Role of Heat Shock Proteins in Glaucoma
Abstract
:1. Introduction
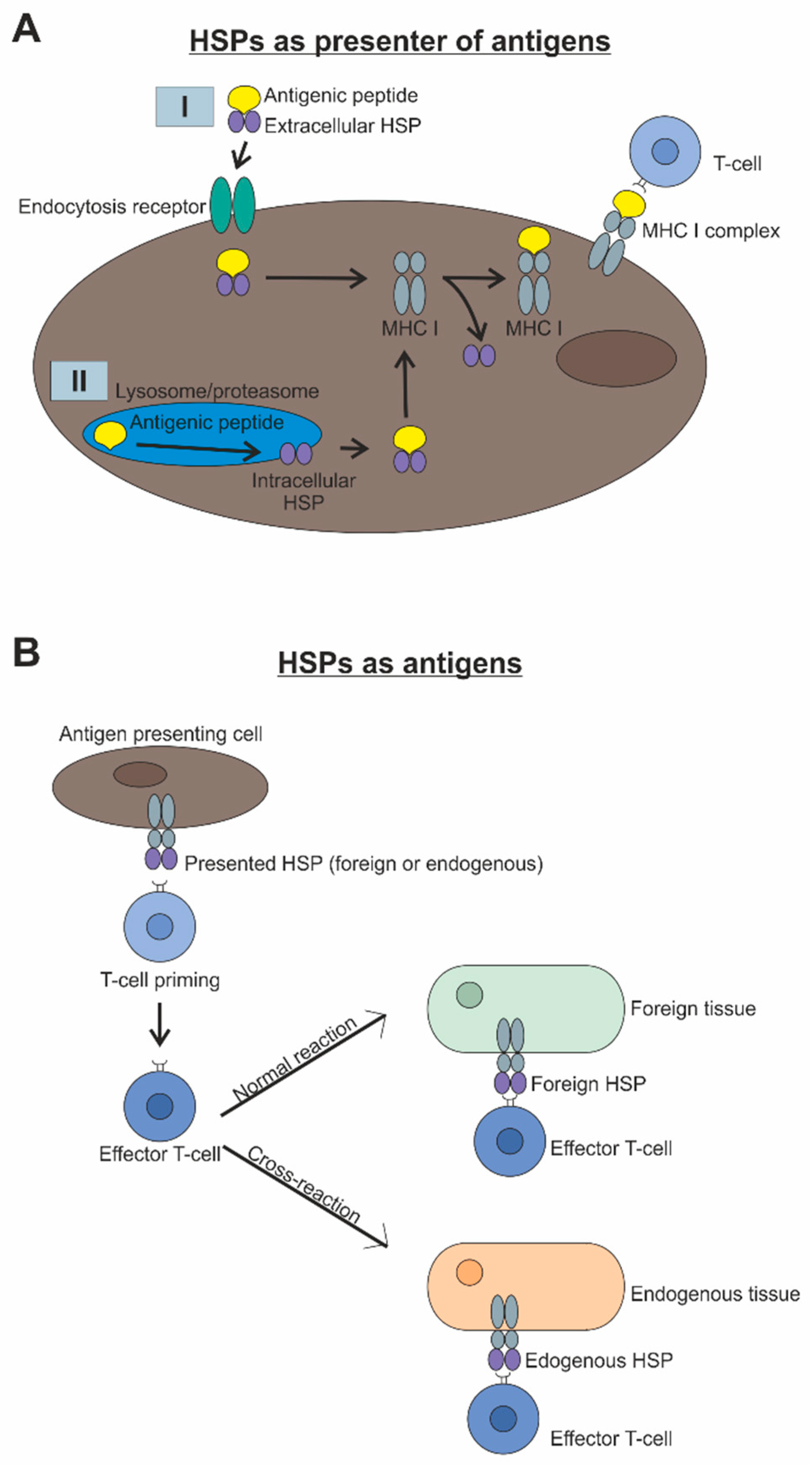
2. Interaction of Heat Shock Proteins and the Immune System
3. Role of Heat Shock Proteins in Glaucoma Patients
4. Heat Shock Proteins Induce Glaucoma-Like Damage in Animal Models
5. Heat Shock Proteins: Friend or Foe
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Michel, G.P.; Starka, J. Effect of ethanol and heat stresses on the protein pattern of Zymomonas mobilis. J. Bacteriol. 1986, 165, 1040–1042. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lindquist, S.; Craig, E.A. The heat-shock proteins. Ann. Rev. Genet. 1988, 22, 631–677. [Google Scholar] [CrossRef] [PubMed]
- Kampinga, H.H.; Hageman, J.; Vos, M.J.; Kubota, H.; Tanguay, R.M.; Bruford, E.A.; Cheetham, M.E.; Chen, B.; Hightower, L.E. Guidelines for the nomenclature of the human heat shock proteins. Cell Stress Chaperones 2009, 14, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Jee, H. Size dependent classification of heat shock proteins: A mini-review. J. Exerc. Rehabil. 2016, 12, 255–259. [Google Scholar] [CrossRef] [PubMed]
- Welch, W.J. Mammalian stress response: Cell physiology, structure/function of stress proteins, and implications for medicine and disease. Physiol. Rev. 1992, 72, 1063–1081. [Google Scholar] [CrossRef]
- Jakob, U.; Gaestel, M.; Engel, K.; Buchner, J. Small heat shock proteins are molecular chaperones. J. Biol. Chem. 1993, 268, 1517–1520. [Google Scholar]
- Cappello, F.; Conway de Macario, E.; Marasa, L.; Zummo, G.; Macario, A.J. Hsp60 expression, new locations, functions and perspectives for cancer diagnosis and therapy. Cancer Biol. Ther. 2008, 7, 801–809. [Google Scholar] [CrossRef]
- Gupta, R.S. Evolution of the chaperonin families (Hsp60, Hsp10 and Tcp-1) of proteins and the origin of eukaryotic cells. Mol. Microbiol. 1995, 15, 1–11. [Google Scholar] [CrossRef]
- Pfister, G.; Stroh, C.M.; Perschinka, H.; Kind, M.; Knoflach, M.; Hinterdorfer, P.; Wick, G. Detection of HSP60 on the membrane surface of stressed human endothelial cells by atomic force and confocal microscopy. J. Cell Sci. 2005, 118, 1587–1594. [Google Scholar] [CrossRef] [Green Version]
- Binder, R.J. Functions of heat shock proteins in pathways of the innate and adaptive immune system. J. Immunol. 2014, 193, 5765–5771. [Google Scholar] [CrossRef]
- Srivastava, P. Roles of heat-shock proteins in innate and adaptive immunity. Nat. Rev. Immunol. 2002, 2, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Castellino, F.; Boucher, P.E.; Eichelberg, K.; Mayhew, M.; Rothman, J.E.; Houghton, A.N.; Germain, R.N. Receptor-mediated uptake of antigen/heat shock protein complexes results in major histocompatibility complex class I antigen presentation via two distinct processing pathways. J. Exp. Med. 2000, 191, 1957–1964. [Google Scholar] [CrossRef] [PubMed]
- Ishii, T.; Udono, H.; Yamano, T.; Ohta, H.; Uenaka, A.; Ono, T.; Hizuta, A.; Tanaka, N.; Srivastava, P.K.; Nakayama, E. Isolation of MHC class I-restricted tumor antigen peptide and its precursors associated with heat shock proteins hsp70, hsp90, and gp96. J. Immunol. 1999, 162, 1303–1309. [Google Scholar] [PubMed]
- Zugel, U.; Kaufmann, S.H. Role of heat shock proteins in protection from and pathogenesis of infectious diseases. Clin. Microbiol. Rev. 1999, 12, 19–39. [Google Scholar] [CrossRef] [PubMed]
- Kaufmann, S.H.; Schoel, B.; van Embden, J.D.; Koga, T.; Wand-Wurttenberger, A.; Munk, M.E.; Steinhoff, U. Heat-shock protein 60: Implications for pathogenesis of and protection against bacterial infections. Immunol. Rev. 1991, 121, 67–90. [Google Scholar] [CrossRef]
- Mayr, M.; Metzler, B.; Kiechl, S.; Willeit, J.; Schett, G.; Xu, Q.; Wick, G. Endothelial cytotoxicity mediated by serum antibodies to heat shock proteins of Escherichia coli and Chlamydia pneumoniae: Immune reactions to heat shock proteins as a possible link between infection and atherosclerosis. Circulation 1999, 99, 1560–1566. [Google Scholar] [CrossRef]
- Massa, M.; Passalia, M.; Manzoni, S.M.; Campanelli, R.; Ciardelli, L.; Yung, G.P.; Kamphuis, S.; Pistorio, A.; Meli, V.; Sette, A.; et al. Differential recognition of heat-shock protein dnaJ-derived epitopes by effector and Treg cells leads to modulation of inflammation in juvenile idiopathic arthritis. Arthritis Rheum. 2007, 56, 1648–1657. [Google Scholar] [CrossRef]
- Grundtman, C.; Kreutmayer, S.B.; Almanzar, G.; Wick, M.C.; Wick, G. Heat shock protein 60 and immune inflammatory responses in atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 960–968. [Google Scholar] [CrossRef]
- Wick, G.; Schett, G.; Amberger, A.; Kleindienst, R.; Xu, Q. Is atherosclerosis an Immunol.ly mediated disease? Immunol. Today 1995, 16, 27–33. [Google Scholar] [CrossRef]
- Juwono, J.; Martinus, R.D. Does Hsp60 Provide a Link between Mitochondrial Stress and Inflammation in Diabetes Mellitus? J. Diabetes Res. 2016, 2016, 8017571. [Google Scholar] [CrossRef]
- Birk, O.S.; Douek, D.C.; Elias, D.; Takacs, K.; Dewchand, H.; Gur, S.L.; Walker, M.D.; van der Zee, R.; Cohen, I.R.; Altmann, D.M. A role of Hsp60 in autoimmune diabetes: Analysis in a transgenic model. Proc. Natl. Acad. Sci. USA 1996, 93, 1032–1037. [Google Scholar] [CrossRef] [PubMed]
- Tukaj, S.; Kaminski, M. Heat shock proteins in the therapy of autoimmune diseases: Too simple to be true? Cell Stress Chaperones 2019, 24, 475–479. [Google Scholar] [CrossRef] [PubMed]
- Van Eden, W.; Jansen, M.A.A.; Ludwig, I.; van Kooten, P.; van der Zee, R.; Broere, F. The Enigma of Heat Shock Proteins in Immune Tolerance. Front. Immunol. 2017, 8, 1599. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, M.E. DAMPs, PAMPs and alarmins: All we need to know about danger. J. Leukoc. Biol. 2007, 81, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Pinar, O.; Ozden, Y.A.; Omur, E.; Muhtesem, G. Heat Shock Proteins in Multiple Sclerosis. Adv. Exp. Med. Biol. 2017, 958, 29–42. [Google Scholar] [CrossRef]
- Wilhelmus, M.M.; Otte-Holler, I.; Wesseling, P.; de Waal, R.M.; Boelens, W.C.; Verbeek, M.M. Specific association of small heat shock proteins with the pathological hallmarks of Alzheimer’s disease brains. Neuropathol. Appl. Neurobiol. 2006, 32, 119–130. [Google Scholar] [CrossRef]
- Tezel, G.; Seigel, G.M.; Wax, M.B. Autoantibodies to small heat shock proteins in glaucoma. Invest. Ophthalmol. Vis. Sci. 1998, 39, 2277–2287. [Google Scholar]
- EGS. European Glaucoma Society Terminology and Guidelines for Glaucoma, 4th Edition—Chapter 2: Classification and terminology. Br. J. Ophthalmol. 2017, 101, 73–127. [Google Scholar] [CrossRef]
- Wax, M.B.; Barrett, D.A.; Pestronk, A. Increased incidence of paraproteinemia and autoantibodies in patients with normal-pressure glaucoma. Am. J. Ophthalmol. 1994, 117, 561–568. [Google Scholar] [CrossRef]
- Wax, M.B.; Tezel, G.; Edward, P.D. Clinical and ocular histopathological findings in a patient with normal-pressure glaucoma. Arch. Ophthalmol. 1998, 116, 993–1001. [Google Scholar] [CrossRef]
- Wax, M.B.; Tezel, G.; Kawase, K.; Kitazawa, Y. Serum autoantibodies to heat shock proteins in glaucoma patients from Japan and the United States. Ophthalmology 2001, 108, 296–302. [Google Scholar] [CrossRef]
- Tezel, G.; Wax, M.B. The mechanisms of hsp27 antibody-mediated apoptosis in retinal neuronal cells. J. Neurosci. 2000, 20, 3552–3562. [Google Scholar] [CrossRef] [PubMed]
- Tezel, G.; Hernandez, R.; Wax, M.B. Immunostaining of heat shock proteins in the retina and optic nerve head of normal and glaucomatous eyes. Arch. Ophthalmol. 2000, 118, 511–518. [Google Scholar] [CrossRef] [PubMed]
- Grus, F.H.; Joachim, S.C.; Hoffmann, E.M.; Pfeiffer, N. Complex autoantibody repertoires in patients with glaucoma. Mol. Vis. 2004, 10, 132–137. [Google Scholar]
- Joachim, S.C.; Bruns, K.; Lackner, K.J.; Pfeiffer, N.; Grus, F.H. Antibodies to alpha B-crystallin, vimentin, and heat shock protein 70 in aqueous humor of patients with normal tension glaucoma and IgG antibody patterns against retinal antigen in aqueous humor. Curr. Eye Res. 2007, 32, 501–509. [Google Scholar] [CrossRef]
- Boehm, N.; Wolters, D.; Thiel, U.; Lossbrand, U.; Wiegel, N.; Pfeiffer, N.; Grus, F.H. New insights into autoantibody profiles from immune privileged sites in the eye: A glaucoma study. Brain Behav. Immun. 2012, 26, 96–102. [Google Scholar] [CrossRef]
- Joachim, S.C.; Wuenschig, D.; Pfeiffer, N.; Grus, F.H. IgG antibody patterns in aqueous humor of patients with primary open angle glaucoma and pseudoexfoliation glaucoma. Mol. Vis. 2007, 13, 1573–1579. [Google Scholar]
- Sacca, S.C.; Centofanti, M.; Izzotti, A. New proteins as vascular biomarkers in primary open angle glaucomatous aqueous humor. Investig. Ophthalmol. Vis. Sci. 2012, 53, 4242–4253. [Google Scholar] [CrossRef]
- Nowak, A.; Szaflik, J.P.; Gacek, M.; Przybylowska-Sygut, K.; Kaminska, A.; Szaflik, J.; Majsterek, I. BDNF and HSP gene polymorphisms and their influence on the progression of primary open-angle glaucoma in a Polish population. Arch. Med. Sci. 2014, 10, 1206–1213. [Google Scholar] [CrossRef] [Green Version]
- Ayub, H.; Khan, M.I.; Micheal, S.; Akhtar, F.; Ajmal, M.; Shafique, S.; Ali, S.H.; den Hollander, A.I.; Ahmed, A.; Qamar, R. Association of eNOS and HSP70 gene polymorphisms with glaucoma in Pakistani cohorts. Mol. Vis. 2010, 16, 18–25. [Google Scholar]
- Tosaka, K.; Mashima, Y.; Funayama, T.; Ohtake, Y.; Kimura, I.; Glaucoma Gene Research, G. Association between open-angle glaucoma and gene polymorphism for heat-shock protein 70-1. Jpn. J. Ophthalmol. 2007, 51, 417–423. [Google Scholar] [CrossRef] [PubMed]
- Cancino-Diaz, M.E.; Sanchez-Becerra, M.; Elizondo-Olascoaga, C.; Rodriguez-Martinez, S.; Cancino-Diaz, J.C. Amino acid regions 357-368 and 418-427 of Streptococcus pyogenes 60-kDa heat shock protein are recognized by antibodies from glaucomatous patient sera. Microb. Pathog. 2010, 48, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Grabska-Liberek, I.; Skonieczna, K.; Olesinska, M.; Terelak-Borys, B.; Kociecki, J.; Sikora, M.; Jamrozy-Witkowska, A.; Tesla, P.; Czarnocka, B. Levels of antibodies against human heat shock protein (HSP) 60 in patients with glaucoma in Poland. Med. Sci. Monit. 2015, 21, 828–832. [Google Scholar] [CrossRef] [PubMed]
- Nowak, A.; Rozpedek, W.; Cuchra, M.; Wojtczak, R.; Siwak, M.; Szymanek, K.; Szaflik, M.; Szaflik, J.; Szaflik, J.; Majsterek, I. Association of the expression level of the neurodegeneration-related proteins with the risk of development and progression of primary open-angle glaucoma. Acta Ophthalmol. 2018, 96, e97–e98. [Google Scholar] [CrossRef] [PubMed]
- Wax, M.B.; Tezel, G.; Yang, J.; Peng, G.; Patil, R.V.; Agarwal, N.; Sappington, R.M.; Calkins, D.J. Induced autoimmunity to heat shock proteins elicits glaucomatous loss of retinal ganglion cell neurons via activated T-cell-derived fas-ligand. J. Neurosci. 2008, 28, 12085–12096. [Google Scholar] [CrossRef]
- Joachim, S.C.; Grus, F.H.; Kraft, D.; White-Farrar, K.; Barnes, G.; Barbeck, M.; Ghanaati, S.; Cao, S.; Li, B.; Wax, M.B. Complex antibody profile changes in an experimental autoimmune glaucoma animal model. Investig. Ophthalmol. Vis. Sci. 2009, 50, 4734–4742. [Google Scholar] [CrossRef]
- Casola, C.; Reinehr, S.; Kuehn, S.; Stute, G.; Spiess, B.M.; Dick, H.B.; Joachim, S.C. Specific Inner Retinal Layer Cell Damage in an Autoimmune Glaucoma Model Is Induced by GDNF With or Without HSP27. Invest. Ophthalmol. Vis. Sci. 2016, 57, 3626–3639. [Google Scholar] [CrossRef] [Green Version]
- Casola, C.; Schiwek, J.E.; Reinehr, S.; Kuehn, S.; Grus, F.H.; Kramer, M.; Dick, H.B.; Joachim, S.C. S100 Alone Has the Same Destructive Effect on Retinal Ganglion Cells as in Combination with HSP 27 in an Autoimmune Glaucoma Model. J. Mol. Neurosci. 2015, 56, 228–236. [Google Scholar] [CrossRef]
- Reinehr, S.; Kuehn, S.; Casola, C.; Koch, D.; Stute, G.; Grotegut, P.; Dick, H.B.; Joachim, S.C. HSP27 immunization reinforces AII amacrine cell and synapse damage induced by S100 in an autoimmune glaucoma model. Cell Tissue Res. 2018, 371, 237–249. [Google Scholar] [CrossRef]
- Joachim, S.C.; Wax, M.B.; Seidel, P.; Pfeiffer, N.; Grus, F.H. Enhanced Characterization of Serum Autoantibody Reactivity Following HSP 60 Immunization in a Rat Model of Experimental Autoimmune Glaucoma. Curr. Eye Res. 2010, 35, 900–908. [Google Scholar] [CrossRef]
- Grotegut, P.; Kuehn, S.; Doepper, H.; Dick, H.B.; Joachim, S.C. Intravitreal HSP27 injection leads to retinal degeneration in rats. Investig. Ophthalmol. Vis. Sci. 2019, 60, 4929. [Google Scholar]
- Sakai, M.; Sakai, H.; Nakamura, Y.; Fukuchi, T.; Sawaguchi, S. Immunolocalization of heat shock proteins in the retina of normal monkey eyes and monkey eyes with laser-induced glaucoma. Jpn. J. Ophthalmol. 2003, 47, 42–52. [Google Scholar] [CrossRef]
- Kalesnykas, G.; Niittykoski, M.; Rantala, J.; Miettinen, R.; Salminen, A.; Kaarniranta, K.; Uusitalo, H. The expression of heat shock protein 27 in retinal ganglion and glial cells in a rat glaucoma model. Neuroscience 2007, 150, 692–704. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, F.; Brown, K.M.; Stephan, D.A.; Morrison, J.C.; Johnson, E.C.; Tomarev, S.I. Microarray analysis of changes in mRNA levels in the rat retina after experimental elevation of intraocular pressure. Investig. Ophthalmol. Vis. Sci. 2004, 45, 1247–1258. [Google Scholar] [CrossRef]
- Chen, H.; Cho, K.S.; Vu, T.H.K.; Shen, C.H.; Kaur, M.; Chen, G.; Mathew, R.; McHam, M.L.; Fazelat, A.; Lashkari, K.; et al. Commensal microflora-induced T cell responses mediate progressive neurodegeneration in glaucoma. Nat. Commun. 2018, 9, 3209. [Google Scholar] [CrossRef]
- Chidlow, G.; Wood, J.P.; Casson, R.J. Expression of inducible heat shock proteins Hsp27 and Hsp70 in the visual pathway of rats subjected to various models of retinal ganglion cell injury. PLoS ONE 2014, 9, e114838. [Google Scholar] [CrossRef]
- Wang, G.H.; Xing, Y.Q. Evaluation of heat shock protein (HSP-72) expression in retinal ganglion cells of rats with glaucoma. Exp. Ther. Med. 2017, 14, 1577–1581. [Google Scholar] [CrossRef] [Green Version]
- Sohn, S.; Im, J.E.; Kim, T.E.; Kee, C. Effect of heat shock protein 72 expression on etoposide-induced cell death of rat retinal ganglion cells. Korean J. Ophthalmol. 2013, 27, 48–51. [Google Scholar] [CrossRef]
- Piri, N.; Song, M.; Kwong, J.M.; Caprioli, J. Modulation of alpha and beta crystallin expression in rat retinas with ocular hypertension-induced ganglion cell degeneration. Brain Res. 2007, 1141, 1–9. [Google Scholar] [CrossRef]
- Naskar, R.; Thanos, S. Retinal gene profiling in a hereditary rodent model of elevated intraocular pressure. Mol. Vis. 2006, 12, 1199–1210. [Google Scholar]
- Piri, N.; Kwong, J.M.; Gu, L.; Caprioli, J. Heat shock proteins in the retina: Focus on HSP70 and alpha crystallins in ganglion cell survival. Prog. Retinal Eye Res. 2016, 52, 22–46. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bruey, J.M.; Ducasse, C.; Bonniaud, P.; Ravagnan, L.; Susin, S.A.; Diaz-Latoud, C.; Gurbuxani, S.; Arrigo, A.P.; Kroemer, G.; Solary, E.; et al. Hsp27 negatively regulates cell death by interacting with cytochrome c. Nat. Cell Biol. 2000, 2, 645–652. [Google Scholar] [CrossRef] [PubMed]
- Mosser, D.D.; Caron, A.W.; Bourget, L.; Denis-Larose, C.; Massie, B. Role of the human heat shock protein hsp70 in protection against stress-induced apoptosis. Mol. Cell. Biol. 1997, 17, 5317–5327. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mao, Y.W.; Liu, J.P.; Xiang, H.; Li, D.W. Human alphaA- and alphaB-crystallins bind to Bax and Bcl-X(S) to sequester their translocation during staurosporine-induced apoptosis. Cell Death Differ. 2004, 11, 512–526. [Google Scholar] [CrossRef]
- Sreekumar, P.G.; Kannan, R.; Kitamura, M.; Spee, C.; Barron, E.; Ryan, S.J.; Hinton, D.R. alphaB crystallin is apically secreted within exosomes by polarized human retinal pigment epithelium and provides neuroprotection to adjacent cells. PLoS ONE 2010, 5, e12578. [Google Scholar] [CrossRef]
- Kamradt, M.C.; Chen, F.; Cryns, V.L. The small heat shock protein alpha B-crystallin negatively regulates cytochrome c- and caspase-8-dependent activation of caspase-3 by inhibiting its autoproteolytic maturation. J. Biol. Chem. 2001, 276, 16059–16063. [Google Scholar] [CrossRef]
- Thanos, S.; Bohm, M.R.; Meyer zu Horste, M.; Prokosch-Willing, V.; Hennig, M.; Bauer, D.; Heiligenhaus, A. Role of crystallins in ocular neuroprotection and axonal regeneration. Prog. Retinal Eye Res. 2014, 42, 145–161. [Google Scholar] [CrossRef]
- Ebrahimi-Fakhari, D.; Saidi, L.J.; Wahlster, L. Molecular chaperones and protein folding as therapeutic targets in Parkinson’s disease and other synucleinopathies. Acta Neuropathol. Commun. 2013, 1, 79. [Google Scholar] [CrossRef]
- Saidi, L.J.; Polydoro, M.; Kay, K.R.; Sanchez, L.; Mandelkow, E.M.; Hyman, B.T.; Spires-Jones, T.L. Carboxy terminus heat shock protein 70 interacting protein reduces tau-associated degenerative changes. J. Alzheimers Dis. 2015, 44, 937–947. [Google Scholar] [CrossRef]
- Petrucelli, L.; Dickson, D.; Kehoe, K.; Taylor, J.; Snyder, H.; Grover, A.; De Lucia, M.; McGowan, E.; Lewis, J.; Prihar, G.; et al. CHIP and Hsp70 regulate tau ubiquitination, degradation and aggregation. Hum. Mol. Genet. 2004, 13, 703–714. [Google Scholar] [CrossRef] [Green Version]
- Dubey, A.; Prajapati, K.S.; Swamy, M.; Pachauri, V. Heat shock proteins: A therapeutic target worth to consider. Vet. World 2015, 8, 46–51. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, A.; Oshitari, T.; Negishi, H.; Dezawa, M.; Mizota, A.; Adachi-Usami, E. Protection of retinal ganglion cells from ischemia-reperfusion injury by electrically applied Hsp27. Investig. Ophthalmol. Vis. Sci. 2001, 42, 3283–3286. [Google Scholar]
- Qing, G.; Duan, X.; Jiang, Y. Heat shock protein 72 protects retinal ganglion cells in rat model of acute glaucoma. Yan Ke Xue Bao Eye Sci. 2005, 21, 163–168. [Google Scholar]
- Park, K.H.; Cozier, F.; Ong, O.C.; Caprioli, J. Induction of heat shock protein 72 protects retinal ganglion cells in a rat glaucoma model. Investig. Ophthalmol. Vis. Sci. 2001, 42, 1522–1530. [Google Scholar]
- Caprioli, J.; Ishii, Y.; Kwong, J.M. Retinal ganglion cell protection with geranylgeranylacetone, a heat shock protein inducer, in a rat glaucoma model. Trans. Am. Ophthalmol. Soc. 2003, 101, 39–50. [Google Scholar]
- Anders, F.; Liu, A.; Mann, C.; Teister, J.; Lauzi, J.; Thanos, S.; Grus, F.H.; Pfeiffer, N.; Prokosch, V. The Small Heat Shock Protein alpha-Crystallin B Shows Neuroprotective Properties in a Glaucoma Animal Model. Int. J. Mol. Sci. 2017, 18, 2418. [Google Scholar] [CrossRef]
- Prakken, B.J.; Samodal, R.; Le, T.D.; Giannoni, F.; Yung, G.P.; Scavulli, J.; Amox, D.; Roord, S.; de Kleer, I.; Bonnin, D.; et al. Epitope-specific immunotherapy induces immune deviation of proinflammatory T cells in rheumatoid arthritis. Proc. Natl. Acad. Sci. USA 2004, 101, 4228–4233. [Google Scholar] [CrossRef] [Green Version]
- Vanags, D.; Williams, B.; Johnson, B.; Hall, S.; Nash, P.; Taylor, A.; Weiss, J.; Feeney, D. Therapeutic efficacy and safety of chaperonin 10 in patients with rheumatoid arthritis: A double-blind randomised trial. Lancet 2006, 368, 855–863. [Google Scholar] [CrossRef]
- Asea, A.; Kraeft, S.K.; Kurt-Jones, E.A.; Stevenson, M.A.; Chen, L.B.; Finberg, R.W.; Koo, G.C.; Calderwood, S.K. HSP70 stimulates cytokine production through a CD14-dependant pathway, demonstrating its dual role as a chaperone and cytokine. Nat. Med. 2000, 6, 435–442. [Google Scholar] [CrossRef]
- Jin, C.; Cleveland, J.C.; Ao, L.; Li, J.; Zeng, Q.; Fullerton, D.A.; Meng, X. Human myocardium releases heat shock protein 27 (HSP27) after global ischemia: The proinflammatory effect of extracellular HSP27 through toll-like receptor (TLR)-2 and TLR4. Mol. Med. 2014, 20, 280–289. [Google Scholar] [CrossRef]
- Lamb, J.R.; Young, D.B. T cell recognition of stress proteins. A link between infectious and autoimmune disease. Mol. Biol. Med. 1990, 7, 311–321. [Google Scholar] [PubMed]
- Benagiano, M.; D’Elios, M.M.; Amedei, A.; Azzurri, A.; van der Zee, R.; Ciervo, A.; Rombola, G.; Romagnani, S.; Cassone, A.; Del Prete, G. Human 60-kDa heat shock protein is a target autoantigen of T cells derived from atherosclerotic plaques. J. Immunol. 2005, 174, 6509–6517. [Google Scholar] [CrossRef] [PubMed]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsai, T.; Grotegut, P.; Reinehr, S.; Joachim, S.C. Role of Heat Shock Proteins in Glaucoma. Int. J. Mol. Sci. 2019, 20, 5160. https://doi.org/10.3390/ijms20205160
Tsai T, Grotegut P, Reinehr S, Joachim SC. Role of Heat Shock Proteins in Glaucoma. International Journal of Molecular Sciences. 2019; 20(20):5160. https://doi.org/10.3390/ijms20205160
Chicago/Turabian StyleTsai, Teresa, Pia Grotegut, Sabrina Reinehr, and Stephanie C. Joachim. 2019. "Role of Heat Shock Proteins in Glaucoma" International Journal of Molecular Sciences 20, no. 20: 5160. https://doi.org/10.3390/ijms20205160
APA StyleTsai, T., Grotegut, P., Reinehr, S., & Joachim, S. C. (2019). Role of Heat Shock Proteins in Glaucoma. International Journal of Molecular Sciences, 20(20), 5160. https://doi.org/10.3390/ijms20205160






