Methyl Donor Deficiency during Gestation and Lactation in the Rat Affects the Expression of Neuropeptides and Related Receptors in the Hypothalamus
Abstract
1. Introduction
2. Results
2.1. Effects of Methyl Donor Deficiency on the Expression of Neuropeptides and Receptors in Mouse and Rat Embryonic Hypothalamic Cell Lines
2.2. Plasma Concentrations of Folate, Vitamin B12 and Homocysteine, and Growth Status of Weaned Rat Pups
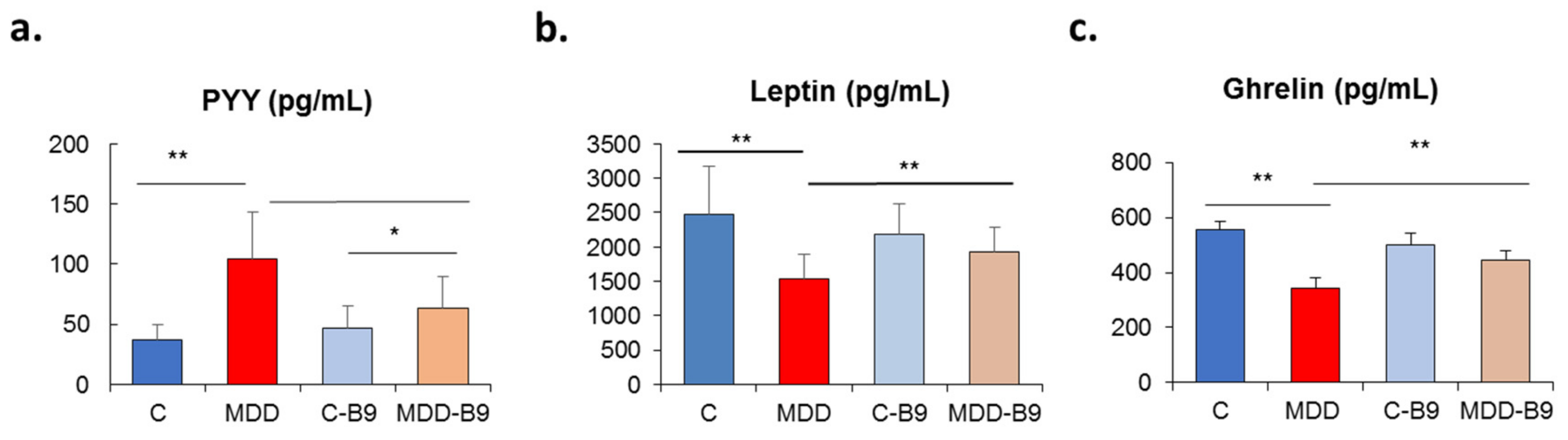
2.3. Plasma Concentrations of Peripheral Hormones
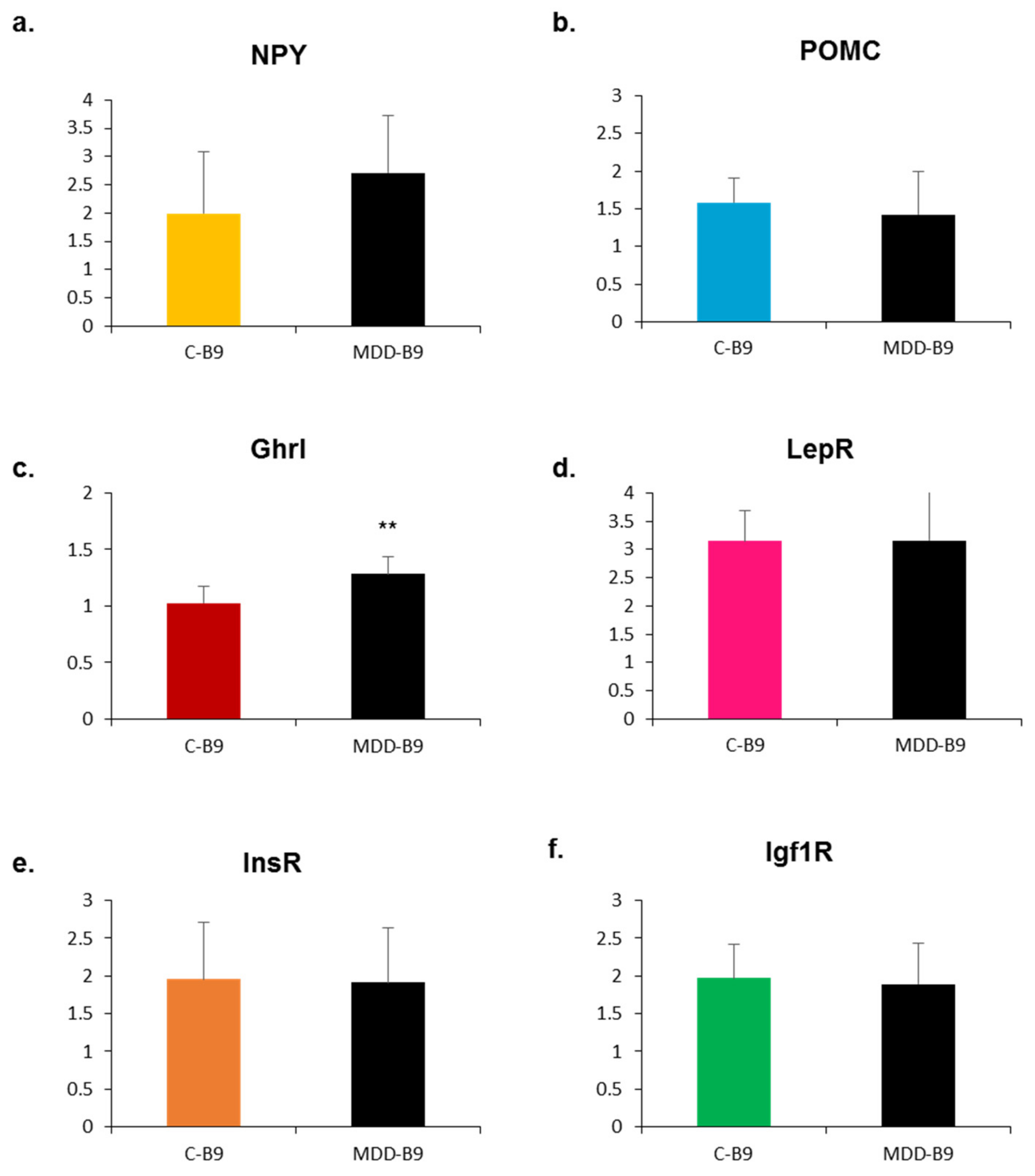
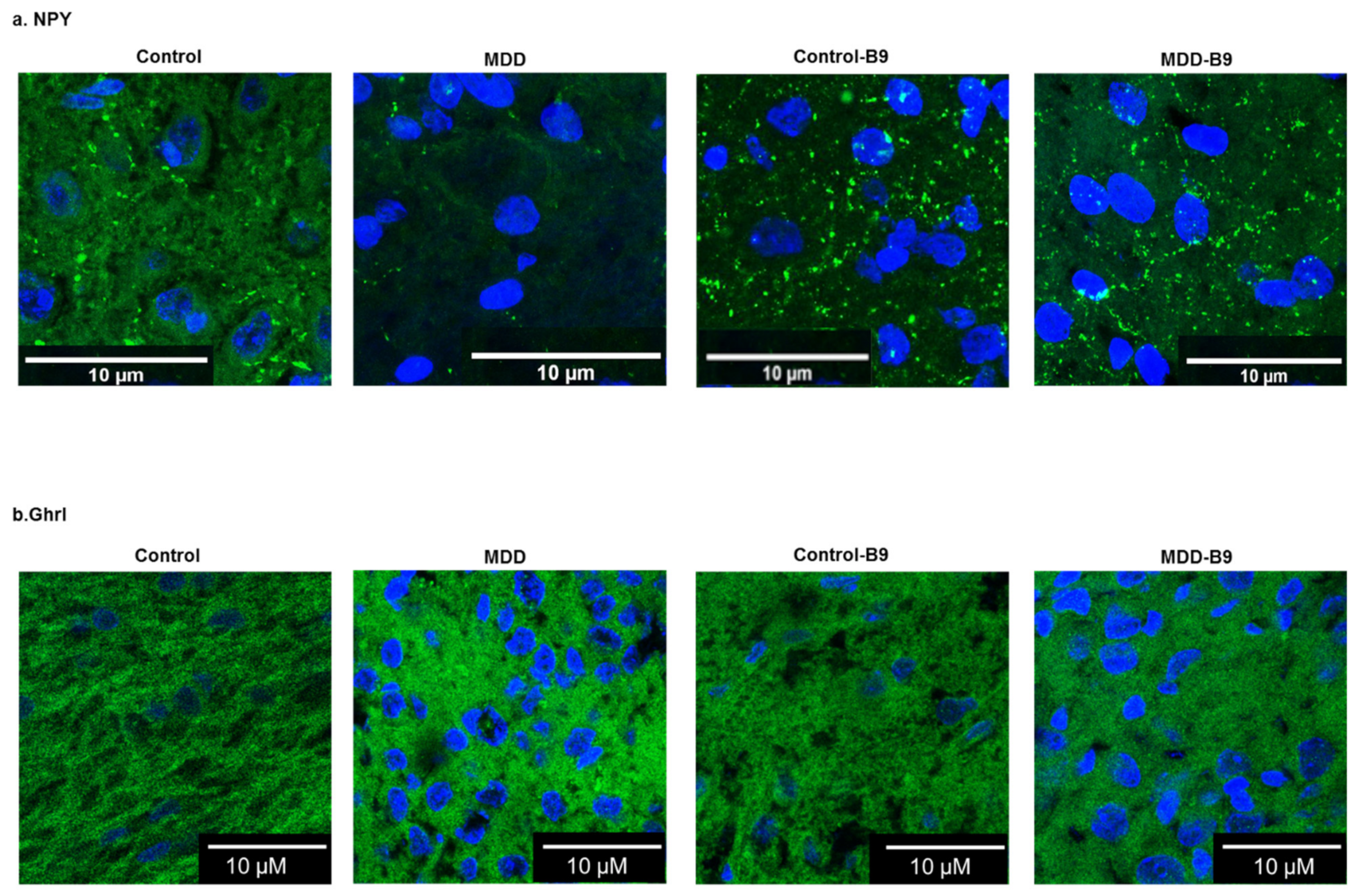
2.4. Effects of Methyl Donor Deficiency on the Expression of Neuropeptides and Receptors in the Hypothalamus of Rat Pups—Consequences of Maternal Folate Supplementation
3. Discussion
4. Materials and Methods
4.1. Cell Cultures
4.2. Animals and Tissue Collection
4.3. Plasma Assays
4.4. RNA Extraction and Quantitative RT-PCR
4.5. Immunohistochemistry
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Guéant, J.L.; Namour, F.; Guéant-Rodriguez, R.M.; Daval, J.L. Folate and fetal programming: A play in epigenomics? Trends Endocrinol. Metab. 2013, 24, 279–289. [Google Scholar] [CrossRef] [PubMed]
- Guéant, J.L.; Caillerez-Fofou, M.; Battaglia-Hsu, S.; Alberto, J.M.; Freund, J.N.; Dulluc, I.; Adjalla, C.; Maury, F.; Merle, C.; Nicolas, J.P.; et al. Molecular and cellular effects of vitamin B12 in brain, myocardium and liver through its role as co-factor of methionine synthase. Biochimie 2013, 95, 1033–1040. [Google Scholar] [CrossRef] [PubMed]
- Mattson, M.P.; Shea, T.B. Folate and homocysteine metabolism in neural plasticity and neurodegenerative disorders. Trends Neurosci. 2003, 26, 137–146. [Google Scholar] [CrossRef]
- Ghemrawi, R.; Pooya, S.; Lorentz, S.; Gauchotte, G.; Arnold, C.; Guéant, J.L.; Battaglia-Hsu, S.F. Decreased vitamin B12 availability induces ER stress through impaired SIRT1-deacetylation of HSF1. Cell Death Dis. 2013, 4, e553. [Google Scholar] [CrossRef]
- Akchiche, N.; Bossenmeyer-Pourié, C.; Kerek, R.; Martin, N.; Pourié, G.; Koziel, V.; Helle, D.; Alberto, J.M.; Ortiou, S.; Camadro, J.M.; et al. Homocysteinylation of neuronal proteins contributes to folate deficiency-associated alterations of differentiation, vesicular transport, and plasticity in hippocampal neuronal cells. FASEB J. 2012, 26, 3980–3992. [Google Scholar] [CrossRef]
- Jeong, S.O.; Son, Y.; Lee, J.H.; Choi, S.W.; Kim, S.H.; Cheong, Y.K.; Chung, H.T.; Pae, H.O. Both nitric oxide and nitrite prevent homocysteine-induced endoplasmic reticulum stress and subsequent apoptosis via cGMP-dependent pathway in neuronal cells. Biochem. Biophys. Res. Commun. 2017, 493, 164–169. [Google Scholar] [CrossRef]
- Kirke, P.N.; Molloy, A.M.; Daly, L.E.; Burke, H.; Weir, D.G.; Scott, J.M. Maternal plasma folate and vitamin B12 are independent risk factors for neural tube defects. QJM Int. J. Med. 1993, 86, 703–708. [Google Scholar]
- Molloy, A.M.; Kirke, P.N.; Brody, L.C.; Scott, J.M.; Mills, J.L. Effects of folate and vitamin B12 deficiencies during pregnancy on fetal, infant, and child development. Food Nutr. Bull. 2008, 29, S101–S111. [Google Scholar] [CrossRef]
- Black, M.M. Effects of vitamin B12 and folate deficiency on brain development in children. Food Nutr. Bull. 2008, 29, S126–S131. [Google Scholar] [CrossRef]
- Blaise, S.A.; Nédélec, E.; Schroeder, H.; Alberto, J.M.; Bossenmeyer-Pourié, C.; Guéant, J.L.; Daval, J.L. Gestational vitamin B deficiency leads to homocysteine-associated brain apoptosis and alters neurobehavioral development in rats. Am. J. Pathol. 2007, 170, 667–679. [Google Scholar] [CrossRef]
- Kerek, R.; Geoffroy, A.; Bison, A.; Martin, N.; Akchiche, N.; Pourié, G.; Helle, D.; Guéant, J.L.; Bossenmeyer-Pourié, C.; Daval, J.L. Early methyl donor deficiency may induce persistent brain defects by reducing Stat3 signaling targeted by miR-124. Cell Death Dis. 2013, 4, e755. [Google Scholar] [CrossRef] [PubMed]
- Bossenmeyer-Pourié, C.; Pourié, G.; Koziel, V.; Helle, D.; Jeannesson, E.; Guéant, J.L.; Beck, B. Early methyl donor deficiency produces severe gastritis in mothers and offspring through N-homocysteinylation of cytoskeleton proteins, cellular stress, and inflammation. FASEB J. 2013, 27, 2185–2197. [Google Scholar] [CrossRef] [PubMed]
- Bossenmeyer-Pourié, C.; Blaise, S.; Pourié, G.; Tomasetto, C.; Audonnet, S.; Ortiou, S.; Koziel, V.; Rio, M.C.; Daval, J.L.; Guéant, J.L.; et al. Methyl donor deficiency affects fetal programming of gastric ghrelin cell organization and function in the rat. Am. J. Pathol. 2010, 176, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Kojima, M.; Kangawa, K. Ghrelin: Structure and function. Physiol. Rev. 2005, 85, 495–522. [Google Scholar] [CrossRef]
- Brunetti, L.; Recinella, L.; Orlando, G.; Michelotto, B.; Di Nisio, C.; Vacca, M. Effects of ghrelin and amylin on dopamine, norepinephrine and serotonin release in the hypothalamus. Eur. J. Pharmacol. 2002, 454, 189–192. [Google Scholar] [CrossRef]
- Brunetti, L.; Di Nisio, C.; Recinella, L.; Orlando, G.; Ferrante, C.; Chiavaroli, A.; Leone, S.; Di Michele, P.; Shohreh, R.; Vacca, M. Obestatin inhibits dopamine release in rat hypothalamus. Eur. J. Pharmacol. 2010, 641, 142–147. [Google Scholar] [CrossRef]
- Morton, G.J.; Schwartz, M.W. The NPY/AgRP neuron and energy homeostasis. Int. J. Obes. Relat. Metab. Disord. 2001, 25, S56–S62. [Google Scholar] [CrossRef]
- Korner, J.; Leibel, R.L. To eat or not to eat - how the gut talks to the brain. N. Engl. J. Med. 2003, 349, 926–928. [Google Scholar] [CrossRef]
- Van den Top, M.; Lee, K.; Whyment, A.D.; Blanks, A.M.; Spanswick, D. Orexigen-sensitive NPY/AgRP pacemaker neurons in the hypothalamic arcuate nucleus. Nat. Neurosci. 2004, 7, 493–494. [Google Scholar] [CrossRef]
- Chaudhri, O.; Small, C.; Bloom, S. Gastrointestinal hormones regulating appetite. Phil. Trans. R. Soc. B. 2006, 361, 1187–1209. [Google Scholar] [CrossRef]
- Bates, S.H.; Stearns, W.H.; Dundon, T.A.; Schubert, M.; Tso, A.W.; Wang, Y.; Banks, A.S.; Lavery, H.J.; Haq, A.K.; Maratos-Flier, E.; et al. STAT3 signalling is required for leptin regulation of energy balance but not reproduction. Nature 2003, 421, 856–859. [Google Scholar] [CrossRef] [PubMed]
- Lau, C.; Rogers, J.M. Embryonic and fetal programming of physiological disorders in adulthood. Birth Defects Res. C Embryo Today 2004, 72, 300–312. [Google Scholar] [CrossRef] [PubMed]
- Breton, C. The hypothalamus-adipose axis is a key target of developmental programming by maternal nutritional manipulation. J. Endocrinol. 2013, 216, R19–R31. [Google Scholar] [CrossRef] [PubMed]
- Cowley, M.A.; Smith, R.G.; Diano, S.; Tschöp, M.; Pronchuk, N.; Grove, K.L.; Strasburger, C.J.; Bidlingmaier, M.; Esterman, M.; Heiman, M.L.; et al. The distribution and mechanism of action of ghrelin in the CNS demonstrates a novel hypothalamic circuit regulating energy homeostasis. Neuron 2003, 37, 649–661. [Google Scholar] [CrossRef]
- Geoffroy, A.; Kerek, R.; Pourié, G.; Helle, D.; Guéant, J.L.; Daval, J.L.; Bossenmeyer-Pourié, C. Late maternal folate supplementation rescues from methyl donor deficiency-associated brain defects by restoring let-7 and miR-34 pathways. Mol. Neurobiol. 2017, 54, 5017–5033. [Google Scholar] [CrossRef]
- Bâ, A. Perinatal thiamine deficiency-induced spontaneous abortion and pup-killing responses in rat dams. Nutr. Neurosci. 2013, 16, 69–77. [Google Scholar] [CrossRef]
- Geoffroy, A.; Saber-Cherif, L.; Pourié, G.; Helle, D.; Umoret, R.; Guéant, J.L.; Bossenmeyer-Pourié, C.; Daval, J.L. Developmental Impairments in a Rat Model of Methyl Donor Deficiency: Effects of a Late Maternal Supplementation with Folic Acid. Int. J. Mol. Sci. 2019, 20, 973. [Google Scholar] [CrossRef]
- Rinaman, L. Visceral sensory inputs to the endocrine hypothalamus. Front. Neuroendocr. 2007, 28, 50–60. [Google Scholar] [CrossRef]
- Warchoł, M.; Krauss, H.; Wojciechowska, M.; Opala, T.; Pięta, B.; Żukiewicz-Sobczak, W.; Kupsz, J.; Grochowalska, A. The role of ghrelin, leptin and insulin in foetal development. Ann. Agric. Environ. Med. 2014, 21, 349–352. [Google Scholar] [CrossRef]
- Chang, G.Q.; Gaysinskaya, V.; Karatayev, O.; Leibowitz, S.F. Maternal high-fat diet and fetal programming: Increased proliferation of hypothalamic peptide-producing neurons that increase risk for overeating and obesity. J. Neurosci. 2008, 28, 12107–12119. [Google Scholar] [CrossRef]
- Gali Ramamoorthy, T.; Begum, G.; Harno, E.; White, A. Developmental programming of hypothalamic neuronal circuits: Impact on energy balance control. Front. Neurosci. 2015, 9, 126. [Google Scholar] [CrossRef]
- Steculorum, S.M.; Collden, G.; Coupe, B.; Croizier, S.; Lockie, S.; Andrews, Z.B.; Jarosch, F.; Klussmann, S.; Bouret, S.G. Neonatal ghrelin programs development of hypothalamic feeding circuits. J. Clin. Investig. 2015, 125, 846–858. [Google Scholar] [CrossRef] [PubMed]
- Kawai, M.; Yamaguchi, M.; Murakami, T.; Shima, K.; Murata, Y.; Kishi, K. The placenta is not the main source of leptin production in pregnant rat: Gestational profile of leptin in plasma and adipose tissues. Biochem. Biophys. Res. Commun. 1997, 240, 798–802. [Google Scholar] [CrossRef] [PubMed]
- Kojima, M.; Hosoda, H.; Date, Y.; Nakazato, M.; Matsuo, H.; Kangawa, K. Ghrelin is a growth-hormone-releasing acylated peptide from stomach. Nature 1999, 402, 656–660. [Google Scholar] [CrossRef] [PubMed]
- Cripps, R.L.; Martin-Gronert, M.S.; Archer, Z.A.; Hales, C.N.; Mercer, J.G.; Ozanne, S.E. Programming of hypothalamic neuropeptide gene expression in rats by maternal dietary protein content during pregnancy and lactation. Clin. Sci. (Lond) 2009, 117, 85–93. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bouret, S.G.; Draper, S.J.; Simerly, R.B. Trophic action of leptin on hypothalamic neurons that regulate feeding. Science 2004, 304, 108–110. [Google Scholar] [CrossRef]
- Abegg, K.; Hermann, A.; Boyle, C.N.; Bouret, S.G.; Lutz, T.A.; Riediger, T. Involvement of amylin and leptin in the development of projections from the area postrema to the nucleus of the solitary tract. Front. Endocrinol. (Lausanne) 2017, 8, 324. [Google Scholar] [CrossRef]
- Pourié, G.; Martin, N.; Bossenmeyer-Pourié, C.; Akchiche, N.; Guéant-Rodriguez, R.M.; Geoffroy, A.; Jeannesson, E.; El Hajj Chehadeh, S.; Mimoun, K.; Brachet, P.; et al. Folate- and vitamin B12-deficient diet during gestation and lactation alters cerebellar synapsin expression via impaired influence of estrogen nuclear receptor α. FASEB J. 2015, 29, 3713–3725. [Google Scholar] [CrossRef]
- Derghal, A.; Djelloul, M.; Trouslard, J.; Mounien, L. The Role of MicroRNA in the modulation of the melanocortinergic system. Front. Neurosci. 2017, 11, 181. [Google Scholar] [CrossRef]
- Croizier, S.; Park, S.; Maillard, J.; Bouret, S.G. Central Dicer-miR-103/107 controls developmental switch of POMC progenitors into NPY neurons and impacts glucose homeostasis. Elife 2018, 7, e40429. [Google Scholar] [CrossRef]
- Yang, N.V.; Pannia, E.; Chatterjee, D.; Kubant, R.; Ho, M.; Hammoud, R.; Pausova, Z.; Anderson, G.H. Gestational folic acid content alters the development and function of hypothalamic food intake regulating neurons in Wistar rat offspring post-weaning. Nutr. Neurosci. 2018, 30, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Remmers, F.; Delemarre-van de Waal, H.A. Developmental programming of energy balance and its hypothalamic regulation. Endocr. Rev. 2011, 32, 272–311. [Google Scholar] [CrossRef] [PubMed]
- Dalvi, P.S.; Nazarians-Armavil, A.; Tung, S.; Belsham, D.D. Immortalized neurons for the study of hypothalamic function. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2011, 300, R1030–R1052. [Google Scholar] [CrossRef] [PubMed]
- Daval, J.L.; Blaise, S.; Guéant, J.L. Vitamin B deficiency causes neural cell loss and cognitive impairment in the developing rat. Proc. Natl. Acad. Sci. USA 2009, 106, E1. [Google Scholar] [CrossRef] [PubMed]
- Ducros, V.; Belva-Besnet, H.; Casetta, B.; Favier, A. A robust liquid chromatography tandem mass spectrometry method for total plasma homocysteine determination in clinical practice. Clin. Chem. Lab. Med. 2006, 44, 987–990. [Google Scholar] [CrossRef] [PubMed]
- Chery, C.; Barbe, F.; Lequere, C.; Abdelmouttaleb, I.; Gerard, P.; Barbarino, P.; Boutroy, J.L.; Gueant, J.L. Hyperhomocysteinemia is related to a decreased blood level of vitamin B12 in the second and third trimester of normal pregnancy. Clin. Chem. Lab. Med. 2002, 40, 1105–1108. [Google Scholar] [CrossRef] [PubMed]
- Paxinos, G.; Watson, C. The Rat Brain in Stereotaxic Coordinates, 7th ed.; Academic Press: London, UK, 2013; p. 472. [Google Scholar]



| Plasma Folate (nmol/L) | Plasma Vitamin B12 (pmol/L) | Plasma Homocysteine (µmol/L) | Body Weight (g) | Brain Weight (g) | |
|---|---|---|---|---|---|
| 21-d Control-vehicle | 53.1 ± 7.5 | 876.6 ± 94.1 | 6.7 ± 1.8 | 48.6 ± 3.2 | 1.43 ± 0.20 |
| 21-d MDD-vehicle | 23.8 ± 7.6 ** | 249.4 ± 211.6 ** | 23.3 ± 7.1 ** | 22.1 ± 2.0 ** | 1.01 ± 0.29 ** |
| 21-d Control + B9 | 103.2 ± 42.7 ** | 899.3 ± 134.6 | 4.2 ± 2.4 | 48.4 ± 3.7 | 1.51 ± 0.11 |
| 21-d MDD + B9 | 94.1 ± 46. 4 **/°° | 249.7 ± 102.2 ** | 4.5 ± 3.7 **/°° | 27.2 ± 3.1 **/°° | 1.32 ± 0.36 **/°° |
| Gene | Forward | Reverse | Species |
|---|---|---|---|
| Neuropeptide Y | AGATCCAGCCCTGAGACACT | TTCAAGCCTTGTTCTGGGGG | Rat |
| Neuropeptide Y receptor 1 | TGC-TAC-TTC-AAG-ATA-TAC-GTT-CGC | ACG-ATG-GAG-AGC-AGC-ATG-AC | Rat |
| Insulin receptor | GGA-CCA-GGC-ATC-CTG-TGA-AA | ATC-CTG-CCC-GTC-AAA-CTC-TG | Rat |
| Leptin receptor | CCC-CCA-CTG-AAA-GAC-AGC-TT | GGC-TTC-ACA-ACA-AGC-ATG-GG | Rat |
| Pro-opiomelanocortin | CGA-CGG-AGG-AGA-AAA-GAG-GTT | CTG-AGG-CTC-TGT-CGC-GGA-A | Rat |
| Insulin growth factor receptor 1 | AAG-GCC-AGA-GGT-GGA-GAA-TAA | TAC-CAT-GCA-GTT-CCG-AGC-AG | Rat |
| Ghrelin | CCA-AGA-AGC-CAC-CAG-CTA-AA | CTG-ATT-TCC-AGC-TCC-TCC-TC | Rat |
| Neuropeptide Y receptor 1 | Reference: qMmuCIP0029884 (BioRad, Marnes-la-Coquette, France) | Mouse | |
| Leptin receptor | Reference: qMmuCID0015266 (BioRad) | Mouse | |
| Agouti-related protein | CGG-AGG-TGC-TAG-ATC-CAC-AGA | AGG-ACT-CGT-GCA-GCC-TTA-CAC | Mouse |
| Insulin growth factor receptor 1 | GCA-CCA-ATG-CTT-CAG-TCC-CT | TTG-GAG-CAG-TAG-TTG-TGC-CG | Mouse |
| Insulin receptor | AGA-TGT-CCC-ATC-AAA-TAT-TGC-CA | CAT-CCG-GCT-GCC-TCT-TTC-TC | Mouse |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saber Cherif, L.; Pourié, G.; Geoffroy, A.; Julien, A.; Helle, D.; Robert, A.; Umoret, R.; Guéant, J.-L.; Bossenmeyer-Pourié, C.; Daval, J.-L. Methyl Donor Deficiency during Gestation and Lactation in the Rat Affects the Expression of Neuropeptides and Related Receptors in the Hypothalamus. Int. J. Mol. Sci. 2019, 20, 5097. https://doi.org/10.3390/ijms20205097
Saber Cherif L, Pourié G, Geoffroy A, Julien A, Helle D, Robert A, Umoret R, Guéant J-L, Bossenmeyer-Pourié C, Daval J-L. Methyl Donor Deficiency during Gestation and Lactation in the Rat Affects the Expression of Neuropeptides and Related Receptors in the Hypothalamus. International Journal of Molecular Sciences. 2019; 20(20):5097. https://doi.org/10.3390/ijms20205097
Chicago/Turabian StyleSaber Cherif, Lynda, Grégory Pourié, Andréa Geoffroy, Amélia Julien, Déborah Helle, Aurélie Robert, Rémy Umoret, Jean-Louis Guéant, Carine Bossenmeyer-Pourié, and Jean-Luc Daval. 2019. "Methyl Donor Deficiency during Gestation and Lactation in the Rat Affects the Expression of Neuropeptides and Related Receptors in the Hypothalamus" International Journal of Molecular Sciences 20, no. 20: 5097. https://doi.org/10.3390/ijms20205097
APA StyleSaber Cherif, L., Pourié, G., Geoffroy, A., Julien, A., Helle, D., Robert, A., Umoret, R., Guéant, J.-L., Bossenmeyer-Pourié, C., & Daval, J.-L. (2019). Methyl Donor Deficiency during Gestation and Lactation in the Rat Affects the Expression of Neuropeptides and Related Receptors in the Hypothalamus. International Journal of Molecular Sciences, 20(20), 5097. https://doi.org/10.3390/ijms20205097





