Beneficial Effects of Neurotensin in Murine Model of Hapten-Induced Asthma
Abstract
:1. Introduction
2. Results
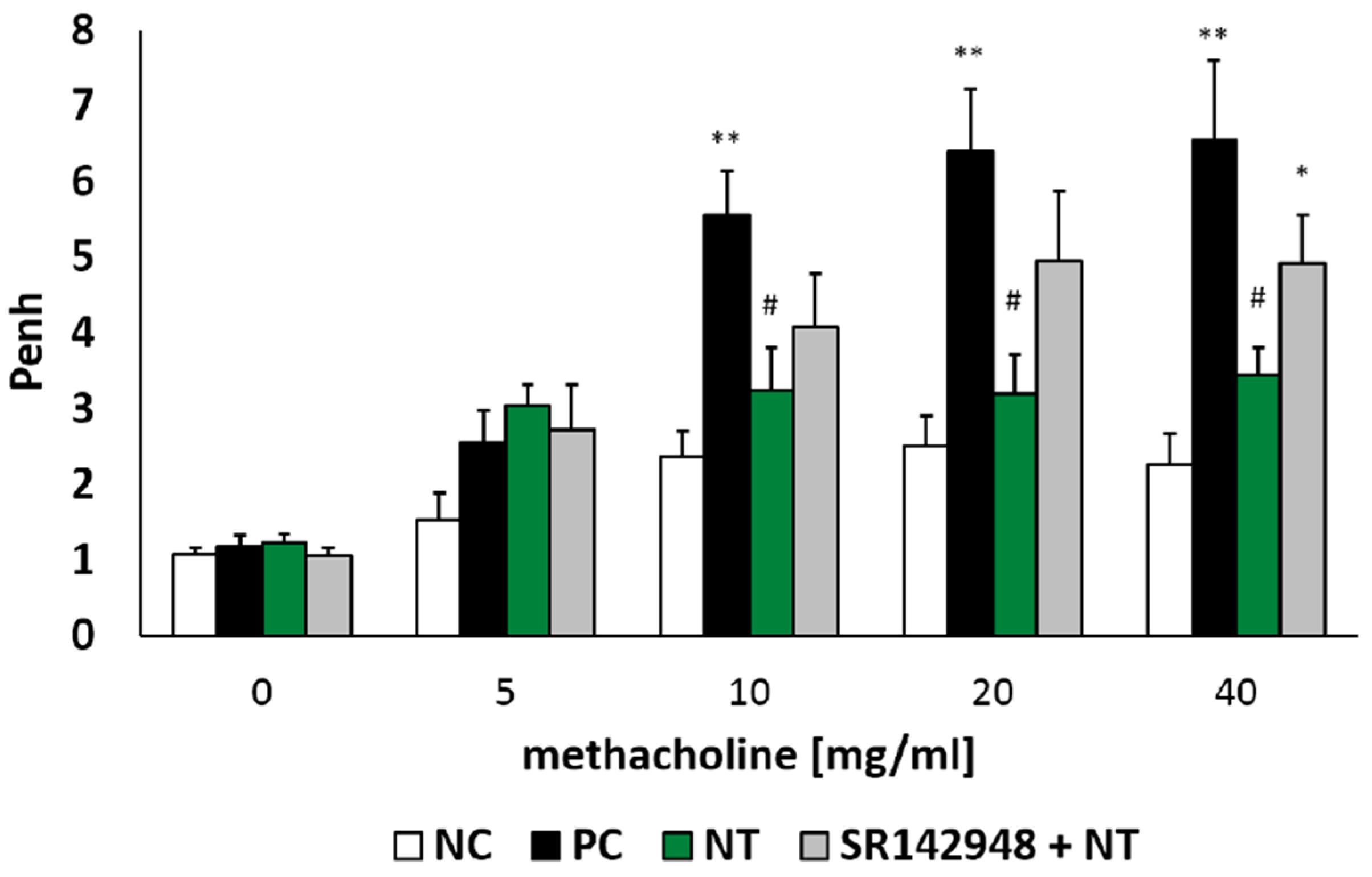
2.1. Airway Hyperresponsiveness (AHR)
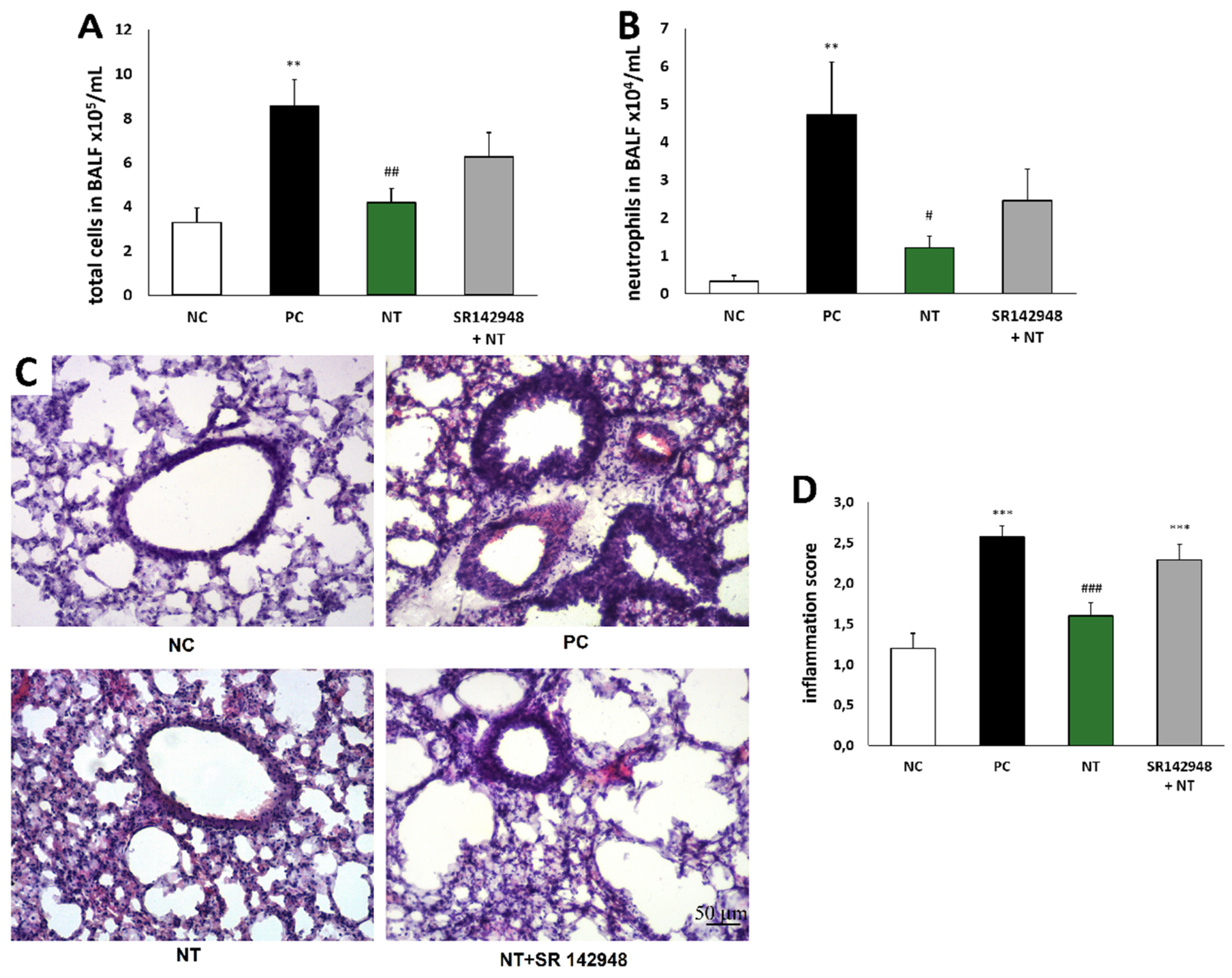
2.2. Leukocyte Accumulation in BALF and Lung Tissue
2.3. Pro-Inflammatory Cytokine Production
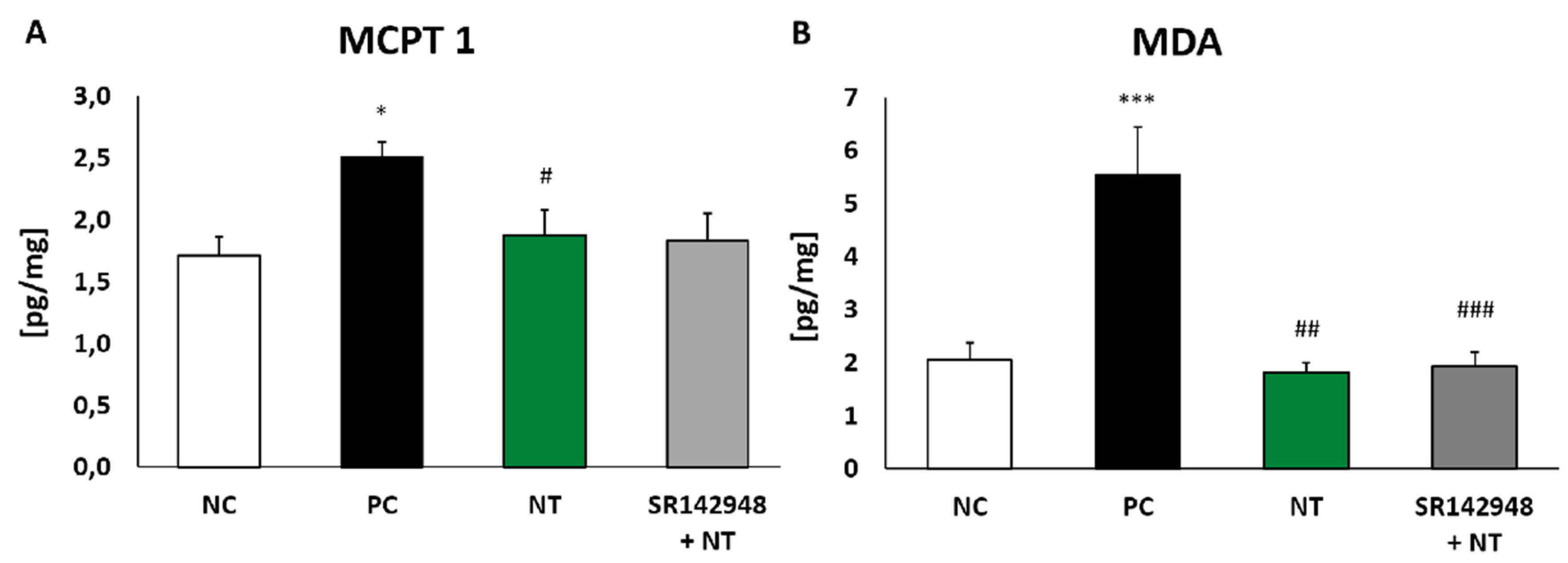
2.4. The Level of Mouse Mast Cell Protease (MCPT 1)
2.5. Oxidative Stress Marker Production
3. Discussion
4. Materials and Methods
4.1. Drugs and Reagents
4.2. Animals and Hapten-Induced Experimental Asthma Model
4.3. Experimental Design
- I
- vehicle-sensitized, DNS-challenged mice treated with physiological saline (NaCl) as the negative control (NC);
- II
- DNFB-sensitized, DNS-challenged mice treated with NaCl as the positive control (PC);
- III
- DNFB-sensitized, DNS-challenged mice treated with neurotensin alone (NT);
- IV
- DNFB-sensitized, DNS-challenged mice pretreated with SR 142948 and treated with NT (SR 142948 + NT).
4.4. Airway Hyperresponsiveness
4.5. Leukocyte Accumulation in BALF
4.6. Pro-Inflammatory Cytokine Production
4.7. The Level of Mouse Mast Cell Protease (MCPT 1)
4.8. Oxidative Stress Marker Production
4.9. Lung Histology
4.10. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
References
- Carraway, R.; Leeman, S.E. The isolation of a new hypotensive peptide, neurotensin, from bovine hypothalami. J. Biol. Chem. 1973, 248, 6854–6861. [Google Scholar]
- Bello, A.R.; Reyes, R.; Hernández, G.; Negrín, I.; González, M.; Tramu, G.; Alonso, R. Developmental expression of neurotensin in thyrotropes and gonadotropes of male and female rats. Neuroendocrinology 2004, 79, 90–99. [Google Scholar] [CrossRef]
- Bissette, G.; Nemeroff, C.B.; Loosen, P.T.; Prange, A.J., Jr.; Lipton, M.A. Hypothermia and intolerance to cold induced by intracisternal administration of the hypothalamic peptide neurotensin. Nature 1976, 262, 607–609. [Google Scholar] [CrossRef] [PubMed]
- Clineschmidt, B.V.; McGuffin, J.C. Neurotensin administered intracisternally inhibits responsiveness of mice to noxious stimuli. Eur. J. Pharmacol. 1977, 46, 395–396. [Google Scholar] [CrossRef]
- Boules, M.; Li, Z.; Smith, K.; Fredrickson, P.; Richelson, E. Diverse roles of neurotensin agonists in the central nervous system. Front. Endocrinol. (Lausanne) 2013, 4, 36. [Google Scholar] [CrossRef] [PubMed]
- Lambert, P.D.; Gross, R.; Nemeroff, C.B.; Kilts, C.D. Anatomy and mechanisms of neurotensin-dopamine interactions in the central nervous system. Ann. N.Y. Acad. Sci. 1995, 757, 377–389. [Google Scholar] [CrossRef] [PubMed]
- Vincent, J.P.; Mazella, J.; Kitabgi, P. Neurotensin and neurotensin receptors. Trends Pharmacol. Sci. 1999, 20, 302–309. [Google Scholar] [CrossRef]
- Tanaka, K.; Masu, M.; Nakanishi, S. Structure and functional expression of the cloned rat neurotensin receptor. Neuron 1990, 4, 847–854. [Google Scholar] [CrossRef]
- Vita, N.; Laurent, P.; Lefort, S.; Chalon, P.; Dumont, X.; Kaghad, M.; Gully, D.; Le Fur, G.; Ferrara, P.; Caput, D. Cloning and expression of a complementary DNA encoding a high affinity human neurotensin receptor. FEBS Lett. 1993, 317, 139–142. [Google Scholar] [CrossRef] [Green Version]
- Chalon, P.; Vita, N.; Kaghad, M.; Guillemot, M.; Bonnin, J.; Delpech, B.; Le Fur, G.; Ferrara, P.; Caput, D. Molecular cloning of a levocabastine-sensitive neurotensin binding site. FEBS Lett. 1996, 386, 91–94. [Google Scholar] [CrossRef] [Green Version]
- Kleczkowska, P.; Lipkowski, A.W. Neurotensin and neurotensin receptors: Characteristic, structure-activity relationship and pain modulation—A review. Eur. J. Pharmacol. 2013, 716, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Mazella, J.; Zsürger, N.; Navarro, V.; Chabry, J.; Kaghad, M.; Caput, D.; Ferrara, P.; Vita, N.; Gully, D.; Maffrand, J.P.; et al. The 100-kDa neurotensin receptor is gp95/sortilin, a non-G-protein-coupled receptor. J. Biol. Chem. 1998, 273, 26273–26276. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, L.; Madsen, P.; Jacobsen, C.; Nielsen, M.S.; Gliemann, J.; Petersen, C.M. Activation and functional characterization of the mosaic receptor SorLA/LR11. J. Biol. Chem. 2001, 276, 22788–22796. [Google Scholar] [CrossRef] [PubMed]
- Moura, L.I.; Cruz, M.T.; Carvalho, E. The effect of neurotensin in human keratinocytes-implication on impaired wound healing in diabetes. Exp. Biol. Med. (Maywood) 2014, 239, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Katsanos, G.S.; Anogianaki, A.; Castellani, M.L.; Ciampoli, C.; De Amicis, D.; Orso, C.; Pollice, R.; Vecchiet, J.; Tetè, S.; Salini, V.; et al. Biology of neurotensin: Revisited study. Int. J. Immunopathol. Pharmacol. 2008, 21, 255–259. [Google Scholar] [CrossRef]
- Schäffer, M.; Beiter, T.; Becker, H.D.; Hunt, T.K. Neuropeptides: Mediators of inflammation and tissue repair? Arch. Surg. 1998, 133, 1107–1116. [Google Scholar] [CrossRef]
- Voisin, T.; Bouvier, A.; Chiu, I.M. Neuro-immune interactions in allergic diseases: Novel targets for therapeutics. Int. Immunol. 2017, 29, 247–261. [Google Scholar] [CrossRef]
- Barnes, P.J. Asthma as an axon reflex. Lancet 1986, 1, 242–245. [Google Scholar] [CrossRef]
- Tränkner, D.; Hahne, N.; Sugino, K.; Hoon, M.A.; Zuker, C. Population of sensory neurons essential for asthmatic hyperreactivity of inflamed airways. Proc. Natl. Acad. Sci. USA. 2014, 111, 11515–11520. [Google Scholar] [CrossRef] [Green Version]
- Kessler, J.P.; Beaudet, A.J. Association of neurotensin binding sites with sensory and visceromotor components of the vagus nerve. J. Neurosci. 1989, 9, 466–472. [Google Scholar] [CrossRef]
- Castagliuolo, I.; Wang, C.C.; Valenick, L.; Pasha, A.; Nikulasson, S.; Carraway, R.E.; Pothoulakis, C.J. Neurotensin is a proinflammatory neuropeptide in colonic inflammation. Clin. Investig. 1999, 103, 843–849. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Margolis, K.G.; Gershon, M.D. Neuropeptides and inflammatory bowel disease. Curr. Opin. Gastroenterol. 2009, 25, 503–511. [Google Scholar] [CrossRef]
- Piliponsky, A.M.; Chen, C.C.; Nishimura, T.; Metz, M.; Rios, E.J.; Dobner, P.R.; Wada, E.; Wada, K.; Zacharias, S.; Mohanasundaram, U.M.; et al. Neurotensin increases mortality and mast cells reduce neurotensin levels in a mouse model of sepsis. Nat. Med. 2008, 14, 392–398. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barchetta, I.; Cimini, F.A.; Capoccia, D.; Bertoccini, L.; Ceccarelli, V.; Chiappetta, C.; Leonetti, F.; Di Cristofano, C.; Silecchia, G.; Orho-Melander, M.; et al. Neurotensin Is a Lipid-Induced Gastrointestinal Peptide Associated with Visceral Adipose Tissue Inflammation in Obesity. Nutrients 2018, 10, 526. [Google Scholar] [CrossRef] [PubMed]
- Law, I.K.; Bakirtzi, K.; Polytarchou, C.; Oikonomopoulos, A.; Hommes, D.; Iliopoulos, D.; Pothoulakis, C. Neurotensin--regulated miR-133α is involved in proinflammatory signalling in human colonic epithelial cells and in experimental colitis. Gut 2015, 64, 1095–1104. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Pothoulakis, C. Effects of NT on gastrointestinal motility and secretion, and role in intestinal inflammation. Peptides 2006, 27, 2434–2444. [Google Scholar] [CrossRef] [PubMed]
- Brun, P.; Mastrotto, C.; Beggiao, E.; Stefani, A.; Barzon, L.; Sturniolo, G.C.; Palù, G.; Castagliuolo, I. Neuropeptide neurotensin stimulates intestinal wound healing following chronic intestinal inflammation. Am. J. Physiol. Gastrointest. Liver Physiol. 2005, 288, G621–G629. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Akcan, A.; Muhtaroglu, S.; Akgun, H.; Akyildiz, H.; Kucuk, C.; Sozuer, E.; Yurci, A.; Yilmaz, N. Ameliorative effects of bombesin and neurotensin on trinitrobenzene sulphonic acid-induced colitis, oxidative damage and apoptosis in rats. World J. Gastroenterol. 2008, 14, 1222–1230. [Google Scholar] [CrossRef] [PubMed]
- Moura, L.I.; Dias, A.M.; Suesca, E.; Casadiegos, S.; Leal, E.C.; Fontanilla, M.R.; Carvalho, L.; de Sousa, H.C.; Carvalho, E. Neurotensin-loaded collagen dressings reduce inflammation and improve wound healing in diabetic mice. Biochim. Biophys. Acta 2014, 1842, 32–43. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- da Silva, L.; Neves, B.M.; Moura, L.; Cruz, M.T.; Carvalho, E. Neurotensin downregulates the pro-inflammatory properties of skin dendritic cells and increases epidermal growth factor expression. Biochim. Biophys. Acta 2011, 1813, 1863–1871. [Google Scholar] [CrossRef] [Green Version]
- Pereira da Silva, L.; Miguel Neves, B.; Moura, L.; Cruz, M.T.; Carvalho, E. Neurotensin decreases the proinflammatory status of human skin fibroblasts and increases epidermal growth factor expression. Int. J. Inflam. 2014, 2014, 248240. [Google Scholar] [CrossRef] [PubMed]
- Van der Kleij, H.P.; Kraneveld, A.D.; van Houwelingen, A.H.; Kool, M.; Weitenberg, A.C.; Redegeld, F.A.; Nijkamp, F.P. Murine model for non-IgE-mediated asthma. Inflammation 2004, 28, 115–125. [Google Scholar] [CrossRef]
- Kaczyńska, K.; Zając, D.; Wojciechowski, P.; Kogut, E.; Szereda-Przestaszewska, M. Neuropeptides and breathing in health and disease. Pulm. Pharmacol. Ther. 2018, 48, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Robbins, R.A.; Nelson, K.J.; Gossman, G.L.; Rubinstein, I. Neurotensin stimulates neutrophil adherence to bronchial epithelial cells in vitro. Life Sci. 1995, 56, 1353–1359. [Google Scholar] [CrossRef]
- Aas, P.; Helle, K.B. Neurotensin receptors in the rat bronchi. Regul. Pept. 1982, 3, 405–413. [Google Scholar] [CrossRef]
- Djokic, T.D.; Dusser, D.J.; Borson, D.B.; Nadel, J.A. Neutral endopeptidase modulates neurotensin-induced airway contraction. J. Appl. Physiol. 1989, 66, 2338–2343. [Google Scholar] [CrossRef] [Green Version]
- Martin, C.A.; Gully, D.; Naline, E.; Advenier, C. Neurotensin modulates cholinergic and noncholinergic neurotransmission in guinea-pig main bronchi in vitro. Neuropeptides 1994, 26, 159–166. [Google Scholar] [CrossRef]
- Tyler-McMahon, B.M.; Boules, M.; Richelson, E. Neurotensin: Peptide for the next millennium. Regul. Pept. 2000, 93, 125–136. [Google Scholar] [CrossRef]
- Gully, D.; Canton, M.; Boigegrain, R.; Jeanjean, F.; Molimard, J.C.; Poncelet, M.; Gueudet, C.; Heaulme, M.; Leyris, R.; Brouard, A.; et al. Biochemical and pharmacological profile of a potent and selective nonpeptide antagonist of the neurotensin receptor. Proc. Natl. Acad. Sci. USA 1993, 90, 65–69. [Google Scholar] [CrossRef]
- Dubuc, I.; Costentin, J.; Terranova, J.P.; Barnouin, M.C.; Soubrié, P.; Le Fur, G.; Rostène, W.; Kitabgi, P. The nonpeptide neurotensin antagonist, SR 48692, used as a tool to reveal putative neurotensin receptor subtypes. Br. J. Pharmacol. 1994, 112, 352–354. [Google Scholar] [CrossRef] [Green Version]
- Schaeffer, P.; Laplace, M.C.; Bernat, A.; Prabonnaud, V.; Gully, D.; Lespy, L.; Herbert, J.M. SR142948A is a potent antagonist of the cardiovascular effects of neurotensin. J. Cardiovasc. Pharmacol. 1998, 31, 545–550. [Google Scholar] [CrossRef] [PubMed]
- Kaczyńska, K.; Szereda-Przestaszewska, M. Cardio-respiratory effects of systemic neurotensin injection are mediated through activation of neurotensin NTS1 receptors. Eur. J. Pharmacol. 2012, 691, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Dobner, P.R. Multitasking with neurotensin in the central nervous system. Cell. Mol. Life Sci. 2005, 62, 1946–1963. [Google Scholar] [CrossRef] [PubMed]
- St-Gelais, F.; Jomphe, C.; Trudeau, L.E. The role of neurotensin in central nervous system pathophysiology: What is the evidence? J. Psychiatry Neurosci. 2006, 31, 229–245. [Google Scholar] [PubMed]
- Trinchieri, G.; Pflanz, S.; Kastelein, R.A. The IL-12 family of heterodimeric cytokines: New players in the regulation of T cell responses. Immunity 2003, 19, 641–644. [Google Scholar] [CrossRef]
- Meyts, I.; Hellings, P.W.; Hens, G.; Vanaudenaerde, B.M.; Verbinnen, B.; Heremans, H.; Matthys, P.; Bullens, D.M.; Overbergh, L.; Mathieu, C.; et al. IL-12 contributes to allergen-induced airway inflammation in experimental asthma. J. Immunol. 2006, 177, 6460–6470. [Google Scholar] [CrossRef] [PubMed]
- Berkman, N.; Krishnan, V.L.; Gilbey, T.; Newton, R.; O’Connor, B.; Barnes, P.J.; Chung, K.F. Expression of RANTES mRNA and protein in airways of patients with mild asthma. Am. J. Respir. Crit. Care Med. 1996, 154, 1804–1811. [Google Scholar] [CrossRef] [PubMed]
- Zietkowski, Z.; Tomasiak, M.M.; Skiepko, R.; Bodzenta-Lukaszyk, A. RANTES in exhaled breath condensate of stable and unstable asthma patients. Respir. Med. 2008, 102, 1198–1202. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agache, I.; Ciobanu, C.; Agache, C.; Anghel, M. Increased serum IL-17 is an independent risk factor for severe asthma. Respir. Med. 2010, 104, 1131–1137. [Google Scholar] [CrossRef] [Green Version]
- Bullens, D.M.; Truyen, E.; Coteur, L.; Dilissen, E.; Hellings, P.W.; Dupont, L.J.; Ceuppens, J.L. IL-17 mRNA in sputum of asthmatic patients: Linking T cell driven inflammation and granulocytic influx? Respir. Res. 2006, 7, 135. [Google Scholar] [CrossRef]
- Dragon, S.; Rahman, M.S.; Yang, J.; Unruh, H.; Halayko, A.J.; Gounni, A.S. IL-17 enhances IL-1beta-mediated CXCL-8 release from human airway smooth muscle cells. Am. J. Physiol. Lung Cell. Mol. Physiol. 2007, 292, L1023–L1029. [Google Scholar] [CrossRef] [PubMed]
- Fossiez, F.; Djossou, O.; Chomarat, P.; Flores-Romo, L.; Ait-Yahia, S.; Maat, C.; Pin, J.J.; Garrone, P.; Garcia, E.; Saeland, S.; et al. T cell interleukin-17 induces stromal cells to produce proinflammatory and hematopoietic cytokines. J. Exp. Med. 1996, 183, 2593–2603. [Google Scholar] [CrossRef] [PubMed]
- Izuhara, K.; Arima, K.; Kanaji, S.; Ohta, S.; Kanaji, T. IL-13: A promising therapeutic target for bronchial asthma. Curr. Med. Chem. 2006, 13, 2291–2298. [Google Scholar] [PubMed]
- Wills-Karp, M. Interleukin-13 in asthma pathogenesis. Immunol. Rev. 2004, 202, 175–190. [Google Scholar] [CrossRef] [PubMed]
- Thomas, P.S.; Yates, D.H.; Barnes, P.J. Tumor necrosis factor-alpha increases airway responsiveness and sputum neutrophilia in normal human subjects. Am. J. Respir. Crit. Care Med. 1995, 152, 76–80. [Google Scholar] [CrossRef] [PubMed]
- Thomas, P.S.; Heywood, G. Effects of inhaled tumour necrosis factor alpha in subjects with mild asthma. Thorax 2002, 57, 774–778. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, Y.S.; Ko, H.M.; Kang, N.I.; Song, C.H.; Zhang, X.; Chung, W.C.; Kim, J.H.; Choi, I.H.; Park, Y.M.; Kim, G.Y.; et al. Mast cells play a key role in the development of late airway hyperresponsiveness through TNF-alpha in a murine model of asthma. Eur. J. Immunol. 2007, 37, 1107–1115. [Google Scholar] [CrossRef] [PubMed]
- Thomas, P.S. Tumour necrosis factor-alpha: The role of this multifunctional cytokine in asthma. Immunol. Cell. Biol. 2001, 79, 132–140. [Google Scholar] [CrossRef]
- Dai, H.; Korthuis, R.J. Mast Cell Proteases and Inflammation. Drug Discov. Today Dis. Models 2011, 8, 47–55. [Google Scholar] [CrossRef]
- Kraneveld, A.D.; van der Kleij, H.P.; Kool, M.; van Houwelingen, A.H.; Weitenberg, A.C.; Redegeld, F.A.; Nijkamp, F.P. Key role for mast cells in nonatopic asthma. J. Immunol. 2002, 169, 2044–2053. [Google Scholar] [CrossRef]
- Sadowska-Woda, I.; Bieszczad-Bedrejczuk, E. Rola stresu oksydacyjnego w patogenezie astmy oskrzelowej. Alergia Astma Immunologia 2011, 16, 80–89. [Google Scholar]
- Abdelaziz, R.R.; Elmahdy, M.K.; Suddek, G.M. Flavocoxid attenuates airway inflammation in ovalbumin-induced mouse asthma model. Chem. Biol. Interact. 2018, 292, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Muti, A.D.; Pârvu, A.E.; Muti, L.A.; Moldovan, R.; Mureşan, A. Vitamin E effect in a rat model of toluene diisocyanate-induced asthma. Clujul. Med. 2016, 89, 499–505. [Google Scholar] [CrossRef] [PubMed]
- Kogut, E.; Kaczyńska, K.; Lipkowski, A.; Kleczkowska, P. Opioid-neurotensin hybrid influences pulmonary inflammatory process in murine model of non-atopic asthma. Eur. Respir. J. 2017, 50, PA1407. [Google Scholar]
- Tournoy, K.G.; Kips, J.C.; Schou, C.; Pauwels, R.A. Airway eosinophilia is not arequirement for allergen-induced airway hyperresponsiveness. Clin. Exp. Allergy 2000, 30, 79–85. [Google Scholar] [CrossRef] [PubMed]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Russjan, E.; Kaczyńska, K. Beneficial Effects of Neurotensin in Murine Model of Hapten-Induced Asthma. Int. J. Mol. Sci. 2019, 20, 5025. https://doi.org/10.3390/ijms20205025
Russjan E, Kaczyńska K. Beneficial Effects of Neurotensin in Murine Model of Hapten-Induced Asthma. International Journal of Molecular Sciences. 2019; 20(20):5025. https://doi.org/10.3390/ijms20205025
Chicago/Turabian StyleRussjan, Ewelina, and Katarzyna Kaczyńska. 2019. "Beneficial Effects of Neurotensin in Murine Model of Hapten-Induced Asthma" International Journal of Molecular Sciences 20, no. 20: 5025. https://doi.org/10.3390/ijms20205025




