The Use of Lupin as a Source of Protein in Animal Feeding: Genomic Tools and Breeding Approaches
Abstract
1. Introduction
2. Economic Importance, Growing Region
2.1. The Genus Lupinus
2.2. The Uses and Production of Lupins
3. The Use of Lupins in Livestock Farming Systems
3.1. Seed Yield Production
3.2. Nutritive Value and Antinutritional Factors
3.3. The Lupins in Livestock Farming Systems
4. Available Genomic Resources and Breeding Trends
5. Breeding of Lupins
5.1. Target Traits for Breeding and Cultivar Release
5.2. Progress and Prospects in Lupin Breeding for Biotic Stresses
5.3. Progress and Prospects in Lupine Breeding for Abiotic Stresses
5.3.1. Salt Stress
5.3.2. Alkaline Soils
5.3.3. Breeding Perspectives
6. Marker-Trait Associations
7. Genome Sequence Efforts in Lupins
8. The Use of Genomic Tools in Breeding Programs
9. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- De Visser, C.L.M.; Schreuder, R.; Stoddard, F. The EU’s dependency on soya bean import for the animal feed industry and potential for EU produced alternatives. Ocl Ol. Corps Gras Lipides 2014, 21. [Google Scholar] [CrossRef]
- Jedrejek, D.; Lević, J.; Wallace, J.; Oleszek, W. Animal by-products for feed: Characteristics, European regulatory framework, and potential impacts on human and animal health and the environment. J. Anim. Feed Sci. 2016, 25, 189–202. [Google Scholar] [CrossRef]
- FEFAC: Statistical Yearbook. 2017. Available online: https://www.fefac.eu/our-publications/statistics (accessed on 26 January 2017).
- COM. Report from the Commission to the Council and the European Parliament on the Development of Plant Proteins in the European Union. 757 Final. pp. 1–15. Available online: https://ec.europa.eu/.../report-plant-proteins-com2018-7 (accessed on 22 November 2018).
- Krautgartner, R.; Lefebvre, L.; Rehder, L.E.; Boshnakova, M.; Dobrescu, M.; Flach, B.; Wilson, W.; Faniadis, D.; Guerrero, M.; Lappin, J.; et al. EU-28: Oilseeds and Products Annual. USDA FOREIGN Agricultural Service, Report Number: AU1803 2018. pp. 1–51. Available online: https://gain.fas.usda.gov (accessed on 3 April 2018).
- Bues, A.; Preißel, S.; Reckling, M.; Zander, P.; Kuhlman, T.; Topp, K.; Watson, C.; Lindström, K.; Stoddard, F.L.; Murphy-Bokern, D. The Environmental Role of Protein Crops in the New Common Agricultural Policy; European Parliament: Brussels, Belgium, 2013.
- FAO. 2006. Available online: www.fao.org/statistics/en/ (accessed on 8 April 2006).
- Geyssens, R. Global Non-GM Soybean Demand to Rise, Driven by EU: Rabobank. The Organic and Non-GMO Reports. 2018. Available online: https://www.agricensus.com/Article/Global-non-GM-soybean-demand-to-rise-driven-by-EU-Rabobank-1367.html (accessed on 11 April 2018).
- Nemecek, T.; von Richthofen, J.-S.; Dubois, G.; Casta, P.; Charles, R.; Pahl, H. Environmental impacts of introducing grain legumes into European crop rotations. Eur. J. Agron. 2008, 28, 380–393. [Google Scholar] [CrossRef]
- Watson, C.A.; Reckling, M.; Preissel, S.; Bachinger, J.; Bergkvist, G.; Kuhlman, T.; Lindström, K.; Nemecek, T.; Topp, C.F.E.; Vanhatalo, A. Grain legume production and use in European agricultural systems. In Advances in Agronomy; Elsevier: Amsterdam, The Netherlands, 2017; Volume 144, pp. 235–303. [Google Scholar]
- Gemede, H.F.; Ratta, N. Antinutritional factors in plant foods: Potential health benefits and adverse effects. Int. J. Nutr. Food Sci. 2014, 3, 284–289. [Google Scholar] [CrossRef]
- Sedláková, K.; Straková, E.; Suchý, P.; Krejcarová, J.; Herzig, I. Lupin as a perspective protein plant for animal and human nutrition—A review. Acta Vet. Brno 2016, 85, 165–175. [Google Scholar] [CrossRef]
- White, C.L.; Staines, V.E. A review of the nutritional value of lupins for dairy cows. Aust. J. Agric. Res. 2007, 58, 185–202. [Google Scholar] [CrossRef]
- Gladstones, J.S. Distribution, origin, taxonomy, history and importance. In Lupins as Crop Plants: Biology, Production, and Utilization; Gladstones, J.S., Atkins, C.A., Hamblin, J., Eds.; CABI: Wallingford, UK, 1998; pp. 1–39. [Google Scholar]
- Naganowska, B.; Wolko, B.; Śliwińska, E.; Kaczmarek, Z. Nuclear DNA content variation and species relationships in the genus Lupinus (Fabaceae). Ann. Bot. 2003, 92, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Ainouche, A.K.; Bayer, R.J. Phylogenetic relationships in Lupinus (Fabaceae: Papilionoideae) based on internal transcribed spacer sequences (its) of nuclear ribosomal DNA. Am. J. Bot. 1999, 86, 590–607. [Google Scholar] [CrossRef]
- Eastwood, R.J.; Drummond, C.S.; Schifino-Wittmann, M.T.; Hughes, C.E. Diversity and evolutionary history of lupins–insights from new phylogenies. In Lupins for Health and Wealth; Palta, J.A., Berger, J.B., Eds.; International Lupin Association: Canterbury, New Zealand, 2008; pp. 346–354. [Google Scholar]
- Wolko, B.; Clements, J.C.; Naganowska, B.; Nelson, M.N.; Yang, H.A. Lupinus. In Wild Crop Relatives: Genomic and Breeding Resources; Springer: Berlin, Germany, 2011; pp. 153–206. [Google Scholar]
- Clements, J.C.; Buirchell, B.J.; Yang, H.; Smith, P.M.C.; Sweetingham, M.W.; Smith, C.G. Lupin. In Genetic Resources, Chromosome Engineering, and Crop Improvement; Grain Legumes; Singh, R.J., Jauhar, P.P., Eds.; CRC Press, Taylor and Francis Group: Boca Raton, FL, USA, 2005; Chapter 9; Volume 1. [Google Scholar]
- Kurlovich, B.S.; Kartuzova, L.T. Lupin breeding. In Lupins (Geography, Classification, Genetic Resources and Breeding); Kurlovich, B.S., Ed.; OY International North Express: St. Petersburg, Russia; Pellosniemi, Finland, 2002; Chapter 13; pp. 351–374. [Google Scholar]
- Petterson, D.S. Composition and food uses of lupins. In Lupins as Crop Plants: Biology, Production, and Utilization; Gladstones, J.S., Atkins, C., Hamblin, J., Eds.; CABI: Wallingford, UK, 1998; pp. 353–384. [Google Scholar]
- Lawrance, L. Lupins: Australia’s role in world markets. Aust. Commod. Forecast. Issues 2007, 14, 353. [Google Scholar]
- Prusinski, J. White lupin (Lupinus albus L.)—Nutritional and health values in human nutrition—A review. Czech J. Food Sci. 2017, 35, 95–103. [Google Scholar]
- Gresta, F.; Wink, M.; Prins, U.; Abberton, M.; Capraro, J.; Scarafoni, A.; Hill, G. Lupins in European cropping systems. In Legumes in Cropping Systems; Murphy-Bokern, D., Stoddard, F.L., Watson, C.A., Eds.; CAB International: Wallingford, UK, 2017; pp. 88–108. [Google Scholar]
- DAFWA. Lupin in Western Australian Farming; Department of Agriculture and Food, Government of Western Australia: Kensington, Australia, 2011. Available online: https://www.agric.wa.gov.au/Crops/ Grains/Lupins (accessed on 22 September 2011).
- Australian Government. The Biology of Lupinus L. (Lupin or Lupine). Department of Health and Ageing Office of the Gene Technology Regulator; 2013. Available online: http://www.ogtr.gov.au (accessed on 19 February 2013).
- FAO. 2018. Available online: http://www.fao.org/faostat/en/#data/QC (accessed on 18 August 2018).
- Lucas, M.M.; Stoddard, F.; Annicchiarico, P.; Frias, J.; Martinez-Villaluenga, C.; Sussmann, D.; Duranti, M.; Seger, A.; Zander, P.; Pueyo, J. The future of lupin as a protein crop in Europe. Front. Plant Sci. 2015, 6, 705. [Google Scholar] [CrossRef] [PubMed]
- Berger, J.D.; Buirchell, B.J.; Luckett, D.J.; Nelson, M.N. Domestication bottlenecks limit genetic diversity and constrain adaptation in narrow-leafed lupin (Lupinus angustifolius L.). Theor. Appl. Genet. 2012, 124, 637–652. [Google Scholar] [CrossRef] [PubMed]
- Annicchiarico, P.; Thami-Alami, I. Enhancing white lupin (Lupinus albus L.) adaptation to calcareous soils through selection of lime-tolerant plant germplasm and bradyrhizobium strains. Plant Soil 2012, 350, 131–144. [Google Scholar] [CrossRef]
- Cernay, C.; Ben-Ari, T.; Pelzer, E.; Meynard, J.-M.; Makowski, D. Estimating variability in grain legume yields across Europe and the Americas. Sci. Rep. 2015, 5, 11171. [Google Scholar] [CrossRef] [PubMed]
- Preissel, S.; Reckling, M.; Schläfke, N.; Zander, P. Magnitude and farm- economic value of grain legume pre-crop benefits in Europe: A review. Field Crop. Res. 2015, 175, 64–79. [Google Scholar] [CrossRef]
- Eurostat. Agriculture, Forestry and Fishery Statistics. 2016. Available online: http://ec.europa.eu/eurostat (accessed on 22 December 2016).
- Kingwell, R. Economic considerations for Lupin Protein Concentrate production. In Proceedings of the 1st Workshop for Seeding a Future for Grains in Aquaculture Feeds, Fremantle, Australia, 28 May 2003; pp. 16–20. [Google Scholar]
- GRDC-Queensland Government, IPM Guidelines for Grains. 10 Pulse Australia, (1 August 2016) Australian Pulse Standards 2016/17, Lupins, L. angustifolius Minimum Receival Standards Farmer Dressed Parameter Requirements. Available online: http://ipmguidelinesforgrains.com.au/wp-content/uploads/Etiella.pdf (accessed on 1 August 2016).
- Beyer, H.; Schmalenberg, A.K.; Jansen, G.; Jürgens, H.U.; Uptmoor, R.; Broer, I.; Huckauf, J.; Dietrich, R.; Michel, V.; Zenk, A.; et al. Evaluation of variability, heritability and environmental stability of seed quality and yield parameters of L. angustifolius. Field Crop. Res. 2015, 174, 40–47. [Google Scholar] [CrossRef]
- Bhardwaj, H.L.; Hamama, A.A.; Merrick, L.C. Genotypic and environmental effects on lupin seed composition. Plant Foods Hum. Nutr. 1998, 53, 1–13. [Google Scholar] [CrossRef]
- Jansen, G.; Jürgens, H.-U.; Schliephake, E.; Ordon, F. Effect of the soil PH on the alkaloid content of Lupinus angustifolius. Int. J. Agron. 2012, 2012, 269878. [Google Scholar] [CrossRef]
- Jansen, G.; Jürgens, H.U.; Ordon, F. Effects of temperature on the alkaloid content of seeds of Lupinus angustifolius cultivars. J. Agron. Crop Sci. 2009, 195, 172–177. [Google Scholar] [CrossRef]
- Tadele, Y. White lupin (Lupinus albus) grain, a potential source of protein for ruminants: A review. Res. J. Agric. Environ. Manag. 2015, 4, 180–188. [Google Scholar]
- Amir, R.; Han, T.; Ma, F. Bioengineering approaches to improve the nutritional values of seeds by increasing their methionine content. Mol. Breed. 2012, 29, 915–924. [Google Scholar] [CrossRef]
- Huyghe, C. White lupin (Lupinus albus L.). Field Crop. Res. 1997, 53, 147–160. [Google Scholar] [CrossRef]
- Suchý, P.; Straková, E.; Kroupa, L.; Večerek, V. The fatty acid content of oil from seeds of some lupin varieties. In Lupins for Health and Wealth; Palta, J.A., Berger, J.B., Eds.; International Lupin Association: Canterbury, New Zealand, 2008; pp. 188–191. [Google Scholar]
- Chiofalo, B.; Presti, V.L.; Chiofalo, V.; Gresta, F. The productive traits, fatty acid profile and nutritional indices of three lupin (Lupinus spp.) species cultivated in a Mediterranean environment for the livestock. Anim. Feed Sci. Technol. 2012, 171, 230–239. [Google Scholar] [CrossRef]
- Sbihi, H.M.; Nehdi, I.A.; Tan, C.P.; Al-Resayes, S.I. Bitter and sweet lupin (lupinus albus l.) seeds and seed oils: A comparison study of their compositions and physicochemical properties. Ind. Crop. Prod. 2013, 49, 573–579. [Google Scholar] [CrossRef]
- Sujak, A.; Kotlarz, A.; Strobel, W. Compositional and nutritional evaluation of several lupin seeds. Food Chem. 2006, 98, 711–719. [Google Scholar] [CrossRef]
- Kasprowicz-Potocka, M.; Borowczyk, P.; Zaworska, A.; Nowak, W.; Frankiewicz, A.; Gulewicz, P. The effect of dry yeast fermentation on chemical composition and protein characteristics of blue lupin seeds. Food Technol. Biotechnol. 2016, 54, 360. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Villaluenga, C.; Frías, J.; Vidal-Valverde, C. Functional lupin seeds (Lupinus albus L. and Lupinus luteus L.) after extraction of α-galactosides. Food Chem. 2006, 98, 291–299. [Google Scholar] [CrossRef]
- Campos, D.; Chirinos, R.; Ranilla, L.G.; Pedreschi, R. Bioactive potential of andean fruits, seeds, and tubers. In Advances in Food and Nutrition Research; Elsevier: Amsterdam, The Netherlands, 2018; Volume 84, pp. 287–343. [Google Scholar]
- Carvajal-Larenas, F.E.; Linnemann, A.R.; Nout, M.J.R.; Koziol, M.; Van Boekel, M. Lupinus mutabilis: Composition, uses, toxicology, and debittering. Crit. Rev. Food Sci. Nutr. 2016, 56, 1454–1487. [Google Scholar] [CrossRef]
- Nalle, C.L.; Ravindran, V.; Ravindran, G. Nutritional value of white lupins (Lupinus albus) for broilers: Apparent metabolisable energy, apparent ileal amino acid digestibility and production performance. Animal 2012, 6, 579–585. [Google Scholar] [CrossRef]
- Erbaş, M.; Certel, M.; Uslu, M.K. Some chemical properties of white lupin seeds (Lupinus albus L.). Food Chem. 2005, 89, 341–345. [Google Scholar] [CrossRef]
- Steenfeldt, S.; González, E.; Knudsen, K.E.B. Effects of inclusion with blue lupins (Lupinus angustifolius) in broiler diets and enzyme supplementation on production performance, digestibility and dietary AME content. Anim. Feed Sci. Technol. 2003, 110, 185–200. [Google Scholar] [CrossRef]
- Palander, S.; Laurinen, P.; Perttilä, S.; Valaja, J.; Partanen, K. Protein and amino acid digestibility and metabolizable energy value of pea (Pisum sativum), faba bean (Vicia faba) and lupin (Lupinus angustifolius) seeds for turkeys of different age. Anim. Feed Sci. Technol. 2006, 127, 89–100. [Google Scholar] [CrossRef]
- Pieper, R.; Taciak, M.; Pieper, L.; Święch, E.; Tuśnio, A.; Barszcz, M.; Vahjen, W.; Skomiał, J.; Zentek, J. Comparison of the nutritional value of diets containing differentially processed blue sweet lupin seeds or soybean meal for growing pigs. Anim. Feed Sci. Technol. 2016, 221, 79–86. [Google Scholar] [CrossRef]
- Borreani, G.; Chion, A.R.; Colombini, S.; Odoardi, M.; Paoletti, R.; Tabacco, E. Fermentative profiles of field pea (Pisum sativum), faba bean (Vicia faba) and white lupin (Lupinus albus) silages as affected by wilting and inoculation. Anim. Feed Sci. Technol. 2009, 151, 316–323. [Google Scholar] [CrossRef]
- Bolland, M.D.A.; Brennan, R.F. Comparing the phosphorus requirements of wheat, lupin, and canola. Aust. J. Agric. Res. 2008, 59, 983–998. [Google Scholar] [CrossRef]
- Fumagalli, P.; Comolli, R.; Ferre, C.; Ghiani, A.; Gentili, R.; Citterio, S. The rotation of white lupin (Lupinus albus L.) with metal-accumulating plant crops: A strategy to increase the benefits of soil phytoremediation. J. Environ. Manag. 2014, 145, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Froidmont, E.; Bartiaux-Thill, N. Suitability of lupin and pea seeds as a substitute for soybean meal in high-producing dairy cow feed. Anim. Res. 2004, 53, 475–487. [Google Scholar] [CrossRef]
- Vicenti, A.; Toteda, F.; Di Turi, L.; Cocca, C.; Perrucci, M.; Melodia, L.; Ragni, M. Use of sweet lupin (Lupinus albus L. var. Multitalia) in feeding for Podolian young bulls and influence on productive performances and meat quality traits. Meat Sci. 2009, 82, 247–251. [Google Scholar] [CrossRef]
- Masucci, F.; Di Francia, A.; Romano, R.; Maresca di Serracapriola, M.T.; Lambiase, G.; Varricchio, M.L.; Proto, V. Effect of Lupinus albus as protein supplement on yield, constituents, clotting properties and fatty acid composition in ewes’ milk. Small Rumin. Res. 2006, 65, 251–259. [Google Scholar] [CrossRef]
- Tadele, Y.; Mekasha, Y.; Tegegne, F. Supplementation with Different Forms of Processed Lupin (Lupinus albus) Grain in Hay Based Feeding of Washera Sheep: Effect on Feed Intake, Digestibility Body Weight and Carcass Parameters. J. Biol. Agric. Healthc. 2014, 4, 213–231. [Google Scholar]
- Tefera, G.; Tegegne, F.; Mekuriaw, Y.; Melaku, S.; Tsumekawa, A. Effects of different forms of white lupin (Lupinus albus) grain supplementation on feed intake, digestibility, growth performance and carcass characteristics of Washera sheep fed Rhodes grass (Chloris gayana) hay-based diets. Trop. Anim. Health Prod. 2015, 47, 1581–1590. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.R.F.; Parkinson, S.; Fleming, H.R.; Theobald, V.J.; Leemans, D.K.; Burgess, T. The potential of blue lupins as a protein source, in the diets of laying hens. Vet. Anim. Sci. 2016, 1, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Laudadio, V.; Tufarelli, V. Influence of substituting dietary soybean meal for dehulled-micronized lupin (Lupinus albus cv. Multitalia) on early phase laying hens production and egg quality. Livest. Sci. 2011, 140, 184–188. [Google Scholar] [CrossRef]
- Koivunen, E.; Partanen, K.; Perttilä, S.; Palander, S.; Tuunainen, P.; Valaja, J. Digestibility and energy value of pea (Pisum sativum L.), faba bean (Vicia faba L.) and blue lupin (narrow-leaf) (Lupinus angustifolius) seeds in broilers. Anim. Feed Sci. Technol. 2016, 218, 120–127. [Google Scholar] [CrossRef]
- Abberton, M.; Batley, J.; Bentley, A.; Bryant, J.; Cai, H.; Cockram, J.; Costa de Oliveira, A.; Cseke, L.J.; Dempewolf, H.; De Pace, C.; et al. Global agricultural intensification during climate change: A role for genomics. Plant Biotechnol. J. 2016, 14, 1095–1098. [Google Scholar] [CrossRef] [PubMed]
- Gomiero, T. Soil degradation, land scarcity and food security: Reviewing a complex challenge. Sustainability 2016, 8, 281. [Google Scholar] [CrossRef]
- Smýkal, P.; Coyne, C.J.; Ambrose, M.J.; Maxted, N.; Schaefer, H.; Blair, M.W.; Berger, J.; Greene, S.L.; Nelson, M.N.; Besharat, N.; et al. Legume crops phylogeny and genetic diversity for science and breeding. Crit. Rev. Plant Sci. 2015, 34, 43–104. [Google Scholar] [CrossRef]
- Clements, J.C.; Cowling, W.A. Patterns of morphological diversity in relation to geographical origins of wild Lupinus angustifolius from the Aegean region. Genet. Resour. Crop Evol. 1994, 41, 109–122. [Google Scholar] [CrossRef]
- Mousavi-Derazmahalleh, M.; Bayer, P.E.; Nevado, B.; Hurgobin, B.; Filatov, D.; Kilian, A.; Kamphuis, L.G.; Singh, K.B.; Berger, J.D.; Hane, J.K.; et al. Exploring the genetic and adaptive diversity of a pan-mediterranean crop wild relative: Narrow-leafed lupin. Theor. Appl. Genet. 2018, 131, 887–901. [Google Scholar] [CrossRef] [PubMed]
- Gladstones, J.S. Lupins as crop plants. Field Crop Abstr. 1970, 23, 123–148. [Google Scholar]
- Nelson, M.N.; Książkiewicz, M.; Rychel, S.; Besharat, N.; Taylor, C.M.; Wyrwa, K.; Jost, R.; Erskine, W.; Cowling, W.A.; Berger, J.D.; et al. The loss of vernalization requirement in narrow-leafed lupin is associated with a deletion in the promoter and de-repressed expression of a flowering locus t (ft) homologue. New Phytol. 2017, 213, 220–232. [Google Scholar] [CrossRef]
- Gross, R.; von Baer, E.; Koch, F.; Marquard, R.; Trugo, L.; Wink, M. Chemical composition of a new variety of the Andean lupin (Lupinus mutabilis cv. Inti) with low-alkaloid content. J. Food Compos. Anal. 1988, 1, 353–361. [Google Scholar] [CrossRef]
- Eastwood, R.J.; Hughes, C.E. Origins of domestication of Lupinus mutabilis in the Andes. In Lupins for Health and Wealth; Palta, J.A., Berger, J.B., Eds.; International Lupin Association: Canterbury, New Zealand, 2008; pp. 373–379. [Google Scholar]
- Święcicki, W.; Kroc, M.; Kamel, K.A. Lupins. In Grain Legumes; Springer: Berlin, Germany, 2015; pp. 179–218. [Google Scholar]
- Annicchiarico, P.; Manunza, P.; Proietti, S. White Lupin Tolerance to Winter Cold, Terminal Drought and Soil Lime: Patterns of Genetic Variation and Their Exploitation in Breeding for Southern Europe; International Lupin Association: Canterbury, New Zealand, 2011; pp. 99–103. [Google Scholar]
- Atnaf, M.; Wegary, D.; Dagne, K.; Tesfaye, K. Genotype by environment interaction and grain yield stability of Ethiopian white lupin (Lupinus albus L.) landraces. Exp. Agric. 2018, 54, 943–956. [Google Scholar] [CrossRef]
- Boschin, G.; D’Agostina, A.; Annicchiarico, P.; Arnoldi, A. Effect of genotype and environment on fatty acid composition of Lupinus albus L. Seed. Food Chem. 2008, 108, 600–606. [Google Scholar] [CrossRef] [PubMed]
- Annicchiarico, P.; Thami-Alami, I. White lupin improvement for countries of the Mediterranean basin. Legume Perspect. 2015, 10, 12–14. [Google Scholar]
- Cowling, W.A.; Huyghe, C.; Swiecicki, W. Lupin breeding. In Lupins as Crop Plants: Biology, Production and Utilization; Gladstones, J., Atkins, C.A., Hamblin, J., Eds.; CAB International: London, UK, 1998; pp. 93–120. [Google Scholar]
- Adhikari, K.N.; Buirchell, B.J.; Thomas, G.J.; Sweetingham, M.W.; Yang, H. Identification of anthracnose resistance in Lupinus albus L. And its transfer from landraces to modern cultivars. Crop Pasture Sci. 2009, 60, 472–479. [Google Scholar] [CrossRef]
- Adhikari, K.N.; Thomas, G.; Diepeveen, D.; Trethowan, R. Overcoming the barriers of combining early flowering and anthracnose resistance in white lupin (Lupinus albus L.) for the northern agricultural region of western australia. Crop Pasture Sci. 2013, 64, 914–921. [Google Scholar] [CrossRef]
- Jacob, I.; Feuerstein, U.; Heinz, M.; Schott, M.; Urbatzka, P. Evaluation of new breeding lines of white lupin with improved resistance to anthracnose. Euphytica 2017, 213, 236. [Google Scholar] [CrossRef]
- Sengbusch, R.V. The breeding of sweet lupins. Herb. Rev. 1938, 6, 1–9. [Google Scholar]
- Golovchenko, V.I. Mutagenesis in Selection of Alkaloidless Varieties of White and Yellow Lupine; Ministri of Agriculture Madrid: Madrid, Spain, 1982; pp. 43–45.
- Si, P.; Buirchell, B.; Sweetingham, M. Improved metribuzin tolerance in narrow-leafed lupin (Lupinus angustifolius L.) by induced mutation and field selection. Field Crop. Res. 2009, 113, 282–286. [Google Scholar] [CrossRef]
- Gladstones, J.S. Breeding lupins in Western Australia. J. Dep. Agric. West. Aust. Ser. 4 1982, 23, 73–76. [Google Scholar]
- Hajjar, R.; Hodgkin, T. The use of wild relatives in crop improvement: A survey of developments over the last 20 years. Euphytica 2007, 156, 1–13. [Google Scholar] [CrossRef]
- Annicchiarico, P.; Harzic, N.; Carroni, A.M. Adaptation, diversity, and exploitation of global white lupin (Lupinus albus L.) landrace genetic resources. Field Crop. Res. 2010, 119, 114–124. [Google Scholar] [CrossRef]
- Atnaf, M.; Tesfaye, K.; Dagne, K.; Wegary, D. Genotype by trait biplot analysis to study associations and profiles of ethiopian white lupin (Lupinus albus L.) landraces. Aust. J. Crop Sci. 2017, 11, 55. [Google Scholar] [CrossRef]
- Fischer, K.; Dieterich, R.; Nelson, M.N.; Kamphuis, L.G.; Singh, K.B.; Rotter, B.; Krezdorn, N.; Winter, P.; Wehling, P.; Ruge-Wehling, B. Characterization and mapping of lanrbo: A locus conferring anthracnose resistance in narrow-leafed lupin (Lupinus angustifolius L.). Theor. Appl. Genet. 2015, 128, 2121–2130. [Google Scholar] [CrossRef] [PubMed]
- Phan, H.T.T.; Ellwood, S.R.; Adhikari, K.; Nelson, M.N.; Oliver, R.P. The first genetic and comparative map of white lupin (Lupinus albus L.): Identification of qtls for anthracnose resistance and flowering time, and a locus for alkaloid content. DNA Res. 2007, 14, 59–70. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Lin, R.; Renshaw, D.; Li, C.; Adhikari, K.; Thomas, G.; Buirchell, B.; Sweetingham, M.; Yan, G. Development of sequence-specific PCR markers associated with a polygenic controlled trait for marker-assisted selection using a modified selective genotyping strategy: A case study on anthracnose disease resistance in white lupin (Lupinus albus L.). Mol. Breed. 2010, 25, 239–249. [Google Scholar] [CrossRef]
- You, M.; Boersma, J.G.; Buirchell, B.J.; Sweetingham, M.W.; Siddique, K.H.; Yang, H. A pcr-based molecular marker applicable for marker-assisted selection for anthracnose disease resistance in lupin breeding. Cellural Mol. Biol. Lett. 2005, 10, 123–134. [Google Scholar]
- Rubiales, D.; Mikic, A. Introduction: Legumes in Sustainable Agriculture; Taylor & Francis: Abingdon, UK, 2015. [Google Scholar]
- Nirenberg, H.I.; Feiler, U.; Hagedorn, G. Description of Colletotrichum lupini comb. nov. in modern terms. Mycologia 2002, 94, 307–320. [Google Scholar] [CrossRef]
- Shea, G.; Cowling, W.A.; Burchell, B.J.; Luckett, D.; Yang, H.; Sweetingham, M.W.; Thomas, G.J. Managing lupin anthracnose. J. Dep. Agric. West. Aust. Ser. 4 1999, 40, 24–30. [Google Scholar]
- Riegel, R.; Véliz, D.; von Baer, I.; Quitral, Y.; Muñoz, M. Genetic diversity and virulence of Colletotrichum lupini isolates collected in chile. Trop. Plant Pathol. 2010, 35, 144–152. [Google Scholar] [CrossRef]
- Talhinhas, P.; Baroncelli, R.; Le Floch, G. Anthracnose of lupins caused by Colletotrichum lupini: A recent disease and a successful worldwide pathogen. J. Plant Pathol. 2016, 98, 5–14. [Google Scholar]
- Yang, H.A.; Sweetingham, M.W. The taxonomy of colletotrichum isolates associated with lupin anthracnose. Aust. J. Agric. Res. 1998, 49, 1213–1224. [Google Scholar] [CrossRef]
- Berger, J.D.; Clements, J.C.; Nelson, M.N.; Kamphuis, L.G.; Singh, K.B.; Buirchell, B. The essential role of genetic resources in narrow-leafed lupin improvement. Crop Pasture Sci. 2013, 64, 361–373. [Google Scholar] [CrossRef]
- Yang, H.; Boersma, J.G.; You, M.; Buirchell, B.J.; Sweetingham, M.W. Development and implementation of a sequence-specific PCR marker linked to a gene conferring resistance to anthracnose disease in narrow-leafed lupin (Lupinus angustifolius L.). Mol. Breed. 2004, 14, 145–151. [Google Scholar] [CrossRef]
- Yang, H.; Renshaw, D.; Thomas, G.; Buirchell, B.; Sweetingham, M. A strategy to develop molecular markers applicable to a wide range of crosses for marker assisted selection in plant breeding: A case study on anthracnose disease resistance in lupin (Lupinus angustifolius L.). Mol. Breed. 2008, 21, 473–483. [Google Scholar] [CrossRef]
- Adhikari, K.; Buirchell, B.; Yan, G.; Sweetingham, M. Two complementary dominant genes control flowering time in albus lupin (Lupinus albus L.). Plant Breed. 2011, 130, 496–499. [Google Scholar] [CrossRef]
- O’Connell, R.J.; Thon, M.R.; Hacquard, S.; Amyotte, S.G.; Kleemann, J.; Torres, M.F.; Damm, U.; Buiate, E.A.; Epstein, L.; Alkan, N. Lifestyle transitions in plant pathogenic Colletotrichum fungi deciphered by genome and transcriptome analyses. Nat. Genet. 2012, 44, 1060–1065. [Google Scholar] [CrossRef] [PubMed]
- Baroncelli, R.; Sreenivasaprasad, S.; Sukno, S.A.; Thon, M.R.; Holub, E. Draft genome sequence of Colletotrichum acutatum sensu lato (Colletotrichum fioriniae). Genome Announc. 2014, 2, e00112-14. [Google Scholar] [CrossRef]
- Yang, H.; Tao, Y.; Zheng, Z.; Li, C.; Sweetingham, M.W.; Howieson, J.G. Application of next-generation sequencing for rapid marker development in molecular plant breeding: A case study on anthracnose disease resistance in Lupinus angustifolius L. BMC Genomics 2012, 13, 318. [Google Scholar] [CrossRef]
- Yang, H.; Tao, Y.; Zheng, Z.; Shao, D.; Li, Z.; Sweetingham, M.W.; Buirchell, B.J.; Li, C. Rapid development of molecular markers by next-generation sequencing linked to a gene conferring phomopsis stem blight disease resistance for marker-assisted selection in lupin (Lupinus angustifolius L.) breeding. Theor. Appl. Genet. 2013, 126, 511–522. [Google Scholar] [CrossRef] [PubMed]
- Zian, A.H.; El-Demardash, I.S.; El-Mouhamady, A.A.; El-Barougy, E. Studies the resistance of lupine for Fusarium oxysporum F. sp Lupini through molecular genetic technique. World Appl. Sci. J. 2013, 26, 1064–1069. [Google Scholar]
- Mohamed, H.I.; El-Rahman, S.S.A.; Mazen, M.M. Influence of some biotic and abiotic inducers on fusarium wilt disease incidence of lupin (Lupinus albus) on disease resistance and protein pattern. Plant Pathol. Quar. 2012, 2, 179–188. [Google Scholar] [CrossRef]
- El-Rahman, S.S.A.; Mohamed, M.M.; Mohamed, H.I.; Mahmoud, N.M. Induction of defence related enzymes and phenolic compounds in lupin (Lupinus albus L.) and their effects on host resistance against Fusarium wilt. Eur. J. Plant Pathol. 2012, 134, 105–116. [Google Scholar] [CrossRef]
- Infantino, A.; Kharrat, M.; Riccioni, L.; Coyne, C.J.; McPhee, K.E.; Grunwald, N.J. Screening techniques and sources of resistance to root diseases in cool season food legumes. Euphytica 2006, 147, 201–221. [Google Scholar] [CrossRef]
- Cowling, W.A.; Sweetingham, M.W.; Diepeveen, D.; Cullis, B.R. Heritability of resistance to brown spot and root rot of narrow-leafed lupins caused by Pleiochaeta setosa (Kirchn.). Hughes in field experiments. Plant Breed. 2006, 116, 341–345. [Google Scholar] [CrossRef]
- Williamson, P.M.; Highet, A.S.; Gams, W.; Sivasithamparam, K.; Cowling, W.A. Diaporthe toxica sp. Nov., the cause of lupinosis in sheep. Mycol. Res. 1994, 98, 1364–1368. [Google Scholar] [CrossRef]
- Wood, P.M.; Allen, J.G. Control of ovine lupinosis: Use of a resistant cultivar of Lupinus albus-cv. Ultra. Aust. J. Exp. Agric. 1980, 20, 316–318. [Google Scholar] [CrossRef]
- Warren, J.M.; Allen, J.; Cowling, W. Economic impact of growing phomopsis-resistant lupins. J. Dep. Agric. West. Aust. Ser. 4 1989, 30, 8–10. [Google Scholar]
- Shankar, M.; Sweetingham, M.W.; Cowling, W.A. Identification of alleles at two loci controlling resistance to phomopsis stem blight in narrow-leafed lupin (Lupinus angustifolius L.). Euphytica 2002, 125, 35–44. [Google Scholar] [CrossRef]
- Wilkie, J.P.; Dye, D.W.; Watson, D.R.W. Further hosts of Pseudomonas viridiflava. N. Z. J. Agric. Res. 1973, 16, 315–323. [Google Scholar] [CrossRef]
- Lu, S.-E.; Gross, D.C. Drippy pod of white lupine: A new bacterial disease caused by a pathovar of brenneria quercina. Plant Dis. 2010, 94, 1431–1440. [Google Scholar] [CrossRef] [PubMed]
- Jones, R.A.C.; McLean, G.D. Virus diseases of lupins. Ann. Appl. Biol. 1989, 114, 609–637. [Google Scholar] [CrossRef]
- Sweetingham, M.W.; Jones, R.A.C.; Brown, A.G.P. Diseases and pests. In Lupins as Crop Plants: Biology, Production and Utilization; Gladstones, J.S., Atkins, C.A., Hamblin, J., Eds.; CAB International: Wallingford, UK, 1998; Chapter 9; pp. 263–289. [Google Scholar]
- Pirone, T.P.; Harris, K.F. Nonpersistent transmission of plant viruses by aphids. Annu. Rev. Phytopathol. 1977, 15, 55–73. [Google Scholar] [CrossRef]
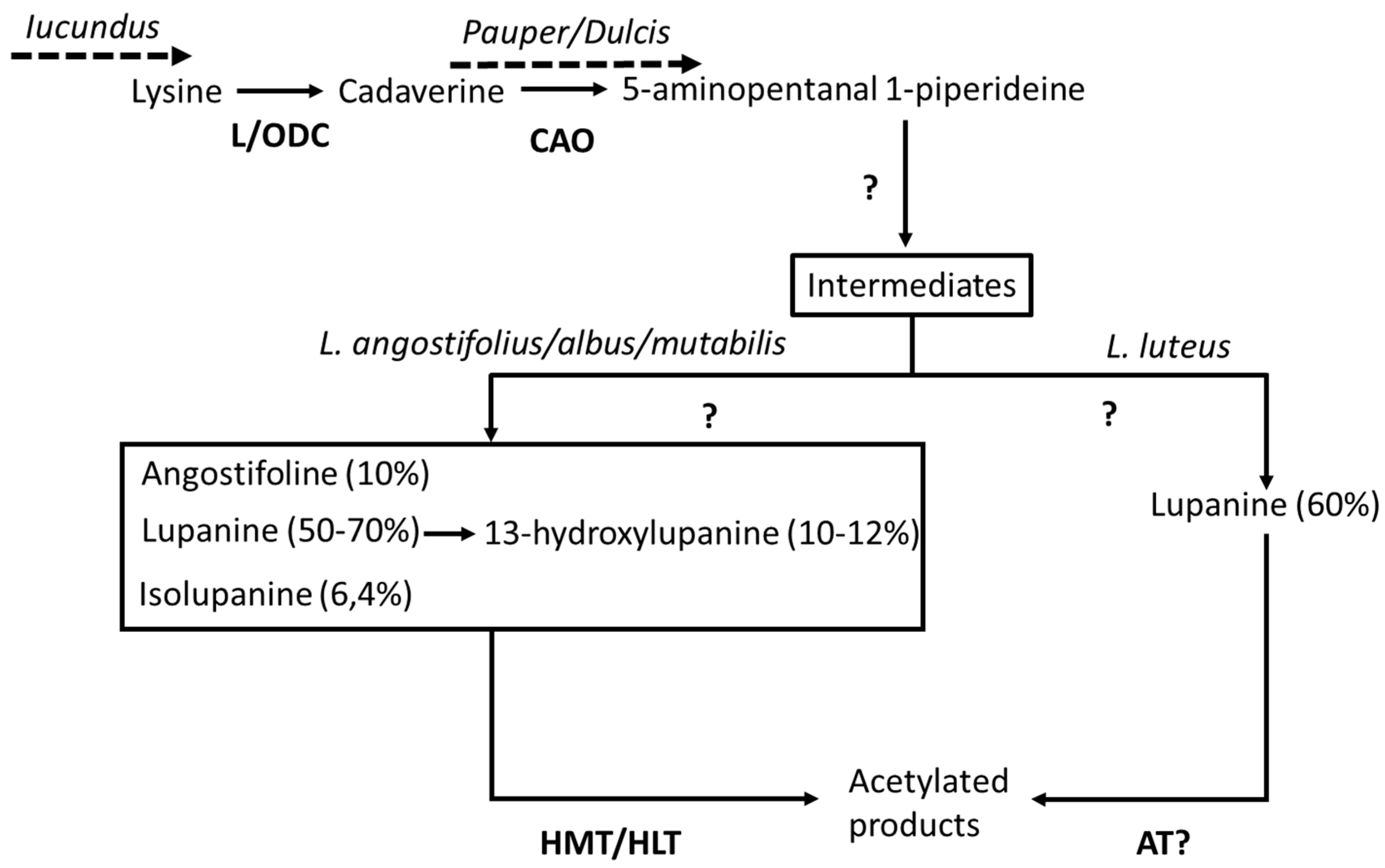
- Frick, K.M.; Kamphuis, L.G.; Siddique, K.H.M.; Singh, K.B.; Foley, R.C. Quinolizidine alkaloid biosynthesis in lupins and prospects for grain quality improvement. Front. Plant Sci. 2017, 8, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Adhikan, K.N.; Edwards, O.R.; Wang, S.; Ridsdill-Smith, T.J.; Buirchell, B. The role of alkaloids in conferring aphid resistance in yellow lupin (Lupinus luteus L.). Crop Pasture Sci. 2012, 63, 444–451. [Google Scholar] [CrossRef]
- Philippi, J.; Schliephake, E.; Jürgens, H.; Jansen, G.; Ordon, F. Feeding behavior of aphids on narrow-leafed lupin (Lupinus angustifolius) genotypes varying in the content of quinolizidine alkaloids. Entomol. Exp. Appl. 2015, 156, 37–51. [Google Scholar] [CrossRef]
- Lee, M.J.; Pate, J.S.; Harris, D.J.; Atkins, C.A. Synthesis, transport and accumulation of quinolizidine alkaloids in Lupinus albus L. and Lupinus angustifolius L. J. Exp. Bot. 2007, 58, 935–946. [Google Scholar] [CrossRef]
- Fernández-Pascual, M.; Pueyo, J.J.; Felipe, M.; Golvano, M.P.; Lucas, M.M. Singular features of the bradyrhizobium-lupinus symbiosis. Dyn. Soil Dyn. Plant 2007, 1, 1–16. [Google Scholar]
- Coba de la Peña, T.; Pueyo, J.J. Legumes in the reclamation of marginal soils, from cultivar and inoculant selection to transgenic approaches. Agron. Sustain. Dev. 2012, 32, 65–91. [Google Scholar] [CrossRef]
- Palta, J.A.; Berger, J.; Bramley, H. Physiology of the yield under drought: Lessons from studies with lupin. In Plant Responses to Drought Stress: From Morphological to Molecular Features; Aroca, R., Ed.; Springer: Berlin, Germany, 2012; pp. 417–440. [Google Scholar]
- Chen, Y.L.; Dunbabin, V.M.; Postma, J.A.; Diggle, A.J.; Palta, J.A.; Lynch, J.P.; Rengel, Z. Phenotypic variability and modelling of root structure of wild Lupinus angustifolius genotypes. Plant Soil 2011, 348, 345–364. [Google Scholar] [CrossRef]
- Roach, D.A.; Wulff, R.D. Maternal effects in plants. Annu. Rev. Ecol. Syst. 1987, 18, 209–235. [Google Scholar] [CrossRef]
- Kalandyk, A.; Waligórski, P.; Dubert, F. Role of the maternal effect phenomena in improving water stress tolerance in narrow-leafed lupine (Lupinus angustifolius). Plant Breed. 2017, 136, 167–173. [Google Scholar] [CrossRef]
- Annicchiarico, P.; Romani, M.; Pecetti, L. White lupin (Lupinus albus) variation for adaptation to severe drought stress. Plant Breed. 2018, 137, 782–789. [Google Scholar] [CrossRef]
- Juzoń, K.; Czyczyło-Mysza, I.; Marcińska, I.; Dziurka, M.; Waligórski, P.; Skrzypek, E. Polyamines in yellow lupin (Lupinus luteus L.) tolerance to soil drought. Acta Physiol. Plant. 2017, 39, 202. [Google Scholar] [CrossRef]
- Lizarazo Torres, C.I. Genetic Variability in the Physiological Responses of Andean Lupin to Drought Stress. Master’s Thesis, University of Helsinki, Helsinki, Finland, 2010. [Google Scholar]
- Fernández-Pascual, M.; de Lorenzo, C.; de Felipe, M.R.; Rajalakshmi, S.; Gordon, A.J.; Thomas, B.J.; Minchin, F.R. Possible reasons for relative salt stress tolerance in nodules of white lupin cv. Multolupa. J. Exp. Bot. 1996, 47, 1709–1716. [Google Scholar] [CrossRef]
- Niu, G.; Rodriguez, D.; Aguiniga, L.; Mackay, W. Salinity tolerance of Lupinus havardii and Lupinus texensis. HortScience 2007, 42, 526–528. [Google Scholar] [CrossRef]
- Mikić, A.; Mihailovic, V.; Cupina, B.; Djordjevic, V.; Stoddard, F. Introduction of Novel Crops in Serbia—White lupin (Lupinus albus). Field Veg. Crops Res. 2010, 47, 21–26. [Google Scholar]
- Vlachostergios, D.; Divanes, I.; Fyntanis, A.; Koseoglou, E.; Mavromatis, A. Selection of white lupine genotypes for yield and tolerance to alkaline soils. In General Eucarpia Congress. “Plant Breeding for Future Generations”; Zurich: Zürich, Switzerland, 2016. [Google Scholar]
- Cowling, W.; Gladstones, J. Lupin Breeding in Australia; Springer: Dordrecht, The Netherlands, 2000; Volume 34, pp. 541–547. [Google Scholar]
- Kroc, M.; Rybiński, W.; Wilczura, P.; Kamel, K.; Kaczmarek, Z.; Barzyk, P.; Święcicki, W. Quantitative and qualitative analysis of alkaloids composition in the seeds of a white lupin (Lupinus albus l.) collection. Genet. Resour. Crop Evol. 2017, 64, 1853–1860. [Google Scholar] [CrossRef]
- Bunsupa, S.; Yamazaki, M.; Saito, K. Quinolizidine alkaloid biosynthesis: Recent advances and future prospects. Front. Plant Sci. 2012, 3, 239. [Google Scholar] [CrossRef]
- Frick, K.M.; Foley, R.C.; Kamphuis, L.G.; Siddique, K.H.M.; Garg, G.; Singh, K.B. Characterization of the genetic factors affecting quinolizidine alkaloid biosynthesis and its response to abiotic stress in narrow-leafed lupin (Lupinus angustifolius L.). Plant Cell Environ. 2018, 41, 2155–2168. [Google Scholar] [CrossRef] [PubMed]
- Hane, J.K.; Ming, Y.; Kamphuis, L.G.; Nelson, M.N.; Garg, G.; Atkins, C.A.; Bayer, P.E.; Bravo, A.; Bringans, S.; Cannon, S.; et al. A comprehensive draft genome sequence for lupin (Lupinus angustifolius), an emerging health food: Insights into plant-microbe interactions and legume evolution. Plant Biotechnol. J. 2017, 15, 318–330. [Google Scholar] [CrossRef] [PubMed]
- Nelson, M.N.; Moolhuijzen, P.M.; Boersma, J.G.; Chudy, M.; Lesniewska, K.; Bellgard, M.; Oliver, R.P.; Swiecicki, W.; Wolko, B.; Cowling, W.A.; et al. Aligning a new reference genetic map of Lupinus angustifolius with the genome sequence of the model legume, lotus japonicus. DNA Res. 2010, 17, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Yang, H.; Buirchell, B.; Yan, G. Development of a DNA marker tightly linked to low-alkaloid gene iucundus in narrow-leafed lupin (Lupinus angustifolius L.) for marker-assisted selection. Crop Pasture Sci. 2011, 62, 218–224. [Google Scholar] [CrossRef]
- Harrison, J.E.M.; Williams, W. Genetical control of alkaloids in Lupinus albus. Euphytica 1982, 31, 357–364. [Google Scholar] [CrossRef]
- Saito, K.; Koike, Y.; Suzuki, H.; Murakoshi, I. Biogenetic implication of lupin alkaloid biosynthesis in bitter and sweet forms of Lupinus luteus and L. albus. Phytochemistry 1993, 34, 1041–1044. [Google Scholar] [CrossRef]
- Lin, R.; Renshaw, D.; Luckett, D.; Clements, J.; Yan, G.; Adhikari, K.; Buirchell, B.; Sweetingham, M.; Yang, H. Development of a sequence-specific pcr marker linked to the gene “pauper” conferring low-alkaloids in white lupin (Lupinus albus L.) for marker assisted selection. Mol. Breed. 2009, 23, 153–161. [Google Scholar] [CrossRef]
- Boersma, J.G.; Buirchell, B.J.; Sivasithamparam, K.; Yang, H. Development of a sequence-specific PCR marker linked to the ku gene which removes the vernalization requirement in narrow-leafed lupin. Plant Breed. 2007, 126, 306–309. [Google Scholar] [CrossRef]
- Li, X.; Yang, H.; Yan, G. Development of a co-dominant DNA marker linked to the gene lentus conferring reduced pod shattering for marker-assisted selection in narrow-leafed lupin (Lupinus angustifolius) breeding. Plant Breed. 2012, 131, 540–544. [Google Scholar] [CrossRef]
- Adhikari, K.N.; Buirchell, B.J.; Sweetingham, M.W. Length of vernalization period affects flowering time in three lupin species. Plant Breed. 2012, 131, 631–636. [Google Scholar] [CrossRef]
- Kroc, M.; Koczyk, G.; Święcicki, W.; Kilian, A.; Nelson, M.N. New evidence of ancestral polyploidy in the genistoid legume Lupinus angustifolius L. (narrow-leafed lupin). Theor. Appl. Genet. 2014, 127, 1237–1249. [Google Scholar] [CrossRef]
- Gladstones, J. Selection for economic characters in Lupinus angustifolius and l. Digitatus. Aust. J. Exp. Agric. 1967, 7, 360. [Google Scholar] [CrossRef]
- Kurlovich, B.S. Genetics of lupins. In Lupins: Geography, Classification, Genetic Resources and Breeding; Kurlovich, B.S., Ed.; Publishing House “Intan”: St. Petersburg, Russia, 2002; p. 468. [Google Scholar]
- Boersma, J.G.; Nelson, M.N.; Sivasithamparam, K.; Yang, H.A. Development of sequence-specific pcr markers linked to the tardus gene that reduces pod shattering in narrow-leafed lupin (Lupinus angustifolius L.). Mol. Breed. 2009, 23, 259–267. [Google Scholar] [CrossRef]
- Li, X.; Renshaw, D.; Yang, H.; Yan, G. Development of a co-dominant DNA marker tightly linked to gene tardus conferring reduced pod shattering in narrow-leafed lupin (Lupinus angustifolius L.). Euphytica 2010, 176, 49–58. [Google Scholar] [CrossRef]
- Li, X.; Buirchell, B.; Yan, G.; Yang, H. A molecular marker linked to the mollis gene conferring soft-seediness for marker-assisted selection applicable to a wide range of crosses in lupin (Lupinus angustifolius L.) breeding. Mol. Breed. 2012, 29, 361–370. [Google Scholar] [CrossRef]
- Yang, H.; Li, C.; Lam, H.-M.; Clements, J.; Yan, G.; Zhao, S. Sequencing consolidates molecular markers with plant breeding practice. Theor. Appl. Genet. 2015, 128, 779–795. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Tao, Y.; Zheng, Z.; Zhang, Q.; Zhou, G.; Sweetingham, M.W.; Howieson, J.G.; Li, C. Draft genome sequence, and a sequence-defined genetic linkage map of the legume crop species Lupinus angustifolius L. PLoS ONE 2013, 8, e64799. [Google Scholar] [CrossRef]
- Gao, L.-L.; Hane, J.K.; Kamphuis, L.G.; Foley, R.; Shi, B.-J.; Atkins, C.A.; Singh, K.B. Development of genomic resources for the narrow-leafed lupin (Lupinus angustifolius): Construction of a bacterial artificial chromosome (bac) library and bac-end sequencing. BMC Genomics 2011, 12, 521. [Google Scholar] [CrossRef]
- Hong, Y.S.; Hogan, J.R.; Wang, X.; Sarkar, A.; Sim, C.; Loftus, B.J.; Ren, C.; Huff, E.R.; Carlile, J.L.; Black, K.; et al. Construction of a BAC library and generation of BAC end sequence-tagged connectors for genome sequencing of the African malaria mosquito Anopheles gambiae. Mol. Genet. Genom. 2003, 268, 720–728. [Google Scholar]
- Kamphuis, L.G.; Hane, J.K.; Nelson, M.N.; Gao, L.; Atkins, C.A.; Singh, K.B. Transcriptome sequencing of different narrow-leafed lupin tissue types provides a comprehensive uni-gene assembly and extensive gene-based molecular markers. Plant Biotechnol. J. 2015, 13, 14–25. [Google Scholar] [CrossRef]
- Kamphuis, L.; Garg, G.; Bayer, P.; Foley, R.; Gao, L.; Nelson, M.; Hane, J.; Edwards, D.; Singh, K. Development of genomic resources for narrow-leafed lupin, including a reference genome and pan-genome and identification of candidate genes for domestication traits. In Proceedings of the International Conference on Legumes Genetics and Genomics, Siófok, Hungary, 18–22 September 2017. [Google Scholar]
- Hufnagel, B.; Marques, A.; Marande, W.; Sallet, E.; Sorriano, A.; Arribat, S.; Berges, H.; Gouzy, J.; Peret, B. Genome Sequence of White Lupin, a Model to Study Root Developmental Adaptations. In Proceedings of the 12th Congress of the International Plant Molecular Biology, Montpellier, France, 5–10 August 2018. [Google Scholar]
- Tian, L.; Peel, G.J.; Lei, Z.; Aziz, N.; Dai, X.; He, J.; Watson, B.; Zhao, P.X.; Sumner, L.W.; Dixon, R.A. Transcript and proteomic analysis of developing white lupin (Lupinus albus L.) roots. BMC Plant Biol. 2009, 9, 1. [Google Scholar] [CrossRef] [PubMed]
- O’Rourke, J.A.; Yang, S.S.; Miller, S.S.; Bucciarelli, B.; Liu, J.; Rydeen, A.; Bozsoki, Z.; Uhde-Stone, C.; Tu, Z.J.; Allan, D.; et al. An rna-seq transcriptome analysis of orthophosphate-deficient white lupin reveals novel insights into phosphorus acclimation in plants. Plant Physiol. 2013, 161, 705–724. [Google Scholar] [CrossRef] [PubMed]
- Wyrwa, K.; Książkiewicz, M.; Szczepaniak, A.; Susek, K.; Podkowiński, J.; Naganowska, B. Integration of Lupinus angustifolius L. (narrow-leafed lupin) genome maps and comparative mapping within legumes. Chromosome Res. 2016, 24, 355–378. [Google Scholar] [CrossRef] [PubMed]
- Boersma, G.J.; Pallotta, M.; Li, C.; Buirchell, B.J.; Sivasithamparam, K.; Yang, H. Construction of a genetic linkage map using MFLP and identification of molecular markers linked to domestication genes in narrow-leafed lupin (Lupinus angustifolius L.). Cell. Mol. Biol. Lett. 2005, 10, 331–344. [Google Scholar] [PubMed]
- Croxford, A.E.; Rogers, T.; Caligari, P.D.S.; Wilkinson, M.J. High-resolution melt analysis to identify and map sequence-tagged site anchor points onto linkage maps: A white lupin (Lupinus albus) map as an exemplar. New Phytol. 2008, 180, 594–607. [Google Scholar] [CrossRef] [PubMed]
- Nelson, M.; Phan, H.T.T.; Ellwood, S.R.; Moolhuijzen, P.M.; Hane, J.; Williams, A.; O’Lone, C.E.; Fosu-Nyarko, J.; Scobie, M.; Cakir, M.; et al. The first gene-based map of Lupinus angustifolius L.-location of domestication genes and conserved synteny with Medicago truncatula. Theor. Appl. Genet. 2006, 113, 225–238. [Google Scholar] [CrossRef] [PubMed]
- Książkiewicz, M.; Czyż, K.; Szczepaniak, A.; Rychel, S.; Majcherkiewicz, K.; Przysiecka, Ł.; Karlowski, W.; Wolko, B.; Naganowska, B. Comparative genomics of Lupinus angustifolius gene-rich regions: Bac library exploration, genetic mapping and cytogenetics. BMC Genomics 2013, 14, 79. [Google Scholar] [CrossRef] [PubMed]
- Książkiewicz, M.; Nazzicari, N.; Yang, H.; Nelson, M.N.; Renshaw, D.; Rychel, S.; Ferrari, B.; Carelli, M.; Tomaszewska, M.; Stawiński, S.; et al. A high-density consensus linkage map of white lupin highlights synteny with narrow-leafed lupin and provides markers tagging key agronomic traits. Sci. Rep. 2017, 7, 15335. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, M.J.; Mamidi, S.; Ahsan, R.; Kianian, S.F.; Coyne, C.J.; Hamama, A.A.; Bhardwaj, H.L. Population structure and linkage disequilibrium in Lupinus albus L. Germplasm and its implication for association mapping. Theor. Appl. Genet. 2012, 125, 517–530. [Google Scholar] [CrossRef]
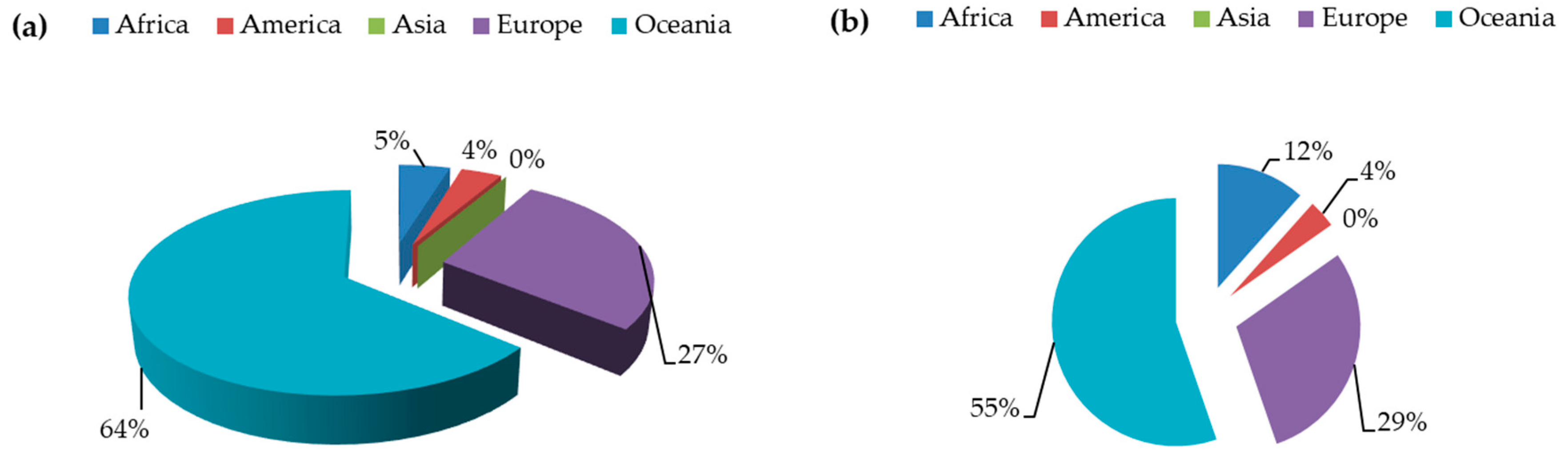
| L. albus a | L. angustifolius b | L. luteus c | L. mutabilis d | |
|---|---|---|---|---|
| Crude Protein (% of DM) | 33–47 | 31–37 | 37–38 | 32–52 |
| Crude fibre (% of DM) | 13–16 | 15–17 | 12–15 | 10 |
| Metabolized Energy (MJ/kg DM) | 13–16 | 12–13 | 10 | na |
| Oil (%) | 6–13 | 6–7 | 5–9 | 13–24 |
| Total oligosaccharides (% of DM) | 7–8 | 8–9 | na | na |
| Non-starch polysaccharides (%) | 18 | 47–51 | na | na |
| Essential amino acids (g/16 gN) | ||||
| Lys | 4.9–5.1 | 4.5–5.0 | 4.2–4.6 | 5.0–7.3 |
| Met | 0.6–0.7 | 0.6–0.7 | 0.6–0.7 | 0.4–1.4 |
| Cys | 1.8–2.1 | 1.3–1.6 | 1.8–2.5 | 1.4–1.7 |
| Leu | 7.5–8.0 | 6.0–7.6 | 6.1–7.3 | 5.7–7.8 |
| Thr | 3.1–4.0 | 3.0–3.3 | 2.6–3.2 | 3.0–4.0 |
| LOCUS | Trait | Species a | Marker | Ref. |
|---|---|---|---|---|
| Iucundus | low alkaloids | NLL | IucLi | [147] |
| pauper | low alkaloids | WL | PauperM1 | [150] |
| mollis | water-permeability of testa | NLL | MoLi | [152] |
| Ku | Early flowering—vernalization | NLL | KuHM1 | [151] |
| Ku | Early flowering—vernalization | NLL | dFTc | [154] |
| tardus | reduced shattering | NLL | TaLi | [158] |
| lentus | reduced shattering | NLL | LeM1, LeM2 | [151] |
| lentus | reduced shattering | NLL | LeLi | [159] |
| Lanr1 | anthracnose disease resistance | NLL | AntjM2 | [95] |
| PSB-res | phomopsis stem blight resistance | NLL | PhtjM4, PhtjM7 | [109] |
| Lupinus albus L. | Lupinus angustifolius L. | ||||
|---|---|---|---|---|---|
| BAC libraries | References | BAC libraries | Two centromeric BAC clones; 111, 360 clones (12× coverage); (26.9× coverage containing 57,807 genes) | [161,162,169] | |
| Genome assemblies | Genome assemblies | Draft genome sequence (609 Mb) | [145] | ||
| Genetic linkage maps | MFLP loci; HRM-STS markers | [170,171] | Genetic linkage maps | STS markers | [146] |
| Transcriptome and proteome assemblies | De Novo Transcriptome assembly, gene annotation and functional classification; EST and protein datasets | [167,168] | Transcriptome and proteome assemblies | 335 transcriptome-derived markers | [164] |
| QTL mapping | seed alkaloid content, flowering time, resistance to Phomopsis stem blight, anthracnose resistance | [93,174] | QTL mapping | ||
| Association mapping | AFLP markers for seed weight | [175] | Association mapping | ||
| Comparative mapping | Lupinus angustifolius; Medicago truncatula | [166,174] | Comparative mapping | Lotus japonicus; synteny analysis among legume species; Glycine max | [146,169,173] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abraham, E.M.; Ganopoulos, I.; Madesis, P.; Mavromatis, A.; Mylona, P.; Nianiou-Obeidat, I.; Parissi, Z.; Polidoros, A.; Tani, E.; Vlachostergios, D. The Use of Lupin as a Source of Protein in Animal Feeding: Genomic Tools and Breeding Approaches. Int. J. Mol. Sci. 2019, 20, 851. https://doi.org/10.3390/ijms20040851
Abraham EM, Ganopoulos I, Madesis P, Mavromatis A, Mylona P, Nianiou-Obeidat I, Parissi Z, Polidoros A, Tani E, Vlachostergios D. The Use of Lupin as a Source of Protein in Animal Feeding: Genomic Tools and Breeding Approaches. International Journal of Molecular Sciences. 2019; 20(4):851. https://doi.org/10.3390/ijms20040851
Chicago/Turabian StyleAbraham, Eleni M., Ioannis Ganopoulos, Panagiotis Madesis, Athanasios Mavromatis, Photini Mylona, Irini Nianiou-Obeidat, Zoi Parissi, Alexios Polidoros, Eleni Tani, and Dimitrios Vlachostergios. 2019. "The Use of Lupin as a Source of Protein in Animal Feeding: Genomic Tools and Breeding Approaches" International Journal of Molecular Sciences 20, no. 4: 851. https://doi.org/10.3390/ijms20040851
APA StyleAbraham, E. M., Ganopoulos, I., Madesis, P., Mavromatis, A., Mylona, P., Nianiou-Obeidat, I., Parissi, Z., Polidoros, A., Tani, E., & Vlachostergios, D. (2019). The Use of Lupin as a Source of Protein in Animal Feeding: Genomic Tools and Breeding Approaches. International Journal of Molecular Sciences, 20(4), 851. https://doi.org/10.3390/ijms20040851










