed]
- Jing, W.; Zhu, J.; Wang, L.; Wang, S. Genome-Wide Association Study Identifies NBS-LRR-Encoding Genes Related with Anthracnose and Common Bacterial Blight in the Common Bean. Front. Plant Sci. 2017, 8, 1398. [Google Scholar]
- Li, X.; Wu, J.; Yin, L.; Zhang, Y.; Qu, J.; Lu, J. Comparative transcriptome analysis reveals defense-related genes and pathways against downy mildew in Vitis amurensis grapevine. Plant Physiol. Biochem. 2015, 95, 1–14. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, F.; Liu, C.H.; Fan, X.C.; Sun, H.S.; Jiang, J.F.; Zhang, G.H. Isolation and Identification of NBS-LRR Resistance Gene Analogs Sequences from Vitis davidii. Sci. Agric. Sin. 2013, 46, 780–789. [Google Scholar]
- Shi, Y.; Niu, K.; Huang, B.; Liu, W.; Ma, H. Transcriptional Responses of Creeping Bentgrass to 2,3-Butanediol, a Bacterial Volatile Compound (BVC) Analogue. Molecules 2017, 22, 1318. [Google Scholar] [CrossRef]
- Kortekamp, A.; Welter, L.; Vogt, S.; Knoll, A.; Schwander, F.; Töpfer, R.; Zyprian, E. Identification, isolation and characterization of a CC-NBS-LRR candidate disease resistance gene family in grapevine. Mol. Breed. 2008, 22, 421–432. [Google Scholar] [CrossRef]
- Mohr, T.J.; Mammarella, N.D.; Hoff, T.; Woffenden, B.J.; Jelesko, J.G.; Mcdowell, J.M. The Arabidopsis Downy Mildew Resistance Gene RPP8 Is Induced by Pathogens and Salicylic Acid and Is Regulated by W Box cis Elements. Mol. Plant Microbe Interact. MPMI 2010, 23, 1303. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.H.; Kapoor, A.; Zhu, J.; Zhu, J.K. STABILIZED1, a stress-upregulated nuclear protein, is required for pre-mRNA splicing, mRNA turnover, and stress tolerance in Arabidopsis. Plant Cell 2006, 18, 1736–1749. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.L.V.; Chekanova, J.A. Small RNAs: Essential regulators of gene expression and defenses against environmental stresses in plants. Wiley Interdiscip. Rev. RNA 2016, 7, 356–381. [Google Scholar] [CrossRef] [PubMed]
- Chung, J.S.; Zhu, J.K.; Bressan, R.A.; Hasegawa, P.M.; Shi, H. Reactive oxygen species mediate Na+-induced SOS1 mRNA stability in Arabidopsis. Plant J. 2008, 53, 554–565. [Google Scholar] [CrossRef] [PubMed]
- Govrin, E.M.; Rachmilevitch, S.; Tiwari, B.S.; Solomon, M.; Levine, A. An Elicitor from Botrytis cinerea Induces the Hypersensitive Response in Arabidopsis thaliana and Other Plants and Promotes the Gray Mold Disease. Phytopathology 2006, 96, 299–307. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Sonbol, F.-M.; Huot, B.; Gu, Y.; Withers, J.; Mwimba, M.; Yao, J.; He, S.Y.; Dong, X. Salicylic acid receptors activate jasmonic acid signalling through a non-canonical pathway to promote effector-triggered immunity. Nat. Commun. 2016, 7, 13099. [Google Scholar] [CrossRef]
- Manosalva, P.M.; Bruce, M.; Leach, J.E. Rice 14-3-3 protein (GF14e) negatively affects cell death and disease resistance. Plant J. 2011, 68, 777–787. [Google Scholar] [CrossRef]
- Temme, N.; Tudzynski, P. Does Botrytis cinerea Ignore H2O2 -Induced Oxidative Stress during Infection? Characterization of Botrytis Activator Protein 1. Mol. Plant Microbe Interact. MPMI 2009, 22, 987. [Google Scholar] [CrossRef]
- Wang, W.; Devoto, A.; Turner, J.G.; Xiao, S. Expression of the Membrane-Associated Resistance Protein RPW8 Enhances Basal Defense Against Biotrophic Pathogens. Mol. Plant Microbe Interact. MPMI 2007, 20, 966. [Google Scholar] [CrossRef]
- Stephens, A.E. The Fusarium mycotoxin deoxynivalenol elicits hydrogen peroxide production, programmed cell death and defence responses in wheat. Mol. Plant Pathol. 2010, 9, 435–445. [Google Scholar]
- Wang, G.; Xiao, Y.; Deng, X.; Zhang, H.; Li, T.; Chen, H. Exogenous Hydrogen Peroxide Contributes to Heme Oxygenase-1 Delaying Programmed Cell Death in Isolated Aleurone Layers of Rice Subjected to Drought Stress in a cGMP-Dependent Manner. Front. Plant Sci. 2018, 9, 84. [Google Scholar] [CrossRef] [PubMed]
- Kotchoni, S.O.; Gachomo, E.W. The reactive oxygen species network pathways: An essential prerequisite for perception of pathogen attack and the acquired disease resistance in plants. J. Biosci. 2006, 31, 389–404. [Google Scholar] [CrossRef] [PubMed]
- Nam-Soo, J.; Kook, H.B. Convergent Evolution of Pathogen Effectors toward Reactive Oxygen Species Signaling Networks in Plants. Front. Plant Sci. 2017, 8, 1687. [Google Scholar]
- Chini, A.; Grant, J.J.; Seki, M.; Shinozaki, K.; Loake, G.J. Drought tolerance established by enhanced expression of the CC–NBS–LRR gene, ADR1, requires salicylic acid, EDS1 and ABI1. Plant J. 2010, 38, 810–822. [Google Scholar] [CrossRef] [PubMed]
- Dangl, J.L.; Jones, J.D.G. Plant pathogens and integrated defence responses to infection. Nature 2001, 411, 826–833. [Google Scholar] [CrossRef]
- Wang, X.; Chen, J.; Yang, Y.; Zhou, J.; Qiu, Y.; Yu, C.; Cheng, Y.; Yan, C.; Chen, J. Characterization of a Novel NBS-LRR Gene Involved in Bacterial Blight Resistance in Rice. Plant Mol. Biol. Report. 2013, 31, 649–656. [Google Scholar] [CrossRef]
- Malik, C.; Agnès, K.; Abdelwahad, E.; Philippe, M.; Dominique, V.F.; Marielle, A. Antimicrobial activity of resveratrol analogues. Molecules 2014, 19, 7679–7688. [Google Scholar]
- Pieterse, C.M.J.; Spoel, S.H.; Ton, J.; Wees, A.C.M.V.; Pelt, J.A.V.; Loon, L.C.V. Cross-talk between salicylate- and jasmonate-dependent induced defenses in Arabidopsis. Biol. Plant Microbe Interact. 2002, 3, 107–112. [Google Scholar]
- Glazebrook, J.; Chen, W.; Estes, B.; Chang, H.S.; Nawrath, C.; Métraux, J.P.; Zhu, T.; Katagiri, F. Topology of the network integrating salicylate and jasmonate signal transduction derived from global expression phenotyping. Plant J. 2010, 34, 217–228. [Google Scholar] [CrossRef]
- Gimenez-Ibanez, S.; Solano, R. Nuclear jasmonate and salicylate signaling and crosstalk in defense against pathogens. Front. Plant Sci. 2013, 4, 59–60. [Google Scholar] [CrossRef]
- Venugopal, S.C.; Jeong, R.D.; Mandal, M.K.; Zhu, S.; Chandra-Shekara, A.C.; Xia, Y.; Hersh, M.; Stromberg, A.J.; Navarre, D.; Kachroo, A. Enhanced Disease Susceptibility 1 and Salicylic Acid Act Redundantly to Regulate Resistance Gene-Mediated Signaling. PLoS Genet. 2009, 5, e1000545. [Google Scholar] [CrossRef] [PubMed]
- Inoue, H.; Hayashi, N.; Matsushita, A.; Liu, X.; Nakayama, A.; Sugano, S.; Jiang, C.-J.; Takatsuji, H. Blast resistance of CC-NB-LRR protein Pb1 is mediated by WRKY45 through protein–protein interaction. Proc. Natl. Acad. Sci. USA 2013, 110, 9577–9582. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Yu, Y.; Zhou, Q.; Ding, J.; Dai, L.; Xie, X.; Xu, Y.; Zhang, C.; Wang, Y. Expression pattern, genomic structure, and promoter analysis of the gene encoding stilbene synthase from Chinese wild Vitis pseudoreticulata. J. Exp. Bot. 2011, 62, 2745–2761. [Google Scholar] [CrossRef] [PubMed]
- Jing, H.; Dan, W.; Zhang, J.; Wang, Y. Functional analysis of the fruit-specific promoter of VqSTS6 from the Chinese wild grape, Vitis quinquangularis. Agri Gene 2016, 1, 38–45. [Google Scholar]
- Li, Y.; Zhang, T.; Zhang, X.; Wang, G.; Wang, Y.; Zhang, Z. Heat shock cognate 70 gene in Haliotis diversicolor: Responses to pathogen infection and environmental stresses and its transcriptional regulation analysis. Cell Stress Chaperones 2018, 90, 335–346. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Zhu, X.; Wang, Y.; Liu, L.; Xu, B.; Li, F.; Fang, J.; Chu, C. Semi-dominant mutations in the CC-NB-LRR-type R gene, NLS1, lead to constitutive activation of defense responses in rice. Plant J. 2011, 66, 996–1007. [Google Scholar] [CrossRef]
- Borhan, M.H.; Holub, E.B.; Kindrachuk, C.; Omidi, M.; Bozorgmaneshfrad, G.; Rimmer, S.R. WRR4, a broad-spectrum TIR-NB-LRR gene from Arabidopsis thaliana that confers white rust resistance in transgenic oilseed brassica crops. Mol. Plant Pathol. 2010, 11, 283–291. [Google Scholar] [CrossRef]
- Eurasian Arabidopsis Stock Centre. Available online: http://arabidopsis.info/ (accessed on 10 April 2017).
- Guo, R.; Tu, M.; Wang, X.; Zhao, J.; Wan, R.; Li, Z.; Wang, Y.; Wang, X. Ectopic expression of a grape aspartic protease gene, AP13, in Arabidopsis thaliana improves resistance to powdery mildew but increases susceptibility to Botrytis cinerea. Plant Sci. 2016, 248, 17–27. [Google Scholar] [CrossRef]
- Clough, S.J.; Bent, A.F. Floral dip: A simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 2010, 16, 735–743. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, D.; Wang, F.; Huang, L.; Tian, X.; Nocker, S.V.; Gao, H.; Wang, X. Expression of the Grape VaSTS19 Gene in Arabidopsis Improves Resistance to Powdery Mildew and Botrytis cinerea but Increases Susceptibility to Pseudomonas syringe pv Tomato DC3000. Int. J. Mol. Sci. 2017, 18, 2000. [Google Scholar] [CrossRef]
- Huang, L.; Yin, X.; Sun, X.; Yang, J.; Rahman, M.; Chen, Z.; Wang, X. Expression of a Grape VqSTS36-Increased Resistance to Powdery Mildew and Osmotic Stress in Arabidopsis but Enhanced Susceptibility to Botrytis cinerea in Arabidopsis and Tomato. Int. J. Mol. Sci. 2018, 19, 2985. [Google Scholar] [CrossRef] [PubMed]
- Vogel, J.; Somerville, S. Isolation and characterization of powdery mildew-resistant Arabidopsis mutants. Proc. Natl. Acad. Sci. USA 2000, 97, 1897–1902. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Woo, D.H.; Kim, J.M.; Lee, S.Y.; Chung, W.S.; Moon, Y.H. Arabidopsis MKK4 mediates osmotic-stress response via its regulation of MPK3 activity. Biochem. Biophys. Res. Commun. 2011, 412, 150–154. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Xu, W.; Wang, J.; Wang, L.; Yao, W.; Xu, Y.; Ding, J.; Wang, Y. A core functional region of the RFP1 promoter from Chinese wild grapevine is activated by powdery mildew pathogen and heat stress. Planta 2013, 237, 293–303. [Google Scholar] [CrossRef]
- PlantCARE. Available online: http://bioinformatics.psb.ugent.be/webtools/plantcare/html/ (accessed on 15 July 2019).
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tian, S.; Yin, X.; Fu, P.; Wu, W.; Lu, J. Ectopic Expression of Grapevine Gene VaRGA1 in Arabidopsis Improves Resistance to Downy Mildew and Pseudomonas syringae pv. tomato DC3000 But Increases Susceptibility to Botrytis cinerea. Int. J. Mol. Sci. 2020, 21, 193. https://doi.org/10.3390/ijms21010193
Tian S, Yin X, Fu P, Wu W, Lu J. Ectopic Expression of Grapevine Gene VaRGA1 in Arabidopsis Improves Resistance to Downy Mildew and Pseudomonas syringae pv. tomato DC3000 But Increases Susceptibility to Botrytis cinerea. International Journal of Molecular Sciences. 2020; 21(1):193. https://doi.org/10.3390/ijms21010193
Chicago/Turabian StyleTian, Shanshan, Xiangjing Yin, Peining Fu, Wei Wu, and Jiang Lu. 2020. "Ectopic Expression of Grapevine Gene VaRGA1 in Arabidopsis Improves Resistance to Downy Mildew and Pseudomonas syringae pv. tomato DC3000 But Increases Susceptibility to Botrytis cinerea" International Journal of Molecular Sciences 21, no. 1: 193. https://doi.org/10.3390/ijms21010193
APA StyleTian, S., Yin, X., Fu, P., Wu, W., & Lu, J. (2020). Ectopic Expression of Grapevine Gene VaRGA1 in Arabidopsis Improves Resistance to Downy Mildew and Pseudomonas syringae pv. tomato DC3000 But Increases Susceptibility to Botrytis cinerea. International Journal of Molecular Sciences, 21(1), 193. https://doi.org/10.3390/ijms21010193



