Intake of Red and Processed Meat, Use of Non-Steroid Anti-Inflammatory Drugs, Genetic Variants and Risk of Colorectal Cancer: A Prospective Study of the Danish “Diet, Cancer and Health” Cohort
Abstract
1. Introduction
2. Results
2.1. Associations between Polymorphisms and CRC
2.2. Gene-Environmental Analyses
3. Discussion
4. Materials and Methods
4.1. Subjects
4.2. Follow-up and Endpoints
4.3. Dietary and Lifestyle Questionnaire
4.4. Genotyping and Selection of Polymorphisms
4.5. Statistics
4.6. Ethics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| NSAID | Non-steroid anti-inflammatory drugs |
| CRC | Colorectal cancer |
| GxE | Gene-environment |
| BMI | Body mass index |
| IRR | Incidence rate ratios |
| CI | Confidence Interval |
References
- World Cancer Research Fund/American Institute for Cancer Research. Food, Nutrition, Physical Activity, and the Prevention of Cancer: A Global Perspective, AIRC. 2018. Available online: https://www.Wcrf.Org/dietandcancer/colorectal-cancer (accessed on 2 March 2019).
- Huxley, R.R.; Nsary-Moghaddam, A.; Clifton, P.; Czernichow, S.; Parr, C.L.; Woodward, M. The impact of dietary and lifestyle risk factors on risk of colorectal cancer: A quantitative overview of the epidemiological evidence. Int. J. Cancer 2009, 125, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Bouvard, V.; Loomis, D.; Guyton, K.Z.; Grosse, Y.; Ghissassi, F.E.; Benbrahim-Tallaa, L.; Guha, N.; Mattock, H.; Straif, K. Carcinogenicity of consumption of red and processed meat. Lancet Oncol. 2015, 16, 1599–1600. [Google Scholar] [CrossRef]
- Friis, S.; Riis, A.H.; Erichsen, R.; Baron, J.A.; Sorensen, H.T. Low-dose aspirin or nonsteroidal anti-inflammatory drug use and colorectal cancer risk: A population-based, case-control study. Ann. Intern. Med. 2015, 163, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Huls, G.; Koornstra, J.J.; Kleibeuker, J.H. Non-steroidal anti-inflammatory drugs and molecular carcinogenesis of colorectal carcinomas. Lancet 2003, 362, 230–232. [Google Scholar] [CrossRef]
- Santarelli, R.L.; Pierre, F.; Corpet, D.E. Processed meat and colorectal cancer: A review of epidemiologic and experimental evidence. Nutr. Cancer. 2008, 60, 131–144. [Google Scholar] [CrossRef] [PubMed]
- Szabo, C. A timeline of hydrogen sulfide (H2S) research: From environmental toxin to biological mediator. Biochem. Pharmacol. 2017, 149, 5–19. [Google Scholar] [CrossRef] [PubMed]
- Erridge, C. Accumulation of stimulants of toll-like receptor (TLR)-2 and TLR4 in meat products stored at 5 °C. J. Food Sci. 2011, 76, H72–H79. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Du, C.; Zhang, N.; Li, M.; Liu, Y.; Zhao, M.; Wang, F.; Luo, F. TGF-β1 mediates the effects of aspirin on colonic tumor cell proliferation and apoptosis. Oncol. Lett. 2018, 15, 5903–5909. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; DuBois, R.N. The role of anti-inflammatory drugs in colorectal cancer. Annu. Rev. Med. 2013, 64, 131–144. [Google Scholar] [CrossRef] [PubMed]
- Alexander, D.D.; Cushing, C.A. Red meat and colorectal cancer: A critical summary of prospective epidemiologic studies. Obes. Rev. 2011, 12, e472–e493. [Google Scholar] [CrossRef] [PubMed]
- Le Leu, R.K.; Young, G.P.; Hu, Y.; Winter, J.; Conlon, M.A. Dietary red meat aggravates dextran sulfate sodium-induced colitis in mice whereas resistant starch attenuates inflammation. Dig. Dis. Sci. 2013, 58, 3475–3482. [Google Scholar] [CrossRef] [PubMed]
- Andersen, V.; Christensen, J.; Overvad, K.; Tjonneland, A.; Vogel, U. Heme oxygenase-1 polymorphism is not associated with risk of colorectal cancer: A Danish prospective study. Eur. J. Gastroenterol. Hepatol. 2011, 23, 282–285. [Google Scholar] [CrossRef] [PubMed]
- Andersen, V.; Christensen, J.; Overvad, K.; Tjonneland, A.; Vogel, U. Polymorphisms in nfkb, pxr, lxr and risk of colorectal cancer in a prospective study of danes. BMC Cancer 2010, 10, 484. [Google Scholar] [CrossRef] [PubMed]
- Andersen, V.; Egeberg, R.; Tjonneland, A.; Vogel, U. Interaction between interleukin-10 (IL-10) polymorphisms and dietary fibre in relation to risk of colorectal cancer in a Danish case-cohort study. BMC Cancer 2012, 12, 183. [Google Scholar] [CrossRef] [PubMed]
- Andersen, V.; Egeberg, R.; Tjonneland, A.; Vogel, U. ABCC2 transporter gene polymorphisms, diet and risk of colorectal cancer: A Danish prospective cohort study. Scand. J. Gastroenterol. 2012, 47, 572–574. [Google Scholar] [CrossRef] [PubMed]
- Andersen, V.; Holst, R.; Kopp, T.I.; Tjonneland, A.; Vogel, U. Interactions between diet, lifestyle and IL10, IL1B, and PTGS2/COX-2 gene polymorphisms in relation to risk of colorectal cancer in a prospective Danish case-cohort study. PLoS ONE 2013, 8, e78366. [Google Scholar] [CrossRef] [PubMed]
- Kopp, T.I.; Andersen, V.; Tjonneland, A.; Vogel, U. Polymorphisms in NFKB1 and TLR4 and interaction with dietary and life style factors in relation to colorectal cancer in a Danish prospective case-cohort study. PLoS ONE 2015, 10, e0116394. [Google Scholar] [CrossRef] [PubMed]
- Kopp, T.I.; Andersen, V.; Tjonneland, A.; Vogel, U. Polymorphisms in ATP-binding cassette transporter genes and interaction with diet and life style factors in relation to colorectal cancer in a Danish prospective case-cohort study. Scand. J. Gastroenterol. 2015, 50, 1469–1481. [Google Scholar] [CrossRef] [PubMed]
- Kopp, T.I.; Vogel, U.; Tjonneland, A.; Andersen, V. Meat and fiber intake and interaction with pattern recognition receptors (TLR1, TLR2, TLR4, and TLR10) in relation to colorectal cancer in a Danish prospective, case-cohort study. Am. J. Clin. Nutr. 2018, 107, 465–479. [Google Scholar] [CrossRef] [PubMed]
- Andersen, V.; Kopp, T.I.; Tjonneland, A.; Vogel, U. No association between Hmox1 and risk of colorectal cancer and no interaction with diet and lifestyle factors in a prospective Danish case-cohort study. Int. J. Mol. Sci. 2015, 16, 1375–1384. [Google Scholar] [CrossRef] [PubMed]
- Andersen, V.; Vogel, U. Systematic review: Interactions between aspirin, and other nonsteroidal anti-inflammatory drugs, and polymorphisms in relation to colorectal cancer. Aliment. Pharmacol. Ther. 2014, 40, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Andersen, V.; Vogel, U. Interactions between meat intake and genetic variation in relation to colorectal cancer. Genes Nutr. 2015, 10, 448. [Google Scholar] [CrossRef] [PubMed]
- Andersen, V.; Holst, R.; Vogel, U. Systematic review: Diet-gene interactions and the risk of colorectal cancer. Aliment. Pharmacol. Ther. 2013, 37, 383–391. [Google Scholar] [CrossRef] [PubMed]
- Nan, H.; Morikawa, T.; Suuriniemi, M.; Imamura, Y.; Werner, L.; Kuchiba, A.; Yamauchi, M.; Hunter, D.J.; Kraft, P.; Giovannucci, E.L.; et al. Aspirin use, 8q24 single nucleotide polymorphism rs6983267, and colorectal cancer according to CTNNB1 alterations. J. Natl. Cancer Inst. 2013, 105, 1852–1861. [Google Scholar] [CrossRef] [PubMed]
- Lemaitre, R.N.; Johnson, C.O.; Hesselson, S.; Sotoodehnia, N.; McKnight, B.; Sitlani, C.M.; Rea, T.D.; King, I.B.; Kwok, P.Y.; Mak, A.; et al. Common variation in fatty acid metabolic genes and risk of incident sudden cardiac arrest. Heart Rhythm. 2014, 11, 471–477. [Google Scholar] [CrossRef] [PubMed]
- Hadj Salem, I.; Kamoun, F.; Louhichi, N.; Trigui, M.; Triki, C.; Fakhfakh, F. Impact of single-nucleotide polymorphisms at the TP53-binding and responsive promoter region of BCL2 gene in modulating the phenotypic variability of LGMD2C patients. Mol. Biol. Rep. 2012, 39, 7479–7486. [Google Scholar] [CrossRef] [PubMed]
- De Angelis, P.M.; Stokke, T.; Thorstensen, L.; Lothe, R.A.; Clausen, O.P. Apoptosis and expression of Bax, Bcl-x, and Bcl-2 apoptotic regulatory proteins in colorectal carcinomas, and association with p53 genotype/phenotype. Mol. Pathol. MP 1998, 51, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Takatsuno, Y.; Mimori, K.; Yamamoto, K.; Sato, T.; Niida, A.; Inoue, H.; Imoto, S.; Kawano, S.; Yamaguchi, R.; Toh, H.; et al. The rs6983267 SNP is associated with MYC transcription efficiency, which promotes progression and worsens prognosis of colorectal cancer. Ann. Surg. Oncol. 2013, 20, 1395–1402. [Google Scholar] [CrossRef] [PubMed]
- Guillem, V.; Amat, P.; Collado, M.; Cervantes, F.; Alvarez-Larran, A.; Martinez, J.; Tormo, E.; Eroles, P.; Solano, C.; Hernandez-Boluda, J.C. Bcl2 gene polymorphisms and splicing variants in chronic myeloid leukemia. Leukemia Res. 2015. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.P.; Zhang, J.; Zhu, H.Y.; Qian, C.L.; Liu, H.; Ji, F.; Shen, Z.Y. Common variation rs6983267 at 8q24.1 and risk of colorectal adenoma and cancer: Evidence based on 31 studies. Tumour. Biol. 2014, 35, 4067–4075. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.L.; Nieters, A.; Hoffmeister, M.; Beckmann, L.; Brenner, H.; Chang-Claude, J. Genetic polymorphisms in TP53, nonsteroidal anti-inflammatory drugs and the risk of colorectal cancer: Evidence for gene-environment interaction? Pharmacogenet. Genom. 2007, 17, 639–645. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.P.; Ha, S.H.; Park, I.S.; Kim, W.H. Induction of apoptosis in colon cancer cells by nonsteroidal anti-inflammatory drugs. Yonsei Med. J. 1998, 39, 287–295. [Google Scholar] [CrossRef] [PubMed]
- Andersen, V.; Ostergaard, M.; Christensen, J.; Overvad, K.; Tjonneland, A.; Vogel, U. Polymorphisms in the xenobiotic transporter multidrug resistance 1 (MDR1) gene and interaction with meat intake in relation to risk of colorectal cancer in a danish prospective case-cohort study. BMC Cancer 2009, 9, 407. [Google Scholar] [CrossRef] [PubMed]
- Cook, R.T. Alcohol abuse, alcoholism, and damage to the immune system—A review. Alcohol. Clin. Exp. Res. 1998, 22, 1927–1942. [Google Scholar] [PubMed]
- Tjonneland, A.; Olsen, A.; Boll, K.; Stripp, C.; Christensen, J.; Engholm, G.; Overvad, K. Study design, exposure variables, and socioeconomic determinants of participation in diet, cancer and health: A population-based prospective cohort study of 57,053 men and women in denmark. Scand. J. Public Health. 2007, 35, 432–441. [Google Scholar] [CrossRef] [PubMed]
- Barlow, W.E.; Ichikawa, L.; Rosner, D.; Izumi, S. Analysis of case-cohort designs. J. Clin. Epidemiol. 1999, 52, 1165–1172. [Google Scholar] [CrossRef]
- Tjonneland, A.; Overvad, K.; Haraldsdottir, J.; Bang, S.; Ewertz, M.; Jensen, O.M. Validation of a semiquantitative food frequency questionnaire developed in Denmark. Int. J. Epidemiol. 1991, 20, 906–912. [Google Scholar] [CrossRef] [PubMed]
- Foodcalc 1.3 computer program 1998. 2009. Available online: http://www.Foodcalc.Dk (accessed on 2 March 2019).
- Prosky, L.; Asp, N.G.; Furda, I.; DeVries, J.W.; Schweizer, T.F.; Harland, B.F. Determination of total dietary fiber in foods and food products: Collaborative study. J. Assoc. Off. Anal. Chem. 1985, 68, 677–679. [Google Scholar] [PubMed]
- Petersen, R.K.; Larsen, S.B.; Jensen, D.M.; Christensen, J.; Olsen, A.; Loft, S.; Nellemann, C.; Overvad, K.; Kristiansen, K.; Tjonneland, A.; et al. Ppargamma-PGC-1α activity is determinant of alcohol related breast cancer. Cancer Lett. 2012, 315, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Miller, S.A.; Dykes, D.D.; Polesky, H.F. A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res. 1988, 16, 1215. [Google Scholar] [CrossRef] [PubMed]
- Langholz, B.; Jiao, J. Computational methods for case–cohort studies. Comput. Stat. Data Anal. 2007, 51, 3737–3748. [Google Scholar] [CrossRef]
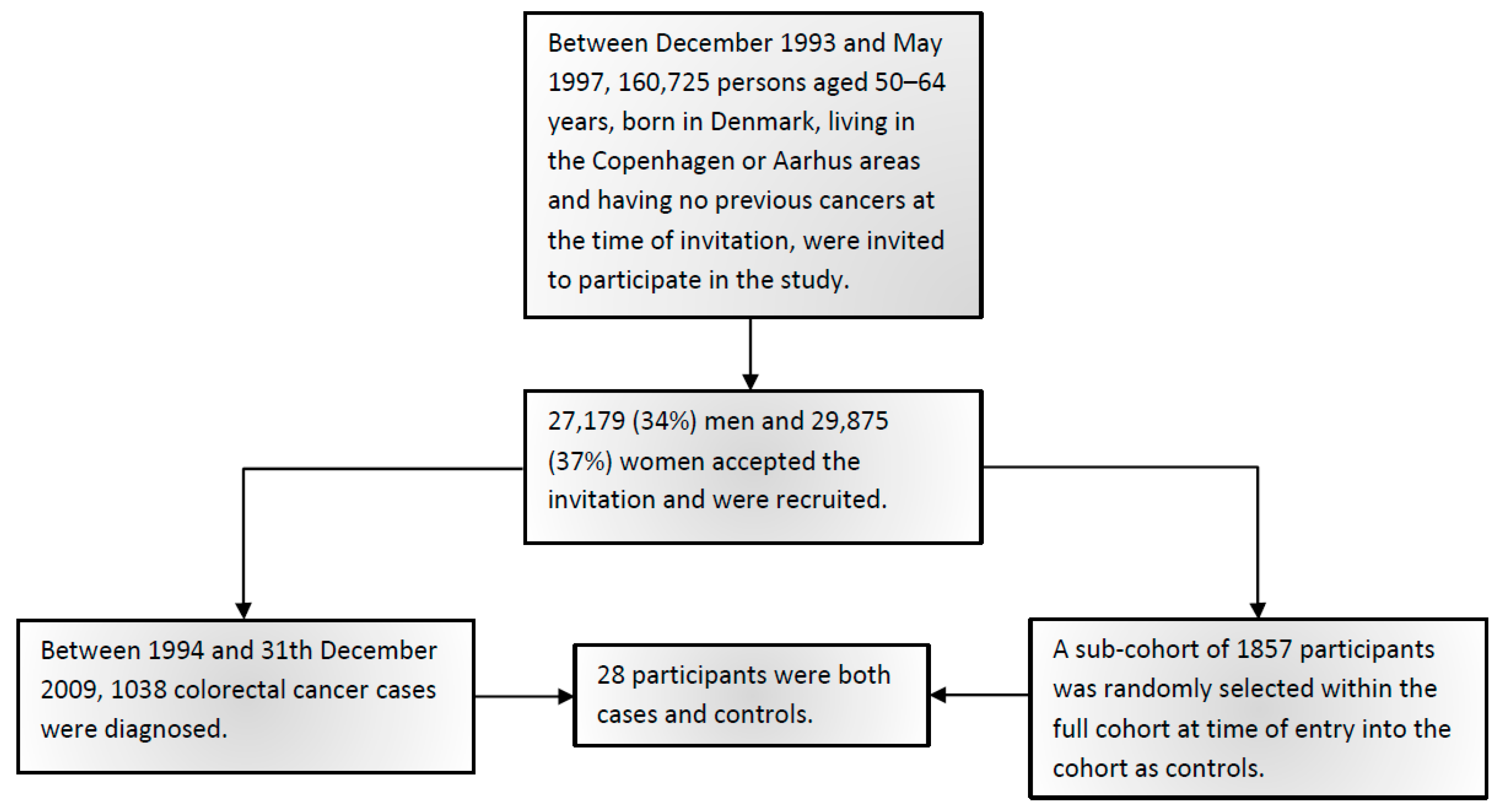
| Variable | Cases | Sub-Cohort | IRR (95%CI) 1 | ||
|---|---|---|---|---|---|
| n (%) | Median (5–95%) | n (%) | Median (5–95%) | ||
| Total | 1038 (100) | 1857 (100) | |||
| Sex | |||||
| Females | 462 (45) | 865 (47) | |||
| Males | 576 (55) | 992 (53) | |||
| Age at entry | 58 (51–65) | 56 (51–64) | |||
| BMI (kg/m2) | 26 (21–34) [3] | 26 (21–33) | 1.05 (1.01–1.10) 4 | ||
| Food intake | |||||
| Alcohol (g/d) 2 | 15 (1–71) | 14 (1–66) | 1.03 (0.98–1.07) 5 | ||
| Dietary fiber (g/d) | 20 (11–33) | 21 (11–34) | 0.83 (0.65–1.08) 6 | ||
| Red and processed meat (g/d) | 112 (46–233) | 109 (41–236) | 1.01 (0.97–1.06) 7 | ||
| Total energy (kJ/d) | 9681 (6115–14712) [4] | 9633 (5922–14820) | 1.00 (1.00–1.00) 8 | ||
| Fruits (g/d) | 166 (24–493) [4] | 176 (27–546) | 0.98 (0.95–1.02) 9 | ||
| Vegetables (g/d) | 153 (46–367) [4] | 163 (50–372) | 1.03 (0.98–1.09) 10 | ||
| Fruit and vegetables (g/d) | 331 (98–796) [4] | 350 (102–818) | 1.00 (0.97–1.02) 11 | ||
| Smoking status | |||||
| Never | 306 (29) | 621 (33) | |||
| Past | 322 (31) | 536 (29) | 1.12 (0.91–1.38) | ||
| Current | 410 (39) | 699 (38) | 1.18 (0.97–1.44) | ||
| NSAID use 3 | |||||
| No | 716 (70) | 1275 (69) | |||
| Yes | 313 (30) | 568 (31) | 0.99 (0.84–1.18) | ||
| HRT use among women | |||||
| Never | 279 (60) | 455 (53) | |||
| Past | 62 (13) | 137 (16) | 0.65 (0.45–0.92) | ||
| Current | 121 (26) | 273 (32) | 0.70 (0.53–0.92) | ||
| Polymorphism | Ncases (%) | Nsub-cohort (%) | IRR (95% CI) 1 | IRR (95% CI) 2 | p-Value 3 |
|---|---|---|---|---|---|
| SLC25A20 rs7623023 | |||||
| AA | 368 (39) | 701 (40) | 1.00 (ref.) | 1.00 (ref.) | |
| GA | 437 (46) | 818 (46) | 1.04 (0.87–1.24) | 1.05 (0.88–1.26) | 0.56 |
| GG | 136 (14) | 245 (14) | 1.08 (0.84–1.38) | 1.07 (0.83–1.38) | 0.62 |
| GA+GG | 573 (61) | 1063 (60) | 1.05 (0.89–1.24) | 1.06 (0.89–1.25) | 0.52 |
| PRKAB1 rs4213 | |||||
| TT | 454 (48) | 875 (50) | 1.00 (ref.) | 1.00 (ref.) | |
| TG | 400 (42) | 728 (41) | 1.02 (0.86–1.21) | 1.01 (0.85–1.20) | 0.95 |
| GG | 90 (10) | 157 (9) | 1.10 (0.82–1.47) | 1.17 (0.87–1.57) | 0.30 |
| TG+GG | 490 (52) | 885 (50) | 1.03 (0.88–1.22) | 1.03 (0.88–1.22) | 0.70 |
| GG vs. TT+TG | 90 (10) | 157 (9) | 1.09 (0.82–1.44) | 1.17 (0.88–1.55) | 0.29 |
| LPCAT1 rs7737692 | |||||
| AA | 378 (40) | 762 (43) | 1.00 (ref.) | 1.00 (ref.) | |
| GA | 437 (46) | 782 (45) | 1.10 (0.92–1.31) | 1.10 (0.92–1.32) | 0.29 |
| GG | 125 (13) | 212 (12) | 1.25 (0.96–1.63) | 1.27 (0.97–1.66) | 0.08 |
| GA+GG | 562 (60) | 994 (57) | 1.13 (0.96–1.33) | 1.14 (0.96–1.34) | 0.14 |
| GG vs. AA+GA | 125 (13) | 212 (12) | 1.19 (0.93–1.52) | 1.21 (0.94–1.55) | 0.14 |
| PLA2G4A rs4402086 | |||||
| AA | 442 (47) | 862 (49) | 1.00 (ref.) | 1.00 (ref.) | |
| GA | 405 (43) | 758 (43) | 1.04 (0.88–1.23) | 1.02 (0.86–1.22) | 0.79 |
| GG | 87 (9) | 141 (8) | 1.22 (0.91–1.65) | 1.23 (0.91–1.66) | 0.19 |
| GA+GG | 492 (53) | 899 (51) | 1.07 (0.91–1.26) | 1.06 (0.89–1.25) | 0.53 |
| GG vs. AA+GA | 87 (9) | 141 (8) | 1.20 (0.90–1.60) | 1.21 (0.90–1.62) | 0.20 |
| ALOX5 rs3780894 | |||||
| AA | 676 (72) | 1264 (72) | 1.00 (ref.) | 1.00 (ref.) | |
| GA | 231 (25) | 463 (26) | 0.93 (0.77–1.13) | 0.92 (0.76–1.12) | 0.43 |
| GG | 27 (3) | 38 (2) | 1.37 (0.81–2.29) | 1.38 (0.81–2.35) | 0.24 |
| GA+GG | 258 (28) | 501 (28) | 0.97 (0.81–1.16) | 0.96 (0.80–1.15) | 0.65 |
| GG vs. AA+GA | 27 (3) | 38 (2) | 1.39 (0.83–2.33) | 1.41 (0.83–2.40) | 0.21 |
| PTGER3 rs6685546 | |||||
| TT | 637 (68) | 1227 (70) | 1.00 (ref.) | 1.00 (ref.) | |
| TC | 276 (29) | 485 (28) | 1.14 (0.95–1.37) | 1.15 (0.96–1.39) | 0.14 |
| CC | 28 (3) | 48 (3) | 0.95 (0.58–1.55) | 0.97 (0.59–1.60) | 0.92 |
| TC+CC | 304 (32) | 533 (30) | 1.12 (0.94–1.34) | 1.13 (0.95–1.36) | 0.18 |
| CC vs. TT+TC | 28 (3) | 48 (3) | 0.91 (0.56–1.49) | 0.93 (0.57–1.54) | 0.79 |
| TP53 rs1042522 | |||||
| GG | 517 (55) | 962 (55) | 1.00 (ref.) | 1.00 (ref.) | |
| GC | 355 (38) | 676 (38) | 0.99 (0.83–1.17) | 0.99 (0.83–1.18) | 0.89 |
| CC | 63 (7) | 120 (7) | 0.94 (0.67–1.31) | 1.00 (0.71–1.40) | 1.00 |
| GC+CC | 418 (45) | 796 (45) | 0.98 (0.83–1.16) | 0.99 (0.84–1.17) | 0.90 |
| CCAT2 rs6983267 | |||||
| GG | 315 (34) | 479 (27) | 1.00 (ref.) | 1.00 (ref.) | |
| TG | 435 (47) | 864 (49) | 0.74 (0.61–0.89) | 0.72 (0.60–0.87) | <0.01 |
| TT | 181 (19) | 413 (24) | 0.68 (0.54–0.85) | 0.66 (0.52–0.83) | <0.01 |
| TG+TT | 616 (66) | 1277 (73) | 0.72 (0.60–0.86) | 0.70 (0.59–0.84) | <0.01 |
| TCF7L2 rs7903146 | |||||
| CC | 492 (53) | 916 (52) | 1.00 (ref.) | 1.00 (ref.) | |
| TC | 366 (39) | 726 (41) | 0.96 (0.81–1.13) | 0.94 (0.79–1.12) | 0.50 |
| TT | 73 (8) | 117 (7) | 1.21 (0.88–1.66) | 1.18 (0.85–1.64) | 0.32 |
| TC+TT | 439 (47) | 843 (48) | 0.99 (0.84–1.17) | 0.97 (0.83–1.15) | 0.76 |
| TT vs. CC+TC | 73 (8) | 117 (7) | 1.23 (0.90–1.68) | 1.21 (0.88–1.66) | 0.24 |
| BCL2 rs2279115 | |||||
| AA | 280 (31) | 508 (29) | 1.00 (ref.) | 1.00 (ref.) | |
| CA | 426 (47) | 861 (50) | 0.86 (0.71–1.04) | 0.84 (0.69–1.02) | 0.09 |
| CC | 196 (22) | 368 (21) | 0.95 (0.75–1.19) | 0.92 (0.73–1.17) | 0.52 |
| CA+CC | 622 (69) | 1229 (71) | 0.89 (0.74–1.06) | 0.87 (0.72–1.04) | 0.13 |
| Polymorphism | Ncases/Nsub-cohort | Ncases/Nsub-cohort | IRR Crude (95%CI) 1 | IRR (95%CI) 2 | p-Value 3 | ||
|---|---|---|---|---|---|---|---|
| No | Yes | No | Yes | No | Yes | ||
| SLC25A20 rs7623023 | |||||||
| AA | 241/469 | 120/216 | 1.00 | 1.09 (0.83–1.45) | 1.00 | 1.07 (0.80–1.42) | |
| GA+GG | 397/717 | 159/320 | 1.08 (0.88–1.32) | 1.04 (0.81–1.34) | 1.09 (0.88–1.33) | 1.04 (0.81–1.35) | 0.59 |
| PRKAB1 rs4213 | |||||||
| TT+TG | 585/1077 | 246/489 | 1.00 | 0.97 (0.81–1.17) | 1.00 | 0.97 (0.80–1.17) | |
| GG | 54/ 104 | 34/ 48 | 0.98 (0.69–1.39) | 1.32 (0.83–2.12) | 1.05 (0.74–1.50) | 1.39 (0.86–2.23) | 0.32 |
| LPCAT1 rs7737692 | |||||||
| AA+GA | 556/1032 | 241/473 | 1.00 | 0.99 (0.82–1.20) | 1.00 | 0.97 (0.80–1.18) | |
| GG | 80/ 148 | 40/ 62 | 1.07 (0.79–1.44) | 1.34 (0.88–2.06) | 1.06 (0.78–1.44) | 1.41 (0.92–2.17) | 0.26 |
| PLA2G4A rs4402086 | |||||||
| AA+GA | 571/1091 | 253/491 | 1.00 | 1.04 (0.86–1.25) | 1.00 | 1.03 (0.85–1.25) | |
| GG | 59/95 | 26/ 42 | 1.22 (0.86–1.73) | 1.22 (0.73–2.05) | 1.25 (0.88–1.77) | 1.20 (0.70–2.05) | 0.83 |
| ALOX5 rs3780894 | |||||||
| AA+GA | 616/1157 | 271/528 | 1.00 | 1.01 (0.84–1.21) | 1.00 | 1.00 (0.83–1.20) | |
| GG | 16/29 | 8/9 | 1.07 (0.57–2.02) | 1.78 (0.66–4.74) | 1.06 (0.55–2.03) | 1.91 (0.71–5.11) | 0.33 |
| PTGER3 rs6685546 | |||||||
| TT+TC | 617/1151 | 273/520 | 1.00 | 1.01 (0.85–1.21) | 1.00 | 1.01 (0.84–1.21) | |
| CC | 20/34 | 7/13 | 0.87 (0.48–1.55) | 1.05 (0.41–2.73) | 0.88 (0.49–1.60) | 1.03 (0.40–2.66) | 0.79 |
| TP53 rs1042522 | |||||||
| GG | 358/632 | 145/308 | 1.00 | 0.86 (0.68–1.09) | 1.00 | 0.85 (0.66–1.08) | |
| GC+CC | 272/549 | 136/228 | 0.87 (0.72–1.07) | 1.11 (0.86–1.44) | 0.87 (0.71–1.07) | 1.11 (0.85–1.44) | 0.04 |
| CCAT2 rs6983267 | |||||||
| GG | 220/318 | 86/152 | 1.00 | 0.88 (0.64–1.21) | 1.00 | 0.87 (0.62–1.20) | |
| TG+TT | 411/862 | 190/383 | 0.69 (0.55–0.85) | 0.74 (0.57–0.95) | 0.67 (0.54–0.83) | 0.71 (0.55–0.92) | 0.31 |
| TCF7L2 rs7903146 | |||||||
| CC+TC | 585/1104 | 251/498 | 1.00 | 1.00 (0.83–1.20) | 1.00 | 0.98 (0.81–1.19) | |
| TT | 45/82 | 27/ 33 | 1.09 (0.74–1.60) | 1.63 (0.95–2.79) | 1.05 (0.71–1.55) | 1.64 (0.95–2.84) | 0.19 |
| BCL2 rs2279115 | |||||||
| AA | 181/339 | 87/159 | 1.00 | 1.13 (0.82–1.56) | 1.00 | 1.07 (0.77–1.48) | |
| CA+CC | 427/833 | 183/364 | 0.95 (0.76–1.18) | 0.96 (0.74–1.24) | 0.91 (0.73–1.14) | 0.93 (0.71–1.21) | 0.82 |
| Polymorphism | IRR (95% CI) 1 | p-Value 2 | IRR (95% CI) 1 | p-Value 2 | IRR (95% CI) 1 | p-value 2 | IRR (95% CI) 1 | p-Value 2 |
|---|---|---|---|---|---|---|---|---|
| Red and Processed Meat (25 g/day) | Fiber (10 g/day) | Fruit and Vegetables (50 g/day) | Alcohol (10 g/day) | |||||
| SLC25A20 rs7623023 | ||||||||
| AA | 1.02 (0.96–1.08) | 0.64 | 0.87 (0.64–1.18) | 0.85 | 0.98 (0.95–1.02) | 0.60 | 1.02 (0.95–1.08) | 0.76 |
| GA+GG | 1.00 (0.95–1.06) | 0.85 (0.64–1.13) | 0.99 (0.96–1.03) | 1.03 (0.97–1.09) | ||||
| PRKAB1 rs4213 | ||||||||
| TT+TG | 1.00 (0.95–1.05) | 0.45 | 0.85 (0.65–1.12) | 0.11 | 0.99 (0.96–1.02) | 0.30 | 1.02 (0.97–1.07) | 0.60 |
| GG | 1.05 (0.93–1.18) | 0.60 (0.37–0.98) | 0.96 (0.90–1.02) | 1.05 (0.95–1.15) | ||||
| LPCAT1 rs7737692 | ||||||||
| AA+GA | 1.02 (0.97–1.07) | 0.06 | 0.88 (0.67–1.16) | 0.09 | 0.99 (0.96–1.02) | 0.65 | 1.02 (0.97–1.07) | 0.87 |
| GG | 0.92 (0.84–1.02) | 0.65 (0.43–0.98) | 0.98 (0.93–1.03) | 1.03 (0.92–1.15) | ||||
| PLA2G4A rs4402086 | ||||||||
| AA+GA | 1.01 (0.96–1.06) | 0.66 | 0.88 (0.67–1.16) | 0.92 | 0.99 (0.96–1.02) | 0.92 | 1.03 (0.98–1.08) | 0.74 |
| GG | 1.03 (0.95–1.12) | 0.90 (0.55–1.48) | 0.99 (0.92–1.06) | 1.01 (0.89–1.14) | ||||
| ALOX5 rs3780894 | ||||||||
| AA+GA | 1.01 (0.96–1.06) | 0.65 | 0.86 (0.65–1.13) | 0.85 | 0.99 (0.96–1.02) | 0.47 | 1.02 (0.98–1.07) | 0.88 |
| GG | 1.06 (0.85–1.32) | 0.80 (0.39–1.67) | 0.96 (0.87–1.05) | 1.07 (0.65–1.75) | ||||
| PTGER3 rs6685546 | ||||||||
| TT+TC | 1.01 (0.96–1.06) | 0.23 | 0.85 (0.65–1.11) | 0.85 | 0.99 (0.96–1.02) | 0.90 | 1.02 (0.98–1.07) | 0.64 |
| CC | 0.91 (0.77–1.08) | 0.90 (0.50–1.62) | 0.99 (0.91–1.08) | 1.08 (0.87–1.34) | ||||
| TP53 rs1042522 | ||||||||
| GG | 1.00 (0.94–1.06) | 0.34 | 0.82 (0.62–1.09) | 0.31 | 1.00 (0.96–1.03) | 0.31 | 0.99 (0.94–1.05) | 0.04 |
| GC+CC | 1.03 (0.97–1.09) | 0.93 (0.68–1.27) | 0.98 (0.94–1.01) | 1.08 (1.01–1.16) | ||||
| CCAT2 rs6983267 | ||||||||
| GG | 1.05 (0.98–1.13) | 0.04 | 0.83 (0.61–1.13) | 0.83 | 1.00 (0.96–1.04) | 0.46 | 1.05 (0.98–1.13) | 0.34 |
| TG+TT | 0.98 (0.93–1.03) | 0.81 (0.60–1.08) | 0.98 (0.95–1.01) | 1.01 (0.96–1.07) | ||||
| TCF7L2 rs7903146 | ||||||||
| CC+TC | 1.01 (0.96–1.06) | 0.68 | 0.84 (0.64–1.11) | 0.30 | 0.99 (0.96–1.02) | 0.61 | 1.02 (0.97–1.07) | 0.68 |
| TT | 1.03 (0.93–1.14) | 1.10 (0.65–1.87) | 0.97 (0.88–1.06) | 1.05 (0.92–1.20) | ||||
| BCL2 rs2279115 | ||||||||
| AA | 1.02 (0.94–1.10) | 0.96 | 0.86 (0.61–1.22) | 0.63 | 1.00 (0.96–1.04) | 0.33 | 1.00 (0.92–1.08) | 0.30 |
| CA+CC | 1.02 (0.97–1.07) | 0.92 (0.70–1.22) | 0.98 (0.95–1.01) | 1.05 (0.99–1.10) |
| Expected Interaction | SNP ID | Nearby Gene | Allele | MAF | Bio Effect | Ref |
|---|---|---|---|---|---|---|
| Meat | rs7623023 | SLC25A20 | G/A | 0.34 | Carnitine acylcarnitine translocase | [26] |
| Meat | rs4213 | PRKAB1 | G/T | 0.31 | AMP-activated protein kinase β1 subunit | - |
| Meat | rs7737692 | LPCAT1 | G/A | 0.36 | Lysophosphatidylcholine acetyltransferase | - |
| Meat | rs4402086 | PLA2G4A | G/A | 0.26 | Phospholipase A2 | - |
| Meat | rs3780894 | ALOX5 | G/A | 0.16 | Arachidonate 5-lipoxygenase | - |
| Meat | rs6685546 | PTGER3 | C/T | 0.14 | Prostaglandin E receptor 3 | - |
| Aspirin | rs1042522 | TP53 | C/G | 0.46 | G allele increase p53 level | [27,28] |
| Aspirin | rs6983267 | CCAT2 | G/T | 0.39 | Aspirin suppresses the binding of TCF7L2 to the T allele | [22,29] |
| Aspirin | rs7903146 | TCF7L2 | T/C | 0.23 | Intron, transcription factor that plays a key role in the Wnt signaling pathway | [22] |
| Aspirin | rs2279115 | BCL2 | G/F | 0.46 | Expression of BCL2 alternative splicing transcripts (BCL2-α, BCL2-β) in healthy donors | [27,30] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Andersen, V.; Halekoh, U.; Tjønneland, A.; Vogel, U.; Kopp, T.I. Intake of Red and Processed Meat, Use of Non-Steroid Anti-Inflammatory Drugs, Genetic Variants and Risk of Colorectal Cancer: A Prospective Study of the Danish “Diet, Cancer and Health” Cohort. Int. J. Mol. Sci. 2019, 20, 1121. https://doi.org/10.3390/ijms20051121
Andersen V, Halekoh U, Tjønneland A, Vogel U, Kopp TI. Intake of Red and Processed Meat, Use of Non-Steroid Anti-Inflammatory Drugs, Genetic Variants and Risk of Colorectal Cancer: A Prospective Study of the Danish “Diet, Cancer and Health” Cohort. International Journal of Molecular Sciences. 2019; 20(5):1121. https://doi.org/10.3390/ijms20051121
Chicago/Turabian StyleAndersen, Vibeke, Ulrich Halekoh, Anne Tjønneland, Ulla Vogel, and Tine Iskov Kopp. 2019. "Intake of Red and Processed Meat, Use of Non-Steroid Anti-Inflammatory Drugs, Genetic Variants and Risk of Colorectal Cancer: A Prospective Study of the Danish “Diet, Cancer and Health” Cohort" International Journal of Molecular Sciences 20, no. 5: 1121. https://doi.org/10.3390/ijms20051121
APA StyleAndersen, V., Halekoh, U., Tjønneland, A., Vogel, U., & Kopp, T. I. (2019). Intake of Red and Processed Meat, Use of Non-Steroid Anti-Inflammatory Drugs, Genetic Variants and Risk of Colorectal Cancer: A Prospective Study of the Danish “Diet, Cancer and Health” Cohort. International Journal of Molecular Sciences, 20(5), 1121. https://doi.org/10.3390/ijms20051121





