Rapid Regulation of Human Mesenchymal Stem Cell Proliferation Using Inducible Caspase-9 Suicide Gene for Safe Cell-Based Therapy
Abstract
1. Introduction
2. Results
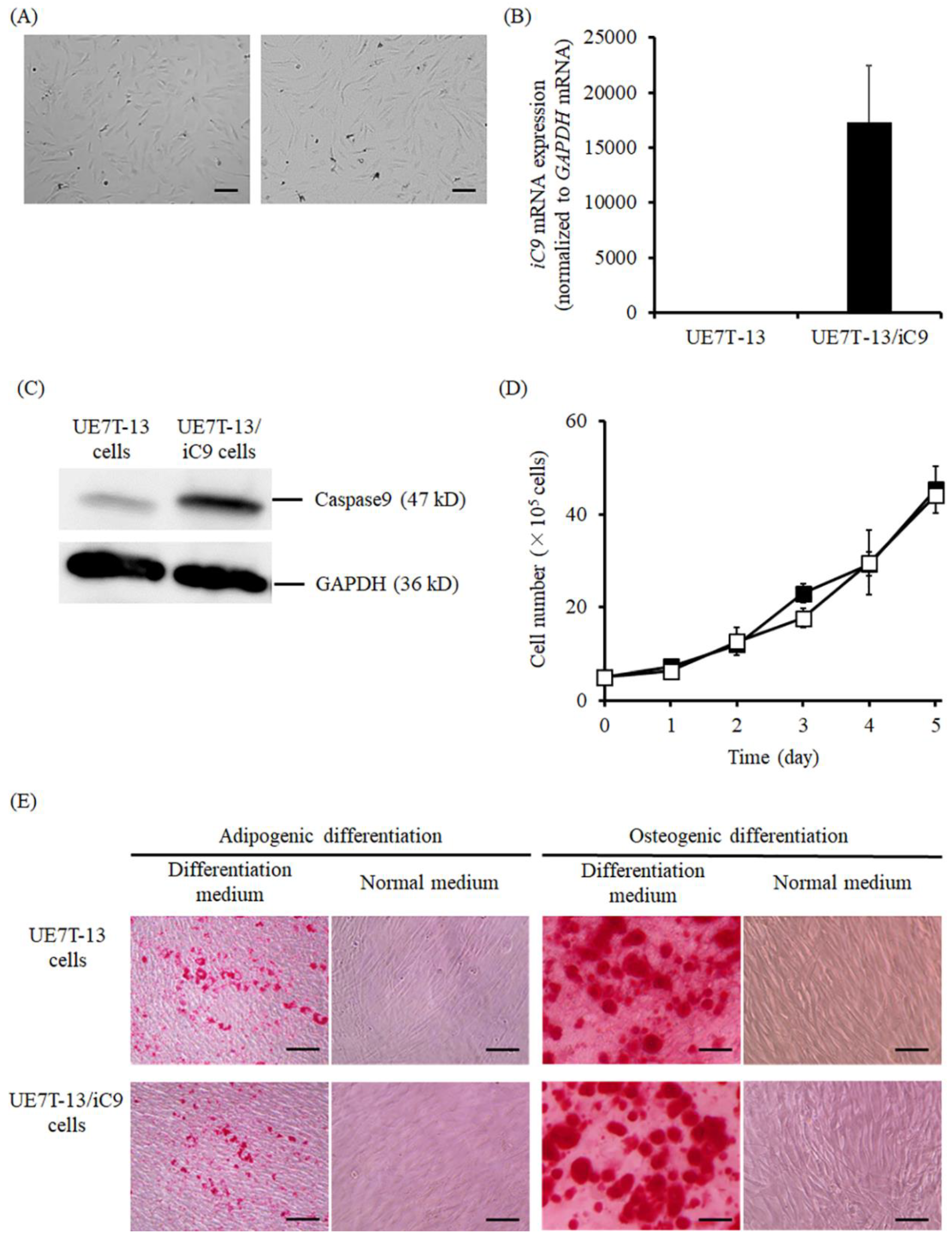
2.1. Characteristics of UE7T-13/iC9 Cells
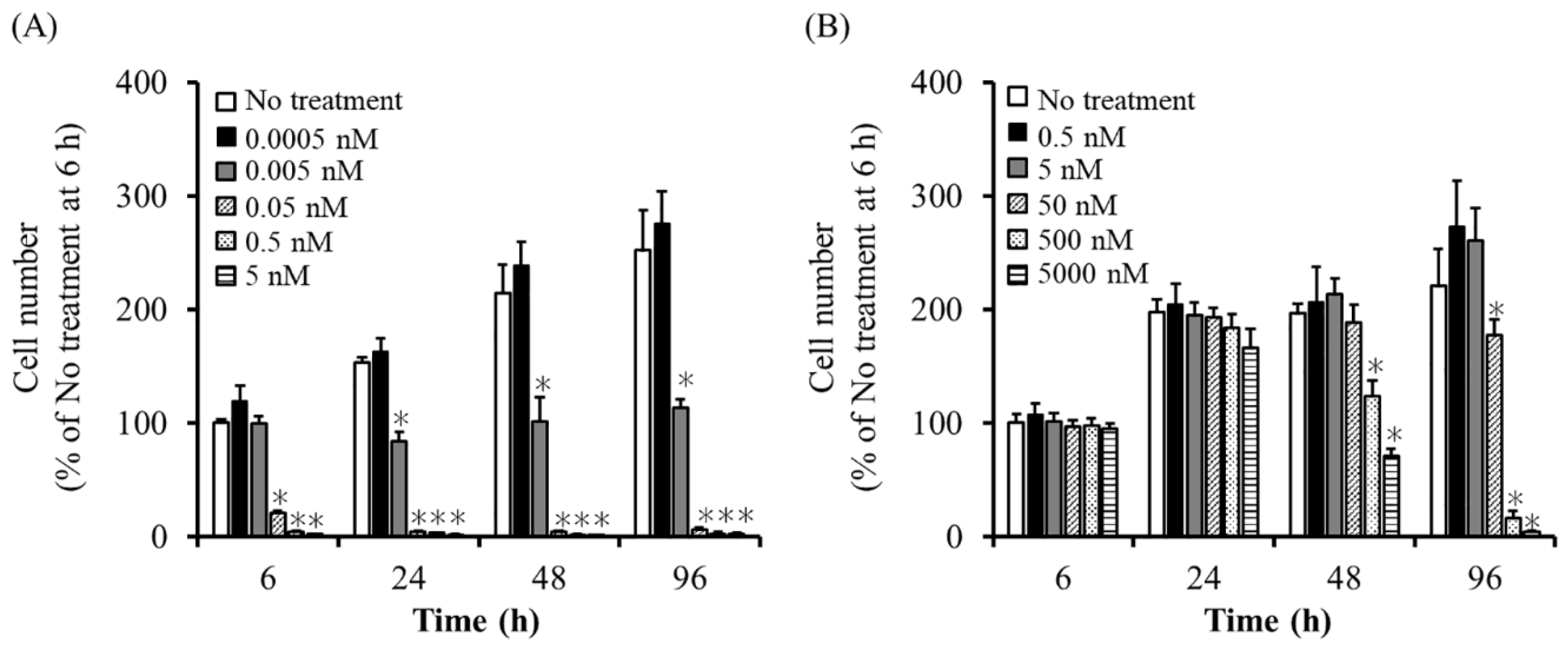
2.2. Sensitivity of UE7T-13/iC9 Cells to AP20187 and UE7T-13/HSVtk Cells to GCV
2.3. Effect of AP20187 on the Proliferation of UE7T-13/iC9 and UE7T-13/iC9/Nluc Cells
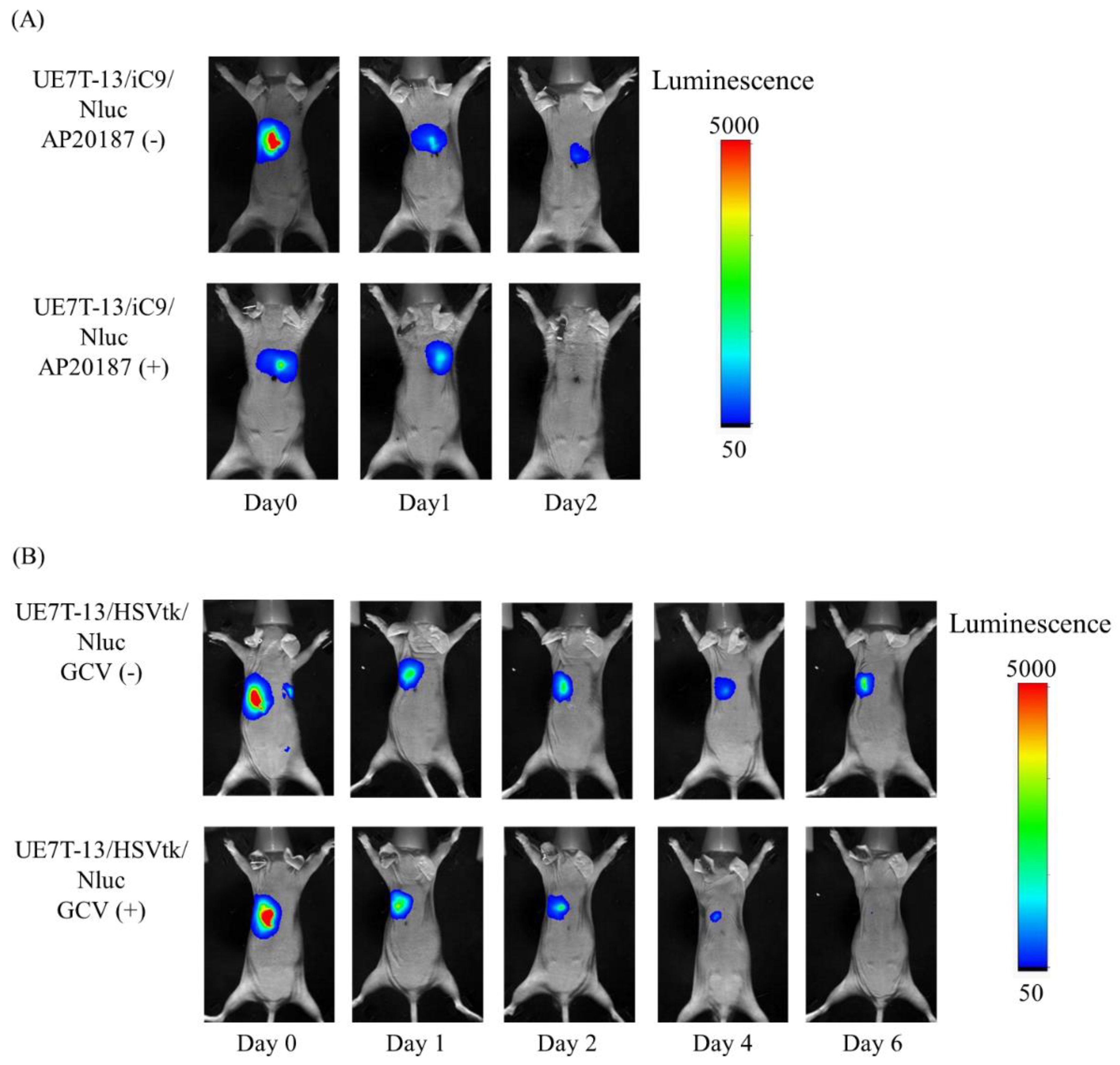
2.4. Elimination of UE7T-13/iC9/Nluc Cells by AP20187 and UE7T-13/HSVtk/Nluc Cells by GCV
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Materials
4.3. Cell Culture
4.4. Construction of Plasmids
4.5. Establishment of Cells
4.6. The mRNA Expression of the iC9 Transgene
4.7. Western Blotting
4.8. Cell Proliferation
4.9. Cell Differentiation
4.10. Sensitivity of UE7T-13/iC9 Cells to AP20187 and UE7T-13/HSVtk Cells to GCV
4.11. In Vitro Regulation of UE7T-13/iC9 Cell Proliferation
4.12. Elimination of UE7T-13/iC9/Nluc Cells and UE7T-13/HSVtk/Nluc Cells in Mice
4.13. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| iPS cell | induced pluripotent stem cell |
| ES cell | embryonic stem cell |
| HSVtk | herpes simplex virus thymidine kinase |
| GCV | ganciclovir |
| iC9 | inducible caspase-9 |
| CID | chemical inducer of dimerization |
| FBS | fetal bovine serum |
| DMEM | Dulbecco’s modified Eagle’s medium |
| PCR | polymerase chain reaction |
| Nluc | NanoLuc luciferase |
References
- Assinck, P.; Duncan, G.J.; Hilton, B.J.; Plemel, J.R.; Tetzlaff, W. Cell transplantation therapy for spinal cord injury. Nat. Neurosci. 2017, 25, 637–647. [Google Scholar] [CrossRef] [PubMed]
- Trounson, A.; McDonald, C. Stem Cell Therapies in Clinical Trials: Progress and Challenges. Cell Stem Cell 2015, 17, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Mead, B.; Berry, M.; Logan, A.; Scott, R.A.; Leadbeater, W.; Scheven, B.A. Stem cell treatment of degenerative eye disease. Stem Cell Res. 2015, 14, 243–257. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Inoue, H.; Wu, J.C.; Yamanaka, S. Induced pluripotent stem cell technology: A decade of progress. Nat. Rev. Drug Discov. 2017, 16, 115–130. [Google Scholar] [CrossRef] [PubMed]
- Mandai, M.; Watanabe, A.; Kurimoto, Y.; Hirami, Y.; Morinaga, C.; Daimon, T.; Fujihara, M.; Akimaru, H.; Sakai, N.; Shibata, Y.; et al. Autologous Induced Stem-Cell-Derived Retinal Cells for Macular Degeneration. N. Engl. J. Med. 2017, 376, 1038–1046. [Google Scholar] [CrossRef]
- Barker, R.A.; Parmar, M.; Studer, L.; Takahashi, J. Human Trials of Stem Cell-Derived Dopamine Neurons for Parkinson’s Disease: Dawn of a New Era. Cell Stem Cell 2017, 21, 569–573. [Google Scholar] [CrossRef]
- Brudno, J.N.; Kochenderfer, J.N. Chimeric antigen receptor T-cell therapies for lymphoma. Nat. Rev. Clin. Oncol. 2018, 15, 31–46. [Google Scholar] [CrossRef]
- Gardner, R.A.; Finney, O.; Annesley, C.; Brakke, H.; Summers, C.; Leger, K.; Bleakley, M.; Brown, C.; Mgebroff, S.; Kelly-Spratt, K.S.; et al. Intent-to-treat leukemia remission by CD19 CAR T cells of defined formulation and dose in children and young adults. Blood 2017, 129, 3322–3331. [Google Scholar] [CrossRef]
- Fry, T.J.; Shah, N.N.; Orentas, R.J.; Stetler-Stevenson, M.; Yuan, C.M.; Ramakrishna, S.; Wolters, P.; Martin, S.; Delbrook, C.; Yates, B.; et al. CD22-targeted CAR T cells induce remission in B-ALL that is naive or resistant to CD19-targeted CAR immunotherapy. Nat. Med. 2018, 24, 20–28. [Google Scholar] [CrossRef]
- Rota, C.; Morigi, M.; Imberti, B. Stem Cell Therapies in Kidney Diseases: Progress and Challenges. Int. J. Mol. Sci. 2019, 20, 2790. [Google Scholar] [CrossRef]
- Wang, J.; Sun, M.; Liu, W.; Li, Y.; Li, M. Stem Cell-Based Therapies for Liver Diseases: An Overview and Update. Tissue Eng. Regen. Med. 2019, 16, 107–118. [Google Scholar] [CrossRef] [PubMed]
- Ghiroldi, A.; Piccoli, M.; Cirillo, F.; Monasky, M.M.; Ciconte, G.; Pappone, C.; Anastasia, L. Cell-Based Therapies for Cardiac Regeneration: A Comprehensive Review of Past and Ongoing Strategies. Int. J. Mol. Sci. 2018, 19, 3194. [Google Scholar] [CrossRef] [PubMed]
- Volkman, R.; Offen, D. Concise Review: Mesenchymal Stem Cells in Neurodegenerative Diseases. Stem Cells 2017, 35, 1867–1880. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez-Aranda, I.; Ramos-Mejia, V.; Bueno, C.; Munoz-Lopez, M.; Real, P.J.; Mácia, A.; Sanchez, L.; Ligero, G.; Garcia-Parez, J.L.; Menendez, P. Human induced pluripotent stem cells develop teratoma more efficiently and faster than human embryonic stem cells regardless the site of injection. Stem Cells 2010, 28, 1568–1570. [Google Scholar] [CrossRef]
- Masuda, S.; Miyagawa, S.; Fukushima, S.; Sougawa, N.; Okimoto, K.; Tada, C.; Saito, A.; Sawa, Y. Eliminating residual iPS cells for safety in clinical application. Protein Cell 2015, 6, 469–471. [Google Scholar] [CrossRef][Green Version]
- Tan, H.L.; Fong, W.J.; Lee, E.H.; Yap, M.; Choo, A. mAb 84, a cytotoxic antibody that kills undifferentiated human embryonic stem cells via oncosis. Stem Cells 2009, 27, 1792–1801. [Google Scholar] [CrossRef]
- Tang, C.; Lee, A.S.; Volkmer, J.P.; Sahoo, D.; Nag, D.; Mosley, A.R.; Inlay, M.A.; Ardehali, R.; Chavez, S.L.; Pera, R.R.; et al. An antibody against SSEA-5 glycan on human pluripotent stem cells enables removal of teratoma-forming cells. Nat. Biotechnol. 2011, 14, 829–834. [Google Scholar] [CrossRef]
- Farney, A.C.; Sutherland, D.E.; Opara, E.C. Evolution of Islet Transplantation for the Last 30 Years. Pancreas 2016, 45, 8–20. [Google Scholar] [CrossRef]
- Shapiro, A.M.; Pokrywczynska, M.; Ricordi, C. Clinical pancreatic islet transplantation. Nat. Rev. Endocrinol. 2017, 13, 268–277. [Google Scholar] [CrossRef]
- Bottino, R.; Bertera, S.; Grupillo, M.; Melvin, P.R.; Humar, A.; Mazariegos, G.; Moser, A.J.; Walsh, R.M.; Fung, J.; Gelrud, A.; et al. Isolation of human islets for autologous islet transplantation in children and adolescents with chronic pancreatitis. J. Transplant. 2012, 2012, 642787. [Google Scholar] [CrossRef]
- Tsujimura, M.; Kusamori, K.; Oda, C.; Miyazaki, A.; Katsumi, H.; Sakane, T.; Nishikawa, M.; Yamamoto, A. Regulation of proliferation and functioning of transplanted cells by using herpes simplex virus thymidine kinase gene in mice. J. Control. Release 2018, 275, 78–84. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, T.; Sifontis, N. Drug interactions and toxicities associated with the antiviral management of cytomegalovirus infection. Am. J. Health Syst. Pharm. 2010, 67, 1417–1425. [Google Scholar] [CrossRef]
- Kimberlin, D.W.; Jester, P.M.; Sánchez, P.J.; Ahmed, A.; Arav-Boger, R.; Michaels, M.G.; Ashouri, N.; Englund, J.A.; Estrada, B.; Jacobs, R.F.; et al. Valganciclovir for symptomatic congenital cytomegalovirus disease. N. Engl. J. Med. 2015, 372, 933–943. [Google Scholar] [CrossRef] [PubMed]
- Berger, C.; Flowers, M.E.; Warren, E.H.; Riddell, S.R. Analysis of transgene-specific immune responses that limit the in vivo persistence of adoptively transferred HSV-TK-modified donor T cells after allogeneic hematopoietic cell transplantation. Blood 2006, 107, 2294–2302. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Hong, S.G.; Winkler, T.; Spencer, D.M.; Jares, A.; Ichwan, B.; Nicolae, A.; Guo, V.; Larochelle, A.; Dunbar, C.E. Development of an inducible caspase-9 safety switch for pluripotent stem cell-based therapies. Mol. Ther. Methods Clin. Dev. 2014, 1, 14053. [Google Scholar] [CrossRef]
- Junker, K.; Koehl, U.; Zimmerman, S.; Stein, S.; Schwabe, D.; Klingebiel, T.; Grez, M. Kinetics of cell death in T lymphocytes genetically modified with two novel suicide fusion genes. Gene Ther. 2003, 10, 1189–1197. [Google Scholar] [CrossRef]
- Straathof, K.C.; Pulè, M.A.; Yotnda, P.; Dotti, G.; Vanin, E.F.; Brenner, M.K.; Heslop, H.E.; Spencer, D.M.; Rooney, C.M. An inducible caspase 9 safety switch for T-cell therapy. Blood 2005, 105, 4247–4254. [Google Scholar] [CrossRef]
- Ramos, C.A.; Asgari, Z.; Liu, E.; Yvon, E.; Heslop, H.E.; Rooney, C.M.; Brenner, M.K.; Dotti, G. An inducible caspase 9 suicide gene to improve the safety of mesenchymal stromal cell therapies. Stem Cells 2010, 28, 1107–1115. [Google Scholar] [CrossRef]
- Di Stasi, A.; Tey, S.K.; Dotti, G.; Fujita, Y.; Kennedy-Nasser, A.; Martinez, C.; Straathof, K.; Liu, E.; Durett, A.G.; Grilley, B.; et al. Inducible apoptosis as a safety switch for adoptive cell therapy. N. Engl. J. Med. 2011, 365, 1673–1683. [Google Scholar] [CrossRef]
- Zhou, X.; Dotti, G.; Krance, R.A.; Martinez, C.A.; Naik, S.; Kamble, R.T.; Durett, A.G.; Dakhova, O.; Savoldo, B.; Di Stasi, A. Inducible caspase-9 suicide gene controls adverse effects from alloreplete T cells after haploidentical stem cell transplantation. Blood 2015, 125, 4103–4113. [Google Scholar] [CrossRef]
- Lukianova-Hleb, E.Y.; Yvon, E.S.; Shpall, E.J.; Lapotko, D.O. All-in-one processing of heterogeneous human cell grafts for gene and cell therapy. Mol. Ther. Methods Clin. Dev. 2016, 3, 16012. [Google Scholar] [CrossRef] [PubMed]
- Ando, M.; Hoyos, V.; Yagyu, S.; Tao, W.; Ramos, C.A.; Dotti, G.; Brenner, M.K.; Bouchier-Hayes, L. Bortezomib sensitizes non-small cell lung cancer to mesenchymal stromal cell-delivered inducible caspase-9-mediated cytotoxicity. Cancer Gene Ther. 2014, 21, 472–482. [Google Scholar] [CrossRef] [PubMed]
- Baron, U.; Bujard, H. Tet repressor-based system for regulated gene expression in eukaryotic cells: Principles and advances. Methods Enzymol. 2000, 327, 401–421. [Google Scholar]
- Lottmann, H.; Vanselow, J.; Hessabi, B.; Walther, R. The Tet-On system in transgenic mice: Inhibition of the mouse pdx-1 gene activity by antisense RNA expression in pancreatic beta-cells. J. Mol. Med. 2001, 79, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Xia, X.; Ayala, M.; Thiede, B.R.; Zhang, S.C. In vitro- and in vivo-induced transgene expression in human embryonic stem cells and derivatives. Stem Cells 2008, 26, 525–533. [Google Scholar] [CrossRef]
- Sakemura, R.; Terakura, S.; Watanabe, K.; Julamanee, J.; Takagi, E.; Miyao, K.; Koyama, D.; Goto, T.; Hanajiri, R.; Nishida, T.; et al. A Tet-On Inducible System for Controlling CD19-Chimeric Antigen Receptor Expression upon Drug Administration. Cancer Immunol. Res. 2016, 4, 658–668. [Google Scholar] [CrossRef] [PubMed]
- Bedel, A.; Beliveau, F.; Lamrissi-Garcia, I.; Rousseau, B.; Moranvillier, I.; Rucheton, B.; Guyonnet-Dupérat, V.; Cardinaud, B.; de Verneuil, H.; Moreau-Gaudry, F.; et al. Preventing Pluripotent Cell Teratoma in Regenerative Medicine Applied to Hematology Disorders. Stem Cells Transl. Med. 2017, 6, 382–393. [Google Scholar] [CrossRef]
- Traversari, C.; Marktel, S.; Magnani, Z.; Mangia, P.; Russo, V.; Ciceri, F.; Bonini, C.; Bordignon, C. The potential immunogenicity of the TK suicide gene does not prevent full clinical benefit associated with the use of TK-transduced donor lymphocytes in HSCT for hematologic malignancies. Blood 2007, 109, 4708–4715. [Google Scholar] [CrossRef]
- Jones, B.S.; Lamb, L.S.; Goldman, F.; Di Stasi, A. Improving the safety of cell therapy products by suicide gene transfer. Front. Pharmacol. 2014, 5, 254. [Google Scholar] [CrossRef]
- Yang, W.; Rozamus, L.W.; Narula, S.; Rollins, C.T.; Yuan, R.; Andrade, L.J.; Ram, M.K.; Phillips, T.B.; van Schravendijk, M.R.; Dalgarno, D.; et al. Investigating protein-ligand interactions with a mutant FKBP possessing a designed specificity pocket. J. Med. Chem. 2000, 43, 1135–1142. [Google Scholar] [CrossRef]
- Richard, R.E.; De Claro, R.A.; Yan, J.; Chien, S.; Von Recum, H.; Morris, J.; Kiem, H.P.; Dalgarno, D.C.; Heimfeld, S.; Clackson, T.; et al. Differences in F36VMpl-based in vivo selection among large animal models. Mol. Ther. 2004, 10, 730–740. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Strober, W. Trypan Blue Exclusion Test of Cell Viability. Curr. Protoc. Immunol. 1997, 21. [Google Scholar] [CrossRef]

| Primers for Construction of Plasmids | |
| iC9 | |
| Forward | 5′- TGCTCTAGAATGCTCGAGGGAGTGC -3′ |
| Reverse | 5′- TAAAGCGGCCGCTTAGTCGAGTGCG -3′ |
| HSVtk | |
| Forward | 5′- CCGGAATTCATGGCTTCTTACCCTG -3′ |
| Reverse | 5′- TAAAGCGGCCGCTTAGTTGGCCTCT -3′ |
| Nluc | |
| Forward | 5′- TGCTCTAGAATGAACTCCTTCTCCACAAG -3′ |
| Reverse | 5′- TAAAGCGGCCGCTTACGCCAGAATGCGTT -3′ |
| Primers for RT-PCR | |
| GAPDH | |
| Forward | 5′- GCACCGTCAAGGCTGAGAAC -3′ |
| Reverse | 5′- ATGGTGGTGAAGACGCCAGT -3′ |
| iC9 | |
| Forward | 5′- TGTGGGTCAGAGAGCCAAAC-3′ |
| Reverse | 5′- CAAATCTGCATTTCCCCTCA -3′ |
| HSVtk | |
| Forward | 5′- AACATCTACACCACCCAGCAC -3′ |
| Reverse | 5′- GAACAGCATCAGTCACAGCATAG -3′ |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsujimura, M.; Kusamori, K.; Nishikawa, M. Rapid Regulation of Human Mesenchymal Stem Cell Proliferation Using Inducible Caspase-9 Suicide Gene for Safe Cell-Based Therapy. Int. J. Mol. Sci. 2019, 20, 5759. https://doi.org/10.3390/ijms20225759
Tsujimura M, Kusamori K, Nishikawa M. Rapid Regulation of Human Mesenchymal Stem Cell Proliferation Using Inducible Caspase-9 Suicide Gene for Safe Cell-Based Therapy. International Journal of Molecular Sciences. 2019; 20(22):5759. https://doi.org/10.3390/ijms20225759
Chicago/Turabian StyleTsujimura, Mari, Kosuke Kusamori, and Makiya Nishikawa. 2019. "Rapid Regulation of Human Mesenchymal Stem Cell Proliferation Using Inducible Caspase-9 Suicide Gene for Safe Cell-Based Therapy" International Journal of Molecular Sciences 20, no. 22: 5759. https://doi.org/10.3390/ijms20225759
APA StyleTsujimura, M., Kusamori, K., & Nishikawa, M. (2019). Rapid Regulation of Human Mesenchymal Stem Cell Proliferation Using Inducible Caspase-9 Suicide Gene for Safe Cell-Based Therapy. International Journal of Molecular Sciences, 20(22), 5759. https://doi.org/10.3390/ijms20225759





