Anticoagulants and Osteoporosis
Abstract
1. Introduction
2. Data Source and Search
3. Heparin
3.1. Effects on Bone Metabolism
3.2. Effect on Bone Mineral Density (BMD)
3.3. Effects on Fractures
4. Oral Anticoagulants (VKAs)
4.1. Effects on Bone Metabolism
4.2. Effect on BMD
4.3. Effects on Fractures
5. Direct Oral Anticoagulants
5.1. Effects on Bone Metabolism
5.2. Effect on BMD
5.3. Effects on Fractures
6. Conclusions
Funding
Conflicts of Interest
References
- Piran, S.; Schulman, S. Management of venous thromboembolism: An update. Thromb. J. 2016, 14, 23. [Google Scholar] [CrossRef] [PubMed]
- Lazrak, H.H.; Ren, E.; Elftouh, N.; Leblanc, M.; Lafrance, J.P. Safety of Low-Molecular-Weight heparin compared to unfractionated heparin in hemodialysis: A systematic review and Meta-Analysis. BMC Nephrol. 2017, 18, 187. [Google Scholar] [CrossRef] [PubMed]
- Cosmi, B. Management of idiopathic venous thromboembolism. Expert Rev. Cardiovasc. Ther. 2016, 14, 1371–1384. [Google Scholar] [CrossRef] [PubMed]
- Thaler, J.; Pabinger, I.; Ay, C. Anticoagulant Treatment of Deep Vein Thrombosis and Pulmonary Embolism: The Present State of the Art. Front. Cardiovasc. Med. 2015, 2, 30. [Google Scholar] [CrossRef] [PubMed]
- Brandjes, D.P.; Heijboer, H.; Buller, H.R.; de Rijk, M.; Jagt, H.; ten Cate, J.W. Acenocoumarol and heparin compared with acenocoumarol alone in the initial treatment of proximal-vein thrombosis. N. Engl. J. Med. 1992, 327, 1485–1489. [Google Scholar] [CrossRef] [PubMed]
- Ageno, W.; Gallus, A.S.; Wittkowsky, A.; Crowther, M.; Hylek, E.M.; Palareti, G. American College of Chest Physicians. Oral anticoagulant therapy: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest 2012, 141, e44S–e88S. [Google Scholar] [CrossRef]
- Bates, S.M.; Jaeschke, R.; Stevens, S.M.; Goodacre, S.; Wells, P.S.; Stevenson, M.D.; Kearon, C.; Schunemann, H.J.; Crowther, M.; Pauker, S.G.; et al. Diagnosis of DVT: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest 2012, 141, e351S–e418S. [Google Scholar] [CrossRef]
- Lim, W.; Le Gal, G.; Bates, S.M.; Righini, M.; Haramati, L.B.; Lang, E.; Kline, J.A.; Chasteen, S.; Snyder, M.; Patel, P.; et al. American Society of Hematology 2018 guidelines for management of venous thromboembolism: Diagnosis of venous thromboembolism. Blood Adv. 2018, 2, 3226–3256. [Google Scholar] [CrossRef]
- Kearon, C.; Akl, E.A.; Ornelas, J.; Blaivas, A.; Jimenez, D.; Bounameaux, H.; Huisman, M.; King, C.S.; Morris, T.A.; Sood, N.; et al. Antithrombotic Therapy for VTE Disease CHEST Guideline and Expert Panel Report. Chest 2016, 149, 315–352. [Google Scholar] [CrossRef]
- Konstantinides, S.V.; Torbicki, A.; Agnelli, G.; Danchin, N.; Fitzmaurice, D.; Galiè, N.; Gibbs, J.S.; Huisman, M.V.; Humbert, M.; Kucher, N.; et al. Task Force for the Diagnosis and Management of Acute Pulmonary Embolism of the European Society of Cardiology (ESC). 2014 ESC guidelines on the diagnosis and management of acute pulmonary embolism. Eur. Heart J. 2014, 35, 3033–3069. [Google Scholar]
- Monreal, M.; Suarez, C.; Fajardo, J.A.; Barba, R.; Uresandi, F.; Valle, R.; Rondón, P.; RIETE investigators. Management of patients with acute venous thromboembolism: Findings from the RIETE registry. Pathophysiol. Haemost. Thromb. 2003, 33, 330–334. [Google Scholar] [CrossRef] [PubMed]
- Nieto, J.A.; Solano, R.; Ruiz-Ribó, M.D.; Ruiz-Gimenez, N.; Prandoni, P.; Kearon, C.; Monreal, M.; Riete Investigators. Fatal bleeding in patients receiving anticoagulant therapy for venous thromboembolism: Findings from the RIETE registry. J. Thromb. Haemost. 2010, 8, 1216–1222. [Google Scholar] [CrossRef] [PubMed]
- Riva, N.; Bellesini, M.; Di Minno, M.N.; Mumoli, N.; Pomero, F.; Franchini, M.; Fantoni, C.; Lupoli, R.; Brondi, B.; Borretta, V.; et al. Poor predictive value of contemporary bleeding risk scores during long-term treatment of venous thromboembolism. A multicentre retrospective cohort study. Thromb. Haemost. 2014, 112, 511–521. [Google Scholar] [PubMed]
- Mischie, A.N.; Chioncel, V.; Droc, I.; Sinescu, C. Anticoagulation in patients with dilated cardiomyopathy, low ejection fraction, and sinus rhythm: Back to the drawing board. Cardiovasc. Ther. 2013, 31, 298–302. [Google Scholar] [CrossRef] [PubMed]
- Knight, S.; Klaskala, W.; Woller, S.C.; Horne, B.D.; Bunch, T.J.; Le, V.T.; Mills, R.M.; Muhlestein, J.B. Antithrombotic therapy in patients with acute coronary syndrome in the intermountain heart collaborative study. Cardiol. Res. Pract. 2015, 2015, 270508. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, R.A.; Otto, C.M.; Bonow, R.O.; Carabello, B.A.; Erwin, J.P., 3rd; Fleisher, L.A.; Jneid, H.; Mack, M.J.; McLeod, C.J.; O’Gara, P.T.; et al. 2017 AHA/ACC Focused Update of the 2014 AHA/ACC Guideline for the Management of Patients With Valvular Heart Disease: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 2017, 135, e1159–e1195. [Google Scholar] [CrossRef]
- Sikorska, J.; Uprichard, J. Direct Oral Anticoagulants: A Quick Guide. Eur. Cardiol. Rev. 2017, 12, 40–45. [Google Scholar] [CrossRef]
- Nelson-Piercy, C. Hazards of heparin: Allergy, Heparin-Induced thrombocytopenia and osteoporosis. Baillieres Clin. Obstet. Gynaecol. 1997, 11, 489–509. [Google Scholar] [CrossRef]
- Alban, S. Adverse effects of heparin. Handb. Exp. Pharmacol. 2012, 207, 211–263. [Google Scholar]
- Schulman, S.; Hellgren-Wångdahl, M. Pregnancy, heparin and osteoporosis. Thromb. Haemost. 2002, 87, 180–181. [Google Scholar]
- Barbour, L.A.; Kick, S.D.; Steiner, J.F.; LoVerde, M.E.; Heddleston, L.N.; Lear, J.L.; Barón, A.E.; Barton, P.L. A prospective study of Heparin-Induced osteoporosis in pregnancy using bone densitometry. Am. J. Obstet. Gynecol. 1994, 170, 862–869. [Google Scholar] [CrossRef]
- Dahlman, T.C.; Sjoberg, H.E.; Ringertz, H. Bone mineral density during long-term prophylaxis with heparin in pregnancy. Am. J. Obstet. Gynecol. 1994, 170, 1315–1320. [Google Scholar] [CrossRef]
- Galambosi, P.; Hiilesmaa, V.; Ulander, V.M.; Laitinen, L.; Tiitinen, A.; Kaaja, R. Prolonged Low-Molecular-Weight heparin use during pregnancy and subsequent bone mineral density. Thromb. Res. 2016, 143, 122–126. [Google Scholar] [CrossRef] [PubMed]
- Rajgopal, R.; Bear, M.; Butcher, M.K.; Shaughnessy, S.G. The effects of heparin and low molecular weight heparins on bone. Thromb. Res. 2008, 122, 293–298. [Google Scholar] [CrossRef]
- Muir, J.M.; Andrew, M.; Hirsh, J.; Weitz, J.I.; Young, E.; Deschamps, P.; Shaughnessy, S.G. Histomorphometric analysis of the effects of standard heparin on trabecular bone in vivo. Blood 1996, 88, 1314–1320. [Google Scholar] [CrossRef]
- Muir, J.M.; Hirsh, J.; Weitz, J.I.; Andrew, M.; Young, E.; Shaughnessy, S.G. A histomorphometric comparison of the effects of heparin and Low-Molecular weight heparin on cancellous bone in rats. Blood 1997, 89, 3236–3242. [Google Scholar] [CrossRef]
- Shaughnessy, S.G.; Young, E.; Deschamps, P.; Hirsh, J. The effects of low molecular weight and standard heparin on calcium loss from fetal rat calvaria. Blood 1995, 86, 1368–1373. [Google Scholar] [CrossRef]
- Handschin, A.E.; Trentz, O.A.; Hoerstrup, S.P.; Kock, H.J.; Wanner, G.A.; Trentz, O. Effect of low molecular weight heparin (dalteparin) and fondaparinux (Arixtra) on human osteoblasts in vitro. Br. J. Surg. 2005, 92, 177–183. [Google Scholar] [CrossRef]
- Osip, S.L.; Butcher, M.; Young, E.; Yang, L.; Shaughnessy, S.G. Differential effects of heparin and low molecular weight heparin on osteoblastogenesis and adipogenesis in vitro. Thromb. Haemost. 2004, 92, 803–810. [Google Scholar] [CrossRef]
- Bhandari, M.; Hirsh, J.; Weitz, J.I.; Young, E.; Venner, T.J.; Shaughnessy, S.G. The effects of standard and low molecular weight heparin on bone nodule formation in vitro. Thromb. Haemost. 1998, 80, 413–417. [Google Scholar] [CrossRef]
- Irie, A.; Takami, M.; Kubo, H.; Sekino-Suzuki, N.; Kasahara, K.; Sanai, Y. Heparin enhances osteoclastic bone resorption by inhibiting osteoprotegerin activity. Bone 2007, 41, 165–174. [Google Scholar] [CrossRef] [PubMed]
- Vik, A.; Brodin, E.; Sveinbjornsson, B.; Hansen, J.B. Heparin induces mobilization of osteoprotegerin into the circulation. Thromb. Haemost. 2007, 98, 148–154. [Google Scholar] [PubMed]
- Douketis, J.D.; Ginsberg, J.S.; Burrows, R.F.; Duku, E.K.; Webber, C.E.; Brill-Edwards, P. The effects of long-term heparin therapy during pregnancy on bone density. A prospective matched cohort study. ThrombHaemost 1996, 75, 254–257. [Google Scholar]
- Nelson-Piercy, C.; Letsky, E.A.; de Swiet, M. Low-Molecular weight heparin for obstetric thromboprophylaxis: Experience of sixty-nine pregnancies in sixty-one women at high risk. Am. J. ObstetGynecol. 1997, 176, 1062–1068. [Google Scholar] [CrossRef]
- Casele, H.L.; Laifer, S.A. Prospective evaluation of bone density in pregnant women receiving the low molecular weight heparin enoxaparin sodium. J. Matern. Fetal Med. 2000, 9, 122–125. [Google Scholar] [PubMed]
- Greer, I.A.; Nelson-Piercy, C. Low-Molecular-Weight heparins for thromboprophylaxis and treatment of venous thromboembolism in pregnancy: A systematic review of safety and efficacy. Blood 2005, 106, 401–407. [Google Scholar] [CrossRef]
- Lefkou, E.; Khamashta, M.; Hampson, G.; Hunt, B.J. Review: Low-Molecular-Weight Heparin-Induced osteoporosis and osteoporotic fractures: A myth or an existing entity? Lupus 2010, 19, 3–12. [Google Scholar] [CrossRef]
- Carlin, A.J.; Farquharson, R.G.; Quenby, S.M.; Topping, J.; Fraser, W.D. Prospective observational study of bone mineral density during pregnancy: Low molecular weight heparin versus control. Hum. Reprod. 2004, 19, 1211–1214. [Google Scholar] [CrossRef]
- Pettila, V.; Leinonen, P.; Markkola, A.; Hiilesmaa, V.; Kaaja, R. Postpartum bone mineral density in women treated for thromboprophylaxis with unfractionated heparin or LMW heparin. ThrombHaemost 2002, 87, 182–196. [Google Scholar] [CrossRef]
- Tsvetov, G.; Levy, S.; Benbassat, C.; Shraga-Slutzky, I.; Hirsch, D. Influence of number of deliveries and total Breast-Feeding time on bone mineral density in premenopausal and young postmenopausal women. Maturitas 2014, 77, 249–254. [Google Scholar] [CrossRef]
- Hirsh, J.; Warkentin, T.E.; Shaughnessy, S.G.; Anand, S.S.; Halperin, J.L.; Raschke, R.; Granger, C.; Ohman, E.M.; Dalen, J.E. Heparin and low-molecular-weight heparin: Mechanisms of action, pharmacokinetics, dosing considerations, monitoring, efficacy, and safety. Chest 1998, 114, 489S–510S. [Google Scholar] [CrossRef] [PubMed]
- Wawrzynska, L.; Tomkowski, W.Z.; Przedlacki, J.; Hajduk, B.; Torbicki, A. Changes in bone density during Long-Term administration of Low-Molecular-Weight heparins or acenocoumarol for secondary prophylaxis of venous thromboembolism. PathophysiolHaemostThromb 2003, 33, 64–67. [Google Scholar] [CrossRef] [PubMed]
- Serra, R.; Buffone, G.; De Franciscis, A.; Mastrangelo, D.; Vitagliano, T.; Greco, M.; de Franciscis, S. Skin grafting followed by low molecular-weight heparin long-term therapy in chronic venous leg ulcers. Ann. Vasc. Surg. 2012, 26, 190–197. [Google Scholar] [CrossRef]
- Bernis, C. Mineral bone density variation in hemodialysis patients: Nonfractioned heparin (NFH) versus low molecular weight heparin (LMWH). Nephrol. Dial. Transplant. 1997, 12, 1789. [Google Scholar]
- Lai, K.N.; Ho, K.; Cheung, R.C.; Lit, L.C.; Lee, S.K.; Fung, K.S.; Tong, M.K.; Lam, C.W. Effect of low molecular weight heparin on bone metabolism and hyperlipidemia in patients on maintenance hemodialysis. Int. J. Artif. Organs. 2001, 24, 447–455. [Google Scholar] [CrossRef]
- Grassman, E.D.; Leya, F.; Fareed, J.; Lewis, B.E.; Bacher, P.; Loeb, H.S.; Moran, J.F. A randomized trial of the low Molecular-Weight heparin certoparin to prevent restenosis following coronary angioplasty. J. Invasive. Cardiol. 2001, 13, 723–728. [Google Scholar]
- Grzegorzewska, A.E.; Mlot-Michalska, M. Low molecular weight heparins and antiplatelet drugs, and bone mineral density in dialysis patients. Adv. Perit. Dial. 2008, 24, 125–131. [Google Scholar]
- Monreal, M.; Olive, A.; Lafoz, E.; del Rio, L. Heparins, coumarin, and bone density. Lancet 1991, 338, 706. [Google Scholar] [CrossRef]
- Pettila, V.; Kaaja, R.; Leinonen, P.; Ekblad, U.; Kataja, M.; Ikkala, E. Thromboprophylaxis with low molecular weight heparin (dalteparin) in pregnancy. Thromb. Res. 1999, 96, 275–282. [Google Scholar] [CrossRef]
- Dahlman, T.C. Osteoporotic fractures and the recurrence of thromboembolism during pregnancy and the puerperium in 184 women undergoing thromboprophylaxis with heparin. Am. J. Obstet. Gynecol. 1993, 168, 1265–1270. [Google Scholar] [CrossRef]
- Monreal, M.; Lafoz, E.; Olive, A.; del Rio, L.; Vedia, C. Comparison of subcutaneous unfractionated heparin with a low molecular weight heparin (Fragmin) in patients with venous thromboembolism and contraindications to coumarin. Thromb. Haemost. 1994, 71, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Gajic-Veljanoski, O.; Chai, W.; Prakesh, S.; Cheung, A.M. Effects of Long-Term Low- Molecular-Weight Heparin on Fractures and Bone Density in Non-Pregnant Adults: A Systematic Review with Meta-Analysis. J. Gen. Intern. Med. 2016, 31, 947–957. [Google Scholar] [CrossRef] [PubMed]
- Karsenty, G.; Olson, E.N. Bone and Muscle Endocrine Functions: Unexpected Paradigms of Inter-Organ Communication. Cell 2016, 164, 1248–1256. [Google Scholar] [CrossRef] [PubMed]
- Price, P.A.; Williamson, M.K. Effects of warfarin on bone. Studies on the vitamin K-dependent protein of rat bone. J. Biol. Chem 1981, 256, 12754–12759. [Google Scholar] [PubMed]
- Ferrière, K.; Rizzoli, R. Anticoagulants and osteoporosis [in French]. Rev. Med. Suisse 2007, 3, 1508–1511. [Google Scholar]
- Pearson, D.A. Bone health and osteoporosis: The role of vitamin K and potential antagonism by anticoagulants. Nutr. Clin. Pract. 2007, 22, 517–544. [Google Scholar] [CrossRef]
- Vermeer, C. Vitamin K: The effect on health beyond coagulation—An overview. Food Nutr. Res. 2012, 56, 5329. [Google Scholar] [CrossRef]
- Cranenburg, E.C.; Schurgers, L.J.; Vermeer, C. Vitamin K: The coagulation vitamin that became omnipotent. Thromb. Haemost. 2007, 98, 120–125. [Google Scholar] [CrossRef]
- Szulc, P.; Arlot, M.; Chapuy, M.C.; Duboeuf, F.; Meunier, P.J.; Delmas, P.D. Serum undercarboxylated osteocalcin correlates with hip bone mineral density in elderly women. J. Bone Miner. Res. 1994, 9, 1591–1595. [Google Scholar] [CrossRef]
- Jamal, S.A.; Browner, W.S.; Bauer, D.C.; Cummings, S.R. Study of Osteoporotic Fractures Research Group. Warfarin use and risk for osteoporosis in elderly women. Ann. Intern. Med. 1998, 128, 829–832. [Google Scholar] [CrossRef]
- Avgeri, M.; Papadopoulou, A.; Platokouki, H.; Douros, K.; Rammos, S.; Nicolaidou, P.; Aronis, S. Assessment of bone mineral density and markers of bone turnover in children under Long-Term oral anticoagulant therapy. J. Pediatr. Hematol. Oncol. 2008, 30, 592–597. [Google Scholar] [CrossRef] [PubMed]
- Rezaieyazdi, Z.; Falsoleiman, H.; Khajehdaluee, M.; Saghafi, M.; Mokhtari-Amirmajdi, E. Reduced bone density in patients on long-term warfarin. Int. J. Rheum. Dis. 2009, 12, 130–135. [Google Scholar] [CrossRef] [PubMed]
- Tufano, A.; Coppola, A.; Contaldi, P.; Franchini, M.; Minno, G.D. Oral Anticoagulant Drugs and the Risk of Osteoporosis: New Anticoagulants Better than Old? Semin. Thromb. Hemost. 2015, 41, 382–388. [Google Scholar] [CrossRef] [PubMed]
- Caraballo, P.J.; Heit, J.A.; Atkinson, E.J.; Silverstein, M.D.; O’Fallon, W.M.; Castro, M.R.; Melton, L.J., 3rd. Long-Term use of oral anticoagulants and the risk of fracture. Arch. Intern. Med. 1999, 159, 1750–1756. [Google Scholar] [CrossRef] [PubMed]
- Caraballo, P.J.; Gabriel, S.E.; Castro, M.R.; Atkinson, E.J.; Melton, L.J., III. Changes in bone density after exposure to oral anticoagulants: A Meta-Analysis. Osteoporos. Int. 1999, 9, 441–448. [Google Scholar] [CrossRef]
- Gage, B.F.; Birman-Deych, E.; Radford, M.J.; Nilasena, D.S.; Binder, E.F. Risk of osteoporotic fracture in elderly patients taking warfarin: Results from the National Registry of Atrial Fibrillation 2. Arch. Intern. Med. 2006, 166, 241–246. [Google Scholar] [CrossRef]
- Fiordellisi, W.; White, K.; Schweizer, M. A Systematic Review and Meta-Analysis of the Association Between Vitamin K Antagonist Use and Fracture. J. Gen. Intern. Med. 2019, 34, 304–311. [Google Scholar] [CrossRef]
- Beaubrun, A.C.; Kilpatrick, R.D.; Freburger, J.K.; Bradbury, B.D.; Wang, L.; Brookhart, M.A. Temporal trends in fracture rates and postdischarge outcomes among hemodialysis patients. J. Am. Soc. Nephrol. 2013, 24, 1461–1469. [Google Scholar] [CrossRef]
- Jadoul, M.; Albert, J.M.; Akiba, T.; Akizawa, T.; Arab, L.; Bragg-Gresham, J.L.; Mason, N.; Prutz, K.G.; Young, E.W.; Pisoni, R.L. Incidence and risk factors for hip or other bone fractures among hemodialysis patients in the Dialysis Outcomes and Practice Patterns Study. Kidney Int. 2006, 70, 1358–1366. [Google Scholar] [CrossRef]
- Gigi, R.; Salai, M.; Dolkart, O.; Chechik, O.; Katzburg, S.; Stern, N.; Somjen, D. The effects of direct factor Xa inhibitor (Rivaroxaban) on the human osteoblastic cell line SaOS2. Connect. Tissue Res. 2012, 53, 446–450. [Google Scholar] [CrossRef]
- Somjen, D.; Katzburg, S.; Gigi, R.; Dolkart, O.; Sharon, O.; Salai, M.; Stern, N. Rivaroxaban, a direct inhibitor of the coagulation factor Xa interferes with hormonal-induced physiological modulations in human female osteoblastic cell line SaSO2. J. Steroid. Biochem. Mol. Biol. 2013, 135, 67–70. [Google Scholar] [CrossRef] [PubMed]
- Winkler, T.; Perka, C.; Matziolis, D.; Matziolis, G. Effect of a direct thrombin inhibitor compared with dalteparin and unfractionated heparin on human osteoblasts. Open Orthop. J. 2011, 5, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Morishima, Y.; Kamisato, C.; Honda, Y.; Furugohri, T.; Shibano, T. The effects of warfarin and edoxaban, an oral direct factor Xa inhibitor, on gammacarboxylated (Gla-osteocalcin) and undercarboxylated osteocalcin (uc-osteocalcin) in rats. Thromb. Res. 2013, 131, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Solayar, G.N.; Walsh, P.M.; Mulhall, K.J. The effect of a new direct Factor Xa inhibitor on human osteoblasts: An in-vitro study comparing the effect of rivaroxaban with enoxaparin. BMC Musculoskelet. Disord. 2011, 12, 247. [Google Scholar] [CrossRef] [PubMed]
- Pilge, H.; Fröbel, J.; Mrotzek, S.J.; Fischer, J.C.; Prodinger, P.M.; Zilkens, C.; Bittersohl, B.; Krauspe, R. Effects of thromboprophylaxis on mesenchymal stromal cells during osteogenic differentiation: An in-vitro study comparing enoxaparin with rivaroxaban. BMC Musculoskelet. Disord. 2016, 17, 108. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Somjen, D.; Sharfman, Z.T.; Katzburg, S.; Sharon, O.; Maman, E.; Salai, M.; Stern, N.; Dolkart, O. Rivaroxaban significantly inhibits the stimulatory effects of bone-modulating hormones: In vitro study of primary female osteoblasts. Connect. Tissue Res. 2017, 58, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Namba, S.; Yamaoka-Tojo, M.; Kakizaki, R.; Nemoto, T.; Fujiyoshi, K.; Hashikata, T.; Kitasato, L.; Hashimoto, T.; Kameda, R.; Meguro, K.; et al. Effects on bone metabolism markers and arterial stiffness by switching to rivaroxaban from warfarin in patients with atrial fibrillation. Heart Vessels. 2017, 32, 977–982. [Google Scholar] [CrossRef]
- Dalle Carbonare, L.; Mottes, M.; Brunelli, A.; Deiana, M.; Cheri, S.; Suardi, S.; Valenti, M.T. Effects of Oral Anticoagulant Therapy on Gene Expression in Crosstalk between Osteogenic Progenitor Cells and Endothelial Cells. J. Clin. Med. 2019, 8, 329. [Google Scholar] [CrossRef]
- Lau, W.C.; Chan, E.W.; Cheung, C.L.; Sing, C.W.; Man, K.K.; Lip, G.Y.; Siu, C.W.; Lam, J.K.; Lee, A.C.; Wong, I.C. Association Between Dabigatran vs Warfarin and Risk of Osteoporotic Fractures Among Patients With Nonvalvular Atrial Fibrillation. JAMA 2017, 317, 1151–1158. [Google Scholar] [CrossRef]
- Treceño-Lobato, C.; Jiménez-Serranía, M.I.; Martínez-García, R.; Corzo-Delibes, F.; Martín Arias, L.H. New Anticoagulant Agents: Incidence of Adverse Drug Reactions and New Signals Thereof. Semin. Thromb. Hemost. 2019, 45, 196–204. [Google Scholar]
| VKA Therapy: Indications |
|---|
| Venous thromboembolism prophylaxis [6] |
| Pulmonary embolism and deep vein thrombosis therapy [9] |
| Thromboembolic prophylaxis in patients with dilated cardiomyopathyc [14] |
| Thromboembolic prophylaxis in patients with biologic heart valve replacement [6] |
| Thromboembolic prophylaxis in patients with myocardial infarction (primary, secondary prevention) [15] |
| Thromboembolic prophylaxis in patients with aortic valvular heart disease [16] |
| Thromboembolic prophylaxis in patients with mechanic heart valve replacement [6] |
| Thromboembolic prophylaxis in patients with anti-phospholipid syndrome [9] |
| Thromboembolic prophylaxis in patients with atrial fibrillation [6] |
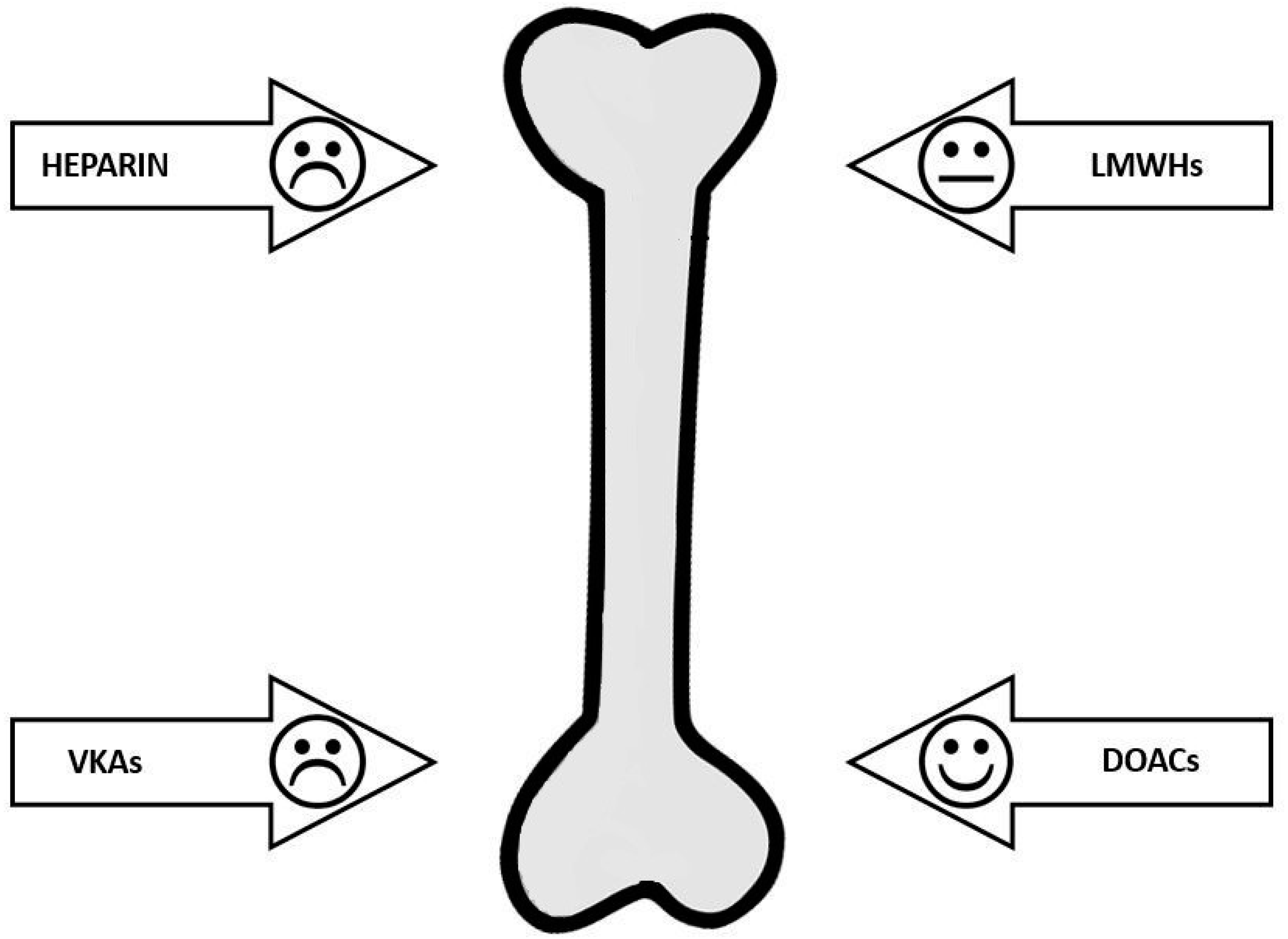
| Drug Class | Effect on Bone Metabolism | Effect on BMD | Effect on Fractures |
|---|---|---|---|
| Heparin | High | High | High |
| Low molecular weight heparins | Low | Uncertain | Uncertain |
| Vitamin K antagonists | High | Low | Low |
| Direct-acting oral anticoagulants | None | N/A | None |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Signorelli, S.S.; Scuto, S.; Marino, E.; Giusti, M.; Xourafa, A.; Gaudio, A. Anticoagulants and Osteoporosis. Int. J. Mol. Sci. 2019, 20, 5275. https://doi.org/10.3390/ijms20215275
Signorelli SS, Scuto S, Marino E, Giusti M, Xourafa A, Gaudio A. Anticoagulants and Osteoporosis. International Journal of Molecular Sciences. 2019; 20(21):5275. https://doi.org/10.3390/ijms20215275
Chicago/Turabian StyleSignorelli, Salvatore Santo, Salvatore Scuto, Elisa Marino, Michele Giusti, Anastasia Xourafa, and Agostino Gaudio. 2019. "Anticoagulants and Osteoporosis" International Journal of Molecular Sciences 20, no. 21: 5275. https://doi.org/10.3390/ijms20215275
APA StyleSignorelli, S. S., Scuto, S., Marino, E., Giusti, M., Xourafa, A., & Gaudio, A. (2019). Anticoagulants and Osteoporosis. International Journal of Molecular Sciences, 20(21), 5275. https://doi.org/10.3390/ijms20215275






