Xenobiotics Formed during Food Processing: Their Relation with the Intestinal Microbiota and Colorectal Cancer
Abstract
1. Introduction
1.1. Impact of Diet on Colorectal Cancer
1.2. Intestinal Microbiota and Human Health
2. Food Processing and Xenobiotics
3. Effect of Food Processing-Borne Xenobiotics on the Gut Microbiota
3.1. Impact of Xenobiotics on Gut Microbiota
3.2. Impact of the Gut Microbiota on the Toxicity of Xenobiotics
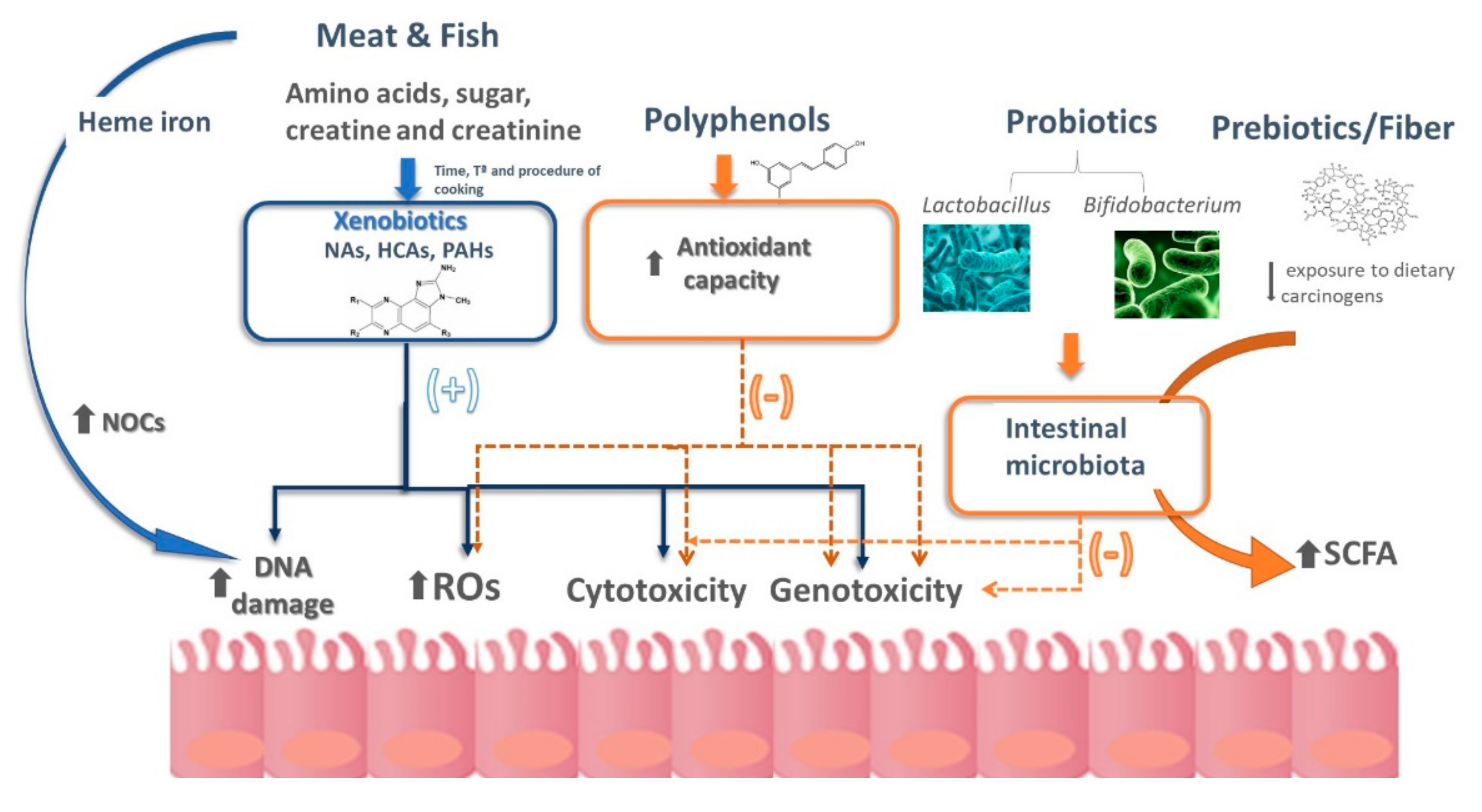
4. Creating a Balance between Xenobiotics and a Healthy Gut
5. Future Perspectives
5.1. Probiotics and Prebiotics to Counteract the Effect of Pro-Carcinogenic Compounds
5.2. Longitudinal Studies on Long Term Impact of Xenobiotics Derived from Food Processing
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ACs | Aminocarbolines |
| AIAs | Aminoimidazoazarenes |
| ATP | Adenosine triphosphate |
| B(a)P | Benzo(a) pyrene |
| CI | Confidence interval |
| CRC | Colorectal cancer |
| CYP1A1 | Aryl-4 monooxigenase |
| CYP450 | Cytochrome P450 |
| DiMeIQx | 2-Amino-3,4,8-trimethylimidazo[4,5-f] quinoxaline |
| EPIC | European Prospective Investigation into Cancer |
| HCAs | Heterocyclic amines |
| IARC | The International Agency for Research on Cancer |
| LAB | Lactic acid bacteria |
| MD | Mediterranean Diet |
| MeIQ | 2-Amino-3,4-dimethylimidazo[4,5-f] quinoline |
| MeIQx | 2-Amino-3,8-dimethylimidazo[4,5-f] quinoxaline |
| MN | Micronucleous assay |
| MTT | (3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) |
| NAs | Nitrosamines |
| NOCs | N-Nitroso compounds |
| OECD | Organization for Economic Cooperation and Development |
| PAHs | Polycyclic aromatic hydrocarbons |
| PhIP | 2-Amino-1-methyl-6-phenylimidazo[4,5-b] pyridine |
| ROS | Reactive oxygen species |
| RR | Relative risk |
| SCE | Sister Chromatid Exchange assay |
| SCFA | Short chain fatty acids |
| WD | Western Diet |
References
- Viera, C.; Rodrigues, M.; Russo, E.; Amedei, A. Role of diet and gut microbiota on colorectal cancer immunomodulation. World J. Gastroenterol. 2019, 25, 151–162. [Google Scholar] [CrossRef]
- Cunnane, S.C. Origins and evolution of the Western diet: Implications of iodine and seafood intakes for the human brain. Am. J. Clin. Nutr. 2005, 82, 483. [Google Scholar] [CrossRef][Green Version]
- Naja, F.; Hwalla, N.; Itani, L.; Karam, S.; Mehio Sibai, A.; Nasreddine, L. A Western dietary pattern is associated with overweight and obesity in a national sample of Lebanese adolescents (13–19 years): A cross-sectional study. Br. J. Nutr. 2015, 114, 1909–1919. [Google Scholar] [CrossRef]
- Di Daniele, N.; Noce, A.; Vidiri, M.F.; Moriconi, E.; Marrone, G.; Annicchiarico-Petruzzelli, M.; D’Urso, G.; Tesauro, M.; Rovella, V.; De Lorenzo, A. Impact of Mediterranean diet on metabolic syndrome, cancer and longevity. Oncotarget 2017, 8, 8947–8979. [Google Scholar] [CrossRef] [PubMed]
- Farinetti, A.; Zurlo, V.; Manenti, A.; Coppi, F.; Mattioli, A. Mediterranean diet and colorectal cancer: A systematic review. Nutrition 2017, 43–44, 82–88. [Google Scholar] [CrossRef]
- Liu, L.; Zhuang, W.; Wang, R.-Q.; Mukherjee, R.; Xiao, S.-M.; Chen, Z.; Wu, X.-T.; Zhou, Y.; Zhang, H.-Y. Is dietary fat associated with the risk of colorectal cancer? A meta-analysis of 13 prospective cohort studies. Eur. J. Nutr. 2011, 50, 173–184. [Google Scholar] [CrossRef] [PubMed]
- Beresford, S.A.A.; Johnson, K.C.; Ritenbaugh, C.; Lasser, N.L.; Snetselaar, L.G.; Black, H.R.; Anderson, G.L.; Assaf, A.R.; Bassford, T.; Bowen, D.; et al. Low-fat dietary pattern and risk of colorectal cancer. JAMA 2006, 295, 643–654. [Google Scholar] [CrossRef]
- Vieira, A.R.; Abar, L.; Chan, D.S.M.; Vingeliene, S.; Polemiti, E.; Stevens, C.; Greenwood, D.; Norat, T. Foods and beverages and colorectal cancer risk: A systematic review and meta-analysis of cohort studies, an update of the evidence of the WCRF-AICR Continuous Update Project. Ann. Oncol. 2017, 28, 1788–1802. [Google Scholar] [CrossRef] [PubMed]
- Zamora-Ros, R.; Barupal, D.K.; Rothwell, J.A.; Jenab, M.; Fedirko, V.; Romieu, I.; Aleksandrova, K.; Overvad, K.; Kyrø, C.; Tjønneland, A.; et al. Dietary flavonoid intake and colorectal cancer risk in the European prospective investigation into cancer and nutrition (EPIC) cohort. Int. J. Cancer 2017, 140, 1836–1844. [Google Scholar] [CrossRef]
- De Stefani, E.; Boffetta, P.; Deneo-Pellegrini, H.; Ronco, A.L.; Aune, D.; Acosta, G.; Brennan, P.; Mendilaharsu, M.; Ferro, G. Meat intake, meat mutagens and risk of lung cancer in Uruguayan men. Cancer Causes Control. 2009, 20, 1635–1643. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, L.R. Meat and cancer. Meat Sci. 2010, 84, 308–313. [Google Scholar] [CrossRef] [PubMed]
- Wie, G.-A.; Cho, Y.-A.; Kang, H.; Ryu, K.-A.; Yoo, M.-K.; Kim, Y.-A.; Jung, K.-W.; Kim, J.; Lee, J.-H.; Joung, H. Red meat consumption is associated with an increased overall cancer risk: A prospective cohort study in Korea. Br. J. Nutr. 2014, 112, 238–247. [Google Scholar] [CrossRef] [PubMed]
- Bouvard, V.; Loomis, D.; Guyton, K.Z.; Grosse, Y.; El Ghissassi, F.; Benbrahim-Tallaa, L.; Guha, N.; Mattock, H.; Straif, K.; Stewart, B.W.; et al. Carcinogenicity of consumption of red and processed meat. Lancet Oncol. 2015, 16, 1599–1600. [Google Scholar] [CrossRef]
- Aune, D.; Chan, D.S.M.; Vieira, A.R.; Navarro Rosenblatt, D.A.; Vieira, R.; Greenwood, D.C.; Kampman, E.; Norat, T. Red and processed meat intake and risk of colorectal adenomas: A systematic review and meta-analysis of epidemiological studies. Cancer Causes Control. 2013, 24, 611–627. [Google Scholar] [CrossRef]
- Chiang, V.S.C.; Quek, S.Y. The relationship of red meat with cancer: Effects of thermal processing and related physiological mechanisms. Crit. Rev. Food Sci. Nutr. 2017, 57, 1153–1173. [Google Scholar] [CrossRef] [PubMed]
- Catsburg, C.E.; Gago-Dominguez, M.; Yuan, J.M.; Castelao, J.E.; Cortessis, V.K.; Pike, M.C.; Stern, M.C. Dietary sources of N-nitroso compounds and bladder cancer risk: Findings from the Los Angeles bladder cancer study. Int. J. Cancer 2014, 134, 125–135. [Google Scholar] [CrossRef]
- Ministerio de Agricultura y Pesca Alimentación y Medio Ambiente (MAPAMA). Informe del Consumo de Alimentación en España; MAPAMA: Madrid, Spain, 2016; pp. 1–225.
- Hirose, M.; Takahashi, S.; Ogawa, K.; Futakuchi, M.; Shirai, T. Phenolics: Blocking agents for heterocyclic amine-induced carcinogenesis. Food Chem. Toxicol. 1999, 37, 985–992. [Google Scholar] [CrossRef]
- Backhed, F.; Ley, R.E.; Sonnenburg, J.L.; Peterson, D.A.; Gordon, J.I. Host-bacterial mutualism in the human intestine. Science 2005, 307, 1915–1920. [Google Scholar] [CrossRef]
- Sender, R.; Fuchs, S.; Milo, R. Revised estimates for the number of human and bacteria cells in the body. PLOS Biol. 2016, 14, e1002533. [Google Scholar] [CrossRef]
- Qin, J.; Li, R.; Raes, J.; Arumugam, M.; Burgdorf, K.S.; Manichanh, C.; Nielsen, T.; Pons, N.; Levenez, F.; Yamada, T.; et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 2010, 464, 59–65. [Google Scholar] [CrossRef]
- Arumugam, M.; Raes, J.; Pelletier, E.; Le Paslier, D.; Yamada, T.; Mende, D.R.; Fernandes, G.R.; Tap, J.; Bruls, T.; Batto, J.-M.; et al. Enterotypes of the human gut microbiome. Nature 2011, 473, 174–180. [Google Scholar] [CrossRef] [PubMed]
- O’Hara, A.M.; Shanahan, F. The gut flora as a forgotten organ. EMBO Rep. 2006, 7, 688–693. [Google Scholar] [CrossRef] [PubMed]
- Guarner, F.; Malagelada, J.-R. Gut flora in health and disease. Lancet 2003, 361, 512–519. [Google Scholar] [CrossRef]
- Knights, D.; Ward, T.L.; McKinlay, C.E.; Miller, H.; Gonzalez, A.; McDonald, D.; Knight, R. Rethinking “Enterotypes” . Cell Host Microbe 2014, 16, 433–437. [Google Scholar] [CrossRef] [PubMed]
- Lloyd-Price, J.; Mahurkar, A.; Rahnavard, G.; Crabtree, J.; Orvis, J.; Hall, A.B.; Brady, A.; Creasy, H.H.; McCracken, C.; Giglio, M.G.; et al. Strains, functions and dynamics in the expanded Human Microbiome Project. Nature 2017, 50, 61–66. [Google Scholar] [CrossRef]
- Li, J.; Jia, H.; Cai, X.; Zhong, H.; Feng, Q.; Sunagawa, S.; Arumugam, M.; Kultima, J.R.; Prifti, E.; Nielsen, T.; et al. An integrated catalog of reference genes in the human gut microbiome. Nat. Biotechnol. 2014, 32, 834–841. [Google Scholar] [CrossRef]
- Integrative HMP (iHMP) Research Network Consortium. The integrative human microbiome project: Dynamic analysis of microbiome-host omics profiles during periods of human health and disease. Cell Host Microbe 2014, 16, 276–289. [Google Scholar] [CrossRef]
- Ridaura, V.K.; Faith, J.J.; Rey, F.E.; Cheng, J.; Duncan, A.E.; Kau, A.L.; Griffin, N.W.; Lombard, V.; Henrissat, B.; Bain, J.R.; et al. Gut microbiota from twins discordant for obesity modulate metabolism in mice. Science 2013, 341, 6150–6172. [Google Scholar] [CrossRef]
- De Palma, G.; Lynch, M.D.J.; Lu, J.; Dang, V.T.; Deng, Y.; Jury, J.; Umeh, G.; Miranda, P.M.; Pigrau Pastor, M.; Sidani, S.; et al. Transplantation of fecal microbiota from patients with irritable bowel syndrome alters gut function and behavior in recipient mice. Sci. Transl. Med. 2017, 9, eaaf6397. [Google Scholar] [CrossRef]
- Shreiner, A.B.; Kao, J.Y.; Young, V.B. The gut microbiome in health and in disease. Curr. Opin. Gastroenterol. 2015, 31, 69–75. [Google Scholar] [CrossRef]
- Le Chatelier, E.; Nielsen, T.; Qin, J.; Prifti, E.; Hildebrand, F.; Falony, G.; Almeida, M.; Arumugam, M.; Batto, J.-M.; Kennedy, S.; et al. Richness of human gut microbiome correlates with metabolic markers. Nature 2013, 500, 541–546. [Google Scholar] [CrossRef]
- Qin, J.; Balzola, F.; Bernstein, C.; Ho, G.T.; Lees, C.; Bork, P.; Dusko, E.; Wang, J. A human gut microbial gene catalogue established by metagenomic sequencing: Commentary. Nature 2010, 464, 59–65. [Google Scholar] [CrossRef]
- Krajmalnik-Brown, R.; Lozupone, C.; Kang, D.-W.; Adams, J.B. Gut bacteria in children with autism spectrum disorders: Challenges and promise of studying how a complex community influences a complex disease. Microb. Ecol. Heal. Dis. 2015, 26, 26914. [Google Scholar] [CrossRef]
- Ahn, J.; Sinha, R.; Pei, Z.; Dominianni, C.; Wu, J.; Shi, J.; Goedert, J.J.; Hayes, R.B.; Yang, L. Human gut microbiome and risk for colorectal cancer. J. Natl. Cancer Inst. 2013, 105, 1907–1911. [Google Scholar] [CrossRef]
- Duvallet, C.; Gibbons, S.M.; Gurry, T.; Irizarry, R.A.; Alm, E.J. Meta-analysis of gut microbiome studies identifies disease-specific and shared responses. Nat. Commun. 2017, 8, 1784. [Google Scholar] [CrossRef]
- O’Keefe, S.J.D. Diet, microorganisms and their metabolites and colon cancer. Nat. Rev. Gastroenterol. Hepatol. 2016, 13, 691–706. [Google Scholar] [CrossRef]
- Zhang, Z.; Li, D. Thermal processing of food reduces gut microbiota diversity of the host and triggers adaptation of the microbiota: Evidence from two vertebrates. Microbiome 2018, 6, 99–113. [Google Scholar] [CrossRef]
- Katsidzira, L.; Laubscher, R.; Gangaidzo, I.T.; Swart, R.; Makunike-Mutasa, R.; Manyanga, T.; Thomson, S.; Ramesar, R.; Matenga, J.A.; Rusakaniko, S. Dietary patterns and colorectal cancer risk in Zimbabwe: A population based case-control study. Cancer Epidemiol. 2018, 57, 33–38. [Google Scholar] [CrossRef]
- Shirai, T.; Tamano, S.; Sano, M.; Masui, T.; Hasegawa, R.; Ito, N. Carcinogenicity of 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine (PhIP) in rats: Dose-response studies. Princess Takamatsu Symp. 1995, 23, 232–239. [Google Scholar]
- Kim, E.; Coelho, D.; Blachier, F. Review of the association between meat consumption and risk of colorectal cancer. Nutr. Res. 2013, 33, 983–994. [Google Scholar] [CrossRef] [PubMed]
- Bastide, N.; Pierre, F.; Corpet, D. Heme iron from meat and risk of colorectal cancer: A meta-analysis and a review of the mechanisms involved. Cancer Prev. Res. 2011, 4, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Viegas, O.; Novo, P.; Pinto, E.; Pinho, O.; Ferreira, I. Effect of charcoal types and grilling conditions on formation of heterocyclic aromatic amines (HAs) and polycyclic aromatic hydrocarbons (PAHs) in grilled muscle foods. Food Chem. Toxicol. 2012, 50, 2128–2134. [Google Scholar] [CrossRef] [PubMed]
- Chiavarini, M.; Bertarelli, G.; Minelli, L.; Fabiani, R. Dietary intake of meat cooking-related mutagens(HCAs) and risk of colorectal adenoma and cancer: A systematic review and meta-analysis. Nutrients 2017, 9, 514. [Google Scholar] [CrossRef] [PubMed]
- Martín-Calero, A.; Tejral, G.; Ayala, J.; González, V.; Afonso, A. Suitability of ionic liquids as mobile-phase additives in HPLC with fluorescence and UV detection for the determination of heterocyclic aromatic amines. J. Sep. Sci. 2010, 33, 182–190. [Google Scholar] [CrossRef]
- Etemadi, A.; Abnet, C.C.; Graubard, B.I.; Beane-Freeman, L.; Freedman, N.D.; Liao, L.; Dawsey, S.M.; Sinha, R. Anatomical subsite can modify the association between meat and meat compounds and risk of colorectal adenocarcinoma: Findings from three large US cohorts. Int. J. Cancer 2018, 143, 2261–2270. [Google Scholar] [CrossRef]
- Martínez Góngora, V.; Matthes, K.L.; Castaño, P.R.; Linseisen, J.; Rohrmann, S. Dietary heterocyclic amine intake and colorectal adenoma risk: A systematic review and meta-analysis. Cancer Epidemiol. Biomark. Prev. 2018, 28, 99–109. [Google Scholar] [CrossRef]
- Helmus, D.S.; Thompson, C.L.; Zelenskiy, S.; Tucker, T.C.; Li, L. Red Meat-derived heterocyclic amines increase risk of colon cancer: A population-based case-control study. Nutr. Cancer 2013, 65, 1141–1150. [Google Scholar] [CrossRef]
- Khan, M.; Busquets, R.; Santos, F.; Puignou, L. New method for the analysis of heterocyclic amines in meat extracts using pressurised liquid extraction and liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 2008, 1194, 155–160. [Google Scholar] [CrossRef]
- Kondjoyan, A.; Chevolleau, S.; Portanguen, S.; Molina, J.; Ikonic, P.; Clerjon, S.; Debrauwer, L. Realation between crust development and heterocyclic aromatic amine formation when air-roasting a meat cylinder. Food Chem. 2016, 213, 641–646. [Google Scholar] [CrossRef]
- Buła, M.; Przybylski, W.; Jaworska, D.; Kajak-Siemaszko, K. Formation of heterocyclic aromatic amines in relation to pork quality and heat treatment parameters. Food Chem. 2019, 276, 511–516. [Google Scholar] [CrossRef]
- Rohrmann, S.; Nimptsch, K.; Sinha, R.; Willett, W.C.; Giovannucci, E.L.; Platz, E.A.; Wu, K. Intake of Meat mutagens and risk of prostate cancer in a cohort of U.S. health professionals. Cancer Epidemiol. Biomark. Prev. 2015, 24, 1557–1563. [Google Scholar] [CrossRef] [PubMed]
- Moorthy, B.; Chu, C.; Carlin, D.J. Polycyclic aromatic hydrocarbons: From metabolism to lung cancer. Toxicol. Sci. 2015, 145, 5–15. [Google Scholar] [CrossRef] [PubMed]
- Larsson, B.; Sahlberg, P.; Eriksson, A.; Busk, L. Polycyclic Aromatic Hydrocarbons in Grilled Food. J. Agric. Food Chem. 1983, 31, 867–873. [Google Scholar] [CrossRef]
- Alomirah, H.; Al-Zenki, S.; Al-Hooti, S.; Zaghloul, S.; Sawaya, W.; Ahmed, N.; Kannan, K. Concentrations and dietary exposure to polycyclic aromatic hydrocarbons (PAHs) from grilled and smoked foods. Food Control 2011, 22, 2028–2035. [Google Scholar] [CrossRef]
- Flores-Balcázar, C.; Rosales-Pérez, S.; Galván-Salazar, G.; López-Navarro, O. Nutrientes de la dieta y apoptosis como mecanismos reguladores del cáncer. iMedPub J. 2015, 11. [Google Scholar] [CrossRef]
- Pérez-Morales, G.; Morales, P.; Haza, A. Polycyclic aromatic hidrocarbons (PAHS) I: Toxicity, population exposure and involved foods. RCCV 2016, 10, 1–15. [Google Scholar] [CrossRef]
- Tamaki, A.; Hayashi, H.; Nakajima, H.; Takii, T.; Katagiri, D.; Miyazawa, K.; Hirose, K.; Onozaki, K. Polycyclic aromatic hydrocarbon increases mRNA level for interleukin 1 beta in human fibroblast-like synoviocyte line via aryl hydrocarbon receptor. Biol. Pharm. Bull. 2004, 27, 407–410. [Google Scholar] [CrossRef]
- Joshi, A.D.; Kim, A.; Lewinger, J.P.; Ulrich, C.M.; Potter, J.D.; Cotterchio, M.; Le Marchand, L.; Stern, M.C. Meat intake, cooking methods, dietary carcinogens, and colorectal cancer risk: Findings from the Colorectal Cancer Family Registry. Cancer Med. 2015, 4, 936–952. [Google Scholar] [CrossRef] [PubMed]
- Busquets, R.; Bordas, M.; Toribio, F.; Puignou, L.; Galceran, M.T. Occurrence of heterocyclic amines in several home-cooked meat dishes of the Spanish diet. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2004, 802, 79–86. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Martínez-González, M.A.; Tong, T.Y.N.; Forouhi, N.G.; Khandelwal, S.; Prabhakaran, D.; Mozaffarian, D.; de Lorgeril, M. Definitions and potential health benefits of the Mediterranean diet: Views from experts around the world. BMC Med. 2014, 12, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Druzhinin, V.G.; Matskova, L.V.; Fucic, A. Induction and modulation of genotoxicity by the bacteriome in mammals. Mutat. Res. 2018, 776, 70–77. [Google Scholar] [CrossRef] [PubMed]
- De Kok, T. Evaluation of fecal mutagenicity and colorectal cancer risk. Mutat. Res. Mutat. Res. 2000, 463, 53–101. [Google Scholar] [CrossRef]
- Grasso, F.; Frisan, T. Bacterial genotoxins: Merging the DNA damage response into infection biology. Biomolecules 2015, 5, 1762–1782. [Google Scholar] [CrossRef] [PubMed]
- Jinadasa, R.N.; Bloom, S.E.; Weiss, R.S.; Duhamel, G.E. Cytolethal distending toxin: A conserved bacterial genotoxin that blocks cell cycle progression, leading to apoptosis of a broad range of mammalian cell lineages. Microbiology 2011, 157, 1851–1875. [Google Scholar] [CrossRef] [PubMed]
- Gagnaire, A.; Nadel, B.; Raoult, D.; Neefjes, J.; Gorvel, J.-P. Collateral damage: Insights into bacterial mechanisms that predispose host cells to cancer. Nat. Rev. Microbiol. 2017, 15, 109–128. [Google Scholar] [CrossRef] [PubMed]
- Ge, Z.; Schauer, D.B.; Fox, J.G. In vivo virulence properties of bacterial cytolethal-distending toxin. Cell. Microbiol. 2008, 10, 1599–1607. [Google Scholar] [CrossRef] [PubMed]
- Nougayrede, J.-P. Escherichia coli induces DNA double-strand breaks in eukaryotic cells. Science 2006, 313, 848–851. [Google Scholar] [CrossRef]
- Cougnoux, A.; Dalmasso, G.; Martinez, R.; Buc, E.; Delmas, J.; Gibold, L.; Sauvanet, P.; Darcha, C.; Déchelotte, P.; Bonnet, M.; et al. Bacterial genotoxin colibactin promotes colon tumour growth by inducing a senescence-associated secretory phenotype. Gut 2014, 63, 1932–1942. [Google Scholar] [CrossRef]
- Buc, E.; Dubois, D.; Sauvanet, P.; Raisch, J.; Delmas, J.; Darfeuille-Michaud, A.; Pezet, D.; Bonnet, R. High prevalence of mucosa-associated E. coli producing cyclomodulin and genotoxin in colon cancer. PLoS ONE 2013, 8, e56964. [Google Scholar] [CrossRef] [PubMed]
- Collins, D.; Hogan, A.M.; Winter, D.C. Microbial and viral pathogens in colorectal cancer. Lancet Oncol. 2011, 12, 504–512. [Google Scholar] [CrossRef]
- Wu, S.; Morin, P.J.; Maouyo, D.; Sears, C.L. Bacteroides fragilis enterotoxin induces c-Myc expression and cellular proliferation. Gastroenterology 2003, 124, 392–400. [Google Scholar] [CrossRef] [PubMed]
- Rubinstein, M.R.; Wang, X.; Liu, W.; Hao, Y.; Cai, G.; Han, Y.W. Fusobacterium nucleatum promotes colorectal carcinogenesis by modulating E-cadherin/β-catenin signaling via its fadA adhesin. Cell Host Microbe 2013, 14, 195–206. [Google Scholar] [CrossRef] [PubMed]
- Horiguchi, Y. Escherichia coli cytotoxic necrotizing factors and Bordetella dermonecrotic toxin: The dermonecrosis-inducing toxins activating Rho small GTPases. Toxicon 2001, 39, 1619–1627. [Google Scholar] [CrossRef]
- Nougayrède, J.-P.; Taieb, F.; De Rycke, J.; Oswald, E. Cyclomodulins: Bacterial effectors that modulate the eukaryotic cell cycle. Trends Microbiol. 2005, 13, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Barrasa, J.I.; Olmo, N.; Lizarbe, M.A.; Turnay, J. Bile acids in the colon, from healthy to cytotoxic molecules. Toxicol. In Vitro 2013, 27, 964–977. [Google Scholar] [CrossRef] [PubMed]
- Tsoi, H.; Chu, E.S.H.; Zhang, X.; Sheng, J.; Nakatsu, G.; Ng, S.C.; Chan, A.W.H.; Chan, F.K.L.; Sung, J.J.Y.; Yu, J. Peptostreptococcus anaerobius induces intracellular cholesterol biosynthesis in colon cells to induce proliferation and causes dysplasia in mice. Gastroenterology 2017, 152, 1419–1433. [Google Scholar] [CrossRef]
- Huycke, M.M.; Abrams, V.; Moore, D.R. Enterococcus faecalis produces extracellular superoxide and hydrogen peroxide that damages colonic epithelial cell DNA. Carcinogenesis 2002, 23, 529–536. [Google Scholar] [CrossRef]
- Owen, R.W. Generation of reactive oxygen species by the faecal matrix. Gut 2000, 46, 225–232. [Google Scholar] [CrossRef]
- Attene-Ramos, M.S. Evidence that hydrogen sulfide is a genotoxic agent. Mol. Cancer Res. 2006, 4, 9–14. [Google Scholar] [CrossRef]
- Andriamihaja, M.; Lan, A.; Beaumont, M.; Audebert, M.; Wong, X.; Yamada, K.; Yin, Y.; Tomé, D.; Carrasco-Pozo, C.; Gotteland, M.; et al. The deleterious metabolic and genotoxic effects of the bacterial metabolite p-cresol on colonic epithelial cells. Free Radic. Biol. Med. 2015, 85, 219–227. [Google Scholar] [CrossRef]
- Blachier, F.; Beaumont, M.; Andriamihaja, M.; Davila, A.-M.; Lan, A.; Grauso, M.; Armand, L.; Benamouzig, R.; Tomé, D. Changes in the luminal environment of the colonic epithelial cells and physiopathological consequences. Am. J. Pathol. 2017, 187, 476–486. [Google Scholar] [CrossRef]
- Szekely, J.; Gates, K.S. Noncovalent DNA binding and the mechanism of oxidative DNA damage by fecapentaene-12. Chem. Res. Toxicol. 2006, 19, 117–121. [Google Scholar] [CrossRef]
- Mouillé, B.; Delpal, S.; Mayeur, C.; Blachier, F. Inhibition of human colon carcinoma cell growth by ammonia: A non-cytotoxic process associated with polyamine synthesis reduction. Biochim. Biophys. Acta Gen. Subj. 2003, 1624, 88–97. [Google Scholar] [CrossRef]
- Federici, E.; Prete, R.; Lazzi, C.; Pellegrini, N.; Moretti, M.; Corsetti, A.; Cenci, G. Bacterial composition, genotoxicity, and cytotoxicity of fecal samples from individuals consuming omnivorous or vegetarian diets. Front. Microbiol. 2017, 8, 1–9. [Google Scholar] [CrossRef]
- Daniela, E.; Sara, S.; Marcella, M.; Giovanni, A.; Meynier, A.; Sophie, V.; Cristina, C.M. Fecal water genotoxicity in healthy free-living young Italian people. Food Chem. Toxicol. 2014, 64, 104–109. [Google Scholar] [CrossRef]
- Vanhaecke, L.; Knize, M.G.; Noppe, H.; De Brabander, H.; Verstraete, W.; Van de Wiele, T. Intestinal bacteria metabolize the dietary carcinogen 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine following consumption of a single cooked chicken meal in humans. Food Chem. Toxicol. 2008, 46, 140–148. [Google Scholar] [CrossRef]
- Kim, J.; Park, S.-H.; Do, K.H.; Kim, D.; Moon, Y. Interference with mutagenic aflatoxin B1-induced checkpoints through antagonistic action of ochratoxin A in intestinal cancer cells: A molecular explanation on potential risk of crosstalk between carcinogens. Oncotarget 2016, 7, 39627–39639. [Google Scholar] [CrossRef]
- Prete, R.; Tofalo, R.; Federici, E.; Ciarrocchi, A.; Cenci, G.; Corsetti, A. Food-associated Lactobacillus plantarum and yeasts inhibit the genotoxic effect of 4-nitroquinoline-1-oxide. Front. Microbiol. 2017, 8, 2349. [Google Scholar] [CrossRef]
- Ribière, C.; Peyret, P.; Parisot, N.; Darcha, C.; Déchelotte, P.J.; Barnich, N.; Peyretaillade, E.; Boucher, D. Oral exposure to environmental pollutant benzo[a]pyrene impacts the intestinal epithelium and induces gut microbial shifts in murine model. Sci. Rep. 2016, 6, 31027. [Google Scholar] [CrossRef]
- Defois, C.; Ratel, J.; Denis, S.; Batut, B.; Beugnot, R.; Peyretaillade, E.; Engel, E.; Peyret, P. Environmental pollutant benzo[a]pyrene impacts the volatile metabolome and transcriptome of the human gut microbiota. Front. Microbiol. 2017, 8, 1562. [Google Scholar] [CrossRef]
- Defois, C.; Ratel, J.; Garrait, G.; Denis, S.; Le Goff, O.; Talvas, J.; Mosoni, P.; Engel, E.; Peyret, P. Food chemicals disrupt human gut microbiota activity and impact intestinal homeostasis as revealed by in vitro systems. Sci. Rep. 2018, 8, 1–12. [Google Scholar] [CrossRef]
- Morotomi, M.; Mutai, M. In vitro binding of potent mutagenic pyrolysates to intestinal bacteria. J. Natl. Cancer Inst. 1986, 77, 195–201. [Google Scholar] [PubMed]
- Orrhage, K.; Sillerström, E.; Gustafsson, J.-Å.; Nord, C.E.; Rafter, J. Binding of mutagenic heterocyclic amines by intestinal and lactic acid bacteria. Mutat. Res. Mol. Mech. Mutagen. 1994, 311, 239–248. [Google Scholar] [CrossRef]
- Vanhaecke, L.; Vercruysse, F.; Boon, N.; Verstraete, W.; Cleenwerck, I.; De Wachter, M.; De Vos, P.; van de Wiele, T. Isolation and characterization of human intestinal bacteria capable of transforming the dietary carcinogen 2-Amino-1-Methyl-6-Phenylimidazo[4,5-b]Pyridine. Appl. Environ. Microbiol. 2008, 74, 1469–1477. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zhang, J.; Empl, M.T.; Schwab, C.; Fekry, M.I.; Engels, C.; Schneider, M.; Lacroix, C.; Steinberg, P.; Sturla, S.J. Gut microbial transformation of the dietary imidazoquinoxaline mutagen MelQx reduces its cytotoxic and mutagenic potency. Toxicol. Sci. 2017, 159, 266–276. [Google Scholar] [CrossRef]
- Carmody, R.N.; Turnbaugh, P.J. Host-microbial interactions in the metabolism of therapeutic and diet-derived xenobiotics. J. Clin. Investig. 2014, 124, 4173–4181. [Google Scholar] [CrossRef] [PubMed]
- Kassie, F. Intestinal microflora plays a crucial role in the genotoxicity of the cooked food mutagen 2-amino-3-methylimidazo[4,5-f]quinoline (IQ). Carcinogenesis 2001, 22, 1721–1725. [Google Scholar] [CrossRef]
- Humblot, C.; Murkovic, M.; Rigottier-Gois, L.; Bensaada, M.; Bouclet, A.; Andrieux, C.; Anba, J.; Rabot, S. Glucuronidase in human intestinal microbiota is necessary for the colonic genotoxicity of the food-borne carcinogen 2-amino-3-methylimidazo[4,5-f]quinoline in rats. Carcinogenesis 2007, 28, 2419–2425. [Google Scholar] [CrossRef] [PubMed]
- Kaderlik, K.R.; Mulder, G.J.; Turesky, R.J.; Lang, N.P.; Teitel, C.H.; Chiarelli, M.P.; Kadlubar, F.F. Glucuronidation of N -hydroxy heterocyclic amines by human and rat liver microsomes. Carcinogenesis 1994, 15, 1695–1701. [Google Scholar] [CrossRef] [PubMed]
- Do, K.N.; Fink, L.N.; Jensen, T.E.; Gautier, L.; Parlesak, A. TLR2 Controls intestinal carcinogen detoxication by CYP1A1. PLoS ONE 2012, 7, e32309. [Google Scholar] [CrossRef]
- Organisation for Economic Co-operation and Development. Genetic Toxicology Guidance Document; OECD: Paris, France, 2015; pp. 1–58. [Google Scholar]
- Thybaud, V.; Lorge, E.; Levy, D.D.; van Benthem, J.; Douglas, G.R.; Marchetti, F.; Moore, M.M.; Schoeny, R. Main issues addressed in the 2014-2015 revisions to the OECD Genetic Toxicology Test Guidelines. Environ. Mol. Mutagen. 2017, 58, 284–295. [Google Scholar] [CrossRef]
- Turkez, H.; Arslan, M.E.; Ozdemir, O. Genotoxicity testing: Progress and prospects for the next decade. Expert Opin. Drug Metab. Toxicol. 2017, 13, 1089–1098. [Google Scholar] [CrossRef]
- Iglesias, T.; López de Cerain, A.; Irache, J.; Martín-Arbella, N.; Wilcox, M.; Pearson, J.; Azqueta, A. Evaluation of the cytotoxicity, genotoxicity and mucus permeation capacity of several surface modified poly(anhydride) nanoparticles designed for oral drug delivery. Int. J. Pharm. 2017, 517, 67–79. [Google Scholar] [CrossRef] [PubMed]
- Róka, E.; Ujhelyi, Z.; Deli, M.; Bocsik, A.; Fenyvesi, É.; Szente, L.; Fenyvesi, F.; Vecsernyés, M.; Váradi, J.; Fehér, P.; et al. Evaluation of the cytotoxicity of α-cyclodextrin derivatives on the Caco-2 Cell line and human erythrocytes. Molecules 2015, 20, 20269–20285. [Google Scholar] [CrossRef]
- Valdés, L.; Gueimonde, M.; Ruas-Madiedo, P. Monitoring in real time the cytotoxic effect of Clostridium difficile upon the intestinal epithelial cell line HT29. J. Microbiol. Methods 2015, 119, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Fair, K.L.; Colquhoun, J.; Hannan, N.R.F. Intestinal organoids for modelling intestinal development and disease. Philos. Trans. R. Soc. B Biol. Sci. 2018, 373, 20170217. [Google Scholar] [CrossRef]
- Kaushik, G.; Ponnusamy, M.P.; Batra, S.K. Concise review: Current status of three-dimensional organoids as preclinical models. Stem Cells 2018, 36, 1329–1340. [Google Scholar] [CrossRef]
- Huang, W.-Y.; Cai, Y.-Z.; Zhang, Y. Natural phenolic compounds from medicinal herbs and dietary plants: Potential use for cancer prevention. Nutr. Cancer 2009, 62, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Dhawan, A.; Anderson, D.; de Pascual-Teresa, S.; Santos-Buelga, C.; Clifford, M.N.; Ioannides, C. Evaluation of the antigenotoxic potential of monomeric and dimeric flavanols, and black tea polyphenols against heterocyclic amine-induced DNA damage in human lymphocytes using the Comet assay. Mutat. Res. 2002, 515, 39–56. [Google Scholar] [CrossRef]
- Quelhas, I.; Petisca, C.; Viegas, O.; Melo, A.; Pinho, O.; Ferreira, I.M.P.L.V.O. Effect of green tea marinades on the formation of heterocyclic aromatic amines and sensory quality of pan-fried beef. Food Chem. 2010, 122, 98–104. [Google Scholar] [CrossRef]
- Bear, W.L.; Teel, R.W. Effects of citrus flavonoids on the mutagenicity of heterocyclic amines and on cytochrome P450 1A2 activity. Anticancer Res. 2000, 20, 3609–3614. [Google Scholar] [PubMed]
- Edenharder, R.; Sager, J.W.; Glatt, H.; Muckel, E.; Platt, K.L. Protection by beverages, fruits, vegetables, herbs, and flavonoids against genotoxicity of 2-acetylaminofluorene and 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine (PhIP) in metabolically competent V79 cells. Mutat. Res. 2002, 521, 57–72. [Google Scholar] [CrossRef]
- Cheng, K.-W.; Wu, Q.; Zheng, Z.P.; Peng, X.; Simon, J.E.; Chen, F.; Wang, M. Inhibitory effect of fruit extracts on the formation of heterocyclic amines. J. Agric. Food Chem. 2007, 55, 10359–10365. [Google Scholar] [CrossRef] [PubMed]
- Sepahpour, S.; Selamat, J.; Khatib, A.; Manap, M.Y.A.; Abdull Razis, A.F.; Hajeb, P. Inhibitory effect of mixture herbs/spices on formation of heterocyclic amines and mutagenic activity of grilled beef. Food Addit. Contam. Part A 2018, 35, 1911–1927. [Google Scholar] [CrossRef] [PubMed]
- Balogh, Z.; Gray, J.I.; Gomaa, E.A.; Booren, A.M. Formation and inhibition of heterocyclic aromatic amines in fried ground beef patties. Food Chem. Toxicol. 2000, 38, 395–401. [Google Scholar] [CrossRef]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef] [PubMed]
- Bindels, L.B.; Delzenne, N.M.; Cani, P.D.; Walter, J. Towards a more comprehensive concept for prebiotics. Nat. Rev. Gastroenterol. Hepatol. 2015, 12, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Rowland, I. Effect of Bifidobacterium longum and inulin on gut bacterial metabolism and carcinogen-induced aberrant crypt foci in rats. Carcinogenesis 1998, 19, 281–285. [Google Scholar] [CrossRef]
- Chen, C.-C.; Lin, W.-C.; Kong, M.-S.; Shi, H.N.; Walker, W.A.; Lin, C.-Y.; Huang, C.-T.; Lin, Y.-C.; Jung, S.-M.; Lin, T.-Y. Oral inoculation of probiotics Lactobacillus acidophilus NCFM suppresses tumour growth both in segmental orthotopic colon cancer and extra-intestinal tissue. Br. J. Nutr. 2012, 107, 1623–1634. [Google Scholar] [CrossRef]
- Gamallat, Y.; Meyiah, A.; Kuugbee, E.D.; Hago, A.M.; Chiwala, G.; Awadasseid, A.; Bamba, D.; Zhang, X.; Shang, X.; Luo, F.; et al. Lactobacillus rhamnosus induced epithelial cell apoptosis, ameliorates inflammation and prevents colon cancer development in an animal model. Biomed. Pharmacother. 2016, 83, 536–541. [Google Scholar] [CrossRef]
- Lili, Z.; Junyan, W.; Hongfei, Z.; Baoqing, Z.; Bolin, Z. Detoxification of cancerogenic compounds by lactic acid bacteria strains. Crit. Rev. Food Sci. Nutr. 2017, 1–16. [Google Scholar] [CrossRef]
- Tsuda, H.; Hara, K.; Miyamoto, T. Binding of mutagens to exopolysaccharide produced by Lactobacillus plantarum mutant strain 301102S. J. Dairy Sci. 2008, 91, 2960–2966. [Google Scholar] [CrossRef]
- Kumar, M.; Kumar, A.; Nagpal, R.; Mohania, D.; Behare, P.; Verma, V.; Kumar, P.; Poddar, D.; Aggarwal, P.K.; Henry, C.J.K.; et al. Cancer-preventing attributes of probiotics: An update. Int. J. Food Sci. Nutr. 2010, 61, 473–496. [Google Scholar] [CrossRef]
- Nowak, A.; Libudzisz, Z. Ability of probiotic Lactobacillus casei DN 114001 to bind or/and metabolise heterocyclic aromatic amines in vitro. Eur. J. Nutr. 2009, 48, 419–427. [Google Scholar] [CrossRef]
- Nowak, A.; Śliżewska, K.; Otlewska, A. Antigenotoxic activity of lactic acid bacteria, prebiotics, and products of their fermentation against selected mutagens. Regul. Toxicol. Pharmacol. 2015, 73, 938–946. [Google Scholar] [CrossRef]
- Beer, F.; Urbat, F.; Steck, J.; Huch, M.; Bunzel, D.; Bunzel, M.; Kulling, S.E. Metabolism of foodborne heterocyclic aromatic amines by Lactobacillus reuteri DSM 20016. J. Agric. Food Chem. 2017, 65, 6797–6811. [Google Scholar] [CrossRef] [PubMed]
- Zhong, L.; Zhang, X.; Covasa, M. Emerging roles of lactic acid bacteria in protection against colorectal cancer. World J. Gastroenterol. 2014, 20, 7878–7886. [Google Scholar] [CrossRef]
- Nowak, A.; Śliżewska, K.; Błasiak, J.; Libudzisz, Z. The influence of Lactobacillus casei DN 114 001 on the activity of faecal enzymes and genotoxicity of faecal water in the presence of heterocyclic aromatic amines. Anaerobe 2014, 30, 129–136. [Google Scholar] [CrossRef]
- Gianfredi, V.; Salvatori, T.; Villarini, M.; Moretti, M.; Nucci, D.; Realdon, S. Is dietary fibre truly protective against colon cancer? A systematic review and meta-analysis. Int. J. Food Sci. Nutr. 2018, 69, 904–915. [Google Scholar] [CrossRef]
- Lim, C.C.; Ferguson, L.R.; Tannock, G.W. Dietary fibres as “prebiotics”: Implications for colorectal cancer. Mol. Nutr. Food Res. 2005, 49, 609–619. [Google Scholar] [CrossRef]
- Mahowald, M.A.; Rey, F.E.; Seedorf, H.; Turnbaugh, P.J.; Fulton, R.S.; Wollam, A.; Shah, N.; Wang, C.; Magrini, V.; Wilson, R.K.; et al. Characterizing a model human gut microbiota composed of members of its two dominant bacterial phyla. Proc. Natl. Acad. Sci. USA 2009, 106, 5859–5864. [Google Scholar] [CrossRef]
- Jakszyn, P.; Agudo, A.; Ibáñez, R.; García-Closas, R.; Pera, G.; Amiano, P.; González, C.A. Development of a food database of nitrosamines, heterocyclic amines, and polycyclic aromatic hydrocarbons. J. Nutr. 2004, 134, 2011–2014. [Google Scholar] [CrossRef]
| Year | No. Subjects | Analytical Category | Source | Dose | Pathology | Ref. |
|---|---|---|---|---|---|---|
| 2018 | 407,270 | HCAs | Red meat | n.a a | MeIQx and DiMeIQx association with all anatomical subsites of colorectal cancer. PhIP associations with total colorectal and colon cancers. Not evidenced an association between ingested B(a)P and CRC | [46] |
| MeIQx | ||||||
| DiMeIQx | ||||||
| PhIP | ||||||
| PAHs | ||||||
| B(a)P | ||||||
| 2018 | 76,657 | HCAs | Red meat | 50 ng/day | Association of HCAs, B(a)P, and mutagenicity index with the risk of colorectal adenomas | [47] |
| MeIQx | n.a a | |||||
| DiMeIQx | 40 ng/day | |||||
| PhIP | n.a a | |||||
| PAHs | ||||||
| B(a)P | ||||||
| 2013 | total 3707: 1062 cases and 1645 controls | HCAs | Red meat | n.a a | Colon cancer | [48] |
| MeIQx | ||||||
| DiMeIQ | ||||||
| PhIP |
| Main Mechanism | Molecules/Compounds Involved | Microbial Group | Experimental Approach Used for Study | Mode of Action | Ref. | |
|---|---|---|---|---|---|---|
| Direct mechanisms | Genotoxins | Typhoid toxin | Salmonella enterica serovar Typhi | In vitro and animal models | DNAse activity; induction of symptoms characteristic of typhoid fever | [64] |
| Cytolethal distending toxin | Proteobacteria | Cell lines and primary cell and mouse models of chronic infections | DNase activity; Proinflamation and carcinogenic potential | [65,66,67] | ||
| Colibactin | Escherichia coli group B | Eukaryotic cells | DNA double-strand breaks | [68] | ||
| Epidemiological and animal model | DNA double-strand breaks in vitro and in vivo; enhanced tumour growth by senescence | [69,70] | ||||
| Alteration of host cellular cycle | Cytotoxin-associated gene A Vacuolating cytotoxin A | Helicobacter pylori | Molecular, experimental and epidemiological | DNA damage; Increases IL-8; produces reactive oxygen species (ROS) and nitric oxide; increases concentrations of cyclo-oxygenase 2; decreases apoptosis; and increases cell proliferation | [66,71] | |
| Enterotoxin | Bacteroides fragilis | In vitro and epidemiological | DNA damage; high levels of ROS; Diarrheal disease, associated with colorectal cancer | [62,72] | ||
| Adhesin A | Fusobacterium nucleatum | In vitro and epidemiological | Activation of β catenin pathway | [66,73] | ||
| ExoS exotoxin | Pseudomonas aeruginosa | In vitro, experimental and epidemiological | Activation of pathways with final mechanism leading to DNA damage; unknown mechanisms in cancer generation | [62,66] | ||
| Cysteine protease-like | Shigella flexneri | In vitro and epidemiological | Potassium outflow conducting to ROS production; induce degradation of p53; DNA damage; dysentery | [62,66] | ||
| Avirulence protein A | Salmonella enterica | In vitro and mouse model of inflammation-associated cancer | Target β-catenin pathway; colonic tumorigenesis and tumour progression | [66] | ||
| Cytotoxic necrotising factor | Escherichia coli | In vitro and animal models | Activates Rho GTPase; modifies cytoskeleton; triggers G1-S transition; downregulate mismatch repair genes; the role of CNF in infections in not clear | [71,74] | ||
| Cycle-inhibiting factor | In vitro | Inhibition of mitosis | [75] | |||
| Secondary bile acids | Anaerobic bacteria with 7-α dehydroxylation activity of primary bile acids | In vitro colon cells and animal models | Changes in physicochemical membrane properties; Apoptosis and genomic damage by ROS; Deoxycholic acid is carcinogenic at high doses and long-term treatment in animal models | [76] | ||
| Indirect mechanisms | Oxidative stress | Reactive oxygen species | Peptostreptococcus anaerobius | In vivo, in vitro and epidemiological | Increase of human colon tumour tissues and adenomas; these bacteria increase colon dysplasia in a mouse model of CRC by induction of ROS levels, which promotes cholesterol synthesis and cell proliferation. | [77] |
| Enterococcus faecalis | In vitro and in vivo models, epidemiological | Induction of ROS, activation of macrophages; promotion of tumorigenesis | [66,78] | |||
| Faecal matrix | In vitro | Unknown reducing agent | [79] | |||
| Formation of H2S | H2S | Sulfate-reducing bacteria | Epidemiological and in vitro models | Promotes instability or cumulative mutations in a predisposed genetic background | [80] | |
| Inflammation | Wall-extracted antigen | Streptococcus bovis | Epidemiological and molecular | Activation of cyclo-oxygenase 2, interleukin 8 production, and cell proliferation | [71] | |
| Disabling cellular DNA repair process | Listeriolysin O | Listeria monocytogenes | In vitro and epidemiological | Pore formation in intestinal host cells; Prevention of recruitment of repair complex to DNA breaks; listeriosis | [66] | |
| Secreted effector protein EspF | Escherichia coli | In vitro | Down-regulation DNA mismatch repair | [66] | ||
| Protein metabolism | Phenol/indol/p-cresol/ | Intestinal bacteria | Colonic cells | Increased anion superoxide production and genotoxic effects | [81,82] | |
| Fecapentanes | Bacteroides sp. | In vitro; In vivo | Cytotoxic and mutagenic effects via ROS production; Controversial in vivo effect | [63,83] | ||
| Ammonium | Intestinal bacteria | In vitro | Antiproliferative effect without decrease of cell viability | [84] | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nogacka, A.M.; Gómez-Martín, M.; Suárez, A.; González-Bernardo, O.; de los Reyes-Gavilán, C.G.; González, S. Xenobiotics Formed during Food Processing: Their Relation with the Intestinal Microbiota and Colorectal Cancer. Int. J. Mol. Sci. 2019, 20, 2051. https://doi.org/10.3390/ijms20082051
Nogacka AM, Gómez-Martín M, Suárez A, González-Bernardo O, de los Reyes-Gavilán CG, González S. Xenobiotics Formed during Food Processing: Their Relation with the Intestinal Microbiota and Colorectal Cancer. International Journal of Molecular Sciences. 2019; 20(8):2051. https://doi.org/10.3390/ijms20082051
Chicago/Turabian StyleNogacka, Alicja M., María Gómez-Martín, Adolfo Suárez, Oscar González-Bernardo, Clara G. de los Reyes-Gavilán, and Sonia González. 2019. "Xenobiotics Formed during Food Processing: Their Relation with the Intestinal Microbiota and Colorectal Cancer" International Journal of Molecular Sciences 20, no. 8: 2051. https://doi.org/10.3390/ijms20082051
APA StyleNogacka, A. M., Gómez-Martín, M., Suárez, A., González-Bernardo, O., de los Reyes-Gavilán, C. G., & González, S. (2019). Xenobiotics Formed during Food Processing: Their Relation with the Intestinal Microbiota and Colorectal Cancer. International Journal of Molecular Sciences, 20(8), 2051. https://doi.org/10.3390/ijms20082051





