Magnesium and Drugs
Abstract
1. Introduction
2. Magnesium and Drugs
3. Influencing Factors
3.1. Age
3.2. pH Value
3.3. Diet, Lifestyle
3.4. Magnesium Transporters
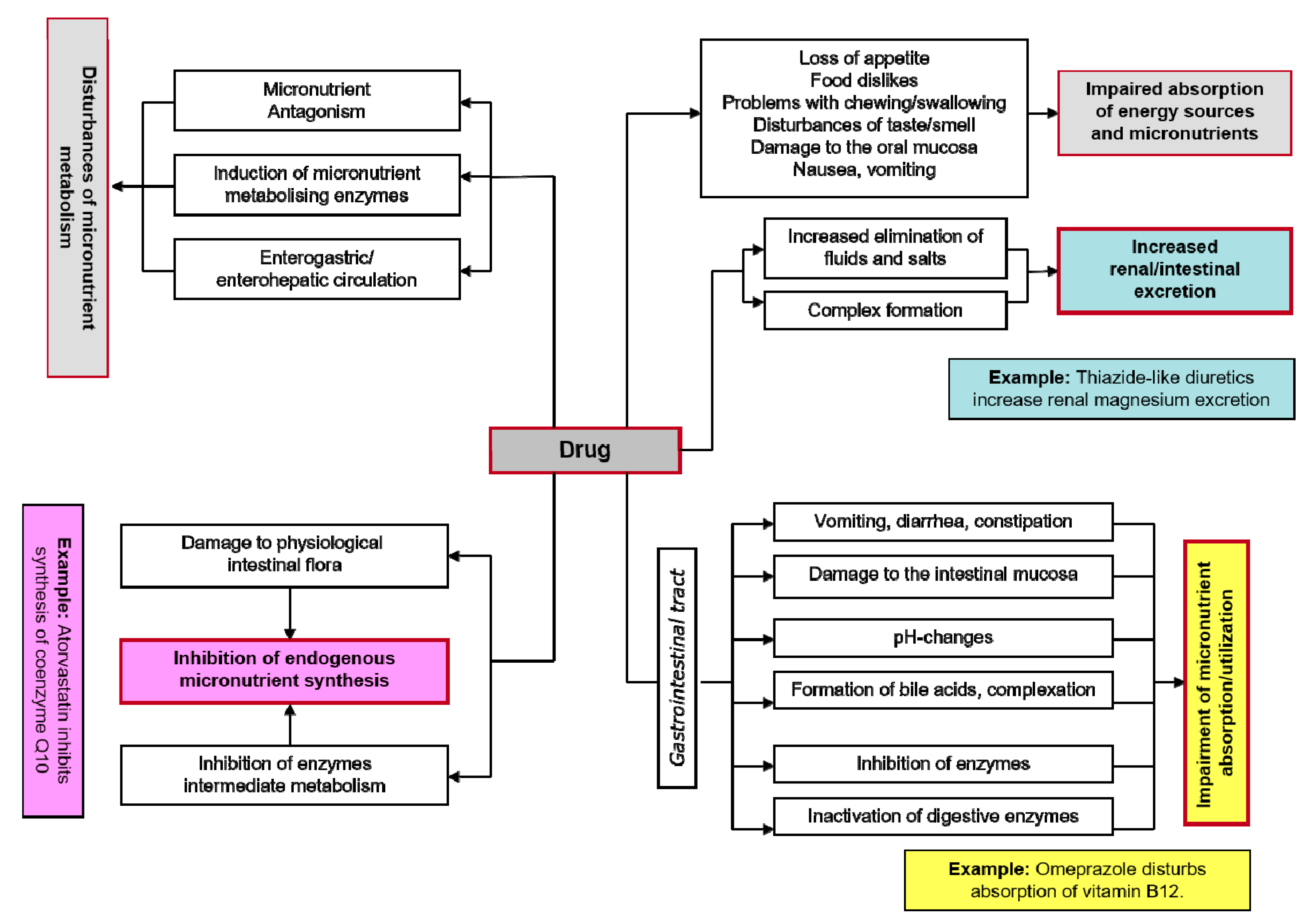
4. Drug-Induced Magnesium Deficiency
4.1. Proton-Pump Inhibitors (PPIs)
4.1.1. PPIs and Magnesium
4.1.2. Diagnostic of Magnesium Deficiency
4.1.3. Recommendations for Clinical Practice
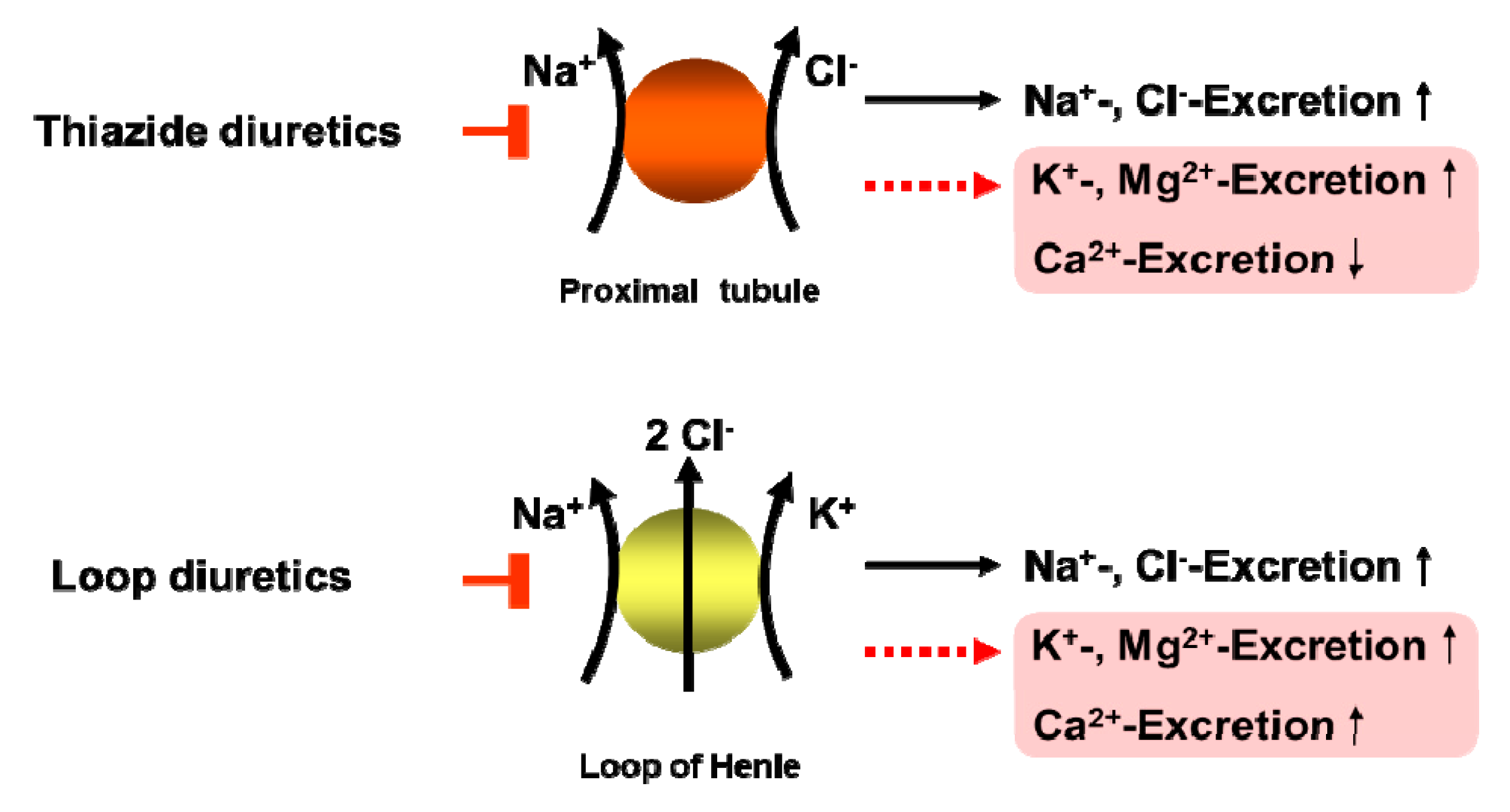
4.2. Thiazide Diuretics
4.2.1. TD and Magnesium
4.2.2. Recommendation for Clinical Practice
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- The 2018 Ageing Report 2018: Underlying Assumptions and Projection Methodologies; Institutional Paper 065; European Commission: Brussels, Belgium, November 2017; p. 3. Available online: https://ec.europa.eu/info/sites/info/files/economy-finance/ip065_en.pdf (accessed on 20 April 2019).
- Hall, M.; Dondo, T.B.; Yan, A.T.; Mamas, M.A.; Timmis, A.D.; Deanfield, J.E.; Jernberg, T.; Hemingway, H.; Fox, K.A.A.; Gale, C.P. Multimorbidity and survival for patients with acute myocardial infarction in England and Wales: Latent class analysis of a nationwide population-based cohort. PLoS Med. 2018, 15, e1002501. [Google Scholar] [CrossRef] [PubMed]
- Barnett, K.; Mercer, S.W.; Norbury, M.; Watt, G.; Wyke, S.; Guthrie, B. Epidemiology of multimorbidity and implications for health care, research, and medical education: A cross-sectional study. Lancet 2012, 380, 37–43. [Google Scholar] [CrossRef]
- Puth, M.T.; Klaschik, M.; Schmid, M.; Weckbecker, K.; Münster, E. Prevalence and comorbidity of osteoporosis- a cross-sectional analysis on 10,660 adults aged 50 years and older in Germany. BMC Musculoskelet. Disord. 2018, 19, 144. [Google Scholar] [CrossRef]
- Hofer-Dückelmann, C. Gender and polypharmacotherapy in the elderly: A clinical challenge. Handb. Exp. Pharmacol. 2012, 214, 169–182. [Google Scholar]
- Harugeri, A.; Joseph, J.; Parthasarathi, G.; Ramesh, M.; Guido, S. Prescribing patterns and predictors of high-level polypharmacy in the elderly population: A prospective surveillance study from two teaching hospitals in India. Am. J. Geriatr. Pharmacother. 2010, 8, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Dwyer, L.L.; Han, B.; Woodwell, D.A.; Rechtsteiner, E.A. Polypharmacy in nursing home residents in the United States: Results of the 2004 National Nursing Home Survey. Am. J. Geriatr. Pharmacother. 2010, 8, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Smithburger, P.L.; Buckley, M.S.; Culver, M.A.; Sokol, S.; Lat, I.; Handler, S.M.; Kirisci, L.; Kane-Gill, S.L. A Multicenter Evaluation of Off-Label Medication Use and Associated Adverse Drug Reactions in Adult Medical ICUs. Crit. Care. Med. 2015, 43, 1612–1621. [Google Scholar] [CrossRef]
- Gröber, U. Interactions between drugs and micronutrients. Med. Monatsschr. Pharm. 2006, 29, 26–35. [Google Scholar] [CrossRef] [PubMed]
- Gröber, U.; Kisters, K.; Schmidt, J. Important drug-micronutrient interactions: A selection for clinical practice. Crit. Rev. Food Sci. Nutr. 2018, 23, 1–19. [Google Scholar] [CrossRef]
- Lazarou, J.; Pomeranz, B.H.; Corey, P.N. Incidence of adverse drug reactions in hospitalized patients: A meta-analysis of prospective studies. JAMA 1998, 279, 1200–1205. [Google Scholar] [CrossRef]
- Kvasz, M.; Allen, I.E.; Gordon, M.J.; Ro, E.Y.; Estok, R.; Olkin, I.; Ross, S.D. Adverse drug reactions in hospitalized patients: A critique of a meta-analysis. Med. Gen. Med. 2000, 2, E3. [Google Scholar]
- Miguel, A.; Azevedo, L.F.; Araújo, M.; Pereira, A.C. Frequency of adverse drug reactions in hospitalized patients: A systematic review and meta-analysis. Pharmacoepidemiol. Drug Saf. 2012, 21, 1139–1154. [Google Scholar]
- Brion, M.; Lambs, L.; Berthon, G. Metal ion-tetracycline interactions in biological fluids. Part 5. Formation of zinc complexes with tetracycline and some of its derivatives and assessment of their biological significance. Agents Actions 1985, 17, 229–242. [Google Scholar] [CrossRef]
- Shea, B.; Swinden, M.V.; Ghogomu, E.T.; Ortiz, Z.; Katchamart, W.; Rader, T.; Bombardier, C.; Wells, G.A.; Tugwell, P. Folic acid and folinic acid for reducing side effects in patients receiving methotrexate for rheumatoid arthritis. J. Rheumatol. 2014, 41, 1049–1060. [Google Scholar] [CrossRef]
- Strolin Benedetti, M.; Baltes, E.L. Drug metabolism and disposition in children. Fundam. Clin. Pharmacol. 2003, 17, 281–299. [Google Scholar] [CrossRef]
- McCarthy, J. Drug metabolism and disposition in pediatric and gerontological stages of life. In Modern Pharmacology with Clinical Applications; Craig, C.R., Stitzel, R.E., Eds.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2004; pp. 56–62. [Google Scholar]
- Turner, M.A.; Catapano, M.; Hirschfeld, S.; Giaquinto, C. Global Research in Paediatrics, Paediatric drug development: The impact of evolving regulations. Adv. Drug Deliv. Rev. 2014, 73, 2–13. [Google Scholar] [CrossRef]
- Freedberg, D.E.; Lebwohl, B.; Abrams, J.A. The impact of proton pump inhibitors on the human gastrointestinal microbiome. Clin. Lab. Med. 2014, 34, 771–785. [Google Scholar] [CrossRef]
- Green, L.K.; Grajam, D.Y. Gastritis in the elderly. Gastroenterol. Clin. North Am. 1990, 19, 273–292. [Google Scholar] [PubMed]
- Sipponen, P.; Maaroos, H.I. Chronic gastritis. Scand. J. Gastroenterol. 2015, 50, 657–667. [Google Scholar] [CrossRef]
- Öztürk, N.; Kurt, N.; Özgeris, F.B.; Baygutalp, N.K.; Tosun, M.S.; Bakan, N.; Bakan, E. Serum Zinc, Copper, Magnesium and Selenium Levels in Children with Helicobacter Pylori Infection. Eurasian J. Med. 2015, 47, 126–129. [Google Scholar] [CrossRef] [PubMed]
- Workinger, J.L.; Doyle, R.P.; Bortz, J. Challenges in the Diagnosis of Magnesium Status. Nutrients 2018, 10, 1202. [Google Scholar] [CrossRef]
- Wang, S.; McDonnell, E.H.; Sedor, F.A.; Toffaletti, J.G. pH effects on measurements of ionized calcium and ionized magnesium in blood. Arch. Pathol. Lab. Med. 2002, 126, 947–950. [Google Scholar]
- Carnauba, R.A.; Baptistella, A.B.; Paschoal, V.; Hübscher, G.H. Diet-Induced Low-Grade Metabolic Acidosis and Clinical Outcomes: A Review. Nutrients 2017, 9, 538. [Google Scholar] [CrossRef]
- Razzaque, M.S. Magnesium: Are We Consuming Enough? Nutrients 2018, 10, 1863. [Google Scholar] [CrossRef]
- de Baaij, J.H.; Hoenderop, J.G.; Bindels, R.J. Magnesium in man: Implications for health and disease. Physiol. Rev. 2015, 95, 1–46. [Google Scholar] [CrossRef]
- Gröber, U.; Schmidt, J.; Kisters, K. Magnesium in Prevention and Therapy. Nutrients 2015, 7, 8199–8226. [Google Scholar] [CrossRef]
- Kolisek, M.; Launay, P.; Beck, A.; Sponder, G.; Serafini, N.; Brenkus, M.; Froschauer, E.M.; Martens, H.A.; Schweigel, M. SLC41A1 is a novel mammalian Mg2+ carrier. J. Biol. Chem. 2008, 283, 16235–16247. [Google Scholar] [CrossRef]
- Ward, K.M.; Niemela, J.E.; Cecco, S.A.; Rehak, N.N.; Elin, R.J.; Balaban, R.S. Determination of the Apparent Dissociation Constant of Mg-ATP (at T=37 °C, pH=7.2, I=0.15) Using a Combination of 31P NMR Spectroscopy and the Fluorescence Indicator Mag-fura-2. Available online: https://cds.ismrm.org/ismrm-1998/PDF6/p1693.pdf (accessed on 20 April 2019).
- Toh, J.W.; Ong, E.; Wilson, R. Hypomagnesaemia associated with long-term use of proton pump inhibitors. Gastroenterol. Rep. 2015, 3, 243–253. [Google Scholar] [CrossRef]
- de Baaij, J.H.; Arjona, F.J.; van den Brand, M.; Lavrijsen, M.; Lameris, A.L.; Bindels, R.J.; Hoenderop, J.G. Identification of SLC41A3 as a novel player in magnesium homeostasis. Sci. Rep. 2016, 6, 28565. [Google Scholar] [CrossRef]
- Rodríguez-Ramírez, M.; Rodríguez-Morán, M.; Reyes-Romero, M.A.; Guerrero-Romero, F. Effect of oral magnesium supplementation on the transcription of TRPM6, TRPM7, and SLC41A1 in individuals newly diagnosed of pre-hypertension. A randomized, double-blind, placebo-controlled trial. Magnes. Res. 2017, 30, 80–87. [Google Scholar]
- Kisters, K.; Gröber, U. Magnesium and thiazide diuretics. Magnes. Res. 2019. [Google Scholar] [CrossRef]
- Mastrototaro, L.; Smorodchenko, A.; Aschenbach, J.R.; Kolisek, M.; Sponder, G. Solute carrier 41A3 encodes for a mitochondrial Mg2+ efflux system. Sci. Rep. 2016, 6, 27999. [Google Scholar] [CrossRef]
- Ledeganck, K.J.; Anné, C.; De Monie, A.; Meybosch, S.; Verpooten, G.A.; Vinckx, M.; Van Hoeck, K.; Van Eyck, A.; De Winter, B.Y.; Trouet, D. Longitudinal Study of the Role of Epidermal Growth Factor on the Fractional Excretion of Magnesium in Children: Effect of Calcineurin Inhibitors. Nutrients 2018, 10, 677. [Google Scholar] [CrossRef]
- Mastrototaro, L.; Tietjen, U.; Sponder, G.; Vormann, J.; Aschenbach, J.R.; Kolisek, M. Insulin Modulates the Na+/Mg2+ Exchanger SLC41A1 and Influences Mg2+ Efflux from Intracellular Stores in Transgenic HEK293 Cells. J. Nutr. 2015, 145, 2440–2447. [Google Scholar] [CrossRef] [PubMed]
- Palmer, B.F.; Clegg, D.J. Electrolyte and acid-base disturbances in patients with diabetes mellitus. N. Engl. J. Med. 2015, 373, 548–559. [Google Scholar] [CrossRef] [PubMed]
- Kolisek, M.; Montezano, A.C.; Sponder, G.; Anagnostopoulou, A.; Vormann, J.; Touyz, R.M.; Aschenbach, J.R. PARK7/DJ-1 dysregulation by oxidative stress leads to magnesium deficiency: Implications in degenerative and chronic diseases. Clin. Sci. 2015, 129, 1143–1150. [Google Scholar] [CrossRef] [PubMed]
- Classen, H.G.; Gröber, U.; Kisters, K. Drug-induced magnesium deficiency. Med. Monatsschr. Pharm. 2012, 35, 274–280. [Google Scholar] [PubMed]
- Parsons, C.; Johnston, S.; Mathie, E.; Baron, N.; Machen, I.; Amador, S.; Goodman, C. Potentially inappropriate prescribing in older people with dementia in care homes: A retrospective analysis. Drugs Aging 2012, 29, 143–155. [Google Scholar] [CrossRef]
- Zhang, X.; Zhou, S.; Pan, K.; Li, X.; Zhao, X.; Zhou, Y.; Cui, Y.; Liu, X. Potentially inappropriate medications in hospitalized older patients: A cross-sectional study using the Beers 2015 criteria versus the 2012 criteria. Clin. Interv. Aging 2017, 12, 1697–1703. [Google Scholar] [CrossRef]
- Boucherie, Q.; Rouby, F.; Frankel, D.; Roll, P.; Micallef, J. Proton pump inhibitors prescriptions in France: Main trends from 2006 to 2016 on French health insurance database. Therapie 2018, 73, 385–388. [Google Scholar] [CrossRef]
- Ito, T.; Jensen, R.T. Association of long-term proton pump inhibitor therapy with bone fractures and effects on absorption of calcium, vitamin B12, iron, and magnesium. Curr. Gastroenterol. Rep. 2010, 12, 448–457. [Google Scholar] [CrossRef] [PubMed]
- Anast, C.S.; Winnacker, J.L.; Forte, L.R.; Burns, T.W. Impaired release of parathyroidhormone in magnesium deficiency. J. Clin. Endocrinol. Metab. 1976, 42, 707–717. [Google Scholar] [CrossRef] [PubMed]
- Zofková, I.; Kancheva, R.L. The relationship between magnesium and calciotrophic hormones. Magnes. Res. 1995, 8, 77–84. [Google Scholar] [PubMed]
- Epstein, M.; McGrath, S.; Law, F. Proton-pump inhibitors and hypomagnesemic hypoparathyroidism. N. Engl. J. Med. 2006, 355, 1834–1836. [Google Scholar] [CrossRef]
- Zittermann, A. Magnesium deficit? overlooked cause of low vitamin D status? BMC Med. 2013, 11, 229. [Google Scholar] [CrossRef]
- Luk, C.P.; Parsons, R.; Lee, Y.P.; Hughes, J.D. Proton pump inhibitor-associated hypomagnesemia: What do FDA data tell us? Ann. Pharmacother. 2013, 47, 773–780. [Google Scholar] [CrossRef]
- Lemon, T.I. Proton pump inhibitors and hypomagnesemia monitoring. Int. J. Gen. Med. 2013, 6, 675. [Google Scholar] [CrossRef][Green Version]
- Henzel, J.H.; DeWeese, M.S.; Ridenhour, G. Significance of magnesium and zinc metabolism in the surgical patient. I. Magnesium. Arch. Surg. 1967, 95, 974–990. [Google Scholar] [CrossRef]
- Tamura, T.; Sakaeda, T.; Kadoyama, K.; Okuno, Y. Omeprazole- and esomeprazole-associated hypomagnesaemia: Data mining of the public version of the FDA Adverse Event Reporting System. Int. J. Med. Sci. 2012, 9, 322–326. [Google Scholar] [CrossRef]
- Hess, M.W.; de Baaij, J.H.; Broekman, M.M.; Bisseling, T.M.; Haarhuis, B.J.; Tan, A.C.; Te Morsche, R.H.; Hoenderop, J.G.; Bindels, R.J.; Drenth, J.P. Common single nucleotide polymorphisms in transient receptor potential melastatin type 6 increase the risk for proton pump inhibitor-induced hypomagnesemia: A case-control study. Pharmacogenet. Genom. 2017, 27, 83–88. [Google Scholar] [CrossRef]
- Abbott, L.G.; Rude, R.K. Clinical manifestations of magnesium deficiency. Miner. Electrolyte Metab. 1993, 19, 314–322. [Google Scholar]
- Durlach, J. Recommended dietary amounts of magnesium: Mg RDA. Magnes. Res. 1989, 2, 195–203. [Google Scholar]
- Costello, R.B.; Elin, R.J.; Rosanoff, A.; Wallace, T.C.; Guerrero-Romero, F.; Hruby, A.; Lutsey, P.L.; Nielsen, F.H.; Rodriguez-Moran, M.; Song, Y.; et al. Perspective: The Case for an Evidence-Based Reference Interval for Serum Magnesium: The Time Has Come. Adv. Nutr. 2016, 7, 977–993. [Google Scholar] [CrossRef]
- Cohen, L.; Kitzes, R. Infrared spectroscopy and magnesium content of bone mineral in osteoporotic women. Isr. J. Med. Sci. 1981, 17, 1123–1125. [Google Scholar]
- Touitou, Y.; Godard, J.P.; Ferment, O.; Chastang, C.; Proust, J.; Bogdan, A.; Auzéby, A.; Touitou, C. Prevalence of magnesium and potassium deficiencies in the elderly. Clin. Chem. 1987, 33, 518–523. [Google Scholar] [PubMed]
- Malon, A.; Brockmann, C.; Fijalkowska-Morawska, J.; Rob, P.; Maj-Zurawska, M. Ionized magnesium in erythrocytes--the best magnesium parameter to observe hypo- or hypermagnesemia. Clin. Chim. Acta 2004, 349, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Schimatschek, H.F.; Rempis, R. Prevalence of hypomagnesemia in an unselected German population of 16,000 individuals. Magnes. Res. 2001, 14, 283–290. [Google Scholar] [PubMed]
- Vormann, J. Magnesium: Nutrition and metabolism. Mol. Aspects Med. 2003, 24, 27–37. [Google Scholar] [CrossRef]
- Florentin, M.; Elisaf, M.S. Proton pump inhibitor-induced hypomagnesemia: A new challenge. World J. Nephrol. 2012, 1, 151–154. [Google Scholar] [CrossRef]
- Famularo, G.; Gasbarrone, L.; Minisola, G. Hypomagnesemia and proton-pump inhibitors. Expert Opin. Drug Saf. 2013, 12, 709–716. [Google Scholar] [CrossRef] [PubMed]
- Schneider, I.; Greupner, T.; Hahn, A. Magnesium bioavailability from mineral waters with different mineralization levels in comparison to bread and a supplement. Food Nutr. Res. 2017, 61, 1384686. [Google Scholar] [CrossRef]
- Roush, G.C.; Sica, D.A. Diuretics for Hypertension: A Review and Update. Am. J. Hypertens. 2016, 29, 1130–1137. [Google Scholar] [CrossRef]
- Chen, P.; Chaugai, S.; Zhao, F.; Wang, D.W. Cardioprotective Effect of Thiazide-Like Diuretics: A Meta-Analysis. Am. J. Hypertens. 2015, 28, 1453–1463. [Google Scholar] [CrossRef] [PubMed]
- Wright, J.M.; Musini, V.M.; Gill, R. First-line drugs for hypertension. Cochrane Database Syst. Rev. 2018, 4, CD001841. [Google Scholar] [CrossRef] [PubMed]
- Shen, L.; Shah, B.R.; Reyes, E.M.; Thomas, L.; Wojdyla, D.; Diem, P.; Leiter, L.A.; Charbonnel, B.; Mareev, V.; Horton, E.S.; et al. Role of diuretics, β blockers, and statins in increasing the risk of diabetes in patients with impaired glucose tolerance: Reanalysis of data from the NAVIGATOR study. BMJ 2013, 347, f6745. [Google Scholar] [CrossRef]
- Martin, B.J.; Milligan, K. Diuretic-associated hypomagnesemia in the elderly. Arch. Intern. Med. 1987, 147, 1768–1771. [Google Scholar] [CrossRef]
- Kroenke, K.; Wood, D.R.; Hanley, J.F. The value of serum magnesium determination in hypertensive patients receiving diuretics. Arch. Intern. Med. 1987, 147, 1553–1556. [Google Scholar] [CrossRef]
- Seelig, C.B. Magnesium deficiency in two hypertensive patient groups. South. Med. J. 1990, 83, 739–742. [Google Scholar] [CrossRef]
- Petri, M.; Cumber, P.; Grimes, L.; Treby, D.; Bryant, R.; Rawlins, D.; Ising, H. The metabolic effects of thiazide therapy in the elderly: A population study. Age Ageing 1986, 15, 151–155. [Google Scholar] [CrossRef]
- Arampatzis, S.; Funk, G.-C.; Leichtle, A.B.; Fiedler, G.-M.; Schwarz, C.; Zimmermann, H.; Exadaktylos, A.K.; Lindner, G. Impact of diuretic therapy-associated electrolyte disorders present on admission to the emergency department: A cross-sectional analysis. BMC Med. 2013, 11, 83. [Google Scholar] [CrossRef]
- Whang, R.; Oei, T.O.; Aikawa, J.K.; Watanabe, A.; Vannatta, J.; Fryer, A.; Markanich, M. Predictors of clinical hypomagnesemia. Hypokalemia, hypophosphatemia, hyponatremia, and hypocalcemia. Arch. Intern. Med. 1984, 144, 1794–1796. [Google Scholar] [CrossRef]
- Dyckner, T.; Wester, P.O. Effects of magnesium infusions in diuretic induced hyponatraemia. Lancet 1981, 1, 585–586. [Google Scholar] [CrossRef]
- Malini, P.L.; Strocchi, E.; Valtancoli, G.; Ambrosioni, E. Angiotensin converting enzyme inhibitors, thiazide diuretics and magnesium balance. A preliminary study. Magnes. Res. 1990, 3, 193–196. [Google Scholar]
- Reinhart, R.A. Magnesium metabolism. A review with special reference to the relationship between intracellular content and serum levels. Arch. Intern. Med. 1988, 148, 2415–2420. [Google Scholar] [CrossRef]
- Paolisso, G.; Scheen, A.; D’Onofrio, F.; Lefèbvre, P. Magnesium and glucose homeostasis. Diabetologia 1990, 33, 511–514. [Google Scholar] [CrossRef] [PubMed]
- Takaya, J.; Higashino, H.; Kobayashi, Y. Intracellular magnesium and insulin resistance. Magnes. Res. 2004, 17, 126–136. [Google Scholar] [PubMed]
- Zou, Z.G.; Rios, F.J.; Montezano, A.C.; Touyz, R.M. TRPM7, Magnesium, and Signaling. Int. J. Mol. Sci. 2019, 20, 1877. [Google Scholar] [CrossRef] [PubMed]
- Gommers, L.M.; Hoenderop, J.G.; Bindels, R.J.; de Baaij, J.H. Hypomagnesemia in Type 2 Diabetes: A Vicious Circle? Diabetes 2016, 65, 3–13. [Google Scholar] [CrossRef]
- Cunha, A.R.; D’El-Rei, J.; Medeiros, F.; Umbelino, B.; Oigman, W.; Touyz, R.M.; Neves, M.F. Oral magnesium supplementation improves endothelial function and attenuates subclinical atherosclerosis in thiazide-treated hypertensive women. J. Hypertens. 2017, 35, 89–97. [Google Scholar] [CrossRef]
- Kisters, K.; Gröber, U. Lowered magnesium in hypertension. Hypertension 2013, 62, e19. [Google Scholar] [CrossRef]
| Drug Group (Drug, Substance) | Examples | Mechanism/Effect |
|---|---|---|
| Anti-diabetic medication | Insulin, insulin mimetic drugs | interference with Na+/Mg2+ exchanger SLC41A1, increased renal magnesium loss |
| Antimicrobials | Aminoglycoside antibiotics (e.g., gentamicin, tobramycin, amikacin) | reduced paracellular reabsorption of magnesium; increased renal magnesium loss, secondary hyperaldosteronism |
| Antimicrobial medication (e.g., Pentamidine) | increased renal magnesium loss | |
| Antiviral medication (e.g., foscarnet) | magnesium chelating, nephrotoxicity, increased renal magnesium loss | |
| Polyene antifungals(e.g., amphotericin B) | nephrotoxicity, increased renal magnesium loss | |
| Beta adrenergic agonists | Fenoterol, salbutamol, theophylline | increased renal magnesium excretion, metabolic abnormalities (magnesium shift into cells) |
| Bisphosphonates | Pamidronate | renal impairment, increased magnesium excretion |
| Cardiac glycoside | Digoxin | reduced renal tubular reabsorption of magnesium, increased magnesium excretion |
| Chemotherapeutic agents | Amsacrine, cisplatin | nephrotoxicity, cisplatin accumulates in renal cortex, increased renal magnesium loss, reduced TRPM6 expression (?) |
| Diuretics | Thiazide diuretics (e.g., HCT) | reduced TRPM6 expression (distal), increased renal magnesium loss, secondary hyperaldosteronism |
| Loop diuretics (e.g,. furosemide) | reduced paracellular magnesium reabsorption (thick ascending limb), increased renal magnesium loss, secondary hyperaldosteronism | |
| EGFR-Inhibitors | Cetuximab | increased renal magnesium loss, reduced TRPM6 activity |
| Immunosuppressants | mTOR-Kinase-Inhibitor (e.g., Rapamycin/Sirolimus) | reduced paracellular magnesium reabsorption |
| Calcineurin inhibitors (e.g., cyclosporine, tacrolimus) | reduced TRPM6 expression (distal), increased renal magnesium loss | |
| Proton-pump inhibitors | Omeprazole, pantoprazole | inhibition of active magnesium absorption by interfering with TRPM6 and TRPM7, increased renal magnesium loss (?) |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gröber, U. Magnesium and Drugs. Int. J. Mol. Sci. 2019, 20, 2094. https://doi.org/10.3390/ijms20092094
Gröber U. Magnesium and Drugs. International Journal of Molecular Sciences. 2019; 20(9):2094. https://doi.org/10.3390/ijms20092094
Chicago/Turabian StyleGröber, Uwe. 2019. "Magnesium and Drugs" International Journal of Molecular Sciences 20, no. 9: 2094. https://doi.org/10.3390/ijms20092094
APA StyleGröber, U. (2019). Magnesium and Drugs. International Journal of Molecular Sciences, 20(9), 2094. https://doi.org/10.3390/ijms20092094