Metabolomic Profile of Oviductal Extracellular Vesicles across the Estrous Cycle in Cattle
Abstract
1. Introduction
2. Results
2.1. Characterization of Oviductal Extracellular Vesicles Used for Metabolomic Profiling
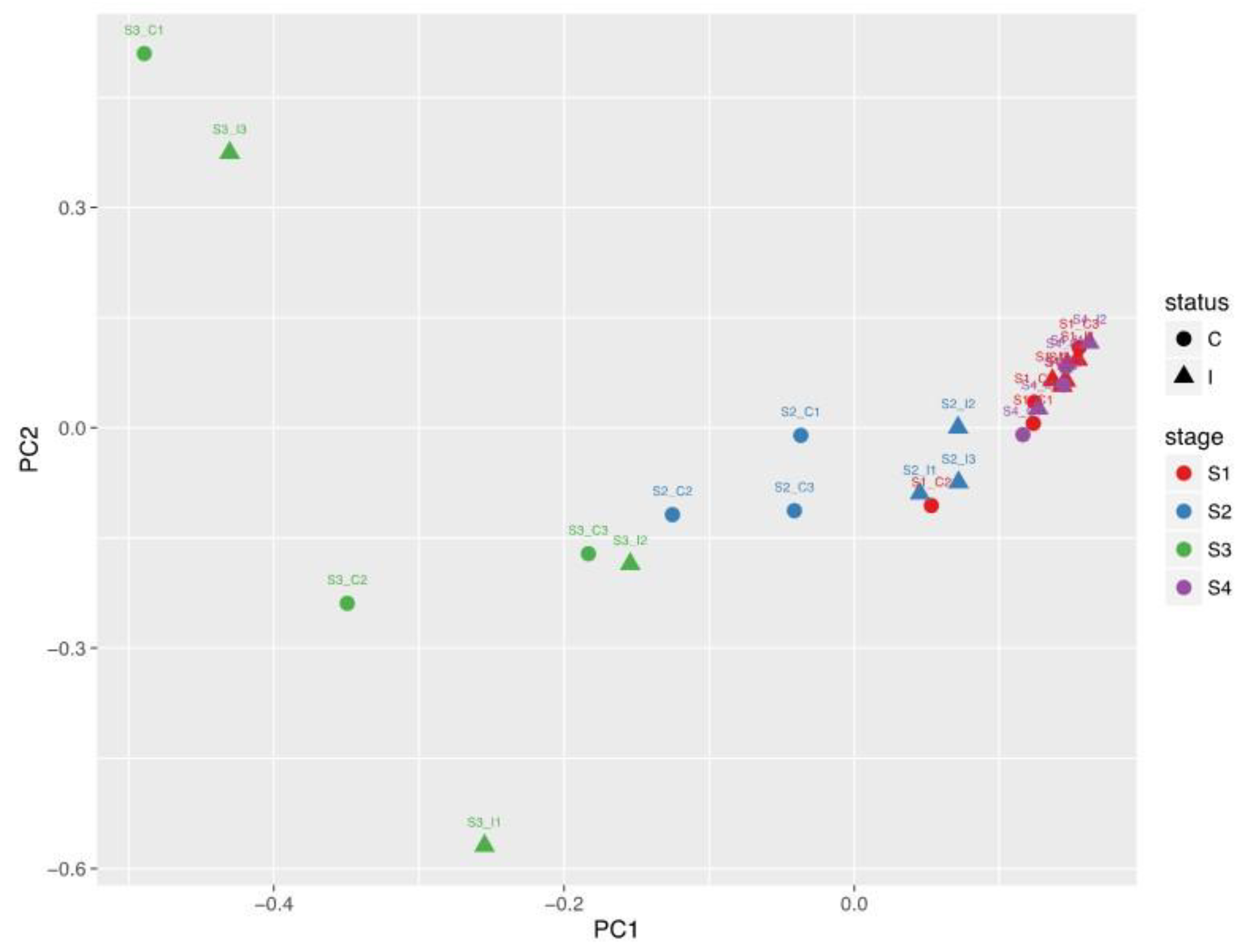
2.2. Metabolite Profile of oEVs across the Estrous Cycle
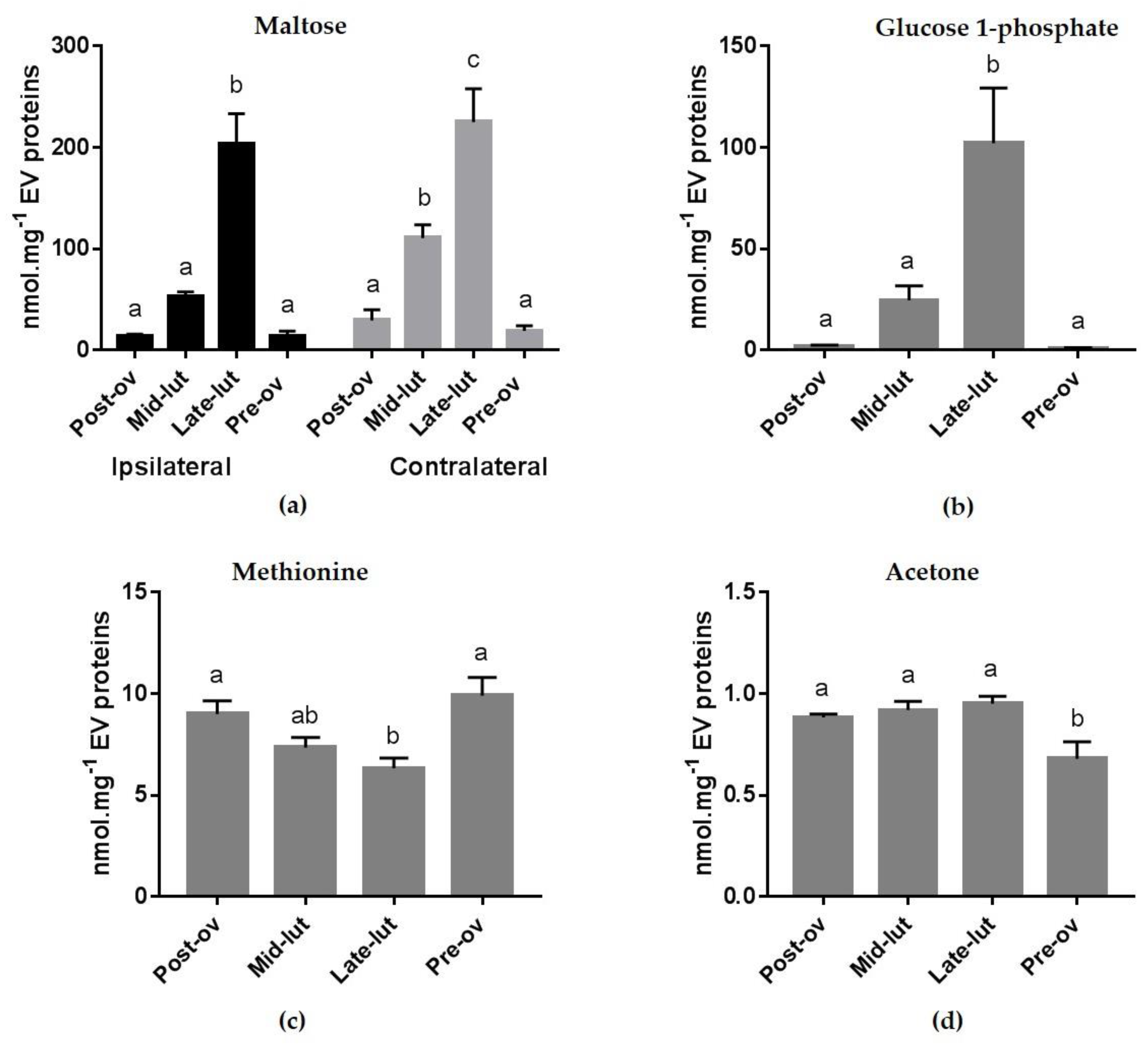
2.3. Effect of the Stage of the Estrous Cycle and Side of Ovulation on Metabolite Concentrations in oEVs
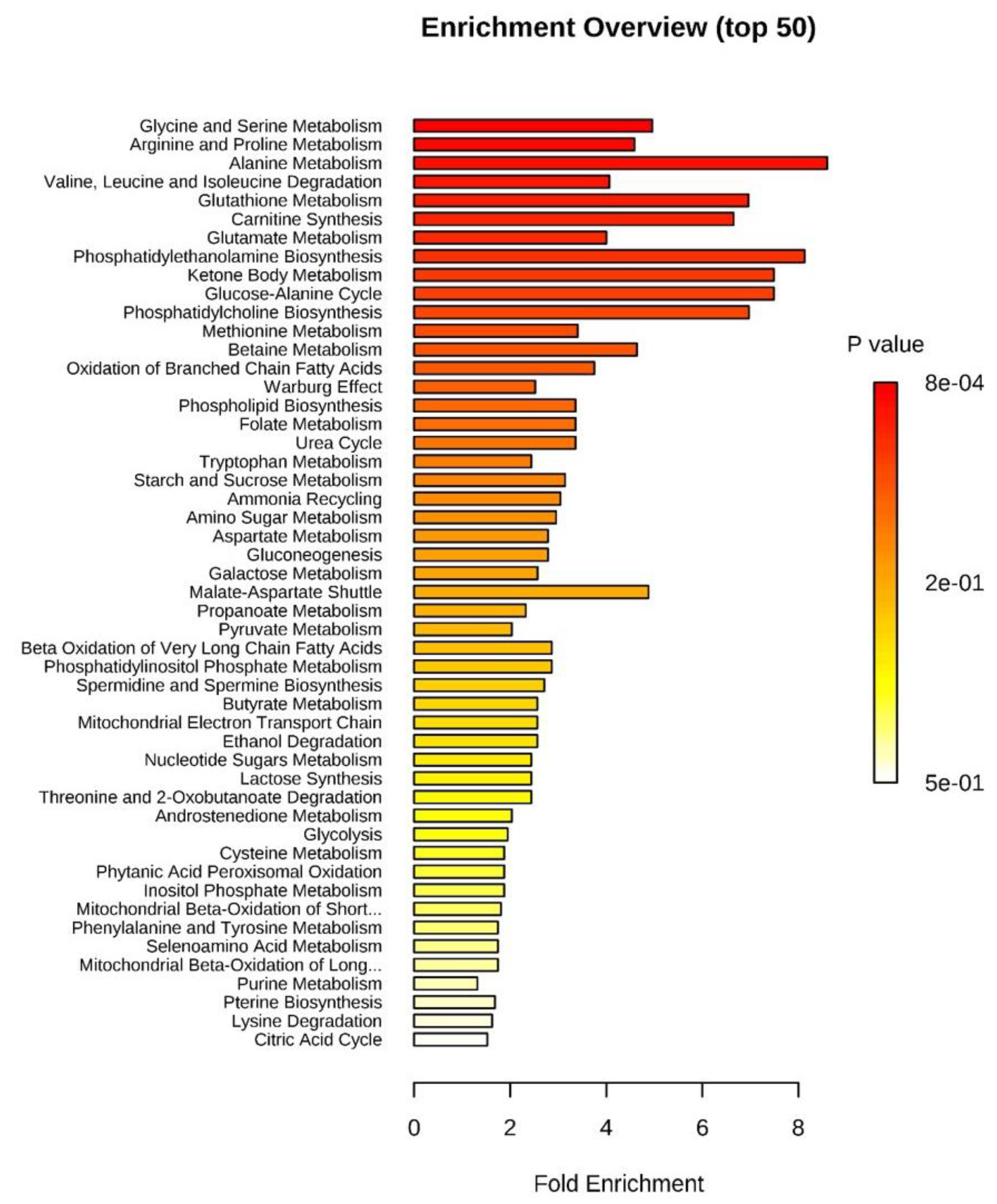
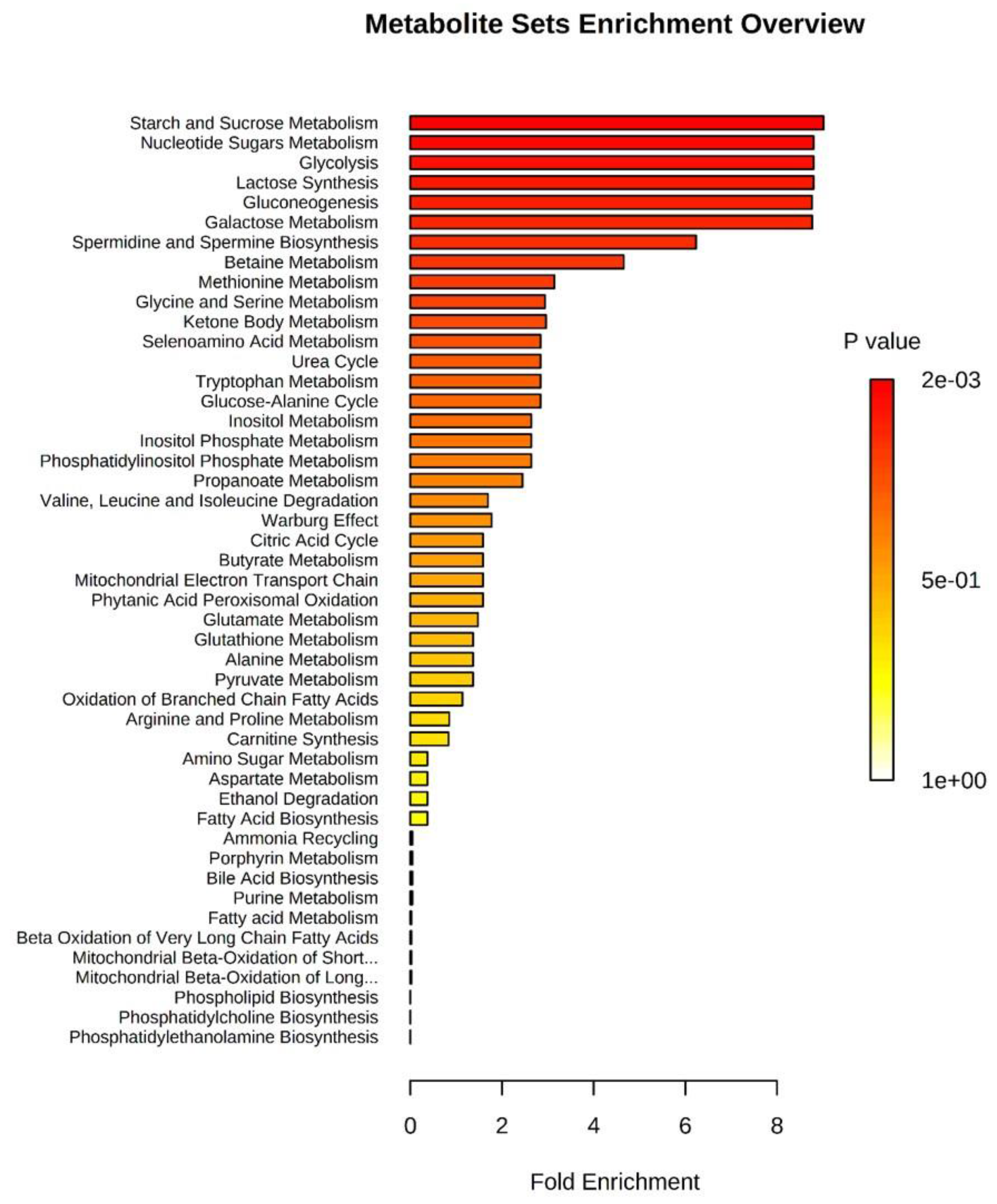
2.4. Pathways Associated with oEVs Metabolites
3. Discussion
4. Materials and Methods
4.1. Collection of Oviductal Fluids
4.2. Isolation and Characterization of oEVs
4.2.1. Preparation of Pools Oviductal Fluids and Isolation of oEVs
4.2.2. Oviductal EVs Observation by TEM
4.2.3. Oviductal EVs Characterization by Western Blotting
4.3. Proton Nuclear Magnetic Resonance Spectroscopy (1H-NMR) Analyses
4.4. Data Processing and Statistical Analysis of NMR Data
4.5. Pathway Enrichment Analyses of NMR Data
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| NMR | Nuclear magnetic resonance |
| oEVs | Oviductal extracellular vesicles |
| OF | Oviductal fluid |
References
- Yanez-Mo, M.; Siljander, P.R.; Andreu, Z.; Zavec, A.B.; Borras, F.E.; Buzas, E.I.; Buzas, K.; Casal, E.; Cappello, F.; Carvalho, J.; et al. Biological properties of extracellular vesicles and their physiological functions. J. Extracell. Vesicles 2015, 4, 27066. [Google Scholar] [CrossRef] [PubMed]
- da Silveira, J.C.; Veeramachaneni, D.N.; Winger, Q.A.; Carnevale, E.M.; Bouma, G.J. Cell-secreted vesicles in equine ovarian follicular fluid contain miRNAs and proteins: A possible new form of cell communication within the ovarian follicle. Biol. Reprod. 2012, 86, 71. [Google Scholar] [CrossRef] [PubMed]
- Ng, Y.H.; Rome, S.; Jalabert, A.; Forterre, A.; Singh, H.; Hincks, C.L.; Salamonsen, L.A. Endometrial exosomes/microvesicles in the uterine microenvironment: A new paradigm for embryo-endometrial cross talk at implantation. PLoS ONE 2013, 8, e58502. [Google Scholar] [CrossRef] [PubMed]
- Lopera-Vasquez, R.; Hamdi, M.; Maillo, V.; Gutierrez-Adan, A.; Bermejo-Alvarez, P.; Ramirez, M.A.; Yanez-Mo, M.; Rizos, D. Effect of bovine oviductal extracellular vesicles on embryo development and quality in vitro. Reproduction 2017, 153, 461–470. [Google Scholar] [CrossRef] [PubMed]
- Alminana, C.; Corbin, E.; Tsikis, G.; Alcantara-Neto, A.S.; Labas, V.; Reynaud, K.; Galio, L.; Uzbekov, R.; Garanina, A.S.; Druart, X.; et al. Oviduct extracellular vesicles protein content and their role during oviduct-embryo cross-talk. Reproduction 2017, 154, 153–168. [Google Scholar] [CrossRef] [PubMed]
- Al-Dossary, A.A.; Strehler, E.E.; Martin-Deleon, P.A. Expression and secretion of plasma membrane Ca2+-ATPase 4a (PMCA4a) during murine estrus: Association with oviductal exosomes and uptake in sperm. PLoS ONE 2013, 8, e80181. [Google Scholar] [CrossRef] [PubMed]
- Lopera-Vasquez, R.; Hamdi, M.; Fernandez-Fuertes, B.; Maillo, V.; Beltran-Brena, P.; Calle, A.; Redruello, A.; Lopez-Martin, S.; Gutierrez-Adan, A.; Yanez-Mo, M.; et al. Extracellular Vesicles from BOEC in In Vitro Embryo Development and Quality. PLoS ONE 2016, 11, e0148083. [Google Scholar] [CrossRef]
- Ferraz, M.; Carothers, A.; Dahal, R.; Noonan, M.J.; Songsasen, N. Oviductal extracellular vesicles interact with the spermatozoon’s head and mid-piece and improves its motility and fertilizing ability in the domestic cat. Sci. Rep. 2019, 9, 9484. [Google Scholar] [CrossRef]
- Alminana, C.; Tsikis, G.; Labas, V.; Uzbekov, R.; da Silveira, J.C.; Bauersachs, S.; Mermillod, P. Deciphering the oviductal extracellular vesicles content across the estrous cycle: Implications for the gametes-oviduct interactions and the environment of the potential embryo. BMC Genom. 2018, 19, 622. [Google Scholar] [CrossRef]
- Alcântara-Neto, A.; Fernandez-Rufete, M.; Corbin, E.; Tsikis, G.; Uzbekov, R.; Garanina, A.S.; Coy, P.; Alminana, C.; Mermillod, P. Oviduct fluid extracellular vesicles regulate polyspermy during porcine in vitro fertilization. Reprod. Fertil. Dev. 2019. [Google Scholar] [CrossRef]
- Qu, P.; Zhao, Y.; Wang, R.; Zhang, Y.; Li, L.; Fan, J.; Liu, E. Extracellular vesicles derived from donor oviduct fluid improved birth rates after embryo transfer in mice. Reprod. Fertil. Dev. 2019, 31, 324–332. [Google Scholar] [CrossRef] [PubMed]
- Alminana, C.; Bauersachs, S. Extracellular Vesicles in the Oviduct: Progress, Challenges and Implications for the Reproductive Success. Bioeng 2019, 6, 32. [Google Scholar] [CrossRef]
- Zebrowska, A.; Skowronek, A.; Wojakowska, A.; Widlak, P.; Pietrowska, M. Metabolome of Exosomes: Focus on Vesicles Released by Cancer Cells and Present in Human Body Fluids. Int. J. Mol. Sci. 2019, 20, 3461. [Google Scholar] [CrossRef] [PubMed]
- Hunter, R.H. Components of oviduct physiology in eutherian mammals. Biol. Rev. Camb. Philos. Soc. 2012, 287, 244–255. [Google Scholar] [CrossRef] [PubMed]
- Lamy, J.; Gatien, J.; Dubuisson, F.; Nadal-Desbarats, L.; Salvetti, P.; Mermillod, P.; Saint-Dizier, M. Metabolomic profiling of bovine oviductal fluid across the oestrous cycle using proton nuclear magnetic resonance spectroscopy. Reprod. Fertil. Dev. 2018, 30, 1021–1028. [Google Scholar] [CrossRef]
- Palomo, L.; Casal, E.; Royo, F.; Cabrera, D.; van-Liempd, S.; Falcon-Perez, J.M. Considerations for applying metabolomics to the analysis of extracellular vesicles. Front. Immunol. 2014, 5, 651. [Google Scholar] [CrossRef] [PubMed]
- Burns, G.; Brooks, K.; Wildung, M.; Navakanitworakul, R.; Christenson, L.K.; Spencer, T.E. Extracellular vesicles in luminal fluid of the ovine uterus. PLoS ONE 2014, 9, e90913. [Google Scholar] [CrossRef] [PubMed]
- Lamy, J.; Labas, V.; Harichaux, G.; Tsikis, G.; Mermillod, P.; Saint-Dizier, M. Regulation of the bovine oviductal fluid proteome. Reproduction 2016, 152, 629–644. [Google Scholar] [CrossRef]
- Lamy, J.; Liere, P.; Pianos, A.; Aprahamian, F.; Mermillod, P.; Saint-Dizier, M. Steroid hormones in bovine oviductal fluid during the estrous cycle. Theriogenology 2016, 86, 1409–1420. [Google Scholar] [CrossRef]
- Hugentobler, S.A.; Diskin, M.G.; Leese, H.J.; Humpherson, P.G.; Watson, T.; Sreenan, J.M.; Morris, D.G. Amino acids in oviduct and uterine fluid and blood plasma during the estrous cycle in the bovine. Mol. Reprod. Dev. 2007, 74, 445–454. [Google Scholar] [CrossRef]
- Hugentobler, S.A.; Humpherson, P.G.; Leese, H.J.; Sreenan, J.M.; Morris, D.G. Energy substrates in bovine oviduct and uterine fluid and blood plasma during the oestrous cycle. Mol. Reprod. Dev. 2008, 75, 496–503. [Google Scholar] [CrossRef] [PubMed]
- Kenny, D.A.; Humpherson, P.G.; Leese, H.J.; Morris, D.G.; Tomos, A.D.; Diskin, M.G.; Sreenan, J.M. Effect of elevated systemic concentrations of ammonia and urea on the metabolite and ionic composition of oviductal fluid in cattle. Biol. Reprod. 2002, 66, 1797–1804. [Google Scholar] [CrossRef] [PubMed]
- Banliat, C.; Tomas, D.; Teixeira-Gomes, A.P.; Uzbekova, S.; Guyonnet, B.; Labas, V.; Saint-Dizier, M. Stage-dependent changes in oviductal phospholipid profiles throughout the estrous cycle in cattle. Theriogenology 2019, 135, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Elhassan, Y.M.; Wu, G.; Leanez, A.C.; Tasca, R.J.; Watson, A.J.; Westhusin, M.E. Amino acid concentrations in fluids from the bovine oviduct and uterus and in KSOM-based culture media. Theriogenology 2001, 55, 1907–1918. [Google Scholar] [CrossRef]
- Condorelli, R.A.; La Vignera, S.; Bellanca, S.; Vicari, E.; Calogero, A.E. Myoinositol: Does it improve sperm mitochondrial function and sperm motility? Urology 2012, 79, 1290–1295. [Google Scholar] [CrossRef]
- Hynes, A.C.; Sreenan, J.M.; Kane, M.T. Uptake and incorporation of myo-inositol by bovine preimplantation embryos from two-cell to early blastocyst stages. Mol. Reprod. Dev. 2000, 55, 265–269. [Google Scholar] [CrossRef]
- Boni, R.; Gallo, A.; Cecchini, S. Kinetic activity, membrane mitochondrial potential, lipid peroxidation, intracellular pH and calcium of frozen/thawed bovine spermatozoa treated with metabolic enhancers. Andrology 2017, 5, 133–145. [Google Scholar] [CrossRef]
- Lim, K.T.; Jang, G.; Ko, K.H.; Lee, W.W.; Park, H.J.; Kim, J.J.; Lee, S.H.; Hwang, W.S.; Lee, B.C.; Kang, S.K. Improved in vitro bovine embryo development and increased efficiency in producing viable calves using defined media. Theriogenology 2007, 67, 293–302. [Google Scholar] [CrossRef]
- Ferramosca, A.; Zara, V. Bioenergetics of mammalian sperm capacitation. Biomed. Res. Int. 2014, 2014, 902953. [Google Scholar] [CrossRef]
- Li, S.; Winuthayanon, W. Oviduct: Roles in fertilization and early embryo development. J. Endocrinol. 2017, 232, R1–R26. [Google Scholar] [CrossRef]
- Redel, B.K.; Spate, L.D.; Lee, K.; Mao, J.; Whitworth, K.M.; Prather, R.S. Glycine supplementation in vitro enhances porcine preimplantation embryo cell number and decreases apoptosis but does not lead to live births. Mol. Reprod. Dev. 2016, 83, 246–258. [Google Scholar] [CrossRef] [PubMed]
- Herrick, J.R.; Lyons, S.M.; Greene, A.F.; Broeckling, C.D.; Schoolcraft, W.B.; Krisher, R.L. Direct and Osmolarity-Dependent Effects of Glycine on Preimplantation Bovine Embryos. PLoS ONE 2016, 11, e0159581. [Google Scholar] [CrossRef] [PubMed]
- Jain, M.; Nilsson, R.; Sharma, S.; Madhusudhan, N.; Kitami, T.; Souza, A.L.; Kafri, R.; Kirschner, M.W.; Clish, C.B.; Mootha, V.K. Metabolite profiling identifies a key role for glycine in rapid cancer cell proliferation. Science 2012, 336, 1040–1044. [Google Scholar] [CrossRef] [PubMed]
- Van Winkle, L.J.; Haghighat, N.; Campione, A.L. Glycine protects preimplantation mouse conceptuses from a detrimental effect on development of the inorganic ions in oviductal fluid. J. Exp. Zool. 1990, 253, 215–219. [Google Scholar] [CrossRef] [PubMed]
- Crow, R.R.; Kumar, S.; Varela, M.F. Maltose Chemistry and Biochemistry. In Dietery Sugars: Analysis, Function and Effects; Preedy, V.R., Ed.; Royal Society of Chemistry: London, UK, 2012. [Google Scholar]
- Dean, M. Glycogen in the uterus and fallopian tubes is an important source of glucose during early pregnancy. Biol. Reprod. 2019, 101, 297–305. [Google Scholar] [CrossRef] [PubMed]
- Gomes, H.; Dias, A.J.B.; Moraes, J.; Sobrinho Paes de Carvalho, C.; Logullo, C. Glucose-6-phosphate Metabolic Preferential Destinations in Bovine Oviduct Cells. Acta Sci. Veternariae 2010, 38, 377–383. [Google Scholar]
- Pillai, V.V.; Weber, D.M.; Phinney, B.S.; Selvaraj, V. Profiling of proteins secreted in the bovine oviduct reveals diverse functions of this luminal microenvironment. PLoS ONE 2017, 12, e0188105. [Google Scholar] [CrossRef]
- Glencross, R.G.; Munro, I.B.; Senior, B.E.; Pope, G.S. Concentrations of oestradiol-17 beta, oestrone and progesterone in jugular venous plasma of cows during the oestrous cycle and in early pregnancy. Acta Endocrinol (Copenh) 1973, 73, 374–384. [Google Scholar] [CrossRef]
- Gregoraszczuk, E.L.; Cala, M.; Witkowska, E. Glycogen distribution in porcine fallopian tube epithelium during the estrus cycle. Folia. Biol. 2000, 48, 85–90. [Google Scholar]
- Gregoire, A.T.; Hafs, H.D. Glycogen content of rabbit tubular genitalia from mating through implantation. Anat. Rec. 1971, 169, 253–258. [Google Scholar] [CrossRef][Green Version]
- Bowman, K.; Rose, J. Estradiol stimulates glycogen synthesis whereas progesterone promotes glycogen catabolism in the uterus of the American mink (Neovison vison). Anim. Sci. J. 2017, 88, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Demers, L.M.; Jacobs, R.D.; Greep, R.O. Comparative effects of ovarian steroids on glycogen metabolism of rat, rabbit and guinea pig uterine tissue. Proc. Soc. Exp. Biol. Med. 1973, 143, 1158–1163. [Google Scholar] [CrossRef] [PubMed]
- Green, C.E.; Bredl, J.; Holt, W.V.; Watson, P.F.; Fazeli, A. Carbohydrate mediation of boar sperm binding to oviductal epithelial cells in vitro. Reproduction 2001, 122, 305–315. [Google Scholar] [CrossRef] [PubMed]
- Sostaric, E.; van de Lest, C.H.; Colenbrander, B.; Gadella, B.M. Dynamics of carbohydrate affinities at the cell surface of capacitating bovine sperm cells. Biol. Reprod 2005, 72, 346–357. [Google Scholar] [CrossRef]
- Thompson, J.G.; Partridge, R.J.; Houghton, F.D.; Cox, C.I.; Leese, H.J. Oxygen uptake and carbohydrate metabolism by in vitro derived bovine embryos. J. Reprod Fertil. 1996, 106, 299–306. [Google Scholar] [CrossRef]
- Uhde, K.; van Tol, H.T.A.; Stout, T.A.E.; Roelen, B.A.J. Exposure to elevated glucose concentrations alters the metabolomic profile of bovine blastocysts. PLoS ONE 2018, 13, e0199310. [Google Scholar] [CrossRef]
- Martinez, Y.; Li, X.; Liu, G.; Bin, P.; Yan, W.; Mas, D.; Valdivie, M.; Hu, C.A.; Ren, W.; Yin, Y. The role of methionine on metabolism, oxidative stress, and diseases. Amino Acids 2017, 49, 2091–2098. [Google Scholar] [CrossRef]
- Zhang, N. Role of methionine on epigenetic modification of DNA methylation and gene expression in animals. Anim. Nutr. 2018, 4, 11–16. [Google Scholar] [CrossRef]
- Ikeda, S.; Sugimoto, M.; Kume, S. Importance of methionine metabolism in morula-to-blastocyst transition in bovine preimplantation embryos. J. Reprod. Dev. 2012, 58, 91–97. [Google Scholar] [CrossRef]
- Shojaei Saadi, H.A.; Gagne, D.; Fournier, E.; Baldoceda Baldeon, L.M.; Sirard, M.A.; Robert, C. Responses of bovine early embryos to S-adenosyl methionine supplementation in culture. Epigenomics 2016, 8, 1039–1060. [Google Scholar] [CrossRef]
- Enjalbert, F.; Nicot, M.C.; Bayourthe, C.; Moncoulon, R. Ketone bodies in milk and blood of dairy cows: Relationship between concentrations and utilization for detection of subclinical ketosis. J. Dairy Sci. 2001, 84, 583–589. [Google Scholar] [CrossRef]
- Reist, M.; Koller, A.; Busato, A.; Kupfer, U.; Blum, J.W. First ovulation and ketone body status in the early postpartum period of dairy cows. Theriogenology 2000, 54, 685–701. [Google Scholar] [CrossRef]
- Roche, J.F. The effect of nutritional management of the dairy cow on reproductive efficiency. Anim. Reprod. Sci. 2006, 96, 282–296. [Google Scholar] [CrossRef] [PubMed]
- Rico, C.; Medigue, C.; Fabre, S.; Jarrier, P.; Bontoux, M.; Clement, F.; Monniaux, D. Regulation of anti-Mullerian hormone production in the cow: A multiscale study at endocrine, ovarian, follicular, and granulosa cell levels. Biol. Reprod. 2011, 84, 560–571. [Google Scholar] [CrossRef]
- Wishart, D.S.; Feunang, Y.D.; Marcu, A.; Guo, A.C.; Liang, K.; Vazquez-Fresno, R.; Sajed, T.; Johnson, D.; Li, C.; Karu, N.; et al. HMDB 4.0: The human metabolome database for 2018. Nucleic Acids Res. 2018, 46, D608–D617. [Google Scholar] [CrossRef]
- Chong, J.; Soufan, O.; Li, C.; Caraus, I.; Li, S.; Bourque, G.; Wishart, D.S.; Xia, J. MetaboAnalyst 4.0: Towards more transparent and integrative metabolomics analysis. Nucleic Acids Res. 2018, 46, W486–W494. [Google Scholar] [CrossRef]
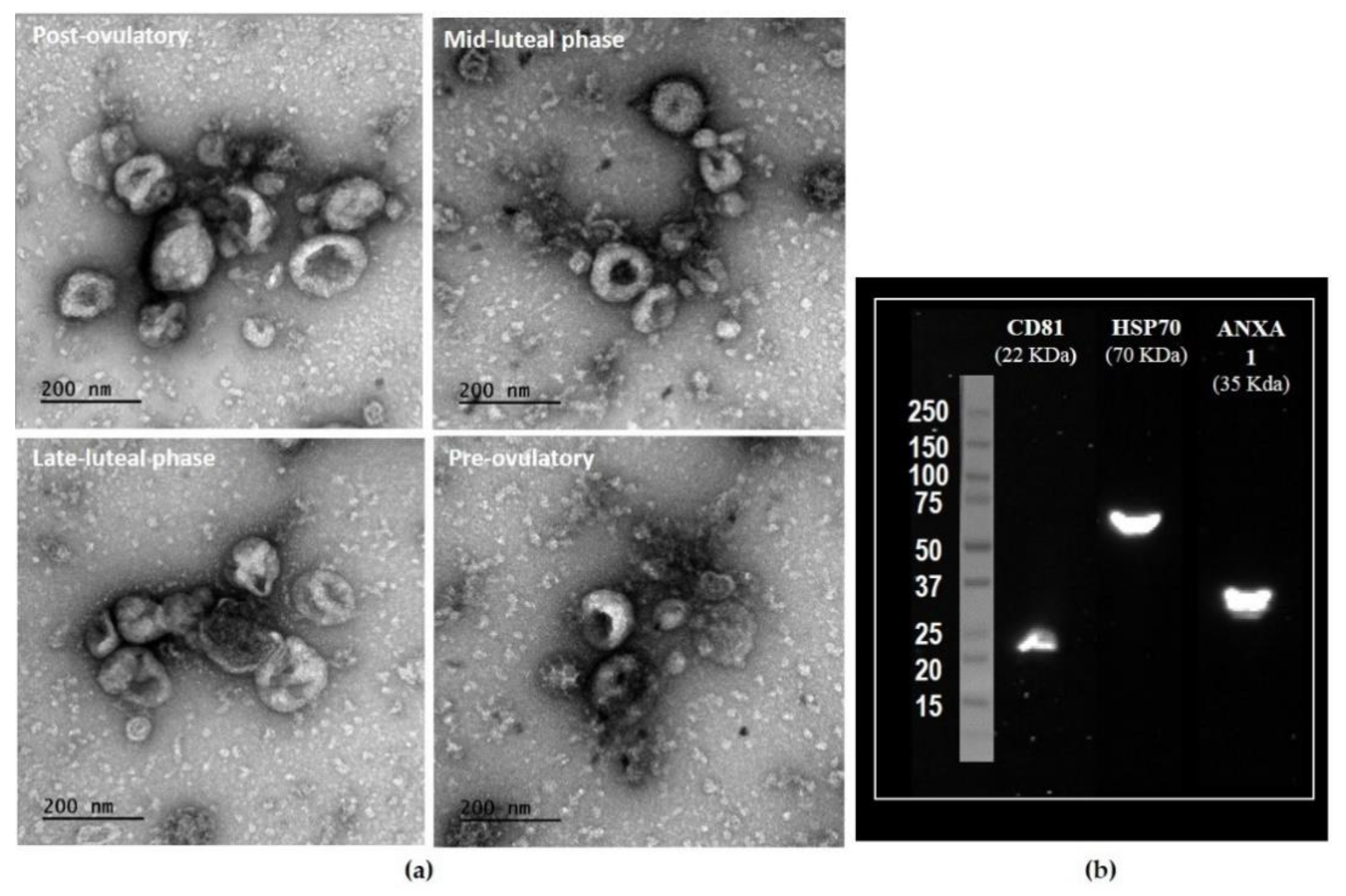
| Stage of the Cycle | No. Animals 1 | Side Relative to Ovulation | Number of OF Pools | Volume (µL) of OF before oEV Isolation | Protein Concentration (mg mL−1) |
|---|---|---|---|---|---|
| Post-ovulatory | 54 | Ipsilateral | 4 | 320–420 | 10.4–13.6 |
| Contralateral | 4 | 288–385 | 9.7–17.4 | ||
| Mid-luteal | 40 | Ipsilateral | 3 | 355–400 | 11.4–12.6 |
| Contralateral | 3 | 305–410 | 11.1–13.5 | ||
| Late-luteal | 34 | Ipsilateral | 3 | 315–470 | 9.4–10.3 |
| Contralateral | 3 | 350–470 | 13.1–15.9 | ||
| Pre-ovulatory | 40 | Ipsilateral | 3 | 450–495 | 10.0–15.2 |
| Contralateral | 3 | 500–580 | 12.0–13.4 |
| Metabolite | Chemical Taxonomy Subclass Description | Identified (I)/Quantified (Q) | Function |
|---|---|---|---|
| Valine | Amino acids, peptides, and analogues (Proteinogenic) Essential amino acids | I/Q | Involved in stress, energy and muscle metabolism |
| Threonine | Amino acids, peptides, and analogues (Proteinogenic) Essential amino acids | I | Involved in biosynthesis of proteins |
| Methionine | Amino acids, peptides, and analogues (Proteinogenic) Essential amino acids | I/Q | Required for normal growth and development |
| Leucine | Amino acids, peptides, and analogues (Proteinogenic) Essential amino acids | I | Involved in biosynthesis of proteins, stress, energy, and muscle metabolism. Stimulates insulin release |
| Isoleucine | Amino acids, peptides, and analogues (Proteinogenic) Essential amino acids | I/Q | Involved in stress, energy and muscle metabolism. |
| Proline | Amino acids, peptides, and analogues Conditionally essential amino acids (Proteinogenic) | I | Involved in biosynthesis of proteins |
| Glycine | Amino acids, peptides, and analogues Non-essential amino acids (Proteinogenic) | I/Q | Involved in the body’s production of DNA, phospholipids, and collagen, and in release of energy. |
| Alanine | Amino acids, peptides, and analogues Non-essential amino acids (Proteinogenic) | I/Q | One of the most important amino acids released by muscle, functioning as a major energy source. Regulator of glucose metabolism, lymphocyte reproduction and immunity. |
| Glutamate | Amino acids, peptides, and analogues (Non-essential alpha Amino Acids) | I | Involved in biosynthesis of proteins Role as neurotransmitter, a chemical used by nerve cells to send signals to other cells. |
| Creatine | Amino acids, peptides, and analogues (endogenous amino acid: synthesized from arginine, glycine, and methionine) | I/Q | Role in energy metabolism. Responsible for the production of ATP in skeletal muscle through the process of oxidative phosphorylation inside the mitochondria. |
| Ethanolamine | Amines | I | Widely distributed in biological tissue and is a component of lecithin. |
| Carnitine | Quaternary ammonium salts Non-proteinogenic amino acids | I/Q | Important in providing energy to muscles Described as a vitamin, an amino acid, or essential metabolite |
| Choline | Quaternary ammonium salts (Essential Vitamin) | I/Q | Considered an essential vitamin. Precursor for the neurotransmitter acetylcholine, which is involved in many functions including memory and muscle control. |
| Lactate | Alpha hydroxy acids and derivatives (Energy Substrate) | I/Q | Plays a role in several biochemical processes and is produced in the muscles during intense activity |
| Myoinositol | Alcohols and polyols | I/Q | Involved in the Inositol phosphate metabolism and the Phosphatidylinositol signaling system. |
| Glucose-1-phosphate | Carbohydrates and carbohydrate conjugates (Energy Substrate) | I/Q | Glycogenolysis produces glucose-1-phosphate and no energy. |
| Maltose | Carbohydrates and carbohydrate conjugates (Energy Substrate) | I/Q | Maltose can be broken down into two glucose molecules |
| Glucose | Carbohydrates and carbohydrate conjugates (Energy Substrate) | I | Primary source of energy |
| Succinate | Dicarboxylic acids and derivatives | I/Q | Component of the citric acid or TCA cycle and is capable of donating electrons to the electron transfer chain. |
| Formate | Carboxylic acids | I | Essential intermediary metabolite in folate-mediated one-carbon metabolism Responsible for both metabolic acidosis and disrupting mitochondrial electron transport and energy production. |
| Acetate | Carboxylic acid derivates | I/Q | Acetate in the form of acetyl CoA is used in metabolism to yield chemical energy. |
| Acetone | Carbonyl compounds | I/Q | One of the ketone bodies produced during ketoacidosis. Since ketosis develops under serious metabolic circumstances, all the mechanisms that balance or moderate the effects of ketosis enhance the chance for survival. |
| Pre-Ovulatory | Post-Ovulatory | Mid-Luteal | Late-Luteal | |||||
|---|---|---|---|---|---|---|---|---|
| Ipsi | Contra | Ipsi | Contra | Ipsi | Contra | Ipsi | Contra | |
| Acetate | 2.5 ± 0.6 | 2.8 ± 0.1 | 2.7 ± 0.2 | 2.7 ± 0.2 | 2.9 ± 0.3 | 2.8 ± 0.0 | 2.8 ± 0.2 | 2.8 ± 0.2 |
| Acetone | 0.6 ± 0.2 | 0.7 ± 0.0 | 0.9 ± 0.0 | 0.9 ± 0.0 | 1.0 ± 0.1 | 0.9 ± 0.0 | 1.0 ± 0.1 | 0.9 ± 0.0 |
| Alanine | 16.7 ± 3.7 | 18.1 ± 0.9 | 15.5 ± 1.6 | 14.9 ± 1.4 | 15.5 ± 2.3 | 13.8 ± 0.3 | 13.4 ± 0.9 | 13.8 ± 1.6 |
| Carnitine | 4.8 ± 0.8 | 6.1 ± 0.2 | 4.3 ± 0.4 | 4.9 ± 0.6 | 5.5 ± 0.6 | 5.7 ± 0.9 | 5.5 ± 0.8 | 5.6 ± 0.8 |
| Choline | 8.8 ± 1.6 | 9.4 ± 0.5 | 10.9 ± 0.9 | 9.7 ± 0.8 | 10.3 ± 1.7 | 10.2 ± 0.4 | 8.6 ± 0.4 | 9.5 ± 1.2 |
| Creatine | 7.8 ± 1.8 | 8.4 ± 0.4 | 7.7 ± 0.6 | 7.8 ± 0.6 | 7.9 ± 0.9 | 7.9 ± 0.5 | 7.7 ± 0.6 | 7.7 ± 0.9 |
| Glucose-1-phosphate | 1.1 ± 0.6 | 0.7 ± 0.5 | 0.8 ± 0.5 | 3.1 ± 1.5 | 9.8 ± 2.1 | 39.5 ± 5.7 | 87.2 ± 41.8 | 117.1 ± 1.4 |
| Glycine | 25.2 ± 5.0 | 31.4 ± 5.7 | 30.8 ± 3.1 | 25.0 ± 2.1 | 39.3 ± 6.4 | 26.5 ± 0.9 | 27.5 ± 1.8 | 25.8 ± 4.2 |
| Isoleucine | 2.6 ± 0.5 | 3.2 ± 0.2 | 2.8 ± 0.3 | 2.6 ± 0.3 | 2.7 ± 0.3 | 2.5 ± 0.0 | 2.5 ± 0.4 | 2.4 ± 0.4 |
| Lactate | 33.0 ± 6.4 | 37.7 ± 2.4 | 36.7 ± 3.1 | 36.9 ± 2.5 | 43.0 ± 4.1 | 41.9 ± 2.7 | 43.3 ± 6.9 | 43.7 ± 6.1 |
| Maltose | 13.9 ± 5.0 | 19.1 ± 5.0 | 14.1 ± 1.7 | 29.3 ± 10.6 | 53.2 ± 4.3 | 110.5 ± 13.1 | 203.7 ± 9.7 | 225.1 ± 32.9 |
| Methionine | 10.0 ± 1.9 | 9.8 ± 0.6 | 8.7 ± 1.0 | 9.2 ± 1.0 | 7.3 ± 0.9 | 7.4 ± 0.6 | 6.2 ± 0.8 | 6.4 ± 0.8 |
| Myoinositol | 37.6 ± 7.9 | 41.5 ± 2.2 | 43.8 ± 4.2 | 44.4 ± 4.2 | 50.9 ± 5.5 | 51.5 ± 3.1 | 47.8 ± 5.7 | 50.4 ± 6.2 |
| Succinate | 0.7 ± 0.2 | 0.7 ± 0.0 | 0.9 ± 0.1 | 0.8 ± 0.1 | 1.1 ± 0.1 | 0.9 ± 0.1 | 1.0 ± 0.2 | 0.8 ± 0.2 |
| Valine | 3.2 ± 0.8 | 3.8 ± 0.2 | 3.3 ± 0.4 | 3.3 ± 0.4 | 3.2 ± 0.5 | 2.9 ± 0.0 | 2.7 ± 0.2 | 2.7 ± 0.2 |
| Pathway Associated Metabolite Sets 1 | Implicated Metabolites 2 | Total 3 | Hits 4 | Raw p 5 | Holm p 6 | FDR 7 |
|---|---|---|---|---|---|---|
| Glycine and serine metabolism | Creatine, Glycine, L-glutamic acid, L-alanine, L-threonine, L-methionine | 59 | 6 | 7.82 × 10−4 | 0.0766 | 0.0766 |
| Arginine and Proline Metabolism | Creatine, Glycine, L-glutamic acid, L-proline, Succinic acid | 53 | 5 | 0.00332 | 0.322 | 0.137 |
| Alanine metabolism | Glycine, L-glutamic acid, L-alanine | 17 | 3 | 0.00421 | 0.404 | 0.137 |
| Valine, Leucine and Isoleucine degradation | L-glutamic acid, L-isoleucine, Succinic acid, L-leucine, L-valine | 60 | 5 | 0.00575 | 0.546 | 0.141 |
| Glutathione Metabolism | Glycine, L-glutamic acid, L-alanine | 21 | 3 | 0.0078 | 0.734 | 0.146 |
| Carnitine Synthesis | L-carnitine, Glycine, Succinic acid | 22 | 3 | 0.00892 | 0.829 | 0.146 |
| Glutamate Metabolism | Glycine, L-glutamic acid, L-alanine, Succinic acid | 49 | 4 | 0.0152 | 1 | 0.212 |
| Phosphatidylethanolamine biosynthesis | Choline, Ethanolamine | 12 | 2 | 0.0234 | 1 | 0.267 |
| Ketone Body Metabolism | Succinic acid, Acetone | 13 | 2 | 0.0273 | 1 | 0.267 |
| Glucose-Alanine Cycle | L-glutamic acid, L-alanine | 13 | 2 | 0.0273 | 1 | 0.267 |
| Phosphatidylcholine biosynthesis | Choline, Ethanolamine | 14 | 2 | 0.0314 | 1 | 0.28 |
| Pathway Associated Metabolite Sets 1 | Implicated Metabolites 2 | Total 3 | Hits 4 | Raw p 5 | Holm p 6 | FDR 7 |
|---|---|---|---|---|---|---|
| Starch and sucrose metabolism | D-maltose, Glucose-1-phosphate | 31 | 2 | 3.21 × 10−5 | 0.0015 | 6.835 × 10−4 |
| Nucleotide sugars metabolism | Glucose-1-phosphate | 20 | 1 | 8.651 × 10−5 | 0.0039 | 6.835 × 10−4 |
| Glycolysis | Glucose-1-phosphate | 25 | 1 | 8.651 × 10−5 | 0.0039 | 6.835 × 10−4 |
| Lactose synthesis | Glucose-1-phosphate | 20 | 1 | 8.651 × 10−5 | 0.0039 | 6.835 × 10−4 |
| Gluconeogenesis | Glucose-1-phosphate, L-lactic acid | 35 | 2 | 8.662 × 10−5 | 0.0039 | 6.835 × 10−4 |
| Galactose metabolism | Myoinositol, Glucose-1-phosphate | 38 | 2 | 8.726 × 10−5 | 0.1933 | 6.835 × 10−4 |
| Spermidine and spermine biosynthesis | L-methionine | 18 | 1 | 0.0047 | 0.4906 | 0.032 |
| Betaine metabolism | Choline, L-methionine | 21 | 2 | 0.0123 | 1 | 0.072 |
| Methionine metabolism | Choline, Glycine, L-methionine | 43 | 3 | 0.0438 | 1 | 1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gatien, J.; Mermillod, P.; Tsikis, G.; Bernardi, O.; Janati Idrissi, S.; Uzbekov, R.; Le Bourhis, D.; Salvetti, P.; Almiñana, C.; Saint-Dizier, M. Metabolomic Profile of Oviductal Extracellular Vesicles across the Estrous Cycle in Cattle. Int. J. Mol. Sci. 2019, 20, 6339. https://doi.org/10.3390/ijms20246339
Gatien J, Mermillod P, Tsikis G, Bernardi O, Janati Idrissi S, Uzbekov R, Le Bourhis D, Salvetti P, Almiñana C, Saint-Dizier M. Metabolomic Profile of Oviductal Extracellular Vesicles across the Estrous Cycle in Cattle. International Journal of Molecular Sciences. 2019; 20(24):6339. https://doi.org/10.3390/ijms20246339
Chicago/Turabian StyleGatien, Julie, Pascal Mermillod, Guillaume Tsikis, Ophélie Bernardi, Sarah Janati Idrissi, Rustem Uzbekov, Daniel Le Bourhis, Pascal Salvetti, Carmen Almiñana, and Marie Saint-Dizier. 2019. "Metabolomic Profile of Oviductal Extracellular Vesicles across the Estrous Cycle in Cattle" International Journal of Molecular Sciences 20, no. 24: 6339. https://doi.org/10.3390/ijms20246339
APA StyleGatien, J., Mermillod, P., Tsikis, G., Bernardi, O., Janati Idrissi, S., Uzbekov, R., Le Bourhis, D., Salvetti, P., Almiñana, C., & Saint-Dizier, M. (2019). Metabolomic Profile of Oviductal Extracellular Vesicles across the Estrous Cycle in Cattle. International Journal of Molecular Sciences, 20(24), 6339. https://doi.org/10.3390/ijms20246339






