Molecular Communication for Coordinated Seed and Fruit Development: What Can We Learn from Auxin and Sugars?
Abstract
1. Auxin in Seed Development
2. Auxin Movement within the Different Seed Structures Triggers Different Developmental Outputs
2.1. Auxin Movement within the Integuments and Funiculus
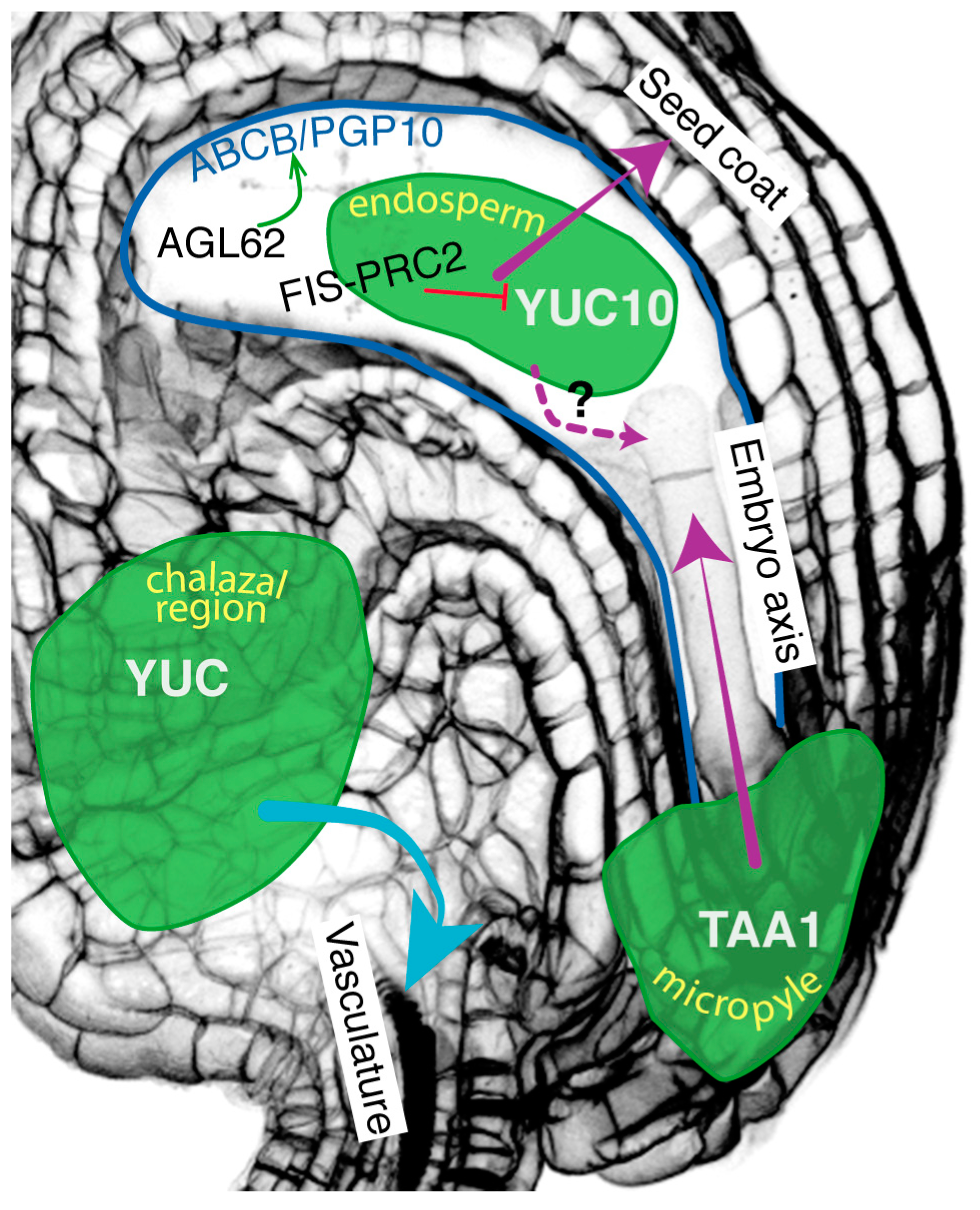
2.2. Auxin Movement from the Endosperm to the Integuments
2.3. Auxin Movement from the Integuments to the Embryo
2.4. Auxin Movement from the Endosperm to the Embryo
3. Symplastic Transport, as an Alternative Route of Communication within the Seed
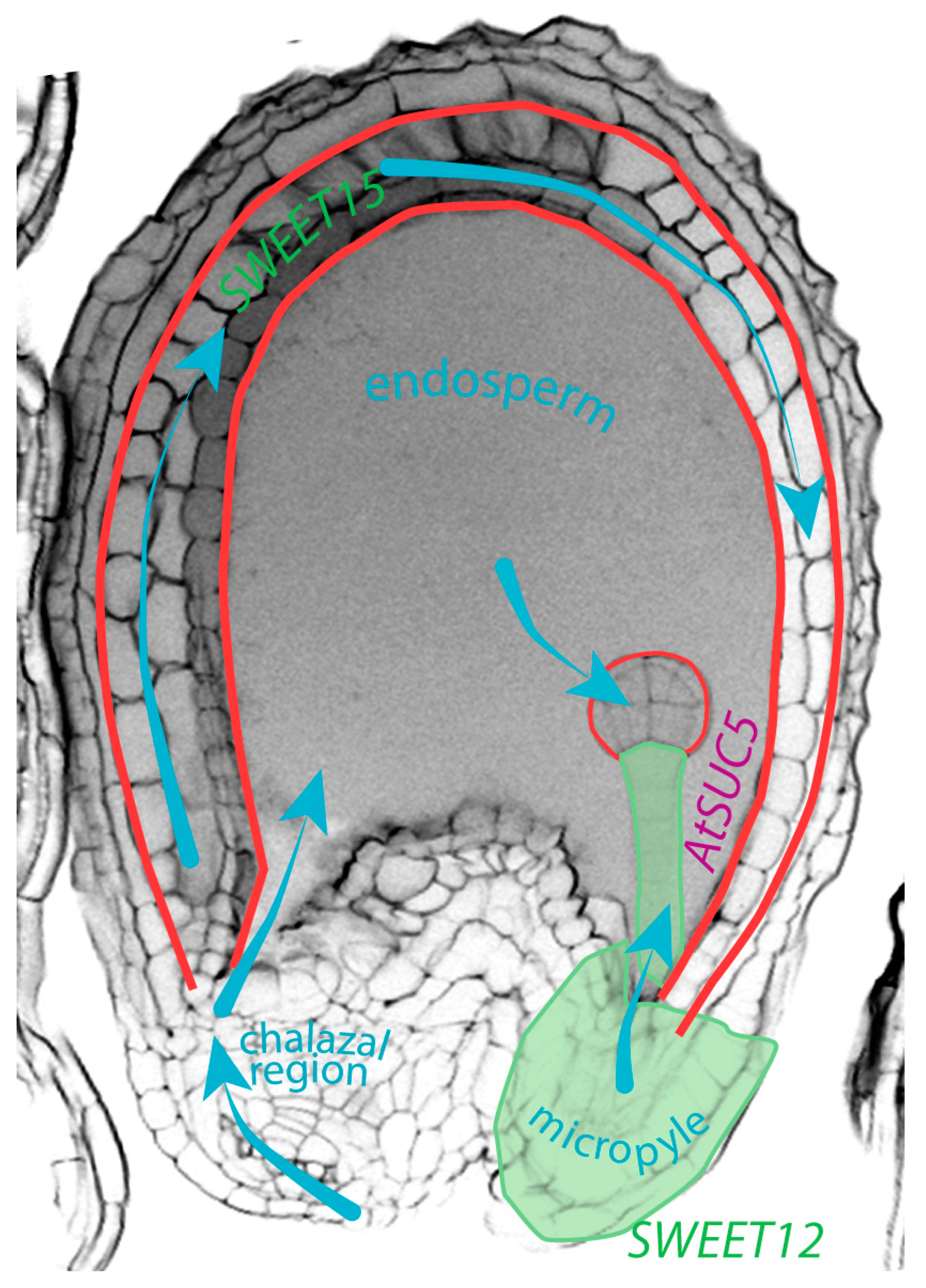
4. Movement of Sucrose within the Seed, Another Example of Molecular Communication
5. Seed to Fruit Communication: Hormonal Crosstalk for Fruit Initiation
6. Conclusions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ABC/PGP | ATP BINDING CASSETTE subfamily B/P-GLYCOPROTEIN |
| AFB | AUXIN RESPONSE F-BOX |
| AGL62 | AGAMOUS-LIKE 62 |
| ALE1 | ABNORMAL LEAF-SHAPE1 |
| ARF | AUXIN RESPONSE FACTOR |
| AUX1 | AUXIN1 |
| Aux/IAA | AUXIN/INDOLE-3-ACETIC ACID |
| AuxRE | auxin response elements |
| AXL1 | AXR1-like1 |
| AXR1 | AUXIN RESPONSE 1 |
| BAN | BANYULS |
| BDL | BODENLOSS–Aux/IAA12 |
| BETL | Basal Endosperm Transfer Layer |
| de18 | defective endosperm18 |
| dk18 | defective kernel18 |
| DII | Domain II of Aux/IAA proteins |
| ESR | Embryo Surrounding Region |
| FG | Female gametophyte |
| FIE | FERTILIZATION INDEPENDENT ENDOSPERM |
| FIS | FERTILIZATION INDEPENDENT SEED |
| GA | gibberellic acid |
| GH3 | GRETCHEN HAGEN3 |
| GID1 | GIBBERELLIN INSENSITIVE DWARF1 |
| GSO1 | GASSHO1 |
| IAA | indole-3-acetic acid |
| IPyA | Indole-3-pyruvic acid |
| KRS | KERBEROS |
| LAX | Like-AUX1 |
| MEA | MEDEA |
| MPK | MAP-kinase |
| MSI1 | MULTICOPY SUPPRESSOR OF IRA1 |
| NPA | N-1-naphthylphthalamic acid |
| PcG | Polycomb group |
| PIN | PIN-FORMED |
| RGA | REPRESSOR OF GA1-3 |
| SUC/SUT | SUCROSE TRANSPORTER |
| TAA1 | TRYPTOPHAN AMINOTRANSFERASE OF ARABIDOPSIS |
| TAR | TAA1-related |
| TIR1 | TRANSPORT INHIBITOR RESISTANT |
| Trp | tryptophan |
| WEI8 | WEAK ETHYLENE INSENSITIVE8 |
| YUC | YUCCA |
| ZOU | ZHOUPI |
References
- Dorcey, E.; Urbez, C.; Blázquez, M.A.; Carbonell, J.; Perez-Amador, M.A. Fertilization-dependent auxin response in ovules triggers fruit development through the modulation of gibberellin metabolism in Arabidopsis. Plant J. 2009, 58, 318–332. [Google Scholar] [CrossRef]
- Cucinotta, M.; Manrique, S.; Cuesta, C.; Benková, E.; Novak, O.; Colombo, L.L. CUP-SHAPED COTYLEDON1 (CUC1) and CUC2 regulate cytokinin homeostasis to determine ovule number in Arabidopsis. J. Exp. Bot. 2018, 69, 5169–5176. [Google Scholar] [CrossRef] [PubMed]
- Cucinotta, M.; Manrique, S.; Guazzotti, A.; Quadrelli, N.E.; Mendes, M.A.; Benková, E.; Colombo, L.L. Cytokinin response factors integrate auxin and cytokinin pathways for female reproductive organ development. Development 2016, 143, 4419–4424. [Google Scholar] [CrossRef] [PubMed]
- Gómez, M.D.; Barro-Trastoy, D.; Escoms, E.; Saura-Sánchez, M.; Sánchez, I.; Briones-Moreno, A.; Vera-Sirera, F.; Carrera, E.; Ripoll, J.J.; Yanofsky, M.F.; et al. Gibberellins negatively modulate ovule number in plants. Development 2018, 145, dev163865. [Google Scholar] [CrossRef] [PubMed]
- Galván-Ampudia, C.S.; Cerutti, G.; Legrand, J.; Azais, R.; Brunoud, G.; Moussu, S.; Wenzl, C.; Lohmann, J.U.; Godin, C.C.; Vernoux, T. From spatio-temporal morphogenetic gradients to rhythmic patterning at the shoot apex. bioRxiv 2019. [Google Scholar] [CrossRef]
- Wybouw, B.; De Rybel, B. Cytokinin—A Developing Story. Trends Plant Sci. 2019, 24, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Robert, H.S.; Khaitova, L.C.; Mroue, S.; Benková, E. The importance of localized auxin production for morphogenesis of reproductive organs and embryos in Arabidopsis. J. Exp. Bot. 2015, 66, 5029–5042. [Google Scholar] [CrossRef]
- Joldersma, D.; Liu, Z. The making of virgin fruit: The molecular and genetic basis of parthenocarpy. J. Exp. Bot. 2018, 69, 955–962. [Google Scholar] [CrossRef] [PubMed]
- Stepanova, A.N.; Yun, J.J.; Robles, L.M.; Novak, O.; He, W.; Guo, H.; Ljung, K.; Alonso, J.M. The Arabidopsis YUCCA1 flavin monooxygenase functions in the indole-3-pyruvic acid branch of auxin biosynthesis. Plant Cell 2011, 23, 3961–3973. [Google Scholar] [CrossRef]
- Mashiguchi, K.; Tanaka, K.; Sakai, T.; Sugawara, S.; Kawaide, H.; Natsume, M.; Hanada, A.; Yaeno, T.; Shirasu, K.; Yao, H.; et al. The main auxin biosynthesis pathway in Arabidopsis. Proc. Natl. Acad. Sci. USA 2011, 108, 18512–18517. [Google Scholar] [CrossRef]
- Won, C.; Shen, X.; Mashiguchi, K.; Zheng, Z.; Dai, X.; Cheng, Y.; Kasahara, H.; Kamiya, Y.; Chory, J.; Zhao, Y. Conversion of tryptophan to indole-3-acetic acid by TRYPTOPHAN AMINOTRANSFERASES OF ARABIDOPSIS and YUCCAs in Arabidopsis. Proc. Natl. Acad. Sci. USA 2011, 108, 18518–18523. [Google Scholar] [CrossRef] [PubMed]
- Stepanova, A.N.; Robertson-Hoyt, J.; Yun, J.J.; Benavente, L.M.; Xie, D.-Y.; Dolezal, K.; Schlereth, A.; Juergens, G.; Alonso, J. M TAA1-mediated auxin biosynthesis is essential for hormone crosstalk and plant development. Cell 2008, 133, 177–191. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y. Auxin biosynthesis. Arab. Book 2014, 12, e0173. [Google Scholar] [CrossRef] [PubMed]
- Figueiredo, D.D.; Batista, R.A.; Roszak, P.J.; Köhler, C.C. Auxin production couples endosperm development to fertilization. Nat. Plants 2015, 1, 15184. [Google Scholar] [CrossRef] [PubMed]
- Figueiredo, D.D.; Batista, R.A.; Roszak, P.J.; Hennig, L.; Köhler, C.C. Auxin production in the endosperm drives seed coat development in Arabidopsis. eLife 2016, 5, e20542. [Google Scholar] [CrossRef] [PubMed]
- Robert, H.S.; Grones, P.; Stepanova, A.N.; Robles, L.M.; Lokerse, A.S.; Alonso, J.M.; Weijers, D.; Friml, J. Local auxin sources orient the apical-basal axis in Arabidopsis embryos. Curr. Biol. 2013, 23, 2506–2512. [Google Scholar] [CrossRef] [PubMed]
- Robert, H.S.; Park, C.; Gutièrrez, C.L.; Wójcikowska, B.; Pencík, A.; Novak, O.; Chen, J.; Grunewald, W.; Dresselhaus, T.; Friml, J.; et al. Maternal auxin supply contributes to early embryo patterning in Arabidopsis. Nat. Plants 2018, 4, 548–553. [Google Scholar] [CrossRef] [PubMed]
- Brumos, J.; Robles, L.M.; Yun, J.J.; Vu, T.C.; Jackson, S.; Alonso, J.M.; Stepanova, A.N. Local Auxin Biosynthesis Is a Key Regulator of Plant Development. Dev. Cell 2018, 47, 306–318.e5. [Google Scholar] [CrossRef]
- Cheng, Y.; Dai, X.; Zhao, Y. Auxin biosynthesis by the YUCCA flavin monooxygenases controls the formation of floral organs and vascular tissues in Arabidopsis. Genes Dev. 2006, 20, 1790–1799. [Google Scholar] [CrossRef]
- Cheng, Y.; Dai, X.; Zhao, Y. Auxin synthesized by the YUCCA flavin monooxygenases is essential for embryogenesis and leaf formation in Arabidopsis. Plant. Cell 2007, 19, 2430–2439. [Google Scholar] [CrossRef]
- Chen, Q.; Dai, X.; DePaoli, H.C.; Cheng, Y.; Takebayashi, Y.; Kasahara, H.; Kamiya, Y.; Zhao, Y. Auxin Overproduction in Shoots Cannot Rescue Auxin Deficiencies in Arabidopsis Roots. Plant. Cell Physiol. 2014, 55, 1072–1079. [Google Scholar] [CrossRef] [PubMed]
- Benková, E.; Michniewicz, M.; Sauer, M.; Teichmann, T.T.; Seifertová, D.; Juergens, G.; Friml, J. Local, efflux-dependent auxin gradients as a common module for plant organ formation. Cell 2003, 115, 591–602. [Google Scholar] [CrossRef]
- Adamowski, M.; Friml, J. PIN-Dependent Auxin Transport: Action, Regulation, and Evolution. Plant Cell 2015, 27, 20–32. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Lee, Y.; Martinoia, E.; Geisler, M. Plant hormone transporters: What we know and what we would like to know. BMC Biol. 2017, 15, 93. [Google Scholar] [CrossRef]
- Péret, B.; Swarup, K.; Ferguson, A.; Seth, M.; Yang, Y.; Dhondt, S.; James, N.; Casimiro, I.; Perry, P.; Syed, A.; et al. AUX/LAX genes encode a family of auxin influx transporters that perform distinct functions during Arabidopsis development. Plant Cell 2012, 24, 2874–2885. [Google Scholar] [CrossRef]
- Weijers, D.; Wagner, D. Transcriptional Responses to the Auxin Hormone. Annu. Rev. Plant Biol. 2016, 67, 539–574. [Google Scholar] [CrossRef] [PubMed]
- Liao, C.-Y.; Smet, W.; Brunoud, G.; Yoshida, S.; Vernoux, T.; Weijers, D. Reporters for sensitive and quantitative measurement of auxin response. Nat. Methods 2015, 12, 207–210. [Google Scholar] [CrossRef] [PubMed]
- Brunoud, G.; Wells, D.M.; Oliva, M.; Larrieu, A.; Mirabet, V.; Burrow, A.H.; Beeckman, T.; Kepinski, S.; Traas, J.; Bennett, M.J.; et al. A novel sensor to map auxin response and distribution at high spatio-temporal resolution. Nature 2012, 482, 103–106. [Google Scholar] [CrossRef] [PubMed]
- Ulmasov, T.; Murfett, J.; Hagen, G.; Guilfoyle, T.J. Aux/IAA proteins repress expression of reporter genes containing natural and highly active synthetic auxin response elements. Plant Cell 1997, 9, 1963–1971. [Google Scholar] [CrossRef]
- Christensen, C.A.; King, E.J.; Jordan, J.; Drews, G.N. Megagametogenesis in Arabidopsis wild type and the Gf mutant. Sex. Plant Reprod. 1997, 10, 49–64. [Google Scholar] [CrossRef]
- Drews, G.N.; Yadegari, R. Development and function of the angiosperm female gametophyte. Annu. Rev. Genet. 2002, 36, 99–124. [Google Scholar] [CrossRef] [PubMed]
- Bencivenga, S.; Colombo, L.L.; Masiero, S. Cross talk between the sporophyte and the megagametophyte during ovule development. Sex. Plant. Reprod. 2011, 24, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Lau, S.S.; Slane, D.; Herud, O.; Kong, J.; Juergens, G. Early embryogenesis in flowering plants: Setting up the basic body pattern. Annu. Rev. Plant. Biol. 2012, 63, 483–506. [Google Scholar] [CrossRef] [PubMed]
- Larsson, E.; Franks, R.G.; Sundberg, E. Auxin and the Arabidopsis thaliana gynoecium. J. Exp. Bot. 2013, 64, 2619–2627. [Google Scholar] [CrossRef] [PubMed]
- Moubayidin, L.L.; Østergaard, L. Gynoecium formation: An intimate and complicated relationship. Curr. Opin. Gen. Dev. 2017, 45, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Figueiredo, D.D.; Köhler, C.C. Auxin: A molecular trigger of seed development. Genes Dev. 2018, 32, 479–490. [Google Scholar] [CrossRef]
- Vivian-Smith, A.; Koltunow, A.M. Genetic analysis of growth-regulator-induced parthenocarpy in Arabidopsis. Plant Physiol. 1999, 121, 437–451. [Google Scholar] [CrossRef]
- Larsson, E.; Vivian-Smith, A.; Offringa, R.; Sundberg, E. Auxin homeostasis in Arabidopsis ovules Is anther-dependent at maturation and changes dynamically upon fertilization. Front. Plant Sci. 2017, 8, 1735. [Google Scholar] [CrossRef]
- Lituiev, D.S.; Krohn, N.G.; Müller, B.; Jackson, D.P.; Hellriegel, B.; Dresselhaus, T.; Grossniklaus, U. Theoretical and experimental evidence indicates that there is no detectable auxin gradient in the angiosperm female gametophyte. Development 2013, 140, 4544–4553. [Google Scholar] [CrossRef]
- Panoli, A.; Martin, M.V.; Alandete-Saez, M.; Simon, M.K.; Neff, C.; Swarup, R.; Bellido, A.; Yuan, L.; Pagnussat, G.C.; Sundaresan, V. Auxin Import and Local Auxin Biosynthesis Are Required for Mitotic Divisions, Cell Expansion and Cell Specification during Female Gametophyte Development in Arabidopsis thaliana. PLoS ONE 2015, 10, e0126164. [Google Scholar] [CrossRef]
- Mozgova, I.; Köhler, C.C.; Hennig, L. Keeping the gate closed: Functions of the polycomb repressive complex PRC2 in development. Plant J. 2015, 83, 121–132. [Google Scholar] [CrossRef] [PubMed]
- Mravec, J.; Kubes, M.; Bielach, A.; Gaykova, V.; Petrásek, J.; Skůpa, P.; Chand, S.; Benková, E.; Zazímalová, E.; Friml, J. Interaction of PIN and PGP transport mechanisms in auxin distribution-dependent development. Development 2008, 135, 3345–3354. [Google Scholar] [CrossRef] [PubMed]
- Forestan, C.; Meda, S.; Varotto, S. ZmPIN1-Mediated Auxin Transport Is Related to Cellular Differentiation during Maize Embryogenesis and Endosperm Development. Plant Physiol. 2010, 152, 1373–1390. [Google Scholar] [CrossRef] [PubMed]
- Bernardi, J.; Lanubile, A.; Li, Q.-B.; Kumar, D.; Kladnik, A.; Cook, S.D.; Ross, J.J.; Marocco, A.; Chourey, P.S. Impaired Auxin Biosynthesis in the defective endosperm18 Mutant Is Due to Mutational Loss of Expression in the ZmYuc1 Gene Encoding Endosperm-Specific YUCCA1 Protein in Maize. Plant Physiol. 2012, 160, 1318–1328. [Google Scholar] [CrossRef] [PubMed]
- Locascio, A.; Roig-Villanova, I.; Bernardi, J.; Varotto, S. Current perspectives on the hormonal control of seed development in Arabidopsis and maize: A focus on auxin. Front. Plant. Sci. 2014, 5, 412. [Google Scholar] [CrossRef] [PubMed]
- Bernardi, J.; Li, Q.-B.; Gao, Y.; Zhao, Y.; Battaglia, R.; Marocco, A.; Chourey, P.S. The Auxin-Deficient Defective Kernel18 (dek18) Mutation Alters the Expression of Seed-Specific Biosynthetic Genes in Maize. J. Plant Growth Regul. 2016, 35, 770–777. [Google Scholar] [CrossRef]
- Chen, J.; Lausser, A.; Dresselhaus, T. Hormonal responses during early embryogenesis in maize. Biochem. Soc. Trans. 2014, 42, 325–331. [Google Scholar] [CrossRef] [PubMed]
- Robert, H.S.; Friml, J. Auxin and other signals on the move in plants. Nat. Chem. Biol. 2009, 5, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Stadler, R.; Lauterbach, C.; Sauer, N. Cell-to-cell movement of green fluorescent protein reveals post-phloem transport in the outer integument and identifies symplastic domains in Arabidopsis seeds and embryos. Plant Physiol. 2005, 139, 701–712. [Google Scholar] [CrossRef]
- Kawashima, T.; Goldberg, R.B. The suspensor: Not just suspending the embryo. Trends Plant Sci. 2010, 15, 23–30. [Google Scholar] [CrossRef]
- Yeung, E.C. Embryogeny of Phaseolus: The Role of the Suspensor. Z. Für Pflanzenphysiol. 1980, 96, 17–28. [Google Scholar] [CrossRef]
- Nagl, W. Translocation of Putrescine in the Ovule, Suspensor and Embryo of Phaseolus coccineus. J. Plant. Physiol. 1990, 136, 587–591. [Google Scholar] [CrossRef]
- Creff, A.; Brocard, L.; Joubes, J.; Taconnat, L.; Doll, N.M.; Pascal, S.; Galletti, R.; Marsollier, A.-C.; Moussu, S.; Widiez, T.; et al. A stress-response-related inter-compartmental signalling pathway regulates embryonic cuticle integrity in Arabidopsis. bioRxiv 2018, 477109-35. [Google Scholar] [CrossRef]
- Ingram, G.C. Family life at close quarters: Communication and constraint in angiosperm seed development. Protoplasma 2010, 247, 195–214. [Google Scholar] [CrossRef] [PubMed]
- Moussu, S.; Doll, N.M.; Chamot, S.; Brocard, L.; Creff, A.; Fourquin, C.; Widiez, T.; Nimchuk, Z.L.; Ingram, G.C. ZHOUPI and KERBEROS Mediate Embryo/Endosperm Separation by Promoting the Formation of an Extracuticular Sheath at the Embryo Surface. Plant Cell 2017, 29, 1642–1656. [Google Scholar] [CrossRef] [PubMed]
- Morley-Smith, E.R.; Pike, M.J.; Findlay, K.; Köckenberger, W.; Hill, L.M.; Smith, A.; Rawsthorne, S. The transport of sugars to developing embryos is not via the bulk endosperm in oilseed rape seeds. Plant Physiol. 2008, 147, 2121–2130. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.-Q.; Lin, I.W.; Qu, X.-Q.; Sosso, D.; McFarlane, H.E.; Londoño, A.; Samuels, A.L.; Frommer, W.B. A cascade of sequentially expressed sucrose transporters in the seed coat and endosperm provides nutrition for the Arabidopsis embryo. Plant Cell 2015, 27, 607–619. [Google Scholar] [CrossRef] [PubMed]
- Baud, S.; Wuillème, S.; Lemoine, R.; Kronenberger, J.; Caboche, M.; Lepiniec, L.; Rochat, C. The AtSUC5 sucrose transporter specifically expressed in the endosperm is involved in early seed development in Arabidopsis. Plant J. 2005, 43, 824–836. [Google Scholar] [CrossRef]
- Aguirre, M.; Kiegle, E.; Leo, G.; Ezquer, I. Carbohydrate reserves and seed development: An overview. Plant Reprod. 2018, 31, 263–290. [Google Scholar] [CrossRef]
- Durand, M.; Mainson, D.; Porcheron, B.; Maurousset, L.; Lemoine, R.; Pourtau, N. Carbon source-sink relationship in Arabidopsis thaliana: The role of sucrose transporters. Planta 2018, 247, 587–611. [Google Scholar] [CrossRef]
- Chen, L.-Q.; Qu, X.-Q.; Hou, B.-H.; Sosso, D.; Osorio, S.S.; Fernie, A.R.; Frommer, W.B. Sucrose Efflux Mediated by SWEET Proteins as a Key Step for Phloem Transport. Science 2012, 335, 207–211. [Google Scholar] [CrossRef] [PubMed]
- Sauer, N. Molecular physiology of higher plant sucrose transporters. FEBS Lett. 2007, 581, 2309–2317. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Ruan, Y.-L. New insights into roles of cell wall invertase in early seed development revealed by comprehensive spatial and temporal expression patterns of GhCWIN1 in cotton. Plant Physiol. 2012, 160, 777–787. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Ruan, Y.-L. Regulation of cell division and expansion by sugar and auxin signaling. Front. Plant Sci. 2013, 4, 163. [Google Scholar] [CrossRef] [PubMed]
- Baud, S.; Boutin, J.-P.; Miquel, M.; Lepiniec, L.; Rochat, C. An integrated overview of seed development in Arabidopsis thaliana ecotype WS. Plant Physiol. Biochem. 2002, 40, 151–160. [Google Scholar] [CrossRef]
- Hehenberger, E.E.; Kradolfer, D.D.; Köhler, C.C. Endosperm cellularization defines an important developmental transition for embryo development. Development 2012, 139, 2031–2039. [Google Scholar] [CrossRef]
- Serrani, J.C.; Ruiz-Rivero, O.; Fos, M.; García-Martínez, J.L. Auxin-induced fruit-set in tomato is mediated in part by gibberellins. Plant J. 2008, 56, 922–934. [Google Scholar] [CrossRef]
- Ozga, J.A.; Reinecke, D.M.; Ayele, B.T.; Ngo, P.; Nadeau, C.; Wickramarathna, A.D. Developmental and Hormonal Regulation of Gibberellin Biosynthesis and Catabolism in Pea Fruit. Plant Physiol. 2009, 150, 448–462. [Google Scholar] [CrossRef]
- Hu, Y.; Zhou, L.; Huang, M.; He, X.; Yang, Y.; Liu, X.; Li, Y.; Hou, X. Gibberellins play an essential role in late embryogenesis of Arabidopsis. Nat. Plants 2018, 4, 289–298. [Google Scholar] [CrossRef]
- Fuentes, S.; Ljung, K.; Sorefan, K.; Alvey, E.; Harberd, N.P.; Østergaard, L. Fruit growth in Arabidopsis occurs via DELLA-dependent and DELLA-independent gibberellin responses. Plant Cell 2012, 24, 3982–3996. [Google Scholar] [CrossRef]
- Rieu, I.; Eriksson, S.; Powers, S.J.; Gong, F.; Griffiths, J.; Woolley, L.; Benlloch, R.; Nilsson, O.; Thomas, S.G.; Hedden, P.; et al. Genetic analysis reveals that C19-GA 2-oxidation is a major gibberellin inactivation pathway in Arabidopsis. Plant Cell 2008, 20, 2420–2436. [Google Scholar] [CrossRef] [PubMed]
- Ueguchi-Tanaka, M.; Ashikari, M.; Nakajima, M.; Itoh, H.; Katoh, E.; Kobayashi, M.; Chow, T.-Y.; Hsing, Y.-I.C.; Kitano, H.; Yamaguchi, I.; et al. GIBBERELLIN INSENSITIVE DWARF1 encodes a soluble receptor for gibberellin. Nature 2005, 437, 693–698. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.-P. The molecular mechanism and evolution of the GA-GID1-DELLA signaling module in plants. Curr. Biol. 2011, 21, R338–R345. [Google Scholar] [CrossRef]
- Hu, J.; Israeli, A.; Ori, N.; Sun, T.-P. The Interaction between DELLA and ARF/IAA Mediates Crosstalk between Gibberellin and Auxin Signaling to Control Fruit Initiation in Tomato. Plant Cell 2018, 30, 1710–1728. [Google Scholar] [CrossRef] [PubMed]
- De Jong, M.; Wolters-Arts, M.; Feron, R.; Mariani, C.; Vriezen, W.H. The Solanum lycopersicum auxin response factor 7 (SlARF7) regulates auxin signaling during tomato fruit set and development. Plant J. 2009, 57, 160–170. [Google Scholar] [CrossRef] [PubMed]
- Goetz, M.; Vivian-Smith, A.; Johnson, S.D.; Koltunow, A.M. AUXIN RESPONSE FACTOR8 is a negative regulator of fruit initiation in Arabidopsis. Plant Cell 2006, 18, 1873–1886. [Google Scholar] [CrossRef] [PubMed]
- Du, L.; Bao, C.; Hu, T.; Zhu, Q.; Hu, H.; He, Q.; Mao, W. SmARF8, a transcription factor involved in parthenocarpy in eggplant. Mol. Genet. Genom. 2016, 291, 93–105. [Google Scholar] [CrossRef]
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Robert, H.S. Molecular Communication for Coordinated Seed and Fruit Development: What Can We Learn from Auxin and Sugars? Int. J. Mol. Sci. 2019, 20, 936. https://doi.org/10.3390/ijms20040936
Robert HS. Molecular Communication for Coordinated Seed and Fruit Development: What Can We Learn from Auxin and Sugars? International Journal of Molecular Sciences. 2019; 20(4):936. https://doi.org/10.3390/ijms20040936
Chicago/Turabian StyleRobert, Hélène S. 2019. "Molecular Communication for Coordinated Seed and Fruit Development: What Can We Learn from Auxin and Sugars?" International Journal of Molecular Sciences 20, no. 4: 936. https://doi.org/10.3390/ijms20040936
APA StyleRobert, H. S. (2019). Molecular Communication for Coordinated Seed and Fruit Development: What Can We Learn from Auxin and Sugars? International Journal of Molecular Sciences, 20(4), 936. https://doi.org/10.3390/ijms20040936




