L-Alanine Exporter, AlaE, of Escherichia coli Functions as a Safety Valve to Enhance Survival under Feast Conditions
Abstract
1. Introduction
2. Results and Discussion
2.1. Susceptibility of MG1655 and Its Derivatives to L-Alanyl-L-Alanine (Ala-Ala)
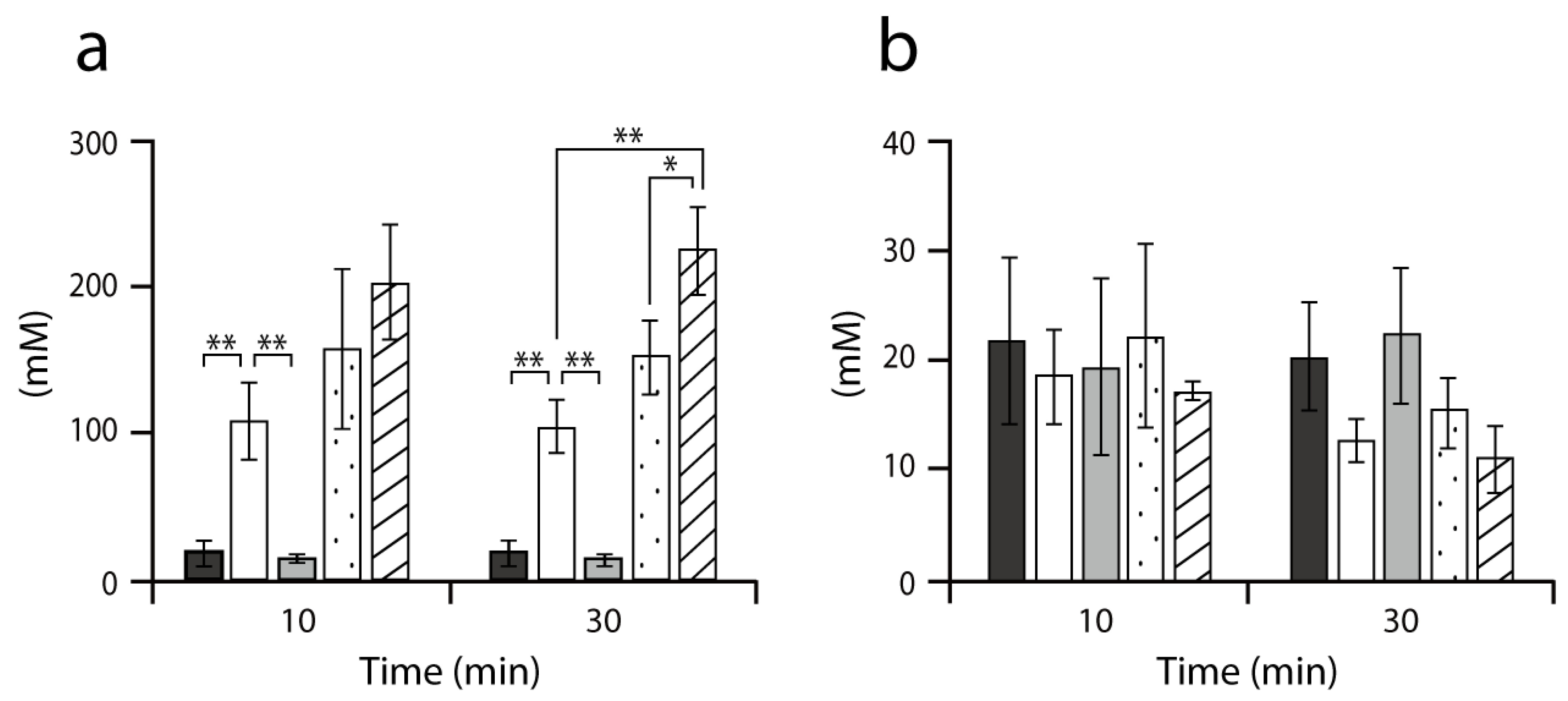
2.2. Accumulation of Intracellular Alanine in MG1655 and its Derivatives
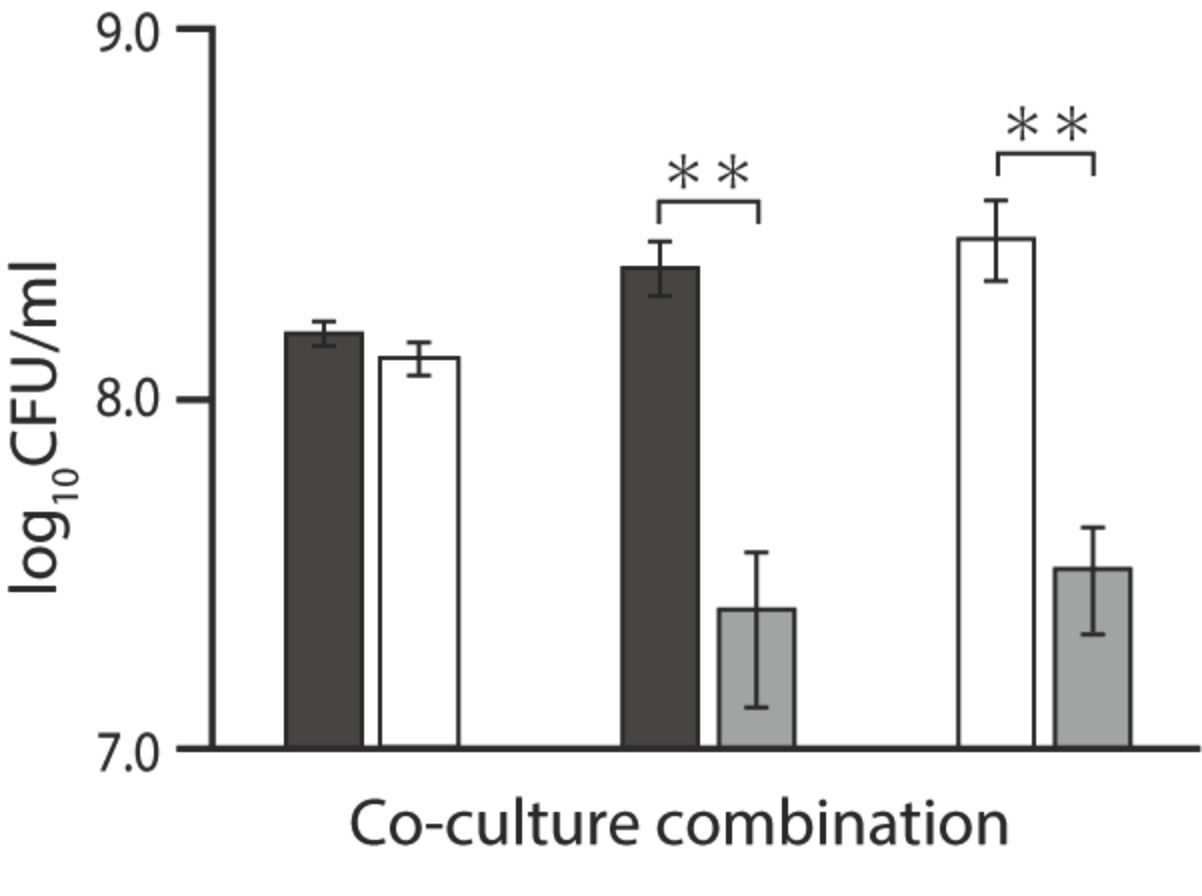
2.3. Coculture Assay in the Presence of Ala-Ala
3. Materials and Methods
3.1. Bacterial Strains, Plasmids, and Culture Conditions
3.2. Construction of a Single Mutant Lacking dadA and Double Mutants Lacking both alaE and dadA
3.3. Susceptibility of the Mutants to L-alanyl-L-alanine (Ala-Ala)
3.4. Determination of Intracellular Amino Acid Concentration
3.5. Coculture Assay
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Reitzer, L.J. Ammonia assimilation and the biosynthesis of glutamine, glutamate, aspartate, asparagine, L-alanine, and D-alanine. In Escherichia Coli and Salmonella: Cellular and Molecular Biology, 2nd ed.; Neidhardt, F.C., Currtiss, R., III, Ingraham, J.L., Lin, E.C.C., Low, K.B., Magasanik, J.B., Reznikoff, W.S., Riley, M., Schaechter, M., Umberger, N.E., Eds.; ASM Press: Washington, DC, USA, 1996; pp. 391–407. [Google Scholar]
- Saier, M.H., Jr. A functional-phylogenetic classification system for transmembrane solute transporters. Microbiol. Mol. Biol. Rev. 2000, 64, 354–411. [Google Scholar] [CrossRef] [PubMed]
- Haney, S.A.; Platko, J.V.; Oxender, D.L.; Calvo, J.M. Lrp, a leucine-responsive protein, regulates branched-chain amino acid transport genes in Escherichia coli. J. Bacteriol. 1992, 174, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, J.; Haneburger, I.; Jung, K. Identification of ArgP and Lrp as Transcriptional Regulators of lysP, the Gene Encoding the Specific Lysine Permease of Escherichia coli. J. Bacteriol. 2011, 193, 2536–2548. [Google Scholar] [CrossRef] [PubMed]
- Mcfall, E.; Newman, E.B. Amino acids as carbon sources. In Escherichia Coli and Salmonella: Cellular and Molecular Biology, 2nd ed.; Neidhardt, F.C., Currtiss, R., III, Ingraham, J.L., Lin, E.C.C., Low, K.B., Magasanik, J.B., Reznikoff, W.S., Riley, M., Schaechter, M., Umberger, N.E., Eds.; ASM Press: Washington, DC, USA, 1996; pp. 358–379. [Google Scholar]
- Reitzer, L.J. Sources of nitrogen and their utilization. In Escherichia Coli and Salmonella: Cellular and Molecular Biology, 2nd ed.; Neidhardt, F.C., Currtiss, R., III, Ingraham, J.L., Lin, E.C.C., Low, K.B., Magasanik, J.B., Reznikoff, W.S., Riley, M., Schaechter, M., Umberger, N.E., Eds.; ASM Press: Washington, DC, USA, 1996; pp. 380–390. [Google Scholar]
- Vrljic, M.; Sahm, H.; Eggeling, L. A new type of transporter with a new type of cellular function: L-lysine export from Corynebacterium glutamicum. Mol. Microbiol. 1996, 22, 815–826. [Google Scholar] [CrossRef] [PubMed]
- Kennerknecht, N.; Sahm, H.; Yen, M.R.; Patek, M.; Saier Jr, M.H., Jr.; Eggeling, L. Export of L-isoleucine from Corynebacterium glutamicum: A two-gene-encoded member of a new translocator family. J. Bacteriol. 2002, 184, 3947–3956. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, J.; Hirano, S.; Ito, H.; Wachi, M. Mutations of the Corynebacterium glutamicum NCgl1221 gene, encoding a mechanosensitive channel homolog, induce L-glutamic acid production. Appl. Env. Microbiol. 2007, 73, 4491–4498. [Google Scholar] [CrossRef] [PubMed]
- Simic, P.; Sahm, H.; Eggeling, L. L-threonine export: Use of peptides to identify a new translocator from Corynebacterium glutamicum. J. Bacteriol. 2001, 183, 5317–5324. [Google Scholar] [CrossRef]
- Zakataeva, N.P.; Aleshin, V.V.; Tokmakova, I.L.; Troshin, P.V.; Livshits, V.A. The novel transmembrane Escherichia coli proteins involved in the amino acid efflux. FEBS Lett. 1999, 452, 228–232. [Google Scholar] [CrossRef]
- Yamada, S.; Awano, N.; Inubushi, K.; Maeda, E.; Nakamori, S.; Nishino, K.; Yamaguchi, A.; Takagi, H. Effect of drug transporter genes on cysteine export and overproduction in Escherichia coli. Appl. Env. Microbiol. 2006, 72, 4735–4742. [Google Scholar] [CrossRef]
- Pittman, M.S.; Corker, H.; Wu, G.; Binet, M.B.; Moir, A.J.; Poole, R.K. Cysteine is exported from the Escherichia coli cytoplasm by CydDC, an ATP-binding cassette-type transporter required for cytochrome assembly. J. Biol. Chem. 2002, 277, 49841–49849. [Google Scholar] [CrossRef]
- Pathania, A.; Sardesai, A.A. Distinct Paths for Basic Amino Acid Export in Escherichia coli: YbjE (LysO) Mediates Export of L-Lysine. J. Bacteriol. 2015, 197, 2036–2047. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Lee, K.H.; Kim, T.Y.; Lee, S.Y. Metabolic engineering of Escherichia coli for the production of L-valine based on transcriptome analysis and in silico gene knockout simulation. Proc. Natl. Acad. Sci. USA 2007, 104, 7797–7802. [Google Scholar] [CrossRef] [PubMed]
- Nandineni, M.R.; Gowrishankar, J. Evidence for an arginine exporter encoded by yggA (argO) that is regulated by the LysR-type transcriptional regulator ArgP in Escherichia coli. J. Bacteriol. 2004, 186, 3539–3546. [Google Scholar] [CrossRef] [PubMed]
- Livshits, V.A.; Zakataeva, N.P.; Aleshin, V.V.; Vitushkina, M.V. Identification and characterization of the new gene rhtA involved in threonine and homoserine efflux in Escherichia coli. Res. Microbiol. 2003, 154, 123–135. [Google Scholar] [CrossRef]
- Liu, Q.; Liang, Y.; Zhang, Y.; Shang, X.; Liu, S.; Wen, J.; Wen, T. YjeH Is a Novel Exporter of l-Methionine and Branched-Chain Amino Acids in Escherichia coli. Appl. Env. Microbiol. 2015, 81, 7753–7766. [Google Scholar] [CrossRef] [PubMed]
- Kutukova, E.A.; Livshits, V.A.; Altman, I.P.; Ptitsyn, L.R.; Zyiatdinov, M.H.; Tokmakova, I.L.; Zakataeva, N.P. The yeaS (leuE) gene of Escherichia coli encodes an exporter of leucine, and the Lrp protein regulates its expression. FEBS Lett. 2005, 579, 4629–4634. [Google Scholar] [CrossRef] [PubMed]
- Hori, H.; Ando, T.; Isogai, E.; Yoneyama, H.; Katsumata, R. Identification of an L-alanine export system in Escherichia coli and isolation and characterization of export-deficient mutants. Fems. Microbiol. Lett. 2011, 316, 83–89. [Google Scholar] [CrossRef][Green Version]
- Franke, I.; Resch, A.; Dassler, T.; Maier, T.; Bock, A. YfiK from Escherichia coli promotes export of O-acetylserine and cysteine. J. Bacteriol. 2003, 185, 1161–1166. [Google Scholar] [CrossRef]
- Doroshenko, V.; Airich, L.; Vitushkina, M.; Kolokolova, A.; Livshits, V.; Mashko, S. YddG from Escherichia coli promotes export of aromatic amino acids. Fems. Microbiol. Lett. 2007, 275, 312–318. [Google Scholar] [CrossRef]
- Dassler, T.; Maier, T.; Winterhalter, C.; Bock, A. Identification of a major facilitator protein from Escherichia coli involved in efflux of metabolites of the cysteine pathway. Mol. Microbiol. 2000, 36, 1101–1112. [Google Scholar] [CrossRef]
- Kim, S.H.; Schneider, B.L.; Reitzer, L. Genetics and regulation of the major enzymes of alanine synthesis in Escherichia coli. J. Bacteriol. 2010, 192, 5304–5311. [Google Scholar] [CrossRef] [PubMed]
- Wild, J.; Hennig, J.; Lobocka, M.; Walczak, W.; Klopotowski, T. Identification of the dadX gene coding for the predominant isozyme of alanine racemase in Escherichia coli K12. Mol. Gen. Genet. 1985, 198, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Wild, J.; Klopotowski, T. D-Amino acid dehydrogenase of Escherichia coli K12: Positive selection of mutants defective in enzyme activity and localization of the structural gene. Mol. Gen. Genet. 1981, 181, 373–378. [Google Scholar] [CrossRef] [PubMed]
- Hori, H.; Yoneyama, H.; Tobe, R.; Ando, T.; Isogai, E.; Katsumata, R. Inducible L-Alanine Exporter Encoded by the Novel Gene ygaW (alaE) in Escherichia coli. Appl. Env. Microb. 2011, 77, 4027–4034. [Google Scholar] [CrossRef] [PubMed]
- Ihara, K.; Sato, K.; Hori, H.; Makino, Y.; Shigenobu, S.; Ando, T.; Isogai, E.; Yoneyama, H. Expression of the alaE gene is positively regulated by the global regulator Lrp in response to intracellular accumulation of l-alanine in Escherichia coli. J. Biosci. Bioeng. 2017, 123, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Ihara, K.; Katsube, S.; Hori, H.; Ando, T.; Isogai, E.; Yoneyama, H. Characterization of the L-alanine exporter AlaE of Escherichia coli and its potential role in protecting cells from a toxic-level accumulation of L-alanine and its derivatives. Microbiologyopen 2015, 4, 632–643. [Google Scholar] [CrossRef] [PubMed]
- Katsube, S.; Sato, K.; Ando, T.; Isogai, E.; Yoneyama, H. Secretion of d-alanine by Escherichia coli. Microbiology 2016, 162, 1243–1252. [Google Scholar] [CrossRef]
- Adibi, S.A.; Mercer, D.W. Protein digestion in human intestine as reflected in luminal, mucosal, and plasma amino acid concentrations after meals. J. Clin. Invest. 1973, 52, 1586–1594. [Google Scholar] [CrossRef]
- Broer, S.; Kramer, R. Lysine excretion by Corynebacterium glutamicum. 1. Identification of a specific secretion carrier system. Eur. J. Biochem. 1991, 202, 131–135. [Google Scholar] [CrossRef]
- Fisher, R.; Tuli, R.; Haselkorn, R. A cloned cyanobacterial gene for glutamine synthetase functions in Escherichia coli, but the enzyme is not adenylylated. Proc. Natl. Acad. Sci. USA 1981, 78, 3393–3397. [Google Scholar] [CrossRef]
- Baba, T.; Ara, T.; Hasegawa, M.; Takai, Y.; Okumura, Y.; Baba, M.; Datsenko, K.A.; Tomita, M.; Wanner, B.L.; Mori, H. Construction of Escherichia coli K-12 in-frame, single-gene knockout mutants: The Keio collection. Mol. Syst. Biol. 2006, 2, 2006.0008. [Google Scholar] [CrossRef] [PubMed]
- Cherepanov, P.P.; Wackernagel, W. Gene disruption in Escherichia coli: TcR and KmR cassettes with the option of Flp-catalyzed excision of the antibiotic-resistance determinant. Gene 1995, 158, 9–14. [Google Scholar] [CrossRef]
- Datsenko, K.A.; Wanner, B.L. One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc. Natl. Acad. Sci. USA 2000, 97, 6640–6645. [Google Scholar] [CrossRef] [PubMed]
- Schneider, F.; Kramer, R.; Burkovski, A. Identification and characterization of the main beta-alanine uptake system in Escherichia coli. Appl. Microbiol. Biotechnol. 2004, 65, 576–582. [Google Scholar] [CrossRef] [PubMed]
| MIC (mg mL −1) | ||
|---|---|---|
| Strains | Glucose | Glycerol |
| MG1655 | >1.25 | >1.25 |
| MG1655∆dadA | >1.25 | >1.25 |
| MG1655∆alaE | 0.04 | 0.16 |
| MG1655∆dadA∆alaE | 0.0025 | 0.0025 |
| MLA301 | >1.25 | >1.25 |
| MLA301∆alaE | 0.0025 | 0.0025 |
| Strain or Plasmid | Characteristics | Reference |
|---|---|---|
| Strain | ||
| E. coli MG1655 | Wild type | Laboratory strain |
| E. coli JW1178 | dadA disruptant derived from BW25113, KMr | [34] |
| E. coli JW2645 | alaE disruptant derived from BW25113, KMr | [34] |
| E. coli MG1655 alaE | alaE disruptant derived from MG1655, KMr | [29] |
| E. coli MG1655 dadA | dadA disruptant derived from MG1655, KMr | This study |
| E. coli MG1655 dadA alaE | MG1655 dadA derivative with a deletion in the alaE gene, KMr | This study |
| E. coli MLA301 | MG1655 alr::FRT, dadX::FRT, yfdZ::FRT, avtA::GM, yfbQ::KM | [20] |
| E. coli MLA301 alaE | MLA301 derivative with a deletion in the alaE gene, GMr, KMr | [27] |
| Plasmid | ||
| pCP20 | FLP+, λcI857−, λpRRepts, Ampr, CPr | [35] |
| pKD46 | Red recombinase expression plasmid | [36] |
| pBR322 | Ampr, TETr, cloning vector | [27] |
| pBR325 | Ampr, TETr, CPr, cloning vector | Laboratory stock |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Katsube, S.; Ando, T.; Yoneyama, H. L-Alanine Exporter, AlaE, of Escherichia coli Functions as a Safety Valve to Enhance Survival under Feast Conditions. Int. J. Mol. Sci. 2019, 20, 4942. https://doi.org/10.3390/ijms20194942
Katsube S, Ando T, Yoneyama H. L-Alanine Exporter, AlaE, of Escherichia coli Functions as a Safety Valve to Enhance Survival under Feast Conditions. International Journal of Molecular Sciences. 2019; 20(19):4942. https://doi.org/10.3390/ijms20194942
Chicago/Turabian StyleKatsube, Satoshi, Tasuke Ando, and Hiroshi Yoneyama. 2019. "L-Alanine Exporter, AlaE, of Escherichia coli Functions as a Safety Valve to Enhance Survival under Feast Conditions" International Journal of Molecular Sciences 20, no. 19: 4942. https://doi.org/10.3390/ijms20194942
APA StyleKatsube, S., Ando, T., & Yoneyama, H. (2019). L-Alanine Exporter, AlaE, of Escherichia coli Functions as a Safety Valve to Enhance Survival under Feast Conditions. International Journal of Molecular Sciences, 20(19), 4942. https://doi.org/10.3390/ijms20194942




