Physiological, Hormonal and Metabolic Responses of two Alfalfa Cultivars with Contrasting Responses to Drought
Abstract
:1. Introduction
2. Results
2.1. Preliminary Study
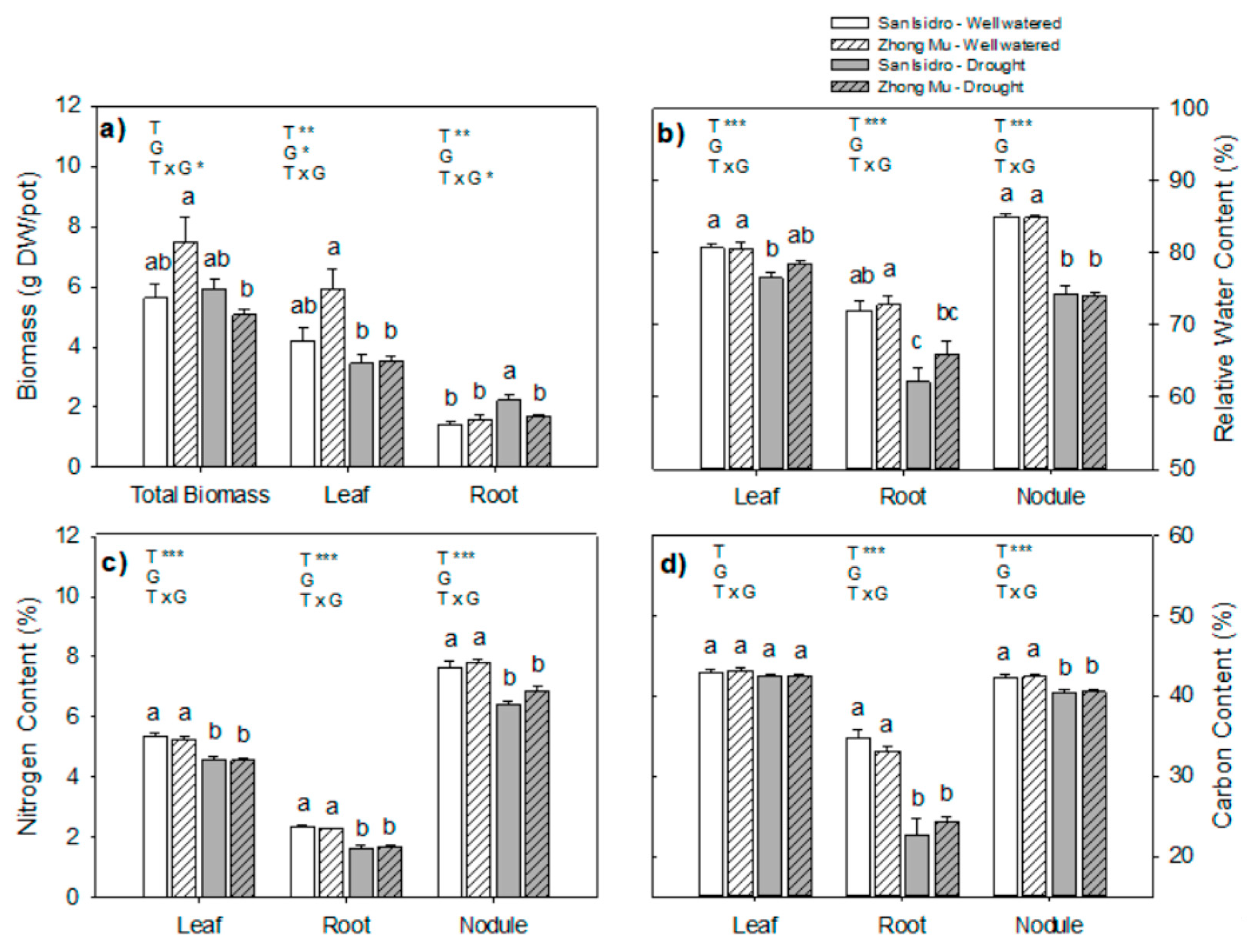
2.2. Water Limitation Effects on Plant Growth
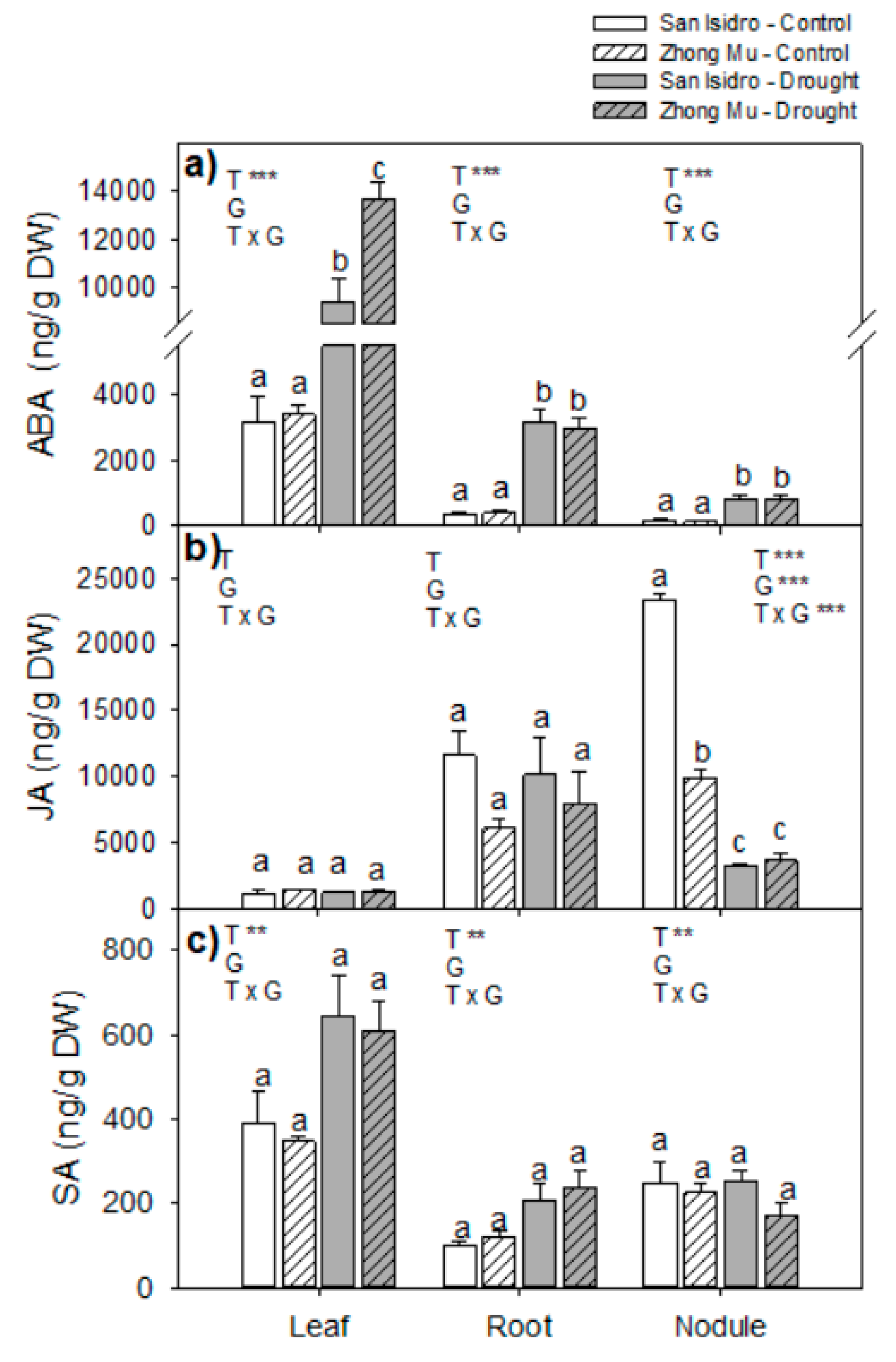
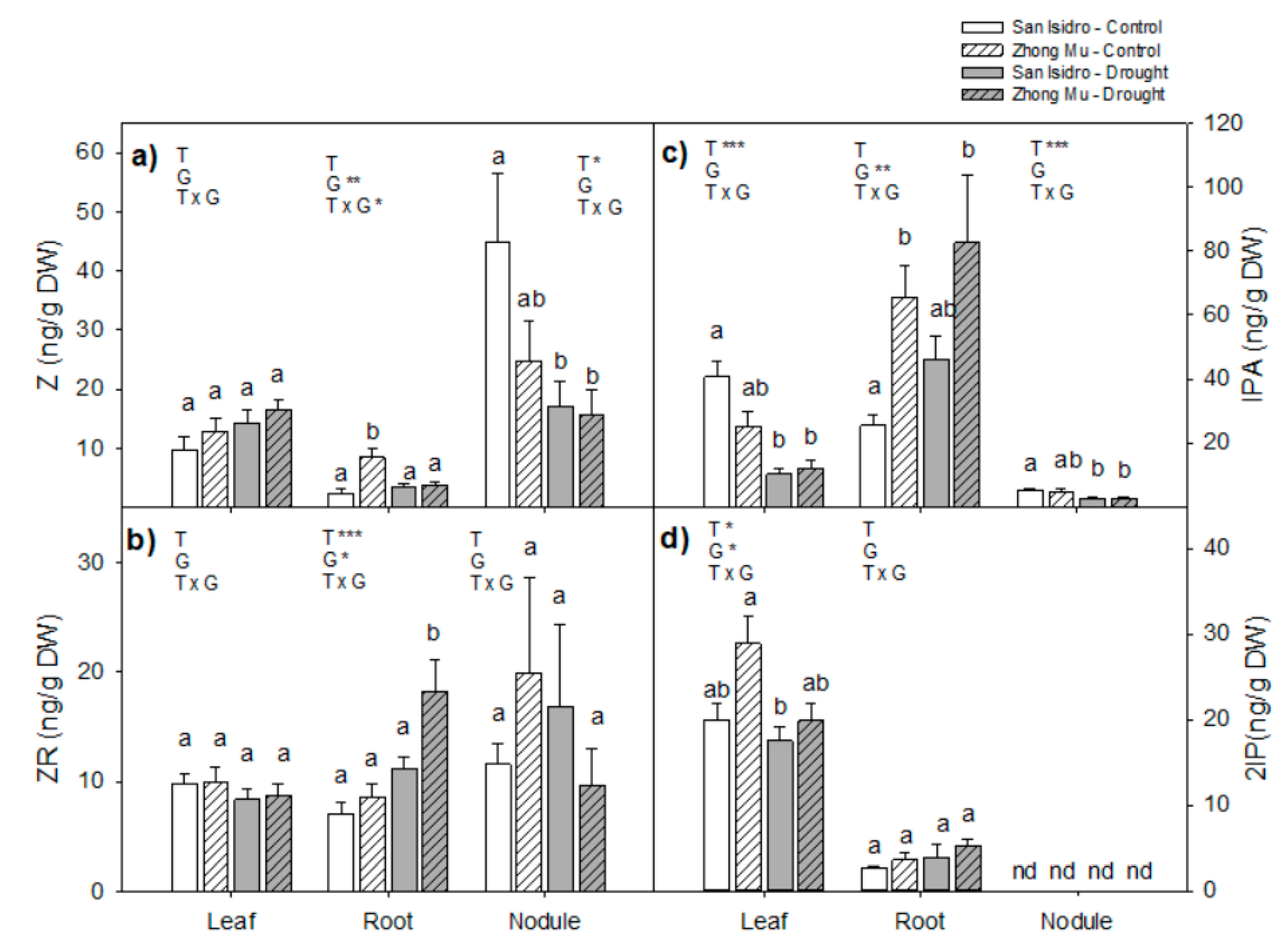
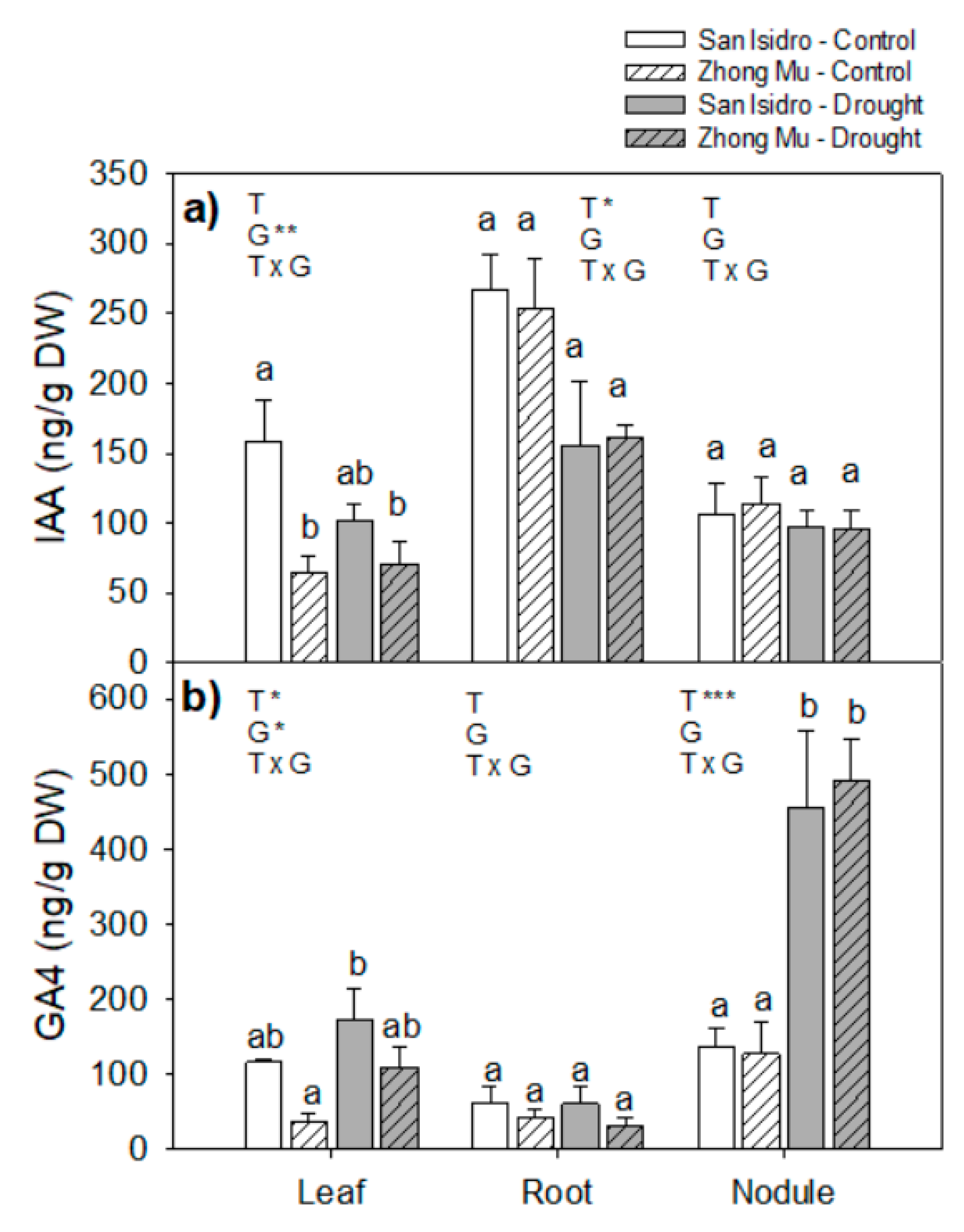
2.3. Hormones
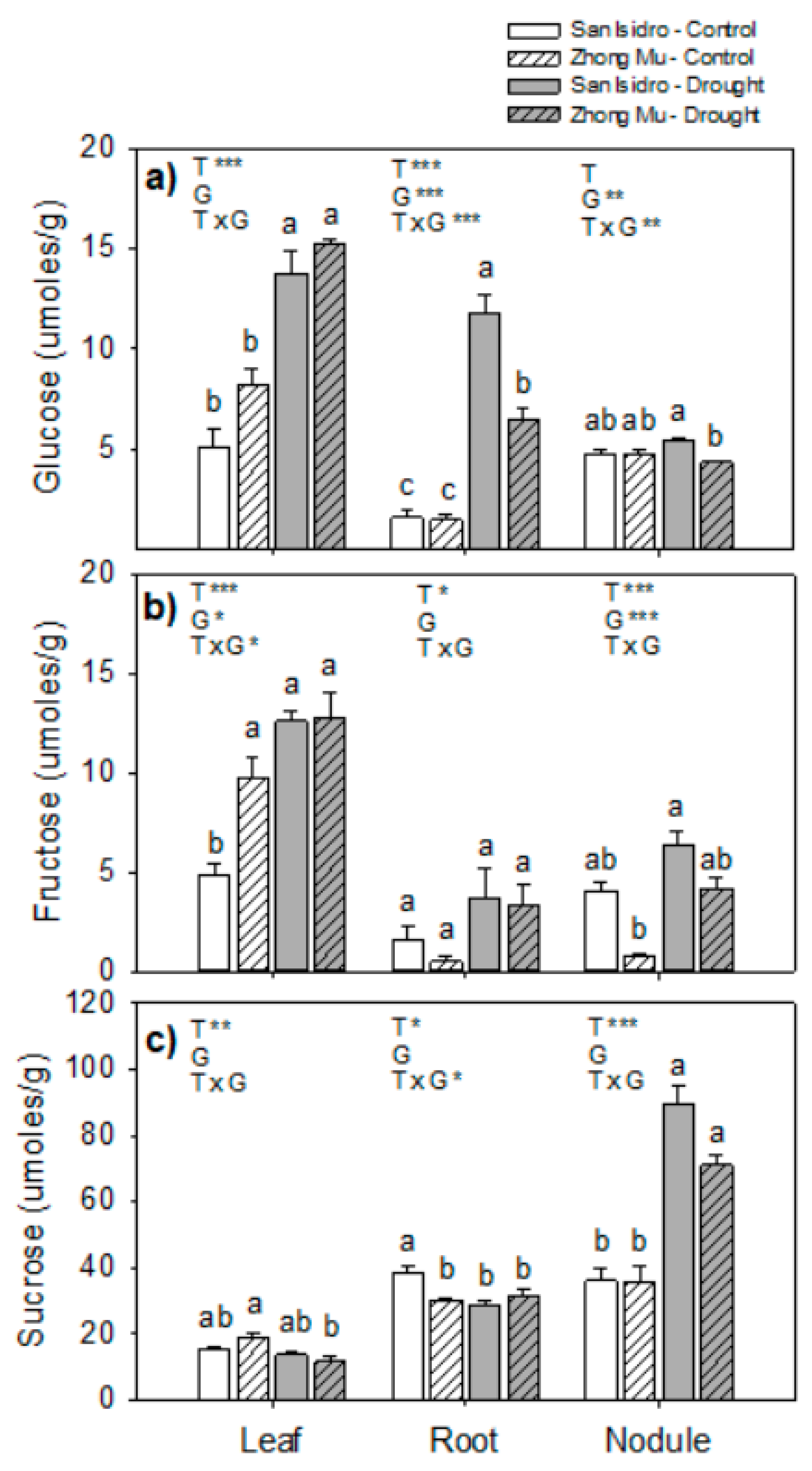
2.4. Amino Acids and Sugars
3. Discussion
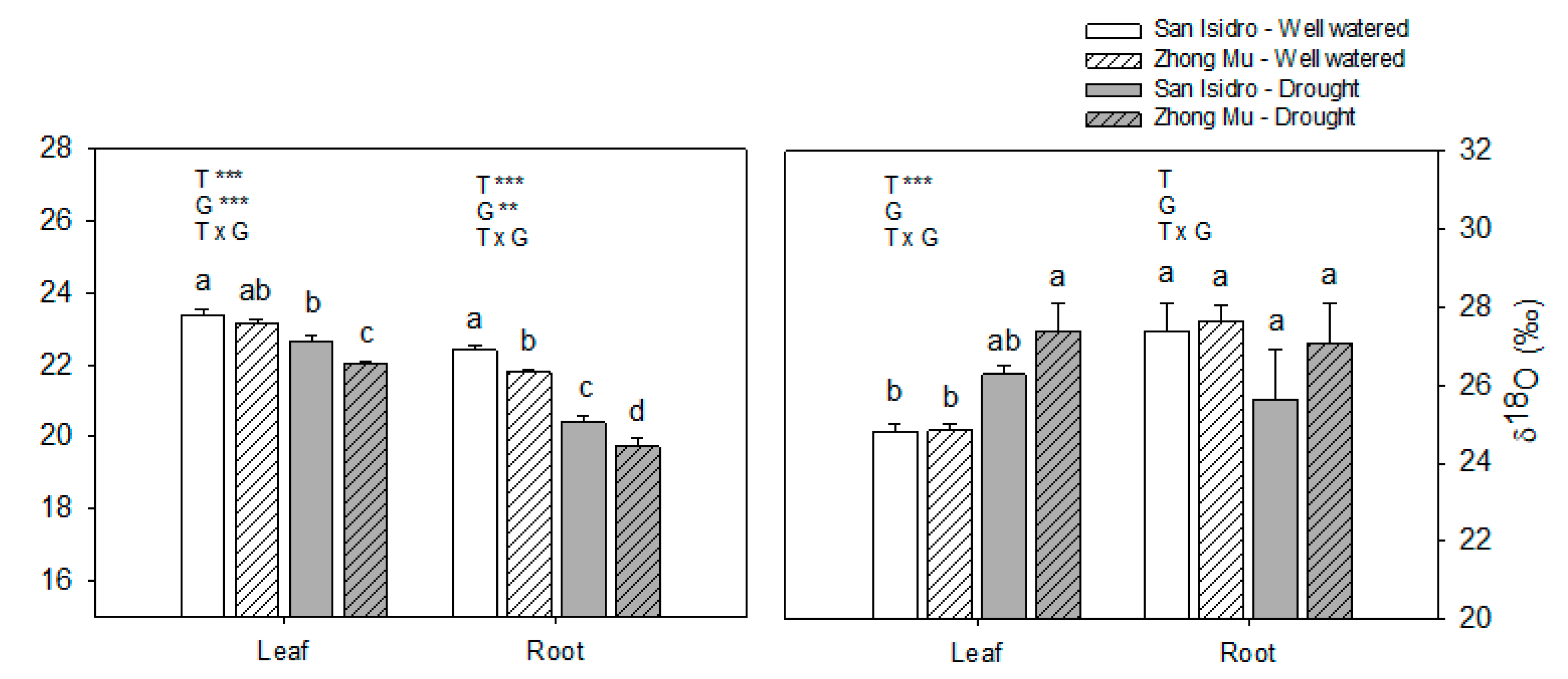
3.1. Drought Induces Different Agrophysiological Responses between Cultivars
3.2. Drought Caused Hormonal Reorchestration at the Whole Plant Level
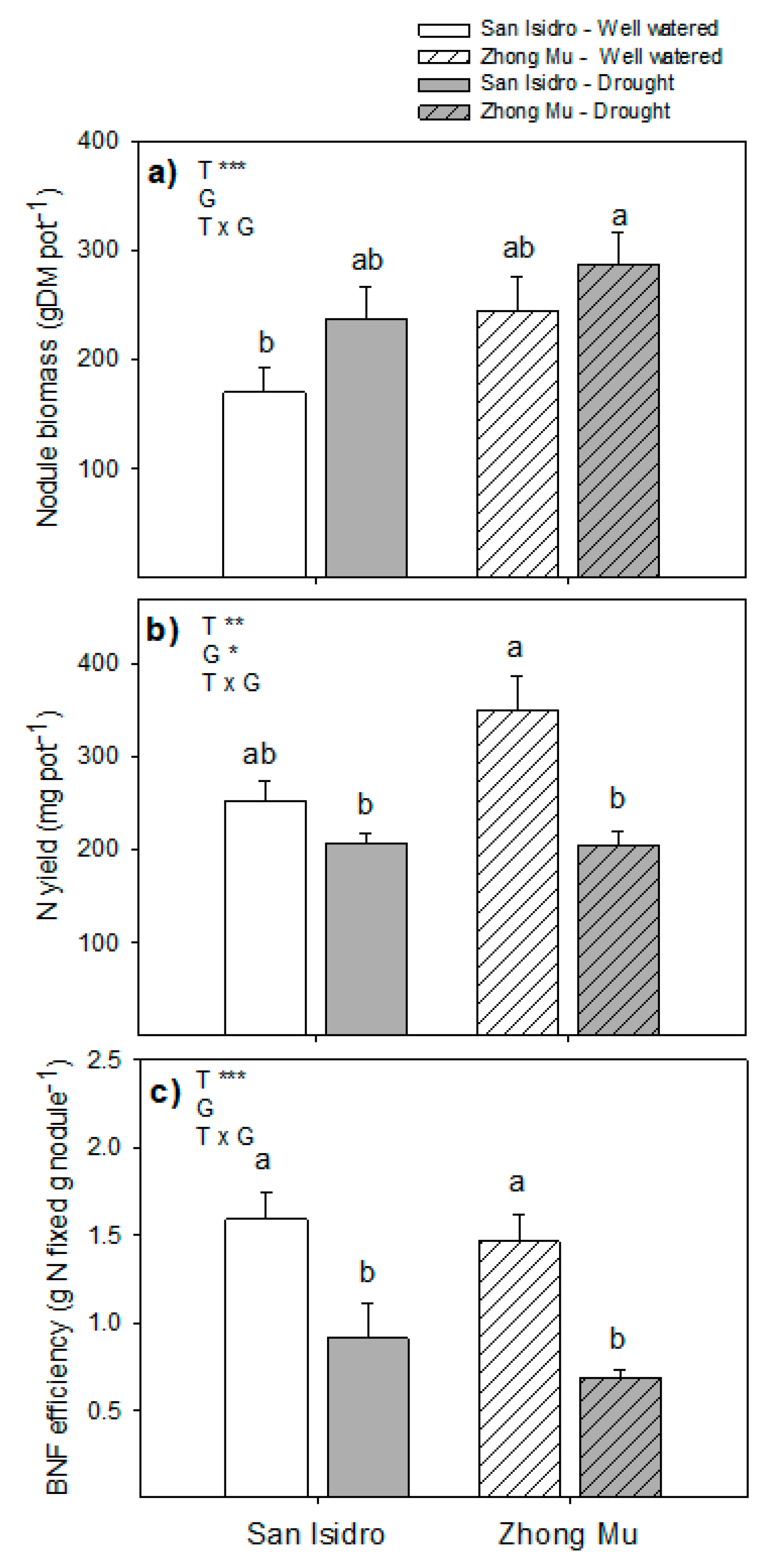
3.3. Drought Induces Lower N2 Fixation, and Cultivar Differences could be Explained by N Feedback
4. Materials and Methods
4.1. Plant Material and Experimental Design
4.2. Gas Exchange and Chlorophyll Fluorescence Determinations
4.3. Hormone Profiling
4.4. Free Amino Acid and Sugar Determinations
4.5. Carbon and Nitrogen Content
4.6. Carbon 13C Discrimination and Oxygen Isotopic Composition Analyses (Δ13C and δ18O Respectively)
4.7. Statistics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| N | Nitrogen |
| BNF | Biological nitrogen fixation |
| C | Carbon |
| ABA | Abscisic acid |
| RWC | Relative water content |
| A | Photosynthetic rates |
| E | Transpiration |
| gs | Stomatal conductance |
| Ci | Substomatic CO2 concentration |
| SA | Salicylic acid |
| JA | Jasmonic acid |
| Z | Trans-zeatin |
| ZR | Trans-zeatin riboside |
| IAA | Indole-3-acetic acid |
| GA1 | Gibberellin 1 |
| GA4 | Gibberellin 4 |
| IPA | Isopentenyl adenosine |
| 2iP | Isopentenyl adenine |
| DW | Dry weight |
| ∆13C | Carbon isotope discrimination |
| Δ18O | Oxygen isotope ratio |
| WUE | Water use efficiency |
| SS | Sucrose synthase |
| ROS | Reactive oxygen species |
| GABA | Gamma-aminobutyric acid |
| Asn | Asparagine |
| Asp | Aspartic acid |
| Glu | Glutamic acid |
| Pro | Proline |
References
- Annicchiarico, P.; Barrett, B.; Brummer, E.C.; Julier, B.; Marshall, A.H. Achievements and challenges in improving temperate perennial forage legumes. Crit. Rev. Plant Sci. 2015, 34, 327–380. [Google Scholar] [CrossRef]
- Radović, J.; Sokolović, D.; Marković, J. Alfalfa—most important perennial forage legume in animal husbandry. Biotech. Anim. Husb. 2009, 25, 465–475. [Google Scholar] [CrossRef]
- Steinfeld, H.; Gerber, P.; Wassenaar, T.D. Livestock’s Long Shadow: Environmental Issues and Options; FAO: Rome, Italy, 2006. [Google Scholar]
- Daryanto, S.; Wang, L.; Jacinthe, P.A. Global synthesis of drought effects on food legume production. PLoS ONE 2015, 10, 6. [Google Scholar] [CrossRef] [PubMed]
- Bray, E.A. Plant responses to water deficit. Trends Plant Sci. 1997, 2, 48–54. [Google Scholar] [CrossRef]
- Cattivelli, L.; Rizza, F.; Badeck, F.W.; Mazzucotelli, E.; Mastrangelo, A.M.; Francia, E.; Marè, C.; Tondelli, A.; Stanca, A.M. Drought tolerance improvement in crop plants: An integrated view from breeding to genomics. Field Crops Res. 2008, 105, 1–14. [Google Scholar] [CrossRef]
- Serraj, R.; Sinclair, T.R. Inhibition of nitrogenase activity and nodule oxygen permeability by water deficit. J. Exp. Bot. 1996, 47, 1067–1073. [Google Scholar] [CrossRef] [Green Version]
- Serraj, R.; Vadez, V.; Sinclair, T.R. Feedback regulation of symbiotic N2 fixation under drought stress. Agronomie 2001, 21, 621–626. [Google Scholar] [CrossRef]
- Durand, J.L.; Sheehy, J.E.; Minchin, F.R. Nitrogenase activity: Photosynthesis and nodule water potential in soybean plants experiencing water-deprivation. J. Exp. Bot. 1987, 38, 311–321. [Google Scholar] [CrossRef]
- Djekoun, A.; Planchon, C. Water status effect on dinitrogen fixation and photosynthesis in soybean. Agron. J. 1991, 83, 316–322. [Google Scholar] [CrossRef]
- Aranjuelo, I.; Arrese-Igor, C.; Molero, G. Nodule performance within a changing environmental context. J. Plant Physiol. 2014, 171, 1076–1090. [Google Scholar] [CrossRef] [Green Version]
- Molero, G.; Tcherkez, G.; Roca, R.; Mauve, C.; Cabrera-Bosquet, L.; Araus, J.L.; Nogués, S.; Aranjuelo, I. Do metabolic changes underpin physiological responses to water limitation in alfalfa (Medicago sativa) plants during a regrowth period? Agric. Water Manag. 2019, 212, 1–11. [Google Scholar] [CrossRef]
- Aranjuelo, I.; Molero, G.; Erice, G.; Avice, J.C.; Nogués, S. Plant physiology and proteomics reveals the leaf response to drought in alfalfa (Medicago sativa L.). J. Exp. Bot. 2011, 62, 111–123. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, S.; Davies, W.J. Drought, ozone, ABA and ethylene: New insights from cell to plant to community. Plant Cell Environ. 2010, 33, 510–525. [Google Scholar] [CrossRef] [PubMed]
- Dodd, I.C. Abscisic acid and stomatal closure: A hydraulic conductance conundrum? New Phytol. 2013, 197, 6–8. [Google Scholar] [CrossRef] [PubMed]
- Sah, S.K.; Kambham, R.R.; Li, J. Abscisic acid and abiotic stress tolerance in crop plants. Front. Plant Sci. 2016, 7, 571. [Google Scholar] [CrossRef] [PubMed]
- Xiong, L.; Schumaker, K.S.; Zhu, J.K. Cell signaling during cold, drought, and salt stress. Plant Cell. 2002, 14, S165–S183. [Google Scholar] [CrossRef] [PubMed]
- Defez, R.; Andreozzi, A.; Dickinson, M.; Charlton, A.; Tadini, L.; Pesaresi, P.; Bianco, C. Improved drought stress response in alfalfa plants modulated by an IAA over-producing rhizobium strain. Front. Microbiol. 2017, 8, 2466. [Google Scholar] [CrossRef]
- Ferguson, B.J.; Mathesius, U. Phytohormone regulation of legume-rhizobia interactions. J. Chem. Ecol. 2014, 40, 770–790. [Google Scholar] [CrossRef]
- Liu, H.; Zhang, C.; Yang, J.; Yu, N.; Wang, E. Hormone modulation of legume-rhizobial symbiosis. J. Integr. Plant Biol. 2018, 60, 632–648. [Google Scholar] [CrossRef]
- Suzaki, T.; Ito, M.; Kawaguchi, M. Genetic basis of cytokinin and auxin functions during root nodule development. Front. Plant Sci. 2013, 4, 42. [Google Scholar] [CrossRef] [Green Version]
- Ng, J.L.P.; Hassan, S.; Truong, T.T.; Hocart, C.H.; Laffont, C.; Frugier, F.; Mathesius, U. Flavonoids and auxin transport inhibitors rescue symbiotic nodulation in the Medicago truncatula cytokinin perception mutant cre1. Plant Cell. 2015, 27, 2210–2226. [Google Scholar] [CrossRef] [PubMed]
- Fisher, J.; Gaillard, P.; Fellbaum, C.R.; Subramanian, S.; Smith, S. Quantitative 3D imaging of cell level auxin and cytokinin response ratios in soybean roots and nodules. Plant Cell Environ. 2018, 41, 2080–2092. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferguson, B.J.; Foo, E.; Ross, J.J.; Reid, J.B. Relationship between gibberellin, ethylene and nodulation in Pisum sativum. New Phytol. 2011, 189, 829–842. [Google Scholar] [CrossRef]
- Sanchez, D.H.; Schwabe, F.; Erban, A.; Udvardi, M.K.; Kopka, J. Comparative metabolomics of drought acclimation in model and forage legumes. Plant Cell Environ. 2012, 35, 136–149. [Google Scholar] [CrossRef] [PubMed]
- Aranjuelo, I.; Molero, G.; Erice, G.; Aldasoro, J.; Arrese-Igor, C.; Nogués, S. Effect of shoot on remobilization of carbon and nitrogen during regrowth of nitrogen-fixing alfalfa. Physiol. Plant 2015, 153, 91–104. [Google Scholar] [CrossRef]
- Larrainzar, E.; Wienkoop, S.; Scherling, C.; Kempa, S.; Ladrera, R.; Arrese-Igor, C.; Weckwerth, W.; Gonzalez, E.M. Carbon metabolism and bacteroid functioning are involved in the regulation of nitrogen fixation in Medicago truncatula under drought and recovery. Mol. Plant Microbe Interact. 2009, 22, 1565–1576. [Google Scholar] [CrossRef]
- King, C.A.; Purcell, L.C. Inhibition of N2 fixation in soybean is associated with elevated ureides and amino acids. Plant Physiol. 2005, 137, 1389–1396. [Google Scholar] [CrossRef]
- Lodwig, E.; Poole, P. Metabolism of rhizobium bacteroids. Crit. Rev. Plant Sci. 2003, 22, 37–78. [Google Scholar] [CrossRef]
- Aldasoro, J.; Larrainzar, E.; Arrese-Igor, C. Application of anti-transpirants temporarily alleviates the inhibition of symbiotic nitrogen fixation in drought-stressed pea plants. Agric. Water Manag. 2019, 213, 193–199. [Google Scholar] [CrossRef]
- Sinclair, T.R.; Nogueira, M.A. Selection of host-plant genotype: The next step to increase grain legume N2 fixation activity. J. Exp. Bot. 2018, 69, 3523–3530. [Google Scholar] [CrossRef]
- Wu, Y.; Cosgrove, D.J. Adaptation of roots to low water potentials by changes in cell wall extensibility and cell wall proteins. J. Exp. Bot. 2000, 51, 1543–1553. [Google Scholar] [CrossRef] [PubMed]
- Furlam, A.L.; Bianucci, E.; Castro, S.; Dietz, K.J. Metabolic features involved in drought stress tolerance mechanisms in peanut nodules and their contribution to biological nitrogen fixation. Plant Sci. 2017, 263, 12–22. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Andersen, M.N.; Jacobsen, S.E.; Jensen, C.R. Stomatal control and water use efficiency of soybean (Glycine max L. Merr.) during progressive soil drying. Environ. Exp. Bot. 2005, 54, 33–40. [Google Scholar] [CrossRef]
- Jongrungklanga, N.; Toomsana, B.; Vorasoot, N.; Jogloy, S.; Boote, K.J.; Hoogenboom, G.; Patanothaia, A. Rooting traits of peanut genotypes with different yield responses to pre-flowering drought stress. Field Crop. Res. 2011, 120, 262–270. [Google Scholar] [CrossRef]
- Silvente, S.; Sobolev, A.P.; Lara, M. Metabolite adjustments in drought tolerant and sensitive soybean genotypes in response to water stress. PLoS ONE. 2012, 7, 6. [Google Scholar] [CrossRef]
- Farquhar, G.D.; Richards, R.A. Isotopic composition of plant carbon correlates with water-use efficiency of wheat genotypes. Aust. J. Plant Physiol. 1984, 11, 539–552. [Google Scholar] [CrossRef]
- Farquhar, G.D.; Ehleringer, J.R.; Hubick, K.T. Carbon isotope discrimination and photosynthesis. Annu. Rev. Plant Physiol. 1989, 40, 503–537. [Google Scholar] [CrossRef]
- Yousfi, S.; Serret, M.D.; Márquez, A.J.; Voltas, J.; Araus, J.L. Combined use of δ13C, δ18O and δ15N tracks nitrogen metabolism and genotypic adaptation of durum wheat to salinity and water deficit. New Phytol. 2012, 194, 230–244. [Google Scholar] [CrossRef]
- Barbour, M.M.; Fischer, R.A.; Sayre, K.D.; Farquhar, G.D. Oxygen isotope ratio of leaf and grain material correlates with stomatal conductance and yield in irrigated, field-grown wheat. Aust. J. Plant Physiol. 2000, 27, 625–637. [Google Scholar]
- Barbour, M.M.; Farquhar, G.D. Relative humidity- and ABA-induced variation in carbon and oxygen isotope ratios of cotton leaves. Plant Cell Environ. 2000, 23, 473–485. [Google Scholar] [CrossRef]
- Del Pozo, A.; Matus, I.; Serret, M.D.; Araus, J.L. Agronomic and physiological traits associated with breeding advances of wheat under high-productive Mediterranean conditions. The case of Chile. Environ. Exp. Bot. 2014, 103, 180–189. [Google Scholar] [CrossRef]
- Cabrera-Bosquet, L.; Molero, G.; Nogués, S.; Araus, J.L. Water and nitrogen conditions affect the relationships of δ13C and δ18O with gas exchange and growth in durum wheat. J. Exp. Bot. 2009, 60, 1633–1644. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, J.I.; Allen, G.J.; Hugouvieux, V.; Kwak, J.M.; Waner, D. Guard cell signal transduction. Annu. Rev. Plant Physiol. Plant Mol. Biol. 2001, 52, 627–658. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Davies, W.J. Sequential responses of whole plant water relations towards prolonged soil drying and the mediation by xylem sap ABA concentration in the regulation of stomatal behaviour of sunflower plants. New Phytol. 1989, 113, 167–174. [Google Scholar] [CrossRef]
- Jia, W.; Zhang, J.; Zhang, D.P. Metabolism of xylem-delivered ABA in relation to ABA flux and concentration in leaves of maize and Commelina communis. J. Exp. Bot. 1996, 47, 1085–1091. [Google Scholar] [CrossRef]
- González, E.M.; Gálvez, L.; Royuela, M.; Aparicio-Tejo, P.M.; Arrese-Igor, C. Insights into the regulation of nitrogen fixation in pea nodules: Lessons from drought, abscisic acid and increased photoassimilate availability. Agronomie 2001, 21, 607–613. [Google Scholar] [CrossRef]
- Abreu, M.E.; Munné-Bosch, S. Photo- and antioxidant protection and salicylic acid accumulation during post-anthesis leaf senescence in Salvia lanigera grown under Mediterranean climate. Physiol. Plant. 2007, 131, 590–598. [Google Scholar] [CrossRef]
- Nakagawa, T.; Kawaguchi, M. Shoot-applied MeJA suppresses root nodulation in Lotus japonicus. Plant Cell Physiol. 2006, 47, 176–180. [Google Scholar] [CrossRef]
- Sun, J.; Cardoza, V.; Mitchell, D.M.; Bright, L.; Oldroyd, G.; Harris, J.M. Crosstalk between jasmonic acid, ethylene and Nod factor signaling allow integration of diverse inputs for regulation of nodulation. Plant J. 2006, 46, 961–970. [Google Scholar] [CrossRef]
- Chen, Y.; Chen, W.; Li, X.; Jiang, H.; Wu, P.; Xia, K.; Yang, Y.; Wu, G. Knockdown of LjIPT3 influences nodule development in Lotus japonicus. Plant Cell Physiol. 2014, 55, 183–193. [Google Scholar] [CrossRef]
- Boivin, S.; Kazmierczak, T.; Brault, M.; Wen, J.; Gamas, P.; Mysore, K.S.; Frugier, F. Different cytokinin CHK receptors regulate nodule initiation as well as later nodule developmental stages in Medicago truncatula. Plant Cell Environ. 2016, 39, 2198–2209. [Google Scholar] [CrossRef] [PubMed]
- Reid, D.E.; Heckmann, A.B.; Novak, O.; Kelly, S.; Stougaard, J. CYTOKININ OXIDASE/DESHYDROGENASE3 maintains cytokinin homeostasis during root and nodule development in Lotus japonicus. Plant Physiol. 2016, 170, 1060–1074. [Google Scholar] [CrossRef] [PubMed]
- Cowan, A.K.; Cairns, A.L.P.; Bartels-Rahm, B. Regulation of abscisic acid metabolism: Towards a metabolic basis for abscisic acid-cytokinin antagonism. J. Exp. Bot. 1999, 50, 595–603. [Google Scholar] [CrossRef]
- Pospíšilová, J.; Synková, H.; Rulcová, J. Cytokinins and water stress. Biol. Plant. 2000, 43, 321–328. [Google Scholar] [CrossRef]
- Davies, W.J.; Zhang, J. Root signals and the regulation of growth and development of plants in drying soil. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1991, 42, 55–76. [Google Scholar] [CrossRef]
- Ferguson, B.J.; Ross, J.J.; Reis, J. Nodulation Phenotypes of gibberellin and brassinosteroid mutants of Pea. Plant physiol. 2005, 138, 2396–2405. [Google Scholar] [CrossRef] [PubMed]
- Aranjuelo, I.; Irigoyen, J.J.; Sánchez-Díaz, M. Effect of elevated temperature and water availability on CO2 exchange and nitrogen fixation of nodulated alfalfa plants. Environ. Exp. Bot. 2007, 59, 99–108. [Google Scholar] [CrossRef]
- Aranjuelo, I.; Irigoyen, J.J.; Sánchez-Díaz, M.; Nogués, S. Carbon partitioning in N2 fixing Medicago sativa plants exposed to different CO2 and temperature conditions. Funct. Plant Biol. 2008, 35, 306–317. [Google Scholar] [CrossRef]
- Gálvez, L.; González, E.M.; Arrese-Igor, C. Evidence for carbon flux shortage and strong carbon/nitrogen interactions in pea nodules at early stages of water stress. J. Exp. Bot. 2005, 56, 2551–2561. [Google Scholar] [CrossRef] [Green Version]
- Schulze, J. How are nitrogen fixation rates regulated in legumes? J. Plant Nutr. Soil Sci. 2004, 167, 125–137. [Google Scholar] [CrossRef]
- Serraj, R. Effects of drought stress on legume symbiotic nitrogen fixation: Physiological mechanisms. Indian J. Exp. Biol. 2003, 41, 1136–1141. [Google Scholar] [PubMed]
- Naya, L.; Ladrera, R.; Ramos, J.; González, E.M.; Arrese-Igor, C.; Minchin, F.R.; Becana, M. The response of carbon metabolism and antioxidant defenses of alfalfa nodules to drought stress and to the subsequent recovery of plants. Plant Physiol. 2007, 144, 1104–1114. [Google Scholar] [CrossRef] [PubMed]
- Egli, D.B.; Ramseur, E.L.; Zhen-wen, Y.; Sullivan, E.H. Source-sink alterations affect the number of cells in soybean cotyledons. Crop. Sci. 1989, 29, 732–735. [Google Scholar] [CrossRef]
- González, E.M.; Gordon, A.J.; James, C.L.; Arrese-Igor, C. The role of sucrose synthase in the response of soybean nodules to drought. J. Exp. Bot. 1995, 46, 1515–1523. [Google Scholar] [CrossRef]
- Gordon, A.J.; Minchin, F.R.; James, C.L.; Komina, O. Sucrose synthase in legume nodules is essential for nitrogen fixation. Plant Physiol. 1999, 120, 867–877. [Google Scholar] [CrossRef]
- Zhang, J.Y.; Cruz de Carvalho, M.H.; Torres-Jerez, I.; Kang, Y.; Allen, S.N.; Huhman, D.V.; Tang, Y.; Murray, J.; Sumner, L.W.; Udvardi, M.K. Global reprogramming of transcription and metabolism in Medicago truncatula during progressive drought and after rewatering. Plant Cell Environ. 2014, 37, 2553–2576. [Google Scholar] [CrossRef]
- Turkan, I.; Bor, M.; Özdemir, F.; Koca, H. Differential responses of lipid peroxidation and antioxidants in the leaves of drought-tolerant P. acutifolius Gray and drought-sensitive P. vulgaris L. subjected to polyethylene glycol mediated water stress. Plant Sci. 2005, 168, 223–231. [Google Scholar] [CrossRef]
- Michaeli, S.; Fromm, H. Closing the loop on the GABA shunt in plants: Are GABA metabolism and signaling entwined? Front. Plant. Sci. 2015, 6, 419. [Google Scholar] [CrossRef]
- Hoagland, D.R.; Arnon, D.I. The water-culture method for growing plants without soil. Calif. Agric. Exp. Stn. 1950, 347, 1–32. [Google Scholar]
- Weatherley, P. Studies in the water relations of the cotton plant. 1. The field measurement of water deficits in leaves. New Phytol. 1950, 49, 81–97. [Google Scholar] [CrossRef]
- Caemmerer, S.; Farquhar, G.D. Some relationships between the biochemistry of photosynthesis and the gas exchange of leaves. Planta 1981, 153, 376–387. [Google Scholar] [CrossRef] [PubMed]
- Harley, P.C.; Loreto, F.; Di Marco, G.; Sharkey, T.D. Theoretical considerations when estimating the mesophyll conductance to CO2 flux by the analysis of the response of photosynthesis to CO2. Plant Physiol. 1992, 98, 1429–1436. [Google Scholar] [CrossRef] [PubMed]
- Müller, M.; Munné-Bosch, S. Rapid and sensitive hormonal profiling of complex plant samples by liquid chromatography coupled to electrospray ionization tandem mass spectrometry. Plant Meth. 2011, 7, 37. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.A.; Michaud, D.P. Synthesis of a fluorescent derivatizing reagent, 6-aminoquinolyl-n-hydroxysuccinimidyl carbamate, and its application for the analysis of hydrolysate amino acids via high-performance liquid chromatography. Anal. Biochem. 1993, 211, 279–287. [Google Scholar] [CrossRef]
| Leaves | Root | Nodule | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| San Isidro | Zhong Mu | San Isidro | Zhong Mu | San Isidro | Zhong Mu | |||||||
| WW | WS | WW | WS | WW | WS | WW | WS | WW | WS | WW | WS | |
| Asp | 0.76 b | 0.45 c | 0.97 a | 0.71 b | 0.86 a | 0.55 b | 0.69 b | 0.60 b | 1.07 | 1.24 | 1.31 | 1.01 |
| Glu | 2.05 a | 0.92 b | 2.37 a | 1.03 b | 1.23 a | 0.75 c | 0.97 ab | 0.86 bc | 4.56 | 3.70 | 4.49 | 3.41 |
| Ser | 1.51 | 1.65 | 1.73 | 1.56 | 0.98 | 0.84 | 0.83 | 0.76 | 0.84 b | 1.55 a | 0.97 b | 1.12 b |
| Asn | 9.88 a | 3.62 c | 7.80 b | 5.07 bc | 56.92 b | 44.20 b | 92.02 a | 59.29 b | 138.73 ab | 150.90 ab | 108.41 b | 179.55 a |
| Gly | 0.50 | 0.59 | 0.58 | 0.62 | 1.39 a | 0.79 b | 0.99 b | 0.84 b | 0.81 ab | 1.01 a | 0.72 b | 0.84 ab |
| Gln | 0.62 | 0.60 | 0.62 | 0.68 | 0.70 | 0.88 | 0.68 | 0.67 | 0.81 | 1.20 | 1.13 | 1.19 |
| His | 0.24 ab | 0.26 ab | 0.21 b | 0.30 a | 0.21 | 0.23 | 0.22 | 0.21 | 0.38 b | 1.06 a | 0.50 b | 0.85 a |
| Thr | 0.97 | 1.60 | 1.51 | 2.03 | 0.93 | 1.25 | 0.88 | 1.11 | 0.77 b | 1.43 a | 0.75 b | 0.95 b |
| Ala | 27.16 a | 23.80 b | 23.43 b | 24.54 b | 8.73 | 8.34 | 10.79 | 10.00 | 19.75 | 19.05 | 21.42 | 26.16 |
| Arg | 1.26 | 1.82 | 1.40 | 1.98 | 1.20 | 1.46 | 0.85 | 1.28 | 0.51 bc | 1.05 a | 0.48 c | 0.85 ab |
| GABA | 1.16 | 1.68 | 1.29 | 1.77 | 1.03 c | 2.85 a | 0.79 c | 1.83 b | 1.25 b | 3.29 a | 1.11 b | 2.56 a |
| Pro | 1.47 b | 5.83 a | 1.56 b | 5.64 a | 3.16 b | 9.98 a | 3.67 b | 12.40 a | 1.80 b | 18.93 a | 2.60 b | 19.86 a |
| Tyr | 0.55 | 0.60 | 0.53 | 0.69 | 0.36 b | 0.43 a | 0.37 b | 0.38 b | 0.56 c | 1.05 a | 0.54 c | 0.81 b |
| Val | 0.56 b | 0.78 ab | 0.56 b | 0.91 b | 0.31 c | 0.62 a | 0.32 c | 0.54 b | 0.53 b | 1.70 a | 0.54 b | 1.24 a |
| Met | 0.40 ab | 0.46 ab | 0.39 b | 0.55 a | 0.33 a | 0.34 b | 0.26 ac | 0.29 c | 0.35 b | 0.51 a | 0.36 b | 0.40 b |
| Ile | 0.54 ab | 0.69 ab | 0.51 b | 0.77 a | 0.33 bc | 0.48 a | 0.32 c | 0.37 b | 0.58 c | 1.62 a | 0.57 c | 1.09 b |
| Leu | 0.86 | 1.08 | 0.84 | 1.29 | 0.30 b | 0.49 a | 0.29 b | 0.35 b | 0.57 c | 1.50 a | 0.54 c | 0.97 b |
| Lys | 0.45 | 0.63 | 0.42 | 0.84 | 0.06 c | 0.21 a | 0.05 c | 0.12b | -- | -- | -- | -- |
| Phe | 0.68 ab | 0.77 ab | 0.59 b | 0.90 a | 0.36 c | 0.49 ab | 0.37 bc | 0.43 a | 0.54 c | 1.26 a | 0.53 c | 0.90 b |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Soba, D.; Zhou, B.; Arrese-Igor, C.; Munné-Bosch, S.; Aranjuelo, I. Physiological, Hormonal and Metabolic Responses of two Alfalfa Cultivars with Contrasting Responses to Drought. Int. J. Mol. Sci. 2019, 20, 5099. https://doi.org/10.3390/ijms20205099
Soba D, Zhou B, Arrese-Igor C, Munné-Bosch S, Aranjuelo I. Physiological, Hormonal and Metabolic Responses of two Alfalfa Cultivars with Contrasting Responses to Drought. International Journal of Molecular Sciences. 2019; 20(20):5099. https://doi.org/10.3390/ijms20205099
Chicago/Turabian StyleSoba, David, Bangwei Zhou, Cesar Arrese-Igor, Sergi Munné-Bosch, and Iker Aranjuelo. 2019. "Physiological, Hormonal and Metabolic Responses of two Alfalfa Cultivars with Contrasting Responses to Drought" International Journal of Molecular Sciences 20, no. 20: 5099. https://doi.org/10.3390/ijms20205099






