Two XMAP215/TOG Microtubule Polymerases, Alp14 and Dis1, Play Non-Exchangeable, Distinct Roles in Microtubule Organisation in Fission Yeast
Abstract
1. Introduction
2. Results
2.1. Creation of Temperature-Sensitive alp14 Mutants in the Absence of Dis1
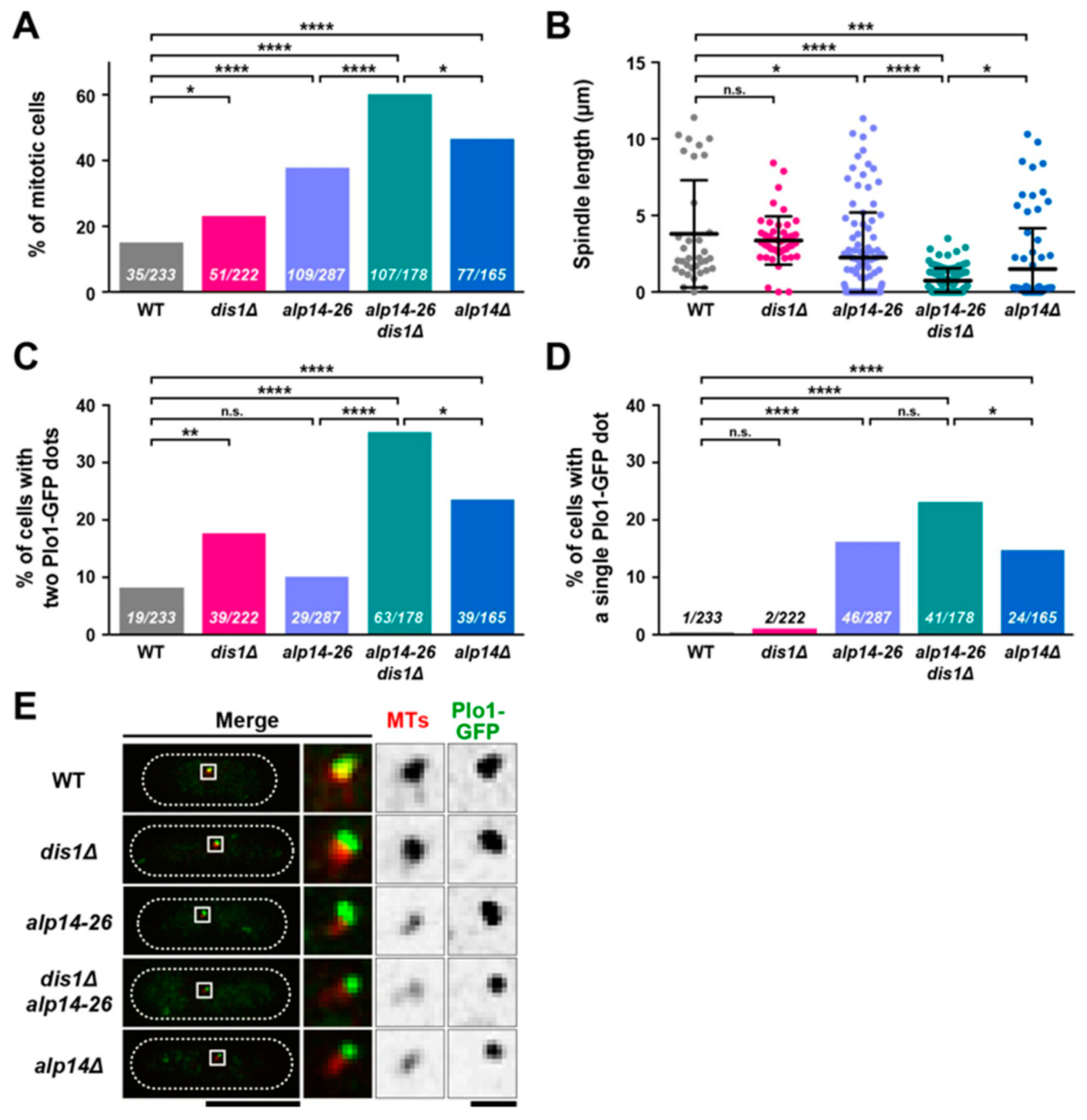
2.2. Alp14 and Dis1 Collaborate to Regulate Proper Mitotic Progression and Promote Spindle Elongation
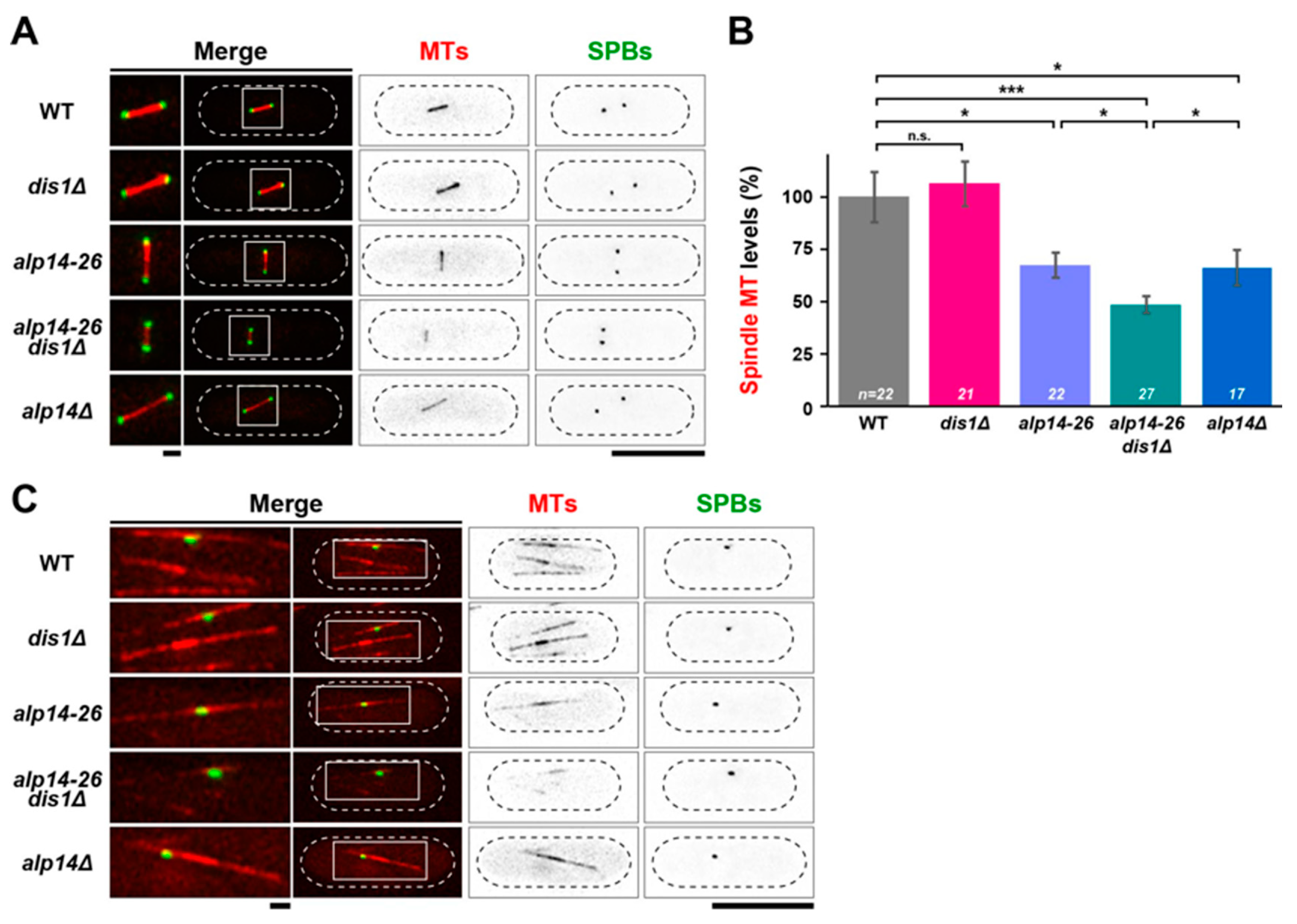
2.3. Alp14, But Not Dis1, Promotes Microtubule Nucleation
2.4. Microtubule Intensities Are Reduced by Simultaneous Inactivation of Alp14 and Dis1
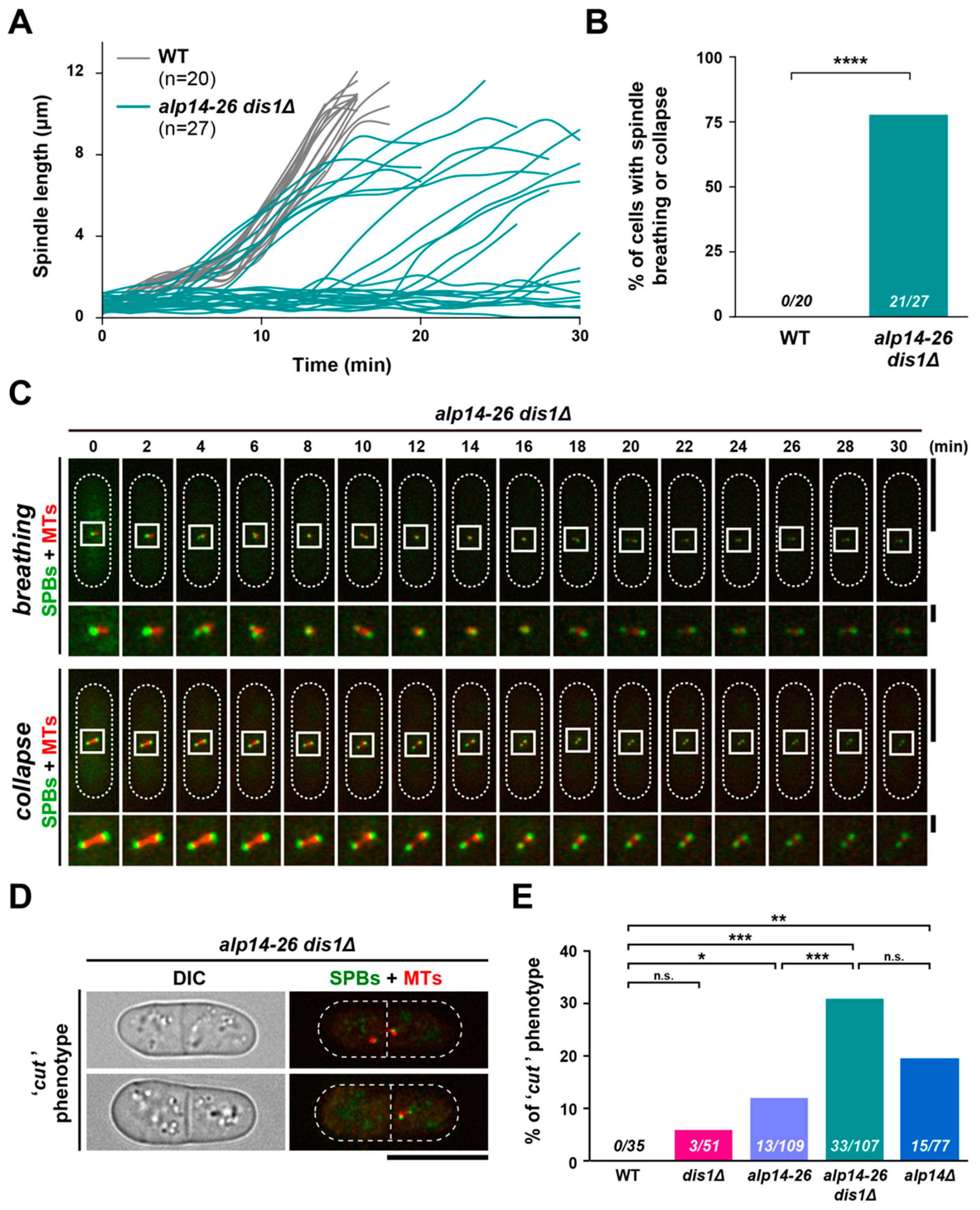
2.5. Short Spindles Often Collapse in alp14-26dis1∆ Cells, Leading to a Lethal “Cut” Phenotype
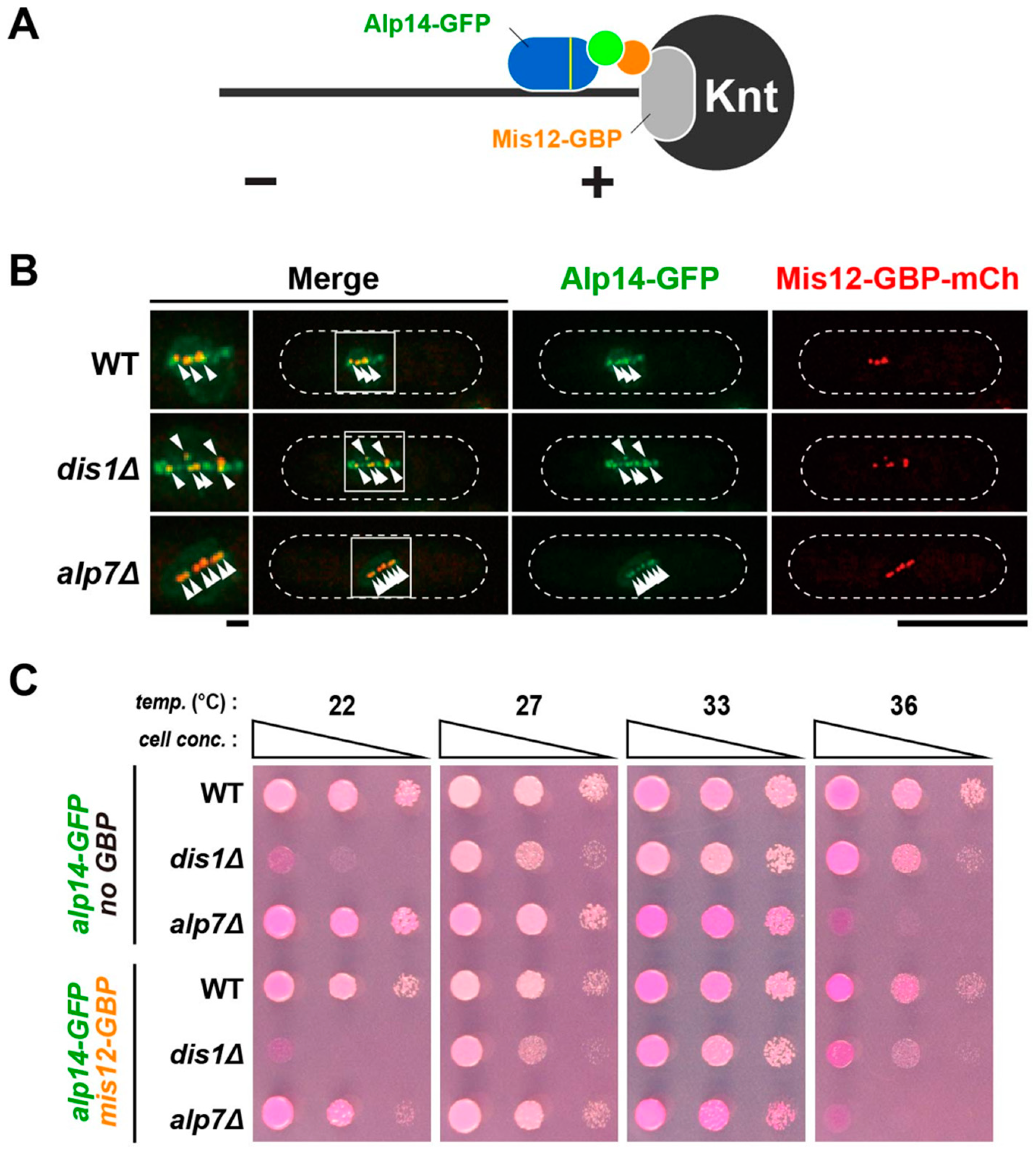
2.6. Alp14 and Dis1 Are Not Functionally Exchangeable
3. Discussion
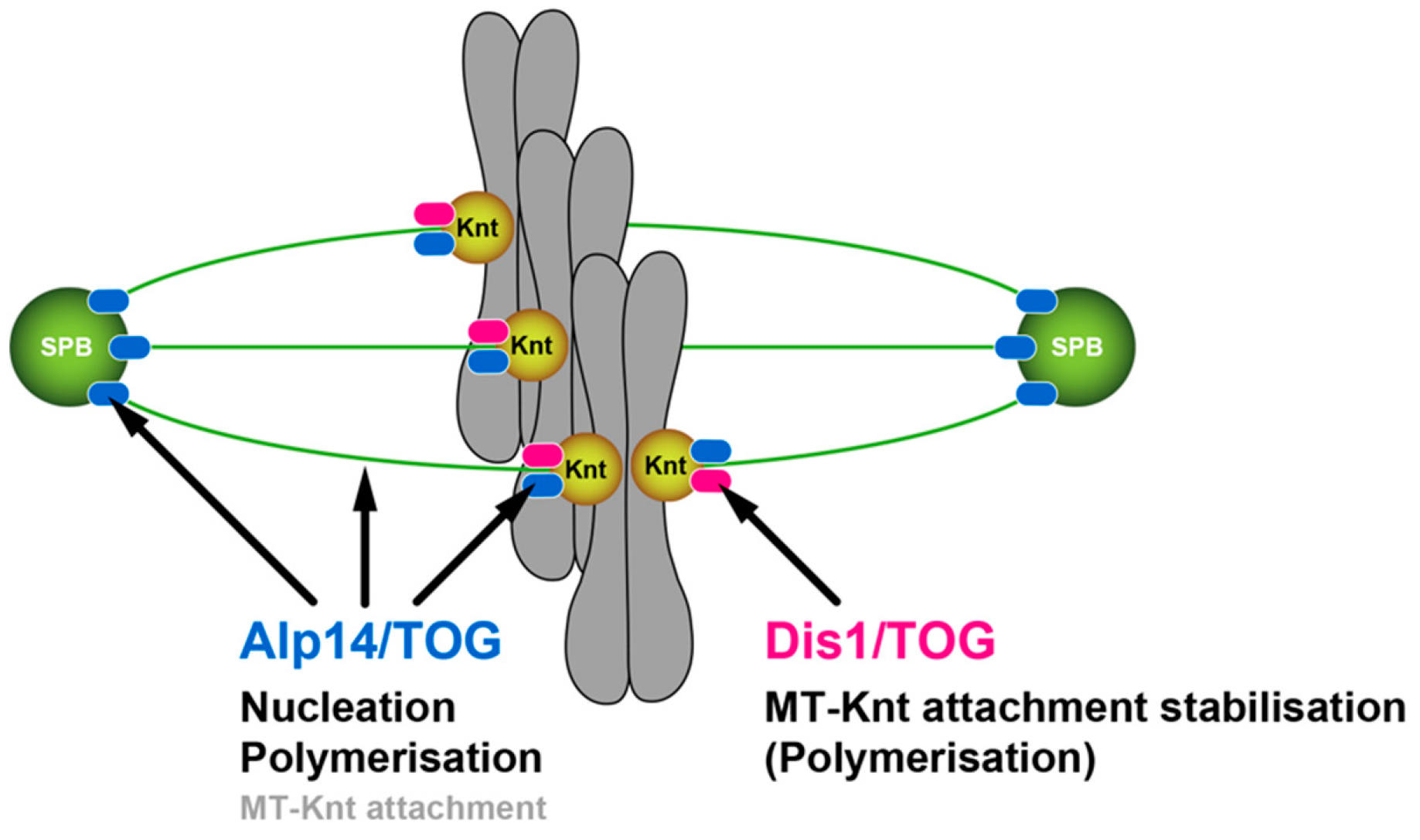
3.1. Alp14 and Dis1 Have Evolved to Execute Functionally Non-Exchangeable Roles
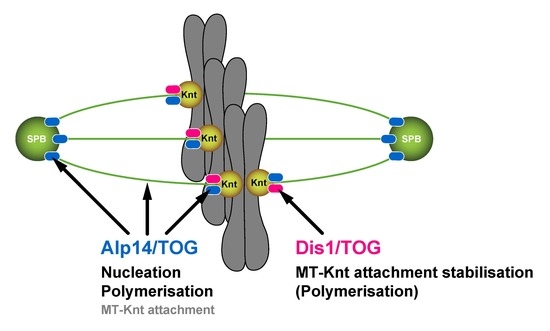
3.2. Dis1 May Be a Specialised Regulator of Microtubule Dynamics at the Kinetochore-Microtubule Interface
4. Materials and Methods
4.1. Strains, Media, and Genetic Methods
4.2. Preparation and Manipulation of Nucleic Acids
4.3. Strain Construction, Gene Disruption and N-Terminal and C-Terminal Epitope Tagging
4.4. Isolation of alp14 Temperature-Sensitive Mutants in the dis1∆ Background
4.5. Fluorescence Microscopy and Time-Lapse Live Cell Imaging
4.6. Quantification of Fluorescent Signal Intensities
4.7. Statistical Data Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| GBP | GFP-binding protein |
| MAPs | Microtubule-Associated Proteins |
| MT | Microtubule |
| NLS | Nuclear Localisation Signal |
| SPB | Spindle Pole Body |
| ts | temperature sensitive |
References
- Akhmanova, A.; Steinmetz, M.O. Control of microtubule organization and dynamics: Two ends in the limelight. Nat. Rev. Mol. Cell Biol. 2015, 16, 711–726. [Google Scholar] [CrossRef] [PubMed]
- Godinho, S.A.; Pellman, D. Causes and consequences of centrosome abnormalities in cancer. Philos. Trans. R. Soc. Lond B Biol. Sci. 2014, 369, 20130467. [Google Scholar] [CrossRef] [PubMed]
- Sheltzer, J.M.; Amon, A. The aneuploidy paradox: Costs and benefits of an incorrect karyotype. Trends Genet. 2011, 27, 446–453. [Google Scholar] [CrossRef] [PubMed]
- Mitchison, T.; Kirschner, M. Dynamic instability of microtubule growth. Nature 1984, 312, 237–242. [Google Scholar] [CrossRef] [PubMed]
- Desai, A.; Mitchison, T.J. Microtubule polymerization dynamics. Annu. Rev. Cell Dev Biol. 1997, 13, 83–117. [Google Scholar] [CrossRef]
- Ohkura, H.; Garcia, M.A.; Toda, T. Dis1/TOG universal microtubule adaptors - one MAP for all? J. Cell Sci. 2001, 114, 3805–3812. [Google Scholar]
- Kinoshita, K.; Habermann, B.; Hyman, A.A. XMAP215: A key component of the dynamic microtubule cytoskeleton. Trends Cell Biol. 2002, 12, 267–273. [Google Scholar] [CrossRef]
- Al-Bassam, J.; Chang, F. Regulation of microtubule dynamics by TOG-domain proteins XMAP215/Dis1 and CLASP. Trends Cell Biol. 2011, 21, 604–614. [Google Scholar] [CrossRef]
- Gard, D.L.; Becker, B.E.; Josh Romney, S. MAPping the eukaryotic tree of life: Structure, function, and evolution of the MAP215/Dis1 family of microtubule-associated proteins. Int. Rev. Cytol. 2004, 239, 179–272. [Google Scholar]
- Gard, D.L.; Kirschner, M.W. A microtubule-associated protein from Xenopus eggs that specifically promotes assembly at the plus-end. J. Cell Biol. 1987, 105, 2203–2215. [Google Scholar] [CrossRef]
- Brouhard, G.J.; Stear, J.H.; Noetzel, T.L.; Al-Bassam, J.; Kinoshita, K.; Harrison, S.C.; Howard, J.; Hyman, A.A. XMAP215 is a processive microtubule polymerase. Cell 2008, 132, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Al-Bassam, J.; van Breugel, M.; Harrison, S.C.; Hyman, A. Stu2p binds tubulin and undergoes an open-to-closed conformational change. J. Cell Biol. 2006, 172, 1009–1022. [Google Scholar] [CrossRef] [PubMed]
- Al-Bassam, J.; Larsen, N.A.; Hyman, A.A.; Harrison, S.C. Crystal structure of a TOG domain: Conserved features of XMAP215/Dis1-family TOG domains and implications for tubulin binding. Structure 2007, 15, 355–362. [Google Scholar] [CrossRef]
- Garcia, M.A.; Vardy, L.; Koonrugsa, N.; Toda, T. Fission yeast ch-TOG/XMAP215 homologue Alp14 connects mitotic spindles with the kinetochore and is a component of the Mad2-dependent spindle checkpoint. EMBO J. 2001, 20, 3389–3401. [Google Scholar] [CrossRef]
- Nakaseko, Y.; Goshima, G.; Morishita, J.; Yanagida, M. M phase-specific kinetochore proteins in fission yeast microtubule-associating Dis1 and Mtc1 display rapid separation and segregation during anaphase. Curr. Biol. 2001, 11, 537–549. [Google Scholar] [CrossRef]
- Matsuo, Y.; Maurer, S.P.; Yukawa, M.; Zakian, S.; Singleton, M.R.; Surrey, T.; Toda, T. An unconventional interaction between Dis1/TOG and Mal3/EB1 in fission yeast promotes the fidelity of chromosome segregation. J. Cell Sci. 2016, 129, 4592–4606. [Google Scholar] [CrossRef]
- Al-Bassam, J.; Kim, H.; Flor-Parra, I.; Lal, N.; Velji, H.; Chang, F. Fission yeast Alp14 is a dose dependent plus end tracking microtubule polymerase. Mol. Biol. Cell 2012, 23, 2878–2890. [Google Scholar] [CrossRef]
- Garcia, M.A.; Koonrugsa, N.; Toda, T. Spindle-kinetochore attachment requires the combined action of Kin I-like Klp5/6 and Alp14/Dis1-MAPs in fission yeast. EMBO J. 2002, 21, 6015–6024. [Google Scholar] [CrossRef]
- Sato, M.; Vardy, L.; Angel Garcia, M.; Koonrugsa, N.; Toda, T. Interdependency of fission yeast Alp14/TOG and coiled coil protein Alp7 in microtubule localization and bipolar spindle formation. Mol. Biol. Cell 2004, 15, 1609–1622. [Google Scholar] [CrossRef]
- Hsu, K.S.; Toda, T. Ndc80 internal loop interacts with Dis1/TOG to ensure proper kinetochore-spindle attachment in fission yeast. Curr. Biol. 2011, 21, 214–220. [Google Scholar] [CrossRef]
- Amin, M.A.; Agarwal, S.; Varma, D. Mapping the kinetochore MAP functions required for stabilizing microtubule attachments to chromosomes during metaphase. Cytoskeleton (Hoboken) 2019, 76, 398–412. [Google Scholar] [CrossRef] [PubMed]
- Tang, N.H.; Toda, T. Ndc80 Loop as a protein-protein interaction motif. Cell Div. 2013, 8, 2. [Google Scholar] [CrossRef] [PubMed]
- Tang, N.H.; Takada, H.; Hsu, K.S.; Toda, T. The internal loop of fission yeast Ndc80 binds Alp7/TACC-Alp14/TOG and ensures proper chromosome attachment. Mol. Biol. Cell 2013, 24, 1122–1133. [Google Scholar] [CrossRef] [PubMed]
- Flor-Parra, I.; Iglesias-Romero, A.B.; Chang, F. The XMAP215 ortholog Alp14 promotes microtubule nucleation in fission yeast. Curr. Biol. 2018, 28, 1681–1691. [Google Scholar] [CrossRef]
- Thawani, A.; Kadzik, R.S.; Petry, S. XMAP215 is a microtubule nucleation factor that functions synergistically with the gamma-tubulin ring complex. Nat. Cell Biol. 2018, 20, 575–585. [Google Scholar] [CrossRef]
- Kakui, Y.; Sato, M.; Okada, N.; Toda, T.; Yamamoto, M. Microtubules and Alp7-Alp14 (TACC-TOG) reposition chromosomes before meiotic segregation. Nat. Cell Biol. 2013, 15, 786–796. [Google Scholar] [CrossRef]
- Pinder, C.L.; Matsuo, Y.; Maurer, S.P.; Toda, T. Kinesin-8 and Dis1/TOG collaborate to limit spindle elongation from prophase to anaphase A for proper chromosome segregation in fission yeast. J. Cell Sci. 2019, 132, 1–15. [Google Scholar] [CrossRef]
- Tang, N.H.; Fong, C.S.; Masuda, H.; Jourdain, I.; Yukawa, M.; Toda, T. Generation of temperature sensitive mutations with error-prone PCR in a gene encoding a component of the spindle pole body in fission yeast. Biosci. Biotechnol. Biochem. 2019, 83, 1717–1720. [Google Scholar] [CrossRef]
- Toda, T.; Adachi, Y.; Hiraoka, Y.; Yanagida, M. Identification of the pleiotropic cell division cycle gene NDA2 as one of two different α-tubulin genes in Schizosaccharomyces pombe. Cell 1984, 37, 233–242. [Google Scholar] [CrossRef]
- Nabeshima, K.; Nakagawa, T.; Straight, A.F.; Murray, A.; Chikashige, Y.; Yamashita, Y.M.; Hiraoka, Y.; Yanagida, M. Dynamics of centromeres during metaphase-anaphase transition in fission yeast: Dis1 is implicated in force balance in metaphase bipolar spindle. Mol. Biol. Cell 1998, 9, 3211–3225. [Google Scholar] [CrossRef]
- Mulvihill, D.P.; Petersen, J.; Ohkura, H.; Glover, D.M.; Hagan, I.M. Plo1 kinase recruitment to the spindle pole body and its role in cell division in Schizosaccharomyces pombe. Mol. Biol. Cell 1999, 10, 2771–2785. [Google Scholar] [CrossRef] [PubMed]
- Grallert, A.; Patel, A.; Tallada, V.A.; Chan, K.Y.; Bagley, S.; Krapp, A.; Simanis, V.; Hagan, I.M. Centrosomal MPF triggers the mitotic and morphogenetic switches of fission yeast. Nat. Cell Biol. 2013, 15, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Bähler, J.; Steever, A.B.; Wheatley, S.; Wang, Y.-l.; Pringle, J.R.; Gould, K.L.; McCollum, D. Role of polo kinase and Mid1p in determining the site of cell division in fission yeast. J. Cell Biol. 1998, 143, 1603–1616. [Google Scholar] [CrossRef]
- Roque, H.; Ward, J.J.; Murrells, L.; Brunner, D.; Antony, C. The fission yeast XMAP215 homolog Dis1p is involved in microtubule bundle organization. PLoS ONE 2010, 5, e14201. [Google Scholar] [CrossRef]
- Yanagida, M. Fission yeast cut mutations revisited: Control of anaphase. Trends Cell Biol. 1998, 8, 144–149. [Google Scholar] [CrossRef]
- Sato, M.; Toda, T. Space shuttling in the cell: Nucleocytoplasmic transport and microtubule organization during the cell cycle. Nucleus 2010, 1, 231–236. [Google Scholar] [CrossRef]
- Aoki, K.; Nakaseko, Y.; Kinoshita, K.; Goshima, G.; Yanagida, M. Cdc2 phosphorylation of the fission yeast Dis1 ensures accurate chromosome segregation. Curr. Biol. 2006, 16, 1627–1635. [Google Scholar] [CrossRef]
- Rothbauer, U.; Zolghadr, K.; Muyldermans, S.; Schepers, A.; Cardoso, M.C.; Leonhardt, H. A versatile nanotrap for biochemical and functional studies with fluorescent fusion proteins. Mol. Cell Proteomics 2008, 7, 282–289. [Google Scholar] [CrossRef]
- Vardy, L.; Toda, T. The fission yeast γ-tubulin complex is required in G1 phase and is a component of the spindle assembly checkpoint. EMBO J. 2000, 19, 6098–6111. [Google Scholar] [CrossRef]
- Goshima, G.; Saitoh, S.; Yanagida, M. Proper metaphase spindle length is determined by centromere proteins Mis12 and Mis6 required for faithful chromosome segregation. Genes Dev. 1999, 13, 1664–1677. [Google Scholar] [CrossRef]
- Kalderon, D.; Roberts, B.L.; Richardson, W.D.; Smith, A.E. A short amino acid sequence able to specify nuclear location. Cell 1984, 39, 499–509. [Google Scholar] [CrossRef]
- Yukawa, M.; Kawakami, T.; Okazaki, M.; Kume, K.; Tang, N.H.; Toda, T. A microtubule polymerase cooperates with the kinesin-6 motor and a microtubule cross-linker to promote bipolar spindle assembly in the absence of kinesin-5 and kinesin-14 in fission yeast. Mol. Biol. Cell 2017, 28, 3647–3659. [Google Scholar] [CrossRef] [PubMed]
- Sato, M.; Toda, T. Alp7/TACC is a crucial target in Ran-GTPase-dependent spindle formation in fission yeast. Nature 2007, 447, 334–337. [Google Scholar] [CrossRef] [PubMed]
- Okada, N.; Toda, T.; Yamamoto, M.; Sato, M. CDK-dependent phosphorylation of Alp7-Alp14 (TACC-TOG) promotes its nuclear accumulation and spindle microtubule assembly. Mol. Biol. Cell 2014, 25, 1969–1982. [Google Scholar] [CrossRef]
- Sato, M.; Okada, N.; Kakui, Y.; Yamamoto, M.; Yoshida, M.; Toda, T. Nucleocytoplasmic transport of Alp7/TACC organizes spatiotemporal microtubule formation in fission yeast. EMBO Rep. 2009, 10, 1161–1167. [Google Scholar] [CrossRef]
- Ling, Y.C.; Vjestica, A.; Oliferenko, S. Nucleocytoplasmic shuttling of the TACC protein Mia1p/Alp7p is required for remodeling of microtubule arrays during the cell cycle. PLoS ONE 2009, 4, e6255. [Google Scholar] [CrossRef]
- Tang, N.H.; Okada, N.; Fong, C.S.; Arai, K.; Sato, M.; Toda, T. Targeting Alp7/TACC to the spindle pole body is essential for mitotic spindle assembly in fission yeast. FEBS Lett. 2014, 588, 2814–2821. [Google Scholar] [CrossRef]
- Zheng, F.; Li, T.; Jin, D.Y.; Syrovatkina, V.; Scheffler, K.; Tran, P.T.; Fu, C. Csi1p recruits alp7p/TACC to the spindle pole bodies for bipolar spindle formation. Mol. Biol. Cell 2014, 25, 2750–2760. [Google Scholar] [CrossRef]
- Yukawa, M.; Yamada, Y.; Yamauchi, T.; Toda, T. Two spatially distinct kinesin-14 proteins, Pkl1 and Klp2, generate collaborative inward forces against kinesin-5 Cut7 in S. pombe. J. Cell Sci. 2018, 131, 1–11. [Google Scholar] [CrossRef]
- Peset, I.; Vernos, I. The TACC proteins: TACC-ling microtubule dynamics and centrosome function. Trends Cell Biol. 2008, 18, 379–388. [Google Scholar] [CrossRef]
- Li, W.; Miki, T.; Watanabe, T.; Kakeno, M.; Sugiyama, I.; Kaibuchi, K.; Goshima, G. EB1 promotes microtubule dynamics by recruiting Sentin in Drosophila cells. J. Cell Biol. 2011, 193, 973–983. [Google Scholar] [CrossRef] [PubMed]
- van der Vaart, B.; Manatschal, C.; Grigoriev, I.; Olieric, V.; Gouveia, S.M.; Bjelic, S.; Demmers, J.; Vorobjev, I.; Hoogenraad, C.C.; Steinmetz, M.O.; et al. SLAIN2 links microtubule plus end-tracking proteins and controls microtubule growth in interphase. J. Cell Biol. 2011, 193, 1083–1099. [Google Scholar] [CrossRef]
- Zanic, M.; Widlund, P.O.; Hyman, A.A.; Howard, J. Synergy between XMAP215 and EB1 increases microtubule growth rates to physiological levels. Nat. Cell Biol. 2013, 15, 688–693. [Google Scholar] [CrossRef] [PubMed]
- Gunzelmann, J.; Ruthnick, D.; Lin, T.C.; Zhang, W.; Neuner, A.; Jakle, U.; Schiebel, E. The microtubule polymerase Stu2 promotes oligomerization of the γ-TuSC for cytoplasmic microtubule nucleation. eLife 2018, 7, e39932. [Google Scholar] [CrossRef]
- Woodruff, J.B.; Ferreira Gomes, B.; Widlund, P.O.; Mahamid, J.; Honigmann, A.; Hyman, A.A. The centrosome is a selective condensate that nucleates microtubules by concentrating tubulin. Cell 2017, 169, 1066–1077 e10. [Google Scholar] [CrossRef]
- George, A.A.; Walworth, N.C. Escape from mitotic arrest: An unexpected connection between microtubule dynamics and epigenetic regulation of centromeric chromatin in Schizosaccharomyces pombe. Genetics 2015, 201, 1467–1478. [Google Scholar] [CrossRef]
- Moreno, S.; Klar, A.; Nurse, P. Molecular genetic analysis of fission yeast Schizosaccharomyces pombe. Methods Enzymol. 1991, 194, 795–823. [Google Scholar]
- Sato, M.; Dhut, S.; Toda, T. New drug-resistant cassettes for gene disruption and epitope tagging in Schizosaccharomyces pombe. Yeast 2005, 22, 583–591. [Google Scholar] [CrossRef]
- Bahler, J.; Wu, J.Q.; Longtine, M.S.; Shah, N.G.; McKenzie, A., 3rd; Steever, A.B.; Wach, A.; Philippsen, P.; Pringle, J.R. Heterologous modules for efficient and versatile PCR-based gene targeting in Schizosaccharomyces pombe. Yeast 1998, 14, 943–951. [Google Scholar] [CrossRef]


| Strains | Genotypes | Figures Used | Derivations |
|---|---|---|---|
| MS345 | h− alp14::ura4+ leu1 ura4 | 1A | Our lab stock |
| MA003 | h+ alp14-GFP-kanR leu1ura4 his2 | 1A | Our lab stock |
| MY2097 | h− alp14-21-GFP-kanR leu1ura4 | 1A | This study |
| TK469 | h− alp14-26-GFP-kanR leu1ura4 | 1A | This study |
| MY2099 | h− alp14-27-GFP-kanR leu1ura4 | 1A | This study |
| TK475 | h− alp14-31-GFP-kanR leu1ura4 | 1A | This study |
| TK484 | h− alp14-32-GFP-kanR leu1ura4 | 1A | This study |
| MY2101 | h− alp14-33-GFP-kanR leu1ura4 | 1A | This study |
| TK358 | h− dis1::hphR alp14-GFP-kanR leu1 ura4 | 1A | This study |
| TK400 | h− alp14-21-GFP-kanR dis1::hphR leu1 ura4 | 1A–B | This study |
| TK405 | h− alp14-26-GFP-kanR dis1::hphR leu1 ura4 | 1A–B | This study |
| TK406 | h− alp14-27-GFP-kanR dis1::hphR leu1 ura4 | 1A–B | This study |
| TK457 | h− alp14-31-GFP-kanR dis1::hphR leu1 ura4 | 1A–B | This study |
| TK458 | h− alp14-32-GFP-kanR dis1::hphR leu1 ura4 | 1A–B | This study |
| TK459 | h− alp14-33-GFP-kanR dis1::hphR leu1 ura4 | 1A–B | This study |
| TK551 | h+ plo1-GFP-HA-kanR aur1R-Pnda3-mCherry-atb2 leu1 ura4 his2 | 2A–E, 4E | This study |
| TK583 | h− dis1::ura4+ plo1-GFP-HA-kanR aur1R-Pnda3-mCherry-atb2 leu1 ura4 | 2A–E, 4E | This study |
| TK556 | h− alp14-26-myc-hphR plo1-GFP-HA-kanR aur1R-Pnda3-mCherry-atb2 leu1 ura4 | 2A–E, 4E | This study |
| TK585 | h− alp14-26-myc-hphR dis1::ura4+ plo1-GFP-HA-kanR aur1R-Pnda3-mCherry-atb2 leu1 ura4 | 2A–E, 4D-E | This study |
| TK586 | h− alp14::ura4+ plo1-GFP-HA-kanR aur1R-Pnda3-mCherry-atb2 leu1 ura4 | 2A–E, 4E | This study |
| MO100 | h− cut12-GFP-ura4+ aur1R-Pnda3-mCherry-atb2 leu1 ura4 | 3A–C, 4A-B | Our lab stock |
| TK547 | h+ dis1::natR cut12-GFP-ura4+ aur1R-Pnda3-mCherry-atb2 leu1 ura4 his2 | 3A–C | This study |
| TK580 | h+ alp14-26-myc-hphR cut12-GFP-ura4+ aur1R-Pnda3-mCherry-atb2 leu1 ura4 | 3A–C | This study |
| TK572 | h− alp14-26-myc-hphR dis1::natR cut12-GFP-ura4+ aur1R-Pnda3-mCherry-atb2 leu1 ura4 | 3A–C, 4A–C | This study |
| TK582 | h+ alp14::hphR cut12-GFP-ura4+ aur1R-Pnda3-mCherry-atb2 leu1 ura4 his2 | 3A–C | This study |
| TK576 | h− dis1-GFP-kanR hphR-GBP-mCherry-alp4 leu1 ura4 | 5B–C | This study |
| TK591 | h− alp14::ura4+ dis1-GFP-kanR hphR-GBP-mCherry-alp4 leu1 ura4 | 5B–C | This study |
| TK590 | h+ alp7::ura4+ dis1-GFP-kanR hphR-GBP-mCherry-alp4 leu1 ura4 his2 | 5B–C | This study |
| TK578 | h− dis1-GFP-kanR leu1 ura4 | 5C | This study |
| TK602 | h+ alp14::ura4+ dis1-GFP-kanR leu1 ura4 his2 | 5C | This study |
| MS187 | h− alp7::ura4+ dis1-GFP-kanR leu1 ura4 | 5C | Our lab stock |
| MY2021 | h− alp14-NLS-GFP-kanR mis12-GBP-6HIS-mCherry-natR leu1 ura4 | 6B–C | This study |
| MY2041 | h− dis1::hphR alp14-NLS-GFP-kanR mis12-GBP-6HIS-mCherry-natR leu1 ura4 | 6B–C | This study |
| MY2027 | h− alp7::ura4+ alp14-NLS-GFP-kanR mis12-GBP-6HIS-mCherry-natR leu1 ura4 | 6B–C | This study |
| MS836 | h+ alp14-NLS-GFP-kanR leu1 ura4 his2 | 6C | Our lab stock |
| TK360 | h− dis1::hphR alp14-NLS-GFP-kanR leu1 ura4 | 6C | This study |
| MS838 | h− alp7::ura4+ alp14-NLS-GFP-kanR leu1 ura4 | 6C | Our lab stock |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yukawa, M.; Kawakami, T.; Pinder, C.; Toda, T. Two XMAP215/TOG Microtubule Polymerases, Alp14 and Dis1, Play Non-Exchangeable, Distinct Roles in Microtubule Organisation in Fission Yeast. Int. J. Mol. Sci. 2019, 20, 5108. https://doi.org/10.3390/ijms20205108
Yukawa M, Kawakami T, Pinder C, Toda T. Two XMAP215/TOG Microtubule Polymerases, Alp14 and Dis1, Play Non-Exchangeable, Distinct Roles in Microtubule Organisation in Fission Yeast. International Journal of Molecular Sciences. 2019; 20(20):5108. https://doi.org/10.3390/ijms20205108
Chicago/Turabian StyleYukawa, Masashi, Tomoki Kawakami, Corinne Pinder, and Takashi Toda. 2019. "Two XMAP215/TOG Microtubule Polymerases, Alp14 and Dis1, Play Non-Exchangeable, Distinct Roles in Microtubule Organisation in Fission Yeast" International Journal of Molecular Sciences 20, no. 20: 5108. https://doi.org/10.3390/ijms20205108
APA StyleYukawa, M., Kawakami, T., Pinder, C., & Toda, T. (2019). Two XMAP215/TOG Microtubule Polymerases, Alp14 and Dis1, Play Non-Exchangeable, Distinct Roles in Microtubule Organisation in Fission Yeast. International Journal of Molecular Sciences, 20(20), 5108. https://doi.org/10.3390/ijms20205108