Allosteric Modulation of Cannabinoid Receptor 1—Current Challenges and Future Opportunities
Abstract
1. Introduction
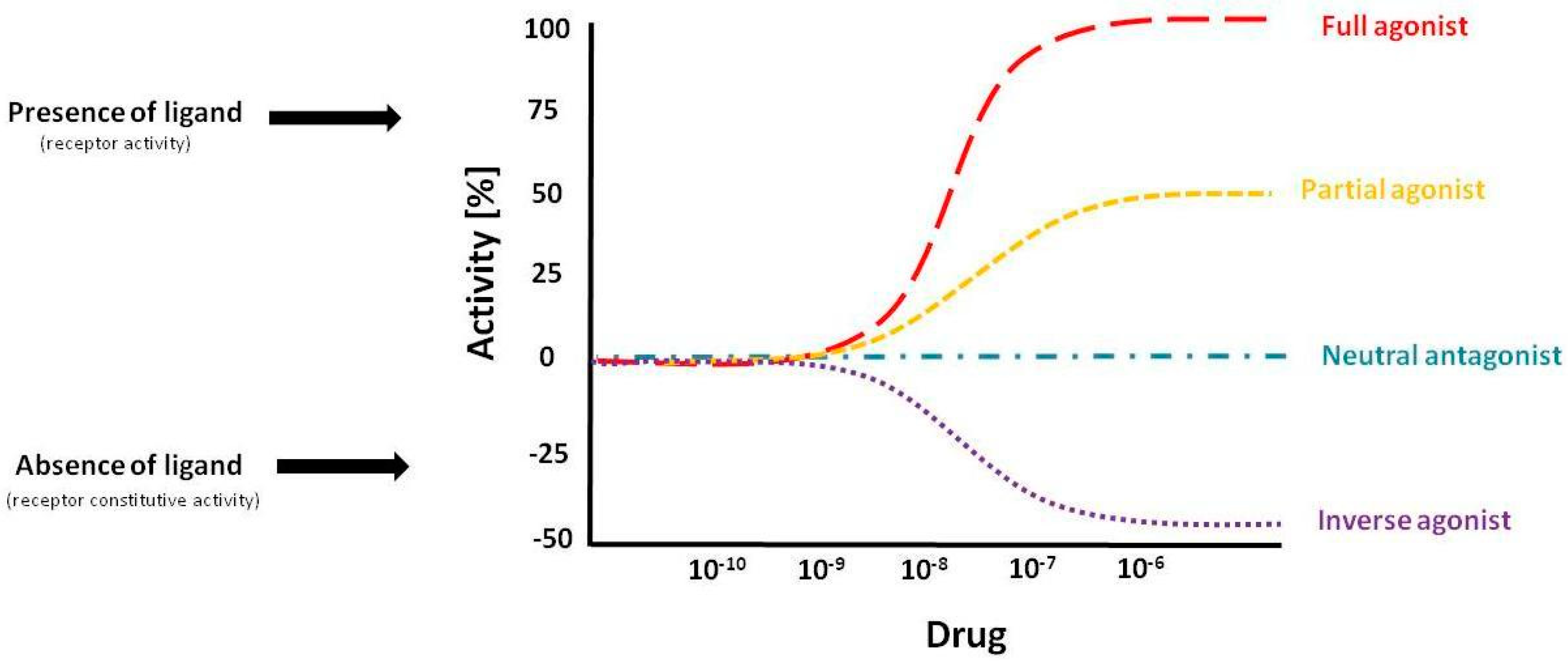
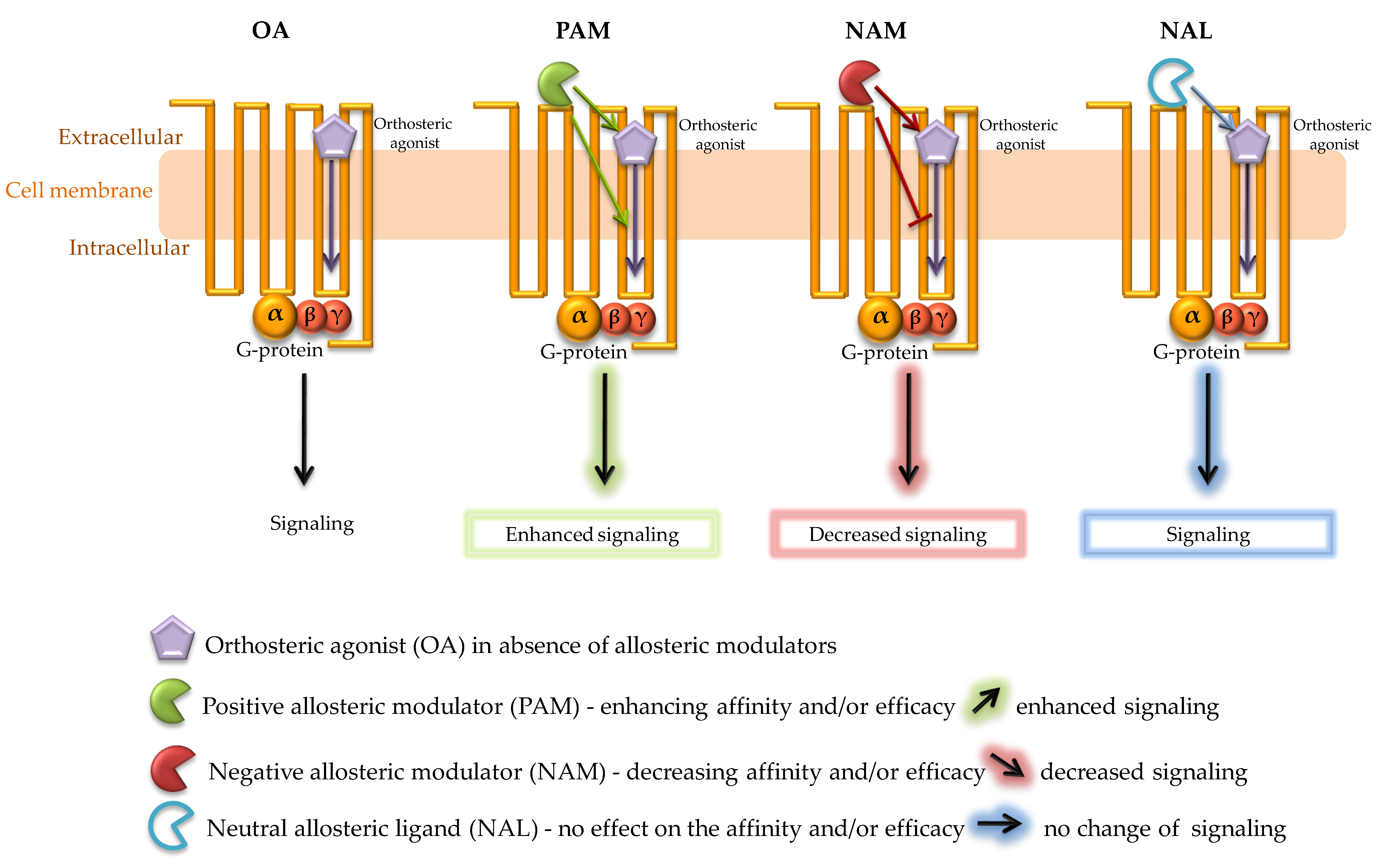
2. Allosteric Modulation of GPCRs
3. CB1R and Its Ligands
4. CB1R Activation and Signaling
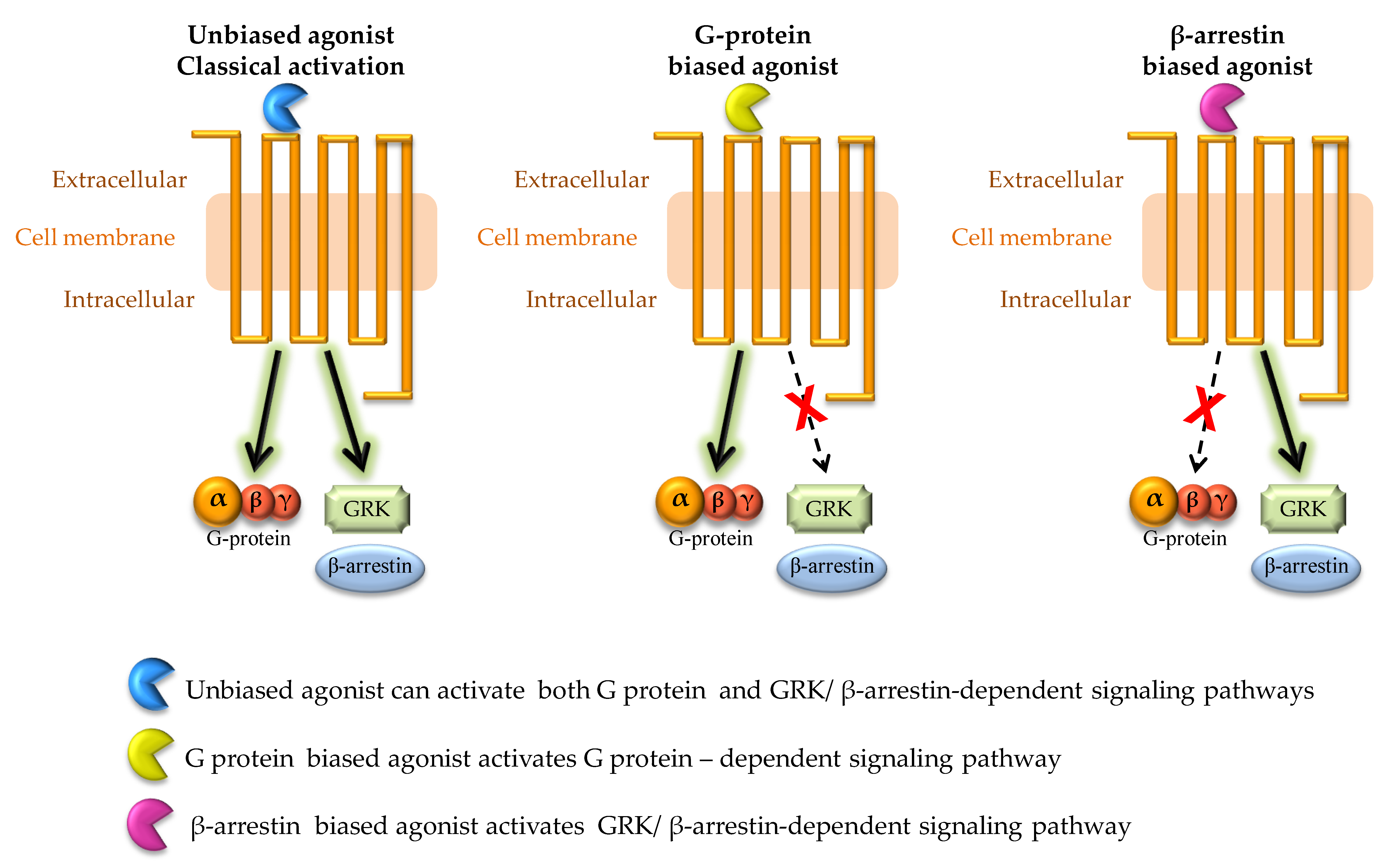
5. CB1R Biased Signaling
6. Other Novel Modes of CB1R Activation
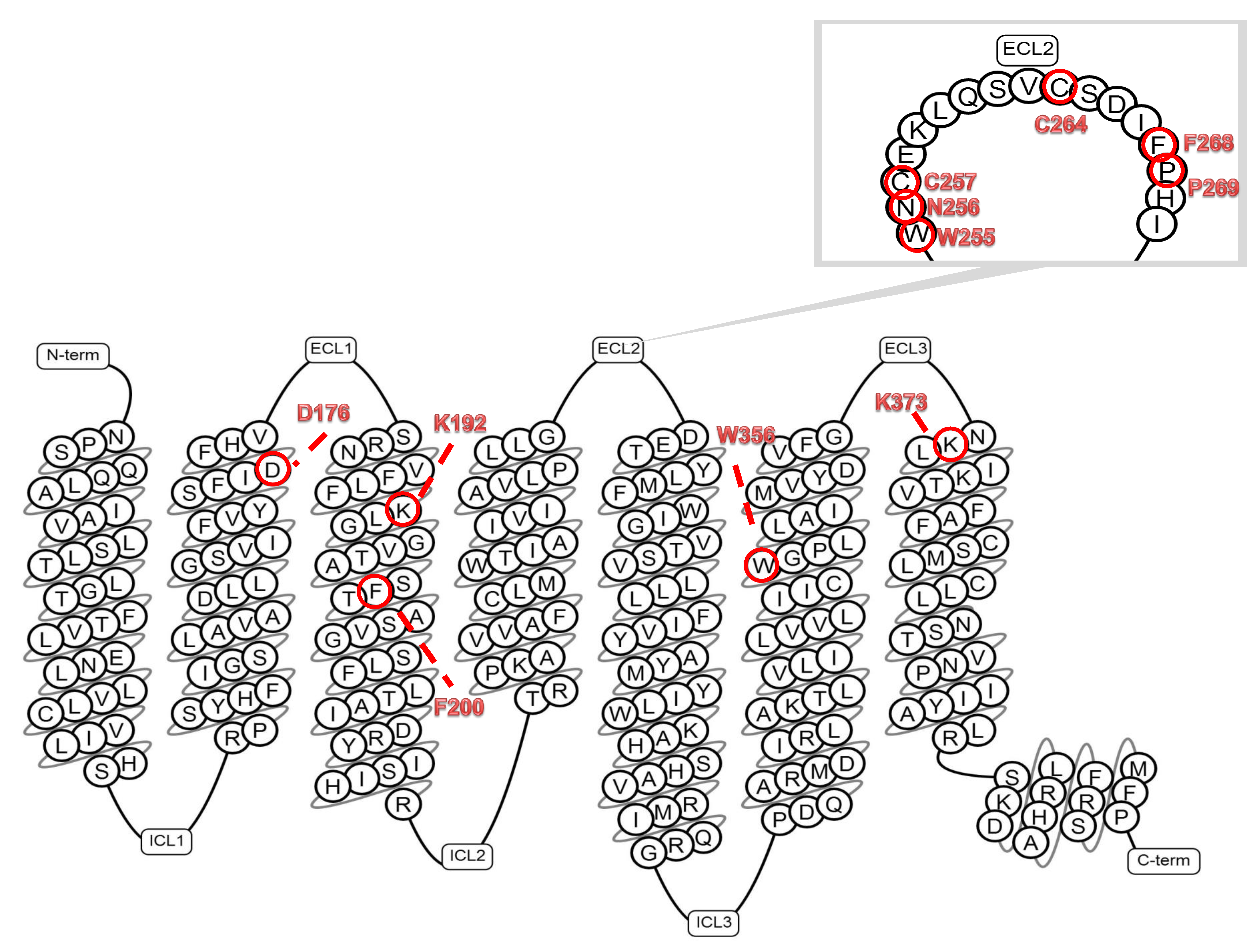
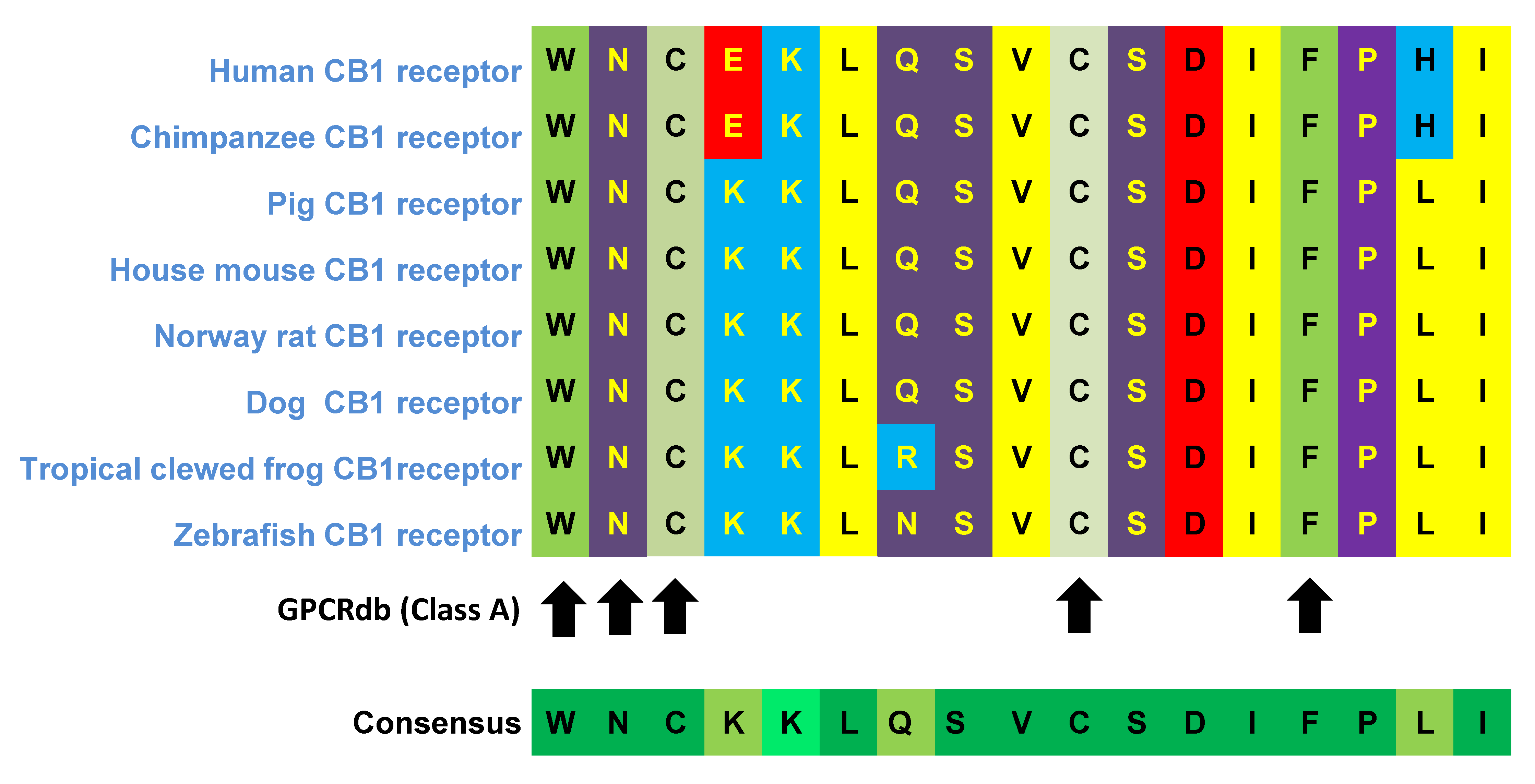
7. A Glimpse into CB1R Structure: The Second Extracellular Loop (ECL2) as a Significant Region of the CB1 Receptor
8. Allosteric Modulators of CB1R
9. Challenges and Perspectives for Translational Use of CB1R Allosteric Modulators
10. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| CB1R | Cannabinoid receptor type 1 |
| GPCR | G protein-coupled receptor |
| ECL2 | Extracellular loop 2 |
| ECS | Endocannabinoid system |
| AEA | Anandamide |
| 2-AG | 2-Arachidonoylglycerol |
| CB2R | Cannabinoid receptor type 2 |
| FAAH | Fatty acid amide hydrolase |
| MGL | Monoacylglycerol lipase |
| NMR | Nuclear magnetic resonance |
| EPR | Electron paramagnetic resonance |
| MS | Mass spectrometry |
| FRET | Fluorescence resonance energy transfer |
| X-ray | X-ray crystallography |
| IUPHAR | The International Union of Basic and Clinical Pharmacology |
| 7TMRs | Seven-Transmembrane Receptors |
| NALs | Neutral allosteric ligands |
| CNR1 | Cannabinoid receptor 1 gene |
| kDa | Kilo Dalton |
| TMH | Transmembrane-helix |
| ICL | Intracellular loop |
| ∆-9-THC | Delta 9 tetrahydrocannabinol |
| CP 55,940 | [2-((1R,2R,5R)-5-hydroxy-2-(3-hydroxypropyl)cyclohexyl)-5-(2-methyloctan-2-yl)phenol] |
| HU-210 | [(6aR,10aR)-9-(hydroxymethyl)-6,6-dimethyl-3-(2-methyloctan-2-yl)-6a,7,10,10a-tetra-hydro-6H-benzo[c]chromen-1-ol]- |
| WIN 55212-2 | [(R)-(5-methyl-3-(morpholinomethyl)-2,3-dihydro-[1,4]oxazino[2,3,4-hi]indol-6-yl)-(naphthalen-1-yl)methanone] |
| JWH-018 | [Naphthalen-1-yl-(1-pentylindol-3-yl)methanone] |
| SR-141716A (rimonabant) | [5-(4-Chlorophenyl)-1-(2,4-dichlorophenyl)-4-methyl-N-(1-piperidinyl)-1H-pyrazole-3-carboxamide] |
| AM251 | [1-(2,4-Dichlorophenyl)-5-(4-iodophenyl)-4-methyl-N-(1-piperidinyl)-1-pyrazole-3-carboxamide] |
| AIDS | Acquired immunodeficiency syndrome |
| FDA | Food and Drug Administration |
| cAMP | Cyclic adenosine monophosphate |
| PKA | Protein kinase A |
| PKB | Protein kinase B |
| MAPKs | Mitogen-activated protein kinases |
| ERK | Extracellular signal-regulated protein kinase |
| JNK | C-JUN N-terminal kinase |
| PI3K | Phosphoinositide 3-kinases |
| AP-3 | Adaptor protein 3 |
| GASP | GPCR-associated sorting proteins |
| FAN | Factor associated with neutral sphingomyelinase activation |
| CRIP1a | Cannabinoid receptor-interacting protein 1a |
| VGCC | Voltage-gated calcium channel |
| GIRK | G-protein-coupled inwardly rectifying potassium channels |
| A2A | Adenosine receptor dopamine D2 |
| D2 | Dopamine receptor |
| CCR2 | Chemokine (C-C motif) receptor type 2 |
| CCR9 | Chemokine (C-C motif) receptor type 9 |
| β-2AR | Beta-2 adrenergic receptor |
| mtCB1 | Mitochondrial cannabinoid receptor |
| sAC | Soluble adenylyl cyclase |
| MECA | Melanocortin, endothelial differentiation, cannabinoid, adenosine cluster |
| AA2AR | adenosine A2A receptor |
| CWxP | Cys-Trp-Xaa-Pro motif |
| EC | Extracellular |
| Cys | Cysteine |
| MPR | Membrane proximal region |
| Org | Organon |
| PAM | Positive allosteric modulator |
| NAM | Negative allosteric modulator |
| SAR | Structure–activity relationship |
| MDMB-Fubinaca | Indazole-based synthetic cannabinoid |
| LXA4 | Lipoxin 4 |
| CBD | Cannabidiol |
| DAT | Dopamine active transporter |
| RTI-371 | 3β-(4-Methylphenyl)-2β-[3-(4-chlorophenyl)isoxazol-5-yl]tropane |
| 7TMR | Seven-Transmembrane Receptors |
References
- Pierce, K.L.; Premont, R.T.; Lefkowitz, R.J. Seven-Transmembrane receptors. Nat. Rev. Mol. Cell Biol. 2002, 3, 639–650. [Google Scholar] [CrossRef]
- Kenakin, T. Allosteric theory: Taking therapeutic advantage of the malleable nature of GPCRs. Curr. Neuropharmacol. 2007, 5, 149–156. [Google Scholar] [CrossRef]
- Rosenbaum, D.M.; Rasmussen, S.G.; Kobilka, B.K. The structure and function of G-protein-coupled receptors. Nature 2009, 459, 356–363. [Google Scholar] [CrossRef]
- Changeux, J.P.; Christopoulos, A. Allosteric modulation as a unifying mechanism for receptor function and regulation. Diabetes Obes. Metab. 2017, 1, 4–21. [Google Scholar] [CrossRef]
- Wacker, D.; Stevens, R.C.; Roth, B.L. How ligands illuminate GPCR molecular pharmacology. Cell 2017, 170, 414–427. [Google Scholar] [CrossRef] [PubMed]
- Packiriswamy, N.; Parameswaran, N. G-Protein-Coupled receptor kinases in inflammation and disease. Genes Immun. 2015, 16, 367–377. [Google Scholar] [CrossRef] [PubMed]
- Insel, P.A.; Tang, C.M.; Hahntow, I.; Michel, M.C. Impact of GPCRs in clinical medicine: Monogenic diseases, genetic variants and drug targets. Biochim. Biophys. Acta 2007, 1768, 994–1005. [Google Scholar] [CrossRef] [PubMed]
- Unal, H.; Karnik, S.S. Domain coupling in GPCRs: The engine for induced conformational changes. Trends Pharm. Sci. 2012, 33, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.C.; Mackie, K. An introduction to the endogenous cannabinoid system. Biol. Psychiatry 2016, 79, 516–525. [Google Scholar] [CrossRef]
- Lu, D.; Potter, D.E. Cannabinoids and the cannabinoid receptors: An overview. In Handbook of Cannabisand Related Pathologies Biology, Pharmacology, Diagnosis, and Treatment; Preedy, V.R., Ed.; Academic Press: Cambridge, MA, USA, 2017; pp. 553–563. ISBN 10: 0128007567. ISBN 13: 9780128007563. [Google Scholar]
- Lu, D.; Immadi, S.S.; Wu, Z.; Kendall, D.A. Translational potential of allosteric modulators targeting the cannabinoid CB1 receptor. Acta Pharmacol. Sin. 2019, 40, 324–335. [Google Scholar] [CrossRef]
- Pertwee, R.G. Endocannabinoids and their pharmacological actions. Handb. Exp. Pharm. 2015, 231, 1–37. [Google Scholar]
- Zou, S.; Kumar, U. Cannabinoid receptors and the endocannabinoid system: Signaling and function in the central nervous system. Int. J. Mol. Sci. 2018, 19, E833. [Google Scholar]
- Pertwee, R.G. Targeting the endocannabinoid system with cannabinoid receptor agonists: Pharmacological strategies and therapeutic possibilities. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2012, 367, 3353–3363. [Google Scholar] [CrossRef] [PubMed]
- Laprairie, R.; Kulkarni, A.; Kulkarni, P.; Hurst, D.; Lynch, D.; Reggio, P.H.; Janero, D.R.; Pertwee, R.G.; Stevenson, L.A.; Kelly, M.E.; et al. Mapping cannabinoid 1 receptor allosteric site(s): Critical molecular determinant and signaling profile of GAT100, a novel, potent, and irreversibly binding probe. ACS Chem. Neurosci. 2016, 7, 776–798. [Google Scholar] [CrossRef] [PubMed]
- Hunter, M.R.; Finlay, D.B.; Glass, M. Signaling and regulation of the cannabinoid CB1 receptor. In Handbook of Cannabis and Related Pathologies Biology, Pharmacology, Diagnosis, and Treatment; Academic Press: Cambridge, MA, USA, 2017; pp. 564–572. ISBN 10: 0128007567. ISBN 13: 9780128007563. [Google Scholar]
- Shao, Z.; Yin, J.; Chapman, K.; Grzemska, M.C.; Wang, J.; Rosenbaum, D.M. High-Resolution crystal structure of the human CB1 cannabinoid receptor. Nature 2016, 540, 602–606. [Google Scholar] [CrossRef]
- Hua, T.; Vemuri, K.; Pu, M.; Qu, L.; Han, G.W.; Wu, Y.; Zhao, S.; Shui, W.; Li, S.; Korde, A.; et al. Crystal structure of the human cannabinoid receptor CB1. Cell 2016, 167, 750–762. [Google Scholar] [CrossRef] [PubMed]
- Hua, T.; Vemuri, K.; Nikas, S.P.; Laprairie, R.B.; Wu, Y.; Qu, L.; Pu, M.; Korde, A.; Jiang, S.; Ho, J.H.; et al. Crystal structures of agonist-bound human cannabinoid receptor CB1. Nature 2017, 547, 468–471. [Google Scholar] [CrossRef]
- Li, X.; Hua, T.; Vemuri, K.; Ho, J.H.; Wu, Y.; Wu, L.; Popov, P.; Benchama, O.; Zvonok, N.; Locke, K.; et al. Crystal structure of the human cannabinoid receptor CB2. Cell 2019, 176, 459–467. [Google Scholar] [CrossRef]
- Kumar, K.K.; Shalev-Benami, M.; Robertson, M.J.; Hu, H.; Banister, S.D.; Hollingsworth, S.A.; Latorraca, N.R.; Kato, H.E.; Hilger, D.; Maeda, S.; et al. Structure of a signaling cannabinoid receptor 1-G protein complex. Cell 2019, 176, 448–458. [Google Scholar] [CrossRef]
- Changeux, J.P. The concept of allosteric modulation: An overview. Drug Discov. Today Technol. 2013, 10, 223–228. [Google Scholar] [CrossRef]
- Kenakin, T. Biased receptor signaling in drug discovery. Pharm. Rev. 2019, 71, 267–315. [Google Scholar] [CrossRef] [PubMed]
- Neubig, R.R.; Spedding, M.; Kenakin, T.; Christopoulos, A. International union pharmacology committee on receptor nomenclature and drug classification. XXXVIII. Update on terms and symbols in quantitative pharmacology. Pharm. Rev. 2003, 55, 597–606. [Google Scholar] [CrossRef] [PubMed]
- Kenakin, T. Inverse, protean, and ligand-selective agonism: Matters of receptor conformation. FASEB J. 2001, 15, 598–611. [Google Scholar] [CrossRef] [PubMed]
- May, L.T.; Leach, K.; Sexton, P.M.; Christopoulos, A. Allosteric modulation of G protein-coupled receptors. Ann. Rev. Pharm. Toxicol. 2007, 47, 1–51. [Google Scholar] [CrossRef]
- Wootten, D.; Christopoulos, A.; Sexton, P.M. Emerging paradigms in GPCR allostery: Implications for drug discovery. Nat. Rev. Drug Discov. 2013, 12, 630–644. [Google Scholar] [CrossRef]
- Alaverdashvili, M.; Laprairie, R.B. The future of type 1 cannabinoid receptor allosteric ligands. Drug Metab. Rev. 2018, 50, 14–25. [Google Scholar] [CrossRef]
- Cawston, E.E.; Hunter, M.R.; Glass, M. Allosteric modulation of the cannabinoid CB1 receptor. In Handbook of Cannabis and Related Pathologies Biology, Pharmacology, Diagnosis, and Treatment; Preedy, V.R., Ed.; Academic Press: Cambridge, MA, USA, 2017; pp. 573–583. ISBN 10: 0128007567. ISBN 13: 9780128007563. [Google Scholar]
- Dopart, R.; Lu, D.; Lichtman, A.H.; Kendall, D.A. Allosteric modulators of cannabinoid receptor 1: Developing compounds for improved specificity. Drug Metab. Rev. 2018, 50, 3–13. [Google Scholar] [CrossRef]
- Janero, D.R.; Thakur, G.A. Leveraging allostery to improve G protein-coupled receptor (GPCR)-directed therapeutics: Cannabinoid receptor 1 as discovery target. Expert Opin. Drug Discov. 2016, 11, 1223–1237. [Google Scholar] [CrossRef]
- Khurana, L.; Mackie, K.; Piomelli, D.; Kendall, D.A. Modulation of CB1 cannabinoid receptor by allosteric ligands: Pharmacology and therapeutic opportunities. Neuropharmacology 2017, 124, 3–12. [Google Scholar] [CrossRef]
- Nguyen, T.; Li, J.X.; Thomas, B.F.; Wiley, J.L.; Kenakin, T.P.; Zhang, Y. Allosteric modulation: An alternate approach targeting the cannabinoid CB1 receptor. Med. Res. Rev. 2017, 37, 441–474. [Google Scholar] [CrossRef]
- Roth, B.L.; Irwin, J.J.; Shoichet, B.K. Discovery of new GPCR ligands to illuminate new biology. Nat. Chem. Biol. 2017, 13, 1143–1151. [Google Scholar] [CrossRef] [PubMed]
- Scott, C.; Kendall, D. Assessing allosteric modulation of CB1 at the receptor and cellular levels. Methods Enzymol. 2017, 593, 317–342. [Google Scholar] [PubMed]
- Christopoulos, A.; Changeux, J.P.; Catterall, W.A.; Fabbro, D.; Burris, T.P.; Cidlowski, J.A.; Olsen, R.W.; Peters, J.A.; Neubig, R.R.; Pin, J.P.; et al. International Union of Basic and Clinical Pharmacology. XC: Multisite pharmacology: Recommendations for the nomenclature of receptor allosterism and allosteric ligands. Pharm. Rev. 2014, 66, 918–947. [Google Scholar] [CrossRef] [PubMed]
- Herkenham, M.; Lynn, A.B.; Little, M.D.; Johnson, M.R.; Melvin, L.S.; de Costa, B.R.; Rice, K.C. Cannabinoid receptor localization in brain. Proc. Natl. Acad. Sci. USA 1990, 87, 1932–1936. [Google Scholar] [CrossRef] [PubMed]
- Sañudo-Peña, M.C.; Strangman, N.M.; Mackie, K.; Walker, J.M.; Tsou, K. CB1 receptor localization in rat spinal cord and roots, dorsal root ganglion, and peripheral nerve. Zhongguo Yao Li Xue Bao Acta Pharmacol. Sin. 1999, 20, 1115–1120. [Google Scholar]
- Abood, M.E. Molecular biology of canabinoid receptors. Handb. Exp. Pharm. 2005, 168, 81–115. [Google Scholar]
- Console-Bram, L.; Marcu, J.; Abood, M.E. Cannabinoid receptors: Nomenclature and pharmacological principles. Progress Neuro Psychopharmacol. Biol. Psychiatry 2012, 38, 4–15. [Google Scholar] [CrossRef]
- Aghazadeh Tabrizi, M.; Baraldi, P.G. Chemistry of cannabinoid receptor agonists. In Handbook of Cannabis and Related Pathologies Biology, Pharmacology, Diagnosis, and Treatment; Academic Press: Cambridge, MA, USA, 2017; pp. 592–605. ISBN 10: 0128007567. ISBN 13: 9780128007563. [Google Scholar]
- Pertwee, R.G. Inverse agonism and neutral antagonism at cannabinoid CB1 receptors. Life Sci. 2005, 76, 1307–1324. [Google Scholar] [CrossRef]
- Morales, P.; Goya, P.; Jagerovic, N.; Hernandez-Folgado, L. Allosteric modulators of the CB1 cannabinoid receptor: A structural update review. Cannabis Cannabinoid Res. 2016, 1, 22–30. [Google Scholar] [CrossRef]
- Howlett, A.C. Cannabinoid receptor signaling. Handb. Exp. Pharm. 2005, 168, 53–79. [Google Scholar]
- Turu, G.; Hunyady, L. Signal transduction of the CB1 cannabinoid receptor. J. Mol. Endocrinol. 2010, 44, 75–85. [Google Scholar] [CrossRef] [PubMed]
- Kenakin, T. Agonist-Receptor efficacy. II. Agonist trafficking of receptor signals. Trends Pharm. Sci. 1995, 16, 232–238. [Google Scholar] [CrossRef]
- Reiter, E.; Ahn, S.; Shukla, A.K.; Lefkowitz, R.J. Molecular mechanism of β-arrestin-biased agonism at seven-transmembrane receptors. Annu. Rev. Pharm. Toxicol. 2012, 52, 179–197. [Google Scholar] [CrossRef]
- Shukla, A.K.; Singh, G.; Ghosh, E. Emerging structural insights into biased GPCR signaling. Trends Biochem. Sci. 2014, 39, 594–602. [Google Scholar] [CrossRef] [PubMed]
- Price, M.R.; Baillie, G.L.; Thomas, A.; Stevenson, L.A.; Easson, M.; Goodwin, R.; McLean, A.; McIntosh, L.; Goodwin, G.; Walker, G.; et al. Allosteric modulation of the cannabinoid CB1 receptor. Mol. Pharm. 2005, 68, 1484–1495. [Google Scholar] [CrossRef] [PubMed]
- Nogueras-Ortiz, C.; Yudowski, G.A. The multiple waves of cannabinoid 1 receptor signaling. Mol. Pharm. 2016, 90, 620–626. [Google Scholar] [CrossRef] [PubMed]
- Vecchio, E.A.; Baltos, J.A.; Nguyen, A.T.N.; Christopoulos, A.; White, P.J.; May, L.T. New paradigms in adenosine receptor pharmacology: Allostery, oligomerization and biased agonism. Br. J. Pharm. 2018, 175, 4036–4046. [Google Scholar] [CrossRef]
- Wang, W.; Qiao, Y.; Li, Z. New insights into modes of GPCR activation. Trends Pharm. Sci. 2018, 39, 367–386. [Google Scholar] [CrossRef]
- Pertwee, R.G.; Howlett, A.C.; Abood, M.E.; Alexander, S.P.; Di Marzo, V.; Elphick, M.R.; Greasley, P.J.; Hansen, H.S.; Kunos, G.; Mackie, K.; et al. International Union of Basic and Clinical Pharmacology. LXXIX. Cannabinoid receptors and their ligands: Beyond CB1 and CB2. Pharm. Rev. 2010, 62, 588–631. [Google Scholar] [CrossRef]
- Callén, L.; Moreno, E.; Barroso-Chinea, P.; Moreno-Delgado, D.; Cortés, A.; Mallol, J.; Casadó, V.; Lanciego, J.L.; Franco, R.; Lluis, C.; et al. Cannabinoid receptors CB1 and CB2 form functional heteromers in brain. J. Biol. Chem. 2012, 287, 20851–20865. [Google Scholar] [CrossRef]
- Sierra, S.; Gupta, A.; Gomes, I.; Fowkes, M.; Ram, A.; Bobeck, E.N.; Devi, L.A. Targeting cannabinoid 1 and delta opioid receptor heteromers alleviates chemotherapy-induced neuropathic pain. ACS Pharm. Transl. Sci. 2019, 2, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Oswald, C.; Rappas, M.; Kean, J.; Doré, A.S.; Errey, J.C.; Bennett, K.; Deflorian, F.; Christopher, J.A.; Jazayeri, A.; Mason, J.S.; et al. Intracellular allosteric antagonism of the CCR9 receptor. Nature 2016, 540, 462–465. [Google Scholar] [CrossRef] [PubMed]
- Bénard, G.; Massa, F.; Puente, N.; Lourenço, J.; Bellocchio, L.; Soria-Gómez, E.; Matias, I.; Delamarre, A.; Metna-Laurent, M.; Cannich, A.; et al. Mitochondrial CB1 receptors regulate neuronal energy metabolism. Nat. Neurosci. 2012, 15, 558–564. [Google Scholar] [CrossRef] [PubMed]
- Hebert-Chatelain, E.; Desprez, T.; Serrat, R.; Bellocchio, L.; Soria-Gomez, E.; Busquets-Garcia, A.; Pagano Zottola, A.C.; Delamarre, A.; Cannich, A.; Vincent, P.; et al. A cannabinoid link between mitochondria and memory. Nature 2016, 539, 555–559. [Google Scholar] [CrossRef] [PubMed]
- Wheatley, M.; Wootten, D.; Conner, M.T.; Simms, J.; Kendrick, R.; Logan, R.T.; Poyner, D.R.; Barwell, J. Lifting the lid on GPCRs: The role of extracellular loops. Br. J. Pharm. 2012, 165, 1688–1703. [Google Scholar] [CrossRef] [PubMed]
- Woolley, M.; Conner, A. Understanding the common themes and diverse roles of the second extracellular loop (ECL2) of the GPCR super-family. Mol. Cell Endocrinol. 2017, 449, 3–11. [Google Scholar] [CrossRef]
- Conner, M.; Hawtin, S.R.; Simms, J.; Wootten, D.; Lawson, Z.; Conner, A.C.; Parslow, R.A.; Wheatley, M. Systematic analysis of the entire second extracellular loop of the V(1a) vasopressin receptor: Key residues, conserved throughout a G-protein coupled receptor family, identified. J. Biol. Chem. 2007, 282, 17405–17412. [Google Scholar] [CrossRef]
- Ahuja, S.; Hornak, V.; Yan, E.C.; Syrett, N.; Goncalves, J.A.; Hirshfeld, A.; Ziliox, M.; Sakmar, T.P.; Sheves, M.; Reeves, P.J.; et al. Helix movement is coupled to displacement of the second extracellular loop in rhodopsin activation. Nat. Struct Mol. Biol. 2009, 16, 168–175. [Google Scholar] [CrossRef]
- Avlani, V.A.; Gregory, K.J.; Morton, C.J.; Parker, M.W.; Sexton, P.M.; Christopoulos, A. Critical role for the second extracellular loop in the binding of both orthosteric and allosteric G protein-coupled receptor ligands. J. Biol. Chem. 2007, 282, 25677–25686. [Google Scholar] [CrossRef]
- Peeters, M.C.; van Westen, G.J.; Li, Q.; Ijzerman, A.P. Importance of the extracellular loops in G protein-coupled receptors for ligand recognition and receptor activation. Trends Pharm. Sci. 2011, 32, 35–42. [Google Scholar] [CrossRef]
- Nguyen, A.T.; Baltos, J.A.; Thomas, T.; Nguyen, T.D.; Muñoz, L.L.; Gregory, K.J.; White, P.J.; Sexton, P.M.; Christopoulos, A.; May, L.T. Extracellular loop 2 of the adenosine A1 receptor has a key role in orthosteric ligand affinity and agonist efficacy. Mol. Pharm. 2016, 90, 703–714. [Google Scholar] [CrossRef] [PubMed]
- Murakami, M.; Kouyama, T. Crystal structure of squid rhodopsin. Nature 2008, 453, 363–367. [Google Scholar] [CrossRef] [PubMed]
- Ahn, K.; Bertalovitz, A.; Mierke, D.; Kendall, D. Dual role of the second extracellular loop of the cannabinoid receptor 1: Ligand binding and receptor localization. Mol. Pharm. 2009, 76, 833–842. [Google Scholar] [CrossRef] [PubMed]
- Shim, J.Y. Understanding functional residues of the cannabinoid CB1. Curr Top. Med. Chem. 2010, 10, 779–798. [Google Scholar] [CrossRef] [PubMed]
- Joost, P.; Methner, A. Phylogenetic analysis of 277 human G-protein- coupled receptors as a tool for the prediction of orphan receptor ligands. Genome Biol. 2002, 3, 0063.1-16. [Google Scholar] [CrossRef]
- Fredriksson, R.; Lagerström, M.C.; Lundin, L.G.; Schiöth, H.B. The G-protein-coupled receptors in the human genome form five main families. Phylogenetic analysis, paralogon groups, and fingerprints. Mol. Pharmacol. 2003, 63, 1256–1272. [Google Scholar] [CrossRef]
- Fay, J.; Dunham, T.; Farrens, D. Cysteine residues in the human cannabinoid receptor: Only C257 and C264 are required for a functional receptor, and steric bulk at C386 impairs antagonist SR141716A binding. Biochemistry 2005, 44, 8757–8769. [Google Scholar] [CrossRef]
- McAllister, S.; Hurst, D.; Barnett-Norris, J.; Lynch, D.; Reggio, P.; Abood, M.E. Structural mimicry in class A G protein-coupled receptor rotamer toggle switches: The importance of the F3.36/W6.48 interaction in cannabinoid CB1 receptor activation. J. Biol. Chem. 2004, 279, 48024–48037. [Google Scholar] [CrossRef]
- Bertalovitz, A.C.; Ahn, K.H.; Kendall, D.A. Ligand binding sensitivity of the extracellular loop two of the cannabinoid receptor 1. Drug Dev. Res. 2010, 71, 404–411. [Google Scholar] [CrossRef]
- Fay, J.; Farrens, D. The membrane proximal region of the cannabinoid receptor CB1 N-terminus can allosterically modulate ligand affinity. Biochemistry 2013, 52, 8286–8294. [Google Scholar] [CrossRef]
- Gyombolai, P.; Tóth, A.D.; Tímár, D.; Turu, G.; Hunyady, L. Mutations in the “DRY” motif of the CB1 cannabinoid receptor result in biased receptor variants. J. Mol. Endocrinol. 2015, 54, 75–89. [Google Scholar] [CrossRef] [PubMed]
- Al-Zoubi, R.; Morales, P.; Reggio, P.H. Structural insights into CB1 receptor biased signaling. Int J. Mol. Sci. 2019, 20, 1837. [Google Scholar] [CrossRef] [PubMed]
- Marcu, J.; Shore, D.; Kapur, A.; Trznadel, M.; Makriyannis, A.; Reggio, P.H.; Abood, M.E. Novel insights into CB1 cannabinoid receptor signaling: A key interaction identified between the extracellular-3 loop and transmembrane helix 2. J. Pharm. Exp. 2013, 345, 189–197. [Google Scholar] [CrossRef]
- Shore, D.; Baillie, G.; Hurst, D.; Navas, F., 3rd; Seltzman, H.H.; Marcu, J.P.; Abood, M.E.; Ross, R.A.; Reggio, P. Allosteric modulation of a cannabinoid G protein-coupled receptor: Binding site elucidation and relationship to G protein signaling. J. Biol. Chem. 2014, 289, 5828–5854. [Google Scholar] [CrossRef] [PubMed]
- Horswill, J.G.; Bali, U.; Shaaban, S.; Keily, J.F.; Jeevaratnam, P.; Babbs, A.J.; Reynet, C.; Wong Kai In, P. PSNCBAM-1, a novel allosteric antagonist at cannabinoid CB1 receptors with hypophagic effects in rats. Br. J. Pharm. 2007, 152, 805–814. [Google Scholar] [CrossRef] [PubMed]
- German, N.; Decker, A.M.; Gilmour, B.P.; Gay, E.A.; Wiley, J.L.; Thomas, B.F.; Zhang, Y. Diarylureas as allosteric modulators of the cannabinoid CB1 receptor: Structure-Activity relationship studies on 1-(4-chlorophenyl)-3-{3-[6-(pyrrolidin-1-yl)pyridin-2-yl]phenyl}urea (PSNCBAM-1). J. Med. Chem. 2014, 57, 7758–7769. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, P.M.; Kulkarni, A.R.; Korde, A.; Tichkule, R.B.; Laprairie, R.B.; Denovan-Wright, E.M.; Zhou, H.; Janero, D.R.; Zvonok, N.; Makriyannis, A.; et al. Novel electrophilic and photoaffinity covalent probes for mapping the cannabinoid 1 receptor allosteric site(s). J. Med. Chem. 2016, 59, 44–60. [Google Scholar] [CrossRef]
- Bertini, S.; Chicca, A.; Gado, F.; Arena, C.; Nieri, D.; Digiacomo, M.; Saccomanni, G.; Zhao, P.; Abood, M.E.; Macchia, M.; et al. Novel analogs of PSNCBAM-1 as allosteric modulators of cannabinoid CB1 receptor. Bioorg. Med. Chem. 2017, 25, 6427–6434. [Google Scholar] [CrossRef]
- Pamplona, F.A.; Ferreira, J.; Menezes de Lima, O., Jr.; Duarte, F.S.; Bento, A.F.; Forner, S.; Villarinho, J.G.; Bellocchio, L.; Wotjak, C.T.; Lerner, R.; et al. Anti-Inflammatory lipoxin a4 is an endogenous allosteric enhancer of CB1 cannabinoid receptor. Proc. Natl Acad Sci. USA 2012, 109, 21134–21139. [Google Scholar] [CrossRef]
- Straiker, A.; Mitjavila, J.; Yin, D.; Gibson, A.; Mackie, K. Aiming for allosterism: Evaluation of allosteric modulators of CB1 in a neuronal model. Pharm. Res. 2015, 99, 370–376. [Google Scholar] [CrossRef]
- Laprairie, R.; Bagher, A.; Kelly, M.; Denovan-Wright, E.M. Cannabidiol is a negative allosteric modulator of the cannabinoid CB1 receptor. Br. J. Pharm. 2015, 172, 4790–4805. [Google Scholar] [CrossRef] [PubMed]
- Navarro, H.; Howard, J.; Pollard, G.; Carroll, F. Positive allosteric modulation of the human cannabinoid (CB) receptor by RTI-371, a selective inhibitor of the dopamine transporter. Br. J. Pharm. 2009, 156, 1178–1184. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Ruiz, J.; Lastres-Becker, I.; Cabranes, A.; González, S.; Ramos, J. Endocannabinoids and basal ganglia functionality. Prostaglandins Leukot. Essent Fat. Acids 2002, 66, 257–267. [Google Scholar] [CrossRef] [PubMed]
- Slivicki, R.A.; Xu, Z.; Kulkarni, P.M.; Pertwee, R.G.; Mackie, K.; Thakur, G.A.; Hohmann, A.G. Positive allosteric modulation of CB1 suppresses pathological pain without producing tolerance or dependence. Biol. Psychiatry 2018, 84, 722–733. [Google Scholar] [CrossRef]
- Laprairie, R.B.; Kulkarni, P.M.; Deschamps, J.R.; Kelly, M.E.M.; Janero, D.R.; Cascio, M.G.; Stevenson, L.A.; Pertwee, R.G.; Kenakin, T.P.; Denovan-Wright, E.M.; et al. Enantiospecific allosteric modulation of cannabinoid 1 receptor. ACS Chem. Neurosci. 2017, 8, 1188–1203. [Google Scholar] [CrossRef]
- Mallipeddi, S.; Janero, D.R.; Zvonok, N.; Makriyannis, A. Functional selectivity at G-protein coupled receptors: Advancing cannabinoid receptors as drug targets. Biochem. Pharm. 2017, 128, 1–11. [Google Scholar] [CrossRef]
- Wootten, D.; Christopoulos, A.; Marti-Solano, M.; Babu, M.M.; Sexton, P.M. Mechanisms of signalling and biased agonism in G protein-coupled receptors. Nat. Rev. Mol. Cell Biol. 2018, 19, 638–653. [Google Scholar] [CrossRef]
- Smith, J.S.; Lefkowitz, R.J.; Rajagopal, S. Biased signalling: From simple switches to allosteric microprocessors. Nat. Rev. Drug Discov. 2018, 17, 243–260. [Google Scholar] [CrossRef]
- Bagher, A.M.; Laprairie, R.B.; Kelly, M.E.; Denovan-Wright, E.M. Antagonism of dopamine receptor 2 long affects cannabinoid receptor 1 signaling in a cell culture model of striatal medium spiny projection neurons. Mol. Pharm. 2016, 89, 652–666. [Google Scholar] [CrossRef]
- Ogawa, G.; Tius, M.A.; Zhou, H.; Nikas, S.P.; Halikhedkar, A.; Mallipeddi, S.; Makriyannis, A. 30-functionalized adamantyl cannabinoid receptor probes. J. Med. Chem. 2015, 58, 3104–3116. [Google Scholar] [CrossRef]
- Nimczick, M.; Decker, M. New approaches in the design and development of cannabinoid receptor ligands: Multifunctional and bivalent compounds. Chem. Med. Chem. 2015, 10, 773–786. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, A.R.; Garai, S.; Janero, D.R.; Thakur, G.A. Design and synthesis of cannabinoid 1 receptor (CB1R) allosteric modulators: Drug discovery applications. Methods Enzym. 2017, 593, 281–315. [Google Scholar]
- Kenakin, T. Signaling bias in drug discovery. Expert Opin. Drug Discov. 2017, 12, 321–333. [Google Scholar] [CrossRef] [PubMed]
| Compound | IUPAC Nomenclature |
|---|---|
| CP 55,940 | [2-((1R,2R,5R)-5-Hydroxy-2-(3-hydroxypropyl) cyclohexyl)-5-(2-methyloctan-2-yl)phenol] |
| HU-210 | [(6aR,10aR)-9-(Hydroxymethyl)-6,6-dimethyl-3-(2-methyloctan-2-yl)-6a,7,10,10a-tetrahydro-6H-benzo [c]chromen-1-ol] |
| WIN 55212-2 | [(R)-(5-Methyl-3-(morpholinomethyl)-2,3-dihydro-[1,4]oxazino [2,3,4-hi]indol-6-yl)(naphthalen-1-yl)methanone] |
| JWH-018 | [Naphthalen-1-yl-(1-pentylindol-3-yl)methanone] |
| SR-141716A (rimonabant) | [5-(4-Chlorophenyl)-1-(2,4-dichlorophenyl)-4-methyl-N-(1-piperidinyl)-1H-pyrazole-3-carboxamide] |
| AM251 | [1-(2,4-Dichlorophenyl)-5-(4-iodophenyl)-4-methyl-N-(1-piperidinyl)-1H-pyrazole-3-carboxamide] |
| Human Homo Sapiens (NC_000006.12) vs | CNR1 Gene | ECL2 Region | |
|---|---|---|---|
| Coverage | Identity | Identity | |
| Chimpanzee Pan troglodytes (NC-006473.4) | 99% | 99% | 99% |
| Pig Sus scrofa (NC_010443.5) | 56% | 85% | 88% |
| House mouse Mus musculus (NC_000070.6) | 29% | 83% | 92% |
| Norway rat Rattus norvegicus(NC_005104.4) | 28% | 83% | 92% |
| Dog Canis lupus familiaris(NC_030681.1) | 50% | 84% | 92% |
| Tropical clawed frog Xenopus tropicalis (NC_030681.1) | 4% | 76% | 78% |
| Zebrafish Danio rerio (NC_007131.7) | 3% | 76% | 72% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hryhorowicz, S.; Kaczmarek-Ryś, M.; Andrzejewska, A.; Staszak, K.; Hryhorowicz, M.; Korcz, A.; Słomski, R. Allosteric Modulation of Cannabinoid Receptor 1—Current Challenges and Future Opportunities. Int. J. Mol. Sci. 2019, 20, 5874. https://doi.org/10.3390/ijms20235874
Hryhorowicz S, Kaczmarek-Ryś M, Andrzejewska A, Staszak K, Hryhorowicz M, Korcz A, Słomski R. Allosteric Modulation of Cannabinoid Receptor 1—Current Challenges and Future Opportunities. International Journal of Molecular Sciences. 2019; 20(23):5874. https://doi.org/10.3390/ijms20235874
Chicago/Turabian StyleHryhorowicz, Szymon, Marta Kaczmarek-Ryś, Angelika Andrzejewska, Klaudia Staszak, Magdalena Hryhorowicz, Aleksandra Korcz, and Ryszard Słomski. 2019. "Allosteric Modulation of Cannabinoid Receptor 1—Current Challenges and Future Opportunities" International Journal of Molecular Sciences 20, no. 23: 5874. https://doi.org/10.3390/ijms20235874
APA StyleHryhorowicz, S., Kaczmarek-Ryś, M., Andrzejewska, A., Staszak, K., Hryhorowicz, M., Korcz, A., & Słomski, R. (2019). Allosteric Modulation of Cannabinoid Receptor 1—Current Challenges and Future Opportunities. International Journal of Molecular Sciences, 20(23), 5874. https://doi.org/10.3390/ijms20235874





