IL-1β Damages Fibrocartilage and Upregulates MMP-13 Expression in Fibrochondrocytes in the Condyle of the Temporomandibular Joint
Abstract
:1. Introduction
2. Results
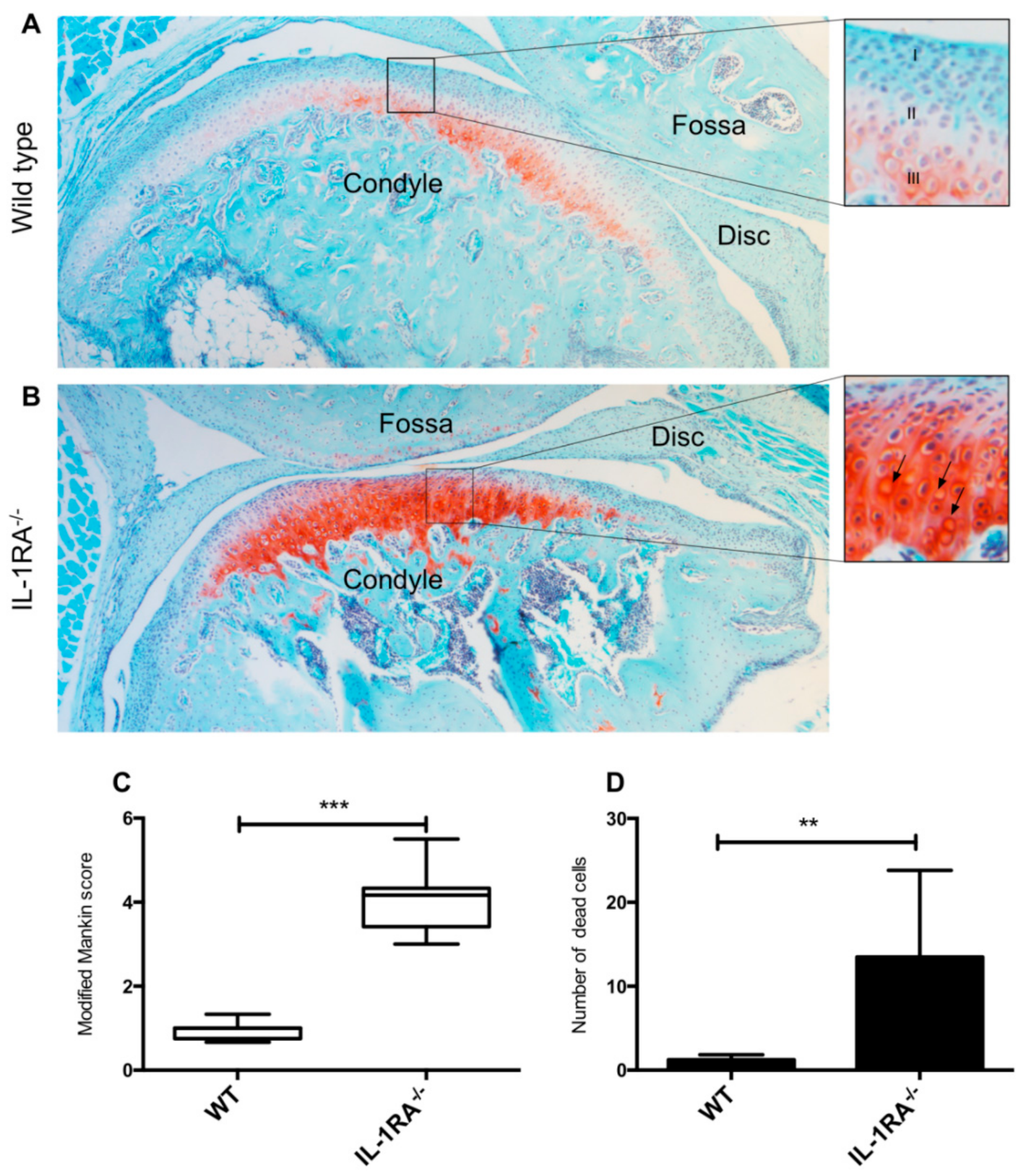
2.1. IL-1βRA−/− Mice Showed Early Signs of Condylar Cartilage Damage
2.2. Cells from the Fossa, Disc, and Condyle Expressed IL-1 Receptors
2.3. IL-1β Increased ADAMTS4 and ADAMTS5 Gene Expression
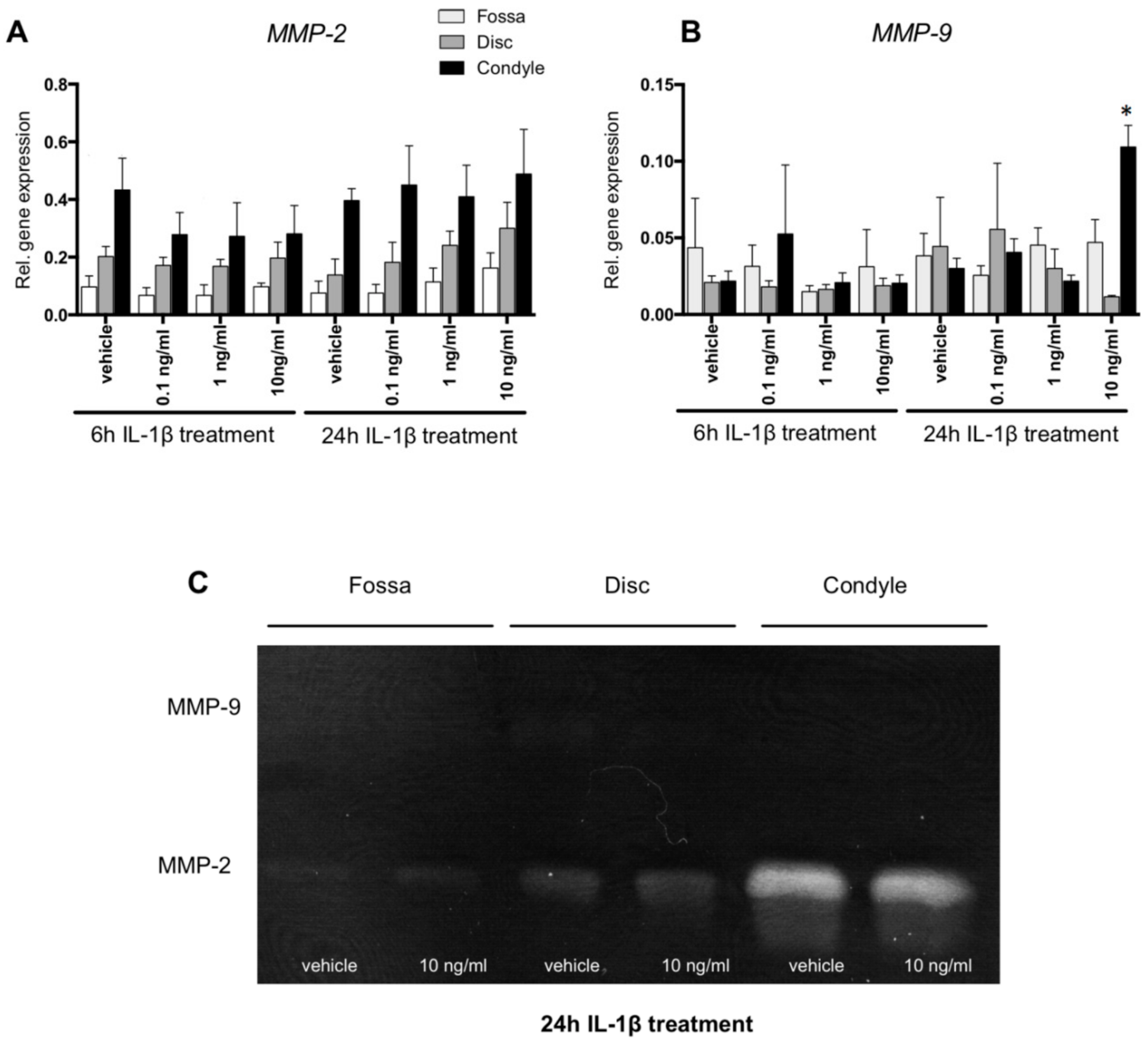
2.4. MMP-2 Activity Was Higher in Condyle Than Disc and Fossa Cells; MMP9 mRNA Upregulated in Condyle by IL-1β
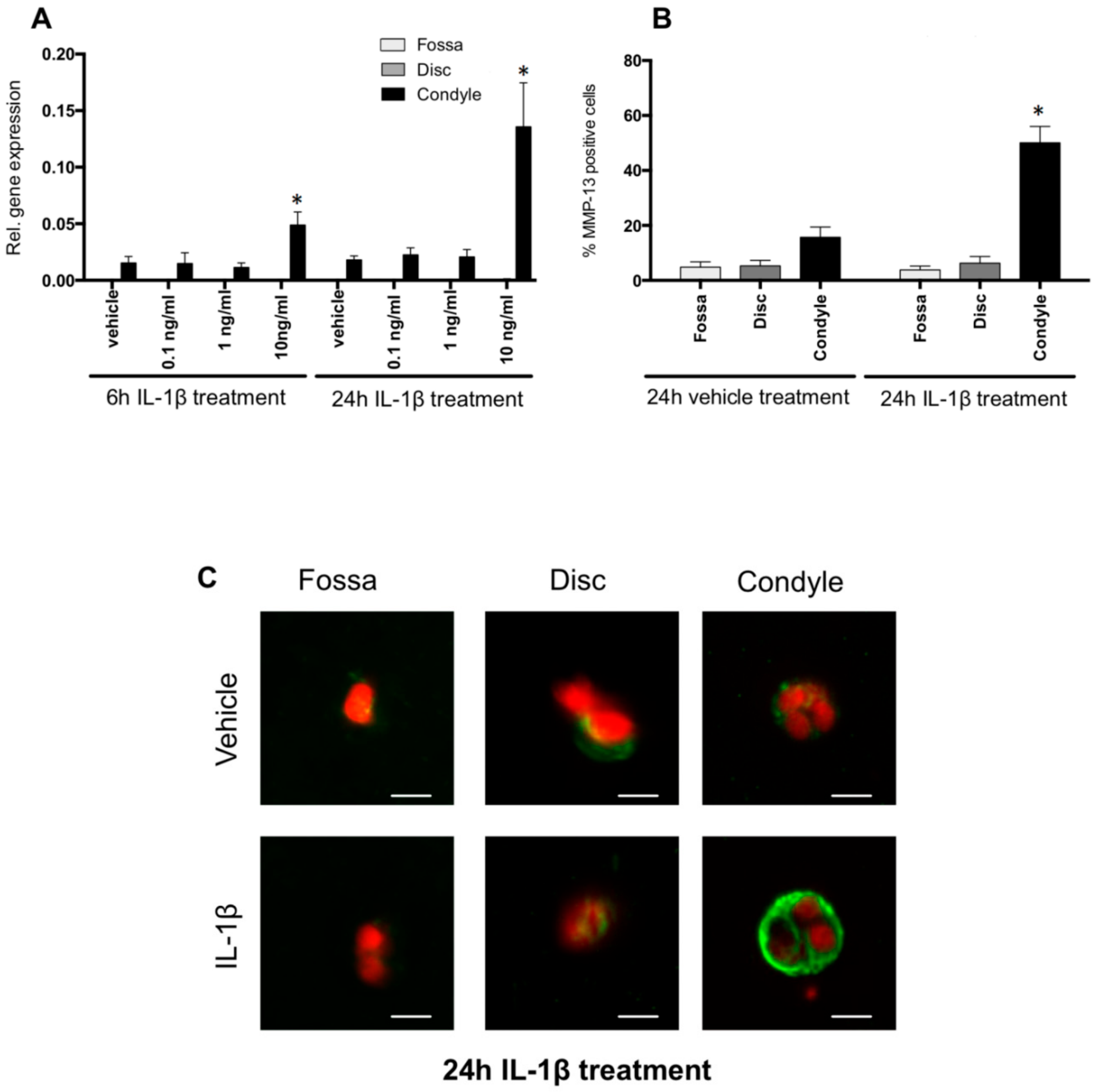
2.5. IL-1β Induced MMP-13 Expression by Condylar Cells Only
2.6. Cyclic Tensile Strain Reduced IL-1β-Induced MMP-13 Expression
3. Discussion
4. Materials and Methods
4.1. Mice
4.2. Histological Analysis of Murine TMJ
4.3. Cell Isolation and Culture
4.4. RNA Extraction and Real-Time Quantitative PCR
4.5. Zymography
4.6. Immunohistochemistry
4.7. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mercuri, L.G. Temporomandibular joint reconstruction. Alpha Omegan 2009, 102, 51–54. [Google Scholar] [CrossRef] [PubMed]
- Tabeian, H.; Bakker, A.D.; De Vries, T.J.; Zandieh-Doulabi, B.; Lobbezoo, F.; Everts, V. Juvenile porcine temporomandibular joint: Three different cartilaginous structures? Arch. Oral Boil. 2016, 72, 211–218. [Google Scholar] [CrossRef]
- Weiss, P.F.; Arabshahi, B.; Johnson, A.; Bilaniuk, L.T.; Zarnow, D.; Cahill, A.M.; Feudtner, C.; Cron, R.Q. High prevalence of temporomandibular joint arthritis at disease onset in children with juvenile idiopathic arthritis, as detected by magnetic resonance imaging but not by ultrasound. Arthritis Rheum. 2008, 58, 1189–1196. [Google Scholar]
- Hu, Y.; Billiau, A.D.; Verdonck, A.; Wouters, C.; Carels, C. Variation in dentofacial morphology and occlusion in juvenile idiopathic arthritis subjects: A case-control study. Eur. J. Orthod. 2009, 31, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Ringold, S.; Torgerson, T.R.; Egbert, M.A.; Wallace, C.A. Intraarticular corticosteroid injections of the temporomandibular joint in juvenile idiopathic arthritis. J. Rheumatol. 2008, 35, 1157–1164. [Google Scholar]
- McInnes, I.B.; Schett, G. Cytokines in the pathogenesis of rheumatoid arthritis. Nat. Rev. Immunol. 2007, 7, 429–442. [Google Scholar] [CrossRef]
- Ge, X.; Ma, X.; Meng, J.; Zhang, C.; Ma, K.; Zhou, C. Role of Wnt-5A in interleukin-1β-induced matrix metalloproteinase expression in rabbit temporomandibular joint condylar chondrocytes. Arthritis Rheum. 2009, 60, 2714–2722. [Google Scholar]
- Su, S.-C.; Tanimoto, K.; Tanne, Y.; Kunimatsu, R.; Hirose, N.; Mitsuyoshi, T.; Okamoto, Y.; Tanne, K. Celecoxib exerts protective effects on extracellular matrix metabolism of mandibular condylar chondrocytes under excessive mechanical stress. Osteoarthr. Cartil. 2014, 22, 845–851. [Google Scholar] [CrossRef]
- Pascual, V.; Allantaz, F.; Arce, E.; Punaro, M.; Banchereau, J. Role of interleukin-1 (IL-1) in the pathogenesis of systemic onset juvenile idiopathic arthritis and clinical response to IL-1 blockade. J. Exp. Med. 2005, 201, 1479–1486. [Google Scholar] [CrossRef] [PubMed]
- Kjellberg, H. Craniofacial growth in juvenile chronic arthritis. Acta Odontol. Scand. 1998, 56, 360–365. [Google Scholar] [CrossRef] [PubMed]
- Twilt, M.; Schulten, A.J.; Nicolaas, P.; Dulger, A.; van Suijlekom-Smit, L.W. Facioskeletal changes in children with juvenile idiopathic arthritis. Ann. Rheum. Dis. 2006, 65, 823–825. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.B. Mechanical loading, cartilage degradation, and arthritis. Ann. N. Y. Acad. Sci. 2010, 1211, 37–50. [Google Scholar] [CrossRef]
- Tani-Ishii, N.; Tsunoda, A.; Teranaka, T.; Umemoto, T. Autocrine regulation of osteoclast formation and bone resorption by IL-1 alpha and TNF alpha. J. Dent. 1999, 78, 1617–1623. [Google Scholar]
- Svensson, B.; Larsson, A.; Adell, R. The mandibular condyle in juvenile chronic arthritis patients with mandibular hypoplasia: A clinical and histological study. Int. J. Oral Surg. 2001, 30, 300–305. [Google Scholar] [CrossRef]
- Herring, S.W. The dynamics of mastication in pigs. Arch. Oral Boil. 1976, 21, 473–480. [Google Scholar] [CrossRef]
- Bermejo, A.; González, O.; Gonzalez, J. The pig as an animal model for experimentation on the temporomandibular articular complex. Oral Surg. Oral Med. Oral Pathol. 1993, 75, 18–23. [Google Scholar] [CrossRef]
- Wang, L.; Detamore, M.S. Effects of growth factors and glucosamine on porcine mandibular condylar cartilage cells and hyaline cartilage cells for tissue engineering applications. Arch. Oral Biol. 2009, 54, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Springer, I.N.; Fleiner, B.; Jepsen, S.; Acil, Y. Culture of cells gained from temporomandibular joint cartilage on non-absorbable scaffolds. Biomaterials 2001, 22, 2569–2577. [Google Scholar] [CrossRef]
- Vapniarsky, N.; Aryaei, A.; Arzi, B.; Hatcher, D.C.; Hu, J.C.; Athanasiou, K.A. The Yucatan Minipig Temporomandibular Joint Disc Structure–Function Relationships Support Its Suitability for Human Comparative Studies. Tissue Eng. Part. C Methods 2017, 23, 700–709. [Google Scholar] [CrossRef] [PubMed]
- Horai, R.; Tanioka, H.; Nakae, S.; Okahara, A.; Ikuse, T.; Iwakura, Y.; Saijo, S.; Sudo, K.; Asano, M. Development of Chronic Inflammatory Arthropathy Resembling Rheumatoid Arthritis in Interleukin 1 Receptor Antagonist-Deficient Mice. J. Exp. Med. 2000, 191, 313–320. [Google Scholar] [CrossRef]
- Xu, L.; Polur, I.; Lim, C.; Servais, J.M.; Dobeck, J.; Li, Y.; Olsen, B.R. Early-onset osteoarthritis of mouse temporomandibular joint induced by partial discectomy. Osteoarthr. Cartil. 2009, 17, 917–922. [Google Scholar] [CrossRef]
- Hu, K.; Xu, L.; Cao, L.; Flahiff, C.M.; Brussiau, J.; Ho, K.; Setton, L.A.; Youn, I.; Guilak, F.; Olsen, B.R.; et al. Pathogenesis of osteoarthritis-like changes in the joints of mice deficient in type IX collagen. Arthritis Rheum. 2006, 54, 2891–2900. [Google Scholar]
- Lam, N.P.; Li, Y.; Waldman, A.B.; Brussiau, J.; Lee, P.L.; Olsen, B.R.; Xu, L. Age-dependent increase of discoidin domain receptor 2 and matrix metalloproteinase 13 expression in temporomandibular joint cartilage of type IX and type XI collagen-deficient mice. Arch. Oral Biol. 2007, 52, 579–584. [Google Scholar] [CrossRef] [PubMed]
- Henry, S.P.; Liang, S.; Akdemir, K.C.; De Crombrugghe, B. The Postnatal Role of Sox9 in Cartilage. J. Bone 2012, 27, 2511–2525. [Google Scholar] [CrossRef]
- Fosang, A.J.; Last, K.; Neame, P.J.; Murphy, G.; Knäuper, V.; Tschesche, H.; Hughes, C.E.; Caterson, B.; Hardingham, T.E. Neutrophil collagenase (MMP-8) cleaves at the aggrecanase site E373–A374 in the interglobular domain of cartilage aggrecan. Biochem. J. 1994, 304, 347–351. [Google Scholar] [CrossRef] [PubMed]
- Takaishi, H.; Kimura, T.; Dalal, S.; Okada, Y.; D’Armiento, J. Joint Diseases and Matrix Metalloproteinases: A Role for MMP-13. Pharm. Biotechnol. 2008, 9, 47–54. [Google Scholar] [CrossRef]
- Aimes, R.T.; Quigley, J.P. Matrix metalloproteinase-2 is an interstitial collagenase. Inhibitor-free enzyme catalyzes the cleavage of collagen fibrils and soluble native type I collagen generating the specific 3/4- and 1/4-length fragments. J. Biol. Chem. 1995, 270, 5872–5876. [Google Scholar] [CrossRef]
- Mort, J.S.; Billington, C. Articular cartilage and changes in arthritis: Matrix degradation. Arthritis Res. 2001, 3, 337–341. [Google Scholar] [CrossRef]
- Neuhold, L.A.; Killar, L.; Zhao, W.; Sung, M.-L.A.; Warner, L.; Kulik, J.; Turner, J.; Wu, W.; Billinghurst, C.; Meijers, T.; et al. Postnatal expression in hyaline cartilage of constitutively active human collagenase-3 (MMP-13) induces osteoarthritis in mice. J. Clin. Investig. 2001, 107, 35–44. [Google Scholar] [CrossRef]
- Yoshihara, Y.; Nakamura, H.; Obata, K.; Yamada, H.; Hayakawa, T.; Fujikawa, K.; Okada, Y. Matrix metalloproteinases and tissue inhibitors of metalloproteinases in synovial fluids from patients with rheumatoid arthritis or osteoarthritis. Ann. Rheum. Dis. 2000, 59, 455–461. [Google Scholar] [CrossRef]
- Inada, M.; Wang, Y.; Byrne, M.H.; Rahman, M.U.; Miyaura, C.; López-Otín, C.; Krane, S.M. Critical roles for collagenase-3 (Mmp13) in development of growth plate cartilage and in endochondral ossification. Proc. Natl. Acad. Sci. USA 2004, 101, 17192–17197. [Google Scholar] [CrossRef]
- Cheleschi, S.; Pascarelli, N.A.; Valacchi, G.; Di Capua, A.; Biava, M.; Belmonte, G.; Giordani, A.; Sticozzi, C.; Anzini, M.; Fioravanti, A. Chondroprotective effect of three different classes of anti-inflammatory agents on human osteoarthritic chondrocytes exposed to IL-1β. Int. Immunopharmacol. 2015, 28, 794–801. [Google Scholar] [CrossRef]
- Bai, X.; Guo, A.; Li, Y. Protective effects of calcitonin on IL-1 stimulated chondrocytes by regulating MMPs/TIMP-1 ratio via suppression of p50-NF-κB pathway. Biosci. Biotechnol. Biochem. 2019, 83, 598–604. [Google Scholar] [CrossRef]
- Tabeian, H.; Bakker, A.D.; Betti, B.F.; Lobbezoo, F.; Everts, V.; de Vries, T.J. Cyclic Tensile Strain Reduces TNF-alpha Induced Expression of MMP-13 by Condylar Temporomandibular Joint Cells. J. Cell. Physiol. 2017, 232, 1287–1294. [Google Scholar] [CrossRef]
- Deschner, J.; Rath-Deschner, B.; Agarwal, S. Regulation of matrix metalloproteinase expression by dynamic tensile strain in rat fibrochondrocytes. Osteoarthr. Cartil. 2006, 14, 264–272. [Google Scholar] [CrossRef]
- Agarwal, S.; Long, P.; Gassner, R.; Piesco, N.P.; Buckley, M.J. Cyclic tensile strain suppresses catabolic effects of interleukin-1β in fibrochondrocytes from the temporomandibular joint. Arthritis. Rheum. 2001, 44, 608–617. [Google Scholar] [CrossRef]
- Singh, M.; Detamore, M.S. Tensile properties of the mandibular condylar cartilage. J. Biomech. Eng. 2008, 130, 011009. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Akyuz, U.; Xu, L.; Pidaparti, R. Stress analysis of the human temporomandibular joint. Med. Eng. Phys. 1998, 20, 565–572. [Google Scholar] [CrossRef]
- Lee, G.M.; Poole, C.A.; Kelley, S.S.; Chang, J.; Caterson, B. Isolated chondrons: A viable alternative for studies of chondrocyte metabolism in vitro. Osteoarthr. Cartil. 1997, 5, 261–274. [Google Scholar] [CrossRef]
- Vonk, L.A.; Doulabi, B.Z.; Huang, C.; Helder, M.N.; Everts, V.; Bank, R.A. Preservation of the chondrocyte’s pericellular matrix improves cell-induced cartilage formation. J. Cell. Biochem. 2010, 110, 260–271. [Google Scholar] [CrossRef]
- Bougault, C.; Paumier, A.; Aubert-Foucher, E.; Mallein-Gerin, F. Investigating conversion of mechanical force into biochemical signaling in three-dimensional chondrocyte cultures. Nat. Protoc. 2009, 4, 928–938. [Google Scholar] [CrossRef] [PubMed]


| 1) Pericellular Safranin O staining | |
|---|---|
| a. Normal | 0 |
| b. Slightly enhanced | 1 |
| c. Intensely enhanced | 2 |
| 2) Background Safranin O staining | |
| a. Normal | 0 |
| b. Slight decrease/increase | 1 |
| c. Severe decrease/increase | 2 |
| d. No staining | 3 |
| 3) Arrangement of Chondrocytes | |
| a. Normal | 0 |
| b. Appearance of clustering | 1 |
| b. Hypocellularity | 2 |
| 4) Cartilage Structure | |
| a. Normal | 0 |
| b. Fibrillation in superficial layer | 1 |
| c. Fibrillation beyond superficial layer | 2 |
| d. Missing articular cartilages | 3 |
| Fetal Bovine Serum (%) | ITS (%) | Ascorbic Acid (µg mL−1) | PSF (%) | |
|---|---|---|---|---|
| Day 0 | 10 | - | 50 | 2 |
| Day 1 | 5 | - | 50 | 2 |
| Day 2 | 1 | 1 | 50 | 1 |
| Day 4 | - | 1 | 50 | 1 |
| Day 6 | - | 1 | 50 | 1 |
| Genes | Primers | Primer Sequences a |
|---|---|---|
| YWHAZ (reference gene) | Forward: | GATGAAGCCATTGCTGAAACTTG |
| Reverse: | CTATTTGTGGGACAGCATGGA | |
| HPRT (reference gene) | Forward: | GCTGACCTGCTGGATTACAT |
| Reverse: | CTTGCGACCTTGACCATCT | |
| IL-1RI | Forward: | CATGACTGCCCATTGTTGAG |
| Reverse: | AGGGCAGAAGCCTAGGAAG | |
| IL-1RII | Forward: | GTGCCTGTTGAGCCTCATT |
| Reverse: | GGCCTTCATGGGCAAATGTCA | |
| ADAMTS4 | Forward: | CATCCTACGCCGGAAGAGTC |
| Reverse: | GGATCACTAGCCGAGTCACCA | |
| ADAMTS5 | Forward: | GTGGAGGAGGAGTCAGTTTG |
| Reverse: | TTCAGTGCCATCGGTCACCTT | |
| MMP-2 | Forward: | CCGTGGTGAGATCTTCTTCTTC |
| Reverse: | GCGGTCAGTGGCTGGGGTA | |
| MMP-9 | Forward: | ACAGGCAGCTGGCAGAGGA |
| Reverse: | GCCGGCAAGTCTTCCGAGTA | |
| MMP-13 | Forward: | GGAGCATGGCGACTTCTAC |
| Reverse: | GAGTGCTCCAGGGTCCTT |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tabeian, H.; Betti, B.F.; dos Santos Cirqueira, C.; de Vries, T.J.; Lobbezoo, F.; ter Linde, A.V.; Zandieh-Doulabi, B.; Koenders, M.I.; Everts, V.; Bakker, A.D. IL-1β Damages Fibrocartilage and Upregulates MMP-13 Expression in Fibrochondrocytes in the Condyle of the Temporomandibular Joint. Int. J. Mol. Sci. 2019, 20, 2260. https://doi.org/10.3390/ijms20092260
Tabeian H, Betti BF, dos Santos Cirqueira C, de Vries TJ, Lobbezoo F, ter Linde AV, Zandieh-Doulabi B, Koenders MI, Everts V, Bakker AD. IL-1β Damages Fibrocartilage and Upregulates MMP-13 Expression in Fibrochondrocytes in the Condyle of the Temporomandibular Joint. International Journal of Molecular Sciences. 2019; 20(9):2260. https://doi.org/10.3390/ijms20092260
Chicago/Turabian StyleTabeian, Hessam, Beatriz F. Betti, Cinthya dos Santos Cirqueira, Teun J. de Vries, Frank Lobbezoo, Anouk V. ter Linde, Behrouz Zandieh-Doulabi, Marije I. Koenders, Vincent Everts, and Astrid D. Bakker. 2019. "IL-1β Damages Fibrocartilage and Upregulates MMP-13 Expression in Fibrochondrocytes in the Condyle of the Temporomandibular Joint" International Journal of Molecular Sciences 20, no. 9: 2260. https://doi.org/10.3390/ijms20092260
APA StyleTabeian, H., Betti, B. F., dos Santos Cirqueira, C., de Vries, T. J., Lobbezoo, F., ter Linde, A. V., Zandieh-Doulabi, B., Koenders, M. I., Everts, V., & Bakker, A. D. (2019). IL-1β Damages Fibrocartilage and Upregulates MMP-13 Expression in Fibrochondrocytes in the Condyle of the Temporomandibular Joint. International Journal of Molecular Sciences, 20(9), 2260. https://doi.org/10.3390/ijms20092260







