Laser-Driven Ultrashort Pulsed Electron Beam Radiation at Doses of 0.5 and 1.0 Gy Induces Apoptosis in Human Fibroblasts
Abstract
:1. Introduction
2. Results
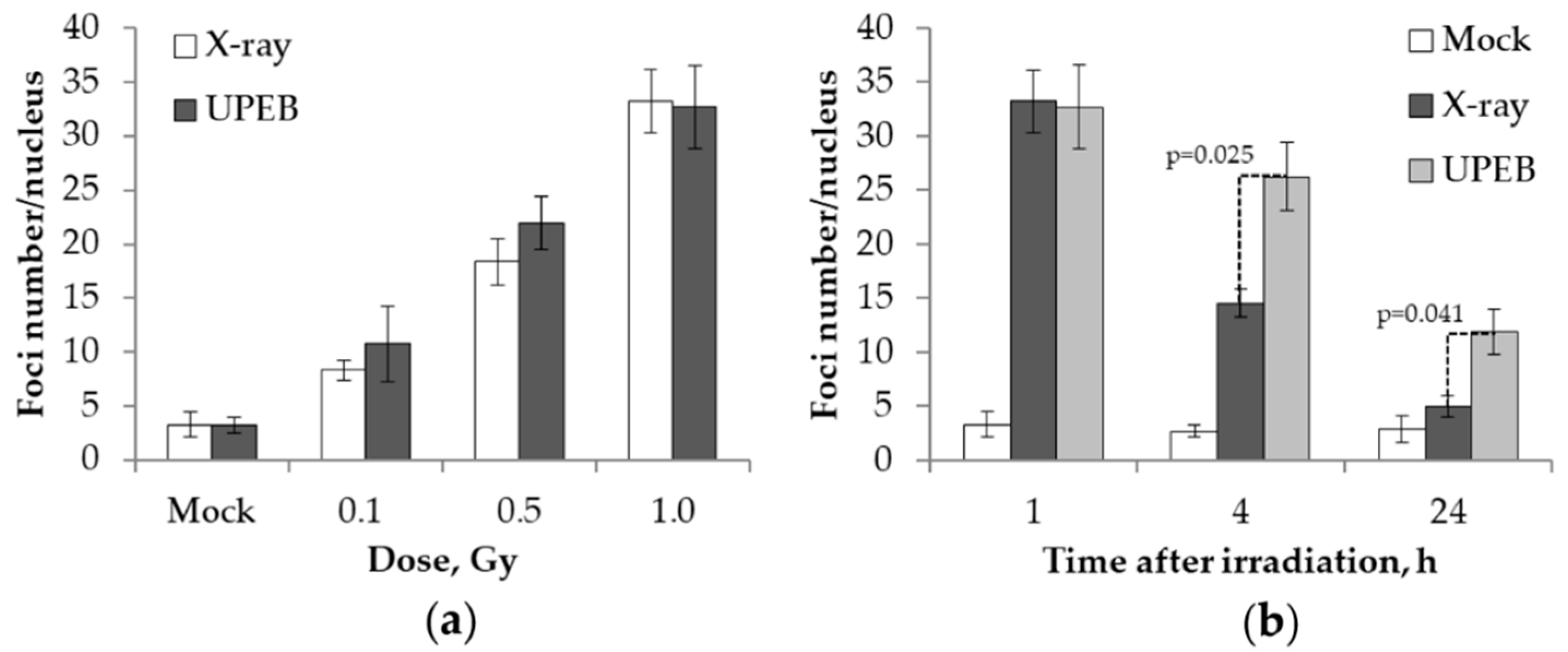
2.1. γH2AX Foci Analysis
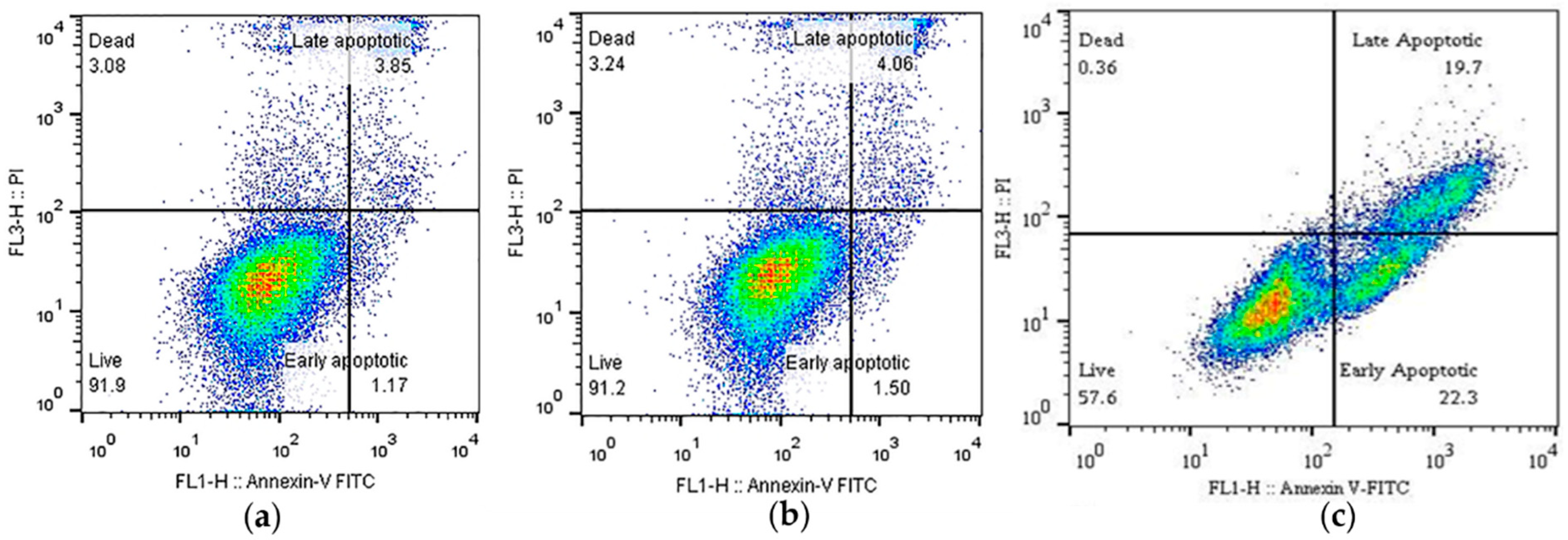
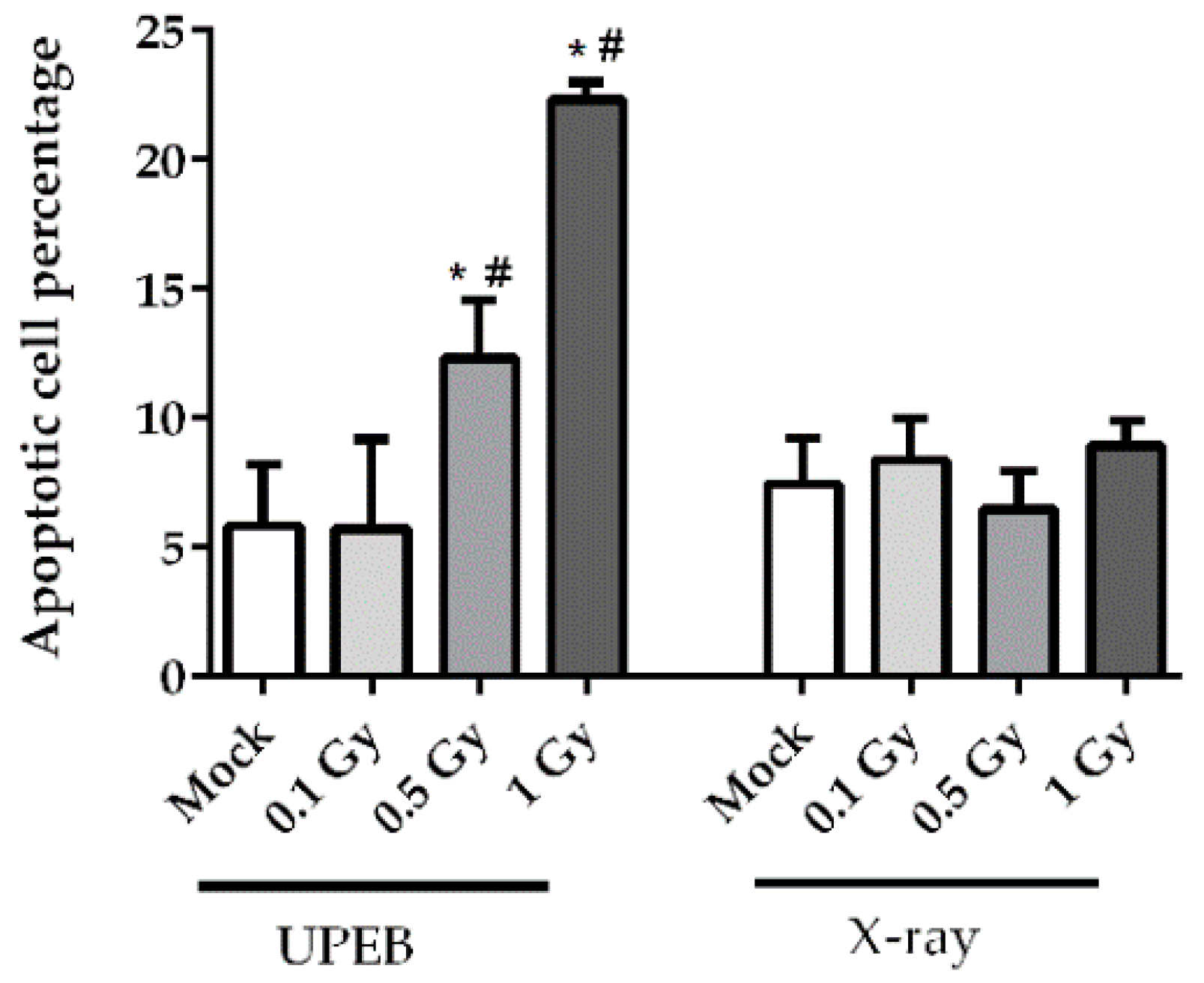
2.2. Cell Viability and Apoptosis Analysis
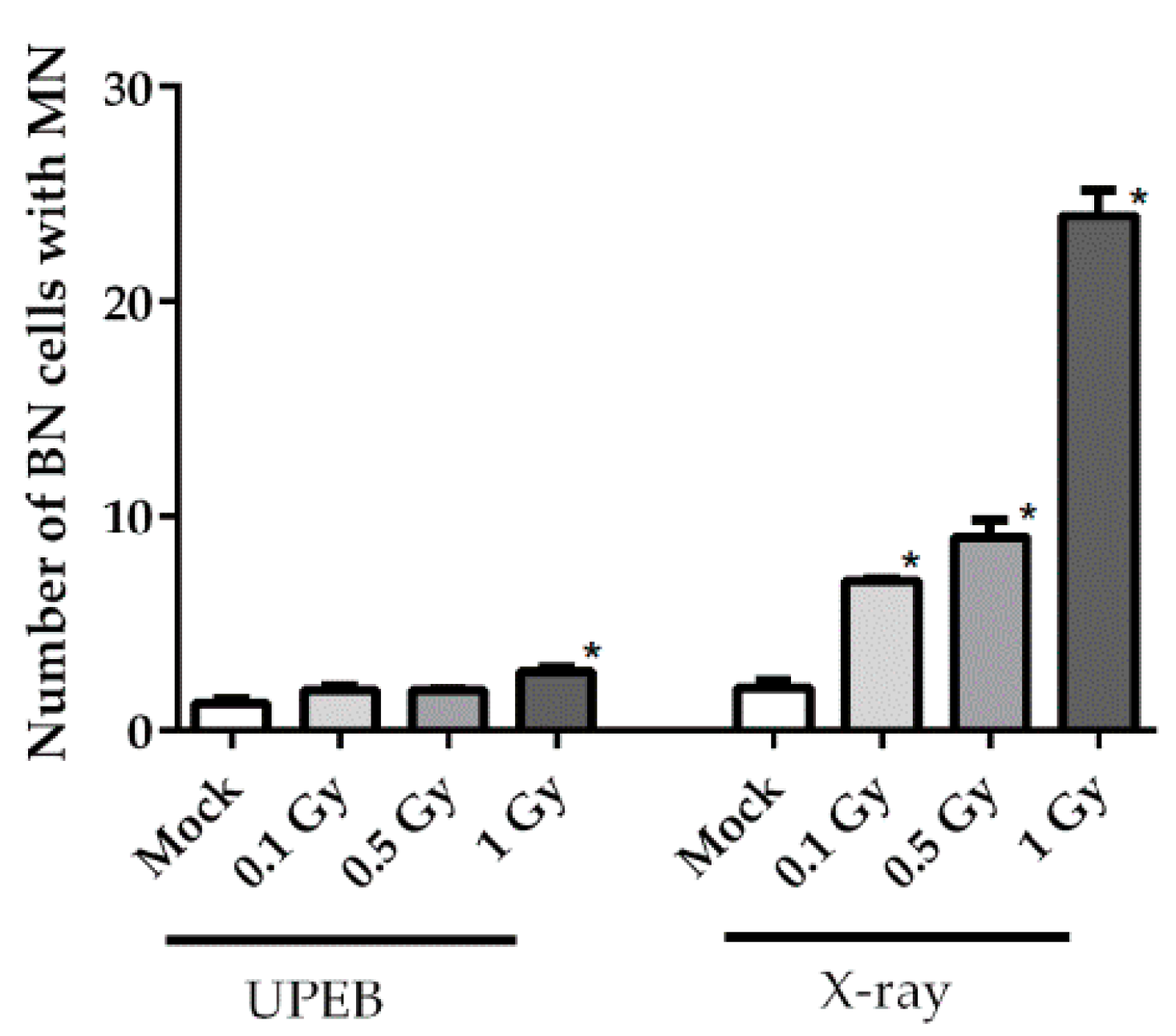
2.3. Micronuclei Formation
3. Discussion
4. Materials and Methods
4.1. Cell Culture and Irradiation
4.2. Apoptosis Analysis
4.3. CBMN Assay
4.4. Immunofluorescence Staining
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AREAL | Advanced Research Electron Accelerator Laboratory |
| BN | Binuclear |
| BSA | Bovine serum albumin |
| CBMN | Cytokinesis-block micronucleus |
| DMEM | Dulbecco’s Modified Eagle Medium |
| DNA | Deoxyribonucleic acid |
| DSBs | Double-strand breaks |
| GHz | Gigahertz |
| Gy | Gray |
| LET | Linear energy transfer |
| MeV | Megaelectronvolt |
| MN | Micronuclei |
| PBS | Phosphate-buffered saline |
| RF | Radio frequency |
| UPEB | Ultrashort pulsed electron beam |
References
- Linz, U.; Alonso, J. What will it take for laser driven proton accelerators to be applied to tumor therapy? Phys. Rev. ST Accel. Beams 2007, 10, 094801–094809. [Google Scholar] [CrossRef]
- Lundh, O.; Lim, J.; Rechatin, C.; Ammoura, L.; Ben-Ismaïl, A.; Davoine, X.; Gallot, G.; Goddet, J.-P.; Lefebvre, E.; Malka, V.; et al. Few femtosecond, few kiloampere electron bunch produced by a laser–plasma accelerator. Nat. Phys. 2011, 7, 219–222. [Google Scholar] [CrossRef]
- Malka, V.; Faure, J.; Glinec, Y.; Lifschitz, A.F. Laser-plasma accelerator: Status and perspectives. Philos. Trans. A. Math. Phys. Eng. Sci. 2006, 364, 601–610. [Google Scholar] [CrossRef] [PubMed]
- Faure, J.; Glinec, Y.; Pukhov, A.; Kiselev, S.; Gordienko, S.; Lefebvre, E.; Rousseau, J.-P.; Burgy, F.; Malka, V. A laser-plasma accelerator producing monoenergetic electron beams. Nature 2004, 431, 541–544. [Google Scholar] [CrossRef] [PubMed]
- Malka, V.; Faure, J.; Glinec, Y.; Lifschitz, A.F. Laser-plasma accelerators: A new tool for science and for society. Plasma Phys. Control. Fusion 2005, 47, B481–B490. [Google Scholar] [CrossRef]
- Gizzi, L.A.; Benedetti, C.; Cecchetti, C.A.; Di Pirro, G.; Gamucci, A.; Gatti, G.; Giulietti, A.; Giulietti, D.; Koester, P.; Labate, L.; et al. Laser-plasma acceleration with FLAME and ILIL ultraintense lasers. Appl. Sci. 2013, 3, 559–580. [Google Scholar] [CrossRef]
- Labate, L.; Andreassi, M.G.; Baffigi, F.; Basta, G.; Bizzarri, R.; Borghini, A.; Candiano, G.C.; Casarino, C.; Cresci, M.; Di Martino, F.; et al. Small-scale laser based electron accelerators for biology and medicine: A comparative study of the biological effectiveness. Proc. SPIE 2013, 8779. [Google Scholar] [CrossRef]
- Hooker, S.M. Developments in laser-driven plasma accelerators. Nat. Photonics 2013, 7, 775–782. [Google Scholar] [CrossRef] [Green Version]
- Andreassi, M.G.; Borghini, A.; Pulignani, S.; Baffigi, F.; Fulgentini, L.; Koester, P.; Cresci, M.; Vecoli, C.; Lamia, D.; Russo, G.; et al. Radiobiological effectiveness of ultrashort laser-driven electron bunches: Micronucleus frequency, telomere shortening and cell viability. Rad. Res. 2016, 186, 245–253. [Google Scholar] [CrossRef]
- Gauduel, Y.A.; Faure, J.; Malka, V. Ultrashort relativistic electron bunches and spatio-temporal radiation biology. Proc. SPIE 2008, 7080, 708002. [Google Scholar] [CrossRef]
- Rigaud, O.; Fortunel, N.O.; Vaigot, P.; Cadio, E.; Martin, M.T.; Lundh, O.; Faure, J.; Rechatin, C.; Malka, V.; Gauduel, Y.A. Exploring ultrashort high-energy electron-induced damage in human carcinoma cells. Cell Death Dis. 2010, 1, e73. [Google Scholar] [CrossRef] [PubMed]
- Acharya, S.; Bhat, N.N.; Joseph, P.; Sanjeev, G.; Sreedevi, B.; Narayana, Y. Dose rate effect on micronuclei induction in human blood lymphocytes exposed to single pulse and multiple pulses of electrons. Radiat. Environ. Biophys. 2011, 50, 253–263. [Google Scholar] [CrossRef] [PubMed]
- Tsakanov, V.; Aroutiounian, R.; Amatuni, G.; Aloyan, L.; Aslanyan, L.; Avagyan, V.; Babayan, N.; Buniatyan, V.; Dalyan, Y.; Davtyan, H.; et al. AREAL low energy electron beam applications in life and materials sciences. Nucl. Instrum. Methods Phys. Res. 2016, 829, 248–253. [Google Scholar] [CrossRef] [Green Version]
- Floettmann, K. Generation of sub-fs electron beams at few-MeV energies. Nucl. Instrum. Methods Phys. Res. A 2014, 740, 34–38. [Google Scholar] [CrossRef] [Green Version]
- Marthandan, S.; Menzel, U.; Priebe, S.; Groth, M.; Guthke, G.; Platzer, M.; Hemmerich, P.; Kaether, C.; Diekmann, S. Conserved genes and pathways in primary human fibroblast strains undergoing replicative and radiation induced senescence. Biol. Res. 2016, 49, 34. [Google Scholar] [CrossRef] [Green Version]
- Fournier, C.; Scholz, M.; Weyrather, W.K.; Rodemann, H.P.; Kraft, G. Changes of fibrosis-related parameters after high- and low-LET irradiation of fibroblasts. Int. J. Radiat. Biol. 2001, 77, 713–722. [Google Scholar] [CrossRef]
- Nasonova, E.; Füssel, K.; Berger, S.; Gudowska-Nowak, E.; Ritter, S. Cell cycle arrest and aberration yield in normal human fibroblasts. I. Effects of X-rays and 195 MeV u(-1) C ions. Int. J. Radiat. Biol. 2004, 80, 621–634. [Google Scholar] [CrossRef] [PubMed]
- Akudugu, J.M.; Bell, R.S.; Catton, C.; Davis, A.M.; Griffin, A.M.; O’Sullivan, B.; Waldron, J.N.; Ferguson, P.C.; Wunder, J.S.; Hill, R.P. Wound healing morbidity in STS patients treated with preoperative radiotherapy in relation to in vitro skin fibroblast radiosensitivity, proliferative capacity and TGF-beta activity. Radiother. Oncol. 2006, 78, 17–26. [Google Scholar] [CrossRef]
- Yim, W.; Kwan, S.W.; Yetisge, M. Classifying tumor event attributes in radiology reports. J. Assoc. Inf. Sci. Technol. 2017, 68, 2662–2674. [Google Scholar] [CrossRef]
- Rothkamm, K.; Löbrich, M. Evidence for a lack of DNA double-strand break repair in human cells exposed to very low x-ray doses. PNAS 2003, 100, 5057–5062. [Google Scholar] [CrossRef] [Green Version]
- Kühne, M.; Riballo, E.; Rief, N.; Rothkamm, K.; Jeggo, P.A.; Löbrich, M. A double-strand break repair defect in ATM-deficient cells contributes to radiosensitivity. Cancer Res. 2004, 64, 500–508. [Google Scholar] [CrossRef] [PubMed]
- Bluwstein, A.; Kumar, N.; Léger, K.; Traenkle, J.; van Oostrum, J.; Rehrauer, H.; Baudis, M.; Hottiger, M.O. PKC signaling prevents irradiation-induced apoptosis of primary human fibroblasts. Cell Death Dis. 2013, 4, e498. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, M.; Suzuki, K.; Kodama, S.; Watanabe, M. Phosphorylated histone H2AX foci persist on rejoined mitotic chromosomes in normal human diploid cells exposed to ionizing radiation phosphorylated histone H2AX foci persist on rejoined mitotic chromosomes in normal human diploid cells exposed to ionizing radiation. Radiat. Res. 2006, 165, 269–276. [Google Scholar] [PubMed]
- Schmid, T.E.; Zlobinskaya, O.; Multhoff, G. Differences in phosphorylated histone H2AX foci formation and removal of cells exposed to low and high linear energy transfer radiation. Curr. Genomics 2012, 13, 418–425. [Google Scholar] [CrossRef] [PubMed]
- Asaithamby, A.; Chen, D.J. Mechanism of cluster DNA damage repair in response to high-atomic number and energy particles radiation. Mutat. Res. 2011, 711, 87–99. [Google Scholar] [CrossRef] [Green Version]
- Laschinsky, L.; Baumann, M.; Beyreuther, E.; Enghardt, W.; Kaluza, M.; Karsch, L.; Lessmann, E.; Naumburger, D.; Nicolai, M.; Richter, C.; et al. Radiobiological effectiveness of laser accelerated electrons in comparison to electron beams from a conventional linear accelerator. J. Radiat. Res. 2012, 53, 395–403. [Google Scholar] [CrossRef]
- Beyreuther, E.; Karsch, L.; Laschinsky, L.; Leßmann, E.; Naumburger, D.; Oppelt, M.; Richter, C.; Schürer, M.; Woithe, J.; Pawelke, J. Radiobiological response to ultra-short pulsed megavoltage electron beams of ultra-high pulse dose rate. Int. J. Radiat. Biol. 2015, 91, 643–652. [Google Scholar] [CrossRef]
- Oppelt, M.; Baumann, M.; Bergmann, R.; Beyreuther, E.; Brüchner, K.; Hartmann, J.; Karsch, L.; Krause, M.; Laschinsky, L.; Leßmann, E.; et al. Comparison study of in vivo dose response to laser-driven versus conventional electron beam. Radiat. Environ. Biophys. 2015, 54, 155–166. [Google Scholar] [CrossRef]
- Scampoli, P.; Carpentieri, C.; Giannelli, M.; Magaddino, V.; Manti, L.; Moriello, C.; Piliero, M.A.; Righi, S.; Di Martino, F. Radiobiological characterization of the very high dose rate and dose per pulse electron beams produced by an IORT (intra operative radiation therapy) dedicated linac. Transl. Cancer Res. 2017, 6, S761–S768. [Google Scholar] [CrossRef]
- Eccles, L.J.; O’Neill, P.; Lomax, M.E. Delayed repair of radiation induced clustered DNA damage: Friend or foe? Mutat. Res. 2011, 711, 134–141. [Google Scholar] [CrossRef]
- Nikitaki, Z.; Nikolov, V.; Mavragani, I.V.; Mladenov, E.; Mangelis, A.; Laskaratou, D.A.; Fragkoulis, G.I.; Hellweg, C.E.; Martin, O.A.; Emfietzoglou, D.; et al. Measurement of complex DNA damage induction and repair in human cellular systems after exposure to ionizing radiations of varying linear energy transfer (LET). Free Rad. Res. 2016, 50, S64–S78. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Durante, M.; Formenti, S.C. Radiation-induced chromosomal aberrations and immunotherapy: Micronuclei, cytosolic DNA, and interferon-production pathway. Front. Oncol. 2018, 8, 192. [Google Scholar] [CrossRef] [PubMed]
- Marder, B.A.; Morgan, W.F. Delayed chromosomal instability induced by DNA damage. Mol. Cell Biol. 1993, 13, 6667–6677. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Snyder, A.R.; Morgan, W.F. Radiation-induced genomic instability and its implications for radiation carcinogenesis. Oncogene 2003, 22, 5848–5854. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Depuydt, J.; Baeyens, A.; Barnard, S.; Beinke, C.; Benedek, A.; Beukes, P.; Buraczewska, I.; Darroudi, F.; De Sanctis, S.; Dominguez, I.; et al. RENEB intercomparison exercises analyzing micronuclei (Cytokinesis-block Micronucleus Assay). Int. J. Radiat. Biol. 2017, 93, 36–47. [Google Scholar] [CrossRef]
- Muller, W.U.; Rode, A. The micronucleus assay in human lymphocytes after high radiation doses (5–15 Gy). Mutat. Res. 2002, 502, 47–51. [Google Scholar] [CrossRef]
- Ojima, M.; Ito, M.; Suzuki, K.; Kai, M. Unstable chromosome aberrations do not accumulate in normal human fibroblast after fractionated X-irradiation. Plos ONE 2015, 10, e0116645. [Google Scholar] [CrossRef]
- Desouky, O.S.; Wenling, Y.; Song, L.; Pei, H.; Zhou, G. Protection against radiation-induced genotoxic damages in cultured human fibroblast cells by treatment with Pallidium-lipoic acid complex. In Proceedings of the Third International Conference on Radiation Sciences and Applications, Hurghada, Egypt, 12–16 November 2012. [Google Scholar]
- Nakamura, A.; Sedelnikova, O.A.; Redon, C.; Pilch, D.R.; Sinogeeva, N.I.; Shroff, R.; Lichten, M.; Bonner, W.M. Techniques for gamma-H2AX detection. Methods Enzymol. 2006, 409, 236–250. [Google Scholar] [CrossRef]
- Fenech, M. Cytokinesis-block micronucleus cytome assay. Nat. Protoc. 2007, 2, 1084–1104. [Google Scholar] [CrossRef] [Green Version]
| AREAL Beam Parameters | UV Laser Parameters | ||
|---|---|---|---|
| Beam charge (pC) | 30 | Wavelength (nm) | 258 |
| Electron energy (MeV) | 3.6 | Pulse energy (μJ) | 200 |
| Pulse duration (fs) | 450 | Repetition rate (Hz) | 2 |
| Pulse repetition rate (Hz) | 2 | Energy stability | <2% |
| Beam spot (mm) | 15 | Beam divergence (mrad) | <0.3 |
| Norm. emittance (mm-mrad) | <0.5 | Beam diameter (mm) | 2.0 |
| RMS energy spread | <1.5% | - | - |
| Online dose information | Faraday cup | - | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Babayan, N.; Grigoryan, B.; Khondkaryan, L.; Tadevosyan, G.; Sarkisyan, N.; Grigoryan, R.; Apresyan, L.; Aroutiounian, R.; Vorobyeva, N.; Pustovalova, M.; et al. Laser-Driven Ultrashort Pulsed Electron Beam Radiation at Doses of 0.5 and 1.0 Gy Induces Apoptosis in Human Fibroblasts. Int. J. Mol. Sci. 2019, 20, 5140. https://doi.org/10.3390/ijms20205140
Babayan N, Grigoryan B, Khondkaryan L, Tadevosyan G, Sarkisyan N, Grigoryan R, Apresyan L, Aroutiounian R, Vorobyeva N, Pustovalova M, et al. Laser-Driven Ultrashort Pulsed Electron Beam Radiation at Doses of 0.5 and 1.0 Gy Induces Apoptosis in Human Fibroblasts. International Journal of Molecular Sciences. 2019; 20(20):5140. https://doi.org/10.3390/ijms20205140
Chicago/Turabian StyleBabayan, Nelly, Bagrat Grigoryan, Lusine Khondkaryan, Gohar Tadevosyan, Natalya Sarkisyan, Ruzanna Grigoryan, Lilit Apresyan, Rouben Aroutiounian, Natalia Vorobyeva, Margarita Pustovalova, and et al. 2019. "Laser-Driven Ultrashort Pulsed Electron Beam Radiation at Doses of 0.5 and 1.0 Gy Induces Apoptosis in Human Fibroblasts" International Journal of Molecular Sciences 20, no. 20: 5140. https://doi.org/10.3390/ijms20205140






