Taste Receptors: New Players in Sperm Biology
Abstract
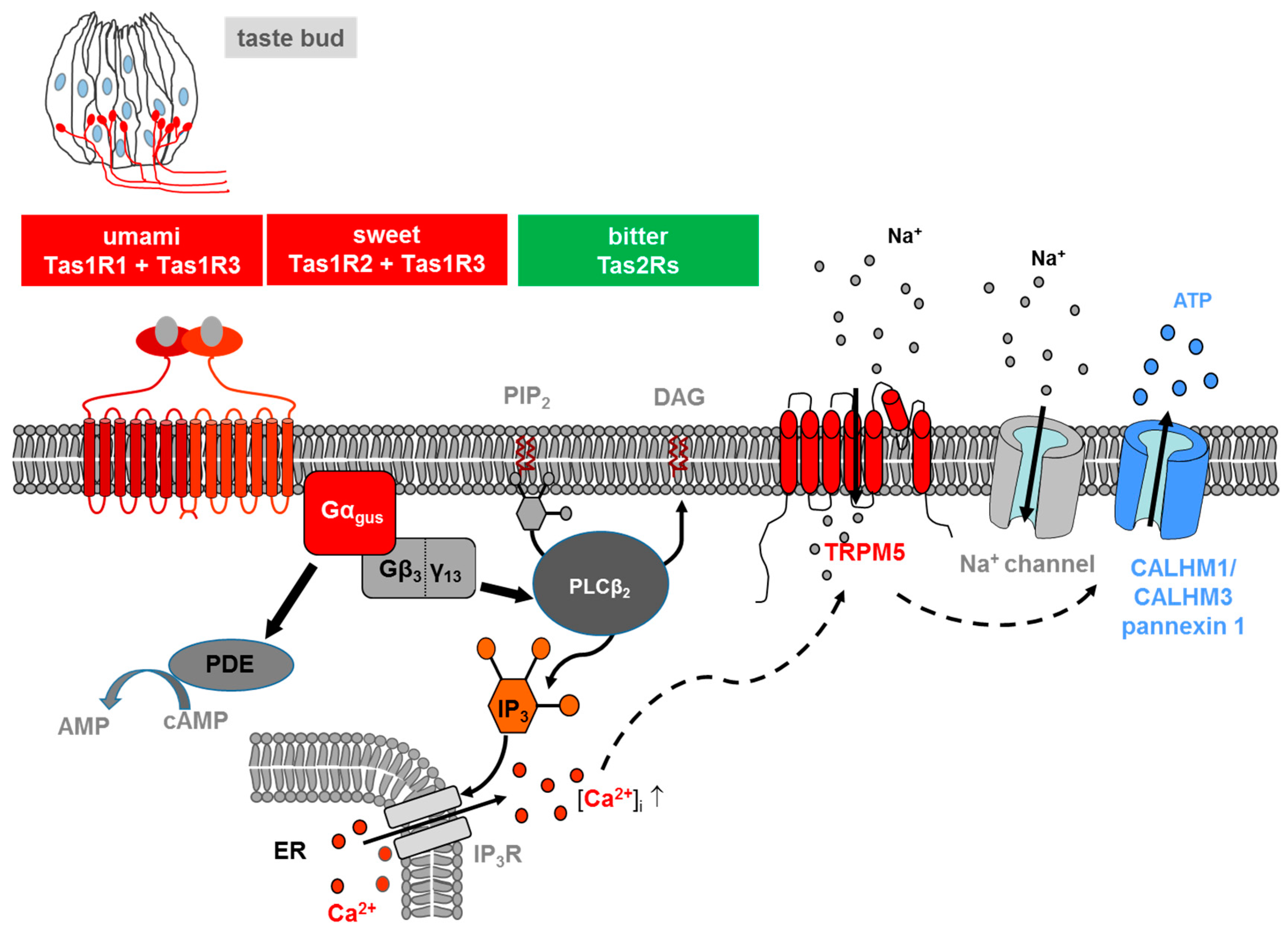
1. Taste Receptors and Signal Transduction
2. Expression of Taste Receptors in Different Tissues
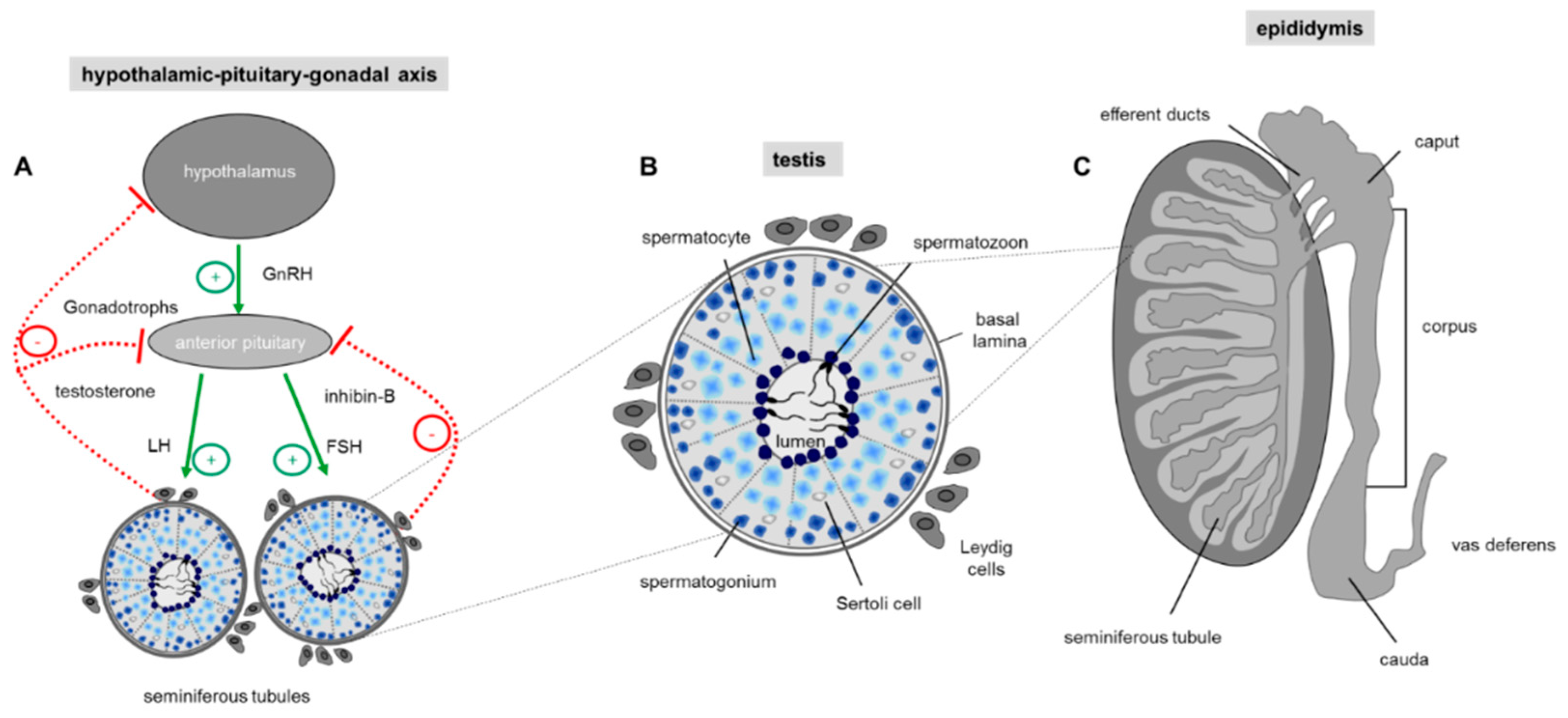
3. Taste Receptors and Spermatogenesis
3.1. Spermatogenesis
3.2. Apoptosis
4. Taste Receptors in Epididymal Maturation and Sperm Function
4.1. Epididymal Sperm Maturation
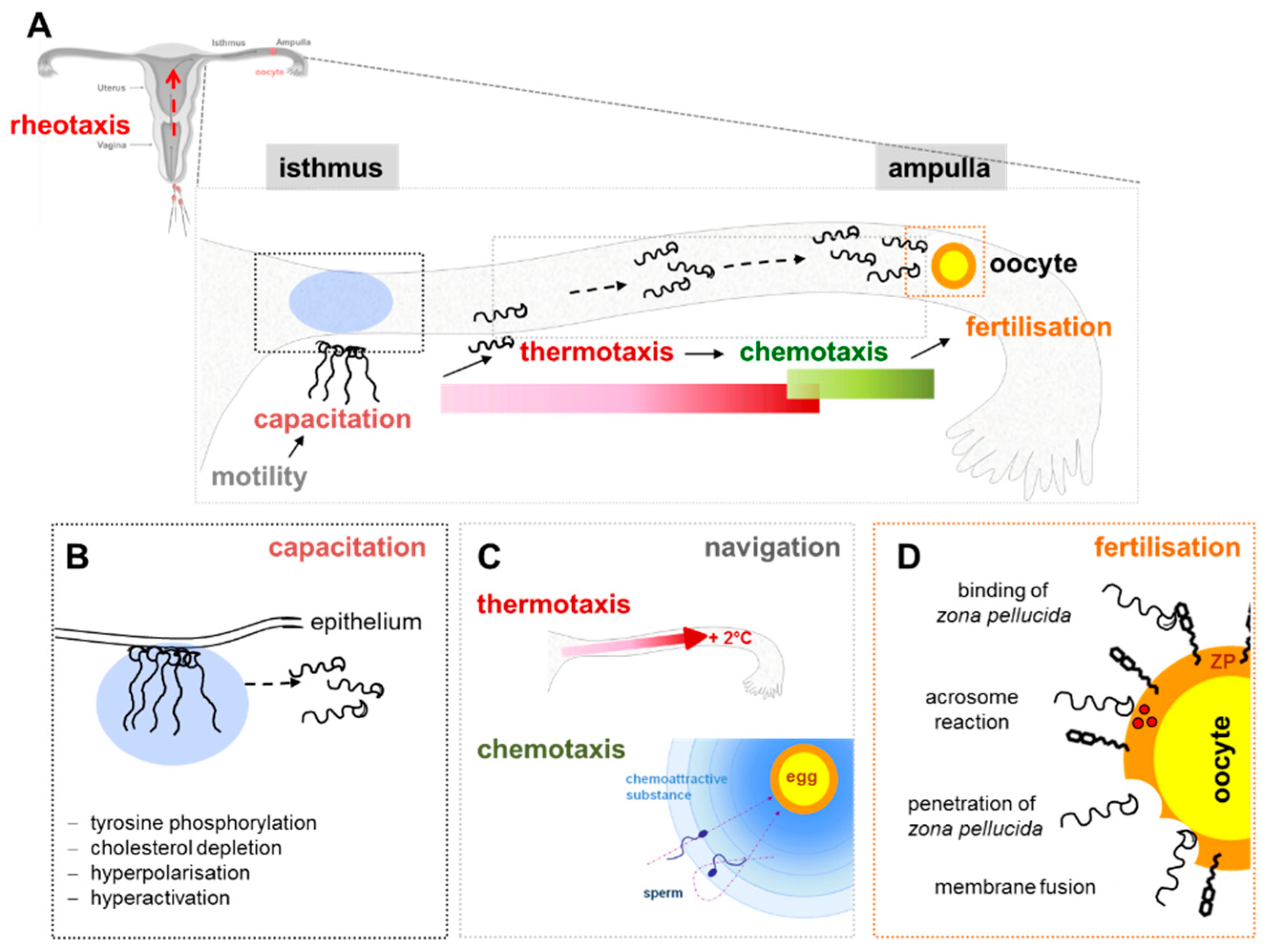
4.2. Sperm Function: Capacitation, Motility, Chemotaxis, Acrosome Reaction
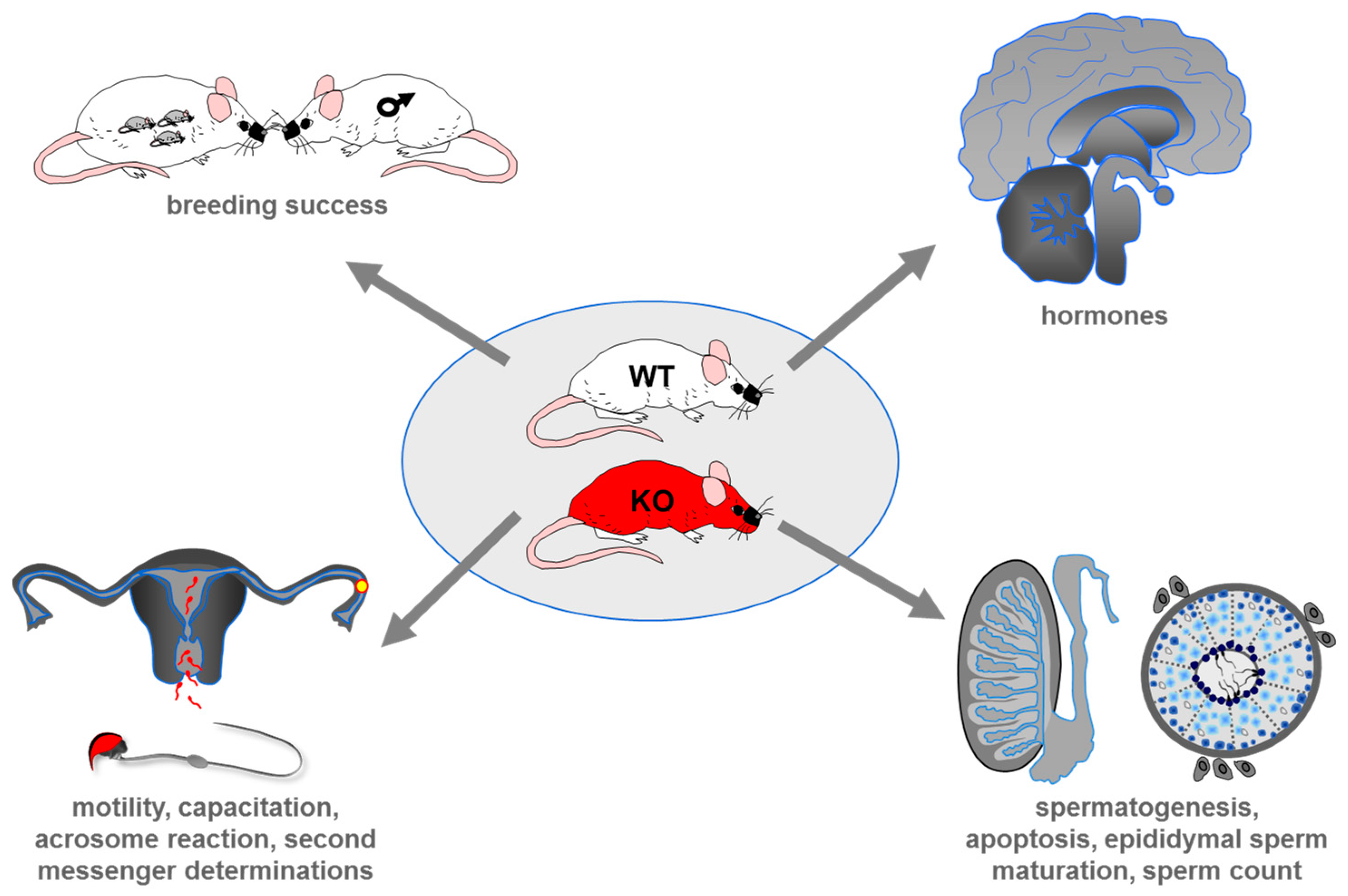
5. Genetic Deletion of Taste Receptors in Mouse and its Impact on Male Reproduction
6. Polymorphisms in Taste Receptor Genesand Male Infertility
7. Conclusions
Funding
Conflicts of Interest
Abbreviations
| ATP | Adenosine triphosphate |
| cAMP | Cyclic adenosine monophosphate |
| HCO3-- | Bicarbonate |
| Ca2+ | Calcium |
| CALHM | Calcium homeostasis modulator |
| CASA | Computer-assisted motility analysis |
| CatSper | Cation channel of sperm |
| CRAC | Cholesterol recognition/interaction amino acid consensus |
| CREM | cAMP responsive element modulator |
| DAG | Diacylglycerol |
| ER | Endoplasmic reticulum |
| FSH | Follicle stimulating hormone |
| GABA | Gamma-aminobutyric acid |
| GnRH | Gonadotropin-releasing hormone |
| GPCRs | G-protein-coupled receptors |
| HPG | Hypothalamic-pituitary-gonadal axis |
| IP3 | Inositol 1,4,5-triphosphate |
| IP3R | Inositol 1,4,5-trisphosphate receptor |
| KO | Knock-out |
| LH | Leutinizing hormone |
| Na+ | Sodium |
| OR | Odorant receptor |
| PDE | Phosphodiesterase |
| PIP2 | Phosphatidylinositol 4,5-bisphosphate |
| PLCβ2 | Phospholipase C β2 |
| P2X2/3 | Purinergic receptor |
| PROP | 6-n-propylthiouracil |
| PTC | Phenylthiocarbamide |
| SNP | Single-nucleotide polymorphism |
| TAS | Taste Receptor |
| TAS1s | Type 1 taste receptors |
| TAS2s | Type 2 taste receptors |
| TRPM5 | Transient receptor potential, melastatin family member 5 |
| TRPV6 | Transient receptor potential vanilloid 6 |
| WEE2-AS1 | WEE2 antisense RNA one gene |
| WT | Wild-type |
References
- Hoon, M.A.; Adler, E.; Lindemeier, J.; Battey, J.F.; Ryba, N.J.; Zuker, C.S. Putative mammalian taste receptors: A class of taste-specific GPCRs with distinct topographic selectivity. Cell 1999, 96, 541–551. [Google Scholar] [CrossRef]
- Roper, S.D.; Chaudhari, N. Taste buds: Cells, signals and synapses. Nat. Rev. Neurosci. 2017, 18, 485–497. [Google Scholar] [CrossRef] [PubMed]
- Lindemann, B. Receptors and transduction in taste. Nature 2001, 413, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Nelson, G.; Chandrashekar, J.; Hoon, M.A.; Feng, L.; Zhao, G.; Ryba, N.J.P.; Zuker, C.S. An amino-acid taste receptor. Nature 2002, 416, 199. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Zhou, M. Depletion of bitter taste transduction leads to massive spermatid loss in transgenic mice. Mol. Hum. Reprod. 2012, 18, 289–297. [Google Scholar] [CrossRef]
- Kuroda, M.; Miyamura, N. Mechanism of the perception of “kokumi” substances and the sensory characteristics of the “kokumi” peptide, γ-Glu-Val-Gly. Flavour 2015, 4, 11. [Google Scholar] [CrossRef]
- Besnard, P.; Passilly-Degrace, P.; Khan, N.A. Taste of Fat: A Sixth Taste Modality? Physiol. Rev. 2016, 96, 151–176. [Google Scholar] [CrossRef]
- Finger, T.E.; Kinnamon, S.C. Taste isn’t just for taste buds anymore. F1000 Biol. Rep. 2011, 3, 20. [Google Scholar] [CrossRef]
- Strosberg, A.D. Structure/function relationship of proteins belonging to the family of receptors coupled to GTP-binding proteins. Eur. J. Biochem. 1991, 196, 1–10. [Google Scholar] [CrossRef]
- Kinnamon, S.C. Taste receptor signalling—From tongues to lungs. Acta Physiol. 2012, 204, 158–168. [Google Scholar] [CrossRef]
- Bachmanov, A.A.; Beauchamp, G.K. Taste receptor genes. Annu. Rev. Nutr. 2007, 27, 389–414. [Google Scholar] [CrossRef] [PubMed]
- Chandrashekar, J.; Hoon, M.A.; Ryba, N.J.P.; Zuker, C.S. The receptors and cells for mammalian taste. Nature 2006, 444, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Montmayeur, J.P.; Matsunami, H. Receptors for bitter and sweet taste. Curr. Opin. Neurobiol. 2002, 12, 366–371. [Google Scholar] [CrossRef]
- Kim, S.-K.; Chen, Y.; Abrol, R.; Goddard, W.A.; Guthrie, B. Activation mechanism of the G protein-coupled sweet receptor heterodimer with sweeteners and allosteric agonists. Proc. Natl. Acad. Sci. USA 2017, 114, 2568–2573. [Google Scholar] [CrossRef] [PubMed]
- Cui, M.; Jiang, P.; Maillet, E.; Max, M.; Margolskee, R.F.; Osman, R. The heterodimeric sweet taste receptor has multiple potential ligand binding sites. Curr. Pharm. Des. 2006, 12, 4591–4600. [Google Scholar] [CrossRef]
- Treesukosol, Y.; Smith, K.R.; Spector, A.C. The functional role of the T1R family of receptors in sweet taste and feeding. Physiol. Behav. 2011, 105, 14–26. [Google Scholar] [CrossRef]
- Margolskee, R.F.; Dyer, J.; Kokrashvili, Z.; Salmon, K.S.H.; Ilegems, E.; Daly, K.; Maillet, E.L.; Ninomiya, Y.; Mosinger, B.; Shirazi-Beechey, S.P. T1R3 and gustducin in gut sense sugars to regulate expression of Na+-glucose cotransporter 1. Proc. Natl. Acad. Sci. USA 2007, 104, 15075–15080. [Google Scholar] [CrossRef]
- Meyerhof, W.; Born, S.; Brockhoff, A.; Behrens, M. Molecular biology of mammalian bitter taste receptors. A review. Flavour Fragr. J. 2011, 26, 260–268. [Google Scholar] [CrossRef]
- Chaudhari, N.; Roper, S.D. The cell biology of taste. J. Cell Biol. 2010, 190, 285–296. [Google Scholar] [CrossRef]
- Liman, E.R.; Zhang, Y.V.; Montell, C. Peripheral Coding of Taste. Neuron 2014, 81, 984–1000. [Google Scholar] [CrossRef]
- Liu, D.; Liman, E.R. Intracellular Ca2+ and the phospholipid PIP2 regulate the taste transduction ion channel TRPM5. Proc. Natl. Acad. Sci. USA 2003, 100, 15160–15165. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, T.; Chubanov, V.; Gudermann, T.; Montell, C. TRPM5 is a voltage-modulated and Ca(2+)-activated monovalent selective cation channel. Curr. Biol. 2003, 13, 1153–1158. [Google Scholar] [CrossRef]
- Taruno, A.; Matsumoto, I.; Ma, Z.; Marambaud, P.; Foskett, J.K. How do taste cells lacking synapses mediate neurotransmission? CALHM1, a voltage-gated ATP channel. Bioessays 2013, 35, 1111–1118. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.-J.; Maruyama, Y.; Dvoryanchikov, G.; Pereira, E.; Chaudhari, N.; Roper, S.D. The role of pannexin 1 hemichannels in ATP release and cell-cell communication in mouse taste buds. Proc. Natl. Acad. Sci. USA 2007, 104, 6436–6441. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Taruno, A.; Ohmoto, M.; Jyotaki, M.; Lim, J.C.; Miyazaki, H.; Niisato, N.; Marunaka, Y.; Lee, R.J.; Hoff, H.; et al. CALHM3 Is Essential for Rapid Ion Channel-Mediated Purinergic Neurotransmission of GPCR-Mediated Tastes. Neuron 2018, 98, 547–561. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.A.; Stone, L.M.; Pereira, E.; Yang, R.; Kinnamon, J.C.; Dvoryanchikov, G.; Chaudhari, N.; Finger, T.E.; Kinnamon, S.C.; Roper, S.D. Knocking out P2X receptors reduces transmitter secretion in taste buds. J. Neurosci. 2011, 31, 13654–13661. [Google Scholar] [CrossRef] [PubMed]
- Finger, T.E.; Danilova, V.; Barrows, J.; Bartel, D.L.; Vigers, A.J.; Stone, L.; Hellekant, G.; Kinnamon, S.C. ATP Signaling Is Crucial for Communication from Taste Buds to Gustatory Nerves. Science 2005, 310, 1495–1499. [Google Scholar] [CrossRef]
- Clapp, T.R.; Trubey, K.R.; Vandenbeuch, A.; Stone, L.M.; Margolskee, R.F.; Chaudhari, N.; Kinnamon, S.C. Tonic activity of Galpha-gustducin regulates taste cell responsivity. FEBS Lett. 2008, 582, 3783–3787. [Google Scholar] [CrossRef]
- Lee, S.-J.; Depoortere, I.; Hatt, H. Therapeutic potential of ectopic olfactory and taste receptors. Nat. Rev. Drug Discov. 2018, 18, 116–138. [Google Scholar] [CrossRef]
- Fujita, T. Taste cells in the gut and on the tongue. Their common, paraneuronal features. Physiol. Behav. 1991, 49, 883–885. [Google Scholar] [CrossRef]
- Rozengurt, E. Taste Receptors in the Gastrointestinal Tract. I. Bitter taste receptors and α-gustducin in the mammalian gut. Am. J. Physiol. Liver Physiol. 2006, 29, G171–G177. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.S.; Yehuda, B.S.; Moninger, T.O.; Kline, J.N.; Welsh, M.J. Motile cilia of human airway epithelia are chemosensory. Science 2009, 325, 1131–1134. [Google Scholar] [CrossRef] [PubMed]
- Deshpande, D.A.; Wang, W.C.H.; McIlmoyle, E.L.; Robinett, K.S.; Schillinger, R.M.; An, S.S.; Sham, J.S.K.; Liggett, S.B. Bitter taste receptors on airway smooth muscle bronchodilate by localized calcium signaling and reverse obstruction. Nat. Med. 2010, 16, 1299. [Google Scholar] [CrossRef] [PubMed]
- Tizzano, M.; Cristofoletti, M.; Sbarbati, A.; Finger, T.E. Expression of taste receptors in Solitary Chemosensory Cells of rodent airways. BMC Pulm. Med. 2011, 11, 3. [Google Scholar] [CrossRef]
- Elliott, R.A.; Kapoor, S.; Tincello, D.G. Expression and distribution of the sweet taste receptor isoforms T1R2 and T1R3 in human and rat bladders. J. Urol. 2011, 186, 2455–2462. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, K. Expression of the Sweet Receptor Protein, T1R3, in the Human Liver and Pancreas. J. Vet. Med. Sci. 2004, 66, 1311–1314. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa, Y.; Nagasawa, M.; Yamada, S.; Hara, A.; Mogami, H.; Nikolaev, V.O.; Lohse, M.J.; Shigemura, N.; Ninomiya, Y.; Kojima, I. Sweet taste receptor expressed in pancreatic β-cells activates the calcium and cyclic AMP signaling systems and stimulates insulin secretion. PLoS ONE 2009, 4, e5106. [Google Scholar] [CrossRef] [PubMed]
- Ren, X. Sweet taste signaling functions as a hypothalamic glucose sensor. Front. Integr. Neurosci. 2009, 3, 12. [Google Scholar] [CrossRef] [PubMed]
- Max, M.; Shanker, Y.G.; Huang, L.; Rong, M.; Liu, Z.; Campagne, F.; Weinstein, H.; Damak, S.; Margolskee, R.F. Tas1r3, encoding a new candidate taste receptor, is allelic to the sweet responsiveness locus Sac. Nat. Genet. 2001, 28, 58. [Google Scholar] [CrossRef] [PubMed]
- Kiuchi, S.; Yamada, T.; Kiyokawa, N.; Saito, T.; Fujimoto, J.; Yasue, H. Genomic structure of swine taste receptor family 1 member 3, TAS1R3, and its expression in tissues. Cytogenet. Genome Res. 2006, 115, 51–61. [Google Scholar] [CrossRef]
- Fehr, J.; Meyer, D.; Widmayer, P.; Borth, H.C.; Ackermann, F.; Wilhelm, B.; Gudermann, T.; Boekhoff, I. Expression of the G-protein α-subunit gustducin in mammalian spermatozoa. J. Comp. Physiol. A Neuroethol. Sens. Neural Behav. Physiol. 2007, 191, 21–34. [Google Scholar] [CrossRef] [PubMed]
- Meyer, D.; Voigt, A.; Widmayer, P.; Borth, H.; Huebner, S.; Breit, A.; Marschall, S.; de Angelis, M.H.; Boehm, U.; Meyerhof, W.; et al. Expression of tas1 taste receptors in mammalian spermatozoa: Functional role of tas1r1 in regulating basal ca2+and camp concentrations in spermatozoa. PLoS ONE 2012, 7, e32354. [Google Scholar] [CrossRef] [PubMed]
- Voigt, A.; Hübner, S.; Lossow, K.; Hermans-Borgmeyer, I.; Boehm, U.; Meyerhof, W. Genetic labeling of Tas1r1 and Tas2r131 taste receptor cells in mice. Chem. Sens. 2012, 37, 897–911. [Google Scholar] [CrossRef]
- Li, F. Taste perception: From the tongue to the testis. Mol. Hum. Reprod. 2013, 19, 349–360. [Google Scholar] [CrossRef] [PubMed]
- Foster, S.R.; Porrello, E.R.; Purdue, B.; Chan, H.W.; Voigt, A.; Frenzel, S.; Hannan, R.D.; Moritz, K.M.; Simmons, D.G.; Molenaar, P.; et al. Expression, Regulation and Putative Nutrient-Sensing Function of Taste GPCRs in the Heart. PLoS ONE 2013, 8, e64579. [Google Scholar] [CrossRef]
- Henquin, J.C. Do pancreatic β cells “taste” nutrients to secrete insulin? Sci. Signal. 2012, 5, pe36. [Google Scholar] [CrossRef]
- Malaisse, W.J.; Vanonderbergen, A.; Louchami, K.; Jijakli, H.; Malaisse-Lagae, F. Effects of artificial sweeteners on insulin release and cationic fluxes in rat pancreatic islets. Cell. Signal. 1998, 10, 727–733. [Google Scholar] [CrossRef]
- Kyriazis, G.A.; Soundarapandian, M.M.; Tyrberg, B. Sweet taste receptor signaling in beta cells mediates fructose-induced potentiation of glucose-stimulated insulin secretion. PNAS 2012, 109, E524–E532. [Google Scholar] [CrossRef] [PubMed]
- Simon, B.R.; Learman, B.S.; Parlee, S.D.; Scheller, E.L.; Mori, H.; Cawthorn, W.P.; Ning, X.; Krishnan, V.; Ma, Y.L.; Tyrberg, B.; et al. Sweet taste receptor deficient mice have decreased adiposity and increased bone mass. PLoS ONE 2014, 9, e86454. [Google Scholar] [CrossRef]
- Iwatsuki, K.; Nomura, M.; Shibata, A.; Ichikawa, R.; Enciso, P.L.M.; Wang, L.; Takayanagi, R.; Torii, K.; Uneyama, H. Generation and characterization of T1R2-LacZ knock-in mouse. Biochem. Biophys. Res. Commun. 2010, 402, 495–499. [Google Scholar] [CrossRef]
- Lu, P.; Zhang, C.-H.; Lifshitz, L.M.; ZhuGe, R. Extraoral bitter taste receptors in health and disease. J. Gen. Physiol. 2017, 149, 181–197. [Google Scholar] [CrossRef] [PubMed]
- Mennella, J.A.; Spector, A.C.; Reed, D.R.; Coldwell, S.E. The bad taste of medicines: Overview of basic research on bitter taste. Clin. Ther. 2013, 35, 1225–1246. [Google Scholar] [CrossRef] [PubMed]
- Griswold, M.D. Spermatogenesis: The Commitment to Meiosis. Physiol. Rev. 2016, 96, 1–17. [Google Scholar] [CrossRef]
- Bergmann, M. Spermatogenesis—Physiology and pathophysiology. Urol. A 2005, 44, 44. [Google Scholar] [CrossRef]
- Choi, D. The consequences of mutations in the reproductive endocrine system. Dev. Reprod. 2012, 16, 235–251. [Google Scholar] [CrossRef] [PubMed]
- O’Shaughnessy, P.J. Hormonal control of germ cell development and spermatogenesis. Semin. Cell Dev. Biol. 2014. [Google Scholar] [CrossRef]
- Ramaswamy, S.; Weinbauer, G.F. Endocrine control of spermatogenesis: Role of FSH and LH/ testosterone. Spermatogenesis 2014, 4, e996025. [Google Scholar] [CrossRef]
- Crisóstomo, L.; Alves, M.G.; Gorga, A.; Sousa, M.; Riera, M.F.; Galardo, M.N.; Meroni, S.B.; Oliveira, P.F. Molecular mechanisms and signaling pathways involved in the nutritional support of spermatogenesis by sertoli cells. In Methods in Molecular Biology; Humana Press: New York, NY, USA, 2018; ISBN 978-1-49-397698-0. [Google Scholar]
- Alves, M.G.; Rato, L.; Carvalho, R.A.; Moreira, P.I.; Socorro, S.; Oliveira, P.F. Hormonal control of Sertoli cell metabolism regulates spermatogenesis. Cell. Mol. Life Sci. 2013, 70, 777–793. [Google Scholar] [CrossRef]
- De Kretser, D.M.; Loveland, K.L.; Meinhardt, A.; Simorangkir, D.; Wreford, N. Spermatogenesis. Hum. Reprod. 1998, 13, 1–18. [Google Scholar] [CrossRef]
- Ubuka, T.; Son, Y.L.; Tobari, Y.; Narihiro, M.; Bentley, G.E.; Kriegsfeld, L.J.; Tsutsui, K. Central and direct regulation of testicular activity by gonadotropin-inhibitory hormone and its receptor. Front. Endocrinol. 2014, 5, 8. [Google Scholar] [CrossRef]
- Barakat, B.; Itman, C.; Mendis, S.H.; Loveland, K.L. Activins and inhibins in mammalian testis development: New models, new insights. Mol. Cell. Endocrinol. 2012, 359, 66–77. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.R.; Dong, H.S.; Yang, W.X. Regulators in the apoptotic pathway during spermatogenesis: Killers or guards? Gene 2016, 582, 97–111. [Google Scholar] [CrossRef]
- Aitken, R.J.; Baker, M.A. Causes and consequences of apoptosis in spermatozoa; contributions to infertility and impacts on development. Int. J. Dev. Biol. 2013, 57, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Boekelheide, K.; Fleming, S.L.; Johnson, K.J.; Patel, S.R.; Schoenfeld, H.A. Role of Sertoli cells in injury-associated testicular germ cell apoptosis (44558). Proc. Soc. Exp. Biol. Med. 2000, 225, 105–115. [Google Scholar] [CrossRef] [PubMed]
- Grunewald, S.; Sharma, R.; Paasch, U.; Glander, H.J.; Agarwal, A. Impact of caspase activation in human spermatozoa. Microsc. Res. Tech. 2009, 72, 878–888. [Google Scholar] [CrossRef] [PubMed]
- Shaha, C. Modulators of spermatogenic cell survival. Soc. Reprod. Fertil. Suppl. 2007, 63, 173–186. [Google Scholar] [PubMed]
- Shukla, K.K. Apoptosis, spermatogenesis and male infertility. Front. Biosci. 2012, 4, 746–754. [Google Scholar] [CrossRef]
- Sinha Hikim, A. Hormonal and genetic control of germ cell apoptosis in the testis. Rev. Reprod. 1999, 4, 38–47. [Google Scholar] [CrossRef]
- Mima, M.; Greenwald, D.; Ohlander, S. Environmental Toxins and Male Fertility. Curr. Urol. Rep. 2018, 19, 50. [Google Scholar] [CrossRef]
- Mathur, P.P.; D’Cruz, S.C. The effect of environmental contaminants on testicular function. Asian J. Androl. 2011, 13, 585. [Google Scholar] [CrossRef]
- Jaggupilli, A.; Howard, R.; Upadhyaya, J.D.; Bhullar, R.P.; Chelikani, P. Bitter taste receptors: Novel insights into the biochemistry and pharmacology. Int. J. Biochem. Cell Biol. 2016, 77, 184–196. [Google Scholar] [CrossRef] [PubMed]
- Behrens, M.; Meyerhof, W. Vertebrate Bitter Taste Receptors: Keys for Survival in Changing Environments. J. Agric. Food Chem. 2018, 66, 2204–2213. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Cao, J.; Iguchi, N.; Riethmacher, D.; Huang, L. Functional characterization of bitter-taste receptors expressed in mammalian testis. Mol. Hum. Reprod. 2013, 19, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Dym, M. Spermatogonial stem cells of the testis. Proc. Natl. Acad. Sci. USA 1994, 91, 11287. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; De Iuliis, G.N.; Dun, M.D.; Nixon, B. Characteristics of the epididymal luminal environment responsible for sperm maturation and storage. Front. Endocrinol. 2018, 9, 59. [Google Scholar] [CrossRef] [PubMed]
- Dacheux, J.L.; Dacheux, F. New insights into epididymal function in relation to sperm maturation. Reproduction 2014, 147, R27–R42. [Google Scholar] [CrossRef]
- Gervasi, M.G.; Visconti, P.E. Molecular changes and signaling events occurring in spermatozoa during epididymal maturation. Andrology 2017, 5, 204–218. [Google Scholar] [CrossRef] [PubMed]
- Cornwall, G.A. New insights into epididymal biology and function. Hum. Reprod. Update 2009, 15, 213–227. [Google Scholar] [CrossRef]
- Verma, R.J. Sperm quiescence in cauda epididymis: A mini-review. Asian J. Androl. 2001, 3, 181–183. [Google Scholar]
- Liu, Y.; Wang, D.-K.; Chen, L.-M. The Physiology of Bicarbonate Transporters in Mammalian Reproduction. Biol. Reprod. 2012, 86, 1–13. [Google Scholar] [CrossRef]
- Weissgerber, P.; Kriebs, U.; Tsvilovskyy, V.; Olausson, J.; Kretz, O.; Stoerger, C.; Vennekens, R.; Wissenbach, U.; Middendorff, R.; Flockerzi, V.; et al. Male fertility depends on Ca2+ absorption by TRPV6 in epididymal epithelia. Sci. Signal. 2011, 4, ra27. [Google Scholar] [CrossRef] [PubMed]
- Yanagimachi, R. Mammalian fertilization. In The Physiology of Reproduction; Knobil, E., Neill, J.D., Eds.; Raven Press: New York, NY, USA, 1994; pp. 189–317. [Google Scholar]
- Yanagimachi, R. Fertility of mammalian spermatozoa: Its development and relativity. Zygote 1994, 2, 371–372. [Google Scholar] [CrossRef] [PubMed]
- Sengupta, D.; Chattopadhyay, A. Molecular dynamics simulations of GPCR-cholesterol interaction: An emerging paradigm. Biochim. Biophys. Acta Biomembr. 2015, 1848, 1775–1782. [Google Scholar] [CrossRef] [PubMed]
- Sengupta, D.; Chattopadhyay, A. Identification of Cholesterol Binding Sites in the Serotonin1A Receptor. J. Phys. Chem. B 2012, 116, 12991–12996. [Google Scholar] [CrossRef] [PubMed]
- Jafurulla, M.; Tiwari, S.; Chattopadhyay, A. Identification of cholesterol recognition amino acid consensus (CRAC) motif in G-protein coupled receptors. Biochem. Biophys. Res. Commun. 2011, 404, 569–573. [Google Scholar] [CrossRef] [PubMed]
- Pydi, S.P.; Jafurulla, M.; Wai, L.; Bhullar, R.P.; Chelikani, P.; Chattopadhyay, A. Cholesterol modulates bitter taste receptor function. Biochim. Biophys. Acta Biomembr. 2016, 1858, 2081–2087. [Google Scholar] [CrossRef] [PubMed]
- Eisenbach, M.; Giojalas, L.C. Sperm guidance in mammals—An unpaved road to the egg. Nat. Rev. Mol. Cell Biol. 2006, 7, 276. [Google Scholar] [CrossRef] [PubMed]
- Bahat, A.; Tur-Kaspa, I.; Gakamsky, A.; Giojalas, L.C.; Breitbart, H.; Eisenbach, M. Thermotaxis of mammalian sperm cells: A potential navigation mechanism in the female genital tract. Nat. Med. 2003, 9, 149. [Google Scholar] [CrossRef]
- Miki, K.; Clapham, D.E. Rheotaxis guides mammalian sperm. Curr. Biol. 2013, 23, 443–452. [Google Scholar] [CrossRef]
- Strünker, T.; Alvarez, L.; Kaupp, U.B. At the physical limit—Chemosensation in sperm. Curr. Opin. Neurobiol. 2015, 34, 110–116. [Google Scholar] [CrossRef]
- Sugiyama, H.; Chandler, D.E. Sperm guidance to the egg finds calcium at the helm. Protoplasma 2014, 251, 461–475. [Google Scholar] [CrossRef] [PubMed]
- Leahy, T.; Gadella, B.M. New insights into the regulation of cholesterol efflux from the sperm membrane. Asian J. Androl. 2015, 17, 561–567. [Google Scholar] [PubMed]
- Perez-Cerezales, S.; Boryshpolets, S.; Eisenbach, M. Behavioral mechanisms of mammalian sperm guidance. Asian J. Androl. 2015, 17, 628–632. [Google Scholar] [PubMed]
- Wachten, D.; Jikeli, J.F.; Kaupp, U.B. Sperm Sensory Signaling. Cold Spring Harb. Perspect. Biol. 2017, 9, a028225. [Google Scholar] [CrossRef] [PubMed]
- De Jonge, C. Biological basis for human capacitation-revisited. Hum. Reprod. Update 2017, 23, 289–299. [Google Scholar] [CrossRef]
- Jin, M.; Fujiwara, E.; Kakiuchi, Y.; Okabe, M.; Satouh, Y.; Baba, S.A.; Chiba, K.; Hirohashi, N. Most fertilizing mouse spermatozoa begin their acrosome reaction before contact with the zona pellucida during in vitro fertilization. Proc. Natl. Acad. Sci. USA 2011, 108, 4892–4896. [Google Scholar] [CrossRef]
- Shaik, F.A.; Medapati, M.R.; Chelikani, P. Cholesterol modulates the signaling of chemosensory bitter taste receptor T2R14 in human airway cells. Am. J. Physiol. Lung Cell. Mol. Physiol. 2019, 316, L45–L57. [Google Scholar] [CrossRef]
- Cohen-Dayag, A.; Tur-Kaspa, I.; Dor, J.; Mashiach, S.; Eisenbach, M. Sperm capacitation in humans is transient and correlates with chemotactic responsiveness to follicular factors. Proc. Natl. Acad. Sci. USA 1995, 92, 11039–11043. [Google Scholar] [CrossRef]
- Ren, D.; Navarro, B.; Perez, G.; Jackson, A.C.; Hsu, S.; Shi, Q.; Tilly, J.L.; Clapham, D.E. A sperm ion channel required for sperm motility and male fertility. Nature 2001, 413, 603–609. [Google Scholar] [CrossRef]
- Lishko, P.V.; Mannowetz, N. CatSper: A Unique Calcium Channel of the Sperm Flagellum. Curr. Opin. Physiol. 2018, 2, 109–113. [Google Scholar] [CrossRef]
- Brenker, C.; Goodwin, N.; Weyand, I.; Kashikar, N.D.; Naruse, M.; Krähling, M.; Müller, A.; Kaupp, U.B.; Strünker, T. The CatSper channel: A polymodal chemosensor in human sperm. EMBO J. 2012, 31, 1654–1665. [Google Scholar] [CrossRef] [PubMed]
- Suarez, S.S. Mammalian sperm interactions with the female reproductive tract. Cell Tissue Res. 2016, 363, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Ralt, D.; Goldenberg, M.; Fetterolf, P.; Thompson, D.; Dor, J.; Mashiach, S.; Garbers, D.L.; Eisenbach, M. Sperm attraction to a follicular factor(s) correlates with human egg fertilizability. Proc. Natl. Acad. Sci. USA 1991, 88, 2840–2844. [Google Scholar] [CrossRef] [PubMed]
- Teves, M.E.; Barbano, F.; Guidobaldi, H.A.; Sanchez, R.; Miska, W.; Giojalas, L.C. Progesterone at the picomolar range is a chemoattractant for mammalian spermatozoa. Fertil. Steril. 2006, 86, 745–749. [Google Scholar] [CrossRef] [PubMed]
- Oren-Benaroya, R.; Orvieto, R.; Gakamsky, A.; Pinchasov, M.; Eisenbach, M. The sperm chemoattractant secreted from human cumulus cells is progesterone. Hum. Reprod. 2008, 23, 2339–2345. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, M.; Yoshida, K. Sperm chemotaxis and regulation of flagellar movement by Ca2+. Mol. Hum. Reprod. 2011, 17, 457–465. [Google Scholar] [CrossRef] [PubMed]
- Cook, S.P.; Brokaw, C.J.; Muller, C.H.; Babcock, D.F. Sperm chemotaxis: Egg peptides control cytosolic calcium to regulate flagellar responses. Dev. Biol. 1994, 165, 10–19. [Google Scholar] [CrossRef]
- Publicover, S.J.; Giojalas, L.C.; Teves, M.E.; de Oliveira, G.S.M.M.; Garcia, A.A.M.; Barratt, C.L.R.; Harper, C.V. Ca2+ signalling in the control of motility and guidance in mammalian sperm. Front. Biosci. 2008, 13, 5623–5637. [Google Scholar] [CrossRef]
- Wistrom, C.A.; Meizel, S. Evidence suggesting involvement of a unique human sperm steroid receptor/Cl−channel complex in the progesterone-initiated acrosome reaction. Dev. Biol. 1993, 159, 679–690. [Google Scholar]
- Ritta, M.N.; Calamera, J.C.; Bas, D.E. Occurrence of GABA and GABA receptors in human spermatozoa. Mol. Hum. Reprod. 1998, 4, 769–773. [Google Scholar] [CrossRef]
- Martín del Rio, R. Gamma-aminobutyric acid system in rat oviduct. J. Biol. Chem. 1981, 256, 9816–9819. [Google Scholar] [PubMed]
- Freund, J.R.; Lee, R.J. Taste receptors in the upper airway. World J. Otorhinolaryngol. Head Neck Surg. 2018. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, C.; Triller, A.; Spehr, M.; Dittrich, R.; Hatt, H.; Buettner, A. Sperm-activating odorous substances in human follicular fluid and vaginal secretion: Identification by gas chromatography-olfactometry and Ca2+ imaging. ChemPlusChem 2013, 78, 695–702. [Google Scholar] [CrossRef]
- Vanderhaeghen, P.; Schurmans, S.; Vassart, G.; Parmentier, M. Olfactory receptors are displayed on dog mature sperm cells. J. Cell Biol. 1993, 123, 1441–1452. [Google Scholar] [CrossRef] [PubMed]
- Milardi, D.; Colussi, C.; Grande, G.; Vincenzoni, F.; Pierconti, F.; Mancini, F.; Baroni, S.; Castagnola, M.; Marana, R.; Pontecorvi, A. Olfactory Receptors in Semen and in the Male Tract: From Proteome to Proteins. Front. Endocrinol. 2017, 8, 379. [Google Scholar] [CrossRef] [PubMed]
- Spehr, M.; Gisselmann, G.; Poplawski, A.; Riffell, J.A.; Wetzel, C.H.; Zimmer, R.K.; Hatt, H. Identification of a testicular odorant receptor mediating human sperm chemotaxis. Science 2003, 299, 2054–2058. [Google Scholar] [CrossRef] [PubMed]
- Storto, M.; Sallese, M.; Salvatore, L.; Poulet, R.; Condorelli, D.F.; Dell’Albani, P.; Marcello, M.F.; Romeo, R.; Piomboni, P.; Barone, N.; et al. Expression of metabotropic glutamate receptors in the rat and human testis. J. Endocrinol. 2001, 170, 71–78. [Google Scholar] [CrossRef]
- Lefkowitz, R.J. Arrestins Come of Age. Prog. Mol. Biol. Transl. Sci. 2013, 118, 3–18. [Google Scholar]
- Józwik, M.; Józwik, M.; Teng, C.; Battaglia, F.C. Amino acid, ammonia and urea concentrations in human pre-ovulatory ovarian follicular fluid. Hum. Reprod. 2006, 21, 2776–2782. [Google Scholar] [CrossRef]
- Buffone, M.G.; Wertheimer, E.V.; Visconti, P.E.; Krapf, D. Central role of soluble adenylyl cyclase and cAMP in sperm physiology. Biochim. Biophys. Acta 2014, 1842, 2610–2620. [Google Scholar] [CrossRef]
- Branham, M.T.; Mayorga, L.S.; Tomes, C.N. Calcium-induced acrosomal exocytosis requires cAMP acting through a protein kinase A-independent, Epac-mediated pathway. J. Biol. Chem. 2006, 281, 8656–8666. [Google Scholar] [CrossRef] [PubMed]
- Visconti, P.E. Understanding the molecular basis of sperm capacitation through kinase design. Proc. Natl. Acad. Sci. USA 2009, 106, 667–668. [Google Scholar] [CrossRef]
- Beltrán, C.; Vacquier, V.D.; Moy, G.; Chen, Y.; Buck, J.; Levin, L.R.; Darszon, A. Particulate and soluble adenylyl cyclases participate in the sperm acrosome reaction. Biochem. Biophys. Res. Commun. 2007, 358, 1128–1135. [Google Scholar] [CrossRef] [PubMed]
- Glassner, M.; Jones, J.; Kligman, I.; Woolkalis, M.J.; Gerton, G.L.; Kopf, G.S. Immunocytochemical and biochemical characterization of guanine nucleotide-binding regulatory proteins in mammalian spermatozoa. Dev. Biol. 1991, 146, 438–450. [Google Scholar] [CrossRef]
- Spinaci, M.; Bucci, D.; Mazzoni, M.; Giaretta, E.; Bernardini, C.; Vallorani, C.; Tamanini, C.; Clavenzani, P.; Galeati, G. Expression of α-gustducin and α-transducin, G proteins coupled with taste receptors, in boar sperm. Theriogenology 2014, 82, 144–151. [Google Scholar] [CrossRef]
- Spinaci, M.; Bucci, D.; Gadani, B.; Porcu, E.; Tamanini, C.; Galeati, G. Pig sperm preincubation and gamete coincubation with glutamate enhance sperm-oocyte binding and in vitro fertilization. Theriogenology 2017, 95, 149–153. [Google Scholar] [CrossRef]
- Mosinger, B.; Redding, K.M.; Parker, M.R.; Yevshayeva, V.; Yee, K.K.; Dyomina, K.; Li, Y.; Margolskee, R.F. Genetic loss or pharmacological blockade of testes-expressed taste genes causes male sterility. Proc. Natl. Acad. Sci. USA 2013, 110, 12319–12324. [Google Scholar] [CrossRef]
- Maillet, E.L.; Margolskee, R.F.; Mosinger, B. Phenoxy herbicides and fibrates potently inhibit the human chemosensory receptor subunit T1R3. J. Med. Chem. 2009, 52, 6931–6935. [Google Scholar] [CrossRef]
- Kochem, M.; Breslin, P.A.S. Lipid-lowering pharmaceutical clofibrate inhibits human sweet taste. Chem. Senses 2016. [Google Scholar] [CrossRef]
- Kochem, M.; Breslin, P.A.S. Clofibrate inhibits the umami-savory taste of glutamate. PLoS ONE 2017, 12, e0172534. [Google Scholar] [CrossRef]
- Kroemer, G.; Galluzzi, L.; Vandenabeele, P.; Abrams, J.; Alnemri, E.S.; Baehrecke, E.H.; Blagosklonny, M.V.; El-Deiry, W.S.; Golstein, P.; Green, D.R.; et al. Classification of cell death: Recommendations of the Nomenclature Committee on Cell Death 2009. Cell Death Differ. 2009, 16, 3. [Google Scholar] [CrossRef] [PubMed]
- Creasy, D.; Bube, A.; de Rijk, E.; Kandori, H.; Kuwahara, M.; Masson, R.; Nolte, T.; Reams, R.; Regan, K.; Rehm, S.; et al. Proliferative and nonproliferative lesions of the rat and mouse male reproductive system. Toxicol. Pathol. 2012, 40, 40S–121S. [Google Scholar] [CrossRef] [PubMed]
- Balbach, M.; Beckert, V.; Hansen, J.N.; Wachten, D. Shedding light on the role of cAMP in mammalian sperm physiology. Mol. Cell. Endocrinol. 2018, 468, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Beigi Harchegani, A.; Irandoost, A.; Mirnamniha, M.; Rahmani, H.; Tahmasbpour, E.; Shahriary, A. Possible Mechanisms for The Effects of Calcium Deficiency on Male Infertility. Int. J. Fertil. Steril. 2019, 12, 267–272. [Google Scholar] [PubMed]
- Stival, C.; Puga Molina, L.D.C.; Paudel, B.; Buffone, M.G.; Visconti, P.E.; Krapf, D. Sperm Capacitation and Acrosome Reaction in Mammalian Sperm. Adv. Anat. Embryol. Cell Biol. 2016, 220, 93–106. [Google Scholar] [PubMed]
- Zitranski, N.; Borth, H.; Ackermann, F.; Meyer, D.; Vieweg, L.; Breit, A.; Gudermann, T.; Boekhoff, I. The “acrosomal synapse”: Subcellular organization by lipid rafts and scaffolding proteins exhibits high similarities in neurons and mammalian spermatozoa. Commun. Integr. Biol. 2010, 3, 513–521. [Google Scholar] [CrossRef]
- DuBois, G.E. Molecular mechanism of sweetness sensation. Physiol. Behav. 2016, 164, 453–463. [Google Scholar] [CrossRef]
- Pydi, S.P.; Bhullar, R.P.; Chelikani, P. Constitutive activity of bitter taste receptors (T2Rs). Adv. Pharmacol. 2014, 70, 303–326. [Google Scholar]
- Du, L.; Xu, Y.; Musson, D.G. Simultaneous determination of clofibrate and its active metabolite clofibric acid in human plasma by reversed-phase high-performance liquid chromatography with ultraviolet absorbance detection. J. Chromatogr. B. Anal. Technol. Biomed. Life Sci. 2003, 794, 343–351. [Google Scholar] [CrossRef]
- Sato, J.; Makita, N.; Iiri, T. Inverse agonism: The classic concept of GPCRs revisited [Review]. Endocr. J. 2016, 63, 507–514. [Google Scholar] [CrossRef]
- Sassone-Corsi, P. CREM: A master-switch regulating the balance between differentiation and apoptosis in male germ cells. Mol. Reprod. Dev. 2000, 56, 228–229. [Google Scholar] [CrossRef]
- Sassone-Corsi, P. CREM: A master-switch governing male germ cell differentiation and apoptosis. Semin. Cell Dev. Biol. 1998, 9, 475–482. [Google Scholar] [CrossRef] [PubMed]
- Sassone-Corsi, P. Regulating the balance between differentiation and apoptosis: Role of CREM in the male germ cells. J. Mol. Med. 1998, 76, 811–817. [Google Scholar] [CrossRef] [PubMed]
- Duffy, V.B.; Davidson, A.C.; Kidd, J.R.; Kidd, K.K.; Speed, W.C.; Pakstis, A.J.; Reed, D.R.; Snyder, D.J.; Bartoshuk, L.M. Bitter receptor gene (TAS2R38), 6-n-propylthiouracil (PROP) bitterness and alcohol intake. Alcohol. Clin. Exp. Res. 2004, 28, 1629–1637. [Google Scholar] [CrossRef] [PubMed]
- Dinehart, M.E.; Hayes, J.E.; Bartoshuk, L.M.; Lanier, S.L.; Duffy, V.B. Bitter taste markers explain variability in vegetable sweetness, bitterness, and intake. Physiol. Behav. 2006, 87, 304–313. [Google Scholar] [CrossRef] [PubMed]
- Sandell, M.; Hoppu, U.; Mikkilä, V.; Mononen, N.; Kähönen, M.; Männistö, S.; Rönnemaa, T.; Viikari, J.; Lehtimäki, T.; Raitakari, O.T. Genetic variation in the hTAS2R38 taste receptor and food consumption among Finnish adults. Genes Nutr. 2014, 9, 433. [Google Scholar] [CrossRef] [PubMed]
- Hayes, J.E.; Feeney, E.L.; Nolden, A.A.; McGeary, J.E. Quinine Bitterness and Grapefruit Liking Associate with Allelic Variants in TAS2R31. Chem. Sens. 2015, 40, 437–443. [Google Scholar] [CrossRef]
- Wang, J.C.; Hinrichs, A.L.; Bertelsen, S.; Stock, H.; Budde, J.P.; Dick, D.M.; Bucholz, K.K.; Rice, J.; Saccone, N.; Edenberg, H.J.; et al. Functional variants in TAS2R38 and TAS2R16 influence alcohol consumption in high-risk families of African-American origin. Alcohol. Clin. Exp. Res. 2007, 31, 209–215. [Google Scholar] [CrossRef]
- Cannon, D.S.; Baker, T.B.; Piper, M.E.; Scholand, M.B.; Lawrence, D.L.; Drayna, D.T.; McMahon, W.M.; Villegas, G.M.M.; Caton, T.C.; Coon, H.; et al. Associations between phenylthiocarbamide gene polymorphisms and cigarette smoking. Nicotine Tob. Res. 2005, 7, 853–858. [Google Scholar] [CrossRef]
- Mangold, J.E.; Payne, T.J.; Ma, J.Z.; Chen, G.; Li, M.D.; Mangold, J.E. Bitter taste receptor gene polymorphisms are an important factor in the development of nicotine dependence in African Americans. J. Med. Genet. 2008, 45, 578–582. [Google Scholar] [CrossRef]
- Padiglia, A.; Zonza, A.; Atzori, E.; Chillotti, C.; Calò, C.; Tepper, B.J.; Barbarossa, I.T. Sensitivity to 6-n-propylthiouracil is associated with gustin (carbonic anhydrase VI) gene polymorphism, salivary zinc, and body mass index in humans. Am. J. Clin. Nutr. 2010, 92, 539–545. [Google Scholar] [CrossRef] [PubMed]
- Aston, K.I.; Carrell, D.T. Genome-wide study of single-nucleotide polymorphisms associated with azoospermia and severe oligozoospermia. J. Androl. 2009, 30, 711–725. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Wen, W.; Yu, J.G.; Qu, S.Q.; Wang, S.S.; Liu, J.; Li, B.S.; Luo, Y. Genetic association of UBE2B variants with susceptibility to male infertility in a Northeast Chinese population. Genet. Mol. Res. 2012, 11, 4226–4234. [Google Scholar] [CrossRef] [PubMed]
- Kosova, G.; Scott, N.M.; Niederberger, C.; Prins, G.S.; Ober, C. Genome-wide association study identifies candidate genes for male fertility traits in humans. Am. J. Hum. Genet. 2012, 90, 950–961. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Wu, W.; Xu, B.; Niu, X.; Cui, H.; Zhang, Y.; Wang, Z.; Wang, X. Variants in the SRD5A2 gene are associated with quality of semen. Mol. Med. Rep. 2012, 6, 639–644. [Google Scholar] [CrossRef]
- Aston, K.I.; Krausz, C.; Laface, I.; Ruiz-Castane, E.; Carrell, D.T.; Ruiz-Castané, E.; Carrell, D.T. Evaluation of 172 candidate polymorphisms for association with oligozoospermia or azoospermia in a large cohort of men of European descent. Hum. Reprod. 2010, 25, 1383–1397. [Google Scholar] [CrossRef] [PubMed]
- Plaseski, T.; Noveski, P.; Popeska, Z.; Efremov, G.D.; Plaseska-Karanfilska, D. Association Study of Single-Nucleotide Polymorphisms in FASLG, JMJDIA, LOC203413, TEX15, BRDT, OR2W3, INSR, and TAS2R38 Genes with Male Infertility. J. Androl. 2012, 33, 675–683. [Google Scholar] [CrossRef]
- Siasi, E.; Aleyasin, A. Four Single Nucleotide Polymorphisms in INSR, SLC6A14, TAS2R38, and OR2W3 Genes in Association with Idiopathic Infertility in Persian Men. J. Reprod. Med. 2016, 61, 145–152. [Google Scholar]
- Krausz, C.; Escamilla, A.R.; Chianese, C. Genetics of male infertility: From research to clinic. Reproduction 2015, 150, R159–R174. [Google Scholar] [CrossRef]
- Nieschlag, E.; Behre, H.M.; Nieschlag, S. Male Reproductive Health and Dysfunction; Springer: New York, NY, USA, 2010; ISBN 978-3-54-078354-1. [Google Scholar]
- Gentiluomo, M.; Crifasi, L.; Luddi, A.; Locci, D.; Barale, R.; Piomboni, P.; Campa, D. Taste receptor polymorphisms and male infertility. Hum. Reprod. 2017, 32, 2324–2331. [Google Scholar] [CrossRef]
- Hwang, K.; Yatsenko, A.N.; Jorgez, C.J.; Mukherjee, S.; Nalam, R.L.; Matzuk, M.M.; Lamb, D.J. Mendelian genetics of male infertility. Ann. N. Y. Acad. Sci. USA 2010, 1214, E1–E17. [Google Scholar] [CrossRef] [PubMed]
- Yoder, B.K. Role of Primary Cilia in the Pathogenesis of Polycystic Kidney Disease. J. Am. Soc. Nephrol. 2007, 18, 1381–1388. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luddi, A.; Governini, L.; Wilmskötter, D.; Gudermann, T.; Boekhoff, I.; Piomboni, P. Taste Receptors: New Players in Sperm Biology. Int. J. Mol. Sci. 2019, 20, 967. https://doi.org/10.3390/ijms20040967
Luddi A, Governini L, Wilmskötter D, Gudermann T, Boekhoff I, Piomboni P. Taste Receptors: New Players in Sperm Biology. International Journal of Molecular Sciences. 2019; 20(4):967. https://doi.org/10.3390/ijms20040967
Chicago/Turabian StyleLuddi, Alice, Laura Governini, Dorke Wilmskötter, Thomas Gudermann, Ingrid Boekhoff, and Paola Piomboni. 2019. "Taste Receptors: New Players in Sperm Biology" International Journal of Molecular Sciences 20, no. 4: 967. https://doi.org/10.3390/ijms20040967
APA StyleLuddi, A., Governini, L., Wilmskötter, D., Gudermann, T., Boekhoff, I., & Piomboni, P. (2019). Taste Receptors: New Players in Sperm Biology. International Journal of Molecular Sciences, 20(4), 967. https://doi.org/10.3390/ijms20040967





