Genome-Wide CpG Island Methylation Profiles of Cutaneous Skin with and without HPV Infection
Abstract
1. Introduction
2. Results
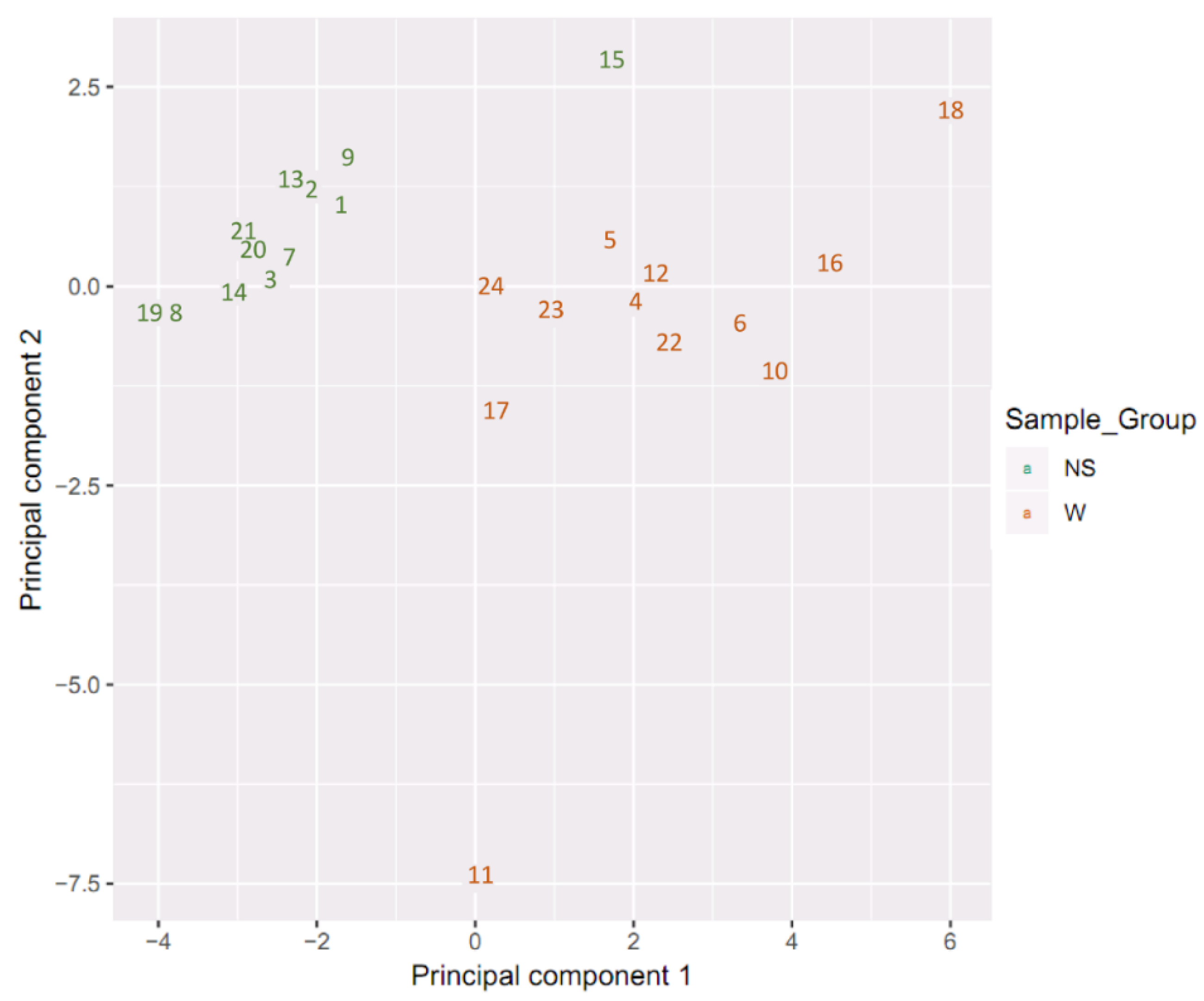
2.1. Sample Clustering
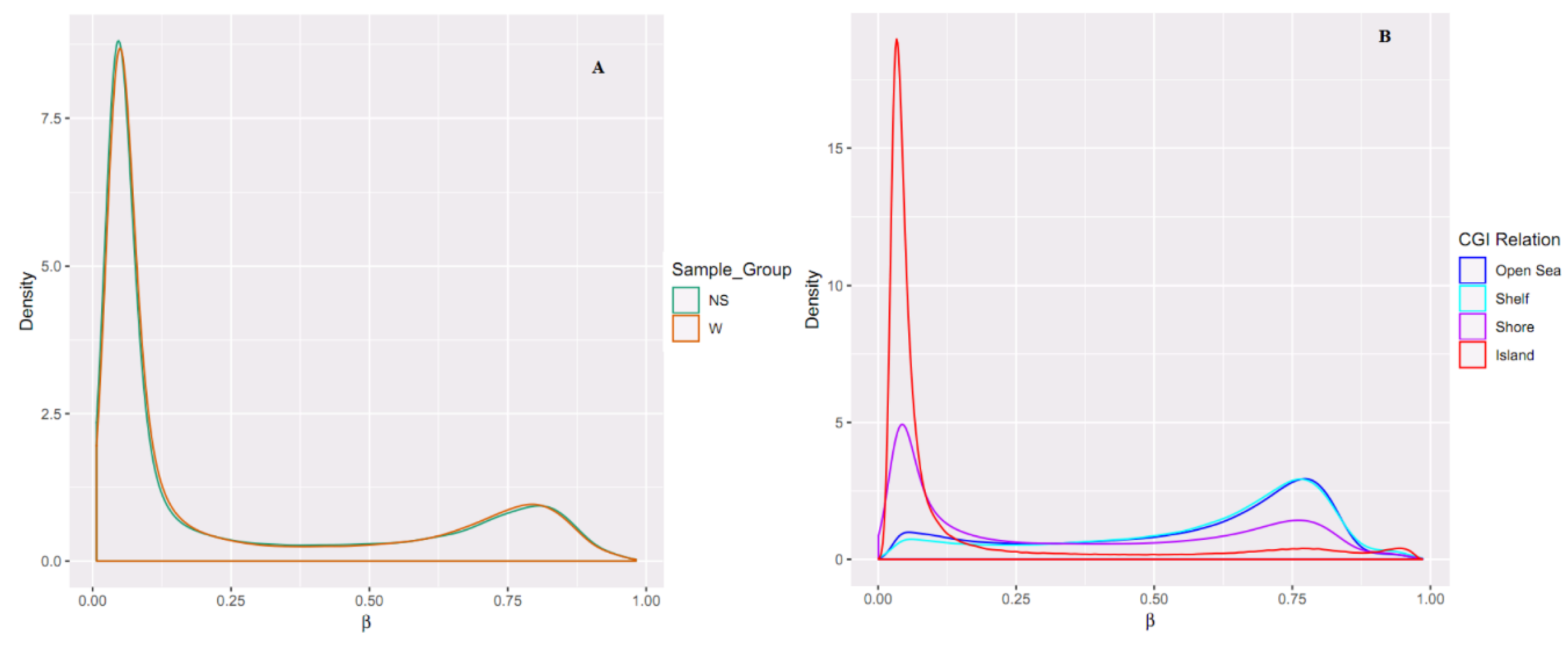
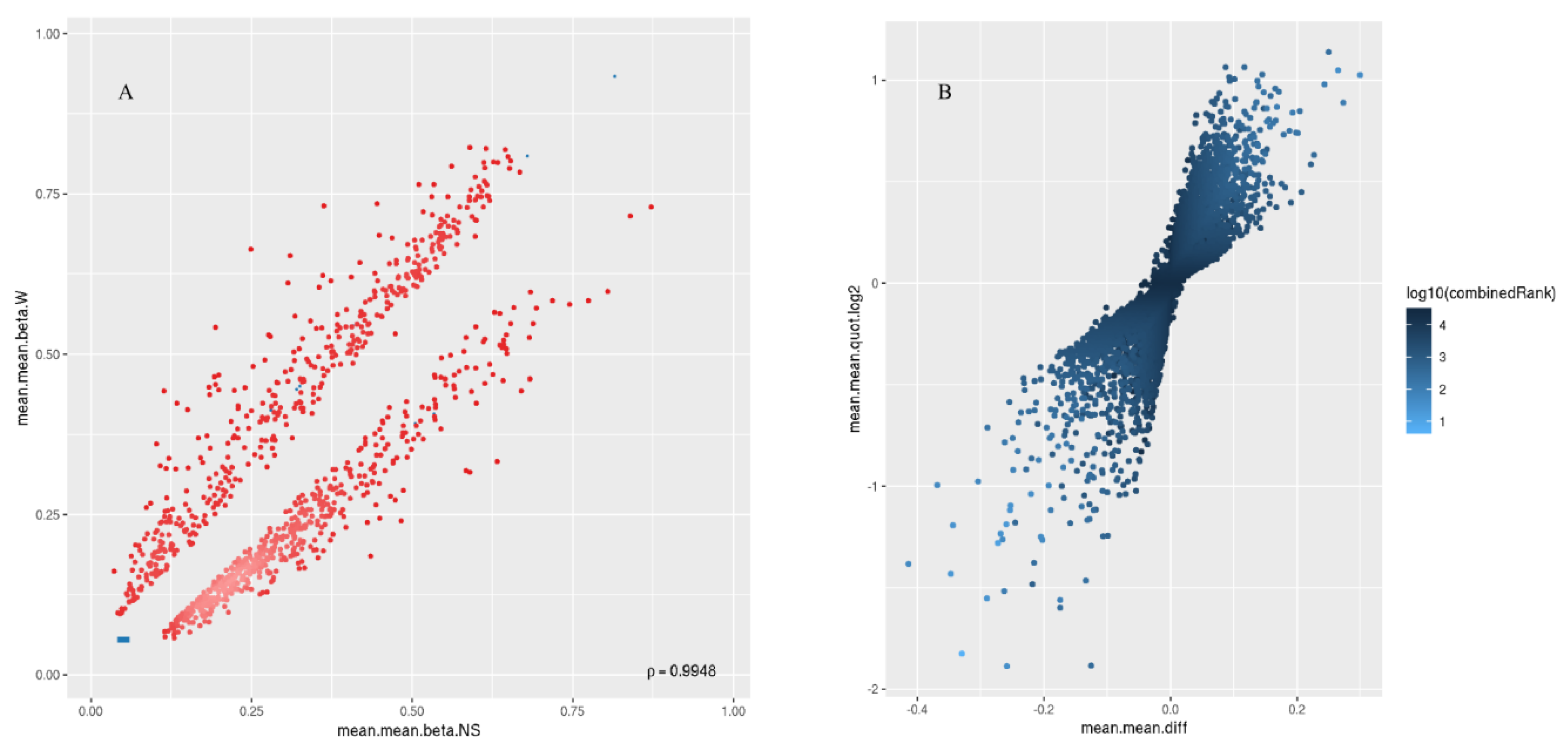
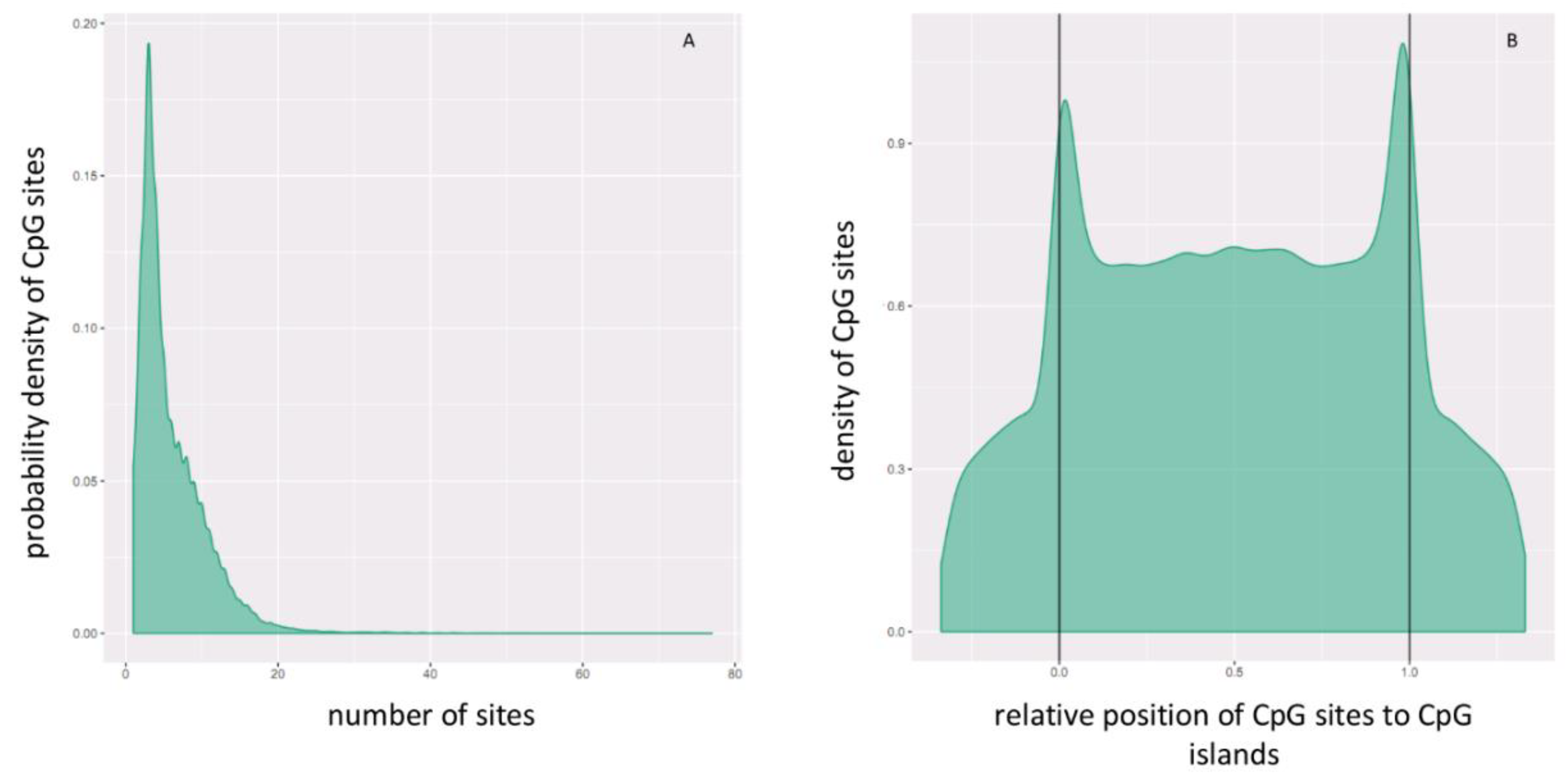
2.2. Differential Methylation of CpG Islands
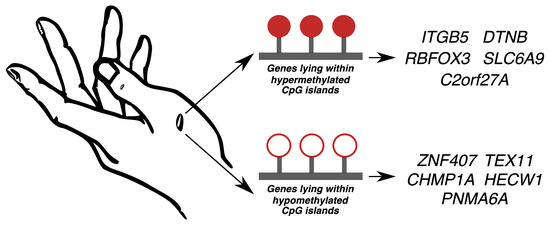
2.3. Hypermethylated and Hypomethylated CpG Islands
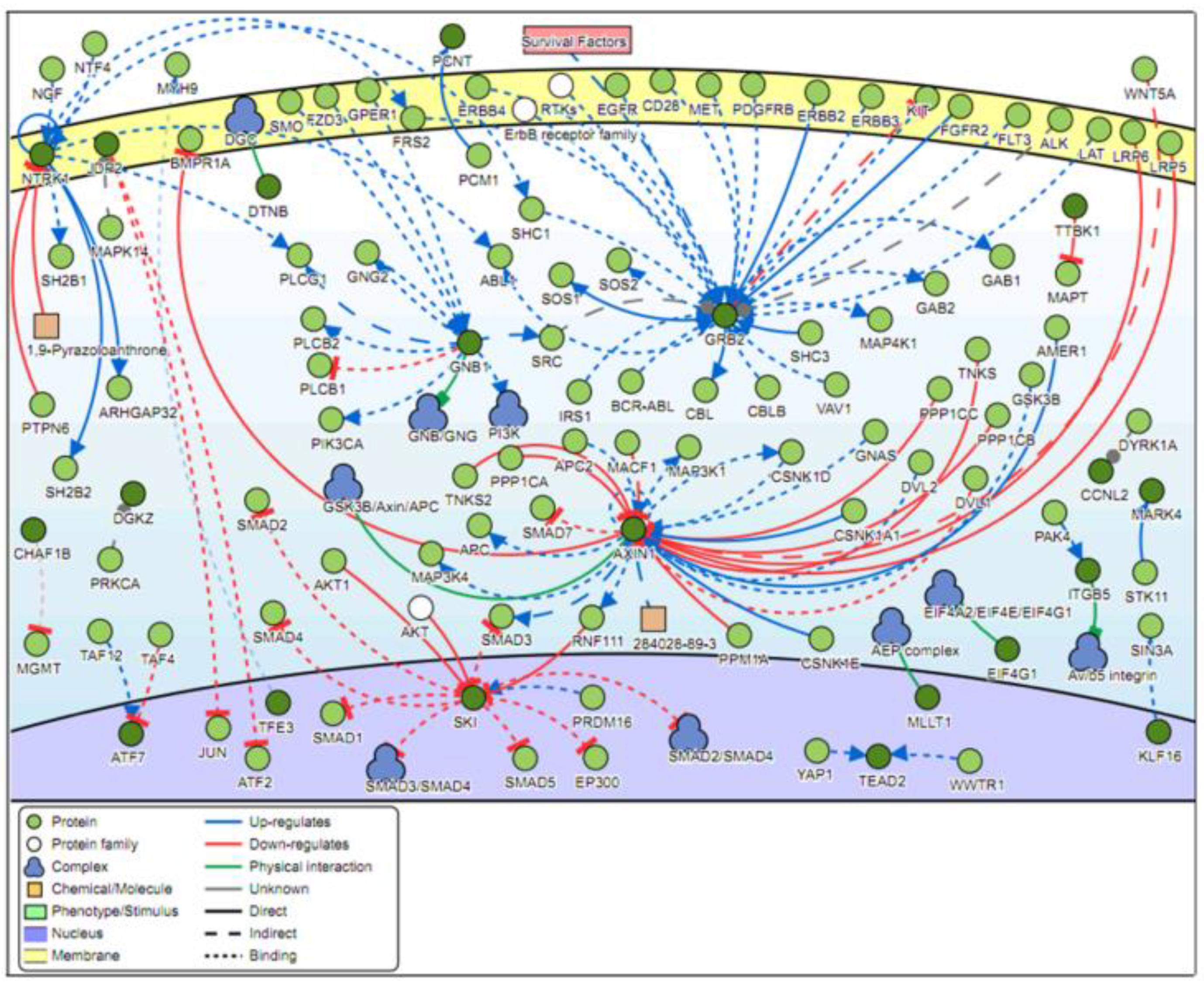
2.4. Signaling Networks
2.5. Validation of the Top Five Genes Associated with Differentially Methylated CpG Islands
3. Discussion
4. Methods
4.1. Subjects
4.2. DNA Extraction and the Infinium MethylationEPIC BeadChip
4.3. Data Analysis and Processing
4.4. Differential Methylation Analysis
4.5. Locus Overlap Analysis
4.6. Signaling Pathway Analysis
4.7. Validation of the Genes Associated with DM CpG Islands
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CpG | 5′-C-phosphate-G-3′ |
| DM | Differentially methylated |
| NS | Normal skin |
| W | Wart |
References
- Van Doorslaer, K. Evolution of the Papillomaviridae. Virology 2013, 445, 11–20. [Google Scholar] [CrossRef]
- Bzhalava, D.; Guan, P.; Franceschi, S.; Dillner, J.; Clifford, G. A systematic review of the prevalence of mucosal and cutaneous human papillomavirus types. Virology 2013, 445, 224–231. [Google Scholar] [CrossRef]
- Durzynska, J.; Lesniewicz, K.; Poreba, E. Human papillomaviruses in epigenetic regulations. Mutat. Res. Mutat. Res. 2017, 772, 36–50. [Google Scholar] [CrossRef]
- Sterling, J.C.; Gibbs, S.; Haque Hussain, S.S.; Mohd Mustapa, M.F.; Handfield-Jones, S.E. British Association of Dermatologists’ guidelines for the management of cutaneous warts 2014. Br. J. Dermatol. 2014, 171, 696–712. [Google Scholar] [CrossRef]
- Accardi, R.; Gheit, T. Cutaneous HPV and skin cancer. Presse Med. 2014, 43, e435–e443. [Google Scholar] [CrossRef]
- De Abreu, P.M.; Có, A.C.G.; Azevedo, P.L.; do Valle, I.B.; de Oliveira, K.G.; Gouvea, S.A.; Cordeiro-Silva, M.F.; Louro, I.D.; de Podestá, J.R.V.; Lenzi, J.; et al. Frequency of HPV in oral cavity squamous cell carcinoma. BMC Cancer 2018, 18, 324. [Google Scholar] [CrossRef]
- Ducatman, B.S. The Role of Human Papillomavirus in Oropharyngeal Squamous Cell Carcinoma. Arch. Pathol. Lab. Med. 2018, 142, 715–718. [Google Scholar] [CrossRef]
- Smith, T.J.; Mendez, A.; Donald, C.; Nagel, T.H. HPV-associated oropharyngeal squamous cell carcinoma. J. Am. Acad. Physician Assist. 2017, 30, 14–19. [Google Scholar] [CrossRef]
- Villagómez-Ortíz, V.J.; Paz-Delgadillo, D.E.; Marino-Martínez, I.; Ceseñas-Falcón, L.Á.; Sandoval-de la Fuente, A.; Reyes-Escobedo, A. Prevalence of human papillomavirus infection in squamous cell carcinoma of the oral cavity, oropharynx and larynx. Cirugía y Cirujanos 2016, 84, 363–368. [Google Scholar] [CrossRef]
- Plesa, A.; Iancu, I.V.; Botezatu, A.; Huica, I.; Stoian, M.; Anton, G. The Involvement of Epigenetic Mechanisms in HPV-Induced Cervical Cancer. In Human Papillomavirus—Research in a Global Perspective; InTech: London, UK, 2016. [Google Scholar]
- Di Domenico, M.; Giovane, G.; Kouidhi, S.; Iorio, R.; Romano, M.; De Francesco, F.; Feola, A.; Siciliano, C.; Califano, L.; Giordano, A. HPV epigenetic mechanisms related to Oropharyngeal and Cervix cancers. Cancer Biol. Ther. 2018, 19, 850–857. [Google Scholar] [CrossRef]
- Jin, Z.; Liu, Y. DNA methylation in human diseases. Genes Dis. 2018, 5, 1–8. [Google Scholar] [CrossRef]
- Heyn, H.; Moran, S.; Hernando-Herraez, I.; Sayols, S.; Gomez, A.; Sandoval, J.; Monk, D.; Hata, K.; Marques-Bonet, T.; Wang, L.; et al. DNA methylation contributes to natural human variation. Genome Res. 2013, 23, 1363–1372. [Google Scholar] [CrossRef]
- Jang, H.S.; Shin, W.J.; Lee, J.E.; Do, J.T. CpG and Non-CpG Methylation in Epigenetic Gene Regulation and Brain Function. Genes 2017, 8, 148. [Google Scholar] [CrossRef]
- Deaton, A.M.; Bird, A. CpG islands and the regulation of transcription. Genes Dev. 2011, 25, 1010–1022. [Google Scholar] [CrossRef]
- Paska, A.V.; Hudler, P. Aberrant methylation patterns in cancer: A clinical view. Biochem. Medica 2015, 25, 161–176. [Google Scholar] [CrossRef]
- Yang, H.-J. Aberrant DNA methylation in cervical carcinogenesis. Chin. J. Cancer 2013, 32, 42–48. [Google Scholar] [CrossRef]
- Verlaat, W.; Van Leeuwen, R.W.; Novianti, P.W.; Schuuring, E.; Meijer, C.J.L.M.; Van Der Zee, A.G.J.; Snijders, P.J.F.; Heideman, D.A.M.; Steenbergen, R.D.M.; Wisman, G.B.A. Host-cell DNA methylation patterns during high-risk HPV-induced carcinogenesis reveal a heterogeneous nature of cervical pre-cancer. Epigenetics 2018, 13, 769–778. [Google Scholar] [CrossRef]
- Small, W.; Bacon, M.A.; Bajaj, A.; Chuang, L.T.; Fisher, B.J.; Harkenrider, M.M.; Jhingran, A.; Kitchener, H.C.; Mileshkin, L.R.; Viswanathan, A.N.; et al. Cervical cancer: A global health crisis. Cancer 2017, 123, 2404–2412. [Google Scholar] [CrossRef]
- Sen, P.; Ganguly, P.; Ganguly, N. Modulation of DNA methylation by human papillomavirus E6 and E7 oncoproteins in cervical cancer (Review). Oncol. Lett. 2017, 15, 11–22. [Google Scholar] [CrossRef]
- Degli Esposti, D.; Sklias, A.; Lima, S.C.; Beghelli-de la Forest Divonne, S.; Cahais, V.; Fernandez-Jimenez, N.; Cros, M.-P.; Ecsedi, S.; Cuenin, C.; Bouaoun, L.; et al. Unique DNA methylation signature in HPV-positive head and neck squamous cell carcinomas. Genome Med. 2017, 9, 33. [Google Scholar] [CrossRef]
- Dankai, W.; Khunamornpong, S.; Siriaunkgul, S.; Soongkhaw, A.; Janpanao, A.; Utaipat, U.; Kitkumthorn, N.; Mutirangura, A.; Srisomboon, J.; Lekawanvijit, S. Role of genomic DNA methylation in detection of cytologic and histologic abnormalities in high risk HPV-infected women. PLoS ONE 2019, 14, e0210289. [Google Scholar] [CrossRef]
- Clarke, M.A.; Gradissimo, A.; Schiffman, M.; Lam, J.; Sollecito, C.C.; Fetterman, B.; Lorey, T.; Poitras, N.; Raine-Bennett, T.R.; Castle, P.E.; et al. Human Papillomavirus DNA Methylation as a Biomarker for Cervical Precancer: Consistency across 12 Genotypes and Potential Impact on Management of HPV-Positive Women. Clin. Cancer Res. 2018, 24, 2194–2202. [Google Scholar] [CrossRef]
- Amaro-Filho, S.M.; Pereira Chaves, C.B.; Felix, S.P.; Basto, D.L.; de Almeida, L.M.; Moreira, M.A.M. HPV DNA methylation at the early promoter and E1/E2 integrity: A comparison between HPV16, HPV18 and HPV45 in cervical cancer. Papillomavirus Res. 2018, 5, 172–179. [Google Scholar] [CrossRef]
- Johannsen, E.; Lambert, P.F. Epigenetics of human papillomaviruses. Virology 2013, 445, 205–212. [Google Scholar] [CrossRef]
- Jeziorska, D.M.; Murray, R.J.S.; De Gobbi, M.; Gaentzsch, R.; Garrick, D.; Ayyub, H.; Chen, T.; Li, E.; Telenius, J.; Lynch, M.; et al. DNA methylation of intragenic CpG islands depends on their transcriptional activity during differentiation and disease. Proc. Natl. Acad. Sci. USA 2017, 114, E7526–E7535. [Google Scholar] [CrossRef]
- Graham, S.V. Keratinocyte Differentiation-Dependent Human Papillomavirus Gene Regulation. Viruses 2017, 9, 245. [Google Scholar] [CrossRef]
- Zhou, C.; Liu, Z.; Liu, Y.; Fu, W.; Ding, X.; Liu, J.; Yu, Y.; Zhang, Q. Gene Silencing of Porcine MUC13 and ITGB5: Candidate Genes towards Escherichia coli F4ac Adhesion. PLoS ONE 2013, 8, e70303. [Google Scholar] [CrossRef]
- Himes, B.E.; Qiu, W.; Klanderman, B.; Ziniti, J.; Senter-Sylvia, J.; Szefler, S.J.; Lemanske, R.F., Jr.; Zeiger, R.S.; Strunk, R.C.; Martinez, F.D.; et al. ITGB5 and AGFG1 variants are associated with severity of airway responsiveness. BMC Med. Genet. 2013, 14, 86. [Google Scholar] [CrossRef]
- Eckhardt, M.; Zhang, W.; Gross, A.M.; Von Dollen, J.; Johnson, J.R.; Franks-Skiba, K.E.; Swaney, D.L.; Johnson, T.L.; Jang, G.M.; Shah, P.S.; et al. Multiple Routes to Oncogenesis Are Promoted by the Human Papillomavirus-Host Protein Network. Cancer Discov. 2018, 8, 1474–1489. [Google Scholar] [CrossRef]
- Quaranta, M.T.; Spinello, I.; Paolillo, R.; Macchia, G.; Boe, A.; Ceccarini, M.; Labbaye, C.; Macioce, P. Identification of β-Dystrobrevin as a Direct Target of miR-143: Involvement in Early Stages of Neural Differentiation. PLoS ONE 2016, 11, e0156325. [Google Scholar] [CrossRef]
- Amin, N.; Allebrandt, K. V.; van der Spek, A.; Müller-Myhsok, B.; Hek, K.; Teder-Laving, M.; Hayward, C.; Esko, T.; van Mill, J.G.; Mbarek, H.; et al. Genetic variants in RBFOX3 are associated with sleep latency. Eur. J. Hum. Genet. 2016, 24, 1488–1495. [Google Scholar] [CrossRef]
- Lal, D.; Reinthaler, E.M.; Altmüller, J.; Toliat, M.R.; Thiele, H.; Nürnberg, P.; Lerche, H.; Hahn, A.; Møller, R.S.; Muhle, H.; et al. RBFOX1 and RBFOX3 Mutations in Rolandic Epilepsy. PLoS ONE 2013, 8, e73323. [Google Scholar] [CrossRef]
- Lulli, D.; Carbone, M.L.; Pastore, S. Epidermal growth factor receptor inhibitors trigger a type I interferon response in human skin. Oncotarget 2016, 7, 47777–47793. [Google Scholar] [CrossRef]
- Teng, Y.; Wang, Y.; Zhang, X.; Liu, W.; Fan, H.; Yao, H.; Lin, B.; Zhu, P.; Yuan, W.; Tong, Y.; et al. Systematic Genome-wide Screening and Prediction of microRNAs in EBOV During the 2014 Ebolavirus Outbreak. Sci. Rep. 2015, 5, 9912. [Google Scholar] [CrossRef]
- Wagner, J.R.; Busche, S.; Ge, B.; Kwan, T.; Pastinen, T.; Blanchette, M. The relationship between DNA methylation, genetic and expression inter-individual variation in untransformed human fibroblasts. Genome Biol. 2014, 15, R37. [Google Scholar] [CrossRef]
- Lichtenberger, B.M.; Gerber, P.A.; Holcmann, M.; Buhren, B.A.; Amberg, N.; Smolle, V.; Schrumpf, H.; Boelke, E.; Ansari, P.; Mackenzie, C.; et al. Epidermal EGFR Controls Cutaneous Host Defense and Prevents Inflammation. Sci. Transl. Med. 2013, 5, 199ra111. [Google Scholar] [CrossRef]
- Wang, H.; Li, S.; Li, H.; Wang, P.; Huang, F.; Zhao, Y.; Yu, L.; Luo, G.; Zhang, X.; Wang, J.; et al. Grb2 interacts with SGEF and antagonizes the ability of SGEF to enhance EGF-induced ERK1/2 activation. Mol. Cell. Biochem. 2014, 389, 239–247. [Google Scholar] [CrossRef]
- Spangle, J.M.; Munger, K. The HPV16 E6 Oncoprotein Causes Prolonged Receptor Protein Tyrosine Kinase Signaling and Enhances Internalization of Phosphorylated Receptor Species. PLoS Pathog. 2013, 9, e1003237. [Google Scholar] [CrossRef]
- Santos, J.; Peixoto da Silva, S.; Costa, N.; Gil da Costa, R.; Medeiros, R. The Role of MicroRNAs in the Metastatic Process of High-Risk HPV-Induced Cancers. Cancers (Basel) 2018, 10, 493. [Google Scholar] [CrossRef]
- Szczałuba, K.; Biernacka, A.; Szymańska, K.; Gasperowicz, P.; Kosińska, J.; Rydzanicz, M.; Płoski, R. Novel GNB1 de novo mutation in a patient with neurodevelopmental disorder and cutaneous mastocytosis: Clinical report and literature review. Eur. J. Med. Genet. 2018, 61, 157–160. [Google Scholar] [CrossRef]
- Bonkowsky, J.L.; Johnson, J.; Carey, J.C.; Smith, A.G.; Swoboda, K.J. An infant with primary tooth loss and palmar hyperkeratosis: A novel mutation in the NTRK1 gene causing congenital insensitivity to pain with anhidrosis. Pediatrics 2003, 112, e237–e241. [Google Scholar] [CrossRef] [PubMed]
- Salahshor, S.; Woodgett, J.R. The links between axin and carcinogenesis. J. Clin. Pathol. 2005, 58, 225–236. [Google Scholar] [CrossRef] [PubMed]
- Pećina-Slaus, N.; Martić, T.N.; Kokotović, T.; Kusec, V.; Tomas, D.; Hrasćan, R. AXIN-1 protein expression and localization in glioblastoma. Coll. Antropol. 2011, 35 (Suppl. 1), 101–106. [Google Scholar] [PubMed]
- Ishiguro, H.; Tsunoda, T.; Tanaka, T.; Fujii, Y.; Nakamura, Y.; Furukawa, Y. Identification of AXUD1, a novel human gene induced by AXIN1 and its reduced expression in human carcinomas of the lung, liver, colon and kidney. Oncogene 2001, 20, 5062–5066. [Google Scholar] [CrossRef] [PubMed]
- Picco, G.; Petti, C.; Centonze, A.; Torchiaro, E.; Crisafulli, G.; Novara, L.; Acquaviva, A.; Bardelli, A.; Medico, E. Loss of AXIN1 drives acquired resistance to WNT pathway blockade in colorectal cancer cells carrying RSPO3 fusions. EMBO Mol. Med. 2017, 9, 293–303. [Google Scholar] [CrossRef] [PubMed]
- Koppert, L.B.; van der Velden, A.W.; van de Wetering, M.; Abbou, M.; van den Ouweland, A.M.W.; Tilanus, H.W.; Wijnhoven, B.P.L.; Dinjens, W.N.M. Frequent loss of the AXIN1 locus but absence of AXIN1 gene mutations in adenocarcinomas of the gastro-oesophageal junction with nuclear β-catenin expression. Br. J. Cancer 2004, 90, 892–899. [Google Scholar] [CrossRef][Green Version]
- Tecalco-Cruz, A.C.; Ríos-López, D.G.; Vázquez-Victorio, G.; Rosales-Alvarez, R.E.; Macías-Silva, M. Transcriptional cofactors Ski and SnoN are major regulators of the TGF-β/Smad signaling pathway in health and disease. Signal Transduct. Target. Ther. 2018, 3, 15. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, A.; Pirisi, L.; Creek, K.E. NFI-Ski Interactions Mediate Transforming Growth Factor β Modulation of Human Papillomavirus Type 16 Early Gene Expression. J. Virol. 2004, 78, 3953–3964. [Google Scholar] [CrossRef]
- Deheuninck, J.; Luo, K. Ski and SnoN, potent negative regulators of TGF-beta signaling. Cell Res. 2009, 19, 47–57. [Google Scholar] [CrossRef]
- Bruggink, S.C.; de Koning, M.N.C.; Gussekloo, J.; Egberts, P.F.; ter Schegget, J.; Feltkamp, M.C.W.; Bavinck, J.N.B.; Quint, W.G.V.; Assendelft, W.J.J.; Eekhof, J.A.H. Cutaneous wart-associated HPV types: Prevalence and relation with patient characteristics. J. Clin. Virol. 2012, 55, 250–255. [Google Scholar] [CrossRef]
- Mulhem, E.; Pinelis, S. Treatment of nongenital cutaneous warts. Am. Fam. Physician 2011, 84, 288–293. [Google Scholar] [PubMed]
- Assenov, Y.; Müller, F.; Lutsik, P.; Walter, J.; Lengauer, T.; Bock, C. Comprehensive analysis of DNA methylation data with RnBeads. Nat. Methods 2014, 11, 1138–1140. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, M.E.; Phipson, B.; Wu, D.; Hu, Y.; Law, C.W.; Shi, W.; Smyth, G.K. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 2015, 43, e47. [Google Scholar] [CrossRef] [PubMed]
- Sheffield, N.C.; Bock, C. LOLA: Enrichment analysis for genomic region sets and regulatory elements in R and Bioconductor. Bioinformatics 2016, 32, 587–589. [Google Scholar] [CrossRef] [PubMed]
- Perfetto, L.; Briganti, L.; Calderone, A.; Cerquone Perpetuini, A.; Iannuccelli, M.; Langone, F.; Licata, L.; Marinkovic, M.; Mattioni, A.; Pavlidou, T.; et al. SIGNOR: A database of causal relationships between biological entities. Nucleic Acids Res. 2016, 44, D548–D554. [Google Scholar] [CrossRef] [PubMed]
- Al-Eitan, L.; Tarkhan, A.H.; Alghamdi, M.A.; Al-Qarqaz, F.A.; Al-Kofahi, H.S. Transcriptome analysis of HPV-induced warts and healthy skin in humans. BMC Med. Genom. 2019. submitted. [Google Scholar]

| Methylation Levels | Expression Levels | |||||
|---|---|---|---|---|---|---|
| Gene | Mean.mean β value (NS) | Mean.mean β value (W) | Mean.mean β value (W-NS) | p-value | logFC | p-value |
| ITGB5 | 0.386 | 0.428 | −0.042 | 0.0002 | 0.418 | 0.0060 |
| DTNB | 0.608 | 0.604 | 0.004 | 0.0138 | −0.029 | 0.8569 |
| RBFOX3 | 0.493 | 0.473 | 0.021 | 0.0543 | n/a | n/a |
| SLC6A9 | 0.497 | 0.499 | −0.002 | 0.0602 | 0.212 | 0.3504 |
| C2orf27A | 0.600 | 0.598 | 0.002 | 0.2827 | n/a | n/a |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
AL-Eitan, L.N.; Alghamdi, M.A.; Tarkhan, A.H.; Al-Qarqaz, F.A. Genome-Wide CpG Island Methylation Profiles of Cutaneous Skin with and without HPV Infection. Int. J. Mol. Sci. 2019, 20, 4822. https://doi.org/10.3390/ijms20194822
AL-Eitan LN, Alghamdi MA, Tarkhan AH, Al-Qarqaz FA. Genome-Wide CpG Island Methylation Profiles of Cutaneous Skin with and without HPV Infection. International Journal of Molecular Sciences. 2019; 20(19):4822. https://doi.org/10.3390/ijms20194822
Chicago/Turabian StyleAL-Eitan, Laith N., Mansour A. Alghamdi, Amneh H. Tarkhan, and Firas A. Al-Qarqaz. 2019. "Genome-Wide CpG Island Methylation Profiles of Cutaneous Skin with and without HPV Infection" International Journal of Molecular Sciences 20, no. 19: 4822. https://doi.org/10.3390/ijms20194822
APA StyleAL-Eitan, L. N., Alghamdi, M. A., Tarkhan, A. H., & Al-Qarqaz, F. A. (2019). Genome-Wide CpG Island Methylation Profiles of Cutaneous Skin with and without HPV Infection. International Journal of Molecular Sciences, 20(19), 4822. https://doi.org/10.3390/ijms20194822