Fibrin-Hyaluronic Acid Hydrogel (RegenoGel) with Fibroblast Growth Factor-18 for In Vitro 3D Culture of Human and Bovine Nucleus Pulposus Cells
Abstract
1. Introduction
2. Results
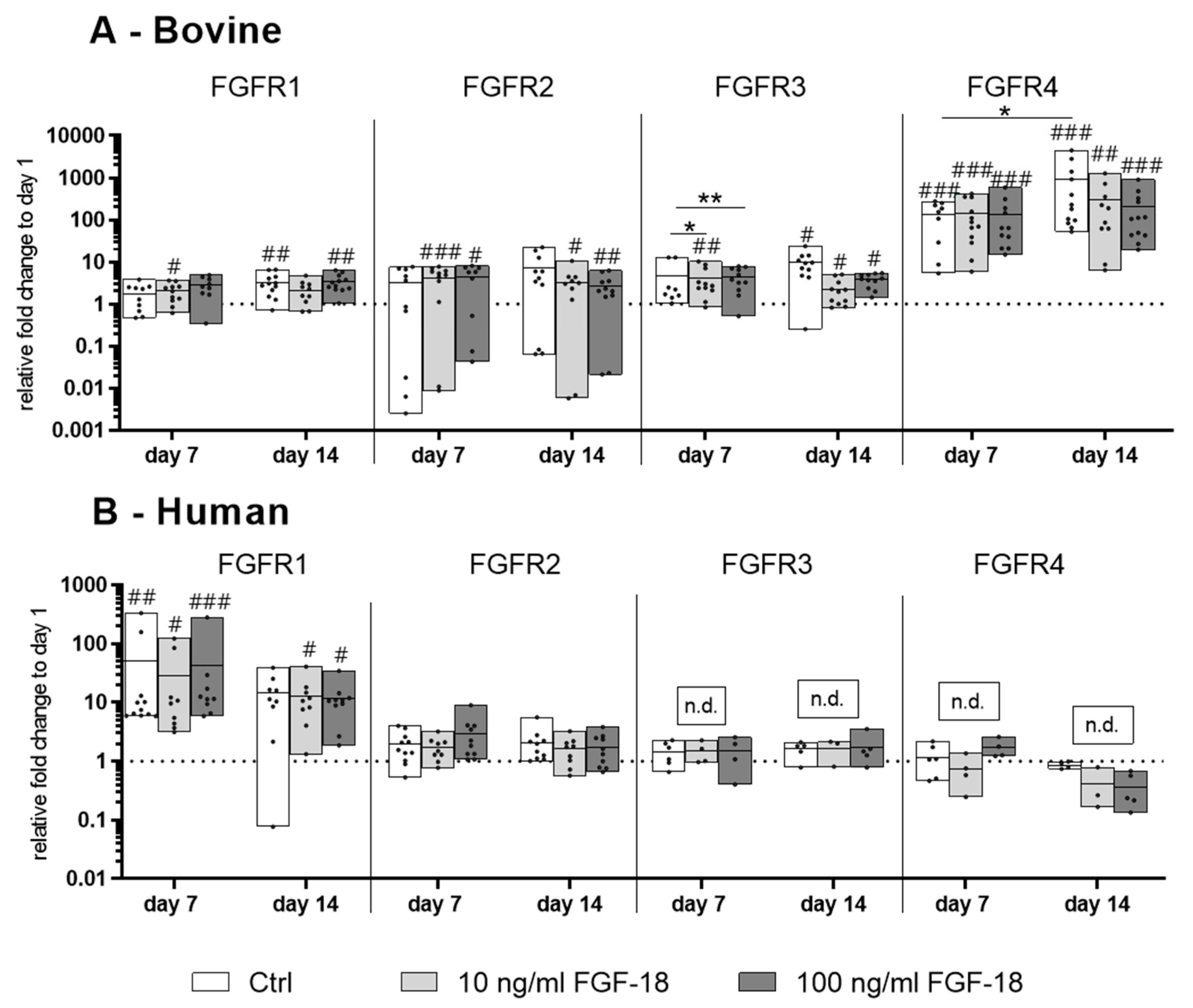
2.1. Expression of the FGF Receptors 1−4 in Bovine and Human NP Cells
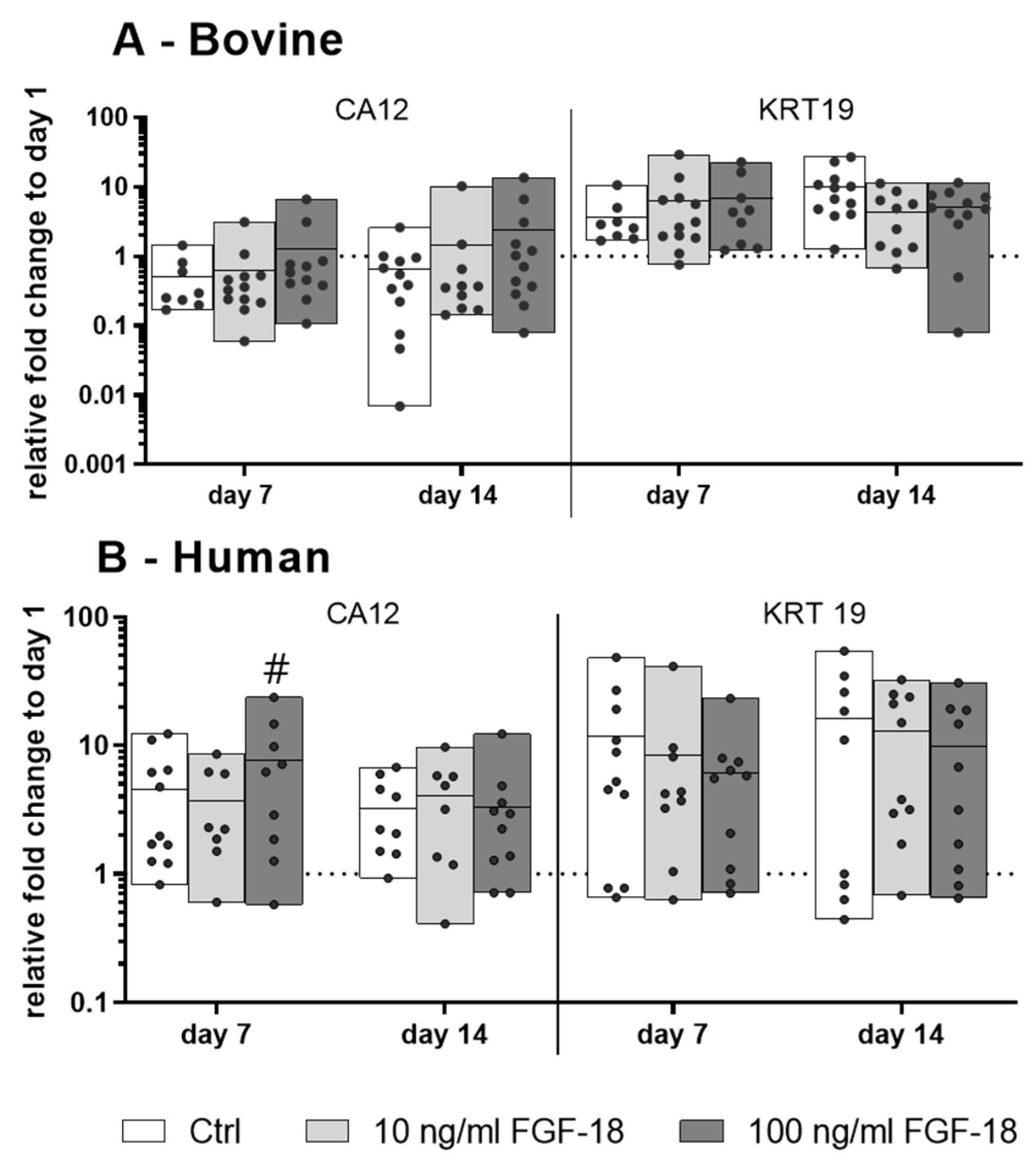
2.2. Expression of NP Cell Markers in Bovine and Human NP Cells
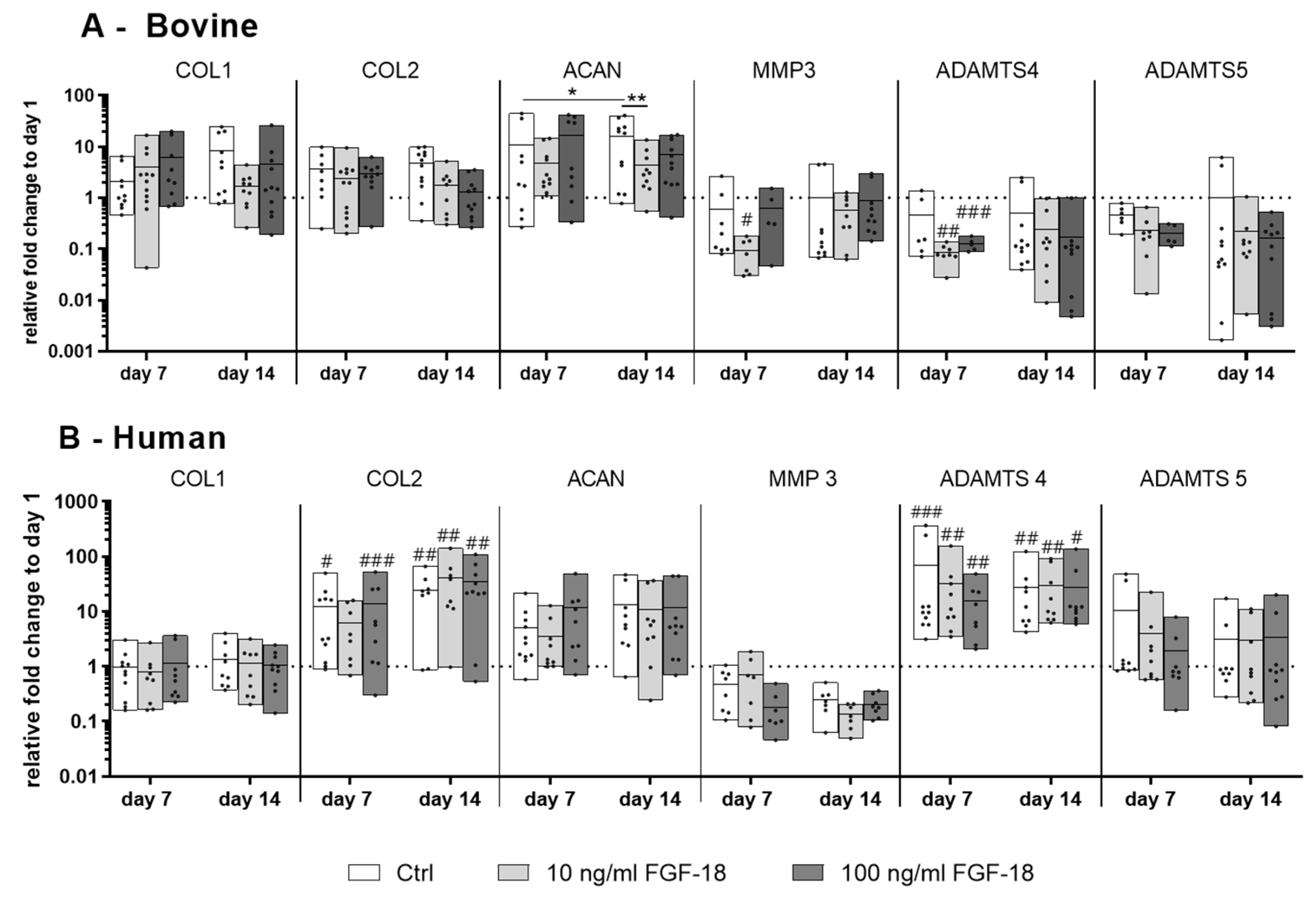
2.3. Gene Expression of IVD Matrix Molecule and Catabolic Markers in Bovine and Human NP Cells
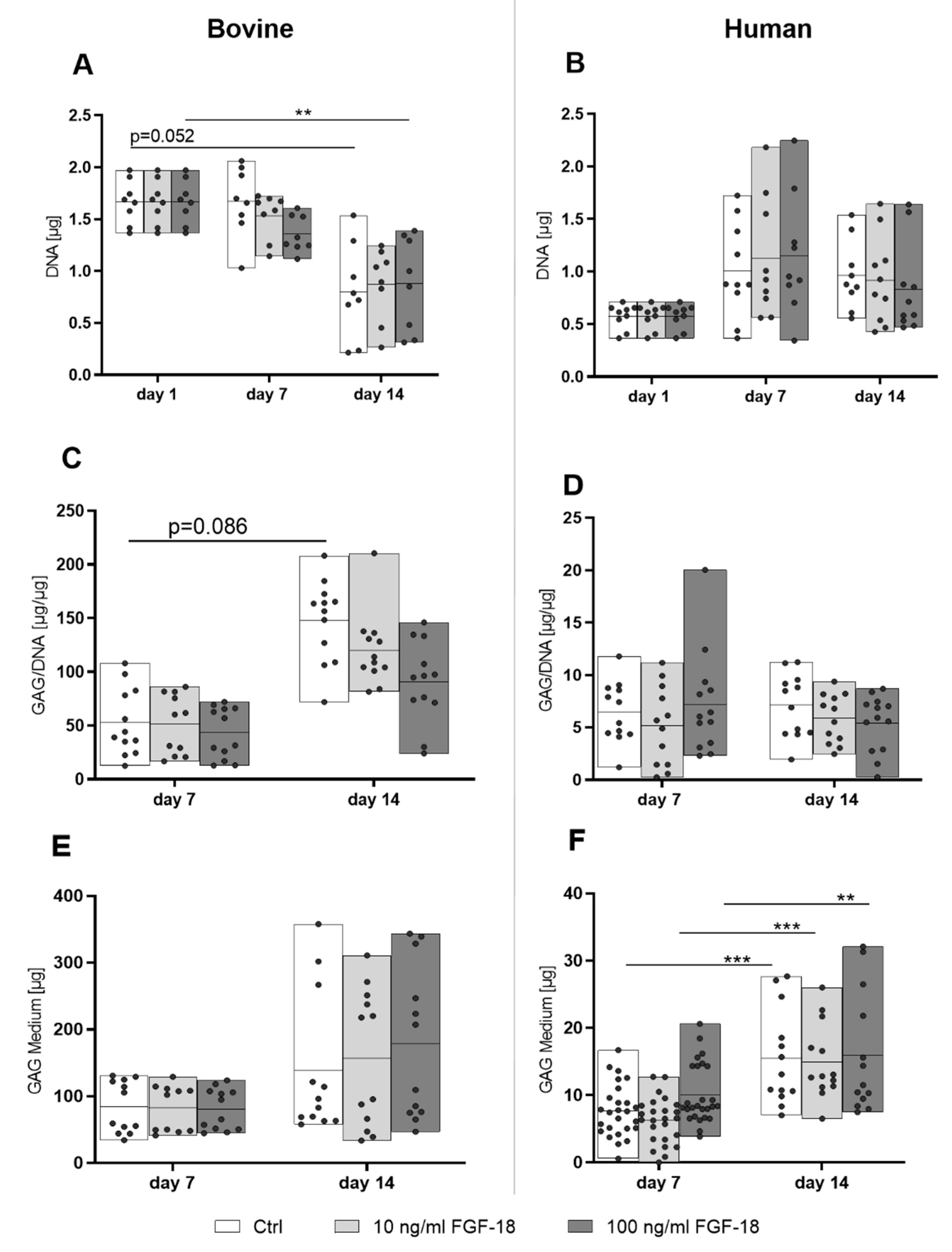
2.4. Proliferation and Matrix Protein Synthesis of Bovine and Human NP Cells
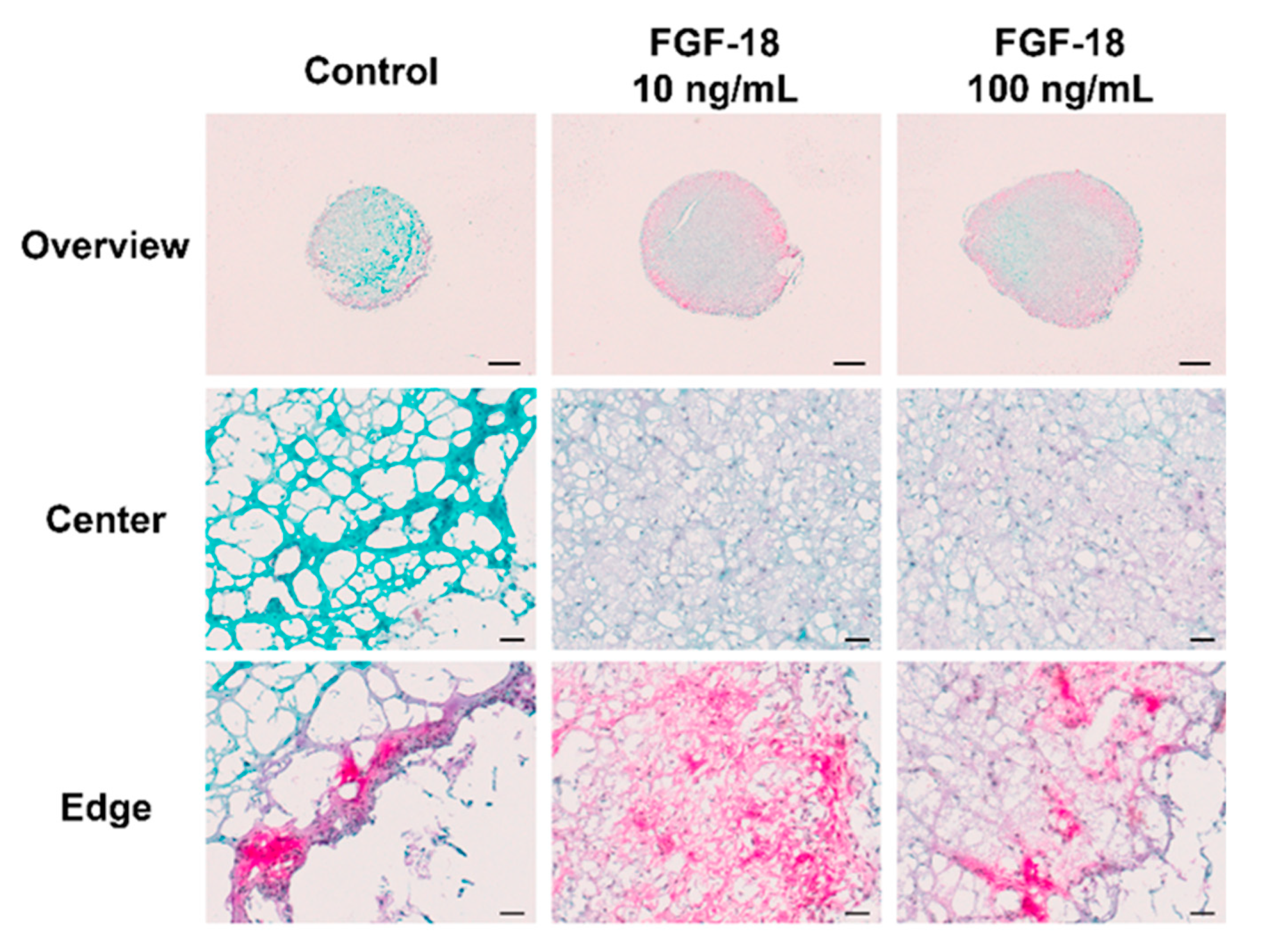
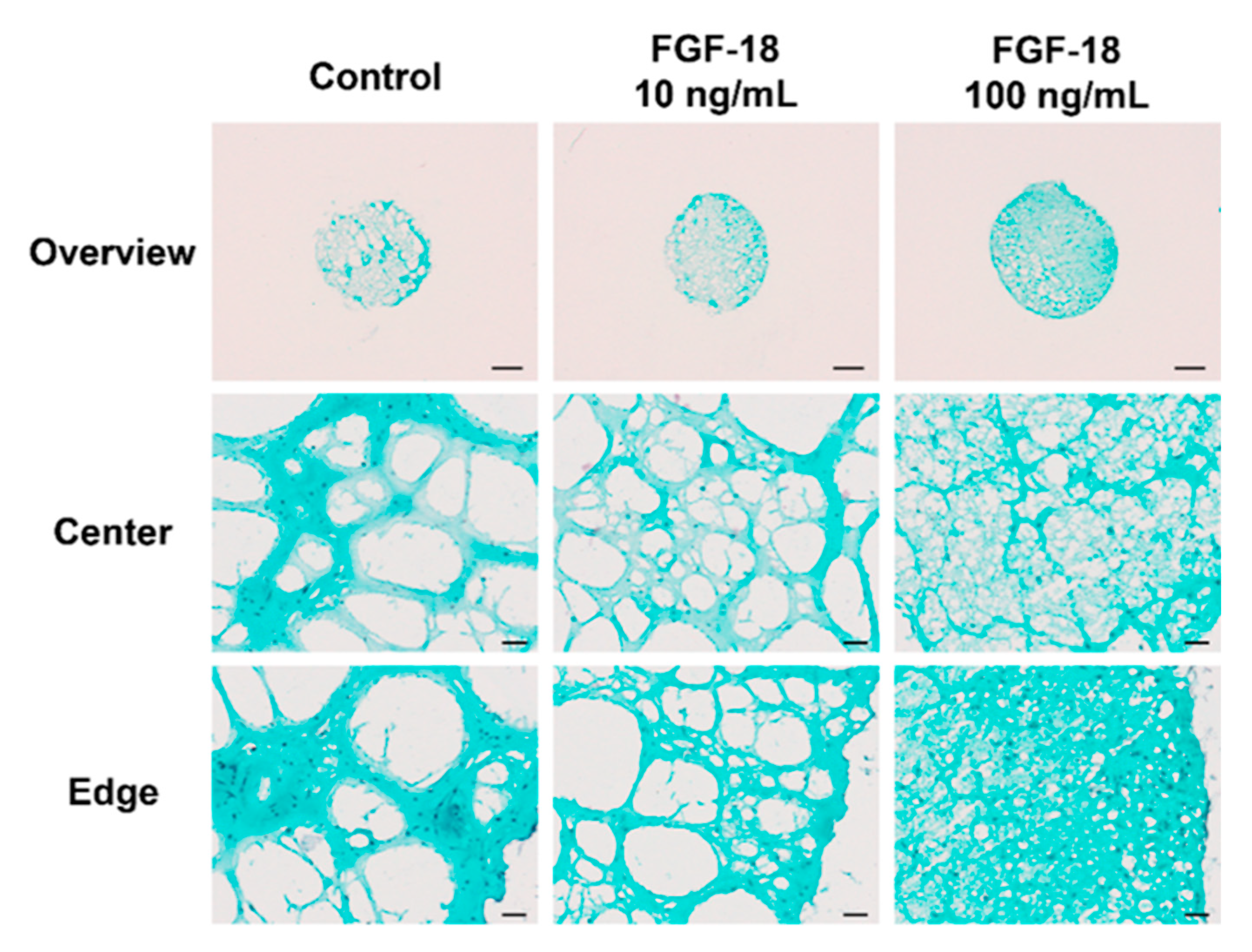
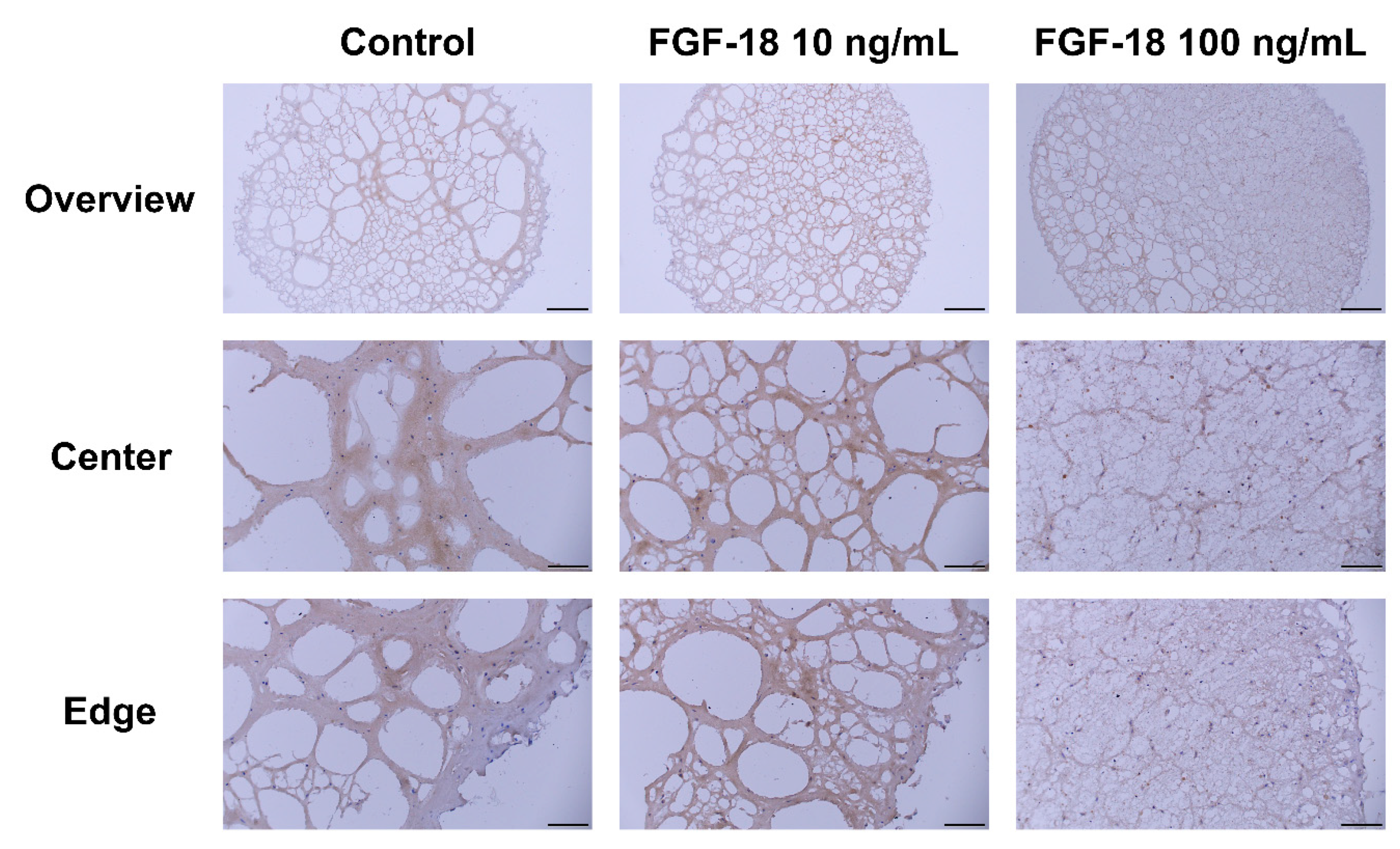
2.5. Histology Staining
3. Discussion
4. Materials and Methods
4.1. Materials and Reagents
4.2. Methods
4.2.1. Cell Isolation and In Vitro Culture in FBG-HA Hydrogel
4.2.2. Biochemical Analysis
4.2.3. Gene Expression Analysis
4.2.4. Safranin O/Fast Green Staining and Immunohistochemistry
4.2.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Richardson, S.M.; Freemont, A.J.; Hoyland, J.A. Pathogenesis of Intervertebral Disc Degeneration. In The Intervertebral Disc; Springer Vienna: Vienna, Austria, 2014; pp. 177–200. [Google Scholar]
- Buckwalter, J.A. Aging and degeneration of the human intervertebral disc. Spine (Phila. Pa. 1976) 1995, 20, 1307–1314. [Google Scholar] [CrossRef] [PubMed]
- Boden, S.; Davis, D.; Dina, T.; Patronas, N.; Wiesel, S. Abnormal MRI of the lumbar spine in asymptomatic subjects. J. Bone Jt. Surg. 1990, 72, 403–408. [Google Scholar] [CrossRef]
- McCann, M.; Séguin, C.; McCann, M.R.; Séguin, C.A. Notochord Cells in Intervertebral Disc Development and Degeneration. J. Dev. Biol. 2016, 4, 3. [Google Scholar] [CrossRef] [PubMed]
- Freemont, A.J.; Peacock, T.E.; Goupille, P.; Hoyland, J.A.; O’Brien, J.; Jayson, M.I. Nerve ingrowth into diseased intervertebral disc in chronic back pain. Lancet 1997, 350, 178–181. [Google Scholar] [CrossRef]
- Shapiro, I.M.; Risbud, M.V. The Intervertebral Disc: Molecular and Structural Studies of the Disc in Health and Disease; Shapiro, I.M., Risbud, M.V., Eds.; Springer Vienna: Vienna, Austria, 2014. [Google Scholar]
- Schwarzer, A.C.; Aprill, C.N.; Derby, R.; Fortin, J.; Kine, G.; Bogduk, N. The prevalence and clinical features of internal disc disruption in patients with chronic low back pain. Spine (Phila. Pa. 1976) 1995, 20, 1878–1883. [Google Scholar] [CrossRef] [PubMed]
- Masuda, K. Biological repair of the degenerated intervertebral disc by the injection of growth factors. Eur. Spine J. 2008, 17, 441. [Google Scholar] [CrossRef] [PubMed]
- Mwale, F. Molecular therapy for disk degeneration and pain. Glob. Spine J. 2013, 3, 185–192. [Google Scholar] [CrossRef]
- Kennon, J.C.; Awad, M.E.; Chutkan, N.; DeVine, J.; Fulzele, S.; Access, O.; Kennon, J.C.; Awad, M.E.; Chutkan, N.; DeVine, J.; et al. Current insights on use of growth factors as therapy for Intervertebral Disc Degeneration. Biomol. Concepts 2018, 9, 43–52. [Google Scholar] [CrossRef]
- Ohbayashi, N.; Hoshikawa, M.; Kimura, S.; Yamasaki, M.; Fukui, S.; Itoh, N. Structure and expression of the mRNA encoding a novel fibroblast growth factor, FGF-18. J. Biol. Chem. 1998, 273, 18161–18164. [Google Scholar] [CrossRef]
- Shimoaka, T.; Ogasawara, T.; Yonamine, A.; Chikazu, D.; Kawano, H.; Nakamura, K.; Itoh, N.; Kawaguchi, H. Regulation of osteoblast, chondrocyte, and osteoclast functions by fibroblast growth factor (FGF)-18 in comparison with FGF-2 and FGF-10. J. Biol. Chem. 2002, 277, 7493–7500. [Google Scholar] [CrossRef] [PubMed]
- Ellman, M.B.; Yan, D.; Ahmadinia, K.; Chen, D.; An, H.S.; Im, H.J. Fibroblast growth factor control of cartilage homeostasis. J. Cell. Biochem. 2013, 114, 735–742. [Google Scholar] [CrossRef] [PubMed]
- Gigout, A.; Guehring, H.; Froemel, D.; Meurer, A.; Ladel, C.; Reker, D.; Bay-Jensen, A.C.; Karsdal, M.A.; Lindemann, S. Sprifermin (rhFGF18) enables proliferation of chondrocytes producing a hyaline cartilage matrix. Osteoarthr. Cart. 2017, 25, 1858–1867. [Google Scholar] [CrossRef] [PubMed]
- Reker, D.; Kjelgaard-Petersen, C.F.; Siebuhr, A.S.; Michaelis, M.; Gigout, A.; Karsdal, M.A.; Ladel, C.; Bay-Jensen, A.C. Sprifermin (rhFGF18) modulates extracellular matrix turnover in cartilage explants ex vivo. J. Transl. Med. 2017, 15, 1–12. [Google Scholar] [CrossRef]
- Roemer, F.W.; Aydemir, A.; Lohmander, S.; Crema, M.D.; Marra, M.D.; Muurahainen, N.; Felson, D.T.; Eckstein, F.; Guermazi, A. Structural effects of sprifermin in knee osteoarthritis: A post-hoc analysis on cartilage and non-cartilaginous tissue alterations in a randomized controlled trial. Bmc Musculoskelet. Disord. 2016, 17, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Krouwels, A.; Melchels, F.; Van Rijen, M.H.P.; Oner, F.C.; Dhert, W.; Tryfonidou, M.; Creemers, L. Effects of the intradiscal implantation of stromal vascular fraction plus platelet rich plasma in patients with degenerative disc disease. J. Transl. Med. 2017, 15, 108. [Google Scholar]
- Rousseau, F.; Bonaventure, J.; Legeai-Mallet, L.; Pelet, A.; Rozet, J.-M.; Maroteaux, P.; Le Merrer, M.; Munnich, A. Mutations of the Fibroblast Growth Factor Receptor-3 Gene in Achondroplasia. Horm. Res. 1996, 45, 108–110. [Google Scholar] [CrossRef] [PubMed]
- Klag, K.A.; Horton, W.A. Advances in treatment of achondroplasia and osteoarthritis. Hum. Mol. Genet. 2016, 25, R2–R8. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.H.; Du, H.; Niu, G.M.; Wang, J.; Weng, L.X. The fibroblast growth factor signaling pathway. Chin. J. Tissue Eng. Res. 2016, 20, 2255–2264. [Google Scholar]
- Horton, W.A.; Garofalo, S.; Lunstrum, G.P. FGFR3 signaling in achondroplasia: A review. Ecm J. 1998, 8, 83–87. [Google Scholar]
- Davidson, D.; Blanc, A.; Filion, D.; Wang, H.; Plut, P.; Pfeffer, G.; Buschmann, M.D.; Henderson, J.E. Fibroblast growth factor (FGF) 18 signals through FGF receptor 3 to promote chondrogenesis. J. Biol. Chem. 2005, 280, 20509–20515. [Google Scholar] [CrossRef]
- Yayon, A.; Moshav, S.; Sirkis, R.; Strauss-Ay Ali, D. Fgf-18 truncated variants having increased receptor specificity and uses thereof. WO 2008/081463, January 2007. [Google Scholar]
- Collin, E.C.; Grad, S.; Zeugolis, D.I.; Vinatier, C.S.; Clouet, J.R.; Guicheux, J.J.; Weiss, P.; Alini, M.; Pandit, A.S. An injectable vehicle for nucleus pulposus cell-based therapy. Biomaterials 2011, 32, 2862–2870. [Google Scholar] [CrossRef]
- Isa, I.L.M.; Srivastava, A.; Tiernan, D.; Owens, P.; Rooney, P.; Dockery, P.; Pandit, A. Hyaluronic acid based hydrogels attenuate inflammatory receptors and neurotrophins in interleukin-1β induced inflammation model of nucleus pulposus cells. Biomacromolecules 2015, 16, 1714–1725. [Google Scholar] [CrossRef]
- Kazezian, Z.; Li, Z.; Alini, M.; Grad, S.; Pandit, A. Injectable hyaluronic acid down-regulates interferon signaling molecules, IGFBP3 and IFIT3 in the bovine intervertebral disc. Acta Biomater. 2017, 52, 118–129. [Google Scholar] [CrossRef]
- Liu, M.; Zeng, X.; Ma, C.; Yi, H.; Ali, Z.; Mou, X.; Li, S.; Deng, Y.; He, N. Injectable hydrogels for cartilage and bone tissue engineering. Bone Res. 2017, 5, 17014. [Google Scholar] [CrossRef] [PubMed]
- News - RegenoGel. Available online: https://regenogel.co.il/news/ (accessed on 2 December 2018).
- Lafont, J.E. Lack of oxygen in articular cartilage: Consequences for chondrocyte biology. Int. J. Exp. Pathol. 2010, 91, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Mwale, F.; Roughley, P.; Antoniou, J. Distinction between the extracellular matrix of the nucleus pulposus and hyaline cartilage: A requisite for tissue engineering of intervertebral disc. Eur. Cells Mater. 2004, 8, 58–64. [Google Scholar] [CrossRef]
- Stockwell, R.A. The cell density of human articular and costal cartilage. J. Anat. 1967, 101, 753–763. [Google Scholar]
- Lee, C.R.; Sakai, D.; Nakai, T.; Toyama, K.; Mochida, J.; Alini, M.; Grad, S. A phenotypic comparison of intervertebral disc and articular cartilage cells in the rat. Eur. Spine J. 2007, 16, 2174–2185. [Google Scholar] [CrossRef]
- Li, Z.; Kaplan, K.M.; Peroglio, M.; Amit, B.; Alini, M.; Grad, S.; Yayon, A. Biomimetic fibrin – hyaluronan hydrogels for nucleus pulposus regeneration. Regen. Med. 2014, 9, 309–326. [Google Scholar] [CrossRef]
- Garcia, J.P.; Stein, J.; Cai, Y.; Riemers, F.; Wexselblatt, E.; Wengel, J.; Tryfonidou, M.; Yayon, A.; Howard, K.A.; Creemers, L.B. Fibrin-hyaluronic acid hydrogel-based delivery of antisense oligonucleotides for ADAMTS5 inhibition in co-delivered and resident joint cells in osteoarthritis. J. Control. Release 2019, 294, 247–258. [Google Scholar] [CrossRef]
- Yue, S.; Li, J.; Lee, S.; Lee, H.J.; Shao, T.; Song, B.; Cheng, L.; Masterson, T.A.; Liu, X.; Ratliff, T.L. Cholesteryl ester accumulation induced by PTEN loss and PI3K/AKT activation underlies human prostate cancer aggressiveness. Cell Metabol. 2014, 19, 393–406. [Google Scholar] [CrossRef] [PubMed]
- Power, K.A.; Grad, S.; Rutges, J.P.H.J.; Creemers, L.B.; Van Rijen, M.H.P.; O’Gaora, P.; Wall, J.G.; Alini, M.; Pandit, A.; Gallagher, W.M. Identification of cell surface-specific markers to target human nucleus pulposus cells: Expression of carbonic anhydrase XII varies with age and degeneration. Arthritis Rheum. 2011, 63, 3876–3886. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues-Pinto, R.; Richardson, S.M.; Hoyland, J.A. Identification of novel nucleus pulposus markers. Bone Jt. Res. 2013, 2, 169–178. [Google Scholar] [CrossRef] [PubMed]
- Haberstroh, K.; Enz, A.; Zenclussen, M.L.; Hegewald, A.A.; Neumann, K.; Abbushi, A.; Thomé, C.; Sittinger, M.; Endres, M.; Kaps, C. Human intervertebral disc-derived cells are recruited by human serum and form nucleus pulposus-like tissue upon stimulation with TGF-β3 or hyaluronan in vitro. Tissue Cell 2009, 41, 414–420. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H.; Martin, J.T.; Elliott, D.M.; Smith, L.J.; Mauck, R.L. Phenotypic stability, matrix elaboration and functional maturation of nucleus pulposus cells encapsulated in photocrosslinkable hyaluronic acid hydrogels. Acta Biomater. 2015, 12, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Vo, N.V.; Hartman, R.A.; Yurube, T.; Jacobs, L.J.; Sowa, G.A.; Kang, J.D. Expression and regulation of metalloproteinases and their inhibitors in intervertebral disc aging and degeneration. Spine J. 2013, 13, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Sztrolovics, R.; Alini, M.; Roughley, P.J.; Mort, J.S. Aggrecan degradation in human intervertebral disc and articular cartilage. Biochem. J. 1997, 326, 235–241. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; An, H.S.; Ellman, M.; Phillips, F.; Thonar, E.J.; Park, D.K.; Udayakumar, R.K.; Im, H.-J. Action of fibroblast growth factor-2 on the intervertebral disc. Arthritis Res. 2008, 10, R48. [Google Scholar] [CrossRef] [PubMed]
- Yan, D.; Chen, D.; Cool, S.M.; van Wijnen, A.J.; Mikecz, K.; Murphy, G.; Im, H.-J. Fibroblast growth factor receptor 1 is principally responsible for fibroblast growth factor 2-induced catabolic activities in human articular chondrocytes. Arthritis Res. 2011, 13, R130. [Google Scholar] [CrossRef]
- Dailey, L.; Ambrosetti, D.; Mansukhani, A.; Basilico, C. Mechanisms underlying differential responses to FGF signaling. Cytokine Growth Factor Rev. 2005, 16, 233–247. [Google Scholar] [CrossRef]
- Kobayashi, S.; Meir, A.; Urban, J. Effect of cell density on the rate of glycosaminoglycan accumulation by disc and cartilage cells in vitro. J. Orthop. Res. 2008, 26, 493–503. [Google Scholar] [CrossRef]
- Chong, K.W.; Chanalaris, A.; Burleigh, A.; Jin, H.; Watt, F.E.; Saklatvala, J.; Vincent, T.L. Fibroblast growth factor 2 drives changes in gene expression following injury to murine cartilage in vitro and in vivo. Arthritis Rheum. 2013, 65, 2346–2355. [Google Scholar] [CrossRef]
- Lolli, A.; Sivasubramaniyan, K.; Vainieri, M.L.; Oieni, J.; Kops, N.; Yayon, A.; van Osch, G.J.V.M. Hydrogel-based delivery of antimiR-221 enhances cartilage regeneration by endogenous cells. J. Control. Release 2019, 309, 220–230. [Google Scholar] [CrossRef]
- Pfirrmann, C.W.A.; Metzdorf, A.; Zanetti, M.; Hodler, J.; Boos, N. Magnetic resonance classification of lumbar intervertebral disc degeneration. Spine (Phila. Pa. 1976) 2001, 26, 1873–1878. [Google Scholar] [CrossRef]
- Antoniou, J.; Steffen, T.; Nelson, F.; Winterbottom, N.; Hollander, A.P.; Poole, R.A.; Aebi, M.; Alini, M. The human lumbar intervertebral disc: Evidence for changes in the biosynthesis and denaturation of the extracellular matrix with growth, maturation, ageing, and degeneration. J. Clin. Invest. 1996, 98, 996–1003. [Google Scholar] [CrossRef]
- Farndale, R.W.; Buttle, D.J.; Barrett, A.J. Improved quantitation and discrimination of sulphated glycosaminoglycans by use of dimethylmethylene blue. Biochim. Biophys. Acta 1986, 883, 173–177. [Google Scholar] [CrossRef]
- Li, Z.; Lang, G.; Karfeld-Sulzer, L.S.; Mader, K.T.; Richards, R.G.; Weber, F.E.; Sammon, C.; Sacks, H.; Yayon, A.; Alini, M.; et al. Heterodimeric BMP-2/7 for nucleus pulposus regeneration—In vitro and ex vivo studies. J. Orthop. Res. 2017, 35, 51–60. [Google Scholar] [CrossRef]

| Gene | Primer/Probe Type | Sequence |
|---|---|---|
| bCOL1 | Primer fw (5′–3′) | TGC AGT AAC TTC GTG CCT AGC A |
| Primer rev (5′–3′) | CGC GTG GTC CTC TAT CTC CA | |
| Probe (5′FAM/3′TAMRA) | CAT GCC AAT CCT TAC AAG AGG CAA CTG C | |
| bCOL2 | Primer fw (5′–3′) | AAG AAA CAC ATC TGG TTT GGA GAA A |
| Primer rev (5′–3′) | TGG GAG CCA GGT TGT CAT C | |
| Probe (5′FAM/3′TAMRA) | CAA CGG TGG CTT CCA CTT CAG CTA TGG | |
| bACAN | Primer fw (5′–3′) | CCA ACG AAA CCT ATG ACG TGT ACT |
| Primer rev (5′–3′) | GCA CTC GTT GGC TGC CTC | |
| Probe (5′FAM/3′TAMRA) | ATG TTG CAT AGA AGA CCT CGC CCT CCA T | |
| bMMP3 | Primer fw (5′–3′) | GGC TGC AAG GGA CAA GGA A |
| Primer rev (5′–3′) | CAA ACT GTT TCG TAT CCT TTG CAA | |
| Probe (5′FAM/3′TAMRA) | CAC CAT GGA GCT TGT TCA GCA ATA TCT AGA AAA C | |
| bADAMTS4 | Primer fw (5′–3′) | CCC CAT GTG CAA CGT CAA G |
| Primer rev (5′–3′) | AGT CTC CAC AAA TCT GCT CAG TGA | |
| Probe (5′FAM/3′TAMRA) | AGC CCC CGA AGG GCT AAG CGC | |
| bADAMTS5 | Primer fw (5′–3′) | GAT GGT CAC GGT AAC TGT TTG CT |
| Primer rev (5′–3′) | GCC GGG ACA CAC CGA GTA C | |
| Probe (5′FAM/3′TAMRA) | AGG CCA GAC CTA CGA TGC CAG CC | |
| bCA12 | Primer fw (5′–3′) | CTG CCA GTC CGC AAT TTG T |
| Primer rev (5′–3′) | CCC CGG ACC TGC ATG TC | |
| Probe (5′FAM/3′TAMRA) | CCA CTC AGT GAA GGT GAA ACT GCC CA | |
| bKRT19 | Bt03219428_m1 | |
| bFGFR1 | Bt03258913_m1 | |
| bFGFR2 | Bt03649230_m1 | |
| bFGFR3 | Bt03259318_m1 | |
| bFGFR4 | Bt04292959_m1 | |
| bRPLP0 | Bt03218086_m1 | |
| hCOL1 | Primer fw (5′–3′) | CCC TGG AAA GAA TGG AGA TGA T |
| Primer rev (5′–3′) | ACT GAA ACC TCT GTG TCC CTT CA | |
| Probe (5′FAM/3′TAMRA) | CGG GCA ATC CTC GAG CAC CCT | |
| hCOL2 | Primer fw (5′–3′) | GGC AAT AGC AGG TTC ACG TAC A |
| Primer rev (5′–3′) | GAT AAC AGT CTT GCC CCA CTT ACC | |
| Probe (5′FAM/3′TAMRA) | CCT GAA GGA TGG CTG CAC GAA ACA TAC | |
| hACAN | Primer fw (5′–3′) | AGT CCT CAA GCC TCC TGT ACT CA |
| Primer rev (5′–3′) | CGG GAA GTG GCG GTA ACA | |
| Probe (5′FAM/3′TAMRA) | CCG GAA TGG AAA CGT GAA TCA GAA TCA ACT | |
| hMMP3 | Hs00968305_m1 | |
| hADAMTS4 | Hs00192708_m1 | |
| hADAMTS5 | Hs01095518_m1 | |
| hKRT19 | Hs00761767_s1 | |
| hFGFR1 | Hs00241111_m1 | |
| hFGFR2 | Hs01552918_m1 | |
| hFGFR3 | Hs00179829_m1 | |
| hFGFR4 | Hs01106910_g1 | |
| hRPLP0 | Primer fw (5′–3′) | TGG GCA AGA ACA CCA TGA TG |
| Primer rev (5′–3′) | CGG ATA TGA GGC AGC AGT TTC | |
| Probe (5′FAM/3′TAMRA) | AGG GCA CCT GGA AAA CAA CCC AGC |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Häckel, S.; Zolfaghar, M.; Du, J.; Hoppe, S.; Benneker, L.M.; Garstka, N.; Peroglio, M.; Alini, M.; Grad, S.; Yayon, A.; et al. Fibrin-Hyaluronic Acid Hydrogel (RegenoGel) with Fibroblast Growth Factor-18 for In Vitro 3D Culture of Human and Bovine Nucleus Pulposus Cells. Int. J. Mol. Sci. 2019, 20, 5036. https://doi.org/10.3390/ijms20205036
Häckel S, Zolfaghar M, Du J, Hoppe S, Benneker LM, Garstka N, Peroglio M, Alini M, Grad S, Yayon A, et al. Fibrin-Hyaluronic Acid Hydrogel (RegenoGel) with Fibroblast Growth Factor-18 for In Vitro 3D Culture of Human and Bovine Nucleus Pulposus Cells. International Journal of Molecular Sciences. 2019; 20(20):5036. https://doi.org/10.3390/ijms20205036
Chicago/Turabian StyleHäckel, Sonja, Mona Zolfaghar, Jie Du, Sven Hoppe, Lorin M. Benneker, Nathalie Garstka, Marianna Peroglio, Mauro Alini, Sibylle Grad, Avner Yayon, and et al. 2019. "Fibrin-Hyaluronic Acid Hydrogel (RegenoGel) with Fibroblast Growth Factor-18 for In Vitro 3D Culture of Human and Bovine Nucleus Pulposus Cells" International Journal of Molecular Sciences 20, no. 20: 5036. https://doi.org/10.3390/ijms20205036
APA StyleHäckel, S., Zolfaghar, M., Du, J., Hoppe, S., Benneker, L. M., Garstka, N., Peroglio, M., Alini, M., Grad, S., Yayon, A., & Li, Z. (2019). Fibrin-Hyaluronic Acid Hydrogel (RegenoGel) with Fibroblast Growth Factor-18 for In Vitro 3D Culture of Human and Bovine Nucleus Pulposus Cells. International Journal of Molecular Sciences, 20(20), 5036. https://doi.org/10.3390/ijms20205036






