Xeno-Free Propagation of Spermatogonial Stem Cells from Infant Boys
Abstract
1. Introduction
2. Results
2.1. Quantification of Germ Cells in Testicular Biopsies
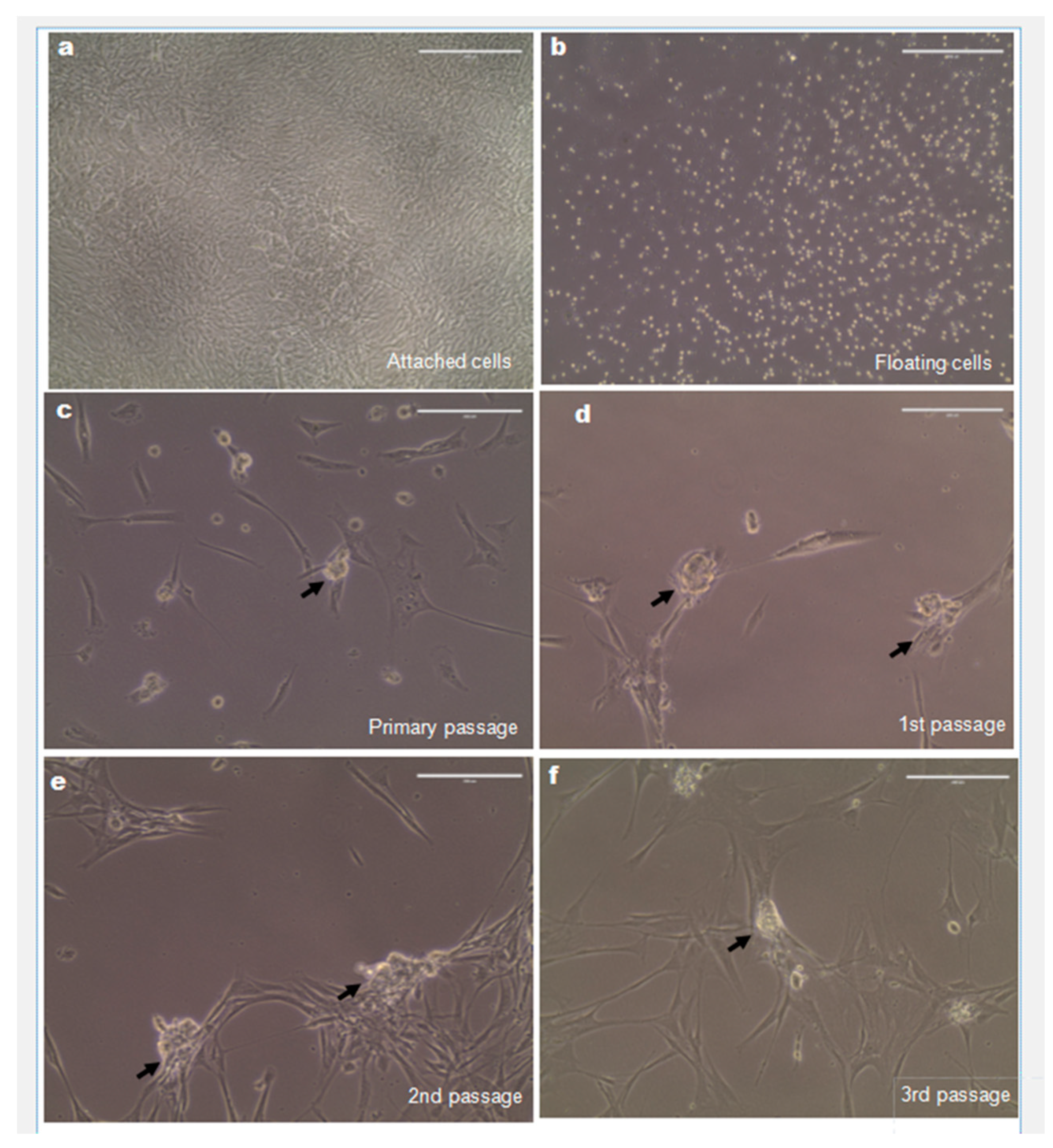
2.2. Expansion of Spermatogonial Stem Cells (SSCs) In Vitro
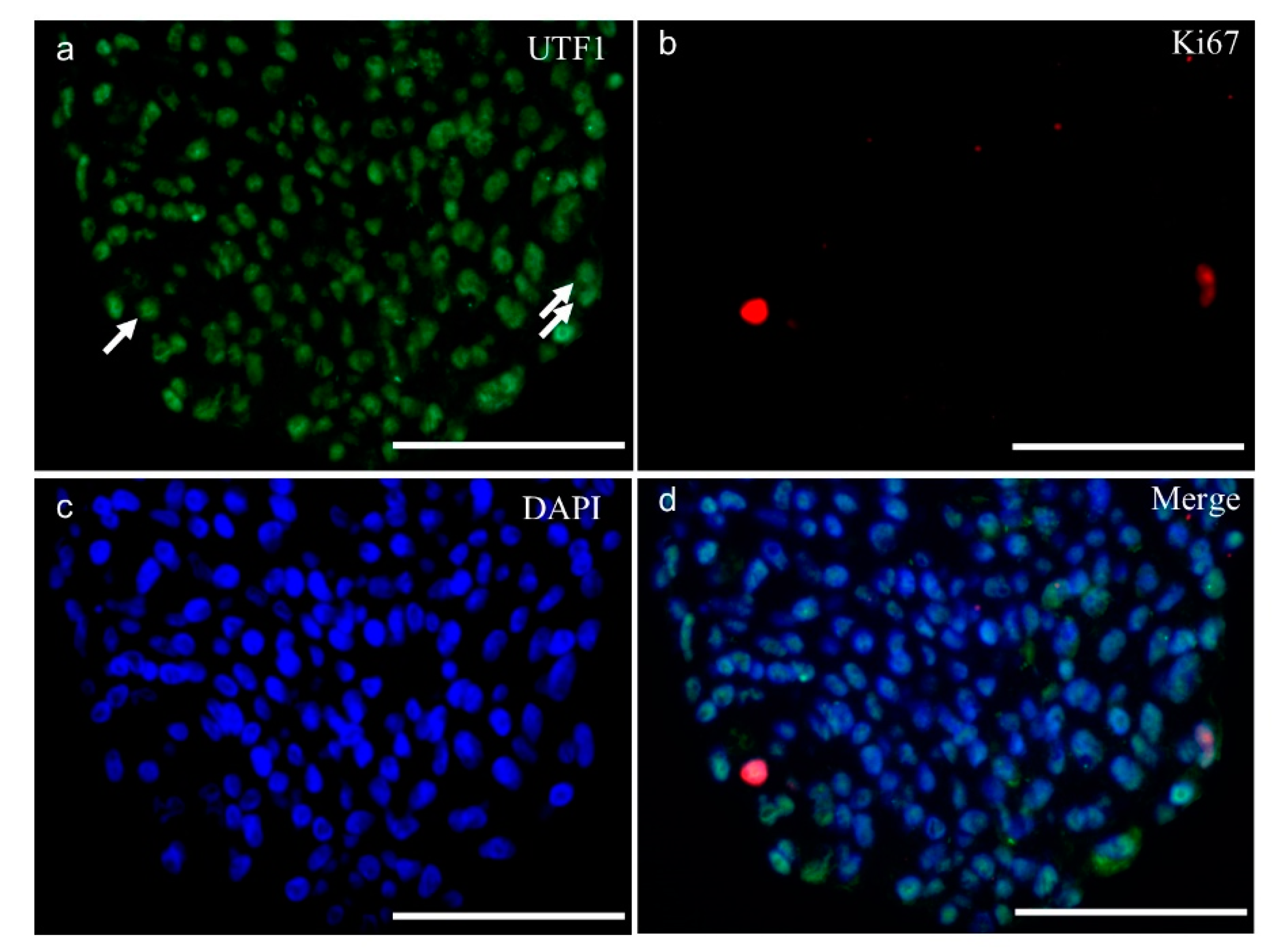
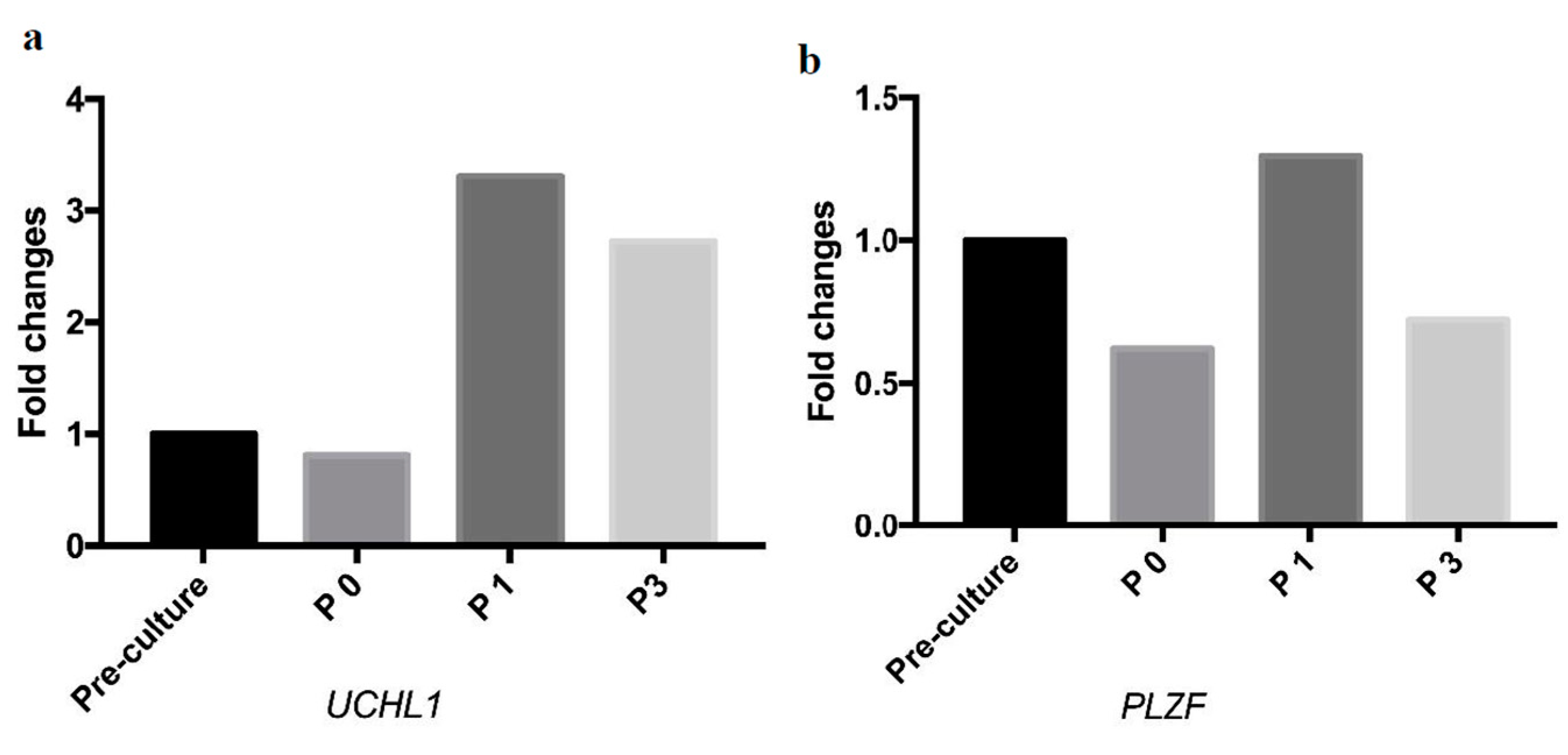
2.3. Molecular Analysis of SSC-Like Cells
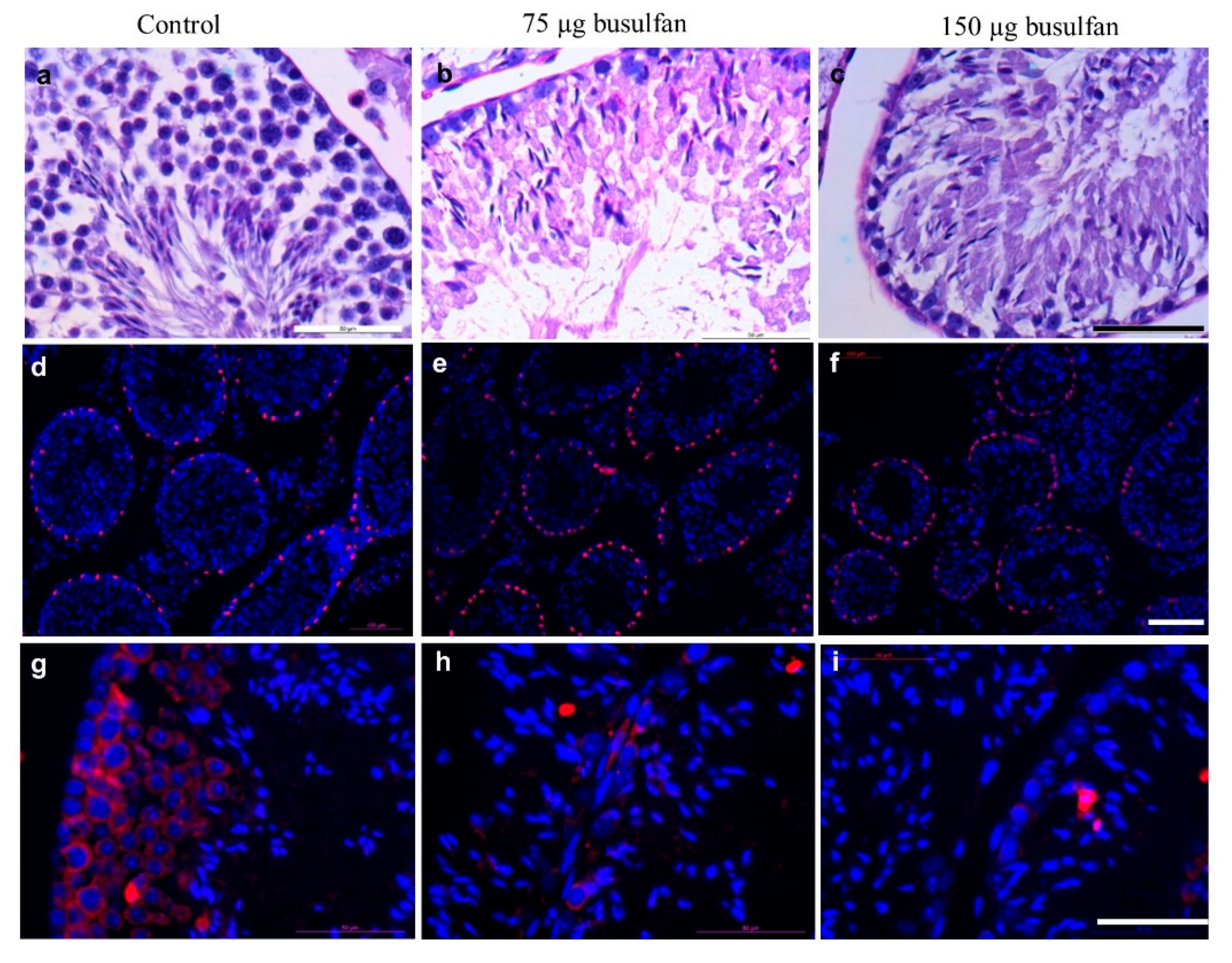
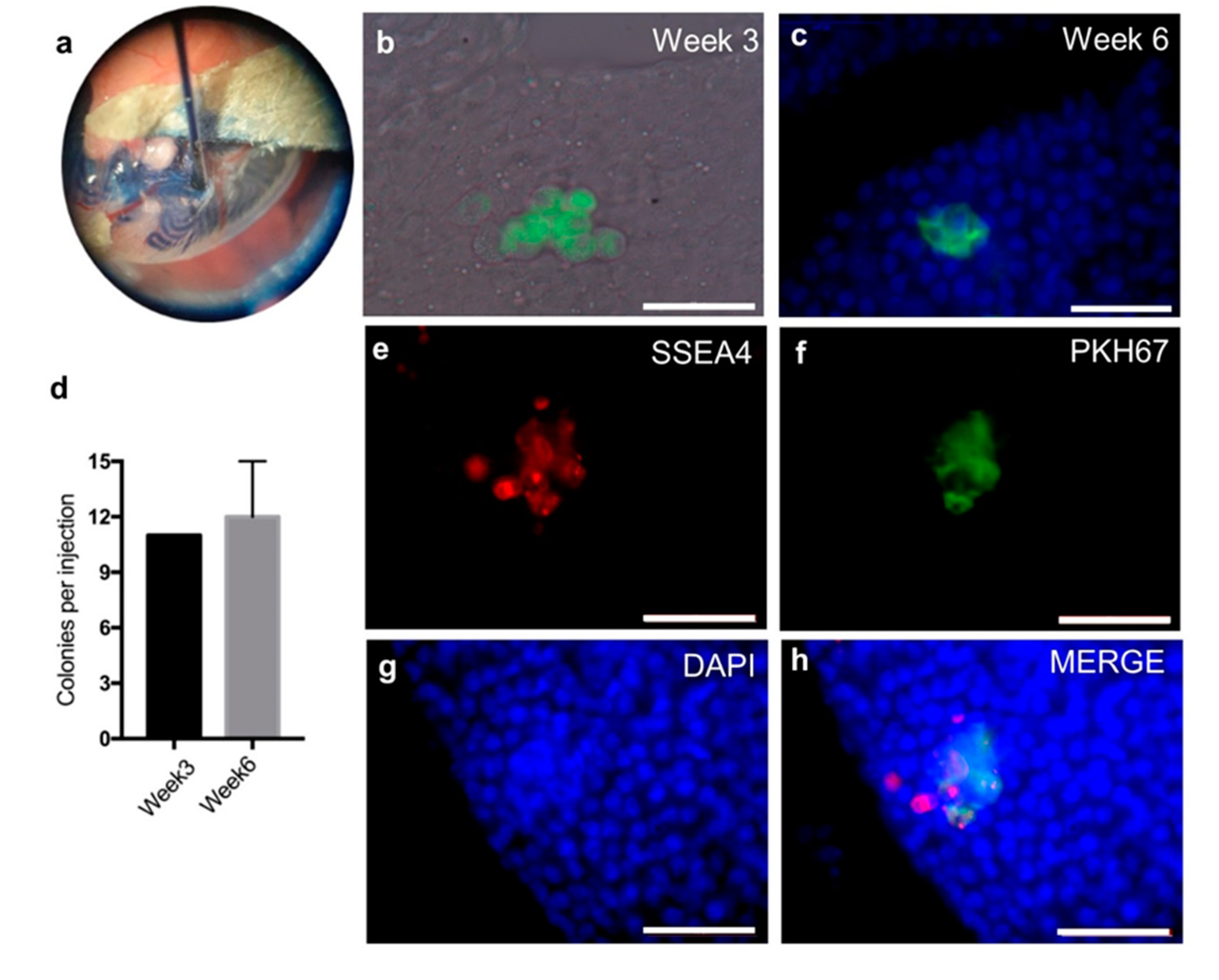
2.4. Sterilization of Nude Mice by Intratesticular Busulfan Injection
2.5. The SSC-Like Cells Settle in the SSC Niche after Xenotransplantation
3. Discussion
4. Materials and Methods
4.1. Testicular Biopsies
4.2. Testicular Cell Isolation, Culture, and Cryopreservation
4.3. Immunostaining
4.4. Gene Expression Analysis
4.5. Xenotransplantation
4.6. Whole-Mount Immunofluorescence Staining of Seminiferous Tubules
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| BSA | Bovine Serum Albumin |
| EGF | Epidermal growth factor |
| FBS | Fetal Bovine Serum |
| FGF2 | Basic fibroblast growth factor |
| GDNF | Glial cell line-derived neurotrophic factor |
| GFRα1 | GDNF family receptor alpha-1 |
| hPL | Human platelet lysate |
| MEM | Minimum Essential Medium |
| ITGA6 | Integrin alpha 6 |
| ITT | Immature testicular testis |
| LIF | Leukaemia inhibitory factor |
| PLZF PTEN | Promyelocytic leukaemia zinc finger protein Phosphatase and tensin homolog |
| SSEA4 | Stage-specific embryonic antigen-4 |
| SSC | Spermatogonial stem cell |
| SSCLC | Spermatogonial stem cell-like cells |
| UCHL1 | Ubiquitin carboxyl-terminal hydrolase isozyme L1 |
| UTF1 | Undifferentiated embryonic cell transcription factor 1 |
References
- Ward, E.; DeSantis, C.; Robbins, A.; Kohler, B.; Jemal, A. Childhood and adolescent cancer statistics, 2014. CA Cancer J. Clin. 2014, 64, 83–103. [Google Scholar] [CrossRef]
- Brannigan, R.E. Risk of infertility in male survivors of childhood cancer. Lancet Oncol. 2014, 15, 1181–1182. [Google Scholar] [CrossRef]
- Schover, L.R.; Brey, K.; Lichtin, A.; Lipshultz, L.I.; Jeha, S. Knowledge and experience regarding cancer, infertility, and sperm banking in younger male survivors. J. Clin. Oncol. 2002, 20, 1880–1889. [Google Scholar] [CrossRef]
- Tournaye, H.; Dohle, G.R.; Barratt, C.L.R. Fertility preservation in men with cancer. Lancet 2014, 384, 1295–1301. [Google Scholar] [CrossRef]
- Picton, H.M.; Wyns, C.; Anderson, R.A.; Goossens, E.; Jahnukainen, K.; Kliesch, S.; Mitchell, R.T.; Pennings, G.; Rives, N.; Tournaye, H.; et al. A European perspective on testicular tissue cryopreservation for fertility preservation in prepubertal and adolescent boys. Hum. Reprod. 2015, 30, 2463–2475. [Google Scholar] [CrossRef] [PubMed]
- Valli-Pulaski, H.; Peters, K.A.; Gassei, K.; Steimer, S.R.; Sukhwani, M.; Hermann, B.P.; Dwomor, L.; David, S.; Fayomi, A.P.; Munyoki, S.K.; et al. Testicular tissue cryopreservation: 8 years of experience from a coordinated network of academic centers. Hum. Reprod. 2019, 34, 966–977. [Google Scholar] [CrossRef] [PubMed]
- Thorup, J.; Cortes, D. Long-Term Follow-Up after Treatment of Cryptorchidism. Eur. J. Pediatr. Surg. 2016, 26, 427–431. [Google Scholar]
- Olesen, I.A.; Andersson, A.-M.; Aksglaede, L.; Skakkebaek, N.E.; Rajpert-de Meyts, E.; Joergensen, N.; Juul, A. Clinical, genetic, biochemical, and testicular biopsy findings among 1,213 men evaluated for infertility. Fertil. Steril. 2017, 107, 74–82.e7. [Google Scholar] [CrossRef]
- Vermeulen, M.; Giudice, M.-G.; Del Vento, F.; Wyns, C. Role of stem cells in fertility preservation: Current insights. Stem Cells Cloning 2019, 12, 27–48. [Google Scholar] [CrossRef]
- Brinster, R.L.; Avarbock, M.R. Germline transmission of donor haplotype following spermatogonial transplantation. Proc. Natl. Acad. Sci. USA 1994, 91, 11303–11307. [Google Scholar] [CrossRef]
- Brinster, R.L.; Zimmermann, J.W. Spermatogenesis following male germ-cell transplantation. Proc. Natl. Acad. Sci. USA 1994, 91, 11298–11302. [Google Scholar] [CrossRef] [PubMed]
- Honaramooz, A.; Megee, S.O.; Dobrinski, I. Germ cell transplantation in pigs. Biol. Reprod. 2002, 66, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Izadyar, F.; Den Ouden, K.; Stout, T.A.E.; Stout, J.; Coret, J.; Lankveld, D.P.K.; Spoormakers, T.J.P.; Colenbrander, B.; Oldenbroek, J.K.; Van der Ploeg, K.D.; et al. Autologous and homologous transplantation of bovine spermatogonial stem cells. Reproduction 2003, 126, 765–774. [Google Scholar] [CrossRef] [PubMed]
- Hermann, B.P.; Sukhwani, M.; Winkler, F.; Pascarella, J.N.; Peters, K.A.; Sheng, Y.; Valli, H.; Rodriguez, M.; Ezzelarab, M.; Dargo, G.; et al. Spermatogonial stem cell transplantation into rhesus testes regenerates spermatogenesis producing functional sperm. Cell Stem Cell 2012, 11, 715–726. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Wistuba, J.; Pock, T.; Schlatt, S.; Neuhaus, N. Spermatogonial stem cells: Updates from specification to clinical relevance. Hum. Reprod. Update 2019, 25, 275–297. [Google Scholar] [CrossRef] [PubMed]
- Zarandi, N.P.; Galdon, G.; Kogan, S.; Atala, A.; Sadri-Ardekani, H. Cryostorage of immature and mature human testis tissue to preserve spermatogonial stem cells (SSCs): A systematic review of current experiences toward clinical applications. Stem Cells Cloning 2018, 11, 23–38. [Google Scholar] [CrossRef]
- Sadri-Ardekani, H.; Homburg, C.H.; van Capel, T.M.M.; van den Berg, H.; van der Veen, F.; van der Schoot, C.E.; van Pelt, A.M.M.; Repping, S. Eliminating acute lymphoblastic leukemia cells from human testicular cell cultures: A pilot study. Fertil. Steril. 2014, 101, 1072–1078.e1. [Google Scholar] [CrossRef]
- Murdock, M.H.; David, S.; Swinehart, I.T.; Reing, J.E.; Tran, K.; Gassei, K.; Orwig, K.E.; Badylak, S.F. Human Testis Extracellular Matrix Enhances Human Spermatogonial Stem Cell Survival In Vitro. Tissue Eng. Part. A 2019, 25, 663–676. [Google Scholar] [CrossRef]
- Chen, B.; Wang, Y.-B.; Zhang, Z.-L.; Xia, W.-L.; Wang, H.-X.; Xiang, Z.-Q.; Hu, K.; Han, Y.-F.; Wang, Y.-X.; Huang, Y.-R.; et al. Xeno-free culture of human spermatogonial stem cells supported by human embryonic stem cell-derived fibroblast-like cells. Asian J. Androl. 2009, 11, 557–565. [Google Scholar] [CrossRef]
- Lim, J.J.; Sung, S.-Y.; Kim, H.J.; Song, S.-H.; Hong, J.Y.; Yoon, T.K.; Kim, J.K.; Kim, K.-S.; Lee, D.R. Long-term proliferation and characterization of human spermatogonial stem cells obtained from obstructive and non-obstructive azoospermia under exogenous feeder-free culture conditions: Long-term proliferation of human SSCs. Cell Prolif. 2010, 43, 405–417. [Google Scholar] [CrossRef]
- Piravar, Z.; Jeddi-Tehrani, M.; Sadeghi, M.R.; Mohazzab, A.; Eidi, A.; Akhondi, M.M. In vitro Culture of Human Testicular Stem Cells on Feeder-Free Condition. J. Reprod. Infertil. 2013, 14, 17–22. [Google Scholar] [PubMed]
- He, Z.; Kokkinaki, M.; Jiang, J.; Dobrinski, I.; Dym, M. Isolation, Characterization, and Culture of Human Spermatogonia1. Biol. Reprod. 2010, 82, 363–372. [Google Scholar] [CrossRef] [PubMed]
- Kokkinaki, M.; Djourabtchi, A.; Golestaneh, N. Long-term Culture of Human SSEA-4 Positive Spermatogonial Stem Cells (SSCs). J. Stem Cell Res. Ther. 2011, 2, 2488. [Google Scholar] [CrossRef] [PubMed]
- Dong, L.; Kristensen, S.G.; Hildorf, S.; Gul, M.; Clasen-Linde, E.; Fedder, J.; Hoffmann, E.R.; Cortes, D.; Thorup, J.; Andersen, C.Y. Propagation of Spermatogonial Stem Cell-Like Cells From Infant Boys. Front. Physiol. 2019, 10, 1155. [Google Scholar] [CrossRef]
- Eildermann, K.; Gromoll, J.; Behr, R. Misleading and reliable markers to differentiate between primate testis-derived multipotent stromal cells and spermatogonia in culture. Hum. Reprod. 2012, 27, 1754–1767. [Google Scholar] [CrossRef]
- Medrano, J.V.; Rombaut, C.; Simon, C.; Pellicer, A.; Goossens, E. Human spermatogonial stem cells display limited proliferation in vitro under mouse spermatogonial stem cell culture conditions. Fertil. Steril. 2016, 106, 1539–1549.e8. [Google Scholar] [CrossRef]
- Kossack, N.; Terwort, N.; Wistuba, J.; Ehmcke, J.; Schlatt, S.; Schöler, H.; Kliesch, S.; Gromoll, J. A combined approach facilitates the reliable detection of human spermatogonia in vitro. Hum. Reprod. 2013, 28, 3012–3025. [Google Scholar] [CrossRef]
- Moraveji, S.-F.; Esfandiari, F.; Sharbatoghli, M.; Taleahmad, S.; Nikeghbalian, S.; Shahverdi, A.; Baharvand, H. Optimizing methods for human testicular tissue cryopreservation and spermatogonial stem cell isolation. J. Cell. Biochem. 2019, 120, 613–621. [Google Scholar] [CrossRef]
- Kvist, K.; Clasen-Linde, E.; Langballe, O.; Hansen, S.H.; Cortes, D.; Thorup, J. The Expression of Markers for Intratubular Germ Cell Neoplasia in Normal Infantile Testes. Front. Endocrinol. 2018, 9, 286. [Google Scholar] [CrossRef]
- Masliukaite, I.; Hagen, J.M.; Jahnukainen, K.; Stukenborg, J.-B.; Repping, S.; van der Veen, F.; van Wely, M.; van Pelt, A.M.M. Establishing reference values for age-related spermatogonial quantity in prepubertal human testes: A systematic review and meta-analysis. Fertil. Steril. 2016, 106, 1652–1657.e2. [Google Scholar] [CrossRef]
- Cortes, D.; Thorup, J.M.; Beck, B.L. Quantitative Histology of Germ Cells in the Undescended Testes of Human Fetuses, Neonates and Infants. J. Urol. 1995, 154, 1188–1192. [Google Scholar] [CrossRef]
- Hutson, J.M.; Li, R.; Southwell, B.R.; Petersen, B.L.; Thorup, J.; Cortes, D. Germ cell development in the postnatal testis: The key to prevent malignancy in cryptorchidism? Front. Endocrinol. 2013, 3, 176. [Google Scholar] [CrossRef] [PubMed]
- Stukenborg, J.-B.; Alves-Lopes, J.P.; Kurek, M.; Albalushi, H.; Reda, A.; Keros, V.; Töhönen, V.; Bjarnason, R.; Romerius, P.; Sundin, M.; et al. Spermatogonial quantity in human prepubertal testicular tissue collected for fertility preservation prior to potentially sterilizing therapy. Hum. Reprod. 2018, 33, 1677–1683. [Google Scholar] [CrossRef]
- Sadri-Ardekani, H.; Mizrak, S.C.; van Daalen, S.K.M.; Korver, C.M.; Roepers-Gajadien, H.L.; Koruji, M.; Hovingh, S.; de Reijke, T.M.; de la Rosette, J.J.M.C.H.; van der Veen, F.; et al. Propagation of human spermatogonial stem cells in vitro. JAMA 2009, 302, 2127–2134. [Google Scholar] [CrossRef]
- Sadri-Ardekani, H.; Akhondi, M.A.; van der Veen, F.; Repping, S.; van Pelt, A.M.M. In vitro propagation of human prepubertal spermatogonial stem cells. JAMA 2011, 305, 2416–2418. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Rebollo, E.; Mentrup, B.; Ebert, R.; Franzen, J.; Abagnale, G.; Sieben, T.; Ostrowska, A.; Hoffmann, P.; Roux, P.-F.; Rath, B.; et al. Human Platelet Lysate versus Fetal Calf Serum: These Supplements Do Not Select for Different Mesenchymal Stromal Cells. Sci. Rep. 2017, 7, 5132. [Google Scholar] [CrossRef] [PubMed]
- van den Boom, V.; Kooistra, S.M.; Boesjes, M.; Geverts, B.; Houtsmuller, A.B.; Monzen, K.; Komuro, I.; Essers, J.; Drenth-Diephuis, L.J.; Eggen, B.J.L. UTF1 is a chromatin-associated protein involved in ES cell differentiation. J. Cell Biol. 2007, 178, 913–924. [Google Scholar] [CrossRef]
- Zhou, W.; Shao, H.; Zhang, D.; Dong, J.; Cheng, W.; Wang, L.; Teng, Y.; Yu, Z. PTEN signaling is required for the maintenance of spermatogonial stem cells in mouse, by regulating the expressions of PLZF and UTF1. Cell Biosci. 2015, 5, 42. [Google Scholar] [CrossRef]
- von Kopylow, K.; Kirchhoff, C.; Jezek, D.; Schulze, W.; Feig, C.; Primig, M.; Steinkraus, V.; Spiess, A.-N. Screening for biomarkers of spermatogonia within the human testis: A whole genome approach. Hum. Reprod. 2010, 25, 1104–1112. [Google Scholar] [CrossRef]
- von Kopylow, K.; Staege, H.; Spiess, A.-N.; Schulze, W.; Will, H.; Primig, M.; Kirchhoff, C. Differential marker protein expression specifies rarefaction zone-containing human Adark spermatogonia. Reproduction 2012, 143, 45–57. [Google Scholar] [CrossRef]
- Di Persio, S.; Saracino, R.; Fera, S.; Muciaccia, B.; Esposito, V.; Boitani, C.; Berloco, B.P.; Nudo, F.; Spadetta, G.; Stefanini, M.; et al. Spermatogonial kinetics in humans. Development 2017, 144, 3430–3439. [Google Scholar] [CrossRef] [PubMed]
- Sharma, M.; Braun, R.E. Cyclical expression of GDNF is required for spermatogonial stem cell homeostasis. Development 2018, 145, dev151555. [Google Scholar] [CrossRef] [PubMed]
- von Kopylow, K.; Spiess, A.-N. Human spermatogonial markers. Stem Cell Res. 2017, 25, 300–309. [Google Scholar] [CrossRef]
- Garbuzov, A.; Pech, M.F.; Hasegawa, K.; Sukhwani, M.; Zhang, R.J.; Orwig, K.E.; Artandi, S.E. Purification of GFRα1+ and GFRα1- Spermatogonial Stem Cells Reveals a Niche-Dependent Mechanism for Fate Determination. Stem Cell Rep. 2018, 10, 553–567. [Google Scholar] [CrossRef]
- Wang, M.; Liu, X.; Chang, G.; Chen, Y.; An, G.; Yan, L.; Gao, S.; Xu, Y.; Cui, Y.; Dong, J.; et al. Single-Cell RNA Sequencing Analysis Reveals Sequential Cell Fate Transition during Human Spermatogenesis. Cell Stem Cell 2018, 23, 599–614.e4. [Google Scholar] [CrossRef]
- Guo, J.; Grow, E.J.; Mlcochova, H.; Maher, G.J.; Lindskog, C.; Nie, X.; Guo, Y.; Takei, Y.; Yun, J.; Cai, L.; et al. The adult human testis transcriptional cell atlas. Cell Res. 2018, 28, 1141–1157. [Google Scholar] [CrossRef]
- Hermann, B.P.; Cheng, K.; Singh, A.; Roa-De La Cruz, L.; Mutoji, K.N.; Chen, I.-C.; Gildersleeve, H.; Lehle, J.D.; Mayo, M.; Westernströer, B.; et al. The Mammalian Spermatogenesis Single-Cell Transcriptome, from Spermatogonial Stem Cells to Spermatids. Cell Rep. 2018, 25, 1650–1667.e8. [Google Scholar] [CrossRef]
- Tan, K.; Wilkinson, M.F. Human Spermatogonial Stem Cells Scrutinized under the Single-Cell Magnifying Glass. Cell Stem Cell 2019, 24, 201–203. [Google Scholar] [CrossRef]
- Valli, H.; Sukhwani, M.; Dovey, S.L.; Peters, K.A.; Donohue, J.; Castro, C.A.; Chu, T.; Marshall, G.R.; Orwig, K.E. Fluorescence- and magnetic-activated cell sorting strategies to isolate and enrich human spermatogonial stem cells. Fertil. Steril. 2014, 102, 566–580.e7. [Google Scholar] [CrossRef]
- Guo, Y.; Liu, L.; Sun, M.; Hai, Y.; Li, Z.; He, Z. Expansion and long-term culture of human spermatogonial stem cells via the activation of SMAD3 and AKT pathways. Exp. Biol. Med. 2015, 240, 1112–1122. [Google Scholar] [CrossRef]
- Zheng, Y.; Thomas, A.; Schmidt, C.M.; Dann, C.T. Quantitative detection of human spermatogonia for optimization of spermatogonial stem cell culture. Hum. Reprod. 2014, 29, 2497–2511. [Google Scholar] [CrossRef] [PubMed]
- Nagano, M.; McCarrey, J.R.; Brinster, R.L. Primate spermatogonial stem cells colonize mouse testes. Biol. Reprod. 2001, 64, 1409–1416. [Google Scholar] [CrossRef] [PubMed]
- Nagano, M.; Brinster, R.L. Long-term survival of human spermatogonial stem cells in mouse testes. Fertil. Steril. 2002, 78, 9. [Google Scholar] [CrossRef]
- Hermann, B.P.; Sukhwani, M.; Lin, C.-C.; Sheng, Y.; Tomko, J.; Rodriguez, M.; Shuttleworth, J.J.; McFarland, D.; Hobbs, R.M.; Pandolfi, P.P.; et al. Characterization, Cryopreservation, and Ablation of Spermatogonial Stem Cells in Adult Rhesus Macaques. Stem Cells 2007, 25, 2330–2338. [Google Scholar] [CrossRef]
- Langenstroth, D.; Kossack, N.; Westernströer, B.; Wistuba, J.; Behr, R.; Gromoll, J.; Schlatt, S. Separation of somatic and germ cells is required to establish primate spermatogonial cultures. Hum. Reprod. 2014, 29, 2018–2031. [Google Scholar] [CrossRef][Green Version]
- Mohaqiq, M.; Movahedin, M.; Mazaheri, Z.; Amirjannati, N. In vitro transplantation of spermatogonial stem cells isolated from human frozen–thawed testis tissue can induce spermatogenesis under 3-dimensional tissue culture conditions. Biol. Res. 2019, 52, 16. [Google Scholar] [CrossRef]
- Dovey, S.L.; Valli, H.; Hermann, B.P.; Sukhwani, M.; Donohue, J.; Castro, C.A.; Chu, T.; Sanfilippo, J.S.; Orwig, K.E. Eliminating malignant contamination from therapeutic human spermatogonial stem cells. J. Clin. Investig. 2013, 123, 1833–1843. [Google Scholar] [CrossRef]
- Kubota, H.; Brinster, R.L. Spermatogonial stem cells. Biol. Reprod. 2018, 99, 52–74. [Google Scholar] [CrossRef]
- Guo, J.; Grow, E.J.; Yi, C.; Mlcochova, H.; Maher, G.J.; Lindskog, C.; Murphy, P.J.; Wike, C.L.; Carrell, D.T.; Goriely, A.; et al. Chromatin and Single-Cell RNA-Seq Profiling Reveal Dynamic Signaling and Metabolic Transitions during Human Spermatogonial Stem Cell Development. Cell Stem Cell 2017, 21, 533–546.e6. [Google Scholar] [CrossRef]
- Dong, L.-H.; Hildorf, S.; Clasen-Linde, E.; Kvist, K.; Cortes, D.; Thorup, J.; Andersen, C.Y. Postnatal germ cell development in cryptorchid boys. Asian J. Androl. 2019. [CrossRef]
- Pors, S.E.; Ramløse, M.; Nikiforov, D.; Lundsgaard, K.; Cheng, J.; Andersen, C.Y.; Kristensen, S.G. Initial steps in reconstruction of the human ovary: Survival of pre-antral stage follicles in a decellularized human ovarian scaffold. Hum. Reprod. 2019, 34, 1523–1535. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Liu, L.; He, Y.; Wang, C.; Liang, M.; Chen, X.; Hao, H.; Qin, T.; Zhao, X.; Wang, D. Testicular Busulfan Injection in Mice to Prepare Recipients for Spermatogonial Stem Cell Transplantation Is Safe and Non-Toxic. PLoS ONE 2016, 11, e0148388. [Google Scholar] [CrossRef] [PubMed]
- Goodyear, S.; Brinster, R. Spermatogonial Stem Cell Transplantation to the Testis. Cold Spring Harb. Protoc. 2017, 2017, pdb.prot094235. [Google Scholar] [CrossRef]
- Bhang, D.H.; Kim, B.-J.; Kim, B.G.; Schadler, K.; Baek, K.-H.; Kim, Y.H.; Hsiao, W.; Ding, B.-S.; Rafii, S.; Weiss, M.J.; et al. Testicular endothelial cells are a critical population in the germline stem cell niche. Nat. Commun. 2018, 9, 4379. [Google Scholar] [CrossRef]
- Gassei, K.; Valli, H.; Orwig, K.E. Whole-mount immunohistochemistry to study spermatogonial stem cells and spermatogenic lineage development in mice, monkeys, and humans. Methods Mol. Biol. 2014, 1210, 193–202. [Google Scholar]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dong, L.; Gul, M.; Hildorf, S.; Pors, S.E.; Kristensen, S.G.; Hoffmann, E.R.; Cortes, D.; Thorup, J.; Andersen, C.Y. Xeno-Free Propagation of Spermatogonial Stem Cells from Infant Boys. Int. J. Mol. Sci. 2019, 20, 5390. https://doi.org/10.3390/ijms20215390
Dong L, Gul M, Hildorf S, Pors SE, Kristensen SG, Hoffmann ER, Cortes D, Thorup J, Andersen CY. Xeno-Free Propagation of Spermatogonial Stem Cells from Infant Boys. International Journal of Molecular Sciences. 2019; 20(21):5390. https://doi.org/10.3390/ijms20215390
Chicago/Turabian StyleDong, Lihua, Murat Gul, Simone Hildorf, Susanne Elisabeth Pors, Stine Gry Kristensen, Eva R. Hoffmann, Dina Cortes, Jorgen Thorup, and Claus Yding Andersen. 2019. "Xeno-Free Propagation of Spermatogonial Stem Cells from Infant Boys" International Journal of Molecular Sciences 20, no. 21: 5390. https://doi.org/10.3390/ijms20215390
APA StyleDong, L., Gul, M., Hildorf, S., Pors, S. E., Kristensen, S. G., Hoffmann, E. R., Cortes, D., Thorup, J., & Andersen, C. Y. (2019). Xeno-Free Propagation of Spermatogonial Stem Cells from Infant Boys. International Journal of Molecular Sciences, 20(21), 5390. https://doi.org/10.3390/ijms20215390




