Acute Spinal Cord Injury: A Systematic Review Investigating miRNA Families Involved
Abstract
1. Introduction
2. Functional and Neurological Implications of the Spinal Cord Injury
2.1. Traumatic SCI: Clinical Findings
2.2. Evaluating Tools
2.2.1. Frankel Scale
2.2.2. ASIA Scale
2.2.3. Barthel Index
2.2.4. Functional Independence Measure
3. Pathophysiology of the Spinal Cord Injury
3.1. Primary Mechanisms
3.2. Secondary Mechanisms
3.3. SCI-Related Pathways
4. Management of the Spinal Cord Injury
4.1. General Management
4.2. The NASCIS Era and Its Legacy
4.3. Surgical Decompression and Treatment of the Vertebral Bony Associated Lesions
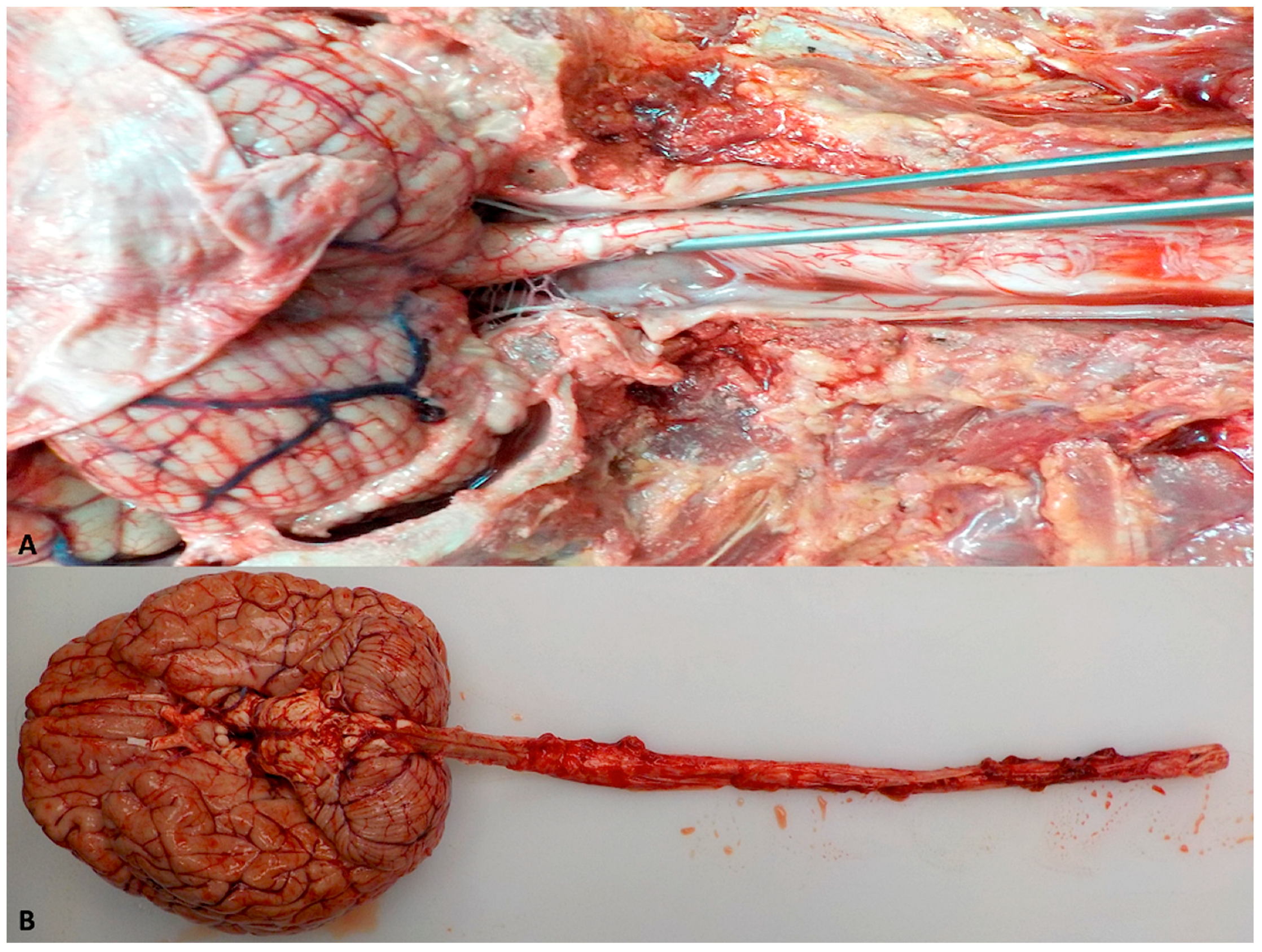
5. Focused Postmortem Techniques of the Human Brain and Spinal Cord
5.1. Removal of the Human Brain and Spinal Cord
5.2. Dissection of the Human Brain and Spinal Cord
5.3. Selection of Tissue Blocks for Histologic Examination
6. Animal Models for the Understanding of SCI
Protocol of Study
7. The Role of miRNA after SCI
7.1. miRNAS: Stucture and Function
7.2. miR-20a and miR-29b
7.3. miR-223
7.4. mir-21
7.5. mir-15 and mir-16
7.6. miR-124
7.7. miR-200c
7.8. miR-486
7.9. Let-7
7.10. miR-96 and miR146a
7.11. miR-107
7.12. miR-1
7.13. miR-129
8. The Future of SCI-Related miRNAs
9. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| SCI | Spinal cord injury |
| CS | Cervical spine |
| TS | Thoracic spine |
| LS | Lumbar spine |
| BSB | Blood–Spinal–Barrier |
| ICU | Intensive care unit |
References
- Sekhon, L.H.; Fehlings, MG. Epidemiology, demographics, and pathophysiology of acute spinal cord injury. Spine 2001, 26, S2–S12. [Google Scholar] [PubMed]
- Dumont, R.J.; Okonkwo, D.O.; Verma, S.; Hurlbert, R.J.; Boulos, P.T.; Ellegala, D.B.; Dumont, A.S. Acute spinal cord injury, part I: Pathophysiologic mechanisms. Clin. Neuropharmacol. 2001, 24, 254–264. [Google Scholar]
- Galeiras Vázquez, R.; Ferreiro Velasco, M.E.; Mourelo Fariña, M.; Montoto Marqués, A.; Salvador de la Barrera, S. Update on traumatic acute spinal cord injury. Part 1. Med. Intensiva 2017, 41, 201–264. [Google Scholar] [CrossRef]
- Witiw, C.D.; Fehlings, M.G. Acute Spinal Cord Injury. J. Spinal Disord. Tech. 2015, 28, 202–210. [Google Scholar] [CrossRef] [PubMed]
- Eckert, M.J.; Martin, M.J. Trauma: Spinal Cord Injury. Surg. Clin. N. Am. 2017, 97, 1031–1045. [Google Scholar] [CrossRef] [PubMed]
- Ahuja, C.S.; Nori, S.; Tetreault, L.; Wilson, J.; Kwon, B.; Harrop, J.; Choi, D.; Fehlings, M.G. Traumatic Spinal Cord Injury-Repair and Regeneration. Neurosurgery 2017, 80, S9–S22. [Google Scholar] [CrossRef] [PubMed]
- Yong, H.Y.F.; Rawji, K.S.; Ghorbani, S.; Xue, M.; Yong, V.W. The benefits of neuroinflammation for the repair of the injured central nervous system. Cell Mol. Immunol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Frankel, H.L.; Hancock, D.O.; Hyslop, G.; Melzak, J.; Michaelis, L.S.; Ungar, G.H.; Vernon, J.D.; Walsh, J.J. The value of postural reduction in the initial management of closed injuries of the spine with paraplegia and tetraplegia. I. Paraplegia 1969, 7, 179–192. [Google Scholar] [CrossRef]
- Roberts, T.T.; Leonard, G.R.; Cepela, D.J. Classifications in Brief: American Spinal Injury Association (ASIA) Impairment Scale. Clin. Orthop. Relat. Res. 2017, 475, 1499–1504. [Google Scholar]
- Maynard, F.M., Jr.; Bracken, M.B.; Creasey, G.; Ditunno, J.F., Jr.; Donovan, W.H.; Ducker, T.B.; Garber, S.L.; Marino, R.J.; Stover, S.L.; Tator, C.H.; et al. International Standards for Neurological and Functional Classification of Spinal Cord Injury. Am. Spinal Inj. Assoc. Spinal Cord. 1997, 35, 266–274. [Google Scholar] [CrossRef]
- Mahoney, F.I.; Barthel, D.W. Functional evaluation: The barthel index. Md. State Med. J. 1965, 14, 61–65. [Google Scholar]
- Shah, S.; Vanclay, F.; Cooper, B. Improving the sensitivity of the Barthel Index for stroke rehabilitation. J. Clin. Epidemiol. 1989, 42, 703–709. [Google Scholar] [CrossRef]
- Zhang, J.L.; Chen, J.; Wu, M.; Wang, C.; Fan, W.X.; Mu, J.S.; Wang, L.; Ni, C.M. Several time indicators and Barthel index relationships at different spinal cord injury levels. Spinal Cord. 2015, 53, 679–681. [Google Scholar] [CrossRef][Green Version]
- Chumney, D.; Nollinger, K.; Shesko, K.; Skop, K.; Spencer, M.; Newton, R.A. Ability of Functional Independence Measure to accurately predict functional outcome of stroke-specific population: Systematic review. J. Rehabil. Res. Dev. 2010, 47, 17–29. [Google Scholar] [CrossRef] [PubMed]
- Horn, S.D.; DeJong, G.; Smout, R.; Gassaway, J.; James, R.; Conroy, B. Stroke rehabilitation patients, practice, and outcomes: Is earlier and more aggressive therapy better? Arch. Phys. Med. Rehabil. 2005, 86, 101–114. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro Neto, F.; Guanais, P.; Lopes, G.H.; Dornelas, E.; de Campos Barbetta, D.; Coutinho, A.C.; Gonçalves, C.W.; Gomes Costa, R.R. Influence of Relative Strength on Functional Independence of Patients with Spinal Cord Injury. Arch. Phys. Med. Rehabil. 2017, 98, 1104–1112. [Google Scholar] [CrossRef] [PubMed]
- Schnake, K.J.; Schroeder, G.D.; Vaccaro, A.R.; Oner, C. AOSpine Classification Systems (Subaxial, Thoracolumbar). J. Orthop. Trauma 2017, 31, S14–S23. [Google Scholar] [CrossRef] [PubMed]
- Tator, C.H. Update on the pathophysiology and pathology of acute spinal cord injury. Brain Pathol. 1995, 5, 407–413. [Google Scholar] [CrossRef]
- Fehlings, M.G.; Sekhon, L.H.S. Cellular, ionic and biomolecular mechanisms of the injury process. In Contemporary Management of Spinal Cord Injury: From Impact to Rehabilitation; Tator, C.H., Benzel, E.C., Eds.; American Association of Neurological Surgeons: New York, NY, USA, 2000; pp. 33–50. [Google Scholar]
- Young, W.; Koreh, I. Potassium and calcium changes in injured spinal cords. Brain Res. 1986, 365, 42–53. [Google Scholar] [CrossRef]
- Noonan, V.K.; Dvorak, M.F.; Fehlings, M.G. Epidemiology of traumatic and nontraumatic spinal cord injury. In Critical Care in Spinal Cord Injury; Fehlings, M.G., Ed.; Future Medicine Ltd.: London, UK, 2013; pp. 6–20. [Google Scholar]
- Singh, A.; Tetreault, L.; Kalsi-Ryan, S.; Nouri, A.; Fehlings, M.G. Global prevalence and incidence of traumatic spinal cord injury. Clin. Epidemiol. 2014, 6, 309–331. [Google Scholar] [PubMed]
- Pineau, I.; Lacroix, S. Proinflammatory cytokine synthesis in the injured mouse spinal cord: Multiphasic expression pattern and identification of the cell types involved. J. Comp. Neurol. 2007, 500, 267–285. [Google Scholar] [CrossRef]
- Tator, C.H.; Fehlings, M.G. Review of the secondary injury theory of acute spinal cord trauma with emphasis on vascular mechanisms. J. Neurosurg. 1991, 75, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Wallace, M.C.; Tator, C.H.; Frazee, P. Relationship between posttraumatic ischemia and hemorrhage in the injured rat spinal cord as shown by colloidal carbon angiography. Neurosurgery 1986, 18, 433–439. [Google Scholar] [CrossRef] [PubMed]
- De la Torre, J.C. Spinal cord injury: Review of basic and applied research. Spine 1981, 6, 315–335. [Google Scholar] [CrossRef]
- Nemecek, S. Morphological evidence of microcirculatory disturbances in experimental spinal cord trauma. Adv. Neurol. 1978, 20, 395–405. [Google Scholar] [PubMed]
- Demopoulos, H.B.; Flamm, E.S.; Pietronigro, D.D.; Seligman, M.L. The free radical pathology and the microcirculation in the major central nervous system disorders. Acta Physiol. Scand. Suppl. 1980, 492, 91–119. [Google Scholar] [PubMed]
- Kwon, B.K.; Tetzlaff, W.; Grauer, J.N.; Beiner, J.; Vaccaro, A.R. Pathophysiology and pharmacologic treatment of acute spinal cord injury. Spine J. 2004, 4, 451–464. [Google Scholar] [CrossRef] [PubMed]
- Hall, E.D.; Braughler, J.M. Effects of intravenous methylprednisolone on spinal cord lipid peroxidation and Na (K)-ATPase activity. Doseresponse analysis during 1st hour after contusion injury in the cat. J. Neurosurg. 1982, 57, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Hall, E.D.; Braughler, J.M. Free radicals in CNS injury. Res. Publ. Assoc. Res. Nerv. Ment. Dis. 1993, 71, 81–105. [Google Scholar] [CrossRef]
- Thuret, S.; Moon, L.D.; Gage, F.H. Therapeutic interventions after spinal cord injury. Nat. Rev. Neurosci. 2006, 7, 628–643. [Google Scholar] [CrossRef]
- Hawthorne, A.L.; Popovich, P.G. Emerging concepts in myeloid cell biology after spinal cord injury. Neurotherapeutics 2011, 8, 252–261. [Google Scholar] [CrossRef] [PubMed]
- Keane, R.W.; Kraydieh, S.; Lotocki, G.; Bethea, J.R.; Krajewski, S.; Reed, J.C.; Dietrich, W.D. Apoptotic and antiapoptotic mechanisms following spinal cord injury. J. Neuropathol. Exp. Neurol. 2001, 60, 422–429. [Google Scholar] [CrossRef]
- Dusart, I.; Schwab, M.E. Secondary cell death and the inflammatory reaction after dorsal hemisection of the rat spinal cord. Eur. J. Neurosci. 1994, 6, 712–724. [Google Scholar] [CrossRef]
- Aimone, J.B.; Leasure, J.L.; Perreau, V.M.; Thallmair, M. Christopher Reeve Paralysis Foundation Research Consortium. Spatial and temporal gene expression profiling of the contused rat spinal cord. Exp. Neurol. 2004, 189, 204–221. [Google Scholar] [CrossRef] [PubMed]
- Downward, J. Ras signalling and apoptosis. Curr. Opin. Genet. Dev. 1998, 8, 49–54. [Google Scholar] [CrossRef]
- Shields, J.M.; Pruitt, K.; McFall, A.; Shaub, A.; Der, C.J. Understanding Ras: ‘it ain’t over ‘til it’s over’. Trends Cell. Biol. 2000, 10, 147–154. [Google Scholar] [CrossRef]
- Vojtek, A.B.; Der, C.J. Increasing complexity of the Ras signaling pathway. J. Biol. Chem. 1998, 273, 19925–19928. [Google Scholar] [CrossRef]
- Secombe, J.; Pierce, S.B.; Eisenman, R.N. Myc: A weapon of mass destruction. Cell 2004, 117, 153–156. [Google Scholar] [CrossRef]
- Lindsay, M.A. MicroRNAs and the immune response. Trends Immunol. 2008, 29, 343–351. [Google Scholar] [CrossRef]
- Sonkoly, E.; Pivarcsi, A. MicroRNAs in inflammation. Int. Rev. Immunol. 2009, 28, 535–561. [Google Scholar] [CrossRef] [PubMed]
- Hausmann, O. Post-traumatic inflawing spinal cord injury. Spinal Cord. 2003, 41, 369–378. [Google Scholar] [CrossRef]
- Carlson, S.L.; Parrish, M.E.; Springer, J.E.; Doty, K.; Dossett, L. Acute inflammatory response in spinal cord following impact injury. Exp. Neurol. 1998, 151, 77–88. [Google Scholar] [CrossRef] [PubMed]
- Taoka, Y.; Okajima, K.; Uchiba, M.; Murakami, K.; Kushimoto, S.; Johno, M.; Naruo, M.; Okabe, H.; Takatsuki, K. Role of neutrophils in spinal cord injury in the rat. Neuroscience 1997, 79, 1177–1182. [Google Scholar] [CrossRef]
- Amar, A.P.; Levy, M.L. Pathogenesis and pharmacological strategies for mitigating secondary damage in acute spinal cord injury. Neurosurgery 1999, 44, 1027–1039. [Google Scholar] [CrossRef]
- Loy, D.N.; Crawford, C.H.; Darnall, J.B.; Burke, D.A.; Onifer, S.M.; Whittemore, S.R. Temporal progression of angiogenesis and basal lamina deposition after contusive spinal cord injury in the adult rat. J. Comp. Neurol. 2002, 445, 308–324. [Google Scholar] [CrossRef]
- Karimi-Abdolrezaee, S.; Billakanti, R. Reactive astrogliosis after spinal cord injury-beneficial and detrimental effects. Mol. Neurobiol. 2012, 46, 251–264. [Google Scholar] [CrossRef]
- Sofroniew, M.V. Molecular dissection of reactive astrogliosis and glial scar formation. Trends Neurosci. 2009, 32, 638–647. [Google Scholar] [CrossRef]
- Silver, J.; Miller, J.H. Regeneration beyond the glial scar. Nat. Rev. Neurosci. 2004, 5, 146–156. [Google Scholar] [CrossRef]
- Faulkner, J.R.; Herrmann, J.E.; Woo, M.J.; Tansey, K.E.; Doan, N.B.; Sofroniew, M.V. Reactive astrocytes protect tissue and preserve function after spinal cord injury. J. Neurosci. 2004, 24, 2143–2155. [Google Scholar] [CrossRef]
- Zamanian, J.L.; Xu, L.; Foo, L.C.; Nouri, N.; Zhou, L.; Giffard, R.G.; Barres, B.A. Genomic analysis of reactive astrogliosis. J. Neurosci. 2012, 32, 6391–6410. [Google Scholar] [CrossRef] [PubMed]
- Nieto-Diaz, M.; Esteban, F.J.; Reigada, D.; Muñoz-Galdeano, T.; Yunta, M.; Caballero-López, M.; Navarro-Ruiz, R.; Del Águila, A.; Maza, R.M. MicroRNA dysregulation in spinal cord injury: Causes, consequences and therapeutics. Front. Cell. Neurosci. 2014, 8, 53. [Google Scholar] [CrossRef]
- Profyris, C.; Cheema, S.S.; Zang, D.; Azari, M.F.; Boyle, K.; Petratos, S. Degenerative and regenerative mechanisms governing spinal cord injury. Neurobiol. Dis. 2004, 15, 415–436. [Google Scholar] [CrossRef] [PubMed]
- Lewen, A.; Matz, P.; Chan, P.H. Free radical pathways in CNS injury. J. Neurotrauma 2000, 17, 871–890. [Google Scholar] [CrossRef] [PubMed]
- Montzka, K.; Lassonczyk, N.; Tschöke, B.; Neuss, S.; Führmann, T.; Rachelle Franzen, R.; Smeets, R.; Brook, G.A.; Wöltje, M. Neural differentiation potential of human bone marrow-derived mesenchymal stromal cells: Misleading marker gene expression. BMC Neurosci. 2009, 10, 16. [Google Scholar] [CrossRef] [PubMed]
- Uittenbogaard, M.; Baxter, K.K.; Chiaramello, A. NeuroD6 genomic signature bridging neuronal differentiation to survival via the molecular chaperone network. J. Neurosci. 2010, 88, 33–54. [Google Scholar] [CrossRef]
- Harkema, S.J. Neural plasticity after human spinal cord injury: Application of locomotor training to the rehabilitation of walking. Neuroscientist 2001, 7, 455–468. [Google Scholar] [CrossRef]
- Resnick, D.K. Guidelines for the Management of Acute Cervical Spine and Spinal Cord Injury: 2013 update. Neurosurgery 2013, 60, 82–91. [Google Scholar]
- Ryken, T.C.; Hurlbert, R.J.; Hadley, M.N.; Aarabi, B.; Dhall, S.S.; Gelb, D.E.; Rozzelle, C.J.; Theodore, N.; Walters, B.C. The acute cardiopulmonary management of patients with cervical spinal cord injuries. Neurosurgery 2013, 72, 84–92. [Google Scholar] [CrossRef]
- Bracken, M.B.; Shepard, M.J.; Holford, T.R.; Leo-Summers, L.; Aldrich, E.F.; Fazl, M.; Fehlings, M.; Herr, D.L.; Hitchon, P.W.; Marshall, L.F.; et al. Administration of methylprednisolone for 24 or 48 h or tirilazad mesylate for 48 h in the treatment of acute spinal cord injury. Results of the Third National Acute Spinal Cord Injury Randomized Controlled Trial. National Acute Spinal Cord Injury Study. JAMA 1997, 277, 1597–1604. [Google Scholar] [CrossRef]
- Bracken, M.B.; Collins, W.F.; Freeman, D.F.; Shepard, M.J.; Wagner, F.W.; Silten, R.M.; Hellenbrand, K.G.; Ransohoff, J.; Hunt, W.E.; Perot, P.L., Jr. Efficacy of methylprednisolone in acute spinal cord injury. JAMA 1984, 251, 45–52. [Google Scholar] [CrossRef]
- Eck, J.C.; Nachtigall, D.; Humphreys, S.C.; Hodges, S.D. Questionnaire survey of spine surgeons on the use of methylprednisolone for acute spinal cord injury. Spine 2006, 20, 250–253. [Google Scholar] [CrossRef] [PubMed]
- Hurlbert, R.J.; Hadley, M.N.; Walters, B.C.; Aarabi, B.; Dhall, S.S.; Gelb, D.E.; Rozzelle, C.J.; Ryken, T.C.; Theodore, N. Pharmacological therapy for acute spinal cord injury. Neurosurgery 2013, 72, 93–105. [Google Scholar] [CrossRef]
- Fehlings, M.G.; Wilson, J.R.; Tetreault, L.A.; Aarabi, B.; Anderson, P.; Arnold, P.M.; Brodke, D.S.; Burns, A.S.; Chiba, K.; Dettori, R.; et al. A Clinical Practice Guideline for the Management of Patients With Acute Spinal Cord Injury: Recommendations on the Use of Methylprednisolone Sodium Succinate. Glob. Spine J. 2017, 7, 203S–211S. [Google Scholar] [CrossRef] [PubMed]
- Frangen, T.M.; Ruppert, S.; Muhr, G.; Schinkel, C. The beneficial effects of early stabilization of thoracic spine fractures depend on trauma severity. J. Trauma 2010, 68, 1208–1212. [Google Scholar] [CrossRef] [PubMed]
- Kerwin, A.J.; Frykberg, E.R.; Schinco, M.A.; Griffen, M.M.; Murphy, T.; Tepas, J.J. The effect of early spine fixation on non-neurologic outcome. J. Trauma 2005, 58, 15–21. [Google Scholar] [CrossRef]
- Zhou, Y.; Wang, X.; Wang, S.; Zheng, H.; Shi, X.; Chai, X. Effects of simple posterior decompression and fusion fixation as treatment strategy for complete thoracic fracture dislocation. Chin. J. Trauma 2017, 33, 890–895. [Google Scholar]
- Phan, K.; Rao, P.J.; Mobbs, R.J. Percutaneous versus open pedicle screw fixation for treatment of thoracolumbar fractures: Systematic review and meta-analysis of comparative studies. Clin. Neurol. Neurosurg. 2015, 135, 85–92. [Google Scholar] [CrossRef]
- Wood, K.B.; Li, W.; Lebl, D.R.; Ploumis, A. Management of thoracolumbar spine fractures. Spine J. 2014, 14, 145–164. [Google Scholar] [CrossRef] [PubMed]
- Bliemel, C.; Lefering, R.; Buecking, B.; Frink, M.; Struewer, J.; Krueger, A.; Ruchholtz, S.; Frangen, T.M. Early or delayed stabilization in severely injured patients with spinal fractures? Current surgical objectivity according to the Trauma Registry of DGU: Treatment of spine injuries in polytrauma patients. J. Trauma Acute Care Surg. 2014, 76, 366–373. [Google Scholar] [CrossRef]
- Schinkel, C.; Frangen, T.M.; Kmetic, A.; Andress, H.J.; Muhr, G. German Trauma Registry. Timing of thoracic spine stabilization in trauma patients: Impact on clinical course and outcome. J. Trauma 2006, 61, 156–160. [Google Scholar] [CrossRef] [PubMed]
- Bellabarba, C.; Fisher, C.; Chapman, J.R.; Dettori, J.R.; Norvell, D.C. Does early fracture fixation of thoracolumbar spine fractures decrease morbidity or mortality? Spine 2010, 35, S138–S145. [Google Scholar] [CrossRef] [PubMed]
- Kato, S.; Murray, J.C.; Kwon, B.K.; Schroeder, G.D.; Vaccaro, A.R.; Fehlings, M.G. Does Surgical Intervention or Timing of Surgery Have an Effect on Neurological Recovery in the Setting of a Thoracolumbar Burst Fracture? J. Orthop. Trauma 2017, 31, S38–S43. [Google Scholar] [CrossRef] [PubMed]
- Fehlings, M.G.; Vaccaro, A.; Wilson, J.R.; Singh, A.; Cadotte, D.; Harrop, J.S.; Aarabi, B.; Shaffrey, C.; Dvorak, M.; Fisher, C.; et al. Early versus delayed decompression for traumatic cervical spinal cord injury: Results of the surgical timing in acute spinal cord injury study (STASCIS). PLoS ONE 2012, e32037. [Google Scholar] [CrossRef]
- Grassner, L.; Wutte, C.; Klein, B.; Mach, O.; Riesner, S.; Panzer, S.; Vogel, M.; Buhre, V.; Strowitzki, M.; Vastmns, J.; et al. Early Decompression (<8 h) after Traumatic Cervical Spinal Cord Injury Improves Functional Outcome as Assessed by Spinal Cord Independence Measure after One Year. J. Neurotrauma 2016, 15, 1658–1666. [Google Scholar]
- Jug, M.; Kejžar, N.; Vesel, M.; Al Mawed, S.; Dobravec, M.; Herman, S.; Bajrovic, F.F. Neurological Recovery after Traumatic Cervical Spinal Cord Injury Is Superior if Surgical Decompression and Instrumented Fusion Are Performed within 8 Hours versus 8 to 24 Hours after Injury: A Single Center Experience. J. Neurotrauma 2015, 32, 1385–1392. [Google Scholar] [CrossRef] [PubMed]
- Mattiassich, G.; Gollwitzer, M.; Gaderer, F.; Blocher, M.; Osti, M.; Lill, M.; Ortmaier, R.; Haider, T.; Hitzl, W.; Resch, H.; et al. Functional Outcomes in Individuals Undergoing Very Early (<5 h) and Early (5-24 h) Surgical Decompression in Traumatic Cervical Spinal Cord Injury: Analysis of Neurological Improvement from the Austrian Spinal Cord Injury Study. J. Neurotrauma 2017, 34, 3362–3371. [Google Scholar] [PubMed]
- Ter Wengel, P.V.; De Witt Hamer, P.C.; Pauptit, J.C.; van der Gaag, N.A.; Oner, F.C.; Vandertop, W.P. Early Surgical Decompression Improves Neurological Outcome after Complete Traumatic Cervical Spinal Cord Injury: A Meta-Analysis. J. Neurotrauma 2018. [Google Scholar] [CrossRef] [PubMed]
- Krishna, V.; Konakondla, S.; Nicholas, J.; Varma, A.; Kindy, M.; Wen, X. Biomaterial-based interventions for neuronal regeneration and functional recovery in rodent model of spinal cord injury: A systematic review. J. Spinal Cord Med. 2013, 36, 174–190. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Alluin, O.; Karimi-Abdolrezaee, S.; Delivet-Mongrain, H.; Leblond, H.; Fehlings, M.G.; Rossignol, S. Kinematic study of locomotor recovery after spinal cord clip compression injury in rats. J. Neurotrauma 2011, 28, 1963–1981. [Google Scholar] [CrossRef] [PubMed]
- Zeman, R.J.; Wen, X.; Ouyang, N.; Rocchio, R.; Shih, L.; Alfieri, A.; Moorthy, C.; Etlinger, J.D. Stereotactic radiosurgery improves locomotor recovery after spinal cord injury in rats. Neurosurgery 2008, 63, 981–987. [Google Scholar] [CrossRef]
- Lee, J.H.; Tigchelaar, S.; Liu, J.; Stammers, A.M.; Streijger, F.; Tetzlaff, W.; Kwon, B.K. Lack of neuroprotective effects of simvastatin and minocycline in a model of cervical spinal cord injury. Exp. Neurol. 2010, 225, 219–230. [Google Scholar] [CrossRef] [PubMed]
- Van Meeteren, N.L.; Eggers, R.; Lankhorst, A.J.; Gispen, W.H.; Hamers, F.P. Locomotor recovery after spinal cord contusion injury in rats is improved by spontaneous exercise. J. Neurotrauma 2003, 20, 1029–1037. [Google Scholar] [CrossRef] [PubMed]
- Metz, G.A.; Curt, A.; van de Meent, H.; Klusman, I.; Schwab, M.E.; Dietz, V. Validation of the weight-drop contusion model in rats: A comparative study of human spinal cord injury. J. Neurotrauma 2000, 17, 1–17. [Google Scholar] [CrossRef]
- Akhtar, A.Z.; Pippin, J.J.; Sandusky, C.B. Animal models in spinal cord injury: A review. Rev. Neurosci. 2008, 19, 47–60. [Google Scholar] [CrossRef] [PubMed]
- Tator, C.H. Review of treatment trials in human spinal cord injury: Issues, difficulties and recommendations. Neurosurgery 2006, 59, 957–982. [Google Scholar] [CrossRef]
- Kwon, B.K.; Okon, E.B.; Plunet, W.; Baptiste, D.; Fouad, K.; Hillyer, J.; Weaver, L.C.; Fehlings, M.G.; Tetzlaff, W. A systematic review of directly applied biologic therapies for acute spinal cord injury. J. Neurotrauma 2011, 28, 1589–1610. [Google Scholar] [CrossRef] [PubMed]
- Dunham, K.A.; Siriphorn, A.; Chompoopong, S.; Floyd, C.L. Characterization of a graded cervical hemicontusion spinal cord injury model in adult male rats. J. Neurotrauma 2010, 27, 2091–2106. [Google Scholar] [CrossRef]
- Stokes, B.T. Experimental spinal cord injury: A dynamic and verifiable injury device. J. Neurotrauma 1992, 9, 129–134. [Google Scholar] [CrossRef]
- Pearse, D.D.; Lo, T.P., Jr.; Cho, K.S.; Lynch, M.P.; Garg, M.S.; Marcillo, A.E.; Sanchez, A.R.; Cruz, Y.; Dietrich, W.D. Histopathological and behavioral characterization of a novel cervical spinal cord displacement contusion injury in the rat. J. Neurotrauma 2005, 22, 680–702. [Google Scholar] [CrossRef]
- Cheriyan, T.; Ryan, D.J.; Weinreb, J.H.; Cheriyan, J.; Paul, J.C.; Lafage, V.; Kirsch, T.; Errico, T.J. Spinal cord injury models: A review. Spinal Cord. 2014, 52, 588–595. [Google Scholar] [CrossRef]
- Gruner, J.A. A monitored contusion model of spinal cord injury in the rat. J. Neurotrauma 1992, 9, 123–128. [Google Scholar] [CrossRef]
- Marcol, W.; Ślusarczyk, W.; Gzik, M.; Larysz-Brysz, M.; Bobrowski, M.; Grynkiewicz-Bylina, B.; Rosicka, P.; Kalita, K.; Węglarz, W.; Barski, J.J.; et al. Air gun impactor—A novel model of graded white matter spinal cord injury in rodents. J. Reconstr. Microsurg. 2012, 28, 561–568. [Google Scholar] [CrossRef] [PubMed]
- Rivlin, A.S.; Tator, C.H. Effect of duration of acute spinal cord compression in a new acute cord injury model in the rat. Surg. Neurol. 1978, 10, 38–43. [Google Scholar] [PubMed]
- Silberstein, M.; McLean, K. Non-contiguous spinal injury: Clinical and imaging features, and postulated mechanism. Paraplegia 1994, 32, 817–823. [Google Scholar] [CrossRef][Green Version]
- Fiford, R.J.; Bilston, L.E.; Waite, P.; Lu, J. A vertebral dislocation model of spinal cord injury in rats. J. Neurotrauma 2004, 21, 451–458. [Google Scholar] [CrossRef] [PubMed]
- Spillmann, A.A.; Amberger, V.R.; Schwab, M.E. High molecular weight protein of human central nervous system myelin inhibits neurite outgrowth: An effect which can be neutralized by the monoclonal antibody IN-1. Eur. J. Neurosci. 2006, 9, 549–555. [Google Scholar] [CrossRef]
- Liu, D.; Xu, G.Y.; Pan, E.; McAdoo, D.J. Neurotoxicity of glutamate at the concentration released upon spinal cord injury. Neuroscience 1999, 93, 1383–1389. [Google Scholar] [CrossRef]
- Bao, F.; DeWitt, D.S.; Prough, D.S.; Liu, D. Peroxynitrite generated in the rat spinal cord induces oxidation and nitration of proteins: Reduction by Mn (III) tetrakis (4–benzoicacid) porphyrin. J. Neurosci. Res. 2002, 71, 220–227. [Google Scholar] [CrossRef]
- Fehlings, M.G.; Tator, C.H. The relationships among the severity of spinal cord injury, residual neurological function, axon counts, and counts of retrogradely labeled neurons after experimental spinal cord injury. Exp. Neurol. 1995, 132, 220–228. [Google Scholar] [CrossRef]
- Bartel, D.P. MicroRNAs: Genomics, biogenesis, mechanism, and function. Cell 2004, 116, 281–297. [Google Scholar] [CrossRef]
- Friedman, R.C.; Farh, K.K.; Burge, C.B.; Bartel, D.P. Most mammalian mRNAs are conserved targets of microRNAs. Genome Res. 2009, 19, 92–105. [Google Scholar] [CrossRef] [PubMed]
- Kosik, K.S. The neuronal microRNA system. Nat. Rev. Neurosci. 2006, 7, 911–920. [Google Scholar] [CrossRef]
- Liu, N.K.; Wang, X.F.; Lu, Q.B.; Xu, X.M. Altered microRNA expression following traumatic spinal cord injury. Exp. Neurol. 2009, 219, 424–429. [Google Scholar] [CrossRef] [PubMed]
- Yunta, M.; Nieto-Díaz, M.; Esteban, F.J.; Caballero-López, M.; Navarro-Ruíz, R.; Reigada, D.; Pita-Thomas, D.W.; del Águila, A.; Muñoz-Galdeano, T.; Maza, R.M. MicroRNA dysregulation in the spinal cord following traumatic injury. PLoS ONE 2012, 7, e34534. [Google Scholar] [CrossRef] [PubMed]
- Redell, J.B.; Liu, Y.; Dash, P.K. Traumatic brain injury alters expression of hippocampal microRNAs: Potential regulators of multiple pathophysiological processes. J. Neurosci. Res. 2009, 87, 1435–1448. [Google Scholar] [CrossRef] [PubMed]
- Lim, L.P.; Lau, N.C.; Garrett-Engele, P.; Grimson, A.; Schelter, J.M.; Castle, J.; Bartel, D.P.; Linsley, P.S.; Johnson, J.M. Microarray analysis shows that some microRNAs downregulate large numbers of target mRNAs. Nature 2005, 433, 769–773. [Google Scholar] [CrossRef]
- Liu, X.J.; Zheng, X.P.; Zhang, R.; Guo, Y.L.; Wang, J.H. Combinatorial effects of miR-20a and miR-29b on neuronal apoptosis induced by spinal cord injury. Int. J. Clin. Exp. Pathol. 2015, 8, 3811–3818. [Google Scholar] [PubMed]
- Arbour, N.; Vanderluit, J.L.; Le Grand, J.N.; Jahani-Asl, A.; Ruzhynsky, V.A.; Cheung, E.C.; Kelly, M.A.; MacKenzie, A.E.; Park, D.S.; Opferman, J.T.; et al. Mcl-1 is a key regulator of apoptosis during CNS development and after DNA damage. J. Neurosci. 2008, 28, 6068–6078. [Google Scholar] [CrossRef]
- Bouillet, P.; Strasser, A. BH3-only proteins—Evolutionarily conserved proapoptotic Bcl-2 family members essential for initiating programmed cell death. J. Cell. Sci. 2002, 115, 1567–1574. [Google Scholar]
- Jee, M.K.; Jung, J.S.; Im, Y.B.; Jung, S.J.; Kang, S.K. Silencing of miR20a is crucial for Ngn1-mediated neuroprotection in injured spinal cord. Hum Gene Ther. 2012, 23, 508–520. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.Z.; Li, L.; Lodish, H.F.; Bartel, D.P. MicroRNAs modulate hematopoietic lineage differentiation. Science 2004, 303, 83–86. [Google Scholar] [CrossRef]
- Johnnidis, J.B.; Harris, M.H.; Wheeler, R.T.; Stehling-Sun, S.; Lam, M.H.; Kirak, O.; Brummelkamp, T.R.; Fleming, M.D.; Camargo, F.D. Regulation of progenitor cell proliferation and granulocyte function by microRNA-223. Nature 2008, 451, 1125–1129. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Huang, Y.; Jia, C.; Li, Y.; Liang, F.; Fu, Q. Administration of antagomir-223 inhibits apoptosis, promotes angiogenesis and functional recovery in rats with spinal cord injury. Cell. Mol. Neurobiol. 2015, 35, 483–491. [Google Scholar] [CrossRef] [PubMed]
- Izumi, B.; Nakasa, T.; Tanaka, N.; Nakanishi, K.; Kamei, N.; Yamamoto, R.; Nakamae, T.; Ohta, R.; Fujioka, Y.; Yamasaki, K.; et al. MicroRNA-223 expression in neutrophils in the early phase of secondary damage after spinal cord injury. Neurosci. Lett. 2011, 492, 114–118. [Google Scholar] [CrossRef] [PubMed]
- Nakanishi, K.; Nakasa, T.; Tanaka, N.; Ishikawa, M.; Yamada, K.; Yamasaki, K.; Kamei, N.; Izumi, B.; Adachi, N.; Miyaki, S.; et al. Responses of microRNAs 124a and 223 following spinal cord injury in mice. Spinal Cord. 2010, 48, 192–196. [Google Scholar] [CrossRef] [PubMed]
- Kaur, P.; Armugam, A.; Jeyaseelan, K. MicroRNAs in Neurotoxicity. J. Toxicol. 2012, 2012, 870150. [Google Scholar] [CrossRef]
- Krichevsky, A.M.; Gabriely, G. miR-21: A small multi-faceted RNA. J. Cell. Mol. Med. 2009, 13, 39–53. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Si, M.L.; Wu, H.; Mo, Y.Y. MicroRNA-21 targets the tumor suppressor gene tropomyosin 1 (TPM1). J. Biol. Chem. 2007, 282, 14328–14336. [Google Scholar] [CrossRef]
- Bhattacharya, B.; Prasad, G.L.; Valverius, E.M.; Salomon, D.S.; Cooper, H.L. Tropomyosins of human mammary epithelial cells: Consistent defects of expression in mammary carcinoma cell lines. Cancer Res. 1990, 50, 2105–2112. [Google Scholar]
- Si, M.L.; Zhu, S.; Wu, H.; Lu, Z.; Wu, F.; Mo, Y.Y. miR-21-mediated tumor growth. Oncogene 2007, 26, 2799–2803. [Google Scholar] [CrossRef]
- Buller, B.; Liu, X.; Wang, X.; Zhang, R.L.; Zhang, L.; Hozeska-Solgot, A.; Chopp, M.; Zhang, Z.G. MicroRNA-21 protects neurons from ischemic death. FEBS J. 2010, 277, 4299–4307. [Google Scholar] [CrossRef] [PubMed]
- Strickland, E.R.; Hook, M.A.; Balaraman, S.; Huie, J.R.; Grau, J.W.; Miranda, R.C. MicroRNA dysregulation following spinal cord contusion: Implications for neural plasticity and repair. Neuroscience 2011, 186, 146–160. [Google Scholar] [CrossRef] [PubMed]
- Corsten, M.F.; Miranda, R.; Kasmieh, R.; Krichevsky, A.M.; Weissleder, R.; Shah, K. MicroRNA-21 knockdown disrupts glioma growth in vivo and displays synergistic cytotoxicity with neural precursor cell delivered S-TRAIL in human gliomas. Cancer Res. 2007, 67, 8994–9000. [Google Scholar] [CrossRef] [PubMed]
- Sathyan, P.; Golden, H.B.; Miranda, R.C. Competing interactions between micro-RNAs determine neural progenitor survival and proliferation after ethanol exposure: Evidence from an ex vivo model of the fetal cerebral cortical neuroepithelium. J. Neurosci. 2007, 27, 8546–8557. [Google Scholar] [CrossRef]
- Bhalala, O.G.; Pan, L.; Sahni, V.; McGuire, T.L.; Gruner, K.; Tourtellotte, W.G.; Kessler, J.A. MicroRNA-21 regulates astrocytic response following spinal cord injury. J. Neurosci. 2012, 32, 17935–17947. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.Z.; Huang, J.H.; Zeng, L.; Wang, G.; Cao, M.; Lu, H.B. Anti-apoptotic effect of microRNA-21 after contusion spinal cord injury in rats. J. Neurotrauma 2013, 30, 1349–1360. [Google Scholar] [CrossRef]
- Strickland, I.T.; Richards, L.; Holmes, F.E.; Wynick, D.; Uney, J.B.; Wong, L.F. Axotomy-induced miR-21 promotes axon growth in adult dorsal root ganglion neurons. PLoS ONE 2011, 6, e23423. [Google Scholar] [CrossRef] [PubMed]
- Calin, G.A.; Dumitru, C.D.; Shimizu, M.; Bichi, R.; Zupo, S.; Noch, E.; Aldler, H.; Rattan, S.; Keating, M.; Rai, K.; et al. Frequent deletions and down-regulation of micro-RNA genes miR15 and miR16 at 13q14 in chronic lymphocytic leukemia. Proc. Natl. Acad. Sci. USA 2002, 99, 15524–15529. [Google Scholar] [CrossRef] [PubMed]
- Finnerty, J.R.; Wang, W.X.; Hébert, S.S.; Wilfred, B.R.; Mao, G.; Nelson, P.T. The miR-15/107 group of microRNA genes: Evolutionary biology, cellular functions, and roles in human diseases. J. Mol. Biol. 2010, 402, 491–509. [Google Scholar] [CrossRef]
- Guo, C.J.; Pan, Q.; Li, D.G.; Sun, H.; Liu, B.W. miR-15b and miR-16 are implicated in activation of the rat hepatic stellate cell: An essential role for apoptosis. J. Hepatol. 2009, 50, 766–778. [Google Scholar] [CrossRef]
- Danial, N.N. BCL-2 family proteins: Critical checkpoints of apoptotic cell death. Clin. Cancer Res. 2007, 13, 7254–7263. [Google Scholar] [CrossRef] [PubMed]
- Eldadah, B.A.; Faden, A.I. Caspase pathways, neuronal apoptosis, and CNS injury. J. Neurotrauma 2000, 17, 811–829. [Google Scholar] [CrossRef] [PubMed]
- Cimmino, A.; Calin, G.A.; Fabbri, M.; Iorio, M.V.; Ferracin, M.; Shimizu, M.; Wojcik, S.E.; Aqeilan, R.I.; Zupo, S.; Dono, M.; et al. miR-15 and miR-16 induce apoptosis by targeting BCL2. Proc. Natl. Acad. Sci. USA 2005, 102, 13944–13949. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Keeler, B.E.; Zhukareva, V.; Houlé, J.D. Cycling exercise affects the expression of apoptosis-associated microRNAs after spinal cord injury in rats. Exp. Neurol. 2010, 226, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Deo, M.; Yu, J.Y.; Chung, K.H.; Tippens, M.; Turner, D.L. Detection of mammalian microRNA expression by in situ hybridization with RNA oligonucleotides. Dev. Dyn. 2006, 235, 2538–2548. [Google Scholar] [CrossRef]
- Lagos-Quintana, M.; Rauhut, R.; Yalcin, A.; Meyer, J.; Lendeckel, W.; Tuschl, T. Identification of tissue-specific microRNAs from mouse. Curr. Biol. 2002, 12, 735–739. [Google Scholar] [CrossRef]
- Makeyev, E.V.; Zhang, J.; Carrasco, M.A.; Maniatis, T. The MicroRNA miR-124 promotes neuronal differentiation by triggering brain-specific alternative pre-mRNA splicing. Mol. Cell. 2007, 27, 435–448. [Google Scholar] [CrossRef] [PubMed]
- Visvanathan, J.; Lee, S.; Lee, B.; Lee, J.W.; Lee, S.K. The microRNA miR-124 antagonizes the anti-neural REST/SCP1 pathway during embryonic CNS development. Genes Dev. 2007, 21, 744–749. [Google Scholar] [CrossRef] [PubMed]
- Louw, A.M.; Kolar, M.K.; Novikova, L.N.; Kingham, P.J.; Wiberg, M.; Kjems, J.; Novikov, L.N. Chitosan polyplex mediated delivery of miRNA-124 reduces activation of microglial cells in vitro and in rat models of spinal cord injury. Nanomedicine 2016, 12, 643–653. [Google Scholar] [CrossRef]
- Wellner, U.; Schubert, J.; Burk, U.C.; Schmalhofer, O.; Zhu, F.; Sonntag, A.; Waldvogel, B.; Vannier, C.; Darling, D.; zur Hausen, A.; et al. The EMT-activator ZEB1 promotes tumorigenicity by repressing stemness-inhibiting microRNAs. Nat. Cell. Biol. 2009, 11, 1487–1495. [Google Scholar] [CrossRef]
- Liu, Y.N.; Yin, J.J.; Abou-Kheir, W.; Hynes, P.G.; Casey, O.M.; Fang, L.; Yi, M.; Stephens, R.M.; Seng, V.; Sheppard-Tillman, H.; et al. MiR-1 and miR-200 inhibit EMT via Slug-dependent and tumorigenesis via Slug-independent mechanisms. Oncogene 2013, 32, 296–306. [Google Scholar] [CrossRef] [PubMed]
- Tellez, C.S.; Juri, D.E.; Do, K.; Bernauer, A.M.; Thomas, C.L.; Damiani, L.A.; Tessema, M.; Leng, S.; Belinsky, S.A. EMT and stem cell-like properties associated with miR-205 and miR-200 epigenetic silencing are early manifestations during carcinogen-induced transformation of human lung epithelial cells. Cancer Res. 2011, 71, 3087–3097. [Google Scholar] [CrossRef]
- Yu, D.S.; Lv, G.; Mei, X.F.; Cao, Y.; Wang, Y.F.; Wang, Y.S.; Bi, Y.L. MiR-200c regulates ROS-induced apoptosis in murine BV-2 cells by targeting FAP-1. Spinal Cord. 2014, 53, 182–189. [Google Scholar] [CrossRef] [PubMed]
- Uittenbogaard, M.; Baxter, K.K.; Chiaramello, A. The neurogenic basic helix-loop-helix transcription factor NeuroD6 confers tolerance to oxidative stress by triggering an antioxidant response and sustaining the mitochondrial biomass. ASN Neuro 2010, 2, e00034. [Google Scholar] [CrossRef] [PubMed]
- Jee, M.K.; Jung, J.S.; Choi, J.I.; Jang, J.A.; Kang, K.S.; Im, Y.B.; Kang, S.K. MicroRNA 486 is a potentially novel target for the treatment of spinal cord injury. Brain 2012, 135, 1237–1252. [Google Scholar] [CrossRef]
- Calin, G.A.; Sevignani, C.; Dumitru, C.D.; Hyslop, T.; Noch, E.; Yendamuri, S.; Shimizu, M.; Rattan, S.; Bullrich, F.; Negrini, M.; et al. Human microRNA genes are frequently located at fragile sites and genomic regions involved in cancers. Proc. Natl. Acad. Sci. USA 2004, 101, 2999–3004. [Google Scholar] [CrossRef]
- Takamizawa, J.; Konishi, H.; Yanagisawa, K.; Tomida, S.; Osada, H.; Endoh, H.; Harano, T.; Yatabe, Y.; Nagino, M.; Nimura, Y.; et al. Reduced expression of the let-7 microRNAs in human lung cancers in association with shortened postoperative survival. Cancer Res. 2004, 64, 3753–3756. [Google Scholar] [CrossRef] [PubMed]
- Asangani, I.A.; Rasheed, S.A.; Nikolova, D.A.; Leupold, J.H.; Colburn, N.H.; Post, S.; Allgayer, H. MicroRNA-21 (miR-21) post-transcriptionally downregulates tumor suppressor Pdcd4 and stimulates invasion, intravasation and metastasis in colorectal cancer. Oncogene 2008, 27, 2128–2136. [Google Scholar] [CrossRef]
- Johnson, S.M.; Grosshans, H.; Shingara, J.; Byrom, M.; Jarvis, R.; Cheng, A.; Labourier, E.; Reinert, K.L.; Brown, D.; Slack, F.J. RAS is regulated by the Let-7 microRNA family. Cell 2005, 120, 635–647. [Google Scholar] [CrossRef] [PubMed]
- Sampson, V.B.; Rong, N.H.; Han, J.; Yang, Q.; Aris, V.; Soteropoulos, P.; Petrelli, N.J.; Dunn, S.P.; Krueger, L.J. MicroRNA Let-7a downregulates MYC and reverts MYC-induced growth in Burkitt lymphoma cells. Cancer Res. 2007, 67, 9762–9770. [Google Scholar] [CrossRef]
- Iliopoulos, D.; Hirsch, H.A.; Struhl, K. An epigenetic switch involving NF-kappaB, Lin28, Let-7 MicroRNA, and IL6 links inflammation to cell transformation. Cell 2009, 139, 693–706. [Google Scholar] [CrossRef] [PubMed]
- Pasquinelli, A.E.; Reinhart, B.J.; Slack, F.; Martindale, M.Q.; Kuroda, M.I.; Maller, B.; Hayward, D.C.; Ball, E.E.; Degnan, B.; Müller, P.; et al. Conservation of the sequence and temporal expression of let-7 heterochronic regulatory RNA. Nature 2000, 408, 86–89. [Google Scholar] [CrossRef]
- Johnson, C.D.; Esquela-Kerscher, A.; Stefani, G.; Byrom, M.; Kelnar, K.; Ovcharenko, D.; Wilson, M.; Wang, X.; Shelton, J.; Shingara, J.; et al. The let-7 microRNA represses cell proliferation pathways in human cells. Cancer Res. 2007, 67, 7713–7722. [Google Scholar] [CrossRef] [PubMed]
- Citron, B.A.; Arnold, P.M.; Sebastian, C.; Qin, F.; Malladi, S.; Ameenuddin, S.; Landis, M.E.; Festoff, B.W. Rapid upregulation of caspase-3 in rat spinal cord after injury: mRNA, protein, and cellular localization correlates with apoptotic cell death. Exp. Neurol. 2000, 166, 213–226. [Google Scholar] [CrossRef] [PubMed]
- Casha, S.; Yu, W.R.; Fehlings, M.G. Oligodendroglial apoptosis occurs along degenerating axons and is associated with FAS and p75 expression following spinal cord injury in the rat. Neuroscience 2001, 103, 203–218. [Google Scholar] [CrossRef]
- Taganov, K.D.; Boldin, M.P.; Chang, K.J.; Baltimore, D. NF-kappaB-dependent induction of microRNA miR-146, an inhibitor targeted to signaling proteins of innate immune responses. Proc. Natl. Acad. Sci. USA 2006, 103, 12481–12486. [Google Scholar] [CrossRef]
- Chen, J.F.; Mandel, E.M.; Thomson, J.M.; Wu, Q.; Callis, T.E.; Hammond, S.M.; Conlon, F.L.; Wang, D.Z. The role of microRNA-1 and microRNA-133 in skeletal muscle proliferation and differentiation. Nat. Genet. 2006, 38, 228–233. [Google Scholar] [CrossRef]
- Ivey, K.N.; Muth, A.; Arnold, J.; King, F.W.; Yeh, R.F.; Fish, J.E.; Hsiao, E.C.; Schwartz, R.J.; Conklin, B.R.; Bernstein, H.S.; et al. MicroRNA regulation of cell lineages in mouse and human embryonic stem cells. Cell Stem Cell 2008, 2, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Strickland, E.R.; Woller, S.A.; Hook, M.A.; Grau, J.W.; Miranda, R.C. The association between spinal cord trauma-sensitive miRNAs and pain sensitivity, and their regulation by morphine. Neurochem. Int. 2014, 77, 40–49. [Google Scholar] [CrossRef]
- Stahlhut, C.; Suárez, Y.; Lu, J.; Mishima, Y.; Giraldez, A.J. miR-1 and miR-206 regulate angiogenesis by modulating VegfA expression in zebrafish. Development 2012, 139, 4356–4365. [Google Scholar] [CrossRef]
- Katada, T.; Ishiguro, H.; Kuwabara, Y.; Kimura, M.; Mitui, A.; Mori, Y.; Ogawa, R.; Harata, K.; Fujii, Y. microRNA expression profile in undifferentiated gastric cancer. Int. J. Oncol. 2009, 34, 537–542. [Google Scholar] [PubMed]
- Gaur, A.; Jewell, D.A.; Liang, Y.; Ridzon, D.; Moore, J.H.; Chen, C.; Ambros, V.R.; Israel, M.A. Characterization of microRNA expression levels and their biological correlates in human cancer cell lines. Cancer Res. 2007, 67, 2456–2468. [Google Scholar] [CrossRef] [PubMed]
- Bandrés, E.; Cubedo, E.; Agirre, X.; Malumbres, R.; Zárate, R.; Ramirez, N.; Abajo, A.; Navarro, A.; Moreno, I.; Monzó, M.; et al. Identification by Real-time PCR of 13 mature microRNAs differentially expressed in colorectal cancer and non-tumoral tissues. Mol Cancer 2006, 19, 29. [Google Scholar] [CrossRef] [PubMed]
- Ferretti, E.; De Smaele, E.; Po, A.; Di Marcotullio, L.; Tosi, E.; Espinola, M.S.; Di Rocco, C.; Riccardi, R.; Giangaspero, F.; Farcomeni, A.; et al. MicroRNA profiling in human medulloblastoma. Int. J. Cancer 2009, 124, 568–577. [Google Scholar] [CrossRef] [PubMed]
- Bottoni, A.; Zatelli, M.C.; Ferracin, M.; Tagliati, F.; Piccin, D.; Vignali, C.; Calin, G.A.; Negrini, M.; Croce, C.M.; Degli Uberti, E.C. Identification of differentially expressed microRNAs by microarray: A possible role for microRNA genes in pituitary adenomas. J. Cell Physiol. 2007, 210, 370–377. [Google Scholar] [CrossRef]
- Huang, Y.W.; Liu, J.C.; Deatherage, D.E.; Luo, J.; Mutch, D.G.; Goodfellow, P.J.; Miller, D.S.; Huang, T.H. Epigenetic repression of microRNA-129-2 leads to overexpression of SOX4 oncogene in endometrial cancer. Cancer Res. 2009, 69, 9038–9046. [Google Scholar] [CrossRef]
- Dyrskjøt, L.; Ostenfeld, M.S.; Bramsen, J.B.; Silahtaroglu, A.N.; Lamy, P.; Ramanathan, R.; Fristrup, N.; Jensen, J.L.; Andersen, C.L.; Zieger, K.; et al. Genomic profiling of microRNAs in bladder cancer: miR-129 is associated with poor outcome and promotes cell death in vitro. Cancer Res. 2009, 69, 4851–4860. [Google Scholar] [CrossRef]
- Bhalala, O.G.; Srikanth, M.; Kessler, J.A. The emerging roles of microRNAs in CNS injuries. Nat. Rev. Neurol. 2013, 9, 328–339. [Google Scholar] [CrossRef]
- Wu, J.; Qian, J.; Li, C.; Kwok, L.; Cheng, F.; Liu, P.; Perdomo, C.; Kotton, D.; Vaziri, C.; Anderlind, C.; et al. miR-129 regulates cell proliferation by downregulating Cdk6 expression. Cell Cycle 2010, 9, 1809–1818. [Google Scholar] [CrossRef]
- Zubakov, D.; Boersma, A.W.; Choi, Y.; van Kuijk, P.F.; Wiemer, E.A.; Kayser, M. MicroRNA markers for forensic body fluid identification obtained from microarray screening and quantitative RT-PCR confirmation. Int. J. Leg. Med. 2010, 124, 217–226. [Google Scholar] [CrossRef]
- Hanson, E.K.; Lubenow, H.; Ballantyne, J. Identification of forensically relevant body fluids using a panel of differentially expressed microRNAs. Anal. Biochem. 2009, 387, 303–314. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, J.; Wei, W.; Zhou, D.; Luo, H.; Chen, X.; Hou, Y. Identification of Saliva Using MicroRNA Biomarkers for Forensic Purpose. J. Forensic Sci. 2015, 60, 702–706. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Luo, H.; Pan, X.; Liao, M.; Hou, Y. A model for data analysis of microRNA expression in forensic body fluid identification. Forensic Sci. Int. Genet. 2012, 6, 419–423. [Google Scholar] [CrossRef] [PubMed]
- Juusola, J.; Ballantyne, J. mRNA profiling for body fluid identification by multiplex quantitative RT-PCR. J. Forensic Sci. 2007, 52, 1252–1262. [Google Scholar] [CrossRef] [PubMed]
- Nussbaumer, C.; Gharehbaghi-Schnell, E.; Korschineck, I. Messenger RNA profiling: A novel method for body fluid identification by real-time PCR. Forensic Sci. Int. 2006, 157, 181–186. [Google Scholar] [CrossRef]
- Haas, C.; Klesser, B.; Maake, C.; Bär, W.; Kratzer, A. mRNA profiling for body fluid identification by reverse transcription endpoint PCR and realtime PCR. Forensic Sci. Int. Genet. 2009, 3, 80–88. [Google Scholar] [CrossRef]
- Pinchi, E.; Frati, A.; Cipolloni, L.; Aromatario, M.; Gatto, V.; La Russa, R.; Pesce, A.; Santurro, A.; Fraschetti, F.; Frati, P.; et al. Clinical-pathological study on β-APP, IL-1β, GFAP, NFL, Spectrin II, 8OHdG, TUNEL, miR-21, miR-16, miR-92 expressions to verify DAI-diagnosis, grade and prognosis. Sci. Rep. 2018, 8, 2387. [Google Scholar] [CrossRef]
- Bedreag, O.H.; Sandesc, D.; Chiriac, S.D.; Rogobete, A.F.; Cradigati, A.C.; Sarandan, M.; Dumache, R.; Nartita, R.; Papurica, M. The Use of Circulating miRNAs as Biomarkers for Oxidative Stress in Critically Ill Polytrauma Patients. Clin. Lab. 2016, 62, 263–274. [Google Scholar] [CrossRef]
- Sun, T.Y.; Chen, X.R.; Liu, Z.L.; Zhao, L.L.; Jiang, Y.X.; Qu, G.Q.; Wang, R.S.; Huang, S.Z.; Liu, L. Expression profiling of MicroRNAs in hippocampus of rats following traumatic brain injury. J. Huazhong Univ. Sci. Technol. Med. Sci. 2014, 34, 548–553. [Google Scholar] [CrossRef]
- Huang, C.; Xiao, X.; Chintagari, N.R.; Breshears, M.; Wang, Y.; Liu, L. MicroRNA and mRNA expression profiling in rat acute respiratory distress syndrome. BMC Med. Genom. 2014, 7, 46. [Google Scholar] [CrossRef]
- Frati, A.; Cerretani, D.; Fiaschi, A.I.; Frati, P.; Gatto, V.; La Russa, R.; Pesce, A.; Pinchi, E.; Santurro, A.; Fraschetti, F.; et al. Diffuse Axonal Injury and Oxidative Stress: A Comprehensive Review. Int. J. Mol. Sci. 2017, 18, 2600. [Google Scholar] [CrossRef] [PubMed]
- Zhao, P.; Wang, S.; Zhou, Y.; Zheng, H.; Zhao, G. MicroRNA-185 regulates spinal cord injuries induced by thoracolumbar spine compression fractures by targeting transforming growth factor-β. Exp. Ther. Med. 2017, 13, 1127–1132. [Google Scholar] [CrossRef] [PubMed]
- Corradini, B.; Alù, M.; Radheshi, E.; Gabbolini, V.; Ferrari, F.; Santunione, A.L.; Silingardi, E. Estimation of the time of death through the analysis of clock miRNAs expression in blood and vitreous humor. Forensic Sci. Int. Genet. Suppl. Ser. 2015, 5, e204–e206. [Google Scholar] [CrossRef]
- Yu, S.; Na, J.Y.; Lee, Y.J.; Kim, K.T.; Park, J.T.; Kim, H.S. Forensic application of microRNA-706 as a biomarker for drowning pattern identification. Forensic Sci. Int. 2015, 255, 96–101. [Google Scholar] [CrossRef]
- Odriozola, A.; Riancho, J.A.; de la Vega, R.; Agudo, G.; García-Blanco, A.; de Cos, E.; Fernández, F.; Sañudo, C.; Zarrabeitia, M.T. miRNA analysis in vitreous humor to determine the time of death: A proof-of-concept pilot study. Int. J. Legal Med. 2013, 127, 573–578. [Google Scholar] [CrossRef] [PubMed]
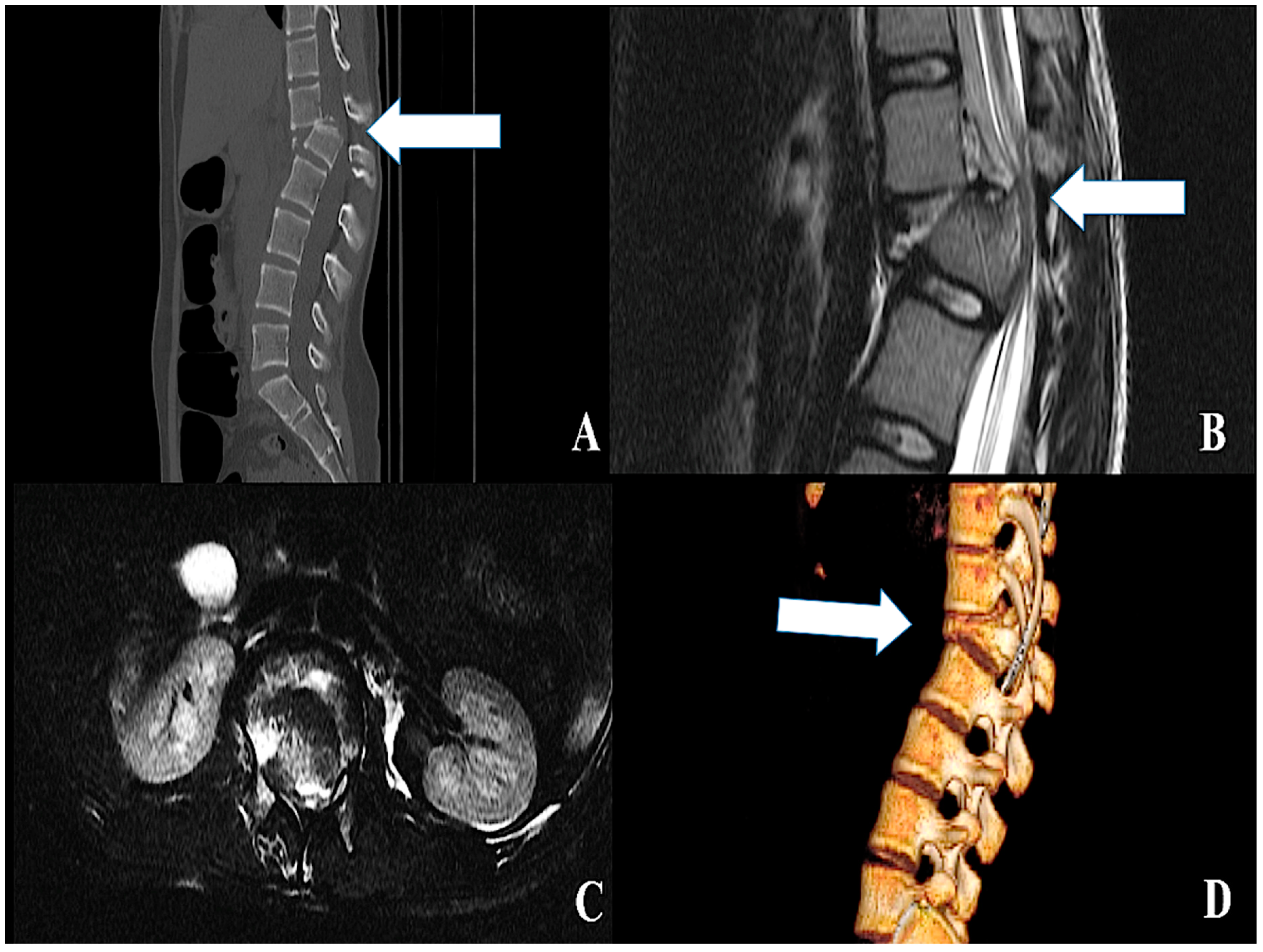
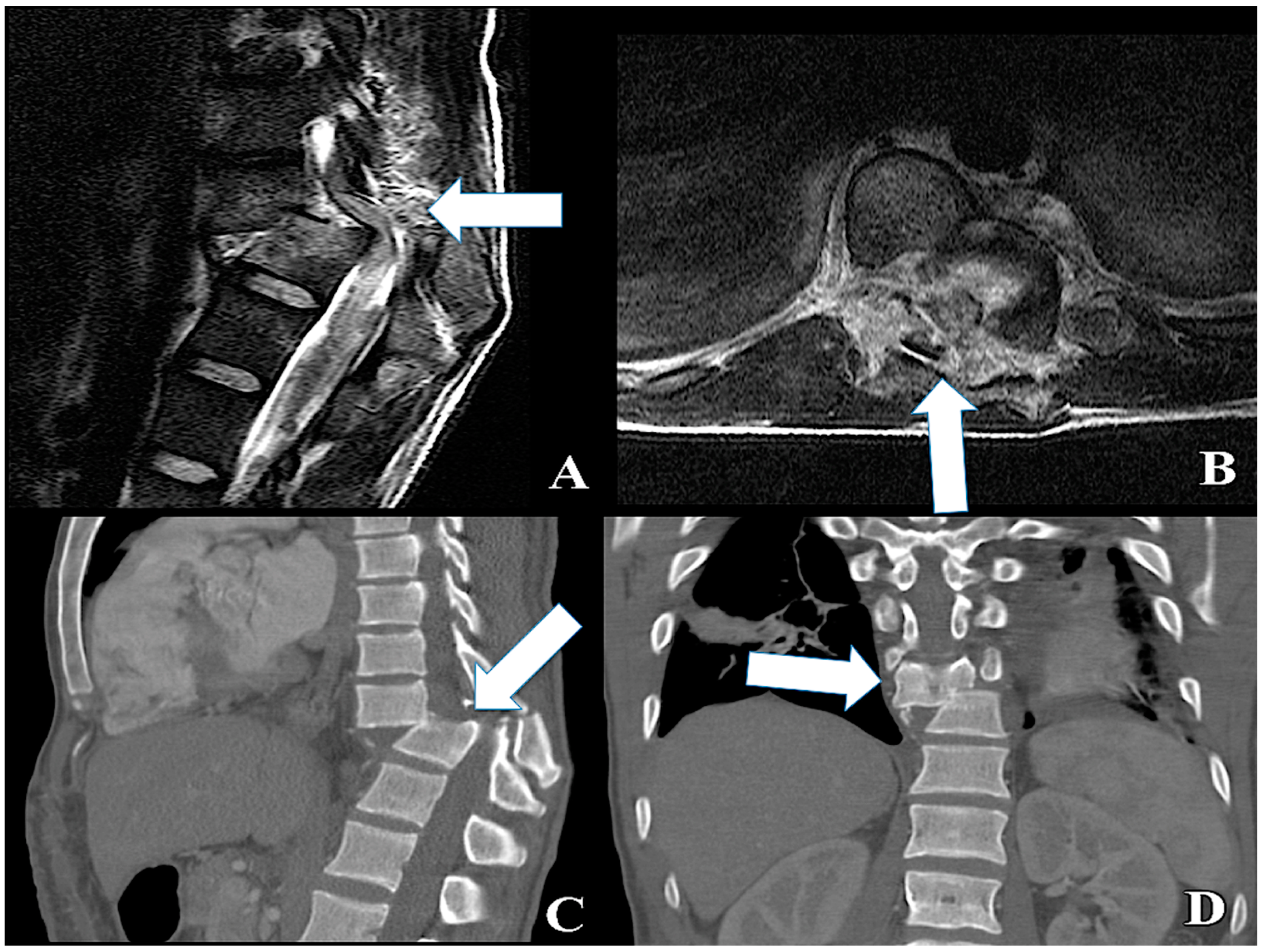

| ID | Model | Expression | Experimental Setup | Method | Site of Injury | Targets | References |
|---|---|---|---|---|---|---|---|
| mir20a mir29b | Adult female C57BL/6 mice | mir20 upregulated mir-29b downregulated | Injection of mir20a and mir-29b in two animal groups of during SCI | Contusive | T10 | downregulation of antiapoptotic myeloid cell leukemia sequence-1 (Mcl-1) and up-regulating proapoptotic BH3-only proteins. | [109] |
| mir-223 | Adult male Sprague Dawley rats | upregulated | 1, 3, 7, and 14 days after SCI | Contusive | T8 | the injection of antagomir-223 reduced Bax and caspase-3 expression levels, ultimately reducing cell apoptosis | [115] |
| Male C57BL/6 mice | upregulated | 12 h after SCI | Compressing the cord laterally from both sides for 10 s with a number 5 forceps | T11–12 | miRNA-223 may reflect inflammatory responses | [117] | |
| Adult male C57BL/6 mice | upregulated | from 6 to 12 h after SCI | Compressing the cord laterally from both sides for 10 s with a number 5 forceps | T11 | miR-223 is expressed in neutrophils that relate to the inflammation in the epicenter after SCI, and inflammatory cytokines were also highly expressed within the same range. | [116] | |
| mir-21 | Adult female SD rats | upregulated | 4 h, 1 day, and 7 days after SCI | Contusive | T10 | Inflammation, oxidation and apoptosis | [105] |
| Adult female Wistar rats | upregulated | 1, 3, and 7 days after SCI | Contusive | T8 | TPM1, PTEN [148], PDCD4 [98], proapoptotic [149] | [106] | |
| Male Sprague Dawley rats | upregulated | 4 and 14 days after SCI | Contusive | T12–T13 | Suppression of miR21 has been shown to cause apoptosis in both cortical progenitor cells and gliomas | [124] | |
| mir-15 mir-16 | Adult female Sprague Dawley rats | downregulated | 12 h after SCI | Compressing the cord laterally from both sides for 10 s with a number 5 forceps | T9–T10 | Target genes: proapoptotic (decreased PTEN, PDCD4 and RAS mRNA) and antiapoptotic (increased Bcl-2 mRNA). Down regulation of mRNA for caspase-7 and caspase-9 and reduced levels of caspase-7 protein. | [136] |
| mir-124 | Male C57BL/6 mice | downregulated | 12 h after SCI | Compressing the cord laterally from both sides for 10 s with a number 5 forceps | T11–12 | reduce the activation of microglial cells, reducing MHC-II, TNFa and ROS production in bone marrow derived macrophages | [117] |
| mir-486 | Adult female ICR mice | upregulated | 0, 1, 2, 3 and 7 days after SCI | Transection | T11 | miR-486 targets NeuroD6 and reflects apoptosis | [147] |
| mir-96 mir146a | Adult female SD rats | upregulated | 4 h, 1 day, and 7 days after SCI | Contusive | T10 | apoptosis through the concomitant increase in expression of the proapoptotic proteins caspase3 | [105] |
| mir-107 | Adult female SD rats | upregulated | 4 h, 1 day, and 7 days after SCI | Contusive | T10 | Apoptosis | [105] |
| mir-1 | Adult female SD rats | upregulation | 4 h, 1 day, and 7 days after SCI | Contusive | T10 | Inflammation, oxidation and apoptosis | [105] |
| mir-129 | Male Sprague Dawley rats | downregulated | 4 and 14 d after SCI | Contusive | T12–T13 | cell cycle, cell proliferation, cell differentiation | [124] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pinchi, E.; Frati, A.; Cantatore, S.; D’Errico, S.; La Russa, R.; Maiese, A.; Palmieri, M.; Pesce, A.; Viola, R.V.; Frati, P.; et al. Acute Spinal Cord Injury: A Systematic Review Investigating miRNA Families Involved. Int. J. Mol. Sci. 2019, 20, 1841. https://doi.org/10.3390/ijms20081841
Pinchi E, Frati A, Cantatore S, D’Errico S, La Russa R, Maiese A, Palmieri M, Pesce A, Viola RV, Frati P, et al. Acute Spinal Cord Injury: A Systematic Review Investigating miRNA Families Involved. International Journal of Molecular Sciences. 2019; 20(8):1841. https://doi.org/10.3390/ijms20081841
Chicago/Turabian StylePinchi, Enrica, Alessandro Frati, Santina Cantatore, Stefano D’Errico, Raffaele La Russa, Aniello Maiese, Mauro Palmieri, Alessandro Pesce, Rocco Valerio Viola, Paola Frati, and et al. 2019. "Acute Spinal Cord Injury: A Systematic Review Investigating miRNA Families Involved" International Journal of Molecular Sciences 20, no. 8: 1841. https://doi.org/10.3390/ijms20081841
APA StylePinchi, E., Frati, A., Cantatore, S., D’Errico, S., La Russa, R., Maiese, A., Palmieri, M., Pesce, A., Viola, R. V., Frati, P., & Fineschi, V. (2019). Acute Spinal Cord Injury: A Systematic Review Investigating miRNA Families Involved. International Journal of Molecular Sciences, 20(8), 1841. https://doi.org/10.3390/ijms20081841







