Resistance to Cereal Cyst Nematodes in Wheat and Barley: An Emphasis on Classical and Modern Approaches
Abstract
:1. Introduction
2. Economic Impact of CCNs in Wheat and Barely
3. Factors Affecting Yield Losses in CCN-Disease
4. Resistance and Tolerance Responses against CCNs
5. Marker-Assisted Breeding and QTL Mapping for Nematode Resistance
6. Genome-Wide Association Studies for CCN Resistance
7. Nematode Resistance through Modern Approaches
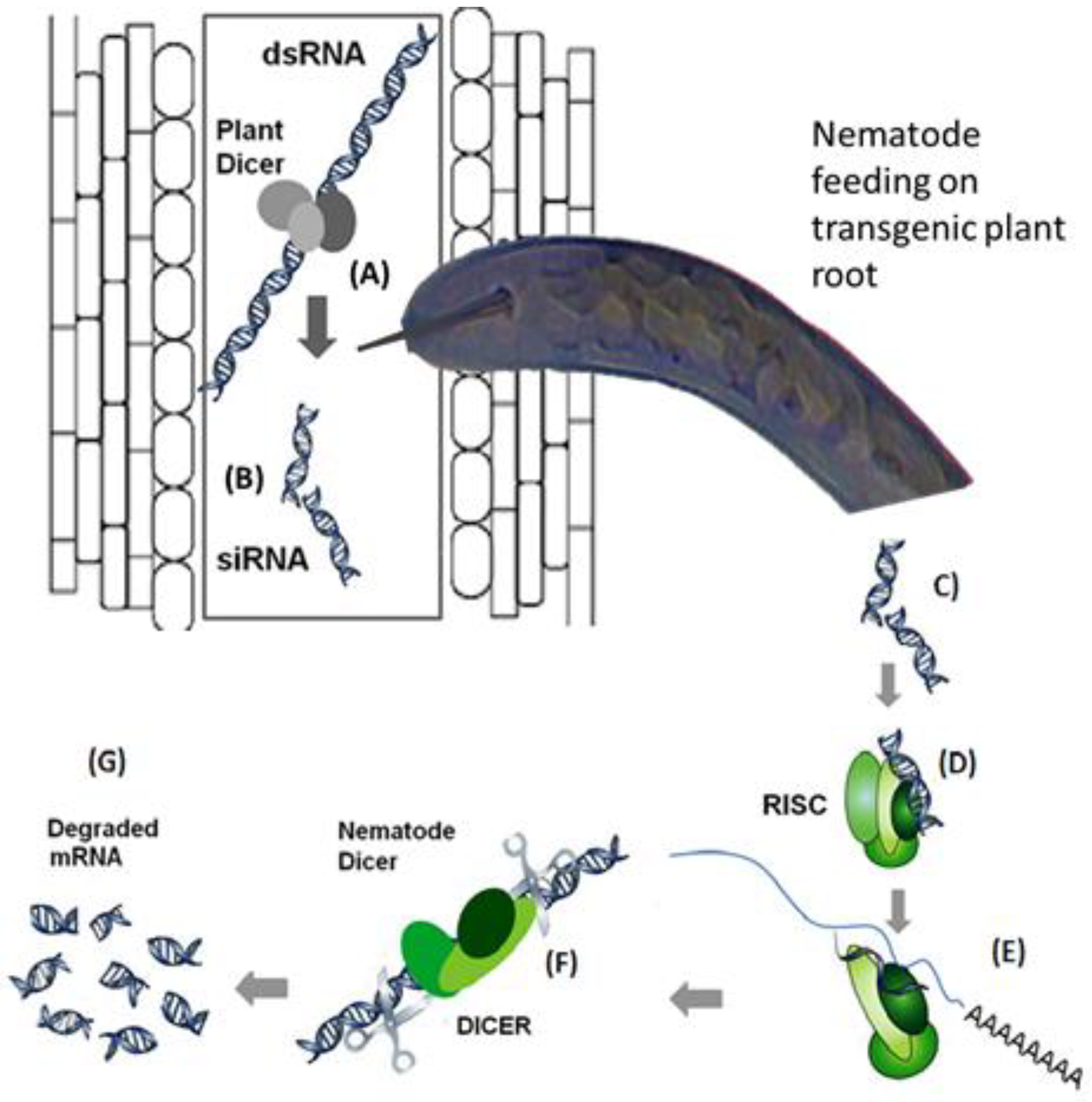
7.1. Gene Silencing for the Enhancement of Nematode Resistance
7.2. Utilization of Proteinase Inhibitors and Chemosensory Disruptive Peptides
7.3. Coupling of Various Resistance Strategies to Augment Nematode Resistance
8. Genome Editing Technologies: A Potential Perspective for Nematode Resistance in Plants
9. Conclusions and Future Perspectives
Funding
Conflicts of Interest
References
- Breiman, A.; Graur, D. Wheat evolution. Israel J. Plant Sci. 1995, 43, 85–98. [Google Scholar] [CrossRef]
- Gustafson, P.; Raskina, O.; Ma, X.-F.; Nevo, E. Wheat evolution, domestication, and improvement. In Wheat Science and Trade; Carver, B., Ed.; Wiley-Blackwell: Ames, IA, USA, 2009; pp. 5–30. [Google Scholar]
- Newman, R.K.; Newman, C.W. Barley history: Relationship of humans and barley through the ages. In Barley for Food and Health: Science, Technology, and Products; Newman, R.K., Walter Newman, C., Eds.; John Wiley and Sons: Hoboken, NJ, USA, 2008; pp. 1–17. [Google Scholar]
- Alexandratos, N.; Bruinsma, J. World Agriculture towards 2030/2050: The 2012 Revision; Division, A.D.E., Ed.; Food and Agriculture Organization of the United Nations: New York, NY, USA, 2012. [Google Scholar]
- OECD-FAO. Agricultural Outlook Organisation for Economic Cooperation and Development (OECD) and Food and Agricultural Organization of the United Nations (FAO). OECD Agricultural Statistics (Database). Available online: http://dx.doi.org/10.1787/agr-data-en (accessed on 7 December 2018).
- Ackerman, F.; Stanton, E.A. Can climate change save lives? A comment on economy-wide estimates of the implications of climate change: Human health. Ecol. Econ. 2008, 66, 8–13. [Google Scholar] [CrossRef]
- Whitehead, A. Plant Nematode Control; CAB International: Oxon, UK; New York, NY, USA, 1998; Volume VIII. [Google Scholar]
- Ali, M.A.; Abbas, A.; Azeem, F.; Javed, N.; Bohlmann, H. Plant-nematode Interactions: From genomics to metabolomics. Int. J. Agric. Biol. 2015, 17, 1071–1082. [Google Scholar]
- Decraemer, W.; Hunt, D.J. Structure and classification. In Plant Nematology; Perry, R.N., Moens, M., Eds.; CABI1: Oxfordshire, UK, 2006; pp. 4–32. [Google Scholar]
- Ali, M.A.; Azeem, F.; Abbas, A.; Joyia, F.A.; Li, H.J.; Dababat, A.A. Transgenic strategies for enhancement of nematode resistance in plants. Front. Plant Sci. 2017, 8, 750. [Google Scholar] [CrossRef]
- Nicol, J.M.; Turner, S.J.; Coyne, D.L.; den Nijs, L.; Hockland, S.; Tahna Maafi, Z. Current nematode threats to world agriculture. In Genomics and Molecular Genetics of Plant-Nematode Interactions; Jones, J., Gheysen, G., Fenoll, C., Eds.; Springer: Dordrecht, The Netherlands, 2011; pp. 21–43. [Google Scholar]
- Smiley, R.W.; Nicol, J.M. Nematodes which challenge global wheat production. In Wheat Science and Trade; Carver, B.F., Ed.; Wiley-Blackwell: Ames, IA, USA, 2009; pp. 171–187. [Google Scholar]
- Dababat, A.A.; Pariyar, S.; Nicol, J.; Erginbasx-Orakçi, G.; Wartin, C.; Klix, M.; Bolat, N.; Braun, H.; Sikora, R. Influence of fungicide seed treatment on the integrated control of Heterodera filipjevi on six wheat germplasm with different levels of genetic resistance under controlled conditions. Nematropica 2014, 44, 25–30. [Google Scholar]
- Dababat, A.A.; Muminjanov, H.; Smiley, R.W. Nematodes of Small Grain Cereals: Current Status and Research; FAO: Ankara, Turkey, 2015. [Google Scholar]
- Nicol, J.M.; Rivoal, R. Global knowledge and its application for the integrated control and management of nematodes on wheat. In Integrated Management and Biocontrol of Vegetable and Grain Crops Nematodes; Ciancio, A., Mukerji, K.G., Eds.; Springer: Dordrecht, The Netherlands, 2008; pp. 243–287. [Google Scholar]
- Smiley, R.W. Occurrence, distribution and control of Heterodera avenae and H. filipjevi in the western USA. In Cereal Cyst Nematodes: Status, Research and Outlook; Riley, I.T., Nicol, J.M., Dababat, A.A., Eds.; CIMMYT: Ankara, Turkey, 2009; pp. 35–40. [Google Scholar]
- Subbotin, S.A.; Sergei, A.; Baldwin, J. Systematics of Cyst Nematodes (Nematoda: Heteroderinae); Brill: Leiden, The Netherlands, 2010; Volume 8A. [Google Scholar]
- Riley, I.T.; McKay, A. Cereal cyst nematode in Australia: Biography of a biological invader. In Cereal Cyst Nematodes: Status, Research and Outlook; Riley, I.T., Nicol, J.M., Dababat, A.A., Eds.; CIMMYT: Ankara, Turkey, 2009; pp. 23–28. [Google Scholar]
- Abidou, H.; El-Ahmed, A.; Nicol, J.M.; Bolat, N.; Rivoal, R.; Yahyaoui, A. Occurrence and distribution of species of the Heterodera avenae group in Syria and Turkey. Nematol. Mediterr. 2005, 33, 195–201. [Google Scholar]
- Elekçioğlu, İ.H.; Nicol, J.; Bolat, N.; Sahin, E.; Yorgancılar, A.; Braun, H.; Yorgancılar, O.; Yılddırım, A.; Kılınç, A.; Toktay, H.; Çalisxkan, M. Longterm studies on the cereal cyst nematode Heterodera filipjevi in Turkey: International collaboration with regional implications. In Cereal Cyst Nematodes: Status, Research and Outlook; Riley, I.T., Nicol, J.M., Dababat, A.A., Eds.; CIMMYT: Ankara, Turkey, 2009; pp. 11–16. [Google Scholar]
- Tanha Maafi, Z.; Nicol, J.M.; Kazemi, H.; Ebrahimi, N.; Gitty, M.; Ghalandar, M.; Mohammadi-Pour, M.; Khoshkhabar, Z. Cereal cyst nematodes, root rot pathogens and root lesion nematodes affecting cereal production in Iran. In Cereal Cyst Nematodes: Status, Research and Outlook; Riley, I.T., Nicol, J.M., Dababat, A.A., Eds.; CIMMYT: Ankara, Turkey, 2009; pp. 51–55. [Google Scholar]
- Smiley, R.W.; Ingham, R.E.; Uddin, W.; Cook, G.H. Crop sequences for managing cereal cyst nematode and fungal populations of winter wheat. Plant Dis. 1994, 78, 1142–1149. [Google Scholar] [CrossRef]
- Rivoal, R.; Cook, R. Nematode pests of cereals. In Plant Parasitic Nematodes in Temperate Agriculture; Evans, K., Trudgill, D.L., Webster, J.M., Eds.; CAB International: Wallingford, UK, 1993; pp. 259–303. [Google Scholar]
- Peng, D.L.; Nicol, J.; Li, H.M.; Hou, S.Y.; Li, H.X.; Chen, S.L.; Ma, P.; Li, H.L.; Riley, I. Current knowledge of cereal cyst nematode (Heterodera avenae) on wheat in China. In Cereal Cyst Nematodes: Status, Research and Outlook; Riley, I.T., Nicol, J.M., Dababat, A.A., Eds.; CIMMYT: Ankara, Turkey, 2009; pp. 29–34. [Google Scholar]
- Riley, I.T.; Qi, R.D. Annotated bibliography of cereal cyst nematodes (Heterodera avenae and H. filipjevi) in China, 1991 to 2014. Australas. Nematol. Newsl. 2015, 26, 1–46. [Google Scholar]
- Murray, G.; Brennan, J. Estimating disease losses to the Australian wheat industry. Australas. Plant Pathol. 2009, 38, 558–570. [Google Scholar] [CrossRef]
- Lewis, J.M.; Matic, M.; McKay, A.C. Success of cereal cyst nematode resistance in Australia: History and status of resistance screening systems. In Cereal Cyst Nematodes: Status, Research and Outlook; Riley, I.T., Nicol, J.M., Dababat, A.A., Eds.; CIMMYT: Ankara, Turkey, 2009; pp. 137–142. [Google Scholar]
- Holgado, R.; Støen, M.; Magnusson, C.; Hammeraas, B. The occurrence and hosts of cereal cyst nematodes (Heterodera spp.) in Norway. Int. J. Nematol. Mediterr. 2003, 13, 1–19. [Google Scholar]
- Smiley, R.W.; Dababat, A.A.; Iqbal, S.; Jones, M.G.K.; Maafi, Z.T.; Peng, D.L.; Subbotin, S.A.; Waeyenberge, L. Cereal cyst nematodes: A complex and destructive group of Heterodera species. Plant Dis. 2017, 101, 1692–1720. [Google Scholar] [CrossRef]
- Riley, I.T.; Nicol, J.M.; Dababat, A.A. Cereal Cyst Nematodes: Status, Research and Outlook; CIMMYT: Ankara, Turkey, 2009. [Google Scholar]
- Dababat, A.; Mokrini, F.; Smiley, R.W. Proceedings of the Sixth International Cereal Nematodes Symposium (Agadir, Morocco); CIMMYT: Ankara, Turkey, 2017. [Google Scholar]
- Hajihasani, A.; Tanha Maafi, Z.; Nicol, J.M.; Rezaee, S. Effect of the cereal cyst nematode, Heterodera filipjevi, on wheat in microplot trials. Nematology 2010, 12, 357–363. [Google Scholar]
- Andersen, S. Resistens mod Havreål Heterodera avenae; I Kommission hos Dansk Videnskabs Forlag: Copenhagen, Denmark, 1961. [Google Scholar]
- Singh, A.; Sharma, A.K.; Shoran, J. Heterodera avenae and its management in India. In Cereal Cyst Nematodes: Status, Research and Outlook; Riley, I.T., Nicol, J.M., Dababat, A.A., Eds.; CIMMYT: Ankara, Turkey, 2009; pp. 17–22. [Google Scholar]
- Trudgill, D.L. Resistance to and tolerance of plant parasitic nematodes in plants. Annu. Rev. Phytopathol. 1991, 29, 167–192. [Google Scholar] [CrossRef]
- Cook, R.; Evans, K. Resistance and tolerance. In Principles and Practice of Nematode Control in Crops; Brown, R.H., Kerry, B.R., Eds.; Academic Press: Sydney, Australia, 1987; pp. 179–231. [Google Scholar]
- Cui, L.; Sun, L.; Gao, X.; Song, W.; Wang, X.M.; Li, H.L.; Liu, Z.Y.; Tang, W.H.; Li, H.J. The impact of resistant and susceptible wheat cultivars on the multiplication of Heterodera filipjevi and H. avenae in parasite-infected soil. Plant Pathol. 2016, 65, 1192–1199. [Google Scholar] [CrossRef]
- McDonald, A.; Nicol, J. Nematode parasites of cereals. In Plant Parasitic Nematodes in Subtropical and Tropical Agriculture; Luc, M., Sikora, R.A., Bridge, J., Eds.; CAB Int.: Wallingford, UK, 2005; pp. 131–191. [Google Scholar]
- Nicol, J.M. Important nematode pests of cereals. In Bread Wheat: Improvement and Production; Curtis, B.C., Rajaram, S., Gómez, M., Eds.; FAO: Rome, Italy, 2002; pp. 345–366. [Google Scholar]
- Nicol, J.M.; Rivoal, R.; Taylor, S.; Zaharieva, M. Global importance of cyst (Heterodera spp.) and lesion nematodes (Pratylenchus spp.) on cereals: Distribution, yield loss, use of host resistance and integration of molecular tools. Nematol. Monogr. Perspect. 2003, 2, 1–19. [Google Scholar]
- Rivoal, R.; Bekal, S.; Valette, S.; Gauthier, J.P.; Fradj, M.B.; Mokabli, A.; Jahier, J.; Nicol, J.M.; Yahyaoui, A. Variation in reproductive capacity and virulence on different genotypes and resistance genes of Triticeae, in the cereal cyst nematode species complex. Nematology 2001, 3, 581–590. [Google Scholar]
- Brown, R.H. Control strategies in low-value crops. In Principles and Practice of Nematode Control in Crops; Brown, R.H., Kerry, B.R., Eds.; Academic Press: Sydney, Australia, 1987; pp. 351–387. [Google Scholar]
- Smiley, R.W.; Marshall, J.M. Detection of dual Heterodera avenae resistance plus tolerance traits in spring wheat. Plant Dis. 2016, 100, 1677–1685. [Google Scholar] [CrossRef]
- Andersson, S. Population dynamics and control of Heterodera avenae—A review with some original results. EPPO Bull. 1982, 12, 463–475. [Google Scholar] [CrossRef]
- O’Brien, P.C.; Fisher, J.M. Development of Heterodera avenae on resistant wheat and barley cultivars. Nematologica 1977, 23, 390–397. [Google Scholar] [CrossRef]
- Ogbonnaya, F.C.; Seah, S.; Delibes, A.; Jahier, J.; Lopez-Braña, I.; Eastwood, R.F.; Lagudah, E.S. Molecular genetic characterisation of a new nematode resistance gene in wheat. Theor. Appl. Genet. 2001, 102, 623–629. [Google Scholar] [CrossRef]
- Oka, Y.; Chet, I.; Speigel, Y. Accumulation of lectins in cereal roots invaded by the cereal cyst nematode Heterodera avenae. Physiol. Mol. Plant Pathol. 1997, 51, 333–345. [Google Scholar] [CrossRef]
- Cui, L.; Gao, X.; Wang, X.M.; Jian, H.; Tang, W.H.; Li, H.L.; Li, H.J. Characterization of interaction between wheat roots with different resistance and Heterodera filipjevi. Acta Agron. Sin. 2012, 38, 1009–1017. [Google Scholar] [CrossRef]
- Stanton, J.M.; Fisher, J.M. Factors of early growth associated with tolerance of wheat to Heterodera avenae. Nematologica 1988, 34, 188–197. [Google Scholar] [CrossRef]
- Volkmar, K.M. The cereal cyst nematode (Heterodera avenae) on oats. I. Identification of attributes useful in early screening for tolerance to H. avenae. Aust. J. Agric. Res. 1990, 41, 39–49. [Google Scholar] [CrossRef]
- Wilson, R.E.; Hollamby, G.J.; Bayraktar, A. Selecting for high yield potential in wheat with tolerance to cereal cyst nematode. Aust. Field Crops Newsl. 1983, 18, 21–25. [Google Scholar]
- Williams, T.D.; Salt, G.A. The effects of soil sterilants on the cereal cyst-nematode (Heterodera avenae Woll.), take-all (Ophiobolus graminis Sacc.) and yields of spring wheat and barley. Ann. Appl. Biol. 1970, 66, 329–338. [Google Scholar] [CrossRef]
- Andres, M.F.; Melillo, M.T.; Delibes, A.; Romero, M.D.; Bleve-Zacheo, T. Changes in wheat root enzymes correlated with resistance to cereal cyst nematodes. New Phytol. 2001, 152, 343–354. [Google Scholar] [CrossRef] [Green Version]
- Montes, M.J.; López-Braña, I.; Delibes, A. Root enzyme activities associated with resistance to Heterodera avenae conferred by gene Cre7 in a wheat/Aegilops triuncalis introgression line. J. Plant Physiol. 2004, 161, 493–495. [Google Scholar] [CrossRef]
- Montes, M.J.; López-Braña, I.; Romero, M.D.; Sin, E.; Andrés, M.F.; Martín-Sánchez, J.; Delibes, A. Biochemical and genetic studies of two Heterodera avenae resistance genes transferred from Aegilops ventricosa to wheat. Theor. Appl. Genet. 2003, 107, 611–618. [Google Scholar] [CrossRef]
- Seah, S.; Miller, C.; Sivasithamparam, K.; Lagudah, E.S. Root responses to cereal cyst nematode (Heterodera avenae) in hosts with different resistance genes. New Phytol. 2000, 146, 527–533. [Google Scholar] [CrossRef]
- Cook, R.; York, P.A. Reaction of some European and Australian oat cultivars to cereal cyst nematode. Ann. Appl. Biol. 1988, 122, 84–85. [Google Scholar]
- Kretschmer, J.M.; Chalmers, K.J.; Manning, S.; Karakousis, A.; Barr, A.R.; Islam, A.K.M.R.; Logue, S.J.; Choe, Y.W.; Barker, S.J.; Lance, R.C.M.; et al. RFLP mapping of the Ha2 cereal cyst nematode resistance gene in barley. Theor. Appl. Genet. 1997, 94, 1060–1064. [Google Scholar] [CrossRef]
- Barr, A.R.; Chalmers, K.J.; Karakousis, A.; Kretschmer, J.M.; Manning, S.; Lance, R.C.M.; Lewis, J.; Jeffries, S.P.; Langridge, P. RFLP mapping of a new cereal cyst nematode resistance locus in barley. Plant Breed 1998, 117, 185–187. [Google Scholar] [CrossRef]
- Barloy, D.; Lemoine, J.; Abelard, P.; Tanguy, A.M.; Rivoal, R.; Jahier, J. Marker-assisted pyramiding of two cereal cyst nematode resistance genes from Aegilops variabilis in wheat. Mol. Breed 2007, 20, 31–40. [Google Scholar] [CrossRef]
- Zhang, R.Q.; Feng, Y.X.; Li, H.F.; Yuan, H.X.; Dai, J.L.; Cao, A.Z.; Xing, X.P.; Li, H.L. Cereal cyst nematode resistance gene CreV effective against Heterodera filipjevi transferred from chromosome 6VL of Dasypyrum villosum to bread wheat. Mol. Breed. 2016, 36, 122. [Google Scholar] [CrossRef]
- Baloch, F.S.; Cömertbay, G.; Özkan, H. DNA molecular markers for disease resistance in plant breeding with example in wheat. In Nematodes of Small Grain Cereals: Current Status and Research; Dababat, A., Muminjanov, H., Smiley, R.W., Eds.; FAO: Ankara, Turkey, 2015; pp. 159–166. [Google Scholar]
- Mokabli, A.; Rivoal, R.; Gauthier, J.P.; Valette, S. Variation in virulence of cereal cyst nematode populations from North Africa and Asia. Nematology 2002, 4, 521–525. [Google Scholar]
- Vanstone, V.A.; Hollaway, G.J.; Stirling, G.R. Managing nematode pests in the southern and western regions of the Australian cereal industry: Continuing progress in a challenging environment. Australas. Plant Pathol. 2008, 37, 220–234. [Google Scholar] [CrossRef]
- De Majnik, J.; Ogbonnaya, F.C.; Moullet, O.; Lagudah, E.S. The Cre1 and Cre3 nematode resistance genes are located at homeologous loci in the wheat genome. Mol. Plant-Microbe Interact. 2003, 16, 1129–1134. [Google Scholar] [CrossRef] [PubMed]
- Safari, E.; Gororo, N.N.; Eastwood, R.F.; Lewis, J.; Eagles, H.A.; Ogbonnaya, F.C. Impact of Cre1, Cre8 and Cre3 genes on cereal cyst nematode resistance in wheat. Theor. Appl. Genet. 2005, 110, 567–572. [Google Scholar] [CrossRef] [PubMed]
- Nicol, J.M.; Rivoal, R.; Trethowan, R.M.; van Ginkel, M.; Mergoum, M.; Singh, R.P. CIMMYT’s approach to identify and use resistance to nematodes and soil-borne fungi, in developing superior wheat germplasm. In Wheat in a Global Environment; Bedo, Z., Lang, L., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2001; pp. 381–389. [Google Scholar]
- Dababat, A.A.; Ferney, G.-B.H.; Erginbas-Orakci, G.; Dreisigacker, S.; Imren, M.; Toktay, H.; Elekçioğlu, İ.H.; Mekete, T.; Nicol, J.M.; Ansari, O.; et al. Association analysis of resistance to cereal cyst nematodes (Heterodera avenae) and root lesion nematodes (Pratylenchus neglectus and P. thornei) in CIMMYT advanced spring wheat lines for semi-arid conditions. Breed. Sci. 2016, 66, 692–702. [Google Scholar] [CrossRef] [PubMed]
- Mulki, M.A.; Jighly, A.; Ye, G.; Emebiri, L.C.; Moody, D.; Ansari, O.; Ogbonnaya, F.C. Association mapping for soilborne pathogen resistance in synthetic hexaploid wheat. Mol. Breed. 2013, 31, 299–311. [Google Scholar] [CrossRef]
- Ogbonnaya, F.C.; Eastwood, R.F.; Lagudah, E. Identification and utilisation of genes for cereal cyst nematode (Heterodera avenae) resistance in wheat: The Australian experience. In Cereal Cyst Nematodes: Status, Research and Outlook; Riley, I.T., Nicol, M.J., Dababat, A.A., Eds.; CIMMYT: Ankara, Turkey, 2009; pp. 166–171. [Google Scholar]
- Ali, M.; Anjam, M.; Nawaz, M.; Lam, H.M.; Chung, G. Signal transduction in plant–nematode interactions. Int. J. Mol. Sci. 2018, 19, 1648. [Google Scholar] [CrossRef]
- Uehara, T.; Sugiyama, S.; Matsuura, H.; Arie, T.; Masuta, C. Resistant and susceptible responses in tomato to cyst nematode are differentially regulated by salicylic acid. Plant Cell Physiol. 2010, 51, 1524–1536. [Google Scholar] [CrossRef]
- Simonetti, E.; Alba, E.; Montes, M.J.; Delibes, A.; López-Braña, I. Analysis of ascorbate peroxidase genes expressed in resistant and susceptible wheat lines infected by the cereal cyst nematode, Heterodera avenae. Plant Cell Rep. 2010, 29, 1169–1178. [Google Scholar] [CrossRef]
- Linsell, K.J.; Rahman, M.S.; Taylor, J.D.; Davey, R.S.; Gogel, B.J.; Wallwork, H.; Forrest, K.L.; Hayden, M.J.; Taylor, S.P.; Oldach, K.H. QTL for resistance to root lesion nematode (Pratylenchus thornei) from a synthetic hexaploid wheat source. Theor. Appl. Genet. 2014, 127, 1409–1421. [Google Scholar] [CrossRef]
- Imren, M.; Toktay, H.; Bozbuğa, R.; Dababat, A.; Elekçioğlu, İ.H. Pathotype determination of the cereal cyst nematode, Heterodera avenae (Wollenweber, 1924) in the Eastern Mediterranean Region in Turkey. Türk. J. Entomol. 2013, 37, 13–19. [Google Scholar]
- Yang, T.M.; Xie, C.J.; Sun, Q.X. Situation of the sources of stripe rust resistance of wheat in the post-CYR32 era in China. Acta Agron. Sin. 2003, 29, 161–168. [Google Scholar] [CrossRef]
- Jia, J.Z.; Zhao, S.C.; Kong, X.Y.; Li, Y.R.; Zhao, G.Y.; He, W.M.; Appels, R.; Pfeifer, M.; Tao, Y.; Zhang, X.Y.; et al. International Wheat Genome Sequencing Consortium, Yang, H.M.; Liu, X.; He, Z.H.; Mao, L.; Wang, J. Aegilops tauschii draft genome sequence reveals a gene repertoire for wheat adaptation. Nature 2013, 496, 91–95. [Google Scholar] [CrossRef]
- Tian, F.; Bradbury, P.J.; Brown, P.J.; Hung, H.; Sun, Q.; Flint-Garcia, S.; Rocheford, T.R.; McMullen, M.D.; Holland, J.B.; Buckler, E.S. Genome-wide association study of leaf architecture in the maize nested association mapping population. Nat. Genet. 2011, 43, 159–162. [Google Scholar] [CrossRef]
- Zhao, K.; Tung, C.-W.; Eizenga, G.C.; Wright, M.H.; Ali, M.L.; Price, A.H.; Norton, G.J.; Islam, M.R.; Reynolds, A.; Mezey, J.; et al. Genome-wide association mapping reveals a rich genetic architecture of complex traits in Oryza sativa. Nat. Commun. 2011, 2, 467. [Google Scholar] [CrossRef]
- Berkman, P.J.; Lai, K.; Lorenc, M.T.; Edwards, D. Next-generation sequencing applications for wheat crop improvement. Am. J. Bot. 2012, 99, 365–371. [Google Scholar] [CrossRef] [Green Version]
- Chia, J.-M.; Song, C.; Bradbury, P.J.; Costich, D.; de Leon, N.; Doebley, J.; Elshire, R.J.; Gaut, B.; Geller, L.; Glaubitz, J.C.; et al. Maize HapMap2 identifies extant variation from a genome in flux. Nat. Genet. 2012, 44, 803–807. [Google Scholar] [CrossRef]
- Xu, X.; Liu, X.; Ge, S.; Jensen, J.D.; Hu, F.Y.; Li, X.; Dong, Y.; Gutenkunst, R.N.; Fang, L.; Huang, L.; et al. Resequencing 50 accessions of cultivated and wild rice yields markers for identifying agronomically important genes. Nat. Biotechnol. 2011, 30, 105–111. [Google Scholar] [CrossRef]
- Allen, A.M.; Barker, G.L.A.; Berry, S.T.; Coghill, J.A.; Gwilliam, R.; Kirby, S.; Robinson, P.; Brenchley, R.C.; D’Amore, R.; McKenzie, N.; et al. Transcript-specific, single-nucleotide polymorphism discovery and linkage analysis in hexaploid bread wheat (Triticum aestivum L.). Plant Biotechnol. J. 2011, 9, 1086–1099. [Google Scholar] [CrossRef]
- Cavanagh, C.R.; Chao, S.; Wang, S.; Huang, B.E.; Stephen, S.; Kiani, S.; Forrest, K.; Saintenac, C.; Brown-Guedira, G.L.; Akhunova, A.; et al. Genome-wide comparative diversity uncovers multiple targets of selection for improvement in hexaploid wheat landraces and cultivars. Proc. Natl. Acad. Sci. USA 2013, 110, 8057–8062. [Google Scholar] [CrossRef] [Green Version]
- Elshire, R.J.; Glaubitz, J.C.; Sun, Q.; Poland, J.A.; Kawamoto, K.; Buckler, E.S.; Mitchell, S.E. A robust, simple genotyping-by-sequencing (GBS) approach for high diversity species. PLoS ONE 2011, 6, e19379. [Google Scholar] [CrossRef]
- Zhang, J.; Wen, Z.X.; Li, W.; Zhang, Y.W.; Zhang, L.F.; Dai, H.Y.; Wang, D.C.; Xu, R. Genome-wide association study for soybean cyst nematode resistance in Chinese elite soybean cultivars. Mol. Breed. 2017, 37, 60. [Google Scholar] [CrossRef]
- Pariyar, S.R.; Dababat, A.A.; Sannemann, W.; Erginbas-Orakci, G.; Elashry, A.; Siddique, S.; Morgounov, A.; Leon, J.; Grundler, F.M.W. Genome-wide association study in wheat identifies resistance to the cereal cyst nematode Heterodera filipjevi. Phytopathology 2016, 106, 1128–1138. [Google Scholar] [CrossRef]
- Urwin, P.E.; Lilley, C.J.; Atkinson, H.J. Ingestion of double-stranded RNA by preparasitic juvenile cyst nematodes leads to RNA interference. Mol. Plant-Microbe Interact. 2002, 15, 747–752. [Google Scholar] [CrossRef]
- Fosu-Nyarko, J.; Jones, M.G.K. Advances in understanding the molecular mechanisms of root lesion nematode host interactions. Annu. Rev. Phytopathol. 2016, 54, 253–278. [Google Scholar] [CrossRef]
- Tan, J.A.C.H.; Jones, M.G.K.; Fosu-Nyarko, J. Gene silencing in root lesion nematodes (Pratylenchus spp.) significantly reduces reproduction in a plant host. Exp. Parasitol. 2013, 133, 166–178. [Google Scholar] [CrossRef]
- Gantasala, N.P.; Kumar, M.; Banakar, P.; Thakur, P.K.; Rao, U. Functional validation of genes in cereal cyst nematode, Heterodera avenae, using siRNA gene silencing. In Nematodes of Small Grain Cereals: Current Status and Research; Dababat, A., Muminjanov, H., Smiley, R.W., Eds.; FAO: Ankara, Turkey, 2015; pp. 353–356. [Google Scholar]
- Ali, M.A.; Azeem, F.; Li, H.J.; Bohlmann, H. Smart parasitic nematodes use multifaceted strategies to parasitize plants. Front. Plant Sci. 2017, 8, 1699. [Google Scholar] [CrossRef]
- Dutta, T.K.; Banakar, P.; Rao, U. The status of RNAi-based transgenic research in plant nematology. Front. Microbiol. 2015, 5, 760. [Google Scholar] [CrossRef]
- Klink, V.P.; Kim, K.-H.; Martins, V.; MacDonald, M.H.; Beard, H.S.; Alkharouf, N.W.; Lee, S.-K.; Park, S.-C.; Matthews, B.F. A correlation between host-mediated expression of parasite genes as tandem inverted repeats and abrogation of development of female Heterodera glycines cyst formation during infection of Glycine max. Planta 2009, 230, 53–71. [Google Scholar] [CrossRef]
- Replogle, A.; Wang, J.; Bleckmann, A.; Hussey, R.S.; Baum, T.J.; Sawa, S.; Davis, E.L.; Wang, X.; Simon, R.; Mitchum, M.G. Nematode CLE signaling in Arabidopsis requires CLAVATA2 and CORYNE. Plant J. 2011, 65, 430–440. [Google Scholar] [CrossRef]
- Replogle, A.; Wang, J.; Paolillo, V.; Smeda, J.; Kinoshita, A.; Durbak, A.; Tax, F.E.; Wang, X.; Sawa, S.; Mitchum, M.G. Synergistic interaction of CLAVATA1, CLAVATA2, and RECEPTOR-LIKE PROTEIN KINASE 2 in cyst nematode parasitism of Arabidopsis. Mol. Plant-Microbe Interact. 2013, 26, 87–96. [Google Scholar] [CrossRef]
- Guo, X.; Chronis, D.; De La Torre, C.M.; Smeda, J.; Wang, X.; Mitchum, M.G. Enhanced resistance to soybean cyst nematode Heterodera glycines in transgenic soybean by silencing putative CLE receptors. Plant Biotechnol. J. 2015, 13, 801–810. [Google Scholar] [CrossRef]
- Ali, M.A.; Plattner, S.; Radakovic, Z.; Wieczorek, K.; Elashry, A.; Grundler, F.M.; Ammelburg, M.; Siddique, S.; Bohlmann, H. An Arabidopsis ATPase gene involved in nematode-induced syncytium development and abiotic stress responses. Plant J. 2013, 74, 852–866. [Google Scholar] [CrossRef]
- Ali, M.A.; Wieczorek, K.; Kreil, D.P.; Bohlmann, H. The beet cyst nematode Heterodera schachtii modulates the expression of WRKY transcription factors in syncytia to favour its development in Arabidopsis roots. PLoS ONE 2014, 9, e102360. [Google Scholar] [CrossRef]
- Chen, C.L.; Liu, S.S.; Liu, Q.; Niu, J.H.; Liu, P.; Zhao, J.L.; Jian, H. An ANNEXIN-like protein from the cereal cyst nematode Heterodera avenae suppresses plant defense. PLoS ONE 2015, 10, e0122256. [Google Scholar] [CrossRef]
- Luo, S.J.; Liu, S.M.; Kong, L.A.; Peng, H.; Huang, W.K.; Jian, H.; Peng, D.L. Two venom allergen-like proteins, HaVAP1 and HaVAP2, are involved in the parasitism of Heterodera avenae. Mol. Plant Pathol. 2018, in press. [Google Scholar] [CrossRef]
- Kumar, M.; Gantasala, N.P.; Roychowdhury, T.; Thakur, P.K.; Banakar, P.; Shukla, R.N.; Jones, M.G.K.; Rao, U. De novo transcriptome sequencing and analysis of the cereal cyst nematode, Heterodera avenae. PLoS ONE 2014, 9, e96311. [Google Scholar] [CrossRef]
- Jones, M.G.K.; Fosu-Nyarko, J. Molecular biology of root lesion nematodes (Pratylenchus spp.) and their interaction with host plants. Ann. Appl. Biol. 2014, 164, 163–181. [Google Scholar] [CrossRef]
- Lilley, C.J.; Devlin, F.; Urwin, P.E.; Atkinson, H.J. Parasitic nematodes, proteinases and transgenic plants. Parasitol. Today 1999, 15, 414–417. [Google Scholar] [CrossRef]
- Vishnudasan, D.; Tripathi, M.N.; Rao, U.; Khurana, P. Assessment of nematode resistance in wheat transgenic plants expressing potato proteinase inhibitor (PIN2) gene. Transgenic Res. 2005, 14, 665–675. [Google Scholar] [CrossRef]
- Winter, M.D.; McPherson, M.J.; Atkinson, H.J. Neuronal uptake of pesticides disrupts chemosensory cells of nematodes. Parasitology 2002, 125, 561–565. [Google Scholar]
- Liu, B.; Hibbard, J.K.; Urwin, P.E.; Atkinson, H.J. The production of synthetic chemodisruptive peptides in planta disrupts the establishment of cyst nematodes. Plant Biotechnol. J. 2005, 3, 487–496. [Google Scholar] [CrossRef] [Green Version]
- Costa, J.C.; Lilley, C.J.; Atkinson, H.J.; Urwin, P.E. Functional characterisation of a cyst nematode acetylcholinesterase gene using Caenorhabditis elegans as a heterologous system. Int. J. Parasitol. 2009, 39, 849–858. [Google Scholar] [CrossRef]
- Wang, D.; Jones, L.M.; Urwin, P.E.; Atkinson, H.J. A synthetic peptide shows retro- and anterograde neuronal transport before disrupting the chemosensation of plant-pathogenic nematodes. PLoS ONE 2011, 6, e17475. [Google Scholar] [CrossRef]
- Tripathi, L.; Babirye, A.; Roderick, H.; Tripathi, J.N.; Changa, C.; Urwin, P.E.; Tushemereirwe, W.K.; Coyne, D.; Atkinson, H.J. Field resistance of transgenic plantain to nematodes has potential for future African food security. Sci. Rep. 2015, 5, 8127. [Google Scholar] [CrossRef]
- Green, J.; Wang, D.; Lilley, C.J.; Urwin, P.E.; Atkinson, H.J. Transgenic potatoes for potato cyst nematode control can replace pesticide use without impact on soil quality. PLoS ONE 2012, 7, e30973. [Google Scholar] [CrossRef]
- Tripathi, L.; Tripathi, J.N.; Roderick, H.; Atkinson, H.J. Engineering nematode resistant plantains for sub-Saharan Africa. Acta Hort. (ISHS) 2013, 974, 99–107. [Google Scholar] [CrossRef]
- Roderick, H.; Tripathi, L.; Babirye, A.; Wang, D.; Tripathi, J.; Urwin, P.E.; Atkinson, H.J. Generation of transgenic plantain (Musa spp.) with resistance to plant pathogenic nematodes. Mol. Plant Pathol. 2012, 13, 842–851. [Google Scholar] [CrossRef]
- Chan, Y.-L.; He, Y.; Hsiao, T.-T.; Wang, C.-J.; Tian, Z.; Yeh, K.-W. Pyramiding taro cystatin and fungal chitinase genes driven by a synthetic promoter enhances resistance in tomato to root-knot nematode Meloidogyne incognita. Plant Sci. 2015, 231, 74–81. [Google Scholar] [CrossRef]
- Tripathi, L.; Atkinson, H.; Roderick, H.; Kubiriba, J.; Tripathi, J.N. Genetically engineered bananas resistant to Xanthomonas wilt disease and nematodes. Food Energy Secur. 2017, 6, 37–47. [Google Scholar] [CrossRef]
- Waltz, E. Nonbrowning GM apple cleared for market. Nat. Biotechnol. 2015, 33, 326–327. [Google Scholar] [CrossRef]
- Kumar, V.; Jain, M. The CRISPR–Cas system for plant genome editing: Advances and opportunities. J. Exp. Bot. 2015, 66, 47–57. [Google Scholar] [CrossRef]
- Belhaj, K.; Chaparro-Garcia, A.; Kamoun, S.; Nekrasov, V. Plant genome editing made easy: Targeted mutagenesis in model and crop plants using the CRISPR/Cas system. Plant Methods 2013, 9, 39. [Google Scholar] [CrossRef]
- Ali, Z.; Abulfaraj, A.; Idris, A.; Ali, S.; Tashkandi, M.; Mahfouz, M.M. CRISPR/Cas9-mediated viral interference in plants. Genome Biol. 2015, 16, 238. [Google Scholar] [CrossRef]
- Baltes, N.J.; Hummel, A.W.; Konecna, E.; Cegan, R.; Bruns, A.N.; Bisaro, D.M.; Voytas, D.F. Conferring resistance to geminiviruses with the CRISPR–Cas prokaryotic immune system. Nat. Plants 2015, 1, 15145. [Google Scholar] [CrossRef]
- Lozano-Juste, J.; Cutler, S.R. Plant genome engineering in full bloom. Trends Plant Sci. 2014, 19, 284–287. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, D.J.; Goldstein, B. CRISPR-based methods for Caenorhabditis elegans genome engineering. Genetics 2016, 202, 885–901. [Google Scholar] [CrossRef] [PubMed]
- Paix, A.; Folkmann, A.; Seydoux, G. Precision genome editing using CRISPR-Cas9 and linear repair templates in C. elegans. Methods 2017, 121–122, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Kang, J. Application of CRISPR/Cas9-Mediated Genome Editing for Studying Soybean Resistance to Soybean Cyst Nematode; University of Missouri-Columbia, University of Missouri-Columbia: Columbia, MO, USA, 2016. [Google Scholar]
- Mayer, K.F.X.; Rogers, J.; Doleel, J.; Pozniak, C.; Eversole, K.; Feuillet, C.; Gill, B.; Friebe, B.; Lukaszewski, A.J.; Sourdille, P.; et al. A chromosome-based draft sequence of the hexaploid bread wheat (Triticum aestivum) genome. Science 2014, 345, 1251788. [Google Scholar]
- Consortium, I.B.G.S. A physical, genetic and functional sequence assembly of the barley genome. Nature 2012, 491, 711–716. [Google Scholar] [Green Version]
- International Wheat Genome Sequencing Consortium. Shifting the limits in wheat research and breeding using a fully annotated reference genome. Science 2018, 361, eaar7191. [Google Scholar] [CrossRef]
- Coskun, D.; Deshmukh, R.; Sonah, H.; Menzies, J.G.; Reynolds, O.; Ma, J.F.; Kronzucker, H.J.; Belanger, R.R. The controversies of silicon’s role in plant biology. New Phytol. 2019, 221, 67–85. [Google Scholar] [CrossRef]
- Fosu-Nyarko, J.; Jones, M.G.K. Application of biotechnology for nematode control in crop plants. In Advances in Botanical Research; Carolina, E., Carmen, F., Eds.; Academic Press: Cambridge, MA, USA, 2015; Volume 73, pp. 339–376. [Google Scholar]
- Naz, F.; Fosu-Nyarko, J.; Jones, M. Improving the effectiveness and delivery of gene silencing triggers to control plant nematode pests. In Proc. 32nd Symp. Eur. Soc. Nematol.; Murdoch University: Braga, Portugal, 2016; p. 69. [Google Scholar]
- Huang, Q.; Li, L.; Zheng, M.; Chen, F.; Long, H.; Deng, G.; Pan, Z.; Liang, J.; Li, Q.; Yu, M.; et al. The Tryptophan decarboxylase 1 gene from Aegilops variabilis No.1 regulate the resistance against cereal cyst nematode by altering the downstream secondary metabolite contents rather than auxin synthesis. Front. Plant Sci. 2018, 9, 1297. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ali, M.A.; Shahzadi, M.; Zahoor, A.; Dababat, A.A.; Toktay, H.; Bakhsh, A.; Nawaz, M.A.; Li, H. Resistance to Cereal Cyst Nematodes in Wheat and Barley: An Emphasis on Classical and Modern Approaches. Int. J. Mol. Sci. 2019, 20, 432. https://doi.org/10.3390/ijms20020432
Ali MA, Shahzadi M, Zahoor A, Dababat AA, Toktay H, Bakhsh A, Nawaz MA, Li H. Resistance to Cereal Cyst Nematodes in Wheat and Barley: An Emphasis on Classical and Modern Approaches. International Journal of Molecular Sciences. 2019; 20(2):432. https://doi.org/10.3390/ijms20020432
Chicago/Turabian StyleAli, Muhammad Amjad, Mahpara Shahzadi, Adil Zahoor, Abdelfattah A. Dababat, Halil Toktay, Allah Bakhsh, Muhammad Azher Nawaz, and Hongjie Li. 2019. "Resistance to Cereal Cyst Nematodes in Wheat and Barley: An Emphasis on Classical and Modern Approaches" International Journal of Molecular Sciences 20, no. 2: 432. https://doi.org/10.3390/ijms20020432
APA StyleAli, M. A., Shahzadi, M., Zahoor, A., Dababat, A. A., Toktay, H., Bakhsh, A., Nawaz, M. A., & Li, H. (2019). Resistance to Cereal Cyst Nematodes in Wheat and Barley: An Emphasis on Classical and Modern Approaches. International Journal of Molecular Sciences, 20(2), 432. https://doi.org/10.3390/ijms20020432





