Effects of Hyperoxia on the Refraction in Murine Neonatal and Adult Models
Abstract
1. Introduction
2. Results
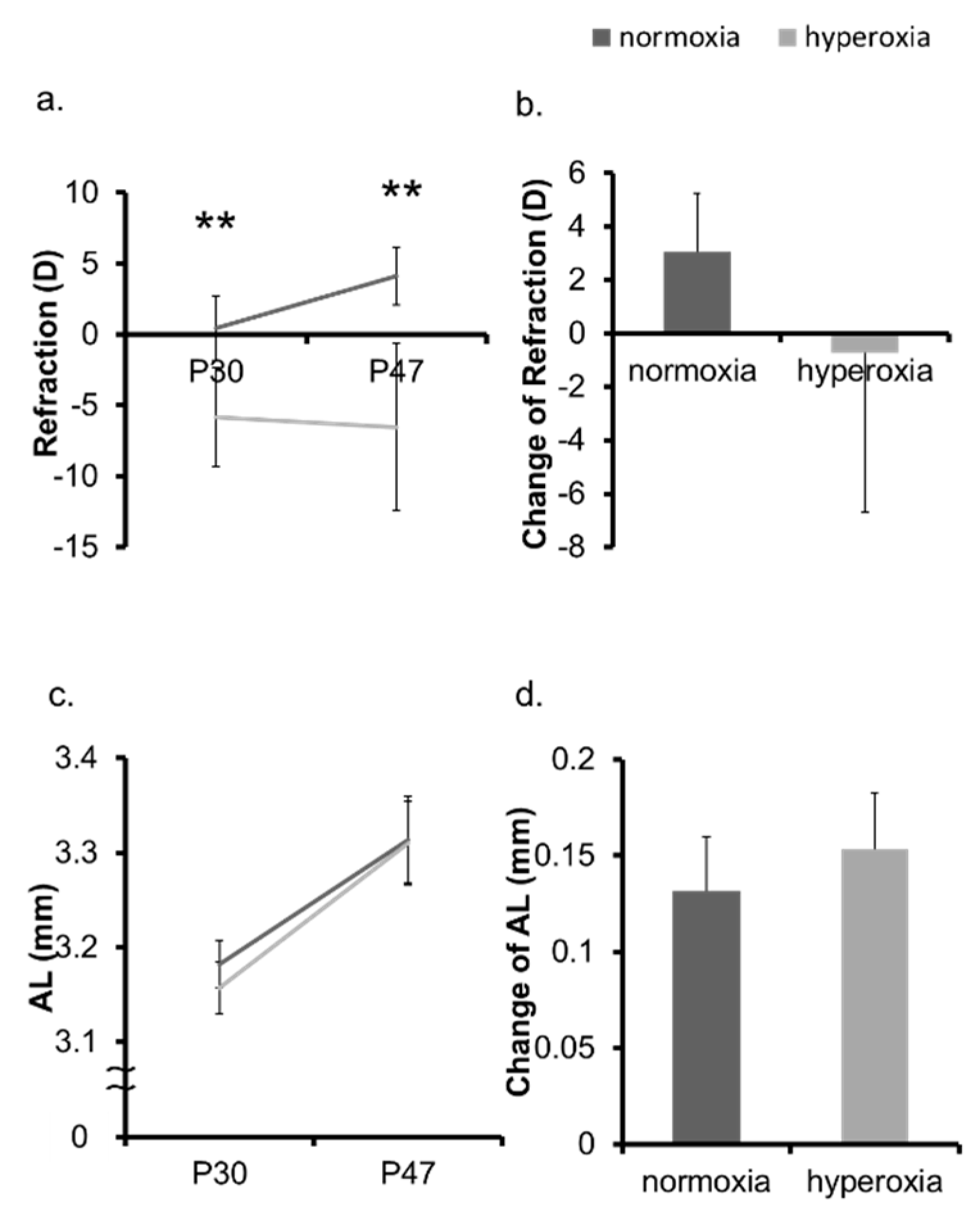
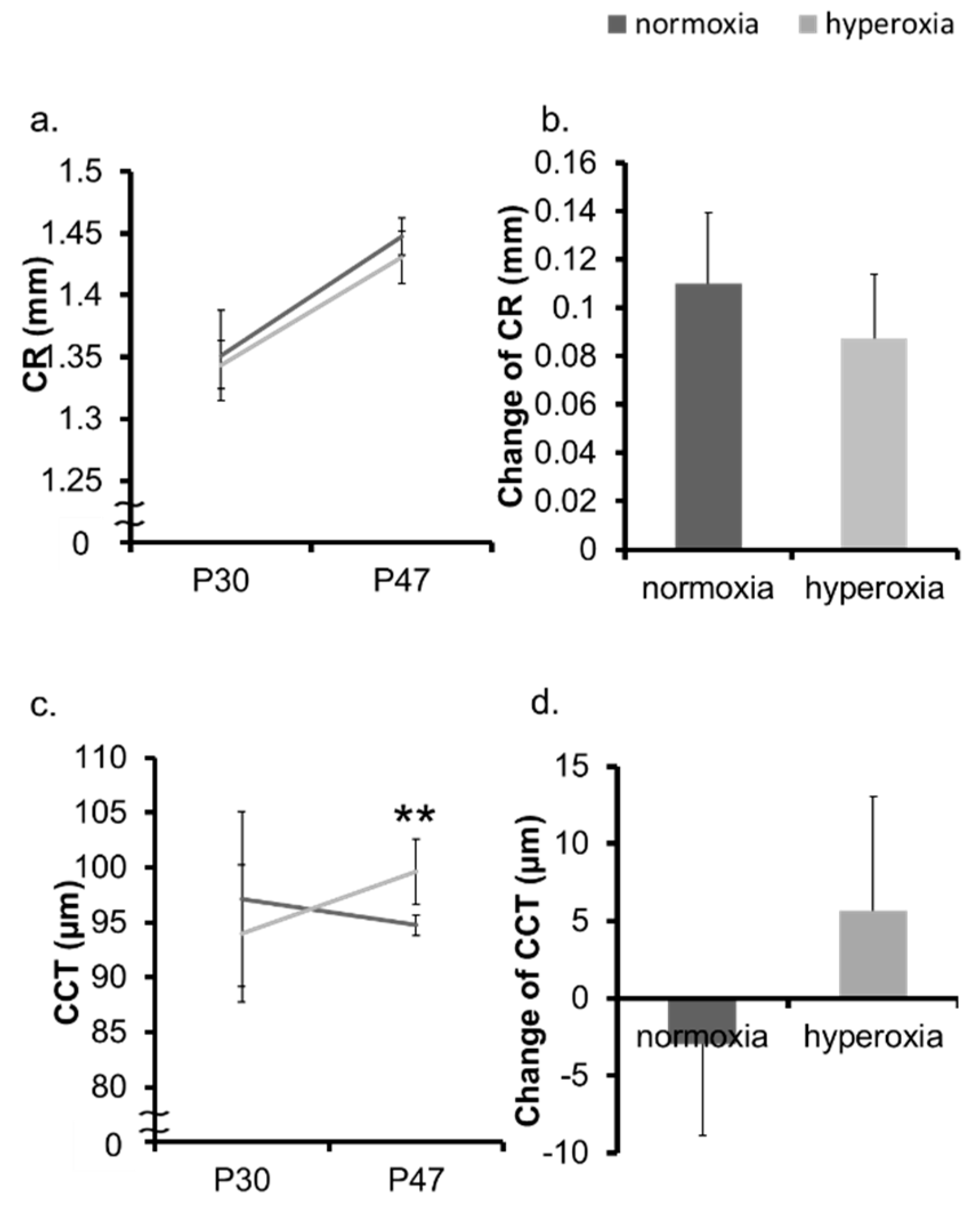
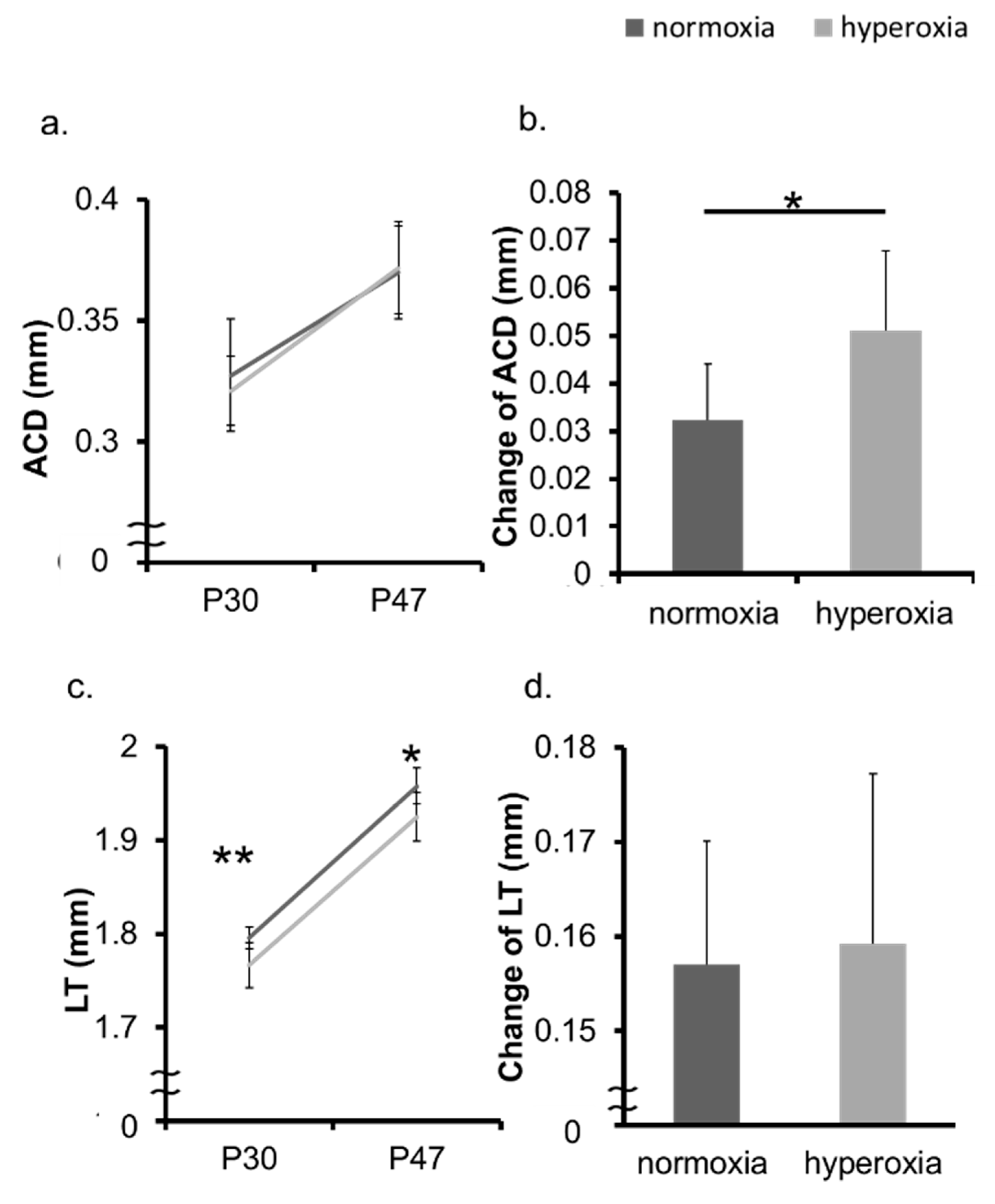
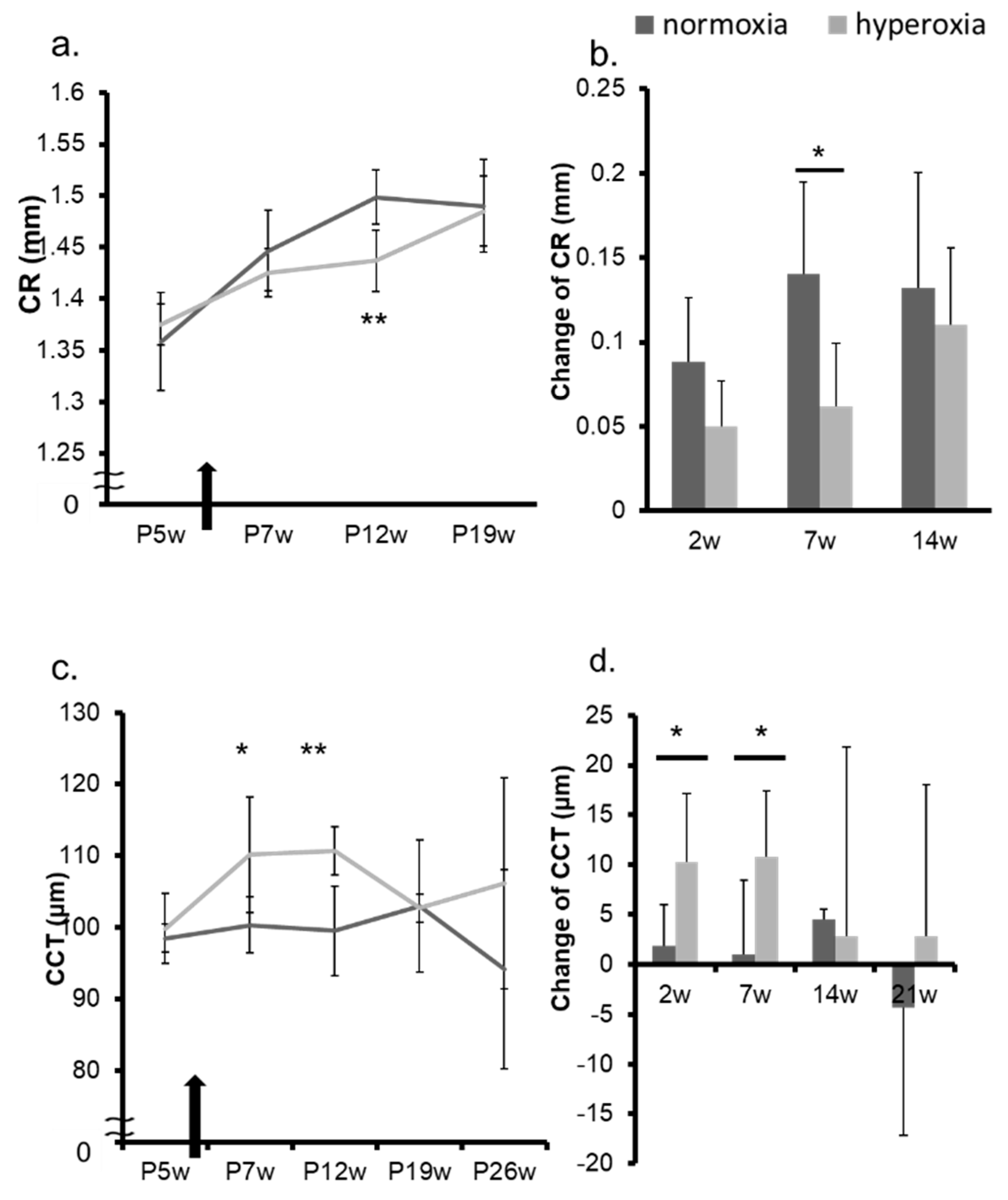
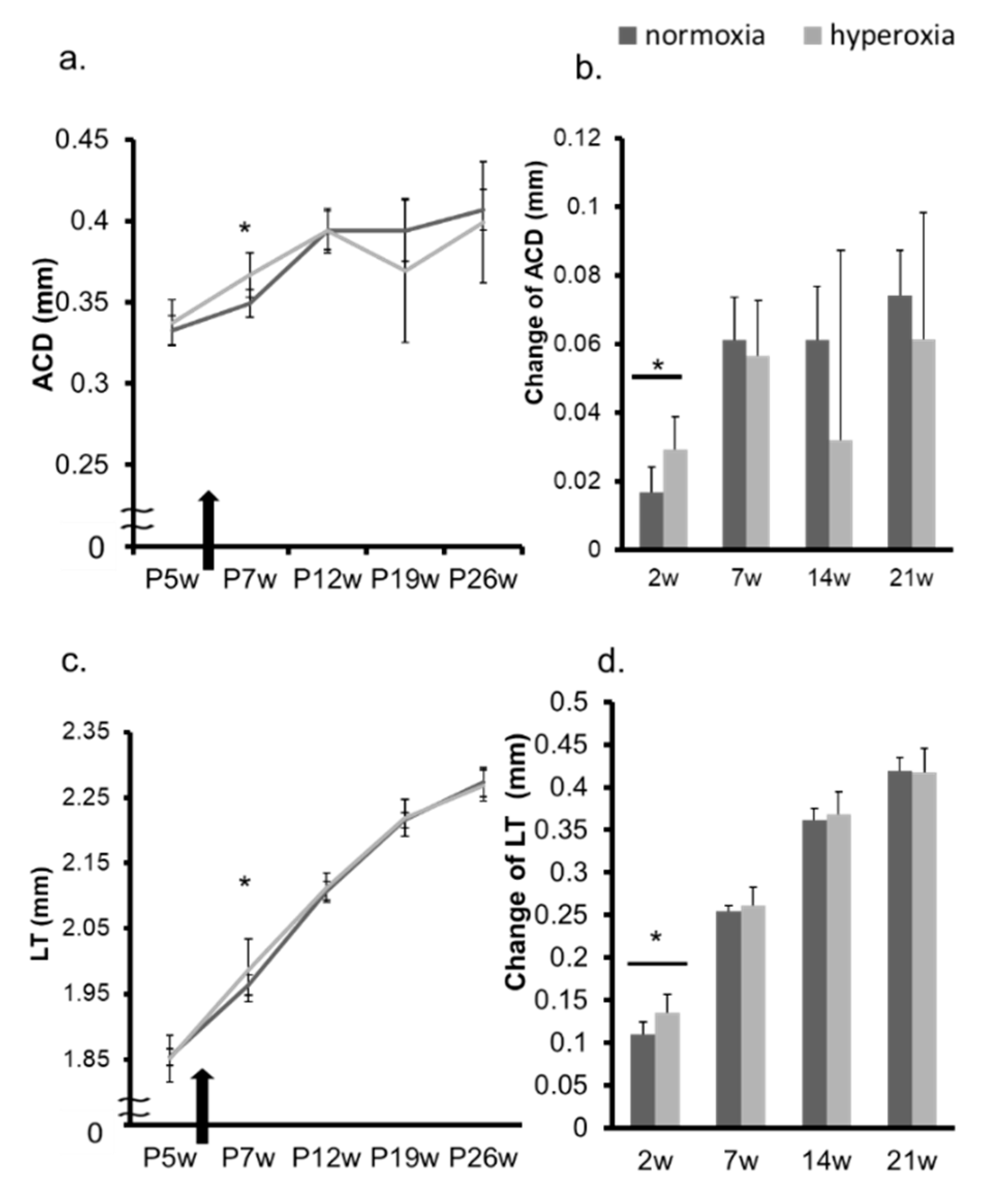
2.1. Neonatal Hyperoxia Model Mice Showed Myopic Shift
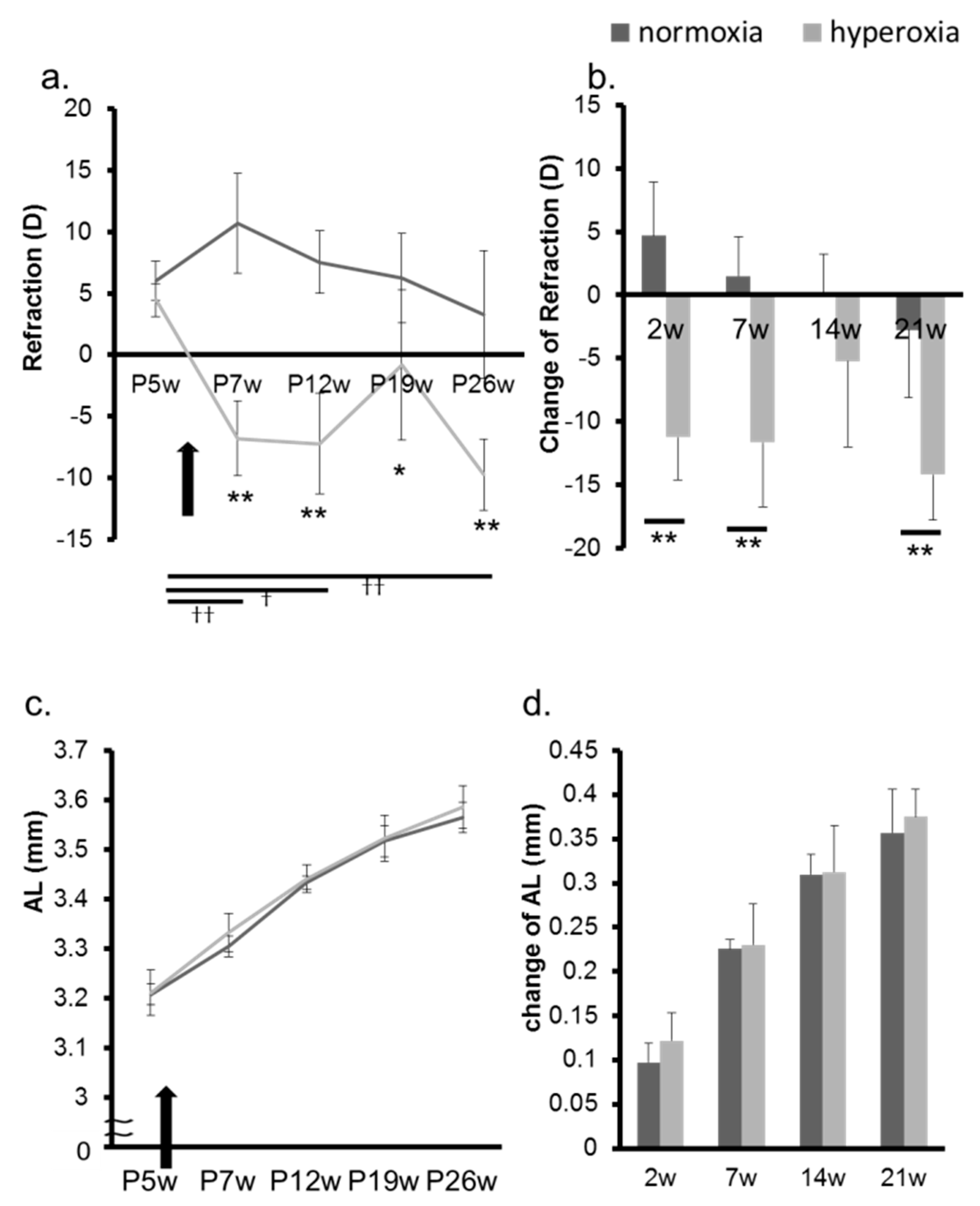
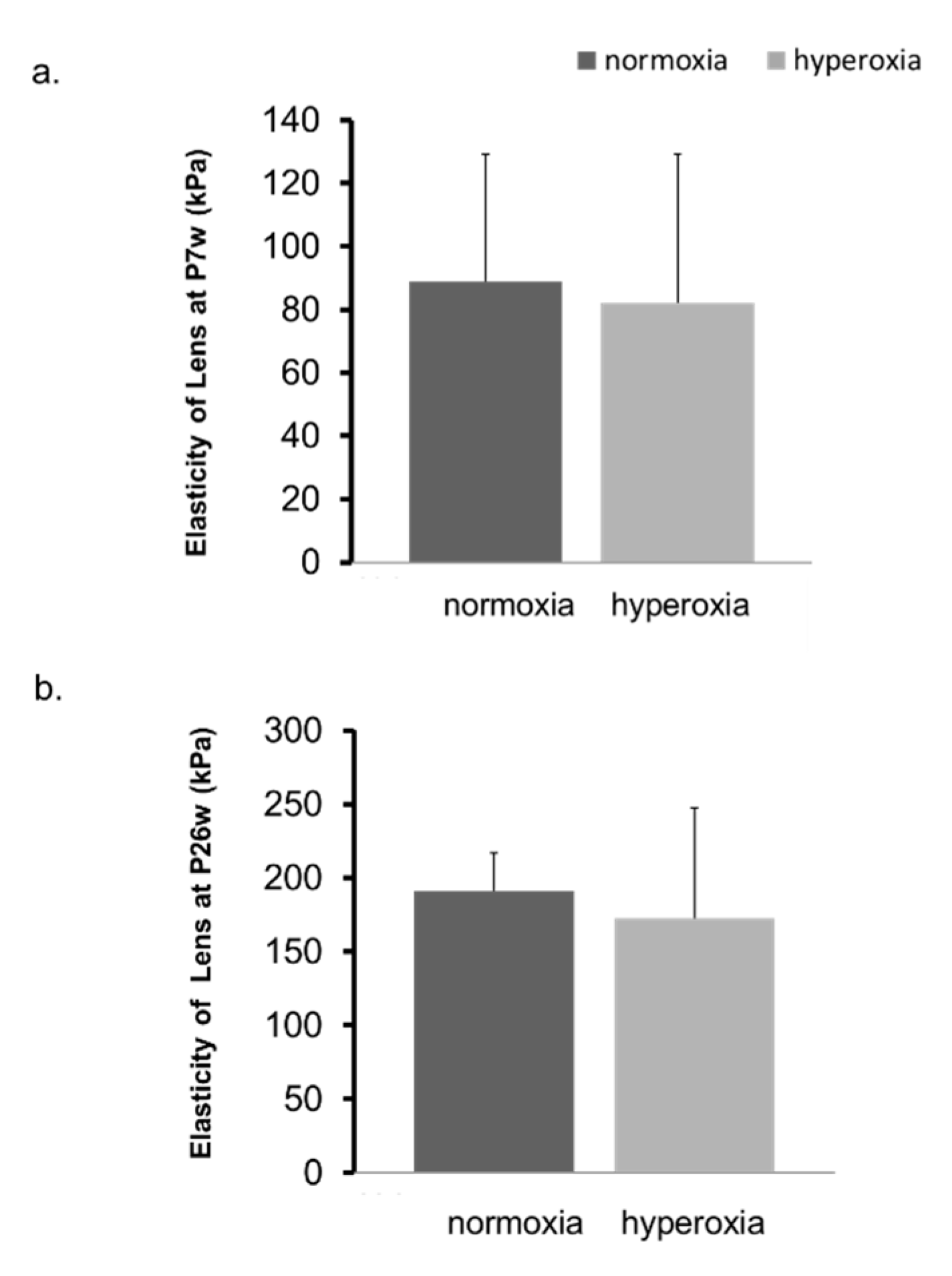
2.2. Hyperoxia Induced Myopia in Adult Mice
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Experimental Design
4.2.1. Exposure to Highly Concentrated Oxygen
4.2.2. Ocular Components Measurement
4.2.3. Measurement of Lens Elasticity
4.3. Statistical Analyses
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| P8d | postnatal 8 days |
| P6w | postnatal 6 weeks |
| CR | corneal curvature radius |
| AL | axial length |
| ROP | retinopathy of prematurity |
| OIR | oxygen-induced retinopathy |
| CCT | central corneal thickness |
| D | diopter |
| ACD | anterior chamber depth |
| LT | lens thickness |
| kPa | kilopascal |
| ROS | reactive oxidative species |
References
- Choi, M.Y.; Park, I.K.; Yu, Y.S. Long term refractive outcome in eyes of preterm infants with and without retinopathy of prematurity: Comparison of keratometric value, axial length, anterior chamber depth, and lens thickness. Br. J. Ophthalmol. 2000, 84, 138–143. [Google Scholar] [CrossRef] [PubMed]
- Hartnett, M.E.; Gilbert, M.M.; Hirose, T.; Richardson, T.M.; Katsumi, O. Glaucoma as a cause of poor vision in severe retinopathy of prematurity. Graefes Arch. Clin. Exp. Ophthalmol. 1993, 231, 433–438. [Google Scholar] [CrossRef] [PubMed]
- Knight-Nanan, D.M.; Algawi, K.; Bowell, R.; O’Keefe, M. Advanced cicatricial retinopathy of prematurity—Outcome and complications. Br. J. Ophthalmol. 1996, 80, 343–345. [Google Scholar] [CrossRef] [PubMed]
- Maly, E. Frequency and natural history of retinopathy of prematurity (ROP): A prospective study in a Swedish city 1986–1990. Acta Ophthalmol. Suppl. 1993, 210, 52–55. [Google Scholar] [CrossRef] [PubMed]
- Baker, P.S.; Tasman, W. Myopia in adults with retinopathy of prematurity. Am. J. Ophthalmol. 2008, 145, 1090–1094. [Google Scholar] [CrossRef] [PubMed]
- Dobson, V.; Fulton, A.B.; Manning, K.; Salem, D.; Petersen, R.A. Cycloplegic refractions of premature infants. Am. J. Ophthalmol. 1981, 91, 490–495. [Google Scholar] [CrossRef]
- Quinn, G.E.; Dobson, V.; Davitt, B.V.; Hardy, R.J.; Tung, B.; Pedroza, C.; Good, W.V. Progression of myopia and high myopia in the early treatment for retinopathy of prematurity study: Findings to 3 years of age. Ophthalmology 2008, 115, 1058–1064. [Google Scholar] [CrossRef]
- Quinn, G.E.; Dobson, V.; Davitt, B.V.; Wallace, D.K.; Hardy, R.J.; Tung, B.; Lai, D.; Good, W.V. Progression of myopia and high myopia in the Early Treatment for Retinopathy of Prematurity study: Findings at 4 to 6 years of age. J. AAPOS Off. Publ. Am. Assoc. Pediatric Ophthalmol. Strabismus 2013, 17, 124–128. [Google Scholar] [CrossRef]
- Quinn, G.E.; Dobson, V.; Repka, M.X.; Reynolds, J.; Kivlin, J.; Davis, B.; Buckley, E.; Flynn, J.T.; Palmer, E.A. Development of myopia in infants with birth weights less than 1251 grams: The Cryotherapy for Retinopathy of Prematurity Cooperative Group. Ophthalmology 1992, 99, 329–340. [Google Scholar] [CrossRef]
- O’Connor, A.R.; Stephenson, T.; Johnson, A.; Tobin, M.J.; Moseley, M.J.; Ratib, S.; Ng, Y.; Fielder, A.R. Long-Term ophthalmic outcome of low birth weight children with and without retinopathy of prematurity. Pediatrics 2002, 109, 12–18. [Google Scholar] [CrossRef]
- O’Connor, A.R.; Stephenson, T.J.; Johnson, A.; Tobin, M.J.; Ratib, S.; Fielder, A.R. Change of refractive state and eye size in children of birth weight less than 1701 g. Br. J. Ophthalmol. 2006, 90, 456–460. [Google Scholar] [CrossRef] [PubMed]
- Al-Otaibi, A.G.; Aldrees, S.S.; Mousa, A.A. Long term visual outcomes in laser treated threshold retinopathy of prematurity in Central Saudi Arabia. Saudi J. Ophthalmol. Off. J. Saudi Ophthalmol. Soc. 2012, 26, 299–303. [Google Scholar] [CrossRef] [PubMed]
- Axer-Siegel, R.; Maharshak, I.; Snir, M.; Friling, R.; Ehrlich, R.; Sherf, I.; Shalev, B.; Sirota, L.; Weinberger, D. Diode laser treatment of retinopathy of prematurity: Anatomical and refractive outcomes. Retina 2008, 28, 839–846. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, P.H.; Catt, C.; Nguyen, T.X.; Pham, V.T. Refractive outcome of prethreshold retinopathy of prematurity treated by diode laser: Follow-up at 5 years. Clin. Ophthalmol. 2015, 9, 1753–1758. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ben Sira, I.; Nissenkorn, I.; Kremer, I. Retinopathy of prematurity. Surv. Ophthalmol. 1988, 33, 1–16. [Google Scholar] [CrossRef]
- Saugstad, O.D. Oxygen and retinopathy of prematurity. J. Perinatol. 2006, 26, S46–S50. [Google Scholar] [CrossRef]
- Saw, S.M.; Chew, S.J. Myopia in children born premature or with low birth weight. Acta Ophthalmol. Scand. 1997, 75, 548–550. [Google Scholar] [CrossRef]
- Chui, T.Y.; Bissig, D.; Berkowitz, B.A.; Akula, J.D. Refractive Development in the “ROP Rat”. J. Ophthalmol. 2012, 2012, 956705. [Google Scholar] [CrossRef]
- Moon, R.E. Hyperbaric oxygen treatment for decompression sickness. Undersea Hyperb. Med. 2014, 41, 151–157. [Google Scholar]
- Whayne, T.F. Medical Management and Risk Reduction of the Cardiovascular Effects of Underwater Diving. Curr. Vasc. Pharm. 2018, 16, 344–354. [Google Scholar] [CrossRef]
- Evanger, K.; Haugen, O.H.; Irgens, A.; Aanderud, L.; Thorsen, E. Ocular refractive changes in patients receiving hyperbaric oxygen administered by oronasal mask or hood. Acta Ophthalmol. Scand. 2004, 82, 449–453. [Google Scholar] [CrossRef] [PubMed]
- Fledelius, H.C.; Jansen, E.C.; Thorn, J. Refractive change during hyperbaric oxygen therapy: A clinical trial including ultrasound oculometry. Acta Ophthalmol. Scand. 2002, 80, 188–190. [Google Scholar] [CrossRef] [PubMed]
- Ashton, N. Some aspects of the comparative pathology of oxygen toxicity in the retina. Ophthalmologica 1970, 160, 54–71. [Google Scholar] [CrossRef] [PubMed]
- Miwa, Y.; Hoshino, Y.; Shoda, C.; Jiang, X.; Tsubota, K.; Kurihara, T. Pharmacological HIF inhibition prevents retinal neovascularization with improved visual function in a murine oxygen-induced retinopathy model. Neurochem. Int. 2019, 128, 21–31. [Google Scholar] [CrossRef]
- Okabe, K.; Kobayashi, S.; Yamada, T.; Kurihara, T.; Tai-Nagara, I.; Miyamoto, T.; Mukouyama, Y.S.; Sato, T.N.; Suda, T.; Ema, M.; et al. Neurons limit angiogenesis by titrating VEGF in retina. Cell 2014, 159, 584–596. [Google Scholar] [CrossRef]
- Smith, L.E.; Wesolowski, E.; McLellan, A.; Kostyk, S.K.; D’Amato, R.; Sullivan, R.; D’Amore, P.A. Oxygen-Induced retinopathy in the mouse. Investig. Ophthalmol. Vis. Sci. 1994, 35, 101–111. [Google Scholar]
- Hashemi, H.; Khabazkhoob, M.; Emamian, M.H.; Shariati, M.; Miraftab, M.; Yekta, A.; Ostadimoghaddam, H.; Fotouhi, A. Association between Refractive Errors and Ocular Biometry in Iranian Adults. J. Ophthalmic Vis. Res. 2015, 10, 214–220. [Google Scholar] [CrossRef]
- Meng, W.; Butterworth, J.; Malecaze, F.; Calvas, P. Axial length of myopia: A review of current research. Ophthalmologica 2011, 225, 127–134. [Google Scholar] [CrossRef]
- Mallen, E.A.; Gammoh, Y.; Al-Bdour, M.; Sayegh, F.N. Refractive error and ocular biometry in Jordanian adults. Ophthalmic Physiol. Opt. J. Br. Coll. Ophthalmic Opt. 2005, 25, 302–309. [Google Scholar] [CrossRef]
- McBrien, N.A.; Adams, D.W. A longitudinal investigation of adult-onset and adult-progression of myopia in an occupational group: Refractive and biometric findings. Investig. Ophthalmol. Vis. Sci. 1997, 38, 321–333. [Google Scholar]
- Shufelt, C.; Fraser-Bell, S.; Ying-Lai, M.; Torres, M.; Varma, R. Refractive error, ocular biometry, and lens opalescence in an adult population: The Los Angeles Latino Eye Study. Investig. Ophthalmol. Vis. Sci. 2005, 46, 4450–4460. [Google Scholar] [CrossRef] [PubMed]
- Ojaimi, E.; Rose, K.A.; Morgan, I.G.; Smith, W.; Martin, F.J.; Kifley, A.; Robaei, D.; Mitchell, P. Distribution of ocular biometric parameters and refraction in a population-based study of Australian children. Investig. Ophthalmol. Vis. Sci. 2005, 46, 2748–2754. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Favazza, T.L.; Baglieri, A.M.; Benador, I.Y.; Noonan, E.R.; Fulton, A.B.; Hansen, R.M.; Iuvone, P.M.; Akula, J.D. The rat with oxygen-induced retinopathy is myopic with low retinal dopamine. Investig. Ophthalmol. Vis. Sci. 2013, 54, 8275–8284. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Garcia-Valenzuela, E.; Kaufman, L.M. High myopia associated with retinopathy of prematurity is primarily lenticular. J. AAPOS Off. Publ. Am. Assoc. Pediatric Ophthalmol. Strabismus 2005, 9, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Giblin, F.J.; Padgaonkar, V.A.; Leverenz, V.R.; Lin, L.R.; Lou, M.F.; Unakar, N.J.; Dang, L.; Dickerson, J.E., Jr.; Reddy, V.N. Nuclear light scattering, disulfide formation and membrane damage in lenses of older guinea pigs treated with hyperbaric oxygen. Exp. Eye Res. 1995, 60, 219–235. [Google Scholar] [CrossRef]
- Palmquist, B.M.; Philipson, B.; Barr, P.O. Nuclear cataract and myopia during hyperbaric oxygen therapy. Br. J. Ophthalmol. 1984, 68, 113–117. [Google Scholar] [CrossRef]
- Francisco, B.M.; Salvador, M.; Amparo, N. Oxidative stress in myopia. Oxidative Med. Cell. Longev. 2015, 2015, 750637. [Google Scholar] [CrossRef]
- McCord, J.M. Superoxide dismutase: Rationale for use in reperfusion injury and inflammation. J. Free Radic. Biol. Med. 1986, 2, 307–310. [Google Scholar] [CrossRef]
- Iuga, C.; Alvarez-Idaboy, J.R.; Vivier-Bunge, A. ROS initiated oxidation of dopamine under oxidative stress conditions in aqueous and lipidic environments. J. Phys. Chem. B 2011, 115, 12234–12246. [Google Scholar] [CrossRef]
- Feldkaemper, M.; Schaeffel, F. An updated view on the role of dopamine in myopia. Exp. Eye Res. 2013, 114, 106–119. [Google Scholar] [CrossRef]
- Kurihara, T. Development and pathological changes of neurovascular unit regulated by hypoxia response in the retina. Prog. Brain Res. 2016, 225, 201–211. [Google Scholar] [CrossRef] [PubMed]
- Yan, S.F.; Lu, J.; Zou, Y.S.; Soh-Won, J.; Cohen, D.M.; Buttrick, P.M.; Cooper, D.R.; Steinberg, S.F.; Mackman, N.; Pinsky, D.J.; et al. Hypoxia-associated induction of early growth response-1 gene expression. J. Biol. Chem. 1999, 274, 15030–15040. [Google Scholar] [CrossRef] [PubMed]
- Bitzer, M.; Kovacs, B.; Feldkaemper, M.; Schaeffel, F. Effects of muscarinic antagonists on ZENK expression in the chicken retina. Exp. Eye Res. 2006, 82, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Bitzer, M.; Schaeffel, F. Defocus-Induced changes in ZENK expression in the chicken retina. Investig. Ophthalmol. Vis. Sci. 2002, 43, 246–252. [Google Scholar]
- Fischer, A.J.; McGuire, J.J.; Schaeffel, F.; Stell, W.K. Light- and focus-dependent expression of the transcription factor ZENK in the chick retina. Nat. Neurosci. 1999, 2, 706–712. [Google Scholar] [CrossRef]
- Schippert, R.; Burkhardt, E.; Feldkaemper, M.; Schaeffel, F. Relative axial myopia in Egr-1 (ZENK) knockout mice. Investig. Ophthalmol. Vis. Sci. 2007, 48, 11–17. [Google Scholar] [CrossRef]
- Wildsoet, C.F.; Wong, R.O. A far-sighted view of myopia. Nat. Med. 1999, 5, 879–880. [Google Scholar] [CrossRef]
- Torii, H.; Ohnuma, K.; Kurihara, T.; Tsubota, K.; Negishi, K. Violet Light Transmission is Related to Myopia Progression in Adult High Myopia. Sci. Rep. 2017, 7, 14523. [Google Scholar] [CrossRef]
- Mori, K.; Kurihara, T.; Miyauchi, M.; Ishida, A.; Jiang, X.; Ikeda, S.I.; Torii, H.; Tsubota, K. Oral crocetin administration suppressed refractive shift and axial elongation in a murine model of lens-induced myopia. Sci. Rep. 2019, 9, 295. [Google Scholar] [CrossRef]
- Jiang, X.; Kurihara, T.; Kunimi, H.; Miyauchi, M.; Ikeda, S.I.; Mori, K.; Tsubota, K.; Torii, H.; Tsubota, K. A highly efficient murine model of experimental myopia. Sci. Rep. 2018, 8, 2026. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mori, K.; Kurihara, T.; Jiang, X.; Ikeda, S.-i.; Ishida, A.; Torii, H.; Tsubota, K. Effects of Hyperoxia on the Refraction in Murine Neonatal and Adult Models. Int. J. Mol. Sci. 2019, 20, 6014. https://doi.org/10.3390/ijms20236014
Mori K, Kurihara T, Jiang X, Ikeda S-i, Ishida A, Torii H, Tsubota K. Effects of Hyperoxia on the Refraction in Murine Neonatal and Adult Models. International Journal of Molecular Sciences. 2019; 20(23):6014. https://doi.org/10.3390/ijms20236014
Chicago/Turabian StyleMori, Kiwako, Toshihide Kurihara, Xiaoyan Jiang, Shin-ichi Ikeda, Ayako Ishida, Hidemasa Torii, and Kazuo Tsubota. 2019. "Effects of Hyperoxia on the Refraction in Murine Neonatal and Adult Models" International Journal of Molecular Sciences 20, no. 23: 6014. https://doi.org/10.3390/ijms20236014
APA StyleMori, K., Kurihara, T., Jiang, X., Ikeda, S.-i., Ishida, A., Torii, H., & Tsubota, K. (2019). Effects of Hyperoxia on the Refraction in Murine Neonatal and Adult Models. International Journal of Molecular Sciences, 20(23), 6014. https://doi.org/10.3390/ijms20236014






