Suitability of GnRH Receptors for Targeted Photodynamic Therapy in Head and Neck Cancers
Abstract
:1. Introduction
2. Results
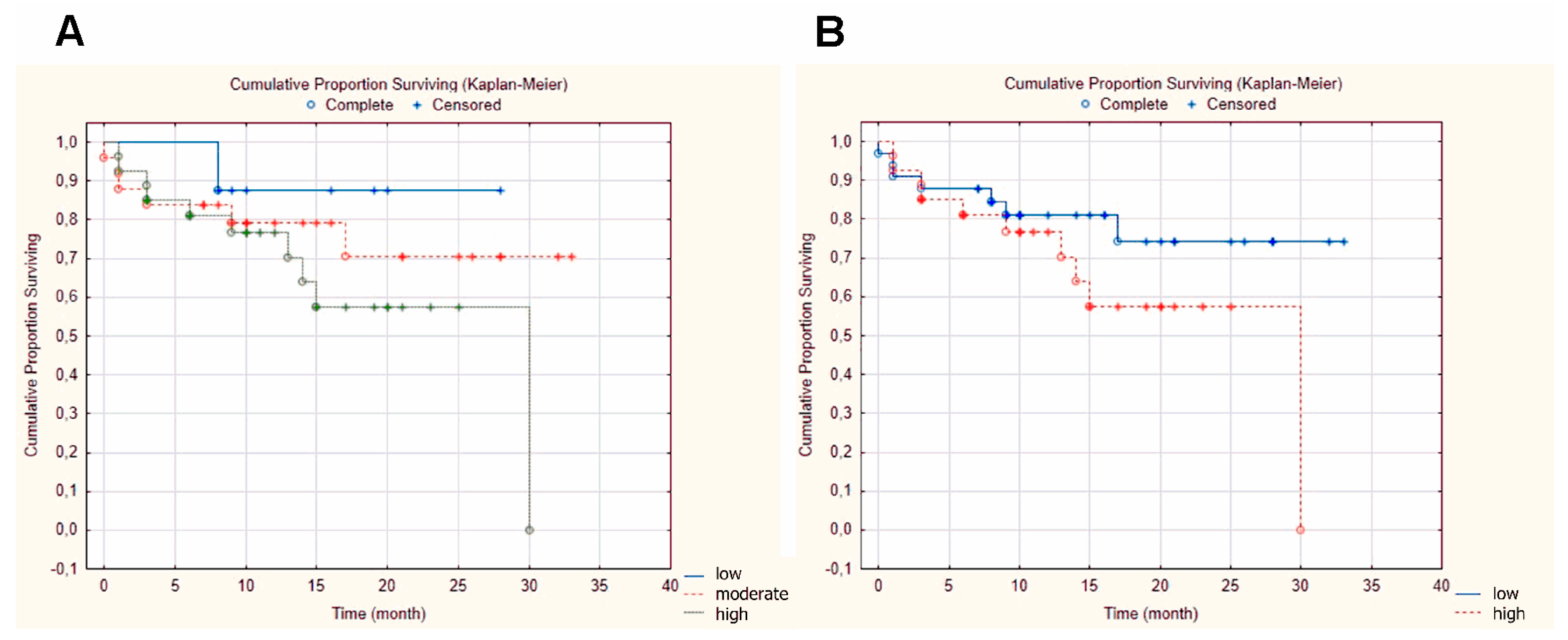
2.1. GnRH receptor (GnRH-R) Expression in Patient-Derived Head and Neck Squamous Cell Carcinoma (HNSCC) Samples
2.2. Peptide Synthesis
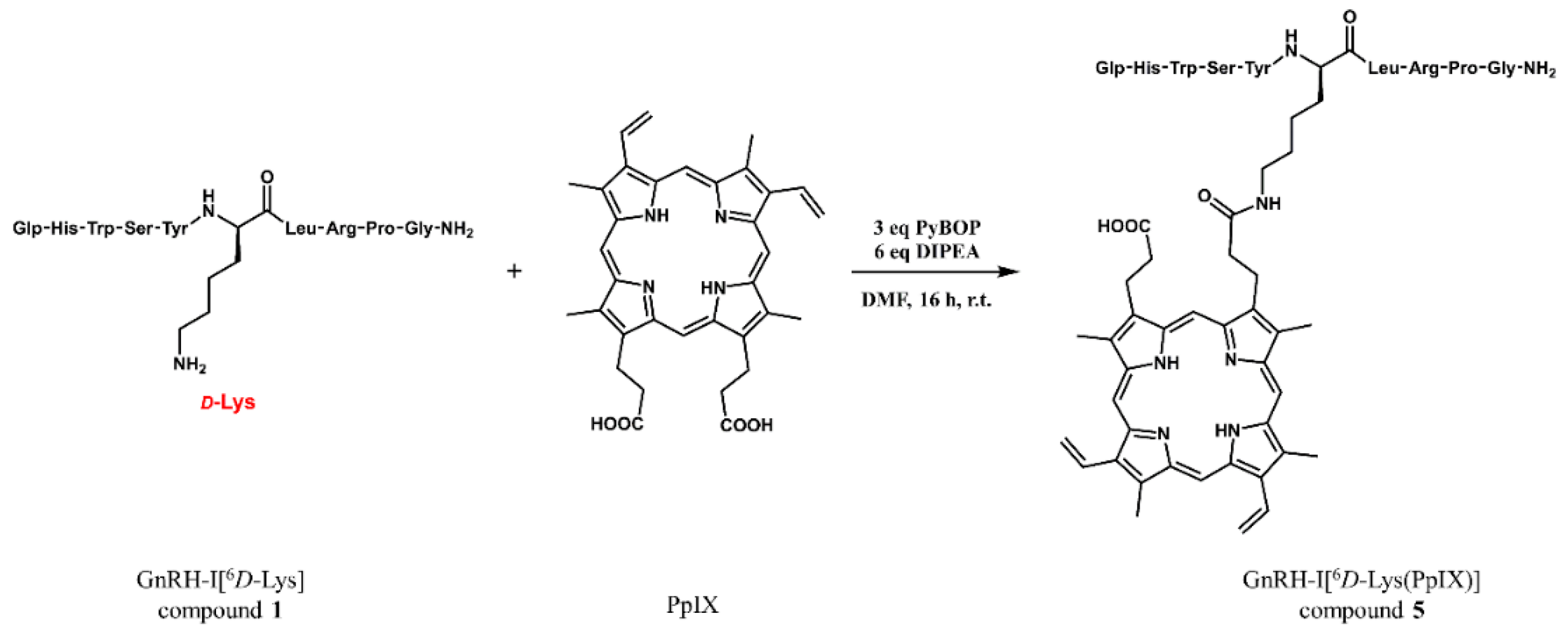
2.3. GnRH-Protoporphyrin IX Conjugate Synthesis
2.4. Receptor Binding Potency of the Novel Compounds
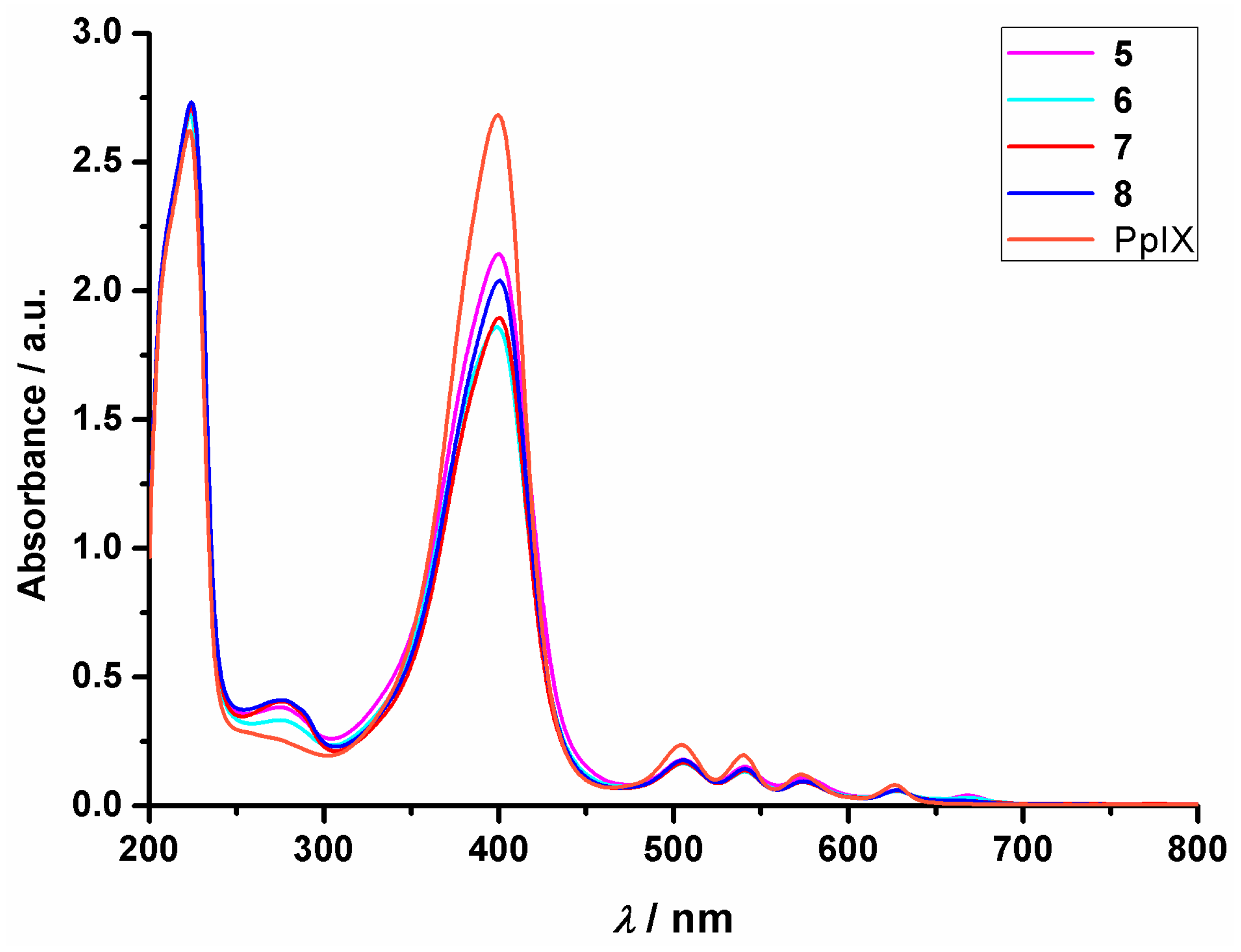
2.5. UV-Vis Absorbance
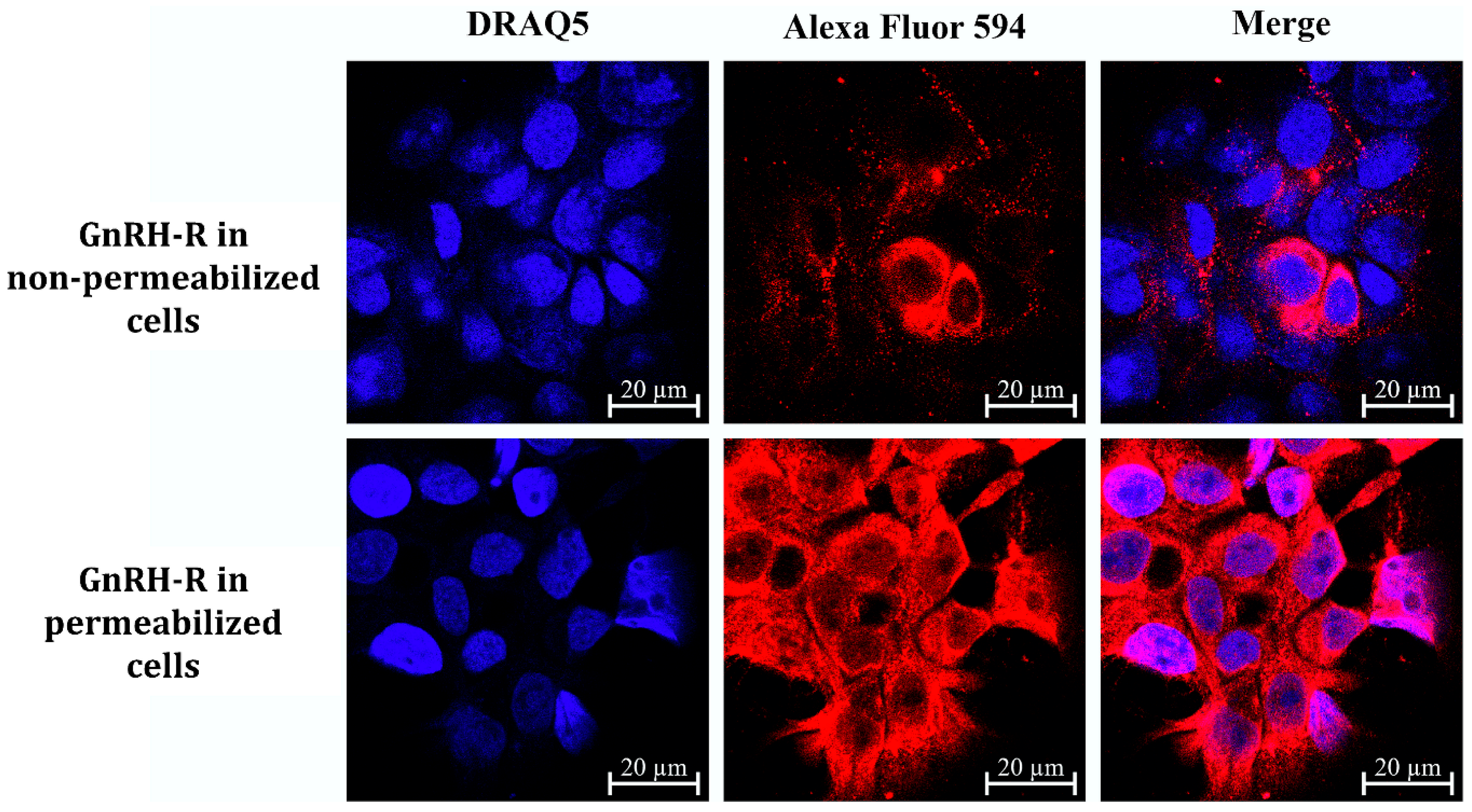
2.6. GnRH-R Expression of Detroit-562 Human Pharyngeal Carcinoma Cells
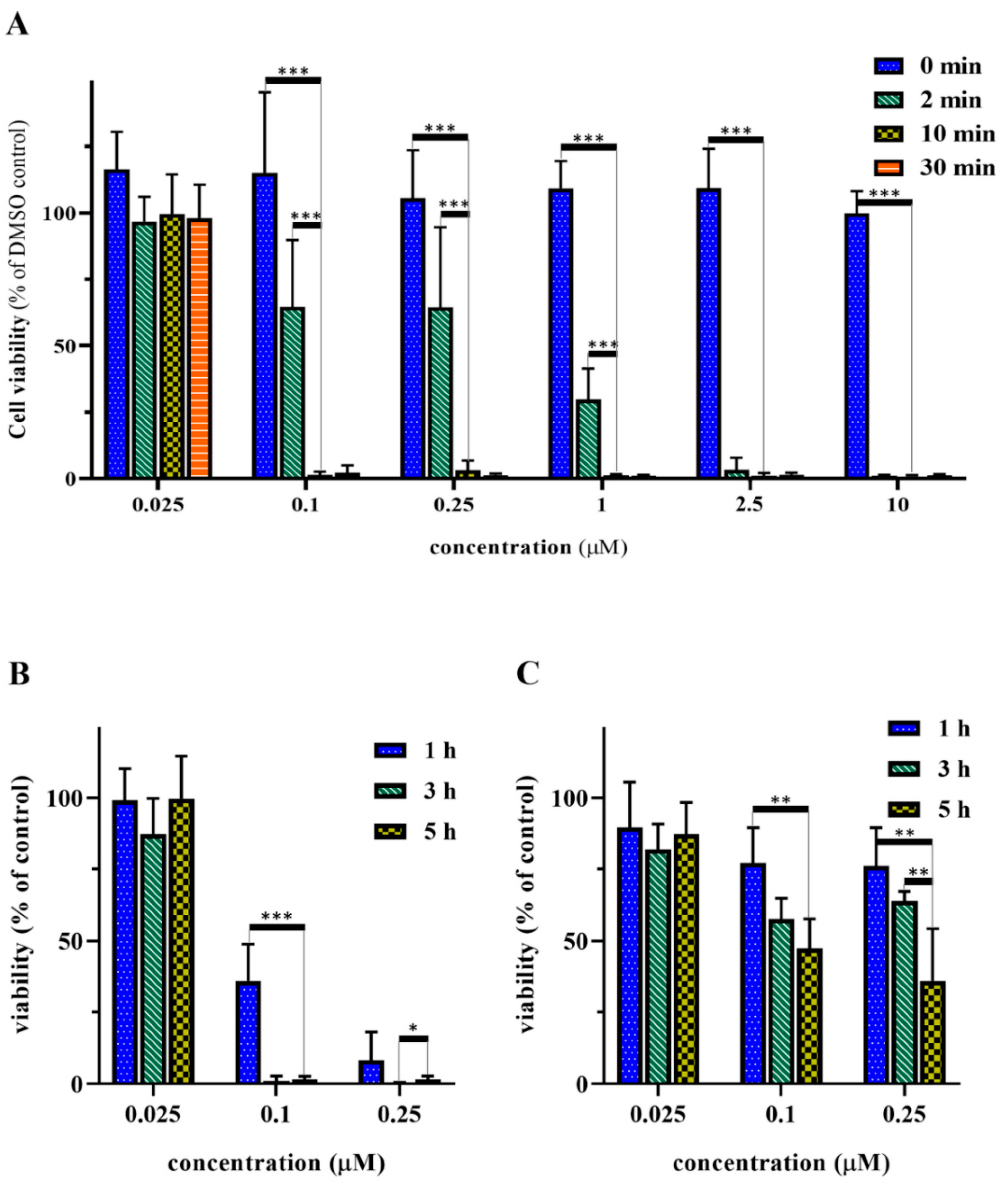
2.7. Optimization of the Irradiation and Incubation Time
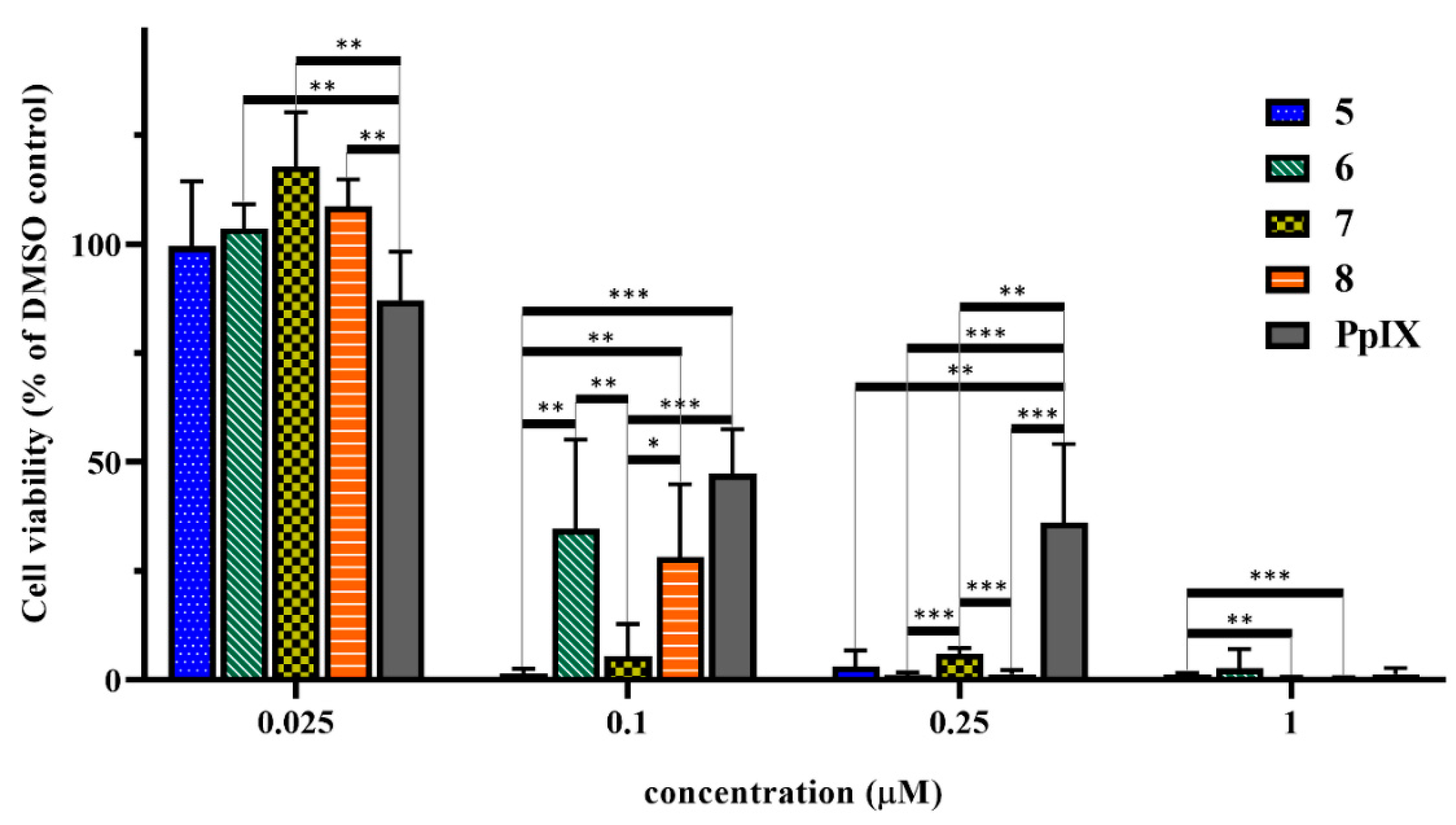
2.8. In Vitro Cell Viability Inhibition Effect of the Peptide-PpIX Conjugates
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Patients
4.3. Tissue Microarray (TMA) and Immunohistochemistry
4.4. Statistical Analysis
4.5. Peptide Synthesis
4.5.1. GnRH-I[6D-Lys] (Compound 1)
4.5.2. GnRH-I[4Lys(Bu),6D-Lys] (Compound 2) and GnRH-II[4Lys(Bu),6D-Lys] (Compound 3)
4.5.3. GnRH-III[4Lys(Bu),6Asp(OMe)] (Compound 4)
4.6. Conjugate Synthesis (Compounds 5–8)
4.7. RP-HPLC
4.8. Mass Spectrometry
4.9. Radioligand Binding Assay
4.10. UV-Vis Spectroscopy
4.11. Cell Cultures
4.12. Confocal Laser Scanning Microscopy
4.13. In Vitro Cell Viability Assay
4.14. Statistical Evaluation of In Vitro Viability Data
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ALA | δ-aminolevulinic acid |
| BSA | Bovine serum albumin |
| Dau | Daunorubicin |
| DBU | 1,8-Diazabicyclo[5.4.0]undec-7-ene |
| DCM | Dichloromethane |
| Dde | N-(1-(4,4-Dimethyl-2,6-dioxocyclohexylidene)ethyl) |
| DIC | N,N′-Diisopropylcarbodiimide |
| DIPEA | Diisopropylethylamine |
| DMF | N,N-Dimethylformamide |
| DSS | Disease-specific survival |
| <E | pyroglutamic acid |
| EGFR | Epidermal growth factor receptor |
| EMEM | Eagle’s Minimum Essential Medium |
| FBS | Fetal bovine serum |
| Fmoc | 9-Fluorenylmethyloxycarbonyl |
| FSH | Follicle stimulating hormone |
| GnRH | Gonadotropin-releasing hormone |
| GnRH-R | Gonadotropin-releasing hormone receptor |
| HOBt | 1-Hydroxybenzotriazole hydrate |
| LH | Luteinizing hormone |
| Mtt | 4-Methyltrityl |
| MTT | 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| NHSCC | Head and neck squamous cell carcinomas |
| PBS | Phosphate-buffered saline |
| PDT | Photodynamic therapy |
| PpIX | Protoporphyrin IX |
| PyBOP | Benzotriazol-1-yl-oxytripyrrolidinophosphonium hexafluorophosphate |
| TFA | Trifluoroacetic acid |
| TIS | Triisopropylsilane |
| TMA | Tissue Microarray |
References
- Dougherty, T.J.; Gomer, C.J.; Henderson, B.W.; Jori, G.; Kessel, D.; Korbelik, M.; Moan, J.; Peng, Q. Photodynamic Therapy. J. Natl. Cancer I 1998, 90, 889–905. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dolmans, D.E.J.G.J.; Fukumura, D.; Jain, R.K. Photodynamic therapy for cancer. Nat. Rev. Cancer 2003, 3, 380–387. [Google Scholar] [CrossRef] [PubMed]
- Agostinis, P.; Berg, K.; Cengel, K.A.; Foster, T.H.; Girotti, A.W.; Gollnick, S.O.; Hahn, S.M.; Hamblin, M.R.; Juzeniene, A.; Kessel, D.; et al. Photodynamic therapy of cancer: An update. Cancer J. Clin. 2011, 61, 250–281. [Google Scholar] [CrossRef] [PubMed]
- Nomoto, T.; Nishiyama, N. Photodynamic Therapy. In Photochemistry for Biomedical Applications; Ito, Y., Ed.; Springer: Singapore, 2018; pp. 301–313. [Google Scholar] [CrossRef]
- Williams, T.M.; Sibrian-Vazquez, M.; Vicente, M.G.H. Design and Synthesis of Photosensitizer-Peptide Conjugates for PDT. In Handbook of Photodynamic Therapy; Pandey, R.K., Kessel, D., Dougherty, T.J., Eds.; World Scientific: Singapore, 2016; pp. 45–93. [Google Scholar] [CrossRef]
- Nagy, A.; Schally, A.V. Targeting of cytotoxic luteinizing hormone-releasing hormone analogs to breast, ovarian, endometrial, and prostate cancers. Biol. Reprod. 2005, 73, 851–859. [Google Scholar] [CrossRef] [PubMed]
- Mező, G.; Manea, M.; Szabó, I.; Vincze, B.; Kovács, M. New derivatives of GnRH as potential anticancer therapeutic agents. Curr. Med. Chem. 2008, 15, 2366–2379. [Google Scholar] [CrossRef] [PubMed]
- White, R.B.; Eisen, J.A.; Kasten, T.L.; Fernald, R.D. Second gene for gonadotropin-releasing hormone in humans. Proc. Natl. Acad. Sci. USA 1998, 95, 305–309. [Google Scholar] [CrossRef] [PubMed]
- Millar, R.P. GnRHs and GnRH receptors. Anim. Reprod. Sci. 2005, 88, 5–28. [Google Scholar] [CrossRef] [PubMed]
- Millar, R.P. GnRH II and type II GnRH receptors. Trends Endocrinol. Metab. 2003, 14, 35–43. [Google Scholar] [CrossRef]
- Lovas, S.; Pályi, I.; Vincze, B.; Horváth, J.; Kovács, M.; Mezö, I.; Tóth, G.; Teplán, I.; Murphy, R.F. Direct anticancer activity of gonadotropin-releasing hormone-III. J. Pept. Res. 1998, 52, 384–389. [Google Scholar] [CrossRef] [PubMed]
- Janáky, T.; Juhász, A.; Bajusz, S.; Csernus, V.; Srkalovic, G.; Bokser, L.; Milovanovic, S.; Redding, T.W.; Rékási, Z.; Nagy, A. Analogues of luteinizing hormone-releasing hormone containing cytotoxic groups. Proc. Natl. Acad. Sci. USA 1992, 89, 972–976. [Google Scholar] [CrossRef] [PubMed]
- Szabó, I.; Bősze, S.; Orbán, E.; Sipos, É.; Halmos, G.; Kovács, M.; Mező, G. Comparative in vitro biological evaluation of daunorubicin containing GnRH-I and GnRH-II conjugates developed for tumor targeting. J. Pept. Sci. 2015, 21, 426–435. [Google Scholar] [CrossRef] [PubMed]
- Nagy, A.; Schally, A.V.; Armatis, P.; Szepesházi, K.; Halmos, G.; Kovács, M.; Zarándi, M.; Groot, K.; Miyazaki, M.; Jungwirth, A.; et al. Cytotoxic analogs of luteinizing hormone-releasing hormone containing doxorubicin or 2-pyrrolinodoxorubicin, a derivative 500–1000 times more potent. Proc. Natl. Acad. Sci. USA 1996, 93, 7269–7273. [Google Scholar] [CrossRef] [PubMed]
- Halmos, G.; Nagy, A.; Lamharzi, N.; Schally, A.V. Cytotoxic analogs of luteinizing hormone-releasing hormone bind with high affinity to human breast cancers. Cancer Lett. 1999, 136, 129–136. [Google Scholar] [CrossRef]
- Westphalen, S.; Kotulla, G.; Kaiser, F.; Krauss, W.; Werning, G.; Elsasser, H.P.; Nagy, A.; Schulz, K.D.; Grundker, C.; Schally, A.V.; et al. Receptor mediated antiproliferative effects of the cytotoxic LHRH agonist AN-152 in human ovarian and endometrial cancer cell lines. Int. J. Oncol. 2000, 17, 1063–1069. [Google Scholar] [CrossRef] [PubMed]
- Szepeshazi, K.; Schally, A.V.; Nagy, A. Effective treatment of advanced estrogen-independent MXT mouse mammary cancers with targeted cytotoxic LH-RH analogs. Breast Cancer Res. Treat. 1999, 56, 267–276. [Google Scholar] [CrossRef] [PubMed]
- Chatzistamou, I.; Schally, A.V.; Nagy, A.; Armatis, P.; Szepeshazi, K.; Halmos, G. Effective treatment of metastatic MDA-MB-435 human estrogen-independent breast carcinomas with a targeted cytotoxic analogue of luteinizing hormone-releasing hormone AN-207. Clin. Cancer Res. 2000, 6, 4158–4165. [Google Scholar] [PubMed]
- Gründker, C.; Völker, P.; Griesinger, F.; Ramaswamy, A.; Nagy, A.; Schally, A.V.; Emons, G. Antitumor effects of the cytotoxic luteinizing hormone-releasing hormone analog AN-152 on human endometrial and ovarian cancers xenografted into nude mice. Am. J. Obstet. Gynecol. 2002, 187, 528–537. [Google Scholar] [CrossRef]
- Bajo, A.M.; Schally, A.V.; Halmos, G.; Nagy, A. Targeted doxorubicin-containing luteinizing hormone-releasing hormone analogue AN-152 inhibits the growth of doxorubicin-resistant MX-1 human breast cancers. Clin. Cancer Res. 2003, 9, 3742–3748. [Google Scholar] [PubMed]
- Emons, G.; Kaufmann, M.; Gorchev, G.; Tsekova, V.; Gründker, C.; Günthert, A.R.; Hanker, L.C.; Velikova, M.; Sindermann, H.; Engel, J.; et al. Dose escalation and pharmacokinetic study of AEZS-108 (AN-152), an LHRH agonist linked to doxorubicin, in women with LHRH receptor-positive tumors. Gynecol. Oncol. 2010, 119, 457–461. [Google Scholar] [CrossRef]
- Engel, J.; Emons, G.; Pinski, J.; Schally, A.V. AEZS-108: A targeted cytotoxic analog of LHRH for the treatment of cancers positive for LHRH receptors. Expert Opin. Investig. Drugs 2012, 21, 891–899. [Google Scholar] [CrossRef]
- Miller, D.S.; Scambia, G.; Bondarenko, I.; Westermann, A.M.; Oaknin, A.; Oza, A.M. ZoptEC: Phase III randomized controlled study comparing zoptarelin with doxorubicin as second line therapy for locally advanced, recurrent, or metastatic endometrial cancer (NCT01767155). J. Clin. Oncol. 2018, 36, 5503. [Google Scholar] [CrossRef]
- Krebs, L.J.; Wang, X.; Nagy, A.; Schally, A.V.; Prasad, P.N.; Liebow, C. A conjugate of doxorubicin and an analog of luteinizing hormone-releasing hormone shows increased efficacy against oral and laryngeal cancers. Oral Oncol. 2002, 38, 657–663. [Google Scholar] [CrossRef]
- Rahimipour, S.; Ben-Aroya, N.; Ziv, K.; Chen, A.; Fridkin, M.; Koch, Y. Receptor-mediated targeting of a photosensitizer by its conjugation to gonadotropin-releasing hormone analogues. J. Med. Chem. 2003, 46, 3965–3974. [Google Scholar] [CrossRef] [PubMed]
- Lev-Goldman, V.; Mester, B.; Ben-Aroya, N.; Hanoch, T.; Rupp, B.; Stanoeva, T.; Gescheidt, G.; Seger, R.; Koch, Y.; Weiner, L.; et al. Conjugates of gonadotropin releasing hormone (GnRH) with carminic acid: Synthesis, generation of reactive oxygen species (ROS) and biological evaluation. Bioorg. Med. Chem. 2008, 16, 6789–6798. [Google Scholar] [CrossRef] [PubMed]
- Mező, G.; Czajlik, A.; Manea, M.; Jakab, A.; Farkas, V.; Majer, Z.; Vass, E.; Bodor, A.; Kapuvári, B.; Boldizsár, M.; et al. Structure, enzymatic stability and antitumor activity of sea lamprey GnRH-III and its dimer derivatives. Peptides 2007, 28, 806–820. [Google Scholar] [CrossRef] [PubMed]
- Kovács, M.; Vincze, B.; Horváth, J.E.; Seprődi, J. Structure-activity study on the LH- and FSH-releasing and anticancer effects of gonadotropin-releasing hormone (GnRH)-III analogs. Peptides 2007, 28, 821–829. [Google Scholar] [CrossRef] [PubMed]
- Mező, I.; Lovas, S.; Pályi, I.; Vincze, B.; Kálnay, A.; Turi, G.; Vadász, Z.; Seprődi, J.; Idei, M.; Tóth, G.; et al. Synthesis of gonadotropin-releasing hormone III analogs. Structure-antitumor activity relationships. J. Med. Chem. 1997, 40, 3353–3358. [Google Scholar] [CrossRef] [PubMed]
- Hegedüs, R.; Manea, M.; Orbán, E.; Szabó, I.; Kiss, É.; Sipos, É.; Halmos, G.; Mező, G. Enhanced cellular uptake and in vitro antitumor activity of short-chain fatty acid acylated daunorubicin-GnRH-III bioconjugates. Eur. J. Med. Chem. 2012, 56, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Kapuvári, B.; Hegedüs, R.; Schulcz, Á.; Manea, M.; Tóvári, J.; Gacs, A.; Vincze, B.; Mező, G. Improved in vivo antitumor effect of a daunorubicin—GnRH-III bioconjugate modified by apoptosis inducing agent butyric acid on colorectal carcinoma bearing mice. Investig. New Drugs 2016, 34, 416–423. [Google Scholar] [CrossRef] [PubMed]
- Sachar, M.; Anderson, K.E.; Ma, X. Phrotoporphyrin IX: The Good, the Bad, and the Ugly. J. Pharmacol. Exp. Ther. 2016, 356, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Azad, B.B.; Cho, C.F.; Lewis, J.D.; Luyt, L.G. Synthesis, radiometal labeling and in vitro evaluation of a targeted PPIX derivative. Appl. Radiat. Isot. 2012, 70, 505–511. [Google Scholar] [CrossRef] [PubMed]
- Eshghi, H.; Sazgarnia, A.; Rahimizadeh, M.; Attaran, N.; Bakavoli, M.; Soudmand, S. Protoporphyrin IX-gold nanoparticle conjugates as an efficient photosensitizer in cervical cancer therapy. Photodiagnosis Photodyn. Ther. 2013, 10, 304–312. [Google Scholar] [CrossRef] [PubMed]
- Ashjari, M.; Dehfuly, S.; Fatehi, D.; Shabani, R.; Koruji, M. Efficient functionalization of gold nanoparticles using cysteine conjugated protoporphyrin IX for singlet oxygen production in vitro. RSC Adv. 2015, 5, 104621–104628. [Google Scholar] [CrossRef]
- Ai, J.; Li, J.; Ga, L.; Yun, G.; Xu, L.; Wang, E. G-quadruplex/protoporphyrin IX-functionalized silver nanoconjugates for targeted cancer cell photodynamic therapy. RSC Adv. 2016, 6, 96942–96945. [Google Scholar] [CrossRef]
- Singh, S.; Jha, P.; Singh, V.; Sinha, K.; Hussain, S.; Singh, M.K.; Das, P. A quantum dot–MUC1 aptamer conjugate for targeted delivery of protoporphyrin IX and specific photokilling of cancer cells through ROS generation. Integr. Biol. 2016, 8, 1040–1048. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.S.; Aruna, P.; Ganesan, S. Influence of protoporphyrin IX loaded phloroglucinol succinic acid dendrimer in photodynamic therapy. Mater. Res. Express 2018, 5, 034004. [Google Scholar] [CrossRef]
- Kojima, C.; Toi, Y.; Harada, A.; Kono, K. Preparation of poly(ethylene glycol)-attached dendrimers encapsulating photosensitizers for application to photodynamic therapy. Bioconjug. Chem. 2007, 18, 663–670. [Google Scholar] [CrossRef]
- Murányi, J.; Gyulavári, P.; Varga, A.; Bökönyi, G.; Tanai, H.; Vántus, T.; Pap, D.; Ludányi, K.; Mező, G.; Kéri, G. Synthesis, characterization and systematic comparison of FITC-labelled GnRH-I, -II and -III analogues on various tumour cells. J. Pept. Sci. 2016, 22, 552–560. [Google Scholar] [CrossRef] [PubMed]
- Schuster, S.; Biri-Kovács, B.; Szeder, B.; Buday, L.; Gardi, J.; Szabó, Z.; Halmos, G.; Mező, G. Enhanced in vitro antitumor activity of GnRH-III-daunorubicin bioconjugates influenced by sequence modification. Pharmaceutics 2018, 10, 223. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Colombet, M.; Soerjomataram, I.; Mathers, C.; Parkin, D.; Piñeros, M.; Znaor, A.; Bray, F. Estimating the global cancer incidence and mortality in 2018: GLOBOCAN sources and methods. Int. J. Cancer 2019, 144, 1941–1953. [Google Scholar] [CrossRef] [PubMed]
- Grandis, J.R.; Tweardy, D.J. Elevated levels of transforming growth factor α and epidermal growth factor receptor messenger RNA are early markers of carcinogenesis in head and neck cancer. Cancer Res. 1993, 53, 3579–3584. [Google Scholar] [PubMed]
- Kuang, J.; Zhao, M.; Li, H.; Dang, W.; Li, W. Identification of potential therapeutic target genes and mechanisms in head and neck squamous cell carcinoma by bioinformatics analysis. Oncol. Lett. 2016, 11, 3009–3014. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brand, T.M.; Iida, M.; Wheeler, D.L. Molecular mechanisms of resistance to the EGFR monoclonal antibody cetuximab. Cancer Biol. Ther. 2011, 11, 777–792. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pappa, E.V.; Zompra, A.A.; Diamantopoulou, Z.; Spyranti, Z.; Pairas, G.; Lamari, F.N.; Katsoris, P.; Spyroulias, G.A.; Cordopatis, P. Structure–activity studies of lGnRH-III through rational amino acid substitution and NMR conformational studies. Biopolymers 2012, 98, 525–534. [Google Scholar] [CrossRef] [PubMed]
- Danos, K.; Brauswetter, D.; Birtalan, E.; Pato, A.; Bencsik, G.; Krenacs, T.; Petak, I.; Tamas, L. The Potential Prognostic Value of Connexin 43 Expression in Head and Neck Squamous Cell Carcinomas. Appl. Immunohisto. Mol. Morphol. 2016, 24, 476–481. [Google Scholar] [CrossRef] [PubMed]
- Cappuzzo, F.; Hirsch, F.R.; Rossi, E.; Bartolini, S.; Ceresoli, G.L.; Bemis, L.; Haney, J.; Witta, S.; Danenberg, K.; Domenichini, I.; et al. Epidermal growth factor receptor gene and protein and gefitinib sensitivity in non-small-cell lung cancer. J. Natl. Cancer I 2005, 97, 643–655. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, F.R.; Varella-Garcia, M.; Bunn, P.A.; Di Maria, M.V.; Veve, R.; Bremmes, R.M.; Baron, A.E.; Zeng, C.; Franklin, W.A. Epidermal growth factor receptor in non-small-cell lung carcinomas: Correlation between gene copy number and protein expression and impact on prognosis. J. Clin. Oncol. 2003, 21, 3798–3807. [Google Scholar] [CrossRef] [PubMed]
- Halmos, G.; Arencibia, J.M.; Schally, A.V.; Davis, R.; Bostwick, D.G. High incidence of receptors for luteinizing hormone-releasing hormone (LHRH) and LHRH receptor gene expression in human prostate cancers. J. Urol. 2000, 163, 623–629. [Google Scholar] [CrossRef]

| Code | Peptide | Code | Conjugate |
|---|---|---|---|
| 1 | GnRH-I[6D-Lys] | 5 | GnRH-I[6D-Lys(PpIX)] |
| 2 | GnRH-I[4Lys(Bu), 6D-Lys] | 6 | GnRH-I[4Lys(Bu), 6D-Lys(PpIX)] |
| 3 | GnRH-II[4Lys(Bu), 6D-Lys] | 7 | GnRH-II[4Lys(Bu), 6D-Lys(PpIX)] |
| 4 | GnRH-III[4Lys(Bu), 6Asp(OMe)] | 8 | GnRH-III[4Lys(Bu), 6Asp(OMe), 8Lys(PpIX)] |
| Code | Compound | IC50 Values/nM | |
|---|---|---|---|
| Human Pituitary | Human Prostate Cancer | ||
| 1 | GnRH-I[6D-Lys] | 6.44 ± 0.95 | 4.31 ± 0.83 |
| 2 | GnRH-I[4Lys(Bu), 6D-Lys] | 9.70 ± 1.07 | 8.61 ± 1.08 |
| 3 | GnRH-II[4Lys(Bu), 6D-Lys] | 5.56 ± 1.07 | 4.65 ± 0.31 |
| 4 | GnRH-III[4Lys(Bu), 6Asp(OMe)] | 7.27 ± 0.92 | 6.56 ± 1.11 |
| 5 | GnRH-I[6D-Lys(PpIX)] | 36.29 ± 3.17 | 42.67 ± 7.04 |
| 6 | GnRH-I[4Lys(Bu), 6D-Lys(PpIX)] | 42.30 ± 4.27 | 41.80 ± 5.74 |
| 7 | GnRH-II[4Lys(Bu), 6D-Lys(PpIX)] | 79.40 ± 8.88 | 84.50 ± 10.30 |
| 8 | GnRH-III[4Lys(Bu), 6Asp(OMe), 8Lys(PpIX)] | 288.6 ± 17.3 | 341.4 ± 21.1 |
| Code | Compound | IC50 Values/nM |
|---|---|---|
| 5 | GnRH-I[6D-Lys(PpIX)] | 62.3 ± 5.9 |
| 6 | GnRH-I[4Lys(Bu), 6D-Lys(PpIX)] | 89.8 ± 19.4 |
| 7 | GnRH-II[4Lys(Bu), 6D-Lys(PpIX)] | 71.1 ± 6.2 |
| 8 | GnRH-III[4Lys(Bu), 6Asp(OMe), 8Lys(PpIX)] | 81.8 ± 12.6 |
| PpIX | Protoporphyrin IX | 209.3 ± 119.5 |
| Variable | No. of Patients |
|---|---|
| Total no. of patients | 60 |
| Sex | |
| Male | 51 |
| Female | 9 |
| Age (year) | |
| Mean | 58.45 (41–77) |
| Localization | |
| Oropharynx | 19 |
| Larynx | 24 |
| Hypopharynx | 16 |
| Lingua | 1 |
| TNM 1 stage | |
| I | 6 |
| II | 11 |
| III | 15 |
| IV A | 20 |
| IV B | 3 |
| IV C | 5 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pethő, L.; Murányi, J.; Pénzes, K.; Gurbi, B.; Brauswetter, D.; Halmos, G.; Csík, G.; Mező, G. Suitability of GnRH Receptors for Targeted Photodynamic Therapy in Head and Neck Cancers. Int. J. Mol. Sci. 2019, 20, 5027. https://doi.org/10.3390/ijms20205027
Pethő L, Murányi J, Pénzes K, Gurbi B, Brauswetter D, Halmos G, Csík G, Mező G. Suitability of GnRH Receptors for Targeted Photodynamic Therapy in Head and Neck Cancers. International Journal of Molecular Sciences. 2019; 20(20):5027. https://doi.org/10.3390/ijms20205027
Chicago/Turabian StylePethő, Lilla, József Murányi, Kinga Pénzes, Bianka Gurbi, Diána Brauswetter, Gábor Halmos, Gabriella Csík, and Gábor Mező. 2019. "Suitability of GnRH Receptors for Targeted Photodynamic Therapy in Head and Neck Cancers" International Journal of Molecular Sciences 20, no. 20: 5027. https://doi.org/10.3390/ijms20205027
APA StylePethő, L., Murányi, J., Pénzes, K., Gurbi, B., Brauswetter, D., Halmos, G., Csík, G., & Mező, G. (2019). Suitability of GnRH Receptors for Targeted Photodynamic Therapy in Head and Neck Cancers. International Journal of Molecular Sciences, 20(20), 5027. https://doi.org/10.3390/ijms20205027









