Insights into Actin-Myosin Interactions within Muscle from 3D Electron Microscopy
Abstract
:1. Introduction
2. Electron Microscopy of Thin Sections
2.1. Preparation of Muscle Tissue for 3D Electron Microscopy
2.2. Methods for 3D Imaging of Muscle Tissue
3. Early 3D Reconstruction Work
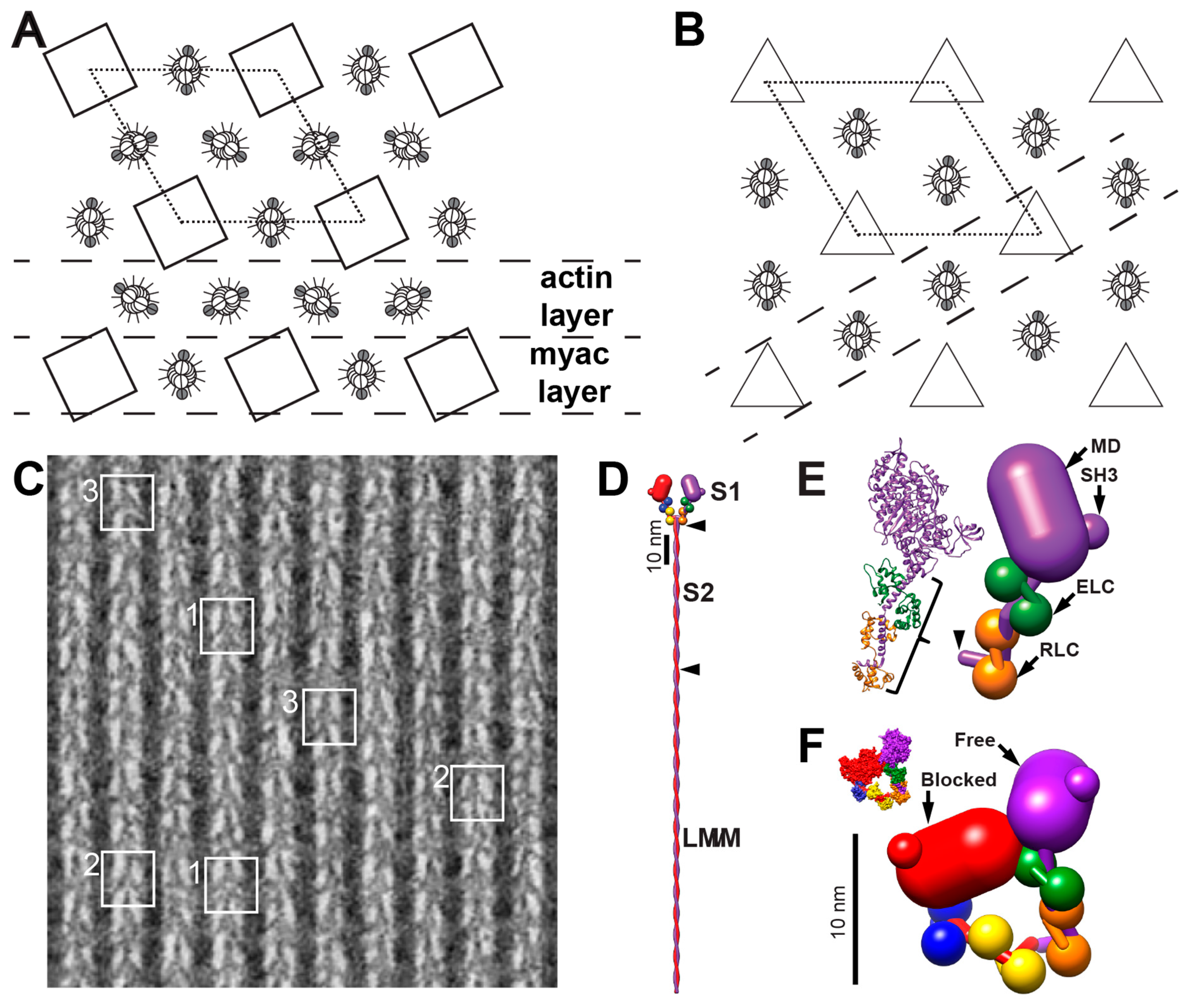
3.1. How Well-Ordered is the Insect Flight Muscle Lattice?
3.2. Rigor Muscle Spatial Average Reconstructions
3.3. Spatial Averages of AMPPNP Treated Muscle
4. 3D Imaging by Electron Tomography of Static States
Impact of New Information
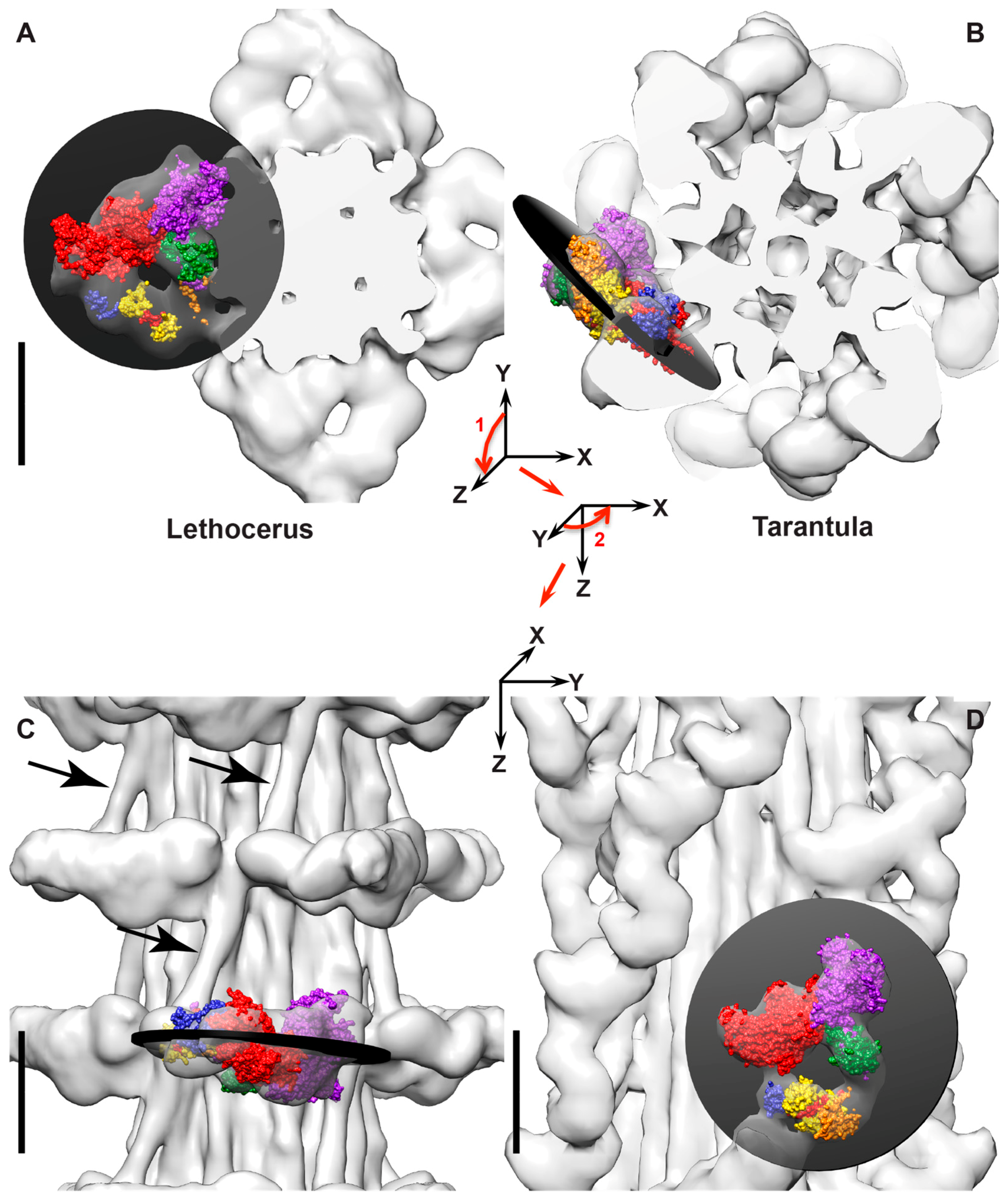
- Assuming the AMPPNP addition to Lethocerus myosin also produces a post-rigor conformation, the head would have its lever arm down and its actin binding cleft open. Such a conformation would explain why lead bridges in aqueous AMPPNP at 23 °C resembled rigor lead bridges when viewed in longitudinal sections. That sometimes they appeared to be single headed would reflect an effect of actin subunit azimuth in relationship to the lead bridge origin on the filament backbone. The comparatively weak actin-binding affinity of myosin•AMPPNP may not sustain large changes in the lever arm required to bind actin with the second head in some instances when one head is already bound. An open actin binding cleft with its correspondingly lower actin-binding affinity when AMPPNP is bound would explain the systematic loss of the highly distorted rigor rear bridges. On the other hand, with the lever arm down, the M-ward cross-bridge of mask motifs and other thin filament attachments outside of the target zone must differ from similar structures identified subsequently from the active muscle described below. The mask motifs of the active muscle have a lever-arm up conformation characteristic of the myosin II transition state, not a lever arm down conformation, and they seem to be contacting TM rather than actin in the active muscle. Conceivably, they might be held near the thin filament by a structure other than the myosin MD, perhaps a long N-terminal extension of the RLC of myosin, in which case, the up or down state of the lever arm would be relatively unimportant. A connection between the RLC and actin seems to occur in the Drosophila flight muscle [82,83]. The Lethocerus flight muscle RLC has a similar N-terminal extension (Belinda Bullard, unpublished).
- In aqueous AMPPNP, there is a stronger periodicity along the 14.5 nm repeat of myosin. Ordering of the RLC at the head rod junction probably contributes too little mass to account for such an effect. Increasing ethylene glycol up to the critical glycol concentration of ~30% moves the appearance even more toward that of the relaxed muscle while retaining rigor stiffness [52]. However, unless ethylene glycol can drive myosin•AMPPNP toward the transition state by cleaving the nucleotide, an unlikely though unproven possibility, the thick filament structure cannot fully relax by forming the IHM. So why does it take on a relaxed appearance in the glycol-stiff state and ultimately take on the appearance of fully relaxed muscle?
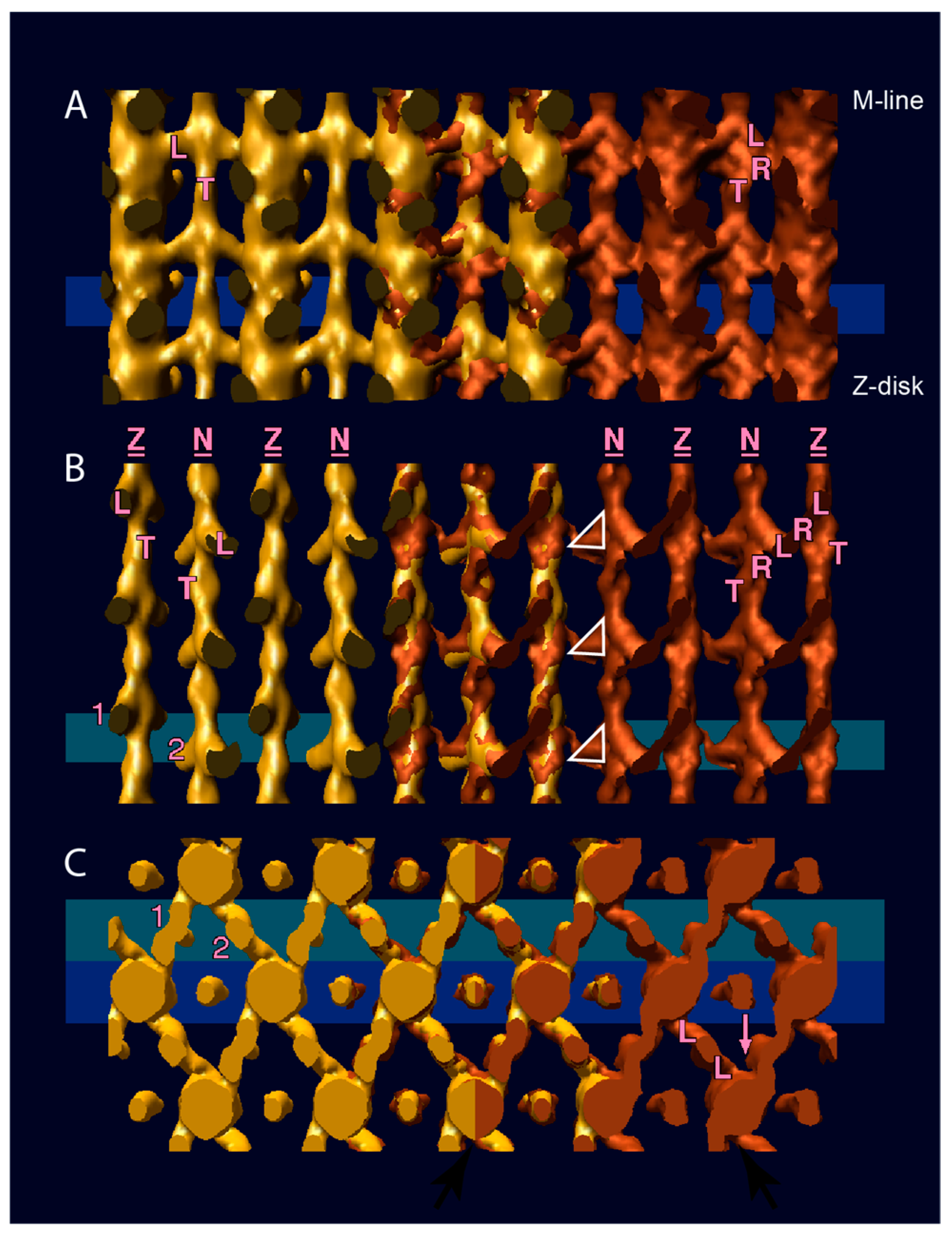
5. Electron Tomography of Frozen Active Muscle
6. Myosin Head Structures in Frozen Active Muscle
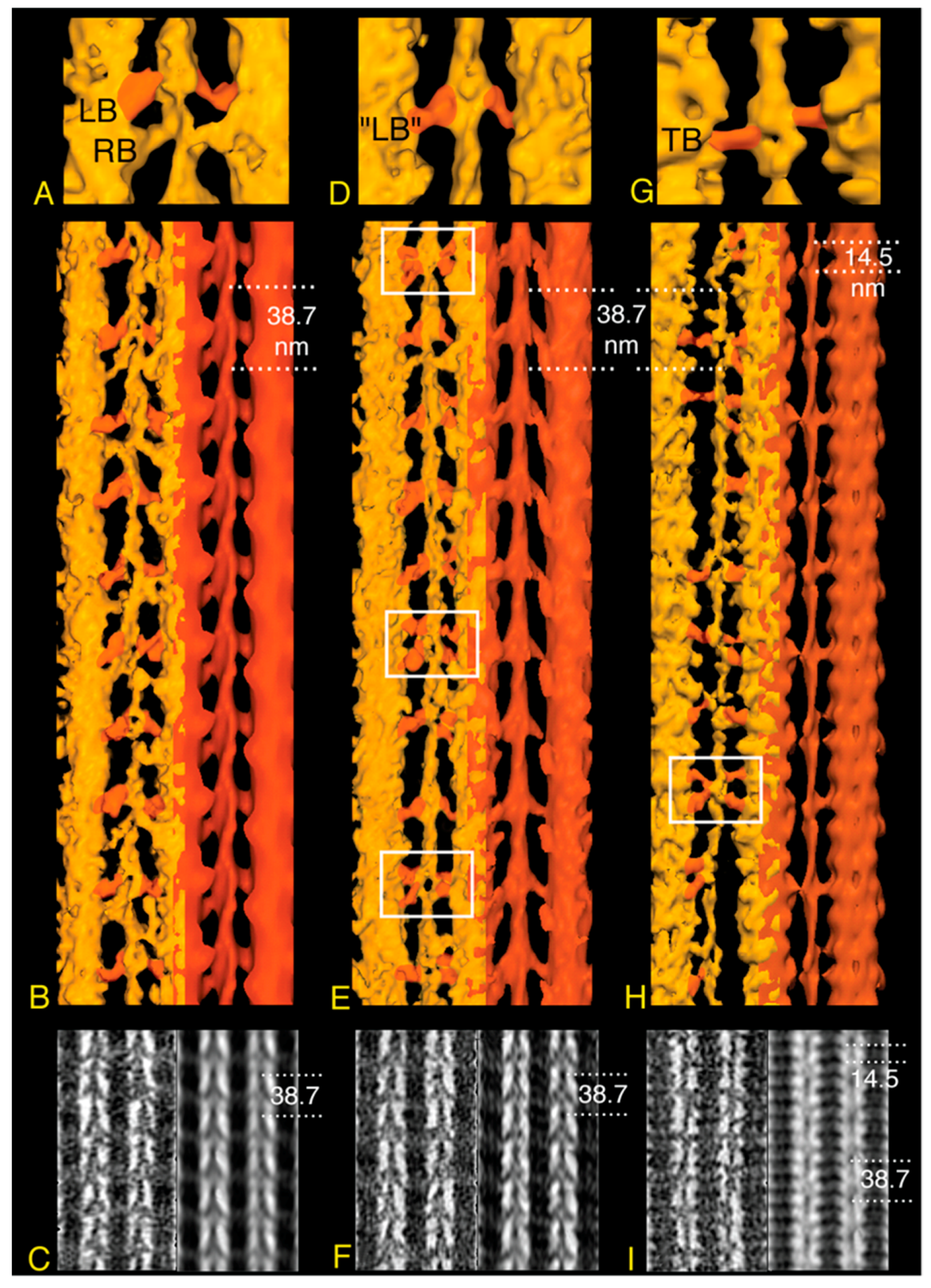
7. Active Muscle Following a Quick Stretch or Release
Impact of Recent Results on the Interpretation of Frozen Active Muscle Imaging
- An X-ray fiber diffraction of the relaxed Lethocerus flight muscle shows that tension applied sinusoidally causes changes in the helical angle between crowns as well as changes in the 14.5 nm meridional reflection. Under tension, relaxed fibers showed a 33.75° helical angle, whereas fibers under low or no tension showed a 33.90° helical angle. The changes in the 14.5 nm meridional intensity are open to interpretation, but could be signaling a change in the IHM resulting from applied tension.
- If tension applied to the relaxed muscle can disorder the IHM, thereby producing a change in the helical angle, a conjecture at this point, then ordering the IHM in isolated filaments should increase the helical angle, which is what is observed. In other words, the rod structure, which defines the filaments helical structure, and the IHM structure appear to be coupled.
- If only the blocked head becomes disordered during a stretch, an ironic statement given its name, the retained order in the free head pins the S2 tether to the backbone thereby restricting the “detached” blocked head to movements about the head-rod junction. Highly restricted movements of the blocked head would require a rather precisely placed target zone actin subunit to initiate force production.
- Reformation of the interacting heads motif after a contraction must be fast enough to sequester the myosin heads from further interaction with the thin filament. The kinetics of ATP cleavage in the Lethocerus myosin in this context have been discussed previously [6], indicating that the ATPase speed is sufficient. However, as noted above (Figure 5), the post rigor, myosin•ATP structure of the free head might be capable of reattaching the thick filament backbone even before ATP cleavage has occurred. Free-head rebinding to the thick filament backbone would be a first order reaction and potentially faster than the ATP cleavage step that recocks the lever arm because it does not involve a covalent bond cleavage.
8. Results from Fast Frozen Vertebrate Muscle Fibers
9. Prospects for Future Improvements
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| OSR | Oblique Section Reconstruction |
| Tn | Troponin |
| TM | Tropomyosin |
| RLC | Regulatory Light Chain |
| ELC | Essential Light Chain |
| myac | A thin, 25 nm thick longitudinal section containing alternating myosin and actin filaments |
| LMM | Light meromyosin; the last ~100 nm of myosin coiled-coil rod domain |
| MD | Motor Domain |
| IHM | Interacting Heads Motif |
| cryoEM | Cryogenic Electron Microscopy |
| S1 | The component of myosin containing the motor domain, light chains and lever arm |
| S2 | Initial ~50 nm of the myosin coiled-coil rod domain |
| ET | Electron Tomography |
| AMPPNP | adenylyl-imidodiphosphate, a non-hydrolysable analog of ATP |
| HST | High Static Tension, i.e., an isometric contraction |
References
- Reedy, M.K.; Holmes, K.C.; Tregear, R.T. Induced changes in orientation of the cross-bridges of glycerinated insect flight muscle. Nature 1965, 207, 1276–1280. [Google Scholar] [CrossRef] [PubMed]
- Huxley, H.E. The mechanism of muscular contraction. Science 1969, 164, 1356–1365. [Google Scholar] [CrossRef]
- Huxley, H.E.; Brown, W. The Low-angle X-ray Diagram of Vertebrate Striated Muscle and its Behaviour during Contraction and Rigor. J. Mol. Biol. 1967, 30, 383–434. [Google Scholar] [CrossRef]
- Holmes, K.C.; Tregear, R.T.; Barrington Leigh, J. Interpretation of the low angle X-ray diffraction from insect muscle in rigor. Proc. Roy. Soc. (Lond.) Ser. B Biol. 1980, 207, 13–33. [Google Scholar]
- Reedy, M.K. Ultrastructure of insect flight muscle. I. Screw sense and structural grouping in the rigor cross-bridge lattice. J. Mol. Biol. 1968, 31, 155–176. [Google Scholar] [CrossRef]
- Hu, Z.; Taylor, D.W.; Reedy, M.K.; Edwards, R.J.; Taylor, K.A. Structure of myosin filaments from relaxed Lethocerus flight muscle by cryo-EM at 6 Å resolution. Sci. Adv. 2016, 2, 9. [Google Scholar] [CrossRef]
- Miller, A.; Tregear, R.T. Structure of insect fibrillar flight muscle in the presence and absence of ATP. J. Mol. Biol. 1972, 70, 85–104. [Google Scholar] [CrossRef]
- Wray, J.S. Structure of the backbone in myosin filaments of muscle. Nature 1979, 277, 37–40. [Google Scholar] [CrossRef]
- Reedy, M.K.; Bahr, G.F.; Fischman, D.A. How Many Myosins per Cross-Bridge? I. Flight Muscle Myofibrils from the Blowfly, Sarcophaga bullata. Cold Spring Harb. Symp. Quant. Biol. 1973, 37, 397–421. [Google Scholar] [CrossRef]
- Morris, E.P.; Squire, J.M.; Fuller, G.W. The 4-stranded helical arrangement of myosin heads on insect (Lethocerus) flight muscle thick filaments. J. Struct. Biol. 1991, 107, 237–249. [Google Scholar] [CrossRef]
- Schmitz, H.; Lucaveche, C.; Reedy, M.K.; Taylor, K.A. Oblique section 3-D reconstruction of relaxed insect flight muscle reveals cross bridge lattice in helical registration. Biophys. J. 1994, 66, A347. [Google Scholar] [CrossRef]
- Reedy, M.K. Cross-Bridges and Periods in Insect Flight Muscle. Am. Zool. 1967, 7, 465–481. [Google Scholar] [CrossRef]
- Pringle, J.W. The Croonian Lecture, 1977. Stretch activation of muscle: Function and mechanism. Proc. R. Soc. Lond. B Biol. Sci. 1978, 201, 107–130. [Google Scholar]
- Bullard, B.; Pastore, A. Regulating the contraction of insect flight muscle. J. Muscle Res. Cell Motil. 2011, 32, 303–313. [Google Scholar] [CrossRef] [PubMed]
- Luther, P.K.; Squire, J.M.; Forey, P.L. Evolution of myosin filament arrangements in vertebrate skeletal muscle. J. Morphol. 1996, 229, 325–335. [Google Scholar] [CrossRef]
- Hirose, K.; Wakabayashi, T. Thin filaments of rabbit skeletal muscle are in helical register. J. Mol. Biol. 1988, 204, 797–801. [Google Scholar] [CrossRef]
- Woodhead, J.L.; Zhao, F.Q.; Craig, R.; Egelman, E.H.; Alamo, L.; Padron, R. Atomic model of a myosin filament in the relaxed state. Nature 2005, 436, 1195–1199. [Google Scholar] [CrossRef]
- Zoghbi, M.E.; Woodhead, J.L.; Moss, R.L.; Craig, R. Three-dimensional structure of vertebrate cardiac muscle myosin filaments. Proc. Natl. Acad. Sci. USA 2008, 105, 2386–2390. [Google Scholar] [CrossRef]
- Al-Khayat, H.A.; Kensler, R.W.; Squire, J.M.; Marston, S.B.; Morris, E.P. Atomic model of the human cardiac muscle myosin filament. Proc. Natl. Acad. Sci. USA 2013, 110, 318–323. [Google Scholar] [CrossRef]
- Wendt, T.; Taylor, D.; Trybus, K.M.; Taylor, K. Three-dimensional image reconstruction of dephosphorylated smooth muscle heavy meromyosin reveals asymmetry in the interaction between myosin heads and placement of subfragment 2. Proc. Natl. Acad. Sci. USA 2001, 98, 4361–4366. [Google Scholar] [CrossRef]
- Henderson, R.; Unwin, P.N. Three-dimensional model of purple membrane obtained by electron microscopy. Nature 1975, 257, 28–32. [Google Scholar] [CrossRef]
- Taylor, K.A.; Reedy, M.C.; Cordova, L.; Reedy, M.K. Three-dimensional reconstruction of rigor insect flight muscle from tilted thin sections. Nature 1984, 310, 285–291. [Google Scholar] [CrossRef]
- Crowther, R.A.; Luther, P.K. Three-dimensional reconstruction from a single oblique section of fish muscle M-band. Nature 1984, 307, 569–570. [Google Scholar] [CrossRef]
- Tsukita, S.; Yano, M. Actomyosin structure in contracting muscle detected by rapid freezing. Nature 1985, 317, 182–184. [Google Scholar] [CrossRef]
- Taylor, K.A.; Reedy, M.C.; Cordova, L.; Reedy, M.K. Three-dimensional image reconstruction of insect flight muscle. I. The rigor myac layer. J. Cell Biol. 1989, 109, 1085–1102. [Google Scholar] [CrossRef]
- Taylor, K.A.; Crowther, R.A. A protocol for 3D image reconstruction from a single image of an oblique section. Ultramicroscopy 1991, 38, 85–103. [Google Scholar] [CrossRef]
- Rayment, I.; Rypniewsky, W.R.; Schmidt-Bäse, K.; Smith, R.; Tomchick, D.R.; Benning, M.M.; Winkelmann, D.A.; Wesenberg, G.; Holden, H.M. Three-dimensional structure of myosin subfragment-1: A molecular motor. Science 1993, 261, 50–58. [Google Scholar] [CrossRef]
- Winkler, H.; Reedy, M.C.; Reedy, M.K.; Tregear, R.; Taylor, K.A. Three-dimensional structure of nucleotide-bearing crossbridges in situ: Oblique section reconstruction of insect flight muscle in AMPPNP at 23 degrees C. J. Mol. Biol. 1996, 264, 302–322. [Google Scholar] [CrossRef]
- Braunfeld, M.B.; Koster, A.J.; Sedat, J.W.; Agard, D.A. Cryo automated electron tomography: Towards high-resolution reconstructions of plastic-embedded structures. J. Microsc. 1994, 174, 75–84. [Google Scholar] [CrossRef]
- Taylor, K.A.; Tang, J.; Cheng, Y.; Winkler, H. The use of electron tomography for structural analysis of disordered protein arrays. J. Struct. Biol. 1997, 120, 372–386. [Google Scholar] [CrossRef]
- Winkler, H.; Taylor, K.A. Multivariate statistical analysis of three-dimensional cross-bridge motifs in insect flight muscle. Ultramicroscopy 1999, 77, 141–152. [Google Scholar] [CrossRef]
- Chen, L.F.; Blanc, E.; Chapman, M.S.; Taylor, K.A. Real space refinement of acto-myosin structures from sectioned muscle. J. Struct. Biol. 2001, 133, 221–232. [Google Scholar] [CrossRef]
- Liu, J.; Reedy, M.C.; Goldman, Y.E.; Franzini-Armstrong, C.; Sasaki, H.; Tregear, R.T.; Lucaveche, C.; Winkler, H.; Baumann, B.A.J.; Squire, J.M.; et al. Electron tomography of fast frozen, stretched rigor fibers reveals elastic distortions in the myosin crossbridges. J. Struct. Biol. 2004, 147, 268–282. [Google Scholar] [CrossRef]
- Wu, S.; Liu, J.; Reedy, M.C.; Tregear, R.T.; Winkler, H.; Franzini-Armstrong, C.; Sasaki, H.; Lucaveche, C.; Goldman, Y.E.; Reedy, M.K.; et al. Electron tomography of cryofixed, isometrically contracting insect flight muscle reveals novel actin-myosin interactions. PLoS ONE 2010, 5, e12643. [Google Scholar] [CrossRef]
- Taylor, K.A.; Wu, S.; Reedy, M.C.; Reedy, M.K. Imaging actomyosin in situ. In Cellular Electron Microscopy; McIntosh, J.R., Ed.; Academic Press: San Diego, CA, USA, 2007; Volume 79, pp. 321–368. [Google Scholar]
- Lehrer, S.S. Damage to actin filaments by glutaraldehyde: Protection by tropomyosin. J. Cell Biol. 1981, 90, 459–466. [Google Scholar] [CrossRef]
- Maupin-Szamier, P.; Pollard, T.D. Actin filament destruction by osmium tetroxide. J. Cell Biol. 1978, 77, 837–852. [Google Scholar] [CrossRef]
- Amos, L.A.; Henderson, R.; Unwin, P.N. Three-dimensional structure determination by electron microscopy of two-dimensional crystals. Prog. Biophys. Mol. Biol. 1982, 39, 183–231. [Google Scholar] [CrossRef]
- Taylor, K.A.; Crowther, R.A. 3D reconstruction from the Fourier transform of a single superlattice image of an oblique section. Ultramicroscopy 1992, 41, 153–167. [Google Scholar] [CrossRef]
- Taylor, K.A.; Reedy, M.C.; Reedy, M.K.; Crowther, R.A. Crossbridges in the complete unit cell of rigor insect flight muscle imaged by three-dimensional reconstruction from oblique sections. J. Mol. Biol. 1993, 233, 86–108. [Google Scholar] [CrossRef]
- Kabsch, W.; Mannherz, H.G.; Suck, D.; Pai, E.F.; Holmes, K.C. Atomic structure of the actin: DNase I complex. Nature 1990, 347, 37–44. [Google Scholar] [CrossRef]
- Rayment, I.; Holden, H.M.; Whittaker, M.; Yohn, C.B.; Lorenz, M.; Holmes, K.C.; Milligan, R.A. Structure of the actin-myosin complex and its implications for muscle contraction. Science 1993, 261, 58–65. [Google Scholar] [CrossRef]
- Chen, L.F.; Winkler, H.; Reedy, M.K.; Reedy, M.C.; Taylor, K.A. Molecular modeling of averaged rigor crossbridges from tomograms of insect flight muscle. J. Struct. Biol. 2002, 138, 92–104. [Google Scholar] [CrossRef]
- Reedy, M.C.; Reedy, M.K.; Leonard, K.R.; Bullard, B. Gold/Fab immuno electron microscopy localization of troponin H and troponin T in Lethocerus flight muscle. J. Mol. Biol. 1994, 239, 52–67. [Google Scholar] [CrossRef]
- Reedy, M.K.; Reedy, M.C. Rigor crossbridge structure in tilted single filament layers and flared-X formations from insect flight muscle. J. Mol. Biol. 1985, 185, 145–176. [Google Scholar] [CrossRef]
- Reedy, M.C.; Reedy, M.K.; Goody, R.S. The structure of insect flight muscle in the presence of AMPPNP. J. Muscle Res. Cell Motil. 1987, 8, 473–503. [Google Scholar] [CrossRef]
- Lovell, S.J.; Knight, P.J.; Harrington, W.F. Fraction of myosin heads bound to thin filaments in rigor fibrils from insect flight and vertebrate muscles. Nature 1981, 293, 664–666. [Google Scholar] [CrossRef]
- Goody, R.S.; Reedy, M.C.; Hofmann, W.; Holmes, K.C.; Reedy, M.K. Binding of myosin subfragment 1 to glycerinated insect flight muscle in the rigor state. Biophys. J. 1985, 47, 151–169. [Google Scholar] [CrossRef]
- Thomas, D.D.; Cooke, R.; Barnett, V.A. Orientation and rotational mobility of spin-labelled myosin heads in insect flight muscle in rigor. J. Muscle Res. Cell Motil. 1983, 4, 367–378. [Google Scholar] [CrossRef]
- Tregear, R.T.; Wakabayashi, K.; Tanaka, H.; Iwamoto, H.; Reedy, M.C.; Reedy, M.K.; Sugi, H.; Amemiya, Y. X-ray diffraction and electron microscopy from Lethocerus flight muscle partially relaxed by adenylylimidodiphosphate and ethylene glycol. J. Mol. Biol. 1990, 214, 129–141. [Google Scholar] [CrossRef]
- Clarke, M.L.; Rodger, C.D.; Tregear, R.T. Modification of crossbridge states by ethylene glycol in insect flight muscle. J. Muscle Res. Cell Motil. 1984, 5, 81–96. [Google Scholar] [CrossRef]
- Reedy, M.C.; Reedy, M.K.; Tregear, R.T. Two attached non-rigor crossbridge forms in insect flight muscle. J. Mol. Biol. 1988, 204, 357–383. [Google Scholar] [CrossRef]
- Williams, D.L., Jr.; Greene, L.E. Comparison of the effects of tropomyosin and troponin-tropomyosin on the binding of myosin subfragment 1 to actin. Biochemistry 1983, 22, 2770–2774. [Google Scholar] [CrossRef]
- Johnson, R.E. Effect of ethylene glycol and Ca2+ on the binding of Mg2+ x adenyl-5′-yl imidodiphosphate to rabbit skeletal myofibrils. J. Biol. Chem. 1986, 261, 728–732. [Google Scholar]
- Marston, S.B.; Rodger, C.D.; Tregear, R.T. Changes in muscle crossbridges when beta, gamma-imido-ATP binds to myosin. J. Mol. Biol. 1976, 104, 263–276. [Google Scholar] [CrossRef]
- Reedy, M.K.; Goody, R.S.; Hofmann, W.; Rosenbaum, G. Co-ordinated electron microscopy and X-ray studies of glycerinated insect flight muscle. I. X-ray diffraction monitoring during preparation for electron microscopy of muscle fibres fixed in rigor, in ATP and in AMPPNP. J. Muscle Res. Cell Motil. 1983, 4, 25–53. [Google Scholar] [CrossRef]
- Reedy, M.C.; Reedy, M.K.; Goody, R.S. Co-ordinated electron microscopy and X-ray studies of glycerinated insect flight muscle. II. Electron microscopy and image reconstruction of muscle fibres fixed in rigor, in ATP and in AMPPNP. J. Muscle Res. Cell Motil. 1983, 4, 55–81. [Google Scholar] [CrossRef]
- Taylor, K.A.; Schmitz, H.; Reedy, M.C.; Goldman, Y.E.; Franzini-Armstrong, C.; Sasaki, H.; Tregear, R.T.; Poole, K.J.V.; Lucaveche, C.; Edwards, R.J.; et al. Tomographic 3-D reconstruction of quick frozen, Ca2+-activated contracting insect flight muscle. Cell 1999, 99, 421–431. [Google Scholar] [CrossRef]
- Tregear, R.T.; Edwards, R.J.; Irving, T.C.; Poole, K.J.; Reedy, M.C.; Schmitz, H.; Towns-Andrews, E.; Reedy, M.K. X-ray diffraction indicates that active cross-bridges bind to actin target zones in insect flight muscle. Biophys. J. 1998, 74, 1439–1451. [Google Scholar] [CrossRef]
- Huxley, H.E.; Kress, M. Crossbridge behaviour during muscle contraction. J. Muscle Res. Cell Motil. 1985, 6, 153–161. [Google Scholar] [CrossRef]
- Winkler, H.; Taylor, K.A. 3D reconstruction by combining data from sections cut oblique to different unit cell axes. Ultramicroscopy 1994, 55, 357–371. [Google Scholar] [CrossRef]
- Schmitz, H.; Reedy, M.C.; Reedy, M.K.; Tregear, R.T.; Winkler, H.; Taylor, K.A. Electron tomography of insect flight muscle in rigor and AMPPNP at 23 degrees C. J. Mol. Biol. 1996, 264, 279–301. [Google Scholar] [CrossRef]
- Schmitz, H.; Reedy, M.C.; Reedy, M.K.; Tregear, R.T.; Taylor, K.A. Tomographic three-dimensional reconstruction of insect flight muscle partially relaxed by AMPPNP and ethylene glycol. J. Cell Biol. 1997, 139, 695–707. [Google Scholar] [CrossRef]
- Winkler, H.; Taylor, K.A. Accurate marker-free alignment with simultaneous geometry determination and reconstruction of tilt series in electron tomography. Ultramicroscopy 2006, 106, 240–254. [Google Scholar] [CrossRef]
- Winkler, H.; Taylor, K.A. Marker-free dual-axis tilt series alignment. J. Struct. Biol. 2013, 182, 117–124. [Google Scholar] [CrossRef]
- Winkler, H.; Zhu, P.; Liu, J.; Ye, F.; Roux, K.H.; Taylor, K.A. Tomographic subvolume alignment and subvolume classification applied to myosin V and SIV envelope spikes. J. Struct. Biol. 2009, 165, 64–77. [Google Scholar] [CrossRef]
- Taylor, K.A.; Liu, J.; Winkler, H. Localization and classification of repetitive structures in electron tomograms of paracrystalline assemblies. In Electron Tomography: Methods for Three-dimensional Visualization of Structures in the Cell, 2nd ed.; Frank, J., Ed.; Springer-Verlag: New York, NY, USA, 2006; pp. 417–439. [Google Scholar]
- Wu, S.; Liu, J.; Reedy, M.C.; Winkler, H.; Reedy, M.K.; Taylor, K.A. Methods for identifying and averaging variable molecular conformations in tomograms of actively contracting insect flight muscle. J. Struct. Biol. 2009, 168, 485–502. [Google Scholar] [CrossRef]
- Winkler, H.; Wu, S.; Taylor, K.A. Electron Tomography of Paracrystalline 2D Arrays. Methods Mol. Biol. 2013, 955, 427–460. [Google Scholar]
- Liu, J.; Wu, S.; Reedy, M.C.; Winkler, H.; Lucaveche, C.; Cheng, Y.; Reedy, M.K.; Taylor, K.A. Electron tomography of swollen rigor fibers of insect flight muscle reveals a short and variably angled S2 domain. J. Mol. Biol. 2006, 362, 844–860. [Google Scholar] [CrossRef]
- Sleep, J.; Glyn, H. Inhibition of myofibrillar and actomyosin subfragment 1 adenosinetriphosphatase by adenosine 5′-diphosphate, pyrophosphate, and adenyl-5′-yl imidodiphosphate. Biochemistry 1986, 25, 1149–1154. [Google Scholar] [CrossRef]
- Schoenberg, M. Equilibrium muscle crossbridge behavior: The interaction of myosin crossbridges with actin. Adv. Biophys. 1993, 29, 55–73. [Google Scholar] [CrossRef]
- Huxley, A.F.; Simmons, R.M. Proposed mechanism of force generation in striated muscle. Nature 1971, 233, 533–538. [Google Scholar] [CrossRef]
- Gulick, A.M.; Bauer, C.B.; Thoden, J.B.; Rayment, I. X-ray structures of the MgADP, MgATPgammaS, and MgAMPPNP complexes of the Dictyostelium discoideum myosin motor domain. Biochemistry 1997, 36, 11619–11628. [Google Scholar] [CrossRef]
- Fisher, A.J.; Smith, C.A.; Thoden, J.B.; Smith, R.; Sutoh, K.; Holden, H.M.; Rayment, I. X-ray structures of the myosin motor domain of Dictyostelium discoideum complexed with MgADP•BeFx and MgADP•AlF4. Biochemistry 1995, 34, 8960–8972. [Google Scholar] [CrossRef]
- Smith, C.A.; Rayment, I. X-ray structure of the magnesium(II)-pyrophosphate complex of the truncated head of Dictyostelium discoideum myosin to 2.7 A resolution. Biochemistry 1995, 34, 8973–8981. [Google Scholar] [CrossRef]
- Holmes, K.C.; Geeves, M.A. The structural basis of muscle contraction. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2000, 355, 419–431. [Google Scholar] [CrossRef]
- Houdusse, A.; Szent-Gyorgyi, A.G.; Cohen, C. Three conformational states of scallop myosin S1. Proc. Natl. Acad. Sci. USA 2000, 97, 11238–11243. [Google Scholar] [CrossRef]
- Himmel, D.M.; Gourinath, S.; Reshetnikova, L.; Shen, Y.; Szent-Gyorgyi, A.G.; Cohen, C. Crystallographic findings on the internally uncoupled and near-rigor states of myosin: Further insights into the mechanics of the motor. Proc. Natl. Acad. Sci. USA 2002, 99, 12645–12650. [Google Scholar] [CrossRef]
- Risal, D.; Gourinath, S.; Himmel, D.M.; Szent-Gyorgyi, A.G.; Cohen, C. Myosin subfragment 1 structures reveal a partially bound nucleotide and a complex salt bridge that helps couple nucleotide and actin binding. Proc. Natl. Acad. Sci. USA 2004, 101, 8930–8935. [Google Scholar] [CrossRef]
- Yang, Y.; Gourinath, S.; Kovacs, M.; Nyitray, L.; Reutzel, R.; Himmel, D.M.; O’Neall-Hennessey, E.; Reshetnikova, L.; Szent-Gyorgyi, A.G.; Brown, J.H.; et al. Rigor-like structures from muscle myosins reveal key mechanical elements in the transduction pathways of this allosteric motor. Structure 2007, 15, 553–564. [Google Scholar] [CrossRef]
- Farman, G.P.; Miller, M.S.; Reedy, M.C.; Soto-Adames, F.N.; Vigoreaux, J.O.; Maughan, D.W.; Irving, T.C. Phosphorylation and the N-terminal extension of the regulatory light chain help orient and align the myosin heads in Drosophila flight muscle. J. Struct. Biol. 2009, 168, 240–249. [Google Scholar] [CrossRef]
- Miller, M.S.; Farman, G.P.; Braddock, J.M.; Soto-Adames, F.N.; Irving, T.C.; Vigoreaux, J.O.; Maughan, D.W. Regulatory light chain phosphorylation and N-terminal extension increase cross-bridge binding and power output in Drosophila at in vivo myofilament lattice spacing. Biophys. J. 2011, 100, 1737–1746. [Google Scholar] [CrossRef]
- Beinbrech, G.; Kuhn, H.J.; Ruegg, J.C. Electron microscope and optical diffraction studies on glycerol-extracted insect flight muscle fibres relaxed by pyrophosphate. Experientia 1972, 28, 511–513. [Google Scholar] [CrossRef]
- Hirose, K.; Wakabayashi, T. Structural change of crossbridges of rabbit skeletal muscle during isometric contraction. J. Muscle Res. Cell Motil. 1993, 14, 432–445. [Google Scholar] [CrossRef]
- Hirose, K.; Franzini-Armstrong, C.; Goldman, Y.E.; Murray, J.M. Structural changes in muscle crossbridges accompanying force generation. J. Cell Biol. 1994, 127, 763–778. [Google Scholar] [CrossRef]
- Sosa, H.; Popp, D.; Ouyang, G.; Huxley, H.E. Ultrastructure of skeletal muscle fibers studied by a plunge quick freezing method: Myofilament lengths. Biophys. J. 1994, 67, 283–292. [Google Scholar] [CrossRef]
- Lenart, T.D.; Murray, J.M.; Franzini-Armstrong, C.; Goldman, Y.E. Structure and periodicities of cross-bridges in relaxation, in rigor, and during contractions initiated by photolysis of caged Ca2+. Biophys. J. 1996, 71, 2289–2306. [Google Scholar] [CrossRef]
- Linari, M.; Reedy, M.K.; Reedy, M.C.; Lombardi, V.; Piazzesi, G. Ca-activation and stretch-activation in insect flight muscle. Biophys. J. 2004, 87, 1101–1111. [Google Scholar] [CrossRef]
- Holmes, K.C. Muscle proteins--their actions and interactions. Curr. Opin. Struct. Biol. 1996, 6, 781–789. [Google Scholar] [CrossRef]
- Molloy, J.E.; Burns, J.E.; Kendrick-Jones, J.; Tregear, R.T.; White, D.C. Movement and force produced by a single myosin head. Nature 1995, 378, 209–212. [Google Scholar] [CrossRef]
- Arakelian, C.; Warrington, A.; Winkler, H.; Perz-Edwards, R.J.; Reedy, M.K.; Taylor, K.A. Myosin S2 origins track evolution of strong binding on actin by azimuthal rolling of motor domain. Biophys. J. 2015, 108, 1495–1502. [Google Scholar] [CrossRef]
- Huxley, H.E.; Stewart, A.; Sosa, H.; Irving, T. X-ray diffraction measurements of the extensibility of actin and myosin filaments in contracting muscle. Biophys. J. 1994, 67, 2411–2421. [Google Scholar] [CrossRef]
- Wakabayashi, K.; Sugimoto, Y.; Tanaka, H.; Ueno, Y.; Takezawa, Y.; Amemiya, Y. X-ray diffraction evidence for the extensibility of actin and myosin filaments during muscle contraction. Biophys. J. 1994, 67, 2422–2435. [Google Scholar] [CrossRef]
- Beausang, J.R.; Schroeder III, H.W.; Nelson, P.C.; Goldman, Y.E. Twirling of actin by myosins II and V observed via polarized TIRF in a modified gliding assay. Biophys. J. 2008, 95, 5820–5831. [Google Scholar] [CrossRef]
- Perz-Edwards, R.J.; Irving, T.C.; Baumann, B.A.; Gore, D.; Hutchinson, D.C.; Krzic, U.; Porter, R.L.; Ward, A.B.; Reedy, M.K. X-ray diffraction evidence for myosin-troponin connections and tropomyosin movement during stretch activation of insect flight muscle. Proc. Natl. Acad. Sci. USA 2011, 108, 120–125. [Google Scholar] [CrossRef]
- Wu, S.; Liu, J.; Reedy, M.C.; Perz-Edwards, R.J.; Tregear, R.T.; Winkler, H.; Franzini-Armstrong, C.; Sasaki, H.; Lucaveche, C.; Goldman, Y.E.; et al. Structural changes in isometrically contracting insect flight muscle trapped following a mechanical perturbation. PLoS ONE 2012, 7, e39422. [Google Scholar] [CrossRef]
- Smith, D.A.; Geeves, M.A.; Sleep, J.; Mijailovich, S.M. Towards a unified theory of muscle contraction. I: Foundations. Ann. Biomed. Eng. 2008, 36, 1624–1640. [Google Scholar] [CrossRef]
- Brunello, E.; Reconditi, M.; Elangovan, R.; Linari, M.; Sun, Y.B.; Narayanan, T.; Panine, P.; Piazzesi, G.; Irving, M.; Lombardi, V. Skeletal muscle resists stretch by rapid binding of the second motor domain of myosin to actin. Proc. Natl. Acad. Sci. USA 2007, 104, 20114–20119. [Google Scholar] [CrossRef]
- Abbott, R.H.; Cage, P.E. A possible mechanism of length activation in insect fibrillar flight muscle. J. Muscle Res. Cell Motil. 1984, 5, 387–397. [Google Scholar] [CrossRef]
- Molloy, J.E. Active Insect Fibrillar Flight Muscle, Its Mechanical Performance and Cross-Bridge Kinetics. Ph.D. Thesis, University of York, York, UK, 1988. [Google Scholar]
- Gilmour, K.M.; Ellington, C.P. Power output of glycerinated bumblebee flight muscle. J. Exp. Biol. 1993, 183, 77–100. [Google Scholar]
- Gilmour, K.M.; Ellington, C.P. In vivo muscle length changes in bumblebees and the in vitro effects on work and power. J. Exp. Biol. 1993, 183, 101–113. [Google Scholar]
- Chan, W.P.; Dickinson, M.H. In vivo length oscillations of indirect flight muscles in the fruit fly Drosophila virilis. J. Exp. Biol. 1996, 199, 2767–2774. [Google Scholar]
- Wray, J.S. Filament geometry and the activation of insect flight muscles. Nature 1979, 280, 325–326. [Google Scholar] [CrossRef]
- Squire, J.M. Muscle filament lattices and stretch-activation: The match-mismatch model reassessed. J. Muscle Res. Cell Motil. 1992, 13, 183–189. [Google Scholar] [CrossRef]
- Hu, Z.; Taylor, D.W.; Edwards, R.J.; Taylor, K.A. Coupling between myosin head conformation and the thick filament backbone structure. J. Struct. Biol. 2017, 200, 325–333. [Google Scholar] [CrossRef]
- AL-Khayat, H.A.; Hudson, L.; Reedy, M.K.; Irving, T.C.; Squire, J.M. Myosin head configuration in relaxed insect flight muscle: X-ray modeled resting cross-bridges in a pre-powerstroke state are poised for actin binding. Biophys. J. 2003, 85, 1063–1079. [Google Scholar] [CrossRef]
- Hirose, K.; Lenart, T.D.; Murray, J.M.; Franzini-Armstrong, C.; Goldman, Y.E. Flash and smash: Rapid freezing of muscle fibers activated by photolysis of caged ATP. Biophys. J. 1993, 65, 397–408. [Google Scholar] [CrossRef]
- Squire, J.M.; Harford, J.J. Actin filament organization and myosin head labelling patterns in vertebrate skeletal muscles in the rigor and weak binding states. J. Muscle Res. Cell Motil. 1988, 9, 344–358. [Google Scholar] [CrossRef]
- Al-Amoudi, A.; Norlen, L.P.; Dubochet, J. Cryo-electron microscopy of vitreous sections of native biological cells and tissues. J. Struct. Biol. 2004, 148, 131–135. [Google Scholar] [CrossRef]
- Al-Amoudi, A.; Studer, D.; Dubochet, J. Cutting artefacts and cutting process in vitreous sections for cryo-electron microscopy. J. Struct. Biol. 2005, 150, 109–121. [Google Scholar] [CrossRef]
- Marko, M.; Hsieh, C.; Schalek, R.; Frank, J.; Mannella, C. Focused-ion-beam thinning of frozen-hydrated biological specimens for cryo-electron microscopy. Nat. Methods 2007, 4, 215–217. [Google Scholar] [CrossRef]
- Rigort, A.; Bauerlein, F.J.; Leis, A.; Gruska, M.; Hoffmann, C.; Laugks, T.; Bohm, U.; Eibauer, M.; Gnaegi, H.; Baumeister, W.; et al. Micromachining tools and correlative approaches for cellular cryo-electron tomography. J. Struct. Biol. 2010, 172, 169–179. [Google Scholar] [CrossRef]
- Rigort, A.; Bauerlein, F.J.; Villa, E.; Eibauer, M.; Laugks, T.; Baumeister, W.; Plitzko, J.M. Focused ion beam micromachining of eukaryotic cells for cryoelectron tomography. Proc. Natl. Acad. Sci. USA 2012, 109, 4449–4454. [Google Scholar] [CrossRef]

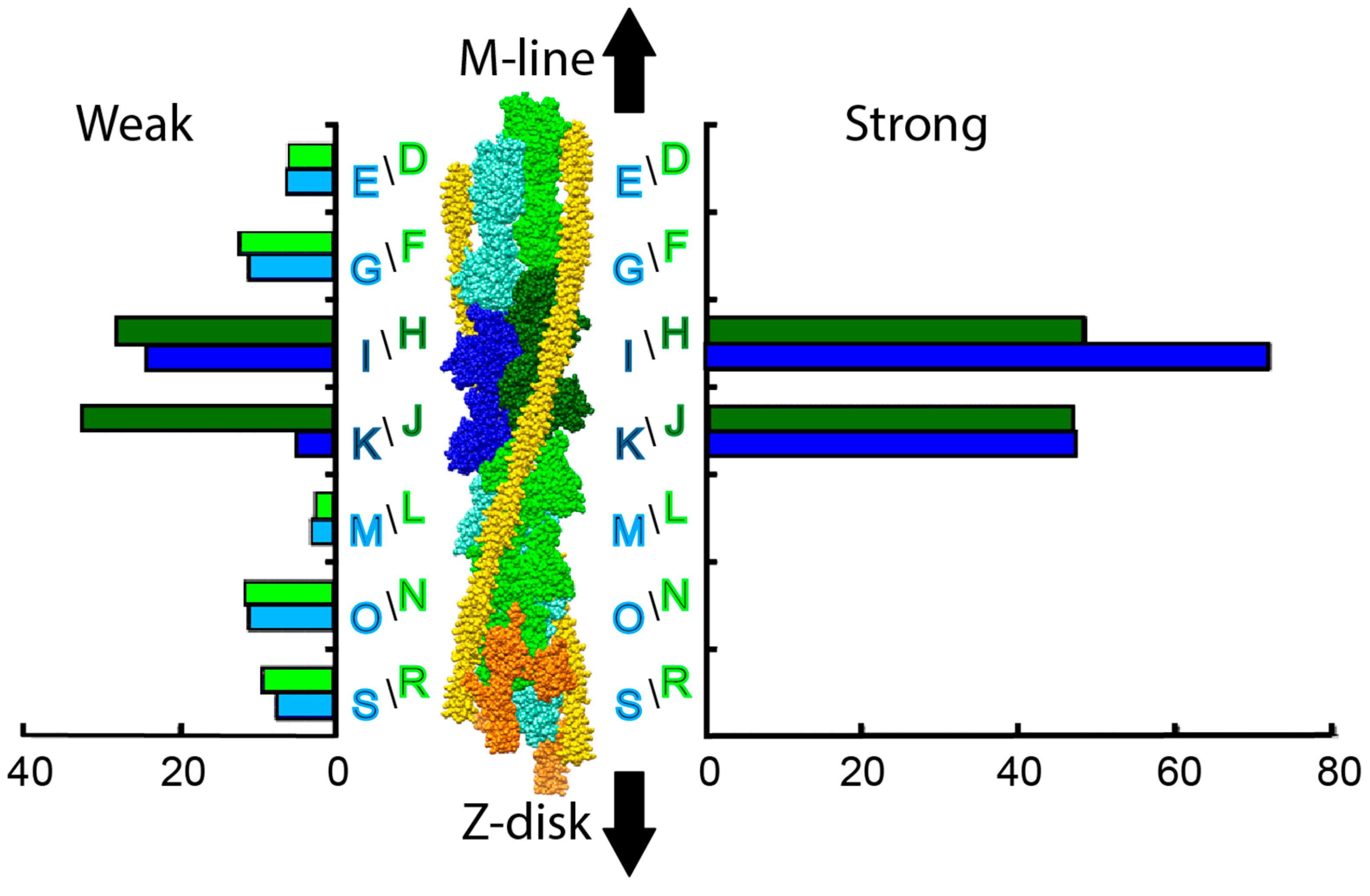
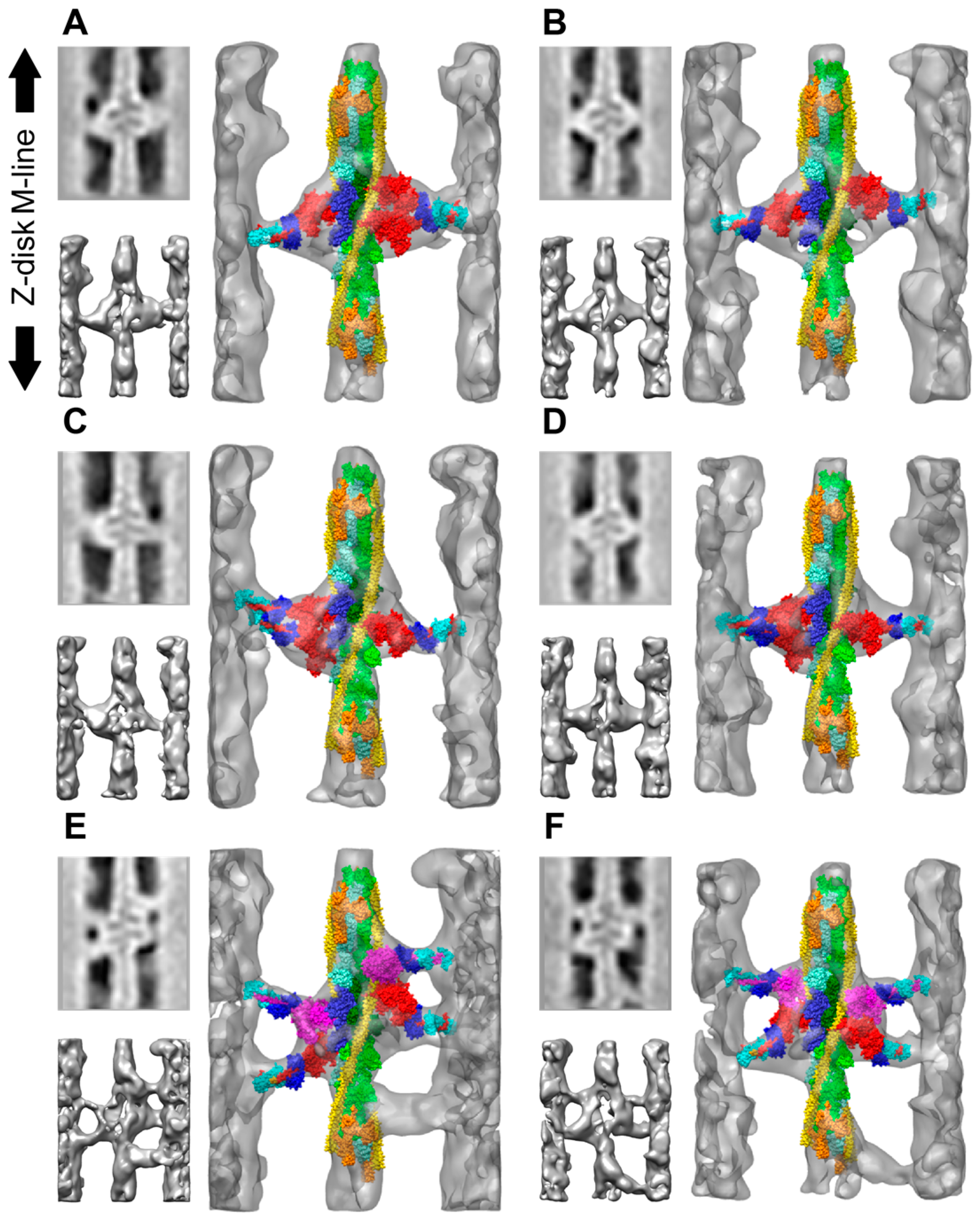
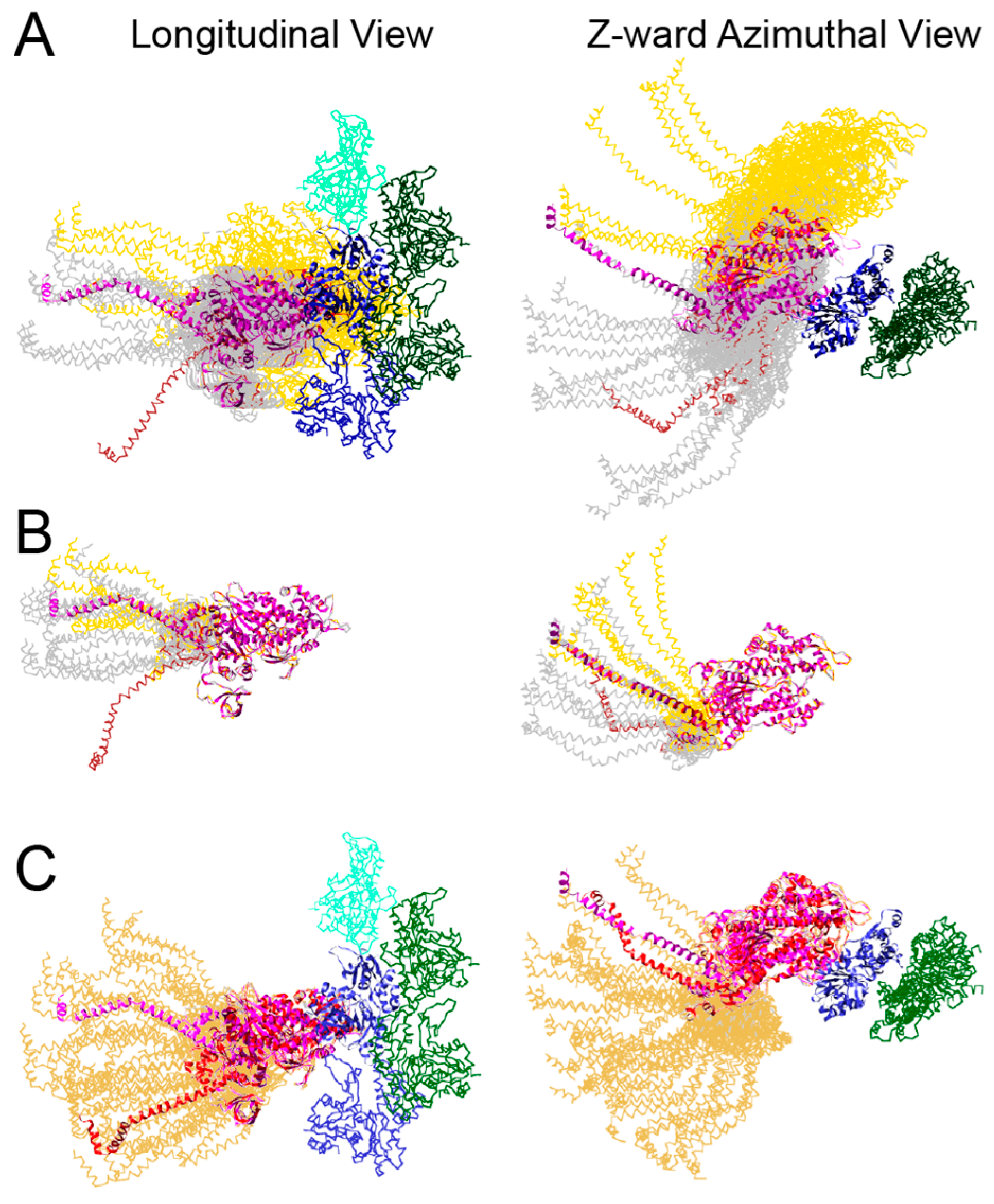
| Year | Event | Reference |
|---|---|---|
| 1975 | 3-D reconstruction from 2-D crystalline arrays | [21] |
| 1984 | 3-D spatial average of Lethocerus rigor flight muscle from tilt series | [22] |
| 1984 | Oblique Section 3-D reconstruction of the vertebrate M-band | [23] |
| 1985 | Freeze substitution of fast frozen active vertebrate skeletal muscle | [24] |
| 1989 | Addition of thick section data to rigor tilt series 3-D reconstruction | [25] |
| 1991 | Oblique section 3-D reconstruction of rigor Lethocerus muscle | [26] |
| 1993 | Crystal structure of a myosin head | [27] |
| 1994 | Oblique section 3-D reconstruction of Lethocerus muscle in AMPPNP | [28] |
| 1994 | Automated data collection for tilt series images | [29] |
| 1997 | Dual axis tilt series tomogram of rigor Lethocerus flight muscle | [30] |
| 1999 | Subvolume classification of Lethocerus flight muscle tomograms | [31] |
| 2001 | Atomic models for rigor Lethocerus flight muscle cross-bridges | [32] |
| 2004 | Tomogram of frozen, freeze substituted rigor Lethocerus flight muscle | [33] |
| 2010 | Tomogram of frozen, freeze substituted, active Lethocerus flight muscle | [34] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Taylor, K.A.; Rahmani, H.; Edwards, R.J.; Reedy, M.K. Insights into Actin-Myosin Interactions within Muscle from 3D Electron Microscopy. Int. J. Mol. Sci. 2019, 20, 1703. https://doi.org/10.3390/ijms20071703
Taylor KA, Rahmani H, Edwards RJ, Reedy MK. Insights into Actin-Myosin Interactions within Muscle from 3D Electron Microscopy. International Journal of Molecular Sciences. 2019; 20(7):1703. https://doi.org/10.3390/ijms20071703
Chicago/Turabian StyleTaylor, Kenneth A., Hamidreza Rahmani, Robert J. Edwards, and Michael K. Reedy. 2019. "Insights into Actin-Myosin Interactions within Muscle from 3D Electron Microscopy" International Journal of Molecular Sciences 20, no. 7: 1703. https://doi.org/10.3390/ijms20071703
APA StyleTaylor, K. A., Rahmani, H., Edwards, R. J., & Reedy, M. K. (2019). Insights into Actin-Myosin Interactions within Muscle from 3D Electron Microscopy. International Journal of Molecular Sciences, 20(7), 1703. https://doi.org/10.3390/ijms20071703





