Astragaloside IV Protects Against Oxidative Stress in Calf Small Intestine Epithelial Cells via NFE2L2-Antioxidant Response Element Signaling
Abstract
1. Introduction
2. Results
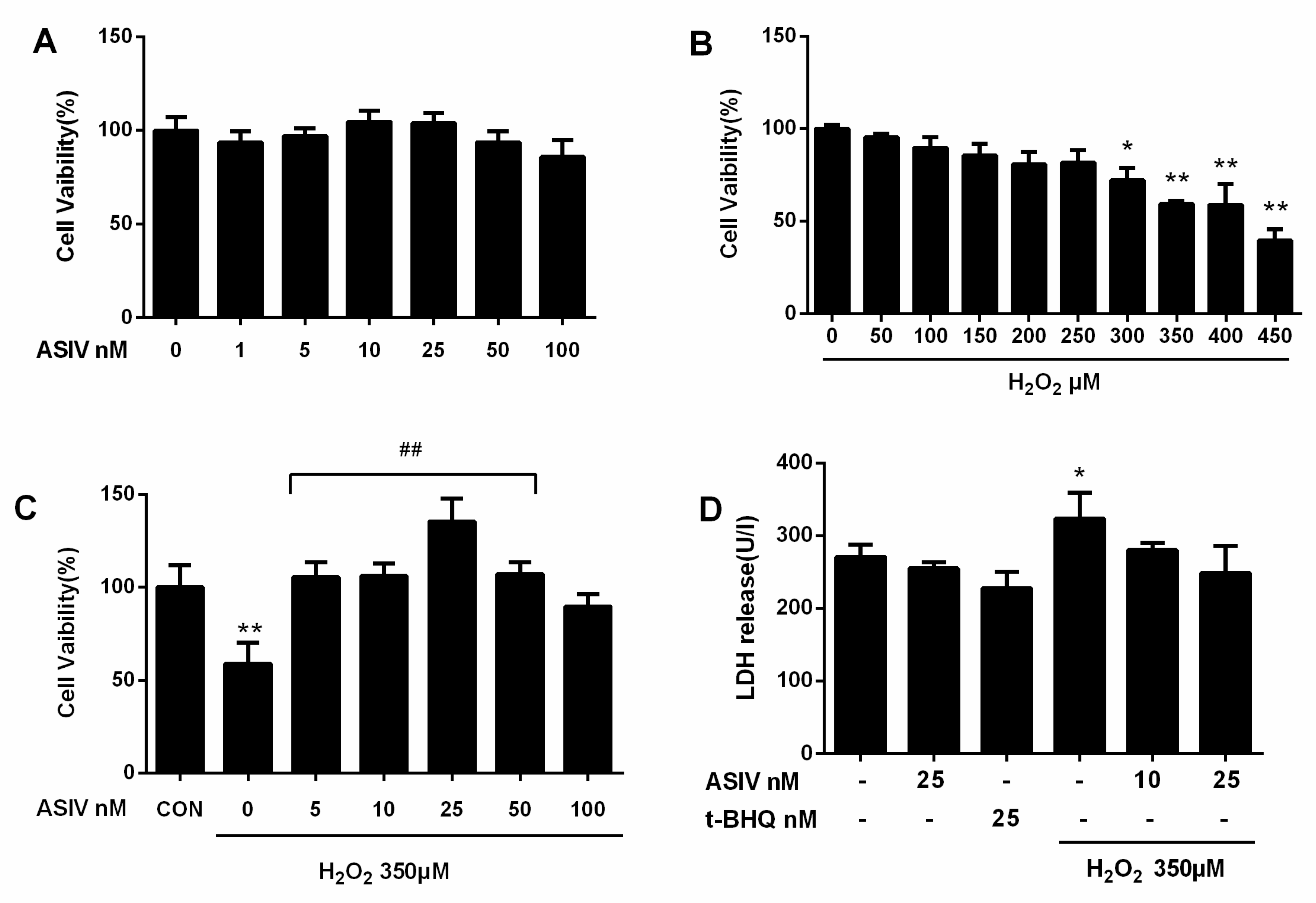
2.1. ASIV Inhibits H2O2-Induced Cell Cytotoxicity in Calf Small Intestine Epithelial Cells
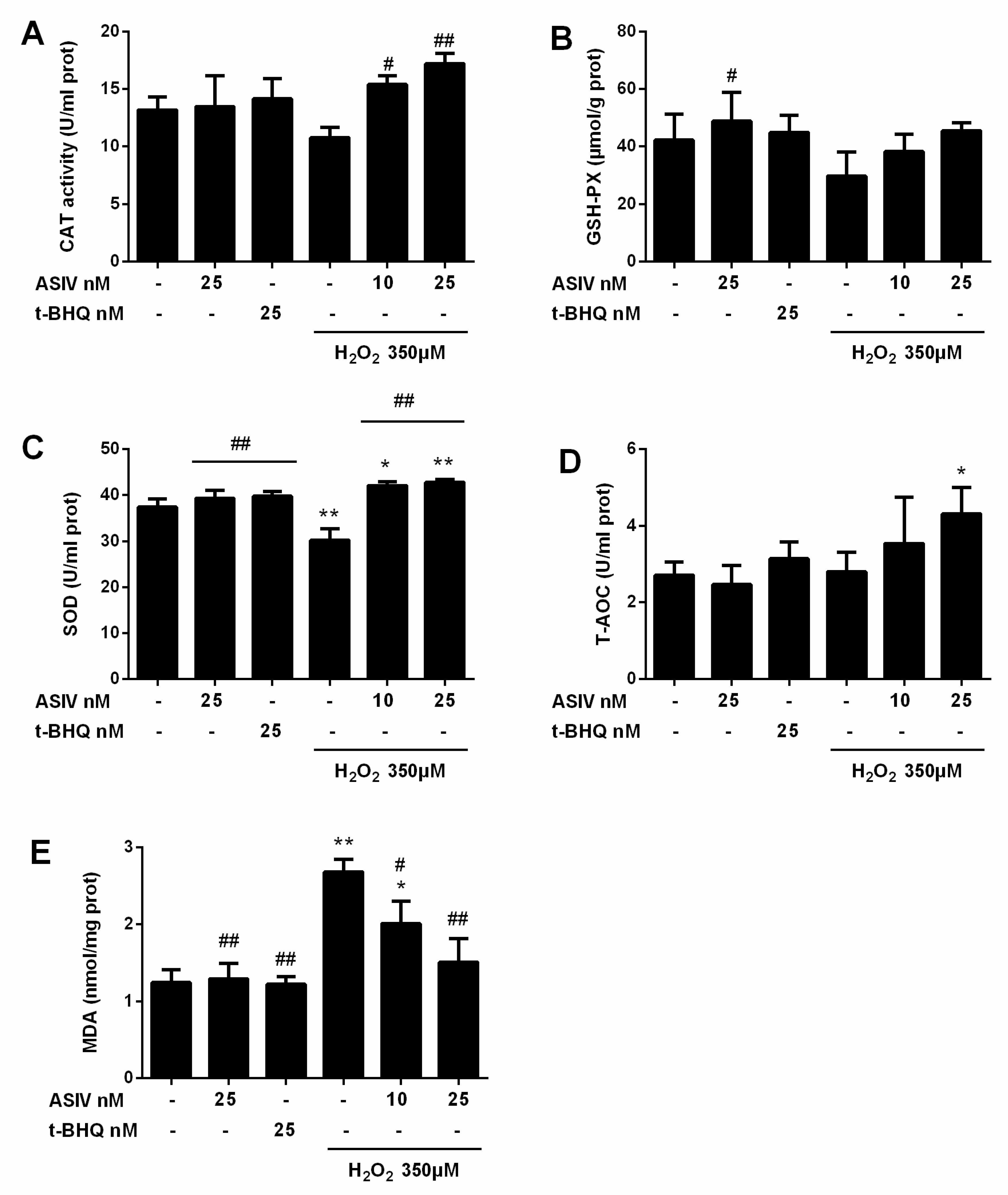
2.2. ASIV Blocks H2O2-Induced Oxidative Stress Injury in Calf Small Intestine Epithelial Cells
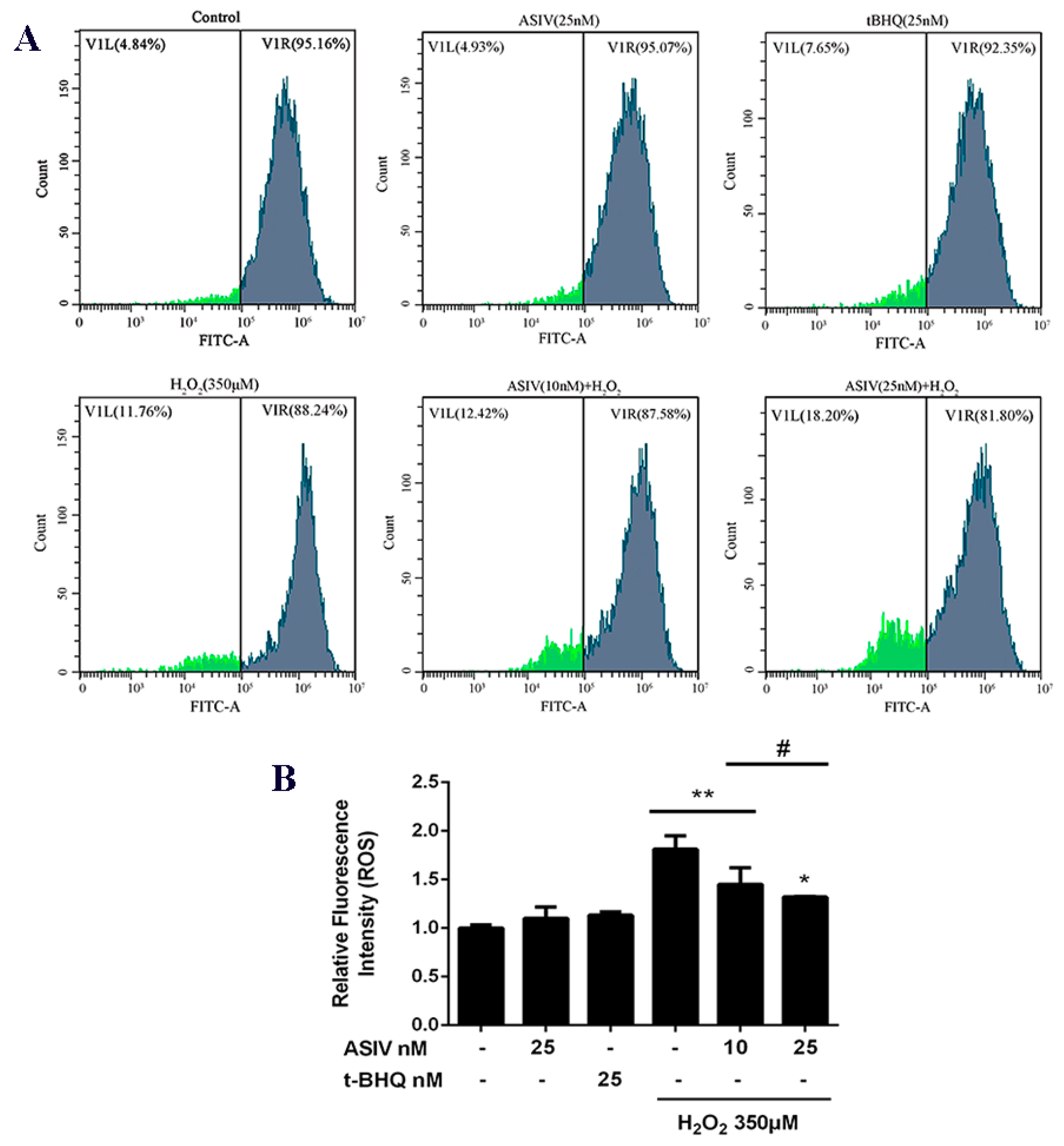
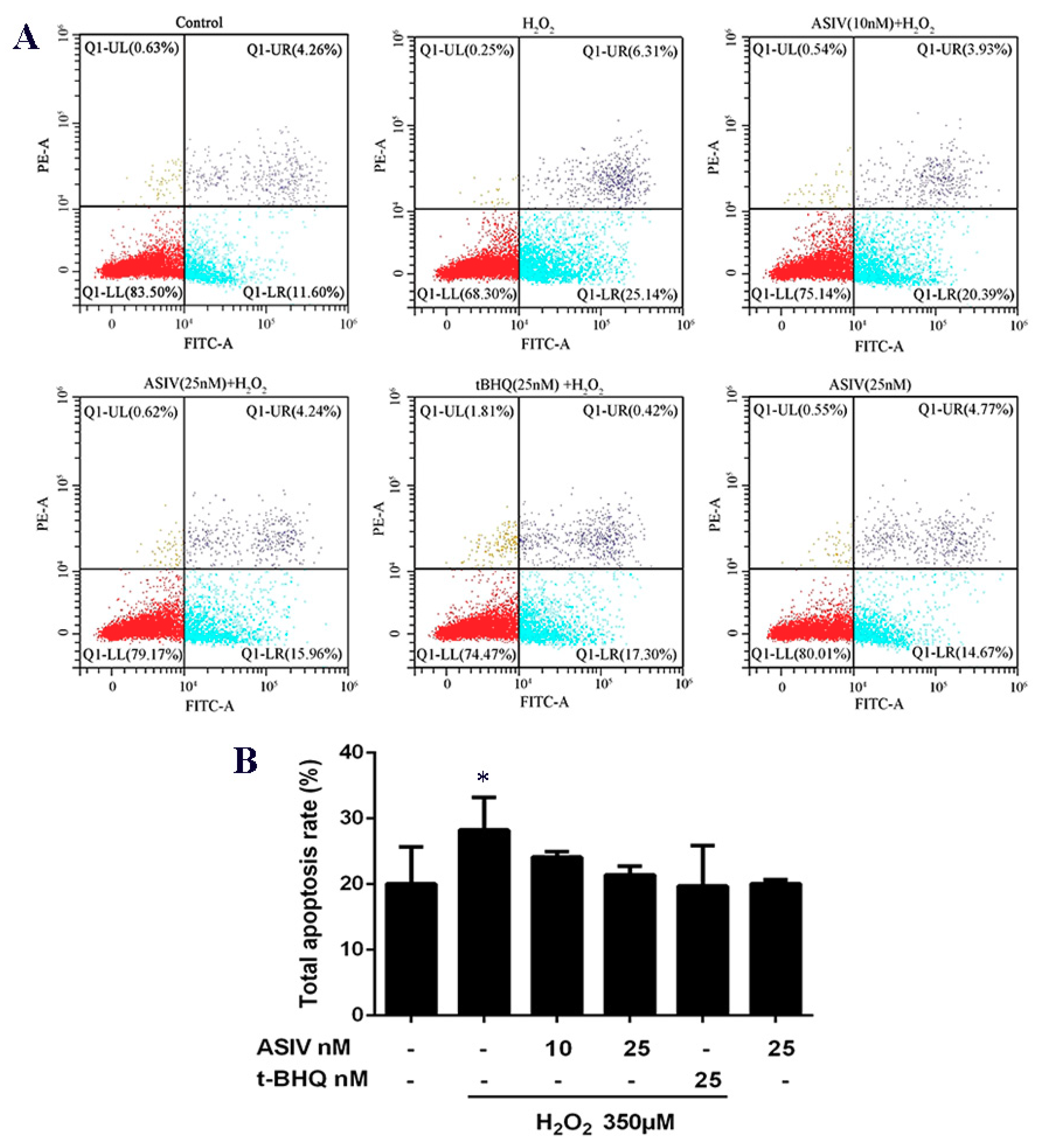
2.3. ASIV Decreases H2O2-Induced Apoptosis in Calf Small Intestine Epithelial Cells
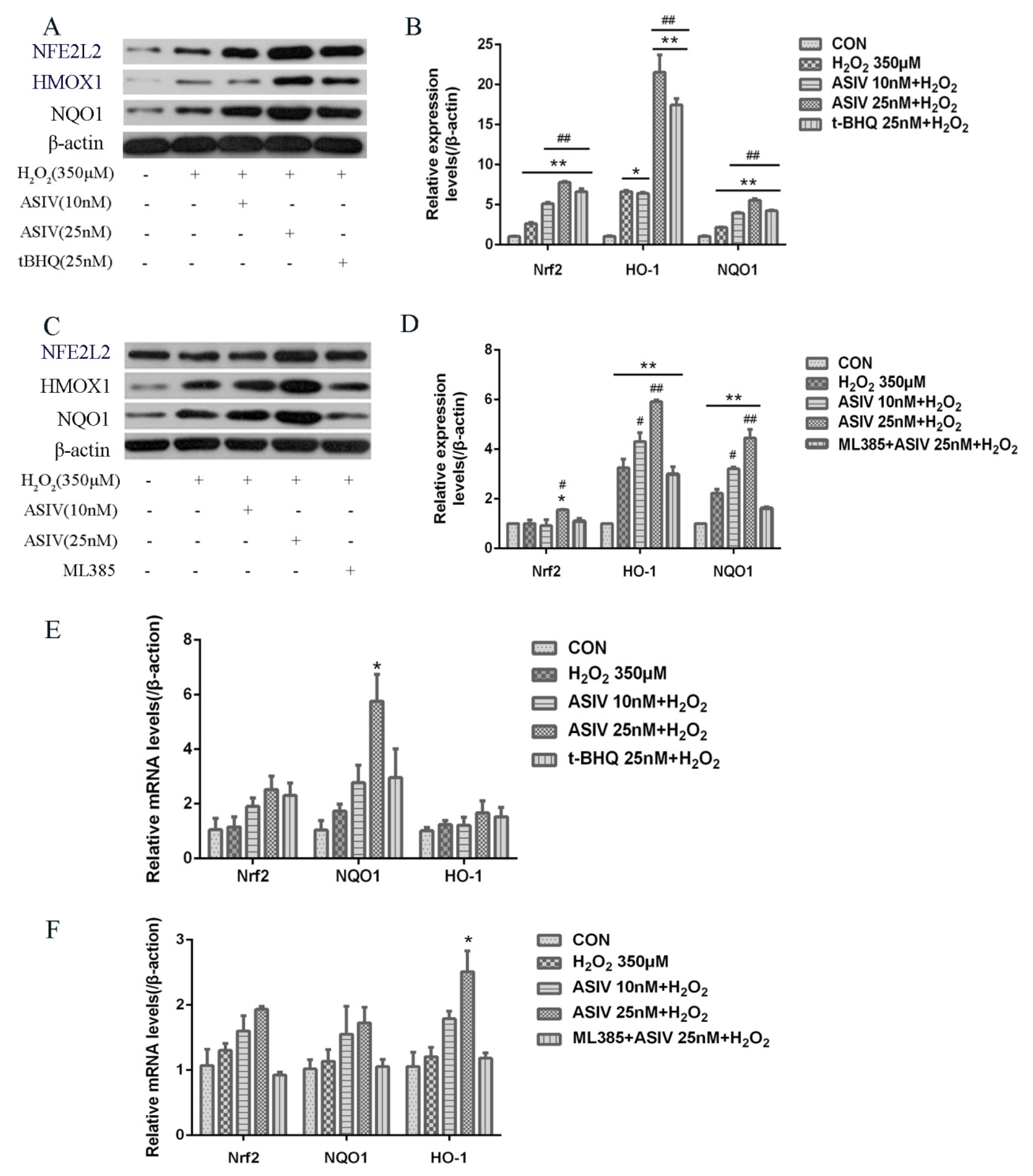
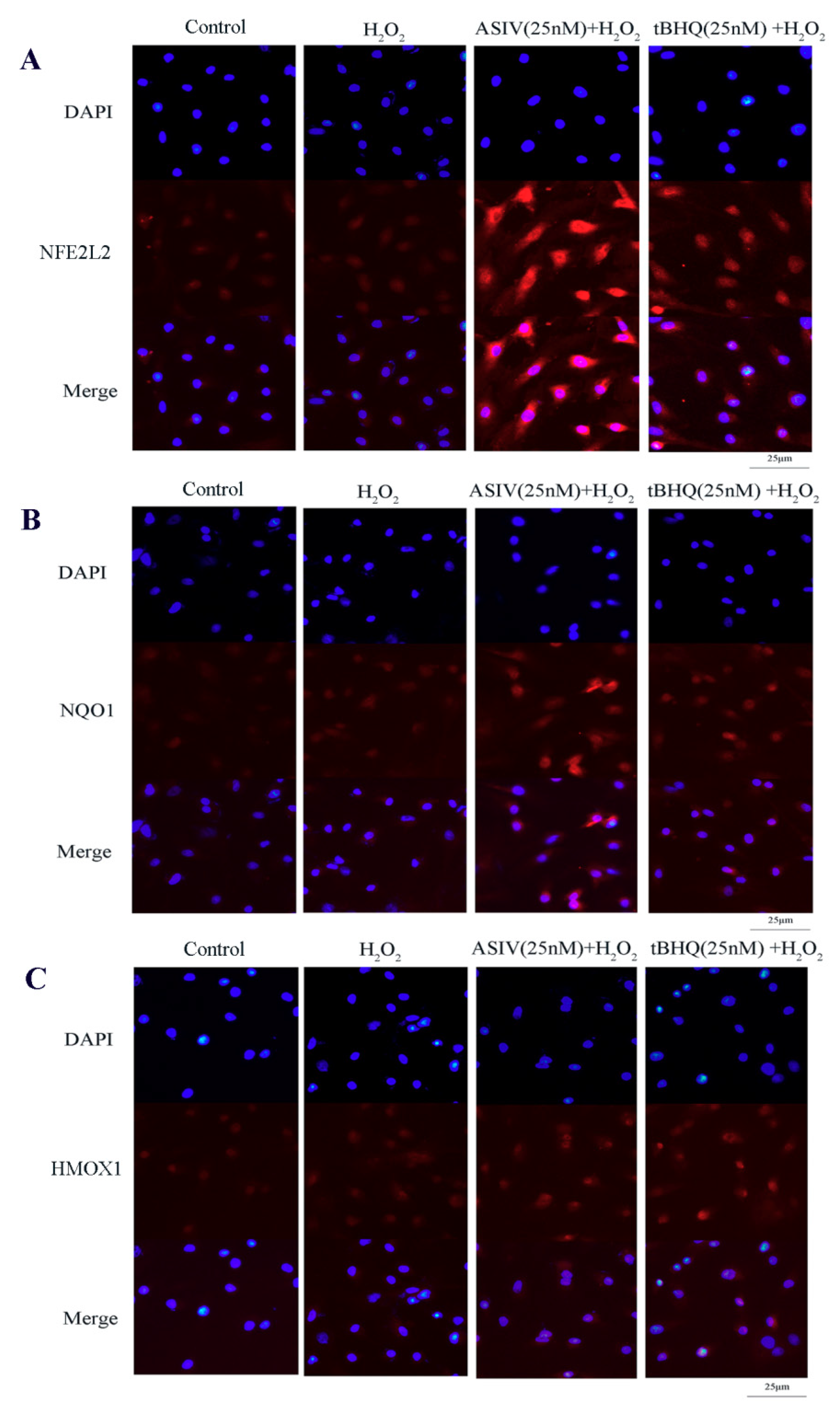
2.4. ASIV Activates NFE2L2-ARE Antioxidative Signaling in Calf Small Intestine Epithelial Cells
3. Discussion
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Cell Culture and Treatments
4.3. Cell Viability and LDH Leakage Assays
4.4. Measurement of Intracellular ROS Levels
4.5. Apoptosis Analysis
4.6. Measurement of the Intracellular GSH-Px, CAT, SOD, T-AOC, and MDA Levels
4.7. RNA Extraction and qRT-PCR Analysis
4.8. NFE2L2, NQO1, and HMOX1 Immunofluorescence
4.9. Western Blot Analysis
4.10. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ARE | antioxidant responsive element |
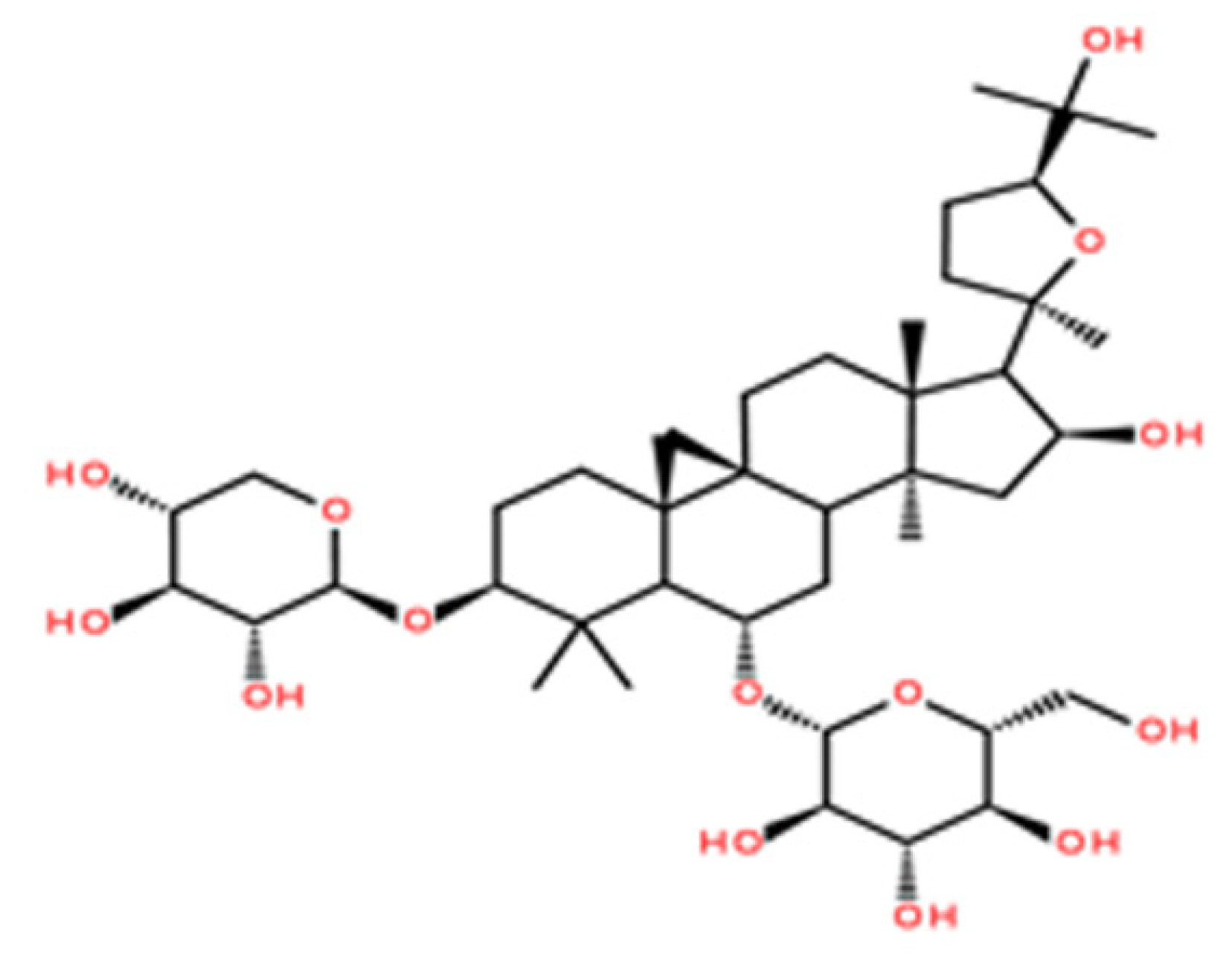
| ASIV | Astragaloside IV |
| CAT | Catalase |
| CCK8 | Cell counting kit 8 |
| DAPI | 4′,6-diamidino-2-phenylindole |
| DCFH-DA | 2′,7′-dichlorodihydrofluorescein diacetate |
| NFE2L2 | nuclear factor erythroid 2-related factor 2 |
| NQO1 | NAD(P)H quinone dehydrogenase 1 |
| O2 | molecular oxygen |
| qRT-PCR | quantitative reverse transcription-polymerase chain reaction |
| ROS | reactive oxygen species |
| SOD | superoxide dismutase |
| T-AOC | total antioxidant capacity |
| tBHQ | tertiary butylhydroquinone |
References
- Wijtten, P.J.A.; van der Meulen, J.; Verstegen, M.W.A. Intestinal barrier function and absorption in pigs after weaning: A review. Br. J. Nutr. 2011, 105, 967–981. [Google Scholar] [CrossRef] [PubMed]
- Xing, S.; Zhang, B.L.; Lin, M.; Li, J.L.; Zhang, L.; Gao, F.; Zhou, G.H. Effects of alanyl-glutamine supplementation on the small intestinal mucosa barrier in weaned piglets. Can. J. Anim. Sci. 2017, 30, 236–245. [Google Scholar] [CrossRef] [PubMed]
- Rao, R.K.; Chaudhry, K. Glutamine protects gi epithelial tight junctions. In Glutamine in Clinical Nutrition; Humana Press: New York, NY, USA, 2015; pp. 323–337. [Google Scholar]
- Zhuang, S.; Yu, R.; Zhong, J.; Liu, P.; Liu, Z. Rhein from rheum rhabarbarum inhibits hydrogen-peroxide-induced oxidative stress in intestinal epithelial cells partly through PI3K/Akt-mediated Nrf2/HO-1 pathways. J. Agric. Food Chem. 2019, 67, 2519–2529. [Google Scholar] [CrossRef] [PubMed]
- Tabei, Y.; Murotomi, K.; Umeno, A.; Horie, M.; Tsujino, Y.; Masutani, B.; Yoshida, Y.; Nakajima, Y. Antioxidant properties of 5-hydroxy-4-phenyl-butenolide via activation of Nrf2/ARE signaling pathway. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2017, 107, 129–137. [Google Scholar] [CrossRef]
- Song, Z.; Tong, G.; Xiao, K.; Jiao, L.F.; Ke, Y.; Hu, C. L-cysteine protects intestinal integrity, attenuates intestinal inflammation and oxidant stress, and modulates NF-κB and Nrf2 pathways in weaned piglets after LPS challenge. Innate Immun. 2016, 22, 152–161. [Google Scholar] [CrossRef] [PubMed]
- Mitsuoka, H.; Schmid-Schonbein, G.W. Mechanisms for blockade of in vivo activator production in the ischemic intestine and multi-organ failure. Shock 2000, 14, 522–527. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chen, Q.; Zhang, S.M.; Liu, H.Y.; Li, Y.Q. Protective effect of glutathione against oxidative injury in intestinal epithelial cells of piglet in vitro. Anim. Biol. 2009, 59, 263–272. [Google Scholar] [CrossRef]
- Wang, Y.; Wu, Y.; Wang, Y.; Fu, A.; Gong, L.; Li, W.; Li, Y. Bacillus amyloliquefaciens SC06 alleviates the oxidative stress of IPEC-1 via modulating Nrf2/Keap1 signaling pathway and decreasing ROS production. Appl. Microbiol. Biotechnol. 2017, 101, 3015–3026. [Google Scholar] [CrossRef]
- Ren, H.; Meng, Q.; Yepuri, N.; Du, X.; Sarpong, J.O.; Cooney, R.N. Protective effects of glutathione on oxidative injury induced by hydrogen peroxide in intestinal epithelial cells. J. Surg. Res. 2018, 222, 39–47. [Google Scholar] [CrossRef]
- Espinosa-Diez, C.; Miguel, V.; Mennerich, D.; Kietzmann, T.; Sanchez-Perez, P.; Cadenas, S.; Lamas, S. Antioxidant responses and cellular adjustments to oxidative stress. Redox Biol. 2015, 6, 183–197. [Google Scholar] [CrossRef]
- Del Río, L.A.; Corpas, F.J.; Sandalio, L.M.; Palma, J.M.; Gόmez, M.; Barroso, J.B. Reactive oxygen species, antioxidant systems and nitric oxide in peroxisomes. J. Exp. Bot. 2002, 53, 1255–1272. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.Y.; Dou, Y.X.; Luo, D.D.; Zhang, Z.B.; Li, C.L.; Zeng, H.F.; Su, Z.R.; Xie, J.H.; Lai, X.P.; Li, Y.C. Beta-Patchoulene from patchouli oil protects against LPS-induced acute lung injury via suppressing NF-kappaB and activating Nrf2 pathways. Int. Immunopharmacol. 2017, 50, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Lu, Y.; Chen, Y.; Cheng, J. The role of Nrf2 in oxidative stress-induced endothelial injuries. J. Endocrinol. 2015, 225, R83–R99. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.D. Mechanistic studies of the Nrf2-Keap1 signaling pathway. Drug Metab. Rev. 2006, 38, 769–789. [Google Scholar] [CrossRef]
- Zhu, C.; Dong, Y.; Liu, H.; Ren, H.; Cui, Z. Hesperetin protects against H2O2-triggered oxidative damage via upregulation of the Keap1-Nrf2/HO-1 signal pathway in ARPE-19 cells. Biomed. Pharmacother. 2017, 88, 124–133. [Google Scholar] [CrossRef]
- Zhang, W.J.; Hufnagl, P.; Binder, B.R.; Wojta, J. Antiinflammatory activity of astragaloside IV is mediated by inhibition of NF-kappaB activation and adhesion molecule expression. Thromb. Haemost. 2003, 90, 904–914. [Google Scholar] [CrossRef]
- Ren, S.; Zhang, H.; Mu, Y.; Sun, M.; Liu, P. Pharmacological effects of Astragaloside IV: A literature review. J. Tradit. Chin. Med. 2013, 33, 413–416. [Google Scholar] [CrossRef]
- Li, L.; Hou, X.; Xu, R.; Liu, C.; Tu, M. Research review on the pharmacological effects of astragaloside IV. Fundam. Clin. Pharmacol. 2017, 31, 17–36. [Google Scholar] [CrossRef]
- Li, H.; Wang, P.; Huang, F.; Jin, J.; Wu, H.; Zhang, B.; Wang, Z.; Shi, H.; Wu, X. Astragaloside IV protects blood-brain barrier integrity from LPS-induced disruption via activating Nrf2 antioxidant signaling pathway in mice. Toxicol. Appl. Pharmacol. 2018, 340, 58–66. [Google Scholar] [CrossRef]
- Wang, F.; Zhao, Y.; Chen, S.; Chen, L.; Sun, L.; Cao, M.; Li, C.; Zhou, X. Astragaloside IV alleviates ammonia-induced apoptosis and oxidative stress in bovine mammary epithelial cells. Int. J. Mol. Sci. 2019, 20, e600. [Google Scholar] [CrossRef]
- Wang, J.; Guo, H.M. Astragaloside IV ameliorates high glucose-induced HK-2 cell apoptosis and oxidative stress by regulating the Nrf2/ARE signaling pathway. Exp. Ther. Med. 2019, 17, 4409–4416. [Google Scholar] [CrossRef] [PubMed]
- Qi, W.; Niu, J.; Qin, Q.; Qiao, Z.; Gu, Y. Astragaloside IV attenuates glycated albumin-induced epithelial-to-mesenchymal transition by inhibiting oxidative stress in renal proximal tubular cells. Cell Stress Chaperones 2014, 19, 105–114. [Google Scholar] [CrossRef] [PubMed]
- Xu, N.; Kan, P.; Yao, X.; Yang, P.; Wang, J.; Xiang, L.; Zhu, Y. Astragaloside IV reversed the autophagy and oxidative stress induced by the intestinal microbiota of ais in mice. J. Microbiol. 2018, 56, 838–846. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhang, J.; Wang, S.; Qiu, J.; Yu, C. Astragaloside IV attenuates the H2O2-induced apoptosis of neuronal cells by inhibiting α-synuclein expression via the p38 MAPK pathway. Int. J. Mol. Med. 2017, 40, 1772–1780. [Google Scholar] [CrossRef]
- Hao, M.; Liu, Y.; Chen, P.; Jiang, H.; Kuang, H.Y. Astragaloside Ⅳ protects RGC-5 cells against oxidative stress. Neural Regen. Res. 2018, 13, 1081–1086. [Google Scholar]
- Cui, W.; Leng, B.; Wang, G. Klotho protein inhibits H2O2-induced oxidative injury in endothelial cells via regulation of PI3K/AKT/Nrf2/HO-1 pathways. Can. J. Physiol. Pharmacol. 2019, 97, 370–376. [Google Scholar] [CrossRef]
- Lu, J.; Zhu, W.; Wu, Y.; Meng, Y.F.; Tao, J.J. Effect of curcumin on aging retinal pigment epithelial cells. Drug Des. Dev. Ther. 2015, 9, 5337. [Google Scholar] [CrossRef][Green Version]
- Wang, C.; Liao, Y.; Wang, S.; Wang, D.; Wu, N.; Xu, Q.; Jiang, W.; Qiu, M.; Liu, C. Cytoprotective effects of diosmetin against hydrogen peroxide-induced L02 cell oxidative damage via activation of the Nrf2-ARE signaling pathway. Mol. Med. Rep. 2018, 17, 7331–7338. [Google Scholar] [CrossRef]
- Cheng, C.H.; Guo, Z.X.; Luo, S.W.; Wang, A.L. Effects of high temperature on biochemical parameters, oxidative stress, DNA damage and apoptosis of pufferfish (Takifugu obscurus). Ecotoxicol. Environ. Saf. 2018, 150, 190–198. [Google Scholar] [CrossRef]
- Pan, Y.; Jia, X.; Song, E.; Peng, X. Astragaloside Ⅳ protects against Aβ1-42-induced oxidative stress, neuroinflammation and cognitive impairment in rats. Chin. Med. Sci. J. 2018, 33, 32–40. [Google Scholar] [CrossRef]
- Liu, X.; Liu, H.; Zhai, Y.; Li, Y.; Zhang, W. Laminarin protects against hydrogen peroxide-induced oxidative damage in mrc-5 cells possibly via regulating nrf2. PeerJ 2017, 5, e3642. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Qin, Z.; Hong, Z.; Zhang, X.; Chen, J. Astragaloside iv protects against ischemic brain injury in a murine model of transient focal ischemia. Neurosci. Lett. 2004, 363, 218–223. [Google Scholar] [CrossRef] [PubMed]
- Qiu, L.H.; Xie, X.J.; Zhang, B.Q. Astragaloside iv improves homocysteine-induced acute phase endothelial dysfunction via antioxidation. Biol. Pharm. Bull. 2010, 33, 641–646. [Google Scholar] [CrossRef] [PubMed]
- Arai, K.; Iizuka, S.; Tada, Y.; Oikawa, K.; Taniguchi, N. Increase in the glucosylated form of erythrocyte Cu-Zn-superoxide dismutase in diabetes and close association of the nonenzymatic glucosylation with the enzyme activity. Biochim. Biophys. Acta (BBA)—Gen. Subj. 1987, 924, 292–296. [Google Scholar] [CrossRef]
- Gui, D.; Huang, J.; Liu, W.; Guo, Y.; Xiao, W.; Wang, N. Astragaloside IV prevents acute kidney injury in two rodent models by inhibiting oxidative stress and apoptosis pathways. Apoptosis 2013, 18, 409–422. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Su, Y.; Ju, Y.; Ma, K.; Li, W.; Li, W. Astragalosides IV protected the renal tubular epithelial cells from free fatty acids-induced injury by reducing oxidative stress and apoptosis. Biomed. Pharmacother. 2018, 108, 679–686. [Google Scholar] [CrossRef] [PubMed]
- Jaramillo, M.C.; Zhang, D.D. The emerging role of the nrf2-keap1 signaling pathway in cancer. Genes Dev. 2013, 27, 2179–2191. [Google Scholar] [CrossRef]
- Young-Sam, K.; Young, C.B. Molecular and chemical regulation of the Keap1-Nrf2 signaling pathway. Molecules 2014, 19, 10074–10089. [Google Scholar]
- Jiang, M.; Ni, J.; Cao, Y.; Xing, X.; Wu, Q.; Fan, G. Astragaloside IV attenuates myocardial ischemia-reperfusion injury from oxidative stress by regulating succinate, lysophospholipid metabolism, and ROS scavenging system. Oxidative Med. Cell. Longev. 2019, 2019, 9137654. [Google Scholar] [CrossRef]
- Gu, D.M.; Lu, P.H.; Zhang, K.; Wang, X.; Sun, M.; Chen, G.Q.; Wang, Q. Egfr mediates astragaloside IV-induced nrf2 activation to protect cortical neurons against in vitro ischemia/reperfusion damages. Biochem. Biophys. Res. Commun. 2015, 457, 391–397. [Google Scholar] [CrossRef]
- LIVAK. Analysis of relative gene expression data using real-time quantitative pcr and the 2 (-delta delta c (t)) method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Tan, X.; Liu, Y.; You, W.; Liu, G.; Liu, X.; Jin, Q.; Wei, C.; Wan, F.; Zhao, H. Alanyl-glutamine ameliorates lipopolysaccharide-induced inflammation and barrier function injury in bovine jejunum epithelial cells. Biochem. Cell Biol. 2019. [Google Scholar] [CrossRef] [PubMed]
| Primer Name | Primer Sequence (5′-3′) | Product Length (bp) |
|---|---|---|
| NFE2L2 | Sense Primer: ACCCAGTCCAACCTTTGTCGT | 143 |
| Anti-sence Primer: GCGGCTTGAATGTTTGTCTTT | ||
| NQO1 | Sense Primer: CGGCTCCATGTACTCTCTGC | 183 |
| Anti-sence Primer: TCCAGGCGTTTCTTCCATCC | ||
| HMOX1 | Sense Primer: CAAGCGCTATGTTCAGCGAC | 198 |
| Anti-sence Primer: TTGGTGGCACTGGCGATATT | ||
| β-actin | Sense Primer: CACCGCAAATGCTTCTAGGC | 186 |
| Anti-sence Primer: TGTCACCTTCACCGTTCCAG |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Jiang, F.; Cheng, H.; Tan, X.; Liu, Y.; Wei, C.; Song, E. Astragaloside IV Protects Against Oxidative Stress in Calf Small Intestine Epithelial Cells via NFE2L2-Antioxidant Response Element Signaling. Int. J. Mol. Sci. 2019, 20, 6131. https://doi.org/10.3390/ijms20246131
Wang Y, Jiang F, Cheng H, Tan X, Liu Y, Wei C, Song E. Astragaloside IV Protects Against Oxidative Stress in Calf Small Intestine Epithelial Cells via NFE2L2-Antioxidant Response Element Signaling. International Journal of Molecular Sciences. 2019; 20(24):6131. https://doi.org/10.3390/ijms20246131
Chicago/Turabian StyleWang, Yafang, Fugui Jiang, Haijian Cheng, Xiuwen Tan, Yifan Liu, Chen Wei, and Enliang Song. 2019. "Astragaloside IV Protects Against Oxidative Stress in Calf Small Intestine Epithelial Cells via NFE2L2-Antioxidant Response Element Signaling" International Journal of Molecular Sciences 20, no. 24: 6131. https://doi.org/10.3390/ijms20246131
APA StyleWang, Y., Jiang, F., Cheng, H., Tan, X., Liu, Y., Wei, C., & Song, E. (2019). Astragaloside IV Protects Against Oxidative Stress in Calf Small Intestine Epithelial Cells via NFE2L2-Antioxidant Response Element Signaling. International Journal of Molecular Sciences, 20(24), 6131. https://doi.org/10.3390/ijms20246131




