Nanomachinery Organizing Release at Neuronal and Ribbon Synapses
Abstract
1. Introduction
2. Synaptic Vesicle Pool Organization at Synapses
3. Filament Organization in the Cytoplasm at Neuronal Compared to Ribbon Synapses
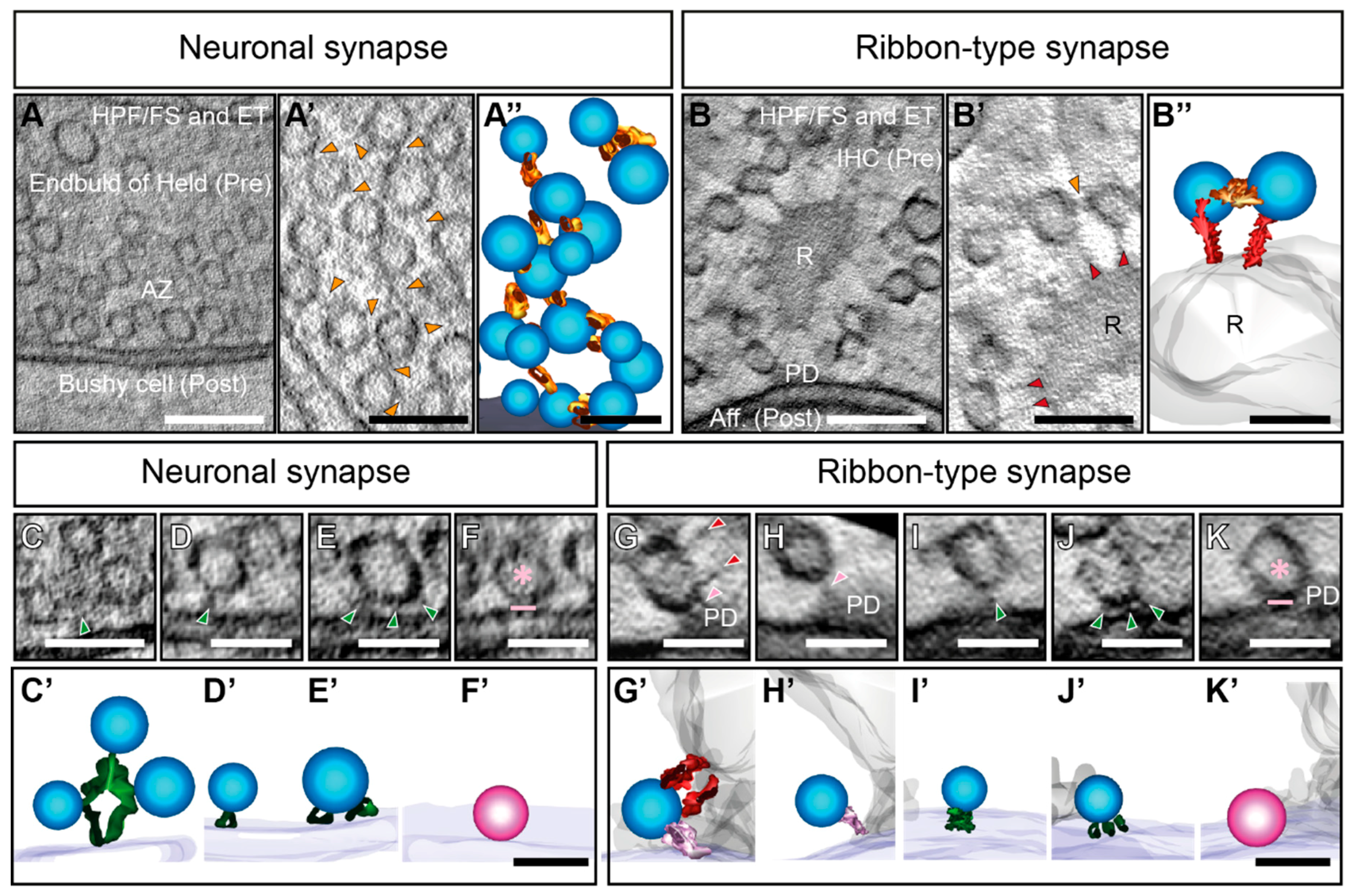
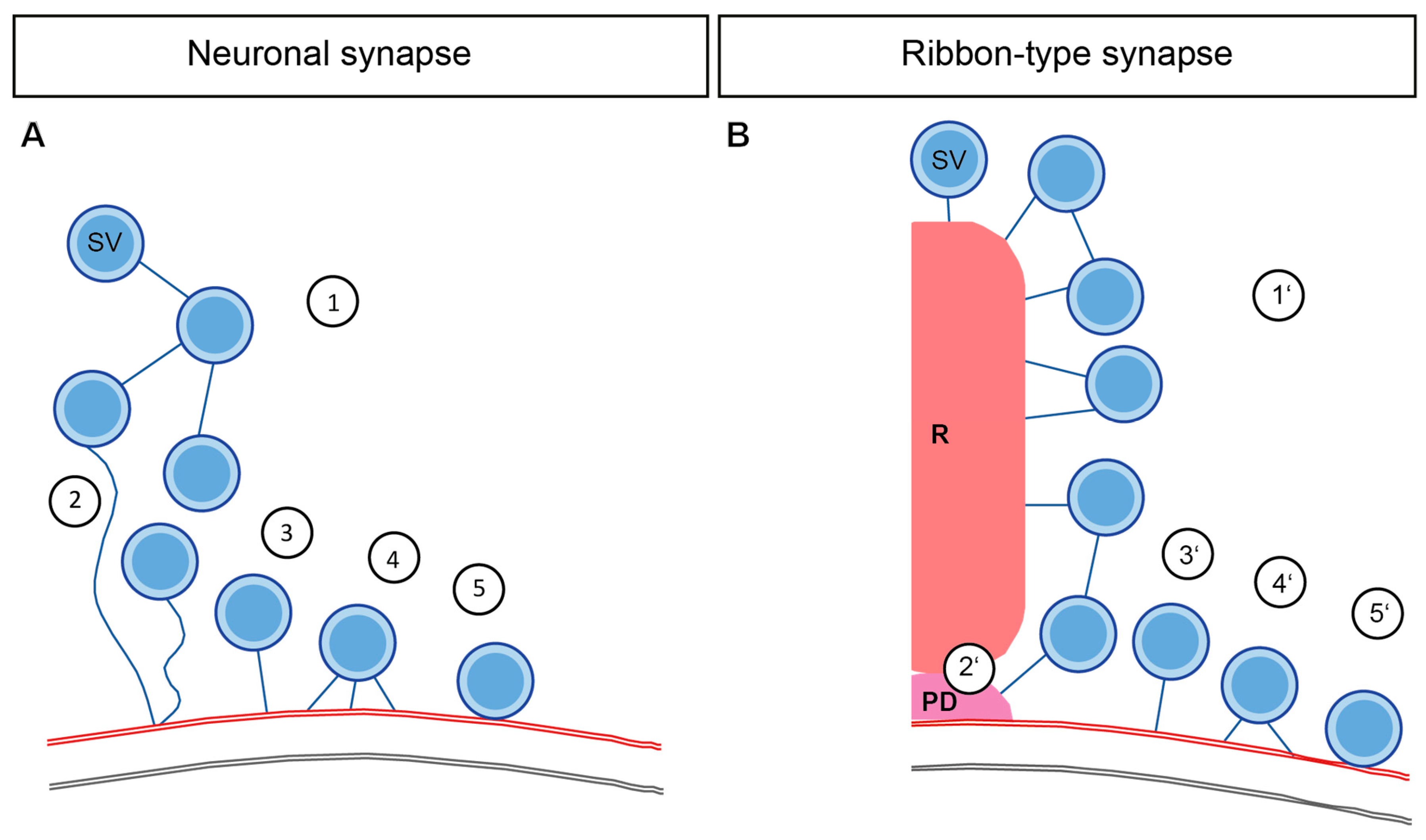
4. Interconnectors in Neuronal Synapses form a Network of SVs
5. Interconnectors and Ribbon-Attached Filaments at Ribbon Synapses Mediate SV Recruitment and Transport towards the AZ Membrane
6. Proteins Potentially Involved in Interconnector Formation of Neuronal Synapses
7. Proteins Potentially Involved in Interconnector and Ribbon-Attached Filament Formation at Ribbon Synapses
8. SV Tethering in Membrane-Proximity
9. Synaptic Vesicle Tethering at the AZ Membrane of Neuronal Synapses to Recruit SVs
10. SV Docking and Priming at Neuronal Synapses
11. Synaptic Vesicle Tethering at the AZ Membrane of Ribbon Synapses Recruit SVs
12. Potential Components of SV Tethers at IHC Ribbon Synapses
13. Synaptic Vesicle Docking at Ribbon Synapses
14. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lin, R.C.; Scheller, R.H. Mechanisms of Synaptic Vesicle Exocytosis. Annu. Rev. Cell Dev. Biol. 2000, 16, 19–49. [Google Scholar] [CrossRef]
- Südhof, T.C. Neurotransmitter release: The last millisecond in the life of a synaptic vesicle. Neuron 2013, 80, 675–690. [Google Scholar] [CrossRef]
- Weimer, R.M. Controversies in synaptic vesicle exocytosis. J. Cell Sci. 2003, 116, 3661–3666. [Google Scholar] [CrossRef]
- Emmanouilidou, E.; Melachroinou, K.; Roumeliotis, T.; Garbis, S.D.; Ntzouni, M.; Margaritis, L.H.; Stefanis, L.; Vekrellis, K. Cell-Produced α-Synuclein Is Secreted in a Calcium-Dependent Manner by Exosomes and Impacts Neuronal Survival. J. Neurosci. 2010, 30, 6838–6851. [Google Scholar] [CrossRef]
- Chivet, M.; Hemming, F.; Pernet-Gallay, K.; Fraboulet, S.; Sadoul, R. Emerging role of neuronal exosomes in the central nervous system. Front. Physiol. 2012, 3, 145. [Google Scholar] [CrossRef] [PubMed]
- Budnik, V.; Ruiz-Cañada, C.; Wendler, F. Extracellular vesicles round off communication in the nervous system. Nat. Rev. Neurosci. 2016, 17, 160–172. [Google Scholar] [CrossRef] [PubMed]
- Surgucheva, I.; Sharov, V.S.; Surguchov, A. γ-Synuclein: Seeding of α-synuclein aggregation and transmission between cells. Biochemistry 2012, 51, 4743–4754. [Google Scholar] [CrossRef]
- Dodge, F.A.; Rahamimoff, R. Co-operative action a calcium ions in transmitter release at the neuromuscular junction. J. Physiol. 1967, 193, 419–432. [Google Scholar] [CrossRef]
- Katz, B.; Miledi, R. Spontaneous and evoked activity of motor nerve endings in calcium Ringer. J. Physiol. 1969, 203, 689–706. [Google Scholar] [CrossRef] [PubMed]
- Llinás, R.; Steinberg, I.Z.; Walton, K. Relationship between presynaptic calcium current and postsynaptic potential in squid giant synapse. Biophys. J. 1981, 33, 323–351. [Google Scholar] [CrossRef]
- Südhof, T.C. Calcium control of neurotransmitter release. Cold Spring Harb. Perspect. Biol. 2012, 4, a011353. [Google Scholar] [CrossRef]
- Moser, T.; Beutner, D. Kinetics of exocytosis and endocytosis at the cochlear inner hair cell afferent synapse of the mouse. Proc. Natl. Acad. Sci. USA 2000, 97, 883–888. [Google Scholar] [CrossRef]
- Beutner, D.; Voets, T.; Neher, E.; Moser, T. Calcium dependence of exocytosis and endocytosis at the cochlear inner hair cell afferent synapse. Neuron 2001, 29, 681–690. [Google Scholar] [CrossRef]
- Cho, S.; von Gersdorff, H. Ca2+ influx and neurotransmitter release at ribbon synapses. Cell Calcium 2012, 52, 208–216. [Google Scholar] [CrossRef][Green Version]
- Castellano-Muñoz, M.; Schnee, M.E.; Ricci, A.J. Calcium-induced calcium release supports recruitment of synaptic vesicles in auditory hair cells. J. Neurophysiol. 2016, 115, 226–239. [Google Scholar] [CrossRef]
- Frank, T.; Rutherford, M.A.; Strenzke, N.; Neef, A.; Pangršič, T.; Khimich, D.; Fejtova, A.; Gundelfinger, E.D.; Liberman, M.C.; Harke, B.; et al. Bassoon and the synaptic ribbon organize Ca2+ channels and vesicles to add release sites and promote refilling. Neuron 2010, 68, 724–738. [Google Scholar] [CrossRef]
- Zhai, R.G.; Bellen, H.J. The Architecture of the Active Zone in the Presynaptic Nerve Terminal. Physiology 2004, 19, 262–270. [Google Scholar] [CrossRef]
- Südhof, T.C. The presynaptic active zone. Neuron 2012, 75, 11–25. [Google Scholar] [CrossRef]
- Szule, J.A.; Harlow, M.L.; Jung, J.H.; De-Miguel, F.F.; Marshall, R.M.; McMahan, U.J. Regulation of synaptic vesicle docking by different classes of macromolecules in active zone material. PLoS ONE 2012, 7, e33333. [Google Scholar] [CrossRef]
- Kittel, R.J.; Heckmann, M. Synaptic Vesicle Proteins and Active Zone Plasticity. Front. Synaptic Neurosci. 2016, 8, 1011–1026. [Google Scholar] [CrossRef]
- Rizo, J.; Rosenmund, C. Synaptic vesicle fusion. Nat. Struct. Mol. Biol. 2008, 15, 665–674. [Google Scholar] [CrossRef] [PubMed]
- Wichmann, C.; Moser, T. Relating structure and function of inner hair cell ribbon synapses. Cell Tissue Res. 2015, 361, 95–114. [Google Scholar] [CrossRef] [PubMed]
- Bornschein, G.; Schmidt, H. Synaptotagmin Ca2+ Sensors and Their Spatial Coupling to Presynaptic Cav Channels in Central Cortical Synapses. Front. Mol. Neurosci. 2019, 11, 494. [Google Scholar] [CrossRef] [PubMed]
- Kittel, R.J.; Wichmann, C.; Rasse, T.M.; Fouquet, W.; Schmidt, M.; Schmid, A.; Wagh, D.A.; Pawlu, C.; Kellner, R.R.; Willig, K.I.; et al. Bruchpilot promotes active zone assembly, Ca2+ channel clustering, and vesicle release. Science 2006, 312, 1051–1054. [Google Scholar] [CrossRef]
- Atwood, H.L.; Karunanithi, S.; Georgiou, J.; Charlton, M.P. Strength of synaptic transmission at neuromuscular junctions of crustaceans and insects in relation to calcium entry. Invert. Neurosci. 1997, 3, 81–87. [Google Scholar] [CrossRef]
- Parsons, T.D.; Lenzi, D.; Almers, W.; Roberts, W.M. Calcium-triggered exocytosis and endocytosis in an isolated presynaptic cell: Capacitance measurements in saccular hair cells. Neuron 1994, 13, 875–883. [Google Scholar] [CrossRef]
- Schneggenburger, R.; Han, Y.; Kochubey, O. Ca(2+) channels and transmitter release at the active zone. Cell Calcium 2012, 52, 199–207. [Google Scholar] [CrossRef]
- Shahrezaei, V.; Cao, A.; Delaney, K.R. Ca2+ from One or Two Channels Controls Fusion of a Single Vesicle at the Frog Neuromuscular Junction. J. Neurosci. 2006, 26, 13240–13249. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Das, B.; Nakamura, Y.; DiGregorio, D.A.; Young, S.M. Ca2+ channel to synaptic vesicle distance accounts for the readily releasable pool kinetics at a functionally mature auditory synapse. J. Neurosci. 2015, 35, 2083–2100. [Google Scholar] [CrossRef]
- Böhme, M.A.; Beis, C.; Reddy-Alla, S.; Reynolds, E.; Mampell, M.M.; Grasskamp, A.T.; Lützkendorf, J.; Bergeron, D.D.; Driller, J.H.; Babikir, H.; et al. Active zone scaffolds differentially accumulate Unc13 isoforms to tune Ca2+ channel–vesicle coupling. Nat. Neurosci. 2016, 19, 1311–1320. [Google Scholar] [CrossRef]
- Neef, J.; Urban, N.T.; Ohn, T.-L.; Frank, T.; Jean, P.; Hell, S.W.; Willig, K.I.; Moser, T. Quantitative optical nanophysiology of Ca 2+ signaling at inner hair cell active zones. Nat. Commun. 2018, 9, 290. [Google Scholar] [CrossRef]
- Neher, E.; Sakaba, T. Multiple roles of calcium ions in the regulation of neurotransmitter release. Neuron 2008, 59, 861–872. [Google Scholar] [CrossRef]
- Dresbach, T.; Qualmann, B.; Kessels, M.M.; Garner, C.C.; Gundelfinger, E.D. The presynaptic cytomatrix of brain synapses. Cell. Mol. Life Sci. 2001, 58, 94–116. [Google Scholar] [CrossRef]
- Bloom, F.E.; Aghajanian, G.K. Cytochemistry of synapses: Selective staining for electron microscopy. Science 1966, 154, 1575–1577. [Google Scholar] [CrossRef]
- Pfenninger, K.; Akert, K.; Moor, H.; Sandri, C. The fine structure of freeze-fractured presynaptic membranes. J. Neurocytol. 1972, 1, 129–149. [Google Scholar] [CrossRef]
- Vrensen, G.; Nunes Cardozo, J.; Müller, L.; Van Der Want, J. The presynaptic grid: A new approach. Brain Res. 1980, 184, 23–40. [Google Scholar] [CrossRef]
- Limbach, C.; Laue, M.M.; Wang, X.; Hu, B.; Thiede, N.; Hultqvist, G.; Kilimann, M.W. Molecular in situ topology of Aczonin/Piccolo and associated proteins at the mammalian neurotransmitter release site. Proc. Natl. Acad. Sci. USA 2011, 108, E392–E401. [Google Scholar] [CrossRef]
- Harlow, M.L.; Ress, D.; Stoschek, A.; Marshall, R.M.; McMahan, U.J. The architecture of active zone material at the frog’s neuromuscular junction. Nature 2001, 409, 479–484. [Google Scholar] [CrossRef]
- Jung, J.H.; Szule, J.A.; Marshall, R.M.; McMahan, U.J. Variable priming of a docked synaptic vesicle. Proc. Natl. Acad. Sci. USA 2016, 113, E1098–E1107. [Google Scholar] [CrossRef]
- Wichmann, C.; Sigrist, S.J. The active zone T-bar--a plasticity module? J. Neurogenet. 2010, 24, 133–145. [Google Scholar] [CrossRef]
- Meinertzhagen, I.A. Ultrastructure and quantification of synapses in the insect nervous system. J. Neurosci. Methods 1996, 69, 59–73. [Google Scholar] [CrossRef]
- Atwood, H.L.; Govind, C.K.; Wu, C.F. Differential ultrastructure of synaptic terminals on ventral longitudinal abdominal muscles in Drosophila larvae. J. Neurobiol. 1993, 24, 1008–1024. [Google Scholar] [CrossRef]
- Jiao, W.; Masich, S.; Franzén, O.; Shupliakov, O. Two pools of vesicles associated with the presynaptic cytosolic projection in Drosophila neuromuscular junctions. J. Struct. Biol. 2010, 172, 389–394. [Google Scholar] [CrossRef]
- Zhan, H.; Bruckner, J.; Zhang, Z.; O’Connor-Giles, K. Three-dimensional imaging of Drosophila motor synapses reveals ultrastructural organizational patterns. J. Neurogenet. 2016, 30, 237–246. [Google Scholar] [CrossRef][Green Version]
- Stigloher, C.; Zhan, H.; Zhen, M.; Richmond, J.; Bessereau, J.-L. The presynaptic dense projection of the Caenorhabditis elegans cholinergic neuromuscular junction localizes synaptic vesicles at the active zone through SYD-2/liprin and UNC-10/RIM-dependent interactions. J. Neurosci. 2011, 31, 4388–4396. [Google Scholar] [CrossRef]
- Watanabe, S.; Liu, Q.; Davis, M.W.; Hollopeter, G.; Thomas, N.; Jorgensen, N.B.; Jorgensen, E.M. Ultrafast endocytosis at Caenorhabditis elegans neuromuscular junctions. eLife 2013, 2, e00723. [Google Scholar] [CrossRef]
- Kittelmann, M. Synaptic Ultrastructure and Regulation of Synaptic Transmission in Caenorhabditis elegans. Ph.D. Thesis, Niedersächsische Staats-und Universitätsbibliothek Göttingen, Göttingen, Germany, 2012. [Google Scholar]
- Kittelmann, M.; Liewald, J.F.; Hegermann, J.; Schultheis, C.; Brauner, M.; Steuer Costa, W.; Wabnig, S.; Eimer, S.; Gottschalk, A. In vivo synaptic recovery following optogenetic hyperstimulation. Proc. Natl. Acad. Sci. USA 2013, 110, E3007–E3016. [Google Scholar] [CrossRef]
- Ackermann, F.; Waites, C.L.; Garner, C.C. Presynaptic active zones in invertebrates and vertebrates. EMBO Rep. 2015, 16, 923–938. [Google Scholar] [CrossRef]
- Bruckner, J.J.; Zhan, H.; O’Connor-Giles, K.M. Advances in imaging ultrastructure yield new insights into presynaptic biology. Front. Cell. Neurosci. 2015, 9, 196. [Google Scholar] [CrossRef]
- Kittelmann, M.; Hegermann, J.; Goncharov, A.; Taru, H.; Ellisman, M.H.; Richmond, J.E.; Jin, Y.; Eimer, S. Liprin-α/SYD-2 determines the size of dense projections in presynaptic active zones in C. elegans. J. Cell Biol. 2013, 203, 849–863. [Google Scholar] [CrossRef]
- Weimer, R.M.; Gracheva, E.O.; Meyrignac, O.; Miller, K.G.; Richmond, J.E.; Bessereau, J.-L. UNC-13 and UNC-10/rim localize synaptic vesicles to specific membrane domains. J. Neurosci. 2006, 26, 8040–8047. [Google Scholar] [CrossRef] [PubMed]
- Weimer, R.M.; Richmond, J.E.; Davis, W.S.; Hadwiger, G.; Nonet, M.L.; Jorgensen, E.M. Defects in synaptic vesicle docking in unc-18 mutants. Nat. Neurosci. 2003, 6, 1023–1030. [Google Scholar] [CrossRef]
- Graf, E.R.; Valakh, V.; Wright, C.M.; Wu, C.; Liu, Z.; Zhang, Y.Q.; DiAntonio, A. RIM Promotes Calcium Channel Accumulation at Active Zones of the Drosophila Neuromuscular Junction. J. Neurosci. 2012, 32, 16586–16596. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Sugita, S.; Südhof, T.C. The RIM/NIM Family of Neuronal C2 Domain Proteins INTERACTIONS WITH Rab3 AND A NEW CLASS OF Src HOMOLOGY 3 DOMAIN PROTEINS. J. Biol. Chem. 2000, 275, 20033–20044. [Google Scholar] [CrossRef] [PubMed]
- Bruckner, J.J.; Gratz, S.J.; Slind, J.K.; Geske, R.R.; Cummings, A.M.; Galindo, S.E.; Donohue, L.K.; O’Connor-Giles, K.M. Fife, a Drosophila Piccolo-RIM Homolog, Promotes Active Zone Organization and Neurotransmitter Release. J. Neurosci. 2012, 32, 17048–17058. [Google Scholar] [CrossRef] [PubMed]
- Bartoletti, T.M.; Jackman, S.L.; Babai, N.; Mercer, A.J.; Kramer, R.H.; Thoreson, W.B. Release from the cone ribbon synapse under bright light conditions can be controlled by the opening of only a few Ca(2+) channels. J. Neurophysiol. 2011, 106, 2922–2935. [Google Scholar] [CrossRef]
- Buran, B.N.; Strenzke, N.; Neef, A.; Gundelfinger, E.D.; Moser, T.; Liberman, M.C. Onset coding is degraded in auditory nerve fibers from mutant mice lacking synaptic ribbons. J. Neurosci. 2010, 30, 7587–7597. [Google Scholar] [CrossRef]
- Glowatzki, E.; Fuchs, P.A. Transmitter release at the hair cell ribbon synapse. Nat. Neurosci. 2002, 5, 147–154. [Google Scholar] [CrossRef]
- Goutman, J.D.; Glowatzki, E. Time course and calcium dependence of transmitter release at a single ribbon synapse. Proc. Natl. Acad. Sci. USA 2007, 104, 16341–16346. [Google Scholar] [CrossRef]
- Heidelberger, R.; Wang, M.M.; Sherry, D.M. Differential distribution of synaptotagmin immunoreactivity among synapses in the goldfish, salamander, and mouse retina. Vis. Neurosci. 2003, 20, 37–49. [Google Scholar] [CrossRef]
- Keen, E.C.; Hudspeth, A.J. Transfer characteristics of the hair cell’s afferent synapse. Proc. Natl. Acad. Sci. USA 2006, 103, 5537–5542. [Google Scholar] [CrossRef]
- Singer, J.H.; Lassová, L.; Vardi, N.; Diamond, J.S. Coordinated multivesicular release at a mammalian ribbon synapse. Nat. Neurosci. 2004, 7, 826–833. [Google Scholar] [CrossRef]
- Moser, T.; Brandt, A.; Lysakowski, A. Hair cell ribbon synapses. Cell Tissue Res. 2006, 326, 347–359. [Google Scholar] [CrossRef]
- Fuchs, P.A.; Glowatzki, E.; Moser, T. The afferent synapse of cochlear hair cells. Curr. Opin. Neurobiol. 2003, 13, 452–458. [Google Scholar] [CrossRef]
- Lysakowski, A.; Goldberg, J.M. A regional ultrastructural analysis of the cellular and synaptic architecture in the chinchilla cristae ampullares. J. Comp. Neurol. 1997, 389, 419–443. [Google Scholar] [CrossRef]
- Eatock, R.A.; Songer, J.E. Vestibular Hair Cells and Afferents: Two Channels for Head Motion Signals. Annu. Rev. Neurosci. 2011, 34, 501–534. [Google Scholar] [CrossRef]
- Goldberg, J.M.; Lysakowski, A.; Fernández, C. Morphophysiological and ultrastructural studies in the mammalian cristae ampullares. Hear. Res. 1990, 49, 89–102. [Google Scholar] [CrossRef]
- Schmitz, F.; Königstorfer, A.; Südhof, T.C. RIBEYE, a component of synaptic ribbons: A protein’s journey through evolution provides insight into synaptic ribbon function. Neuron 2000, 28, 857–872. [Google Scholar] [CrossRef]
- Morgans, C.W. Presynaptic proteins of ribbon synapses in the retina. Microsc. Res. Tech. 2000, 50, 141–150. [Google Scholar] [CrossRef]
- Heidelberger, R.; Thoreson, W.B.; Witkovsky, P. Synaptic transmission at retinal ribbon synapses. Prog. Retin. Eye Res. 2005, 24, 682–720. [Google Scholar] [CrossRef]
- Reuss, S. Pineal ribbon synapses: Regulated by the gland’s central innervation. Biog. Amines 2011, 25, 50–58. [Google Scholar]
- Kindt, K.S.; Sheets, L. Transmission Disrupted: Modeling Auditory Synaptopathy in Zebrafish. Front. Cell Dev. Biol. 2018, 6, 114. [Google Scholar] [CrossRef] [PubMed]
- Sheets, L.; Trapani, J.G.; Mo, W.; Obholzer, N.; Nicolson, T. Ribeye is required for presynaptic CaV1.3a channel localization and afferent innervation of sensory hair cells. Development 2011, 138, 1309–1319. [Google Scholar] [CrossRef]
- Bensouilah, M.; Schugardt, C.; Roesler, R.; Kirschbaum, F.; Denizot, J.-P. Larval electroreceptors in the epidermis of mormyrid fish: I. Tuberous organs of type A and B. J. Comp. Neurol. 2002, 447, 309–322. [Google Scholar] [CrossRef]
- Sejnowski, T.J.; Yodlowski, M.L. A freeze-fracture study of the skate electro receptor. J. Neurocytol. 1982, 11, 897–912. [Google Scholar] [CrossRef]
- Schnee, M.E.; Lawton, D.M.; Furness, D.N.; Benke, T.A.; Ricci, A.J. Auditory hair cell-afferent fiber synapses are specialized to operate at their best frequencies. Neuron 2005, 47, 243–254. [Google Scholar] [CrossRef]
- Tucker, T.; Fettiplace, R. Confocal imaging of calcium microdomains and calcium extrusion in turtle hair cells. Neuron 1995, 15, 1323–1335. [Google Scholar] [CrossRef]
- Raviola, E.; Gilula, N.B. Intramembrane organization of specialized contacts in the outer plexiform layer of the retina. A freeze-fracture study in monkeys and rabbits. J. Cell Biol. 1975, 65, 192–222. [Google Scholar] [CrossRef]
- Rao-Mirotznik, R.; Harkins, A.B.; Buchsbaum, G.; Sterling, P. Mammalian rod terminal: Architecture of a binary synapse. Neuron 1995, 14, 561–569. [Google Scholar] [CrossRef]
- Pangrsic, T.; Lasarow, L.; Reuter, K.; Takago, H.; Schwander, M.; Riedel, D.; Frank, T.; Tarantino, L.M.; Bailey, J.S.; Strenzke, N.; et al. Hearing requires otoferlin-dependent efficient replenishment of synaptic vesicles in hair cells. Nat. Neurosci. 2010, 13, 869–876. [Google Scholar] [CrossRef]
- Holt, M.; Cooke, A.; Neef, A.; Lagnado, L. High Mobility of Vesicles Supports Continuous Exocytosis at a Ribbon Synapse. Curr. Biol. 2004, 14, 173–183. [Google Scholar] [CrossRef] [PubMed]
- Schnee, M.E.; Santos-Sacchi, J.; Castellano-Muñoz, M.; Kong, J.-H.; Ricci, A.J. Calcium-Dependent Synaptic Vesicle Trafficking Underlies Indefatigable Release at the Hair Cell Afferent Fiber Synapse. Neuron 2011, 70, 326–338. [Google Scholar] [CrossRef]
- Nouvian, R.; Beutner, D.; Parsons, T.D.; Moser, T. Structure and function of the hair cell ribbon synapse. J. Membr. Biol. 2006, 209, 153–165. [Google Scholar] [CrossRef] [PubMed]
- tom Dieck, S.; Altrock, W.D.; Kessels, M.M.; Qualmann, B.; Regus, H.; Brauner, D.; Fejtová, A.; Bracko, O.; Gundelfinger, E.D.; Brandstätter, J.H. Molecular dissection of the photoreceptor ribbon synapse: Physical interaction of Bassoon and RIBEYE is essential for the assembly of the ribbon complex. J. Cell Biol. 2005, 168, 825–836. [Google Scholar] [CrossRef] [PubMed]
- Khimich, D.; Nouvian, R.; Pujol, R.; tom Dieck, S.; Egner, A.; Gundelfinger, E.D.; Moser, T. Hair cell synaptic ribbons are essential for synchronous auditory signalling. Nature 2005, 434, 889–894. [Google Scholar] [CrossRef] [PubMed]
- Dick, O.; tom Dieck, S.; Altrock, W.D.; Ammermüller, J.; Weiler, R.; Garner, C.C.; Gundelfinger, E.D.; Brandstätter, J.H. The Presynaptic Active Zone Protein Bassoon Is Essential for Photoreceptor Ribbon Synapse Formation in the Retina. Neuron 2003, 37, 775–786. [Google Scholar] [CrossRef]
- Jing, Z.; Rutherford, M.A.; Takago, H.; Frank, T.; Fejtova, A.; Khimich, D.; Moser, T.; Strenzke, N. Disruption of the presynaptic cytomatrix protein bassoon degrades ribbon anchorage, multiquantal release, and sound encoding at the hair cell afferent synapse. J. Neurosci. 2013, 33, 4456–4467. [Google Scholar] [CrossRef]
- Wong, A.B.; Rutherford, M.A.; Gabrielaitis, M.; Pangršič, T.; Göttfert, F.; Frank, T.; Michanski, S.; Hell, S.; Wolf, F.; Wichmann, C.; et al. Developmental refinement of hair cell synapses tightens the coupling of Ca2+ influx to exocytosis. EMBO J. 2014, 33, 247–264. [Google Scholar] [CrossRef]
- Vogl, C.; Cooper, B.H.; Neef, J.; Wojcik, S.M.; Reim, K.; Reisinger, E.; Brose, N.; Rhee, J.-S.; Moser, T.; Wichmann, C. Unconventional molecular regulation of synaptic vesicle replenishment in cochlear inner hair cells. J. Cell. Sci. 2015, 128, 638–644. [Google Scholar] [CrossRef]
- Nouvian, R.; Neef, J.; Bulankina, A.V.; Reisinger, E.; Pangršič, T.; Frank, T.; Sikorra, S.; Brose, N.; Binz, T.; Moser, T. Exocytosis at the hair cell ribbon synapse apparently operates without neuronal SNARE proteins. Nat. Neurosci. 2011, 14, 411–413. [Google Scholar] [CrossRef] [PubMed]
- Babai, N.; Sendelbeck, A.; Regus-Leidig, H.; Fuchs, M.; Mertins, J.; Reim, K.; Brose, N.; Feigenspan, A.; Brandstätter, J.H. Functional Roles of Complexin3 and Complexin4 at Mouse Photoreceptor Ribbon Synapses. J. Neurosci. 2016, 36, 6651–6667. [Google Scholar] [CrossRef] [PubMed]
- Vaithianathan, T.; Henry, D.; Akmentin, W.; Matthews, G. Functional roles of complexin in neurotransmitter release at ribbon synapses of mouse retinal bipolar neurons. J. Neurosci. 2015, 35, 4065–4070. [Google Scholar] [CrossRef] [PubMed]
- Reim, K.; Wegmeyer, H.; Brandstätter, J.H.; Xue, M.; Rosenmund, C.; Dresbach, T.; Hofmann, K.; Brose, N. Structurally and functionally unique complexins at retinal ribbon synapses. J. Cell Biol. 2005, 169, 669. [Google Scholar] [CrossRef] [PubMed]
- Reim, K.; Regus-Leidig, H.; Ammermüller, J.; El-Kordi, A.; Radyushkin, K.; Ehrenreich, H.; Brandstätter, J.H.; Brose, N. Aberrant Function and Structure of Retinal Ribbon Synapses in the Absence of Complexin 3 and Complexin 4. J. Cell Sci. 2009, 122, 1352–1361. [Google Scholar] [CrossRef] [PubMed]
- Strenzke, N.; Chanda, S.; Kopp-Scheinpflug, C.; Khimich, D.; Reim, K.; Bulankina, A.V.; Neef, A.; Wolf, F.; Brose, N.; Xu-Friedman, M.A.; et al. Complexin-I Is Required for High-Fidelity Transmission at the Endbulb of Held Auditory Synapse. J. Neurosci. 2009, 29, 7991–8004. [Google Scholar] [CrossRef]
- Roux, I.; Safieddine, S.; Nouvian, R.; Grati, M.; Simmler, M.-C.; Bahloul, A.; Perfettini, I.; Le Gall, M.; Rostaing, P.; Hamard, G.; et al. Otoferlin, defective in a human deafness form, is essential for exocytosis at the auditory ribbon synapse. Cell 2006, 127, 277–289. [Google Scholar] [CrossRef]
- Strenzke, N.; Chakrabarti, R.; Al-Moyed, H.; Muller, A.; Hoch, G.; Pangrsic, T.; Yamanbaeva, G.; Lenz, C.; Pan, K.T.; Auge, E.; et al. Hair cell synaptic dysfunction, auditory fatigue and thermal sensitivity in otoferlin Ile515Thr mutants. EMBO J. 2016, 35, 2519–2535. [Google Scholar] [CrossRef]
- Pangršič, T.; Reisinger, E.; Moser, T. Otoferlin: A multi-C2 domain protein essential for hearing. Trends Neurosci. 2012, 35, 671–680. [Google Scholar] [CrossRef]
- Hams, N.; Padmanarayana, M.; Qiu, W.; Johnson, C.P. Otoferlin is a multivalent calcium-sensitive scaffold linking SNAREs and calcium channels. Proc. Natl. Acad. Sci. USA 2017, 114, 8023–8028. [Google Scholar] [CrossRef]
- Dulon, D.; Safieddine, S.; Jones, S.M.; Petit, C. Otoferlin Is Critical for a Highly Sensitive and Linear Calcium-Dependent Exocytosis at Vestibular Hair Cell Ribbon Synapses. J. Neurosci. 2009, 29, 10474–10487. [Google Scholar] [CrossRef]
- Dunlap, K.; Luebke, J.I.; Turner, T.J. Exocytotic Ca2+ channels in mammalian central neurons. Trends Neurosci. 1995, 18, 89–98. [Google Scholar] [CrossRef]
- Mintz, I.M.; Sabatini, B.L.; Regehr, W.G. Calcium control of transmitter release at a cerebellar synapse. Neuron 1995, 15, 675–688. [Google Scholar] [CrossRef]
- Qian, J.; Noebels, J.L. Presynaptic Ca2+ channels and neurotransmitter release at the terminal of a mouse cortical neuron. J. Neurosci. 2001, 21, 3721–3728. [Google Scholar] [CrossRef]
- Brandt, A.; Khimich, D.; Moser, T. Few CaV1. 3 channels regulate the exocytosis of a synaptic vesicle at the hair cell ribbon synapse. J. Neurosci. 2005, 25, 11577. [Google Scholar] [CrossRef] [PubMed]
- Platzer, J.; Engel, J.; Schrott-Fischer, A.; Stephan, K.; Bova, S.; Chen, H.; Zheng, H.; Striessnig, J. Congenital deafness and sinoatrial node dysfunction in mice lacking class D L-type Ca2+ channels. Cell 2000, 102, 89–97. [Google Scholar] [CrossRef]
- Brandt, A.; Striessnig, J.; Moser, T. CaV1. 3 channels are essential for development and presynaptic activity of cochlear inner hair cells. J. Neurosci. 2003, 23, 10832–10840. [Google Scholar] [CrossRef]
- Pangrsic, T.; Singer, J.H.; Koschak, A. Voltage-Gated Calcium Channels: Key Players in Sensory Coding in the Retina and the Inner Ear. Physiol. Rev. 2018, 98, 2063–2096. [Google Scholar] [CrossRef] [PubMed]
- Baig, S.M.; Koschak, A.; Lieb, A.; Gebhart, M.; Dafinger, C.; Nürnberg, G.; Ali, A.; Ahmad, I.; Sinnegger-Brauns, M.J.; Brandt, N.; et al. Loss of Ca(v)1.3 (CACNA1D) function in a human channelopathy with bradycardia and congenital deafness. Nat. Neurosci. 2011, 14, 77–84. [Google Scholar] [CrossRef]
- Bech-Hansen, N.T.; Naylor, M.J.; Maybaum, T.A.; Pearce, W.G.; Koop, B.; Fishman, G.A.; Mets, M.; Musarella, M.A.; Boycott, K.M. Loss-of-function mutations in a calcium-channel alpha1-subunit gene in Xp11.23 cause incomplete X-linked congenital stationary night blindness. Nat. Genet. 1998, 19, 264–267. [Google Scholar] [CrossRef]
- Dou, H.; Vazquez, A.E.; Namkung, Y.; Chu, H.; Cardell, E.L.; Nie, L.; Parson, S.; Shin, H.-S.; Yamoah, E.N. Null mutation of alpha1D Ca2+ channel gene results in deafness but no vestibular defect in mice. J. Assoc. Res. Otolaryngol. 2004, 5, 215–226. [Google Scholar] [CrossRef] [PubMed]
- Lodha, N.; Loucks, C.M.; Beaulieu, C.; Parboosingh, J.S.; Bech-Hansen, N.T. Congenital Stationary Night Blindness: Mutation Update and Clinical Variability. In Retinal Degenerative Diseases; LaVail, M.M., Ash, J.D., Anderson, R.E., Hollyfield, J.G., Grimm, C., Eds.; Springer: Boston, MA, USA, 2012; pp. 371–379. [Google Scholar]
- Liu, X.; Kerov, V.; Haeseleer, F.; Majumder, A.; Artemyev, N.; Baker, S.A.; Lee, A. Dysregulation of Ca(v)1.4 channels disrupts the maturation of photoreceptor synaptic ribbons in congenital stationary night blindness type 2. Channels 2013, 7, 514–523. [Google Scholar] [CrossRef]
- Lenzi, D.; von Gersdorff, H. Structure suggests function: The case for synaptic ribbons as exocytotic nanomachines. Bioessays 2001, 23, 831–840. [Google Scholar] [CrossRef]
- Lenzi, D.; Crum, J.; Ellisman, M.H.; Roberts, W.M. Depolarization redistributes synaptic membrane and creates a gradient of vesicles on the synaptic body at a ribbon synapse. Neuron 2002, 36, 649–659. [Google Scholar] [CrossRef]
- Lenzi, D.; Runyeon, J.W.; Crum, J.; Ellisman, M.H.; Roberts, W.M. Synaptic Vesicle Populations in Saccular Hair Cells Reconstructed by Electron Tomography. J. Neurosci. 1999, 19, 119–132. [Google Scholar] [CrossRef]
- Graydon, C.W.; Cho, S.; Li, G.-L.; Kachar, B.; Gersdorff, H. von Sharp Ca2+ Nanodomains beneath the Ribbon Promote Highly Synchronous Multivesicular Release at Hair Cell Synapses. J. Neurosci. 2011, 31, 16637–16650. [Google Scholar] [CrossRef] [PubMed]
- Sobkowicz, H.M.; Rose, J.E.; Scott, G.E.; Slapnick, S.M. Ribbon synapses in the developing intact and cultured organ of Corti in the mouse. J. Neurosci. 1982, 2, 942–957. [Google Scholar] [CrossRef] [PubMed]
- Sobkowicz, H.M.; Rose, J.E.; Scott, G.L.; Levenick, C.V. Distribution of synaptic ribbons in the developing organ of Corti. J. Neurocytol. 1986, 15, 693–714. [Google Scholar] [CrossRef] [PubMed]
- Jean, P.; Lopez de la Morena, D.; Michanski, S.; Jaime Tobón, L.M.; Chakrabarti, R.; Picher, M.M.; Neef, J.; Jung, S.; Gültas, M.; Maxeiner, S.; et al. The synaptic ribbon is critical for sound encoding at high rates and with temporal precision. eLife 2018, 7, e29275. [Google Scholar] [CrossRef]
- Jung, S.; Oshima-Takago, T.; Chakrabarti, R.; Wong, A.B.; Jing, Z.; Yamanbaeva, G.; Picher, M.M.; Wojcik, S.M.; Göttfert, F.; Predoehl, F.; et al. Rab3-interacting molecules 2α and 2β promote the abundance of voltage-gated CaV1.3 Ca2+ channels at hair cell active zones. Proc. Natl. Acad. Sci. USA 2015, 112, E3141–E3149. [Google Scholar] [CrossRef] [PubMed]
- Chakrabarti, R.; Michanski, S.; Wichmann, C. Vesicle sub-pool organization at inner hair cell ribbon synapses. EMBO Rep. 2018, e44937. [Google Scholar] [CrossRef]
- Kroll, J.; Jaime Tobón, L.M.; Vogl, C.; Neef, J.; Kondratiuk, I.; König, M.; Strenzke, N.; Wichmann, C.; Milosevic, I.; Moser, T. Endophilin-A regulates presynaptic Ca2+ influx and synaptic vesicle recycling in auditory hair cells. EMBO J. 2019, 38. [Google Scholar] [CrossRef]
- Michanski, S.; Smaluch, K.; Steyer, A.M.; Chakrabarti, R.; Setz, C.; Oestreicher, D.; Fischer, C.; Möbius, W.; Moser, T.; Vogl, C.; et al. Mapping developmental maturation of inner hair cell ribbon synapses in the apical mouse cochlea. Proc. Natl. Acad. Sci. USA 2019, 201812029. [Google Scholar] [CrossRef] [PubMed]
- Graydon, C.W.; Cho, S.; Diamond, J.S.; Kachar, B.; von Gersdorff, H.; Grimes, W.N. Specialized Postsynaptic Morphology Enhances Neurotransmitter Dilution and High-Frequency Signaling at an Auditory Synapse. J. Neurosci. 2014, 34, 8358–8372. [Google Scholar] [CrossRef] [PubMed]
- Zampighi, G.A.; Schietroma, C.; Zampighi, L.M.; Woodruff, M.; Wright, E.M.; Brecha, N.C. Conical Tomography of a Ribbon Synapse: Structural Evidence for Vesicle Fusion. PLoS ONE 2011, 6, e16944. [Google Scholar] [CrossRef] [PubMed]
- Müller, T.M.; Gierke, K.; Joachimsthaler, A.; Sticht, H.; Izsvák, Z.; Hamra, F.K.; Fejtová, A.; Ackermann, F.; Garner, C.C.; Kremers, J.; et al. A multiple Piccolino-RIBEYE interaction supports plate-shaped synaptic ribbons in retinal neurons. J. Neurosci. 2019, 39, 2606–2619. [Google Scholar] [CrossRef] [PubMed]
- Cooper, B.; Hemmerlein, M.; Ammermüller, J.; Imig, C.; Reim, K.; Lipstein, N.; Kalla, S.; Kawabe, H.; Brose, N.; Brandstätter, J.H.; et al. Munc13-Independent Vesicle Priming at Mouse Photoreceptor Ribbon Synapses. J. Neurosci. 2012, 32, 8040–8052. [Google Scholar] [CrossRef]
- Landis, D.M.; Hall, A.K.; Weinstein, L.A.; Reese, T.S. The organization of cytoplasm at the presynaptic active zone of a central nervous system synapse. Neuron 1988, 1, 201–209. [Google Scholar] [CrossRef]
- Usukura, J.; Yamada, E. Ultrastructure of the synaptic ribbons in photoreceptor cells of Rana catesbeiana revealed by freeze-etching and freeze-substitution. Cell Tissue Res. 1987, 247, 483–488. [Google Scholar] [CrossRef]
- Hirokawa, N.; Sobue, K.; Kanda, K.; Harada, A.; Yorifuji, H. The cytoskeletal architecture of the presynaptic terminal and molecular structure of synapsin 1. J. Cell Biol. 1989, 108, 111–126. [Google Scholar] [CrossRef]
- Siksou, L.; Rostaing, P.; Lechaire, J.-P.; Boudier, T.; Ohtsuka, T.; Fejtová, A.; Kao, H.-T.; Greengard, P.; Gundelfinger, E.D.; Triller, A.; et al. Three-dimensional architecture of presynaptic terminal cytomatrix. J. Neurosci. 2007, 27, 6868–6877. [Google Scholar] [CrossRef]
- Fernández-Busnadiego, R.; Asano, S.; Oprisoreanu, A.-M.; Sakata, E.; Doengi, M.; Kochovski, Z.; Zürner, M.; Stein, V.; Schoch, S.; Baumeister, W.; et al. Cryo-electron tomography reveals a critical role of RIM1α in synaptic vesicle tethering. J. Cell Biol. 2013, 201, 725–740. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Busnadiego, R.; Zuber, B.; Maurer, U.E.; Cyrklaff, M.; Baumeister, W.; Lučić, V. Quantitative analysis of the native presynaptic cytomatrix by cryoelectron tomography. J. Cell Biol. 2010, 188, 145–156. [Google Scholar] [CrossRef] [PubMed]
- Cole, A.A.; Chen, X.; Reese, T.S. A Network of Three Types of Filaments Organizes Synaptic Vesicles for Storage, Mobilization, and Docking. J. Neurosci. 2016, 36, 3222–3230. [Google Scholar] [CrossRef] [PubMed]
- Bullen, A.; West, T.; Moores, C.; Ashmore, J.; Fleck, R.A.; MacLellan-Gibson, K.; Forge, A. Association of intracellular and synaptic organization in cochlear inner hair cells revealed by 3D electron microscopy. J. Cell Sci. 2015, 128, 2529–2540. [Google Scholar] [CrossRef]
- Helmprobst, F.; Frank, M.; Stigloher, C. Presynaptic architecture of the larval zebrafish neuromuscular junction. J. Comp. Neurol. 2015, 523, 1984–1997. [Google Scholar] [CrossRef]
- Perkins, G.A.; Jackson, D.R.; Spirou, G.A. Resolving Presynaptic Structure by Electron Tomography. Synapse 2015, 69, 268–282. [Google Scholar] [CrossRef]
- Siksou, L.; Triller, A.; Marty, S. Ultrastructural organization of presynaptic terminals. Curr. Opin. Neurobiol. 2011, 21, 261–268. [Google Scholar] [CrossRef]
- Rosenmund, C.; Stevens, C.F. The rate of aldehyde fixation of the exocytotic machinery in cultured hippocampal synapses. J. Neurosci. Methods 1997, 76, 1–5. [Google Scholar] [CrossRef]
- Smith, J.E.; Reese, T.S. Use of aldehyde fixatives to determine the rate of synaptic transmitter release. J. Exp. Biol. 1980, 89, 19–28. [Google Scholar]
- Moor, H. Theory and Practice of High Pressure Freezing. In Cryotechniques in Biological Electron Microscopy; Steinbrecht, P.D.R.A., Zierold, D.K., Eds.; Springer: Berlin/Heidelberg, Germany, 1987; pp. 175–191. ISBN 978-3-642-72817-4. [Google Scholar]
- Dubochet, J. High-pressure freezing for cryoelectron microscopy. Trends Cell Biol. 1995, 5, 366–368. [Google Scholar] [CrossRef]
- Kanno, H.; Speedy, R.J.; Angell, C.A. Supercooling of Water to −92 °C Under Pressure. Science 1975, 189, 880–881. [Google Scholar] [CrossRef] [PubMed]
- Leunissen, J.L.M.; Yi, H. Self-pressurized rapid freezing (SPRF): A novel cryofixation method for specimen preparation in electron microscopy. J. Microsc. 2009, 235, 25–35. [Google Scholar] [CrossRef]
- Feder, N.; Sidman, R.L. Methods and principles of fixation by freeze-substitution. J. Biophys. Biochem. Cytol. 1958, 4, 593–600. [Google Scholar] [CrossRef]
- Steinbrecht, R.A.; Müller, M. Freeze-Substitution and Freeze-Drying. In Cryotechniques in Biological Electron Microscopy; Steinbrecht, R.A., Zierold, K., Eds.; Springer: Berlin/Heidelberg, Germany, 1987; pp. 149–172. ISBN 978-3-642-72815-0. [Google Scholar]
- Hobot, J.A.; Villiger, W.; Escaig, J.; Maeder, M.; Ryter, A.; Kellenberger, E. Shape and fine structure of nucleoids observed on sections of ultrarapidly frozen and cryosubstituted bacteria. J. Bacteriol. 1985, 162, 960–971. [Google Scholar]
- Kellenberger, E. The potential of cryofixation and freeze substitution: Observations and theoretical considerations. J. Microsc. 1991, 161, 183–203. [Google Scholar] [CrossRef]
- Siksou, L.; Varoqueaux, F.; Pascual, O.; Triller, A.; Brose, N.; Marty, S. A common molecular basis for membrane docking and functional priming of synaptic vesicles. Eur. J. Neurosci. 2009, 30, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Imig, C.; Min, S.-W.; Krinner, S.; Arancillo, M.; Rosenmund, C.; Südhof, T.C.; Rhee, J.; Brose, N.; Cooper, B.H. The morphological and molecular nature of synaptic vesicle priming at presynaptic active zones. Neuron 2014, 84, 416–431. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, S.; Rost, B.R.; Camacho-Pérez, M.; Davis, M.W.; Söhl-Kielczynski, B.; Rosenmund, C.; Jorgensen, E.M. Ultrafast endocytosis at mouse hippocampal synapses. Nature 2013, 504, 242–247. [Google Scholar] [CrossRef]
- Rostaing, P.; Real, E.; Siksou, L.; Lechaire, J.-P.; Boudier, T.; Boeckers, T.M.; Gertler, F.; Gundelfinger, E.D.; Triller, A.; Marty, S. Analysis of synaptic ultrastructure without fixative using high-pressure freezing and tomography. Eur. J. Neurosci. 2006, 24, 3463–3474. [Google Scholar] [CrossRef]
- Imig, C.; Cooper, B.H. 3D Analysis of Synaptic Ultrastructure in Organotypic Hippocampal Slice Culture by High-Pressure Freezing and Electron Tomography. In Synapse Development: Methods and Protocols; Poulopoulos, A., Ed.; Methods in Molecular Biology; Springer: New York, NY, USA, 2017; pp. 215–231. ISBN 978-1-4939-6688-2. [Google Scholar]
- Möbius, W.; Cooper, B.; Kaufmann, W.A.; Imig, C.; Ruhwedel, T.; Snaidero, N.; Saab, A.S.; Varoqueaux, F. Chapter 20—Electron Microscopy of the Mouse Central Nervous System. In Methods in Cell Biology; Müller-Reichert, T., Ed.; Electron Microscopy of Model Systems; Academic Press: Cambridge, MA, USA, 2010; Volume 96, pp. 475–512. [Google Scholar]
- Tao, C.-L.; Liu, Y.-T.; Sun, R.; Zhang, B.; Qi, L.; Shivakoti, S.; Tian, C.-L.; Zhang, P.; Lau, P.-M.; Zhou, Z.H.; et al. Differentiation and Characterization of Excitatory and Inhibitory Synapses by Cryo-electron Tomography and Correlative Microscopy. J. Neurosci. 2018, 38, 1493–1510. [Google Scholar] [CrossRef]
- Fernández-Busnadiego, R.; Schrod, N.; Kochovski, Z.; Asano, S.; Vanhecke, D.; Baumeister, W.; Lučić, V. Insights into the molecular organization of the neuron by cryo-electron tomography. J. Electron Microsc. (Tokyo) 2011, 60, S137–S148. [Google Scholar] [CrossRef] [PubMed]
- Lucić, V.; Kossel, A.H.; Yang, T.; Bonhoeffer, T.; Baumeister, W.; Sartori, A. Multiscale imaging of neurons grown in culture: From light microscopy to cryo-electron tomography. J. Struct. Biol. 2007, 160, 146–156. [Google Scholar] [CrossRef] [PubMed]
- Katz, B.; Miledi, R. The Effect of Calcium on Acetylcholine Release from Motor Nerve Terminals. Proc. R. Soc. Lond. Ser. B Biol. Sci. 1965, 161, 496–503. [Google Scholar] [CrossRef]
- Rizzoli, S.O.; Betz, W.J. Synaptic vesicle pools. Nat. Rev. Neurosci. 2005, 6, 57–69. [Google Scholar] [CrossRef] [PubMed]
- Denker, A.; Kröhnert, K.; Rizzoli, S.O. Revisiting synaptic vesicle pool localization in the Drosophila neuromuscular junction. J. Physiol. 2009, 587, 2919–2926. [Google Scholar] [CrossRef] [PubMed]
- Denker, A.; Rizzoli, S.O. Synaptic Vesicle Pools: An Update. Front. Synaptic Neurosci. 2010, 2. [Google Scholar] [CrossRef]
- Alabi, A.A.; Tsien, R.W. Synaptic vesicle pools and dynamics. Cold Spring Harb. Perspect. Biol. 2012, 4. [Google Scholar] [CrossRef]
- Neher, E. Vesicle pools and Ca2+ microdomains: New tools for understanding their roles in neurotransmitter release. Neuron 1998, 20, 389–399. [Google Scholar] [CrossRef]
- Neher, E. Merits and Limitations of Vesicle Pool Models in View of Heterogeneous Populations of Synaptic Vesicles. Neuron 2015, 87, 1131–1142. [Google Scholar] [CrossRef]
- Stevens, C.F.; Tsujimoto, T. Estimates for the pool size of releasable quanta at a single central synapse and for the time required to refill the pool. Proc. Natl. Acad. Sci. USA 1995, 92, 846–849. [Google Scholar] [CrossRef] [PubMed]
- Rosenmund, C.; Stevens, C.F. Definition of the readily releasable pool of vesicles at hippocampal synapses. Neuron 1996, 16, 1197–1207. [Google Scholar] [CrossRef]
- Borges, S.; Gleason, E.; Turelli, M.; Wilson, M. The kinetics of quantal transmitter release from retinal amacrine cells. Proc. Natl. Acad. Sci. USA 1995, 92, 6896–6900. [Google Scholar] [CrossRef]
- Zampighi, G.A.; Zampighi, L.M.; Fain, N.; Lanzavecchia, S.; Simon, S.A.; Wright, E.M. Conical Electron Tomography of a Chemical Synapse: Vesicles Docked to the Active Zone are Hemi-Fused. Biophys J. 2006, 91, 2910–2918. [Google Scholar] [CrossRef]
- Ikeda, K.; Bekkers, J.M. Counting the number of releasable synaptic vesicles in a presynaptic terminal. Proc. Natl. Acad. Sci. USA 2009, 106, 2945–2950. [Google Scholar] [CrossRef] [PubMed]
- Kuromi, H.; Kidokoro, Y. Two synaptic vesicle pools, vesicle recruitment and replenishment of pools at the Drosophila neuromuscular junction. J. Neurocytol. 2003, 32, 551–565. [Google Scholar] [CrossRef] [PubMed]
- Von Gersdorff, H.; Sakaba, T.; Berglund, K.; Tachibana, M. Submillisecond kinetics of glutamate release from a sensory synapse. Neuron 1998, 21, 1177–1188. [Google Scholar] [CrossRef]
- Von Gersdorff, H.; Vardi, E.; Matthews, G.; Sterling, P. Evidence that vesicles on the synaptic ribbon of retinal bipolar neurons can be rapidly released. Neuron 1996, 16, 1221–1227. [Google Scholar] [CrossRef]
- Von Gersdorff, H.; Matthews, G. Dynamics of synaptic vesicle fusion and membrane retrieval in synaptic terminals. Nature 1994, 367, 735–739. [Google Scholar] [CrossRef] [PubMed]
- Zenisek, D.; Steyer, J.A.; Almers, W. Transport, capture and exocytosis of single synaptic vesicles at active zones. Nature 2000, 406, 849–854. [Google Scholar] [CrossRef] [PubMed]
- Jackman, S.L.; Choi, S.-Y.; Thoreson, W.B.; Rabl, K.; Bartoletti, T.M.; Kramer, R.H. Role of the synaptic ribbon in transmitting the cone light response. Nat. Neurosci. 2009, 12, 303. [Google Scholar] [CrossRef]
- Thoreson, W.B.; Miller, R.F. Removal of extracellular chloride suppresses transmitter release from photoreceptor terminals in the mudpuppy retina. J. Gen. Physiol. 1996, 107, 631–642. [Google Scholar] [CrossRef]
- Mennerick, S.; Matthews, G. Ultrafast Exocytosis Elicited by Calcium Current in Synaptic Terminals of Retinal Bipolar Neurons. Neuron 1996, 17, 1241–1249. [Google Scholar] [CrossRef]
- Vaithianathan, T.; Henry, D.; Akmentin, W.; Matthews, G. Nanoscale dynamics of synaptic vesicle trafficking and fusion at the presynaptic active zone. eLife 2016, 5, e13245. [Google Scholar] [CrossRef]
- Bunt, A.H. Enzymatic digestion of synaptic ribbons in amphibian retinal photoreceptors. Brain Res. 1971, 25, 571–577. [Google Scholar] [CrossRef]
- Gray, E.G.; Pease, H.L. On understanding the organisation of the retinal receptor synapses. Brain Res. 1971, 35, 1–15. [Google Scholar] [CrossRef]
- Kantardzhieva, A.; Liberman, M.C.; Sewell, W.F. Quantitative analysis of ribbons, vesicles, and cisterns at the cat inner hair cell synapse: Correlations with spontaneous rate. J. Comp. Neurol. 2013, 521, 3260–3271. [Google Scholar] [CrossRef]
- Verhage, M.; Sørensen, J.B. Vesicle Docking in Regulated Exocytosis. Traffic 2008, 9, 1414–1424. [Google Scholar] [CrossRef] [PubMed]
- Gotow, T.; Miyaguchi, K.; Hashimoto, P.H. Cytoplasmic architecture of the axon terminal: Filamentous strands specifically associated with synaptic vesicles. Neuroscience 1991, 40, 587–598. [Google Scholar] [CrossRef]
- Burette, A.C.; Lesperance, T.; Crum, J.; Martone, M.; Volkmann, N.; Ellisman, M.H.; Weinberg, R.J. Electron Tomographic Analysis of Synaptic Ultrastructure. J. Comp. Neurol. 2012, 520. [Google Scholar] [CrossRef]
- Gustafsson, J.S.; Birinyi, A.; Crum, J.; Ellisman, M.; Brodin, L.; Shupliakov, O. Ultrastructural organization of lamprey reticulospinal synapses in three dimensions. J. Comp. Neurol. 2002, 450, 167–182. [Google Scholar] [CrossRef] [PubMed]
- Perkins, G.A.; Tjong, J.; Brown, J.M.; Poquiz, P.H.; Scott, R.T.; Kolson, D.R.; Ellisman, M.H.; Spirou, G.A. The Micro-Architecture of Mitochondria at Active Zones: Electron Tomography Reveals Novel Anchoring Scaffolds and Cristae Structured for High-Rate Metabolism. J. Neurosci. 2010, 30, 1015–1026. [Google Scholar] [CrossRef]
- Wolff, G.; Hagen, C.; Grünewald, K.; Kaufmann, R. Towards correlative super-resolution fluorescence and electron cryo-microscopy. Biol. Cell 2016, 108, 245–258. [Google Scholar] [CrossRef]
- Schaffer, M.; Mahamid, J.; Engel, B.D.; Laugks, T.; Baumeister, W.; Plitzko, J.M. Optimized cryo-focused ion beam sample preparation aimed at in situ structural studies of membrane proteins. J. Struct. Biol. 2017, 197, 73–82. [Google Scholar] [CrossRef]
- Mahamid, J.; Schampers, R.; Persoon, H.; Hyman, A.A.; Baumeister, W.; Plitzko, J.M. A focused ion beam milling and lift-out approach for site-specific preparation of frozen-hydrated lamellas from multicellular organisms. J. Struct. Biol. 2015, 192, 262–269. [Google Scholar] [CrossRef]
- Arnold, J.; Mahamid, J.; Lucic, V.; de Marco, A.; Fernandez, J.-J.; Laugks, T.; Mayer, T.; Hyman, A.A.; Baumeister, W.; Plitzko, J.M. Site-Specific Cryo-focused Ion Beam Sample Preparation Guided by 3D Correlative Microscopy. Biophys. J. 2016, 110, 860–869. [Google Scholar] [CrossRef]
- Villa, E.; Schaffer, M.; Plitzko, J.M.; Baumeister, W. Opening windows into the cell: Focused-ion-beam milling for cryo-electron tomography. Curr. Opin. Struct. Biol. 2013, 23, 771–777. [Google Scholar] [CrossRef]
- Fletcher, T.; Cameron, P.; De Camilli, P.; Banker, G. The distribution of synapsin I and synaptophysin in hippocampal neurons developing in culture. J. Neurosci. 1991, 11, 1617–1626. [Google Scholar] [CrossRef]
- Takamori, S.; Holt, M.; Stenius, K.; Lemke, E.A.; Grønborg, M.; Riedel, D.; Urlaub, H.; Schenck, S.; Brügger, B.; Ringler, P. Molecular Anatomy of a Trafficking Organelle. Cell 2006, 127, 831–846. [Google Scholar] [CrossRef]
- Huttner, W.B.; Schiebler, W.; Greengard, P.; De Camilli, P. Synapsin I (protein I), a nerve terminal-specific phosphoprotein. III. Its association with synaptic vesicles studied in a highly purified synaptic vesicle preparation. J. Cell Biol. 1983, 96, 1374–1388. [Google Scholar] [CrossRef]
- Tatsuoka, H.; Reese, T.S. New structural features of synapses in the anteroventral cochlear nucleus prepared by direct freezing and freeze-substitution. J. Comp. Neurol. 1989, 290, 343–357. [Google Scholar] [CrossRef]
- Takei, Y.; Harada, A.; Takeda, S.; Kobayashi, K.; Terada, S.; Noda, T.; Takahashi, T.; Hirokawa, N. Synapsin I deficiency results in the structural change in the presynaptic terminals in the murine nervous system. J. Cell Biol. 1995, 131, 1789–1800. [Google Scholar] [CrossRef]
- Betz, W.J.; Henkel, A.W. Okadaic acid disrupts clusters of synaptic vesicles in frog motor nerve terminals. J. Cell Biol. 1994, 124, 843–854. [Google Scholar] [CrossRef]
- Milovanovic, D.; De Camilli, P. Synaptic vesicle clusters at synapses: A distinct liquid phase? Neuron 2017, 93, 995–1002. [Google Scholar] [CrossRef]
- Doussau, F.; Augustine, G.J. The actin cytoskeleton and neurotransmitter release: An overview. Biochimie 2000, 82, 353–363. [Google Scholar] [CrossRef]
- Levitan, E.S. Signaling for vesicle mobilization and synaptic plasticity. Mol. Neurobiol. 2008, 37, 39–43. [Google Scholar] [CrossRef] [PubMed]
- Shakiryanova, D.; Tully, A.; Hewes, R.S.; Deitcher, D.L.; Levitan, E.S. Activity-dependent liberation of synaptic neuropeptide vesicles. Nat. Neurosci. 2005, 8, 173–178. [Google Scholar] [CrossRef]
- Allersma, M.W.; Bittner, M.A.; Axelrod, D.; Holz, R.W. Motion matters: Secretory granule motion adjacent to the plasma membrane and exocytosis. Mol. Biol. Cell 2006, 17, 2424–2438. [Google Scholar] [CrossRef]
- Gaffield, M.A.; Rizzoli, S.O.; Betz, W.J. Mobility of synaptic vesicles in different pools in resting and stimulated frog motor nerve terminals. Neuron 2006, 51, 317–325. [Google Scholar] [CrossRef]
- Mandell, J.W.; Townes-Anderson, E.; Czernik, A.J.; Cameron, R.; Greengard, P.; De Camilli, P. Synapsins in the vertebrate retina: Absence from ribbon synapses and heterogeneous distribution among conventional synapses. Neuron 1990, 5, 19–33. [Google Scholar] [CrossRef]
- Safieddine, S.; Wenthold, R.J. SNARE complex at the ribbon synapses of cochlear hair cells: Analysis of synaptic vesicle- and synaptic membrane-associated proteins. Eur. J. Neurosci. 1999, 11, 803–812. [Google Scholar] [CrossRef]
- Guillet, M.; Sendin, G.; Bourien, J.; Puel, J.-L.; Nouvian, R. Actin Filaments Regulate Exocytosis at the Hair Cell Ribbon Synapse. J. Neurosci. 2016, 36, 649–654. [Google Scholar] [CrossRef] [PubMed]
- Vincent, P.F.; Bouleau, Y.; Petit, C.; Dulon, D. A synaptic F-actin network controls otoferlin-dependent exocytosis in auditory inner hair cells. eLife 2015, 4. [Google Scholar] [CrossRef]
- Schwarz, K.; Natarajan, S.; Kassas, N.; Vitale, N.; Schmitz, F. The Synaptic Ribbon Is a Site of Phosphatidic Acid Generation in Ribbon Synapses. J. Neurosci. 2011, 31, 15996–16011. [Google Scholar] [CrossRef] [PubMed]
- Schmitz, F. The Making of Synaptic Ribbons: How They Are Built and What They Do. Neurosci. 2009, 15, 611–624. [Google Scholar] [CrossRef]
- Schwarz, K.; Schmitz, F. RIBEYE(B)-domain binds to lipid components of synaptic vesicles in an NAD(H)-dependent, redox-sensitive manner. Biochem. J. 2017, 474, 1205–1220. [Google Scholar] [CrossRef]
- Maxeiner, S.; Luo, F.; Tan, A.; Schmitz, F.; Südhof, T.C. How to make a synaptic ribbon: RIBEYE deletion abolishes ribbons in retinal synapses and disrupts neurotransmitter release. EMBO J. 2016, 35, 1098–1114. [Google Scholar] [CrossRef] [PubMed]
- Becker, L.; Schnee, M.E.; Niwa, M.; Sun, W.; Maxeiner, S.; Talaei, S.; Kachar, B.; Rutherford, M.A.; Ricci, A.J. The presynaptic ribbon maintains vesicle populations at the hair cell afferent fiber synapse. eLife Sci. 2018, 7, e30241. [Google Scholar] [CrossRef] [PubMed]
- Lv, C.; Stewart, W.J.; Akanyeti, O.; Frederick, C.; Zhu, J.; Santos-Sacchi, J.; Sheets, L.; Liao, J.C.; Zenisek, D. Synaptic Ribbons Require Ribeye for Electron Density, Proper Synaptic Localization, and Recruitment of Calcium Channels. Cell Rep. 2016, 15, 2784–2795. [Google Scholar] [CrossRef] [PubMed]
- Muresan, V.; Lyass, A.; Schnapp, B.J. The Kinesin Motor KIF3A Is a Component of the Presynaptic Ribbon in Vertebrate Photoreceptors. J. Neurosci. 1999, 19, 1027–1037. [Google Scholar] [CrossRef] [PubMed]
- Uthaiah, R.C.; Hudspeth, A.J. Molecular Anatomy of the Hair Cell’s Ribbon Synapse. J. Neurosci. 2010, 30, 12387–12399. [Google Scholar] [CrossRef] [PubMed]
- Roux, I.; Hosie, S.; Johnson, S.L.; Bahloul, A.; Cayet, N.; Nouaille, S.; Kros, C.J.; Petit, C.; Safieddine, S. Myosin VI is required for the proper maturation and function of inner hair cell ribbon synapses. Hum. Mol. Genet. 2009, 18, 4615–4628. [Google Scholar] [CrossRef] [PubMed]
- Regus-Leidig, H.; Fuchs, M.; Löhner, M.; Leist, S.R.; Leal-Ortiz, S.; Chiodo, V.A.; Hauswirth, W.W.; Garner, C.C.; Brandstätter, J.H. In vivo knockdown of Piccolino disrupts presynaptic ribbon morphology in mouse photoreceptor synapses. Front. Cell. Neurosci. 2014, 8, 259. [Google Scholar] [CrossRef]
- Regus-Leidig, H.; Ott, C.; Löhner, M.; Atorf, J.; Fuchs, M.; Sedmak, T.; Kremers, J.; Fejtová, A.; Gundelfinger, E.D.; Brandstätter, J.H. Identification and Immunocytochemical Characterization of Piccolino, a Novel Piccolo Splice Variant Selectively Expressed at Sensory Ribbon Synapses of the Eye and Ear. PLoS ONE 2013, 8, e70373. [Google Scholar] [CrossRef]
- Schoch, S.; Castillo, P.E.; Jo, T.; Mukherjee, K.; Geppert, M.; Wang, Y.; Schmitz, F.; Malenka, R.C.; Südhof, T.C. RIM1alpha forms a protein scaffold for regulating neurotransmitter release at the active zone. Nature 2002, 415, 321–326. [Google Scholar] [CrossRef] [PubMed]
- Schoch, S.; Mittelstaedt, T.; Kaeser, P.S.; Padgett, D.; Feldmann, N.; Chevaleyre, V.; Castillo, P.E.; Hammer, R.E.; Han, W.; Schmitz, F.; et al. Redundant functions of RIM1[alpha] and RIM2[alpha] in Ca2+-triggered neurotransmitter release. EMBO J. 2006, 25, 5852–5863. [Google Scholar] [CrossRef] [PubMed]
- Kaeser, P.S.; Kwon, H.-B.; Chiu, C.Q.; Deng, L.; Castillo, P.E.; Südhof, T.C. RIM1α and RIM1β Are Synthesized from Distinct Promoters of the RIM1 Gene to Mediate Differential But Overlapping Synaptic Functions. J. Neurosci. 2008, 28, 13435–13447. [Google Scholar] [CrossRef]
- Deng, L.; Kaeser, P.S.; Xu, W.; Südhof, T.C. RIM Proteins Activate Vesicle Priming by Reversing Autoinhibitory Homodimerization of Munc13. Neuron 2011, 69, 317–331. [Google Scholar] [CrossRef]
- Li, W.; Ma, C.; Guan, R.; Xu, Y.; Tomchick, D.R.; Rizo, J. The crystal structure of a Munc13 C-terminal module exhibits a remarkable similarity to vesicle tethering factors. Structure 2011, 19, 1443–1455. [Google Scholar] [CrossRef]
- Altrock, W.D.; tom Dieck, S.; Sokolov, M.; Meyer, A.C.; Sigler, A.; Brakebusch, C.; Fässler, R.; Richter, K.; Boeckers, T.M.; Potschka, H.; et al. Functional inactivation of a fraction of excitatory synapses in mice deficient for the active zone protein bassoon. Neuron 2003, 37, 787–800. [Google Scholar] [CrossRef]
- Mukherjee, K.; Yang, X.; Gerber, S.H.; Kwon, H.-B.; Ho, A.; Castillo, P.E.; Liu, X.; Südhof, T.C. Piccolo and bassoon maintain synaptic vesicle clustering without directly participating in vesicle exocytosis. Proc. Natl. Acad. Sci. USA 2010, 107, 6504–6509. [Google Scholar] [CrossRef]
- Waites, C.L.; Leal-Ortiz, S.A.; Okerlund, N.; Dalke, H.; Fejtova, A.; Altrock, W.D.; Gundelfinger, E.D.; Garner, C.C. Bassoon and Piccolo maintain synapse integrity by regulating protein ubiquitination and degradation. EMBO J. 2013, 32, 954–969. [Google Scholar] [CrossRef]
- Gundelfinger, E.D.; Reissner, C.; Garner, C.C. Role of Bassoon and Piccolo in Assembly and Molecular Organization of the Active Zone. Front. Synaptic Neurosci. 2015, 7, 19. [Google Scholar] [CrossRef]
- Mendoza Schulz, A.; Jing, Z.; Sánchez Caro, J.M.; Wetzel, F.; Dresbach, T.; Strenzke, N.; Wichmann, C.; Moser, T. Bassoon-disruption slows vesicle replenishment and induces homeostatic plasticity at a CNS synapse. EMBO J. 2014, 33, 512–527. [Google Scholar] [CrossRef] [PubMed]
- Butola, T.; Wichmann, C.; Moser, T. Piccolo promotes vesicle replenishment at a fast central auditory synapse. Front. Synaptic Neurosci. 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- Stanley, E.F. Single calcium channels and acetylcholine release at a presynaptic nerve terminal. Neuron 1993, 11, 1007–1011. [Google Scholar] [CrossRef]
- Stanley, E.F. The calcium channel and the organization of the presynaptic transmitter release face. Trends Neurosci. 1997, 20, 404–409. [Google Scholar] [CrossRef]
- Stanley, E.F. The Nanophysiology of Fast Transmitter Release. Trends Neurosci. 2016, 39, 183–197. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.H.C.; Li, Q.; Snidal, C.A.; Gardezi, S.R.; Stanley, E.F. The Calcium Channel C-Terminal and Synaptic Vesicle Tethering: Analysis by Immuno-Nanogold Localization. Front. Cell. Neurosci. 2017, 11. [Google Scholar] [CrossRef]
- Wong, F.K.; Nath, A.R.; Chen, R.H.C.; Gardezi, S.R.; Li, Q.; Stanley, E.F. Synaptic vesicle tethering and the CaV2.2 distal C-terminal. Front. Cell. Neurosci. 2014, 8, 71. [Google Scholar] [CrossRef]
- Sutton, R.B.; Fasshauer, D.; Jahn, R.; Brunger, A.T. Crystal structure of a SNARE complex involved in synaptic exocytosis at 2.4 A resolution. Nature 1998, 395, 347–353. [Google Scholar] [CrossRef]
- Martens, S.; McMahon, H.T. Mechanisms of membrane fusion: Disparate players and common principles. Nat. Rev. Mol. Cell Biol. 2008, 9, 543–556. [Google Scholar] [CrossRef]
- Südhof, T.C.; Rothman, J.E. Membrane Fusion: Grappling with SNARE and SM Proteins. Science 2009, 323, 474–477. [Google Scholar] [CrossRef] [PubMed]
- Risselada, H.J.; Kutzner, C.; Grubmüller, H. Caught in the Act: Visualization of SNARE-Mediated Fusion Events in Molecular Detail. ChemBioChem 2011, 12, 1049–1055. [Google Scholar] [CrossRef] [PubMed]
- Jahn, R.; Fasshauer, D. Molecular machines governing exocytosis of synaptic vesicles. Nature 2012, 490, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Lindau, M. Synaptotagmin function illuminated. J. Gen. Physiol. 2003, 122, 251–253. [Google Scholar] [CrossRef]
- Fox, M.A.; Sanes, J.R. Synaptotagmin I and II are present in distinct subsets of central synapses. J. Comp. Neurol. 2007, 503, 280–296. [Google Scholar] [CrossRef]
- Fernández-Chacón, R.; Königstorfer, A.; Gerber, S.H.; García, J.; Matos, M.F.; Stevens, C.F.; Brose, N.; Rizo, J.; Rosenmund, C.; Südhof, T.C. Synaptotagmin I functions as a calcium regulator of release probability. Nature 2001, 410, 41–49. [Google Scholar] [CrossRef]
- Yoshihara, M.; Littleton, J.T. Synaptotagmin I Functions as a Calcium Sensor to Synchronize Neurotransmitter Release. Neuron 2002, 36, 897–908. [Google Scholar] [CrossRef]
- Nishiki, T.; Augustine, G.J. Synaptotagmin I synchronizes transmitter release in mouse hippocampal neurons. J. Neurosci. 2004, 24, 6127–6132. [Google Scholar] [CrossRef]
- Koh, T.-W.; Bellen, H.J. Synaptotagmin I, a Ca2+ sensor for neurotransmitter release. Trends Neurosci. 2003, 26, 413–422. [Google Scholar] [CrossRef]
- Geppert, M.; Goda, Y.; Hammer, R.E.; Li, C.; Rosahl, T.W.; Stevens, C.F.; Südhof, T.C. Synaptotagmin I: A major Ca2+ sensor for transmitter release at a central synapse. Cell 1994, 79, 717–727. [Google Scholar] [CrossRef]
- Südhof, T.C. Synaptotagmins: Why So Many? J. Biol. Chem. 2002, 277, 7629–7632. [Google Scholar] [CrossRef]
- Tucker, W.C.; Chapman, E.R. Role of synaptotagmin in Ca2+-triggered exocytosis. Biochem. J. 2002, 366, 1–13. [Google Scholar] [CrossRef]
- Lee, H.-K.; Yang, Y.; Su, Z.; Hyeon, C.; Lee, T.-S.; Lee, H.-W.; Kweon, D.-H.; Shin, Y.-K.; Yoon, T.-Y. Dynamic Ca2+-Dependent Stimulation of Vesicle Fusion by Membrane-Anchored Synaptotagmin 1. Science 2010, 328, 760–763. [Google Scholar] [CrossRef]
- Weber, T.; Zemelman, B.V.; McNew, J.A.; Westermann, B.; Gmachl, M.; Parlati, F.; Söllner, T.H.; Rothman, J.E. SNAREpins: Minimal Machinery for Membrane Fusion. Cell 1998, 92, 759–772. [Google Scholar] [CrossRef]
- Wickner, W.; Schekman, R. Membrane fusion. Nat. Struct. Mol. Biol. 2008, 15, 658–664. [Google Scholar] [CrossRef]
- Wickner, W. Membrane fusion: Five lipids, four SNAREs, three chaperones, two nucleotides, and a Rab, all dancing in a ring on yeast vacuoles. Annu. Rev. Cell Dev. Biol. 2010, 26, 115–136. [Google Scholar] [CrossRef]
- Ramakrishnan, N.A.; Drescher, M.J.; Drescher, D.G. The SNARE complex in neuronal and sensory cells. Mol. Cell. Neurosci. 2012, 50, 58–69. [Google Scholar] [CrossRef]
- Hohl, T.M.; Parlati, F.; Wimmer, C.; Rothman, J.E.; Söllner, T.H.; Engelhardt, H. Arrangement of Subunits in 20 S Particles Consisting of NSF, SNAPs, and SNARE Complexes. Mol. Cell 1998, 2, 539–548. [Google Scholar] [CrossRef]
- Zhao, M.; Brunger, A.T. Recent Advances in Deciphering the Structure and Molecular Mechanism of the AAA+ ATPase N-Ethylmaleimide-Sensitive Factor (NSF). J. Mol. Biol. 2016, 428, 1912–1926. [Google Scholar] [CrossRef]
- Zhou, Q.; Lai, Y.; Bacaj, T.; Zhao, M.; Lyubimov, A.Y.; Uervirojnangkoorn, M.; Zeldin, O.B.; Brewster, A.S.; Sauter, N.K.; Cohen, A.E.; et al. Architecture of the Synaptotagmin-SNARE Machinery for Neuronal Exocytosis. Nature 2015, 525, 62–67. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.; Huang, X.; Sun, S.; Li, X.; Wang, H.-W.; Sui, S.-F. Cryo-EM structure of SNAP-SNARE assembly in 20S particle. Cell Res. 2015, 25, 551–560. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.; Zhou, P.; Wang, A.L.; Wu, D.; Zhao, M.; Südhof, T.C.; Brunger, A.T. The primed SNARE-complexin-synaptotagmin complex for neuronal exocytosis. Nature 2017, 548, 420–425. [Google Scholar] [CrossRef] [PubMed]
- Brose, N.; Hofmann, K.; Hata, Y.; Südhof, T.C. Mammalian Homologues of Caenorhabditis elegans unc-13 Gene Define Novel Family of C-domain Proteins. J. Biol. Chem. 1995, 270, 25273. [Google Scholar] [CrossRef] [PubMed]
- Varoqueaux, F.; Sons, M.S.; Plomp, J.J.; Brose, N. Aberrant morphology and residual transmitter release at the Munc13-deficient mouse neuromuscular synapse. Mol. Cell. Biol. 2005, 25, 5973–5984. [Google Scholar] [CrossRef] [PubMed]
- Lipstein, N.; Sakaba, T.; Cooper, B.H.; Lin, K.-H.; Strenzke, N.; Ashery, U.; Rhee, J.-S.; Taschenberger, H.; Neher, E.; Brose, N. Dynamic control of synaptic vesicle replenishment and short-term plasticity by Ca(2+)-calmodulin-Munc13-1 signaling. Neuron 2013, 79, 82–96. [Google Scholar] [CrossRef]
- Lai, Y.; Choi, U.B.; Leitz, J.; Rhee, H.J.; Lee, C.; Altas, B.; Zhao, M.; Pfuetzner, R.A.; Wang, A.L.; Brose, N.; et al. Molecular mechanisms of synaptic vesicle priming by Munc13 and Munc18. Neuron 2017, 95, 591–607.e10. [Google Scholar] [CrossRef]
- Quade, B.; Camacho, M.; Zhao, X.; Orlando, M.; Trimbuch, T.; Xu, J.; Li, W.; Nicastro, D.; Rosenmund, C.; Rizo, J. Membrane bridging by Munc13-1 is crucial for neurotransmitter release. eLife 2019, 8, e42806. [Google Scholar] [CrossRef]
- Ma, C.; Su, L.; Seven, A.B.; Xu, Y.; Rizo, J. Reconstitution of the Vital Functions of Munc18 and Munc13 in Neurotransmitter Release. Science 2013, 339, 421–425. [Google Scholar] [CrossRef]
- Dulubova, I.; Khvotchev, M.; Liu, S.; Huryeva, I.; Südhof, T.C.; Rizo, J. Munc18-1 binds directly to the neuronal SNARE complex. Proc. Natl. Acad. Sci. USA 2007, 104, 2697–2702. [Google Scholar] [CrossRef]
- Dulubova, I.; Lou, X.; Lu, J.; Huryeva, I.; Alam, A.; Schneggenburger, R.; Südhof, T.C.; Rizo, J. A Munc13/RIM/Rab3 tripartite complex: From priming to plasticity? EMBO J. 2005, 24, 2839–2850. [Google Scholar] [CrossRef]
- Han, Y.; Babai, N.; Kaeser, P.; Südhof, T.C.; Schneggenburger, R. RIM1 and RIM2 redundantly determine Ca2+ channel density and readily releasable pool size at a large hindbrain synapse. J. Neurophysiol. 2015, 113, 255–263. [Google Scholar] [CrossRef]
- Betz, A.; Thakur, P.; Junge, H.J.; Ashery, U.; Rhee, J.-S.; Scheuss, V.; Rosenmund, C.; Rettig, J.; Brose, N. Functional Interaction of the Active Zone Proteins Munc13-1 and RIM1 in Synaptic Vesicle Priming. Neuron 2001, 30, 183–196. [Google Scholar] [CrossRef]
- Kaeser, P.S. Pushing synaptic vesicles over the RIM. Cell. Logist. 2011, 1, 106–110. [Google Scholar] [CrossRef]
- Wang, Y.; Südhof, T.C. Genomic definition of RIM proteins: Evolutionary amplification of a family of synaptic regulatory proteins. Genomics 2003, 81, 126–137. [Google Scholar] [CrossRef]
- Kaeser, P.; Südhof, T. RIM function in short- and long-term synaptic plasticity. 2005, 33, 1345–1349. Biochem. Soc. Trans. 2005, 33, 1345–1349. [Google Scholar] [CrossRef] [PubMed]
- Kaeser, P.S.; Deng, L.; Fan, M.; Südhof, T.C. RIM genes differentially contribute to organizing presynaptic release sites. Proc. Natl. Acad. Sci. USA 2012, 109, 11830–11835. [Google Scholar] [CrossRef] [PubMed]
- Wong, M.Y.; Liu, C.; Wang, S.S.H.; Roquas, A.C.F.; Fowler, S.C.; Kaeser, P.S. Liprin-α3 controls vesicle docking and exocytosis at the active zone of hippocampal synapses. Proc. Natl. Acad. Sci. USA 2018, 115, 2234–2239. [Google Scholar] [CrossRef]
- Richmond, J.E.; Davis, W.S.; Jorgensen, E.M. UNC-13 is required for synaptic vesicle fusion in C. elegans. Nat. Neurosci. 1999, 2, 959–964. [Google Scholar] [CrossRef]
- Broadie, K.; Prokop, A.; Bellen, H.J.; O’Kane, C.J.; Schulze, K.L.; Sweeney, S.T. Syntaxin and synaptobrevin function downstream of vesicle docking in drosophila. Neuron 1995, 15, 663–673. [Google Scholar] [CrossRef]
- Hess, S.D.; Doroshenko, P.A.; Augustine, G.J. A functional role for GTP-binding proteins in synaptic vesicle cycling. Science 1993, 259, 1169–1172. [Google Scholar] [CrossRef]
- Schikorski, T.; Stevens, C.F. Quantitative ultrastructural analysis of hippocampal excitatory synapses. J. Neurosci. 1997, 17, 5858–5867. [Google Scholar] [CrossRef]
- Stein, A.; Weber, G.; Wahl, M.C.; Jahn, R. Helical extension of the neuronal SNARE complex into the membrane. Nature 2009, 460, 525–528. [Google Scholar] [CrossRef]
- Varoqueaux, F.; Sigler, A.; Rhee, J.-S.; Brose, N.; Enk, C.; Reim, K.; Rosenmund, C. Total arrest of spontaneous and evoked synaptic transmission but normal synaptogenesis in the absence of Munc13-mediated vesicle priming. Proc. Natl. Acad. Sci. USA 2002, 99, 9037–9042. [Google Scholar] [CrossRef]
- Jockusch, W.J.; Speidel, D.; Sigler, A.; Sørensen, J.B.; Varoqueaux, F.; Rhee, J.-S.; Brose, N. CAPS-1 and CAPS-2 Are Essential Synaptic Vesicle Priming Proteins. Cell 2007, 131, 796–808. [Google Scholar] [CrossRef]
- Schoch, S.; Deák, F.; Königstorfer, A.; Mozhayeva, M.; Sara, Y.; Südhof, T.C.; Kavalali, E.T. SNARE function analyzed in synaptobrevin/VAMP knockout mice. Science 2001, 294, 1117–1122. [Google Scholar] [CrossRef]
- Bronk, P.; Deák, F.; Wilson, M.C.; Liu, X.; Südhof, T.C.; Kavalali, E.T. Differential Effects of SNAP-25 Deletion on Ca2+ -Dependent and Ca2+ -Independent Neurotransmission. J. Neurophysiol. 2007, 98, 794–806. [Google Scholar] [CrossRef]
- Arancillo, M.; Min, S.-W.; Gerber, S.; Münster-Wandowski, A.; Wu, Y.-J.; Herman, M.; Trimbuch, T.; Rah, J.-C.; Ahnert-Hilger, G.; Riedel, D.; et al. Titration of Syntaxin1 in mammalian synapses reveals multiple roles in vesicle docking, priming, and release probability. J. Neurosci. 2013, 33, 16698–16714. [Google Scholar] [CrossRef]
- Löhner, M.; Babai, N.; Müller, T.; Gierke, K.; Atorf, J.; Joachimsthaler, A.; Peukert, A.; Martens, H.; Feigenspan, A.; Kremers, J.; et al. Analysis of RIM Expression and Function at Mouse Photoreceptor Ribbon Synapses. J. Neurosci. 2017, 37, 7848–7863. [Google Scholar] [CrossRef]
- Rutherford, M.A.; Roberts, W.M. Frequency selectivity of synaptic exocytosis in frog saccular hair cells. Proc. Natl. Acad. Sci. USA 2006, 103, 2898. [Google Scholar] [CrossRef]
- Ullrich, B.; Südhof, T.C. Distribution of synaptic markers in the retina: Implications for synaptic vesicle traffic in ribbon synapses. J. Physiol. Paris 1994, 88, 249–257. [Google Scholar] [CrossRef]
- Hu, K.; Carroll, J.; Rickman, C.; Davletov, B. Action of Complexin on SNARE Complex. J. Biol. Chem. 2002, 277, 41652–41656. [Google Scholar] [CrossRef] [PubMed]
- Ishizuka, T.; Saisu, H.; Odani, S.; Abe, T. Synaphin: A Protein Associated with the Docking/Fusion Complex in Presynaptic Terminals. Biochem. Biophys. Res. Commun. 1995, 213, 1107–1114. [Google Scholar] [CrossRef]
- Martin, J.A.; Hu, Z.; Fenz, K.M.; Fernandez, J.; Dittman, J.S. Complexin has opposite effects on two modes of synaptic vesicle fusion. Curr. Biol. 2011, 21, 97–105. [Google Scholar] [CrossRef]
- Cao, P.; Yang, X.; Südhof, T.C. Complexin Activates Exocytosis of Distinct Secretory Vesicles Controlled by Different Synaptotagmins. J. Neurosci. 2013, 33, 1714–1727. [Google Scholar] [CrossRef] [PubMed]
- Scholz, N.; Ehmann, N.; Sachidanandan, D.; Imig, C.; Cooper, B.H.; Jahn, O.; Reim, K.; Brose, N.; Meyer, J.; Lamberty, M.; et al. Complexin cooperates with Bruchpilot to tether synaptic vesicles to the active zone cytomatrix. J. Cell Biol. 2019, 218, 1011–1026. [Google Scholar] [CrossRef] [PubMed]
- Thoreson, W.B.; Rabl, K.; Townes-Anderson, E.; Heidelberger, R. A highly Ca2+-sensitive pool of vesicles contributes to linearity at the rod photoreceptor ribbon synapse. Neuron 2004, 42, 595–605. [Google Scholar] [CrossRef]
- Singer, J.H.; Diamond, J.S. Vesicle Depletion and Synaptic Depression at a Mammalian Ribbon Synapse. J. Neurophysiol. 2006, 95, 3191–3198. [Google Scholar] [CrossRef]
- Johnson, S.L.; Marcotti, W.; Kros, C.J. Increase in efficiency and reduction in Ca2+ dependence of exocytosis during development of mouse inner hair cells. J. Physiol. 2005, 563, 177–191. [Google Scholar] [CrossRef] [PubMed]
- Vincent, P.F.Y.; Bouleau, Y.; Charpentier, G.; Emptoz, A.; Safieddine, S.; Petit, C.; Dulon, D. Different CaV1.3 Channel Isoforms Control Distinct Components of the Synaptic Vesicle Cycle in Auditory Inner Hair Cells. J. Neurosci. 2017, 37, 2960–2975. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.-H.; Moser, T. Ca2+ Regulates the Kinetics of Synaptic Vesicle Fusion at the Afferent Inner Hair Cell Synapse. Front. Cell. Neurosci. 2018, 12, 364. [Google Scholar] [CrossRef]
- Beurg, M.; Michalski, N.; Safieddine, S.; Bouleau, Y.; Schneggenburger, R.; Chapman, E.R.; Petit, C.; Dulon, D. Control of exocytosis by synaptotagmins and otoferlin in auditory hair cells. J. Neurosci. 2010, 30, 13281–13290. [Google Scholar] [CrossRef]
- Reisinger, E.; Bresee, C.; Neef, J.; Nair, R.; Reuter, K.; Bulankina, A.; Nouvian, R.; Koch, M.; Bückers, J.; Kastrup, L.; et al. Probing the functional equivalence of otoferlin and synaptotagmin 1 in exocytosis. J. Neurosci. 2011, 31, 4886–4895. [Google Scholar] [CrossRef] [PubMed]
- Krinner, S.; Butola, T.; Jung, S.; Wichmann, C.; Moser, T. RIM-binding protein 2 promotes a large number of CaV1.3 Ca2+-channels and contributes to fast synaptic vesicle replenishment at hair cell active zones. Front. Cell. Neurosci. 2017, 11, 334. [Google Scholar] [CrossRef] [PubMed]

| Neuronal Synapse | Ribbon Synapse | ||
|---|---|---|---|
| Step 1 | Synapsins [131,132,134,135,196] | Step 1′ | RIBEYE-Piccolino [127] |
| F-Actin [132,200,201,202,203,204] | RIBEYE [69,210,211] | ||
| Myosin6 [217] | |||
| F-Actin [200,207,208] | |||
| Step 2 | Bassoon and Piccolo [226] | Step 2′ | Bassoon [16,120,122] |
| F-Actin [132,200,201,202,203,204] | |||
| Liprinα3 [274] | |||
| Step 3 | RIM1α [133] Liprinα3 [274] | Step 3′ | RIM2α [121,285] |
| Complexin3 (excluding IHC ribbon synapses) [292] | |||
| RIM-BP2 [300] | |||
| Step 4 | RIM1α [133] | Step 4′ | Otoferlin (only in HC ribbon synapses) [90] |
| Munc13 [133,224] | |||
| Liprinα3 [274] | |||
| Cav2.2 channels [234,235] | |||
| Step 5 | Munc13 [133,150,151,224] | Step 5′ | Otoferlin (only in HC ribbon synapses) [90,97] |
| Liprinα3 [274] | |||
| Neuronal SNAREs [135,151] | |||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chakrabarti, R.; Wichmann, C. Nanomachinery Organizing Release at Neuronal and Ribbon Synapses. Int. J. Mol. Sci. 2019, 20, 2147. https://doi.org/10.3390/ijms20092147
Chakrabarti R, Wichmann C. Nanomachinery Organizing Release at Neuronal and Ribbon Synapses. International Journal of Molecular Sciences. 2019; 20(9):2147. https://doi.org/10.3390/ijms20092147
Chicago/Turabian StyleChakrabarti, Rituparna, and Carolin Wichmann. 2019. "Nanomachinery Organizing Release at Neuronal and Ribbon Synapses" International Journal of Molecular Sciences 20, no. 9: 2147. https://doi.org/10.3390/ijms20092147
APA StyleChakrabarti, R., & Wichmann, C. (2019). Nanomachinery Organizing Release at Neuronal and Ribbon Synapses. International Journal of Molecular Sciences, 20(9), 2147. https://doi.org/10.3390/ijms20092147





