Niemann-Pick Type A Disease: Behavior of Neutral Sphingomyelinase and Vitamin D Receptor
Abstract
1. Introduction
2. Results
2.1. aSMase-KO Mice Exhibit the Increase in SOX2 Expression in Hippocampal GD
2.2. aSMase Deficiency Leads to Overexpression of TLRs mRNA
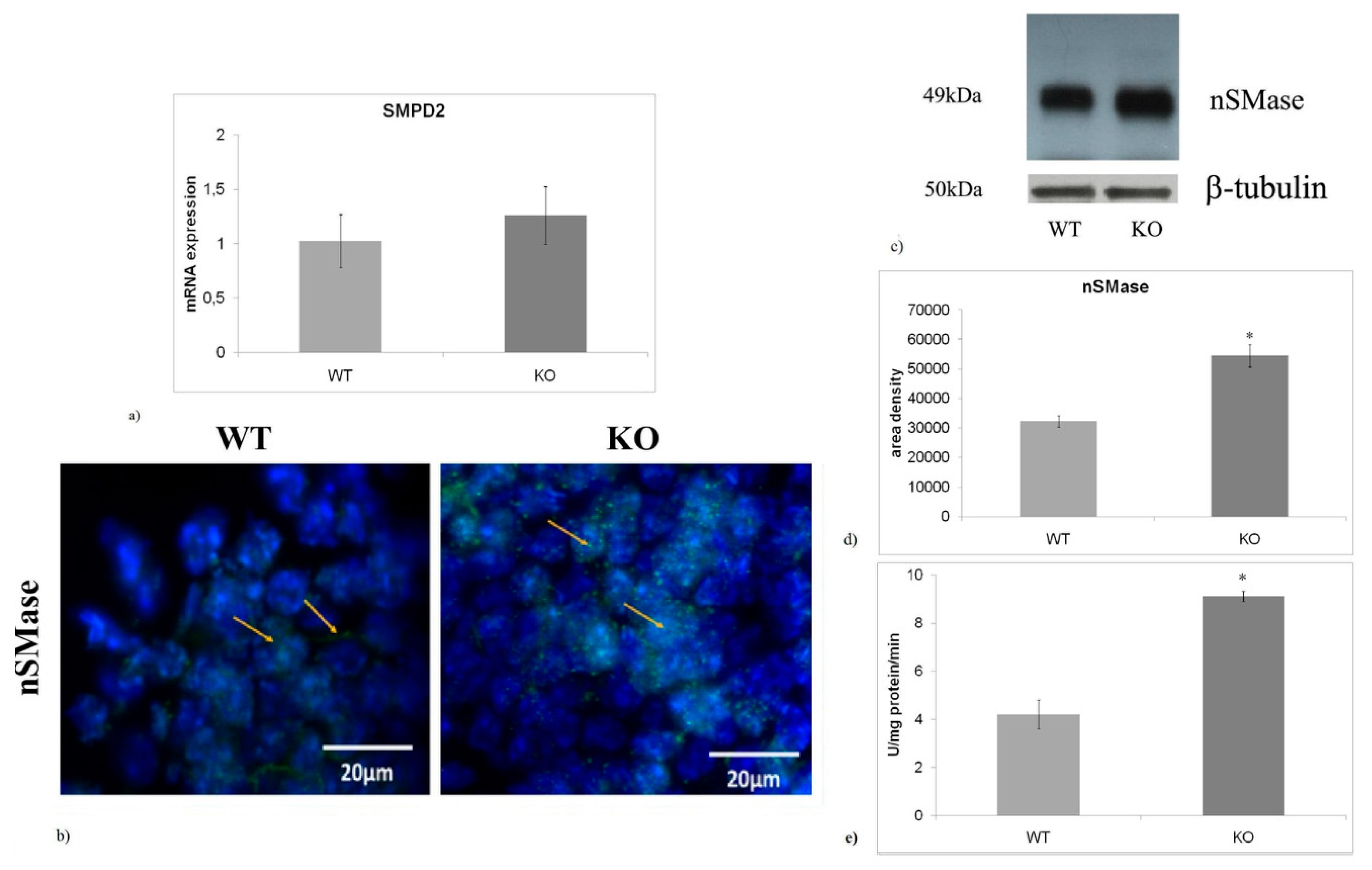
2.3. nSMase is Up-Regulated in aSMase-KO Mice
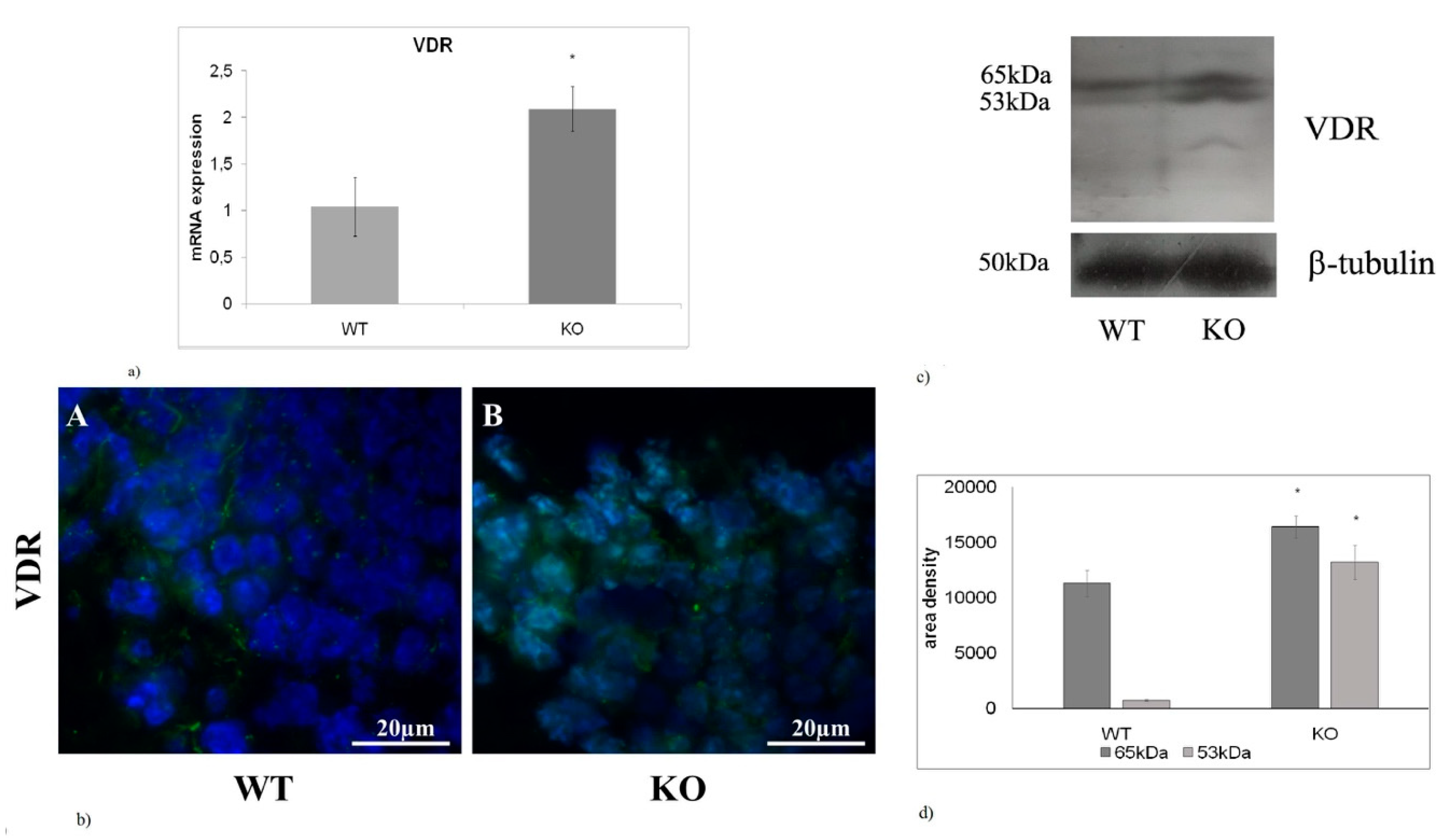
2.4. The absence of aSMase Causes VDR Overexpression and Nuclear Localization
3. Discussion
4. Material and Methods
4.1. Materials
4.2. Mice Hippocampi
4.3. Reverse Transcription Quantitative PCR (RT-qPCR)
4.4. Western Blotting
4.5. Immunofluorescence
4.6. Nuclear Sphingomyelinase Activity Assay
4.7. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Charruyer, A.; Grazide, S.; Bezombes, C.; Müller, S.; Laurent, G.; Jaffrézou, J.P. UV-C light induces raft-associated acid sphingomyelinase and JNK activation and translocation independently on a nuclear signal. J. Biol. Chem. 2005, 280, 19196–19204. [Google Scholar] [CrossRef]
- Vanier, M.T. Niemann-Pick diseases. Handb. Clin. Neurol. 2013, 113, 1717–1721. [Google Scholar]
- Schuchman, E.H.; Desnick, R.J. Types A and B Niemann-Pick disease. Mol. Genet. Metab. 2017, 120, 27–33. [Google Scholar] [CrossRef]
- Ridgway, N.D. Interactions between metabolism and intracellular distribution of cholesterol and sphingomyelin. Biochim. Biophys. Acta 2000, 1484, 129–141. [Google Scholar] [CrossRef]
- McGovern, M.M.; Aron, A.; Brodie, S.E.; Desnick, R.J.; Wasserstein, M.P. Natural history of Type A Niemann-Pick disease: Possible endpoints for therapeutic trials. Neurology 2006, 66, 228–232. [Google Scholar] [CrossRef]
- Otterbach, B.; Stoffel, W. Acid sphingomyelinase-deficient mice mimic the neurovisceral form of human lysosomal storage disease (Niemann-Pick disease). Cell 1995, 81, 1053–1061. [Google Scholar] [CrossRef]
- Horinouchi, K.; Erlich, S.; Perl, D.P.; Ferlinz, K.; Bisgaier, C.L.; Sandhoff, K.; Desnick, R.J.; Stewart, C.L.; Schuchman, E.H. Acid sphingomyelinase deficient mice: A model of types A and B Niemann-Pick disease. Nat. Genet. 1995, 10, 288–293. [Google Scholar] [CrossRef] [PubMed]
- Camoletto, P.G.; Vara, H.; Morando, L.; Connell, E.; Marletto, F.P.; Giustetto, M.; Sassoè-Pognetto, M.; Van Veldhoven, P.P.; Ledesma, M.D. Synaptic vesicle docking: Sphingosine regulates syntaxin1 interaction with Munc18. PLoS ONE 2009, 4, e5310. [Google Scholar] [CrossRef] [PubMed]
- Arroyo, A.I.; Camoletto, P.G.; Morando, L.; Sassoe-Pognetto, M.; Giustetto, M.; Van Veldhoven, P.P.; Schuchman, E.H.; Ledesma, M.D. Pharmacological reversion of sphingomyelin-induced dendritic spine anomalies in a Niemann-Pick disease type A mouse model. EMBO Mol. Med. 2014, 6, 398–413. [Google Scholar] [CrossRef]
- Cataldi, S.; Arcuri, C.; Hunot, S.; Légeron, F.P.; Mecca, C.; Garcia-Gil, M.; Lazzarini, A.; Codini, M.; Beccari, T.; Tasegian, A.; et al. Neutral Sphingomyelinase Behaviour in Hippocampus Neuroinflammation of MPTP-Induced Mouse Model of Parkinson’s Disease and in Embryonic Hippocampal Cells. Mediators Inflamm. 2017, 2017, 2470950. [Google Scholar] [CrossRef]
- Cataldi, S.; Arcuri, C.; Hunot, S.; Mecca, C.; Codini, M.; Laurenti, M.E.; Ferri, I.; Loreti, E.; Garcia-Gil, M.; Traina, G.; et al. Effect of Vitamin D in HN9.10e Embryonic Hippocampal Cells and in Hippocampus from MPTP-Induced Parkinson’s Disease Mouse Model. Front. Cell Neurosci. 2018, 12, 31. [Google Scholar] [CrossRef]
- Byun, K.; Kim, D.; Bayarsaikhan, E.; Oh, J.; Kim, J.; Kwak, G.; Jeong, G.B.; Jo, S.M.; Lee, B. Changes of calcium binding proteins, c-Fos and COX in hippocampal formation and cerebellum of Niemann-Pick, type C mouse. J. Chem. Neuroanat. 2013, 52, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Lo Castro, A.; Murdocca, M.; Pucci, S.; Zaratti, A.; Greggi, C.; Sangiuolo, F.; Tancredi, V.; Frank, C.; D’Arcangelo, G. Early Hippocampal i-LTP and LOX-1 Overexpression Induced by Anoxia: A Potential Role in Neurodegeneration in NPC Mouse Model. Int. J. Mol. Sci. 2017, 18, 1442. [Google Scholar] [CrossRef] [PubMed]
- Causevic, M.; Dominko, K.; Malnar, M.; Vidatic, L.; Cermak, S.; Pigoni, M.; Kuhn, P.-H.; Colombo, A.; Havas, D.; Flunkert, S.; et al. BACE1-cleavage of Sez6 and Sez6L is elevated in Niemann-Pick type C disease mouse brains. PLoS ONE 2018, 13, e0200344. [Google Scholar] [CrossRef] [PubMed]
- Conte, C.; Roscini, L.; Sardella, R.; Mariucci, G.; Scorzoni, S.; Beccari, T.; Corte, L. Toll Like Receptor 4 Affects the Cerebral Biochemical Changes Induced by MPTP Treatment. Neurochem. Res. 2017, 42, 493–500. [Google Scholar] [CrossRef]
- Mariucci, G.; Pagiotti, R.; Galli, F.; Romani, L.; Conte, C. The Potential Role of Toll-Like Receptor 4 in Mediating Dopaminergic Cell Loss and Alpha-Synuclein Expression in the Acute MPTP Mouse Model of Parkinson’s Disease. J. Mol. Neurosci. 2018, 64, 611–618. [Google Scholar] [CrossRef]
- Guo, H.; Zhao, M.; Qiu, X.; Deis, J.A.; Huang, H.; Tang, Q.Q.; Chen, X. Niemann-Pick type C2 deficiency impairs autophagy-lysosomal activity, mitochondrial function, and TLR signaling in adipocytes. J. Lipid Res. 2016, 57, 1644–1658. [Google Scholar] [CrossRef] [PubMed]
- Barak, B.; Feldman, N.; Okun, E. Toll-like receptors as developmental tools that regulate neurogenesis during development: An update. Front. Neurosci. 2014, 28, 272. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Li, X.; Yue, H.; Ji, J.; You, M.; Ding, L.; Fan, H.; Hou, Y. TLR-Induced SMPD3 Defects Enhance Inflammatory Response of B Cell and Macrophage in the Pathogenesis of SLE. Scand. J. Immunol. 2017, 86, 377–388. [Google Scholar] [CrossRef]
- Luan, W.; Hammond, L.A.; Vuillermot, S.; Meyer, U.; Eyles, D.W. Maternal Vitamin D Prevents Abnormal Dopaminergic Development and Function in a Mouse Model of Prenatal Immune Activation. Sci. Rep. 2001, 27, 9741. [Google Scholar] [CrossRef]
- Gage, F.H. Mammalian neural stem cells. Science 2000, 287, 1433–1438. [Google Scholar] [CrossRef]
- Ortiz-López, L.; Pérez-Beltran, C.; Ramírez-Rodríguez, G. Chronic administration of a melatonin membrane receptor antagonist, luzindole, affects hippocampal neurogenesis without changes in hopelessness-like behavior in adult mice. Neuropharmacology 2016, 103, 211–221. [Google Scholar] [CrossRef] [PubMed]
- Rolls, A.; Shechter, R.; London, A.; Ziv, Y.; Ronen, A.; Levy, R.; Schwartz, M. Toll-like receptors modulate adult hippocampal neurogenesis. Nat. Cell Biol. 2007, 9, 1081–1088. [Google Scholar] [CrossRef]
- Okun, E.; Griffioen, K.; Barak, B.; Roberts, N.J.; Castro, K.; Pita, M.A.; Cheng, A.; Mughal, M.R.; Wan, R.; Ashery, U.; et al. Toll-like receptor3 inhibits memory retention and constrains adult hippocampal neurogenesis. Proc. Natl. Acad. Sci. USA 2010, 107, 15625–15630. [Google Scholar] [CrossRef]
- Kaul, D.; Habbel, P.; Derkow, K.; Krüger, C.; Franzoni, E.; Wulczyn, F.G.; Bereswill, S.; Nitsch, R.; Schott, E.; Veh, R.; et al. Expression of Toll-Like Receptors in the Developing Brain. PLoS ONE 2012, 7, e37767. [Google Scholar] [CrossRef]
- Albi, E.; Viola Magni, M.P. The role of intranuclear lipids. Biol. Cell. 2004, 96, 657–667. [Google Scholar] [CrossRef]
- Bartoccini, E.; Marini, F.; Damaskopoulou, E.; Lazzarini, R.; Cataldi, S.; Cascianelli, G.; Gil Garcia, M.; Albi, E. Nuclear lipid microdomains regulate nuclear vitamin D3 uptake and influence embryonic hippocampal cell differentiation. Mol. Biol. Cell 2011, 22, 3022–3031. [Google Scholar] [CrossRef] [PubMed]
- Pike, J.W.; Meyer, M.B. The vitamin D receptor: New paradigms for the regulation of gene expression by 1,25-dihydroxyvitamin D(3). Endocrinol. Metab. Clin. North. Am. 2010, 39, 255–269. [Google Scholar] [CrossRef]
- Féron, F.; Burne, T.H.; Brown, J.; Smith, E.; McGrath, J.J.; Mackay-Sim, A.; Eyles, D.W. Developmental vitamin D3 deficiency alters the adult rat brain. Brain Res. Bull. 2005, 65, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Mancuso, F.; Arato, I.; Lilli, C.; Bellucci, C.; Bodo, M.; Calvitti, M.; Aglietti, M.C.; dell’Omo, M.; Nastruzzi, C.; Calafiore, R.; et al. Acute effects of lead on porcine neonatal Sertoli cells in vitro. Toxicol In Vitro 2018, 48, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Codini, M.; Cataldi, S.; Ambesi-Impiombato, F.S.; Lazzarini, A.; Floridi, A.; Lazzarini, R.; Curcio, F.; Beccari, T.; Albi, E. Gentamicin arrests cancer cell growth: The intriguing involvement of nuclear sphingomyelin metabolism. Int. J. Mol. Sci. 2015, 16, 2307–2319. [Google Scholar] [CrossRef]
- Albi, E.; Cataldi, S.; Ferri, I.; Sidoni, A.; Traina, G.; Fettucciari, K.; Ambesi-Impiombato, F.S.; Lazzarini, A.; Curcio, F.; Ceccarini, M.R.; et al. VDR independent induction of acid-sphingomyelinase by 1,23(OH)2 D3 in gastric cancer cells: Impact on apoptosis and cell morphology. Biochimie 2017, 146, 35–42. [Google Scholar] [CrossRef]
- Arcuri, C.; Giambanco, I.; Bianchi, R.; Donato, R. Annexin V, annexin VI, S100A1 and S100B in developing and adult avian skeletal muscles. Neuroscience 2002, 109, 371–388. [Google Scholar] [CrossRef]
- Codini, M.; Conte, C.; Cataldi, S.; Arcuri, C.; Lazzarini, A.; Ceccarini, M.R.; Patria, F.; Floridi, A.; Mecca, C.; Ambesi-Impiombato, F.S.; et al. Nuclear Lipid Microdomains Regulate Daunorubicin Resistance in Hepatoma Cells. Int. J. Mol. Sci. 2018, 19, 3424. [Google Scholar] [CrossRef]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Conte, C.; Arcuri, C.; Cataldi, S.; Mecca, C.; Codini, M.; Ceccarini, M.R.; Patria, F.F.; Beccari, T.; Albi, E. Niemann-Pick Type A Disease: Behavior of Neutral Sphingomyelinase and Vitamin D Receptor. Int. J. Mol. Sci. 2019, 20, 2365. https://doi.org/10.3390/ijms20092365
Conte C, Arcuri C, Cataldi S, Mecca C, Codini M, Ceccarini MR, Patria FF, Beccari T, Albi E. Niemann-Pick Type A Disease: Behavior of Neutral Sphingomyelinase and Vitamin D Receptor. International Journal of Molecular Sciences. 2019; 20(9):2365. https://doi.org/10.3390/ijms20092365
Chicago/Turabian StyleConte, Carmela, Cataldo Arcuri, Samuela Cataldi, Carmen Mecca, Michela Codini, Maria Rachele Ceccarini, Federica Filomena Patria, Tommaso Beccari, and Elisabetta Albi. 2019. "Niemann-Pick Type A Disease: Behavior of Neutral Sphingomyelinase and Vitamin D Receptor" International Journal of Molecular Sciences 20, no. 9: 2365. https://doi.org/10.3390/ijms20092365
APA StyleConte, C., Arcuri, C., Cataldi, S., Mecca, C., Codini, M., Ceccarini, M. R., Patria, F. F., Beccari, T., & Albi, E. (2019). Niemann-Pick Type A Disease: Behavior of Neutral Sphingomyelinase and Vitamin D Receptor. International Journal of Molecular Sciences, 20(9), 2365. https://doi.org/10.3390/ijms20092365






