Anti-Apoptotic Effects of Recombinant Human Hepatocyte Growth Factor on Hepatocytes Were Associated with Intrahepatic Hemorrhage Suppression Indicated by the Preservation of Prothrombin Time
Abstract
:1. Introduction
2. Results
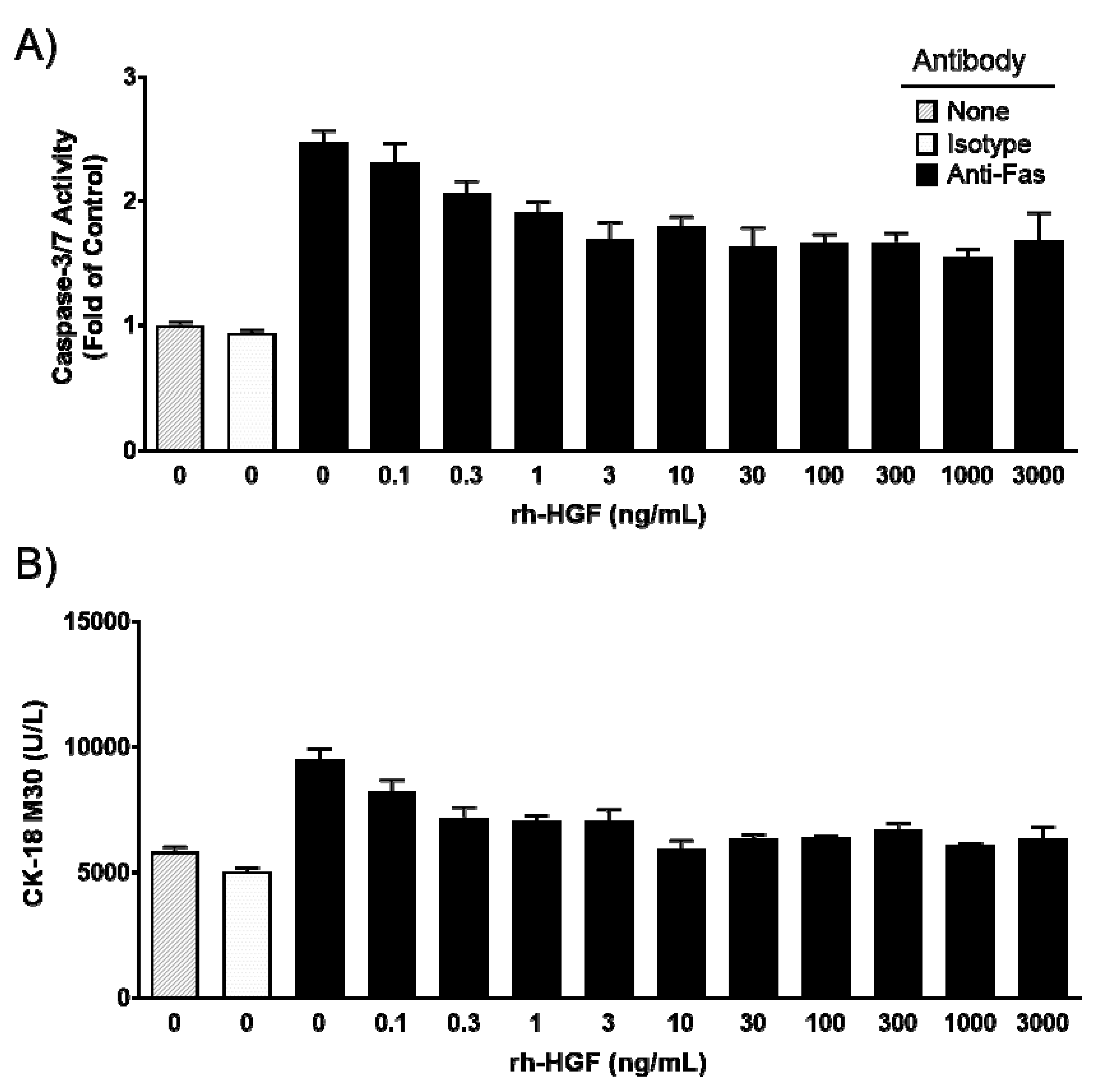
2.1. Recombinant Human Hepatocyte Growth Factor (rh-HGF) Inhibited Fas-Induced Apoptosis in Primary Cultured Human Hepatocytes
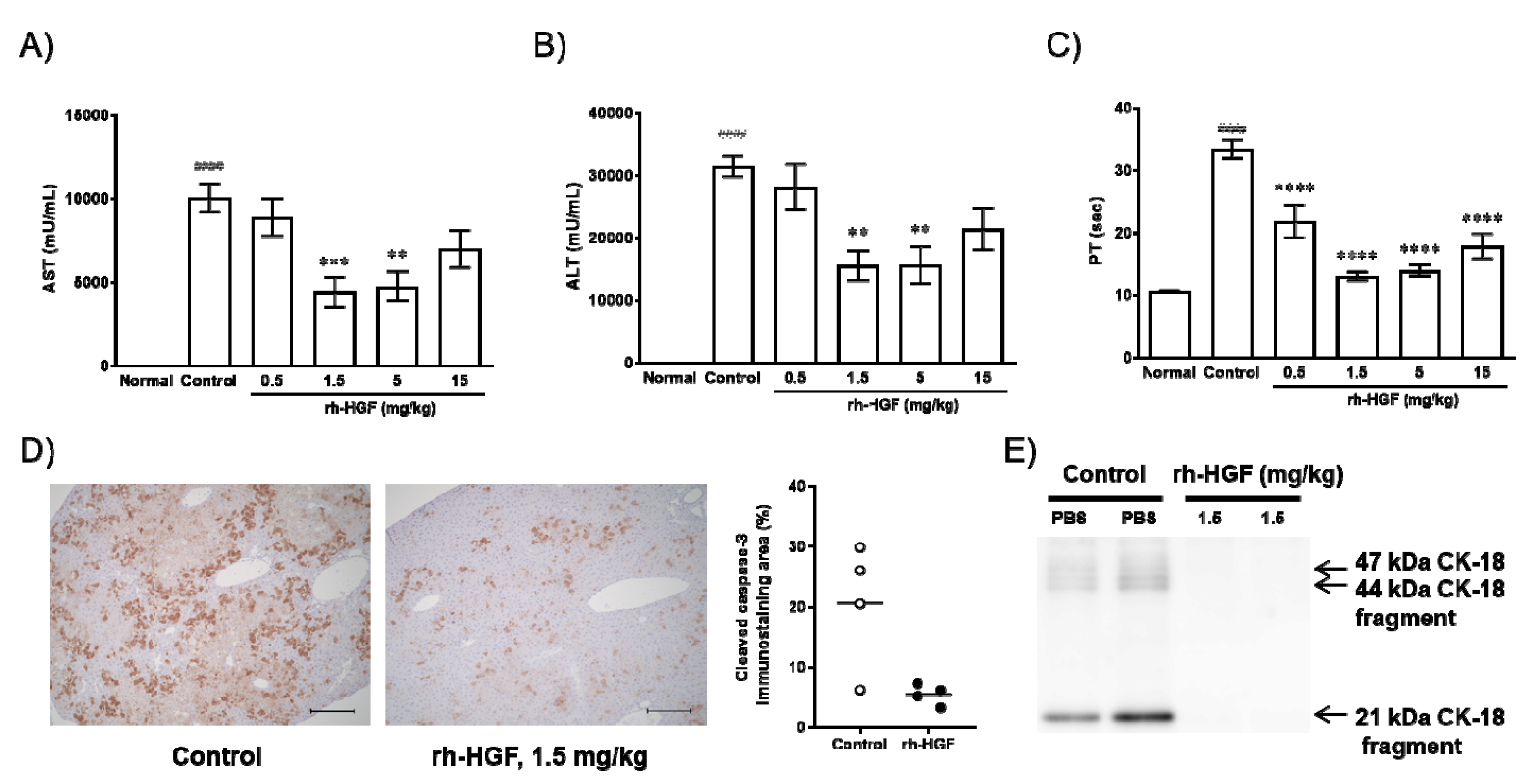
2.2. rh-HGF Suppressed Mouse Acute Liver Failure (ALF) via Anti-Apoptotic Effects and Preserved Prothrombin Time (PT)
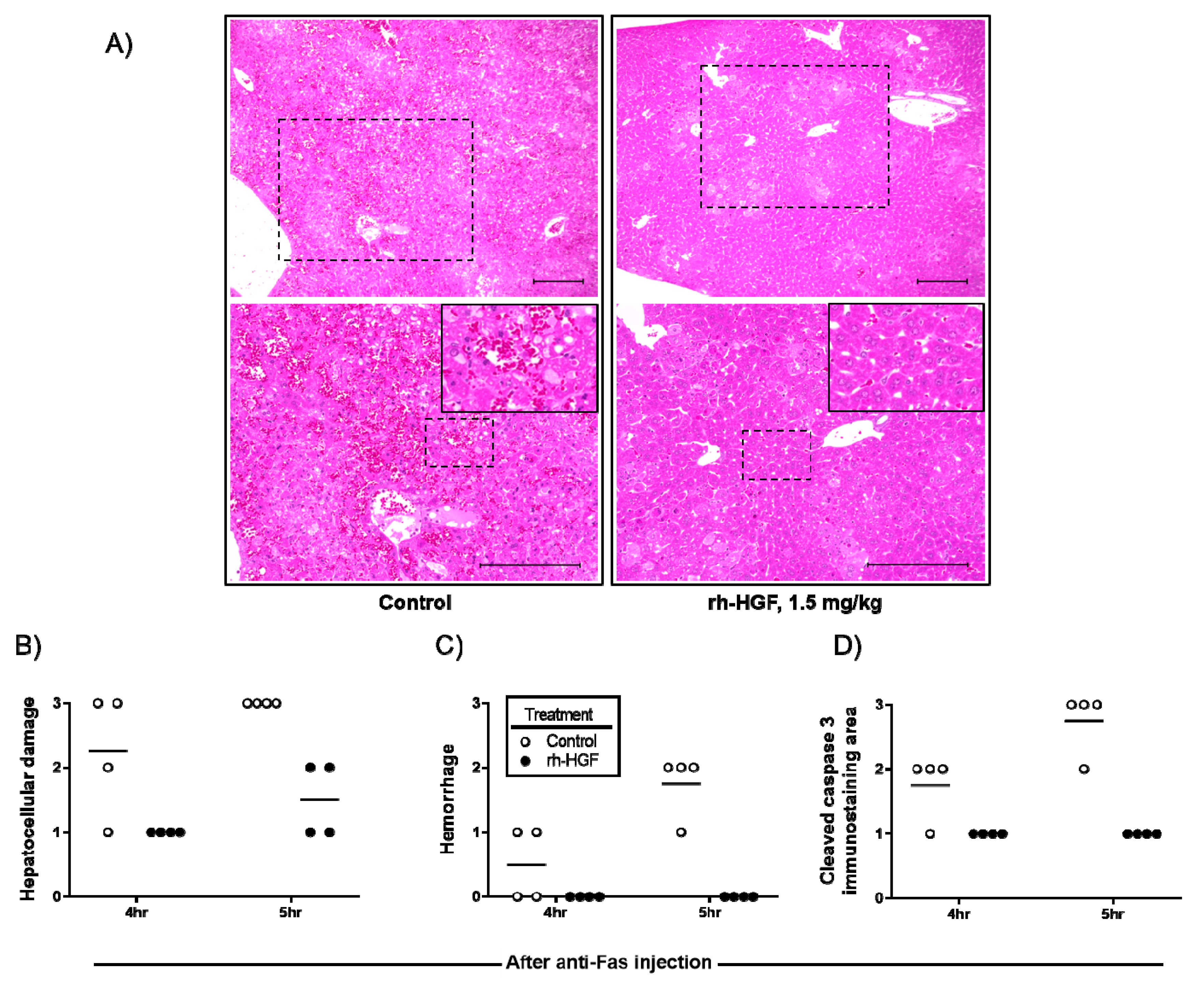
2.3. rh-HGF Treatment Reduced Hepatocellular Damage and Prevented Diffuse Hemorrhage in the Liver
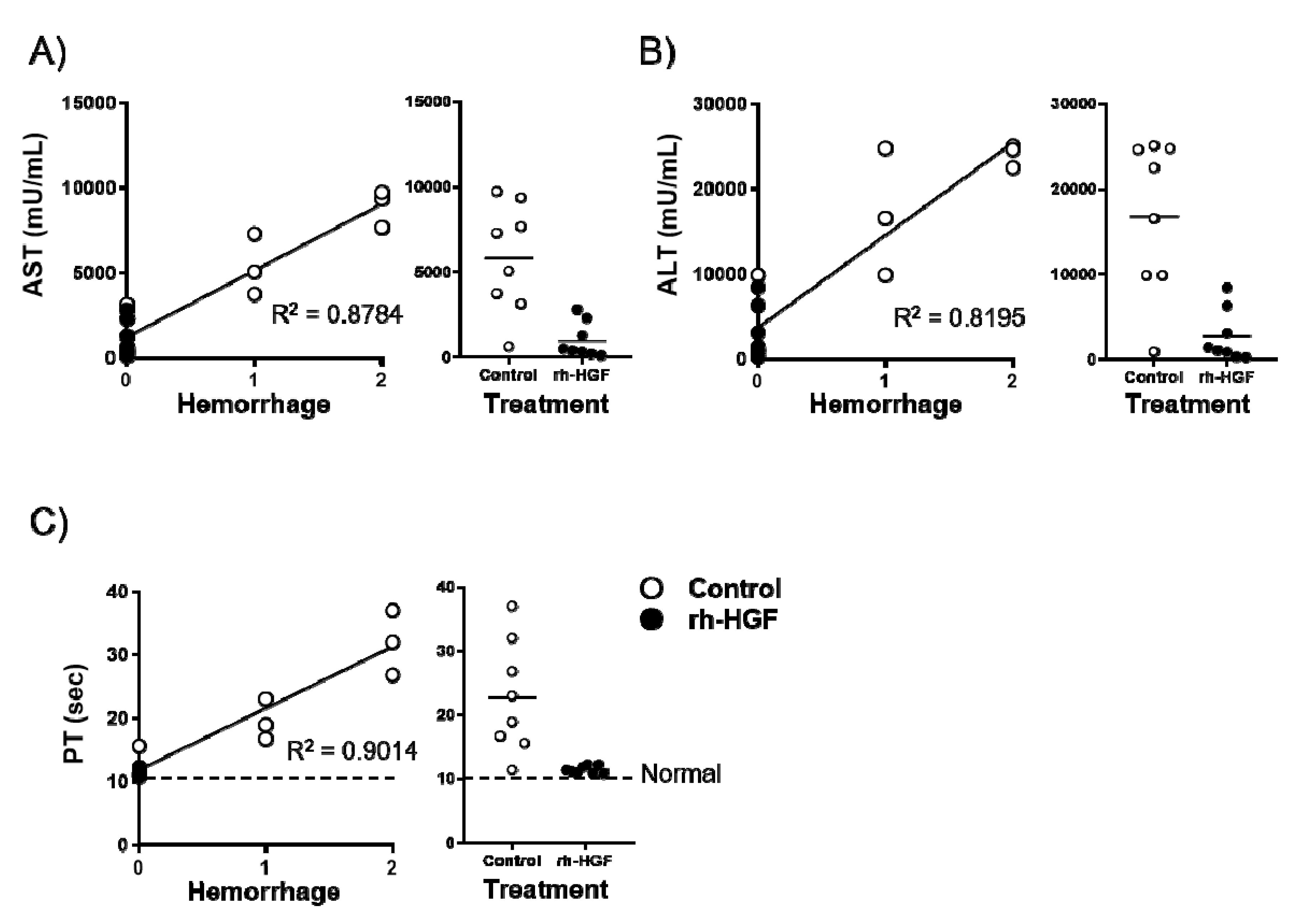
2.4. Intrahepatic Hemorrhage Suppression was Well Correlated with PT Preservation
3. Discussion
4. Materials and Methods
4.1. Recombinant Human HGF
4.2. In vitro Human Hepatocyte Injury Model
4.3. Mouse ALF Model
4.4. Histopathological Analysis and Immunohistochemistry
4.5. Western Blot Analysis of Serum CK-18 in Mice
4.6. Statistics
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| HGF | Hepatocyte growth factor |
| ALF | Acute liver failure |
| CK-18 | Cytokeratin 18 |
| PT | Prothrombin time |
| INR | International normalized ratio |
| rh-HGF | Recombinant human HGF |
| IgG | Immunoglobulin G |
| AST | Aspartate transaminase |
| ALT | Alanine transaminase |
| PBS | Phosphate-buffered saline |
| hr | Hour |
| AGE | Advanced glycation end products |
| ROS | Reactive oxygen species |
| PI3K | Phosphatidylinositol 3-kinase |
| Akt | Protein kinase B |
| GMP | Good manufacturing practices |
| RLU | Relative luminescent unit |
| HE | Hematoxylin-eosin |
References
- Gohda, E.; Tsubouchi, H.; Nakayama, H.; Hirono, S.; Takahashi, K.; Koura, M.; Hashimoto, S.; Daikuhara, Y. Human hepatocyte growth factor in plasma from patients with fulminant hepatic failure. Exp. Cell Res. 1986, 166, 139–150. [Google Scholar] [CrossRef]
- Gohda, E.; Tsubouchi, H.; Nakayama, H.; Hirono, S.; Sakiyama, O.; Takahashi, K.; Miyazaki, H.; Hashimoto, S.; Daikuhara, Y. Purification and partial characterization of hepatocyte growth factor from plasma of a patient with fulminant hepatic failure. J. Clin. Investig. 1988, 81, 414–419. [Google Scholar] [CrossRef] [PubMed]
- Tsubouchi, H.; Hirono, S.; Gohda, E.; Nakayama, H.; Takahashi, K.; Sakiyama, O.; Miyazaki, H.; Sugihara, J.; Tomita, E.; Muto, Y.; et al. Clinical significance of human hepatocyte growth factor in blood from patients with fulminant hepatic failure. Hepatology 1989, 9, 875–881. [Google Scholar] [CrossRef] [PubMed]
- Kosai, K.; Matsumoto, K.; Nagata, S.; Tsujimoto, Y.; Nakamura, T. Abrogation of Fas-induced fulminant hepatic failure in mice by hepatocyte growth factor. Biochem. Biophys. Res. Commun. 1998, 244, 683–690. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, H.; Toyoda, M.; Horiguchi, N.; Kakizaki, S.; Ohyama, T.; Takizawa, D.; Ichikawa, T.; Sato, K.; Takagi, H.; Mori, M. Hepatocyte growth factor protects against Fas-mediated liver apoptosis in transgenic mice. Liver Int. 2009, 29, 1562–1568. [Google Scholar] [CrossRef] [PubMed]
- O’Grady, J.G.; Schalm, S.W.; Williams, R. Acute liver failure: Redefining the syndromes. Lancet 1993, 342, 273–275. [Google Scholar] [PubMed]
- Bernal, W.; Hyyrylainen, A.; Gera, A.; Audimoolam, V.K.; McPhail, M.J.; Auzinger, G.; Rela, M.; Heaton, N.; O’Grady, J.G.; Wendon, J.; et al. Lessons from look-back in acute liver failure? A single centre experience of 3300 patients. J. Hepatol. 2013, 59, 74–80. [Google Scholar] [CrossRef] [PubMed]
- O’Grady, J.G. Acute liver failure. Postgrad. Med. J. 2005, 81, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Mawatari, S.; Moriuchi, A.; Ohba, F.; Kawano, T.; Oda, K.; Takikawa, Y.; Takikawa, H.; Ido, A.; Tsubouchi, H. The recovery of the PT-INR to less than 1.3 predicts survival in patients with severe acute liver injury. J. Gastroenterol. 2017, 53, 861–872. [Google Scholar] [CrossRef] [PubMed]
- Kosai, K.; Matsumoto, K.; Funakoshi, H.; Nakamura, T. Hepatocyte growth factor prevents endotoxin-induced lethal hepatic failure in mice. Hepatology 1999, 30, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Ding, S.; Merkulova-Rainon, T.; Han, Z.C.; Tobelem, G. HGF receptor up-regulation contributes to the angiogenic phenotype of human endothelial cells and promotes angiogenesis in vitro. Blood 2003, 101, 4816–4822. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.J.; Yang, H.W.; Wang, X.G.; Zhang, H. Hepatocyte growth factor prevents advanced glycation end products-induced injury and oxidative stress through a PI3K/Akt-dependent pathway in human endothelial cells. Life Sci. 2009, 85, 670–677. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Motoi, S.; Toyoda, H.; Obara, T.; Ohta, E.; Arita, Y.; Negishi, K.; Moriya, K.; Kuboi, Y.; Soejima, M.; Imai, T.; et al. Anti-Apoptotic Effects of Recombinant Human Hepatocyte Growth Factor on Hepatocytes Were Associated with Intrahepatic Hemorrhage Suppression Indicated by the Preservation of Prothrombin Time. Int. J. Mol. Sci. 2019, 20, 1821. https://doi.org/10.3390/ijms20081821
Motoi S, Toyoda H, Obara T, Ohta E, Arita Y, Negishi K, Moriya K, Kuboi Y, Soejima M, Imai T, et al. Anti-Apoptotic Effects of Recombinant Human Hepatocyte Growth Factor on Hepatocytes Were Associated with Intrahepatic Hemorrhage Suppression Indicated by the Preservation of Prothrombin Time. International Journal of Molecular Sciences. 2019; 20(8):1821. https://doi.org/10.3390/ijms20081821
Chicago/Turabian StyleMotoi, Sotaro, Hiroko Toyoda, Takashi Obara, Etsuko Ohta, Yoshihisa Arita, Kana Negishi, Katsuhiro Moriya, Yoshikazu Kuboi, Motohiro Soejima, Toshio Imai, and et al. 2019. "Anti-Apoptotic Effects of Recombinant Human Hepatocyte Growth Factor on Hepatocytes Were Associated with Intrahepatic Hemorrhage Suppression Indicated by the Preservation of Prothrombin Time" International Journal of Molecular Sciences 20, no. 8: 1821. https://doi.org/10.3390/ijms20081821
APA StyleMotoi, S., Toyoda, H., Obara, T., Ohta, E., Arita, Y., Negishi, K., Moriya, K., Kuboi, Y., Soejima, M., Imai, T., Ido, A., Tsubouchi, H., & Kawano, T. (2019). Anti-Apoptotic Effects of Recombinant Human Hepatocyte Growth Factor on Hepatocytes Were Associated with Intrahepatic Hemorrhage Suppression Indicated by the Preservation of Prothrombin Time. International Journal of Molecular Sciences, 20(8), 1821. https://doi.org/10.3390/ijms20081821



