Co-Stimulation of Purinergic P2X4 and Prostanoid EP3 Receptors Triggers Synergistic Degranulation in Murine Mast Cells
Abstract
1. Introduction
2. Results
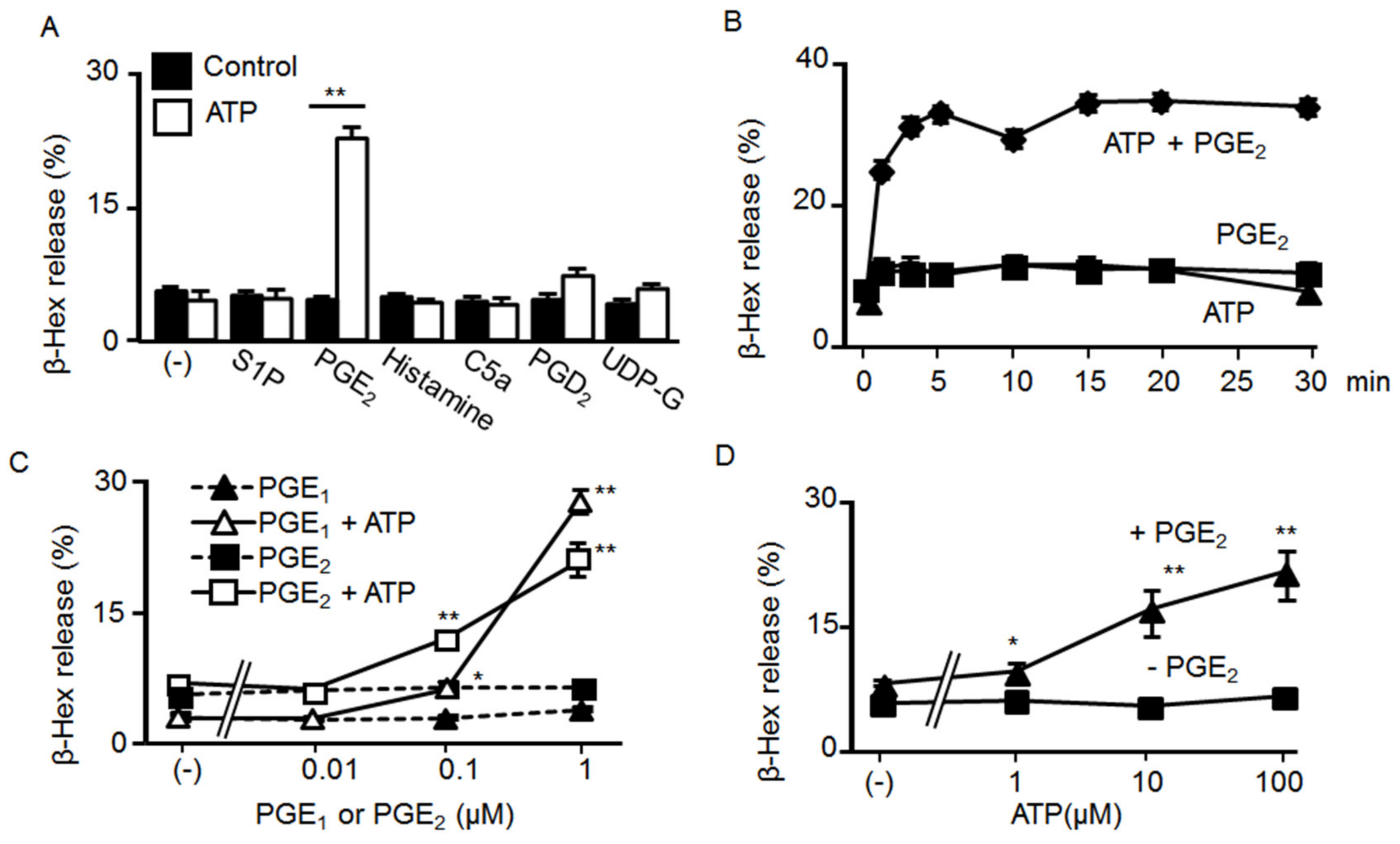
2.1. Effects of ATP and GPCR Agonist co-Stimulation on BMMC Degranulation
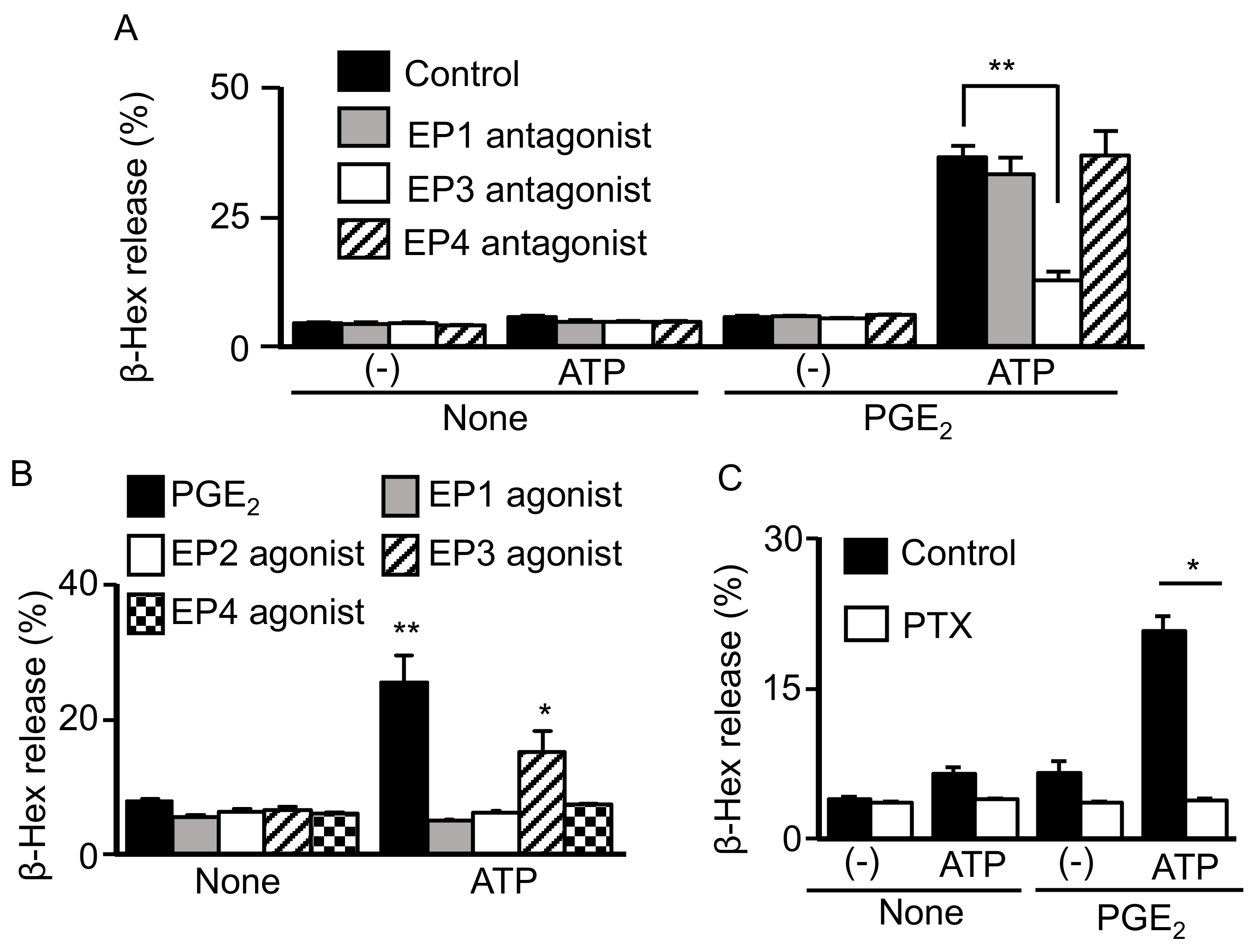
2.2. Involvement of Gi-Coupled EP3R in Synergistic Degranulation Induced by PGE2 and ATP
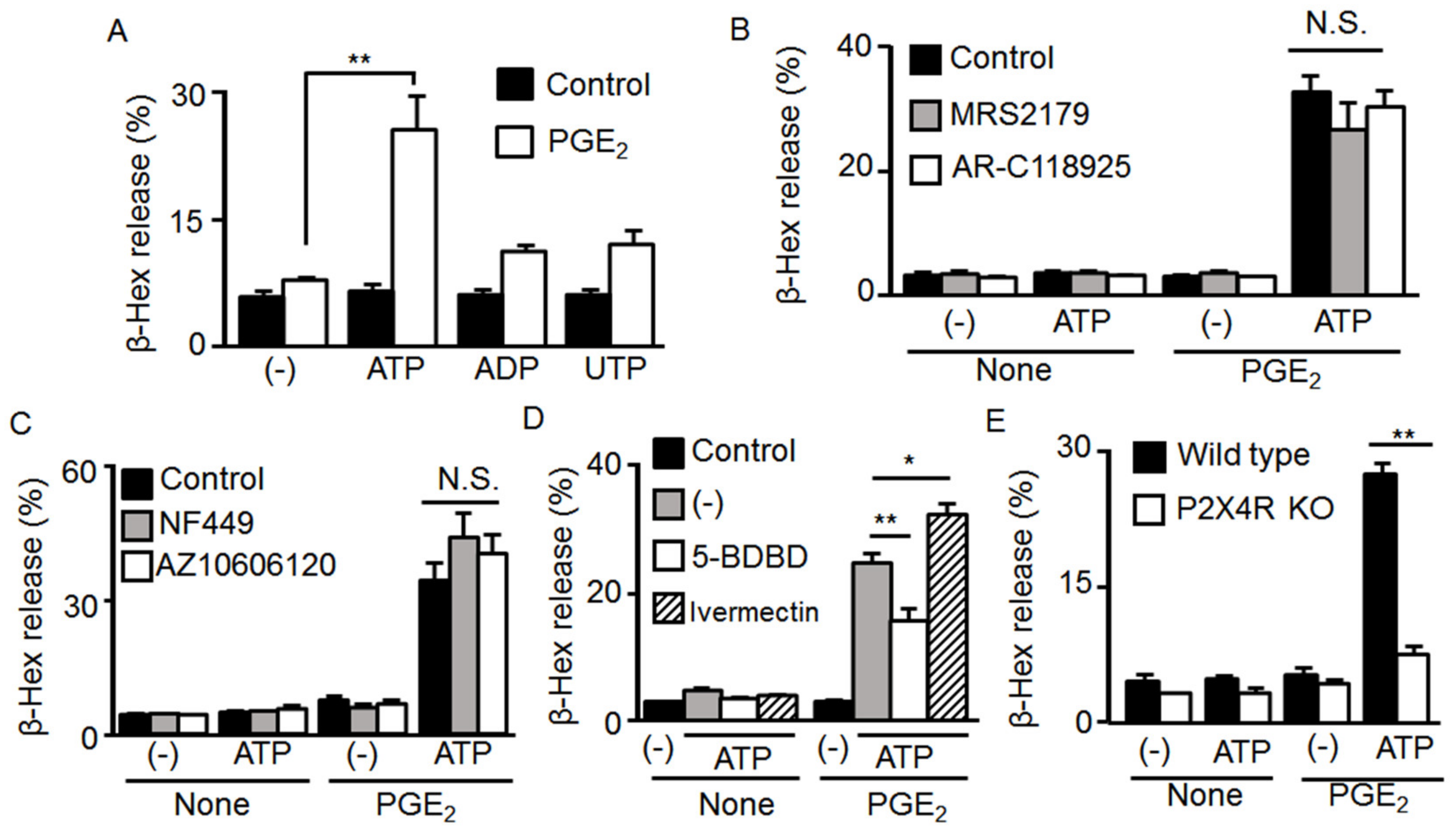
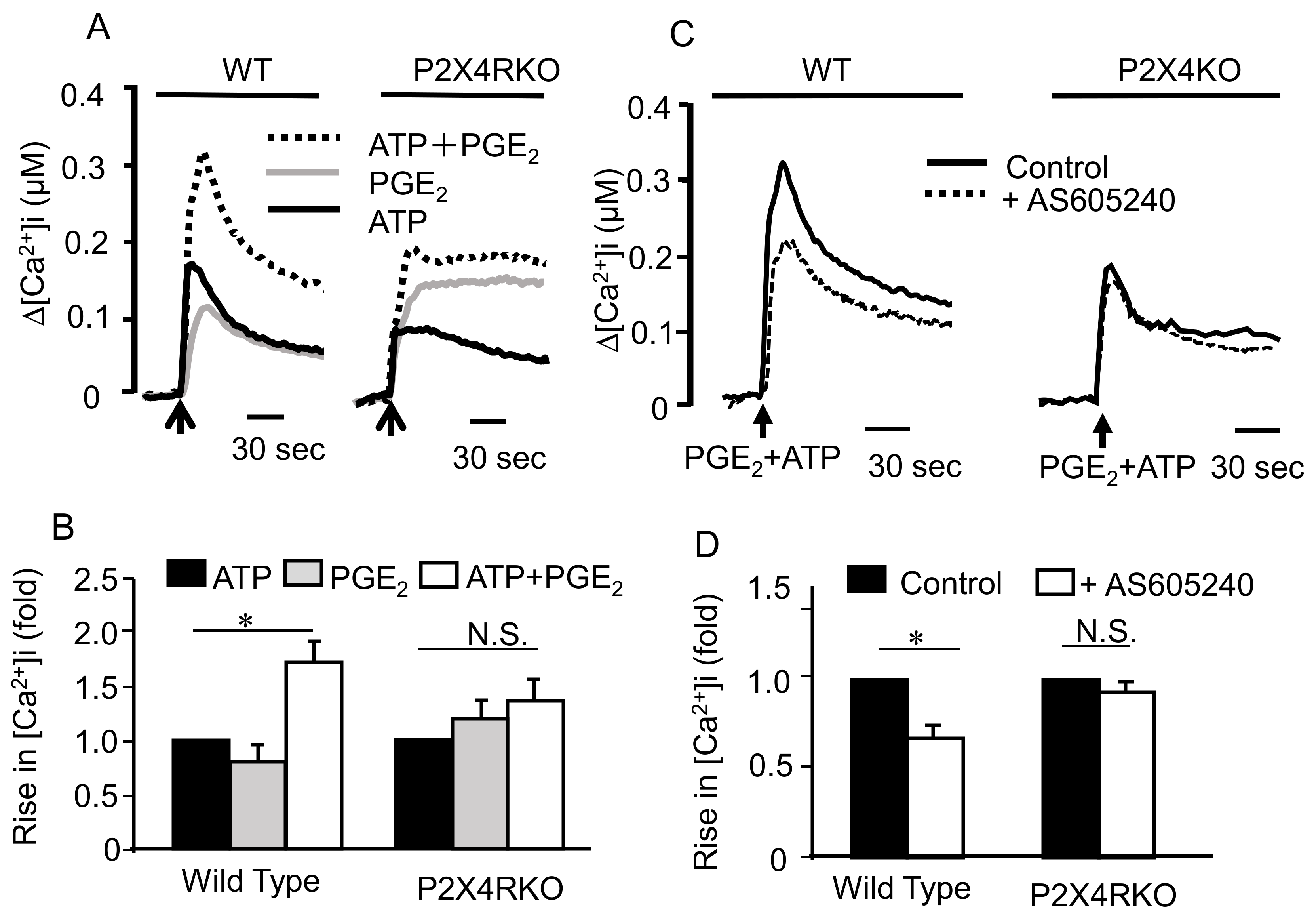
2.3. Involvement of Ionotropic P2X4R in the Effect of ATP and PGE2 on Degranulation
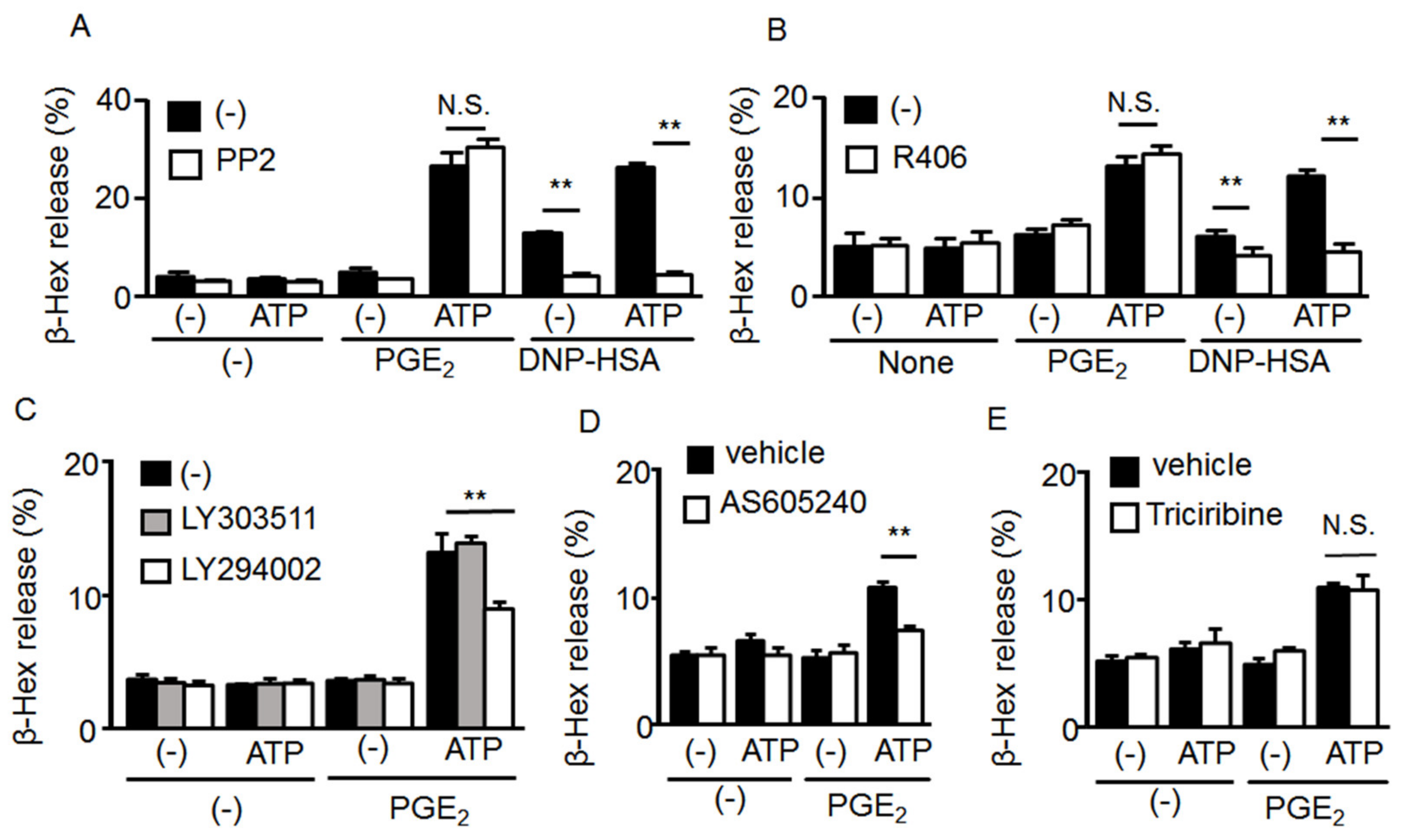
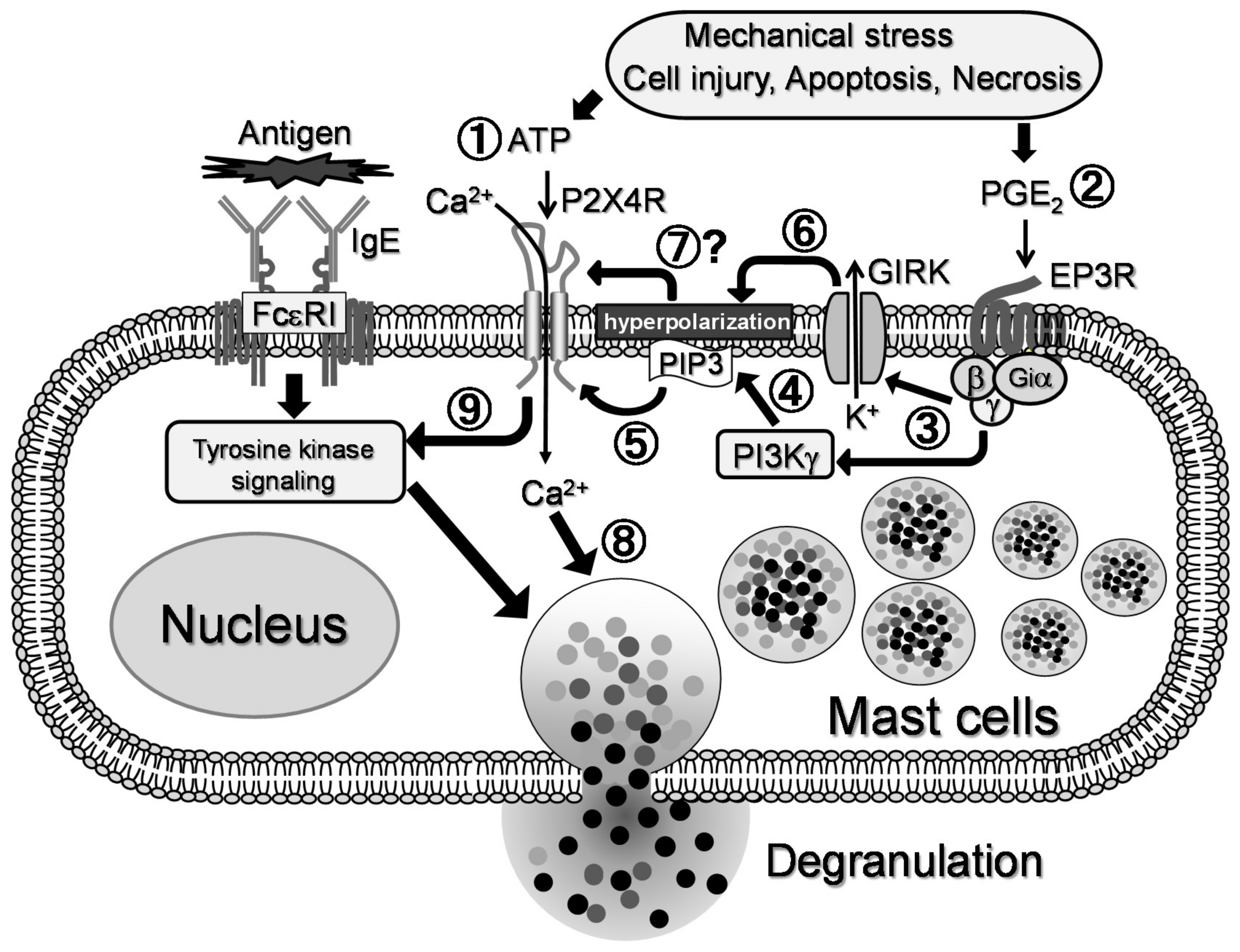
2.4. Mechanism underlying the Synergistic Degranulation Induced by ATP and PGE2
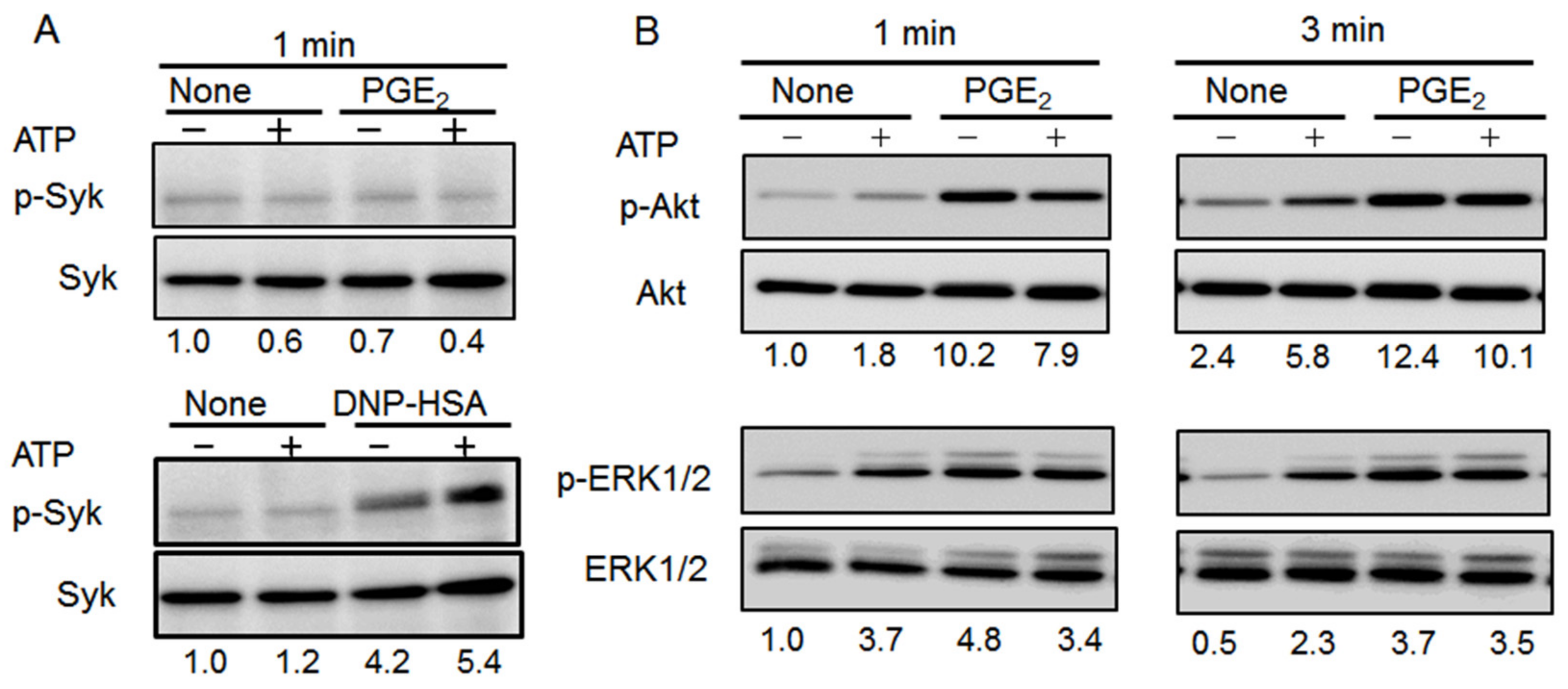
2.5. Effects of Co-Stimulating BMMCs with ATP and PGE2 on ERK1/2, Akt, and Syk Phosphorylation
2.6. Effects of Co-Stimulating BMMCs with ATP and PGE2 on [Ca2+]i
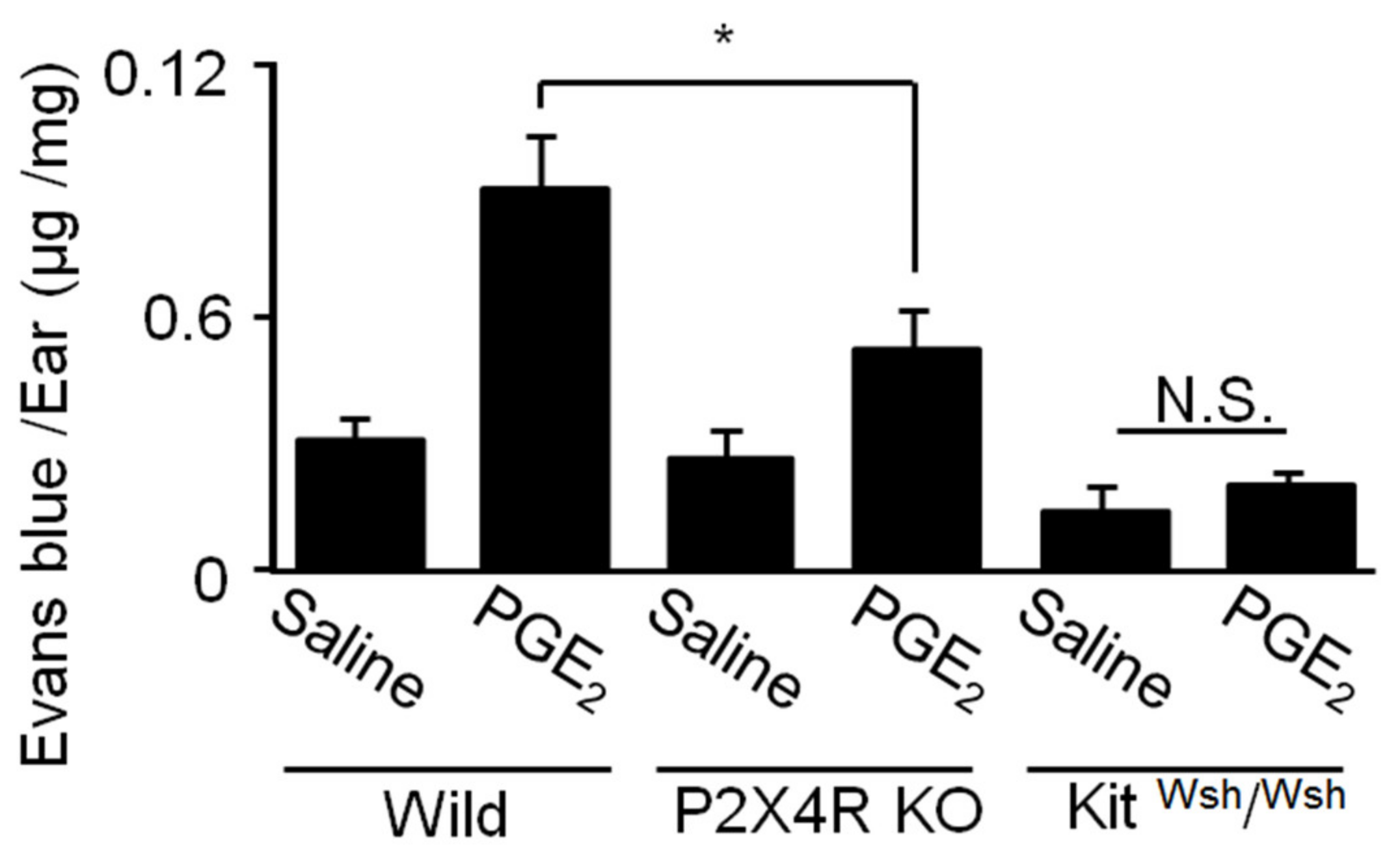
2.7. Role of P2X4R Signaling in PGE2-Induced Skin MC Activation in Vivo
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Animals
4.3. Cell Culture
4.4. Degranulation Assay
4.5. [Ca2+]i Measurement
4.6. Western Blot
4.7. Quantitative RT-PCR (qRT-PCR)
4.8. PGE2-Induced Skin Edema
4.9. Statistics
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| [Ca2+]i | Intracellular Ca2+ concentration |
| A3R | A3 receptor |
| Ag | Antigen |
| APC | Allophycocyanin |
| BMMCs | Bone marrow-derived mast cells |
| DNP-HSA | 2,4-dinitrophenyl human serum albumin |
| EP3R | EP3 receptor |
| FcεRI | High affinity IgE receptors |
| GPCR | G-protein-coupled receptors |
| IL | Interleukin |
| KRH | Krebs-Ringer-HEPES |
| MC | Mast cell |
| P2X4R | P2X4 receptor |
| PE | Phycoerythrin |
| PG | Prostaglandin |
| PI3K | Phosphoinositide 3-kinase |
| PLC | Phospholipase C |
| PTX | Pertussis toxin |
| PVDF | Immobilon-P polyvinylidene fluoride |
| qRT-PCR | Quantitative reverse transcription-polymerase chain reaction |
| S1P | Sphingosine-1-phosphate |
| SDS-PAGE | Sodium dodecyl sulfate polyacrylamide gel electrophoresis |
References
- Metcalfe, D.D.; Baram, D.; Mekori, Y. Mast cells. Pharmacol. Rev. 1997, 77, 1033–1079. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, E.Z.M.; Jamur, M.C.; Oliver, C. Mast cell function: A new vision of an old cell. J. Histochem. Cytochem. 2014, 62, 698–738. [Google Scholar] [CrossRef] [PubMed]
- Metcalfe, D.D.; Peavy, R.D.; Gilfillan, A.M. Mechanisms of mast cell signaling in anaphylaxis. J. Allergy Clin. Immunol. 2009, 124, 639–646. [Google Scholar] [CrossRef] [PubMed]
- Metzger, H. The receptor with high affinity for IgE. Immunol. Rev. 1992, 125, 37–48. [Google Scholar] [CrossRef]
- Gilfillan, A.M.; Tkaczyk, C. Integrated signalling pathways for mast-cell activation. Nat. Rev. Immunol. 2006, 6, 218–230. [Google Scholar] [CrossRef]
- Kuehn, H.S.; Gilfillan, A.M. G protein-coupled receptors and the modification of FcεRI-mediated mast cell activation. Immunol. Lett. 2007, 113, 59–69. [Google Scholar] [CrossRef]
- Moncada, S.; Ferreira, S.; Vane, J. Prostaglandins, aspirin-like drugs and the oedema of inflammation. Nature 1973, 246, 217–219. [Google Scholar] [CrossRef]
- Borea, P.A.; Gessi, S.; Merighi, S.; Varani, K. Adenosine as a multi-signalling guardian angel in human diseases: When, where and how does it exert its protective Effects? Trends Pharmacol. Sci. 2016, 37, 419–434. [Google Scholar] [CrossRef]
- Kuehn, H.S.; Beaven, M.A.; Ma, H.-T.; Kim, M.-S.; Metcalfe, D.D.; Gilfillan, A.M. Synergistic activation of phospholipases Cγ and Cβ: A novel mechanism for PI3K-independent enhancement of FcεRI-induced mast cell mediator release. Cell. Signal. 2008, 20, 625–636. [Google Scholar] [CrossRef]
- Kuehn, H.S.; Jung, M.-Y.; Beaven, M.A.; Metcalfe, D.D.; Gilfillan, A.M. Prostaglandin E 2 activates and utilizes mTORC2 as a central signaling locus for the regulation of mast cell chemotaxis and mediator release. J. Biol. Chem. 2011, 286, 391–402. [Google Scholar] [CrossRef]
- Laffargue, M.; Calvez, R.; Finan, P.; Trifilieff, A.; Barbier, M.; Altruda, F.; Hirsch, E.; Wymann, M.P. Phosphoinositide 3-kinase γ is an essential amplifier of mast cell function. Immunity 2002, 16, 441–451. [Google Scholar] [CrossRef]
- Yoshida, K.; Ito, M.; Matsuoka, I. Divergent regulatory roles of extracellular ATP in the degranulation response of mouse bone marrow-derived mast cells. Int. Immunopharmacol. 2017, 43, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, K.; Ito, M.; Yamamoto, K.; Koizumi, S.; Tanaka, S.; Furuta, K.; Matsuoka, I. Extracellular ATP augments antigen-induced murine mast cell degranulation and allergic responses via P2X4 receptor activation. J. Immunol. 2019. submitted for publication. [Google Scholar]
- Redegeld, F.A.; Yu, Y.; Kumari, S.; Charles, N.; Blank, U. Non-IgE mediated mast cell activation. Immunol. Rev. 2018, 282, 87–113. [Google Scholar] [CrossRef] [PubMed]
- Fitz, J.G. Regulation of cellular ATP release. Trans. Am. Clin. Climatol. Assoc. 2007, 118, 199–208. [Google Scholar]
- Gallenga, C.; Pandolfi, F.; Caraffa, A.; Ronconi, G.; Toniato, E.; Martinotti, S.; Conti, P. Interleukin-1 family cytokines and mast cells: activation and inhibition. J. Biol. Regul. Homeost. Agents 2019, 33, 1–6. [Google Scholar]
- Caraffa, A.; Gallenga, C.E.; Kritas, S.K.; Ronconi, G.; Conti, P. Impact of mast cells in systemic lupus erythematosus: can inflammation be inhibited? J. Biol. Regul. Homeost. Agents 2019, 33, 669–673. [Google Scholar]
- Antonopulos, D.; Tsilioni, I.; Balatsos, N.A.A.; Gourgoulianis, K.I.; Theoharides, T.C. The mast cell—Neurofibromatosis connection. J. Biol. Regul. Homeost. Agents 2019, 33, 657–659. [Google Scholar]
- Lazarowski, E.R. Vesicular and conductive mechanisms of nucleotide release. Purinergic. Signal 2012, 8, 359–373. [Google Scholar] [CrossRef]
- Dosch, M.; Gerber, J.; Jebbawi, F.; Beldi, G. Mechanisms of ATP Release by inflammatory cells. Int. J. Mol. Sci. 2018, 19, 1222. [Google Scholar] [CrossRef]
- Burnstock, G. Purinergic signalling: Therapeutic developments. Front. Pharmacol. 2017, 8, 661. [Google Scholar] [CrossRef] [PubMed]
- Morimoto, K.; Shirata, N.; Taketomi, Y.; Tsuchiya, S.; Segi-Nishida, E.; Inazumi, T.; Kabashima, K.; Tanaka, S.; Murakami, M.; Narumiya, S.; et al. Prostaglandin E2–EP3 signaling induces inflammatory swelling by mast cell activation. J. Immunol. 2014, 192, 1130–1137. [Google Scholar] [CrossRef] [PubMed]
- Needleman, P.; Minkes, M.S.; Douglas, J.R. Stimulation of prostaglandin biosynthesis by adenine nucleotides: Profile of prostaglandin release by perfused organs. Circ. Res. 1974, 34, 455–460. [Google Scholar] [CrossRef] [PubMed]
- Loredo, G.A.; Benton, H.P. ATP and UTP activate calcium-mobilizing P2U-like receptors and act synergistically with interleukin-1 to stimulate prostaglandin E2 release from human rheumatoid synovial cells. Arthritis Rheum. 1998, 41, 246–255. [Google Scholar] [CrossRef]
- Sugimoto, Y.; Narumiya, S. Prostaglandin E receptors. J. Biol. Chem. 2007, 282, 11613–11617. [Google Scholar] [CrossRef]
- Khakh, B.S.; Proctor, W.R.; Dunwiddie, T.V.; Labarca, C.; Lester, H.A. Allosteric control of gating and kinetics at P2X 4 receptor channels. J. Neurosci. 1999, 19, 7289–7299. [Google Scholar] [CrossRef]
- Bernier, L.-P.; Ase, A.R.; Chevallier, S.; Blais, D.; Zhao, Q.; Boue-Grabot, E.; Logothetis, D.; Seguela, P. Phosphoinositides regulate P2X4 ATP-gated channels through direct interactions. J. Neurosci. 2008, 28, 12938–12945. [Google Scholar] [CrossRef]
- Qian, Y.X.; McCloskey, M.A. Activation of mast cell K+ channels through multiple G protein-linked receptors. Proc. Natl. Acad. Sci. USA 1993, 90, 7844–7848. [Google Scholar] [CrossRef]
- Ashmole, I.; Bradding, P. Ion channels regulating mast cell biology. Clin. Exp. Allergy 2013, 43, 491–502. [Google Scholar] [CrossRef]
- Shumilina, E.; Lam, R.S.; Wölbing, F.; Matzner, N.; Zemtsova, I.M.; Sobiesiak, M.; Mahmud, H.; Sausbier, U.; Biedermann, T.; Ruth, P.; et al. Blunted IgE-mediated activation of mast cells in mice lacking the Ca2+ -Activated K+ channel K Ca 3.1. J. Immunol. 2008, 180, 8040–8047. [Google Scholar] [CrossRef]
- Tsuda, M.; Shigemoto-Mogami, Y.; Koizumi, S.; Mizokoshi, A.; Kohsaka, S.; Salter, M.W.; Inoue, K. P2X4 receptors induced in spinal microglia gate tactile allodynia after nerve injury. Nature 2003, 424, 778–783. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Guo, N.; Ma, Y.; Ning, B.; Wang, Y.; Kou, L. Inhibition of P2X4 suppresses joint inflammation and damage in collagen-induced arthritis. Inflammation 2014, 37, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Ledderose, C.; Fakhari, M.; Lederer, J.A.; Robson, S.C.; Visner, G.A.; Junger, W.G.; Liu, K.; Kondo, Y.; Slubowski, C.J.; Dertnig, T.; et al. Purinergic P2X4 receptors and mitochondrial ATP production regulate T cell migration. J. Clin. Invest. 2018, 128, 3583–3594. [Google Scholar] [CrossRef] [PubMed]
- Zech, A.; Wiesler, B.; Ayata, C.K.; Schlaich, T.; Dürk, T.; Hoßfeld, M.; Ehrat, N.; Cicko, S.; Idzko, M.; Zech, A.; et al. P2rx4 deficiency in mice alleviates allergen-induced airway inflammation. Oncotarget 2016, 7, 80288–80297. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, K.; Sokabe, T.; Matsumoto, T.; Yoshimura, K.; Shibata, M.; Ohura, N.; Fukuda, T.; Sato, T.; Sekine, K.; Kato, S.; et al. Impaired flow-dependent control of vascular tone and remodeling in P2X4-deficient mice. Nat. Med. 2006, 12, 133–137. [Google Scholar] [CrossRef]
- Yoshida, K.; Ito, M.; Matsuoka, I. P2X7 receptor antagonist activity of the anti-allergic agent oxatomide. Eur. J. Pharmacol. 2015, 767, 41–51. [Google Scholar] [CrossRef]
- Ito, M.; Matsuoka, I. Inhibition of P2Y6 receptor-mediated phospholipase C activation and Ca2+ signalling by prostaglandin E2 in J774 murine macrophages. Eur. J. Pharmacol. 2015, 749, 124–132. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yoshida, K.; Tajima, M.; Nagano, T.; Obayashi, K.; Ito, M.; Yamamoto, K.; Matsuoka, I. Co-Stimulation of Purinergic P2X4 and Prostanoid EP3 Receptors Triggers Synergistic Degranulation in Murine Mast Cells. Int. J. Mol. Sci. 2019, 20, 5157. https://doi.org/10.3390/ijms20205157
Yoshida K, Tajima M, Nagano T, Obayashi K, Ito M, Yamamoto K, Matsuoka I. Co-Stimulation of Purinergic P2X4 and Prostanoid EP3 Receptors Triggers Synergistic Degranulation in Murine Mast Cells. International Journal of Molecular Sciences. 2019; 20(20):5157. https://doi.org/10.3390/ijms20205157
Chicago/Turabian StyleYoshida, Kazuki, Makoto Tajima, Tomoki Nagano, Kosuke Obayashi, Masaaki Ito, Kimiko Yamamoto, and Isao Matsuoka. 2019. "Co-Stimulation of Purinergic P2X4 and Prostanoid EP3 Receptors Triggers Synergistic Degranulation in Murine Mast Cells" International Journal of Molecular Sciences 20, no. 20: 5157. https://doi.org/10.3390/ijms20205157
APA StyleYoshida, K., Tajima, M., Nagano, T., Obayashi, K., Ito, M., Yamamoto, K., & Matsuoka, I. (2019). Co-Stimulation of Purinergic P2X4 and Prostanoid EP3 Receptors Triggers Synergistic Degranulation in Murine Mast Cells. International Journal of Molecular Sciences, 20(20), 5157. https://doi.org/10.3390/ijms20205157



